Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reassessed the safety of 2-pyrrolidone-5-carboxylic acid (PCA) and sodium PCA; the Panel added 3 previously unreviewed salts (calcium, magnesium, and potassium) of PCA to this safety assessment. 2-Pyrrolidone-5-carboxylic acid and its salts are reported to function in cosmetics as skin conditioning agents—humectants. The Panel reviewed the data from the 1999 report of PCA and sodium PCA, as well as additional data included in this report, to determine the safety of these ingredients. The Panel concluded that PCA and its salts are safe in cosmetics in the present practices of use and concentration; additionally, these ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed.
Introduction
In 1999, the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) published a safety assessment of 2-pyrrolidone-5-carboxylic acid (PCA) and sodium PCA. 1 Based on the data presented in that assessment, the Panel concluded these ingredients are safe as used in cosmetic formulations; additionally, the Panel stated that these ingredients should not be used in cosmetic products containing N-nitrosating agents.
The Panel found that the data included in the original safety assessment also supported the safety of the calcium, magnesium, and potassium salts of PCA as used in cosmetics; therefore, the safety assessment was reopened to include these salts. The 5 ingredients are reported to function as skin conditioning agents—humectants in cosmetic formulations 2 (Table 1).
Definition, Structure, and Function.
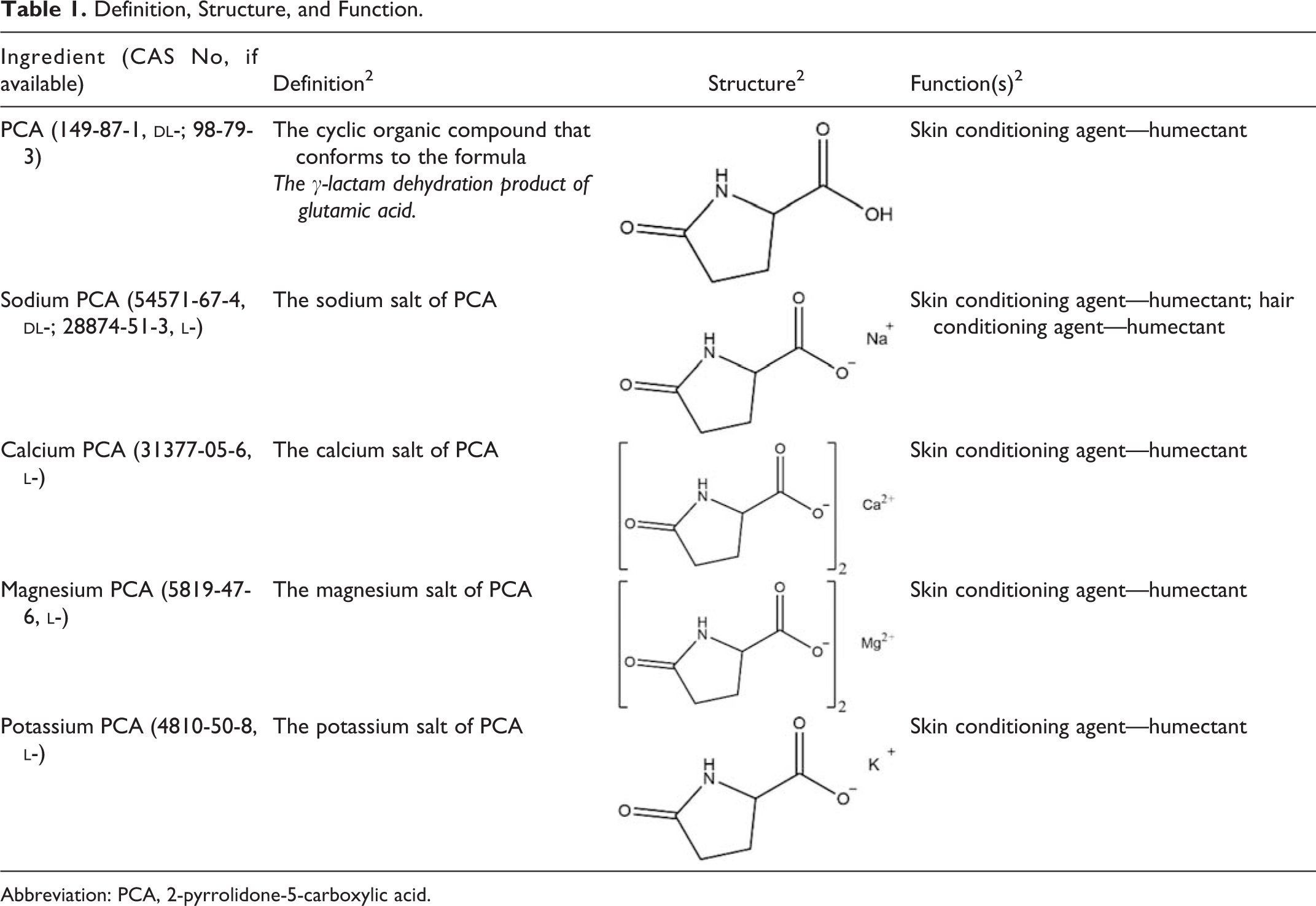
Abbreviation: PCA, 2-pyrrolidone-5-carboxylic acid.
