Abstract
Coumarin is a naturally occurring sweet-smelling benzopyrone that may be extracted from plants or synthesized for commercial uses. Its uses include as a flavoring agent, fragrance enhancer, and odor-masking additive. We reviewed and evaluated the scientific evidence on the carcinogenicity of coumarin, integrating information from carcinogenicity studies in animals with mechanistic and other relevant data, including data from toxicogenomic, genotoxicity, and metabolism studies, and studies of human variability of a key enzyme, CYP2A6. Increases in tumors were observed in multiple studies in rats and mice in multiple tissues. Our functional pathway analysis identified several common cancer-related biological processes/pathways affected by coumarin in rat liver following in vivo exposure and in human primary hepatocytes exposed in vitro. When coumarin 7-hydroxylation by CYP2A6 is compromised, this can lead to a shift in metabolism to the 3,4-epoxidation pathway and increased generation of electrophilic metabolites. Mechanistic data align with 3 key characteristics of carcinogens, namely formation of electrophilic metabolites, genotoxicity, and induction of oxidative stress. Considerations of metabolism, human variability in CYP2A6 activity, and coumarin hepatotoxicity in susceptible individuals provide additional support for carcinogenicity concern. Our analysis illustrates the importance of integrating information on human variability in the cancer hazard identification process.
Keywords
Introduction
Coumarin (1,2-benzopyrone, CAS No.: 91-64-5) is a naturally occurring compound found in many plants, such as tonka beans, green tea leaves, some fruits (eg, strawberries, apricots), and some spices and herbs (eg, cinnamon, lavender, sweet woodruff, sage, dill, chamomile, peppermint). 1,2 It has a pleasant sweet odor resembling that of vanilla beans or fresh cut grass. 3 Coumarin (Figure 1) is a crystalline solid that is freely soluble in ethanol, chloroform, and oils and is slightly soluble in water. 4 It can be extracted from plants (eg, it was first isolated from tonka beans in 1822) or synthesized from ortho cresol, phenol, and salicylaldehyde for commercial uses. 5 Coumarin is not to be confused with “coumarins” used by the pharmaceutical industry (eg, coumadin or warfarin).
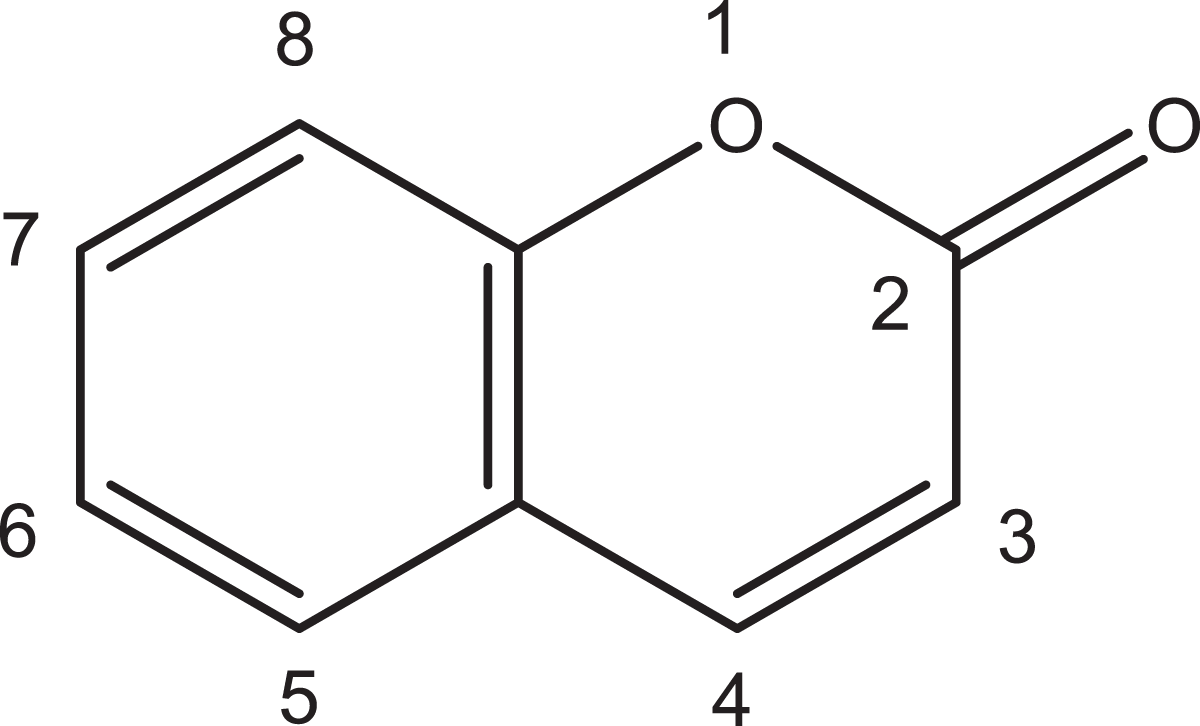
Chemical structure of coumarin.
Cinnamon is one of the more common dietary sources of coumarin. Of the 2 major types of cinnamon used in food, levels of coumarin are higher in Cassia cinnamon, ranging from 85 to 9,300 ppm, than in Ceylon (“true”) cinnamon (5-90 ppm). 6 Wang et al 6 measured coumarin in cinnamon-flavored foods (eg, rolls, cereal; 3-56 ppm) and cinnamon-based dietary supplements sold in the United States (2,450-3,610 ppm).
Coumarin is used as an industrial chemical to mask odors in plastic materials and paints. 4,7 Coumarin is also used as a fragrance enhancer in a variety of personal care products (eg, perfume, cosmetics, hair spray, detergents, soaps) and as a flavoring agent in tobacco products, including US electronic cigarettes, 8 cigarette tobacco, and Indian bidi cigarette tobacco. 9 Coumarin is on the US Food and Drug Administration (FDA) list of Harmful and Potentially Harmful Constituents in tobacco products and tobacco smoke. 10
In the United States, the use of coumarin as a direct food additive (in pure form, and as a constituent of tonka beans or tonka bean extracts) has been banned since 1954, due to its reported severe hepatotoxicity in animal studies. 10 The FDA has issued warnings for coumarin found in some artificial vanilla extracts or flavorings purchased or imported from Mexico that contained tonka bean extracts. 11
In 2000, the International Agency for Research on Cancer (IARC) evaluated coumarin as to its potential carcinogenicity, and classified it in group 3 (not classifiable as to its carcinogenicity to humans), based on no epidemiological data and limited evidence in experimental animals. 5 Since the IARC review, several studies relevant to the carcinogenicity of coumarin have been published and have been included as part of this review. Here, we examine the carcinogenicity of coumarin, summarizing and integrating evidence from a variety of study types, including long-term animal cancer bioassays and co-carcinogenicity studies, followed by data from mechanistic studies, including pharmacokinetics and metabolism, genotoxicity and cell transformation studies, toxicogenomic analysis, ToxCast high-throughput in vitro assays, and structure activity comparisons. We also discuss information on human genetic polymorphisms and variability of CYP2A6, a key enzyme for coumarin metabolism. Evidence from all data streams is integrated in considering the carcinogenicity of coumarin.
Methods
Literature Search Strategy
General searches of the literature on the carcinogenicity of coumarin were conducted to identify peer-reviewed open source and proprietary journal articles, print and digital books, and reports and gray literature that potentially reported relevant toxicological and epidemiological information on the carcinogenicity of this chemical. Additional focused searches were performed as needed. For example, searches on the pathology and spontaneous incidence of selected rodent tumors were conducted, as well as focused searches of toxicogenomic databases. Relevant literature was also identified from citations in individual articles.
Databases searched
The literature search utilized the following search platforms/databases: ChemSpider (http://www.chemspider.com/) PubMed (National Library of Medicine) EMIC (National Library of Medicine) SciFinder® TOXNET (National Library of Medicine): Toxicology Literature Online (TOXLINE), Genetic Toxicology Data Bank (GENE-TOX) Web of Knowledge: BIOSIS Previews, Web of Science (Thomson-Reuters, Inc) Other databases include Google search engine, PubChem BioAssay (National Library of Medicine), ChemoTyper (https://chemotyper.org/, Molecular Networks GmbH and Altamira LLC 2013), Tox21 chemical structure database (ftp://ftp.epa.gov/dsstoxftp/DSSTox_Archive_20150930/TOX21S_DownloadFiles/), ChEBI chemical structure database (ftp://ftp.ebi.ac.uk/pub/databases/chebi/SDF/), Pharmacogene Variation Consortium CYP2A6 Allele Nomenclature (https://www.pharmvar.org/gene/CYP2A6), iCSS Dashboard V2 (US EPA ToxCast Phase II data), CTD (Comparative Toxicogenomics Database, http://ctdbase.org/), and DAVID (Database for Annotation, Visualization and Integrated Discovery) (https://david.ncifcrf.gov/home.jsp).
Search Process
The following search strings, in whole or in part, were applied to the databases listed above, when applicable: (“coumarin” [MeSH] OR “1,2-benzopyrone” OR “91-64-5 [RN]”) AND (“Neoplasms” [MeSH] OR “Cancer” [MeSH] OR “Mutation” [MeSH] AND “Toxicity” [MeSH] OR “Mechanism” [MeSH] OR “CYP2A6” [MeSH] OR “Metabolism” [MeSH] OR “Polymorphism” [MeSH]).
Additional databases listed above were then searched with appropriate search terms. Web of Science, for example, was searched by entering chemical terms (name, synonyms or CAS number) and refining the search by applying Web of Science categories Toxicology and/or Public, Environmental and Occupational Health.
In summary, more than 700 references, including government reports, peer-reviewed journal articles, and books, were identified through these search strategies up to September 2017. Among these, 257 references were cited in this document.
Pathway Analysis Using Gene Ontology and Kyoto Encyclopedia of Genes and Genomes Pathway Databases
We used the following approaches to analyze one set of toxicogenomic data published by Uehara et al, 12 and the data were from the rat liver following in vivo exposure to an oral dose of coumarin (150 mg/kg). To identify biological pathways altered by coumarin, we analyzed 136 probe sets of statistically significantly upregulated genes and 79 probe sets of statistically significantly downregulated genes.
We chose the National Institutes of Health-developed DAVID, an open-access web-based program with broad databases containing over 50 annotation categories from dozens of public databases, including Gene Ontology (GO) terms, PANTHER (Protein ANalysis THrough Evolutionary Relationships) GO terms, and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways 13 to perform GO and KEGG pathway analysis on these data.
Lists of the upregulated and downregulated genes identified by Uehara et al 12 were uploaded into DAVID 6.8 analysis wizard (https://david.ncifcrf.gov/tools.jsp), along with each gene’s unique Affy gene code (Affymetrix, Santa Clara, California). We analyzed these data using 2 different approaches selected from among those available in DAVID. In the first approach, we applied a functional annotation clustering tool to the gene lists. The DAVID functional annotation clustering tool uses a novel algorithm to measure relationships among the annotation terms based on the degrees of their coassociation genes, in order to group similar annotation terms into annotation clusters. This clustering function groups similar annotations together and makes the interpretation of multiple pathways easier, as compared with the second approach, the traditional chart report, which focuses on the individual annotation terms.
In the second approach, we applied the traditional functional annotation chart to the gene lists. The DAVID traditional functional annotation chart is an annotation-term-focused view which lists annotation terms and their associated genes. All results of the chart reports have to pass the default threshold criteria: P value ≤0.1 and gene count ≥2. For both of the above approaches, we used all default options in DAVID and chose a medium level of clustering options and stringency.
In DAVID, a more conservative modified Fisher exact test is used to measure gene enrichment by annotation terms. A selected annotation pathway with a P value of less than 0.1 (by default) means this pathway has more than a random gene enrichment association compared to the background rat transcriptome. The P values presented in Supplementary Tables B1 to B4 correspond to this more conservative version of the one-tailed Fisher exact test (online Functional Annotation Tool, https://david.ncifcrf.gov/helps/functional_annotation.html#).
Data output from the approach using functional annotation clustering are presented in Supplementary Table B1 for the upregulated genes (111 out of the 136 upregulated genes from Uehara et al 12 were recognized by DAVID) and supplementary Table B2 for the downregulated genes (69 out of the 79 downregulated genes from Uehara et al 12 were recognized by DAVID).
Data output from the approach using the traditional functional annotation chart, presenting only the pathways with P values less than 0.1; ie, DAVID default) is presented in Supplementary Table B3 for the upregulated genes and supplementary Table B4 for the downregulated genes.
Application of the Comparative Toxicogenomics Database to Identify Cancer-Associated Pathways and Biological Processes
The Comparative Toxicogenomics Database (CTD; http://ctdbase.org/), a public database providing manually curated information about chemical gene/protein interactions, and chemical–disease, gene–disease, GO–gene, and pathway–gene associations, was consulted to help interpret the results of our pathway analysis of the Uehara et al 12 data described above, in order to identify those pathways and biological processes that are likely associated with cancer.
The CTD uses text mining to sort literature. Each reference (abstract or full text) is read by a biocurator to identify interactions and relationships, and all curated data are supported by their source citations. The CTD also provides indirect “inferred” associations that are established via CTD-curated gene–disease, GO–gene, and pathway–gene associations. The CTD ratio of cancer to (all) disease categories (%) of a GO/pathway cluster represents its percentage of the associations with diseases that were specifically associated with cancer. For example, CTD inferred a cancer association with DNA replication. The number of inferred associations for DNA replication and all disease categories is 1,185, and the number of inferred associations for DNA replication and cancers is 418. Therefore, the cancer association ratio for DNA replication is 418 to 1,185, or 35.27%. The higher the CTD ratio of cancer to all disease, the stronger the cancer association. We used the CTD to identify the CTD ratio of cancer to all disease for each of the enriched gene annotation clusters (ie, P values <.05) identified through our pathway analysis (see Supplementary Tables B1 and B2).
Results
The results described here summarize the scientific literature we determined to be relevant to the carcinogenicity of coumarin. Included among these results are the findings from Office of Environmental Health Hazard Assessment’s (OEHHA) further examination of some data sets.
Carcinogenicity Studies in Humans
No cancer epidemiological studies on the effects of human exposure to coumarin were identified in literature searches conducted through September 2017.
Carcinogenicity Studies in Animals
A review of the carcinogenicity studies of coumarin in experimental animals identified 2 gavage studies in Fischer 344/N (F344/N) rats (1 in male Fisher rats and 1 in female Fisher rats), 6 feed studies in rats (3 in S-D, 1 in Osborne-Mendel, and 2 in an unspecified strain), 2 gavage studies in B6C3F1 mice, 2 dietary studies in CD-1 mice, and 2 dietary studies in Syrian golden hamsters. Table 1 lists these studies by species, strain, and route of administration, and each is described briefly below. A number of these studies were limited by small numbers of animals per group and inadequate reporting. Statistically significant and/or biologically important tumor findings are summarized in the text. In addition, a less-than-lifetime study in baboons 22 and 4 co-carcinogenicity studies with either the known carcinogens, 7,12-dimethylbenz[a]anthracene (DMBA) or benzo[a]pyrene (BP), were identified. The co-carcinogenicity studies were in Wistar rats, 23 S-D rats, ICR/Ha mice, 24 and Syrian golden hamsters. 25
Overview of Coumarin Animal Carcinogenicity Studies.
Abbreviations: F, female; M, male; NTP, National Toxicology Program.
a Calculated by Office of Environmental Health Hazard Assessment (OEHHA) based on gavage dosing of 5 days per week.
b As reported by study authors.
c Average calculated by OEHHA using values reported by study authors.
d Calculated by OEHHA using administered dose reported by study authors and standard body weight and food intake values from Gold and Zeiger. 21
Each study design is described in detail below, followed by information on the survival and body weight of the treated animals. Tumor findings and the pathology of the tumor types are described. A brief discussion of non-neoplastic findings may be included if the findings are considered treatment related and possibly related to tumor development.
Studies in rats
One hundred three–week gavage studies in male and female F344/N rats 14
Male and female F344/N rats were administered coumarin (>97% purity) in corn oil by gavage for up to 103 weeks. An additional 10 rats per sex per group were necropsied at 15 months for interim evaluation, except for the mid-dose group of male rats, where 9 additional male rats were necropsied. Additionally, a stop-exposure evaluation was conducted in male rats. For treatment-related tumor findings in the continuous exposure and stop-exposure regimens, see Tables 2 and 3, respectively.
Kidney Tumor Incidencea in Male F344/N Rats Administered Coumarin via Gavage 5 d/wk for 103 Weeks. 14,b
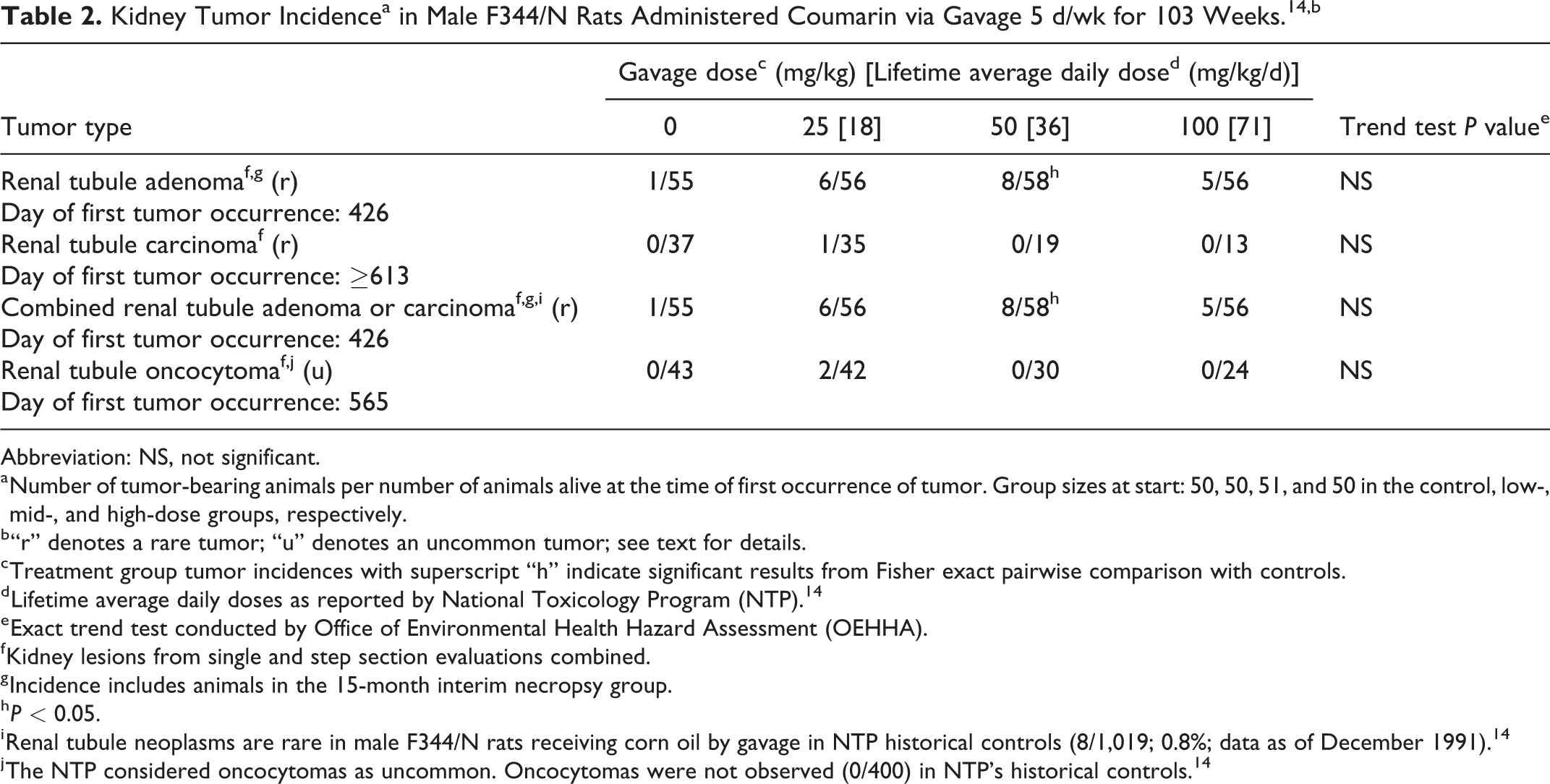
Abbreviation: NS, not significant.
a Number of tumor-bearing animals per number of animals alive at the time of first occurrence of tumor. Group sizes at start: 50, 50, 51, and 50 in the control, low-, mid-, and high-dose groups, respectively.
b “r” denotes a rare tumor; “u” denotes an uncommon tumor; see text for details.
c Treatment group tumor incidences with superscript “h” indicate significant results from Fisher exact pairwise comparison with controls.
d Lifetime average daily doses as reported by National Toxicology Program (NTP). 14
e Exact trend test conducted by Office of Environmental Health Hazard Assessment (OEHHA).
f Kidney lesions from single and step section evaluations combined.
g Incidence includes animals in the 15-month interim necropsy group.
h P < 0.05.
i Renal tubule neoplasms are rare in male F344/N rats receiving corn oil by gavage in NTP historical controls (8/1,019; 0.8%; data as of December 1991). 14
j The NTP considered oncocytomas as uncommon. Oncocytomas were not observed (0/400) in NTP’s historical controls. 14
Kidney Tumor Incidencea in Male F344/N Rats in 9- and 15-Month Stop Exposure Groupsb (Incidence From Control and 103-Week Continuous Exposure Groups Provided for Comparison). 14,c
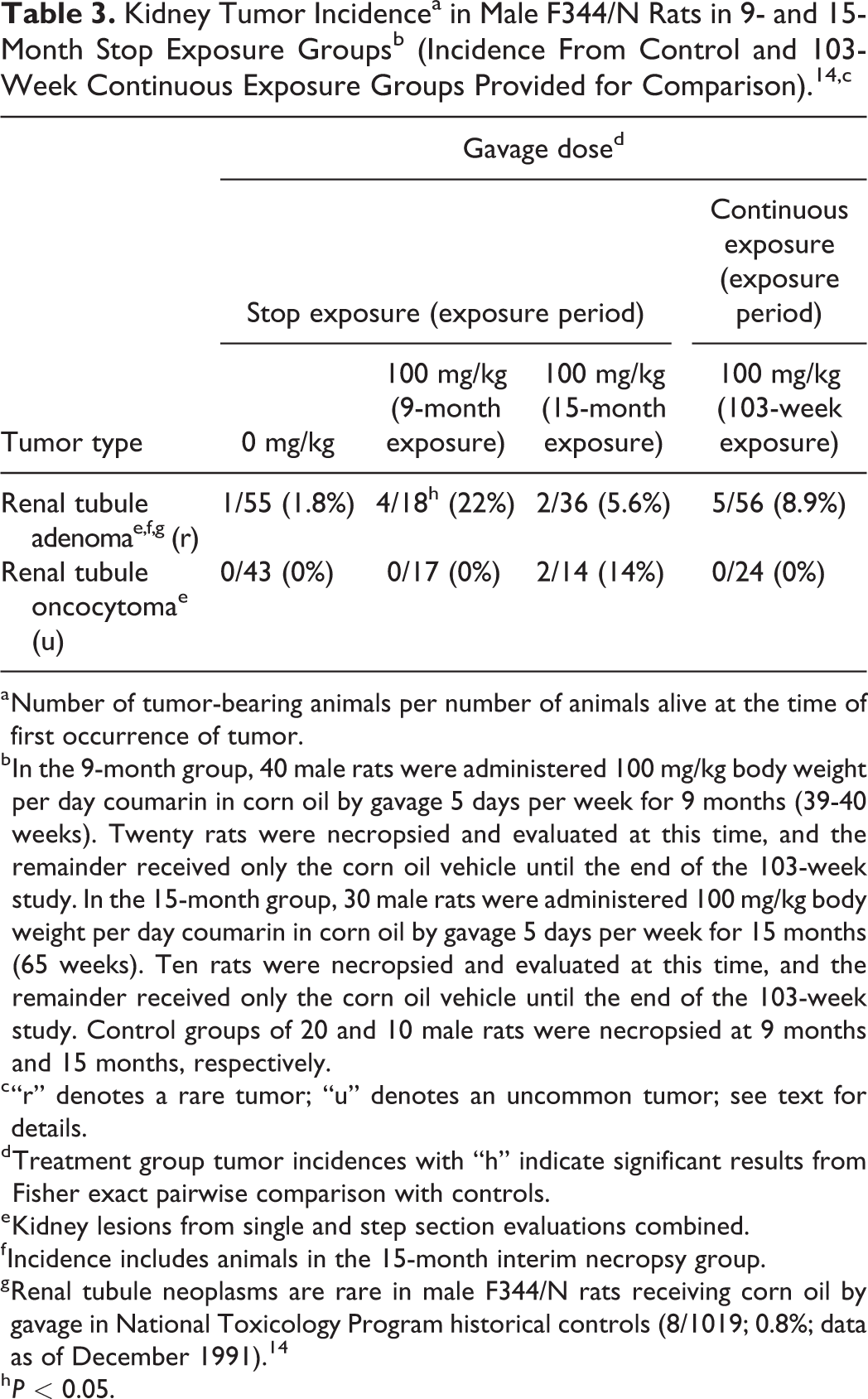
a Number of tumor-bearing animals per number of animals alive at the time of first occurrence of tumor.
b In the 9-month group, 40 male rats were administered 100 mg/kg body weight per day coumarin in corn oil by gavage 5 days per week for 9 months (39-40 weeks). Twenty rats were necropsied and evaluated at this time, and the remainder received only the corn oil vehicle until the end of the 103-week study. In the 15-month group, 30 male rats were administered 100 mg/kg body weight per day coumarin in corn oil by gavage 5 days per week for 15 months (65 weeks). Ten rats were necropsied and evaluated at this time, and the remainder received only the corn oil vehicle until the end of the 103-week study. Control groups of 20 and 10 male rats were necropsied at 9 months and 15 months, respectively.
c “r” denotes a rare tumor; “u” denotes an uncommon tumor; see text for details.
d Treatment group tumor incidences with “h” indicate significant results from Fisher exact pairwise comparison with controls.
e Kidney lesions from single and step section evaluations combined.
f Incidence includes animals in the 15-month interim necropsy group.
g Renal tubule neoplasms are rare in male F344/N rats receiving corn oil by gavage in National Toxicology Program historical controls (8/1019; 0.8%; data as of December 1991). 14
h P < 0.05.
Continuous exposure for 103 weeks in male rats
In the continuous exposure groups, survival of treated male rats was significantly lower than that of controls. Survival rates at week 77 were 43 (86%) of 50 in the control group, 42 (84%) of 50 in the low-dose group, 36 (71%) of 51 in the mid-dose group, and 29 (58%) of 50 in the high-dose group. Survival rates in the mid- and high-dose groups dropped precipitously as the study progressed after week 77. At week 89, survival rates were 35 (70%) of 50 in the control group, 32 (64%) of 50 in the low-dose group, 14 (28%) of 51 in the mid-dose group, and 10 (20%) of 50 in the high-dose group. Decreased survival of treated groups was attributed to treatment-related increases in severity of nephropathy. 14 Mean body weights of the mid- and high-dose groups were lower than those of controls. At 53 weeks, mean body weights in the mid- and high-dose groups were 6% and 14% lower, respectively, than controls, and 17% and 22% lower, respectively, at 89 weeks.
Kidney tumors were observed in male rats (Table 2). The kidneys in this study were initially examined by preparing a single section of each kidney. Since rare renal tubule adenomas were observed in all groups, additional step sections of the kidney were prepared, revealing additional kidney tumors. Renal tubule adenomas were characterized as “discrete, sometimes multinodular masses at least three times greater in diameter than an average tubule and composed of somewhat pleomorphic epithelial cells arranged in complex tubular structures and solid clusters.” 14 Renal tubule carcinomas were described as being larger than adenomas, with cellular pleomorphism, atypia, and central necrosis. Renal tubule adenomas and carcinomas are aggregated when evaluating study results. 26
Increases in renal tubule adenomas were observed in all treatment groups, and a rare renal tubule carcinoma was seen in 1 rat in the low-dose group. The renal tubule tumors (adenomas and adenoma and carcinomas combined) were considered rare by National Toxicology Program (NTP). 14 The increases in the incidence of renal tubule adenoma and renal tubule adenoma and carcinoma combined were statistically significant only in the mid-dose group compared to controls, but dose-related trends were not statistically significant at the P < 0.05 level. Two renal tubule oncocytomas, which are recognized as neoplasms distinct from renal tubule adenomas and carcinomas, 27,28 were observed in 2 males in the low-dose group. These tumors are considered uncommon by NTP. 14 Oncocytomas were characterized as “small, discrete nodules of uniform cells with dense, hyperchromatic nuclei and granular eosinophilic cytoplasm.” 14 Oncocytomas are reported to arise in the collecting ducts from oncocytic hyperplasia, usually grow very slowly in rats, and are considered benign and uncommon in F344 rats. 28 -30
Non-neoplastic pathology findings included significantly increased relative kidney weights of male rats in the high-dose group compared to controls at the 15-month interim necropsy. While nephropathy was observed in all groups, even controls, the severity grade of nephropathy increased with dose. All treated groups were statistically significantly different from controls in severity (assessed by the Mann-Whitney U test as reported by NTP 14 ). The NTP 14 characterized nephropathy by “glomerulosclerosis, thickening of tubule basement membrane, degeneration and atrophy of tubule epithelium, dilatation of tubule lumens by pale pink acellular material (hyaline casts), interstitial fibrosis, and chronic inflammation.” Degenerative changes were often accompanied by regeneration of tubule epithelium, the extent of which reflected the overall severity of the degenerative changes. Statistically significant increases in incidences of renal tubule hyperplasia were observed in the low- and mid-dose groups, but not the high-dose group, by pairwise comparison with controls (P ≤ 0.01). Hyperplasia was described as a single tubule filled with normal or slightly enlarged epithelial cells. The NTP 14 states that hyperplasia “was distinguished from the common regenerative epithelial changes commonly seen as a part of nephropathy and was considered a preneoplastic lesion. Hyperplasia, adenoma, and carcinoma were part of a morphological continuum and occurred in the cortex of the kidney.”
Several non-neoplastic findings were observed in the livers of treated male rats. Absolute and relative liver weights were increased in the high-dose group compared to controls at the 15-month interim evaluation. At this evaluation, a statistically significant increase in the incidence of hepatocellular degeneration was observed in treated rats, most often located in the centrilobular region, and characterized by “the presence of multiple small, clear, intracytoplasmic vacuoles” or fewer larger vacuoles typical of fatty change. Minimal to mild necrosis often accompanied hepatocellular degeneration; in a few rats, moderate to marked necrosis was present. Cytologic alterations were characterized by the presence of enlarged hepatocytes in the peripheral regions of liver lobules, with increased cytoplasmic basophilia, enlarged vesicular nuclei, and an increase in the number of cells in mitosis. Statistically significant increases in coagulative necrosis and fibrosis were observed in the livers of male rats in the treated groups at the end of the 2-year continuous exposure study. Liver fibrosis, characterized by bands of connective tissue, was considered a consequence of necrosis. 14 Bile duct hyperplasia increased in severity with increasing dose and was significantly increased in the high-dose group compared to controls (P ≤ 0.01). 14 The bile duct hyperplasia observed in this study was characterized by “increased profiles of well-differentiated bile ductules in the portal areas” but “did not exhibit the mucus cell metaplasia or epithelial dysplasia typical of cholangiofibrosis”. 14 Bile duct hyperplasia, a common aging lesion in rats, does not often progress to neoplasia. 31,32
Stop-exposure groups
Stop-exposure groups were included in the NTP 14 male rat study. Groups of 40 and 30 male rats were administered 100 mg/kg coumarin via gavage 5 d/wk for 9 and 15 months, when 20 and 10 rats were necropsied, respectively. The remaining 20 rats in each group were administered corn oil via gavage until the end of the 103-week study. Treatment-related effects on survival and body weight in the stop-exposure groups were consistent with those observed with 103-week continuous exposure. Survival was decreased in both the 9- and 15-month stop-exposure groups compared to controls. Nine (45%) of 20 males treated for 9 months and 2 (10%) of 20 males treated for 15 months survived until the end of the study. Decreased survival was attributed to treatment-related increases in the severity of renal nephropathy. Mean body weight of rats treated for 9 months was 16% less than controls at week 41 and 15% less at week 103, while mean body weight of rats treated for 15 months was 14% less than controls at week 41 and 22% less at week 103.
Tumor findings in the stop-exposure groups, presented in Table 3, were also consistent with findings observed with 103-week continuous exposure (see Table 2) in which rare renal tubule tumors were observed. A statistically significant increase in the incidence of renal tubule adenomas was observed in the 9-month stop-exposure group by pairwise comparison with controls at the end of the 103-week study. One renal tubule adenoma was observed at the 15-month interim evaluation, and 2 renal tubule adenomas were observed in the 15-month stop-exposure group at the end of the 103-week study. Two rats in the 15-month stop-exposure group also had uncommon renal tubule oncocytomas at the end of the 103-week study. Besides the single rare renal tubule adenoma observed at the 15-month interim evaluation, no additional treatment-related tumors were observed at the 9- and 15-month interim evaluations for the stop-exposure groups.
