Abstract
Because dogs are widely used in drug development as nonrodent experimental animals, using a dog model for drug-induced adverse reactions is considered to be relevant for an evaluation and investigation of a mechanism and a biomarker of clinical drug-induced adverse reactions. Skeletal muscle injury occurs by various drugs, including statins and fibrates, during drug development. However, there is almost no report of a dog model for drug-induced skeletal muscle injury. In the present study, we induced skeletal muscle injury in dogs by oral coadministration of lovastatin (LV) and fenofibrate (FF) for 4 weeks. Increases in plasma levels of creatine phosphokinase, myoglobin, miR-1, and miR-133a and degeneration/necrosis of myofibers in skeletal muscles but not in the heart were observed in LV- and FF-coadministered dogs. Plasma levels of lovastatin lactone and lovastatin acid were higher in LV- and FF-coadministered dogs than LV-administered dogs. Taken together, FF coadministration is considered to affect LV metabolism and result in skeletal muscle injury.
Introduction
Drug-induced skeletal muscle injury occurs by various drugs, and it becomes an obstacle to the development of the drugs. Statins, 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors, in particular, have been reported to cause skeletal muscle injury including rhabdomyolysis in clinical study. It was also reported that the combination therapies with fibrates, peroxisome proliferator-activated receptor agonists, increase the risk of skeletal muscle injury. 1 For example, cerivastatin was withdrawn from the market due to the high risk of rhabdomyolysis in the combination therapy with gemfibrozil. 2
Several drug-induced skeletal muscle injury models have been reported in mice and rats. 3 -7 On the other hand, in dogs, muscular dystrophy models have been reported as a muscle injury model 8,9 ; however, very few drug-induced skeletal muscle injury models have been reported in dogs. 10 Because dogs are widely used in drug development as a nonrodent experimental animal, a drug-induced skeletal muscle injury model in dogs is considered to be useful for evaluation and investigation of a mechanism and a biomarker of a clinical drug-induced skeletal muscle injury.
Clinical skeletal muscle injuries were reported in each monotherapy of lovastatin (LV) and fenofibrate (FF). 1,11 In rats, skeletal muscle injuries were also reported by each mono-administration of these drugs. 3,4,12 However in dogs, skeletal muscle injury was not observed in oral mono-administration of each drug, 13 -15 and a coadministration study of these drugs has never been reported.
In this study, we intended to induce skeletal muscle injury in the dog by coadministration of LV and FF.
Materials and Methods
Chemicals and Reagents
Both LV and FF were purchased from Tokyo Chemical Industry (Tokyo, Japan). The drugs were packed in 1 or 2 capsules (1/2-oz gelatin capsule, Torpac Inc, Fairfield, New Jersey) for daily dosing. The capsules were prepared 1 day before the first administration, on days 1, 7, 13, and 20, and stored at room temperature under protection from light until dosing.
The miRNeasy serum/plasma kit was purchased from Qiagen (Valencia, California). The TaqMan microRNA (mRNA) reverse transcription (RT) kit and TaqMan miRNA assays, including the miRNA-specific primer (ath-miR159a-3p, cfa-miR-1, and cfa-miR-133a) and TaqMan Universal Master Mix II with no uracil-N-glycosylase (UNG) were obtained from Applied Biosystems (Foster City, California). Ath-miR159a-3p was purchased from Thermo Fisher Scientific (Waltham, Massachusetts). The dog myoglobin (MYO) enzyme-linked immunosorbent assay (ELISA) kit was purchased from Life Diagnostics (West Chester, Pennsylvania).
Animals and Drug Treatment
Six male beagle dogs and 6 female beagle dogs (conventional) were purchased from Japan Laboratory Animals (Tokyo, Japan). Each dog was individually housed in a stainless-steel cage in an air-conditioned room with a 12-hour light–dark cycle, ventilated with an air-exchange rate of 13 to 17 times per hour, and maintained at 23°C ± 2°C with a relative humidity of 60% ± 10%. Each dog was provided with 300 g/d of food (CD-5M, Clea Japan, Tokyo, Japan) in the morning. Each dog was also provided in-house tap water ad libitum via an automatic watering device. Dogs were divided into 3 groups: LV, FF, and LV/FF combination (Table 1). On the first day of dosing, the male dogs were 25 to 34 months old, and the female dogs were 30 to 34 months old.