The complete original report on PCA can be found on the CIR website, http://www.cir-safety.org/ingredients. Therefore, only brief summary data from the original report are included, as appropriate; these summaries are identified by italicized text. Please refer to the original report for detailed information.
Additional data were found on the European Chemicals Agency (ECHA) website
3
and are included in this report. The ECHA website provides summaries of information generated by industry, and it is those summary data that are reported in this safety assessment when ECHA is cited. Unless indicated otherwise in this assessment, all of the ECHA data pertain to the
Chemistry
Definition and Structure
2-Pyrrolidone-5-carboxylic acid, also known as pyroglutamic acid, is an internal amide of
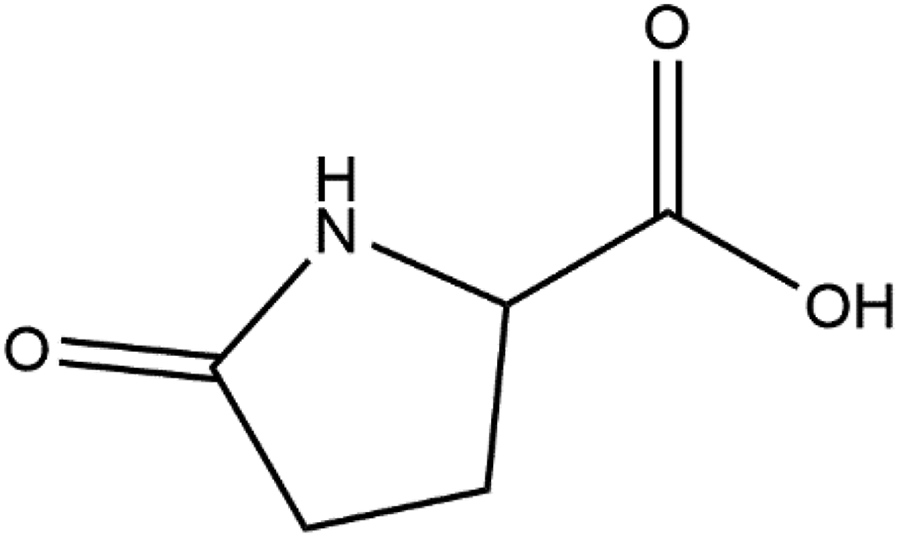
2-Pyrrolidone-5-carboxylic acid (PCA).
Chemical and Physical Properties
2-Pyrrolidone-5-carboxylic acid is an orthorhombic bisphenoidal crystal with a molecular weight of 129.11 Da. 1 It is soluble in water, alcohol, and acetone. 2-Pyrrolidone-5-carboxylic acid is nonhygroscopic, but its sodium salt is extremely hygroscopic. The ultraviolet absorption spectrum of PCA indicates very weak absorption from 320 to 240 nm and strong absorption from 240 nm to shorter wavelengths.
Nitrosation
The N-nitrosation of PCA was investigated under conditions simulating those in the stomach following a meal. 1 2-Pyrrolidone-5-carboxylic acid was reacted with sodium nitrite at pH 2.5 and 37°C, and sulfamic acid was added to the mixture. The initial rate of reaction was very slow, and the rate constant was 1.23 × 10− 3 M/min. The investigators noted that this rate value was 1.7% of that observed with hydantoic acid and 0.03% of that of nitrosomethylurea formation.
