Abstract
N-nitrosodimethylamine (NDMA) is a toxicant found in foods and drinking water. Several synthetic agents used in alleviation of NDMA toxicity have been associated with serious side effects. Therefore, a safe and less toxic agent is desirable. In this study, betulinic acid (BA), a triterpenoid antioxidant, is proposed as a better and alternative agent to modulate NDMA-induced toxicity. Twenty-four Wistar rats were assigned into four groups of six rats each and treated with normal saline (control), BA (25 mg/kg), NDMA (5 mg/kg) and (BA + NDMA). BA was given by oral gavage for 14 consecutive days, while NDMA was administered intraperitoneally on days 7 and 12. Results showed that administration of NDMA significantly (p < 0.05) elevated the activities of serum alanine aminotransferase (ALT), aspartate aminotransferase and gamma-glutamyl transferase by 51%, 48% and 81%, respectively. Also, NDMA intoxication significantly (p < 0.05) increased the levels of serum urea and creatinine by 64% and 82%, respectively, and decreased urinary creatinine by 67%. In addition, administration of NDMA significantly (p < 0.05) increased the levels of hepatic and renal DNA fragmentation by 44% and 61%, respectively, relative to control. The number of micronucleated polychromatic erythrocytes (mnPCEs) in NDMA-treated rats (11.1 ± 2.6 mnPCE/1000PCE) was significantly higher than control (4.3 ± 1.1 mnPCE/1000 PCE). Immunohistochemistry revealed strong expressions of Bcl-2 and nuclear p53 in NDMA-intoxicated rats. Interestingly, pretreatment with BA significantly (p < 0.05) ameliorated NDMA-induced changes in serum biochemical indices, mnPCEs, DNA fragmentation and expressions of Bcl-2 and p53 proteins. These findings suggest that BA protects against NDMA-induced toxicity via anti-oxidative and anti-apoptotic activities.
Introduction
N-nitrosamines such as N-nitrosodimethylamine (NDMA) are potent carcinogens present in foods, drinking water and ground water. 1 They are capable of generating deleterious radicals in cellular systems. 2 However, these compounds can also be formed in man as a result of nitrosation of nitrosable precursors in diets, drinking water and tobacco smoke. 3 In mammalian cells, NDMA is metabolically demethylated through a cytochrome P450-dependent α-hydroxylation reaction to form methyldiazohydroxide. 4,5 The methyldiazohydroxide is finally converted to methyldiazonium ion, which can methylate DNA and other macromolecules to initiate carcinogenesis. 6 During N-nitrosamines metabolism, nitric oxide is generated, 7 which may lead to the formation of reactive oxygen species (ROS) and initiation of tumourigenesis. 8
Betulinic acid (BA) (3β-hydroxy-lup-20(29)-en-28-oic acid) is a pentacyclic lupane – type triterpene, found in edible vegetables, fruits and nuts. It has been abundantly isolated from the bark of the birch tree (Betula pendula Roth). This phytochemical has been reported to exhibit a variety of biological activities including cytotoxicity against cells such as melanoma, leukaemia, carcinoma (cancers of head, neck, colon, liver, lungs, prostate, ovary or kidney), glioblastoma and neuroblastoma. 9 –13 Importantly, BA exhibited cytotoxicity against tumours that are resistant to conventional chemotherapeutic drugs. 10 Also, BA demonstrated selectivity for metastatic over non-metastatic cancer cells and selectivity for cancer cells over normal cells. 14,15 Safety evaluation studies by Sandberg et al. 16 and Pisha et al. 9 in rats and mice, respectively, at the dose range of 200–500 mg/kg found BA to be non-toxic to experimental rodents. In quest for better agent to ameliorate NDMA-induced toxicity and, the fact that there is paucity of information on the possible modulatory effects of BA against NDMA-induced toxicity. This study was designed, in addition to the above objectives, to unravel the mechanism of chemopreventive effects of BA against NDMA-induced toxicity.