The NTP considered the non-neoplastic pathology findings of treatment-related liver and kidney lesions observed in the stop-exposure groups at the 9- and 15-month interim evaluations to be similar to those observed at the end of the 103-week continuous exposure study. The incidences and/or severity of the hepatic lesions observed in the stop-exposure groups returned to levels similar to controls following termination of coumarin exposure, indicating that the hepatocellular and biliary lesions were reversible. In contrast, the kidney lesions were largely irreversible in the stop-exposure groups and increased in severity with age. However, the severity of the nephropathy observed in the stop-exposure groups was less than that observed in the 103-week continuous exposure high-dose group.
Continuous exposure for 103 weeks in female rats
Mortality in dosed female rats was similar to that of controls. Mean body weights of the high-dose group were slightly lower than those of controls but were not statistically significantly different. Each kidney was examined by single and step sectioning. Rare kidney tumors were observed only in treated mid- and high-dose female rats in this study (Table 4). No kidney tumors were observed in the 15-month interim necropsy groups. 14
Kidney Tumor Incidencea in Female F344/N Rats Administered Coumarin via Gavage 5 d/wk for 103 Weeks. 14,b

a Number of tumor-bearing animals per number of animals alive at the time of first occurrence of tumor (day 699).
b “r” denotes a rare tumor; see text for details.
c Exact trend test conducted by Office of Environmental Health Hazard Assessment.
d Kidney lesions from single and step section evaluations combined. Renal tubule neoplasms are rare in female F344/N rats receiving corn oil by gavage in National Toxicology Program historical controls (2/1018; 0.2%; data as of December 1991). 14
Non-neoplastic pathology findings included a significant increase in relative kidney weights of female rats in the high-dose group compared to controls at the 15-month interim evaluation (organ weights not reported for other time points). Statistically significant increases in the incidence of nephropathy was observed in all treatment groups by pairwise comparison with controls, in both the 15-month interim evaluation groups (P ≤ 0.05) and the groups on test for 103 weeks (P ≤ 0.01). Additionally, nephropathy severity grade increased with dose, and all treated groups were statistically significantly different from controls by the Mann-Whitney U test (P ≤ 0.01; as reported by NTP 14 ). There was an increase in renal tubule hyperplasia in the treated rats compared to controls, although it did not reach statistical significance.
In female rats, absolute and relative liver weights were increased in the high-dose group compared to controls at the 15-month interim evaluation. A dose-related increase in severity of hepatocellular degeneration was observed in treated rats in both the 15-month interim evaluation groups and the 103-week test groups. Incidences of coagulative necrosis, fibrosis, and cytologic alterations of the liver were statistically significantly increased in the high-dose group at 103 weeks compared to controls.
Two-year feeding studies in male and female S-D rats (Animals in the 3 lowest dose groups were also exposed in utero and via lactation) 15
Male and female S-D rats (50/sex/group) were administered coumarin (>98% purity) in the diet at doses of 0, 333, 1,000, 2,000, 3,000, or 5,000 ppm. Rats receiving 333, 1,000, and 2,000 ppm coumarin were exposed in utero, during lactation, and following weaning until being euthanized. Rats receiving 3,000 and 5,000 ppm were exposed after weaning, beginning at 21 to 28 days of age, until being euthanized. Male and female rats were euthanized after 104 and 110 weeks of postweaning exposure, respectively. Additional “satellite groups” of animals (15/group) were included in each study and euthanized at week 104. These satellite groups were evaluated for a subset of the parameters examined in the main studies (hematology, clinical chemistry, urinalysis, gross necropsy, organ weight determinations, and microscopic examination only of gross lesions). Achieved intakes of coumarin were reported by the study authors to be 13, 42, 87, 130, and 234 mg/kg/d” in male rats and 16, 50, 107, 156, and 283 mg/kg/d in female rats. The lower doses (expressed as average daily dose) in these feeding studies are fairly comparable to the average daily doses in the NTP 14 gavage studies in Fischer rats, which were 18, 36, and 71 mg/kg/d.
Males: Survival at 104 weeks was below 50% in the controls and the groups dosed in utero and during lactation and was significantly decreased in male rats in the 333 ppm group compared to controls. Survival was greater than controls in the 2 highest dose groups, in which dosing began postweaning. Food consumption was significantly lower in the 3 highest dose groups compared to controls throughout the entire study, and dose-related reductions in body weight gain were observed in these same dose groups. Mean body weight in the high-dose group was approximately 43% lower than controls at 52 weeks and 35% lower at 104 weeks.
The study reported treatment-related increases in multiple types of liver tumors, as shown in Table 5. There were significant increases in incidences of metastasizing cholangiocarcinomas, nonmetastasizing cholangiocarcinomas, and hepatocellular adenomas and carcinomas (combined; referred to as benign and malignant parenchymal tumors by the study authors) in the highest dose group by pairwise comparison with controls. Significant dose–response trends were observed for each of these tumor types. Satellite groups were examined only for gross lesions, which potentially resulted in underascertainment of the number of liver tumors in these animals. Carlton et al 15 referred to liver tumors as benign and malignant parenchymal tumors, which is an older term for hepatocellular tumors. Hepatocellular adenomas and carcinomas arise from the same cell type, and adenomas can progress to carcinomas. For this reason, these 2 tumor phenotypes are aggregated when evaluating study results. 26 Rare cholangiocarcinomas were also observed, which are bile duct tumors that often metastasize and generally show invasive growth into blood vessels, lymph vessels, and connective tissue in the liver. 35 These tumors usually develop after application of high doses of chemicals, cause marked necrosis in the liver parenchyma, are often associated with significant liver toxicity, and generally only occur in the presence of hepatocellular neoplasms. 36
Liver Tumor Incidencea in Male S-D Rats Administered Coumarin in Feed for 104 Weeks (With In Utero and Lactational Exposure in the 3 Lowest Dose Groups). 15
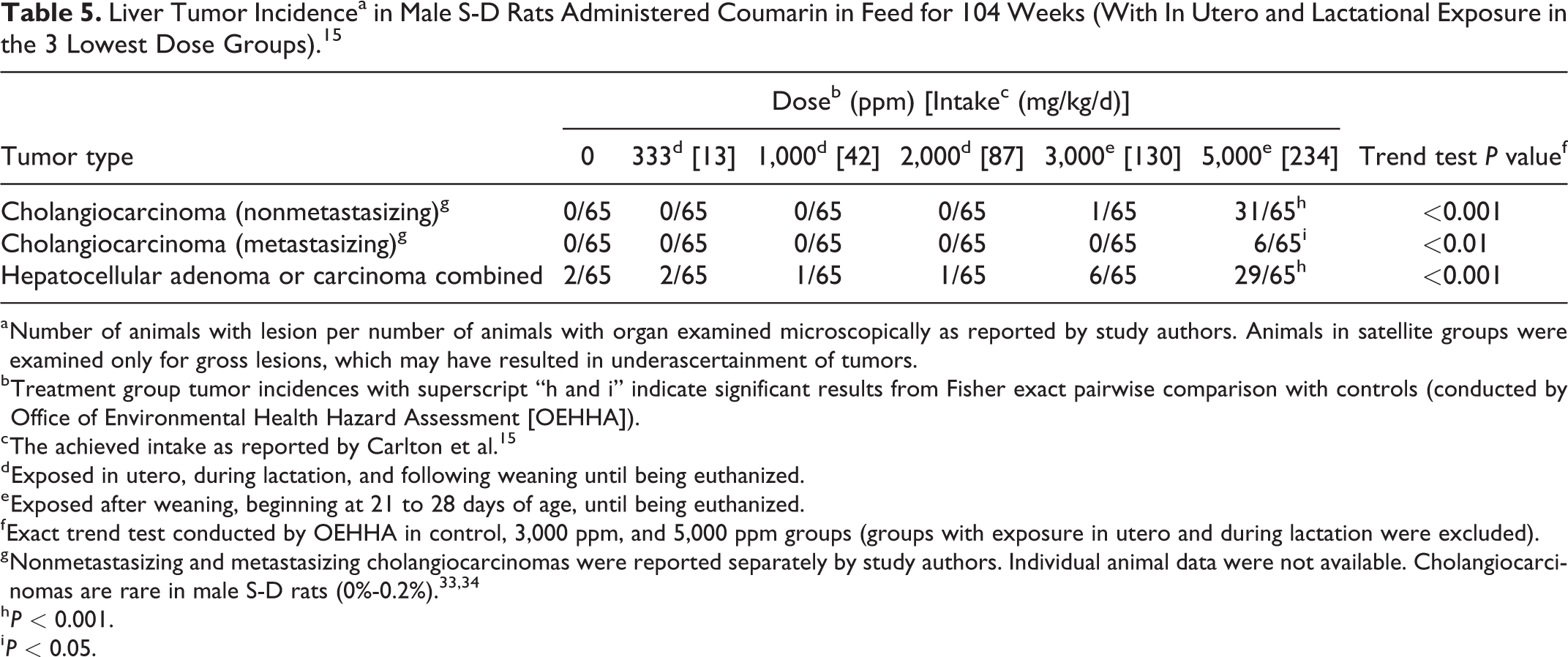
a Number of animals with lesion per number of animals with organ examined microscopically as reported by study authors. Animals in satellite groups were examined only for gross lesions, which may have resulted in underascertainment of tumors.
b Treatment group tumor incidences with superscript “h and i” indicate significant results from Fisher exact pairwise comparison with controls (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c The achieved intake as reported by Carlton et al. 15
d Exposed in utero, during lactation, and following weaning until being euthanized.
e Exposed after weaning, beginning at 21 to 28 days of age, until being euthanized.
f Exact trend test conducted by OEHHA in control, 3,000 ppm, and 5,000 ppm groups (groups with exposure in utero and during lactation were excluded).
g Nonmetastasizing and metastasizing cholangiocarcinomas were reported separately by study authors. Individual animal data were not available. Cholangiocarcinomas are rare in male S-D rats (0%-0.2%). 33,34
h P < 0.001.
i P < 0.05.
Carlton et al 15 considered the liver tumors at the 5,000 ppm group to be caused by exceedance of the maximum tolerated dose that led to hepatotoxicity, stating that “tumors were not metastatic and survival was significantly increased among rats in the two highest dose groups.” However, the increase in metastasizing cholangiocarcinomas in the high-dose group was statistically significant (P < 0.05). Additionally, survival was significantly better compared to controls in the 2 highest dose groups. Body weight gain was decreased in the 3 highest dose groups in this study. This, however, is not by itself an indication of an excessive high dose. It is possible that a reduction in food consumption and consequent reduced body weight gain may have contributed to the greater survival rates observed in the highest 2 dose groups. Feed restriction studies have shown that reduced body weight is associated with increased survival and reductions in spontaneous liver tumor incidence. 37 Since increased incidences were observed in this study, it appears that these liver tumors are treatment related.
With regard to dose selection in animal carcinogenicity studies, the US Environmental Protection Agency (EPA) Guidelines for Carcinogen Risk Assessment 38 explain that “an adequate high dose would generally be one that produces some toxic effects without unduly affecting mortality from effects other than cancer or producing significant adverse effects on the nutrition and health of the test animals.” At the same time, the US EPA Guidelines state that the high dose should not produce “significant increases in mortality from effects other than cancer,” to ensure that any effects observed are not due to excessive toxicity. Based on these guidelines, in the male rat study of Carlton et al, 15 the greater survival of the treated groups compared to the control group indicates that the maximum tolerated dose was not exceeded.
Non-neoplastic liver effects reported in this study were an increase in relative liver weights in the 2 highest dose groups, an increased incidence of cholangiofibrosis (considered to be a preneoplastic lesion) in the highest dose group, and an increase in alkaline phosphatase (ALP; doses not specified). This lesion can be difficult to diagnose and is sometimes characterized as cholangiocarcinoma when there is extensive involvement of the liver. However, cholangiofibrosis is considered to be an early proliferative lesion on the continuum of proliferative lesions that progress to cholangiofibroma and then to cholangiocarcinoma with time. 36,39 Cholangiofibrosis is not considered to be a spontaneous lesion and is not typically seen in untreated rats. 39 Cholesterol levels of all treated groups except the highest dose group were elevated throughout the study, an indication of altered liver function.
Females: Survival at 110 weeks was below 50% in the controls and the groups dosed in utero and during lactation. Survival was greater than controls in the 2 highest dose groups, in which dosing began postweaning. Food consumption was significantly lower in the 3 highest dose groups compared to controls throughout the entire study, and dose-related reductions in body weight gain were observed in these same dose groups. Mean body weight in the high-dose group was approximately 44% lower than controls at 52 weeks and 46% lower at 110 weeks. These reductions in food consumption and body weight may have contributed to the greater survival rates observed in the highest 2 dose groups.
The study reported treatment-related increases in multiple types of liver tumors as shown in Table 6. There were significant increases in incidences of nonmetastasizing cholangiocarcinoma and hepatocellular adenoma and carcinoma (combined; referred to as benign and malignant parenchymal tumors by the study authors) in the highest dose group by pairwise comparison with controls. Significant dose–response trends were observed for each of these tumor types. One metastasizing cholangiocarcinoma was observed in the highest dose group. Satellite groups were examined only for gross lesions, which potentially resulted in underascertainment of the number of liver tumors in these animals.
Liver Tumor Incidencea in Female S-D Rats Administered Coumarin in Feed for 104 or 110 Weeks (With In Utero and Lactational Exposure in the 3 Lowest Dose Groups). 15
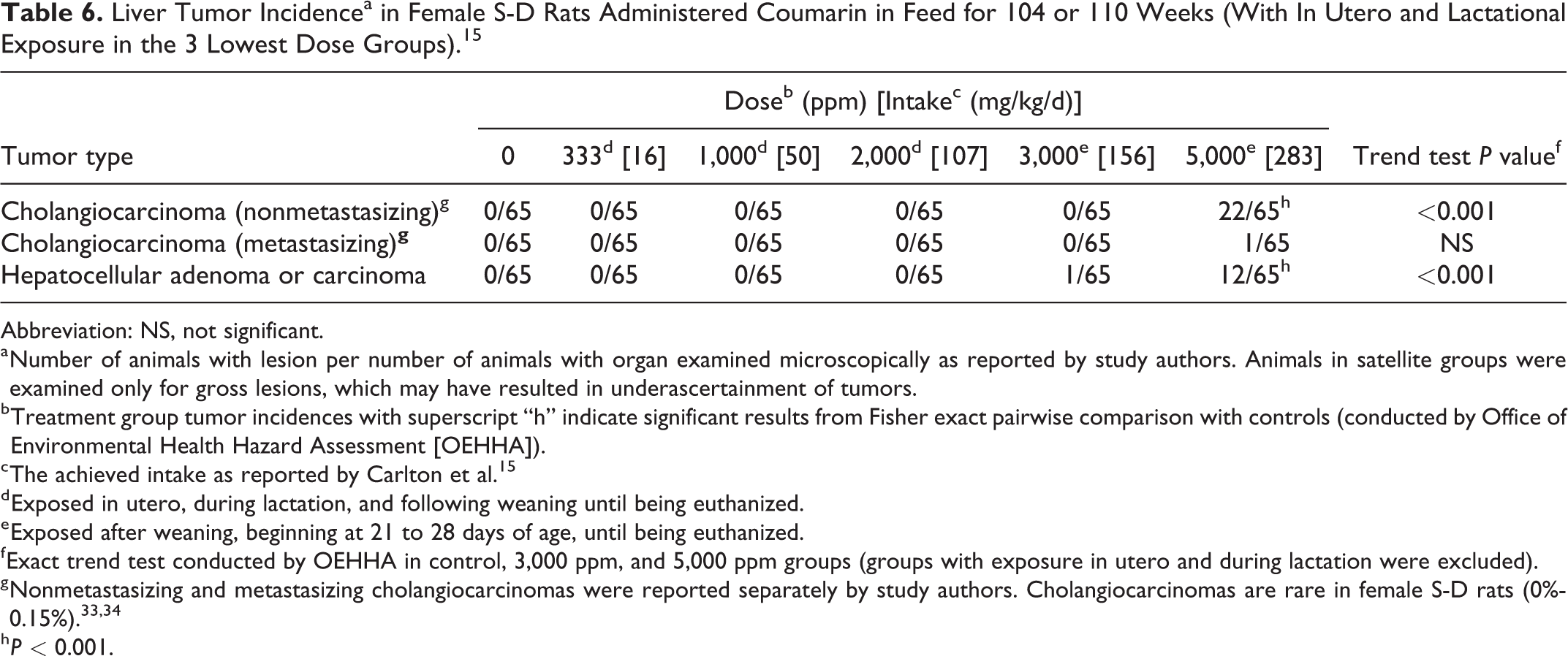
Abbreviation: NS, not significant.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by study authors. Animals in satellite groups were examined only for gross lesions, which may have resulted in underascertainment of tumors.
b Treatment group tumor incidences with superscript “h” indicate significant results from Fisher exact pairwise comparison with controls (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c The achieved intake as reported by Carlton et al. 15
d Exposed in utero, during lactation, and following weaning until being euthanized.
e Exposed after weaning, beginning at 21 to 28 days of age, until being euthanized.
f Exact trend test conducted by OEHHA in control, 3,000 ppm, and 5,000 ppm groups (groups with exposure in utero and during lactation were excluded).
g Nonmetastasizing and metastasizing cholangiocarcinomas were reported separately by study authors. Cholangiocarcinomas are rare in female S-D rats (0%-0.15%). 33,34
h P < 0.001.
Similar to the male rat study, the study authors implied that the maximum tolerated dose was exceeded in this study in the 3 highest dose groups and cited the large body weight decrements in these groups. Increased liver weights were found in the 4 highest dose groups, increased incidences of cholangiofibrosis were observed in the highest dose group, and increases in blood potassium, ALP, and glutamic-pyruvic transaminase were noted (doses not specified). However, female rats in the 2 highest dose groups had increased survival compared to controls. As explained above in the discussion of the male rat study, these observations do not support a conclusion that the liver tumors observed in the higher dose groups were the result of excessive toxicity.
Seventy-eight-week feeding study in male S-D rats 16
Male S-D rats were given the control diet or administered coumarin in the diet at a dose of 5,000 ppm. Groups of 5 control and 5 treated animals were necropsied at 4, 12, 14, 18, 22, 26, 30, 52, and 78 weeks. There was no difference in survival between the treated and control groups, but food intake and body weights were reduced in the treated animals. No treatment-related tumors were reported. Cholangiofibrosis was observed in the livers of the majority of rats treated from 18 to 78 weeks. Evans et al 16 reported that cholangiofibrotic lesions “were particularly prominent in animals killed at 18 months (78 weeks) and were reminiscent of cholangiocarcinoma in other species although no evidence of local invasion or of metastasis was found.” 16 The utility of this study for assessing the carcinogenicity of coumarin is limited by a number of factors, including the less-than-lifetime study duration, numerous early interim necropsies, small numbers of animals per interim necropsy, administration of coumarin at a single dietary concentration, and inadequate reporting. Cholangiofibrosis is a potential precursor to biliary neoplasia. As noted earlier, cholangiofibrosis, cholangiofibroma, and cholangiocarcinoma is a morphological continuum, and there are not specific criteria for separation of the various categories. 36
Two-year feeding study in male and female Osborne-Mendel rats 17
Coumarin was administered in the diet at 0, 1,000, 2,500, or 5,000 ppm to groups of 5 to 7 male and female Osborne-Mendel rats for 2 years. Reporting of the results for male and female rats was combined. No information on survival of rats was mentioned in the study. The study noted there was “growth retardation” and that food consumption, which was measured over the first year, was normal. However, the study publication did not provide the data on body weight or food consumption. No treatment-related tumors were reported. 17
Liver damage was observed as focal proliferation of bile ducts with cholangiofibrosis, fatty change, and focal necrosis in the 5,000 ppm dose group. The study reported that the 2,500 ppm dose group had minimal to slight proliferation of the bile ducts with fatty change and focal necrosis in the hepatic parenchyma, but cholangiofibrosis was not observed in this group. It reported that there was no effect in the 1,000 ppm dose group. This study was limited by the small number of animals per group and inadequate reporting.
Two-year feeding studies in male and female Albino rats 18,19
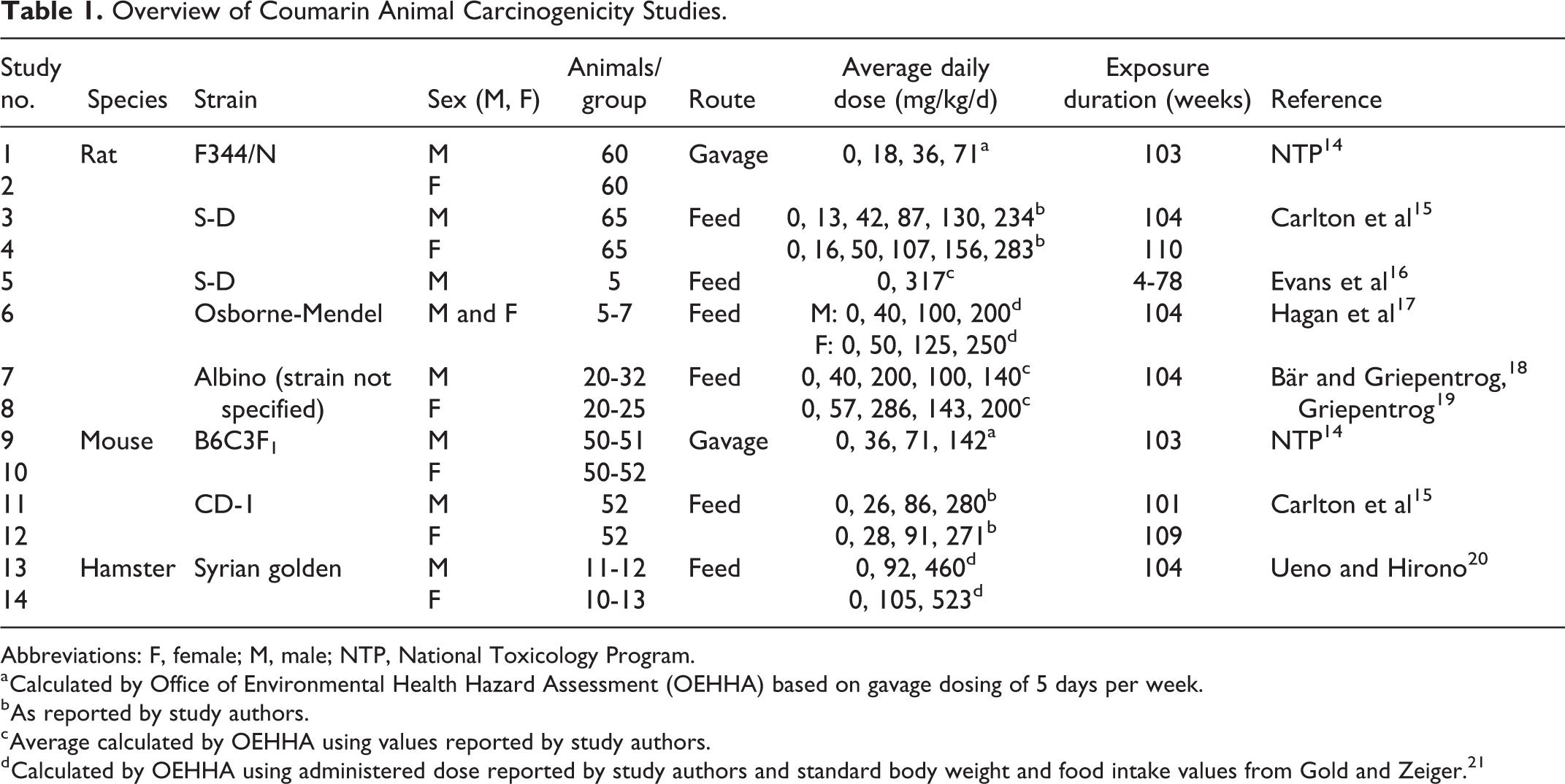
Coumarin was administered to male and female albino rats (strain not specified) in feed for up to 2 years. The “main studies” were comprised of 3 groups: control, 1,000 ppm, and 5,000 ppm coumarin. The “additional studies” were comprised of 4 groups: control, 2,500 ppm, and two 6,000 ppm coumarin groups, and were started after the “main studies.” Bär and Griepentrog 18 was the original publication; Griepentrog 19 provided additional detail about the studies. The papers did not specify why there were two 6,000 ppm dose groups per sex. Food consumption was reduced by approximately half in the 6,000 ppm groups. The OEHHA estimated the lifetime average daily doses using standard body weights and reported food intake values. In the “main studies,” the lifetime average daily doses for the 1,000 ppm dose groups were estimated to be 40 and 57 mg/kg/d for males and females, respectively, and for the 5,000 ppm groups were 200 and 286 mg/kg/d for males and females, respectively. In the “additional studies,” lifetime average daily doses for the 2,500 ppm groups were estimated to be 100 and 143 mg/kg/d for males and females, respectively, and for the 6,000 ppm groups were 140 and 200 mg/kg/d for males and females, respectively. Table 7 indicates the doses, number of animals per group, incidences of cholangiocarcinoma, and number of animals alive at 18 months. The authors reported that survival was decreased in treated animals compared to controls at 1.5 and 2 years.
Abbreviation: NR, not reported.
a Food intake was reduced in these 6,000 ppm dose groups.
b Some animals had both benign bile duct adenomas and carcinomas.
c Denominator not reported.
The reported liver carcinomas are shown in Table 7. In addition, Griepentrog 19 stated that rats in the 1,000 and 2,500 ppm dose groups developed a few small benign bile duct adenomas and displayed bile duct proliferation; however, the numbers of animals with these lesions were not reported. All carcinomas were reported to be bile duct carcinomas (ie, cholangiocarcinomas), with some evidence of extrahepatic metastasis, and extensive expansion and destruction of the liver parenchyma. 18 Griepentrog 19 stated that the cell and nucleus types, infiltration and destruction of liver tissues, bile duct structures, and lack of bile pigment observed in these lesions indicated that they were bile duct carcinomas.
The diagnosis of the lesions in these studies of Bär and Griepentrog 18 and Griepentrog 19 as cholangiocarcinomas has been questioned 16,40 and controversy surrounding the actual diagnosis of these lesions persists. 4 Cohen 40 noted that 2 external pathologists from the British Industrial Biological Research Association had examined the slides and had concluded that “the cytological changes in the bile ducts were not regarded as unequivocal evidence of a carcinomatous process and the possible occurrence of metastasis were excluded” (Cohen, 40 citing personal communication with DM Conning and JC Evans). Cohen 40 also referenced a note published in Food and Cosmetics Toxicology 41 that suggested the photomicrographs published by Bär and Griepentrog 18 were more consistent with the diagnosis of cholangiofibrosis than with cholangiocarcinoma. However, the distinction between the diagnosis of cholangiofibrosis, cholangiofibroma, or cholangiocarcinoma is not well defined, and there are not specific criteria for making these determinations. 36
Studies in mice
One hundred three-week gavage studies in male and female B6C3F1 mice 14
Male and female B6C3F1 mice were administered coumarin (> 97% purity) in corn oil by gavage for up to 103 weeks (Tables 8 and 9). An additional 20, 20, 20, and 19 male mice and 18, 20, 19, and 19 female mice from the control, low-, mid-, and high-dose groups, respectively, were necropsied at 15 months for interim evaluation. Only 5 to 10 animals per dose group in the 15-month interim evaluation groups were examined microscopically.
Incidencea of Treatment-Related Lesions in Male B6C3F1 Mice Administered Coumarin via Gavage 5 d/wk for 103 Weeks. 14,b
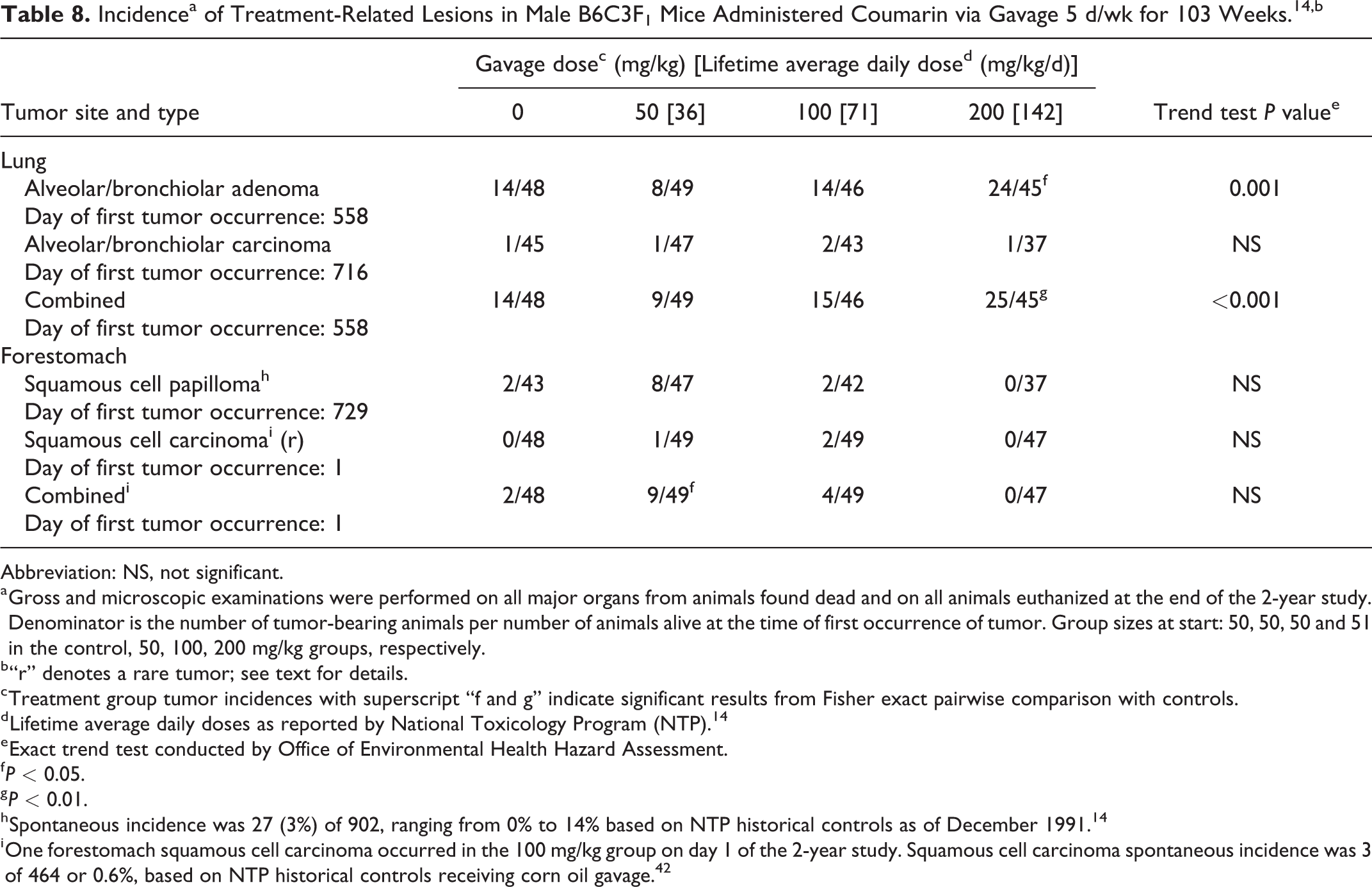
Abbreviation: NS, not significant.
a Gross and microscopic examinations were performed on all major organs from animals found dead and on all animals euthanized at the end of the 2-year study. Denominator is the number of tumor-bearing animals per number of animals alive at the time of first occurrence of tumor. Group sizes at start: 50, 50, 50 and 51 in the control, 50, 100, 200 mg/kg groups, respectively.
b “r” denotes a rare tumor; see text for details.
c Treatment group tumor incidences with superscript “f and g” indicate significant results from Fisher exact pairwise comparison with controls.
d Lifetime average daily doses as reported by National Toxicology Program (NTP). 14
e Exact trend test conducted by Office of Environmental Health Hazard Assessment.
f P < 0.05.
g P < 0.01.
