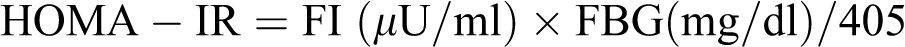Abstract
4-Methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid (bioactive compound (BAC)), a novel thiazole derivative, is a xanthine oxidase inhibitor and free radical scavenging agent. Effects of BAC on hyperglycemia, insulin sensitivity, oxidative stress, and inflammatory mediators were evaluated in streptozotocin (STZ)-induced neonatal models of non-insulin-dependent diabetes mellitus (NIDDM) rats where NIDDM was induced in neonatal pups with single intraperitoneal injection of STZ (100 mg/kg). The effect of BAC (10 and 20 mg/kg, p.o.) for 3 weeks was evaluated by the determination of blood glucose, oral glucose tolerance test (OGTT), HbA1c level, insulin level, insulin sensitivity, and insulin resistance (IR). Furthermore, inflammatory mediators (tumor necrosis factor-alpha and interleukin-6) and oxidative stress were estimated in serum and pancreatic tissue, respectively. Significant alteration in the level of blood glucose, OGTT, HbA1c, insulin level, insulin sensitivity, in addition variation in the antioxidant status and inflammatory mediators, and alteration in histoarchitecture of pancreatic tissue confirmed the potential of BAC in STZ-induced neonatal models of NIDDM rats. Pretreatment with BAC restored the level of glucose by decreasing the IR and increasing the insulin sensitivity. Furthermore, BAC balanced the antioxidant status and preserved the inflammatory mediators. Histological studies of pancreatic tissues showed normal architecture after BAC administration to diabetic rats. Altogether, our results suggest that BAC successfully reduces the blood glucose level and possesses antioxidant as well as anti-inflammatory activities. This leads to decreased histological damage in diabetic pancreatic tissues, suggesting the possibility of future diabetes treatments.
Keywords
Introduction
Diabetes mellitus (DM) is a metabolic disorder associated with an array of dysfunctions characterized by elevated levels of glucose in blood and paucity in production or action of insulin produced by the pancreatic beta cells. 1 Non-insulin-dependent diabetes mellitus (NIDDM) is due to a combination of resistance to insulin action and impaired secretion of this hormone, and it accounts for 90% of total cases of DM. 2 NIDDM is more prevalent than ever, particularly in developing countries, and more than 415 million people suffered from diabetes worldwide in 2015, and if current rate of growth continues, the number is projected to exceed 642 million cases by 2040. 3 Insulin resistance (IR) is the key pathophysiological factor behind the etiology and progression of NIDDM. 4 IR mainly attenuated insulin signaling leading to abundant metabolic deregulation, cellular alterations including impaired stimulation of diminished cellular glucose uptake in skeletal muscle and adipocytes, and impaired inhibition of glucose production from the liver or increased liver gluconeogenesis resulting in hyperglycemia and alterations in lipid metabolism. 5,6
Streptozotocin (STZ), a glucose analog (2-deoxy-2-[3-methyl-e-nitrosourido]-D-glucopyranose) extracted from Streptomyces achromogenes, possesses a diabetogenic effect via its specific damaging action against pancreatic β-cells. 7 Neonatal STZ-induced diabetic rat model is a better tool for elucidating the importance of the β-cell dysfunction in the development of NIDDM. 8 Additionally, it has also been well documented that blood glucose and insulin level significantly increases after administration of single dose of STZ in 2-day-old pups. 9 Previous finding suggests that oxidative stress actively involved in the etiology of diabetic complications that arise from chronic hyperglycemia and increased lipogenesis. 10 Cytotoxic action of STZ leads to the generation of reactive oxygen species (ROS), which is responsible for oxidative damage in pancreatic tissues. 11 Association between NIDDM and inflammation has been previously well documented. 12 Tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6), a pro-inflammatory cytokine, as well as oxidative stress are widely recognized markers of vascular inflammation, 13,14 and the level of these cytokines and oxidative stress are elevated in the blood of diabetic animals. 15