Materials and methods
Chemicals
NDMA and BA were purchased from Sigma Chemical Co. (Saint Louis, Missouri, USA). Potassium chloride and sodium hydroxide were procured from British Drug House Chemical Ltd. (Poole, UK). Other chemicals were of analytical grade and purest quality available.
Experimental animals
Male Wistar rats weighing 110–130 g were obtained from Animal House of the Faculty of Basic Medical Sciences, University of Ibadan, Nigeria. They were housed in plastic cages and fed on rats’ pellets and given drinking water ad libitum. The rats were allowed to acclimatize for 7 days before the commencement of the experiment and kept at 12-h light/dark cycle and temperature of 29°C ± 2°C. The Faculty of Basic Medical Sciences, University of Ibadan Animal Ethics Committee, approved this study.
Study design
Twenty-four rats were assigned into four groups of six rats each. The first group (control) received drug vehicle (normal saline), second group (BA) received BA alone (25 mg/kg), third group (NDMA) received NDMA alone (5 mg/kg) and fourth group received (BA + NDMA). The BA and NDMA were dissolved/diluted with normal saline. BA was given by oral gavage for 14 consecutive days, while NDMA was given intraperitoneally on days 7 and 12 of the study.
Collection of blood and tissues
The rats were fasted overnight after last dose of BA on day 14 and then killed by cervical decapitation on day 15 under light ether anaesthesia. Blood was collected by ocular bleeding, allowed to clot and centrifuged at 3000 × g for 10 min to obtain serum. The femurs of the rats were collected immediately after killing and used for micronucleus assay. The liver and kidney were quickly excised and washed in ice-cold 1.15% potassium chloride solution to remove blood stains, dried and weighed. A section of these tissues were homogenized in Tris–ethylenediaminetetraacetic acid (EDTA; TE) buffer at pH 7.4 and centrifuged at 27,000 × g for 10 min and were used for DNA fragmentation assay, while another section cut from liver was stored in 10% formalin and used for immunohistochemical staining for Bcl-2 and p53 proteins.
Biochemical assays
Protein determination
Serum, kidney and liver protein levels were determined according to the method of Lowry et al. 17 using bovine serum albumin as standard.
Alanine and aspartate aminotransferases determination
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined using a combination of the methods of Mohun and Cook 18 and Reitman and Frankel. 19
Creatinine and urea determination
Serum creatinine and urea levels were estimated by the methods of Jaffe 20 and Talke and Schubert, 21 respectively.
Determination of γ-GT activity
Serum gamma-glutamyl transferase (γ-GT) activity was assayed by the method of Fossati et al. 22
Micronucleus assay
Bone marrow fluids containing cells were harvested, prepared and stained with Giemsa stain. The number of micronucleated polychromatic erythrocytes (mnPCEs) per 1000 PCEs was counted under light microscope. The ratio of PCE to normochromatic erythrocytes was calculated. While normal cells appeared pink, damaged cells appeared blue, with the micronucleus appearing as a black spot in the cells.
DNA fragmentation by DPA assay
The colorimetric determination of fragmented DNA was done according to the method described by Wu et al. 23 with slight modification. The liver and kidney were homogenized in TE buffer and centrifuged at 27,000 × g for 10 min to separate the intact DNA (pellet) from the fragmented DNA (supernatant). Both the pellet and supernatant were treated with freshly prepared diphenylamine (DPA) reagent for colour development. Incubation was carried out at 37°C for 20–24 h. The absorbance was read spectrophotometrically at 620 nm. The percentage fragmented DNA was calculated using the formula:
Immunohistochemical assay
The immunochemical staining of liver for expressions of Bcl-2 and p53 were done according to the method of Chakravarthi et al. 24 with slight modification. Slides were observed under a binocular microscope. Cells with specific brown colour in the cytoplasm, cell membrane or nuclei, depending on the antigenic sites, were considered positive.
