Abstract
The acidic and non-acidic fractions of Boswellia serrata gum resin extracts were combined to prepare a unique product, LI13019F1 (Serratrin). The present series of studies evaluated LI13019F1 for acute and subchronic (28-day) toxicity in Wistar rats and acute dermal and eye irritation in New Zealand white rabbits. The mutagenicity and clastogenicity of LI13019F1 were evaluated in bacteria and mouse bone marrow erythrocytes, respectively. All studies were performed following the Organization for Economic Co-operation and Development guidelines. Acute oral and acute dermal toxicity studies did not show mortality or signs of toxicity in Wistar rats at a limit dose of 2,000 mg/kg LI13019F1. LI13019F1 did not cause irritation to the skin or the eyes of New Zealand white rabbits. In a repeated dose 28-day oral toxicity study, LI13019F1-treated Wistar rats did not show dose-related signs of toxicity on their body weights, organ weights, and on the hematology and clinical chemistry parameters. The estimated no observed adverse effect level for LI13019F1 was 1,000 mg/kg/day in both male and female rats. The bacterial reverse mutation test and a micronucleus assay in mouse bone marrow erythrocytes revealed that LI13019F1 was neither mutagenic nor clastogenic. Together, the present observations demonstrate a broad-spectrum safety of LI13019F1.
Keywords
Introduction
For the last 2 decades, various preparations of the oleo gum resin of Boswellia serrata or Indian Frankincense, commonly known as olibanum, have gained popularity among the consumers in the management of a variety of inflammatory diseases, including arthritis, inflammatory bowel disease, allergy, and asthma. 1,2 The pentacyclic triterpenic acids, called boswellic acids (BAs), are the active phytochemicals accountable for the anti-inflammatory and other biological activities of the resin. 3 The 6 major BAs in B serrata resin are α-BA, β-BA, 3-O-acetyl-α-BA, 3-O-acetyl-β-BA, 11-keto-β-boswellic acid (KBA), and 3-O-acetyl-11-keto-β-boswellic acid (AKBA). 3 Among the BAs, KBA and AKBA are considered as the most active modulators for the anti-inflammatory properties of the Boswellia gum resin extracts. 3 They reduce leukotriene synthesis via blocking 5-lipoxygenase activity, a major pro-inflammatory enzyme; this event has been considered as a fundamental basis of anti-inflammatory properties of the Boswellia extracts. 3,4 In addition, AKBA and KBA also reduce the Nuclear factor kappa B regulated pro-inflammatory cytokine production. 5 -7
LI13019F1 (also known as Serratrin) is a unique composition prepared using a novel combination of the acidic and nonacidic fractions of B serrata gum resin. This unique Boswellia composition is standardized to at least 30% of the 6 major BAs containing at least 5% of the KBAs.
Here we present a systematic toxicological evaluation of LI13019F1, including acute oral toxicity, acute dermal toxicity, and repeated dose 28-day oral toxicity studies in Wistar rats, as well as acute dermal irritation and eye irritation studies in New Zealand Albino rabbits. Along with these, a bacterial reverse mutation assay and an erythrocyte micronucleus assay in mouse bone marrow present the safety of LI13019F1.
Materials and Methods
Test Item
LI13019F1 (Serratrin) is a unique composition of acidic and non-acidic components derived from an aqueous ethanol extract of B serrata gum resin. The gum resin was extracted with aqueous ethanol, and the concentrated extract was subjected to phase separation to obtain Boswellia nonacidic resin extract and the acidic extract containing BAs. Two parts of the BAs containing resin extract and one part of the nonacidic resin extract were combined, milled, and sieved using a unique process to obtain LI13019F1. To maintain the quality and batch-to-batch consistency, LI13019F1 was standardized to contain at least 30% of total BAs with not less than 5% KBAs.
The phytochemical marker compounds in the herbal composition were analyzed on high-performance liquid chromatography (HPLC) system (Waters Corporation, Milford, Massachusetts) using Empower 3 software (Waters Corporation). Briefly, the methanol-extracted LI13019F1 solution was filtered through 0.22 µm polyvinylidene difluoride membrane and transferred into an autosampler vial. The sample was eluted through a Kinetex C18 2.6 µm (100 mm × 4.6 mm) column (Phenomenex, Torrance, California) using a gradient system consisting of solvent A (0.1% vol/vol orthophosphoric acid in water) and solvent B (acetonitrile and methanol at 45:55 vol/vol). Following the sample injection, the column was eluted initially with 20% A and 80% B solvent for 16 minutes, followed by a linear gradient reaching up to 18% A and 82% B in 2 minutes, and finally maintained an isocratic run at 18% A and 82% B for further 12 minutes. The column was eluted at a uniform flow rate of 1.3 mL/min at 45°C. Figure 1 shows a representative HPLC chromatogram of LI13019F1 presenting an elution profile of the major BAs monitored at 210 nm. The peaks 1 to 6 with the retention times of 2.973, 5.416, 10.154, 10.972, 22.660, and 24.305 minutes represent KBA, 3-acetyl-11-keto-β-BA, α-BA, β-BA, 3-O-acetyl-α-BA, and 3-O-acetyl-β-BA, respectively (Figure 1).
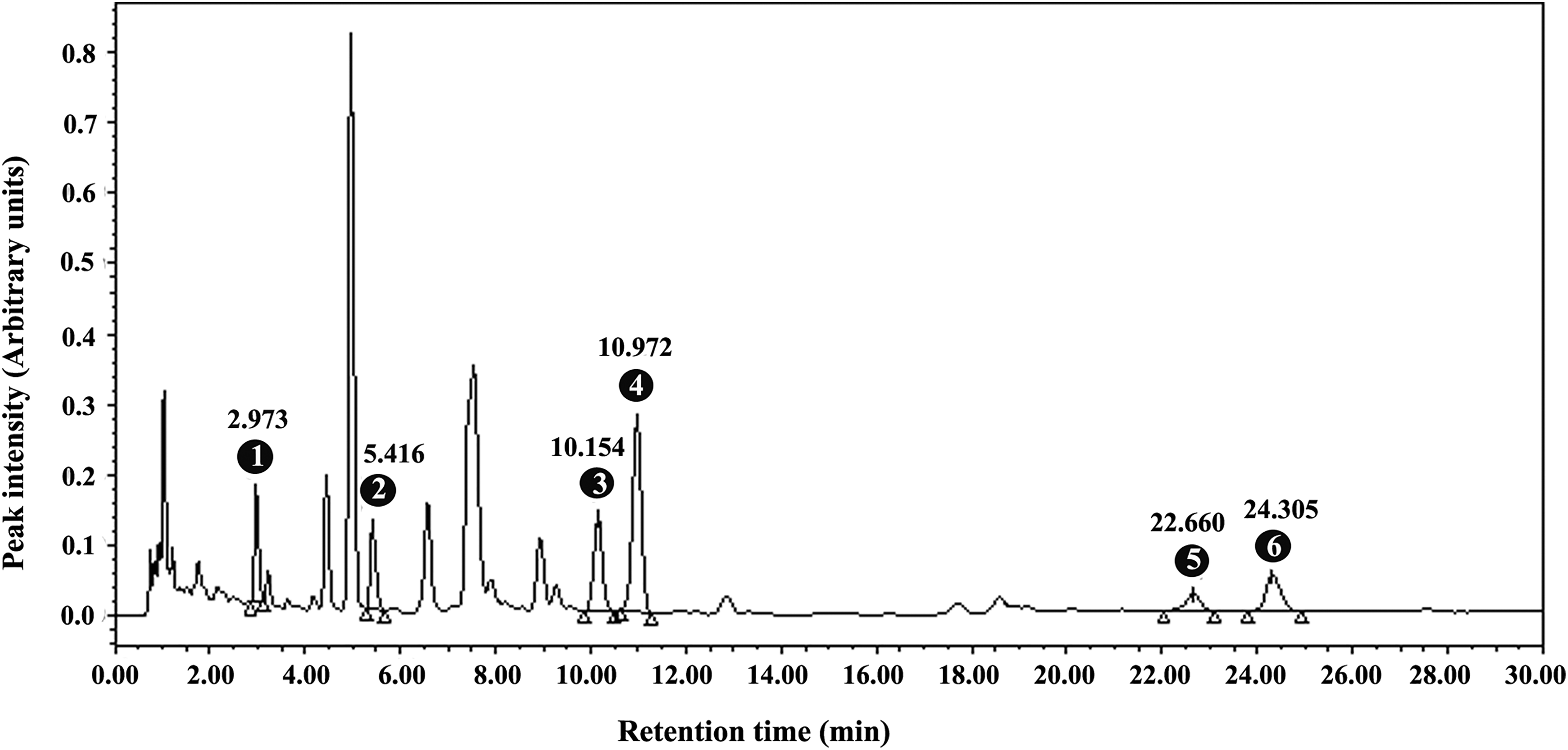
A representative high-performance liquid chromatography shows the elution profile of the major boswellic acids of LI13019F1 at 210 nm. The peaks 1 to 6 represent 11-keto-β-boswellic acid, 3-acetyl-11-keto-β-boswellic acid, α-boswellic acid, β-boswellic acid, 3-O-acetyl-α-boswellic acid, and 3-O-acetyl-β-boswellic acid, respectively with their retention times as indicated.
Chemicals, Reagents, and Bacterial Strains
The sources of the clinical chemistry and hematology reagents were Ilab Aries (Bergamo, Italy) and SIEMENS (Munich, Germany), respectively. Bacto Agar and fetal bovine serum were purchased from Becton Dickinson (Sparks, Maryland) and Sigma-Aldrich (St Louis, Missouri), respectively. The Salmonella typhimurium strains (TA98, TA100, TA1535, and TA1537) and Escherichia coli WP2 uvrA/pKM101 were purchased from the National Collection of Type Cultures (London, England) and the National Collection of Industrial, Food and Marine Bacteria (Edinburgh, Scotland), respectively. Lyophilized rat liver S9 fraction was procured from Celsis In vitro Technologies (Baltimore, Maryland). All other analytical and laboratory reagents were purchased from the authorized local vendors.