Managements of the NIDDM mainly focus on lowering the blood glucose level through diverse mechanisms including glucose assimilation, insulin secretion, regulation, or inhibition of biochemical pathways involved in carbohydrate and lipid metabolisms 16 with lesser associated side effects. The new remedies for NIDDM such as herbal remedies and bioactive compound (BAC) are viewed as a promising agent due to the lesser associated side effects and their economical value. Although the precise mechanism of action of current available therapy for NIDDM, that is, thiazolidinediones (TZDs), has not fully elucidated, the end results of their physiologic effects are to improve insulin-mediated peripheral glucose removal and reduce hepatic glucose output, as demonstrated by in vitro and in vivo studies. 17 However, these TZDs have several side effects such as increased risk of heart attacks, risk of bladder cancer, and cardiac failure. 18 Therefore, the search for new agents that minimize these undesirable effects and are still effective in regulating the disturbances observed in NIDDM has been receiving increasing attention from the scientific community. In this regard, several thiazole compounds have emerged as promising treatments for NIDDM. 19 Thiazoles are important heterocyclics compounds with array of therapeutic potentials such as antidiabetic, antioxidant, antibacterial, antifungal, anti-inflammatory, antitumor, antitubercular, antiviral, and as substituents on a particular position of the thiazole ring which affects the biological outcomes to a great extent. Currently, many researchers have been driven toward the synthesis of thiazole compound and evaluation of its biological activities. 20 In addition, it has also been previously documented that N-(4-phe-nylthiazol-2-yl) benzenesulfonamide derivatives show antidiabetic activities in STZ-induced rat models. 21
4-Methyl-2-[(2-methylbenzyl)amino]-1,3-thiazole-5-carboxylic acid (BAC) is a new thiazole derivative and was designed and synthesized as a structural analogue of pioglitazone. Recently, in vitro study showed that BAC possesses enzyme xanthine oxidase inhibitors, antioxidant, as well as free radical scavenging activities. 22 In addition, from the prediction of activity spectra of species (PASS) analysis, BAC is predicted to have antidiabetic property. Accordingly, this study investigated the antidiabetic, antioxidant, and anti-inflammatory potential of BAC, a new thiazole derivative in STZ-induced NIDDM rats for the first time.
Materials and methods
Synthesis of 4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid (BAC)
Substituted ethyl 2-(benzylamino)-4-methyl-1,3-thiazole-5-carboxylate
The chemical properties of 4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid (BAC) (Figure 1) were as follows: yield: 67%; m.p.: 106–108°C; IR (KBr v max): 2926, 1607,1505,1238, 995, and 821 cm−1; 1H-NMR (proton nuclear magnetic resonance) (400 MHz, CDCl3); δ 2.29 (s, 3 H, Ar-CH3), 2.30 (d, 3 H, CH3), 4.34 (d, 2 H, CH2), 6.16 (bs, 1 H, D2O exchangeable NH), 7.03 (s, 1 H, Ar-H), 7.21 (d, 2 H, J = 6.0 Hz, Ar-H), and 7.57 (s, 1 H, Ar-H); 13C-NMR (carbon- nuclear magnetic resonance) (100 MHz, CDCl3); 17.31 (thiazole CH3), 19.82 (Ar-CH3), 51.19 (CH2NH), 117.04, 126.19, 127.07, 128.63, 130.21, 134.13, 140.61, 155.43, 162.37(C=N), and 168.51(C=O); electrospray ionization-mass spectrometer (ESI-MS): m/z: 263.27 (M + H); Anal. Calcd. for C13H14N2O2S: C, 59.52; H, 5.38; N, 10.68%. Found: C, 59.61, H, 5.37, and N, 10.71%.

Structure of 4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid (BAC). BAC: bioactive compound.
Drugs and chemicals
4-Methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid (BAC) was designed and synthesized by the Department of Pharmaceutical Chemistry, Jamia Hamdard, and the standard drug pioglitazone was procured from the Intas Pharmaceutical Ltd. (Ahmedabad, Gujarat, India).
STZ was procured from Sigma-Aldrich Chemicals Co., St. Louis, Missouri, USA. The enzyme-linked immunosorbent assay (ELISA) kit for insulin was purchased from Mercodia (Uppsala, Sweden); TNF-α and IL-6 were purchased from the eBioscience (San Diego, California, USA). 1,1′,3,3′-tetraethoxypropane, crystalline beef liver catalase (CAT), reduced glutathione (GSH), 5,5-dithiobis (2-nitrobenzoic acid), bovine serum albumin (BSA), thiobarbituric acid, Tris buffer, sucrose, trichloroacetic acid, citric acid monohydrate, sodium nitrate, copper sulfate, sodium potassium tartrate, ethylenediaminetetraacetic acid disodium salt, Folin’s phenol reagent, hydrochloric acid, and concentrated sulfuric acid were purchased from S.D. Fine Chemicals, Mumbai, Maharashtra, India. All the other biochemicals and chemicals used for the experiment were of analytical grade.
