Abstract
Exposure to persistent organic pollutants including dichlorodiphenyltrichloroethane (DDT) induces insulin resistance. But the mechanism is not clearly known. The present study was designed to explore the effect of subtoxic DDT exposure on (1) insulin-stimulated glucose uptake, (2) malondialdehyde (MDA) level and total antioxidant content, (3) activation of redox sensitive kinases (RSKs), and (4) insulin signaling in rat L6 myoblast-derived myotubes. Exposure to 30 mg/L and 60 mg/L of DDT for 18 hours dose dependently decreased glucose uptake and antioxidant content in myotubes and increased MDA levels. The exposures did not alter tumor necrosis factor α (TNF-α) level as determined by enzyme-linked immunosorbent assay, despite decreased messenger RNA expression following DDT exposures. Phosphorylation of c-Jun N-terminal kinases and IκBα, an inhibitory component of nuclear factor κB (NFκB), was increased, suggesting activation of RSKs. The level of tyrosine phosphorylation of insulin receptor substrate 1 and serine phosphorylation of protein kinase B (Akt) on insulin stimulation decreased in myotubes with exposure to subtoxic concentrations of DDT, but there was no change in tyrosine phosphorylation level of insulin receptors. We conclude that subtoxic DDT exposure impairs insulin signaling and thereby induces insulin resistance in muscle cells. Data show that oxidative stress-induced activation of RSKs is responsible for impairment of insulin signaling on DDT exposure.
Introduction
Rachel Carson in her environmental classic Silent Spring claimed that the organochlorine pesticides, especially dichlorodiphenyltrichloroethane (DDT), enter the biosphere after their use in agriculture and health programs. These pesticides accumulate in the environment due to their long half-life. These organochlorines not only kill the pests but also enter into food chain and reach nontarget organisms including humans at high concentration to harm them. 1 -4 Exposures to persistent organic pollutants (POPs) are not only associated with pathogenesis of cancer, 5 genotoxicity, 6 immunotoxicity 5,7,8 and intrauterine growth retardation (IUGR), 9 but they have recently been shown to play a role in increased prevalence of type 2 diabetes mellitus (T2DM). There are many cross-sectional and longitudinal studies, which report that exposure to POPs increases the risk of insulin resistance and diabetes. 10 -13
Some previous studies reported that DDT exposure causes oxidative stress. 14,15 DDT exposure is known to increase the level of serum tumor necrosis factor α (TNF-α), a proinflammatory cytokine. 16 Oxidative stress and TNF-α which is produced from adipocytes and other immunocytes activate redox-sensitive kinases (RSKs; eg, nuclear factor κB [NFκB], c-Jun N terminal kinase [JNK], etc). Nuclear factor κB has an inhibitory component (IκBα) attached to p50–p65 complex. In the presence of various stimuli (eg, oxidative stress, TNF-α, and so on), IκBα is phosphorylated and separated from p50–p65 complex. Then p50–p65 complex is translocated to nucleus to modulate expression of various genes. 17 Hence, the level of phosphorylation of IκBα is considered to be an index of activation of NFκB. These RSKs interfere with insulin signaling and cause insulin resistance. 18 -20
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder characterized by persistent high glucose level in blood. It is caused by insulin resistance and/or defective insulin secretion from pancreatic β-cells (β-cells dysfunction). Glucose homeostasis is maintained when the production and the utilization of glucose are balanced whereas in diabetes it is impaired. 21 -23 Skeletal muscles play an important role in maintenance of blood glucose homeostasis by taking up glucose from blood in response to insulin stimulation. 24,25 The elevation of glucose level in blood triggers the release of insulin from pancreatic β-cells. Then insulin enters into the bloodstream and binds to insulin receptor (IR) that causes phosphorylation of tyrosine moieties of β subunits of IR (IR β). This triggers insulin signaling cascade leading to tyrosine phosphorylation of insulin receptor substrates (IRSs) including IRS-1. That, in turn, phosphorylates Akt (also known as protein kinase B) at its serine moieties. Finally, the phosphorylated Akt mediates translocation of glucose transporter type 4 (GLUT4) to the membrane of insulin-sensitive cells. Glucose transporter type 4 transports circulating glucose from blood to the tissues by facilitated diffusion. In T2DM, insulin-signaling mechanism is reported to be impaired. 24,26
Rat L6 myoblast cells can be differentiated into myotubes that behave like skeletal muscles. L6-derived myotubes possess complete insulin-signaling cascade and take up glucose on insulin stimulation. L6-derived myotubes are widely used in diabetic research for studying insulin-signaling cascade and insulin-stimulated glucose uptake. 27 In view of the above, in this study we tested the hypothesis that exposure to subtoxic levels of DDT would create oxidative stress, activate the RSKs (e.g., NFκB and JNK), alter insulin signaling, and impair insulin-induced glucose uptake in L6-derived myotubes.
Materials and Methods
Chemicals and Reagents
Dichlorodiphenyltrichloroethane (DDT; 99% pure) was obtained as gift from Hindustan Insecticide Limited, India. Dimethyl sulfoxide (DMSO), insulin, protease-phosphatase inhibitor cocktail (PPI), phenylmethylsulfonyl fluoride (PMSF), radioimmunoprecipitation assay (RIPA) lysis buffer, and agarose were purchased from Sigma-Aldrich (St Louis, Missouri). Total RNA extraction kit was procured from Geneaid Biotech Ltd. (New Taipei City, Taiwan). High-glucose Dulbecco modified Eagle medium (DMEM), fetal bovine serum (FBS), and penicillin–streptomycin–amphotericin B were purchased from Gibco, Invitrogen (Carlsbad, California). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and Trypsin-EDTA were purchased from Hi-media laboratories (Mumbai, India). Bicinchoninic Acid (BCA)-based protein assay kit, complementary DNA (cDNA) synthesis kit and SYBR Green master mix were procured from Thermo Scientific (Waltham, Massachusetts, USA). 3,3’,5,5’-Tetramethylbenzidine (TMB) was obtained from GeNei (Bangalore, India). Total anti-oxidant assay kit of Randox (Crumlin, County Antrim, United Kingdom) was used for assay of total anti-oxidant content.
Primary antibodies against phospho-IR (tyrosine 1361) and phospho-Akt (serine 473) were purchased from Abcam (Cambridge, Massachusetts). Primary antibody against phospho-IRS-1 (tyrosine 1222) and horseradish peroxidase (HRP)-conjugated anti-rabbit secondary antibody were from Cell Signaling Technologies (Danvers, Massachusetts). Primary antibodies against phospho-JNK (threonine 183/tyrosine 185), phospho-IκBα (serine 32/serine 36), and HRP-conjugated anti-mouse secondary antibody were from Sigma Aldrich.