Statistical analysis
All values were expressed as the mean ± standard deviationSD of six animals per group. Data were analysed using one-way analysis of variance followed by the post hoc Duncan multiple range test for analysis of biochemical data using SPSS (10.0). Values were considered statistically significant at p < 0.05.
Results
Effects of BA on body weight and serum biochemical indices in NDMA-treated rats
As shown in Table 1, NDMA significantly (p < 0.05) decreased body weight gain of rats by 76% when compared to control. However, pretreatment with BA significantly (p < 0.05) increased body weight gain by 50% relative to NDMA-only-treated group. Also, NDMA significantly increased the relative weight of kidney and liver of the rats relative to the control. The relative weight of both organs in NDMA-treated group was significantly (p < 0.05) decreased upon pretreatment with BA (Table 1). Administration of NDMA significantly (p < 0.05) increased the activities of serum ALT, AST and γ-GT by 51%, 48% and 81%, respectively, when compared to the control. However, when the rats were pretreated with BA, the activities of these enzymes were significantly (p < 0.05) attenuated relative to NDMA-treated group, and the values were comparable to controls (Table 2). Furthermore, NDMA intoxication significantly (p < 0.05) increased serum total bilirubin by 72% relative to control. Pretreatment with BA was noticed to significantly reduced total bilirubin in NDMA-treated rats (Table 2). In Table 3, administration of NDMA caused a significant (p < 0.05) increase in the serum levels of urea and creatinine by 64% and 82%, respectively, relative to control. In contrast, NDMA intoxication significantly (p < 0.05) reduced urinary creatinine by 67% when compared to the control. Pretreatment of NDMA-treated rats with BA significantly attenuated the serum and urinary creatinine as well as serum urea in the rats.
Changes in body weight, organs’ weight and relative weight of organs of rats administered with BA and NDMA treatments weight (g), weight of organs (g) and relative weight (as % of body weight).a

BA: betulinic acid; NDMA: N-nitrosodimethylamine.
aData expressed in mean ± SD, n = 6.
bStatistically different (p < 0.05) compared with control.
cStatistically different (p < 0.05) compared with NDMA.
Effects of BA on serum enzymes and total bilirubin in rats intoxicated with NDMA.a

BA: betulinic acid; NDMA: N-nitrosodimethylamine; ALT: alanine aminotransferase; AST: aspartate aminotransferase; γ-GT: gamma-glutamyl transferase.