h Spontaneous incidence was 27 (3%) of 902, ranging from 0% to 14% based on NTP historical controls as of December 1991. 14
i One forestomach squamous cell carcinoma occurred in the 100 mg/kg group on day 1 of the 2-year study. Squamous cell carcinoma spontaneous incidence was 3 of 464 or 0.6%, based on NTP historical controls receiving corn oil gavage. 42
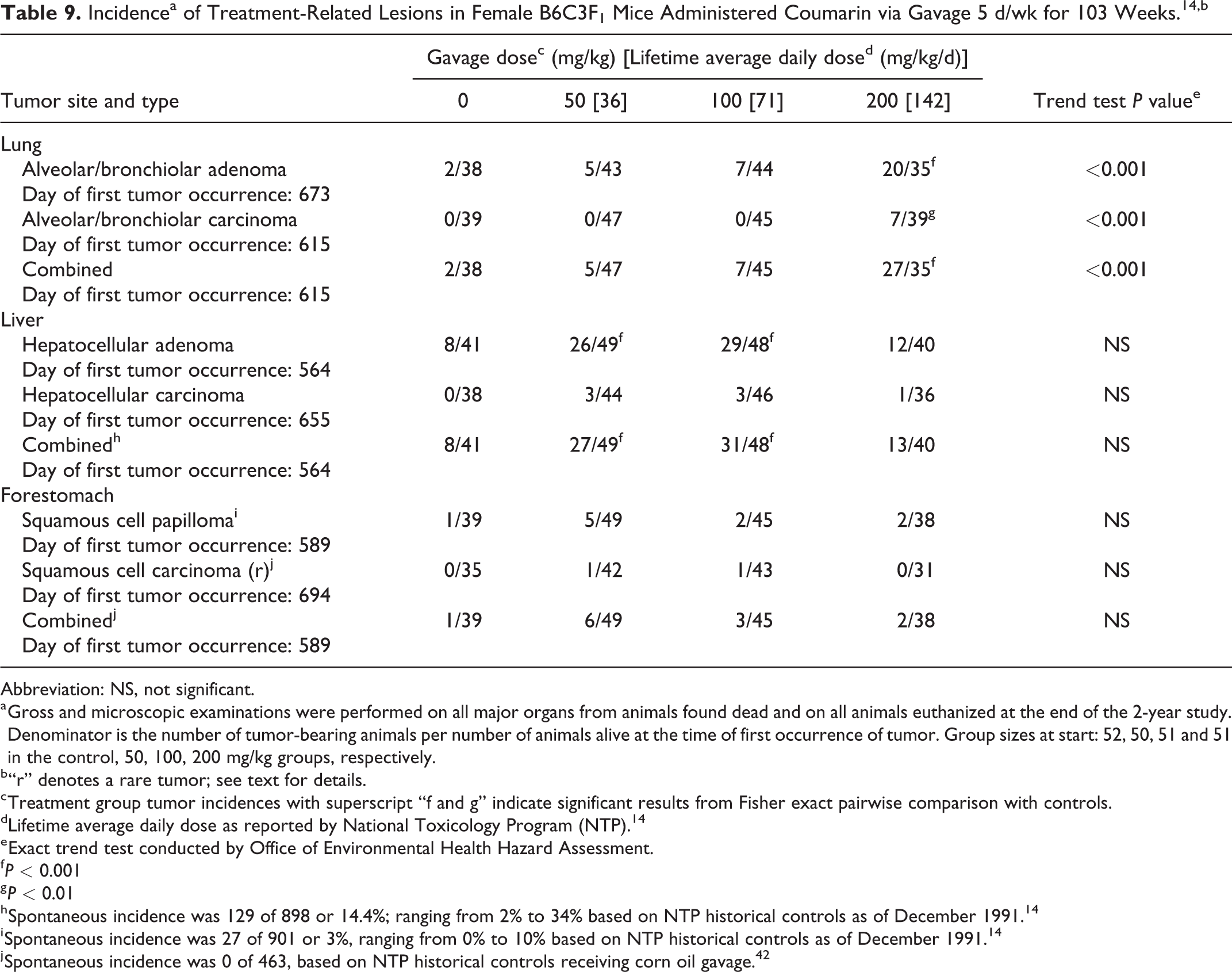
Incidencea of Treatment-Related Lesions in Female B6C3F1 Mice Administered Coumarin via Gavage 5 d/wk for 103 Weeks. 14,b
Abbreviation: NS, not significant.
a Gross and microscopic examinations were performed on all major organs from animals found dead and on all animals euthanized at the end of the 2-year study. Denominator is the number of tumor-bearing animals per number of animals alive at the time of first occurrence of tumor. Group sizes at start: 52, 50, 51 and 51 in the control, 50, 100, 200 mg/kg groups, respectively.
b “r” denotes a rare tumor; see text for details.
c Treatment group tumor incidences with superscript “f and g” indicate significant results from Fisher exact pairwise comparison with controls.
dLifetime average daily dose as reported by National Toxicology Program (NTP). 14
e Exact trend test conducted by Office of Environmental Health Hazard Assessment.
f P < 0.001
g P < 0.01
h Spontaneous incidence was 129 of 898 or 14.4%; ranging from 2% to 34% based on NTP historical controls as of December 1991. 14
i Spontaneous incidence was 27 of 901 or 3%, ranging from 0% to 10% based on NTP historical controls as of December 1991. 14
j Spontaneous incidence was 0 of 463, based on NTP historical controls receiving corn oil gavage. 42
Males: No significant differences in survival were observed between the control and treated male B6C3F1 mice. The mean body weights of the high-dose group were 3% to 10% lower but not statistically significantly different from those of controls from week 10 to 81 and were similar to controls at the end of the study.
Treatment-related tumor incidences observed in male mice are summarized in Table 8. Statistically significant increases in alveolar/bronchiolar adenomas and alveolar/bronchiolar adenomas and carcinomas combined were observed in the high-dose group compared to controls, with positive dose–response trends. Alveolar/bronchiolar adenomas and carcinomas may originate from alveolar type II cells or Clara cells. 43 Alveolar/bronchiolar adenomas in mice are considered to have the potential to progress to carcinomas and are aggregated when evaluating study results. 26 In the 15-month interim evaluation groups, alveolar/bronchiolar adenomas were observed in 2 out of 2 low-dose animals examined and 3 out of 9 high-dose animals examined. Since not all lung tissues were examined histopathologically in the interim evaluation groups, lung tumor findings in these groups are not included in Table 8.
In addition, the incidence of forestomach squamous cell papillomas and carcinomas combined was significantly increased (P < 0.05) in the low-dose group. Forestomach squamous cell papillomas occurred more frequently in the low-dose group than in controls and slightly exceeded the range of NTP historical controls (Table 8). The incidence in the mid-dose group was similar to controls, and no forestomach papillomas were observed in the high-dose group. One forestomach squamous cell carcinoma was observed in the low-dose group, 2 in the mid-dose group, and none in the high-dose group or in controls. Forestomach tumors were not reported in the 15-month interim evaluation groups. Forestomach carcinomas are considered rare in untreated male B6C3F1 mice. 14 The forestomach squamous cell papillomas observed in these studies were described as consisting of thickened, folded epithelium with a fibrovascular core. Differentiation of the epithelium within the papillomas was normal and there were no atypical cellular changes. The squamous cell carcinomas consisted of cords of stratified squamous epithelium, which invaded the submucosa and muscularis. 14 Forestomach squamous cell papillomas are considered to have the potential to progress to carcinomas. 26
The NTP report concluded that the increase in forestomach papillomas in male mice may have been related to coumarin administration but noted that the incidence in the low-dose group was not significantly higher than the controls and there was not a corresponding increase over a 4-fold dose range from 50 to 200 mg/kg. 14 There was a statistically significant increase by pairwise comparison of combined papillomas and carcinomas in the low-dose group with the controls (Table 8).
Females: No significant differences in survival were observed between the control and treated female B6C3F1 mice. The mean body weights of the high-dose group were slightly, but not statistically significantly, lower than those of controls from week 11 to 49 (3%-18% lower) and were about 12% lower at the end of the study.
Treatment-related tumor incidences observed in female mice are summarized in Table 9. Statistically significant increases in alveolar/bronchiolar adenomas, carcinomas, and adenomas and carcinomas combined were observed in the high-dose group compared to controls, with positive dose–response trends. In the 15-month interim evaluation groups, alveolar/bronchiolar adenomas were observed in one out of 1 mid-dose animals examined and 2 out of 9 high-dose animals examined. Since not all lung tissues were examined histopathologically in the interim evaluation groups, lung tumor findings in these groups are not included in Table 9. As noted previously, alveolar/bronchiolar adenomas in mice are considered to have the potential to progress to carcinomas and are aggregated when evaluating study results. 26
There were significant increases in hepatocellular adenomas and adenomas and carcinomas combined in the low- and mid-dose groups compared to the controls. The incidences of liver tumors in the low- and mid-dose groups exceeded the NTP historical control incidence (Table 9). According to NTP, the lower incidence of hepatocellular neoplasms in the high-dose group may be related to the reduced body weight of this group. In the 15-month interim evaluation groups, hepatocellular adenomas were observed in 1 out of 8 control animals examined. Since not all liver tissues were examined histopathologically in the interim evaluation groups, these findings are not included in Table 9. Hepatocellular adenomas arise from the same cell type as carcinomas and are considered to have the potential to progress to carcinomas. These 2 tumor phenotypes are aggregated when evaluating study results. 26,44 An increased incidence of eosinophilic foci of the liver, which are morphologically similar to adenomas and are considered to be preneoplastic lesions in mice, was observed in low- and mid-dose females.
There were increased incidences of forestomach squamous cell papillomas and carcinomas in each dose group, but these increases were not statistically significantly different from controls. The observed increases in forestomach squamous cell papillomas were within the range of NTP historical control incidence. One forestomach squamous cell carcinoma was observed in each of the low- and mid-dose groups. Forestomach squamous cell carcinomas are considered rare in female B6C3F1 mice. 14 No forestomach tumors were reported in the 15-month interim evaluation groups. The NTP report concluded that the increase in forestomach papillomas in female mice may have been related to coumarin administration but noted that the incidences in the low-dose group were not significantly higher than the controls and there was no corresponding increase over a 4-fold dose range from 50 to 200 mg/kg.
Two-year feeding studies in male and female CD-1 mice
Male and female CD-1 mice (52/sex/group) were administered coumarin (>98% purity) in the diet at doses of 0, 300, 1,000, and 3,000 ppm for 2 years. 15 The 2 lower doses (expressed as average daily dose) in these feeding studies are fairly comparable to the average daily doses received by the low- and mid-dose groups in the NTP 14 gavage studies in B6C3F1 mice, which were 36 and 71 mg/kg/d (Tables 10 and 11).
Incidencea of Treatment-Related Lesions in Male CD-1 Mice Administered Coumarin in Feed for 101 Weeks. 15
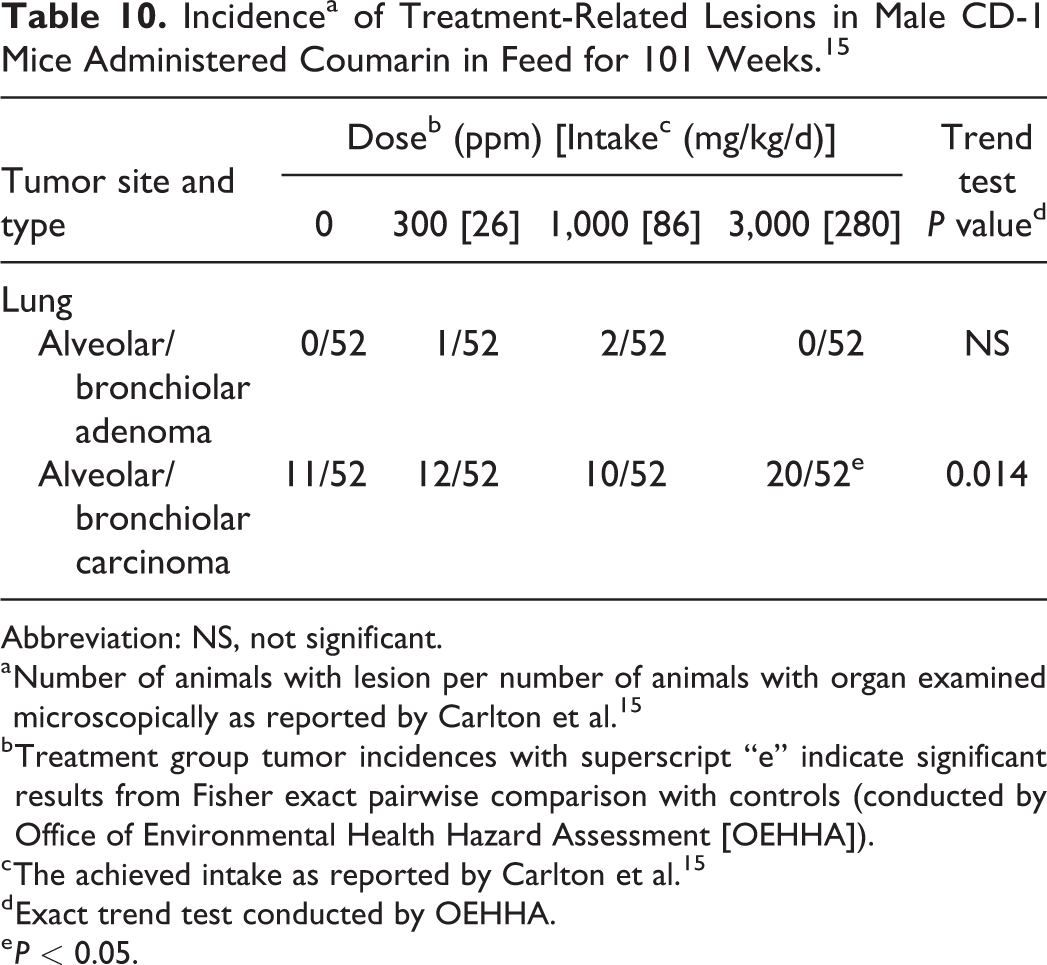
Abbreviation: NS, not significant.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by Carlton et al. 15
b Treatment group tumor incidences with superscript “e” indicate significant results from Fisher exact pairwise comparison with controls (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c The achieved intake as reported by Carlton et al. 15
d Exact trend test conducted by OEHHA.
e P < 0.05.
Incidencea of Treatment-Related Lesions in Female CD-1 Mice Administered Coumarin in Feed for 109 Weeks. 15
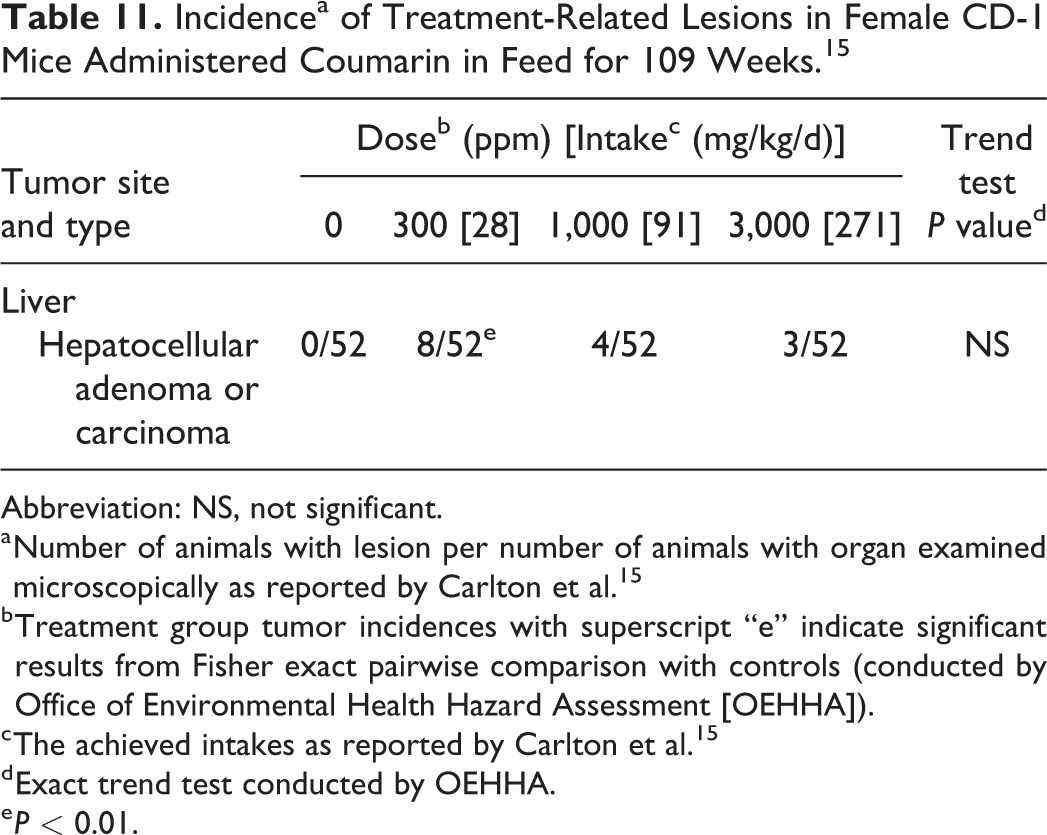
Abbreviation: NS, not significant.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by Carlton et al. 15
b Treatment group tumor incidences with superscript “e” indicate significant results from Fisher exact pairwise comparison with controls (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c The achieved intakes as reported by Carlton et al. 15
d Exact trend test conducted by OEHHA.
e P < 0.01.
Males: Survival of treated male mice was similar to that of controls. Body weight gains in treated males were significantly reduced compared to controls, with an 18% reduction in the 3,000 ppm group and a 10% reduction in the 1,000 ppm group at week 52. Food intake in the 3,000 ppm group was marginally lower than the controls.
Lung tumors were observed in male CD-1 mice (Table 10). Carlton et al 15 referred to the observed lung tumors as pulmonary adenomas and adenocarcinomas, but these tumors are typically referred to as alveolar/bronchiolar adenomas and carcinomas. A statistically significant increase in alveolar/bronchiolar carcinomas was observed in the high-dose group compared to controls, with a positive dose–response trend. The authors reported that the incidence of alveolar/bronchiolar carcinomas was within the laboratory historical control range for CD-1 male mice (range not reported). There were no effects on organ weights, as reported by the study authors.
Females: Survival of treated female CD-1 mice was similar to that of controls, as were body weights. Food consumption was reported to be similar across all dose groups.
Liver tumors were observed in female CD-1 mice (Table 11). A statistically significant increase in benign and malignant parenchymal tumors combined (hepatocellular adenomas and carcinomas) was observed in low-dose female mice compared to controls. The increases in liver tumors observed in the mid- and high-dose groups did not reach statistical significance. Non-neoplastic pathology findings included a significant increase in absolute/relative liver weights in the high-dose group compared to controls.
Studies in hamsters
Two-year feeding studies in male and female Syrian golden hamsters 20
Coumarin was administered to 8-week old male and female Syrian golden hamsters (group sizes of 12, 11, 11 in males, and 12, 13, 10 in females for the control, low-, and high-dose group, respectively) via the diet at levels of 0%, 0.1%, and 0.5% for up to 2 years. The OEHHA estimated the average daily doses to be 0, 92, and 460 mg/kg/d for the males and 0, 105, and 523 mg/kg/d for the females, based on default body weights of 125 g for male and 110 g for female hamsters and food intake of 11.5 g/d for both males and females. 21
In both the male and female studies, a transient 20% reduction in food intake was observed in the coumarin-treated groups after 1 month. Food intakes returned to control levels by month 5 in both studies. No treatment-related effects on growth were observed in either study.
In the male hamster study, survival was poor in the low-dose treatment group. At 22 months, only 2 (18%) of 11 survived in the low-dose group compared to 10 (90%) of 11 in the high-dose and 9 (75%) of 12 in the control group. No treatment-related tumors were observed. The utility of this study for assessing the carcinogenicity of coumarin is limited by the small numbers of animals per group and poor survival in the low-dose group.
In the female hamster study, survival was poor in all groups, including the control group. Survival at 22 months was 0% in the low-dose group and 2/10 (20%) in the high-dose group; survival in the controls was 3 (25%) of 12. No significant increases in tumors were observed in the treated groups; however, 2 pancreatic islet cell carcinomas were observed in the high-dose group, with none in the control or low-dose groups. Pancreatic islet cell tumors are uncommon in female hamsters. 45 The utility of this study for assessing the carcinogenicity of coumarin is limited by the small numbers of animals per group and poor survival in the control and treated groups.
Less-than-lifetime study in baboons 22
Coumarin was administered in the diet to 34 male baboons, each of 3 different species (Papio anubis, Papio hamadryas, Papio cynocephalus), for up to 2 years. Groups of 8, 8, 8, 6, and 4 animals were fed diets containing 0, 2.5, 7.5, 22.5, or 67.5 mg/kg/d coumarin, respectively. The daily intake of one animal receiving 22.5 mg/kg/d was increased to 67.5 mg/kg/d at 18 months. One animal from each of the groups treated with 0, 22.5, and 67.5 mg/kg/d was necropsied at 16 months. All animals in the 2.5 mg/kg/d group and 4 animals in the 7.5 mg/kg/d group were necropsied at 18 months. All remaining animals were euthanized at 24 months. The typical life span of these species of baboon ranges from 15 to 40 years. Thus, the length of this study was significantly less than lifetime.
No treatment-related tumors were reported. This is not unexpected given the small numbers of animals per group and the short study duration, which, depending on the baboon species, ranged from approximately 5% to 13% of the animals’ expected life span.
Non-neoplastic pathology findings included a significant increase in relative liver weights in the 67.5 mg/kg/d group compared to the controls. Liver hypertrophy and dilatation of the endoplasmic reticulum were also observed in the 67.5 mg/kg/d group. There was no evidence of treatment-related biliary hyperplasia or fibrosis. Although there was not a statistically significant difference in biochemical or histochemical parameters between treated and control groups, the authors characterized the effects on relative liver weight, liver hypertrophy, and dilatation of the endoplasmic reticulum as evidence of early cell damage in the liver.
Co-carcinogenicity studies
Co-carcinogenicity study in female Wistar rats 23
This study investigated the effects of coumarin on mammary gland carcinogenesis induced by DMBA in female Wistar rats. All 4 groups of female Wistar rats (32/group) received DMBA (2 mg/injection) intravenously (IV) via the tail vein on days 50, 53, and 56 of life. Group 1 received only DMBA. Groups 2 and 3 received coumarin before, during, and after dosing with DMBA, with coumarin administered via drinking water to group 2 and via gavage to group 3. Group 4 received coumarin only after DMBA administration, via gavage. There were no significant differences in the mean body weights between groups. Six rats from each group were necropsied on day 50 (after receiving the first DMBA injection) and 10 rats from each group were necropsied on day 57. The remaining rats were euthanized on day 198 and examined for mammary tumors.
Table 12 shows the mammary tumor incidences observed in the study. All animals in group 1 (receiving DMBA only) had mammary gland adenocarcinomas and 2 animals also had mammary gland fibroadenomas. Administration of coumarin (0.06-0.07 mM/kg/d) via drinking water on days 44 to 61 (before, during, and after DMBA administration; group 2) resulted in slight decreases in mammary gland adenocarcinoma incidence and multiplicity, but no difference in size or growth rate of tumors compared to rats treated with DMBA only (group 1). A higher dose of coumarin (group 3, 1 mM/kg/d) administered during the same time period via gavage resulted in a statistically significant reduction in the incidence of mammary gland adenocarcinomas compared to group 1, as well as a reduction in tumor size and multiplicity. Administration of coumarin on days 56 to 73 (coadministered and after DMBA administration; group 4, 1 mM/kg/d) did not affect mammary tumor incidence, size, or multiplicity. Results from this 28-week study suggest that coadministration of coumarin with DMBA may reduce the carcinogenic effect of DMBA. The authors suggest that these results are consistent with the possibility that there may be competition between the metabolism of coumarin and DMBA and that a reduction in the bioactivation of DMBA to the active carcinogenic species might have resulted in a decrease in tumor incidence.
Mammary Tumor Incidencea in Female Wistar Rats Administered DMBA (All Groups) and Coumarin. 23
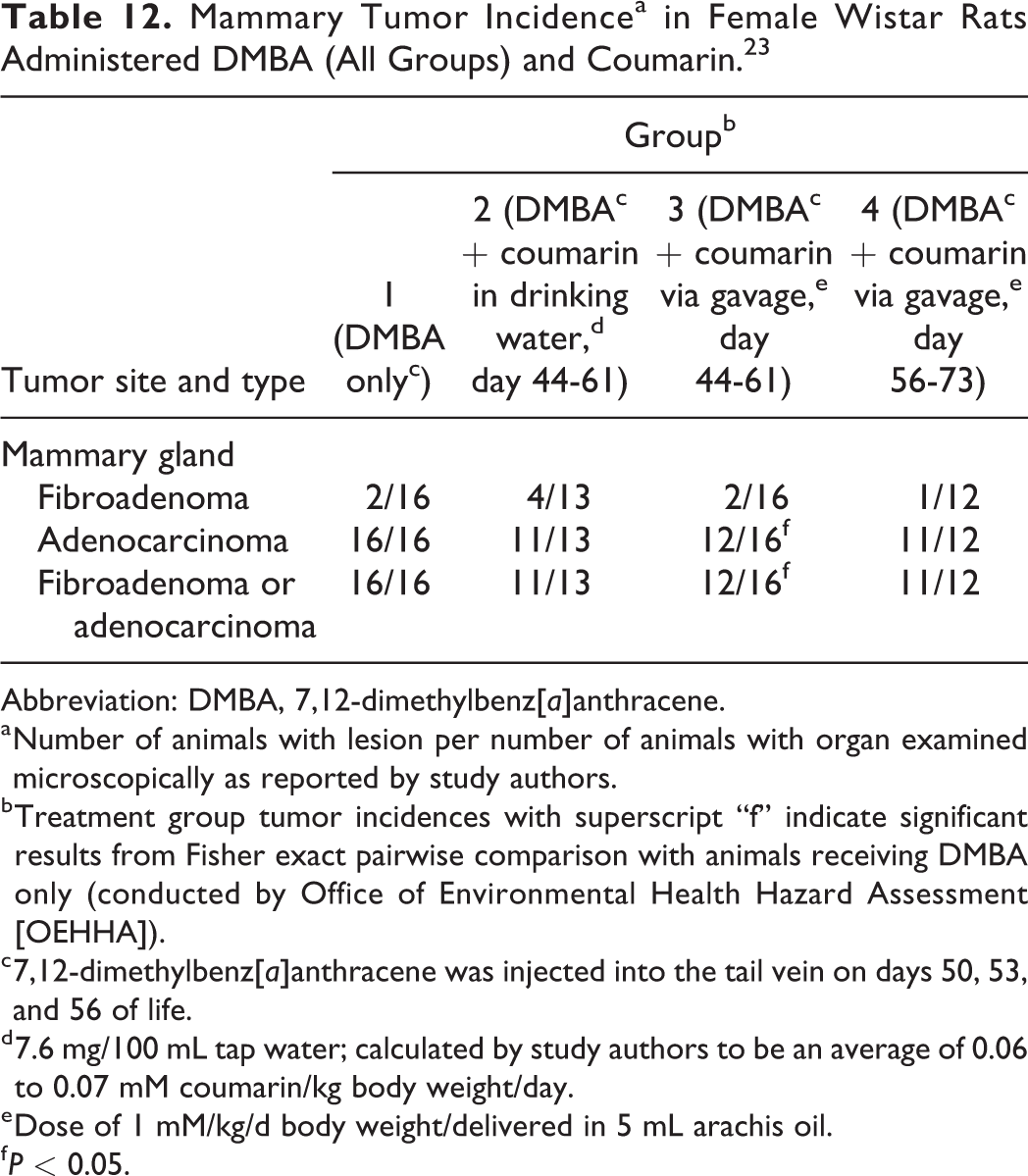
Abbreviation: DMBA, 7,12-dimethylbenz[a]anthracene.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by study authors.
b Treatment group tumor incidences with superscript “f” indicate significant results from Fisher exact pairwise comparison with animals receiving DMBA only (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c 7,12-dimethylbenz[a]anthracene was injected into the tail vein on days 50, 53, and 56 of life.
d 7.6 mg/100 mL tap water; calculated by study authors to be an average of 0.06 to 0.07 mM coumarin/kg body weight/day.
e Dose of 1 mM/kg/d body weight/delivered in 5 mL arachis oil.
f P < 0.05.
Co-carcinogenicity study in female S-D rats 24
This study investigated the effects of coumarin on mammary gland carcinogenesis induced by DMBA in female S-D rats. Two groups of 6-week old female S-D rats (15/group) were administered coumarin in the diet (low and high dose groups) for 8 days; a third group received feed without coumarin. On the seventh day, all rats were given one dose of DMBA by gavage. All rats were necropsied at 23 weeks of age. There were no significant differences in the mean body weight gains between groups.
Table 13 shows the mammary tumor incidences (tumor type not specified) observed in the study. Rats that received coumarin in feed for 7 days prior to administration of DMBA had statistically significantly fewer mammary tumors than rats administered only DMBA.
Mammary Tumor Incidencea in Female S-D Rats Administered DMBA (All Groups) and Coumarin. 24

Abbreviation: DMBA, 7,12-dimethylbenz[a]anthracene.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by study authors.
b Treatment group tumor incidences with superscript “f” indicate significant results from Fisher exact pairwise comparison with animals receiving DMBA only (group 1) (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c Average daily dose estimated to be 248 mg/kg/d, calculated by OEHHA.
d 12 mg DMBA in 1 mL olive oil by gavage.
e Average daily dose estimated to be 497 mg/kg/d, calculated by OEHHA.
f P < 0.001.
Co-carcinogenicity study in female ICR/Ha mice 24
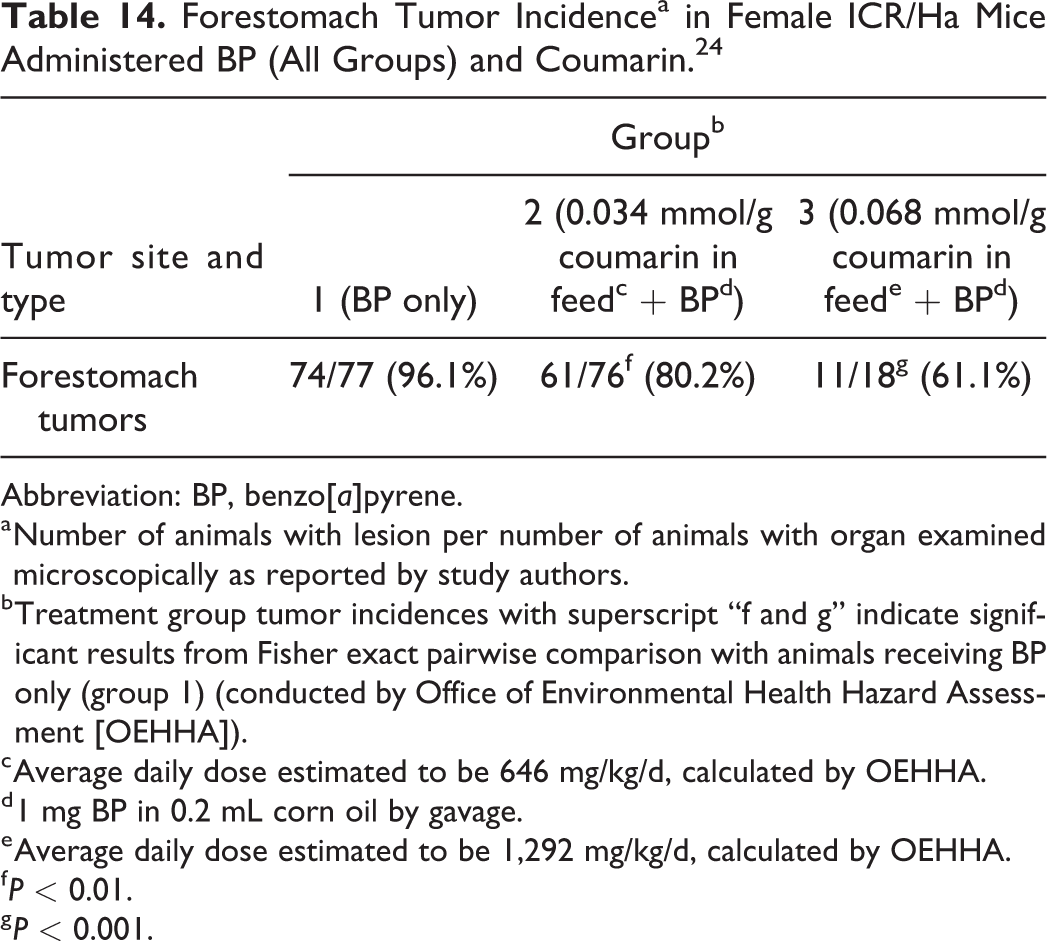
This study investigated the effects of coumarin on forestomach tumor formation induced by benzo(a)pyrene (BP) in female ICR/Ha mice. Nine-week-old female ICR/Ha mice (groups of 18, 76, or 77) were administered coumarin in the diet. Control mice received feed without coumarin. On the eighth day, all mice were given BP by gavage. Mice received a total of 8 doses of BP (2 times per week for 4 weeks). Mice administered coumarin in the diet were switched to control diets 3 days after the last BP dose. All mice were necropsied at 30 weeks of age.