Rat Myoblast (L6) Culture and Differentiation
Rat myoblast (L6) cell line was purchased from the National Centre for Cell Sciences (Pune, Maharashtra, India). L6 myoblast cells were cultured in high-glucose DMEM supplemented with 10% FBS and appropriate antibiotic–antimycotic (100 units/mL of penicillin G, 100 μg/mL of streptomycin sulfate, 0.25 μg/mL amphotericin B) in a humidified atmosphere with 5% CO2 at 37°C. For differentiation, cells were allowed to reach 70% to 80% confluence and then the medium was replaced with 2% FBS containing high-glucose DMEM (containing 100 units/mL penicillin, 100 μg/mL of streptomycin sulfate, and 0.25 μg/mL amphotericin B). The differentiation of myoblasts into myotubes was confirmed by the changes in morphology and fusion of cells as seen under the inverted microscope (Motic, model: AE31; Viking Way Richmond, British Columbia, Canada). In this study, DDT treatment was started after successful completion of differentiation. To assay the insulin signaling, RSKs, malondialdehyde (MDA) level, antioxidant content, and TNF-α secretion, L6 myoblasts were grown and differentiated in 75-cm2 culture flasks. Glucose uptake and MTT assay were done in 96-well culture plates (Tarsons India Pvt Ltd., Kolkata, India).
Subtoxic Dose Determination
Rat L6 myoblast cells (104 cells) were seeded in 96-well culture plates in high-glucose DMEM (containing 10% FBS and 1% antibiotic plus antimycotic) and kept in humidified atmosphere of 5% CO2 incubator at 37°C to achieve 70% to 80% confluence. At 70% to 80% confluence, differentiation was induced with high-glucose DMEM containing 2% FBS. The stock of DDT (1000 mg/mL) was prepared in DMSO and further diluted in high-glucose DMEM (containing 2% FBS). Differentiated myotubes in 96-well culture plates were treated for 18 hours with various concentrations of DDT (0 [vehicle-treated control], 1, 3, 6, 10, 30, 60, 100, 300, 600, 1000 mg/L) for cytotoxicity assay. Maximum DMSO concentration in 2% DMEM including that in control cultures was 0.1%. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 1 mg/mL stock) was prepared in phosphate-buffered saline (PBS) and sterilized by filtering through 0.22-µm syringe filter. After 18 hours of DDT treatment, MTT solution was added (with final concentration of 0.3 mg/mL), and then, the cells were incubated in CO2 incubator for 3 hours. Then, the medium was removed, and formazan crystals were dissolved by adding DMSO (100 μL) to each well. The absorbance was measured at 570 nm 15 minutes after adding DMSO using a microplate reader (Biorad 680 XR; Biorad, California). The concentration at which there was 50% decrease in optical density (OD) from the control was defined as half maximal inhibitory concentration 50 (IC50) of DDT. The cytotoxic effect (IC50) of DDT as measured by MTT assay was observed at concentration of 1000 mg/L. No observable effect level (NOEL) was found to be 300 mg/L (Figure 1). In this study, subtoxic dose was defined as 1/5th and 1/10th of highest concentration of NOEL. Therefore, we considered the concentration of 30 mg/L and 60 mg/L of DDT to be subtoxic to myotubes. In the present experiment, myotubes were exposed to the above-mentioned concentrations of DDT for 18 hours.
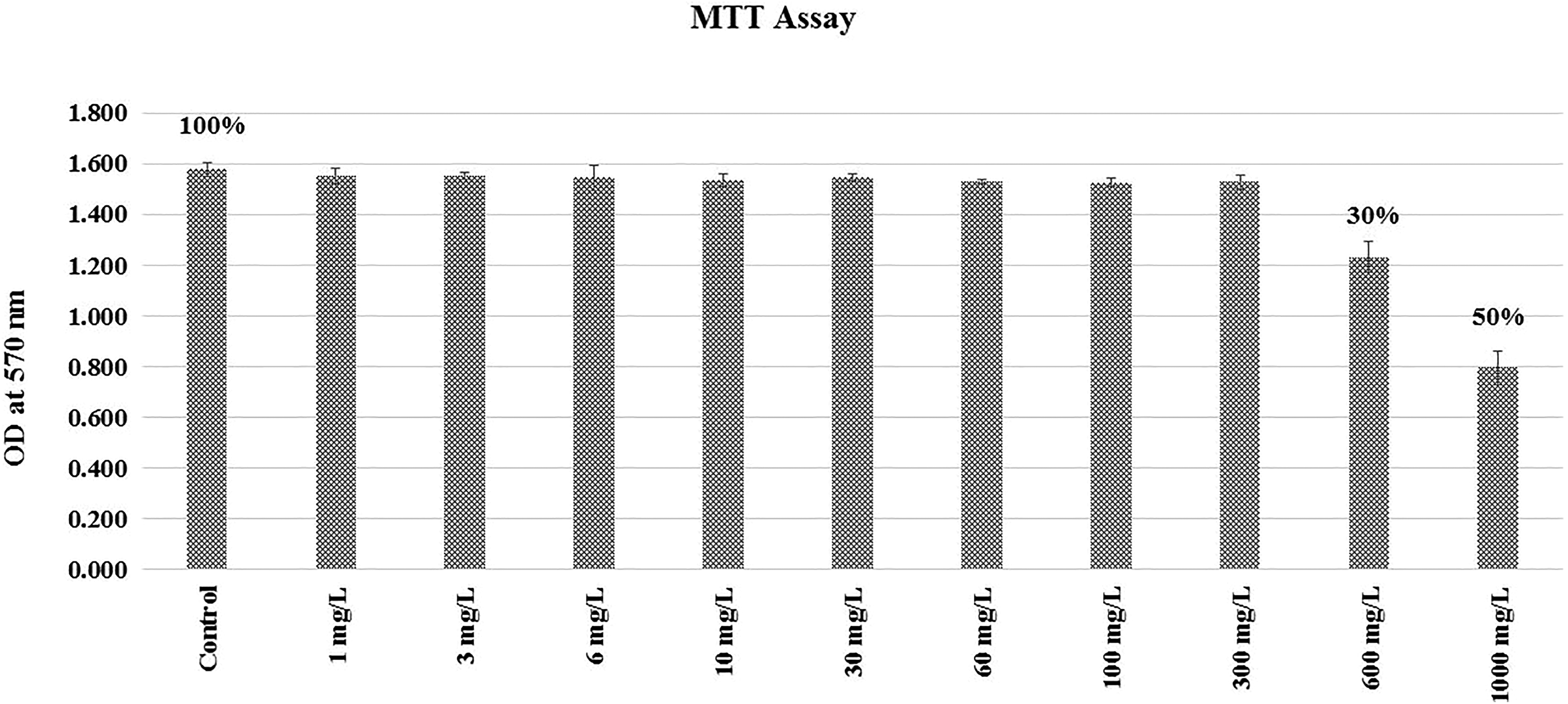
Cytotoxic effect (half maximal inhibitory concentration [IC50]) of dichlorodiphenyltrichloroethane (DDT) measured by 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay.