Experimental Animals and Housing Conditions
The toxicity studies included acute oral and acute dermal toxicity in Wistar rats, acute dermal and acute eye irritation in New Zealand white rabbits, repeated dose 28-day oral toxicity in Wistar rats, and a micronucleus assay in mouse bone marrow erythrocytes. Male and female Wistar rats were purchased from Vivo BioTech Limited (Hyderabad, India). Swiss albino mice and New Zealand white rabbits were sourced from Palamur Biosciences Private Limited (Hyderabad, India) and Mahaveera Enterprises (Hyderabad, India), respectively. The experimental animals were specific pathogen-free. The animals were acclimatized to the laboratory conditions for 7 days and maintained in a controlled environment (21°C ± 2°C, 40%-70% relative humidity, and 12 hour/12 hour light/dark cycle). During the study period, the animals were allowed free access to a standard diet and bottled mineral water. The Institutional Animal Ethics Committee approved the experimental protocols. The animal care operating procedure followed the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals, India. All studies followed the regulations of the good laboratory practices (GLPs), and the test procedures were in agreement with guidelines from the Organization for Economic Co-operation and Development (OECD) for the testing of chemicals. A GLP-certified animal facility at Laila Nutraceuticals (Vijayawada, Andhra Pradesh, India) performed these studies. Advinus Therapeutics Ltd, a GLP-certified test facility in Bengaluru, India, performed the Ames assay.
Acute Oral Toxicity Study in Female Rats
The acute oral toxicity test was conducted according to the OECD guideline-425 for testing of chemicals. 8 Briefly, a limit test was performed utilizing 9-week-old female (n = 5) Wistar rats. LI13019F1 suspended in CMC-Na (0.5% wt/vol) was administered by gavage at a dose of 2,000 mg/kg body weight (BW). Before the test item administration, the animals were observed for clinical signs. The animals were monitored for morbidity/mortality or clinical signs at 30 minutes, 1 hour, 2 hours, 3 hours, and 4 hours after the test item administration. Thereafter, all animals were observed once daily for 14 consecutive days. Body weights were measured once a week during the observation period. The vital organs and tissues were subjected to gross pathological examinations following CO2 euthanasia on day 15.
Acute Dermal Toxicity Study in Rats
An acute dermal limit toxicity study using five 9-week-old rats of each sex was conducted following OECD guideline-402. 9 A day before the commencement of the study, the skin on the dorsal area and the trunk were clipped. Five hundred milligrams (a dose equivalent to 2,000 mg/kg BW) of LI13019F1 was moistened in distilled water and applied to the skin with the help of a surgical gauze patch secured with a bandage. The gauze was removed after 24 hours, and the skin was wiped off using distilled water-soaked cotton to remove the remaining test item. Animals were observed daily for mortality/morbidity and clinical signs of toxicity at 30 minutes, 1 hour, 2 hours, 3 hours, and 4 hours after the test item administration on day 1 and once a day for the subsequent 14 days. Their body weights were measured once a week. The vital organs and tissues were subjected to gross pathological examinations following CO2 euthanasia on day 15.
Acute Dermal Irritation Study in Rabbits
The acute dermal irritation test was conducted as per OECD guideline-404. 10 LI13019F1 was applied to the shaved skin on the dorsal trunk of healthy young adult male New Zealand rabbits. Five hundred milligrams of LI13019F1 was moistened with distilled water and was applied to the shaved skin (6 cm × 6 cm) and the application area was covered with a gauze patch secured with a bandage. The patch was removed after 4 hours. Signs of skin reactions and irritation effects were assessed at 1, 24, 48, and 72 hours after removal of the gauze patch. The test was performed on one animal and then the same procedure was repeated on 2 more animals. The untreated skin of the adjacent areas of each animal served as controls. After 72-hour observation, the animals were euthanized using sodium thiopentone. The vital organs and the tissues were examined for gross pathological observations.
Acute Eye Irritation Study in Rabbits
The acute eye irritation study on healthy young adult male New Zealand white rabbits (2.0-2.4 kg) followed the OECD test guideline-405. 11 Three animals were selected, and both eyes were examined 24 hours before LI13019F1 application. Briefly, a neat sample of 100 mg LI13019F was applied into the conjunctival sac of the left eye. The eyelids were then gently held together for a few seconds to prevent loss of the test item. The untreated right eye served as controls. After 24 hours, both eyes of the animal were washed with distilled water. After 1, 24, 48, and 72 hours of the test item removal, the conjunctiva, iris, and cornea of both eyes were examined and scored according to the Draize method. 12 The test was performed on one animal, and after 72 hours, the same procedure was repeated on 2 more animals. After 72 hours of observation, the animals were euthanized and examined as described in the previous section.
Twenty-Eight-Day Repeated Dose Oral Toxicity Study
A 28-day repeated dose subchronic toxicity study evaluated the safety of LI13019F1 as per OECD Guidelines 407. 13 Briefly, 7-week-old Wistar rats were randomized into 6 groups (n = 10; 5 males and 5 females per group). The groups were as follows: vehicle control (G1), 250 mg/kg (G2), 500 mg/kg (G3), 1,000 mg/kg (G4) of LI13019F1; 2 recovery groups, namely, vehicle control (G1R) and 1,000 mg/kg LI13019F1 (G4R). The animals received either 0.5% (wt/vol) Na-CMC as vehicle control or LI13019F1 through oral gavage for 28 days. The animals in G1, G2, G3, and G4 were subjected to CO2 euthanasia on day 29. The animals in the recovery groups (ie, G1R and G4R) continued their in-life phase for an additional 14 days with the regular diet feed, without the test item or vehicle supplementation. The animals were examined for morbidity/mortality and clinical signs. The body weight of the animals was recorded during the treatment and the recovery period. After 16- to 18-hour fast, blood samples from all animals in the treatment and recovery groups were collected by retro-orbital puncture under isoflurane anesthesia.
Serum biochemistry and hematology assessments were parts of the safety evaluation of LI13019F1. The serum biochemistry parameters included glucose, blood urea nitrogen, urea, creatinine, total cholesterol, triglycerides, total bilirubin, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, total protein, albumin, calcium, phosphorus, sodium, potassium, and chloride. The hematology parameters were total leukocyte count, red blood cell count, hemoglobin, hematocrit, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, mean corpuscular volume, differential count, platelet count, and clotting time.
Serum biochemical parameters and hematological parameters were measured using an automated analyzer clinical chemistry analyzer (ILab Aries, Bergamo, Italy) and a hematology analyzer ADVIA 2120 (Siemens Healthcare Private Limited, Munich, Germany).
After CO2 euthanasia, the animals’ vital organs and tissues were weighed on an electronic balance with 0.01 g accuracy (Mettler Toledo, Columbus, Ohio). The relative organ weights were calculated by comparing the organ weight with the body weight of the respective animal on the day of sacrifice. Small pieces of the vital organs such as liver, kidney, heart, lung, pancreas, testes, and ovaries were fixed in phosphate-buffered 10% formalin for histopathological examinations. The paraffin-embedded tissue sections of 5-µm thickness were stained with hematoxylin and eosin. The stained tissue sections were examined under a light microscope (Zeiss AXIO Scope.A1; Carl Zeiss AG, Munich, Germany).
Bacterial Reverse Mutation Assay
The Ames bacterial reverse mutation test was conducted according to the OECD test guideline 471. 14 The assay was carried out on a panel of Salmonella typhimurium strains (TA98, TA100, TA1535, and TA1537) and Escherichia coli (WP2 uvrA/pKM101) strain with or without S9 metabolic activation. 15,16 The bacterial cells were incubated with increasing concentrations of LI13019F1 in 100, 266, 707, 1,880 and 5,000 μg per plate, in triplicate. The bacterial suspensions were mixed with an overlay agar and plated immediately onto the minimal agar medium. The plates were kept at 37°C ± 1°C for 48 to 72 hours. The number of the revertant colonies on the LI13019F1 treated plates and on the vehicle control plates were counted. The strain-specific mutagenic compounds were run in parallel as positive controls.
Mouse Bone Marrow Erythrocyte Micronucleus Assay
The in vivo erythrocyte micronucleus assay in the mouse bone marrow was conducted following the OECD Test Guideline 474. 17 Healthy mice (aged 6-10 weeks) were randomly allocated into 5 groups (n = 10; 5 males and 5 females per group). The control group animals received 0.5% wt/vol CMC-Na as the vehicle. LI13019F1 was administered orally at 500, 1,000, and 2,000 mg/kg BW. The animals in the control and LI13019F1 groups received 2 oral doses at an interval of 24 hours. Only the animals in the positive control group received a single intraperitoneal administration of cyclophosphamide (40 mg/kg BW) on the second day, 24 hours before euthanasia. The bone marrow samples were smeared on randomly coded slides. The slides were stained with Giemsa and examined under a 20× objective of an ECLIPSE E200 light microscope (Nikon Corp, Tokyo, Japan). Polychromatic erythrocytes (PCEs), normochromatic erythrocytes (NCE), and micronucleated polychromatic erythrocytes (MNPCE) were counted in each bone marrow sample. A total of 4,000 PCEs was scored in each sample, and the frequency of MNPCE was expressed as a percentage. Besides, the number of PCE counted in 1,000 total number of erythrocytes (TE = PCE + NCE), and the frequency of PCE was expressed as a ratio between PCE and TE.
Statistical Analysis
The collected data were organized and computed using GraphPad Prism software version 6 (GraphPad Software, La Jolla, California). One-way analysis of variance followed by Dunnett post hoc test was employed to determine the difference between dosage groups and the control group. Student t-test analyzed the data from the recovery groups for comparison. All values expressed as mean (standard deviation), the confidence level set at 95% with a statistical significance of P < 0.05.
Results
Acute Oral Toxicity
Administration of LI13019F1 at 2,000 mg/kg BW did not cause any mortality/morbidity. Also, the animals did not show any clinical signs of toxicity during the 15-day follow-up period. They did not show abnormal changes in their body weights during the observation period. A gross pathological examination also did not reveal any abnormalities. Together, these findings suggested that the median oral lethal dose (LD50) of LI13019F1 was at least 2,000 mg/kg BW in female Wistar rats.
Acute Dermal Toxicity
The experimental animals did not exhibit any signs of gross toxicity, dermal irritation, adverse pharmacological effects, or any behavioral abnormality. These findings suggested that the dermal LD50 of LI13019F1 was at least 2,000 mg/kg BW in Wistar rats of either sex.