Table 14 shows the forestomach tumor incidences (tumor type not specified) observed in the study. Mice that received coumarin in the feed had statistically significantly fewer forestomach tumors than mice administered only BP. Coumarin is metabolized by CYP2A5, 46 which is involved in bioactivation of BP. 47 Thus, it is possible that there may be competition between the metabolism of coumarin and BP that results in reduced bioactivation of coumarin to the active carcinogenic species.
Forestomach Tumor Incidencea in Female ICR/Ha Mice Administered BP (All Groups) and Coumarin. 24
Abbreviation: BP, benzo[a]pyrene.
a Number of animals with lesion per number of animals with organ examined microscopically as reported by study authors.
b Treatment group tumor incidences with superscript “f and g” indicate significant results from Fisher exact pairwise comparison with animals receiving BP only (group 1) (conducted by Office of Environmental Health Hazard Assessment [OEHHA]).
c Average daily dose estimated to be 646 mg/kg/d, calculated by OEHHA.
d 1 mg BP in 0.2 mL corn oil by gavage.
e Average daily dose estimated to be 1,292 mg/kg/d, calculated by OEHHA.
f P < 0.01.
g P < 0.001.
Co-carcinogenicity study in male Syrian golden hamsters 25
This study investigated the effect of coumarin on buccal pouch carcinogenesis induced by DMBA in male hamsters. Four groups (10/group) of male hamsters received treatment for 14 weeks. Group I animals served as controls and were painted with liquid paraffin 3 times a week for 14 weeks on their left buccal pouches. Groups II and III were painted with 0.5% DMBA in liquid paraffin 3 times a week for 14 weeks on their left buccal pouches. Group II received no other treatment. Group III received oral administration of coumarin at a dose of 100 mg/kg body weight/day, starting 1 week before exposure to DMBA and continuing on days alternate to DMBA painting, until study termination at week 16. Group IV received oral administration of coumarin (100 mg/kg body weight/day) alone throughout the experimental period.
7,12-Dimethylbenz[a]anthracene induced epithelial tumors in the buccal mucosa in 100% of the animals in group II (DMBA only). No buccal mucosal tumors were observed in group I (control), group III (DMBA plus coumarin), or group IV (coumarin). These results indicate that coadministration of coumarin with DMBA reduces the carcinogenic effect of DMBA and are consistent with the possibility that there may be competition between the metabolism of coumarin and DMBA that results in reduced bioactivation of DMBA to the active carcinogenic species.
Summary of animal carcinogenicity study findings
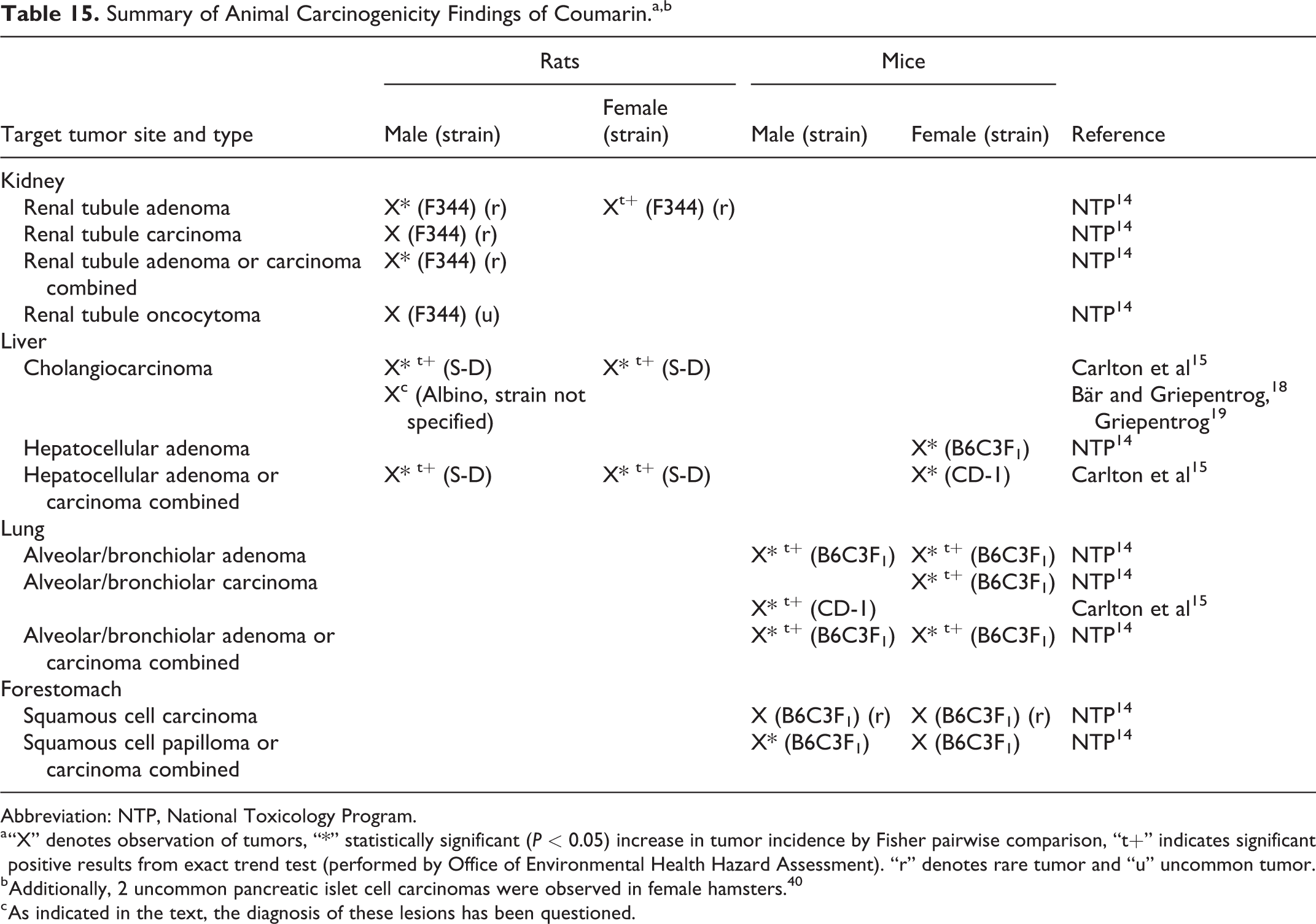
No tumors were reported in limited studies of coumarin carcinogenicity conducted in rats 16,17 and baboons. 22 A number of tumor findings reported in other animal carcinogenicity studies of coumarin are summarized in Table 15. Tumor findings in these studies that were judged likely to provide evidence of treatment-related effects, either because the tumors observed were rare or uncommon or because the incidence was increased with treatment at the P < 0.05 level of statistical significance in tests for trend or pairwise comparisons with controls (or both) are discussed below, organized by tumor type.
Summary of Animal Carcinogenicity Findings of Coumarin.a,b
Abbreviation: NTP, National Toxicology Program.
a “X” denotes observation of tumors, “*” statistically significant (P < 0.05) increase in tumor incidence by Fisher pairwise comparison, “t+” indicates significant positive results from exact trend test (performed by Office of Environmental Health Hazard Assessment). “r” denotes rare tumor and “u” uncommon tumor.
b Additionally, 2 uncommon pancreatic islet cell carcinomas were observed in female hamsters. 40
c As indicated in the text, the diagnosis of these lesions has been questioned.
Tumor site concordance is not expected across species, strains, or sex, 48 and only 52% of rat carcinogens are positive in the same site in mice, and 31% of test substances are gender-specific in rats. 49,50 Additionally, differences in study design and conduct, including dosing regimen, laboratory conditions, animal diet, and other factors, such as animal colony origin, can also result in differences in tumor findings across studies. However, multiple observations of tumor induction at a particular site (and of a particular cell type) across studies generally provide increased evidence of carcinogenicity.
Hepatocellular tumors were observed in both sexes of S-D rats and in 2 strains of female mice. More specifically, increases in hepatocellular adenoma or carcinoma combined were observed in male and female S-D rats and female CD-1 mice, and increases in hepatocellular adenoma were observed in female B6C3F1 mice. Increases in alveolar/bronchiolar tumors were observed in 2 strains of male mice (B6C3F1: adenoma; CD-1: carcinoma) and 1 strain of female mice (B6C3F1: adenoma, carcinoma). Increases in liver cholangiocarcinoma were observed in both sexes of S-D rats, and observations of cholangiocarcinoma were also reported in another study in male albino rats of an unspecified strain, 18,19 although the findings in this latter study have been questioned. Increases in rare renal tubule adenomas were observed in both sexes of F344 rats, and additionally increases in rare renal tubule adenoma and carcinoma combined and observations of uncommon renal tubule oncocytomas occurred in males. Observations of rare forestomach squamous cell carcinoma and of forestomach squamous cell papilloma or carcinoma combined occurred in both sexes of B6C3F1 mice, with the increase in papilloma or carcinoma combined reaching statistical significance in males.
Findings in the co-carcinogenicity studies include a decrease in the number of mammary tumors induced by DMBA in female Wistar rats 23 and S-D rats, 24 a decrease in forestomach tumors induced by BP in female ICR/Ha mice, 24 and an absence of buccal mucosal tumors induced by DMBA in male Syrian golden hamsters 25 when animals also received coumarin.
Other Relevant Data
Pharmacokinetics and metabolism
This section summarizes the absorption, distribution, metabolism, and excretion of coumarin in humans and animals. It first discusses key findings from studies in humans, followed by studies in animals, and then discusses genetic polymorphisms of CYP2A6, a key enzyme involved in human coumarin metabolism. Additional details on CYP2A6 polymorphisms are presented in Appendix A.
Studies in humans
The pharmacokinetics and metabolism of coumarin have been studied in humans in vivo and in vitro. Many of these studies have been reviewed previously. 4,5,51,52 Briefly, absorption studies have been conducted in vivo by the oral and dermal routes and in vitro with a human skin absorption model. 53 -58 Distribution studies were conducted in human volunteers by the IV and oral routes. 40,53,59 Studies of coumarin metabolism were conducted by oral, dermal, and IV routes in vivo and were tested in human liver microsomal samples, human liver slice cultures, and recombinant human cytochrome P-450s (CYPs) in vitro. 53,57,60 -73 Excretion studies were conducted in human volunteers by the IV, oral, and dermal routes. 3,40,57,61,72 -77
Coumarin is quickly absorbed by the oral and dermal routes. Following oral administration, coumarin is rapidly and completely absorbed. 3,78 In a dermal application study, 60% of the coumarin dose was absorbed within 6 hours. 57 Similarly, in vitro studies with human skin found that 66% of the applied dose was absorbed after 72 hours. 56
Coumarin and its metabolites are distributed throughout the body in humans. 40,59,79,80 Coumarin is rapidly and extensively metabolized, and only a small amount of the parent compound (about 3% of the administered dose) is detected in the blood following oral administration. 78 The half-life of coumarin in the blood is similar following administration via either the IV or oral routes, ranging from 1 to 1.5 hours. 40,53 The plasma half-life following dermal exposure is 1.7 hours. 57
A number of coumarin metabolites have been identified in humans either in vivo from urine or blood samples or in vitro with human liver microsomes, liver slices, or recombinant cytochromes (Table 16). Metabolites identified in humans in vivo are 7-hydroxycoumarin (7-HC), its conjugated glucuronides or sulfates, 3-hydroxycoumarin (3-HC) and o-hydroxyphenylacetic acid (o-HPAA). 53,57,60,72,73 Additional metabolites that have been identified in vitro include 4-, 5-, 6-, and 8-HC, 6,7-dihydroxycoumarin (6,7-DiHC), o-coumaric acid (o-CA), o-hydroxyphenylpropionic acid (o-HPPA), o-hydroxyphenylethanol (o-HPE), and coumarin 3,4-epoxide glutathione (GSH) conjugate (CE-SG). 62,64 -69,71,81 Other metabolic products of coumarin have been detected in humans, but their structures have not been identified. 57,62,63
Summary of Coumarin Metabolites Detected in Humans In Vivo and In Vitro.a
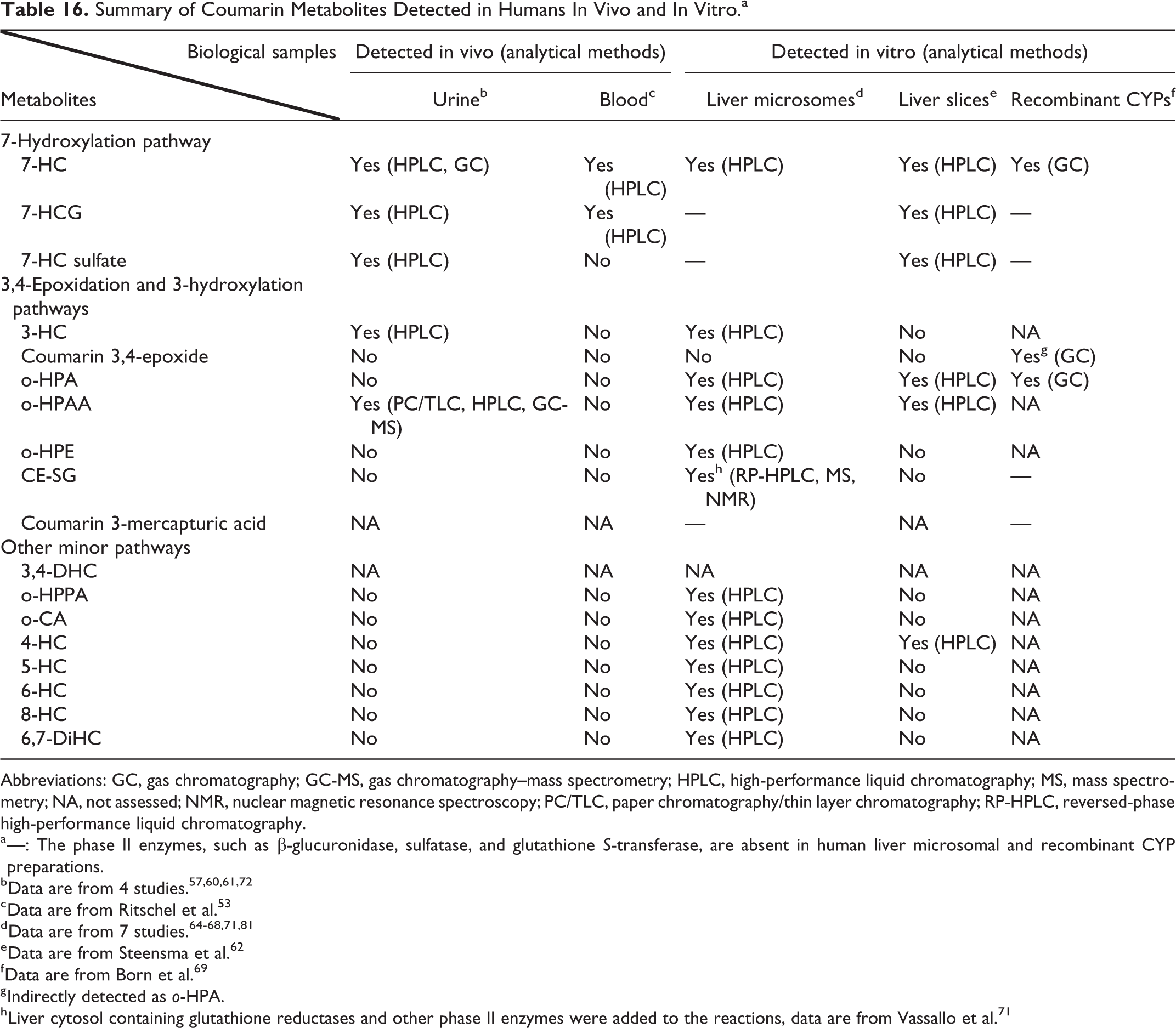
Abbreviations: GC, gas chromatography; GC-MS, gas chromatography–mass spectrometry; HPLC, high-performance liquid chromatography; MS, mass spectrometry; NA, not assessed; NMR, nuclear magnetic resonance spectroscopy; PC/TLC, paper chromatography/thin layer chromatography; RP-HPLC, reversed-phase high-performance liquid chromatography.
a —: The phase II enzymes, such as β-glucuronidase, sulfatase, and glutathione S-transferase, are absent in human liver microsomal and recombinant CYP preparations.
c Data are from Ritschel et al. 53
e Data are from Steensma et al. 62
f Data are from Born et al. 69
g Indirectly detected as o-HPA.
h Liver cytosol containing glutathione reductases and other phase II enzymes were added to the reactions, data are from Vassallo et al. 71
As shown in Figure 2, coumarin is metabolized in humans via several different pathways. The primary pathways of coumarin metabolism are the 7-hydroxylation pathway and the 3,4-epoxidation pathway, although coumarin can be hydroxylated at other possible positions (ie, carbons 3, 4, 5, 6, and 8) and the opening of the lactone ring can yield various products (Figure 2). Multiple CYP enzymes can catalyze the 7-hydroxylation reaction to form 7-HC, including CYP2A6, CYP2A13, and CYP2B6, with CYP2A6 being the most active. 82 CYP2A6, also known as coumarin 7-hydroxylase, is a polymorphic enzyme. Multiple CYP enzymes can catalyze the 3,4-epoxidation reaction, including CYP1A1, CYP1A2, CYP2B6, CYP2A13, CYP3A4, and CYP2E1, with CYP1A1, CYP1A2, and CYP2E1 thought to be the most active. 46,60,66,69,83
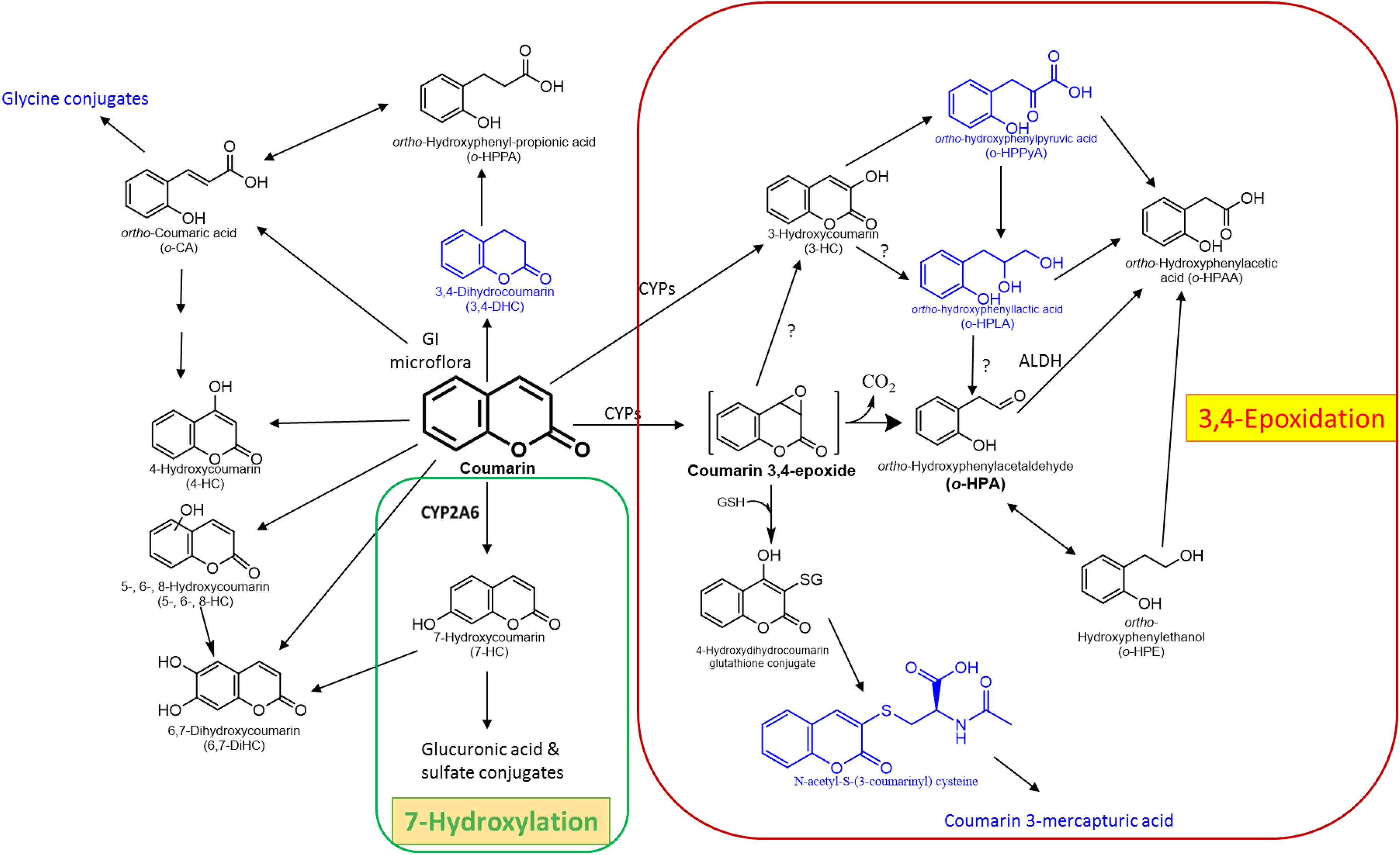
Proposed coumarin metabolism in humans and animals. Human and animal metabolism of coumarin is qualitatively similar and occurs through a number of enzymatic pathways. Metabolites observed in animals that have not yet been looked for in humans are shown in blue. The 2 predominant metabolic pathways are 7-hydroxylation (shown in the green box) and 3,4-epoxidation (shown in the red box). CYP2A6, the key enzyme involved in coumarin 7-hydroxylation, is highlighted in green.
Coumarin 3,4-epoxide (CE) is an unstable, highly reactive electrophile and can react with GSH to form a GSH conjugate (CE-SG) or degrade spontaneously to form the reactive aldehyde ortho-hydroxyphenylacetaldehyde (o-HPA) via opening of the lactone ring and cleavage of CO2. 70 Ortho-hydroxyphenylacetaldehyde can be oxidized to o-HPAA by aldehyde dehydrogenases 5,71 or reduced to o-HPE. Coumarin 3,4-epoxide has a short half-life (4 seconds) and is generally measured indirectly as either o-HPA or o-HPAA. 69,70 Both CE and o-HPA are toxic electrophilic metabolites of coumarin that covalently bind to cellular macromolecules and are associated with hepatotoxicity. 51,65,71,81,84,85
It is unclear if 3-HC can be oxidized to o-HPA. Studies by Kaighen and Williams 86 provide indirect support for this pathway with their detection of urinary o-HPAA in rabbits and rats administered 3-HC. o-Hydroxyphenylacetic acid is formed from o-HPA; however, it is also formed from o-hydroxyphenylpyruvic acid (o-HPPyA) and o-hydroxyphenyllactic acid (o-HPLA). On the other hand, studies by Born et al 68,84 and Normal et al 87 suggest that 3-HC is not converted to o-HPA.
The 7-hydroxylation pathway is often but not always the predominant pathway of coumarin metabolism in humans. As discussed in greater detail in the section below on CYP2A6 genetic polymorphisms, the relative importance of the 7-hydroxylation pathway versus the 3,4-epoxidation pathway in the metabolism of coumarin is determined primarily by an individual’s CYP2A6 phenotype. The shift in the quantities of specific coumarin metabolites formed, either by decrease-of-function or loss-of-function CYP2A6 genetic polymorphisms, or nongenetic factors, can be significant. For example, 7-HC can constitute up to 92% of coumarin metabolites in some humans. 61 However, in an individual homozygous for a loss-of-function CYP2A6 variant allele, the amount of 7-HC measured in the urine represented less than 0.02% of the applied dose, while o-HPAA (a product of the 3,4-epoxidation pathway) accounted for up to 54.6% of the total urinary metabolites. 60
In humans, excretion of coumarin and its metabolites is rapid and proceeds primarily via urine. After oral administration, 95% was excreted within 4 hours in one study. 75 Dermal application results in slower excretion, with 26% of the applied coumarin dose excreted in 2 hours and 59% excreted in 120 hours. 57 The primary urinary metabolites were conjugates of 7-HC and small amounts of unconjugated 7-HC and o-HPAA. Fecal excretion has been measured only following dermal exposure and amounted to 1% of the applied dose in 120 hours. 57 This finding suggests that very little biliary excretion of coumarin metabolites occurs in humans.
Studies in animals
The pharmacokinetics and metabolism of coumarin have been studied in animals in vivo and in vitro, and many of these studies have been reviewed previously. 4,5,51,52 Briefly, in vivo studies have been conducted in several animal species, including rats (intraperitoneal [IP], IV, oral, dermal), mice (oral), rabbits (oral), dogs (IV, oral), gerbils (IP), rhesus monkeys (IV, oral), baboons (oral), squirrel monkeys (oral), ferrets (oral), guinea pigs (oral), pigs (oral), hamsters (oral), dogs (oral), and cats (oral). 4,5,57,88 -92 In vitro studies were conducted with rat and mouse skin 56,58 ; liver slices from rats, cynomolgus monkeys, mice, Syrian hamsters, rabbits, and baboons 62 ; lung cytosolic fractions from rats and mice 71 ; recombinant CYP enzymes 69 ; and microsomal fractions from rats, mice, and other species. 46,65,66,68 -70,81,85,93 -99
Coumarin is rapidly absorbed in rats and mice following oral or dermal administration and is distributed throughout the body and extensively metabolized, with little excretion of the parent compound. 57,89,91,100,101 In mice, peak plasma concentrations can be reached within 10 minutes following oral gavage and within 9 hours when coumarin is added to the diet. 100 In baboons, 90% of the dose was absorbed within 45 minutes after oral administration. 89 When applied dermally to rats, absorption was considered complete within 6 hours. 91
Following dermal or oral absorption, coumarin is rapidly distributed throughout the body and metabolized, as shown in studies conducted in baboons, rats, and gerbils. 57,89,91,102,103 Following dermal application of radiolabeled coumarin to rats, the highest tissue concentrations of radioactivity were found in the small intestine, large intestine, and stomach, with lower concentrations observed in the kidney and liver. 57 These results are indicative of significant biliary excretion of coumarin in the rat 57 and are supported by IP injection studies in rats. 102 The plasma half-life of coumarin varies greatly by species, with values of 4 hours reported for mice and 5 to 20 hours for rats. 57,100 By comparison, the plasma half-life in humans ranged from 1 to 1.7 hours, depending on the route of exposure. 40,53,57
Coumarin metabolism in animals is qualitatively similar to that in humans, although there are quantitative differences between humans and some species (eg, rats), as well as quantitative differences among some nonhuman species. For example, in baboons, 7-hydroxylation is the main pathway by which coumarin is metabolized, leading to 7-HC as the primary initial metabolite. In contrast, rats, hamsters, guinea pigs, and some strains of mice form little or no 7-HC and instead form CE and 3-HC. 4,40,86 Animal studies have identified some additional coumarin metabolites that have not been reported in human studies, including o-HPLA, o-hydroxyphenylpyruvic acid, and N acetyl-S-(3-coumarinyl) cysteine. 86,87,101,104 Other metabolic products of coumarin have been detected in animals, but the structures of these products have not yet been identified. 86,101
Briefly, metabolism of coumarin in animals occurs primarily via either epoxidation or hydroxylation. These specific reactions are catalyzed by species- and tissue-specific CYP enzymes and result in the formation of either the reactive intermediate CE or various hydroxylated coumarins, with 7- and 3-HC being the most frequently formed hydroxylated metabolites. 4,40,64,88 As discussed previously, CE is reactive and unstable and can spontaneously rearrange to form o-HPA, followed by further oxidation to o-HPAA or reduction to o-HPE. 5,68,87 Coumarin 3,4-epoxide may also form a GSH conjugate and be excreted. 4,105 Other metabolites resulting from ring-opening reactions include o-HPLA, o-HPPyA, and o-HPPA. 4,85,100,103 3,4-Dihydrocoumarin (3,4-DHC) is another metabolite of coumarin which is formed in the intestine by microflora. 14,106 Scheline 107 incubated coumarin with microflora from the caeca of rats and rabbits under anaerobic conditions and demonstrated the formation of 3,4-DHC and o-HPPA. Hydroxycoumarins and other coumarin metabolites can be conjugated with glucuronic acid or sulfate and excreted, and o-CA can be conjugated with glycine and excreted. 108,109
As discussed in the section on human metabolism, it is unclear if 3-HC can be metabolized to o-HPA or if this reactive aldehyde (ie, o-HPA) is produced from coumarin solely through the 3,4-epoxidation pathway. The urinary excretion studies of Kaighen and Williams 86 in rats and rabbits administered radiolabeled 3-HC provide some indirect support for the formation of o-HPA from 3-HC. These investigators looked for several possible coumarin metabolites, based on scientific understanding and analytical methods available at that time. Ortho-hydroxyphenylacetaldehyde was not one of the analytes assessed; however, o-HPAA was assessed and it was detected in the urine of rats and rabbits dosed with 3-HC. o-Hydroxyphenylacetic acid is formed from o-HPA; however, it is also formed from o-HPPyA and o-HPLA, both of which were also detected in rats and rabbits administered 3-HC. 86 Other studies conducted using various in vitro model systems suggest that 3-HC is not converted to o-HPA. These studies include those of Norman and Wood, 87 where the production of o-HPE, which is formed from o-HPA, was assessed in rat liver 10,000g supernatants incubated with either coumarin or 3-HC. While the production of o-HPE was observed in incubations with coumarin, no production of o-HPE was observed in incubations with 3-HC. 87 Born et al 68 investigated the formation of o-HPA from either CE or 3-HC in an aqueous cell- and enzyme-free incubation system and found that o-HPA was formed from CE, but not from 3-HC. These authors also studied o-HPA formation from CE and 3-HC in the presence of mouse liver microsomes and found that o-HPA was formed in microsomes incubated with CE, but not with 3-HC. 68 Comparative toxicity studies in which coumarin and o-HPA were found to be cytotoxic to rat hepatocytes, but 3-HC was not, provide additional, albeit indirect evidence that 3-HC is not converted to o-HPA. 84,110
Coumarin toxicity is associated with the formation of toxic and electrophilic metabolites that can bind covalently to cellular macromolecules, including CE and o-HPA. 51,65,71,84,85 While CE and o-HPA are formed in all animal species studied, differences among species in the toxicity of coumarin may be explained not only by differences in metabolic activation but also by differences in detoxification reactions. Mice appear to catalyze the oxidation of o-HPA to o-HPAA more efficiently than rats, as o-HPAA may account for up to 41% of the administered dose in mice and only 12% in rats. 100 A faster clearance rate for the oxidation of o-HPA to o-HPAA in mice as compared to rats is supported by findings from studies with liver microsomal and cytosolic fractions. The total clearance of coumarin in microsomal incubations was 4-fold greater in mice than in rats 85 ; similarly, the total clearance of coumarin in cytosolic incubations was 20 times higher in mice compared to rats. 71 Both mice and rats reduced o-HPA to o-HPE; however, this is only a major reaction in rats. Vasallo et al 71 suggest that a cycle of oxidation and reduction from o-HPA to o-HPE and back may contribute to slower hepatic clearance of the toxic aldehyde in the rat. The extent and kinetics of additional detoxification reactions such as conjugation with GSH may also determine the extent to which electrophilic metabolites bind covalently with cellular macromolecules in a given tissue.
Excretion of coumarin and its metabolites occurs via expired air, urine, and feces. About 30% of the radioactivity associated with a dose of [2-14C] coumarin administered to rats was recovered in expired air as CO2. 92 Little or no radioactivity was found in expired air of rats (and other species) when [3-14C or 4-14C] coumarin was administered. 4,86 These findings are consistent with loss of carbon number 2 on the lactone ring during coumarin metabolism to form o-HPA. Fecal excretion is attributed to biliary excretion of metabolites, including unidentified ring opened compounds, and is greater in many of the animal species studied than in humans. 57,86,92,101,102 The highest fecal excretion has been observed in rats (38%), followed by hamsters (12%), baboons (3.4%), and rabbits (1%). 86,89,101 Excretion of coumarin and its metabolites via urine and feces is rapid, with the majority occurring within 24 hours, and excretion being essentially complete within 96 hours.
Genetic polymorphisms in CYP2A6, a key enzyme involved in human coumarin metabolism
As discussed above, CYP2A6 is one of the key enzymes involved in the metabolism of coumarin in humans. CYP2A6 catalyzes the 7-hydroxylation of coumarin to form 7-HC. CYP2A6-mediated 7-hydroxylation is considered to be the major metabolic detoxification pathway for coumarin in humans, as 7-HC and its glucuronide and sulfate conjugates are rapidly excreted, primarily in the urine. CYP2A6 is a highly polymorphic gene. Some polymorphisms affect CYP2A6 enzyme activity, with some resulting in increased activity, others resulting in reduced activity, and still others resulting in complete loss of enzyme activity (loss of function). Individuals with CYP2A6 polymorphisms that confer reduced enzyme function or loss of function may have increased susceptibility to coumarin toxicity.