aData expressed as mean ± SD, n = 6.
bStatistically different (p < 0.05) compared with control.
cStatistically different (p < 0.05) compared with NDMA.
Effects of BA on the levels of serum urea and creatinine, and urinary creatinine in rats treated with NDMA.a
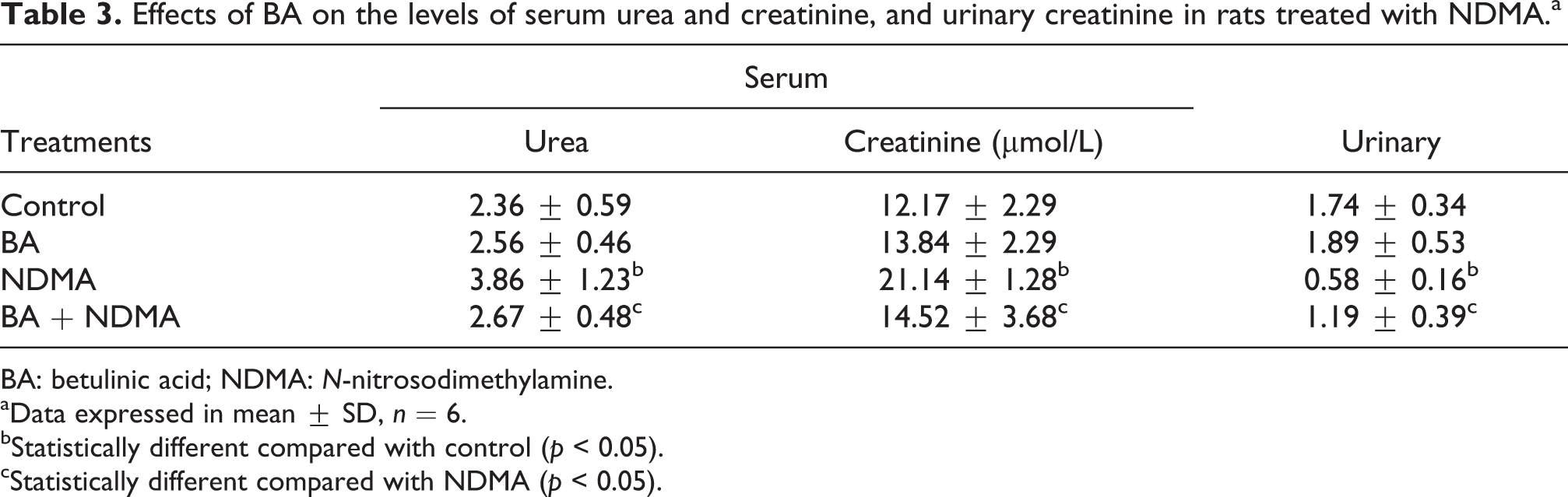
BA: betulinic acid; NDMA: N-nitrosodimethylamine.
aData expressed in mean ± SD, n = 6.
bStatistically different compared with control (p < 0.05).
cStatistically different compared with NDMA (p < 0.05).
Effects of BA on NDMA-induced genotoxicity in rats
As shown in Figure 1, NDMA significantly (p < 0.05) increased the frequency of mnPCE (11.1 ± 2.6 mnPCE/1000 PCE) in the rats relative to the control (4.3 ± 1.1 mnPCE/1000 PCE). Pretreatment with BA significantly (p < 0.05) reduced the frequency of mnPCE (7.5 ± 4.1 mnPCE/1000 PCE) relative to NDMA-treated group (11.1 ± 2.6 mnPCE/1000 PCE). Furthermore, NDMA intoxication significantly (p < 0.05) elevated DNA fragments in the liver (43.63 ± 11.74) and kidney (60.88 ± 13.40) when compared to their controls (22.10 ± 8.44 and 16.37 ± 76, respectively; Figure 2). Pretreatment with BA significantly (p < 0.05) reduced the levels of DNA fragments in the liver (18.03 ± 5.49) and kidney (34.08 ± 9.38) relative to the NDMA-treated group (43.63 ± 11.74 and 60.88 ± 13.40, respectively). The immunohistochemical findings revealed strong expression of Bcl-2 in the liver of NDMA-treated group (Figure 3), while moderate expressions were observed in the control, BA alone and (BA + NDMA) groups. In Figure 4, NDMA intoxication caused strong nuclear expression of p53 protein. However, moderate nuclear expressions of p53 were seen in slides from the control, BA alone and (BA + NDMA) groups.

Effects of BA on frequency of micronucleated polychromatic erythrocytes in bone marrow of NMDA-treated rats. BA: betulinic acid; NDMA: N-nitrosodimethylamine. *p < 0.05: significantly different from control; #p < 0.05: significantly different from NDMA group (p < 0.05).

Effects of BA on DNA fragmentation in liver and kidney of NDMA-treated rats. BA: betulinic acid; NDMA: N-nitrosodimethylamine. *p < 0.05: significantly different from control; #p < 0.05: significantly different from NDMA group (p < 0.05).

Effects of BA on Bcl-2 expression of liver of rats treated with NDMA. BA: betulinic acid; NDMA: N-nitrosodimethylamine. *Significantly different from control (p < 0.05), #Significantly different from NDMA group (p < 0.05).