NIDDM rat model
Two-day neonatal Wistar rat pups were raised in our animal facility. The rats were maintained under a standard 12-h light/dark cycle and were housed in a controlled temperature (24°C ± 2°C) and humidity (50% ± 5%) environment. Food and water were available ad libitum. Severe NIDDM was induced by a single intraperitoneal injection of STZ (100 mg/kg body weight) that was freshly dissolved in 0.01 M sodium citrate buffer (pH 4.5) after overnight fasting. 9 The animals were awake during the administration of saline or STZ. Rats with blood glucose level ≥250 mg/dl were considered diabetic and used in this study. Citrate buffer was injected as vehicle in the control groups. The experimental study was reviewed and approved by the Institutional Animal Ethics Committee of Jamia Hamdard (Hamdard University), New Delhi, India.
Experimental design
The rats were divided into six groups (n = 6 per group): (I) normal control, (II) NIDDM control rats, (III) NIDDM rats treated with BAC (10 mg/kg, p.o.), (IV) NIDDM rats treated with BAC (20 mg/kg, p.o.), (V) per se rats were treated with BAC (20 mg/kg, p.o.), and (VI) NIDDM rats treated with standard drug pioglitazone (10 mg/kg, p.o.). Dose of pioglitazone (10 mg/kg) has been selected on the basis of previous study, 23 and the dose of BAC (10 and 20 mg/kg) has been selected to compare its activity with the standard pioglitazone. BAC and pioglitazone (10 mg/kg) solutions were dissolved in 1% carboxymethylcellulose (CMC) suspension, and oral gavage method was used to administer drug in groups III, IV, V, and VI for 3 weeks after confirmation of diabetes. At the end of the study, blood samples were collected by nicking the tip of tail and all animals were killed; pancreases were isolated for biochemical estimations. Isolated pancreas is rinsed in ice-cold saline and stored in −80°C freezer for further biochemical studies. A pancreas of rat from each group was isolated and fixed in 10% formalin solution for histopathological examination. Plan of the work is shown in Figure 2.
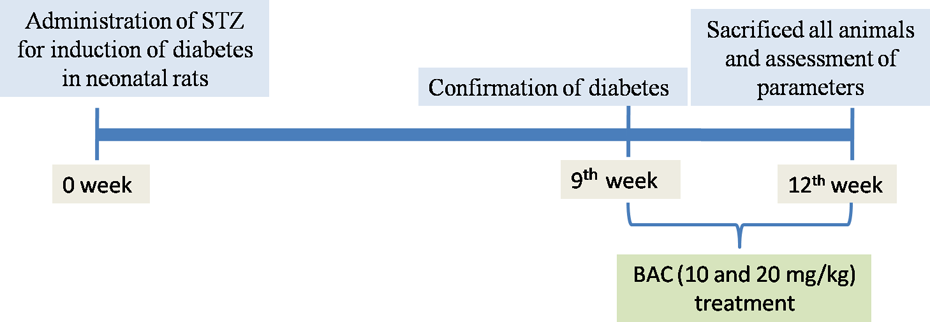
Plan of work. STZ: streptozotocin; BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid).
Measurement of glucose level in the serum
The serum glucose level was examined using a commercial kit from Span Diagnostic Limited (Surat, Gujarat, India), using a previously established method. 24
Determination of oral glucose tolerance test
Glucose solution (2 g/kg, p.o.) was given to 12-h overnight fasted rats, and blood samples were taken at 0, 15, 30, 60, and 120 min after glucose administration. All the blood samples were collected for glucose estimation. 9
Determination of HbA1c level
Glycosylated hemoglobin (HbA1c) was estimated by using a diagnostic kit from Monozyme India Limited (Hyderabad, Telangana, India). 9
Determination of insulin level
Plasma insulin level was estimated by ELISA kit method. For this purpose, insulin ELISA kit from Mercodia (Uppsala, Sweden) was used.
Determination of insulin sensitivity
Insulin sensitivity was measured using K ITT as an index of insulin-mediated glucose metabolism. For this purpose, rats were fasted for 15 h before giving insulin challenge. Insulin (0.2 U/100 g body weight human regular insulin) was administered by slow intravenous injection through tail vein. Blood samples were collected at 0, 15, 30, 60, and 120 min after administration of insulin injection. K ITT was determined from the slope of a linear portion of the regression line of natural logarithm of glucose versus time 9 and using the formula
where t 1/2 represents the half-life of plasma glucose decay.