Clinical trials with coumarin have shown that certain individuals are more susceptible to coumarin hepatotoxicity. 111 -114 Studies in humans or human tissues have also demonstrated interindividual variability in coumarin metabolism to 7-HC. CYP2A6 polymorphisms are thought to be largely responsible for this variability. It has been hypothesized that reduced or loss-of-function CYP2A6 polymorphisms make individuals more susceptible to coumarin toxicity, including carcinogenicity, by increasing the metabolism of coumarin through the 3,4-epoxidation pathway and increasing the formation of the reactive metabolites CE and o-HPA.
This shift in metabolism from 7-hydroxylation to the 3,4-epoxidation pathway and other pathways (ie, 3-hydroxylation) has been observed in microsomes prepared from some human liver samples, and in vivo, in an individual that lacked functional CYP2A6. In one study of coumarin metabolism using human liver microsomes prepared from 12 individuals, van Iersel et al 63 demonstrated the presence of the following metabolites: 3-HC, 4-HC, 7-HC, 6,7-DiHC, o-CA, o-HPPA, o-HPA, o-HPE, and o-HPAA. For 11 of the 12 individuals, 7-HC was the major liver microsomal metabolite, accounting for 76% to 92% of the total polar products (ie, all metabolites except the ones covalently bound to the microsomal proteins). However, in microsomes prepared from the 12th subject, 7-HC accounted for only 1.2% of the total polar products, while products of the 3,4-epoxidation and 3-hydroxylation pathways (o-HPA, o-HPE, o-HPAA, and 3-HC) accounted for 69% of the total polar products. In another study of coumarin metabolism using human liver microsomes prepared from 4 individuals from the United Kingdom, o-HPA was identified as the most abundant metabolite of coumarin (about 6 times that of 7-HC) in each of the 4 microsomal preparations. 64 This shift in coumarin metabolism to the 3,4-epoxidation and 3-hydroxylation pathways was also evident in vivo in an individual with the CYP2A6*2/*2 genotype (ie, no CYP2A6 coumarin 7-hydroxylase activity), in which 45.9% to 54.6% of an administered 2 mg dose of coumarin was excreted as o-HPAA, with less than 0.02% excreted as 7-HC. 60
Consequently, it is of critical importance to identify the subgroups of the human population where this shift occurs, as these subgroups are likely to be more susceptible to coumarin toxicity. Besides genetic polymorphisms, the modulation of CYP2A6 activity by age, gender, and lifestyle factors (such as drugs and dietary factors) could also lead to a shift of coumarin metabolism toward increased production of the reactive metabolites CE and o-HPA.
This section summarizes the available information on the coumarin 7-hydroxylase activity of CYP2A6 variants, the distribution of certain loss of function and decrease of function CYP2A6 variants in different ethnic populations, CYP2A6 genotype–phenotype correlation studies, and other factors that affect the CYP2A6 enzyme’s ability to metabolize coumarin.
CYP2A6 variants and associated coumarin 7-hydroxylase activities
CYP2A6 is a highly polymorphic gene. These genetic polymorphisms can affect the coumarin 7-hydroxylase activity of CYP2A6. To date, there are at least 45 identified allelic variants of CYP2A6, with many subgroups within some of the variants. 115 The complete list of nucleotide changes for each CYP2A6 allele is available at https://www.pharmvar.org/gene/CYP2A6.
Coumarin is one of the most well-studied substrates of CYP2A6, and measurement of 7-HC is commonly used to characterize CYP2A6-mediated 7-hydroxylase activity. 46,116 -118 While coumarin can also undergo 7-hydroxylation via CYP2A13, 7-HC formation is thought to occur primarily via CYP2A6 due to coumarin’s high affinity as a substrate for CYP2A6. 7-hydroxycoumarin is the primary metabolite of coumarin in most individuals. For example, in a small study of 8 healthy volunteers, about 79% of the total coumarin was excreted as 7-HC (the analytical method used did not distinguish between conjugated and unconjugated 7-HC) and about 4% was excreted as o-HPAA. 61 Additional studies with human volunteers have demonstrated that considerable interindividual variability in CYP2A6 coumarin 7-hydroxylase activity exists within the human population, and several studies have associated this variability with specific CYP2A6 genetic polymorphisms. 60,119 -125
Three model systems for measuring 7-hydroxylase activity of CYP2A6 variants are commonly used in research: the heterologous expression of recombinant protein in bacteria or cultured cell lines, the use of human liver microsomes, and the measurement of 7-HC in human subjects. Additional discussion of these methods is presented in Supplementary Material A.
The genetic variations of CYP2A6, caused by either single-nucleotide polymorphisms in coding or noncoding regions or gene conversions, duplications, or deletions, can result in an increase, decrease, or lack of coumarin 7-hydroxylase activity of the enzyme (Table 17).
Human CYP2A6 Variants and Their Coumarin 7-Hydroxylase Activities.a
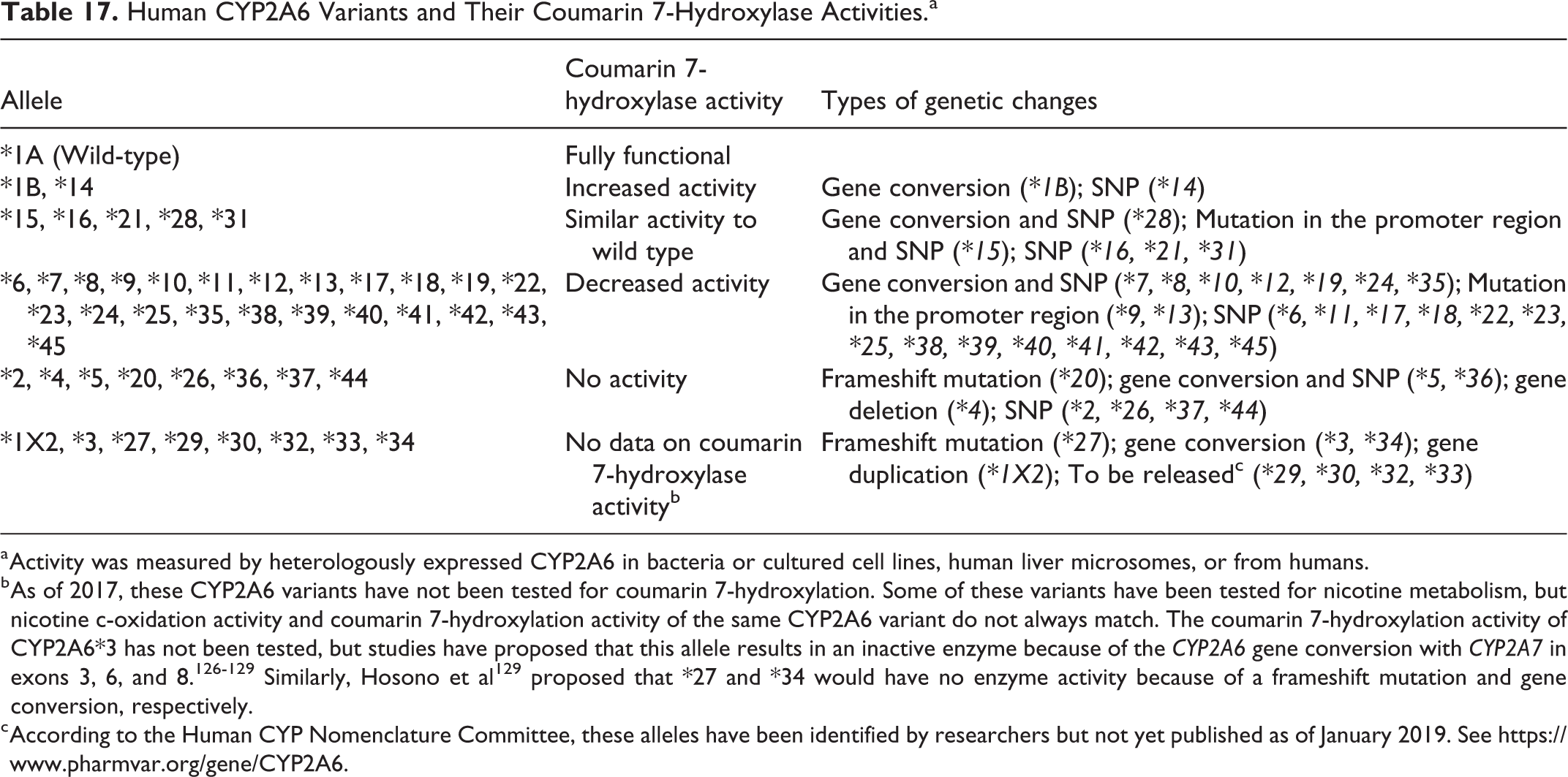
a Activity was measured by heterologously expressed CYP2A6 in bacteria or cultured cell lines, human liver microsomes, or from humans.
b As of 2017, these CYP2A6 variants have not been tested for coumarin 7-hydroxylation. Some of these variants have been tested for nicotine metabolism, but nicotine c-oxidation activity and coumarin 7-hydroxylation activity of the same CYP2A6 variant do not always match. The coumarin 7-hydroxylation activity of CYP2A6*3 has not been tested, but studies have proposed that this allele results in an inactive enzyme because of the CYP2A6 gene conversion with CYP2A7 in exons 3, 6, and 8. 126 -129 Similarly, Hosono et al 129 proposed that *27 and *34 would have no enzyme activity because of a frameshift mutation and gene conversion, respectively.
c According to the Human CYP Nomenclature Committee, these alleles have been identified by researchers but not yet published as of January 2019. See https://www.pharmvar.org/gene/CYP2A6.
A detailed summary of the ability of individual CYP2A6 allele variants to catalyze 7-hydroxylation of coumarin is provided in Supplementary Table A1. Overall, there is consistency among studies reporting enzyme activity for specific CYP2A6 variants.
Recently, Tanner et al 130 studied the correlation between several CYP2A6 allele variants (*2, *4, *7, *8, *9, *10, *12, *17, *20, *23, *25, *28, and *35) and enzyme activity using human liver tissues from 360 donors of various ethnicities. They found a strong correlation between genotypes with one or more variant alleles and decreased CYP2A6 protein expression as well as coumarin 7-hydroxylation activity. 130 This study also shows that, although the frequency of individual loss of function or decrease of function alleles can be low, the proportion of variant allele carriers that are slow coumarin metabolizers can be significant.
Tanner et al 130 also observed a wide range of CYP2A6 messenger RNA expression, protein expression, and enzyme activity levels within the wild-type (*1/*1) group. This variation in the coumarin 7-hydroxylation phenotype in CYP2A6 wild-type individuals is consistent with observations from other studies. For example, in a group of Thai individuals, there were 17 poor coumarin 7-hydroxylators among the 55 wild-type (CYP2A6*1A/*1A) individuals. 119 The variation of enzyme activity that was unaccounted for by the genotype was possibly due to unknown (or unassessed) genetic variations in this gene or upstream regulatory genes or nongenetic factors. Overall, the collection of evidence shows that individuals with loss of function or decrease of function CYP2A6 alleles are probably poor metabolizers for coumarin 7 hydroxylation, and in these individuals, the metabolism of coumarin could shift toward increased production of the reactive metabolites CE and o-HPA.
Distribution of CYP2A6 alleles in different ethnicities and populations
CYP2A6 genetic variation directly alters the enzymatic activity of the protein and is therefore important for evaluating individual susceptibility to toxicants that are CYP2A6 substrates, such as coumarin. CYP2A6 shows genotypic polymorphisms in populations across the world. Knowledge of the allele frequencies within different ethnic populations does not directly predict the genotype frequencies, but it does provide information on the potential for poor metabolizer genotypes (homozygotes with 2 inactive alleles or heterozygotes with 1 inactive allele and 1 intermediate allele) to exist in those populations. We have summarized the findings on CYP2A6 allele frequencies in different ethnicities and geographical areas (see Supplementary Table A2), focusing on variants with loss-of-function (CYP2A6*2, *4, *5, and *20) and the 3 most studied decrease-of-function variants (CYP2A6*7, *9, and *10).
A graphic depiction of population frequencies for alleles 4 and 9, 2 of the most studied alleles, is provided in Figure 3. Overall, there is a diverse distribution of these 2 alleles. The frequencies in African individuals and African North Americans are similar, as shown by the overlapping of the first 2 dark blue bars for allele 4 and the first 2 orange bars for allele 9. Between East or Southeast Asians and Asian North Americans, the frequencies for allele 4 also overlap and go up to over 22%. The rest of the populations shown in Figure 3 contain different levels of these 2 alleles. Defective CYP2A6 alleles are present in all of these populations tested, and the carriers of these alleles are the subpopulations that may lose part or all of their coumarin 7-hydroxylation activity.
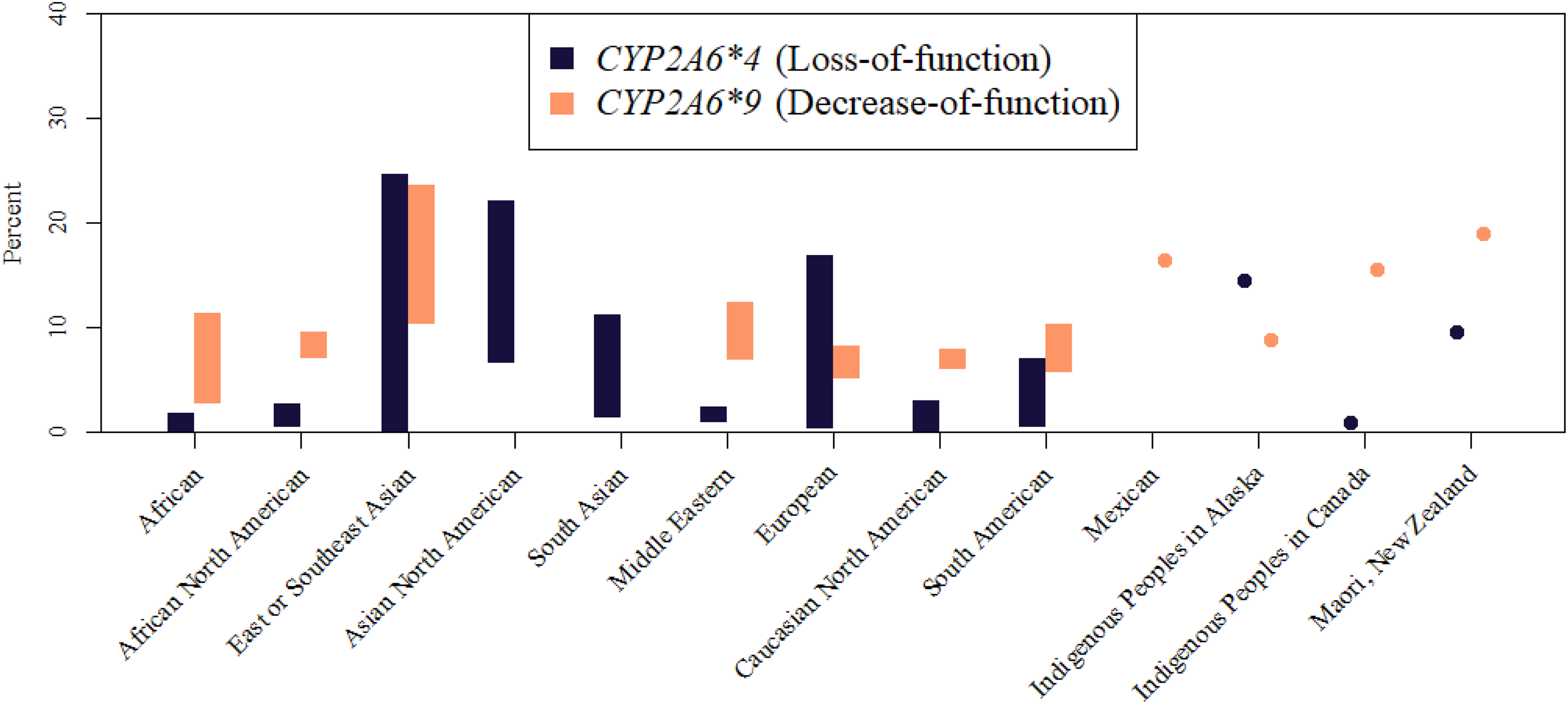
Distribution of 2 CYP2A6 alleles reported in different populations around the world. Allele 4 (dark blue), a loss-of-function allele; Allele 9 (orange), a decrease-of-function allele; x axis, populations that were genotyped; y axis, percentage found in each population in the genotyping studies. Each bar represents a range of frequencies found in a population, based on multiple studies, with the bottom of the bar starting at the minimum of the range and the top of the bar showing the maximum of the range. A dot means the frequency came from one study.
Table 18 summarizes the detailed findings from Supplementary Table A2 and presents them as ranges in each population/ethnicity. The results show that certain ethnic populations carry significant frequencies of some decrease of function or loss-of-function alleles.
Frequencies of Several Loss-of-Function or Decrease-of-Function CYP2A6 Alleles in Different Ethnicities and Populations.a
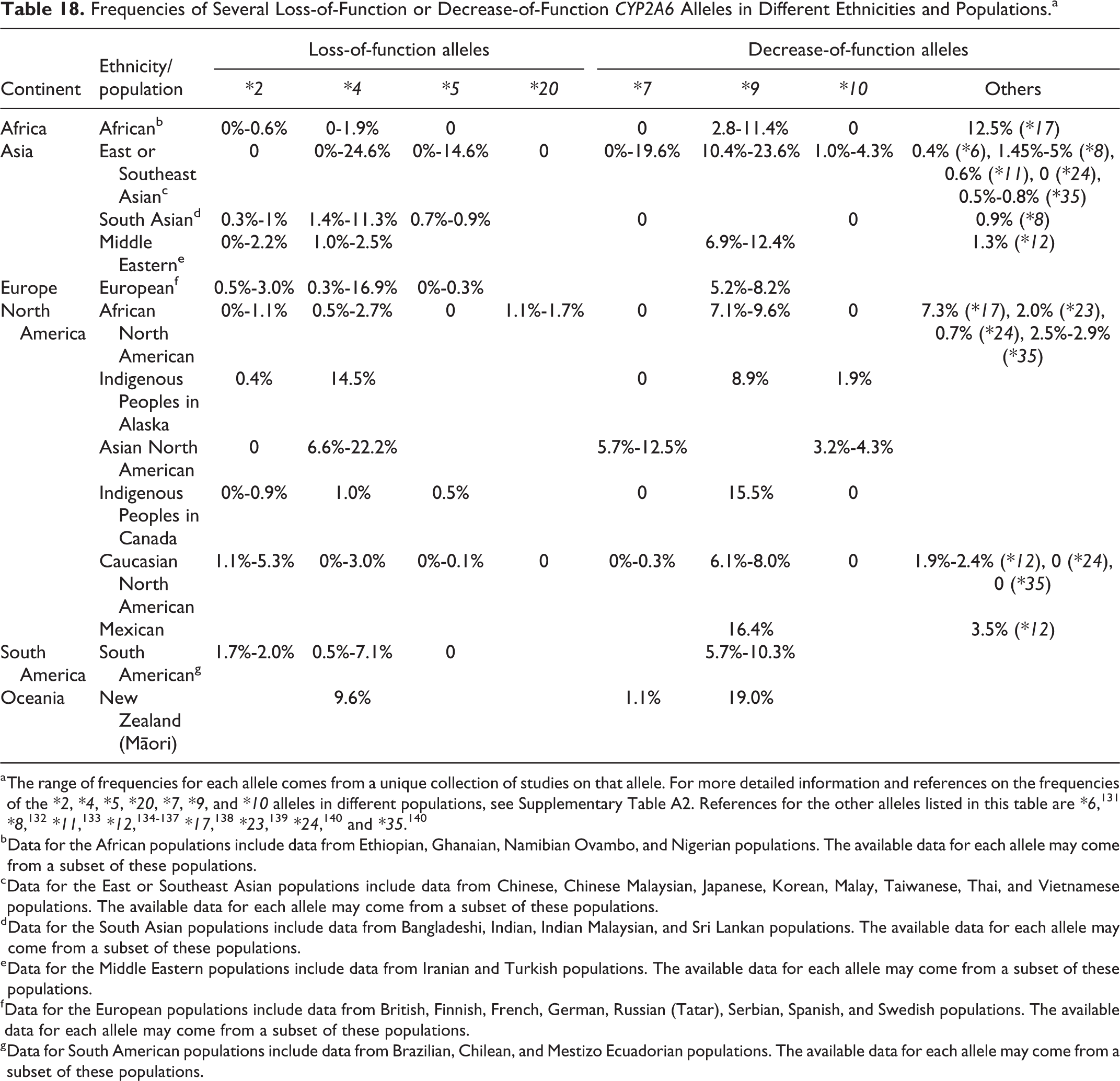
a The range of frequencies for each allele comes from a unique collection of studies on that allele. For more detailed information and references on the frequencies of the *2, *4, *5, *20, *7, *9, and *10 alleles in different populations, see Supplementary Table A2. References for the other alleles listed in this table are *6, 131 *8, 132 *11, 133 *12, 134 -137 *17, 138 *23, 139 *24, 140 and *35. 140
b Data for the African populations include data from Ethiopian, Ghanaian, Namibian Ovambo, and Nigerian populations. The available data for each allele may come from a subset of these populations.
c Data for the East or Southeast Asian populations include data from Chinese, Chinese Malaysian, Japanese, Korean, Malay, Taiwanese, Thai, and Vietnamese populations. The available data for each allele may come from a subset of these populations.
d Data for the South Asian populations include data from Bangladeshi, Indian, Indian Malaysian, and Sri Lankan populations. The available data for each allele may come from a subset of these populations.
e Data for the Middle Eastern populations include data from Iranian and Turkish populations. The available data for each allele may come from a subset of these populations.
f Data for the European populations include data from British, Finnish, French, German, Russian (Tatar), Serbian, Spanish, and Swedish populations. The available data for each allele may come from a subset of these populations.
g Data for South American populations include data from Brazilian, Chilean, and Mestizo Ecuadorian populations. The available data for each allele may come from a subset of these populations.
The frequency of CYP2A6*4, an allele that results in an absence of functional enzyme in homozygous individuals, is elevated in East and Southeast Asian populations. The highest frequency of CYP2A6*4 appears to be in the Japanese population (the frequency varies from 16% to 24.6%, based on 12 studies). The frequency of this allele is also high in Asian Americans (15.3% for a group of Asian Americans with unspecified lineage, 6.6% in a group of Chinese Americans, and 22.2% in a group of Japanese Americans). One study showed that there is considerable variation among 4 different ethnic groups in China, with the frequency of CYP2A6*4 ranging from 0% to 15%. 141 East or Southeast Asians also have elevated frequencies of the CYP2A6*7, CYP2A6*9, and CYP2A6*10 alleles, which are decrease-of-function variants. These populations would be expected to be more susceptible to coumarin-induced toxicity because of the reduced capacity of the coumarin 7-hydroxylation detoxification pathway. In South Asian and Middle Eastern populations, CYP2A6*4 is present, but at a lower frequency than in East Asians.
In general, Caucasians from Europe and North America carry relatively low levels of these 7 loss-of-function and decrease-of-function alleles. CYP2A6*9 seems to be present in most of the populations tested and at higher frequencies in certain populations (eg, 16.4% in Mexicans and up to 23.6% in Asians). Lower frequencies of these 4 loss-of-function and 3 decrease-of-function polymorphisms were found in African populations, with no detection of CYP2A6*5, CYP2A6*7, or CYP2A6*10 and very low frequencies of CYP2A6*2 reported.
The presence of other decrease-of-function alleles has been assessed in various ethnicities/populations. For example, a decrease-of-function allele, CYP2A6*12, was found in Caucasian Americans (1.9%-2.4%), Iranians (1.3%), and Mexicans (3.5%). 134 -137 The decrease-of-function alleles CYP2A6*17 and CYP2A6*23 appeared to be unique to populations of African descent, 138,139,142 and the decrease-of-function allele CYP2A6*35 appears to be more common in populations of African descent than other populations. 140
The allele frequencies mentioned above show that there is great variability of the CYP2A6 genotype in populations throughout the world. Our findings are consistent with a recent worldwide distribution study on CYP2A6 alleles by population-scale sequencing, which found considerable variability and highlighted the prevalence of deficient CYP2A6 alleles in East Asians. 143 The number of carriers of loss-of-function or decrease-of-function alleles can be significant, indicating that a significant number of individuals are poor metabolizers of coumarin (ie, poor 7-hydroxylators) and are thus more susceptible to hepatotoxicity from products of the coumarin 3,4-epoxidation pathway, such as CE and o-HPA.
In vivo CYP2A6 genotype–phenotype correlation studies using coumarin as the substrate in the Thai population
Correlation studies are direct evidence of the impact of the CYP2A6 genotype on an individual’s 7-hydroxylation of coumarin, which is considered the main detoxifying pathway. Three such studies, all in the Thai population, have been summarized here. 119 -121 The findings on CYP2A6 genetic polymorphisms and urinary excretion of 7-HC from these studies are summarized in Table 19.
In Vivo CYP2A6 Genotype–Phenotype Correlation Studies Using Coumarin as the Substrate in the Thai Population.
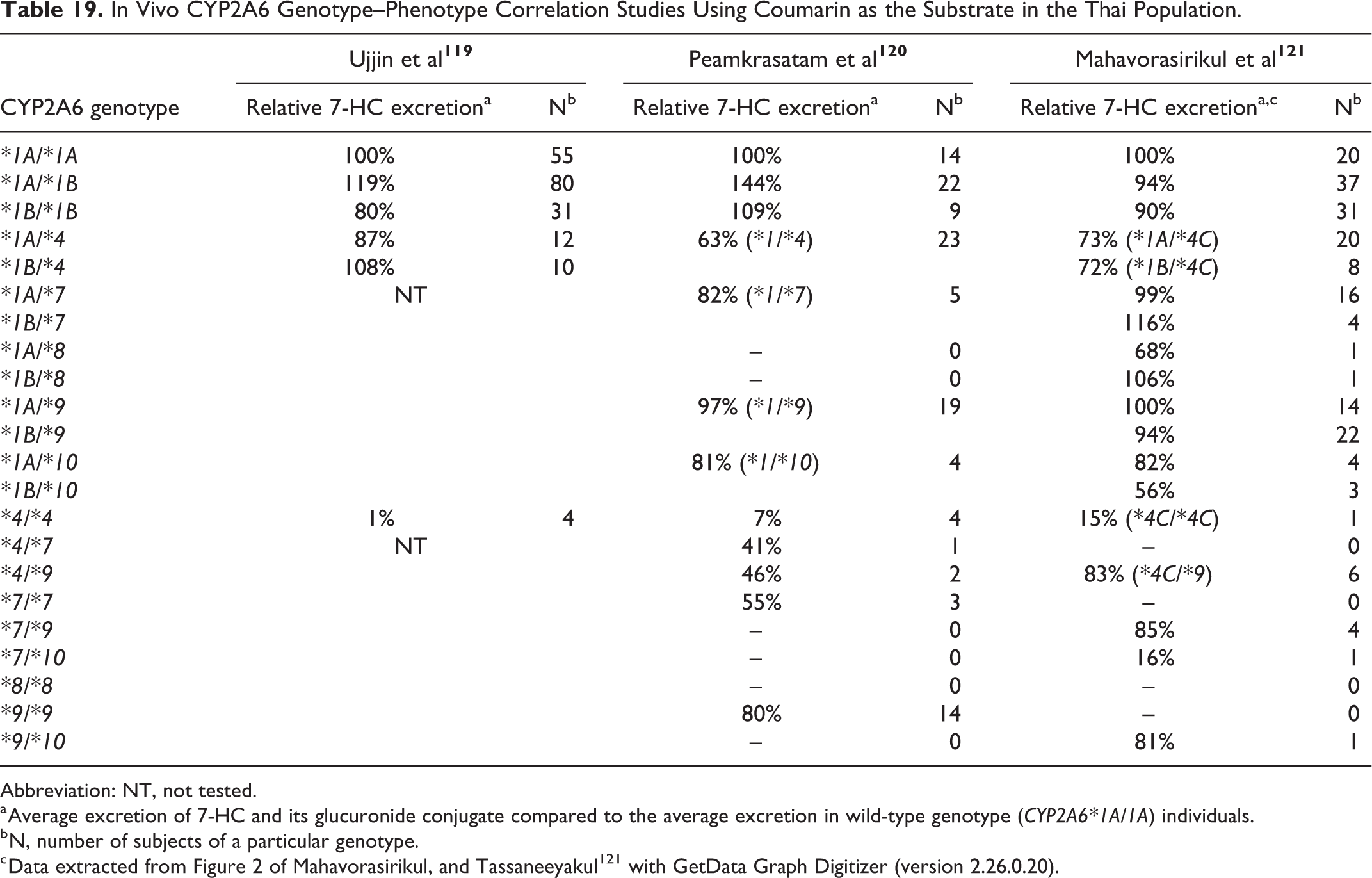
Abbreviation: NT, not tested.
a Average excretion of 7-HC and its glucuronide conjugate compared to the average excretion in wild-type genotype (CYP2A6*1A/1A) individuals.
b N, number of subjects of a particular genotype.
These studies examined the amount of 7-HC (measured together with its glucuronide conjugate) excreted in the urine after an oral dose of coumarin. The coumarin doses used by Ujjin et al, 119 Peamkrasatam et al, 120 and Mahavorasirikul et al 121 were 15, 15, and 5 mg, respectively, and the urine samples were collected 3 hours, 8 hours, and 2 hours after dosing, respectively. In another study, coumarin metabolism was tested in 10 healthy volunteers with different doses and time intervals. 75 With doses of 5, 10 and 30 mg coumarin, the amount of 7-HC (measured together with its glucuronide conjugate) recovered in the first 2 hours comprised 80.6%, 80.1%, and 79.2%, respectively, of the total 7-HC excretion in 8 hours. Two tests within 1 to 3 months in the same individuals showed that the 2-hour 7-HC excretion test is repeatable and a stable representation of total excretion. These data demonstrated that 2 hours is sufficient time for the coumarin metabolism assay and that there is very little difference among different doses below 30 mg.
The 3 studies summarized in Table 19 show some similarities. Wild-type individuals, including *1A/*1A, *1A/*1B, and *1B/*1B, were expected to be extensive metabolizers of coumarin. However, there was considerable variation among the wild-type individuals. For example, among the 55 individuals of the *1A/*1A genotype in Ujjin et al, 119 only 12 were extensive or very extensive metabolizers, 26 were moderate metabolizers, and 17 were actually poor metabolizers. Heterozygous individuals with 1 wild-type allele and 1 variant allele, such as *1A/*4, *1B/*4, *1A/*7, *1B/*7, and so on, showed variable levels of coumarin 7-hydroxylase activity, ranging from intermediate to extensive (56%-116%). Individuals with the deletion allele *4 and any of the *7, *8, or *9 alleles were generally poor metabolizers, with the exception of 6 *4C/*9 individuals showing 83% activity from the Mahavorasirikul et al’s 121 study. As shown in Table 19, 8 individuals homozygous for *4 excreted 1% to 7% 7-HC relative to wild type, while 1 person with *4C/*4C excreted 15%. The individuals with 2 decrease-of-function CYP2A6 alleles produced 7-HC with reduced activity, ranging from 16% to 85%.
Interpretation of these results is limited by small numbers of subjects for several of the variant CYP2A6 genotypes and by the fact that many other CYP2A6 alleles were discovered after these studies were completed. The presence of unknown/assessed CYP2A6 variant alleles could have confounded the results and likely contributed to the interindividual variation in coumarin 7-hydroxylase activity reported among the subjects categorized as “wild type.” In spite of these limitations, the data showed large differences in CYP2A6 activity across the different CYP2A6 genotypes. CYP2A6 enzyme activity among subjects who are heterozygous for 1 functional allele and 1 defective allele varied considerably (see Table 19 and Supplementary Table A1). Besides the genetic (known and unknown) and nongenetic factors, background exposures to coumarin (eg, from diet, tobacco, or fragrances, see the section describing occurrence, use, and exposure) and 7-HC (also known as umbelliferone, a sunscreen ingredient also present in some foods, such as carrots and golden apples) may also have contributed to the reported variation in phenotype within certain CPY2A6 genotypes. One study measured 7-HC in “blank” urine samples from 14 healthy individuals from Norway not administered coumarin and found that background levels of 7-HC in the urine averaged 254 ng/mL with a range of 0 to 2,799 ng/mL. 60
While these results provide information on identifying genotypes associated with poor metabolizers (ie, reduced or no coumarin 7-hydroxylase activity), more studies are needed regarding the distribution of CYP2A6 alleles and genotypes within specific populations, as well as the identification and characterization of presently unknown CYP2A6 genetic variants in order to better predict the susceptibility of different populations to coumarin toxicity.