Subtoxic DDT Treatment and 2-Deoxy-D-glucose-Uptake Assay
L6 myotubes that were grown in 96-well plate were exposed to 30 mg/L and 60 mg/L of DDT for 18 hours. Then, the cells were made to starve of serum for 30 minutes that was followed by stimulation of cells with 1 µM insulin for 15 minutes. Glucose uptake by the cells was assayed by incubation of the cells with 2-deoxyglucose-6-phosphate (2DG6P) which was provided in BioVision’s (Milpitas, California) colorimetric glucose uptake assay kit. The manufacturer protocol that was provided in the kit was followed to measure the glucose uptake by myotubes. Absorbance was measured at 412 nm by a microplate reader (Biorad 680-XR) for calculation of glucose uptake which was expressed as uptake of glucose in pmol per well.
Subtoxic Exposure of Myotubes to DDT and Cell Lysate Preparation
L6 myoblast cells were grown and differentiated into myotubes in 75-cm2 culture flasks. Then, the cells were treated with DDT (at concentration of 30mg/L and 60mg/L after dissolving it in DMSO) for 18 hours at 37°C in a 5% CO2 incubator. Control myotubes were treated with only vehicle (ie, DMSO 0.01%). After 18 hours treatment, the media were decanted and kept for TNF-α assay. Then, the cells were serum starved by adding serum-free media to the culture flasks and incubating those at 37°C for 30 minutes. Then, the cells were stimulated with 1 μM insulin for 15 minutes. After that, harvesting of the cells in ice-cold RIPA lysis buffer containing 1 mM PMSF and PPI was done using cell scrapper. Cells were further lysed mechanically using a hand-held homogenizer, and then the homogenate was centrifuged at 10 000×g for 20 minutes at 4°C to obtain a supernatant (i.e., lysate) for further analysis. Concentration of protein in the lysate was determined using BCA protein assay kit.
Total Antioxidant, MDA, and TNF-α Estimation
Estimation of total antioxidant from the above-mentioned DDT-treated cell lysate was done using a total antioxidant assay kit (Randox, Catalog Number NX 2332) adopted to a random access fully automatic clinical chemistry analyzer (Randox immola), and total antioxidant level was expressed in mmol/gm of proteins. This assay utilizes the suppression by antioxidants of the reaction that converts ABTS (2,2’-Azino-di-[3-ethylbenzthiazoline sulfonate]) in presence of peroxidase (met myoglobin) and H2O2 to a relatively stable blue–green compound that is measured at 600 nm.
Malondialdehyde level was assayed in cell lysate from thiobarbituric acid reactivity using a method as described by Ohkawa et al. 28 The amount of thiobarbituric acid–MDA adduct was measured from OD taken at 532 nm using its extinction coefficient (1.56 × 105 M-1 cm-1) and expressed in nmol/mg of protein.
Tumor necrosis factor α was assayed from the differentiation media (i.e., DMEM with 2% FBS in which pesticide treatment was administered to the cells for 18 hours) and from the cell lysates as well. Tumor necrosis factor α was measured using Rat TNF Alpha PicoKine enzyme-linked immunosorbent assay (ELISA) Kit (BosterBio, Pleasanton CA, USA, Catalog Number EK0526) and expressed in pg/mL. Rat serum was used as a positive control for the assay. Tumor necrosis factor α was measured from undiluted FBS and from FBS diluted to different concentrations with normal saline to check cross reactivity of the assay method with bovine TNF-α.
Polymerase Chain Reaction and Quantitative Reverse Transcription Polymerase chain reaction (qRT-PCR) for TNF-α Messenger RNA Expression
Myotubes with and without DDT treatment were harvested in PBS using cell scrapper. RNA from the harvested myotubes was extracted by using Geneaid total RNA extraction kit (Geneaid, New Taipei, Taiwan) and for that, the kit manufacturer’s protocol was followed. The cDNA was synthesized from extracted RNAs (100 ng) using cDNA synthesis kit. Standard polymerase chain reaction (PCR) for detection of cDNA of TNF-α was carried out for 35 cycles at the annealing temperature of 53°C. The product was visualized on 2% agarose gel electrophoresis. Quantitative PCR (qPCR) was carried out to assay TNF-α messenger RNA (mRNA) using SYBR Green master mix in a Rotor-Gene Q (Qiagen, Hilden, Germany). β-actin was used as a reference house-keeping gene.
The following forward and reverse primers were used. TNF-α: forward primer: 5’-CCCTGGTACTAACTCCCAGAAA-3’, reverse primer: 5’-TGTATGAGAGGGACGGAACC-3’. β-actin: Forward- 5’-CCCGCGAGTACAACCTTCT-3’, Reverse- 5’-CGTCATCCATGGCGAACT-3’. The fold change of TNF-α was calculated by relative quantification method as described by Livak et al. 29
Western Blot Analysis
Western blot analysis was used to investigate the effect of DDT exposure on IκBα and JNK activation and insulin-induced phosphorylation of IR-β, IRS-1, and Akt in the differentiated L6 myotubes. The proteins (30 µg/well) of L6 myotube cell lysate were separated on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and electrophoretically transferred to a polyvinylidene difluoride (PVDF) membrane. Nonspecific binding was blocked by incubating the PVDF membrane with 5% bovine serum albumin dissolved in Tris-buffered saline containing 0.1% Tween 20 at 25°C for 1 hour. After blocking, the membrane was incubated overnight with primary antibody (against phospho-IR/phospho-IRS-1/phospho-Akt/phospho-IκBα/phospho-JNK) at 4°C. Subsequently, the membrane was washed thrice and incubated with HRP-conjugated secondary antibodies for 1 hour at 25°C. The bands were visualized using a chromogenic substrate (TMB), and the band densities were measured with ChemiDoc XRS+ (Bio-Rad) using Image lab software (Bio-Rad).
