Abstract
The Expert Panel assessed the safety of 28 alkyl phosphates and concluded that these ingredients are safe in the current practices of use and concentration when formulated to be nonirritating. The ingredients in the alkyl phosphate family share a common phosphate core structure, and vary by the identity of the alkyl chains attached therein. Most of the alkyl phosphates function as surfactants in cosmetic ingredients; however, the triesters function as plasticizers rather than surfactants. The Panel reviewed the available animal and clinical data to determine the safety of these ingredients.
Keywords
Introduction
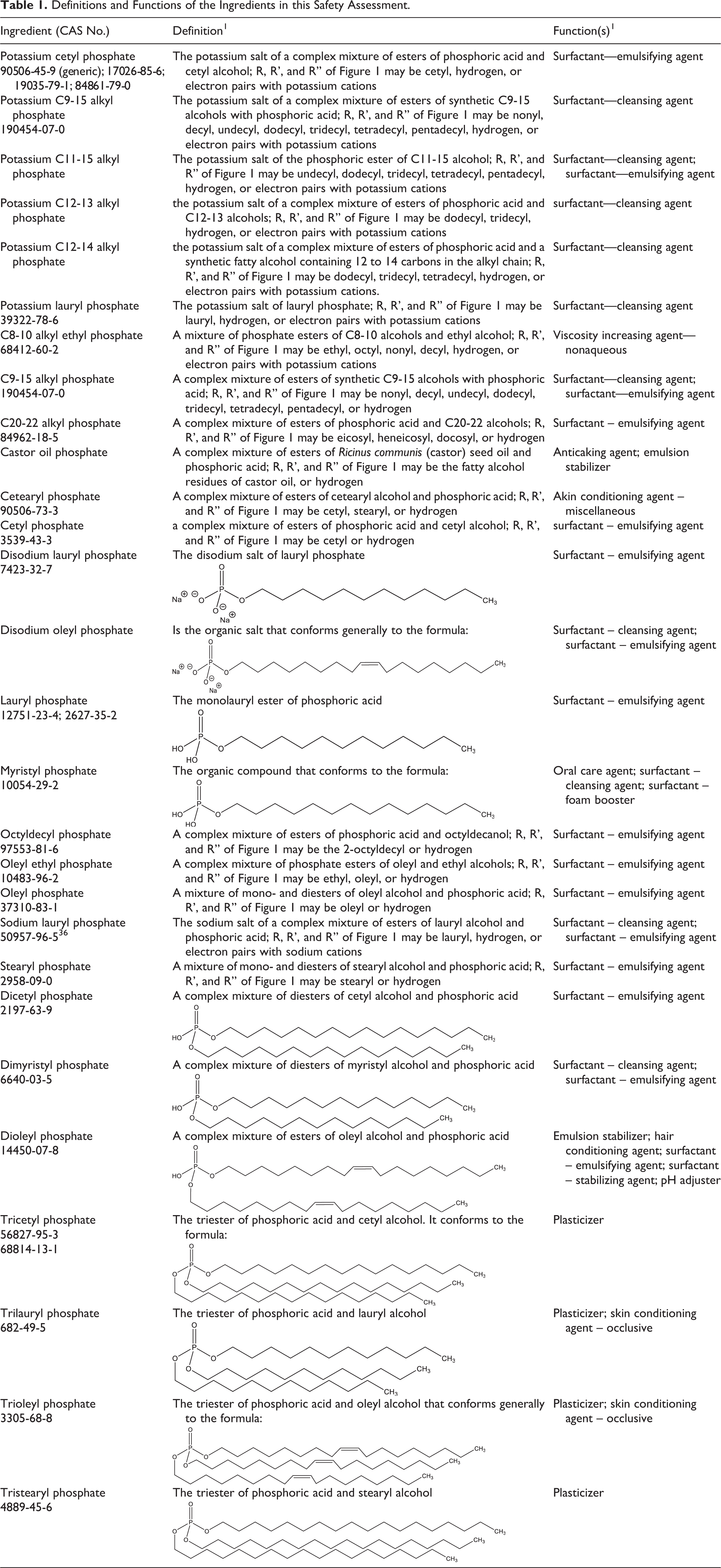
This report is a safety assessment of the following 28 alkyl phosphates as used in cosmetic formulations: Potassium cetyl phosphate Potassium C9-15 alkyl phosphate Potassium C11-15 alkyl phosphate Potassium C12-13 alkyl phosphate Potassium C12-14 alkyl phosphate Potassium lauryl phosphate C8-10 alkyl ethyl phosphate C9-15 alkyl phosphate C20-22 alkyl phosphate Castor oil phosphate Cetearyl phosphate Cetyl phosphate Disodium lauryl phosphate Disodium oleyl phosphate Lauryl phosphate Myristyl phosphate Octyldecyl phosphate Oleyl ethyl phosphate Oleyl phosphate Sodium lauryl phosphate Stearyl phosphate Dicetyl phosphate Dimyristyl phosphate Dioleyl phosphate Tricetyl phosphate Trilauryl phosphate Trioleyl phosphate Tristearyl phosphate
The ingredients in the alkyl phosphate family share a common phosphate core structure, and vary by the identity of the alkyl chains (ranging from 8-22 carbons in length) attached. Most of the alkyl phosphates are reported to function as surfactants in cosmetic ingredients; however, the triesters function as plasticizers rather than surfactants (Table 1). 1
Definitions and Functions of the Ingredients in this Safety Assessment.
Much of the data included in this safety assessment were found on the European Chemicals Agency (ECHA) website. 2 The ECHA website provides summaries of information generated by industry, and it is those summary data that are reported in this safety assessment when ECHA is cited. In several instances, structural analogs were used as supporting substances to provide read-across. Specifically, phosphoric acid, C16-18 alkyl esters, and potassium salts are used as read-across for potassium cetyl phosphate. Although not identical, the distribution of chain-lengths for these 2 chemicals will have a great deal of overlap, for example, both will contain 16-carbon chain lengths. 1-Octadecanol, phosphate, and potassium salt also are relevant to the safety of potassium cetyl phosphate because potassium cetyl phosphate is a distribution of chain-lengths (a mixture) attached to phosphate, with a mean peak at 16-carbons in length, and 1 octadecanol, phosphate, potassium salt is a distribution of chain-lengths attached to phosphate, with a mean peak at 18-carbons in length. Both include some longer and some shorter fatty acid residues (eg, 14- and 18-carbon chains and 16- and 20-carbon chains, respectively). Accordingly, some read-across may be accessible between these 2 ingredients, as their mean chain-lengths only differ by 2 carbons and there are at least some literally identical chain-lengths shared by the 2 ingredients (in light of the complete length-distribution of each ingredient). Additionally, phosphoric acid and 2-ethylhexyl ester were justified as read-across for potassium lauryl phosphate because both are members of the phosphoric acid, alkyl ester family, and the characteristic and functional active center of both substances is the ester binding between the alcoholic compound and phosphate. When providing information on a structural analog, the name of that analog is italicized to indicate read-across is being employed. However, the names of the ingredients in this report are written in accordance with the International Nomenclature naming conventions, that is, capitalized without italics or abbreviations.
Chemistry
Definition and Structure
Alkyl phosphates are the organic esters of ortho-phosphoric acid. These ingredients are mixtures of esters and salts wherein a phosphate may have 1 to 3 alkylations and 1 to 2 potassium or sodium cations (Figures 1 and 2; Table 1).