Dosage of LV and FF and Animal Number in the Present Animal Groups.

Abbreviations: FF, fenofibrate; LV, lovastatin.
Body weight measurement and drug preparation were performed on days 0, 1, 7, 13, and 20. An appropriate quantity of the drug was calculated based on the most recent body weight measured, and the drug was placed in gelatin capsule for daily dosing (LV: 180 mg/kg/d, FF: 400 mg/kg/d) and stored until dosing. The gelatin capsules containing LV or FF were administered once daily for 4 weeks (28 days) before providing food in the morning by placing them into the pharynx to ensure swallowing by the dogs.
All procedures performed on the animals were approved by the Institutional Guidelines for Animal Care and Use of Otsuka Pharmaceutical Co, Ltd (approval no. 16-0370).
Plasma Biochemical Analysis
A blood biochemical test was performed on days 0, 7, 13, 20, and 27. Approximately 2 mL of the blood sample was obtained via a heparinized syringe from the cephalic vein of each dog before providing food in the morning. The heparinized plasma obtained after centrifugation (3,000 rpm, 4°C, 20 minutes) was used for the analyses of creatine kinase (CK) and MYO. Activity of CK in plasma was measured using the Hitachi-7180 (Hitachi High-Technologies Corp, Tokyo, Japan) according to the manufacturer’s instructions. Concentrations of MYO were measured using dog MYO ELISA, a microplate reader (
Histopathological Examination
Dogs were anesthetized with sodium pentobarbital and euthanized by exsanguination from the carotid artery on the day after the final dosing (day 29). The following organs and tissues were fixed in 10% neutral-buffered formalin: quadriceps, intercostal muscle, gastrocnemius, diaphragm, tongue, and heart. The tissues obtained were dehydrated with a graded series of ethyl alcohol, dealcoholized with xylene, and embedded in paraffin. These tissues in paraffin blocks were thinly sectioned and stained with hematoxylin and eosin (H&E). The specimens were examined with a light microscope.
Micro-RNA Extraction and Real-Time Reverse Transcription Polymerase Chain Reaction
The miRNA extraction from plasma was performed on all dogs on days 0, 7, 13, 20, and 27. Approximately 1 mL of the blood sample was obtained from the cephalic vein of each dog and mixed with EDTA-2K before providing food in the morning. The plasma obtained after centrifugation (3,000 rpm, 4°C, 20 minutes) was used. The plasma samples were mixed with QIAzol lysis reagent (Qiagen), and as a spike in control,
Concentration of Drugs in Plasma
A blood sample (1 mL) was obtained via a heparinized syringe from the cephalic vein of each dog at 1, 2, 4, 8, and 24 hours after the administration on day 1 and at 0, 1, 2, 4, 8, and 24 hours after the drug administration on day 27. The heparinized plasma obtained after centrifugation (3,000 rpm, 4°C, 20 minutes) was used for the measurement of lovastatin lactone (LVL), lovastatin acid (LVA), and fenofibric acid (FFA). Fifty microliters of plasma sample was mixed with 10 μL of methanol, 50 μL of 100 mM sodium acetate buffer solution (pH 4.7), 0.2 mL of methanol/acetic acid (1000:1, v/v), and 10 μL of glimepiride and simvastatin (500 ng/mL and 100 ng/mL, respectively) as an internal standard. After incubation at 10°C for 30 minutes, the mixture was centrifuged at 21 600 rpm at 4°C for 5 minutes, and the supernatant was analyzed by liquid chromatography (LC) with tandem mass spectrometry (MS/MS). The LC used Atlantis T3 column (3 μm, 50 mm × 2.1 mm id; Waters Corp, Milford, Massachusetts) with a binary gradient solvent system consisting of the following: (a) water/formic acid (1,000:1, v/v); or (b) acetonitrile/formic acid (1,000:1, v/v). The chromatography was performed using a Nanospace SI-2 HPLC system (Osaka Soda Co, Ltd, Osaka, Japan). The column temperature was maintained at 40°C, and the flow rate was 0.4 mL/min. The LC eluate was introduced directly into a TSQ Quantum Ultra triple-quadrupole mass spectrometer (Thermo Fisher Scientific Inc), equipped with an electrospray ionization interface operated in negative-ion mode and positive-ion mode. Quantification was performed using selected reaction monitoring (SRM). The LC–MS/MS parameters are shown in Table 2. The detailed MS conditions were as follows: spray voltage, −2,000 V (negative) and 2,000 V (positive); vaporizer temperature, 220°C; sheath gas pressure (nitrogen), 30; auxiliary gas pressure (nitrogen), 60; ion sweep gas pressure (nitrogen), 0; capillary temperature, 250°C; collision pressure (argon), 1.0 mTorr; skimmer offset, 10 V (negative) and −10 V (positive).