Acute Dermal Irritation
Dermal application of LI13019F1 did not show any cutaneous reactions in the New Zealand white rabbits 24, 48, and 72 hours after patch removal. Under the specified experimental conditions, the acute dermal irritation index for LI13019F1 is 0. According to the Harmonized Integrated Classification System, these findings suggest that LI13019F1 is a “nonirritant” to the rabbit skin. 18
Acute Eye Irritation
After 1 hour of a single application, LI13019F1-treated rabbits exhibited conjunctival redness. The effect was reversible within 72 hours of the test product application. No signs of corneal opacity or iritis observed due to the LI13019F1 application. As per the Harmonized Integrated Classification System, these findings suggest that LI13019F1 is a “nonirritant” to the rabbit eyes. 12
Repeated Dose 28-Day Oral Toxicity Study
Mortality and clinical signs
The animals in the primary and recovery groups of the study did not show treatment-related clinical signs of toxicity. Mortality was limited to one male rat in the high-dose group G4 (1,000 mg/kg BW), which died on day 19 of the treatment period. The signs of mortality suggested that the animal died due to gavage error. The remaining animals in the primary groups survived through observation period. In this study, all observations in the G4 male group are calculated considering the group size of 4 (n = 4), while the remaining groups are of n = 5.
Body weight and feed consumption
Tables 1 and 2 present the mean body weights and changes in the body weights of the male and female rats following supplementation of LI13019F1. The male and female rats did not show treatment-related changes in the overall body weight. In the recovery groups, after completion of the first week of the study, the G4R male rats showed a significant reduction in their body weight compared with the G1R animals. However, the G4R animals regained their body weights in the following weeks of the study and the difference were not statistically significant from the G1R animals.
Effect of LI13019F1 on Body Weight of Male Wistar Rats in a 28-Day Repeated Dose Oral Toxicity Study Including a Recovery Period of 14 Days.
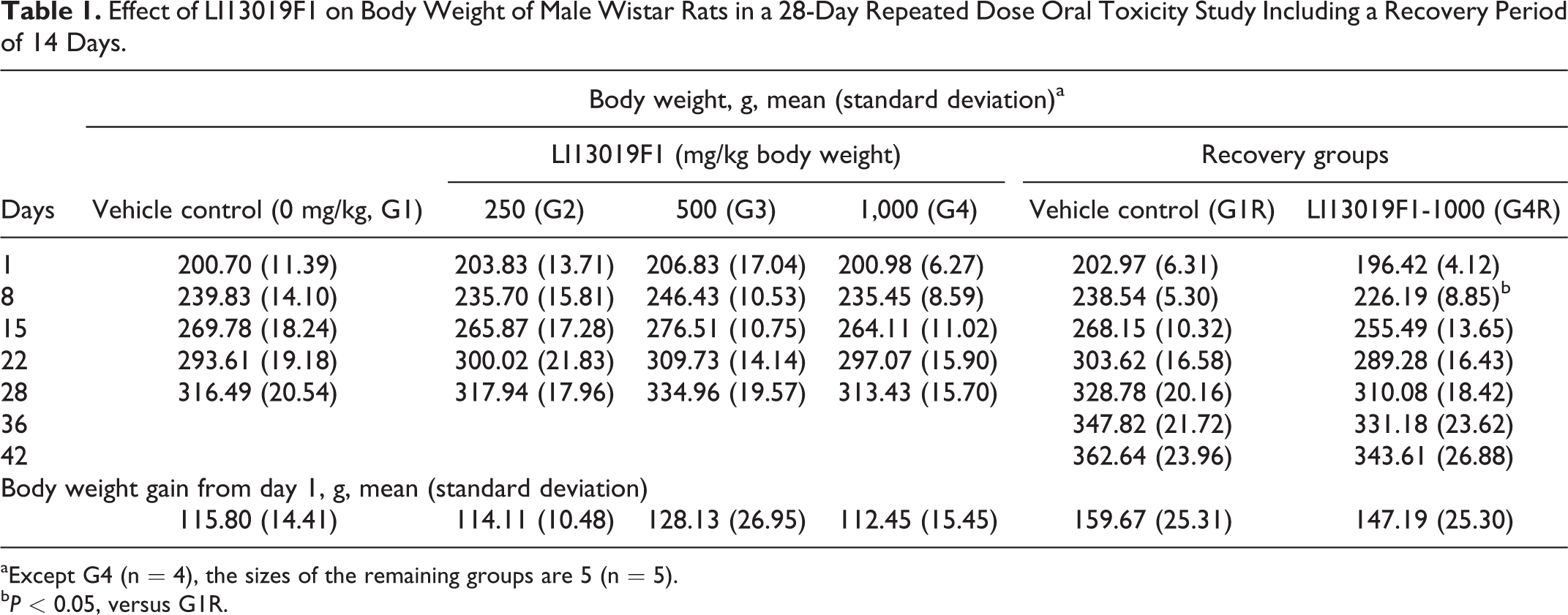
aExcept G4 (n = 4), the sizes of the remaining groups are 5 (n = 5).
bP < 0.05, versus G1R.
Effect of LI13019F1 on Body Weight of Female Wistar Rats in a 28-Day Repeated Dose Oral Toxicity Study Including a Recovery Period of 14 Days.a
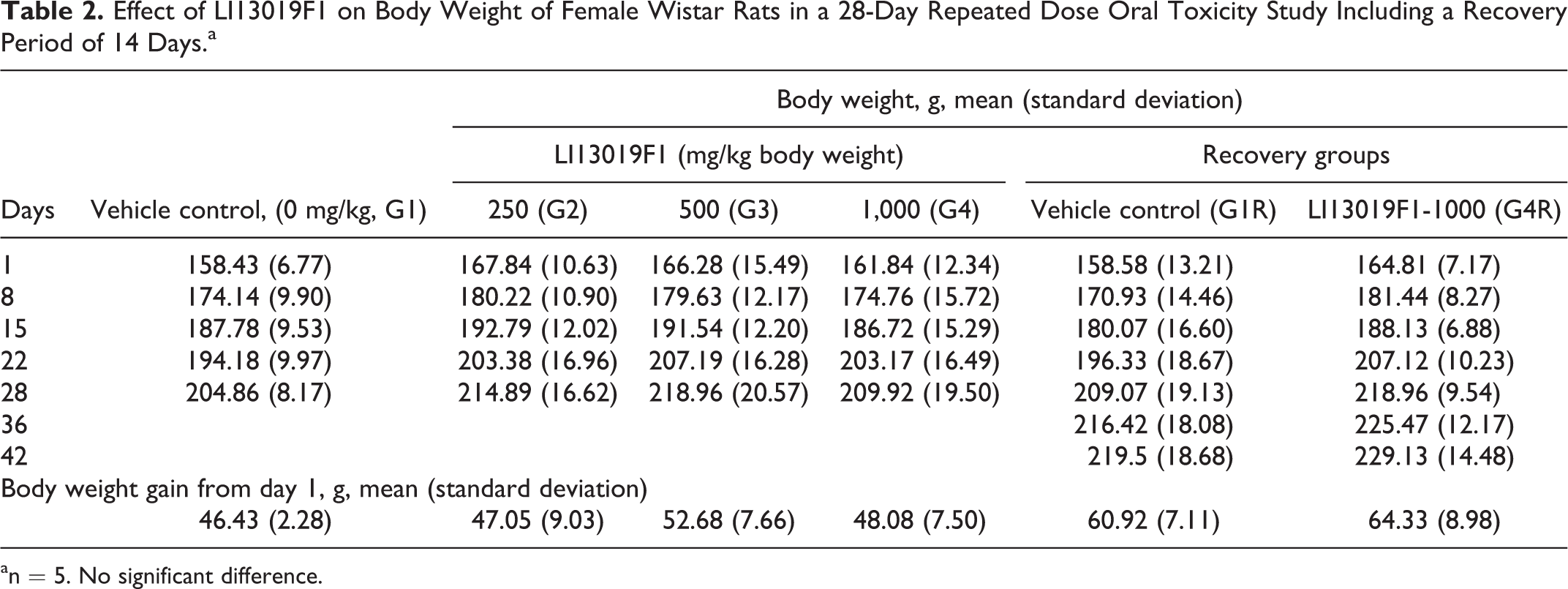
an = 5. No significant difference.
Tables 3 and 4 present the weekly feed consumption by the male and female rats, respectively. The weekly feed consumption data showed some significant variations in the primary and recovery groups of the male rats (Table 3). However, the total feed consumption by the male animals in the treatment groups is not significantly different from the respective control animals (Table 3). The total and weekly feed consumption by the female rats in the primary and recovery groups was not statistically different from the respective controls (Table 4).
Effect of LI13019F1 on Feed Consumption by Male Wistar Rats.
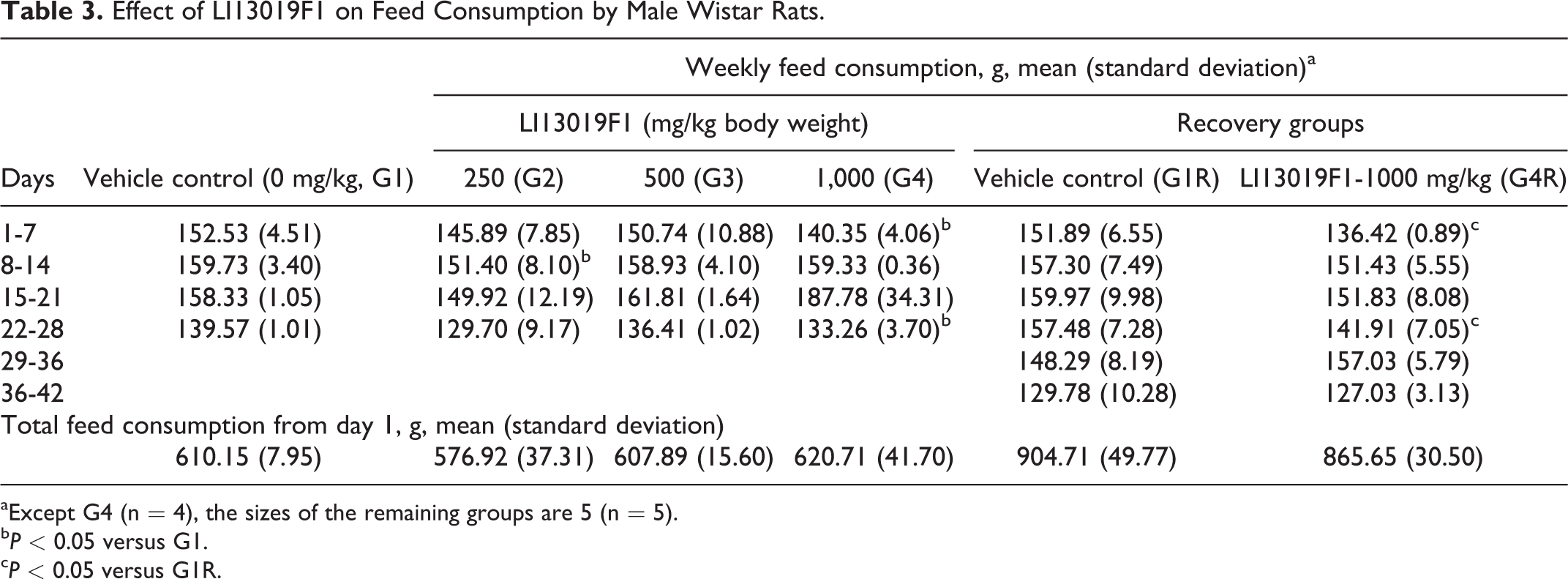
aExcept G4 (n = 4), the sizes of the remaining groups are 5 (n = 5).
bP < 0.05 versus G1.
cP < 0.05 versus G1R.