Effects of BA on the expression of nuclear p53 protein in liver of rats treated with NDMA. BA: betulinic acid; NDMA: N-nitrosodimethylamine. *p < 0.05: significantly different from control; #p < 0.05: significantly different from NDMA group (p < 0.05).
Discussion
N-nitrosamines generate deleterious radicals in cellular systems 2 and have been linked to induction of cancers. 25,26 NDMA and diethylnitrosamines have been reported to cause hepatic carcinogenesis in animals. 27,28
In this study, administration of NDMA to rats caused significantly reduction in body weight gain relative to control. Similar observations were reported by Sivaramakrishnan et al. 29 and Singh et al. 30 who noted body weight loss after administration of N-nitrosodiethylamine (NDEA), a closely related toxicant. The decrease in body weight gain of the NDMA-treated rats may be due to loss of appetite, digestive disorder or malabsorption. 31 Furthermore, the toxicant caused significant increase in the relative weight of liver and kidney in the rats. This observation is consistent with the findings of Sivaramakrishnan et al. 29 and Singh et al. 30 who observed that NDEA caused a significant increase in the relative weight of liver in rats. However, pretreatment with BA significantly increased body weight gain and, decreased relative weight of liver and kidney of NDMA-treated rats. The increase in body weight gain of rats upon pretreatment with BA could be as a result of improved appetite and increased intestinal absorption of digested foods. However, this suggestion is not supported by our data and, is one of the limitations of this study. Administration of NDMA resulted in elevation of serum ALT and AST and γ-GT, which is an indication of hepatic damage in the rats. Similar findings were reported by Farombi et al. 32 and Lin et al. 33 Also, in agreement with other studies, nitrosamines such as N-butyl-N-(4-hydroxybutyl) nitrosamine, 34 NDEA 35 and NDBA 36 have been reported to cause significant increase in serum ALT and AST activities. Elevated levels of these enzymes may be an indication of impaired liver function in the NDMA-treated group. Both ALT and AST are sensitive markers of hepatic damage and are therefore used in determining occurrence of hepatocellular diseases. The results indicate that NDMA or its toxic metabolites may damage the hepatocytes leading to alterations in membrane permeability and resulting in leakages of these enzymes into the serum. 37 Interestingly, pretreatment with BA significantly attenuated the activities of these enzymes to values that were statistical similar to the control. Thus, BA may exert protection against NDMA-induced hepatocellular damage. Bilirubin is a lipid-soluble pigment formed in the reticuloendothelial cells, which may serve as a marker of hepatic function. Therefore, an increase in plasma/serum total bilirubin level is a measure of integrity of the liver. In this study, NDMA intoxication led to elevated serum total bilirubin in the rats. Early studies by Singh et al. 30 and Hussain et al. 38 observed that NDEA caused a significant increase in serum total bilirubin level, which is in agreement with the present study. The increased level of total bilirubin observed in this study indicates that NDMA may induce hyperbilirubinemia in the rats. Pretreatment with BA lowered serum total bilirubin in NDMA-treated rats, indicating hepatoprotective effect of this triterpenoid. In order to investigate the effect of NDMA on renal function in the treated rats, serum levels of urea and creatinine and urinary creatinine were examined. NDMA intoxication significantly increased serum urea and creatinine and decreased urinary creatinine in the rats. The elevation of urea and creatinine or decreased creatinine clearance in NDMA-treated rats is a sign of renal impairment which may be due to tubular damage or reduced glomerular filtration. 39 The results of this study showed renal protective effects of BA, since the elevated levels of both urea and creatinine in NDMA-treated rats were significantly ameliorated in rats given (BA + NDMA).