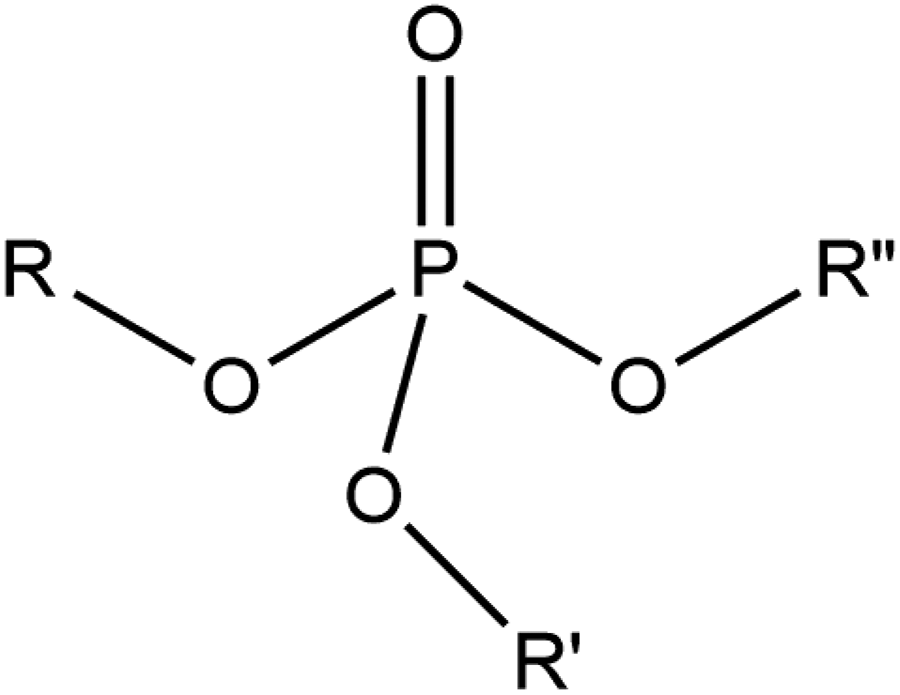
Alkyl phosphates, wherein R, R’, and R’’ may be alkyl groupings (eg, cetyl), hydrogen, or shared electron pairs with potassium or sodium cations.
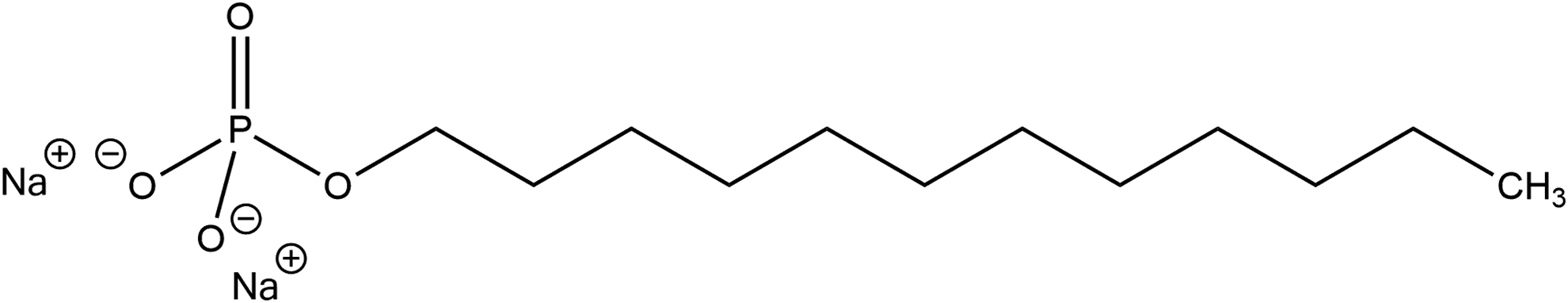
Disodium lauryl phosphate, for example, has sodium cations for R and R’, and a lauryl chain for R”.
These ingredients share some similarities, in structures and properties, with the natural phosphatides, lecithin, and cephalin. 3 But these ingredients differ by the lack of core glyceryl structures and the resultant lack of susceptibility to enzymatic degradation. With solubility across polar and nonpolar solvents, it is not surprising that these ingredients are commonly used as surfactants, wetting agents, and emulsifiers in cosmetic applications.
Chemical and Physical Properties
The alkyl phosphates can be liquids or solids (Table 2). They have solubility in both polar and nonpolar solvents.
Chemical and Physical Properties.

Methods of Manufacture
Alkyl phosphates can be prepared by reactions of fatty alcohols with polyphosphoric acid to yield the corresponding alkyl phosphates. 4 Dialkyl phosphates can be prepared by a stepwise procedure via the monoalkyl phosphate from pyrophosphoric acid using tetramethylammonium hydroxide as a base. 5 They also can be prepared by the reaction of 2 equivalents of alcohol with phosphorus oxychloride followed by hydrolysis of the intermediate phosphoroxychloride. Dialkyl phosphates also have been synthesized by the reaction of the appropriate alcohol with phosphorus trichloride followed by treatment with pyridine and carbon tetrachloride, which provides the corresponding trichloromethyl ester. Reaction of the triethylamine salt of acetic acid, followed by hydrolysis of the mixed anhydride that formed, yields the dialkyl phosphate.
C20-22 alkyl phosphate is obtained from the reaction of alcohols, C20-22 with phosphoric anhydride. 6
Constituents/Impurities
C20-22 alkyl phosphate contains <1% phosphoric acid. 6 No other published constituent data were found, and no unpublished data were submitted.
Use
Cosmetic
Most of the alkyl phosphates are reported to function as surfactants in cosmetic ingredients; however, the triesters (ie, R, R’, and R” are all alkyl; eg, tricetyl phosphate) function as plasticizers rather than surfactants (Table 1). 1 Surfactants, or surface-active agents, have the ability to lower the surface tension of water or to reduce the interfacial tension between 2 immiscible substances. Plasticizers are materials that soften synthetic polymers.
The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). Voluntary Cosmetic Registration Program data obtained from the FDA in 2014, 7 and data received in 2013 to 2014 in response to surveys of the maximum reported use concentration by category that were conducted by the Personal Care Products Council (Council), 8,9 indicate that 13 of the 28 ingredients included in this safety assessment are currently used in cosmetic formulations.
According to the VCRP data, potassium cetyl phosphate is reported to be used in 375 formulations, the majority of which are leave-on formulations, dicetyl phosphate is reported to be used in 109 formulations, and cetyl phosphate in 94 formulations. 7 All other in-use ingredients are reported to be used in less than 15 formulations. The results of the concentration of use surveys conducted by the Council indicate potassium cetyl phosphate also has the highest concentration of use in a leave-on formulation; it is used at up to 8.3% in mascara products. 8 The highest concentration of use reported for products resulting in leave-on dermal exposure is 4.2% trioleyl phosphate in “other” make-up preparations. 9 Lauryl phosphate is used at 8.7% in a skin cleaning product, which is most likely a rinse-off formulation. 8 All available frequency and concentration of use data, for those ingredients currently in use, are reported in Table 3. The ingredients not reported to be used, according to VCRP data and the Council survey, are listed in Table 4.
Frequency and Concentration of Use According to Duration and Type of Exposure.