Effect of LI13019F1 on Feed Consumption by Female Wistar Rats.a
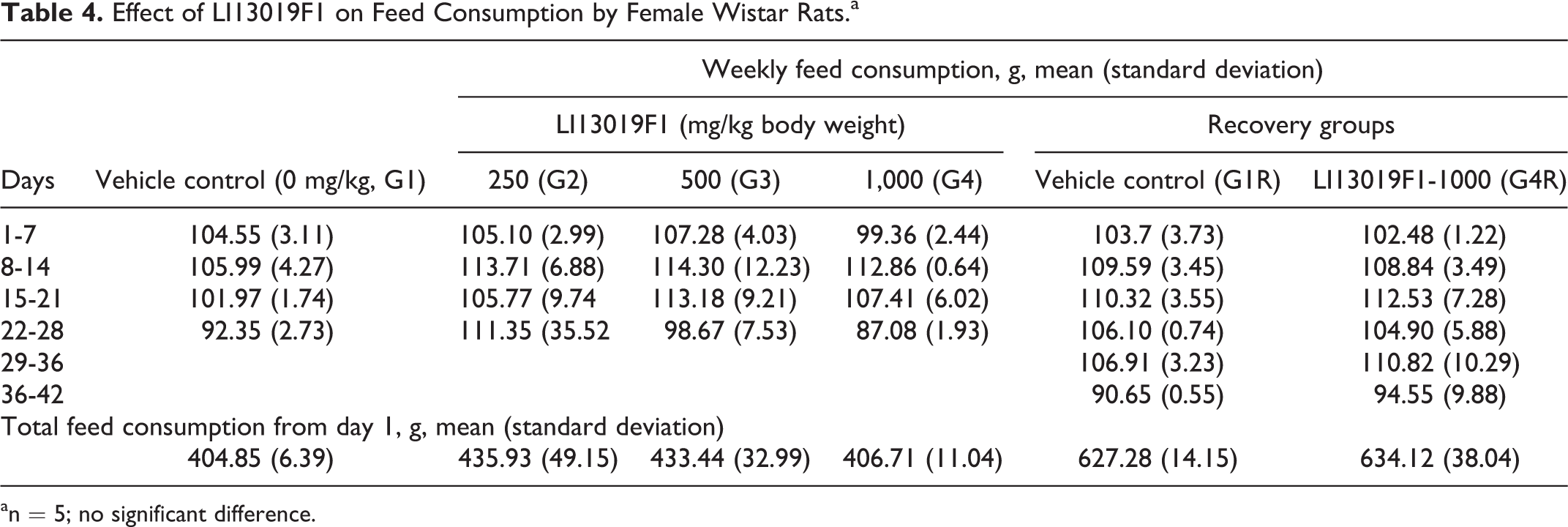
an = 5; no significant difference.
Hematology and clinical chemistry
Tables 5 -8 present observations on various parameters of hematology and clinical chemistry in the experimental male and female animals. The G3 and G4 males showed a significant increase in platelet count (Table 5), and G2 females showed a substantial decrease in lymphocyte count (Table 6), in comparison with the respective sex-matched controls. However, the data were within the normal physiological range and not related to the treatment doses.
Effect of LI13019F1 on Hematology Parameters in Male Wistar Rats.a
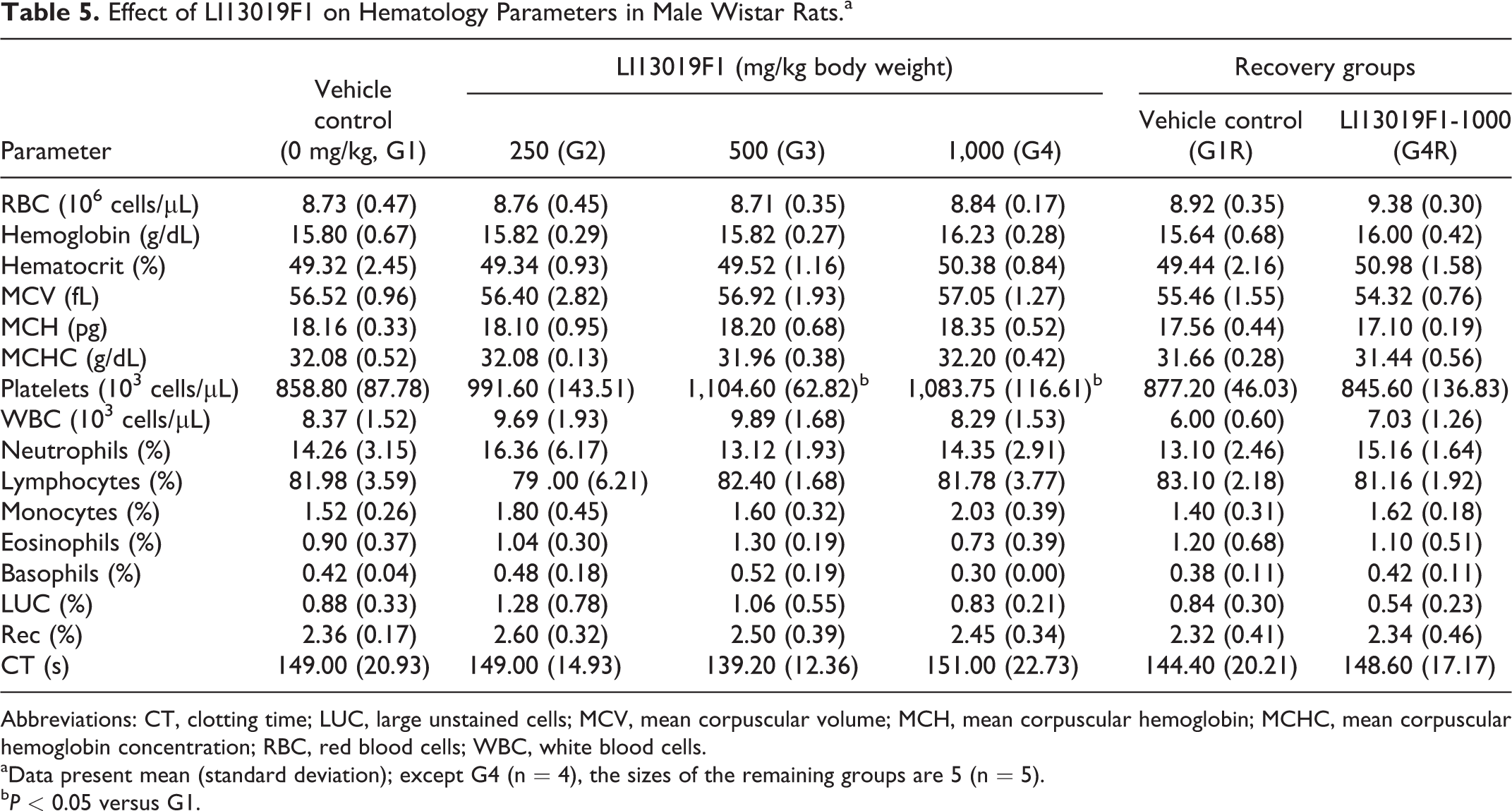
Abbreviations: CT, clotting time; LUC, large unstained cells; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RBC, red blood cells; WBC, white blood cells.
aData present mean (standard deviation); except G4 (n = 4), the sizes of the remaining groups are 5 (n = 5).
bP < 0.05 versus G1.
