Abstract
Ocular toxicity studies are the bedrock of nonclinical ocular drug and drug–device development, and there has been an evolution in experience, technologies, and challenges to address that ensures safe clinical trials and marketing authorization. The expectations of a well-designed ocular toxicity study and the generation of a coherent, integrative ocular toxicology report and subreports are high, and this article provides a pathology/toxicology consultant’s perspective on achieving that goal. The first objective is to cover selected aspects of study designs for ocular toxicity studies including considerations for contract research organization selection, minipig species selection, unilateral versus bilateral dosing, and in-life parameters based on fit-for-purpose study objectives. The main objective is a focus on a high-quality ocular pathology report that includes ocular histology procedures to meet regulatory expectations and a report narrative and tables that correlate microscopic findings with key ophthalmic findings and presents a clear interpretation of test article-, vehicle-, and procedure-related ocular and extraocular findings with identification of adversity and a pathology peer review. The last objective covers considerations for a high-quality ophthalmology report, which in concert with a high-quality pathology report, will pave the way for a best quality toxicology report for an ocular toxicity study.
Keywords
Introduction: Recently Approved Ocular Drugs and Ocular Toxicity Studies
The ocular drug landscape for small molecules, biologics and gene and cell therapies and novel drug delivery platforms continues to expand at a rapid trajectory. Results from a 2015 internet survey identified 190 companies worldwide developing 436 ocular drug products, of which 78% were pharmaceuticals and 22% were devices, 1 and the number of companies and ocular drug products has likely increased since then. With pharmaceuticals, they identified 46 separate indications with age-related macular degeneration (AMD), glaucoma, and dry eye as most common; anti-vascular endothelial growth factor, hormone therapy, and anti-inflammatory products were also common classes. With devices, there were 30 indications with glaucoma, AMD, and dry eye as most common; drug delivery, ocular implants, and prostheses were fewer classes. Results from a subanalysis identified 63 ophthalmic pharmaceutical start-up or mid-sized (<200 employees) companies, of which, after 5 years of follow-up, there were 3 categories: unsuccessful (19%), unchanged (43%), and successful (38%), and this success rate compares favorably with other therapeutic areas and their start-up success rate. 2
In ophthalmology, the number of approved medicines has decreased from an average of 4 per year in 1995 to 2000 to 2 per year in 2009 to 2019. 3 Between 2014 and May 2020, 33 drugs for ophthalmic use have been approved as an original new drug application (NDA) by the US Food and Drug Administration (FDA) and 7 NDA supplements have been approved for new drug indications (Table 1), which is an update of a review of FDA-approved drugs for ophthalmic use from 2001 to 2014. 4 Of 33 ophthalmic drugs approved by FDA since 2014, 20 (61%) were reformulations of previously approved ocular drugs with a new formulation, new dosage form, or new combination, 9 (27%) were new molecular entities (NME), and 5 (15%) were small molecule topical ocular solutions that were approved for a new manufacturer or a drug already marketed without an approved NDA. All 20 reformulations were small molecules and most (15) were topical ocular drugs, but 4 were steroidal drugs that included 2 intravitreal implants (Iluvien, Yutiq), 1 intravitreal suspension (Dexycu Kit), and 1 intracanalicular insert (Dextenza), and the remaining drug reformulation was a recently approved intracameral implant of bimatoprost (Durysta) for sustained release in reducing intraocular pressure (IOP). Six of 9 NME drugs were small molecules: 3 were topical ocular ophthalmic solutions for dry eye disease (Xiidra) or reduction of IOP (Vyzulta, Rhopressa), 1 was an intravitreal solution for selective staining of the internal limiting membrane for vitrectomy surgery (TissueBlue), and 2 were oral dugs, one for non-24-hour sleep–wake disorder in the totally blind (Hetlioz) and one for onchocerciasis (Moxidectin). Two of 9 NME drugs were biologics, one was intravitreal drug Beovu, a low-molecular-weight anti-VEGF single-chain Fv for AMD, which shows the difficultly for demonstrating advantages over other anti-VEGF biologic therapies Eylea and Lucentis that were approved for the same indication in 2011 and 2012, respectively. The other was recently approved Tepezza, an insulin-like growth factor 1 receptor inhibitor administered intravenously (IV) for thyroid eye disease. The remaining NME, Luxturna, was approved to treat biallelic retinal pigment epithelium-specific 65 kDa (RPE65) mutation-associated retinal dystrophy for retinal blindness, which was the first directly administered gene therapy approved in the United States that targets a disease caused by mutations in a specific gene.
FDA-Approved Drugs for Ophthalmology (2014-2020).a
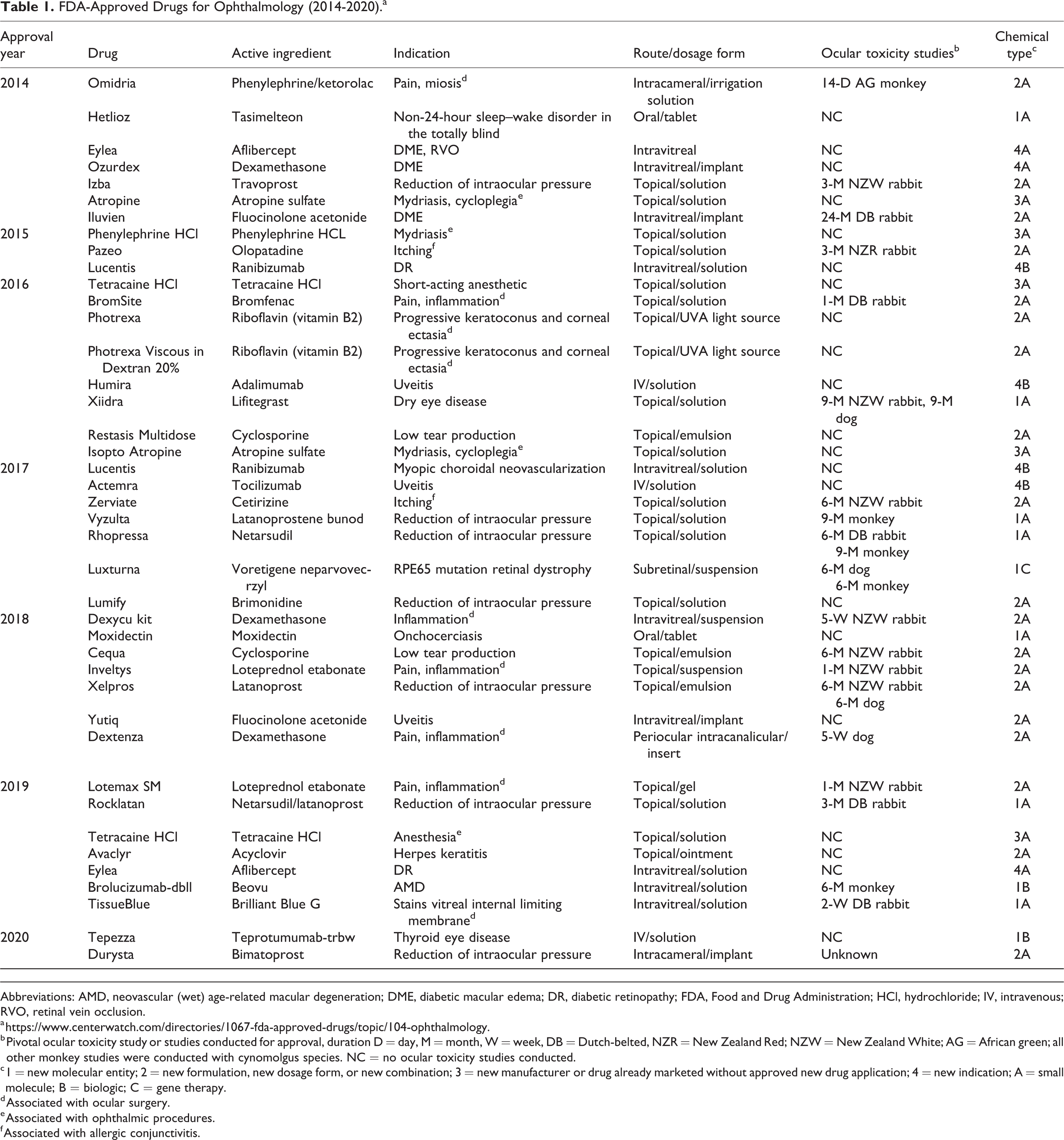
Abbreviations: AMD, neovascular (wet) age-related macular degeneration; DME, diabetic macular edema; DR, diabetic retinopathy; FDA, Food and Drug Administration; HCl, hydrochloride; IV, intravenous; RVO, retinal vein occlusion.
a https://www.centerwatch.com/directories/1067-fda-approved-drugs/topic/104-ophthalmology.
b Pivotal ocular toxicity study or studies conducted for approval, duration D = day, M = month, W = week, DB = Dutch-belted, NZR = New Zealand Red; NZW = New Zealand White; AG = African green; all other monkey studies were conducted with cynomolgus species. NC = no ocular toxicity studies conducted.
c 1 = new molecular entity; 2 = new formulation, new dosage form, or new combination; 3 = new manufacturer or drug already marketed without approved new drug application; 4 = new indication; A = small molecule; B = biologic; C = gene therapy.
d Associated with ocular surgery.
e Associated with ophthalmic procedures.
f Associated with allergic conjunctivitis.
Ocular toxicity studies are the bedrock of nonclinical ocular drug and drug–device development as they are conducted for drugs administered by the clinical ocular route for all NMEs and almost for all reformulations. The longest duration of ocular toxicity studies in various species that were conducted to support these recently approved ocular drugs and drug–devices is given in Table 1. This list does not include ocular studies conducted earlier in drug development or any other nonclinical study types conducted. Ocular toxicity studies were not conducted for ocularly administered drugs approved with a new manufacturer or indication or systemically administered drugs approved for an ophthalmic indication. However, for ocularly administered biosimilar drugs approved with a new manufacturer that are currently in development, there are potential formulation issues, including endotoxins, or clinical differences in immunogenicity, 5 –7 and an ocular toxicity study that compares the biosimilar with that from the approved manufacturer will most likely be expected for NDA approval.
There are many ocular delivery routes and formulation technologies used in ocular toxicity studies. 4,8 –14 Topical ocular instillation (eye drops) is the most common route for anterior segment ocular diseases, and there are many new drug delivery systems based on in situ forming gels, nanoparticles, and combinations of both as well as other formulation approaches. 15,16 Ocular toxicity issues are not as common with eye drops as with other routes of administration for anterior segment diseases. More novel ocular routes and/or sustained release technologies for anterior segment diseases with robust pharmacokinetic/pharmacodynamic studies in rabbits, dogs, and/or monkeys include bimatoprost, difluprednate, and other intracameral implants 15,17 –19 ; dexamethasone intracanalicular insert 19–20 ; and periocular and subconjunctival sustained release formulations. 15,21 Unfortunately, published literature on ocular toxicity studies for these novel ocular anterior segment drug products including rationale for species selection, study design, findings, and challenges encountered are lacking. Insight to ocular toxicity studies usually relies on FDA reviews of approved ocular drug products.
Safety evaluation of intraocular routes of administration, especially those with sustained release technologies, is especially challenging. Several articles highlight these challenges and/or important factors to consider for successful development. 8,10 –12,14–15,22 This is not a trivial matter, as it has been pointed out by Thackaberry et al 12 that marketing approval of sustained delivery of nonsteroidal molecules in an injectable formulation (small molecule or protein based) has not been achieved, with the exception of a few long-acting VEGF therapies, including recently approved brolucizumab 23–24 and the ranibizumab port delivery system (PDS) implant, a drug–device combination in late clinical development. 25 For the purposes of this review, test article refers to drugs and drug–devices. Discussion of ocular medical devices for anterior or posterior segment diseases is beyond the scope of this article; however, many principles and recommendations presented in this publication apply to ocular medical devices, which have been recently reviewed. 26
Nonclinical development of ocular drugs, including pharmacology, pharmacokinetic, and toxicity studies, to support clinical trials based on International Conference on Harmonization and FDA guidance, species selection, study design considerations, and ocular safety margins have been previously addressed 4,8,11,27 –29 and will not be reviewed here. From a toxicology and pathology consultant’s perspective, selected aspects of study design of ocular toxicity studies and the quality of ocular toxicity reports and contributing scientist reports, including correlations between ophthalmology and pathology findings and establishing adversity, have room for improvement. The major goal of this article is to cover aspects of sound, fit-for-purpose (FFP) nonpivotal (dose range) and pivotal (Good Laboratory Practice [GLP]) ocular toxicity studies from the perspective of the sponsor’s toxicologist and/or sponsor’s peer review pathologist to deliver high-quality contributing pathology and ophthalmology reports that will ensure a high-quality toxicology report. The FFP concept for ocular toxicity studies is that the level of evaluation should be appropriate for the intended objective of the study and not “one size fits all.” The first objective of this article is to discuss a few select aspects of study designs and in-life parameters, including minipig species selection considerations, deciding on unilateral or bilateral dosing, and ocular and systemic in-life parameters. The second objective is to cover pathology parameters and procedures for ocular toxicity studies, including eye fixation, trimming, and sectioning procedures, since adequate evaluation of all major ocular structures and extraocular tissues is a regulatory expectation. The third objective is to provide an overview of a high-quality pathology reports for ocular toxicity studies. This objective covers pathology considerations for selecting a contract research organization (CRO), study design, study-related data review, microscopic examination, pathology report narrative including determining and establishing adversity and correlating microscopic and ophthalmic findings, and pathology peer review. The fourth objective is to provide a pathologist’s and toxicologist’s perspective on high-quality ophthalmology report, which in concert with a high-quality pathology report, will pave the way for a high-quality toxicology report. Roles and responsibilities of the study pathologist and sponsor’s peer review pathologist that employ a number of recommended approaches and best practice principles including sound communication with the study ophthalmologist, toxicologist, and other members of the study team to achieve these objectives will also be discussed.