Methods of Manufacture
2-Pyrrolidone-5-carboxylic acid is prepared from
Impurities
No by-products are reported in the production of PCA and sodium PCA from glutamic acid and sodium glutamate, respectively. 1 It could be expected that some dimer or polymer of glutamic acid would be found, but none was detected. However, glutamic acid and sodium glutamate are possible impurities.
Natural Occurrence
2-Pyrrolidone-5-carboxylic acid is a naturally occurring component of mammalian tissue; 270 µmol/g wet weight was found using ion-exchange chromatography in epidermal scrapings taken from albino guinea pigs.
1
In further studies with epidermal samples from guinea pigs, humans, dogs, rats, and mice, the total amount of free PCA was 186.0, 44.9, 30.9, 21.3, and 19.0 µmol/g wet weight, respectively. Optical rotatory dispersion studies of PCA isolated from guinea pig skin indicated that the epidermal PCA was the
Use
Cosmetic
2-Pyrrolidone-5-carboxylic acid and its salts are reported to function in cosmetics as skin conditioning agents—humectants 2 (Table 1). The intended use of ingredients that function as humectant skin conditioning in cosmetics is to increase the water content of the top layers of skin.
The US Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetic formulations as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). In 2014, VCRP data obtained from the FDA 4 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 5 indicate that PCA and its salts are all in-use in cosmetic formulations.
The frequency of use of PCA and sodium PCA has increased since the original assessment. 2-Pyrrolidone-5-carboxylic acid and sodium PCA currently are reported to be used in 106 and 1289 formulations, respectively; in 1996, PCA and sodium PCA were reported to be used in 25 and 437 formulations, respectively (Table 2). The maximum concentration of use of sodium PCA in rinse-off formulations has