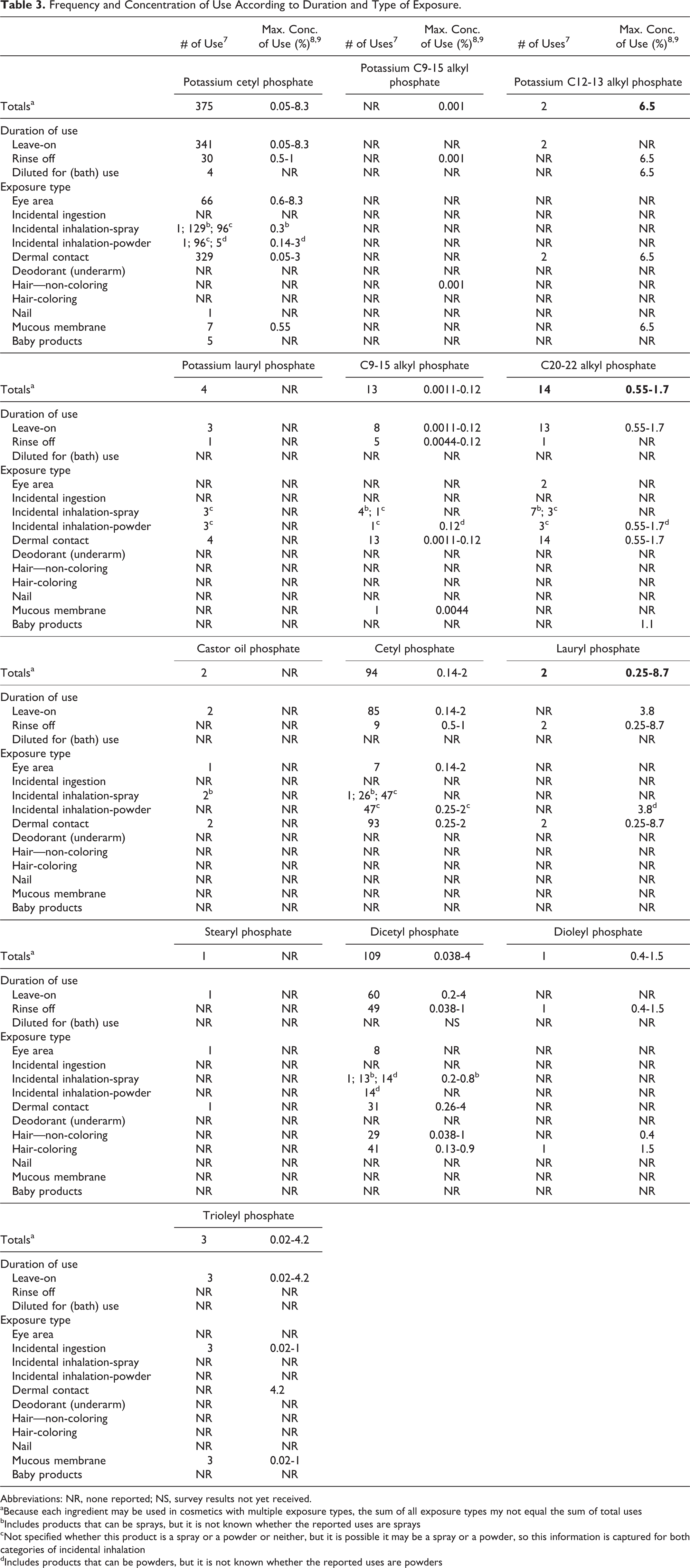
Abbreviations: NR, none reported; NS, survey results not yet received.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses
bIncludes products that can be sprays, but it is not known whether the reported uses are sprays
cNot specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation
dIncludes products that can be powders, but it is not known whether the reported uses are powders
Ingredients Not Reported to be Used.

A few of the ingredients are used in products that could be incidentally ingested (eg, up to 1% trioleyl phosphate in lipsticks) or used near the eye or mucous membranes (eg, up to 8.3% potassium cetyl phosphate in mascara formulations). One ingredient, C20-22 alkyl phosphate, is reported to be used at 1.1% in a baby product. Additionally, according to the VCRP, dicetyl phosphate is used in a hair spray, which is a product that can be incidentally inhaled; however, the Council survey did not report a concentration of use for this product type. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 10,11 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 12,13
All of the alkyl phosphates named in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 14
Noncosmetic
Potassium lauryl phosphate can be used as an optional finish component in poly(phenyleneterephthalamide) resins, which are indirect food additives intended for repeated contact with food; the total weight of potassium lauryl phosphate is not to exceed 1% of the base polymer [21CFR177.1632]. Tristearyl phosphate is approved as an indirect food additive as a substance permitted to be used in the formulation of defoaming agents used in the manufacture of paper and paperboard [21CFR176.210].
The use of dicetyl phosphate in niosomes (nonionic surfactant-based vesicles) 15 -17 and solid lipid nanoparticles 18 has been investigated. Niosomes are microscopic vesicles composed of nonionic surface-active agent bilayers, and the intended use of these vesicles is as a drug delivery system. 16,17 Solid lipid nanoparticles are another possible dermal delivery system. 18
Toxicokinetics
Oral
Potassium lauryl phosphate
Five male and 5 female F344 rats were given a single dose of 200 mg/kg bw phosphoric acid, 2-ethylhexyl ester in corn oil by gavage. 19 (As stated previously, information on phosphoric acid, 2-ethylhexyl ester is being provided as read-across for potassium lauryl phosphate. Specifically, with reference to the occurrence of esterases which take part in the mammalian phase I metabolism, it can be assumed that both phosphoric acid esters are hydrolyzed independent from the constitution of the alcoholic part. Since the ester binding is the specific target of endogenous esterases, it is justified to perform a read across between both ester-type substances in order to estimate potential metabolism. Urine and feces were collected every 12 hours for 72 hours after dosing. Analysis of the samples via 31 P-nuclear magnetic resonance spectroscopy indicated the ester was completely hydrolyzed to phosphate and 2-ethylhexanol; only a phosphate peak was found in the urine samples. The conclusion of this summary report stated phosphoric acid, 2-ethylhexyl ester was efficiently absorbed, metabolized, and excreted quantitatively by the body, and there was no indication of accumulation; however, no details were provided.
Toxicological Studies
Single Dose (Acute) Toxicity
Dermal, oral, and inhalation single-dose toxicity testing has been performed with some alkyl phosphates (Table 5). These ingredients are relatively nontoxic. The dermal LD50 in rats was >2 g/kg bw for C20-22 alkyl phosphate, 20 oleyl phosphate, 21 and for 45.45% and 80% dicetyl phosphate. 22,23 In rats, the oral LD50 was >2 g/kg for 1 octadecanol, phosphate, potassium salt, 22 potassium C9-15 alkyl phosphate, 22 C20-22 alkyl phosphate, 20 oleyl phosphate, 21 and dimyristyl phosphate; 24 the oral LD50 of 25% potassium lauryl phosphate was 10.49 g/kg; 19 and for 10% cetyl phosphate it was > 4.7 g/kg. 22 In both the mouse 25 and rat, 26 the oral LD50 of a 25% suspension of dicetyl phosphate was > 5 g/kg. In a 4-hour exposure inhalation study, the LC50 of 1% aq. phosphoric acid, C16-18 alkyl esters, potassium salts was > 200 µl/L. 22
Single-Dose Toxicity Studies.