Ocular Toxicity Studies: Selecting a CRO, Study Designs, and Parameters
The first objective of this review is to discuss the types of ocular toxicity studies, selecting a CRO, and discuss a few select aspects of ocular toxicity designs and in-life parameters. Pathology-related aspects of selecting a CRO are discussed later in this article under pathology reports. Ocular tolerability studies are generally nonterminal studies with in-life ocular evaluation and no pathology end points. Ocular tolerability studies are usually adequate FFP for topical (eye drop) drug products since in-life ocular evaluation is usually adequate to determine the ocular toxicity profile and design pivotal topical ocular toxicity studies. In many cases for topical ocular drug products, an ocular tolerability study in one species (rabbits) is adequate for understanding the ocular toxicity profile before conducting pivotal topical ocular toxicity studies in 2 species, if needed. On the other hand, dose range toxicity studies are terminal studies to include pathology evaluation, which is critical for most other ocular routes, including periocular, intracameral, and intravitreal administration since in-life ocular evaluation may not be sufficient for full characterization and correlation between ophthalmologic and pathological end points which is important to establish before designing pivotal ocular toxicity studies by these routes.
Many pharmaceutical companies, especially small- or medium-sized ones, are not equipped or staffed to conduct ocular tolerability or dose range toxicity studies and almost all do not currently conduct pivotal ocular toxicity studies. The number and type of ocular studies a test facility CRO has conducted in the past 5 years, broken down by species, route, duration, and GLP status, should be shared with the sponsor to gauge if the CRO has the experience and expertise to conduct non-GLP and/or GLP ocular toxicity studies by the route and species selected by the sponsor to support the clinical plan. Intraocular studies, especially those with sustained release technologies, may require specialized ocular examination techniques and expertise discussed below that are not available at some CRO test facilities. Ideally, before a request for proposal (RFP) for ocular toxicity studies is generated by the sponsor and submitted to one or several CROs that will present quotes contained within a scope of work (SOW), it is best for the sponsor to put together FFP protocol outlines. Protocol outlines to support the clinical plan should include as much detail as possible including objective of the study, species, type of molecule (small molecule or biologic), NME or reformulation, route, frequency, and duration of administration, unilateral or bilateral dosing, number of animals/group, and termination time point(s) and parameters and their frequency of evaluation, including any nonstandard ocular evaluation techniques. Requests for board-certified ophthalmologists and pathology peer review (see below) by an in-house or sponsor-provided pathologist for most dose range ocular toxicity studies and all pivotal ocular toxicity studies are highly recommended and need to be specified in protocol outlines. As with any single-dose or repeat dose toxicity study, protocol outline details should indicate if the test facility or sponsor is responsible for drug dose preparation, drug formulation analysis, drug bioanalysis, toxicokinetics, anti-drug antibody (ADA) analysis, and if a common technical document summary table is requested. This approach results in an accurate quote that will not need SOW amendments during protocol development and an “apples to apples” and not “apples to oranges” comparison between CRO quotes since there can be variability in standard practices for ocular toxicity studies between CRO test facilities.
Selected aspects in this review that have not been covered in previous publications include minipig species selection considerations for ocular toxicity studies, comparison of advantages of unilateral versus bilateral dosing, and FFP considerations in ocular and systemic in-life parameters for tolerability/dose range and pivotal ocular toxicity studies.
Minipig Species Selection Considerations
Species selection considerations of albino and pigmented rabbits, beagle dogs, and cynomolgus monkeys for designing ocular toxicity studies have been well covered 4,8,10,13,28–29 and are beyond the scope of this article. There is increasing interest in the use of minipigs for ocular toxicity studies because they have advantages of a favorable replacement, refinement, and reduction of animal testing (3Rs) profile, their eyes lack a tapetum, they have holangiotic retinal vasculature unlike the rabbit, they have a cone to rod ratio similar to that of humans and a large vitreal volume, 14,30,31 and they have other ocular features comparable to humans as recently reviewed by Shrader and Greentree. 32 Gottingen, Yucatan, Hanford, and Sinclair minipigs have been used in research settings, and the smallest of these minipig models is the Gottingen, which has rapidly gained popularity among researchers for ocular studies, including those investigating ocular diseases, novel therapeutics, surgical techniques, and implantable materials/devices. 32 Other strains have successfully been used for ocular toxicity studies, which may be CRO or institutional preference. The author is not aware of advantages or disadvantages of particular strains other than the size advantage and greater amount of literature available on Göttingen minpigs. Göttingen and Yucatan minipigs have primarily been used for intravitreal and subretinal toxicity studies of small molecules, biologics, and gene and cell therapies. The FDA-approved intravitreal drug ocriplasmin 33 and ranibizumab PDS implant in late clinical development 25 used minipigs for ocular toxicity studies. A minipig surgical model for vitreous hemorrhage with ranibizumab PDS implant was useful for investigation of the insertion of long-acting, indwelling devices into the vitreous cavity and provided important information that may be relevant to similar surgical approaches in different settings. 34 As discussed further in the section on pathology reports, intravitreal injection of some saline solutions in minipigs can cause retinal and other ocular changes that can be difficult to discern from test article-related findings. 35 Minipigs have not been used for intracameral toxicity studies to the author’s knowledge, and they have a smaller anterior chamber angle opening distance and angle recess area, which are disadvantages to the more similar angle size in dogs and humans. 36 The minipig was shown to be a promising model for endothelial cell evaluation during the development of ophthalmic viscosurgical devices since minipigs, like humans, do not have the ability to regenerate the corneal endothelium post injury, unlike rabbits. 37 However, minipigs were not suitable for corneal transplantation studies because of intraoperative challenges and development of retrocorneal membrane postoperatively. 38 Minipigs should be considered for ocular toxicity studies by periocular routes of injection, including topical dermal eyelid administration. Hanford minipigs were successfully used for ocular toxicity studies by the topical dermal eyelid route in the author’s experience. Minipig is the closest and most relevant model for human eyelid skin based on similarities in cutaneous anatomy, biochemistry, and physiology, 39 and topical dermal eyelid administration is not practical in rats, rabbits, or dogs due to increased follicular density of eyelids, smaller eyelid area, and thinner eyelid skin thickness compared to humans. Minipigs were used to demonstrate the protective effect of 7 days of twice-daily topical ocular (eye drop) administration of a kinase inhibitor on retinal pigment epithelium against sodium iodate retinal injury 40 ; however, this species is not practical for topical ocular (eye drop) toxicity studies due to excessive blinking and blepharospasm following topical ocular dose administration, which complicates accurate evaluation of ocular observations by technical personnel.
With the growing popularity of minipigs in ocular research and drug development, there are numerous recent minipig publications useful for pathologists and toxicologists involved in ocular toxicity studies. Recent publications with relevance to ocular toxicity studies in minipigs, primarily Göttingen minipigs, include basic ocular histomorphometry, 41 pre- and postnatal development of the eye, 42 ocular immunohistochemistry and in situ hybridization biomarkers, 43 electroretinography (ERG), 44 and optical coherence tomography (OCT). 43,45 Immunology 46 and ocular immunogenicity testing 47 of minipigs has been characterized, and although the minipig may be suitable for biologics, this species can mount a substantial immunogenic response to human proteins. 13 Vitreous volume in the Gottingen minipig eye using a frozen eye collection technique by Struble et al 48 was approximately 2.3 mL, which is important for calculating a human equivalent dose (HED) and corresponding ocular dose safety margins for intravitreal drugs in this species. Minipig vitreal volume of 2.3 mL is the best estimate to use since it is more similar to other estimates compared to the outlier of 3.0 mL used in other publications. 14,49 For comparison in vitreal HED calculations from intravitreal toxicity studies, vitreal volume in minipigs is comparable to beagle dogs (2.2 mL) and larger than cynomolgus monkeys (2.0 mL) and albino and pigmented rabbits (1.3-1.4 mL) based on the frozen eye collection technique. 48
Unilateral Versus Bilateral Dosing
Whether to administer an ocular drug product unilaterally or bilaterally in ocular toxicity studies is one of the most important aspects of study design, especially from a pathologist’s and ophthalmologist’s perspective. Both unilateral and bilateral ocular dosing are acceptable in ocular toxicity studies depending on the objectives, but from a pathology/toxicology consultant’s perspective, unilateral dosing is preferred to bilateral dosing in many cases depending on the route of ocular administration and main objectives of the study. Unilateral dosing allows a valuable comparison between eyes treated with test or vehicle control article and untreated eyes and is generally acceptable to clinicians and regulatory agencies for bilateral ocular indications. The main advantage of unilateral dosing is that it allows clearer determination if ocular findings are test article-related, vehicle/placebo (formulation)-related, or procedure-related or if ocular findings represent spontaneous (incidental) background changes or are artifacts of fixation, trimming, and/or sectioning. Unilateral dosing with an untreated or sham-treated (injected) contralateral eye in all groups allows a clearer understanding of all potential sources of ophthalmologic and ocular microscopic findings. Ophthalmologist’s and pathologist’s interpretations of the causes of various findings in an ocular toxicity study are usually the main drivers of the no-observed-adverse-effect-level (NOAEL), which impacts clinical trial dose selection and may determine success or failure of an ocular drug product. Many procedure-related effects in ocular toxicity studies do not translate to the clinic due to anatomical considerations or greater experience in clinical ophthalmologic procedures, including intraocular injection. 13 Understanding complex ocular formulations, especially sustained delivery systems or formulations containing novel excipients or excipients used at higher concentrations than approved by the route of administration, and differentiating these findings from procedure-related findings or background spontaneous findings, or ocular histological artifacts with the help of an untreated contralateral eye are as important as differentiating drug (test article)-related effects from vehicle-related effects in treated eyes. In the author’s experience, high-quality ocular sections in ocular toxicity studies are difficult to achieve. Many ocular histology artifacts (folded cornea, lens shatter, retinal detachment, inconsistent dorsal ventral globe orientation on slides) can be present in eye slides from ocular toxicity studies and are more easily identified as such in untreated compared to treated eyes. In summary, correct interpretation of the cause(s) and contributing factors to ophthalmologic and ocular microscopic findings is easier, with fewer potential interpretive errors unilateral compared to bilateral ocular dosing designs in ocular toxicity studies, resulting in greater certainty and confidence in interpretation of pathology and ophthalmology reports. A theoretical concern for unilateral dosing in ocular toxicity studies is that toxicological effects may occur since pharmacological effects and/or ocular absorption in the untreated contralateral eye have been observed following ipsilateral ocular administration in animals and humans 50 –54 ; therefore, the “untreated” eye is not truly an untreated control in ocular toxicity studies. Since ocular exposure to the contralateral eye through systemic absorption or other potential mechanisms in nonrodents is generally a small fraction (<-0.01% - 5%) to that observed in the treated eye, the potential for this effect to confound interpretation is considered low and can be reconciled with untreated control eye of vehicle-treated group.
Some reviews on ocular toxicity study design advocate bilateral dosing and CROs commonly design ocular toxicity studies with bilateral dosing with the aim of “the more eyes, the better,” but a justification for bilateral dosing in ocular toxicity studies other than the potential for increased systemic exposure above unilateral dosing has not been presented. 28 Increased number of eyes injected with bilateral dosing compared to unilateral dosing has a theoretical advantage based on increased number of eyes evaluated; however, in the author’s experience, differences in ocular effects from ocular administration are usually greater between animals (inter-animal variability) than between eyes in the same animal (intra-animal variability) within a dose group. Therefore, the potential advantage of bilateral dosing to increase the number of treated eyes does not always outweigh the advantages of unilateral dosing.
Bilateral dosing has the advantage of potentially doubling systemic exposure compared to unilateral dosing. Repeat dose ocular toxicity studies of some NME, especially biologics, have used bilateral dosing and cited its justification as increasing systemic exposure in an effort to characterize any potential systemic risk of on- or off-target organ toxicity. 55 Bilateral intravitreal dosing of NME biologics or gene or cell therapies with simple solution formulations that contain no novel excipients and/or excipient concentrations approved by the same route of ocular administration may be justified in ocular toxicity studies especially if ocular toxicity testing is limited to one species (nonhuman primates) and no systemic toxicity studies are conducted in other species. However, even simple solution formulation vehicles can sometimes cause interpretive issues for vehicle-related ocular microscopic findings in some species as discussed in this article. Finally, based on species differences in size, systemic dose and exposure margins in ocular toxicity studies are generally adequate to support systemic safety for clinical trials of NME. Bilateral dosing does not double the ocular safety margin compared to unilateral dosing. For ocular drug reformulations that have been approved by ocular or systemic routes, bilateral dosing in ocular toxicity studies offers no advantage over unilateral dosing since comparative pharmacokinetics in animals and/or humans usually demonstrates lower systemic exposure from the ocular route compared to the approved nonocular or ocular route with established clinical systemic safety.
In general, even for ocular drug programs that choose bilateral dosing for pivotal ocular toxicity studies, unilateral dosing should be considered for dose range ocular toxicity studies. The major objective of ocular tolerability and dose range ocular toxicity studies is to understand local tolerance and toxicity and not systemic effects. In the author’s experience, systemic effects do not influence ocular dose selection for pivotal ocular toxicity studies. Toxicokinetics following unilateral dosing and/or ocular pharmacokinetic studies that are usually conducted with bilateral dosing with 2 to 3 animals/time point (N = 4-6 eyes/time point) are sufficient to understand the systemic and ocular pharmacokinetic profile to predict if systemic exposure in pivotal ocular toxicity studies will be negligible and to determine the duration of ocular exposure, which helps in designing pivotal ocular toxicity studies.