Effect of LI13019F1 on Hematology Parameters in Female Wistar Rats.a
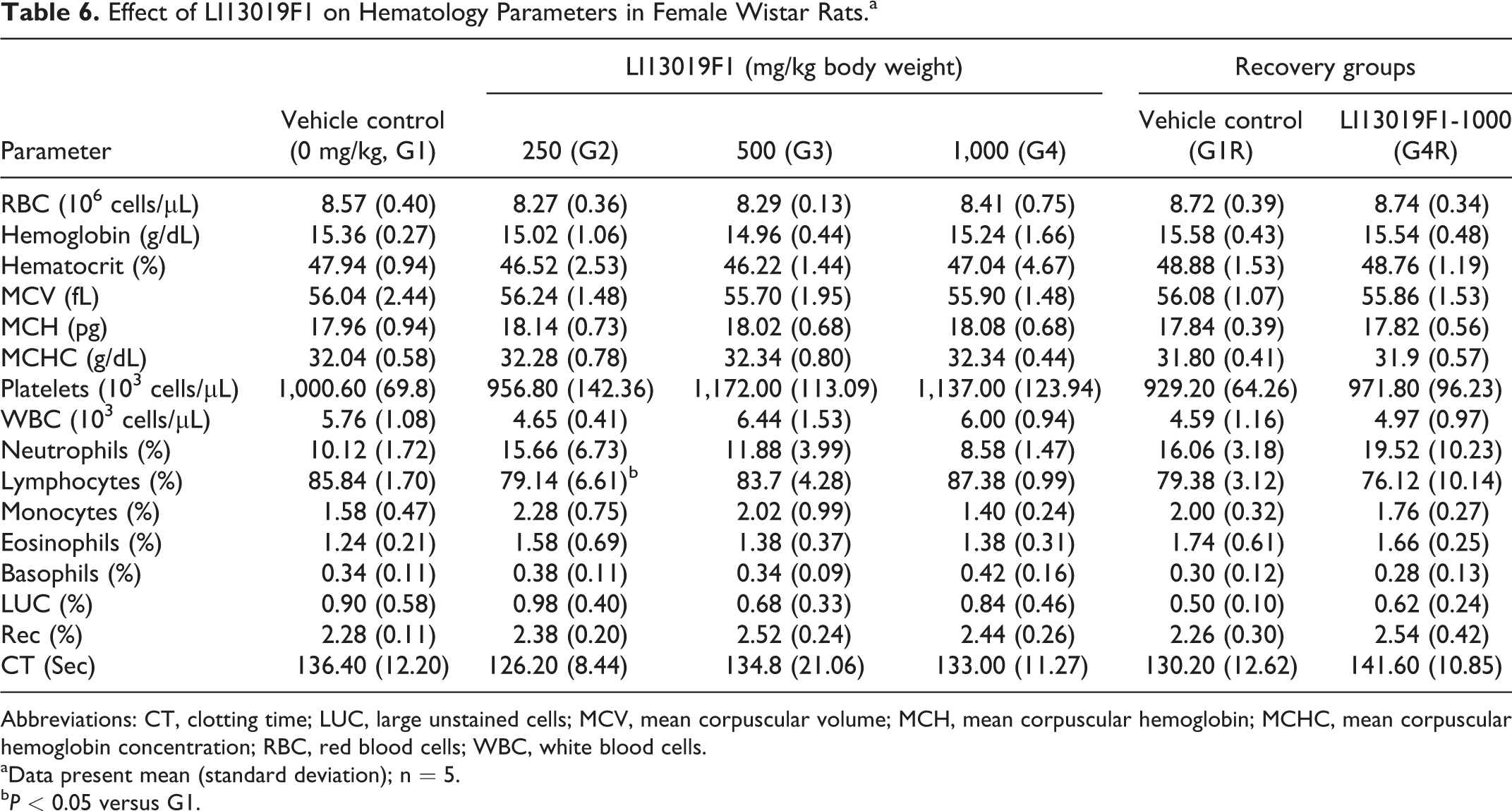
Abbreviations: CT, clotting time; LUC, large unstained cells; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RBC, red blood cells; WBC, white blood cells.
aData present mean (standard deviation); n = 5.
bP < 0.05 versus G1.
Effect of LI13019F1 on Clinical Chemistry Data in Male Wistar Rats.a
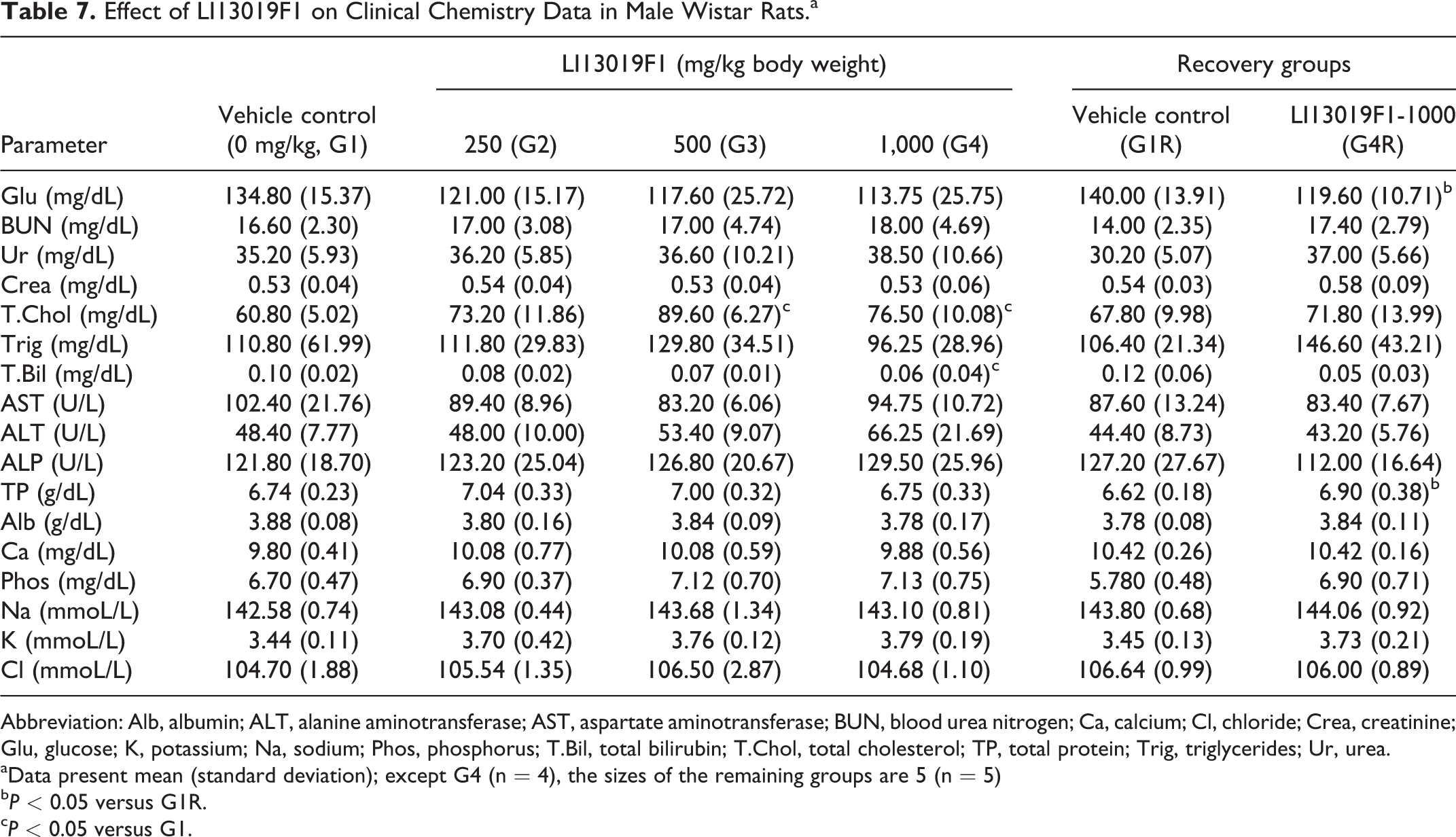
Abbreviation: Alb, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Ca, calcium; Cl, chloride; Crea, creatinine; Glu, glucose; K, potassium; Na, sodium; Phos, phosphorus; T.Bil, total bilirubin; T.Chol, total cholesterol; TP, total protein; Trig, triglycerides; Ur, urea.
aData present mean (standard deviation); except G4 (n = 4), the sizes of the remaining groups are 5 (n = 5)
bP < 0.05 versus G1R.
cP < 0.05 versus G1.
Effect of LI13019F1 on Clinical Chemistry Data in Female Waster Rats.a
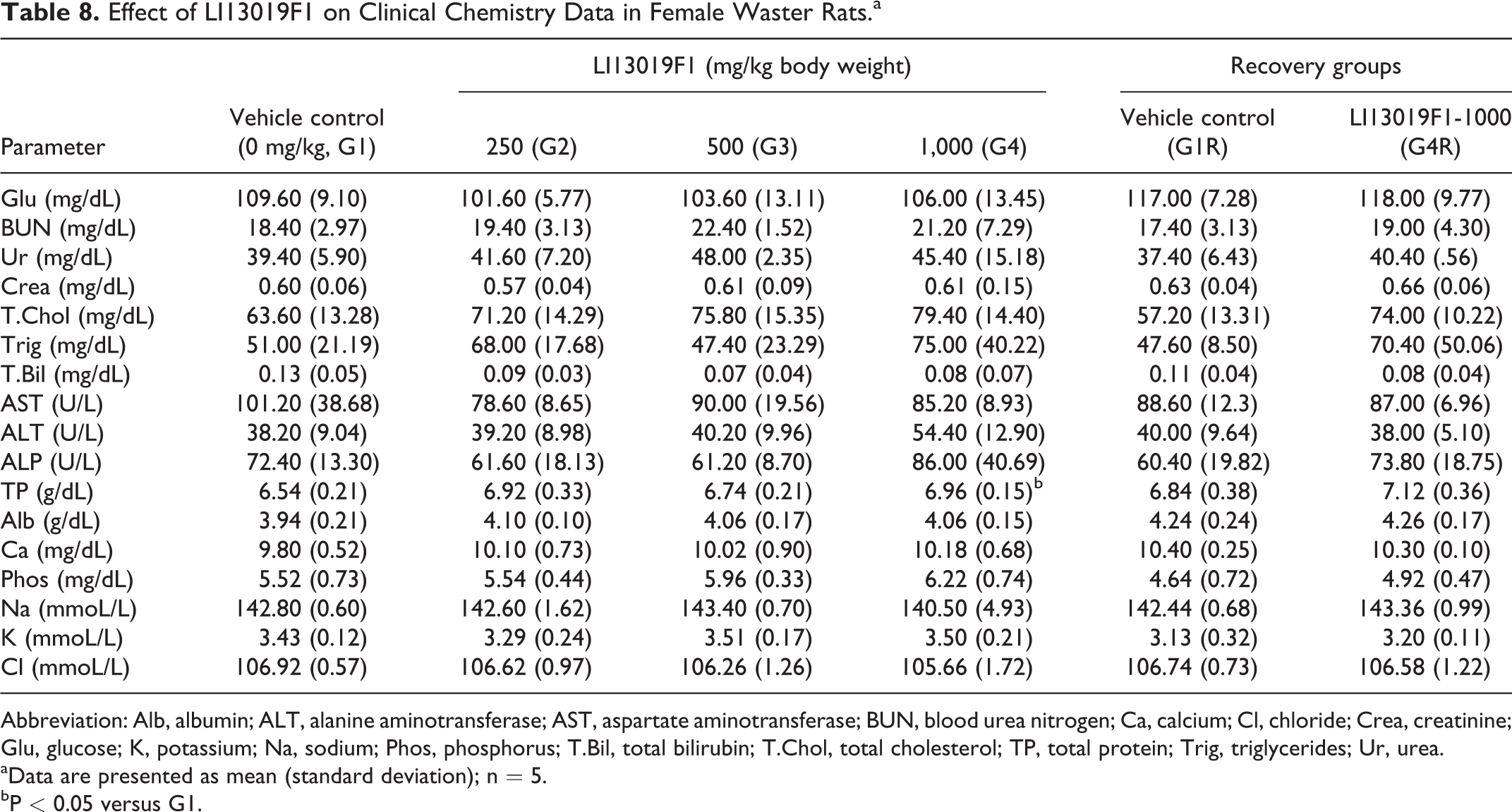
Abbreviation: Alb, albumin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Ca, calcium; Cl, chloride; Crea, creatinine; Glu, glucose; K, potassium; Na, sodium; Phos, phosphorus; T.Bil, total bilirubin; T.Chol, total cholesterol; TP, total protein; Trig, triglycerides; Ur, urea.
aData are presented as mean (standard deviation); n = 5.
bP < 0.05 versus G1.