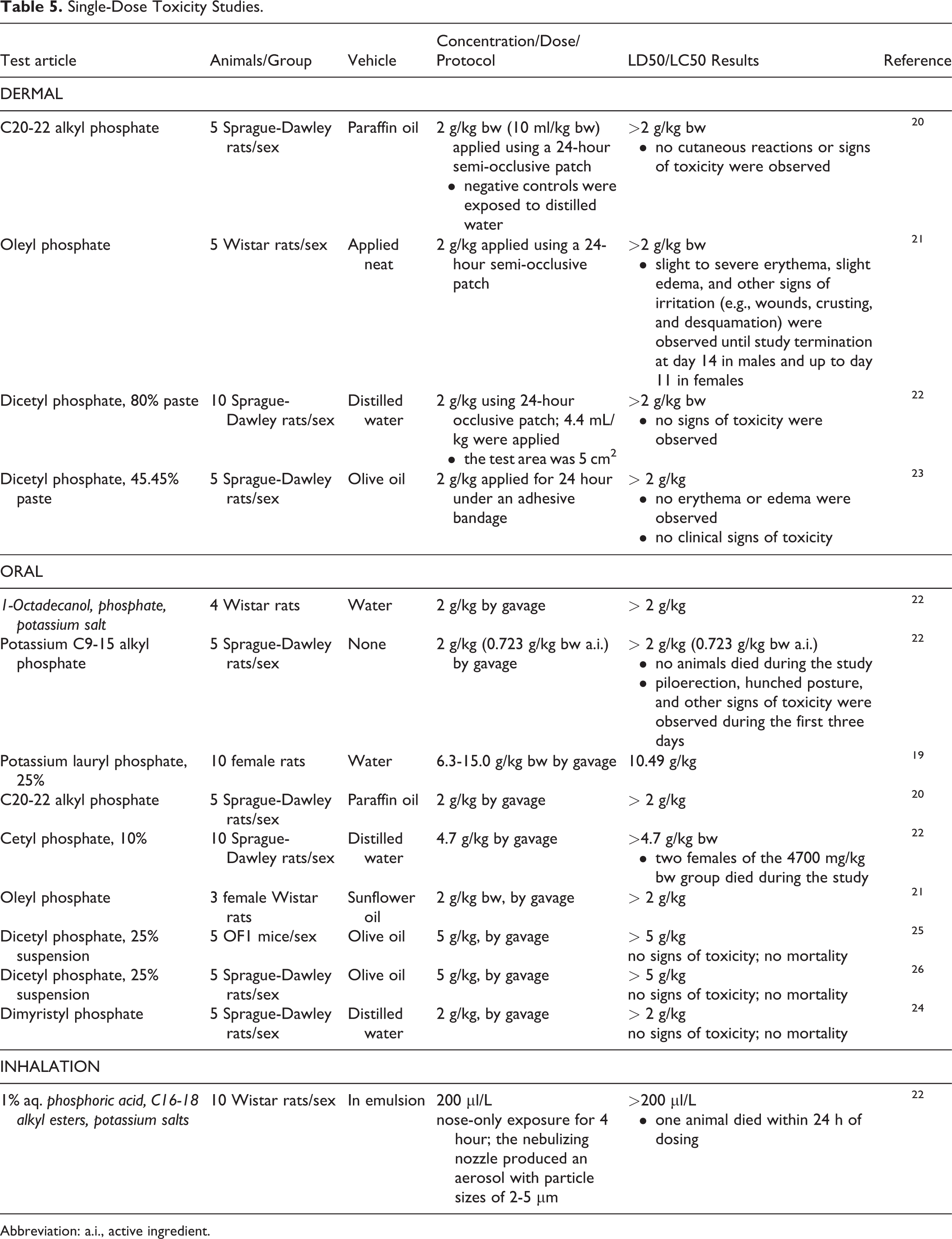
Abbreviation: a.i., active ingredient.
Repeated Dose Toxicity
Repeated dose oral toxicity studies were performed in rats for several alkyl phosphates (Table 6). In 14-day studies, potassium lauryl phosphate had a no-observable adverse effect level (NOAEL) of 600 mg/kg bw/d for both males and females, and oral administration of up to 1000 mg/kg bw/d sodium lauryl phosphate for 14 days did not result in any adverse effects; 19 no remarkable effects were observed with up to 1000 mg/kg bw/d C20-22 alkyl phosphate by gavage. 20 The NOELs of myristyl phosphate in a 28-day dietary study were 1564 mg/kg bw/d for males and 227 mg/kg bw/d for females, and the NOAEL was 1564 mg/kg bw/d for females. 22 Oleyl phosphate had a NOAEL of 1000 mg/kg bw/d for male and female rats in a 28-day gavage study. 21 In a 91-day gavage study, potassium C9-15 alkyl phosphate had a benchmark dose lower confidence limit of 240.3 mg/kg bw/d in males and females. 22
Repeated Dose Toxicity Studies.
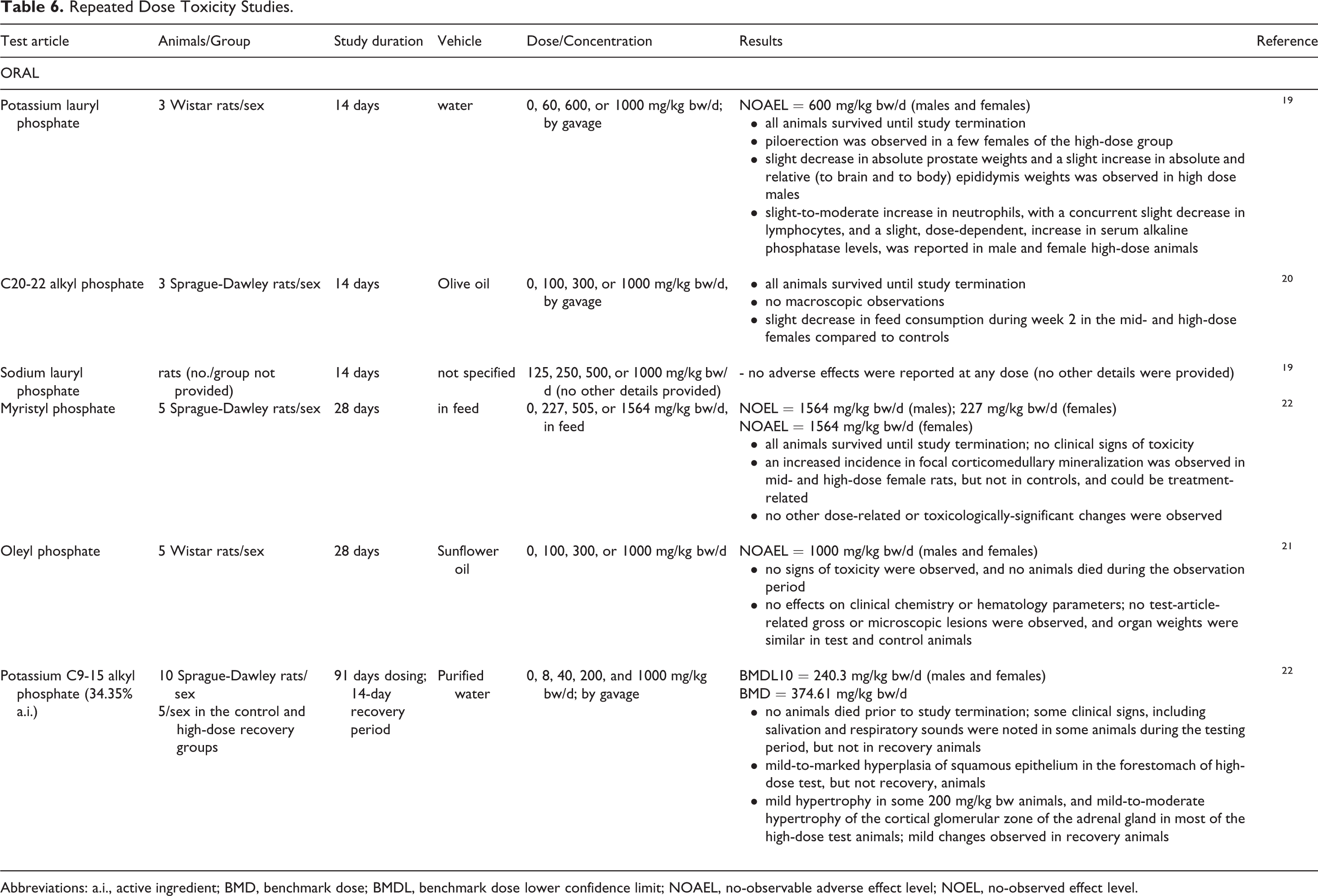
Abbreviations: a.i., active ingredient; BMD, benchmark dose; BMDL, benchmark dose lower confidence limit; NOAEL, no-observable adverse effect level; NOEL, no-observed effect level.