To determine the half-life of plasma glucose, plasma glucose concentrations versus time on semilogarithmic graph paper were plotted. Fasting blood glucose (FBG) and fasting insulin (FI) levels were used for the calculation of homeostasis model assessment of insulin resistance (HOMA-IR) and were used for the determination of hepatic IR. 25 Sensitivity level of insulin was calculated using the following formula:
Biochemical estimations
The pancreatic tissue homogenate was used to assess lipid peroxidation (LPO) and antioxidant enzymes. Tissue homogenates were prepared with 0.1 M Tris HCl buffer (pH 7.4). Supernatant of homogenates was used to estimate LPO (malondialdehyde (MDA)) according to the method of Ohkawa et al., 26 reduced GSH by Moron et al., 27 superoxide dismutase (SOD) activity by Misra and Fridovich, 28 and CAT activity by Sinha. 29 Protein concentration was determined using BSA as standard by Lowry et al. 30
Estimation of TNF-α and IL-6
The quantifications of TNF-α and IL-6 were performed in the serum according to instructions provided by the rat TNF-α and IL-6 ELISA kit manufacturer eBioscience.
Histopathological examination of the pancreas
For microscopic evaluation, the pancreas was cut to isolate the middle segment. These segments were fixed in 10% neutral buffered formalin, and after embedding in paraffin, the segments were cut into 2 μm sections and stained using hematoxylin and eosin (H&E) for microscopic evaluation.
Statistical analysis
Data were expressed as the mean ± standard error of the mean (SEM). For a statistical analysis of the data, group means were compared by one-way analysis of variance (ANOVA) with post hoc analysis. The Tukey–Kramer post hoc test was applied to identify significance among groups; p < 0.05 was considered to be statistically significant. Statistical analysis was carried out using Prism software package (version 7; GraphPad, San Diego, California, USA).
Results
Effect of BAC on relative pancreas weight in STZ-induced NIDDM rats
Table 1 shows the relative pancreas weight of experimental animals. In the current study, we observed that STZ administration leads to significant decrease in relative pancreas weight of NIDDM control rats when compared to normal control rats. At the end of the experimental period, relative pancreas weight of BAC-treated NIDDM rats was slightly elevated in comparison to the NIDDM control rats. On the other hand, we observed no significant differences between the relative pancreas weight of pioglitazone-treated rats and normal control rats.
Effect of BAC on relative organ weight, blood glucose, and HbA1c levels in STZ-induced NIDDM rats.a

BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
aData are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.), NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group; NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group.
b p < 0.001 as compared to the normal control group.
c p < 0.001 as compared to STZ group.
d p < 0.01 as compared to STZ group.
Effect of BAC on blood glucose and HbA1c levels in STZ-induced NIDDM rats
As shown in Table 1, serum glucose and HbA1c levels in NIDDM control rats were markedly higher than those in normal control rats, but treatment with BAC significantly prevented the rise in serum glucose and HbA1c levels in the NIDDM-treated rats when compared to the NIDDM control rats. On the other hand, only BAC treatment in normal rats does not register any significant changes in serum glucose and HbA1c levels when compared to normal control rats.
Effect of BAC on oral glucose tolerance test in STZ-induced NIDDM rats
Oral glucose tolerance of the nondiabetic and diabetic rats after administration of glucose (2 mg/kg) is illustrated in Table 2. NIDDM control rats demonstrated a peak blood glucose levels after 30 min of glucose administration. The blood glucose level remains high over the next 90 min. However, after treatments with BAC, glucose utilization became enhanced, and hyperglycemic condition of the diabetic rats was significantly reduced as shown in Table 2. On the other hand, no significant changes were noted in blood glucose levels of per se BAC rats in comparison to normal control rats.
Effect of BAC on OGTT in STZ-induced NIDDM rats.a

BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); OGTT: oral glucose tolerance test; STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
aData are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.), NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group; NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group.
b p < 0.001 as compared to the normal control group.
c p < 0.01 as compared to STZ group.
d p < 0.001 as compared to STZ group.