The male rats in G3 (P < 0.001) and G4 (P < 0.05) showed a significant increase in serum total cholesterol level, compared with the control animals. Also, the male rats in G4 showed a significant decrease (P < 0.05) in total bilirubin in comparison with the control rats (Table 7). The G4R male rats showed a significant increase in total protein and a substantial reduction in glucose levels in comparison with the respective control rats (Table 7). LI13019F1 supplementation did not show any significant changes in the clinical chemistry parameters of the female rats allocated in the primary and recovery groups when compared with the respective controls (Table 8).
Organ weights
Tables 9 and 10 present the absolute organ weights of the male and female rats belonging to the primary and recovery groups of the study. The male G3 rats showed an increase (P < 0.05) in the absolute weight of the kidneys, compared with the controls (Table 9). The absolute weights of the remaining vital organs of LI13019F1-supplemented animals in the primary groups (G2, G3, and G4) and in the recovery group (G4R) were not statistically different, compared with the respective controls (Tables 9 and 10). The relative organ weights of the male and female rats in the LI13019F1-supplemented groups were not significantly different in comparison with the control animals (Tables 9 and 10).
Effect of Oral Administration of LI13019F1 on Absolute and Relative Organ Weights of Male Wistar Rats.a
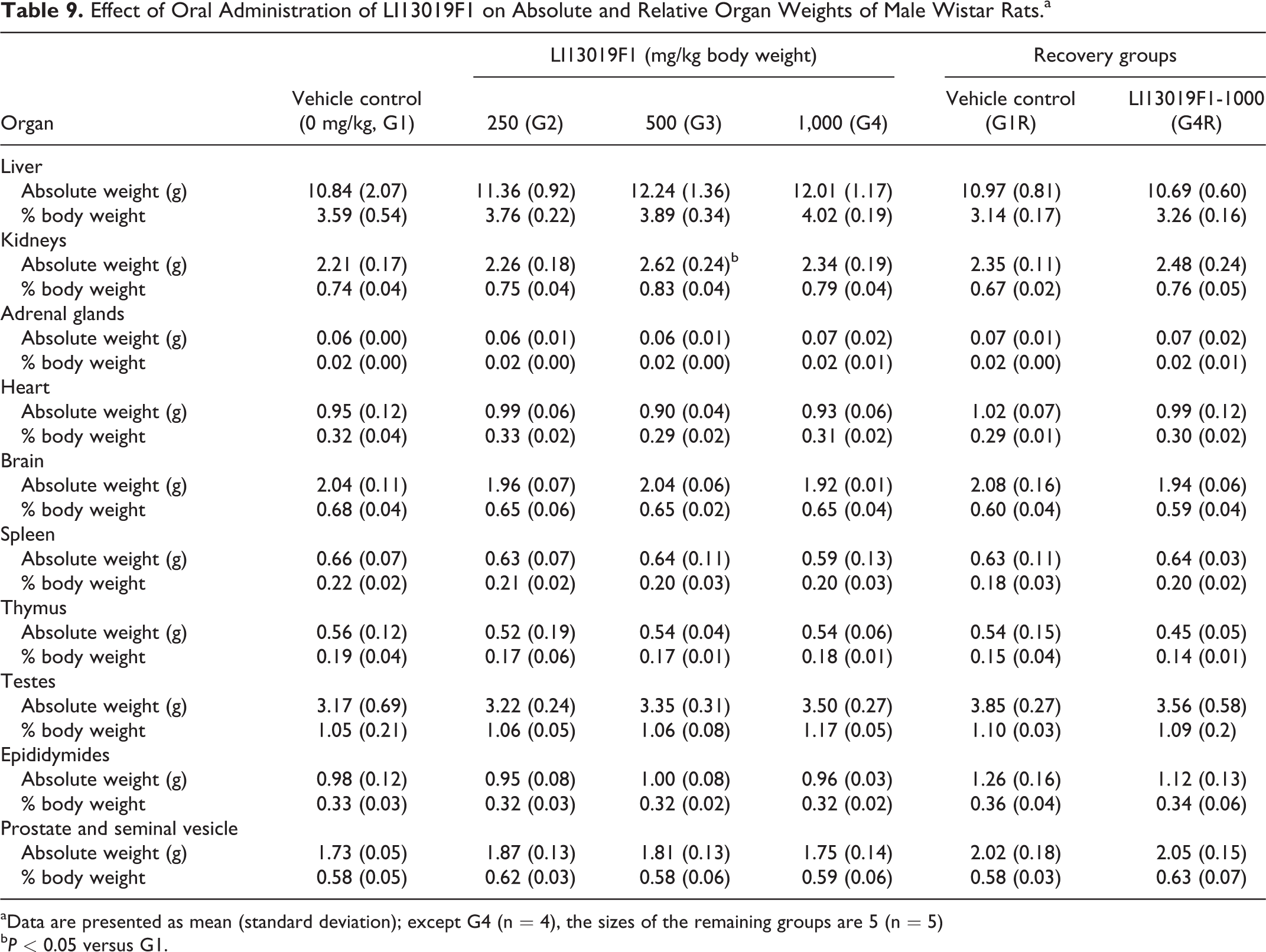
aData are presented as mean (standard deviation); except G4 (n = 4), the sizes of the remaining groups are 5 (n = 5)
bP < 0.05 versus G1.
Effect of Oral Administration of LI13019F1 on Absolute and Relative Organ Weights of Female Wistar Rats.a
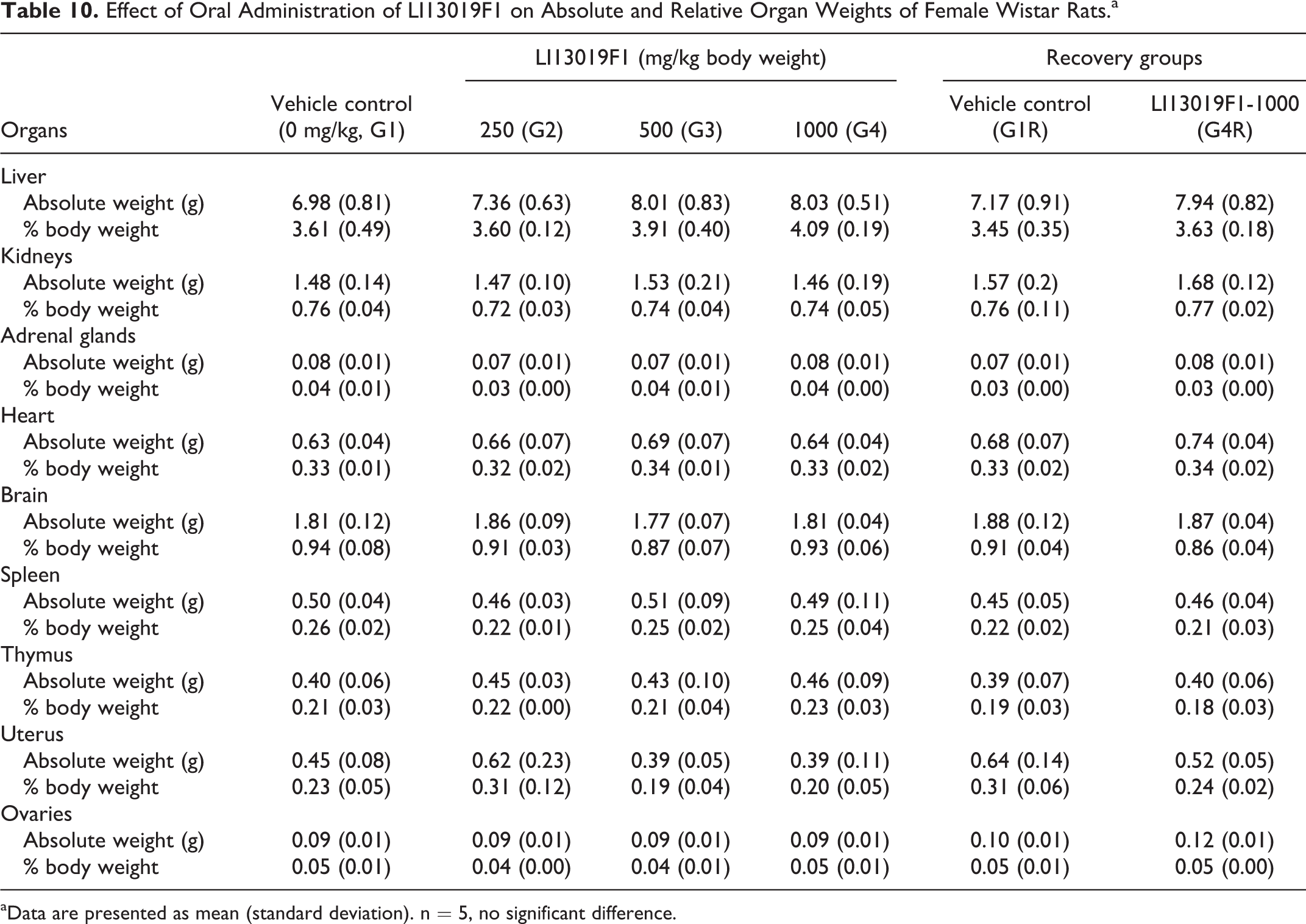
aData are presented as mean (standard deviation). n = 5, no significant difference.