Ocular In-Life Parameters
Characterization of standard or specialized ocular examination techniques used for in-life evaluation in ocular tolerability/dose range and pivotal ocular toxicity studies including basic principles, performance parameters, and use in ocular and other toxicity studies have been reviewed in greater detail elsewhere, 4,10,56 –60 and only a few practical points or recent developments will be covered in this article. Recent reviews of species anatomical differences in anterior segment that affect anterior chamber scoring systems, 61 –63 which is a revision to the ocular scoring systems of McDonald-Shadduck and Hackett-McDonald, have greatly contributed to our understanding and standardization of ocular evaluation by ophthalmologists in ocular toxicity studies. A book that provides standards and harmonization for procedures, terminology, and scoring schemes for ocular toxicity studies which was endorsed by American College of Veterinary Pathology (ACVO) has been recently published. 64
A brief, noninclusive list of standard and specialized ocular examination techniques (parameters) for topical (eye drop), periocular (eyelid, subconjunctival, sub-Tenon), intracameral, and intravitreal or subretinal toxicity studies is presented in Table 2. This list is also divided between nonpivotal and pivotal (GLP) ocular toxicity studies since specialized examination techniques should be FFP and case-by-case and not “checkbox” since specialized techniques are generally route-specific and depend on the type of ocular toxicity study and its objectives. There may be scientific advantages to inclusion of some parameters in dose range and/or pivotal ocular toxicity studies which needs to be balanced by value-added cost considerations as well as limitations in capabilities of some CROs. Although ocular observations are generally conducted at frequent intervals, most other standard and specialized ocular in-life parameters are best evaluated at intervals that take into consideration the in vitro and/or ocular pharmacokinetic profile for drug release and biodegradation profile for sustained release technologies 10 although more frequent intervals depend upon the objective of the study.
Ocular In-Life Parameters in Ocular Toxicity Studies.
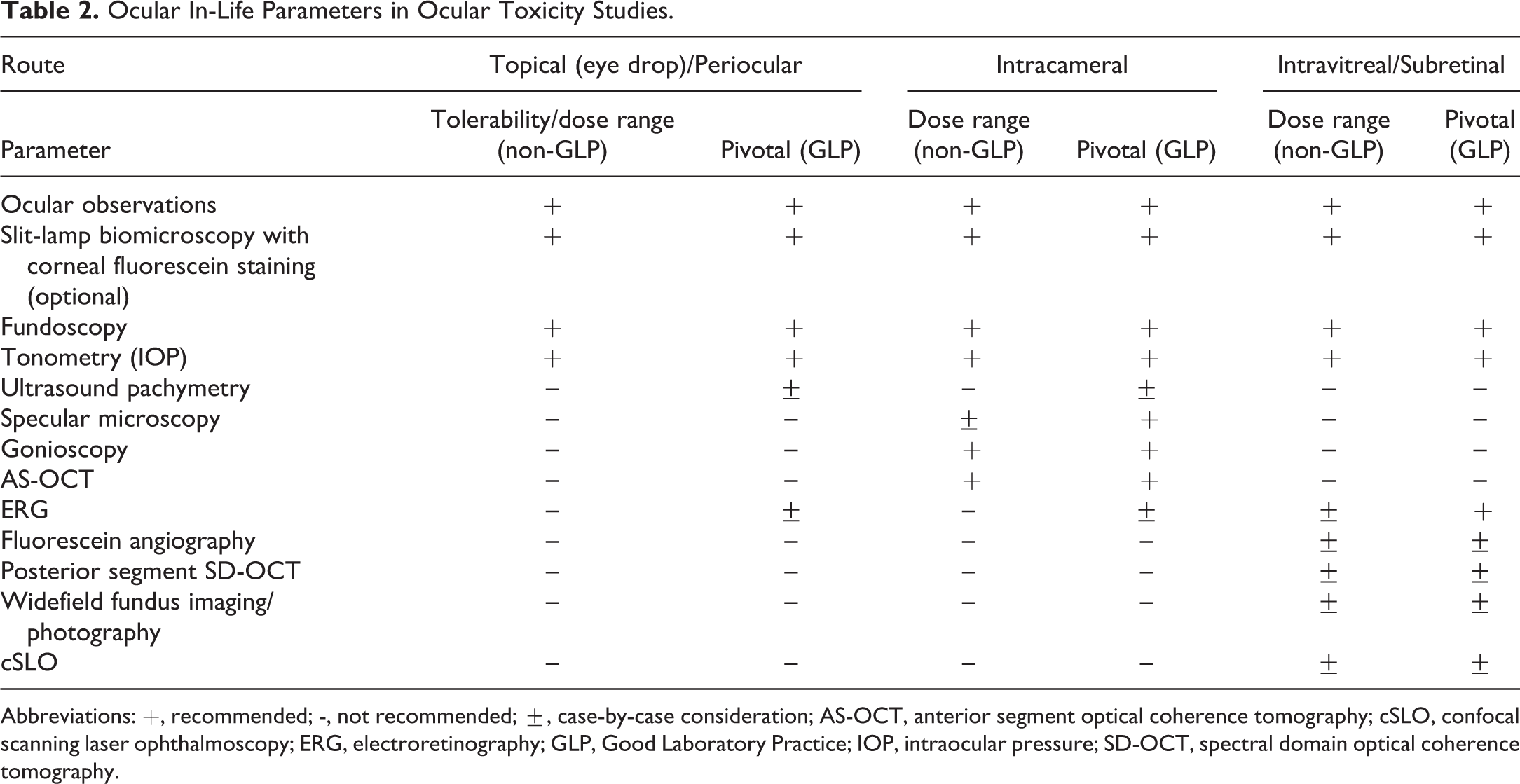
Abbreviations: +, recommended; -, not recommended; ±, case-by-case consideration; AS-OCT, anterior segment optical coherence tomography; cSLO, confocal scanning laser ophthalmoscopy; ERG, electroretinography; GLP, Good Laboratory Practice; IOP, intraocular pressure; SD-OCT, spectral domain optical coherence tomography.
Standard ocular examination techniques for tolerability/dose range and pivotal ocular toxicity studies include ocular gross observations, slit-lamp biomicroscopy, fundoscopy (direct or indirect), and tonometry. Ultrasound pachymetry is sometimes included for pivotal topical and intracameral ocular studies since a change in corneal thickness may occur by these routes but is seldom included in nonpivotal studies and intravitreal toxicity studies. Specular microscopy is important for intracameral toxicity studies to evaluate corneal endothelium in pivotal ocular toxicity studies and is optional for understanding potential corneal endothelial effects in dose range intracameral toxicity studies since sequelae of these effects that impact dose selection for pivotal intracameral toxicity studies can be observed by ophthalmology (corneal edema) and/or microscopic examination (corneal endothelial attenuation). Gonioscopy is invaluable to follow biodegradation of intracameral sustained release technologies since they migrate to the inferior anterior chamber angle, which is important to understand in dose range and pivotal ocular intracameral toxicity studies. Gonioscopy provides limited quantitative information to compare the iridocorneal anatomy across different species, so for intracameral sustained release technologies, it is important to consider the comparative size of the anterior chamber angle and corresponding angle opening distance and angle recess area across species. 14 Anterior chamber angle dimensions are larger in dogs and more like humans compared to rabbits, minipigs and monkeys, although there is much variability among humans. Anterior segment OCT for cross-sectional evaluation of the anterior eye segment is the gold standard over gonioscopy for quantitative measurements of the iridocorneal angle in humans. 65 Anterior segment OCT was used in a intracameral pharmacokinetic study with the sustained release technology, Bimatoprost SR, in dogs. 18 Dogs were grouped according to their iridocorneal angle size; those with larger iridocorneal angles were included in the treatment group to minimize the possibility of implant contact with the corneal endothelium. Therefore, this technology can be useful to justify dogs as the species of choice for intracameral toxicity studies with sustained release technologies since they have the most similar iridocorneal angle measurement compared to humans. 14,65
Electroretinography is considered a standard parameter to evaluate retinal function for all pivotal ocular toxicity studies, 27 and full-field or “flash” ERG is most commonly used although it is limited to detecting functional deficits in a relatively large area of the retina. 58,66 Multifocal ERG is useful for detecting localized retinal changes, but it is not currently offered by many CROs as a standardized procedure, and therefore, the relatively more insensitive full-field ERG used for ocular toxicity studies is often not as sensitive as ophthalmic and/or ocular microscopic examination for local retinal effects. 8,67 Certainly, full-field ERG is a regulatory expectation in most pivotal ocular toxicity studies, but based on its lack of sensitivity, it should be used judiciously in dose range intravitreal/subretinal toxicity studies and some pivotal ocular toxicity studies by other routes as a case-by-case consideration. Inclusion of ERG in dose range intravitreal/subretinal toxicity studies should be considered for certain drug classes that have known ERG effects, such as aminoglycosides. 67 Electroretinography has the advantage that it is a noninvasive method and can provide evidence that a drug is affecting retina function prior to changes in retinal structure, 58 but it is generally only used in this context in investigative intravitreal toxicity studies. Most FDA-approved topical ocular drug products with pivotal toxicity studies conducted in the past 10 years included ERG evaluation, but most topical older pivotal topical ocular studies did not. These topical ocular studies did not have ERG findings with the exception of decreases in a- and b-wave amplitude observed in a 26-week study at the high dose of bepotastine, a histamine H1 receptor antagonist, in dogs without ocular inflammation or microscopic changes. 68 Similar ERG findings were not observed at 13 weeks in dogs, and ERG was not measured in rabbits. Since ERG is important only if drug reaches the retina, if an ocular pharmacokinetic study shows that drug does not reach the retina, or the ocular drug product is an ocular reformulation that has previously evaluated ERG at higher doses and retinal tissue concentrations with no ERG effects, this information may be useful in justifying exclusion of ERG from pivotal ocular toxicity studies by other ocular routes (topical, periocular, and intracameral) from a regulatory standpoint.
Intravitreal and subretinal toxicity studies need to consider one or more specialized ocular imaging techniques in most cases, especially with small molecule formulations using sustained release technologies biologics, and gene and cell therapies with all formulations. Heidelberg Spectralis ultra-widefield fluorescein angiography (FA; Heidelberg Engineering, Heidelberg, Germany) is used to determine retinal vascular morphology and permeability following IV dye injection and fundus photography. Fluorescein angiography is suited to monitoring intravitreal/subretinal biologics or gene/cell therapies that may induce ocular inflammation secondary to an innate or adaptive immune response, which is usually manifested in and surrounding the retinal vasculature. Spectral domain OCT (SD-OCT) for cross-sectional evaluation of the posterior eye segment has been increasingly employed as a noninvasive imaging modality to evaluate wide-field retinal and choroidal anatomy in intravitreal and subretinal toxicity studies, especially in cynomolgus monkeys. 55,57 Normative macular thickness measurements and common and uncommon spontaneous findings have recently been published for cynomolgus monkeys with SD-OCT using the Spectralis HRA + OCT Heidelberg platform to aid in distinguishing normal retinal thickness and retina and choroidal features from toxic outcomes in ocular toxicity studies. 69,70 The Heidelberg Spectralis platform is the most commonly used technology for widefield fundus imaging, but the handheld RetCam (Natus Medical) that contacts the ocular surface for widefield fundus color imaging and FA is another device in use at CROs for ocular toxicity studies. In the author’s experience, SD-OCT and/or RetCam3 widefield fundus imaging technologies are also useful for tracking location, changing dimensions and appearance, and bioerosion of sustained release technologies, especially biodegradable intravitreal implants and microspheres in dose range ocular toxicity studies, which is important for determining the duration of the recovery phase for GLP ocular toxicity studies, among other useful information for ophthalmologists, pathologists, and toxicologists. Some advantages of the Heidelberg Spectralis platform are that there is no contact with the ocular surface, which avoids compression artifact, the posterior pole and periphery of the retina can be captured in a 1-shot image, and there are other imaging modalities with the standard lens that have utility in ocular toxicity studies, such as confocal scanning laser ophthalmoscopy (cSLO) and autofluorescence. 71,72 Confocal scanning laser ophthalmoscopy is useful for an en face fundus reflectance image of the outer retina and retinal pigment epithelium and has shown utility in nonrodents. 73,74 Confocal scanning laser ophthalmoscopy combined with OCT (cSLO-OCT) has been shown to improve toxicity readouts and facilitate longitudinal examination of single animals at multiple time points in rats 59,75 ; therefore, combined cSLO-OCT to enable location of retinal changes and then structural lesion evaluation in cross sections has promise in ocular toxicity studies in nonrodents as well. The cSLO-guided OCT also has the potential to assist navigation to area of interest in tissue blocks prepared for histology, and facilitation of longitudinal examination of single animals at multiple time points can lead to a marked reduction of animals in ocular toxicity studies according to the 3R principles. 75
Systemic In-Life Parameters
A list of systemic in-life parameters utilizing the FFP concept are presented in Table 3. A NME or an ocular drug product reformulation that has not been given by the proposed route or that contains a novel excipient will need evaluation of systemic in-life parameters in ocular toxicity studies. 28,76 As with all general toxicity studies, standard in-life parameters of general health including clinical observations, body weights, and food consumption should be evaluated for all tolerability/dose range and pivotal ocular toxicity studies. Bioanalytical analysis of drug concentrations in plasma is usually recommended for all ocular toxicity studies regardless of study type, and toxicokinetic analysis should be conducted if there are measurable concentrations at a sufficient number of time points. For an NME, systemic exposure in pivotal ocular toxicity studies is important for demonstrating lower levels compared to systemic exposure at the NOAEL in pivotal systemic toxicity studies, which helps in establishing a safe first-in-human ocular dose, especially for topical ocular drugs with significant systemic exposure. Comparison of systemic exposure obtained after administration of an ocular reformulation to systemic exposure achieved with the previously approved ocular or nonocular formulation in animals and humans is important for demonstrating that the ocular reformulation has lower or equal systemic exposure compared to a previously approved route, which may justify limiting microscopic examination to ocular tissues. 76 In general, systemic exposure following ocular administration that is shown to be negligible has a host of implications for the toxicology program for an ocular drug product. A systemic exposure level generally is considered negligible if it produces no activity in adequately controlled functional activity assays and produces no systemic effects in animal toxicity studies or clinical studies. Therefore, it is imperative that the lower limit of quantitation for bioanalysis in ocular toxicity studies be below the level that would detect functional activity. Bioanalytical analysis of ocular fluids, such as vitreous humor (VH) or aqueous humor (AH) in ocular toxicity studies by intraocular routes, is not a regulatory expectation since these data are collected in pharmacokinetic studies. Bioanalytical drug analysis of VH was conducted for several approved intravitreal biological ocular drug products, including ranibizumab, aflibercept, and brolucizumab 23,77,78 but not for other intravitreal biological ocular drug products, such as anti-factor D monoclonal antibody lampalizumab and an anti-VEGF/ANGPT1/2 monoclonal antibody. 54,79 For gene therapy ocular drugs, biodistribution profile of the vector and viral genome DNA with quantitative polymerase chain reaction in ocular tissues, including the target tissue (retina), biofluids, and systemic tissues for potential off-target biodistribution, is conducted in ocular toxicity studies. 80–81
Systemic In-Life Parameters in Ocular Toxicity Studies.