Effect of BAC on insulin levels in STZ-induced NIDDM rats
Figure 3(a) shows the effect of BAC on insulin level. NIDDM control rats demonstrated a significant (p < 0.001) increase in insulin level when compared to the normal control rats. Administration of BAC to NIDDM rats continuously for 3 weeks significantly prevented the rise in insulin level in comparison to NIDDM control rats suggesting BAC might be effective in preventing hyperinsulinemia. There were no significant changes in insulin level of per se BAC rats when compared to normal control rats

Effect of BAC on insulin (a) and K ITT (b) level in STZ-induced NIDDM rats. Data are expressed as mean ± SEM (n = 6) and analyzed by two-way ANOVA followed by Tukey’s multiple range test. ### p < 0.001 as compared to the normal control group, ***p < 0.001 as compared to STZ group, and **p < 0.01 as compared to STZ group. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.); NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group; NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group. BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
Effect of BAC on insulin sensitivity in STZ-induced NIDDM rats
Figure 3(b) shows the levels of K ITT, an index of insulin sensitivity, and Figure 4 shows the levels of HOMA-IR, an index of hepatic IR. NIDDM control rats demonstrated a significant (p < 0.001) decrease in K ITT level as well as significant increase in HOMA-IR levels when compared to normal control rats. NIDDM group treated with BAC for a duration of 3 weeks demonstrated increased level of K ITT level and decreased level of HOMA-IR when compared to NIDDM control rats. No significant changes were noted in K ITT and HOMA-IR levels of per se BAC rats when compared to normal control rats.
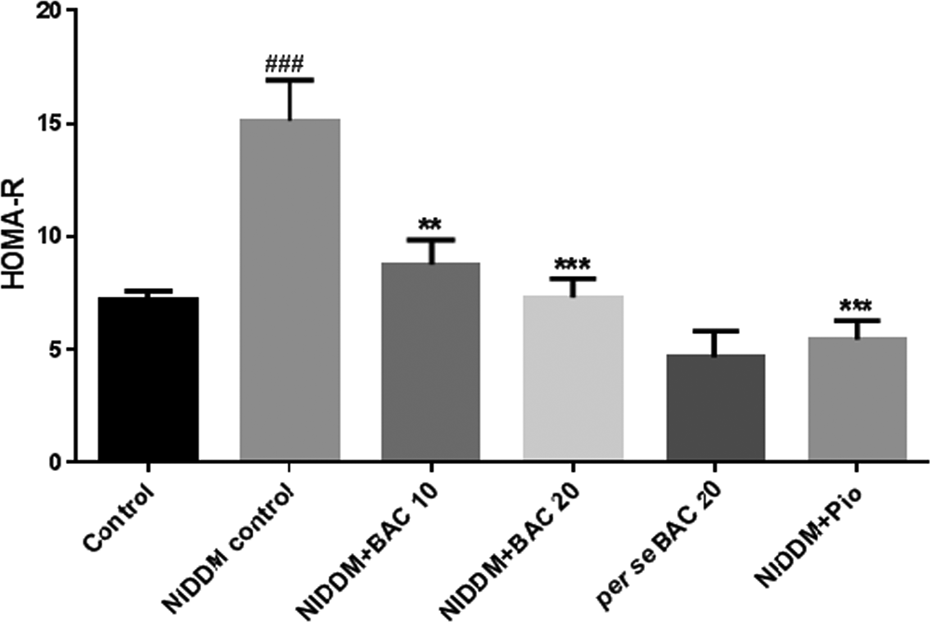
Effect of BAC on HOMA-IR level in STZ-induced NIDDM rats. Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. ### p < 0.001 as compared to the normal control group, ***p < 0.001 as compared to STZ group, and **p < 0.01 as compared to STZ group. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.), NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group; NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group. BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); HOMA-IR: homeostasis model assessment of insulin resistance; STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
Effect of BAC on pro-inflammatory cytokines (TNF-α and IL-6) in STZ-induced NIDDM rats
To analyze the effect of BAC on pro-inflammatory cytokines production, TNF-α and IL-6 were assayed using ELISA. Figure 5(a) and (b) shows the effect of BAC on pro-inflammatory cytokines TNF-α and IL-6, respectively. The levels of these two cytokines were considerably increased in NIDDM control rats when compared to normal control rats. BAC-administered group exhibited TNF-α and IL-6 values that are lower than NIDDM control groups and higher than the normal control values, and both the findings are statistically significant. These results indicate that BAC successfully inhibits the production of pro-inflammatory cytokines in STZ-induced NIDDM rats. On the positive note, pioglitazone treatment also significantly reduced TNF-α and IL-6 levels when compared with NIDDM control rats, and no significant changes were noted in the values of TNF-α and IL-6 in per se BAC rats when compared to normal control rats.