The genotoxicity of NDMA was examined in the bone marrow of experimental rats using micronuclei induction assay. NDMA intoxication increased the number of mnPCEs in bone marrow and thus confirmed its genotoxicity. This assay has been found useful in evaluating the ability of environmental chemicals to induce chromosomal aberrations (clastogenicity) or causing mitotic distortion in cells. 40 Micronuclei formation results from abnormal chromosomal processes due to aberration in chromosomal segregation in mitosis or when the frequency of chromosomal breakage exceeds the capacity of the normal DNA repair machinery. 41 It has been shown that acentric chromosome fragments from unrepaired DNA strand which may lag behind during anaphase to form micronuclei. 42 The observed gentotoxic effect of NDMA in this study was in consonance with the studies of Akira et al. 43 who reported the cytogenetic potentials of nitrosamines including NDMA, NDEA and NDBA in human hepatocytes (FLC-4). Furthermore, the effect of NDMA on apoptosis was established using DPA assay of DNA fragmentation 23 in hepatic and renal tissues. It was observed that NDMA intoxication increased the levels of DNA fragments in tissues. Importantly, pretreatment with BA significantly reduced the number of mnPCE and levels of DNA fragments induced by NDMA. The present study also examined the modulatory effects of BA on the expression of apoptotic proteins (Bcl-2 and p53) during NDMA intoxication. One of the physiological roles of anti-apoptotic (Bcl-2) is to prevent the action of pro-apoptotic proteins responsible for the opening of mitochondrial membrane permeability transition pore. 44 The dynamic balance between Bcl-2 (anti-apoptotic) and the pro-apoptotic members significantly determines initiation of apoptosis. 45 In the present study, there was strong expression of Bcl-2 in the liver of NDMA-treated rats which may represent an adaptive mechanism during the generation of ROS or metabolites of NDMA, while moderate expressions were found in BA alone and (BA + NDMA) groups. It is known that Bcl-2 protein is overexpressed in a variety of solid tumours, 46 such as acute myeloid leukaemia, 47 chronic lymphocytic leukaemia 48 and follicular lymphoma. 49 Repressing the expression of Bcl-2 protein has been suggested to be a target in cancer chemotherapy due to its anti-apoptotic and tumour cell priming effects. 44 A well-established function of p53 gene is in the induction of both intrinsic and extrinsic pathways of apoptosis. The p53-mediated apoptosis primarily utilizes the intrinsic, while the extrinsic pathway complements the apoptotic response. 50 During intrinsic apoptosis, the genes encoding the Bcl-2 family proteins are the primary target of p53 gene. While the genes of pro-apoptotic ones (e.g. Bax, Bak, Bcl-xl, Bid, puma and Noxa) are upregulated, the genes of anti-apoptotic ones (e.g. Bcl-2) are downregulated by p53. 51 In a transcription-independent manner, cytoplasmic p53 may form complexes with pro-apoptotic Bax family members, being accumulated in the mitochondria, inducing loss of mitochondrial membrane potential which may result in a release of cytochrome C and formation of apoptosome complex. 52 In the present study, nuclear p53 protein was moderately expressed in control, BA alone and (BA + NDMA) groups while strong nuclear expression of p53 protein was observed in NDMA-treated rats. It has been reported that a normal cell moderately expressed the wild-type p53, which is in an inactive form, 53 and this level is maintained during cell cycle progression. 54 However, during cellular (genotoxic) stress, such as DNA damage, hypoxia, heat shock and oncogene overexpression, there is activation of the wild-type p53 gene, leading to an extensive accumulation of p53 protein in the nucleus. 55 This explanation supports the strong expression of p53 protein in NDMA-treated rats. The moderate nuclear expression of p53 in (BA + NDMA) group shows that this triterpenoid induces physiologically mediated apoptosis against NDMA-induced toxicity in the rats.
In conclusion, administration of NDMA caused gentotoxic, hepatotoxic and nephrotoxic effects in rats which were mitigated via mechanism that involves anti-oxidative and anti-apoptotic roles of BA.
Footnotes
Acknowledgements
The authors thankfully acknowledge the assistance of technical staff of the Department of Biochemistry, University of Ibadan, Nigeria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