Reproductive and Developmental Toxicity
Potassium C9-15 alkyl phosphate was not embryotoxic, fetotoxic, or teratogenic in rats dosed by gavage on days 6 to 15 of gestation; the NOELs for developmental toxicity, embryotoxicity, fetotoxicity, and teratogenicity were 361 mg/kg bw/d (active ingredient [a.i.]), and the NOEL and NOAEL for maternal toxicity were 36.1 and 361 mg/kg bw/d (a.i.), respectively (Table 7). 22 For C20-22 alkyl phosphate, the NOELs for reproduction (mating and fertility) and neonatal toxicity, and the NOAEL for parental toxicity, were 1000 mg/kg bw/d in rats. 20 Oleyl phosphate also was not a reproductive toxicant in rats; in a gavage study, the NOAELs were 1000 mg/kg bw/d for maternal toxicity, reproductive performance in male and female rats, and development in F1 offspring. 21 In a reproductive study with sodium lauryl phosphate in rats, the NOAEL for parental male and female animals and the NOEL for the F1 generation were 1000 mg/kg bw/d. 19
Reproductive and Developmental Toxicity Studies.
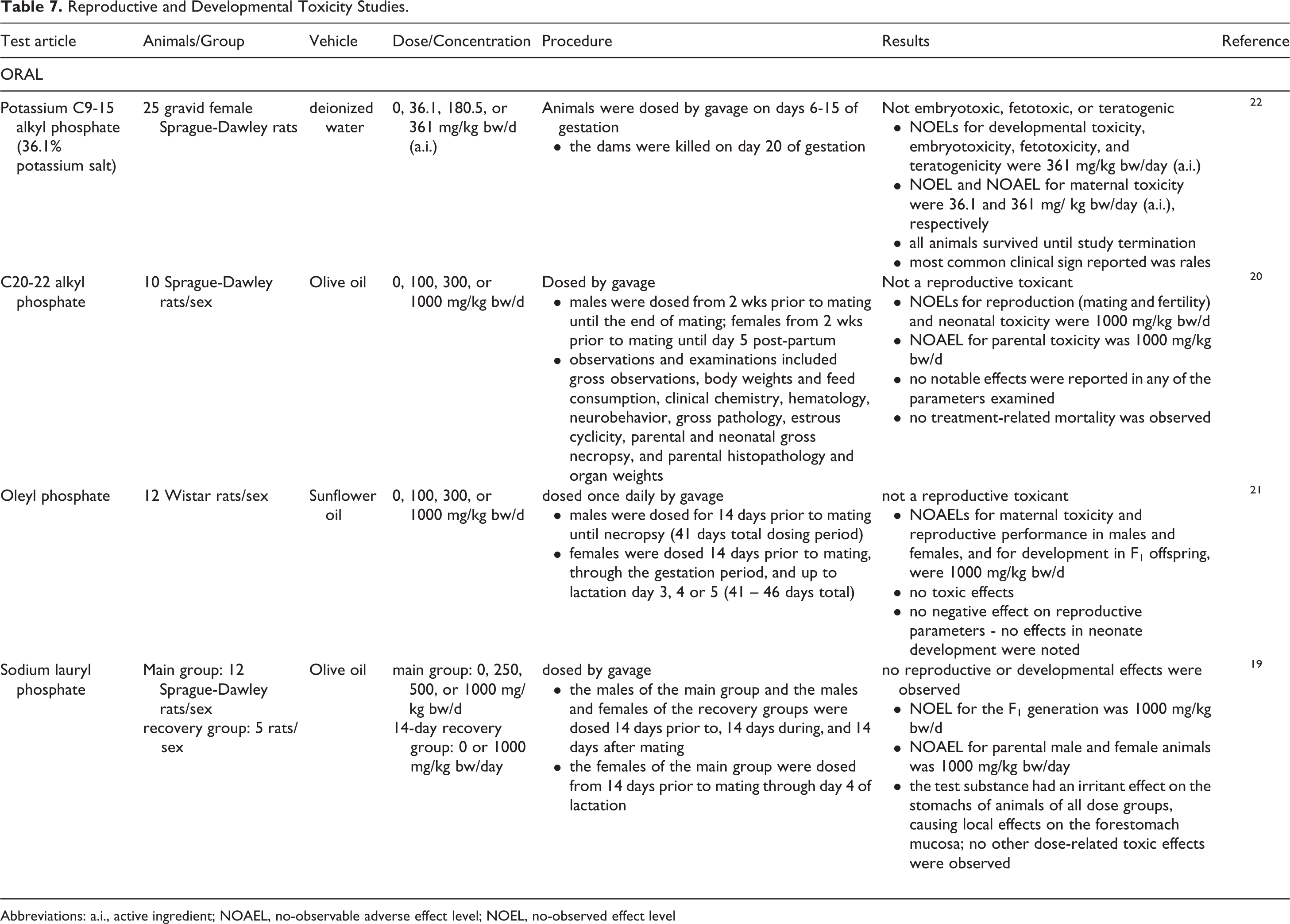
Abbreviations: a.i., active ingredient; NOAEL, no-observable adverse effect level; NOEL, no-observed effect level
Genotoxicity
In vitro genotoxicity assays have been performed on several of the alkyl phosphates and the results of all these assays were negative (Table 8). 1-Octadecanol, phosphate, and potassium salt were negative in an Ames test, 22 and cetyl phosphate was not genotoxic in a mammalian cell gene mutation assay. 22 Potassium lauryl phosphate, 19 C20 22 alkyl phosphate, 20 and oleyl phosphate 21 were not mutagenic in the Ames test, mammalian cell gene mutation assay, or chromosomal aberration assay.
Genotoxicity Studies.
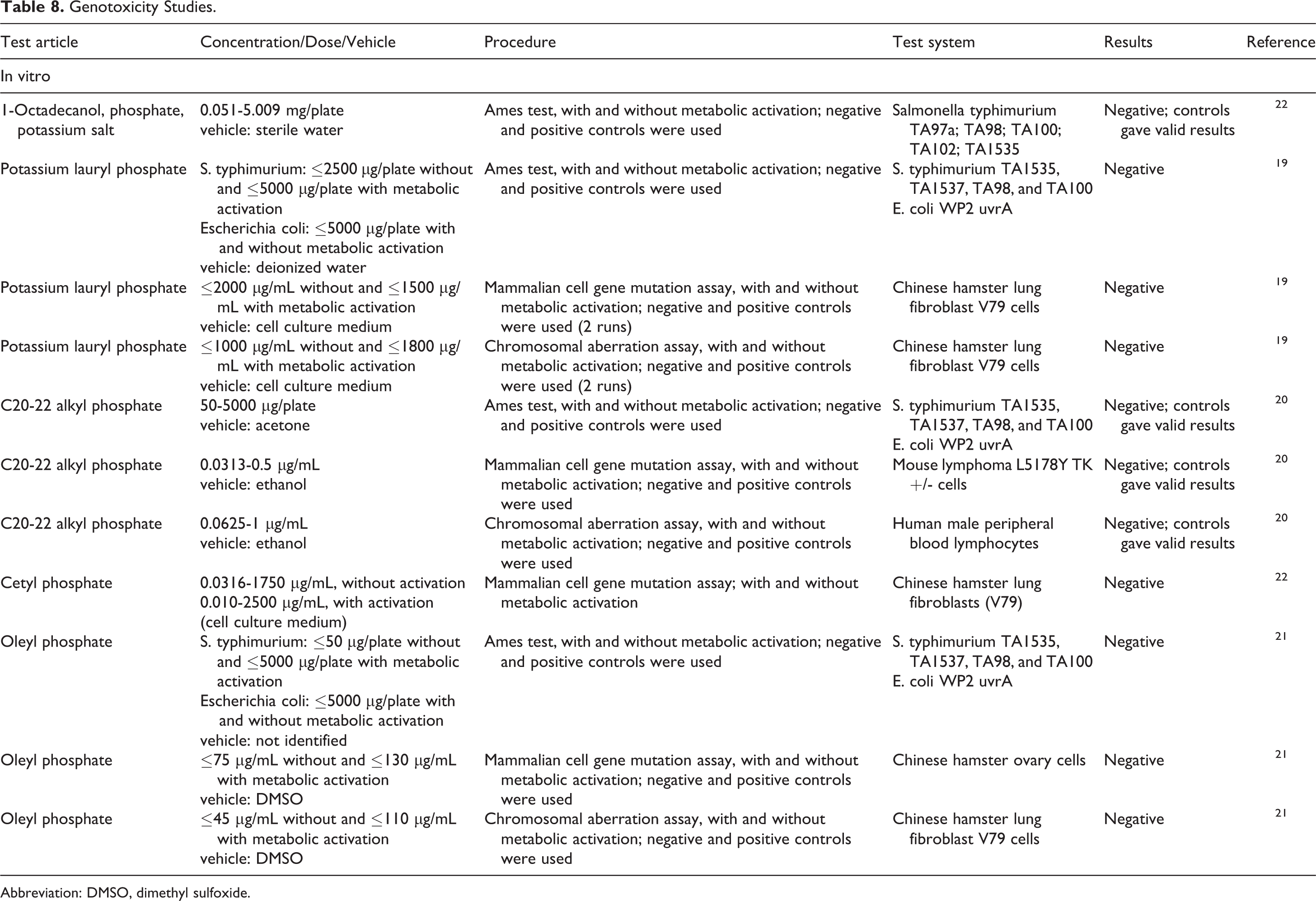
Abbreviation: DMSO, dimethyl sulfoxide.