Effect of BAC on TNF-α (a) and IL-6 (b) level in STZ-induced NIDDM rats. Data are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. ### p < 0.001 as compared to the normal control group, ***p < 0.001 as compared to STZ group, and **p < 0.01 as compared to STZ group. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.), NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group; NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group. BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
Effect of BAC on LPO (MDA) contents, GSH contents, activity of SOD, and CAT in the pancreas of STZ-induced NIDDM rats
Table 3 illustrates the level of MDA, a secondary product of LPO, level of GSH, activity of SOD, and CAT in pancreatic tissue homogenate. NIDDM control rats demonstrated the decreased CAT and SOD activity and GSH content and increased MDA level in pancreatic tissue when compared to the normal control rats. NIDDM rats treated with BAC for 3 weeks demonstrated increase in the activity of CAT and SOD as well as the level of GSH, respectively, and reduced the MDA level when compared to NIDDM control rats. On the other hand, no significant changes were noted in the levels of MDA, GSH, SOD, and CAT with per se BAC-treated rats.
Effect of BAC on MDA contents, GSH, SOD, and CAT activities in the pancreas of STZ-induced NIDDM rats.a

BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone; i.p.: intraperitoneally; SEM: standard error of the mean; ANOVA: analysis of variance.
aData are expressed as mean ± SEM (n = 6) and analyzed by one-way ANOVA followed by Tukey’s multiple range test. Control: Control group, citrate buffer (0.1 ml/10 g, i.p.); NIDDM control: NIDDM control group, STZ (100 mg/kg, i.p.), NIDDM + BAC 10: STZ and BAC (10 mg/kg, p.o.) treated group; NIDDM + BAC 20: STZ and BAC (20 mg/kg, p.o.) treated group; per se BAC: BAC (20 mg/kg, p.o.) treated group and NIDDM + Pio: STZ + Pio (10 mg/kg, p.o.) treated group.
b p < 0.001 as compared to the normal control group.
c p < 0.01 as compared to STZ group.
d p < 0.05 as compared to STZ group.
e p < 0.001 as compared to STZ group.
Effect of BAC on histological changes in the pancreas
In the histopathological examinations using H&E staining, pancreatic tissue of NIDDM control rats (Figure 6(b)) exhibited a decrease in pancreatic islet numbers and size as well as extensive destruction of islet cells when compared to normal control rats (Figure 6(a)). BAC-administered group (Figure 6(c) and (d)) for 3 weeks distinctly improved this abnormal histopathological phenomenon as evidenced by the regeneration of β-cells suggesting protective effects of BAC against pancreatic β-cells damage. Per se BAC (20 mg/kg)-treated rats show the normal histology similar to the control rat (Figure 6(e)). In addition, NIDDM rats treated with pioglitazone (10 mg/kg) show the regeneration of β-cells as well as the number of β-cells is increased, and it resembled the pancreatic morphology similar to the normal rat (Figure 6(f)).