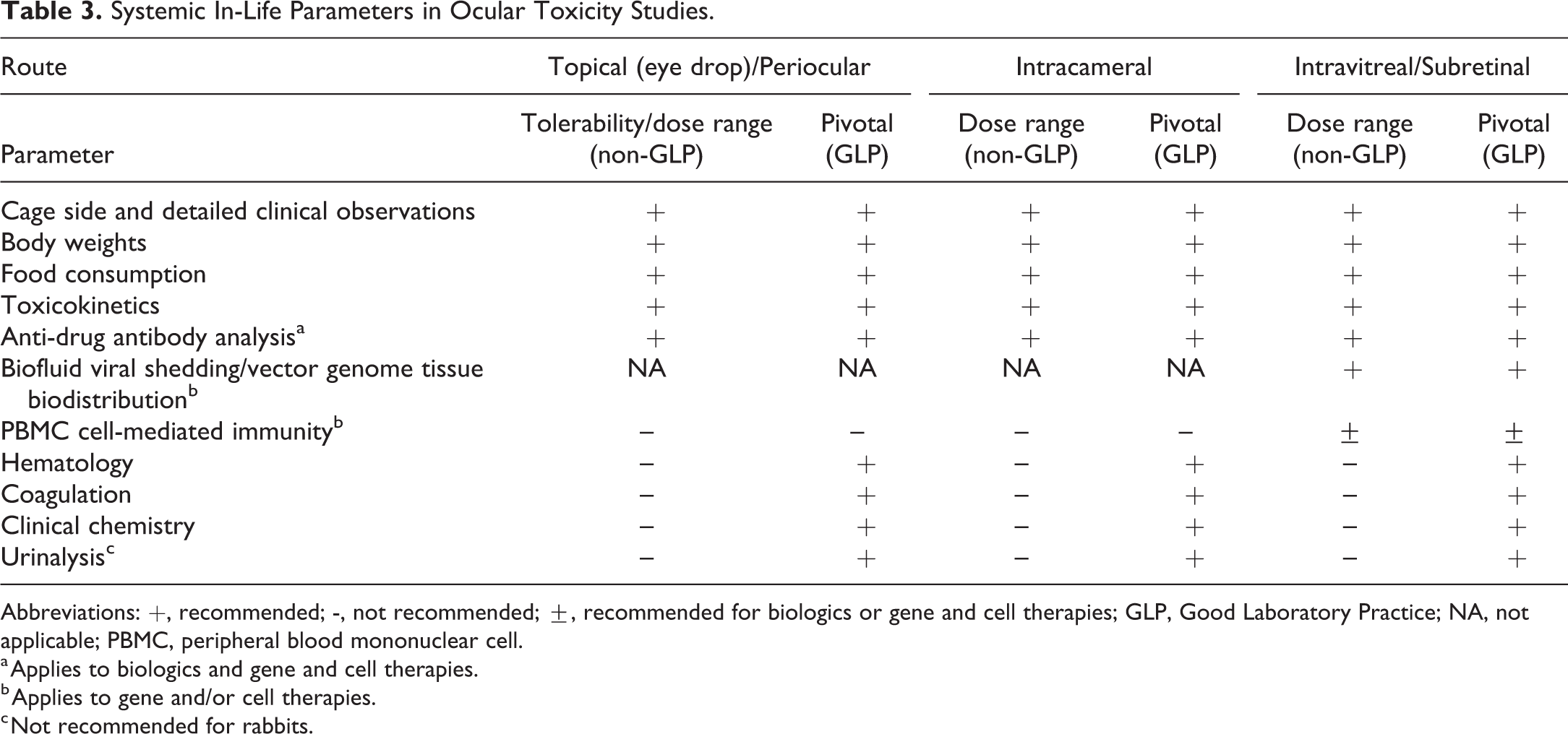
Abbreviations: +, recommended; -, not recommended; ±, recommended for biologics or gene and cell therapies; GLP, Good Laboratory Practice; NA, not applicable; PBMC, peripheral blood mononuclear cell.
a Applies to biologics and gene and cell therapies.
b Applies to gene and/or cell therapies.
c Not recommended for rabbits.
Antidrug antibodies in serum, plasma or blood are important to evaluate in dose-range and pivotal toxicity studies by intraocular routes for biologic ocular drug products, including single-dose administration, to understand potential effects on toxicokinetics or correlation with ocular inflammation to understand if it may be related to a potential adaptive immune reaction to the test article. 10,12–13,29 For gene therapy ocular drug products, blood ADA to the viral vector and genome are conducted to understand humoral immunity, along with evaluation of cell-mediated immunity assays using peripheral blood mononulcear cells or other assays. 80,81 For gene therapy with adeno-associated viral (AAV) vectors in cynomolgus monkeys, intravitreal injections are associated with a higher risk of humoral immune responses and a less favorable biodistribution profile with more viral shedding to blood and draining lymphatic tissue, increased off-target deposition, and less efficient gene transfer compared to subretinal delivery of AAV vectors. 82,83 Detection of ADA in VH or AH is not currently a regulatory expectation for ocular toxicity studies with biologics and gene or cell therapies, and serum, plasma or blood ADA is generally sufficient for evaluating correlations between immunogenicity and ocular immune complex-related effects observed by ophthalmic or microscopic examination. Hurdles with poor drug tolerance and related false-negative results with state-of-the-art bridging assays to detect ADA in ocular fluid samples have recently been overcome with more sensitive immune complex assays. 47 Immune complex assays of VH and AH samples with high residual drug concentrations following intravitreal injection in minipigs and monkeys detected ADA, although systemic ADA were detected earlier than in ocular fluids and systemic ADA response was not always accompanied by the presence of ADA in ocular fluids, indicating systemic ADA detection is adequate in most cases for ocular toxicity studies. Aqueous humor was a good predictive matrix for ADA detection in VH, which is important for ease of sampling in AH compared to VH in ocular toxicity studies and minimizing potential contamination issues in VH.
While clinical pathology is considered a regulatory expectation to monitor for systemic effects in all pivotal ocular toxicity studies regardless of systemic exposure, it is generally not needed for tolerability or dose range ocular toxicity studies since clinical pathology parameters are seldom affected and do not contribute to rationale for dose selection in pivotal ocular toxicity studies. Urinalysis in rabbits is not recommended for pivotal ocular toxicity studies since rabbit urine usually has a thick consistency due to the presence of mucus, sex gland secretions, and calcium carbonate crystals making voided samples often unsuitable for analysis. 84 In the author’s experience, handling and procedures (intraocular injections, ocular examinations, and blood collections) in ocular toxicity studies of rabbits can occasionally elevate routine acute phase response clinical pathology parameters, including C-reactive protein and creatine kinase, and exacerbate the frequency and severity of myocardial inflammatory cell infiltrates. These effects are similar to that seen in with handling and study-related procedures such as blood collection and intramuscular injection in female NZW rabbits and included elevated stress biomarkers (norepinephrine, epinephrine, cortisol, and corticosterone) and cardiac troponin I. 85
Ocular Toxicity Studies: Pathology Parameters and Procedures
The second objective of this review is to cover pathology parameters and procedures for ocular toxicity studies, including ocular histology sectioning procedures to adequately evaluate all major ocular structures and extraocular tissues. Necropsy examination and gross findings for ocular toxicity studies depend upon the objective of the study and may be limited to ocular examination and enucleation for dose range ocular toxicity studies, if only eyes will be collected, to a full necropsy for pivotal ocular toxicity studies. Pathology parameters including collection and histopathology of ocular and systemic tissues and organ weights for ocular toxicity studies based on the FFP concept are listed in Table 4. Ocular dose range toxicity studies usually need to collect and conduct histopathology only on the eye and attached optic nerve, since evaluation of extraocular and systemic tissues is not informative in selection of doses for pivotal ocular toxicity studies and there is no cause for concern for potential nonocular targets of toxicity. Exceptions exist for periocular routes of administration, such as evaluation of eyelids for topical eyelid administration or subconjunctival extraocular tissues for the subconjunctival route. Pivotal ocular toxicity studies should evaluate eyes and all extraocular structures from all main study and recovery animals on the study are listed in Table 4. It is recommended that a full tissue list and organ weights be collected for all pivotal ocular toxicity studies regardless of the level of systemic exposure. For pivotal ocular toxicity studies with NME or ocular drug product reformulations approved by another route, microscopic evaluation of a full systemic organ/tissue list is usually conducted on control and high-dose main study animals, with evaluation of target organs in intermediate groups, if present. As discussed previously, FDA’s reformulation guidance 75 states that if systemic exposure from an ocular drug product reformulation has systemic exposure that is equivalent or less than that of an approved route, and there are no novel excipient concerns, microscopic evaluation may be limited to the eye and extraocular tissues. For this case, it is still advised to collect and retain a full systemic tissue list in case there are systemic effects or to address potential regulatory concerns.
Pathology Parameters in Ocular Toxicity Studies.

Abbreviations: +, recommended for all groups, including recovery; -, not recommended; ±, case-by-case recommendation; GLP, Good Laboratory Practice.
a Present in rabbits, minipigs.
b Present in rabbits, dogs, minipigs.
c Collection of organs/tissues and organ weights in all groups and histopathology of control and high-dose groups and gross observations and systemic target organs in intermediate groups.
Eye Fixation, Trimming, and Sectioning
Details regarding fixation and trimming techniques for ocular and extraocular tissues in ocular toxicity studies have been extensively covered, 4,10,60,86,87 and this section will focus on selected aspects and a brief update on fixation and recommended trimming and sectioning for eyes as a “hot topic” based on new regulatory expectations. Left and right eyes should be collected separate and marked to designate the 12-o′ clock (dorsal midsagittal) position, and any site of injection using indelible dye, tattoo ink, or suture, and placed in individual jars so that unilateral ophthalmologic or trimming correlations can be matched to the correct eye. 4 The most common eye fixation techniques for ocular toxicity studies is immersion fixation of the eye and attached optic nerve in Davidson’s for 18 to 24 hours or modified Davidson’s for 48 to 96 hours with subsequent storage in 70% ethanol or 10% neutral-buffered formalin before trimming. 4,66,86,87 Trimming of eye and determining the number of sections for microscopic evaluation is currently undergoing review and revision within the pharmaceutical industry based on recent regulatory expectations to ensure adequate microscopic evaluation of the eye in ocular toxicity studies, regardless of species, route, or NME or ocular drug product reformulation. In the author’s experience, regulatory expectations include (1) adequate ocular microscopic assessment of a full range of ocular tissues, (2) microscopic evaluation from a sufficient number of sections, with a full assessment of the macula/fovea or equivalent (visual streak), (3) consideration of in-life observations, including ophthalmology, in regard to microscopic sectioning, and (4) the study pathologist’s justification for the section/assessment approach. The Society of Toxicologic Pathology’s (STP) Scientific and Regulatory Policy Committee (SPRC) has formed a Non-Rodent Ocular Trimming & Sectioning (NOTS) Working Group to survey common ocular histology sampling/sectioning procedures for nonrodent species (rabbits, dogs, minipigs, monkeys) among industry in ocular toxicity studies, and the NOTS Working Group is charged with drafting recommendations on ocular sectioning paradigms to satisfy regulatory expectations and standardize approaches for each species with a publication. Histology sectioning recommendations are presented below based on the author’s experience; however, more detailed and authoritative recommendations with diagrams for rabbits, dogs, minipigs, and monkeys will be published by the NOTS Working Group.
The major recommendation is that each eye and attached optic nerve should be trimmed and at least 5 H & E sections per eye should be made for all ocular toxicity studies in rabbits, dogs, minipigs, and monkeys. A total of 5 sections or more should allow adequate evaluation of a full range of ocular tissues including the cone-rich visual streak/macula which varies by species as diagramed in Figure 1.8 by Vezina. 88 The visual streak (macula equivalent) is inferior to the optic disc in rabbits, 89,90 superior/temporal to the optic disc in dogs, 91,92 and superior to the optic disc in domestic pigs and minipigs 93 –95 and inadvertently referred to as area centralis in domestic pigs and minipigs by several laboratories. 96,97 Sagittal (vertical) trimming with 2-step H & E-stained sections for temporal (200-500 µm apart) and central calottes (500-1000 µm apart) and 1 H&E-stained section for the nasal calotte should be made for eyes from these species, which should capture the visual streak identified by high ganglion cell density in retina from multiple sagittal sections from all 3 species. Additional sections may be collected and left as unstained slides for special stains if desired. The area centralis (fovea equivalent), which has the highest cone density in monkeys and humans, is not present in rabbits and minipigs. In dog, area centralis has a higher cone density than the cone-rich visual streak and is located 0.6 mm superior and 1.5 mm temporal to the optic disc. 91 Histology sectioning to reliably capture the area centralis in dogs is impractical, and examination of the cone-rich visual streak is adequate for evaluation of the cone-rich area of retina in ocular toxicity studies with this species similar to that for rabbits and minipigs that lack an area centralis. The temporal calotte usually contains the temporal/superior injection sites in intraocular studies of rabbits, dogs, and minipigs, but it may not always be practical to identify them in histological sections, especially in single-dose studies with solution formulations. Since ocular sustained release technologies usually migrate to the inferior portion of the anterior or posterior segment in intraocular toxicity studies, sagittal sections from the central calotte are adequate to capture the inferior region of the anterior and posterior segments in ocular sections from rabbits, dogs, and minipigs.