Then, the hybridized antibodies on PVDF membrane were stripped with the stripping buffer at 50°C for 20 minutes. For the normalization of phosphoproteins, the rehybridization of the proteins in the PVDF membrane with antibodies (primary) raised against the corresponding non-phosphoproteins was done which was visualized by incubating the PVDF membrane first with the HRP-conjugated secondary antibodies and then with TMB as mentioned earlier. Similarly, the non-phosphoproteins were normalized with β-actin.
Statistical Analysis
The data are expressed as mean (standard deviation [SD]) of 3 independent experiments carried out in duplicate. All data are analyzed by one way analysis of variance followed by a post hoc (i.e., Tukey honestly significant difference) test using the IBM SPSS version 23 software with significance set at the P < 0.05. The photographs of agarose gel electrophoresis of PCR products and western blots are representative of the results observed in three experiments.
Results
Insulin-Stimulated Glucose Uptake in DDT-Treated L6-Derived Myotubes
Insulin-stimulated glucose uptake was significantly (P < 0.05) decreased in L6-derived myotubes with exposure to subtoxic doses of DDT in comparison to that of untreated controls. Subtoxic DDT exposure dose-dependently (P < 0.05) suppressed insulin-stimulated glucose uptake in L6-derived myotubes (Table 1).
Glucose Uptake by Myotubes Following DDT Exposures (n = 3/group).
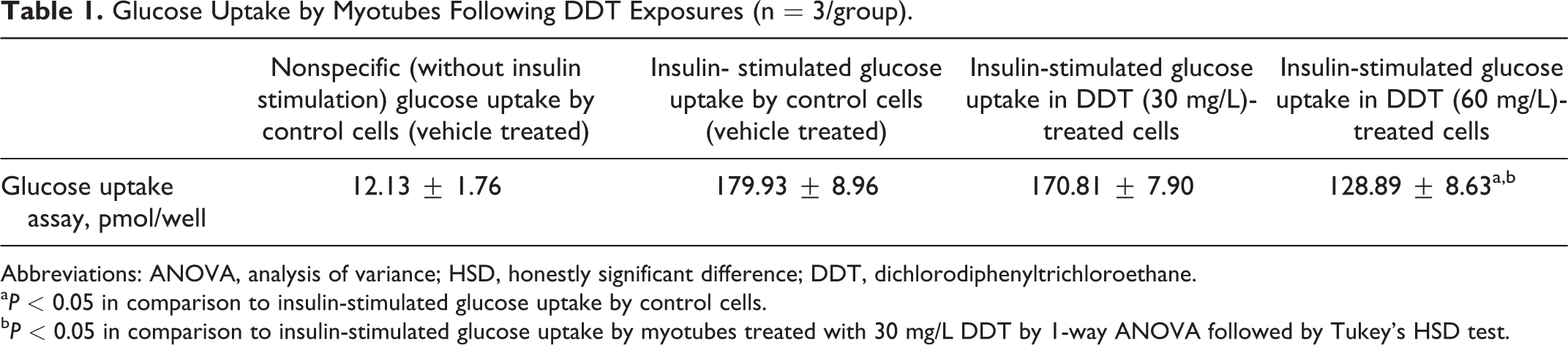
Abbreviations: ANOVA, analysis of variance; HSD, honestly significant difference; DDT, dichlorodiphenyltrichloroethane.
a P < 0.05 in comparison to insulin-stimulated glucose uptake by control cells.
b P < 0.05 in comparison to insulin-stimulated glucose uptake by myotubes treated with 30 mg/L DDT by 1-way ANOVA followed by Tukey’s HSD test.
Total Antioxidant Content, MDA Level, TNF-α Content, and its Secretion in Media in DDT-Treated L6-Derived Myotubes
The level of total antioxidants in controls (2.42 ± 0.39 mmol/gm of proteins) was significantly (P < 0.05) higher than that of DDT-exposed cells (1.9 ± 0.2 and 1.68 ± 0.23 mmol/gm of proteins when exposed to 30 mg/L and 60 mg/L of DDT, respectively). Exposure to DDT was found to dose dependently (P < 0.05) reduce total antioxidant level in cell lysate. Malondialehyde level was found to be significantly (P < 0.05) increased in the cell lysate obtained from myotubes treated with 30 mg/L and 60 mg/L of DDT in comparison to that of controls. It was not dose dependent. The concentration of TNF-α in DMEM with 2% FBS was 0.08 ± 0.01 ng/mL. The DMEM with 2% FBS in which myotubes were treated with the subtoxic doses of DDT for 18 hours also had TNF-α level of 0.08 ± 0.01 ng/mL irrespective of concentration of DDT used. There was no difference in TNF-α level in DMEM with 2% FBS before and after DDT treatment of the cells. TNF-α level in the cell lysate was significantly (P < 0.05) decreased in myotubes treated with DDT (30 mg/L and 60 mg/L) in comparison to that of controls. The above-mentioned results are shown in Table 2. The TNF-α level in pure FBS was measured to be 4 ng/mL. On dilution of positive control (rat serum) and FBS up to 2% level, the absorbance was found to be decreased in linear manner (data not shown) indicating a possible cross reactivity of bovine TNF-α with the assay method.
Total Antioxidant Level, Malondialdehyde (MDA) Level, TNF-α Content in Cell Lysate and its Secretion in Media, and TNF-α mRNA Expression in Myotubes Following Subtoxic DDT Exposures for 18 hours (n = 3 per Group).
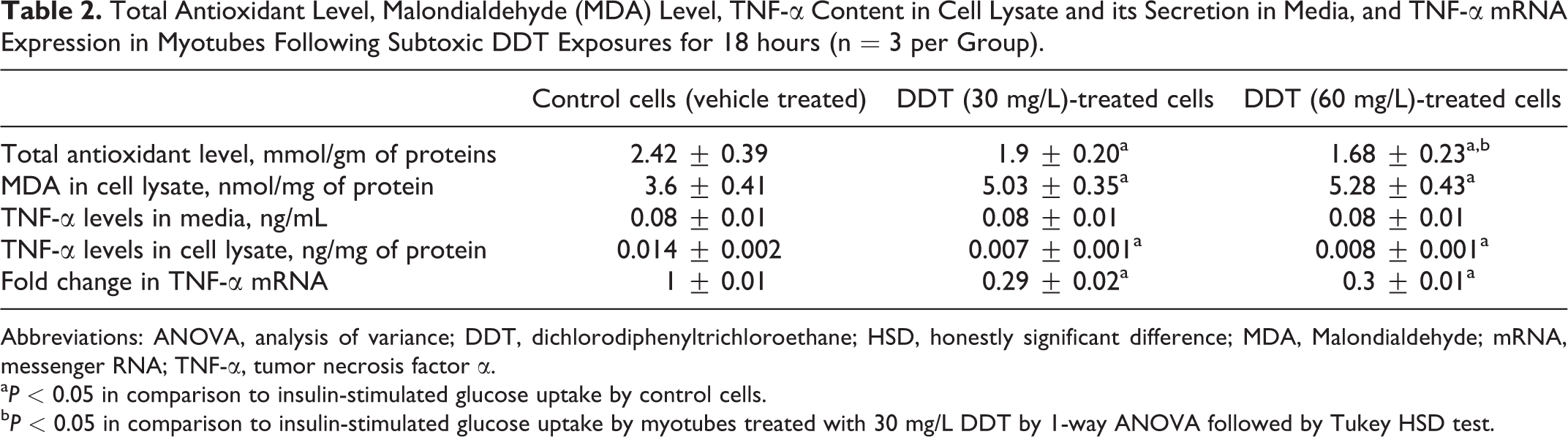
Abbreviations: ANOVA, analysis of variance; DDT, dichlorodiphenyltrichloroethane; HSD, honestly significant difference; MDA, Malondialdehyde; mRNA, messenger RNA; TNF-α, tumor necrosis factor α.