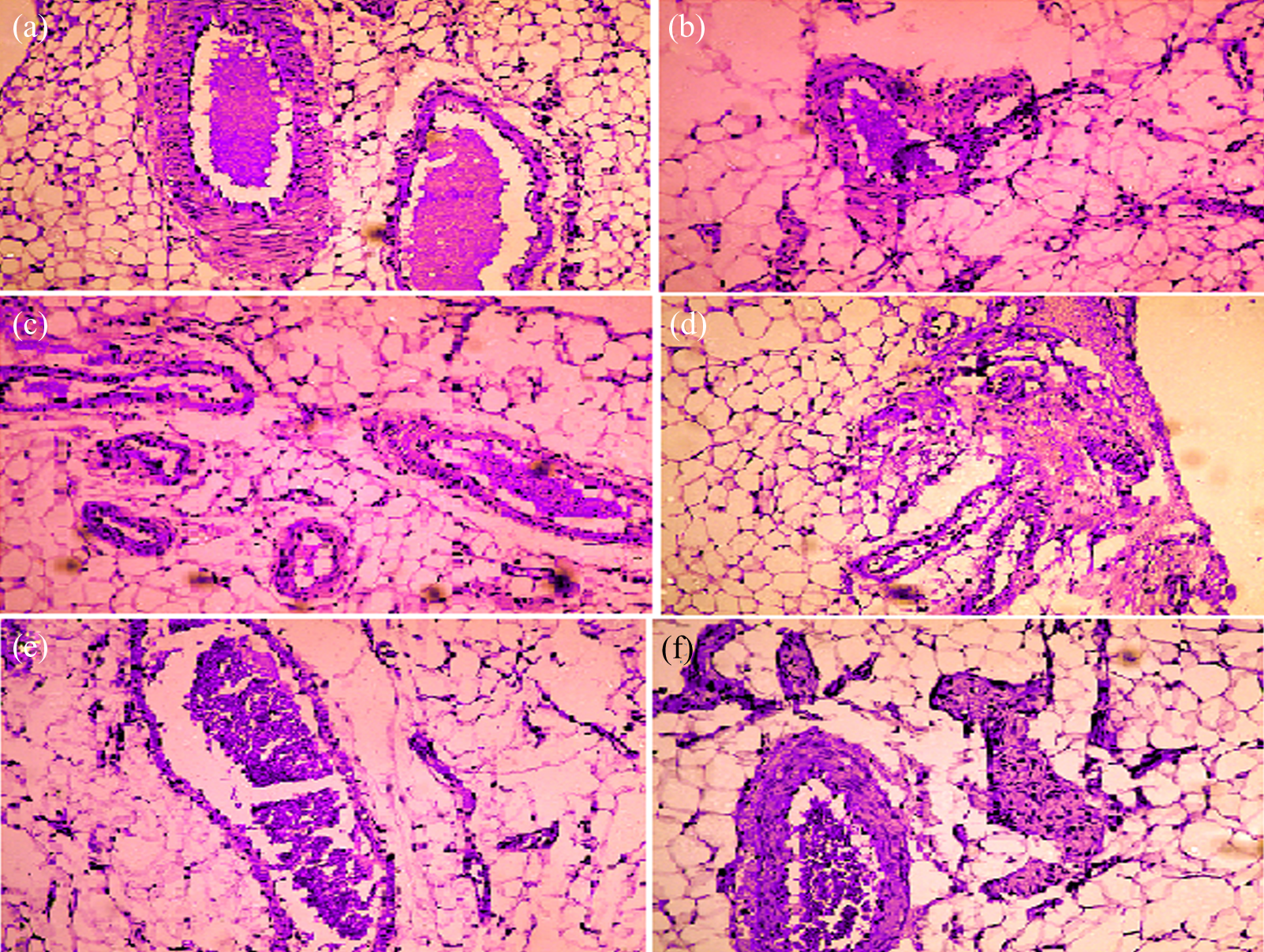
Effect of BAC on histopathological changes in STZ-induced NIDDM rats. Photomicrograph of sections of pancreatic tissue of normal group (a), NIDDM control group (b), NIDDM + BAC (10 mg/kg) treated group (c), NIDDM + BAC (20 mg/kg) treated group (d), per se BAC (20 mg/kg) treated group (e), and NIDDM + Pio (10 mg/kg) treated group (f) (×100). BAC: bioactive compound (4-methyl-2-[(2-methylbenzyl) amino]-1,3-thiazole-5-carboxylic acid); STZ: streptozotocin; NIDDM: non-insulin-dependent diabetes mellitus; Pio: pioglitazone.
Discussion
DM is the most challenging health problem of the current century, 3 recommending a great need to focus on additional improvements. Neonatal STZ-induced rat model resembles the natural course DM in human as it exhibits hyperglycemia, abnormal glucose tolerance, and mild hypoinsulinemia through an insidious process which is almost asymptomatic at the beginning and becomes manifested at the adult age. 31 Previously, several lines of evidence indicate the key role of free radicals in the mechanism of β-cell damage and diabetogenic effect of STZ. 32 The present study investigated the preventive effect of BAC on hyperglycemia, insulin sensitivity, inflammation, and oxidative stress in STZ-induced NIDDM rats. Previously, it has been well reported that chronic hyperglycemia develops in neonatal rats after STZ injection on the second day. 9 It has been also documented that STZ administration causes oxidative stress and free radical generation which damages the pancreatic β-cells and results in hyperglycemia and glycosylation of various proteins including hemoglobin. 33 In the present study, NIDDM control rats showed significantly higher levels of blood glucose and HbA1c levels as compared with normal control rats, which is consistent with the earlier report. 34 However, administering BAC, for 3 weeks of treatment duration, remarkably attenuates the altered blood glucose and HbA1c levels in STZ-induced diabetic rats showing its antihyperglycemic activity. This effect of BAC might be due to antioxidant and free radical scavenging property. 22 In addition, during the oral glucose tolerance test, BAC significantly improved glucose tolerance, as indicated by reduction in peak blood glucose level at 1 and 2 h in NIDDM-treated rats. BAC might enhance glucose utilization by peripheral tissues and increase the glycogen stores in the liver due to restoration of delayed insulin response.