Gross pathology and histopathology
The external and internal pathological examinations of all terminally euthanized animals (allocated in G1-G4, G1R, and G4R) showed no abnormalities following the treatment and the recovery periods. One male rat in the high-dose group (G4) of LI13019F1 died after 18 days of treatment. It showed a reddish discharge from the nose, and the thymus and lungs were pale. The histopathological examinations also revealed that the animal suffered from alveolar hemorrhage/disruption in the lungs and mucosal atrophy in the stomach. Tables 11 and 12 summarize the histological observations on the male and female animals, respectively.
Histopathological Observations on the Vital Organs of Male Wistar Rats After 28 Days Oral Supplementation of LI13019F1.a
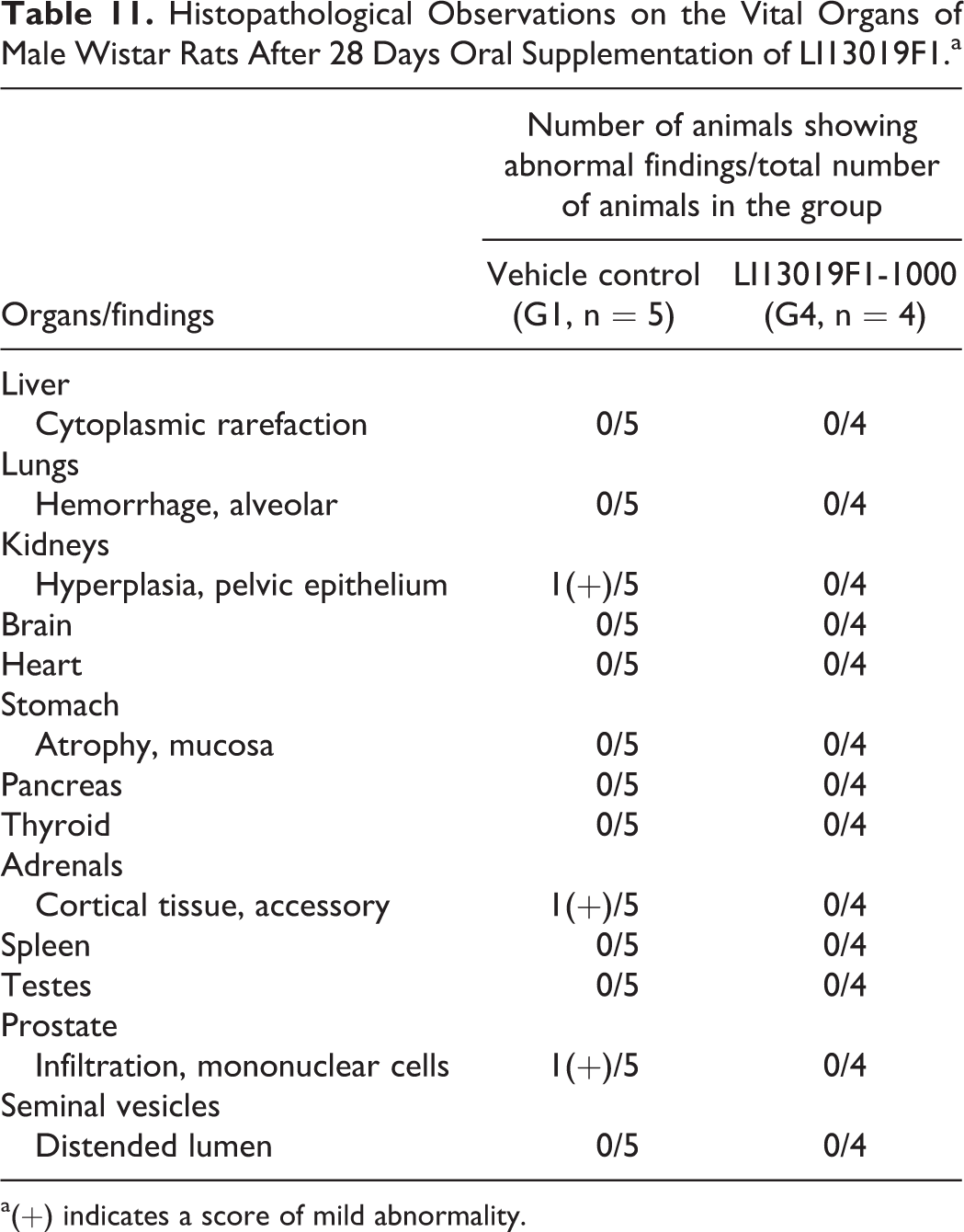
a(+) indicates a score of mild abnormality.
Histopathological Observations on the Vital Organs of Female Wistar Rats After 28 Days Oral Supplementation of LI13019F1.a
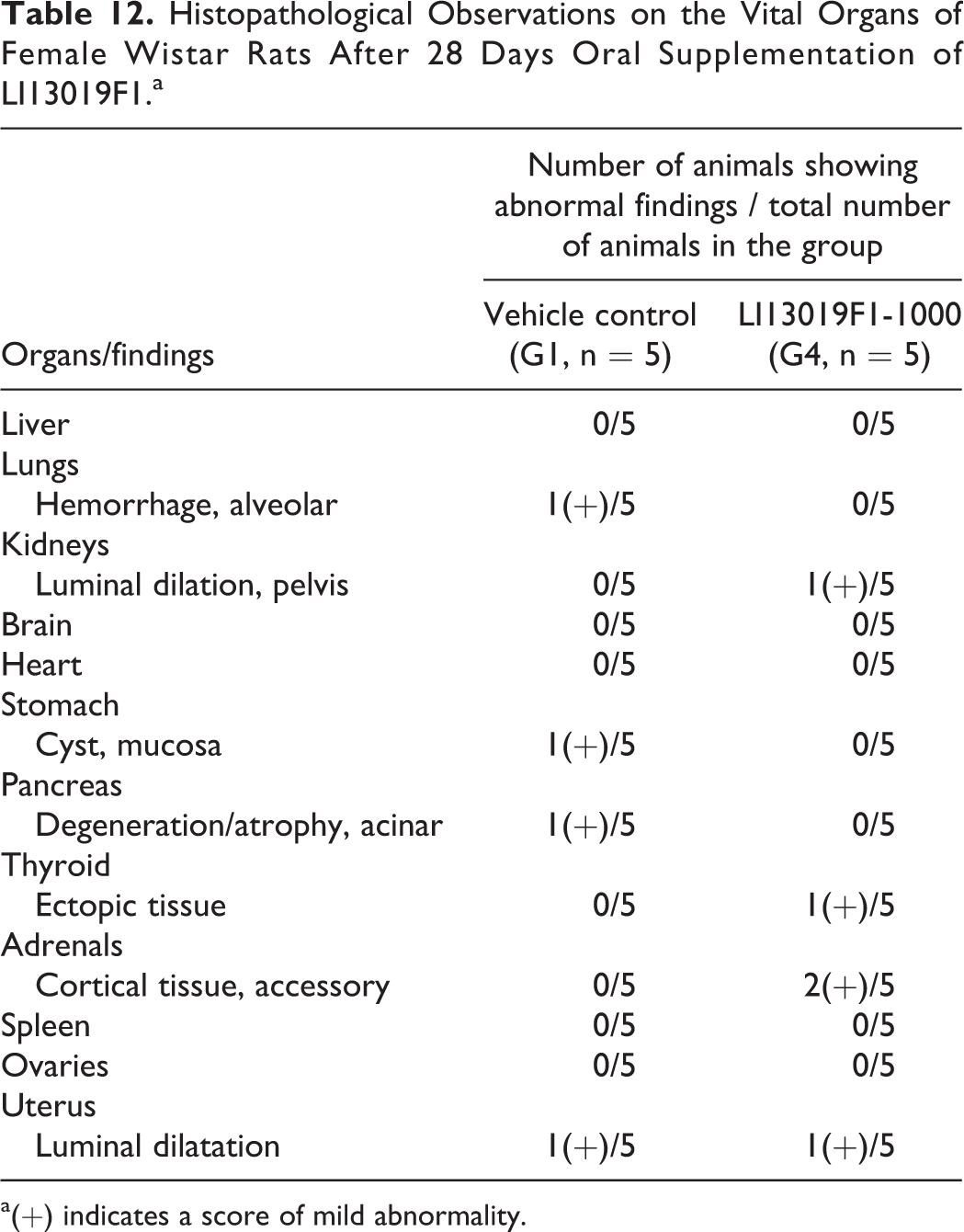
a(+) indicates a score of mild abnormality.
The microscopic examinations did not show any significant histopathological changes in the hematoxylin-eosin-stained sections of the vital organs of the survived rats at high-dose LI13019F1-treated groups (data not shown).
Bacterial Reverse Mutation Assay
Table 13 presents data from the mutagenicity assay. The assay tested up to 5,000 µg/plate LI13019F1, and even the highest dose did not show an increased number of revertant colonies of any of the tested bacterial strains in the presence or absence of metabolic activation. LI13019F1 did not precipitate on the experimental plates. The culture plates containing the strain-specific mutagenic compounds, served as positive controls, showed typically increased number of revertant colonies in the presence or absence of metabolic activation, as expected. These observations suggest that LI13019F1 lacks mutagenic potential.
Genotoxicity Potential of LI13019F1 in Bacterial Reverse Mutation Assay.