a P < 0.05 in comparison to insulin-stimulated glucose uptake by control cells.
b P < 0.05 in comparison to insulin-stimulated glucose uptake by myotubes treated with 30 mg/L DDT by 1-way ANOVA followed by Tukey HSD test.
Polymerase Chain Reaction and qRT-PCR of TNF-α mRNA in Myotubes
Figure 2 shows that TNF-α gene is expressed in myotubes with and without DDT treatment. As shown in Table 2, the TNF-α mRNA was found to be significantly (P < 0.05) decreased by 0.29- and 0.3-fold, respectively, in myotubes following 30 mg/L and 60 mg/L of DDT treatment for 18 hours. The decrease in TNF-α mRNA following DDT treatment was not dose dependent.
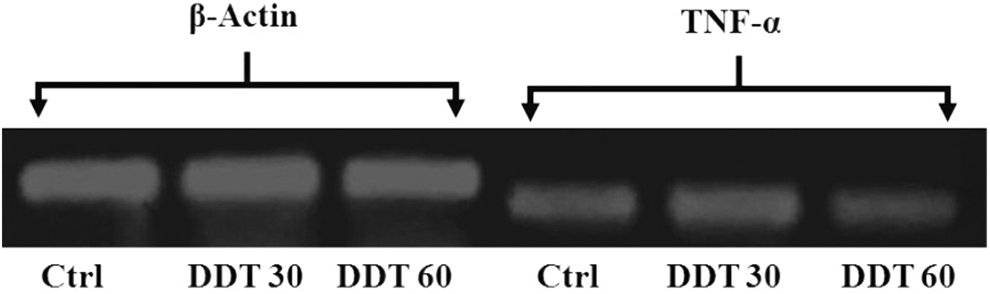
The electrophoretogram showing the band of complementary DNA (cDNA) of β-actin and tumor necrosis factor α (TNF-α) messenger RNA (mRNA) derived from myotubes with and without dichlorodiphenyltrichloroethane (DDT) treatment. Ctrl indicates control, DDT30, 30 mg/L DDT exposure; DDT60, 60 mg/L DDT exposure.
Phosphorylation of IR, IRS-1, and Akt in DDT-Treated L6-Derived Myotubes
Subtoxic DDT exposure did not alter the level of tyrosine phosphorylation of β-subunit of insulin receptor (IR) and IRS-1 and serine phosphorylation of Akt when cells were not stimulated with insulin (Figure 3).
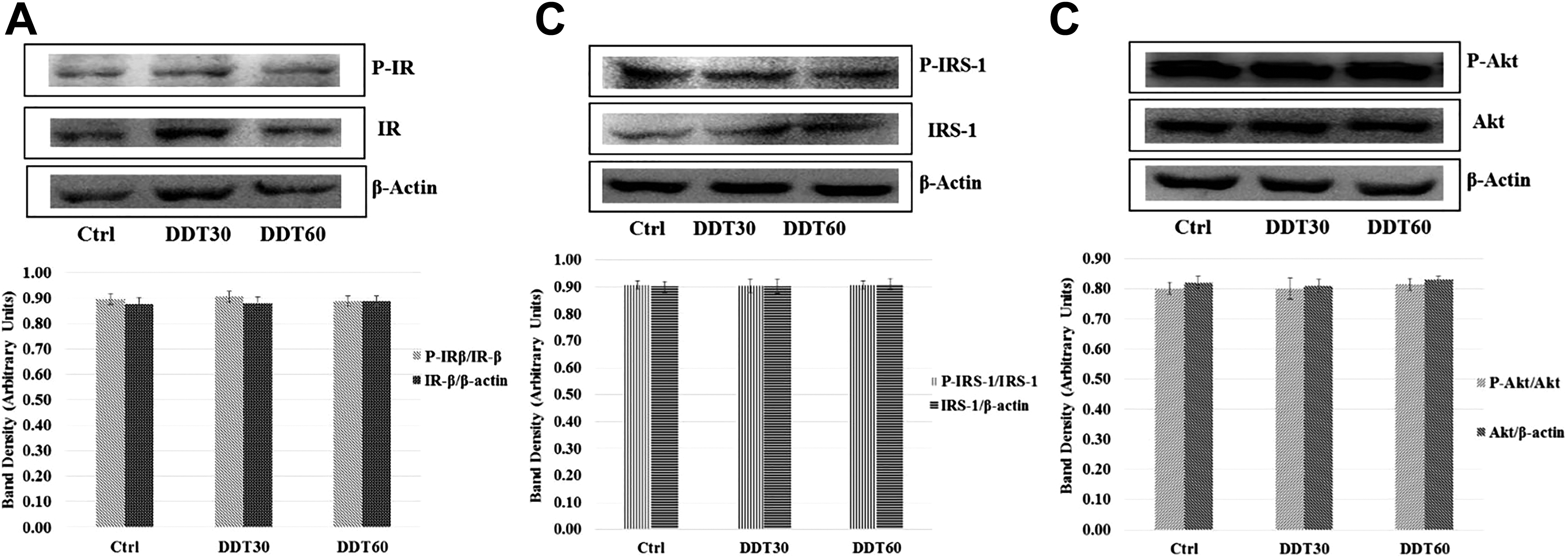
Effect of dichlorodiphenyltrichloroethane (DDT) exposure for 18 hours on basal level of (A) tyrosine phosphorylation of β-subunit of insulin receptor (IR-β), (B) tyrosine phosphorylation of insulin receptor substrate-1 (IRS-1), and (C) serine phosphorylation of Akt in L6 myoblast-derived myotubes without insulin stimulation after normalization with corresponding nonphospho proteins. Nonphospho proteins bands were normalized with β-actin. DDT exposure of 60 mg/L (DDT60) and of 30 mg/L (DDT30) did not alter the above-mention protein levels and phosphorylation of IR-β, IRS-1, and Akt in comparison to that of control (Ctrl).