Other factors that influence coumarin 7-hydroxylase activity of CYP2A6
Besides genetic polymorphisms, other factors can influence the activity of CYP2A6. These include age, gender, diet, and drugs. These factors, along with uncharacterized genetic polymorphisms, likely explain the variation in activity measured in humans with the same genotype.
Age: There are different results regarding the effect of age on CYP2A6 activity. One study with a group of 153 Spanish individuals aged 18 to 57 years concluded that age should be controlled for in epidemiological studies looking at CYP2A6 activity; older people in that study (>40 years of age) were shown to have significantly higher CYP2A6 metabolic activity, as measured using caffeine as the substrate. 144 In another study, Sotaniemi et al 145 reported that coumarin metabolism is slower in elderly people (>65 years old) compared with young adults (<25 years old). The urinary excretion of 7-HC 2 hours after a 5 mg oral dose of coumarin was 65% to 68.1% in the young subjects and 44.8% to 46.5% in the older subjects. 145 Yet in a study consisting of 100 Turkish individuals (ages 19-56), no correlation was observed between age and urinary excretion of 7-HC. 76 In a recent US study, the age of liver donors (0-87 years old) was weakly positively correlated with CYP2A6 activity using coumarin as the substrate (Spearman r = 0.13, P < 0.05). 130 Any effect of age is probably masked by genetic polymorphisms and environmental factors in the populations studied, as well as by the inconsistent age ranges the studies used for evaluation.
Gender: The effect of gender on CYP2A6 activity has been investigated in different populations. The amount of 7-HC excreted in the first 4 hours after administration was higher in females than males in a study from Finland. 75 In a study of 100 Turkish individuals, females had significantly higher urinary 7-HC excretion after 2 hours compared with males. 76 A Thai study with 101 females and 101 males showed that the average amount of 7-HC excreted by females 3 hours after dosing was 17% higher than males, and more female (N = 8) than male (N = 3) “very extensive metabolizers” were observed in the study. 119 “Very extensive metabolizers” refers to those individuals who excreted more than 96% of the administered coumarin as 7-HC. 146 In a group of African Canadians, the CYP2A6 activity (measured by nicotine metabolism) was significantly higher in females than males when smoking was controlled for. 147
Other studies have shown marginally higher activities in females or no difference between the 2 genders. In a Thai study of 120 subjects, females showed marginally higher excretion of 7-HC. 120 In a study consisting of 120 Chinese subjects, the females showed higher CYP2A6 activity than the males, though the mean ratio of 7-HC:coumarin dose for men versus women (0.637 vs 0.699, respectively) was not statistically significantly different. In a study with 50 Turkish subjects, again, there was no significant difference between male and female subjects in the percentage of dose excreted as 7-HC in an 8-hour time period. 148 A recent in vitro study using human liver bank samples from the United States reported that the formation of 7-HC in 139 female samples was higher than the 197 male samples, although the difference was not statistically significant. 130
The use of oral contraceptives in women has also been correlated with increased CYP2A6 activity. For example, the clearance of nicotine was higher in women than in men, and the use of oral contraceptives further accelerated nicotine clearance by induction of CYP2A6. 149 In a study with 178 Spanish subjects using caffeine as a CYP2A6 substrate, 26 women who were taking oral contraceptives had significantly higher CYP2A6 activity (based on measurement of caffeine metabolites in the urine) than either women who were not taking oral contraceptives or men. 144 The women who were not taking oral contraceptives had higher CYP2A6 activity than men, but the increase was not significant.
Taken together, these data suggest that CYP2A6 activity is higher in women than men. The ability of some studies to detect a gender difference may have been blunted by the distribution of genotypic polymorphisms in the test subjects (eg, uneven occurrence of defective alleles in male and female subjects in the samples tested). In studies where a gender effect was observed, lifestyle factors such as the use of oral contraceptives might have contributed to the higher level of activity seen in females.
Diet: Compounds present in vegetables and fruits have been shown to inhibit CYP2A6 activity. Grapefruit juice, a potent inhibitor of CYP3A4, also inhibits CYP2A6 mediated nicotine-to-cotinine metabolism 150 and inhibits the formation of 7-HC in humans. 151,152 Celery extract has been shown to irreversibly inhibit the coumarin 7-hydroxylation activity of human CYP2A6. 153 Phenethyl isothiocyanate, a constituent of cruciferous vegetables, competitively inhibited coumarin 7-hydroxylase activity with a Ki value of 18.2 ± 2.5 µM. 154 Additionally, diallyl disulfide, an organosulfuric compound present in garlic oil, has been shown to be a potent inhibitor of CYP2A6. 155
On the other hand, a study with 21 Finnish subjects on a strict, uncooked (raw) vegan diet and 20 omnivorous controls found no significant effect of the raw vegan diet on coumarin 7-hydroxylase activity and no difference in the incidence of phenotypically defined poor metabolizers between the 2 groups. 156
Thus, while certain compounds present in the diet have been shown to affect the metabolism of coumarin by CYP2A6, the overall effect of diet on coumarin metabolism is unclear, as diet can also affect the activity of other CYP enzymes, such as CYP3A4.
Drugs: Studies have shown that certain pharmaceutical agents can inhibit coumarin 7-hydroxylation by CYP2A6. Individuals taking these medications are more susceptible to coumarin toxicity. Isoniazid, a drug used in the treatment and prophylaxis of tuberculosis, inhibits CYP2A6 coumarin 7-hydroxylation in vitro in a time- and concentration-dependent manner. 157 Similarly, valproic acid, an antiepilepsy drug, inhibits CYP2A6 coumarin 7-hydroxylation activity in vitro in a time- and concentration-dependent manner. 158 In another study, hepatotoxicity as measured by abnormal liver function tests (LFTs) in patients taking valproate (a salt of valproic acid) was significantly higher in CYP2A6*4/*4 (loss-of-function) (P = 0.006) and CYP2A6*1/*4 (P=0.035) individuals, compared to patients with wild-type CYP2A6*1/*1. 143 The odds ratio (OR) for hepatotoxicity in CYP2A6*4/*4 individuals was 20.27 (95% confidence interval [CI]: 2.38-172.62), and the OR for hepatotoxicity in CYP2A6*1/*4 individuals was 2.46 (95% CI: 1.01-5.69). These results indicate that patients with reduced or no CYP2A6 activity are more susceptible to hepatotoxicity induced by the CYP2A6 inhibitor valproate. It is reasonable to hypothesize that poor 7-hydroxylators of coumarin who are taking drugs such as valproic acid and are simultaneously exposed to coumarin are particularly susceptible to liver toxicity induced by both chemicals.
Human hepatotoxicity
In the 1980s and 1990s, coumarin was evaluated for the treatment of lymphedema and various types of cancer. In many of these studies, a fraction of subjects treated orally with coumarin presented symptoms of hepatotoxicity and elevated LFTs. The severity of hepatotoxicity ranged from abnormal liver function as observed in blood tests to jaundice, pruritus, nausea and/or vomiting, to severe liver damage. In the mid-1990s, the Adverse Drug Reactions Advisory Committee 159 of Australia estimated that the incidence of hepatotoxicity was at least 34 cases per 10,000 users (0.34%), which is greater than the incidence for other known hepatotoxins (eg, flucloxacillin, which has approximately 0.7 cases per 10,000 users). 159 Other clinical trials of coumarin saw even higher incidences of hepatotoxicity. Some smaller clinical trials reported that up to 6% of the participants treated with coumarin developed elevated aminotransferase levels (defined as greater than double the upper limit of normal). Following these reports of hepatotoxic effects attributed to treatment, coumarin was banned in Australia in 1996 and France in 1997. 160 Coumarin is not approved to be used as a prescription drug in the United States. Only one human study, conducted in Germany, 161 investigated the association between coumarin-induced hepatotoxicity and CYP2A6 genotype. This study was limited by the small number of CYP2A6 allelic variants assessed (wild-type [*1] and 3 loss-of-function alleles [*2, *3, *4]), by the absence of individuals studied who were homozygous for loss-of-function alleles, and by the small number of heterozygous individuals who carried 1 copy of the wild-type allele and 1 copy of a loss-of-function allele (n = 6). The following summarizes clinical trials and case reports of hepatotoxicity involving orally administered coumarin.
Cox et al 162 conducted a clinical trial of 2,173 patients taking coumarin for a variety of diagnoses (chronic brucellosis, breast cancer, melanoma, advanced renal cell carcinoma, glioma, chronic infections, and chronic fatigue syndrome) in Ireland. All patients underwent medical examinations and LFTs every 3 months throughout the study. The dose of coumarin ranged from 25 to 2,000 mg daily (the majority received 50 mg/d for 2 years). Five patients developed elevated LFTs within 1 to 4 months that returned to normal while still taking coumarin (50 or 100 mg/d), of which the authors could not determine the significance. Eight patients developed elevated LFTs (including elevated aspartate aminotransferase (AST), alanine aminotransferase (ALT), ALP, and/or bilirubin) within 1 to 8 months, which was attributed to coumarin treatment (25 to 1,600 mg/d). The LFTs returned to normal after coumarin was stopped. Seven of these individuals were retreated with coumarin and developed elevated LFTs a second time, which again returned to normal after treatment was stopped. 162 This study was the basis for the incidence of 0.34% cited by Adverse Drug Reactions Advisory Committee. 159
Subsequent clinical studies observed additional cases of hepatotoxicity. Fifty-four patients in the United States with advanced malignancies were treated with 400 to 7,000 mg coumarin daily in combination with cimetidine. One patient receiving a daily dose of 5,000 mg coumarin developed hepatotoxicity and elevated LFTs (ALT, AST, gamma-glutamyl transferase [GGT]). Treatment was discontinued after 14 months, and LFTs returned to normal 3 weeks later. 163 In a clinical trial for treatment of lymphedema in the United States, 9 of 140 patients presented with elevated LFTs, including AST, ALT, GGT, ALP, and/or bilirubin, after 2 to 6 months of treatment with 400 mg coumarin daily. 111,164 The LFTs returned to normal after coumarin was withdrawn. In a study in Spain, 2 of 77 patients treated with 90 or 135 mg coumarin daily developed increased ALT after 6 months, which returned to normal without requiring treatment discontinuation. 165 In a different study, 9 of 114 patients in Germany treated with 90 mg coumarin daily in combination with troxerutin presented with elevated LFTs within 16 weeks. 166 Three of these cases were assessed as possibly due to coumarin treatment, and 1 was assessed as probably due to coumarin treatment.
A number of case reports describe patients who presented with elevated LFTs and/or hepatotoxicity following treatment with coumarin, and a few cases resulted in severe liver toxicity. Table 20 summarizes the case reports.
Summary of Case Reports of Patients Who Presented With Elevated LFTs and/or Hepatotoxicity Following Treatment With Coumarin.
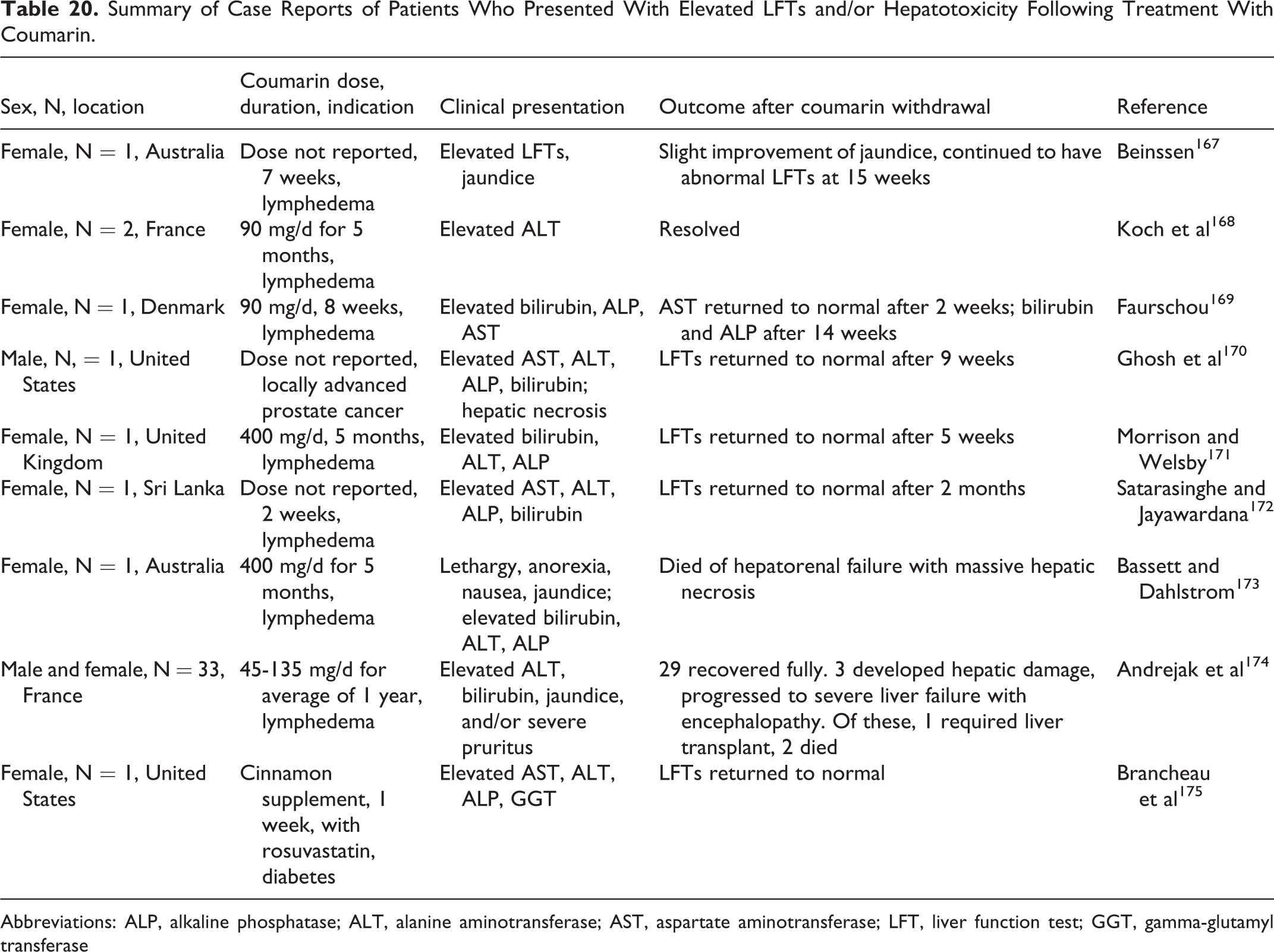
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; LFT, liver function test; GGT, gamma-glutamyl transferase
A number of clinical trials in the literature did not report hepatotoxic effects. Forty-five renal cell carcinoma patients receiving 100 mg coumarin daily in combination with cimetidine in the United States reported no liver toxicity. 176 Subsequent trials with similar dosing regimens also did not report hepatotoxicity, including clinical trials of 24 non-small cell lung cancer patients in the United States, 177 22 and 17 malignant melanoma patients in the United States and Denmark, respectively. 178 -180 14 prostate cancer patients in the United States. 181 and 31 and 50 advanced renal cell carcinoma patients in Germany and the United States, respectively. 182,183 Dexeus et al 183 reported a mild increase in ALP in 7 patients but did not report other LFTs and did not consider this to be a significant adverse effect. Three studies of 52 patients in Australia, 184 64 patients in China, 185 and 30 patients in China 186 treated with 400 mg daily did not report hepatotoxic effects. A study of 47 patients treated with 400 mg coumarin daily and 45 patients taking 400 mg coumarin daily in combination with diethylcarbamazine did not report liver toxicity. 187 Many of these studies were limited by small treated groups and incomplete reporting of side effects. The majority of these studies did not monitor patients for LFTs.
A retrospective study by Iwata et al 188 investigated the effects of Japanese herbal medications (Kampo medicines) that commonly contain Cassia cinnamon bark (which is known to contain high levels of coumarin). This study quantitatively determined the coumarin content of Kampo preparations and cinnamon bark using high-performance liquid chromatography. Of 129 patients, the authors estimated an average intake of 0.113 mg/kg/d. Twenty-three cases had abnormal LFTs, but the authors concluded that none of the cases were related to intake of cinnamon bark. It is of note that the estimated doses of coumarin from the herbal medications studied were considerably less than what patients received in the clinical trials reported above.
It is clear that a portion of the population (as high as 6.4% in US studies 111,164 ) is at risk for hepatotoxicity from pharmaceutical coumarin exposure. The abovementioned reports of adverse effects resulted in the withdrawal of coumarin for use as a pharmaceutical in Australia, France, and the United States. Hepatotoxicity does not appear to be simply dose related; some patients who were taking lower levels of coumarin presented with severe liver damage, while others who were taking high levels did not develop elevated LFTs. The hepatotoxicity appears to occur in susceptible subjects and is possibly related to genetic polymorphisms of CYP2A6 which result in decreased coumarin-7-hydroxylase activity. Farinola and Piller 113 proposed the use of pharmacogenomics to identify patients who are poor CYP2A6 metabolizers to reduce the incidence of toxicity in patients being treated for lymphedema.
There is only one study available in the literature that genotyped patients taking coumarin. 161 This study is of limited relevance since it did not test for all allele variants, and it was conducted in a homogenous population with no homozygous individuals carrying 2 copies of the variant alleles. This study genotyped 104 German patients receiving coumarin (90 mg/d) and troxerutin (540 mg/d) from the clinical trial conducted by Schmeck-Lindenau et al 166 to determine if susceptibility to coumarin-induced hepatotoxicity is determined by a polymorphism in CYP2A6. The authors tested for 3 loss-of-function CYP2A6 alleles, namely CYP2A6*2, CYP2A6*3, and CYP2A6*4. Of the 104 patients, 98 had the wild-type genotype CYP2A6*1/*1 and 6 had 1 copy of the variant allele and 1 copy of the wild-type allele. It is possible that the individuals marked as wild type in this study actually carried decrease-of-function or loss-of-function alleles the authors did not test for. Eight of the 9 patients who exhibited elevated LFTs carried the wild-type alleles. The ninth patient carried one copy of the CYP2A6*2 allele. There was no statistically significant difference in incidence of hepatotoxicity between carriers of a single loss-of-function CYP2A6 allele and those homozygous for the wild-type allele. 161
In summary, a number of clinical trials and case reports indicate that coumarin causes hepatotoxicity in susceptible individuals. The majority of cases of hepatotoxicity occurred at doses that ranged from 45 to 400 mg/d (2 occurred at doses of 1,600 and 5,000 mg/d). Although it is unclear if a direct connection between susceptibility to coumarin-induced hepatotoxicity and liver cancer can be made, it is important to consider the potential consequences of liver damage in humans following coumarin exposure, given that hepatotoxicity and liver tumors have been observed in rats and mice treated with coumarin. Hepatotoxicity caused by coumarin can lead to extensive liver damage, including necrosis, which has pro-inflammatory and tumor-promoting potential. 189 However, additional factors may also play a role in potential carcinogenesis. Susceptible individuals who are poor 7-hydroxylators may form higher amounts of the electrophilic metabolites CE and o-HPA. These compounds may also conjugate with GSH or bind covalently to cellular macromolecules resulting in hepatotoxicity. 65 A reduction or depletion of the reduced GSH pool may shift the redox balance and impact overall ability to detoxify additional reactive species (eg, reactive oxygen species [ROS]), leading to oxidative stress. Thus, multiple potential risk factors, including deficient CYP2A6 activity, liver damage, GSH depletion, and covalent binding to cellular macromolecules could contribute to the development of cancer.
Genotoxicity
The genotoxicity of coumarin has been studied in a variety of assay systems, including bacteria and fungi, in vitro systems of plant cells, mammalian cells, and human liver slices, cell-free systems, and in vivo in insects, mice, and rats. End points assessed include gene mutations, chromosomal damage (micronuclei [MN], chromosomal aberrations [CA], sister chromatid exchange [SCE]), chromosome instability (eg, deletions), and DNA damage and other effects (DNA strand breaks, DNA repair, binding to DNA, unscheduled DNA synthesis [UDS]). The findings from these assays are summarized in Tables 21 to 26.
Genotoxicity Studies of Coumarin in Salmonella typhimurium, Escherichia Coli, and Aspergillus nidulans.
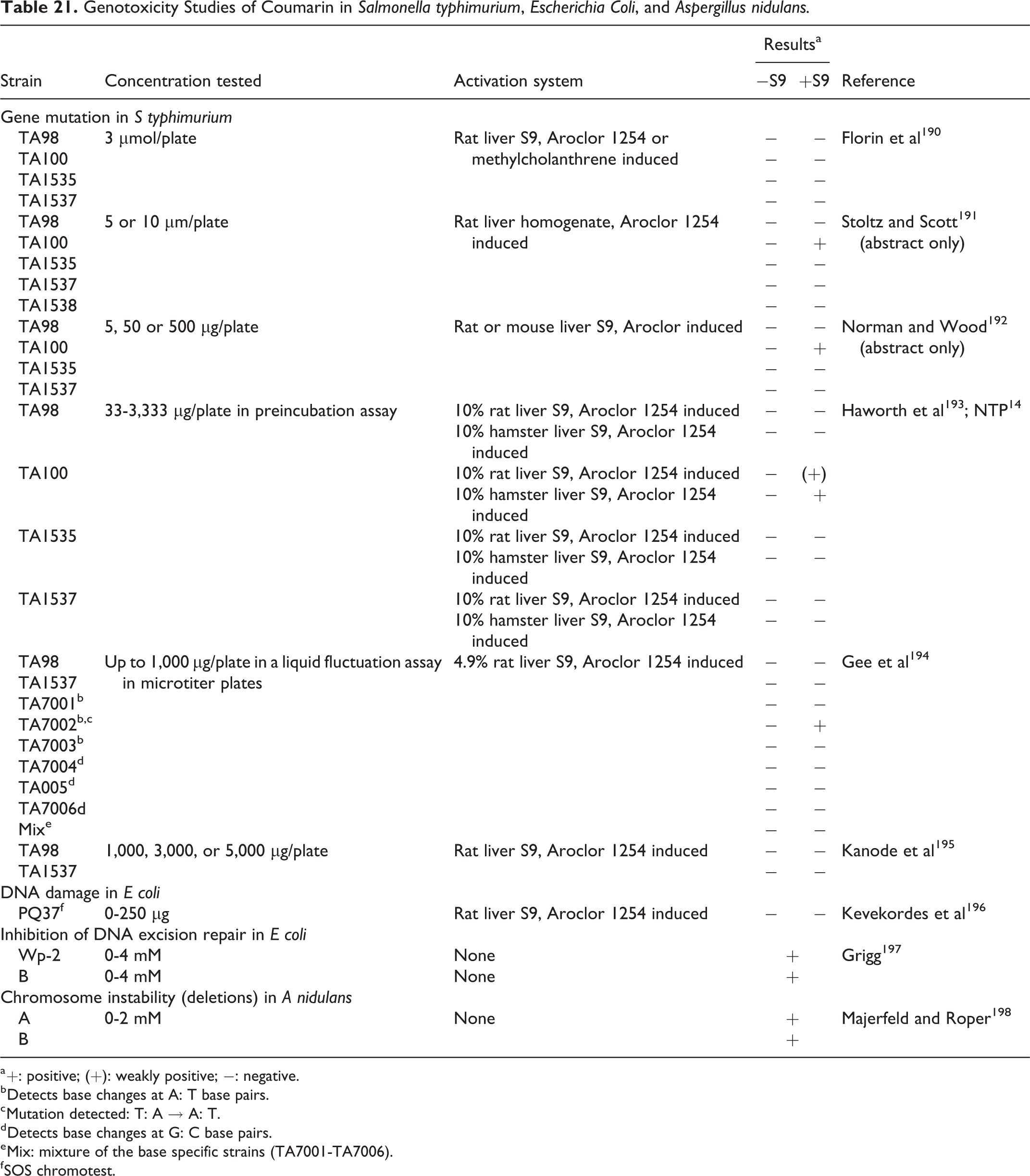
a +: positive; (+): weakly positive; −: negative.
b Detects base changes at A: T base pairs.
c Mutation detected: T: A → A: T.
d Detects base changes at G: C base pairs.
e Mix: mixture of the base specific strains (TA7001-TA7006).
f SOS chromotest.
In Vitro Genotoxicity Studies of Coumarin in Mammalian Cells, Plant Cells, and Cell-Free Systems.
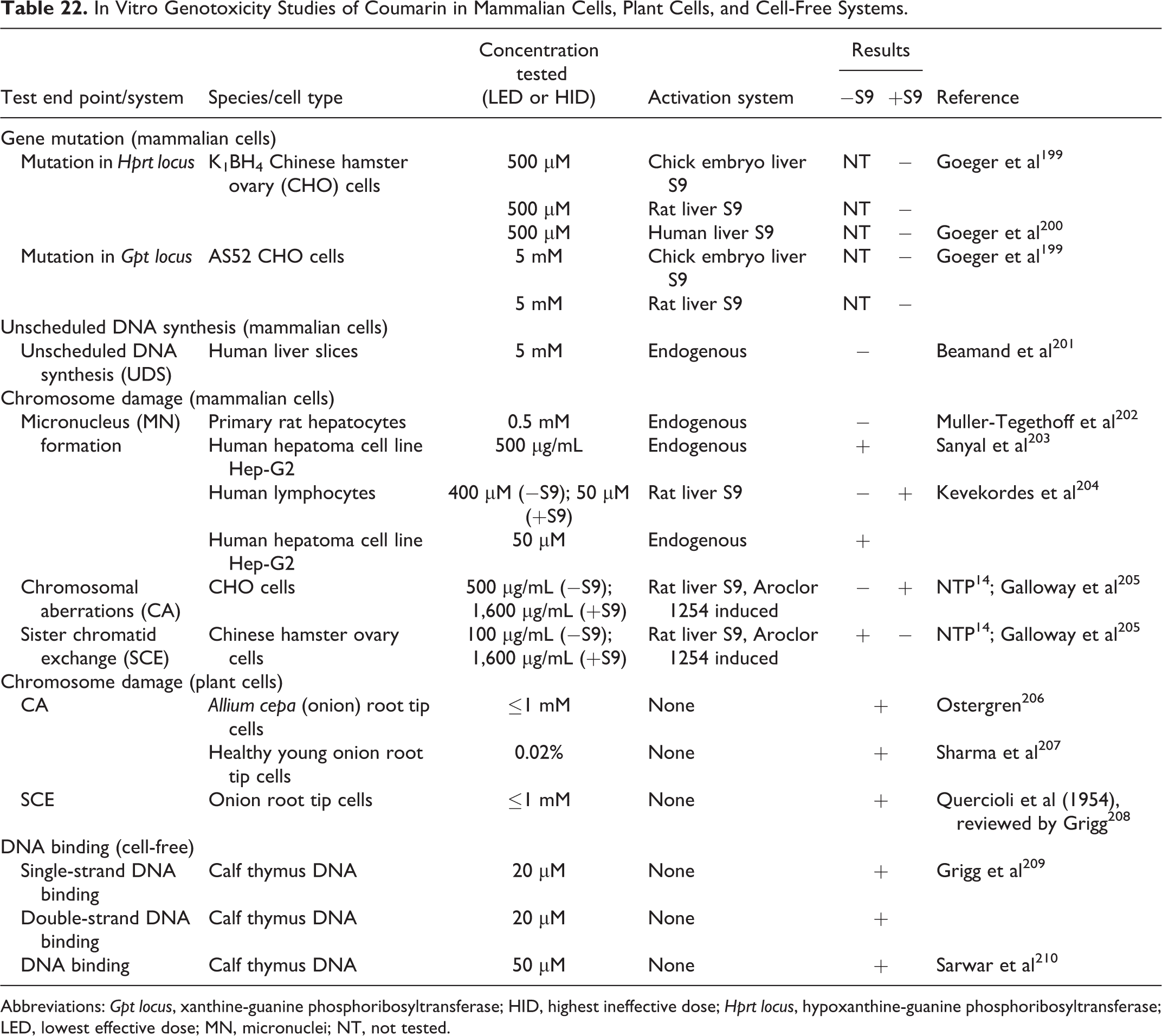
Abbreviations: Gpt locus, xanthine-guanine phosphoribosyltransferase; HID, highest ineffective dose; Hprt locus, hypoxanthine-guanine phosphoribosyltransferase; LED, lowest effective dose; MN, micronuclei; NT, not tested.
In Vitro Genotoxicity Studies of Plant Extracts Containing Coumarin in Mammalian Cells.

Abbreviations: HID, highest ineffective dose; LED, lowest effective dose; MN, micronuclei.
In Vivo Genotoxicity Studies of Coumarin.
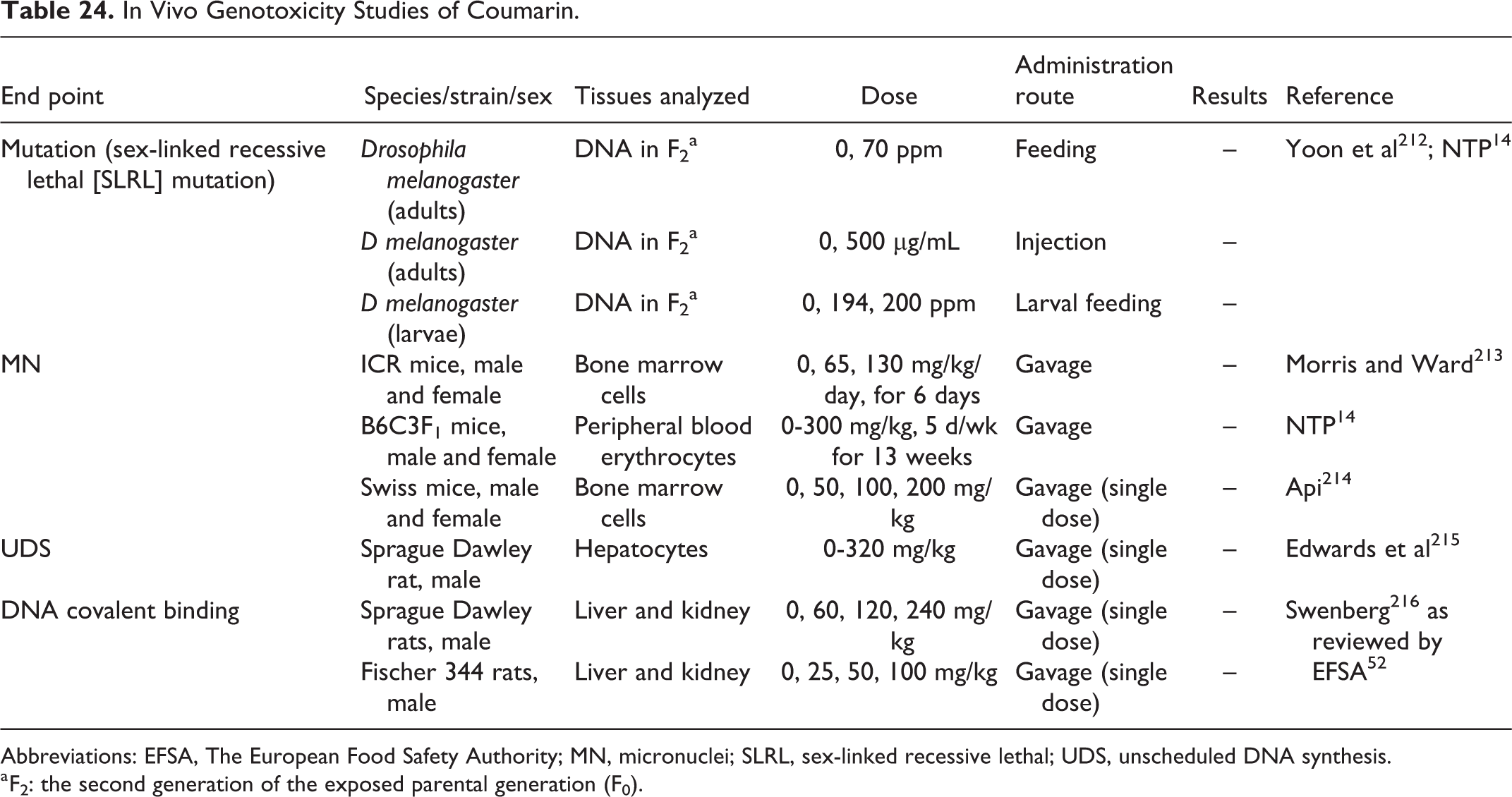
Abbreviations: EFSA, The European Food Safety Authority; MN, micronuclei; SLRL, sex-linked recessive lethal; UDS, unscheduled DNA synthesis.
a F2: the second generation of the exposed parental generation (F0).
Summary of Coumarin Genotoxicity Findings.