Carcinogenicity
Published carcinogenicity data were not found, and no unpublished data were submitted.
Irritation and Sensitization
Some alkyl phosphates were not dermal irritants, whereas several were irritating but not sensitizing, in nonhuman studies (Table 9). C20-22 alkyl phosphate, applied neat, was not irritating to rat skin, 20 nor was it a sensitizer in a guinea pig maximization test (GPMT). 20 Undiluted phosphoric acid, C16-18 alkyl esters, potassium salts produced some signs of irritation in the abraded skin of rabbits. 22 Potassium lauryl phosphate was irritating to rabbit skin as a 77% paste in one study, and highly irritating to rabbit skin in another (concentration not specified); it was not a sensitizer in a GPMT. 19 Cetyl phosphate and lauryl phosphate were not sensitizers in GPMTs, but challenge concentrations of 10% and 40% cetyl phosphate and an epidermal induction concentration of 12.5% lauryl phosphate were irritating. 22 Undiluted oleyl phosphate was irritating to rat skin; concentrations up to 5% did not demonstrate a potential for sensitization in a local lymph node assay. 21 (Alternative studies with oleyl phosphate did not demonstrate a potential for skin irritation or corrosion.) Dicetyl phosphate was not irritating to rat skin as an 80% paste, 22 was not irritating to rabbit skin when prepared as a 46.5% paste in olive oil (w/w), 27 and was not a sensitizer in a GPMT. 28 Dimyristyl phosphate, applied under an occlusive patch for 4 hours, was not irritating to rabbit skin, 29 nor was it an irritant or sensitizer in a GPMT at a concentration of 75% in distilled water. 30
Irritation and Sensitization Studies.
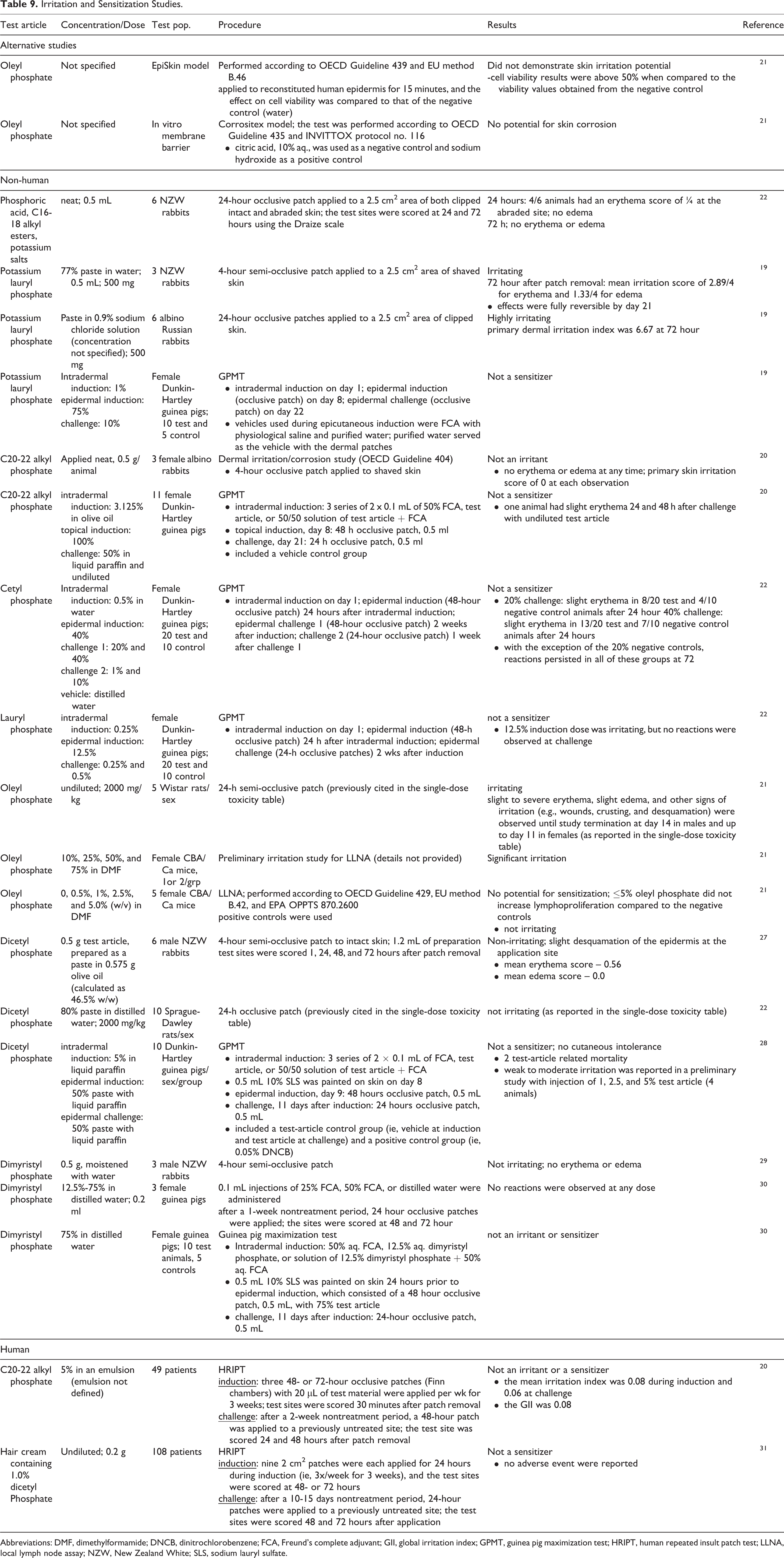
Abbreviations: DMF, dimethylformamide; DNCB, dinitrochlorobenzene; FCA, Freund’s complete adjuvant; GII, global irritation index; GPMT, guinea pig maximization test; HRIPT, human repeated insult patch test; LLNA, local lymph node assay; NZW, New Zealand White; SLS, sodium lauryl sulfate.