The exposures to subtoxic doses of DDT did not alter insulin-stimulated tyrosine phosphorylation of β-subunits of IR in L6-derived myotubes (Figure 4A). But, insulin-stimulated tyrosine phosphorylation of IRS-1 (Figure 4B) and serine phosphorylation of Akt (Figure 4C) were found to be significantly (P < 0.05) attenuated in the myotubes exposed to subtoxic doses of DDT when compared to that of controls.
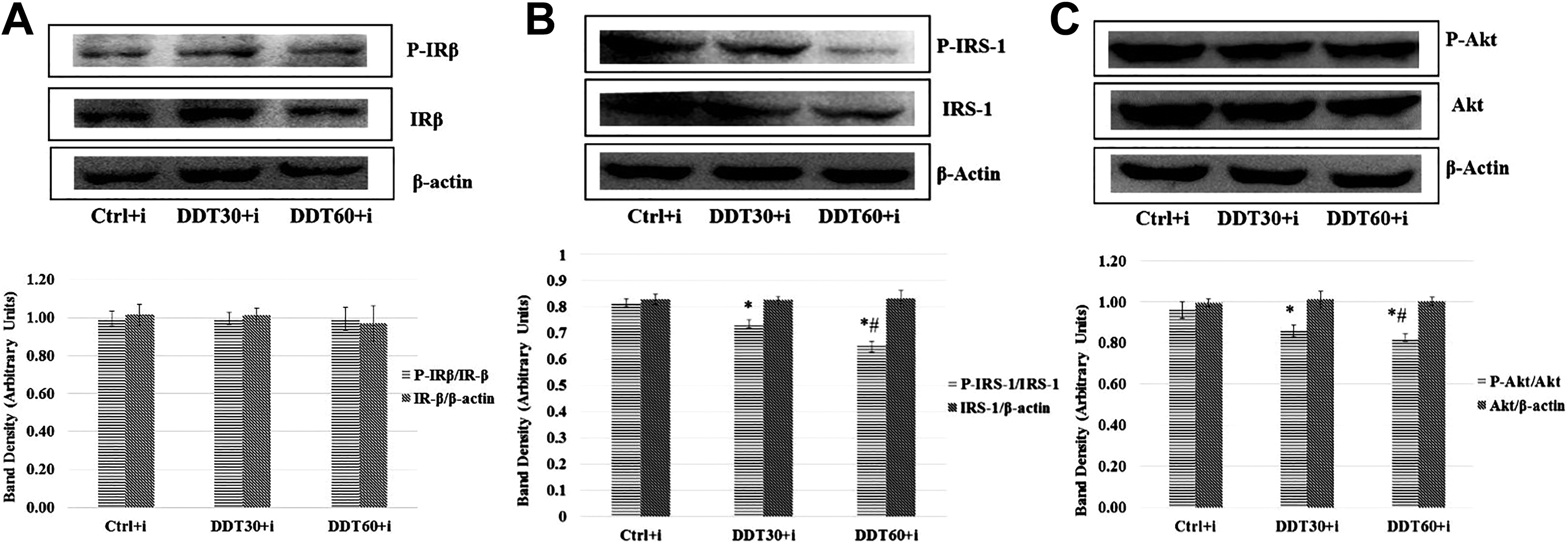
Effect of dichlorodiphenyltrichloroethane (DDT) exposure for 18 hours on insulin-induced (A) tyrosine phosphorylation of β-subunit of insulin receptor (IR-β), (B) tyrosine phosphorylation of insulin receptor substrate-1 (IRS-1), and (C) serine phosphorylation of Akt in L6 myoblast-derived myotubes after normalization with corresponding nonphospho proteins. Nonphospho protein bands were normalized with β-actin. *P < 0.05 in comparison to control (Ctrl) and #P < .05 in comparison to 30mg/L of DDT exposure followed by insulin stimulation (DDT30+i) by 1-way analysis of variance (ANOVA) followed by Tukey honestly significant difference (HSD) test. DDT60+i = 60 mg/L DDT exposure followed by insulin stimulation.
Phosphorylation of IκBα and JNK in DDT-Treated L6-Derived Myotubes
Subtoxic DDT exposure to L6-derived myotubes significantly (P < 0.05) increased phosphorylation of JNK and IκBα, a component of NFκB (Figure 3B and A). The increase in phosphorylation of these RSKs was dose dependent (P < 0.05).
Discussion
In the present study, DDT treatment was administered at 30 mg/L and 60 mg/L concentrations to explore the effect of exposure to subtoxic concentrations of DDT on glucose uptake, antioxidant level, MDA level, TNF-α content in cell lysate and its secretion in the culture media, activation of RSKs, and activation of insulin-signaling pathway. Table 1 shows that insulin-induced glucose uptake by L6-derived myotubes is decreased in a dose-dependent manner following subtoxic DDT exposure for 18 hours. This indicates the induction of insulin resistance in these muscle cells by subtoxic DDT exposures. This is in agreement with the finding that exposures to POPs including DDT as measured from their blood levels correlate with a surrogate marker of insulin resistance (i.e., HOMA-IR) in human population. 11,12,27 As shown in the result section, the antioxidant content of the cell lysate from DDT-exposed myotubes is found to be decreased in a dose-dependent manner, and the MDA level is increased in myotubes on DDT exposures. This reveals that oxidative stress prevails in myotubes on subtoxic DDT exposure. Our previous studies showed that subchronic DDT treatment induces oxidative stress in experimental animals. 14 Oxidative stress is known to induce insulin resistance. 19 Hence, we hypothesize that subtoxic DDT exposure leads to insulin resistance in muscle cells by induction of oxidative stress.