LC-MS/MS Parameters for the Analytes and Internal Standards.
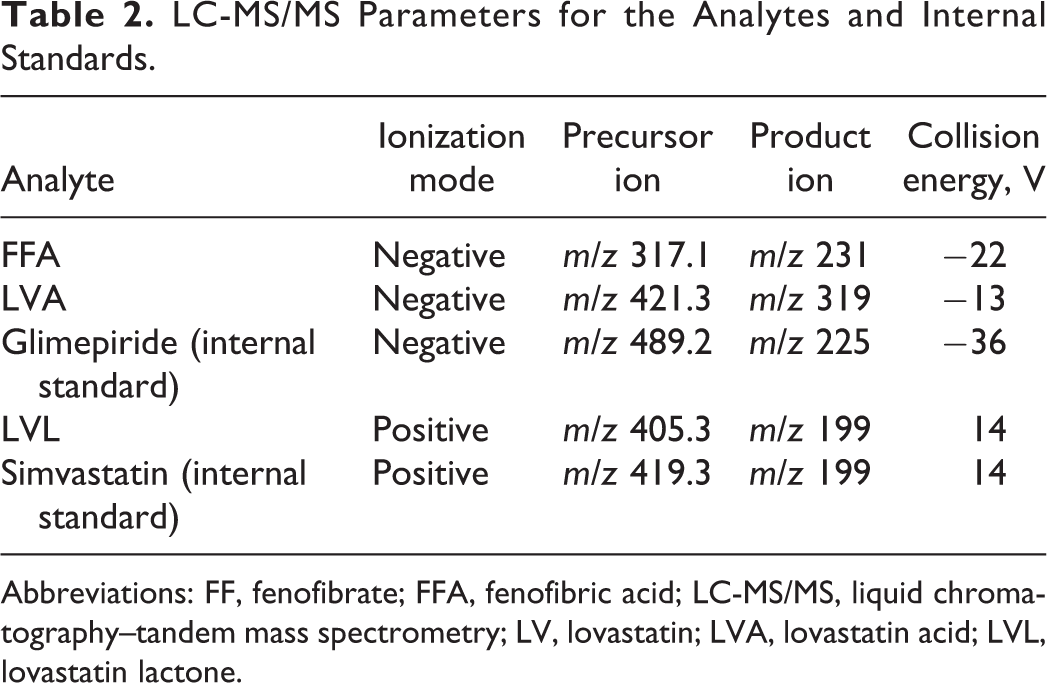
Abbreviations: FF, fenofibrate; FFA, fenofibric acid; LC-MS/MS, liquid chromatography–tandem mass spectrometry; LV, lovastatin; LVA, lovastatin acid; LVL, lovastatin lactone.