increased; however, the maximum concentration of use of PCA in leave-on formulations and of PCA and sodium PCA in rinse-off formulations has not increased. Sodium PCA currently is reported to be used at a maximum of 2.5% in leave-on formulations (ie, nail creams and lotions) and 3% in rinse-off formulations (ie, in skin cleansing preparations); the current reported maximum concentration of use of PCA is 1.9% in face powders. In the original assessment, PCA and sodium PCA were reported to be used at a maximum of 2.5% in leave-on formulations (ie, a moisturizer) and 1.2% in rinse-off formulations (a cleanser); however, it was stated that the recommended concentration of use was in the range of 0.2% to 4%. The calcium, magnesium, and potassium salts of PCA are reported to be used at 0.75% or less in cosmetic formulations (Table 3).
Current and Historical Frequency and Concentration of Use According to Duration and Exposure.
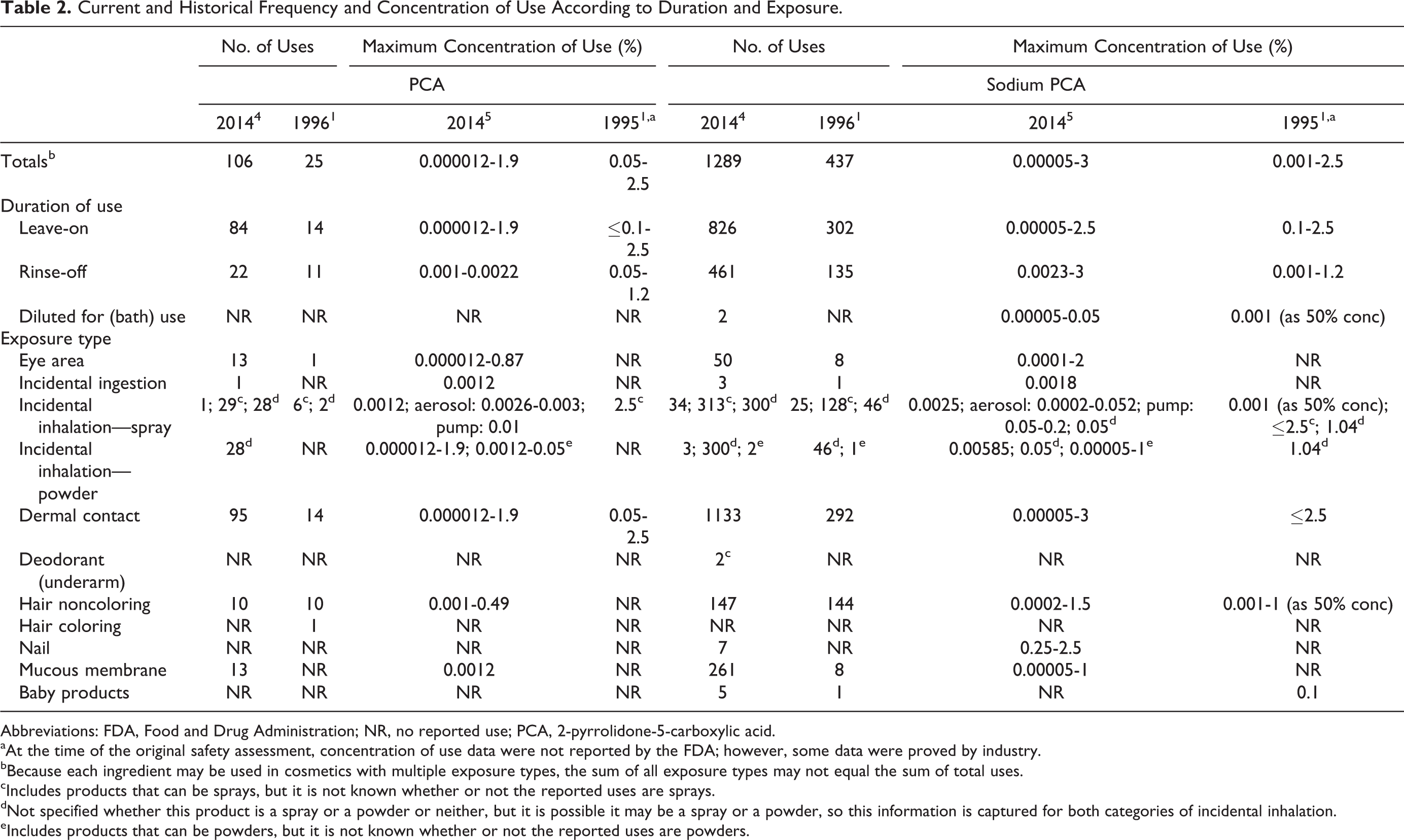
Abbreviations: FDA, Food and Drug Administration; NR, no reported use; PCA, 2-pyrrolidone-5-carboxylic acid.
aAt the time of the original safety assessment, concentration of use data were not reported by the FDA; however, some data were proved by industry.
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
cIncludes products that can be sprays, but it is not known whether or not the reported uses are sprays.
dNot specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
eIncludes products that can be powders, but it is not known whether or not the reported uses are powders.
Current Frequency and Concentration of Use According to Duration and Exposure.
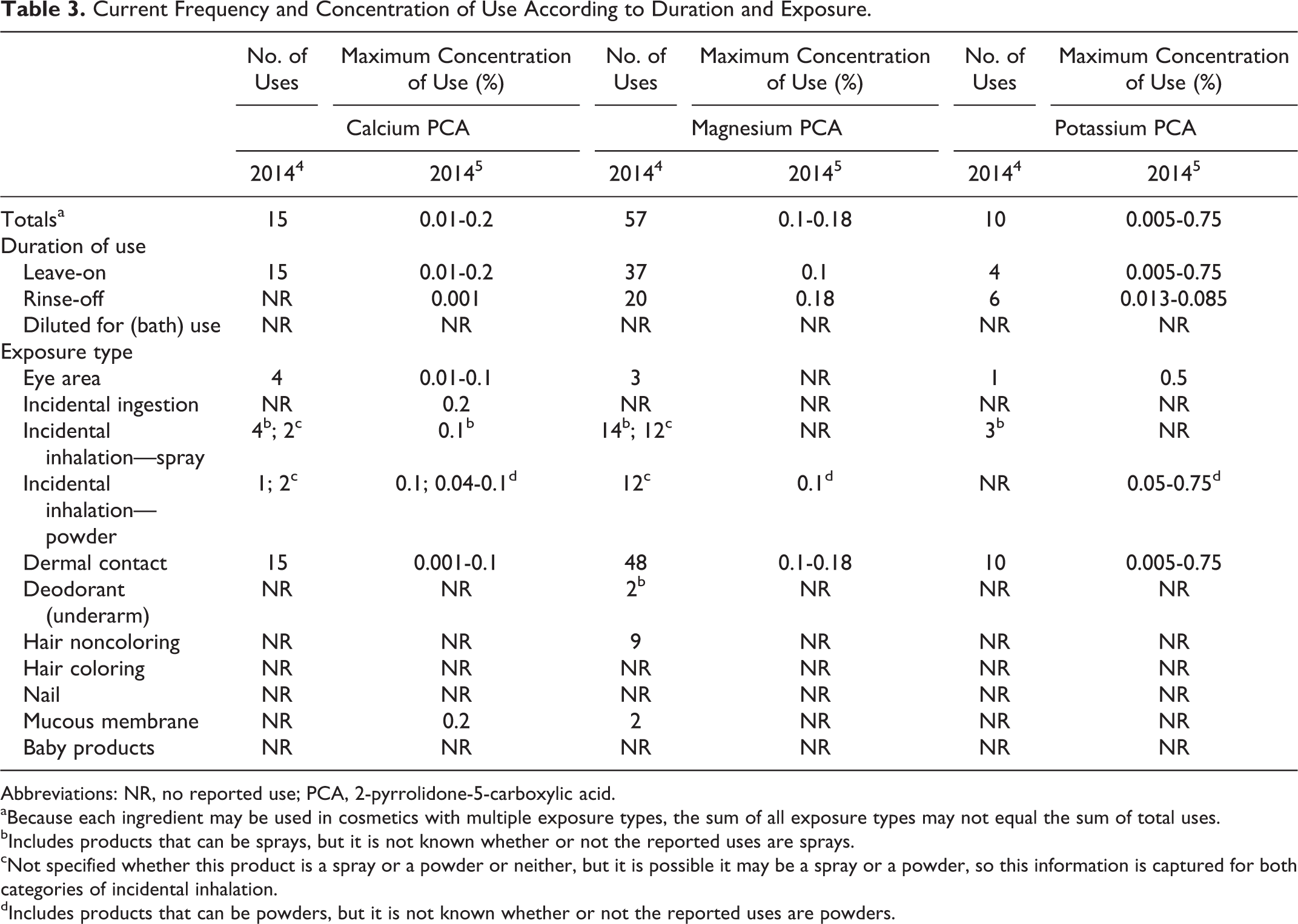
Abbreviations: NR, no reported use; PCA, 2-pyrrolidone-5-carboxylic acid.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes products that can be sprays, but it is not known whether or not the reported uses are sprays.