Figures 3 and 4 (A, B, and C) show that density of protein bands produced by IR-β, IRS-1, and Akt when normalized to β-actin do not differ after subtoxic DDT exposure when compared to that in controls. This indicates that the gene expression of these proteins is not altered following DDT exposure. The phosphorylation of these proteins without insulin stimulation do not alter after DDT exposure indicating that basal level of phosphorylation of insulin-signaling molecules remains unaffected following subtoxic DDT exposure. But on insulin stimulation, the density of phosphorylated IRS-1 (at tyrosine 1222) and phosphorylated Akt (at serine 307) after normalization with their corresponding nonphosphorylated protein bands decreases in a dose-dependent manner following DDT exposure. This indicates that insulin signaling is impaired in L6-derived myotubes following subtoxic DDT exposure. The activation of Akt is required for the recruitment of GLUT4 on cell membrane that leads to insulin-induced glucose uptake in muscle cells. Hence, we infer that this attenuation of phosphorylation of these signaling molecules due to DDT exposure is probably responsible for decreased insulin-induced glucose uptake by myotubes indicating the induction of an insulin resistant state. But there is no change in degree of phosphorylation of IR-β following DDT exposure. This indicates that DDT exposures do not interfere with the binding of insulin to its receptor or the receptor phosphorylation. This indicates that the impairment of insulin signaling following DDT exposure occurs at the level of IRS-1 and not at the level of IR.
Figure 5 (A and B) shows that the band density of IκBα and JNK after normalization with the band of β-actin is increased following DDT exposure. This indicates that probably NFκB and JNK genes are induced following subtoxic DDT exposure to myotubes. The band density of phosphorylated IκBα and phosphorylated JNK after normalization with their nonphosphorylated counterparts is found to be increased following DDT exposure in a dose-dependent manner. This indicates that the gene of these 2 RSKs are induced and the phosphorylation leading to activation of these RSKs is also increased following DDT exposure. These activated RSKs are known to impair activation and function of IRS-1. 19,20,30 Hence, we infer that DDT-induced impairment of insulin signaling is mediated via the gene induction and enzymatic activation of the RSKs.
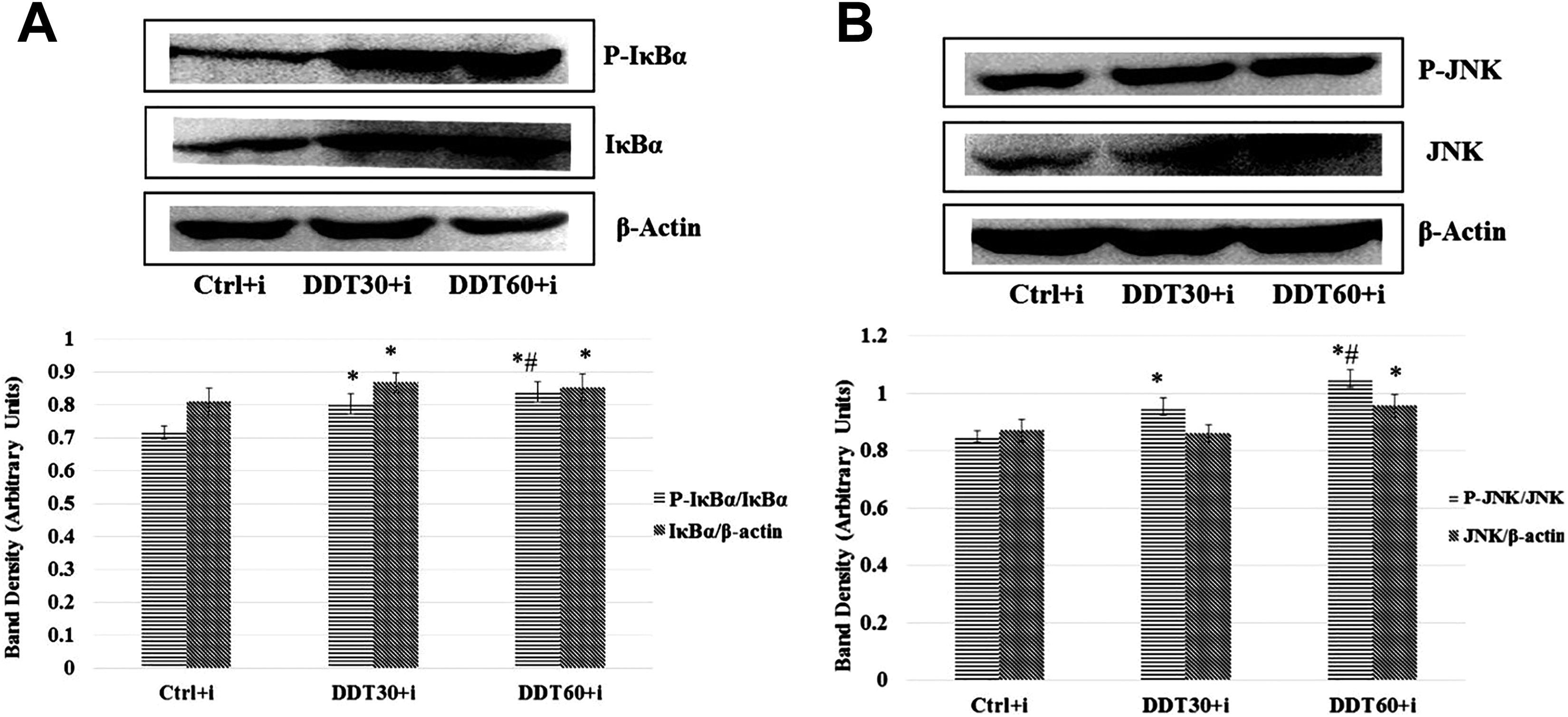
Effect of dichlorodiphenyltrichloroethane (DDT) exposure for 18 hours on (A) serine phosphorylation of IκBα and (B) phosphorylation of JNK in L6 myoblast-derived myotubes after normalization with corresponding nonphospho proteins. Nonphospho protein bands were normalized with β-actin. *P < 0.05 in comparison to control (Ctrl) and #P < 0.05 in comparison to 30 mg/L of DDT exposure followed by insulin stimulation (DDT30+i) by 1-way analysis of variance (ANOVA) followed by Tukey honestly significant difference (HSD) test. DDT60+i = 60 mg/L of DDT exposures followed by insulin stimulation.