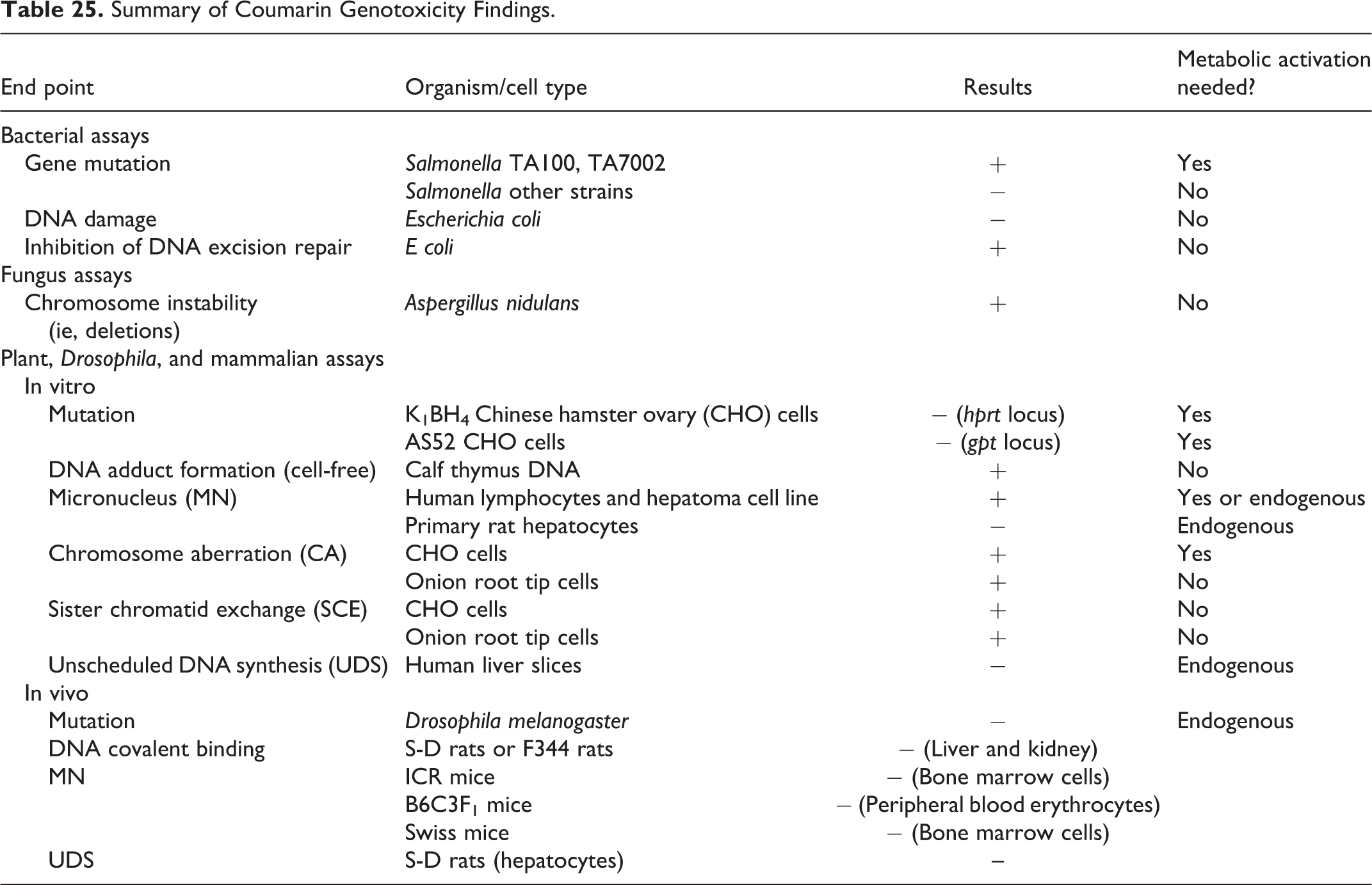
Genotoxicity Studies of Coumarin Metabolites in Salmonella Typhimurium and Escherichia Coli.
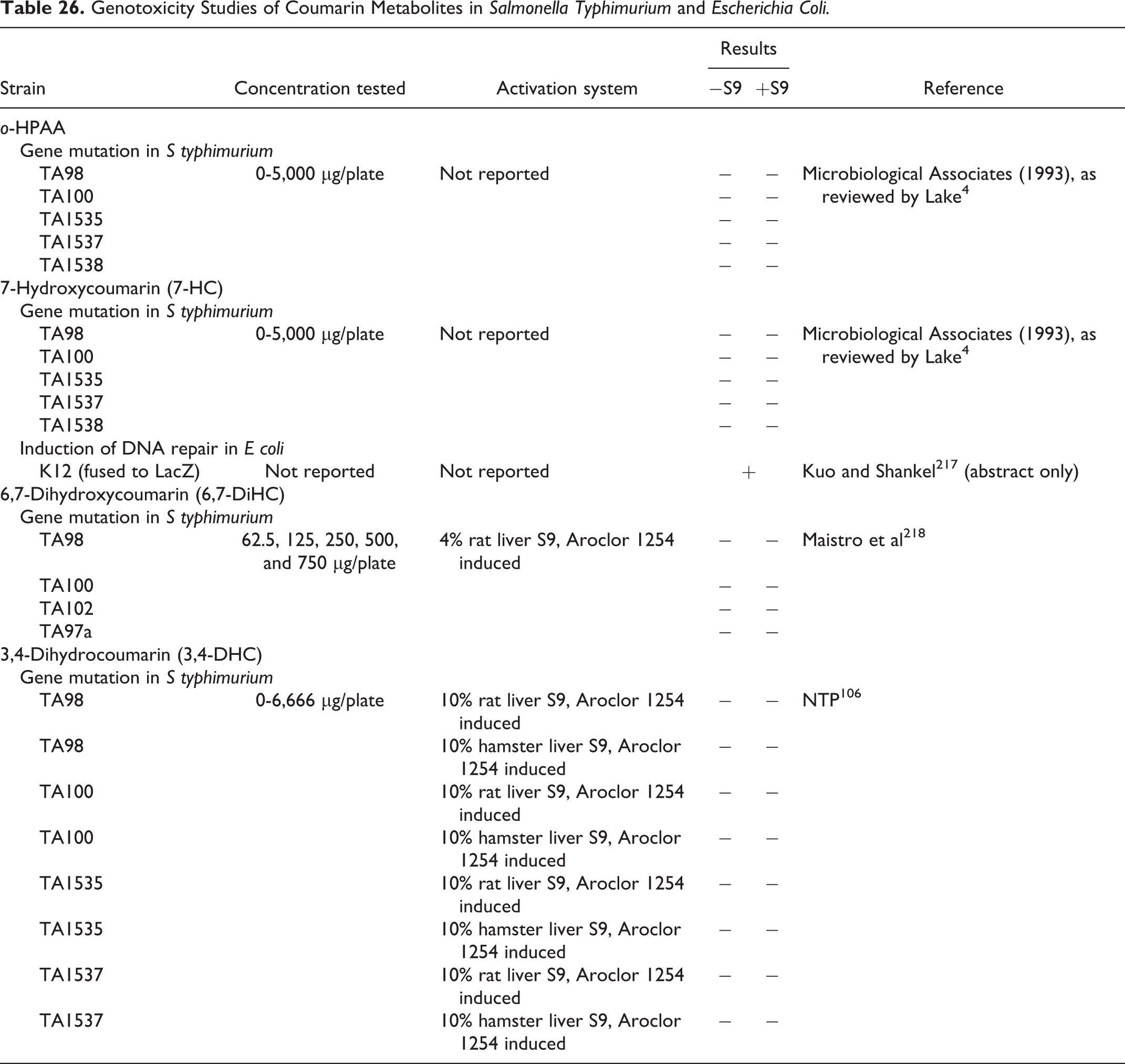
The coumarin metabolites o-HPAA, 7-HC, 6,7-DiHC and 3,4-DHC have been studied in a limited number of genotoxicity assays for a limited number of end points. Specifically, both o-HPAA and 7-HC have been studied in bacteria (gene mutation) and in mammalian cells in vitro (CA, UDS); in addition, 7-HC has been studied in 1 Escherichia coli DNA repair assay and 1 cell-free system for DNA adduct formation; 6,7-DiHC has been studied in bacteria (gene mutation), human cells in vitro (DNA strand breaks, MN), and mice in vivo (DNA strand breaks, MN), and 3,4-DHC has been studied in bacteria (gene mutation), in mammalian cells in vitro (CA, SCE), and mice in vivo (MN). The findings from these assays are summarized in Tables 27 to 29.
In Vitro Genotoxicity Studies of Coumarin Metabolites in Mammalian Cells and Cell-Free Systems.
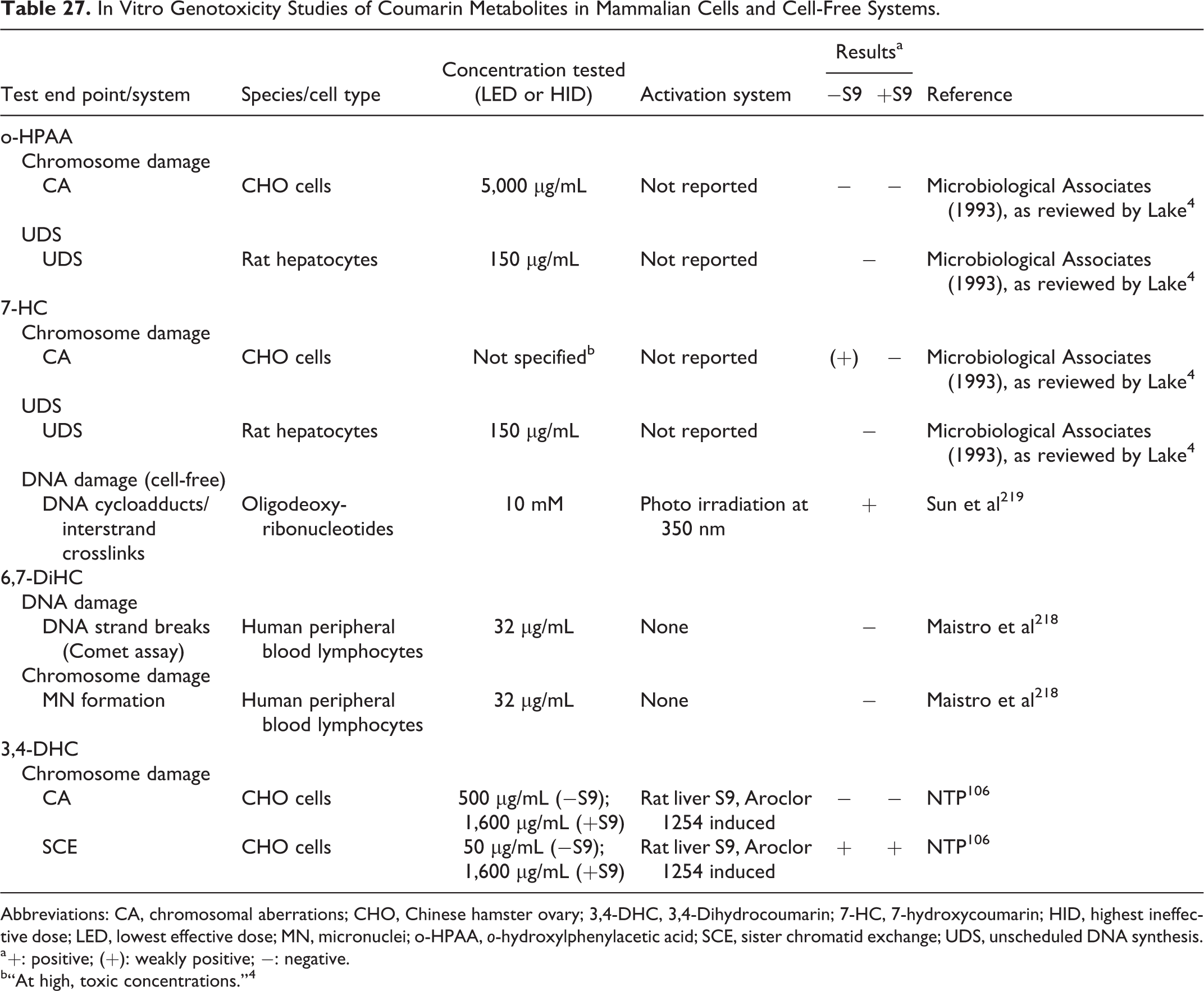
Abbreviations: CA, chromosomal aberrations; CHO, Chinese hamster ovary; 3,4-DHC, 3,4-Dihydrocoumarin; 7-HC, 7-hydroxycoumarin; HID, highest ineffective dose; LED, lowest effective dose; MN, micronuclei; o-HPAA, o-hydroxylphenylacetic acid; SCE, sister chromatid exchange; UDS, unscheduled DNA synthesis.
a +: positive; (+): weakly positive; −: negative.
b“At high, toxic concentrations.” 4
In Vivo Genotoxicity Studies of Coumarin Metabolites.

Abbreviations: bw, body weight; 3,4-DHC, 3,4-dihydrocoumarin; 6,7-DiHC, 6,7-dihydroxycoumarin; MN, micronuclei.
OEHHA’s GO and KEGG Pathway Analysis of Up- and Downregulated Genes in the Liver of Rats Administered 150 mg/kg Coumarin by Gavage 5 Times Over 28 Days (Day 1, 3, 7, 14, 28) and Euthanized 24 Hours After the Last Dose.a
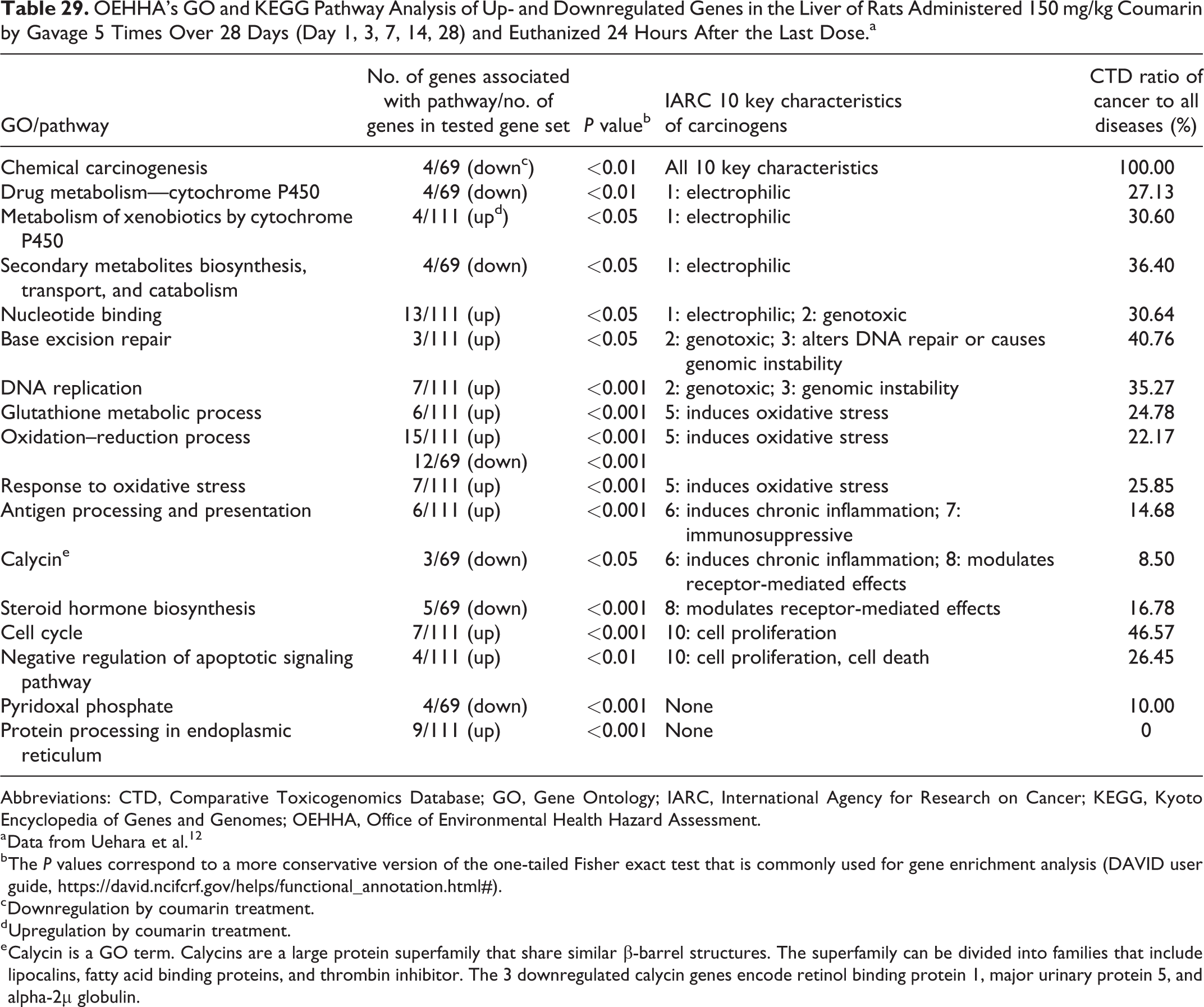
Abbreviations: CTD, Comparative Toxicogenomics Database; GO, Gene Ontology; IARC, International Agency for Research on Cancer; KEGG, Kyoto Encyclopedia of Genes and Genomes; OEHHA, Office of Environmental Health Hazard Assessment.
a Data from Uehara et al. 12
b The P values correspond to a more conservative version of the one-tailed Fisher exact test that is commonly used for gene enrichment analysis (DAVID user guide, https://david.ncifcrf.gov/helps/functional_annotation.html#).
c Downregulation by coumarin treatment.
d Upregulation by coumarin treatment.
e Calycin is a GO term. Calycins are a large protein superfamily that share similar β-barrel structures. The superfamily can be divided into families that include lipocalins, fatty acid binding proteins, and thrombin inhibitor. The 3 downregulated calycin genes encode retinol binding protein 1, major urinary protein 5, and alpha-2µ globulin.
As shown in Table 21, coumarin has been shown to be mutagenic in the presence of rat, mouse, or hamster liver S9 in the Salmonella typhimurium reverse mutation assay in strain TA100, which detects base pair substitution mutations, in multiple studies. 191 -193 When tested in a series of S typhimurium strains derived from TA100 that detect specific base pair substitutions, Gee et al 194 found that coumarin was positive in the presence of rat liver S9 in strain TA7002, which detects T: A → A: T mutations. Both strains TA100 and TA1535 contain the hisG46 marker, which is a mutation formed by the substitution of a leucine by a proline. This mutation can be reverted to the wild-type state by mutagens that cause base pair substitution mutations primarily at a GC pair. 221 TA100 and strains derived from TA100 are different from TA1535 because they also contain the plasmid PKM101, which enhances mutagenesis via an increase in the error-prone recombinational DNA repair pathway. Thus, strain TA100 is more sensitive at detecting mutagens than strain TA1535. 222 Coumarin did not induce reverse mutations in the other strains of S typhimurium in which it was tested, which detect frameshifts, not base pair substitutions (see Table 21). Coumarin was found to inhibit DNA excision repair in 2 strains of E coli 197 and to induce chromosome instability (deletions) in Aspergillus nidulans. 198 Coumarin did not induce DNA damage in the SOS chromotest in E coli.
As shown in Table 22, coumarin was positive in many, but not all in vitro genotoxicity studies, causing DNA and chromosomal damage in human cells, other mammalian cells, and plant cells in vitro, and binding to calf thymus DNA (both single-stranded and double-stranded DNA).
Coumarin did not induce mutations in the Hprt locus or the Gpt locus in Chinese hamster ovary (CHO) cells in the presence of chick embryo, rat, or human liver S9. 199,200 Coumarin did not induce UDS in human liver slices. 201
Coumarin induced micronucleus formation (MN) in human lymphocytes in the presence of rat liver S9 204 and in 2 studies with the Hep-G2 human hepatoma cell line. 203,204 Coumarin did not induce MN in primary rat hepatocytes. 202 It is unclear whether the type of metabolic activation system used in the individual studies can explain the different findings for MN induction. In CHO cells, coumarin induced CA in the presence and SCE in the absence of rat liver S9. 14,205 In studies with Allium cepa (onion) root tip cells, coumarin induced CA in 2 studies 206,207 and SCE in another (Quercioli et al 1954, as reviewed by Grigg. 208 Coumarin binds to calf thymus DNA. 209,210 In the studies by Grigg et al, 209 coumarin was shown to bind to both single-stranded and double-stranded DNA. In the studies by Sarwar et al, 210 the mode of binding of coumarin with calf thymus DNA was investigated through various biophysical techniques, including iodide-induced quenching, competitive binding assays with ethidium bromide, acridine orange, and Hoechst 33258, and in silico molecular docking studies. The authors concluded that coumarin possibly binds to the minor groove of DNA, likely by forming hydrogen bonds.
Costa et al 211 investigated the genotoxicity of an infusion (prepared in phosphate-buffered saline) and an ethanol extract (80% ethanol) of the South American medicinal plant Mikania glomerata, known as “guaco,” which contains coumarin. As shown in Table 23, both the coumarin-containing infusion and the ethanol extract induced DNA strand breaks in the comet assay in rat hepatoma cells. The infusion also induced MN in rat hepatoma cells, but the ethanol extract did not.
As shown in Table 24, in vivo studies of coumarin have not demonstrated genotoxicity. Specifically, coumarin did not induce sex-linked recessive lethal (SLRL) mutations in Drosophila melanogaster exposed to coumarin in feed as adults or larvae or by injection as adults. 14,212 Coumarin did not increase MN formation in peripheral blood erythrocytes of male and female B6C3F1 mice after 13 weeks of exposure, 14 bone marrow cells of male or female Swiss mice after a single gavage dose, 214 or bone marrow cells of male and female ICR mice after 6 daily gavage doses. 213 Coumarin did not induce UDS in the hepatocytes of S-D rats following a single gavage dose, 215 and covalent binding of [14C] coumarin to DNA was not observed in the liver or kidney of S-D or Fischer 344 rats (Swenberg, 216 as reviewed by The European Food Safety Authority 52 ). An overall summary of the genotoxicity findings for coumarin is presented in Table 25.
Studies of coumarin metabolites
As shown in Table 26, the coumarin metabolite 7-HC induced expression of ada, a gene associated with DNA repair, in E coli. 217 None of the 4 coumarin metabolites tested in the S typhimurium reverse mutation assay (ie, o-HPAA, 7-HC, 6,7-DiHC, and 3,4-DHC) induced mutations in various S typhimurium tester strains (with or without exogenous metabolic activation (S9); see Table 26).
As shown in Table 27, 7-HC was weakly positive in the absence of exogenous metabolic activation in an assay for the induction of CA in CHO cells (Microbiological Associates, 1993, as reviewed by Lake 4 ). Sun et al 219 found that incubation of 7-HC with oligodeoxyribonucleotides and photoirradiation at 350 nm results in the formation of 7-HC-DNA cycloadducts with thymine and cytosine, and DNA interstrand crosslinks. Cycloadduct formation was reversible with photoirradiation at 254 nm, however. 7-hydroxycoumarin did not induce UDS in rat hepatocytes (Microbiological Associates, 1993, as reviewed by Lake 4 ).
The metabolite 3,4-DHC induced a dose-related increase in SCE in the absence of exogenous metabolic activation, as well as an increase in the presence of exogenous metabolic activation (which was dose related in one of 2 replicate studies). 106 3,4-Dihydrocoumarin did not induce CA in CHO cells (see Table 27).
Two in vitro mammalian genotoxicity studies have been reported to date for o-HPAA (CA in CHO cells and UDS in rat hepatocytes), each of which was negative. In addition, 6,7-DiHC did not induce DNA strand breaks or MN formation in human peripheral blood lymphocytes (see Table 27).
As shown in Table 27, only 2 coumarin metabolites (6,7-DiHC and 3,4-DHC) have been tested for genotoxicity in vivo, and these studies have been negative. Specifically, 6,7-DiHC did not induce DNA strand breaks in Swiss albino mice in peripheral blood cells at 4 or 24 hours or in liver, bone marrow, or testicular cells at 24 hours after a single gavage dose and did not induce MN in bone marrow cells at 24 or 48 hours after a single gavage dose. 220 3,4-Dihydrocoumarin did not induce MN in peripheral blood cells of B6C3F1 mice following 13 weeks of exposure. 106
Cell transformation studies
Cell transformation assays are designed to detect a change in the growth pattern of cells that is indicative of loss of contact inhibition, a phenotype that is characteristic of cancer cells. The effect of coumarin on cell transformation has not been adequately studied.
Nashed and Brendel 223 studied in vitro cell transformation of rat peritoneal cells from animals exposed to coumarin in vivo, using the peritoneal cell test. This assay was developed as a short-term in vivo–in vitro alternative to rodent cancer bioassays, and the testing of coumarin was an early effort at assay validation. This test system was never widely used, and interpretation of results from use of this assay are uncertain. Coumarin was administered as a single gavage dose of 0, 7.5, 30 and 60 mg/kg bw to groups of 10 male and female Wister rats. Six hours after dosing, the animals received 10 mL of a mitogenic medium by IP injection to stimulate peritoneal cell proliferation. Animals received a second IP injection of mitogenic medium 7 days after dosing. Two weeks after dosing, peritoneal cells were harvested and cultured for colony growth in soft agar for 7 days. Colony growth in soft agar is indicative of cell transformation. 224,225 Cultures were scored for colony size, colony frequency, and colony survival. Positive evidence of colony growth was observed in cultures from 1 out of 10 low- and high-dose males, 1 out of 10 mid-dose females, and 2 out of 10 high-dose females, as compared to 2 of 10 male and 3 of 10 female positive controls (dimethylnitrosamine) and no evidence of colony growth in cultures from water controls of both sexes.
Milo et al 226 studied the inhibition of N-methyl-N-nitro-N-nitrosoguanidine (MNNG) or methylazoxymethanol acetate (MAMA)-induced cell transformation of human fibroblasts by coumarin and other chemicals that inhibit the nuclear poly(ADP-ribose) polymerase system. Coumarin in combination with MNNG or MAMA inhibited the formation of transformed colonies at 69 µM (a nontoxic dose that inhibits transformation by 85%-95%). A slight, but not statistically significant, elevation in the number of transformed colonies per 50,000 cells (n = 13.0 ± 4) was observed with coumarin (69 µM) treatment alone.
Toxicogenomic data
This section summarizes the findings from studies that generated or analyzed toxicogenomic data on coumarin. These toxicogenomic data come from multiple research groups and include gene expression data in mouse lung following exposure in vivo from 1 publication, 227 in rat liver following exposure in vivo from 5 publications, 12,228 -231 in rat primary hepatocytes exposed in vitro from 2 publications, 12,228 and in human primary hepatocytes exposed in vitro from 2 publications. 12,232 An additional analysis by OEHHA of the in vivo rat liver toxicogenomic data from Uehara et al 12 is also presented.
In vivo data
Mouse lung: Thomas et al 227 identified lung cancer biomarkers using microarray data from female B6C3F1 mice. Thirteen diverse chemicals were chosen based on lung tumor findings in NTP carcinogenesis studies in female B6C3F1 mice: 7 lung carcinogens, including coumarin, and 6 noncarcinogens (ie, chemicals that did not induce lung tumors in female mice). The coumarin treatment group consisted of 5 female mice administered coumarin in corn oil by gavage at a dose of 200 mg/kg body weight per day, 5 days per week for 13 weeks. The study also included an appropriate vehicle control group. After 13 weeks, animals were euthanized and the lung tissues collected for total RNA extraction and gene expression analysis with Affymetrix Mouse Genome 430 2.0 arrays.
A comparison of gene expression data from mice exposed to lung carcinogens and noncarcinogens revealed a total of 82 probe sets corresponding to 75 unique transcripts that were significantly altered (65 were upregulated and 10 were downregulated). A GO analysis using DAVID indicated that these gene expression changes are associated with several different biological processes categories (eg, GSH metabolism and lipid metabolism) and molecular function categories (eg, GSH transferase activity, oxidoreductase activity). The top 6 gene expression biomarkers identified as discriminating between lung carcinogens and noncarcinogens were from the following genes: UDP-glucuronosyltransferase 1a (Ugt1a) family, carboxylesterase 1 (Ces1), fibroblast growth factor receptor 2 (Fgfr2), epoxide hydrolase 1, microsomal (Ephx1), GSH S-transferase mu 1 (Gstm1), and an unannotated gene. Four gene products are enzymes involved in endogenous and xenobiotic metabolism. One gene product is a growth factor receptor involved in lung development. In validation studies, coumarin treatment was shown to result in significant changes in gene expression of 2 of these biomarkers (Ces1 and Ephx1) in female mouse lung by quantitative reverse transcription polymerase chain reaction.
Rat liver: Kienhuis et al 228 reported the gene expression changes in 9- to 12-week-old male Wistar rats administered coumarin dissolved in corn oil by a single IP injection at 0, 17.5, 75, or 200 mg/kg (5 animals per dose group). Animals were euthanized 24 hours after dosing and liver samples were prepared for RNA extraction. RNA samples were extracted and labeled prior to hybridization for gene expression analysis with QIAGEN Operon oligonucleotide microarrays containing approximately 5,800 different 70-mer oligonucleotide fragments. A total of 321 significantly altered genes and 6 pathways (ie, 3 metabolic-related pathways [methionine metabolism, tryptophan metabolism, and fatty acid metabolism], γ-hexachlorocyclohexane degradation, complement and coagulation cascades, and citrate cycle) were identified in vivo.
Kiyosawa et al 229 developed 161 GSH depletion-responsive gene probe sets to identify chemicals that perturb GSH homeostasis in rat liver. The authors grouped the probe sets into the following 5 categories: “antioxidant, phase II drug metabolizing enzymes and oxidative stress markers,” “transporter,” “metabolism,” “transcription factors and signal transduction-related and protein turnover-related genes,” and “miscellaneous.” The authors tested the usefulness of the GSH depletion-responsive gene probe sets using several prototypical GSH depletors, including coumarin. Five six-week-old male Crj: CD(S-D)IGS rats were treated with a single dose of 150 mg/kg coumarin orally. Blood samples were collected at 3, 6, 9 and 24 hours after treatment to assay for markers of liver toxicity; no indicators of liver toxicity were observed in coumarin-treated animals. Animals were euthanized 24 hours after treatment, livers removed, and total RNA extracted for gene expression analysis with Affymetrix GeneChip RAE 230A probe arrays. Principal component analysis was applied to the gene expression data using the GSH depletion-responsive gene probe sets. Coumarin-treated rats showed the second most affected gene expression profile among the 15 chemicals studied (after bromobenzene, the most potent GSH depletor). This is consistent with previous reports of reactive metabolites generated from coumarin oxidation in the liver being involved in coumarin-induced GSH depletion. 110,233
In the Uehara et al’s 12 study, 6-week-old male S-D rats were exposed to coumarin in corn oil by the oral route at 15, 50, or 150 mg/kg (5 animals per dose) on days 1, 3, 7, 14, and 28. The rats were euthanized 24 hours after the last dose and liver samples were obtained immediately after euthanasia for total RNA extraction and gene expression analysis with Affymetrix GeneChip RAE 230A probe arrays. This study is part of the Genomics Assisted Toxicity Evaluation System, for the Toxicogenomics Project conducted in Japan (TG-GATEs).
Statistically differentially expressed genes were identified, including 136 upregulated and 79 downregulated probe sets related to GSH metabolism and oxidative stress response. The most sensitive genes identified at the lowest dose were aldo-keto reductase family 7, member A3 (Akr7a3), NAD(P)H dehydrogenase, quinone 1 (Nqo1), GSH reductase (Gsr), GSH-S-transferase, pi 1/2 (Gstp1/Gstp2), and GSH S-transferase Yc2 subunit (Gsta5). These 5 genes are involved in GSH metabolism and cellular responses to oxidative stress.
Uehara et al 230 applied a toxicogenomics approach to develop a Prediction Analysis of Microarray (PAM) classifier, consisting of 112 rat liver gene expression probe sets, to identify nongenotoxic hepatocarcinogens causing oxidative stress. Validation studies on the PAM classifier were conducted with 30 chemicals classified by the authors as either nongenotoxic rat liver carcinogens that cause oxidative stress or noncarcinogens. Coumarin was included as one of the rat liver carcinogens. This study treated 6-week-old male S-D rats orally with 150 mg/kg coumarin in corn oil using 2 dosing schemes: (1) a single dose administered, with animals being euthanized at 3, 6, 9, or 24 hours after dosing, and (2) repeated doses administered daily for 3, 7, 14 or 28 days with euthanasia 24 hours after the last dose (corresponding to days 4, 8, 15, and 29, respectively). Liver samples were obtained immediately after euthanasia for total RNA extraction and gene expression analysis with Affymetrix GeneChip RAE 230A probe arrays. The authors concluded that the PAM classifier correctly predicted coumarin to be a hepatocarcinogen causing oxidative stress and noted that time-dependent increases in the PAM score were observed with coumarin treatment.