cNot specified whether this product is a spray or a powder or neither, but it is possible it may be a spray or a powder, so this information is captured for both categories of incidental inhalation.
dIncludes products that can be powders, but it is not known whether or not the reported uses are powders.
Many of the reported uses are in the eye area; the highest concentration of use reported for eye products is 2% sodium PCA in an eye lotion. According to VCRP data, sodium PCA is used in 5 baby products; however, concentration of use data were not reported for this use category. Use in products applied to the mucous membranes or in products that could possibly be ingested has also been reported at low concentrations.
2-Pyrrolidone-5-carboxylic acid and sodium PCA are used in cosmetic sprays and could possibly be inhaled; for example, sodium PCA is used at up to 0.2% in pump hair sprays and PCA is used at up to 1.9% in face powders. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 6,7 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 8,9 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 9 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays. 2-Pyrrolidone-5-carboxylic acid and its salts are listed in the European Union inventory of cosmetic ingredients. 10
Toxicokinetics
The major pathway by which
Toxicological Studies
The oral LD50 of sodium PCA was 10.4 g/kg for male mice, and the oral LD50 of 50% sodium PCA was >2.0 g/kg for rats.
1
No adverse effects were observed in either a short-term study using rats fed 1.5% PCA or in subchronic studies with rats fed diets containing up to 8%
Single-Dose (Acute) Toxicity
Dermal
Occlusive patches with 2 g/kg sodium PCA were applied to the backs of 5 male and 5 female rats for 24 hours. 3 No mortality was reported.
Oral
Two groups of 3 female rats were given a single dose of 2 g/kg sodium PCA in distilled water by gavage. 3 None of the animals died.