Oxidative stress and TNF-α, a proinflammatory cytokine, are known to activate RSKs. 20,30 In the result section, it is shown that TNF-α level is not altered in DMEM media on exposure of the myotubes to DDT, although TNF-α gene in the myotubes is found to be expressed and it is in agreement with Chen et al 31 who showed that cardiac myocytes produce TNF- α that mediates myocardial inflammatory response to stretch. Contrary to the observation made in a previous study, 16 the expression of mRNA of TNF-α is observed to be significantly decreased by subtoxic DDT exposure in myotubes. Although TNF-α is a secretary protein, we tried to measure TNF-α content in cell lysate and found a significant decrease in intracellular TNF-α content in the myotubes on DDT treatment. The decrease in intracellular TNF-α is because of the observed decrease in levels of mRNA of TNF-α by DDT exposure. However, TNF-α level is not changed in the media in 18 hours with or without DDT treatment indicating that secretion of TNF-α from the myotubes is so minimal that it is not quantifiable by ELISA kit used in this study, or else this may be due to dilution of secreted TNF-α in the media beyond a measurable range. The kit used is found to have cross reactivity with bovine TNF-α. So, we presume that measured TNF-α in media is contributed by FBS which is added to the differentiating media and a relatively higher concentration of TNF-α in FBS might have masked minor difference in TNF-α level attributed by difference in its secretion by the myotubes due to DDT treatment. Major source of TNF-α is adipocytes and immunocytes. On POP exposure, TNF-α level was reported to be elevated in blood in human population, which was correlated with insulin resistance. 32 But in the present in vitro study, TNF-α level is decreased in the cell lysate but not in the media. Hence, we conclude that in the present study, TNF-α has not contributed to the observed activation of RSKs in myotubes following DDT exposure. But oxidative stress (as assessed from total antioxidant content and MDA level) prevails in myotubes following DDT exposure. Hence, we infer that the activation of RSKs in myotubes following subtoxic DDT exposures is attributed to oxidative stress induced by DDT in this in vitro study.
We conclude that subtoxic DDT exposure induces insulin resistance in muscles by inducing oxidative stress and that in turn activates RSKs and thereby attenuates insulin signaling (Figure 6). Recently, we showed that lindane has similar mechanism of inducing insulin resistance. 33
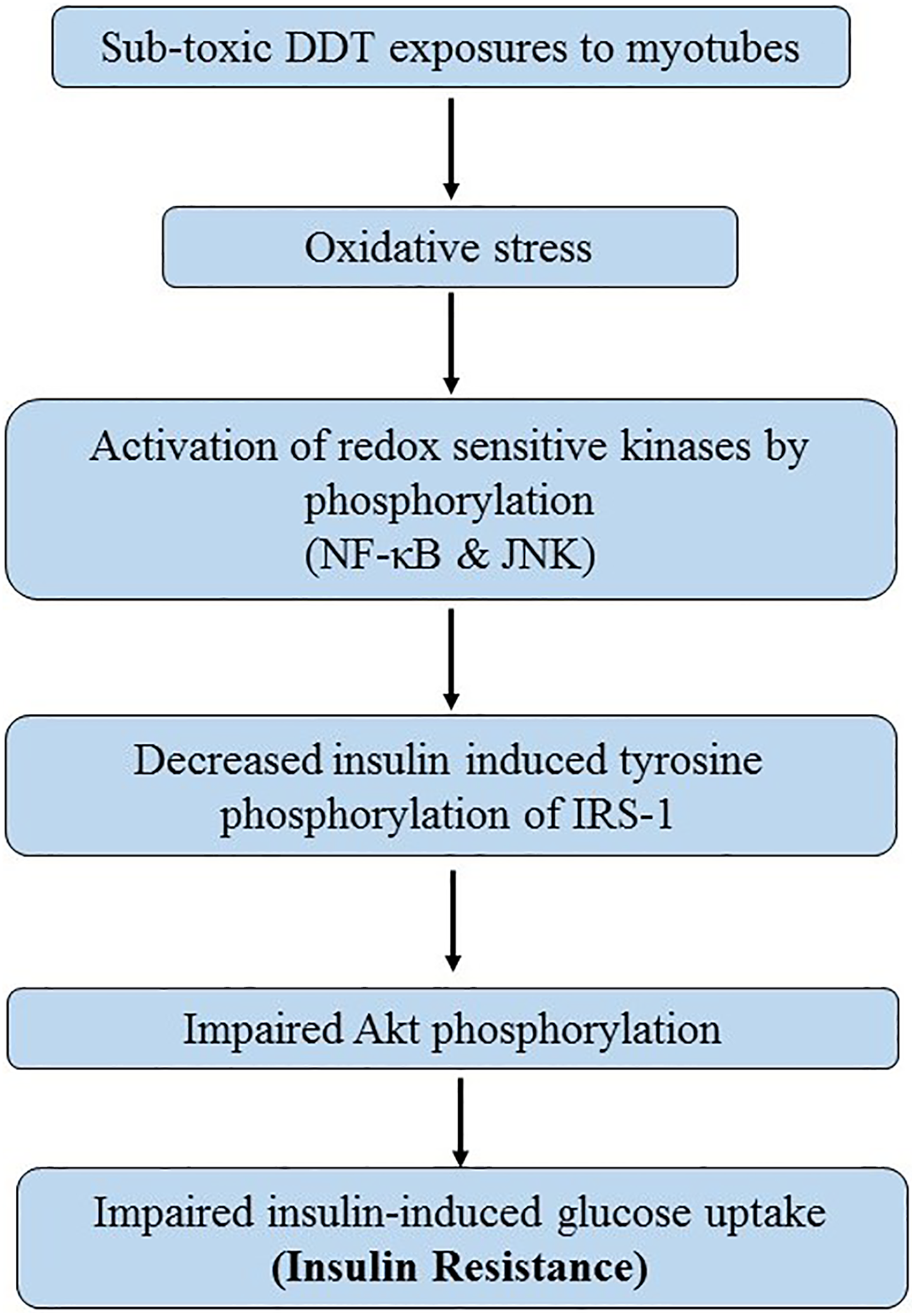
Proposed mechanism of subtoxic dichlorodiphenyltrichloroethane (DDT) exposure induced insulin resistance in myotubes.
In human populations, blood and tissue levels of DDT are much lower than the concentrations used in this in vitro study. Mean blood levels of DDT in the geographical area where the study was conducted was found to vary between 1 and 3 ppb. 34 Exposure duration is also very long in human. The present study used an exposure to DDT for a very short duration of exposure (18 hours). These limitations should be kept in mind while extrapolating the present data for human population.
Footnotes
Acknowledgment
We acknowledge Science and Engineering Research Board (SERB), Department of Science and Technology, Government of India for funding the project to carry out this study (SB/SO/HS/0024/2013) and MRU, MAMC for allowing us to use their instrumentation facility for the present work.
Author Contribution
Singh, V.K. and Sarkar, S.K. contributed to design, contributed to acquisition, drafted manuscript, and critically revised manuscript. Saxena, A. contributed to conception, contributed to analysis, drafted manuscript, and critically revised manuscript. Koner, B.C. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Dr. BC Koner received funding to carry out the research project from SERB, DST, Govt. of India.