The C20-22 alkyl phosphate, 5% in an emulsion, was not an irritant or a sensitizer in a human repeated insult patch test (HRIPT) completed in 49 patients. 20 In an HRIPT completed in 108 patients, a hair cream containing 1.0% dicetyl phosphate was not a sensitizer. 31
Case Report
Trioleyl phosphate
A female patient with severe contact dermatitis on the eyelids was patch-tested with ingredients from the cosmetic formulation suspected of causing the reaction; the product was a lipstick that was mistaken for an eyeshadow. 22 The patient had positive reactions to 3 ingredients, one of which was trioleyl phosphate. The patch testing was repeated using patch test chambers secured to the back. Positive reactions were observed with 0.5% and 1% trioleyl phosphate in petrolatum on days 4 and 7, but not on day 2. The patient did not react to 1% to 10% cetyl phosphate in petrolatum. Negative results were reported in 20 control patients patch tested with 1% trioleyl phosphate in petrolatum.
Ocular Irritation
Some of the alkyl phosphates are reported to be ocular irritants (Table 10). A 10% solution of potassium lauryl phosphate, tested as a 50% dilution in the hen’s egg test utilizing the chorioallantoic membrane (HET-CAM), demonstrated moderate ocular irritation potential, 32 and undiluted oleyl phosphate demonstrated the potential to be corrosive and a severe ocular irritant in an in vitro eye corrosive and severe irritant study. 21 In rabbit eyes, potassium lauryl phosphate was an irritant, 19 C20-22 alkyl phosphate was a moderate irritant, 20 and dicetyl phosphate was slightly irritating. 33 However, a 3% potassium cetyl phosphate solution, tested as a 50% dilution in the HET-CAM, demonstrated practically no ocular irritation potential, 34 and phosphoric acid, C16-18 alkyl esters, potassium salts 22 and dimyristyl phosphate 35 were classified as nonirritating to rabbit eyes.
Ocular Irritation Studies.
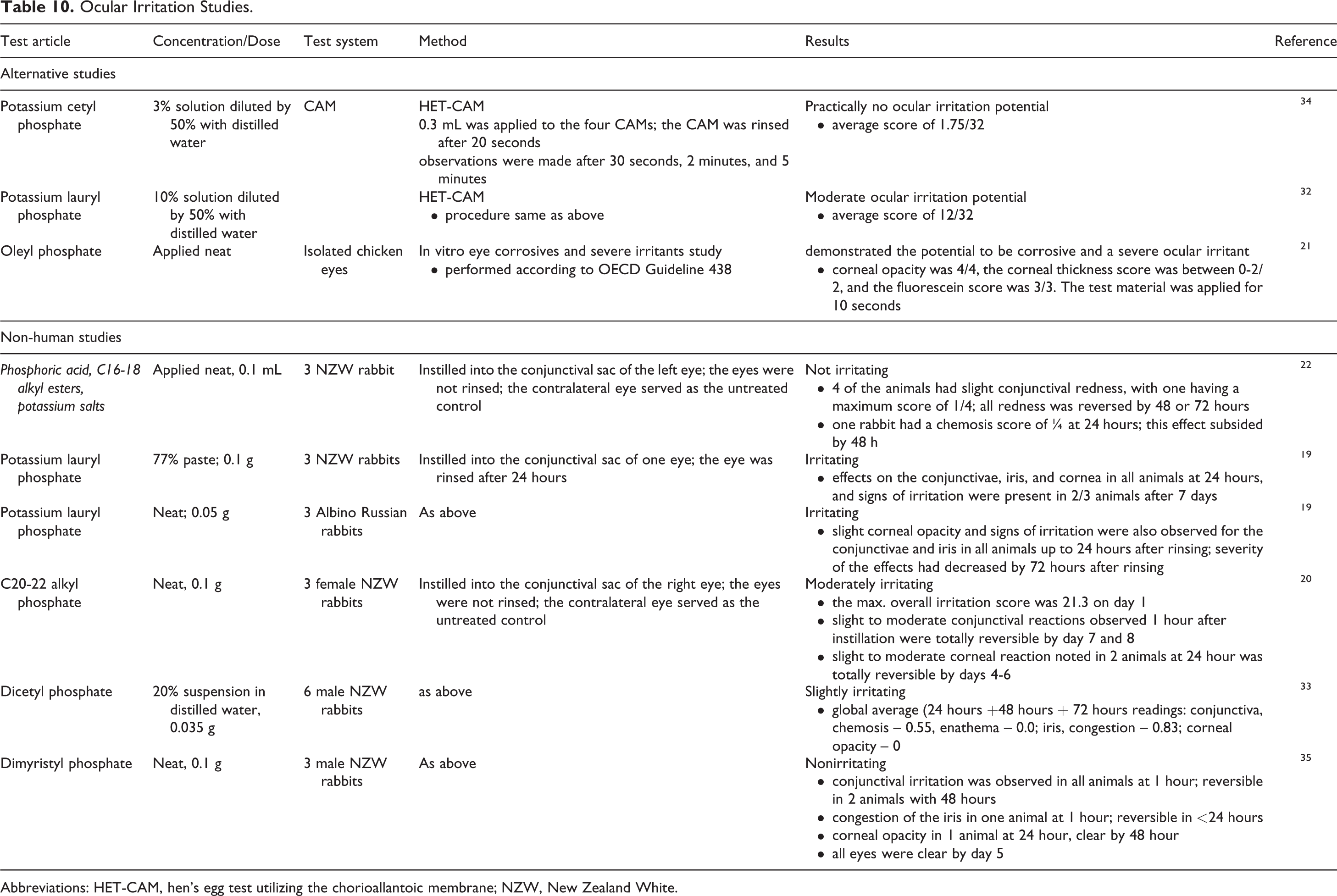
Abbreviations: HET-CAM, hen’s egg test utilizing the chorioallantoic membrane; NZW, New Zealand White.
Summary
This report addresses the safety of 28 alkyl phosphates as used in cosmetics. The ingredients in the alkyl phosphate family share a common phosphate core structure, and vary by the identity of the alkyl chains attached therein. In some instances, structural analogs were used as supporting substances to provide read-across. Specifically, phosphoric acid, C16-18 alkyl esters, potassium salts and 1-octadecanol, phosphate, potassium salt provided read-across for potassium cetyl phosphate, and phosphoric acid, 2-ethylhexyl ester provided read-across for potassium lauryl phosphate.
Most of the alkyl phosphates function as surfactants in cosmetic ingredients; however, the triesters function as plasticizers rather than surfactants. Voluntary Cosmetic Registration Program data obtained from the FDA in 2014, and data received in response to a survey of the maximum reported use concentration by category conducted by Council in 2013 to 2014, indicate that 13 of the 28 ingredients included in this safety assessment are used in cosmetic formulations. Potassium cetyl phosphate is reported to be used in 375 formulations, dicetyl phosphate in 109 formulations, and cetyl phosphate in 94 formulations. All other in-use ingredients are reported to be used in less than 15 formulations. Potassium cetyl phosphate has the highest concentration of use in a leave-on formulation, that is, up to 8.3% in mascara products. The highest concentration of use reported for products resulting in leave-on dermal exposure is 4.2% trioleyl phosphate in “other” make-up preparations.
A single oral dose of phosphoric acid, 2-ethylhexyl ester to F344 rats was completely hydrolyzed to phosphate and 2-ethylhexanol. The ester was reported to be efficiently absorbed, metabolized, and excreted quantitatively by the body and there was no indication of accumulation.
The alkyl phosphate ingredients are relatively nontoxic in single-dose studies. The dermal LD50 in rats was >2 g/kg bw for C20-22 alkyl phosphate, oleyl phosphate, and 45.45% and 80% dicetyl phosphate. The oral LD50 in rats was >2 g/kg for 1-octadecanol, phosphate, potassium salt, potassium C9-15 alkyl phosphate, C20-22 alkyl phosphate, oleyl phosphate, and dimyristyl phosphate. The oral LD50 of 25% potassium lauryl phosphate was 10.49 g/kg, and for 10% cetyl phosphate it was >4.7 g/kg. In both the mouse and rat, the oral LD50 of a 25% suspension of dicetyl phosphate was >5 g/kg. In a 4-hour inhalation study, the LC50 of 1% aq phosphoric acid, C16-18 alkyl esters, potassium salts was >200 µl/L.