Based on the temporal (lateral) location of macula/fovea with respect to the optic disc in monkeys, horizontal trimming is usually conducted. The central calotte should be embedded and several step sections (∼20 µm apart) should be made to collect macula/fovea and optic disc and 2 additional step sections (∼500-1000 µm apart) should be made to include another section of optic disc and one section superior to the optic disc. For intraocular studies, a vertical section of the superior calotte to contain injection sites is warranted. For intraocular studies with sustained release technologies, a vertical section of the inferior calotte to include the inferior region of the anterior and posterior segments to evaluate migration and potential test article effects is warranted. Depending on the type of study, 5 to 7 H&E sections per eye should be made and additional sections may be collected and left unstained for special stains if desired.
The cone-rich visual streak/macula is identified histologically by increased numbers of ganglion cells. It is important that consistent orientation of sections from each eye is made so that the pathologist can easily identify inferior/superior (dorsal/ventral) orientation in rabbits, dogs, and minipigs studies and temporal/nasal (lateral/medial) orientation in monkey studies. Contralateral eyes may should be sectioned as mirror images and slide block diagrams should explain orientation for right and left eyes. This will allow the study and peer review pathologists to determine location (superior/inferior/temporal/nasal) of regional ocular microscopic findings if present and assist in correlation with ophthalmologic or gross (trimming) observations.
Special staining procedures on ocular sections are not routinely done in ocular toxicity studies for small molecules and biologics, unless there is a focused investigative aim to understand the pathological response. For gene and cell therapy ocular toxicity studies, special pathology procedures in ocular sections such as in immunohistochemistry for target protein and/or situ hybridization to identify the target gene or cell transfer in the eye is commonly conducted, along with other specialized immunological techniques such as lymph node immunophenotyping. 75
Extraocular Tissues
Left and right extraocular tissues including upper and lower eyelids, lacrimal gland, harderian gland (rabbits and minipigs), nictitating membrane (rabbits, dogs and minipigs), mandibular lymph node, and nasolacrimal ducts should be collected and evaluated in all groups in pivotal ocular toxicity studies as listed in Table 4 but are generally not collected in dose range ocular toxicity studies unless there is justification. Extraocular tissues should be collected and processed separately and examined as separate protocol organs/tissues for microscopic evaluation to allow correlation of ophthalmologic or trimming findings in pivotal ocular toxicity studies. There is sufficient justification to evaluate the nasal lacrimal system in ocular toxicity studies since there is ample exposure through lacrimal duct drainage, especially with topical administration. Treatment-related findings can occur in the nasolacrimal duct following administration of topical, intravitreal, and systemic drugs, 78,87 which justifies evaluation in pivotal ocular toxicity studies with all routes of administration in the authors’ experience, although some CROs prefer to evaluate the nasolacrimal duct microscopically only if warranted by clinical signs. Although ocular muscles are recommended to be collected and evaluated, 4 inclusion of collection and evaluation of ocular muscles in pivotal ocular toxicity studies usually only occurs at the sponsor’s request since ocular muscle microscopic findings in ocular toxicity studies are rare.
Ocular Toxicity Studies: Pathology Report
The third and most important objective of this review is to provide an overview of a high-quality pathology reports for ocular toxicity studies. Generating a high-quality pathology report includes activities prior to placement and initiation at a CRO, review of study-related data, microscopic examination, the pathology report narrative, and pathology peer review to ensure that ocular pathology data and interpretations are accurate and include integration of key ophthalmology to help produce a high-quality ocular toxicity report. Many of these principles build upon STP’s best practices for reporting pathology interpretations within GLP toxicity studies 98 and industry-CRO pathology interactions that facilitate creation of the best quality pathology report. 99,100 The scope of this section is to address industry-CRO interactions and the author’s perspective on pathology report best practices with specific reference to ocular pathology evaluation for dose range and pivotal ocular toxicity studies and not to restate industry or CRO perspectives and best practice recommendations in reviews cited above other than providing context if needed. The sponsor-CRO relationship must be a collaborative and nonadversarial relationship that balances the perspectives and needs of both CROs and sponsors, 99 and the success of this relationship and the generation of a pathology report of high quality will greatly depend on thorough communication at all stages of a toxicity study. 100 In most cases, the study director is an employee of the test facility CRO, the study pathologist and histologist are employees of the test facility or pathology CRO, the study ophthalmologist is an employee or independent contractor of the test facility CRO, and the peer review pathologist is an employee or independent contractor of the sponsor. This current pharmaceutical industry business model requires extremely close and effective communication between individuals at different sites across multiple time zones to work efficiently as a team to produce high-quality pathology and ophthalmology contributing reports and toxicology reports. It is unwise to wait to engage the study pathologist and peer review pathologist until “the end of the parade” when slides are ready for microscopic examination and pathology peer review, respectively.
Prior to Placement and Initiation of Study at CRO
Submission of an RFP to one or several test facility CROs to receive a quote for one or more ocular toxicity studies is the first step as discussed above. As with all general toxicity studies, sponsors and CROs should engage in communication of the procedures and processes that affect the generation of anatomic pathology and ophthalmic data, and before placing studies, sponsors should fully communicate their expectations, which may affect the decision to select one CRO over another, as well as cost and timing. 100 A critical part of toxicity studies is the selection of a study pathologist, and with the large number of ocular toxicity studies conducted over the past 10 years, there is usually one or several anatomic pathologists with experience and expertise in ocular pathology at each test facility or pathology CRO. The ACVP or European College of Veterinary Pathology certification is not a regulatory requirement but is recommended for ocular toxicity studies. The sponsor should inquire as to the experience of potential study pathologists with ocular toxicity studies at the CRO test facility or pathology CRO. The sponsor should consider providing names on one or several preferred study pathologists based on ocular experience and expertise, with all the caveats for potential delays or scheduling complications mentioned for any general toxicity study. 100 In many cases, the study pathologist is added by protocol amendment based on scheduling constraints, but providing the sponsor’s study pathologist preferences during protocol development ensures selection of a study pathologist with ocular experience and expertise.
Contract research organizations in the United States and Canada who regularly conduct general toxicity studies have 1 or 2 employees who are ACVO-certified ophthalmologists or enlist an independently contracted ACVO ophthalmologist. There is no European board certification for veterinary ophthalmologists. Ophthalmologic examination and specialized ophthalmic procedures by an ACVO ophthalmologist is highly recommended for all ocular toxicity studies conducted in the United States and Canada and outlying regions. For independently contracted ophthalmologists, the sponsor should inquire about their experience with ocular toxicity studies since some are primarily engaged in practice at veterinary clinics or universities, which may be a disadvantage for complex ocular toxicity studies. Most CROs have highly trained technical staff who conduct ocular observations (modified Draize) for all ocular toxicity studies. A few CROs have a highly trained scientist who conducts ophthalmic examinations for nonpivotal ocular toxicity studies, but this is usually the exception and the sponsor may request an ACVO ophthalmologist depending on the objectives of the study.
It has long been advocated that providing information about the test article to the CRO, in particular the study pathologist, is clearly beneficial and facilitates the work of the study pathologist and the peer review pathologist, 98,100,101 and this information should also be shared with the study ophthalmologist. Information to consider providing to the CRO includes literature references, results, or reports from previous ocular toxicity studies with the same compound and publicly available information on similar compounds, such as NDA pharmacology/toxicity reviews. Sponsors should notify the CRO in advance if a specific report format or style is necessary, including the extent to which ophthalmologic findings are to be correlated with microscopic findings, which is discussed later. Currently, most CROs are willing to state this correlation in pathology reports, while others prefer that this correlation be made only in the toxicology report for ocular toxicity studies, so discussions with CRO management and scientific personnel should occur beforehand.
How sponsors handled characterization of pathology findings as adverse findings used to vary widely due to the complex and subjective nature of the process. 102,103 There are now best practices and recommendations for determining and communicating adverse effect data in nonclinical studies as published by STP, the European STP, and an STP continuing education course. 104 –106 In the author’s experience, there is reluctance to characterize adversity of ocular findings by some pathologists; however, adversity should be addressed in pathology reports for all ocular toxicity studies. From a pathology/toxicology consultant’s perspective, adversity should also be addressed in ophthalmology reports as discussed below. Therefore, CRO policies and practices regarding establishing adversity in pathology and ophthalmology reports should be a topic for discussion between the sponsor and the CRO. Consistent with industry and CRO practice, the NOAEL for pivotal ocular toxicity studies should be determined in the context of the complete study data set of the toxicology report and not within the pathology and ophthalmology reports. 99–100,104–105
The decision to process all tissues from all animals to blocks and/or slides is driven by the study protocol, should be at the discretion of the sponsor, and can result in important benefits, particularly when time lines are important to the sponsor. 100 As discussed above, this is generally not an issue for eyes and extraocular tissues since they should be examined microscopically from all animals. For pivotal ocular toxicity studies with systemic tissue evaluation, examination of systemic tissues from high dose and control groups is most common and test article-related findings in systemic tissues are uncommon. Therefore, processing systemic tissues from intermediate-dose groups and/or recovery groups to slides is usually not conducted based on cost/benefit considerations. However, if clinical observations and/or gross necropsy findings indicate potential systemic effects, then processing of systemic tissues from all groups is recommended to avoid delays in microscopic examination.
Lastly, the sponsor should indicate if they would like a pathology peer review and if they will be providing a sponsor’s pathologist or if they would like the CRO to provide a pathologist within the same organization or company, or a third party. The eye is one of the tissues most prone to diagnostic error in toxicology studies, and one of the steps that sponsors can take to reduce risk associated with subjectivity or error in anatomic pathology evaluation is to use pathology peer review. 107 The sponsor’s peer review pathologist for an ocular toxicity study frequently has access to information that is not available to the CRO’s study pathologist, including ocular target biology and pharmacology, ocular pharmacokinetic data, previous findings with the same ocular drug or drug–device product including mechanisms of target-mediated and off-target toxicity, experience with ocular vehicle excipients, ocular formulation impurities such as endotoxin or host cell proteins, sustained release technologies, and knowledge of procedure-related effects for intraocular toxicity studies. As with all toxicity studies, this additional experience and knowledge by a sponsor’s peer review pathologist significantly improves the quality of the pathology peer-review process and the final pathology interpretations and in no way inappropriately influences the study pathologist’s interpretations. 108
In the author’s experience, pathology peer review should be conducted for all intraocular and periocular toxicity studies including dose range studies, since intraocular or periocular injection or topical application to periocular structures usually result in procedural-related ocular findings and often test-article and/or vehicle-related findings that should be characterized and interpreted as such with agreed-upon terminology and identification of potential adverse effects that are crucial to understand for justification of dose selection in pivotal intraocular toxicity studies. Ocular tolerability studies for topical (eye drop) ocular drug products, usually conducted in one species (rabbits), typically do not have terminal end points including microscopic evaluation, and dose range toxicity studies are not usually conducted prior to pivotal ocular studies unless there is cause for concern. Pathology peer review of pivotal topical ocular toxicity studies is highly recommended; however, they are not always conducted at the sponsor’s discretion if test article- and/or vehicle-related effects can be predicted to be absent or of minimal severity and nonadverse based on results from ocular tolerability studies. For the purposes of study design and conduct and data evaluation below, it is assumed that a pathology peer review is included and details regarding pathology peer review are described in another section below.
Study Design and Review of Study-Related Data
It is essential that both the study pathologist and the sponsor’s peer review pathologist participate and contribute to study design to ensure that study objectives can be achieved. 100 In some cases, the study pathologist has not been assigned during protocol development; therefore, it is even more critical that the sponsor’s peer review pathologist participate in study design, especially with recent regulatory expectations regarding ocular histology in ocular toxicity studies. As with all toxicity studies, the CRO should provide the study pathologist and peer review pathologist with all study-related information and data, including protocol and amendments, clinical pathology data tables and draft clinical pathology report, and in-life data such as clinical signs, body weights, and food consumption. 98,100 Toxicokinetic and ADA information should be provided to the study pathologist and peer review pathologist preferably before slide evaluation or later during the reporting process. For ocular toxicity studies, the CRO should provide the study pathologist and peer review pathologist with data tables from ocular observations and ophthalmic examinations including data tables and ophthalmology exam data sheets from each animal prior to necropsy that contain diagrams to indicate the location of ocular findings and/or the character (color, shape, size, and/or number) of sustained release technologies or other descriptions of test article that may help in nonroutine ocular sectioning by the histologist and correlation with microscopic findings by the pathologist and peer review pathologist. Data or images from specialized ocular examination techniques listed in Table 2 should also be provided by the CRO to the study pathologist and peer review pathologist. It is highly desirable that the ophthalmology report is available for review before microscopic examination, but this is not usually the case and the study pathologist and peer review pathologist need to rely on close communication at this stage to understand the ophthalmologist’s interpretation of ophthalmology data and their perspective on potential test article-, vehicle-, or procedural-related ocular effects and if any of these ophthalmologic findings are considered adverse. As with any general toxicity study, organ weights and gross findings of all organs and tissues, including those in ocular and extraocular tissues found at necropsy, should be reviewed by the study pathologist and peer review pathologist prior to microscopic examination. 98 This should include gross findings noted at trimming that were not observed at necropsy, and these trimming findings should be entered into the gross pathology database. This is especially important for intraocular toxicity studies with sustained release technologies, since gross findings noted at trimming are generally more revealing than gross findings noted at necropsy, as discussed previously. It is wise for the study pathologist and peer review pathologist to review individual histology sheets in case gross findings at trimming for ocular and extraocular tissues were inadvertently not entered in the gross pathology database. Key ophthalmology findings at the last examination time point before termination can be considered more important than gross pathology findings at necropsy, as ocular necropsy observations are usually uncommon except for some corneal findings. Postfixation gross findings at trimming are uncommon but important especially for intraocular studies with sustained release technologies since findings may be present that were not observed during ophthalmologic examination, such as vitreal or retinal findings in the inferior portion of the globe, which may be out of the field of view with routine indirect ophthalmoscopy but would be detected with wide-angle fundus imaging and OCT.