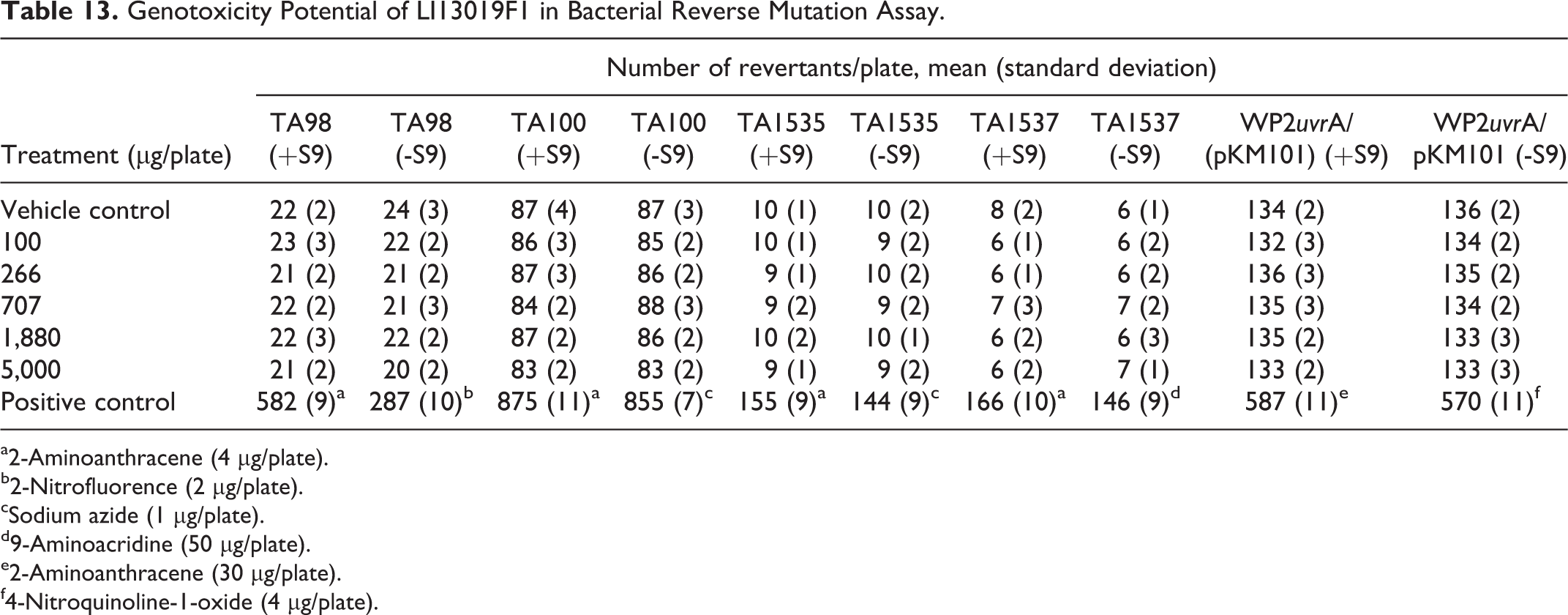
a2-Aminoanthracene (4 µg/plate).
b2-Nitrofluorence (2 µg/plate).
cSodium azide (1 µg/plate).
d9-Aminoacridine (50 µg/plate).
e2-Aminoanthracene (30 µg/plate).
f4-Nitroquinoline-1-oxide (4 µg/plate).
Mouse Bone Marrow Erythrocytes Micronucleus Assay
Oral doses of LI13019F1 up to 2,000 mg/kg BW did not alter the number of immature PCE in male and female mice (Table 14). Also, the frequencies of the MNPCEs did not alter significantly in the LI13019F1-supplemented animals’ bone marrow when compared with vehicle control group (Table 14). The cyclophosphamide-treated animals showed a significant increase in MNPCEs, compared with the vehicle control. Overall, the observations indicate that LI13019F1 is not cytotoxic or genotoxic.
Effect of LI13019F1 on Micronucleus Frequency in Mouse Bone Marrow Erythrocytes.a
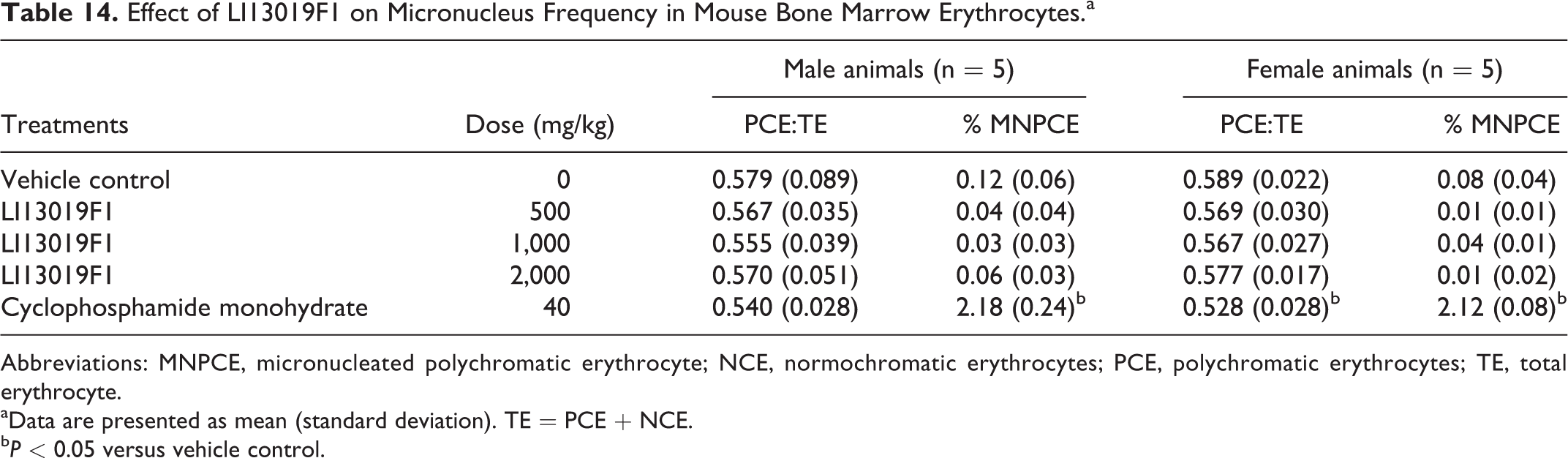
Abbreviations: MNPCE, micronucleated polychromatic erythrocyte; NCE, normochromatic erythrocytes; PCE, polychromatic erythrocytes; TE, total erythrocyte.
aData are presented as mean (standard deviation). TE = PCE + NCE.
bP < 0.05 versus vehicle control.
Discussion
Use of B serrata gum resin extract is a popular alternative strategy for the management of various inflammatory diseases, including osteoarthritis and chronic bowel diseases. 3,19 -23 Earlier, we and others have shown the broad-spectrum safety of different preparations of Boswellia resin extracts in series of preclinical models. 24 -26 In addition to the long history of human use, a series of human clinical studies conducted on various preparations of B serrata gum resin extracts have firmly established a broad safety profile of the resin extracts. 19 -23 The US Food and Drug Administration has allowed its use as a food additive. 27 In spite of lack of safety concerns on various preparations of B serrata resin extracts, we explored a broad-spectrum safety profile of LI13019F1, a new formulation containing a combination of selective acidic and nonacidic fractions of the gum resin.
In the present study, the observations from the acute oral toxicity experiment suggest that the oral median LD50 of LI13019F1 would be at least 2,000 mg/kg BW in the rats. The dermal toxicity and primary eye irritation studies suggest that LI13019F1 is nonirritating to the skin and eyes.
The observations from the present 28-day repeated dose toxicity study revealed that oral supplementation of LI13019F1 didn’t cause meaningful dose-dependent changes in rats of either sex. The animals did not show any physiological abnormalities after 14 days of the recovery period. Potentially toxic substances cause reduction in body weight gain. 28 In the present 28-day toxicity study, all animals in the treatment groups continued gaining their body weights throughout the study. Also, the amount of food consumption by the treated animals was not significantly different from the control animals, during the study period. These suggest that LI13019F1 supplementation did not affect the healthy growth and metabolism of the rats.
Popularly, nonsteroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen, naproxen, are used as a pain management strategy for symptomatic relief of osteoarthritis. Use of traditional NSAIDs causes serious gastrointestinal toxicity, renal toxicity, and adversely affects platelet function. 29,30 In preclinical toxicity studies, oral administrations of ibuprofen, indomethacin, and vitacoxib showed significant alterations in serum biomarkers along with histological changes associated with liver and kidney toxicity in rats. 31 -33 In the present 28-day repeated dose toxicity study, LI13019F1 supplementation did not show significant changes in the biochemical parameters associated with liver or renal toxicity. Besides, gross and microscopic examinations revealed that LI13019F1-supplemented rats had normal architecture of the liver, presence of the sufficient number of glomeruli and normal tubules in the kidney, presence of normal glandular structures, presence of mucosal layer, and absence of lesion in the stomach. Together, these findings suggest that the oral administration of LI13019F1 did not cause toxicity in the liver, kidneys, and stomach.
It is of note that one male rat in the LI13019F1-1000 group died on the 19th day of the study, due to gavage error. We excluded the deceased animal’s data from the calculations of all evaluations in the study. We anticipate that the loss of animal impacted the statistical analyses of the data but believe that this incidence is not related to the toxicity of the test product. Although some animals showed significant changes in some hemato-biochemical parameters, those variations were within the physiological limits. 34 Also, the gross pathological observations of the vital organs and their microscopic examinations suggest that oral treatment with this Boswellia product did not cause systemic toxicity in the experimental rats. Together, these findings establish that the “no observed adverse effect level” of LI13019F1 in rats of both sexes is 1,000 mg/kg body weight per day.
Furthermore, as a part of the broad range of safety profile of LI13019F1, we also evaluated the genotoxic potential of this novel preparation of Boswellia gum resin. Our observations demonstrate that LI13019F1 does not induce mutagenesis and also does not possess clastogenic or DNA damage potential. These observations suggest that LI13019F1 lacks genotoxic potential when consumed orally. Together, these data support the safety of the human oral ingestion of LI13019F1 when consumed at a dose of not greater than 167 mg per kg BW.
Footnotes
Acknowledgments
The authors greatly acknowledge the management of Laila Nutraceuticals, Vijayawada, India, for sponsoring the present research. The authors are also thankful to the staff of the Pathology and Toxicology departments of Laila Nutraceuticals R&D Centre for their technical support.
Author Contributions
K.V.A. contributed to designing the study, contributed to raw data handling, contributed to statistical analyses, and drafted the manuscript. S.D. performed the experiment works and drafted the manuscript. E.K. helped in designing the study and reviewed the manuscript. T.G. contributed to the production and phytochemical characterization of the test item and reviewed the manuscript. K.S. analyzed the data, helped in data presentation, and critically revised the manuscript. All authors read and approved the contents of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.V.A., S.D., T.G., and K.S. are employees of Laila Nutraceuticals R&D Center, Vijayawada, India. E.K. is a faculty in an independent institution, Andhra University, Visakhapatnam, India.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Laila Nutraceuticals, India, sponsored the study. The authors K.V.A., S.D., T.G., and K.S. received financial support for the research.