Eichner et al 231 developed 2 new approaches to select robust gene expression signatures to predict nongenotoxic carcinogens in rat liver using the TG-GATEs database (http://toxico.nibiohn.go.jp/english/). Rat liver gene expression data for 2 chemicals classified as genotoxic carcinogens, 9 as nongenotoxic carcinogens (including coumarin), and 11 as noncarcinogens were analyzed using these approaches. Both approaches predicted that coumarin is a hepatocarcinogen. The top 5 genes incorporated into prediction models were phosphatidylinositol-3,4,5-trisphosphate binding protein (Phlda3), cyclin-dependent protein kinase (Cdkn1a), NADP aldo-keto reductase (Akr7a3), Cyclin G1 (Ccng1) and ATP-binding cassette (Abcb4), all of which were altered by treatment with coumarin. These genes are related to either p53 (a tumor suppressor) signaling or to specific changes in anabolic processes or energy metabolism that are typically found in tumor cells. The NADP aldo-keto reductase gene Akr7a3 was also among the affected genes in vitro and among the 5 most sensitive genes identified at the lowest dose in vivo as having altered expression in the analysis by Uehara et al 12 .
The OEHHA’s analysis of toxicogenomic data from Uehara et al (2008) 12
In order to further characterize the effects of coumarin on cancer-associated biological processes and pathways, OEHHA conducted additional analyses of the toxicogenomic data of Uehara et al 12 using DAVID 233 and the CTD (http://ctdbase.org/, assessed June 14, 2017). Details of the output from DAVID are presented in Appendix B.
Among the 136 upregulated genes, 111 were recognized by DAVID and grouped into 17 annotation clusters of GO biological processes or KEGG pathways. Among the 79 downregulated genes, 69 were recognized by DAVID and grouped into 12 annotation clusters. In Table 29, findings with statistical significance as determined by a modified Fisher exact test (P < 0.05) are presented, consisting of 10 annotation clusters of upregulated genes and 8 annotation pathways of downregulated genes. The modified Fisher exact test was used to determine whether the number of upregulated (or downregulated) genes in a given annotation pathway were significantly different (or “enriched”; see Supplementary Tables B1-B4).
The CTD was used to ascertain if any of the annotation clusters identified in the DAVID functional annotation clustering analysis are associated with cancer. The percentage of disease associations for a given GO or pathway cluster that were specifically related to cancer is shown in Table 29 as the CTD ratio of cancer to all diseases. The higher the CTD ratio of cancer to all diseases, the stronger the association of the annotation cluster with cancer.
The results of our analysis indicate that 10 of the 11 GO or pathway clusters identified as genes upregulated by coumarin in rat liver are cancer-associated pathways or biological processes with CTD cancer association ratios ranging from 47% to 15% (Table 29). Ranked in order of highest association to lowest, these are cell cycle, base excision repair, DNA replication, aging, nucleotide binding, metabolism of xenobiotics by CYP, negative regulation of apoptotic signaling pathway, response to oxidative stress, GSH metabolic process, oxidation–reduction process, and antigen processing and presentation.
Five of the seven GOs or pathways identified as genes downregulated by coumarin in rat liver have CTD cancer association ratios ranging from 100% to 17%. Ranked from highest association to lowest, these are chemical carcinogenesis, two metabolic-related pathways (“secondary metabolites, biosynthesis, transport, and catabolism”, and “drug metabolism—CYP”), oxidation–reduction process, and steroid hormone biosynthesis.
The 10 key characteristics of carcinogens 234 were also applied to assist in recognizing cancer-associated pathway clusters among those identified by the DAVID analysis (Table 29). Each of the 10 key characteristics was associated with at least one annotation cluster of genes with significantly altered expression in rat liver following in vivo coumarin treatment.
To summarize, our analysis provides additional evidence that administration of coumarin to rats in vivo induces changes in gene expression associated with cancer pathways and biological processes and is consistent with the findings of Carlton et al 15 that coumarin induces liver tumors in male and female S-D rats. Our analysis identified multiple pathways that may be involved in coumarin-induced liver tumor formation in rats, including 2 major pathways of response to oxidative stress and the GSH metabolic process that have also been identified by other researchers using either toxicogenomics 229,230 or traditional toxicological approaches. 110,233,235,236
In vitro data
Rat hepatocytes: Because of the expected differences in the extent to which coumarin is metabolically activated in hepatocytes in vitro versus in liver in vivo, the information reported in these in vitro studies may provide a limited picture of what happens in the in vivo system. In general, hepatocytes have less metabolic capacity in vitro than in vivo. Nevertheless, Kienhuis et al 228 and Uehara et al 12 reported some common genes that were significantly modulated by coumarin exposure in both rat liver in vivo and rat hepatocytes in vitro.
In the Kienhuis et al’s 228 study, rat primary hepatocytes were prepared from 3 untreated 9- to 12-week-old male Wistar rats. The primary rat hepatocytes were sandwich-cultured between 2 collagen layers in either a standard medium or an enhanced medium containing low concentrations of CYP inducers (phenobarbitol, dexamethasone, and β-naphthoflavone) for 72 hours and then exposed to coumarin at doses of 0, 70, 200, and 600 µM for 24 hours. RNA samples were extracted and labeled prior to hybridization for gene expression analysis with QIAGEN Operon oligonucleotide microarrays containing approximately 5,800 different 70-mer oligonucleotide fragments. o-Hydroxyphenylacetic acid, a metabolite of coumarin, was measured in the culture media after 24 hours of exposure to coumarin. Comparisons of significantly modulated genes and biological pathways were made among hepatocytes cultured with the standard versus the enhanced medium and with the findings from the in vivo rat hepatic gene expression studies. Similar to the in vivo finding that o-HPAA was detected in rat urine, o-HPAA was only identified from hepatocytes cultured in the enhanced medium, but not the standard medium. The number of genes with significantly altered expression in response to coumarin treatment was 321 in liver in vivo, 13 in hepatocytes cultured with standard medium, and 92 in hepatocytes cultured with enhanced medium. Only one gene was altered in rat liver in vivo and in both in vitro hepatocyte systems, and an additional 23 were altered both in vivo and in the enhanced system. This demonstrates that the enhanced in vitro system with added metabolic activation capacity is better able to simulate the in vivo system and indicates that metabolic capacity is an important factor to consider when evaluating and comparing results from in vivo and in vitro toxicogenomic studies. Biological pathway analysis of the genes with altered expression identified 4 biological pathways that were shared between the in vivo liver response and the hepatocyte response in the enhanced in vitro system: methionine metabolism, fatty acid metabolism, γ-hexachlorocyclohexane degradation, and complement and coagulation cascades.
In the Uehara et al’s 12 study, rat primary hepatocytes were prepared from livers of 6-week-old male S-D rats following IP injection with 120 mg/kg sodium pentobarbital. The primary hepatocytes were exposed to coumarin at doses of 0, 12, 60, and 300 µM for 24 hours and the total RNA extracted for gene expression analysis with Affymetrix GeneChip RAE 230A probe arrays. The authors compared the changes in gene expression observed in response to coumarin in rat liver in vivo with those in primary rat hepatocytes exposed in vitro. Fewer responsive genes were observed in the primary rat hepatocytes than in rat liver in vivo, and smaller fold changes were observed in vitro in those responsive gene probe sets, possibly due to limited metabolic activation in the in vitro system. The authors identified 37 upregulated and 29 downregulated gene probe sets as being differentially expressed in response to coumarin in both the rat liver in vivo and in vitro data sets. Many of these genes are involved in pathways related to the oxidative stress response or GSH metabolism. For example, the upregulated genes include hypoxia upregulated 1 (Hyou1), aldo-keto reductase family 7-member A3 (Akr7a3), ischemia/reperfusion inducible protein (Yrdc), GSH reductase (Gsr), glutamate-cysteine ligase-catalytic subunit (Gclc), NAD(P)H dehydrogenase, quinone 1 (Nqo1), and DNA-damage inducible transcript 4-like (Ddit4 l).
Human hepatocytes: In the study by Uehara et al, 12 frozen human hepatocytes were obtained from a commercial source. After thawing and plating, hepatocytes were exposed to coumarin at doses of 0, 12, 60, and 300 µM for 24 hours, and total RNA extracted for gene expression analysis with Affymetrix U133 Plus 2.0 arrays. Based on the differentially expressed probe sets identified in response to coumarin treatment as common in both rat liver in vivo and rat primary hepatocytes in vitro, human orthologs were identified for 14 upregulated and 11 downregulated probe sets. Many of these genes are involved in pathways related to the oxidative stress response or GSH metabolism. Similar expression patterns were observed in these 14 upregulated and 11 downregulated probe sets in cultured human hepatocytes treated with coumarin as in rat primary hepatocytes in vitro and rat liver in vivo. Smaller fold changes were observed in response to coumarin in human hepatocytes than in rat hepatocytes; however, interpretation of this finding is limited in the absence of information on the relative metabolic competencies of the human and rat hepatocytes.
Using different methods, Kienhuis et al 232 obtained complete data sets of coumarin-induced gene expression profiles in primary human hepatocytes from 5 donors. The sandwich-cultured primary human hepatocytes were grown between 2 collagen layers on collagen gel precoated plates. The human hepatocytes were then exposed to coumarin at 2 doses for 24 hours: 200 µM (equivalent to 100 mg/kg/d for a 70-kg person) and 600 µM (a proposed toxic dose). The labeled cRNA samples were hybridized on Agilent 22 K format 60-mer oligo microarrays (∼20,000 probes, G4110B for human from Agilent Technologies, Palo Alto, California); then gene expression data were analyzed. A total of 198 genes and 619 genes were significantly modulated at 200 and 600 µM coumarin, respectively, with an overlap of 135 differentially expressed genes. A clear dose–response relationship of differential gene expression was observed at the 2 doses. No cytotoxicity was observed in human hepatocytes at either of the doses.
The gene expression data were analyzed by T-profiler (http://www.t-profiler.org and Boorsma et al 237 ). At the 200 µM dose, downregulated complement and coagulation cascades were observed. At 600 µM (Table 30), several pathways and processes were affected, including upregulation of transcription and protein folding-related pathways, and downregulated complement and coagulation cascades, lipid metabolism pathways, oxidoreductase activity, metabolism of xenobiotics by CYPs, repression of energy-consuming biochemical pathways, and impairment of mitochondrial function. Oxidoreductase activity and metabolism of xenobiotics by CYPs are related to the 10 key characteristics of carcinogens identified by an IARC working group. 234 Reprogramming energy metabolism was defined as one of the 6 cancer hallmarks in a review published by Hanahan and Weinberg. 189 Decreased mitochondrial membrane potential is observed in cancer cells in vitro and linked with cellular properties associated with cancer progression. 238 -240
Up- and Downregulated Pathways and Biological Processes by Coumarin in Sandwich-Cultured Primary Human Hepatocytes In Vitro 232 and Liver Tissues of S-D Rats In Vivo.a
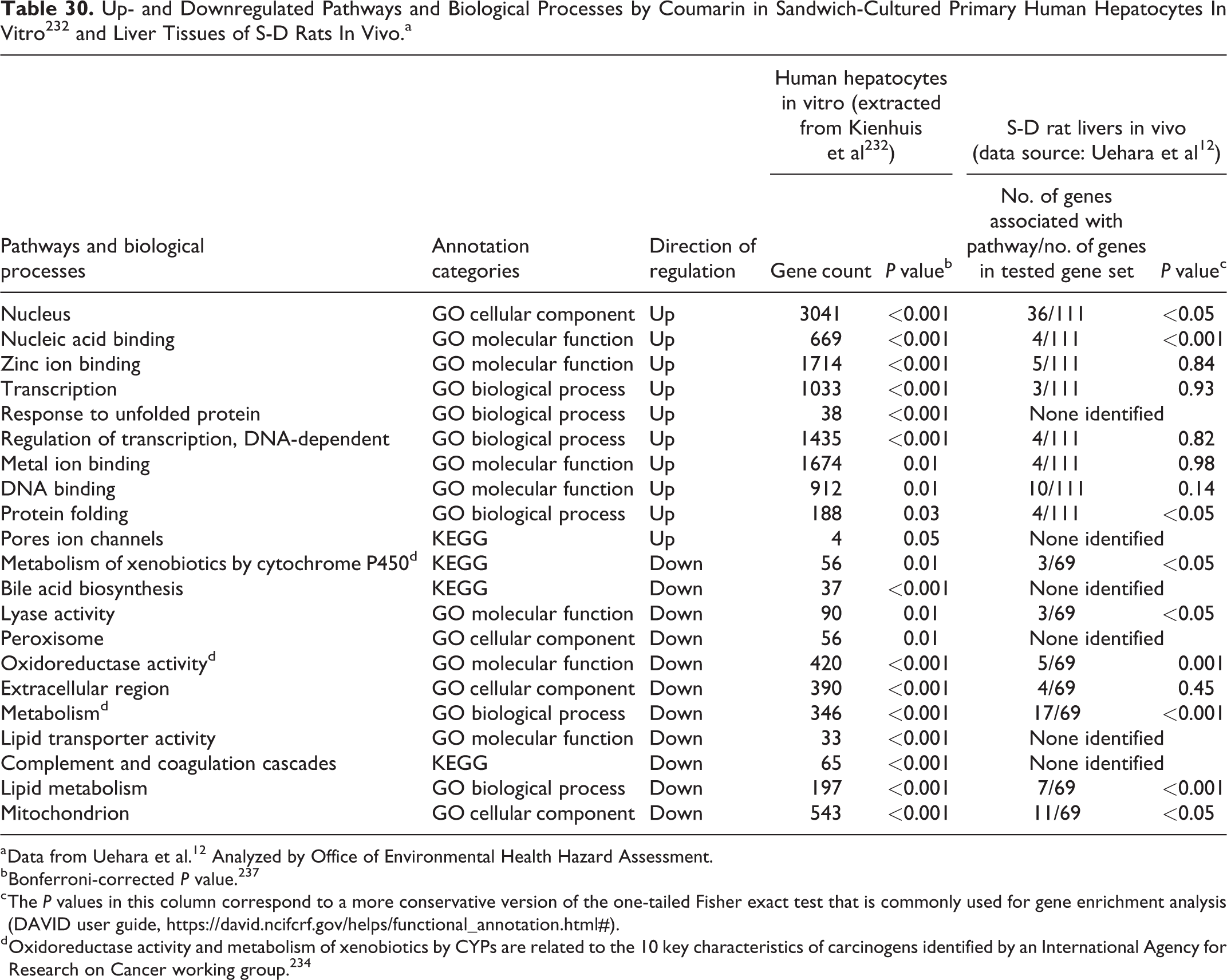
a Data from Uehara et al. 12 Analyzed by Office of Environmental Health Hazard Assessment.
b Bonferroni-corrected P value. 237
c The P values in this column correspond to a more conservative version of the one-tailed Fisher exact test that is commonly used for gene enrichment analysis (DAVID user guide, https://david.ncifcrf.gov/helps/functional_annotation.html#).
d Oxidoreductase activity and metabolism of xenobiotics by CYPs are related to the 10 key characteristics of carcinogens identified by an International Agency for Research on Cancer working group. 234
Comparison of toxicogenomic data from rat liver in vivo and primary human hepatocytes
Among the biological processes or biochemical pathways identified, several pathways identified in human hepatocytes by Kienhuis et al 232 are similar to OEHHA’s analysis of Uehara et al 12 rat data in vivo by DAVID, including oxidoreductase activity, metabolism of xenobiotics by CYPs, nuclear acid binding, and DNA binding (Tables 29 and 30).
Table 30 summarizes the up- and downregulated pathways and biological processes affected by coumarin in the sandwich-cultured primary human hepatocytes in vitro 232 and in liver tissues of S-D rats in vivo (data presented in Uehara et al 12 and analyzed by OEHHA; see Supplementary Tables B3 and B4). Different from Table 29, the microarray data from rat livers in vivo 12 were analyzed by the general gene functional annotation approach, not the clustering approach in DAVID to accommodate the annotation categories defined in Kienhuis et al. 232 Several upregulated cancer-associated pathways and biological processes induced by coumarin are similar in the livers of rats and humans, including nucleic acid binding and protein binding (Table 30). Several downregulated cancer-associated pathways and biological processes induced by coumarin are similar in the livers of rats and humans, including metabolism of xenobiotics by CYPs, oxidoreductase activity, and mitochondrial functions (Table 30). In addition, genes involved in lipid metabolism, including one that reprograms energy metabolism and is identified as a cancer hallmark by Hanahan and Weinberg, 189 were also downregulated by coumarin in both rat and human livers.
Summary of toxicogenomic data
In summary, several toxicogenomic studies and OEHHA’s functional pathways analysis show that multiple biological processes/pathways could be involved in the hepatocarcinogenicity of coumarin, such as GSH metabolism, and the oxidative stress response. In addition, as shown in Table 30, there are several common cancer-related biological processes/pathways altered by coumarin in rat liver and in human primary hepatocytes, including upregulated pathways related to nucleic acid binding and protein binding, and downregulated pathways related to metabolism of xenobiotics by CYPs, oxidoreductase activity, and mitochondrial functions.
ToxCast high-throughput in vitro assays
ToxCast is a chemical prioritization research program developed by the US EPA. 241 It is a multiyear project that launched in 2007. It utilizes various in vitro systems to identify chemical activity in a battery of high-throughput screening (HTS) assays. As of 2017, more than 9,000 chemicals have been tested and there have been more than 700 high-throughput assays that cover a range of high-level cell responses and approximately 300 signaling pathways in the ToxCast database.
This section highlights the ToxCast HTS assays in which coumarin and its metabolite 3,4-DHC were active were reported as active on ToxCast database. The OEHHA has searched the ToxCast database via the Interactive Chemical Safety for Sustainability (iCSS) Dashboard (accessed on May 19, 2017) (note 1) and identified chemical activity data on coumarin and one of its metabolites (3,4-DHC). There are 13 active ToxCast assays for coumarin and 1 active assay for 3,4-DHC (Table 31).
Overview of ToxCast High-Throughput Screening Assay Activity for Coumarin and 3,4-Dihydrocoumarin (3,4-DHC).

Information on each of the active ToxCast HTS assays reported for coumarin and 3,4-DHC is presented in Tables 32 and 33, respectively, with the assays sorted by lowest to highest AC50 (the concentration that induces a half-maximal assay response).
Active ToxCast High-Throughput Screening Assays for Coumarin.
Abbreviation: NA, not applicable.
Active ToxCast High-Throughput Screening Assays for 3,4-Dihydrocoumarin.

Coumarin was active in assays related to several biological processes or intended target families, including cell cycle, nuclear receptor, cytokine and DNA binding in various human cell lines, and oxidoreductase regulation in rat cell-free assays. 3,4-Dihydrocoumarin was active in an assay related to cytotoxicity in a human cell line.
Genes involved in active coumarin ToxCast HTS assays
For each of the active ToxCast assays, we identified the genes, functions, and related pathways associated with the assay, as well as the curated associations of the gene with cancer that have been identified in the CTD (http://ctdbase.org/; accessed on May 26, 2017). This information is summarized below for each identified gene (those with curated evidence are underlined). For coumarin, the following genes have curated associations with cancer, as compared with indirect or inferred associations: CCL2, MAOA, MAOB, and ESR1. The CTD ratios of cancer to all diseases for each of the genes with curated associations were below 10%, ranging from 7.3% to 9.2%.
The gene descriptions provided below were adapted from CTD.
PTGER2 (prostaglandin E receptor 2) encodes a receptor for prostaglandin E2, a metabolite of arachidonic acid, which has different biologic activities in a wide range of tissues. Two pathways involved are neuroactive ligand-receptor interaction and signal transduction. The GO annotations related to this gene include G-protein-coupled receptor activity and prostaglandin E receptor activity.
SAA1 (serum amyloid A1) encodes an apolipoprotein, which is a member of the serum amyloid A family. The encoded preproprotein is proteolytically processed to generate the mature protein. The protein is highly expressed in response to inflammation and tissue injury. High levels of this protein are associated with chronic inflammatory diseases including atherosclerosis and Alzheimer disease. This protein may also be a potential biomarker for certain tumors.
Other Mechanistic Studies
Effects on cell proliferation and cell cycle
Cell proliferation and apoptosis
The effects of coumarin and its metabolites on cell proliferation and apoptosis have been studied in cultured rat hepatocytes and various human cancer cell lines.
Coumarin was reported to increase cell proliferation in cultured rat hepatocytes, increasing the mitotic index 1.4-fold above controls. 202 Other studies, however, have reported that coumarin and its metabolite 7-HC inhibited proliferation of various human cancer cell lines. 244 -247 The inhibition seemed to be concentration dependent and the IC50 for 7-HC tended to be lower than for coumarin.
One study found that coumarin induced apoptosis in human cervical cancer HeLa cells. 248 In this study, coumarin treatment led to cellular morphological changes and signs of cellular apoptosis, including internucleosomal DNA laddering fragmentation and an increase in cells in the sub-G1 phase of the cell cycle. On the molecular level, coumarin decreased the expression of antiapoptotic proteins such as Bcl-2 and Bcl-xl, increased the expression of proapoptotic proteins such as P21, P53, Bax, cytochrome c, and the active forms of caspase-3 and caspase-9, increased intracellular Ca2+ concentration and ROS production, and decreased mitochondrial membrane potential. 248 The coumarin metabolite 7-HC has been shown to induce apoptosis in 2 studies. In one study in human promyelocytic leukemia HL-60 cells, 7-HC induced the appearance of DNA “ladder” patterns, a sign of apoptotic cell death. 244 In the second study in human lung cancer A549 cells, treatment of 7-HC significantly increased caspase-3 activity. 249
Cell cycle regulation
Coumarin and 7-HC have been shown to induce G1 cell cycle arrest (thus blocking the G1/S transition) in human cancer cell lines, such as lung carcinoma cell lines 246,250 and HeLa cells, 248 but not in cultured peripheral blood mononuclear cells. 246 Induction of G1 cell cycle arrest is consistent with the cytostatic effects of coumarin and 7-HC reported in these cell lines. The cell cycle arrest observed in HeLa cells was shown to be accompanied by decreased expression of cyclin D1, cdk2, and Cdc25A, 248 while in human lung carcinoma A-427 cells, cell cycle arrest induced by 7-HC, but not coumarin, was accompanied by decreased expression of cyclin D1. 250
Effects on ROS production and glutathione depletion
Multiple toxicogenomic studies, conducted in both in vivo and in vitro models, have reported that coumarin exposure alters the expression of genes involved in the oxidative stress response and GSH metabolism pathways (see the section describing the toxicogenomic data). Traditional toxicology studies of coumarin and its effects on ROS production and GSH depletion are briefly summarized here.
When human cervical cancer HeLa cells were treated with coumarin at 0, 1, 5, 10, 25, 50, or 100 µM for 24 hours, there were significant increases in ROS at 25 µM or higher doses. 248 In another study, treatment of HeLa cells with the metabolite 6,7-DiHC at 50 µM for 2, 4, or 6 hours induced significant increases in mitochondrial ROS. 251 An increase in ROS is an upstream event that may lead to oxidative stress and DNA damage, as well as caspase activation and apoptosis. 248,251
Coumarin depletes GSH in rat liver in vivo, 110 in freshly isolated rat hepatocytes, 252 and in rat primary hepatocyte cultures. 110 These studies demonstrated that coumarin does not react directly with GSH. Coumarin metabolism by CYP enzymes is required, and GSH depletion results from the formation of metabolite-GSH conjugates, rather than the oxidation of GSH to GSSG (GSH disulfide). Formation of multiple coumarin metabolite-derived GSH conjugates has been reported in other model systems, including in rat olfactory mucosal microsomes, rat liver microsomes, and human liver microsomes. 105
Structure Activity Considerations
Coumarin consists of an aromatic ring fused to a condensed lactone ring. The OEHHA used Chemotyper (https://chemotyper.org/, accessed May 24, 2017), a tool available from Molecular Networks GmbH and Altamira LLC for searching and highlighting chemotypes (chemical structures or subgraphs), to identify chemicals that share structural similarities with coumarin. Eight structurally similar chemicals were chosen for structure activity comparison with coumarin, based on the following criteria: (1) presence of the basic coumarin structure (1,2-benzopyrone), or a metabolite of coumarin; (2) no methoxy groups; (3) no halogen, nitrogen, or sulfur groups; (4) no additional aromatic rings; (5) testing for genotoxicity or animal carcinogenicity. The selected compounds were 3,4-DHC, 3-methylcoumarin, 4-methylcoumarin, 6-methylcoumarin, 6,7-DiHC, 7-HC, 7,8-DiHC, and 4-methyl-7-hydroxycoumarin. Information on the genotoxicity and carcinogenicity of the 8 comparison chemicals is briefly described below and in Table 34. No human cancer epidemiology studies were identified for any of the 8 comparison chemicals.
Structure Activity Comparison Between Coumarin and 8 Structurally Related Chemicals.a
Abbreviations: CA, chromosomal aberrations; CHO, Chinese hamster ovary; 3,4-DHC, 3,4-Dihydrocoumarin; 7-HC, 7-hydroxycoumarin; MN, micronuclei; NT, not tested; SCE, sister chromatid exchange.
a +, positive; (+), weakly positive;
b Observed in low-dose group.
c Male and female rats.
As summarized in Table 34, only 2 of these comparison chemicals have been tested for carcinogenicity in animals. One of these, 3,4-DHC, induced tumors at similar sites to coumarin, including kidney tumors in rats and liver tumors in mice. Five comparison chemicals have been tested for mutagenicity in Salmonella, and only one, 6-methylcoumarin, induced mutations. Similar to coumarin, 6-methylcoumarin was judged to be weakly positive as a mutagen in Salmonella. Seven of the comparison chemicals were tested in chromosomal effects assays, and 6 (3,4-DHC, 3-methylcoumarin, 4-methylcoumarin, 6-methylcoumarin, 7-HC, 4-methyl-7-hydroxycoumarin) were reported to induce CA or SCEs, similar to coumarin. DNA binding was observed with coumarin and 2 of the 3 comparison chemicals tested, namely 7-HC and 7,8-DiHC.
Summary and Conclusions
Tumors in animal cancer bioassays. Coumarin increased the incidence of tumors in multiple studies conducted in rats and mice. These observations include statistically significant increases in rare kidney tumors in male and female rats of one strain, liver hepatocellular tumors and cholangiocarcinomas in male and female rats of another strain, liver hepatocellular tumors in female mice, lung tumors in 2 strains of male mice and one strain of female mice, and forestomach tumors, including rare squamous cell carcinomas, in male mice.
Formation of electrophilic metabolites. Metabolism of coumarin in humans and animals can proceed via 7-hydroxylation, which is generally considered a detoxification pathway, and via 3,4-epoxidation, which generates the electrophilic metabolites CE and the aldehyde o-HPA. Both CE and o-HPA have been shown to bind covalently to microsomal proteins in rats and humans. 81,98,233 In humans, 7-hydroxylation of coumarin is catalyzed primarily by CYP2A6, and populations around the world carry certain allelic variants of CYP2A6 that are associated with either no enzyme function or reduced function, resulting in poor 7-hydroxylator phenotypes. Compromised coumarin 7-hydroxylation by CYP2A6 can lead to increased metabolism through the 3,4-epoxidation pathway and increased generation of the reactive electrophilic metabolites CE and o-HPA. The kinetics of these reactions and subsequent detoxification reactions, including conjugation of CE with GSH (GSH), and metabolism of o-HPA to either o-HPE or o-HPAA, may determine the ultimate toxic effects of these metabolites. A number of clinical trials and case reports indicate that coumarin causes hepatotoxicity in susceptible individuals, which can lead to extensive liver damage with pro-inflammatory and tumor-promoting potential.
Genotoxicity. Coumarin’s genotoxic effects include the induction of base-pair substitution mutations in two strains of Salmonella, chromosome aberrations and SCE in CHO and onion root tip cells; MN in human lymphocytes and a human hepatoma cell line; and chromosome instability (i.e., deletions) in Aspergillus. In addition, coumarin inhibits DNA excision repair in E. coli and binds to single- and double-stranded calf thymus DNA. There is also some evidence for the genotoxicity of two coumarin metabolites. Specifically, 3,4-DHC induced SCEs in CHO cells, and 7-HC was weakly positive in a CA assay in CHO cells, induced expression of a DNA repair gene in E. coli, and formed DNA cycloadducts with photoirradiation and DNA interstrand crosslinks in a cell-free assay. However, in vivo studies of coumarin were not positive for genotoxicity, including SLRL mutations in Drosophila, MN in mice, and UDS and DNA covalent binding in rats.
Induction of oxidative stress. Coumarin depletes cellular GSH by forming GSH conjugates. Depletion of GSH may result in cellular redox imbalance and impaired capacity to detoxify additional reactive species, and ultimately lead to oxidative stress. Indeed, significant increases in cellular and mitochondrial ROS have been measured with coumarin and its metabolite 6,7-DiHC, respectively, in HeLa cells. In addition, in vivo and in vitro toxicogenomic studies have reported that coumarin alters the expression of genes involved in the oxidative stress response and GSH metabolism pathways.
Cancer-related toxicogenomic pathways. As already noted, altered expression of genes in the oxidative stress response and GSH metabolism pathways has been observed in multiple studies of coumarin. In addition, functional pathways analysis and comparison of toxicogenomic data from rat liver following in vivo exposure and human primary hepatocytes exposed in vitro identified several cancer-related pathways and biological processes that were similarly affected by coumarin. These include up-regulated pathways related to nucleic acid binding and protein binding, and down-regulated pathways related to metabolism of xenobiotics by CYP enzymes, oxidoreductase activity, and mitochondrial function.
Other possible effects. It is unclear what effects coumarin has on cell proliferation and cell death. While coumarin inhibits cell proliferation and induces apoptosis in some human cancer cell lines, it was observed to increase cell proliferation (as measured by an increase in the mitotic index) in cultured rat hepatocytes. A toxicogenomic study found that coumarin up-regulated expression of seven cell-cycle related genes in rat liver in vivo.
Structure activity considerations. The biological activity of coumarin was compared to eight structurally related compounds. Two of the comparison chemicals were tested in animal cancer bioassays, and one (the coumarin metabolite 3,4-DHC) induced tumors at similar sites to coumarin, including kidney tumors in male rats and liver tumors in female mice. Five of the comparison chemicals were tested for mutagenicity in Salmonella, and one (6-methylcoumarin) tested positive. Seven comparison chemicals were tested for chromosomal effects and six (3,4-DHC, 3-methylcoumarin, 4-methylcoumarin, 6-methylcoumarin, 7-HC, and 4-methyl-7-hydroxycoumarin) tested positive. Three comparison chemicals were tested for DNA damage/binding, and two (7-HC and 7,8-DiHC) tested positive.
In summary, a review of the available evidence suggests that coumarin may pose a cancer hazard to humans. When tested in long-term animal carcinogenicity studies, coumarin increased the incidence of tumors in multiple studies in male and female rats and mice. Several of the mechanistic findings for coumarin are associated with three of the key characteristics of carcinogens described by Smith et al 234 namely coumarin can form electrophilic metabolites, it is genotoxic, and it can induce oxidative stress. Considerations of metabolism, human variability in CYP2A6 activity, and reports of coumarin hepatotoxicity in susceptible individuals provide additional support for carcinogenicity concern. Our analysis illustrates the importance of integrating information on human variability in the cancer hazard identification process.
Supplemental Material
Supplemental Material, Coumarin_IJT_supplementary - Cancer Hazard Identification Integrating Human Variability: The Case of Coumarin
Supplemental Material, Coumarin_IJT_supplementary for Cancer Hazard Identification Integrating Human Variability: The Case of Coumarin by ChingYi Jennifer Hsieh, Meng Sun, Gwendolyn Osborne, Karin Ricker, Feng C. Tsai, Kate Li, Rajpal Tomar, Jimmy Phuong, Rose Schmitz and Martha S. Sandy in International Journal of Toxicology
Footnotes
Authors’ Note
The views expressed are those of the authors and do not necessarily represent those of the Office of Environmental Health Hazard Assessment (OEHHA), the California Environmental Protection Agency, or the State of California.
Acknowledgments
The authors thank Drs Lauren Zeise, Shoba Iyer, and Lori Lim at OEHHA for their thoughtful review and helpful discussion and input on this work.
Author Contributions
ChingYi Jennifer Hsieh and Meng Sun contributed equally to this work. ChingYi Jennifer Hsieh and Meng Sun contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. Gwendolyn Osborne contributed to conception, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. Karin Ricker contributed to design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. Feng C. Tsai and Kate Li contributed to design, contributed to analysis, drafted the manuscript, and critically revised the manuscript. Rajpal Tomar contributed to design, contributed to analysis, and drafted the manuscript. Jimmy Phuong contributed to conception and design and contributed to analysis. Rose Schmitz contributed to analysis. Martha S. Sandy contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Note
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.