As with all toxicity studies, the study pathologist and the peer review pathologist should communicate freely and directly during the primary microscopic evaluation to assist refinement of terminology of preliminary ocular findings and determine how significant ocular pathology findings, particularly unexpected adverse findings, will be communicated to the sponsor during the evaluation of data. 100 If significant findings are identified, there must be timely communication to the study director and sponsor’s study monitor since they may be considered as reportable findings when safety of patients is at risk per 21CFR§312.32(c)(1)(iii). Unexpected adverse ocular findings are usually first observed during ophthalmologic examination, but in the author’s experience, this is not always the case as discussed above for findings in the inferior portion of the globe.
Microscopic Examination: Data Entry and Tables
The use of standardized terminology for pathology observations between multiple studies for any pathology report facilitates the peer review process, avoids confusion when discussing findings with the sponsor, and ensures continuity between pathology reports within a project as well as within the toxicologic pathology community in general. The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria) for Lesions in Rats and Mice, www.toxpath.org/inhand.asp, is a joint initiative between STPs from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for nonproliferative and proliferative lesions in laboratory animals. The INHAND nomenclature for special sense organs (ocular, olfactory, and otic) has recently been published for rats and mice. 109 An INHAND project for special sense organs in nonrodents (rabbits, dogs, minipigs, and monkeys) is ongoing. Therefore, the INHAND publication on special sense organs in rodents should be consulted for standardized terminology for nonrodents until publication of the INHAND publication for nonrodents is available. If the microscopic finding is not covered within the INHAND for nonrodents, additional references in the next section should be reviewed.
Computer system data entry for gross and microscopic findings in eye and extraocular tissues should be separated by right and left for each tissue. Ocular microscopic findings should all indicate the location of each finding by the ocular tissue compartment listed in Table 5, and if there are similar findings among several ocular tissue compartments, such as “inflammatory infiltrates, mixed cell, minimal” or “inflammation, mononuclear cell, mild,” the microscopic finding should be entered separately for each ocular tissue (cornea, vitreous, retina, etc) so the pathologist can determine if there are any differences in incidence between various ocular structures for a similar finding in summary tables. It is accepted practice in microscopic data collection for the eye that the entry “no abnormality” or “within normal limits” means that all ocular structures listed in Table 5 have been adequately examined and there is no need to list each ocular structure separately in tables or create separate protocol tissues for each ocular structure to record findings or note the absence of findings. The location of an ocular microscopic finding should be further defined if there is regionality to its location within the anatomical structure. Including inferior (ventral), superior (dorsal), temporal (lateral), nasal (medial), anterior, and posterior locators for anterior chamber and vitreous findings; anterior, posterior, and subcapsular locators for lens findings; central or peripheral locators for corneal and retinal findings are some examples. For intraocular studies with sustained release technologies, inclusion of these locators can help the pathologist to understand the relationship between initial deposition or migration of the drug depot in anterior chamber or vitreous and a surrounding foreign body inflammatory response and/or adjacent tissue reaction that reflects physical, chemical, or immune-mediated injury to the retina, lens, cornea, or other ocular structures. 11 –13 Another advantage in recording these regional locators is that it may help correlate microscopic with ophthalmic findings in a specific region of the ocular tissue.
Major Anatomical Eye Structures Evaluated by Microscopic Examination in Ocular Toxicity Studies.
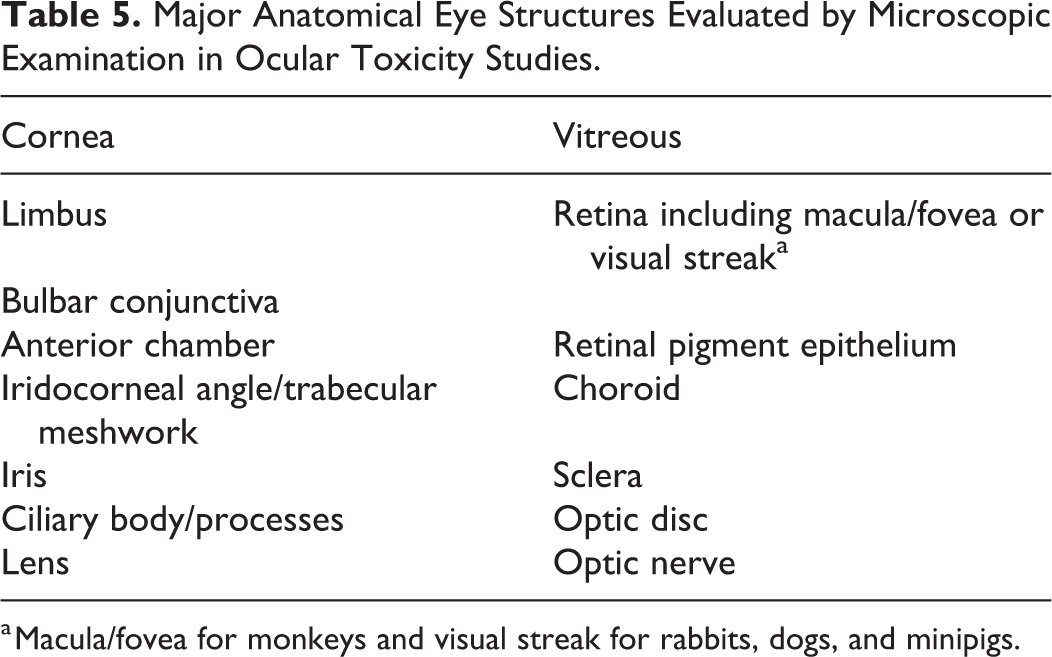
a Macula/fovea for monkeys and visual streak for rabbits, dogs, and minipigs.
The use of severity grades as a semiquantitative assessment of the relative extent of ocular and extraocular microscopic findings should follow that published by the STP Working Group commissioned by the SPRC. 110 Grading is subjective, and one of the prime advantages of pathology peer review by the same pathologist is controlling grading within and between studies, but minor differences of opinion within one grade are usually acceptable. This “Points to Consider” article states that it is simply not feasible or necessary to apply lesion-specific grading criteria to the vast majority of microscopic findings, and this includes eyes and extraocular tissues unless the study pathologist believes this step is warranted for transparency for a particular key finding. It is understood that documentation of grading criteria for critical findings helps the reviewer follow the pathologist’s observations and interpretation of changes, particularly for histopathologic changes that are important in determining adversity or dose-limiting effects. For example, specific grading criteria for retinal degeneration based on the specific layers affected, and degree of thinning and decreased cellularity has been proposed. 108 For some findings, it is sufficient to record histopathologic changes only as “present” without the assignment of a severity grade, if the severity of the change is difficult to assess in the tissue section and/or will not impact the interpretation of the finding or the relationship of that finding to the test article. 110 For example, presence of injection sites or descriptions of drug depot material from sustained release technologies (ie, “implant” or “implant remnants”) do not need to be assigned a severity grade and “present” with location is sufficient.
Computer systems used by CROs to enter animal data, including gross findings, microscopic findings, and organ weights, such as Provantis (Instem), Path/Tox System Pristima (Xybion), and Ascentos (PDS), may or may not be configured by CROs to enter all ophthalmic examination findings for ophthalmology reports. Regardless, with all systems, there is no correlative function for microscopic and ophthalmic findings in individual animal data entry for pathology tables as is present for microscopic and gross findings. Therefore, the pathologist for an ocular toxicity study should carefully review ophthalmic findings recorded prior to necropsy from each eye during microscopic data entry and decide which ophthalmic findings merit consideration as key ophthalmic findings to include as a correlative comment with that ocular tissue. The most common example is a “cataract” observed by ophthalmic examination in intraocular studies and usually listed as lens opacity in data tables, which can usually be correlated with “lens fiber degeneration” microscopically (cataracts are clinical findings and are not diagnosed as such microscopically). Cataracts are key findings in ophthalmology reports because they may be test article-, vehicle-, or procedure-related or spontaneous and they are usually considered adverse and irreversible; therefore, it is an important study objective for the ophthalmologist and pathologist to determine the potential cause of cataracts and lens fiber degeneration, respectively. Close communication between both scientists and the peer review pathologist before microscopic examination starts should address potential key ophthalmic findings that may benefit from a correlative comment in microscopic data entry, since this step may add value in supporting qualitative correlative statements in the discussion of the pathology and toxicology reports. For a positive correlation, a comment for the microscopic observation of “lens degeneration” such as “Lens fiber degeneration correlates with ophthalmic examination finding of cataract” is recommended. For no microscopic correlate for a cataract, a tissue comment for the affected eye such as “No correlate with ophthalmic examination finding of cataract” is suggested. Other key ophthalmic findings such as corneal opacity, vitreal cells, retinal detachment, or descriptions of sustained release technologies (ie, implants) should be considered if there are microscopic findings that may be correlated with ophthalmic findings. For example, a comment for the microscopic observation of “corneal injection site” for an intracameral toxicity study such as “Correlates with ophthalmic examination finding of corneal opacity” is suggested. An ophthalmic finding of vitreal cells may have a microscopic correlate of vitreal inflammatory infiltrates or inflammation. An ophthalmic finding of anterior chamber aqueous flare and/or aqueous cells may have a microscopic correlate of anterior chamber proteinaceous fluid and/or inflammatory cell infiltrate, respectively. However, anterior chamber ophthalmic findings usually arise within days of intraocular injection and are transient and may not be present by ophthalmic examination prior to necropsy, unlike vitreal cells that usually appear 1 to 2 weeks following intraocular injection and persist for weeks or months and therefore are more frequently observed by microscopic examination. Other examples of key ophthalmic and ocular microscopic correlates are presented below. It is beyond the scope of this article to present a comprehensive list of potential correlates for consideration. Furthermore, only key ophthalmic findings should be considered and it is not necessary or advised to indicate microscopic correlates for all ophthalmic findings in a study. For most ophthalmic and microscopic findings, it is not considered necessary to enter comments for negative correlation in microscopic data entry.
The study pathologist and peer review pathologist must be experienced in recognizing many artifacts, especially in ocular sections, and avoid entering these findings. Handling, fixation, trimming, and/or sectioning artifacts are present in most ocular toxicity studies. Each step of tissue preparation introduces artifacts in eye sections. Corneal wrinkles, corneal epithelial tears, separation or vacuoles, corneal stromal clefts, corneal endothelial vacuoles, ciliary epithelial vacuoles, lens shattering, shredding, cracking, lens vacuoles or spherules, retinal detachment, photoreceptor inconsistent appearance, vacuoles, or eosinophilic bodies, and optic nerve vacuoles are examples of artifacts that may be observed in ocular toxicity studies that must not be confused with test article-, vehicle-, or procedure-related findings. The preponderance of artifacts are one of the reasons why untreated eyes should be included in ocular toxicity design as mentioned above.
Pathology Report Narrative
Report organization and style
There are specific components that should be included in the results and discussion section of a pathology report narrative for a general toxicity study such as a clear description of treatment-related findings, their significance, and their relationships to corresponding information in control animals and stating clear reasons for deciding that specific observations are not related to treatment. 98 For an ocular toxicity study, there can be added complexity to determine if findings are related to the drug or drug–device (test article), the vehicle (ie, formulation, slow-releasing device, or polymer implant placebo) or the procedure (injection, ocular implantation, or periocular administration, if applicable), or if findings are incidental, background changes or if findings are a histological artifact of fixation, trimming, or sectioning. Therefore, there are up to 5 categories for ocular microscopic findings and the contributing role of some of them may be difficult to sort out. Intraocular toxicity studies commonly have injection-related findings and formulation-related inflammatory or foreign body responses that can be exacerbated at higher doses of test article and/or the drug delivery system material. 12 Procedure-related retinal microscopic and ERG changes occur with subretinal dosing of solution formulations. 111,112 Periocular application that involves injection or eyelid application also has the potential for procedural-related findings. Topical (eye drop) ocular toxicity studies do not usually have procedural-related findings. The pathologist and peer review pathologist must be aware of incidental ocular findings, which occur in all nonrodent species and are discussed below.
Ocular microscopic findings should be presented before extraocular microscopic findings, and most important findings (ie, test article-related) should be discussed first and grouped together if related. In other words, ocular microscopic findings of various tissues do not need to be organized from anterior to posterior segments (ie, “front to back” as in cornea to retina). Vehicle-related ocular microscopic findings (if any) should be presented next, followed by procedure-related ocular microscopic findings, and incidental ocular microscopic findings should be presented last. Extraocular microscopic findings should be presented following ocular findings, and usually, these findings are restricted to test article-related findings and incidental, background changes since vehicle- and procedure-related findings are uncommon unless there is direct application to the extraocular tissue, such as eyelids.
Inclusion of brief descriptions of toxicologically important ocular and extraocular microscopic findings is encouraged in the report and severity grades should be briefly explained in certain cases, such as retinal degeneration. 110 Ocular microscopic findings should be described to include location with the range of severity within a group or groups and identify group or groups using dose levels (rabbits administered ≥0.05 mg/right eye) rather than group numbers or terms such as “high dose,” although the latter is usually CRO preference unless the sponsor indicates a more specific term or value. Presentation of incidence in sentence format is encouraged if there are only a few findings and presented among eyes and not animals (“minimal or moderate retinal degeneration was observed in 2 of 14 eyes (14%) injected with 0.05 mg/right eye”]. If there are a greater number of test-article-, vehicle-, and/or procedural-related ocular and extraocular findings, a text table indicating severity and incidence of ocular and extraocular microscopic findings is recommended. Findings in the table should be listed in the same order as presented in text, and common findings among different ocular tissues should be listed separately by ocular tissue and not lumped together. Ocular and extraocular microscopic findings in males and females should be combined to evaluate overall group incidence if no gender-related differences are observed, which is usually the case in ocular toxicity studies. It is also preferable to combine findings in right and left eyes in a summary text table if bilateral dosing was conducted, since both eyes were treated similarly and the incidence of findings among the total number of eyes treated can be compared among vehicle- and test article-treated groups. For ocular toxicity studies with unilateral dosing, it is important to keep findings separate between right and left eyes in text tables of the report narrative to highlight differences between vehicle- or test article-treated and untreated eyes.