Reproductive and Developmental Toxicity
Sodium PCA was not a developmental or reproductive toxicant in rats. 3 Groups of 12 male and 12 female Wistar rats were dosed by gavage once daily with 0, 62.5, 250, or 1000 mg/kg body weight/day sodium PCA in water. The females were dosed for 2 weeks prior to mating until day 4 of lactation; the males were dosed for 14 days prior to mating and for 14 days during mating. No adverse effects on any measured parameters in the parents or offspring were observed. The no observable adverse effect level (NOAEL) for maternal and reproductive toxicity was listed as 1000 mg/kg body weight/day.
Ocular Irritation
No ocular irritation was observed when 50% aqueous sodium PCA were instilled into the conjunctival sac of the eye of rabbits. 1
The ocular irritation potential of sodium PCA was evaluated in vitro using a chicken eye test method for identifying ocular corrosives and severe irritants; sodium PCA was classified as nonirritating. 3 Sodium PCA also was found to be nonirritating to rabbit eyes in an in vivo study. Sodium PCA, 0.1 g neat, was instilled into the conjunctival sac of one eye of 3 New Zealand white rabbits, and the eyes were rinsed after 1 hour. Slight conjunctival redness and discharge were observed in all 3 animals 1 hour after administration; all reactions were reversed within 48 hours, and sodium PCA was considered nonirritating.
Genotoxicity
2-Pyrrolidone-5-carboxylic acid and sodium PCA were not mutagenic in a Salmonella mutagenicity assay with or without metabolic activation, and PCA was not considered clastogenic in a chromosome damage assay. 1
Sodium PCA in distilled water was not genotoxic in an Ames test, mammalian cell gene mutation assay, or chromosomal aberration assay, with or without metabolic activation. 3 In the Ames test, Salmonella typhimurium strains TA1535, TA1537, TA98, and TA100 and Escherichia coli strain WP2 uvrA were exposed to 78.125 to 5,000 µg/plate. Mouse lymphoma L5178Y cells were exposed to 20.58 to 5,000 µg/mL in the mammalian cell gene mutation assay, and Chinese hamster lung fibroblasts V79 cells were exposed to 312.5 to 5,000 µg/mL in the chromosomal aberration assay. Appropriate solvent and positive controls gave valid results in each study.
Carcinogenicity
Carcinogenicity data were not found in a search of the published literature, nor were unpublished data provided.
Irritation and Sensitization
Dermal Irritation and Sensitization
In vitro
Undiluted sodium PCA was considered to be nonirritating in a reconstructed human epidermis model test using the EpiSkin model. 3 Twenty milligrams of the test article was applied to the skin model for 15 minutes.
Nonhuman
Sodium PCA was nonirritating when applied to the skin of guinea pigs and rabbits at concentrations up to 50%. 1 No evidence of dermal sensitization was observed when guinea pigs were induced with 2% to 50% aqueous sodium PCA and challenged with 5% aqueous sodium PCA. Sodium PCA was noncomedogenic in rabbits.
Sodium PCA was not a sensitizer in a guinea pig maximization test (GPMT). 3 Ten female Hartley guinea pigs were exposed to 20% aqueous sodium PCA at both intradermal and epidermal induction. Intradermal induction involved paired injections of 0.1 mL Freund complete adjuvant (FCA), 0.1 mL of the test solution, and 0.1 mL of the test material and FCA. Four days later, 10% sodium lauryl sulfate in petrolatum was applied to the skin of the animals, and a 48-hour occlusive patch with 0.5 mL sodium PCA was applied the next day. Twenty days after the epidermal induction, the animals were challenged using a 24-hour occlusive patch with 10% sodium PCA. Two challenge control groups (5 animals/dose) and a positive control group (10 animals exposed to 2,4-dinitrochlorobenzene) were used. No reactions were observed at challenge with sodium PCA.