Furthermore, it has been well described earlier that hyperinsulinemia is strongly associated with IR through compensatory mechanism. 35 Previously, it has been reported that hyperinsulinemia is associated with decreased hepatic insulin clearance and hypersecretion of β-cells in mild glucose intolerance obese subjects. 36 Results of the present study suggest that hyperinsulinemia was seen in NIDDM control rats that might be due to reduced hepatic clearance of insulin or even by downregulation of insulin receptors and desensitizing post-receptor pathways. 37 However, BAC treatment was found to be effective in reducing insulin level of NIDDM rats, thereby preventing hyperinsulinemia. K ITT is an insulin sensitivity index used to determine peripheral IR, and HOMA-IR is a clinical index of hepatic IR. 33 It has been previously documented that STZ administration impaired the K ITT and HOMA-IR levels in the diabetic rats. 9 The results obtained from our study clearly showed that K ITT was significantly improved by BAC treatment in STZ-induced NIDDM rats. Additionally, BAC treatment significantly prevented the rise in HOMA-IR level in NIDDM-treated rats. These findings suggest that BAC might be pharmacologically effective in improving insulin sensitivity.
STZ administration causes generation of ROS, ultimately leading to increased oxidative stress in a variety of tissues. 38 Several lines of studies reported that oxidative stress disturbs the oxidant and antioxidant balance. 39 In addition, antioxidant enzymes such as SOD and GSH play an important role against the oxidative stress. 40 In oxidative stress condition, decreased SOD enzymes excessively generate the superoxide and hydrogen peroxide free radicals. 38 However, overproduction of free radicals or increased consumption in the defense of –SH group-containing proteins is responsible for the depletion of GSH contents. 41 It has been reported that decreased SOD and CAT levels are important risk factor for the initiation of LPO. 42 In accordance with previous findings, our result showed altered SOD, GSH, CAT, and MDA levels in the pancreatic tissues of diabetic rats. 38,42 In the present study, we showed that BAC significantly increased the SOD, GSH, and CAT activities, whereas decreased the MDA contents in the pancreas of STZ-induced diabetic rats. In line with previous study, BAC, a thiazole-containing compound, normalizes the SOD, GSH, and CAT activities and decreased the MDA contents in the pancreas of diabetic rats. Attenuation of oxidative stress may be due to antioxidant and free radical scavenging property of BAC. 22
Production of pro-inflammatory cytokines (TNF-α and IL-6) is an important indicator of chronic low-grade inflammation, which is another pathogenic factor for the NIDDM. 43 It has been previously reported that these increased cytokines levels in diabetic rats might be due to IR and inflammatory processes. 44 TNF-α increases serine/threonine phosphorylation of the insulin receptor substrate, which negatively affects insulin signal transduction, 45 whereas increased IL-6 production in NIDDM may reduce tyrosine phosphorylation and eventually decrease insulin sensitivity. 46 In agreement with previous findings, we also observed significantly raised serum TNF-α and IL-6 levels in the NIDDM rats, which were significantly reversed by the BAC supplementation, claiming its potential role in inflammation. The H&E staining of pancreatic tissues corroborates the above discussed molecular changes that BAC maintained the almost normal architecture of the pancreas in NIDDM rats.
In conclusion, our result shows that BAC has an insulin sensitizing effect comparable to that of the TZDs, pioglitazone. These findings demonstrate that BAC attenuated glucose intolerance and IR through its antioxidant and anti-inflammatory effects in STZ-induced neonatal NIDDM animal model. Our study provides new insights about preventive effects of BAC on hyperglycemia, insulin sensitivity, inflammation, and oxidative stress for the very first time. However, further studies are suggested to investigate the molecular changes to explore the antidiabetic properties of BAC, which may provide a new lead compound for the treatment of NIDDM.
Footnotes
Acknowledgement
The authors are indebted to Department of Pharmacology and Central Instrumentation Facility (CIF), Jamia Hamdard, for providing necessary facilities as well as they are thankful to Central Animal house, Jamia Hamdard, for providing facilities for animal studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.