Microscopic findings in systemic organs and tissues, if present, should be presented following presentation of ocular and extraocular microscopic findings in the pathology report. For many ocular toxicity studies, test article-related microscopic findings in systemic tissues and organs are uncommon. However, systemic exposure is observed with all ocular routes of administration, although the ratio of local to systemic exposure varies by route. Topical and sub-Tenon administration will provide the highest systemic exposures for small molecule drugs and most likely to be associated with systemic toxicity, followed by intracameral, periocular route such as subconjunctival and eyelid administration and the lowest potential for systemic exposure by intravitreal and subretinal administration. Toxicokinetic information is not always available for the pathologist and peer review pathologist to evaluate for discussion of potential relationships in the pathology report, so a discussion of these relationships may need to be added later during the pathology report process or included in the discussion of the toxicity report with input from the study pathologist and peer review pathologist. Vehicle- or procedure-related microscopic findings in systemic organs and tissues is also uncommon, except for procedure-related intramuscular findings in any species and stress-associated inflammatory cell findings in rabbit heart discussed above. 85
Report interpretation of findings: Test article-, vehicle-, or procedure-related or incidental background?
It is beyond the scope of this article to cover the wide array of test article-related pathology findings and underlying modes of action in ocular toxicity studies of nonrodents and there are comprehensive articles and book chapters on toxicologic pathology of the eye and extraocular tissues that are helpful. 4,66,87,113 –117 The difficulty of safety assessment of sustained release technologies in ocular toxicity studies highlights the challenges to these complex systems including target tissues and types of ocular toxicities including local chemical- and immune-related toxicity and local physical toxicity with foreign body responses. 10 –13
Microscopic findings of various ophthalmic vehicle formulations have not been widely addressed in ocular toxicologic pathology publications other than as single excipients or isolated reviews as discussed below and are beyond the scope of this article. Phrases that include “treatment-related” should be avoided in ocular toxicity studies since the vehicle is also a treatment, so the report should specify the effect as test article- or vehicle-related to avoid confusion. Frequently, a vehicle formulation may contain one or several excipients that are novel (unapproved) for a given route or that are present at a higher concentrations than currently approved in ophthalmic formulations for a given route, and these novel or higher concentrations of excipients need to be qualified as safe for regulatory purposes. Vehicle-related effects can sometimes be difficult to sort out from incidental background changes, procedural-related effects, and histology artifacts, especially if untreated eyes are not included in the study. In any case, the study pathologist and peer review pathologist need to address the safety of the vehicle in the report, especially if one of the objectives is to qualify one or more vehicle excipients. Vehicle-related microscopic effects depend on several factors, including route of administration, species, type of formulation as a solution, suspension, or sustained release technology. 12 It can be quite challenging to determine the causative role in ocular formulations that contain several excipients and/or complex sustained release technology. Over 20 vehicles in intravitreal solutions have been evaluated in rabbits, including various concentrations of sodium chloride (NaCl) and different osmolarities and pH. 118 For intravitreal solutions, phosphate-buffered saline is much better tolerated than isotonic NaCl, especially in minipigs and less so in rabbits. 35 In the author’s experience, monthly injection of a balanced salt solution in minipigs caused retinal atrophy and similar but less severe changes in rabbits. This suggests that minipigs and rabbits may be more susceptible to adverse ocular effects from repeated intravitreal injection of simple salt solutions considered safe in other species, including humans. Chemical toxicity produced by excipients within an sustained release technology, such as solvents, polymers, or surfactants, and their metabolites or degradation products ideally should be present as a vehicle formulation to test in a control group to determine the impact of these excipients as well as size and shape of nano- or microparticles or implant rods over the life cycle of the sustained release formulation. 11–12 Non-ADA-related general mixed cell inflammation is common with both particle and/or implant and suspension sustained delivery ocular formulations that is usually present with vehicle formulations. 12 Unfortunately, the combination of drug with the sustained release technology can alter size and shape and affect the biodegradation profile; therefore, a true “control” vehicle formulation with the same characteristics as the test article is not always possible. With some sustained release ocular formulations, the chemistry is such that a true vehicle or placebo cannot be formulated, and therefore, a vehicle control group is not possible.
Procedural-related effects, usually by the result of iatrogenic intervention (injection) in intraocular toxicity studies, depend on the route of injection. The cornea may be affected following intracameral injection with microscopic needle tracks or from an incision site and sutures 87 or following sampling of AH, and these effects may be characterized as corneal opacity by ophthalmology. Less commonly, intracameral injection trauma can cause microscopic anterior capsule lens fiber degeneration, manifested as lens opacity (cataract) by ophthalmology, or microscopic iris adhesion to the cornea or lens, manifested as anterior or posterior synechiae, respectively, by ophthalmoscopy. Intracameral injection-related inflammatory responses can also be present in other anterior segment tissues, including anterior chamber, limbus, trabecular meshwork or iris, and conjunctiva. Intravitreal injections, drug–device implantation, or other surgical trauma from posterior segment delivery procedures will create a needle track in the sclera and form a scleral fibrous plug as part of the wound repair process, which may transect the choroid and retina (depending on the site of injection and species differences width of the pars plana) and extend into the interior ocular space and comingle with endogenous cells, such as zonule fibers suspending the lens or cells within the vitreous, with both populations contributing to the membranous process or vitreal prolapse. 114 Microscopic posterior capsular lens fiber degeneration can occur with needle prick by intravitreal injection and correlates with a cataract by ophthalmoscopy in some cases. Differentiating injection procedure-related inflammatory findings in various ocular tissues from vehicle-related and sometimes test article-related inflammatory findings can be difficult, especially since sham injection control eyes are not commonly included in ocular toxicity studies, but nevertheless, procedure-related findings must be identified as such in the report using the pathologist’s best judgment.
Incidental background or spontaneous ocular and extraocular microscopic findings are generally random or sporadic across dose groups but those that occur with increased incidence or severity in treated eyes including the high dose group should include a rationale why they were considered as such and not test article-, vehicle-, or procedure-related. For key incidental findings that may be misinterpreted by regulatory reviewers as due to other causes, consider citing literature references to support the incidental nature of these findings, such as Langevin et al 119 for albino and pigmented rabbits and Woicke et al 120 for cynomolgus monkeys. Spontaneous ocular and extraocular findings in laboratory beagles are uncommon, and there are several findings mentioned in references cited above for ocular toxicologic pathology by Schafer and Render, 87 individual case reports, and a recent article on background retinal vacuolation in neonatal dogs. 121 Spontaneous ocular and extraocular findings are uncommon in minipigs and there is one reference by Helke et al 122 for a limited number of animals evaluated in this survey.
Report Discussion Hot Topic: Determining and Communicating Adversity
As discussed above, determining and communicating adversity in ocular toxicity studies is a key objective and another “hot topic” in this article that should be addressed in pathology reports. Anatomic and clinical pathologists are well aware of best practices and recommendations for determining and communicating adverse effect data in nonclinical studies as published by STP. 104,123 The CRO study team personnel, including study directors, toxicologists, ophthalmologists, and management, as well as sponsor representatives, study monitors, and toxicologists should also be aware of STP best practices and recommendations for determining and communicating adversity. Interpretation of test article-related effects as adverse and assigning an NOAEL is a cardinal feature of GLP studies, including ocular toxicity studies. Consistent with industry and CRO practice, the NOAEL for pivotal ocular toxicity studies should be determined in the context of the complete study data set and not within contributing reports. 99–100,104–105,123 , Although assigning an NOAEL is not usually done for dose ranging studies, interpretation of test article effects as adverse or nonadverse is important for determining the relevance of animal toxicity data in these studies, including ocular dose range toxicity studies. From the author’s perspective, adversity should be addressed in pathology reports for all ocular toxicity studies regardless of GLP status. It is important for the sponsor and the CRO to understand early on in drug development if ocular microscopic and other study-related findings are considered adverse or not to aid in dose selection for pivotal ocular toxicity studies and to begin internal discussions on human risk assessment and confront any issues with developability of the ocular drug product including the formulation or procedures used to deliver it. In the author’s experience, there is reluctance to characterize adversity of ocular findings in pathology reports from dose range and pivotal ocular toxicity studies by some study pathologists, and this should be addressed in the pathology peer review.
It is beyond the scope of this article to review all 10 major recommendations for communicating adverse findings and nonadverse findings 104 and further characterization of adversity 105 in the context of pathology reports for ocular toxicity studies or provide numerous examples of adverse effects in ocular and extraocular tissues caused by drug treatment or toxins found elsewhere. 4,87,114 Only a few STP best practice and recommendations on adversity will be emphasized in this section with some examples from ocular toxicity studies in the author’s experience, with the understanding that adversity of ocular and extraocular findings is controversial and needs more careful consideration and discussion among pathologists, ophthalmologists, and toxicologists. Publications on adversity guidelines refer to adversity in the context of test article effects. One of the unique aspects of ocular toxicity studies discussed throughout this review is that an adverse effect may also be vehicle-related due to chemical toxicity of an excipient or chemical or physical toxicity of a sustained release technology placebo or an adverse effect may be procedure-related due to trauma from an ocular injection or drug–device implantation. Therefore, determining adversity in pathology reports of ocular toxicity studies as discussed below applies to all categories of ocular microscopic findings whether they are considered test article-, vehicle, - or procedure-related.
Adversity is a term indicating “harm” to the test animal within the constraints of the study design (recommendation 1), and decisions about adversity or nonadversity should be unambiguously stated and justified in subreports and/or the study report (recommendation 2) and should be applied only to the test species and under the conditions of the study (recommendation 3). 104 It is preferable to address adversity of a key microscopic ocular finding or group of findings in the pathology report with a brief justification of the finding(s) as adverse or nonadverse. Retinal degeneration may be considered “inherently adverse” regardless of severity or distribution of the finding. 105 Lens fiber degeneration regardless of severity or distribution is usually considered adverse as well since it too is generally not reversible and may be progressive within the context of the study with potential for visual deficits. As with other tissues and organs, for ocular findings that are not inherently adverse, severity of the change can be an important factor in deciding upon adversity. 105 An example is minimal or mild ocular inflammation which is usually considered nonadverse and moderate or severe ocular inflammation which is usually considered adverse. Although the lack of reversibility could indicate additional cause for concern and consequently increases the likelihood that a change will be considered adverse, reversibility does not automatically indicate nonadversity. 105 For example, corneal epithelium is highly regenerative and most corneal lesions will recover but that does not mean all corneal findings would be considered nonadverse. The decision on adversity should not take into account whether this finding may occur in humans; however, conjecture about human susceptibility to such effects is perfectly valid and even expected to be discussed within the report narrative, especially where the pathologist is aware of human/animal correlates of a particular toxicity or even where literature is available showing that certain target effects in animals have no counterpart in humans. 106 One example is adverse vacuolation/demyelination of the medullary ray in rabbit eyes. In one case, the pathology report stated that the retina of other species including humans does not have an equivalent myelinated structure and that this finding may be of limited relevance when translated to humans.
Communication of what is considered adverse and assignment of NOAEL in the overall study report should be consistent with and supported by the information provided in the subreports (recommendation 5) and should include direct interaction between staff within different scientific disciplines (recommendation 6). 104 The toxicology report for an ocular toxicity study should discuss adversity with regard to ophthalmic examination findings as well as ocular microscopic findings and they should be consistent with what was provided in the ophthalmology and pathology reports, which may benefit from direct interaction between the study director, study ophthalmologist, and study pathologist and others including the peer review pathologist. This recommendation also infers that the ophthalmology report should also address adversity in the same manner that a pathology report addresses adversity, although recommendations as such have not been published other than STP best practices that refer to other subreports as stated above in recommendation 5. It is particularly important that there is early and clear communication between such authors so that the various manifestations of toxicity due to a single effect may not be thought to be due to multiple different toxicities. 105 This does not mean that a pathology report and ophthalmology report have to agree on adversity of correlative findings. For determining the NOAEL in a toxicology report for a pivotal ocular toxicity study, an adverse finding in one subreport will generally overrule the correlative findings as nonadverse in the other subreport. For example, test article-related lens fiber degeneration may be considered adverse in a pathology report, but a correlative test article-related pinpoint lens opacity (cataract) may not be considered adverse in the ophthalmology report since the ophthalmologist’s interpretation was that the cataract did not impair the field of vision. In this case, the NOAEL would be set according to the adversity call in the pathology report.
As stated above, recommended best practices on adversity refer to test article-related effects. One of the more difficult and controversial decisions for a study team in establishing the NOAEL for pivotal ocular toxicity studies that has not been addressed in publications of STP best practices or recommendations on adversity is if the NOAEL applies strictly to test article-related adverse effects since vehicle- or procedure-related adverse effects are not mentioned. In the author’s perspective and experience, the NOAEL should apply only to test article-related effects and that vehicle- or procedure-related adverse effects should not influence the NOAEL. Therefore, the adversity of vehicle- or procedure-related effects should be clearly stated in pathology and toxicology reports of dose range and pivotal ocular toxicity reports and should not influence the test article-related NOAEL nor should a separate NOAEL be defined for vehicle- or procedure-related effects for pivotal ocular toxicity studies.
Similar to communication of the test article-related NOAEL in an overview document (recommendation 7) with active participation of nonclinical scientists who interpret data from nonclinical studies in assessing and communicating human risk (recommendation 9), 104 vehicle- or procedure-related adverse effects observed in ocular toxicity studies should also be communicated in an overview document for human risk assessment that takes into account interaction from toxicologists, pathologists, and ophthalmologists. For example, an intracameral toxicity study with procedure-related anterior cataracts in vehicle and test article groups in rabbits and dogs should not influence the NOAEL but would be clearly described in the overview document with the potential human risk. In this case, the potential for cataracts in humans following intracameral injection may be low based on species differences in anatomy including smaller lens size to anterior humor volume ratio in humans compared to those species or other considerations that arise following discussions with nonclinical and clinical scientists.