The sensitization potential of the
Human
In a clinical study of dermal irritation using open patch test methods on various sites of the body, 2 of 13 volunteers had reactions to 6.25% sodium PCA applied to their backs and 3 volunteers developed erythema when concentrations of ≥12.5% sodium PCA were applied. 1 These reactions disappeared within 30 minutes. No reactions were observed when sodium PCA was applied to the skin of the forehead, cheek, or neck. A formulation containing 2.0% sodium PCA was negative in a mini-cumulative irritation test. In another study, no significant irritation was observed when 46 volunteers were treated with 30% sodium PCA using open patch test methods. Negative results were also obtained when 46 volunteers were tested with concentrations up to 32% sodium PCA using occlusive patches. Provocative tests of 0.2% sodium PCA using occlusive patches were also negative.
Clinical studies using 39 subjects indicated that 32% aqueous sodium PCA is not a sensitizer. A maximization test of a cosmetic formulation containing 2.0% sodium PCA was also negative.
Phototoxicity
No phototoxic effects were observed in 10 guinea pigs treated topically with 1% aqueous sodium PCA. 1 In a clinical study using 39 subjects, 32% aqueous sodium PCA was not a photosensitizer.
Summary
In 1999, the Panel concluded that PCA and sodium PCA were safe as used in cosmetics and that these ingredients should not be used in cosmetic products containing nitrosating agents. This report was reopened to add 3 previously unreviewed salts of PCA, that is, calcium PCA, magnesium PCA, and potassium PCA. All 5 ingredients are reported to function as skin-conditioning agents—humectant in cosmetic formulations.
Voluntary Cosmetic Registration Program data obtained from the FDA, and data received in response to surveys of the maximum reported use concentration by category that were conducted by the Council, indicate that PCA and the 4 PCA salts are all in use in cosmetic formulations. Sodium PCA has the most reported uses and the highest concentration of use; it is reported to be used in 1289 formulations, and the maximum leave-on and rinse-off concentrations of use are 2.5% in nail creams and lotions and 3% in skin cleansing preparations, respectively. The frequency of use of PCA and sodium PCA has increased since the original safety assessment; the concentration of use of sodium PCA in rinse-off products has increased from 2.5% to 3.0%, but there has been no increase in use concentrations of PCA or sodium PCA in leave-on formulations.
Sodium PCA (tested as the
Sodium PCA (
Sodium PCA was not a dermal or ocular irritant, nor was it a sensitizer. Dermally, undiluted sodium PCA was considered to be nonirritating in a reconstructed human epidermis model and in ocular testing. Sodium PCA was classified as nonirritating in both in vitro and rabbit eyes. Neither the
Discussion
A safety assessment of PCA and sodium PCA was published in 1999, with the conclusion that these ingredients are safe as used in cosmetic formulations; additionally, the Panel stated that these ingredients should not be used in cosmetic products in which N-nitroso compounds can be formed. Publicly available data found on the ECHA website provided additional data that supported the original conclusion, and therefore, the additional data were not a reason to reopen the safety assessment. However, the Panel found that the existing data do support the safety of 3 previously unreviewed salts of PCA and thus reopened the safety assessment to add these salts.
The Panel noted that PCA and sodium PCA are used in products that could be incidentally inhaled; reported concentrations of use include 0.2% sodium PCA in pump hair sprays and up to 1.9% PCA in face powders; this fact was addressed in the discussion of the original safety assessment as well. Although there were no inhalation data available, the Panel was not concerned with the use of these ingredients in such formulations. The Panel noted that in aerosol products, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that PCA (ie, 2-pyrrolidone-5-carboxylic acid), calcium PCA, magnesium PCA, potassium PCA, and sodium PCA are safe in cosmetics in the present practices of use and concentration described in this safety assessment. 2-Pyrrolidone-5-carboxylic acid and its salts should not be used in cosmetic products in which N-nitroso compounds can be formed.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Authors Contribution
M.M.F., W.F.B., D.V.B., R.A.H., C.D.K., D.C.L., J.G.M., R.C.S., T.J.S., P.W.S., and L.J.G. contributed to conception and design. M.M.F. contributed to acquisition, analysis, and interpretation. W.F.B., D.V.B., R.A.H., C.D.K., D.C.L., J.G.M., R.C.S., T.J.S., P.W.S., L.J.G., and B.H. contributed to analysis and interpretation. MMF drafted the manuscript. M.M.F., W.F.B., D.V.B., R.A.H., C.D.K., D.C.L., J.G.M., R.C.S., T.J.S., P.W.S., L.J.G., and B.H. critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