In 14-day studies, potassium lauryl phosphate had a NOAEL of 600 mg/kg bw/d for both males and females, and oral administration of up to 1000 mg/kg bw/d sodium lauryl phosphate for 14 days did not result in any adverse effects; no remarkable effects were observed with up to 1000 mg/kg bw/d C20-22 alkyl phosphate by gavage. The NOELs of myristyl phosphate in a 28-day dietary study were 1564 mg/kg bw/d for males and 227 mg/kg bw/d for females; because an increased incidence in focal corticomedullary mineralization was observed in females fed 505 and 1564 mg/kg bw/d, the NOAEL was 1564 mg/kg bw/d for females. Oleyl phosphate had a NOAEL of 1000 mg/kg bw/d for male and female rats in a 28-day gavage study. In a 91-day gavage study, potassium C9-15 alkyl phosphate had a benchmark dose lower confidence limit of 240.3 mg/kg bw/d in males and females.
Potassium C9-15 alkyl phosphate was not embryotoxic, fetotoxic, or teratogenic in rats dosed by gavage on days 6 to 15 of gestation; the NOELs for developmental toxicity, embryotoxicity, fetotoxicity, and teratogenicity were 361 mg/kg bw/d (a.i.), and the NOEL and NOAEL for maternal toxicity were 36.1 and 361 mg/ kg bw/d (a.i.), respectively. For C20-22 alkyl phosphate, the NOELs for reproduction (mating and fertility) and neonatal toxicity, and the NOAEL for parental toxicity, were 1000 mg/kg bw/d in rats. Oleyl phosphate also was not a reproductive toxicant in rats; in a gavage study, the NOAELs were 1000 mg/kg bw/d for maternal toxicity, reproductive performance in male and female rats, and development in F1 offspring. In a reproductive study in rats with sodium lauryl phosphate, the NOAEL for parental male and female animals and the NOEL for the F1 generation was 1000 mg/kg bw/d.
1-Octadecanol, phosphate, and potassium salt were negative in an Ames test, and cetyl phosphate was not genotoxic in a mammalian cell gene mutation assay. Potassium lauryl phosphate, C20-22 alkyl phosphate, and oleyl phosphate were not mutagenic in the Ames test, mammalian cell gene mutation assay, or chromosomal aberration assay.
Some alkyl phosphates were not irritating to the skin, whereas several were irritating, but not sensitizing, in nonhuman studies. C20-22 alkyl phosphate, applied neat, was not irritating to rat skin, nor was it a sensitizer in a GPMT. Undiluted phosphoric acid, C16-18 alkyl esters, potassium salts produced some signs of irritation in the abraded skin of rabbits. Potassium lauryl phosphate was irritating to rabbit skin as a 77% paste in one study, and highly irritating to rabbit skin in another (concentration not specified); it was not a sensitizer in a GPMT. Cetyl phosphate and lauryl phosphate were not sensitizers in GPMTs, but challenge concentrations of 10% and 40% cetyl phosphate and an epidermal induction concentration of 12.5% lauryl phosphate were irritating. Undiluted oleyl phosphate was irritating to rat skin; concentrations up to 5% did not demonstrate a potential for sensitization in a local lymph node assay. (Alternative studies with oleyl phosphate did not demonstrate a potential for skin irritation or corrosion.) Dicetyl phosphate was not irritating to rat skin as an 80% paste, was not irritating to rabbit skin when prepared as a 46.5% paste in olive oil (w/w), and was not a sensitizer in a GPMT. Dimyristyl phosphate, applied under an occlusive patch for 4 hours, was neither irritating to rabbit skin, nor was it an irritant or sensitizer in a GPMT at a concentration of 75% in distilled water.
C20-22 alkyl phosphate, 5% in an emulsion, was not an irritant or a sensitizer in an HRIPT completed in 49 patients. In an HRIPT completed in 108 patients, a hair cream containing 1.0% dicetyl phosphate was not a sensitizer.
Some of the alkyl phosphates are reported to be ocular irritants. A 10% solution of potassium lauryl phosphate, tested as a 50% dilution in the HET-CAM, demonstrated moderate ocular irritation potential, and oleyl phosphate demonstrated the potential to be corrosive and a severe ocular irritant in an in vitro eye corrosive and severe irritant study. In rabbit eyes, potassium lauryl phosphate was an irritant, C20-22 alkyl phosphate was a moderate irritant and dicetyl phosphate was slightly irritating. However, a 3% potassium cetyl phosphate solution, tested as a 50% dilution in a HET-CAM, demonstrated practically no ocular irritation potential, and phosphoric acid, C16-18 alkyl esters, potassium salts and dimyristyl phosphate 35 were classified as non-irritating to rabbit eyes.
Discussion
The Panel reviewed the safety of 28 ingredients in the alkyl phosphate family; these ingredients share a common phosphate core structure, varying by the identity of the alkyl chains attached. The Panel acknowledged that much of the data were obtained from ECHA summaries, and in several instances, structural analogs were used as supporting substances to provide read-across. The Panel found this read-across appropriate to support the safety of the alkyl phosphates named in this report because the analogs contained chain length distributions that had a great deal of overlap with the alkyl phosphate ingredients.
The Panel noted there were little to no safety test data on the triester phosphates included in this safety assessment. However, based on the molecular weights of the triesters (≥603), and the calculated log P values (≥17.02), the Panel does not expect these ingredients to penetrate through the skin. Therefore, the Panel determined that it was appropriate to include the triesters among the ingredients in this safety assessment and to conclude on their safety.
Although there were no impurities data, based on the method of manufacture and the absence of adverse effects in repeat oral toxicity studies, the Panel was not concerned with the absence of these data.
Finally, the Panel was concerned that the potential exists for ocular and/or dermal irritation with the use of products formulated using alkyl phosphates, and the Panel specified that products containing alkyl phosphates must be formulated to be non-irritating. Specifically, the Panel recognized the potential for ocular irritation when potassium cetyl phosphate is used at up to 8.3% in mascara products. Additionally, some of the alkyl phosphates were irritating to the skin of animals; however, these studies were conducted with concentrations that were much greater than the concentrations reported to be used in cosmetics.
Conclusion
The CIR Expert Panel concluded the following 28 alkyl phosphates are safe in the present practices of use and concentration in cosmetics when formulated to be non-irritating: Potassium cetyl phosphate Potassium C9-15 alkyl phosphate Potassium C11-15 alkyl phosphate* Potassium C12-13 alkyl phosphate Potassium C12-14 alkyl phosphate* Potassium lauryl phosphate C8-10 alkyl ethyl phosphate* C9-15 alkyl phosphate C20-22 alkyl phosphate Castor oil phosphate Cetearyl phosphate* Cetyl phosphate Disodium lauryl phosphate* Disodium oleyl phosphate* Lauryl phosphate Myristyl phosphate* Octyldecyl phosphate* Oleyl ethyl phosphate* Oleyl phosphate* Sodium lauryl phosphate* Stearyl phosphate Dicetyl phosphate Dimyristyl phosphate* Dioleyl phosphate Tricetyl phosphate* Trilauryl phosphate* Trioleyl phosphate Tristearyl phosphate*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Fiume M. contributed to conception and design, acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld W. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Belsito D. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Hill R. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Klaassen C. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Liebler D. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Marks J. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Shank R. contributed to conception and design, and critically revised manuscript; Slaga T. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Snyder P. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Gill L. contributed to conception and design, analysis and interpretation, and critically revised manuscript; Heldreth B. contributed to analysis and interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