Other Report Discussion Topics
A discussion of relationships between various significant study related findings (eg, in-life findings, gross findings, body weight changes, organ weight changes, clinical pathology changes, and microscopic findings) needs to be considered for any pathology report of a general toxicity study. 98 For an ocular toxicity study, the most important discussion is between ocular microscopic findings and standard and specialized ophthalmic examination findings and ocular trimming observations, since these findings represent ocular structural “gross” or “macroscopic” findings or functional findings (ie, IOP, ERG). This discussion is best positioned in the results section, similar to that followed in most pathology reports that describe correlative relationships between macroscopic observations at necropsy with microscopic findings, but this preference is up to the study pathologist and CRO practices. The ophthalmology report may or may not be available at this point; however, a discussion with the ophthalmologist on their interpretation of test article-, vehicle-, and/or procedure-related effects and potential positive and/or negative correlations with ocular microscopic findings before the pathology report is drafted is highly recommended. Most CROs will agree to placement of the discussion of the correlation between key ophthalmic findings and ocular microscopic findings in the pathology report, although in the author’s experience, this is not universal at present and some CROs mandate that this discussion takes place only in the toxicology report. Omitting a discussion of key ophthalmic findings in the pathology report has the disadvantage that the study pathologist and peer review pathologists may misinterpret ocular microscopic findings by not addressing the relationship with ophthalmic findings with the ophthalmologist’s interpretation, especially with test article-related versus procedural-related effects. Another disadvantage is that there is usually a delay in communication between the study pathologist, peer review pathologist, ophthalmologist, and study director to address these correlative relationships until the discussion is drafted in the toxicology report. This delay in communication can impose a burden on the study director and other scientists mentioned above who may not have given much thought to correlative findings until long after contributing reports were drafted.
A statement should be provided in the discussion section of the pathology report confirming full assessment of retinal macula/fovea (monkeys) or equivalent retinal visual streak (cone-rich region for rabbits, dogs, and minipigs) in sections from all eyes in the study, which is a regulatory agency expectation as discussed above.
Review of ADA-related immunogenicity data in the toxicology report is crucial in defining the mechanism of ocular inflammation with biologics and gene and cell therapies. 10,12 The ADA data may not be available for the pathologist and peer review pathologist to review at the time of slide evaluation. Therefore, discussion of the correlation between ADA data and ocular microscopic findings for individual animals as supportive evidence for the role of an adaptive immune response in ocular inflammation may need to be added later to the pathology report. Alternatively, this integrated discussion could wait until the toxicology report with the study pathologist’s and peer review pathologist’s input. Plasma toxicokinetic data commonly collected in ocular toxicity studies are seldom helpful in understanding ocular microscopic findings. If collection of ocular fluids (tears, AH, and VH) was conducted in the study, then analyte data including drug concentrations or biomarkers should be provided to the study pathologist and peer review pathologist for discussion of relationships to ocular microscopic findings in the pathology report.
Pathology Peer Review
The STP has carefully considered the topic of pathology peer review for several decades and published reviews and commentaries on the topic, including comprehensive recommendations 106 and a review of Guidance on the GLP Requirements for Peer Review of Histopathology (Guidance No. 16) issued by the Organisation for Economic Co-operation and Development (OECD, 2014) by Fikes et al. 124 The FDA has recently issued draft guidance on Questions and Answers on Pathology Peer Review in Nonclinical Toxicology Studies, 125 and STP has provided comments for consideration and incorporation of these comments into the final guidance. 126 These documents as well as publications from a sponsor’s or CRO’s perspective 99–100 fully apply to ocular toxicity studies, and areas for consideration that have not been addressed for ocular toxicity studies are discussed below.
As emphasized in previous sections for data review prior to microscopic examination of an ocular toxicity study, the peer-review pathologist generally is responsible for reviewing all relevant pathology-related data that the study pathologist reviewed, including ophthalmology data and the draft ophthalmology report if available and ocular histology trimming macroscopic observations if any. The peer review pathologist should have familiarity with standardized terminology of ocular and extraocular tissues, especially within the context of pathology reports from prior ocular toxicity studies with the test article, and should have established communication with the study pathologist prior to their microscopic examination and may benefit from discussions with the study pathologist and study ophthalmologist prior to peer review.
For toxicity studies in general, the peer review pathologist must have broad flexibility to select materials for review within wide general guidelines (standard operating procedures and the objectives of the peer review) and must have latitude to examine additional organs and other study data as necessary to thoroughly evaluate all pathology findings. 108 In most nonrodent toxicity studies, the peer review pathologist usually examines all protocol organs in at least half the high dose and control animals in the treatment phase, and target organs are examined from all animals on the study including the recovery phase. In the author’s experience for ocular toxicity studies, these recommendations hold true for systemic tissue evaluation, but for ocular and extraocular tissue evaluation, pathology peer review should be conducted on all animals on the study based on the low numbers of nonrodents compared to rodents and the complexity in interpretation and determining the source of ocular findings as test article-, vehicle-, or procedure-related or incidental background changes or artifact.
The peer review pathologist should ensure that the individual animal tables contain accurate and appropriate terminology for ocular and extraocular microscopic findings and that locators are included for each ocular tissues rather than “lumping” similar microscopic findings among different ocular locations and that severity grades have been adequately assigned. As discussed previously, individual animal tables should have a comment for a microscopic finding in an ocular tissue that correlates with a key ophthalmic finding or if no microscopic correlate exists, a general comment for the eye that no microscopic correlate exists for that key ophthalmic finding. For most ophthalmic and microscopic findings, it is not considered necessary to enter comments for negative correlation in microscopic data entry. Pathology peer review of ocular toxicity studies should also evaluate quality of histology sections of ocular and extraocular tissues discussed previously, including artifacts, consistency of globe orientation, and adequacy of full assessment of all major ocular structures as listed in Table 5, including the macula/fovea or visual streak. Feedback on histology quality of eyes and extraocular tissues should be provided to the study pathologist to forward the histology department to improve ocular and extraocular histology techniques for future studies.
The peer review pathologist’s responsibilities for an ocular toxicity study are to ensure that the draft pathology report narrative accurately reflects and is supported by the individual animal data tables and summary tables; identifies test article-, vehicle-, and/or procedure-related microscopic observations and the dose groups in which these findings occur in treated eyes and extraocular tissues; and identifies incidental background changes and the dose groups in which these findings occur in treated and untreated eyes and extraocular tissues. The peer review pathologist should also ensure that all the other features described above for the presentation and interpretation for the draft pathology report narrative are covered, including organization and presentation of microscopic ocular and extraocular findings, addressing adversity, and discussion of correlations of ocular microscopic findings with ophthalmic findings. Following review of the ophthalmology report, the peer review pathologist may discover that there is a discrepancy between it and the pathology report in categorization of findings as test article-, vehicle-, and/or procedure-related or incidental and background changes. More commonly, the ophthalmology report is not available at this point, and a discussion with the study ophthalmologist, study pathologist, and study director is highly recommended, if this has not occurred during the study pathologist’s examination. The purpose of this discussion is to reconcile the ophthalmologist’s interpretation of key ophthalmology findings in the context of the study pathologist’s and peer review pathologist’s interpretation of correlative microscopic ocular findings and their categorization. Lastly, based on regulatory expectations, the peer review pathologist should check to see that draft pathology report narrative contains a statement that the macula/fovea or retinal visual streak was observed and if the presence of microscopic findings in this region was noted.
As with other toxicity studies that include a pathology peer review, 108 the study pathologist and peer-review pathologist for ocular toxicity studies should reach consensus on target ocular and extraocular tissues; terminology for test article-, vehicle-, and/or procedure-related findings; dose groups with these findings; and other major conclusions within the study pathologist’s report. When differences in opinion regarding pathology findings or interpretations exist that could affect risk assessment, informal consultations and/or formal (documented) consultations with additional experienced pathologists and/or subject matter experts often lead to resolution of discrepancies. If consultation results in agreement by the study pathologist and peer review pathologist on the major pathology findings and dose groups affected, documentation of the informal consultations with other experts should not be required because consensus has been reached and is documented in the peer review statement. 108
Ocular Toxicity Studies: Ophthalmology Report
Recommended practices to produce a high-quality ophthalmology report have not been published, and the author’s perspective on these practices is briefly provided here. The author’s perspective is not that of an ophthalmologist but represents the perspective of a pathology/toxicology consultant who has reviewed many ophthalmology reports and addressed integration of ophthalmic and ocular microscopic findings in pathology and toxicology reports from ocular toxicity studies. There are excellent publications that deal with ophthalmology in the context of ocular toxicity studies as noted above in the discussion of ocular in-life parameters, including a recent book that provides standards and harmonization for procedures, terminology, and scoring schemes for ophthalmic evaluation in ocular toxicity studies that have been endorsed by ACVO. 64 The Standard for the Exchange of Nonclinical Data (SEND), for study data tabulation of nonclinical studies with standardized terminology that enables FDA to modernize and streamline the review process, has not yet been implemented for ophthalmology data as it has for pathology 127 and most other parameters for general toxicity studies. In the author’s experience, the lack of SEND implementation for ophthalmology data does not preclude regulatory expectations for SEND-compliant formats for all other parameters (domains) from dose range and pivotal ocular toxicity studies.
In the author’s experience, publications for best practices for anatomic pathology and clinical pathology reports 98,128 are useful for ophthalmology reports. The inclusion of specific elements, formatting, and arrangement of ophthalmology reports varies depending on the CRO and individual study circumstances and usually minimally contains study design, results, and discussion of standard and specialized ophthalmic examination techniques presented in Table 2, scoring schemes, and individual and summary tables of ophthalmology data. From a pathology/toxicology consultant’s perspective, results and discussion narrative section for ophthalmology reports should contain description and characterization of ophthalmic examination findings that include severity expressed as terms that are consistent with the semiquantitative ocular scoring scheme. This includes stating the score (0-3+, 0-4+) or a range of scores for an observation or group of observations as well as providing a severity term that is consistent with the ocular scoring scheme for that observation (ie, very slight, slight, moderate, severe or trace, mild, moderate, severe). Unfortunately, not all observations in ocular scoring schemes have semiquantitative severity terms; therefore, in these cases, the ophthalmologist should be encouraged to use their best judgment and provide severity terms such as “minimal, mild, moderate, or severe,” similar to widely accepted severity terms used for anatomic and clinical pathology data. The rationale for providing widely accepted severity terms along with grading scores is that severity scores are usually removed in overview documents from multiple studies, but severity terms are provided for anatomic and clinical pathology and most other parameters from ocular toxicity studies. Therefore, for consistency in describing findings across parameters, inclusion of severity terms for ophthalmology data is preferred over severity scores in overview documents.
It can be a challenge to summarize key ophthalmic findings accurately and concisely in narrative form since there may be several intervals for ophthalmic examination findings during the dosing phase. The specific incidence of the various scores, by category and by interval, should be shown in the report with text tables, and combining scores across findings for one aggregate score are generally not recommended (Paul Miller, DVM, ACVO, personal communication). Providing text tables for the incidence of findings is commonly done in the pathology report narrative as discussed above. Ophthalmology reports usually include interpretation of data from other standard examination parameters, such as IOP evaluation and ERG, and interpretation of images from specialized examination parameters, such as wide-field fundus imaging and SD-OCT for intravitreal toxicity studies and gonioscopy for intracameral toxicity studies. For some CROs, another principal investigator with expertise in ERG provides a separate contributing scientist report for that parameter. Ocular observations conducted by technical staff are not included in the ophthalmology report and they are summarized by the study director in the ocular toxicity report.
There are no publications that specifically address determining and communicating adverse effect data in ophthalmology reports as exists for anatomic pathology and clinical pathology reports, 104,123 although STP’s recommended best practices for establishing and communicating adversity in toxicity studies encompass subreports in general so these recommendations are applicable to ophthalmology reports, although they are not specifically mentioned. In the author’s experience, adversity of key ophthalmic findings is not addressed in a majority of draft ophthalmology reports and the current approach at most CROs is to include it in the discussion of the ocular toxicity report. However, from a pathology/toxicology consultant’s perspective, it is advantageous for the study ophthalmologist to address adversity in the ophthalmology report, similar to that recommended for study pathologists in pathology reports. Most ophthalmologists and CROs are receptive to addressing adversity in the draft ophthalmology report but may need guidance with references discussed above.
There are no publications that specifically address integration and correlation of ophthalmology and anatomic pathology results in toxicity studies, similar to that described for integration and correlation of clinical and anatomic pathology results. 129 Many of the basic principles presented and illustrated for correlating anatomic and clinical pathology findings and integrating these with other study endpoints can be applied to integrating anatomic pathology and in-life ophthalmic end points. As discussed above, integration and correlation of ophthalmic findings and ocular microscopic findings is advised for pathology reports but not recommended for ophthalmology reports, which would be a duplication of effort.
Ocular Toxicity Study: Toxicology Report
The study pathologist, ophthalmologist, and director for an ocular toxicity study have similar responsibilities to interact with each other and other study team members to achieve high-quality contributing reports that will ensure a high-quality toxicology report. Study pathologists and ophthalmologists must ensure their interpretations and discussion including adversity and/or correlations are accurately incorporated and integrated in the toxicology report. For toxicology reports in general, the scientific judgment needed to interpret test article–related findings and to assign an NOAEL for a nonclinical study requires communication among key members of the study team responsible for data interpretation. In such deliberations, due weight must be given to the opinions of the experts who wrote each subreport rather than deferring the decision solely to the study director. 104 For the overall toxicology report, the study director is also ultimately responsible for integrating test article-, vehicle-, and procedure-related findings and correlating ocular microscopic and ophthalmic findings as well as leading discussions on decisions defining adverse findings and the NOAEL that combine the expertise of the study pathologist and ophthalmologist. If pathology and ophthalmology reports have adequately addressed test article-, vehicle-, and procedure-related finding and their adversity and the pathology report has also addressed correlations between microscopic and ophthalmic findings, then the study director’s burden for primary authorship of the toxicology report is reduced. In this case, the study director can direct their focus to carefully review draft pathology and ophthalmology reports and use the discussion on correlations in the pathology report and make a final determination on the NOAEL in the toxicology report. Finally, the study team including the study pathologist, study ophthalmologist, quality control and/or quality assurance staff, and sponsor’s team, including the representative, study monitor, and peer review pathologist, should review the draft toxicology report to provide input. In the generation of high-quality pathology, ophthalmology, and toxicology reports for ocular toxicity studies, the sponsor-CRO relationship must be collaborative with direct collegial communication that balances the perspectives and needs of both parties. 100
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