Statistical Analysis
All values were represented as the mean ± standard error of the mean. Statistical analysis was performed using JMP Software (JMP Pro, version 13.0.0; SAS Institute Inc, Cary, North Carolina). Comparisons of 2 groups were performed using a 2-tailed
Results
Increase in Plasma CK and MYO in LV/FF-Coadministered Dogs
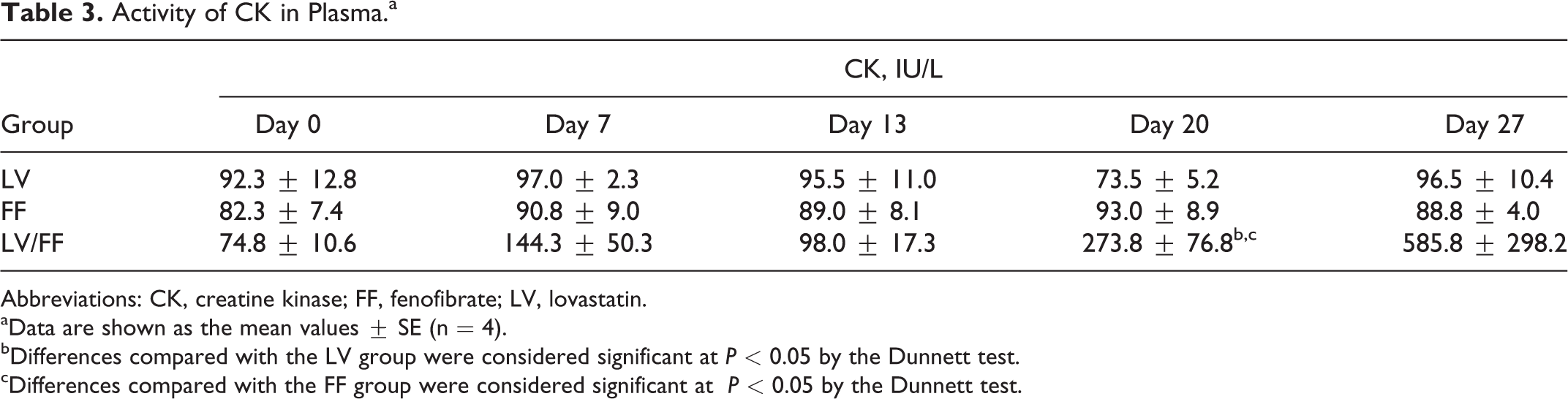
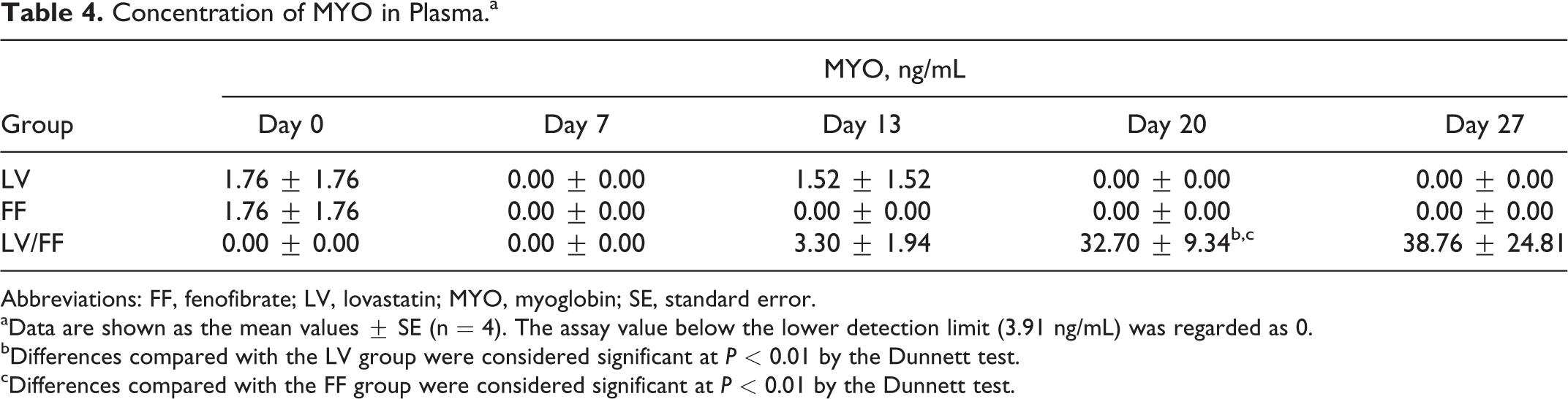
The plasma CK and MYO were measured on days 0, 7, 13, 20, and 27. Plasma CK activity (Figure 1A and Table 3) and MYO concentration (Figure 1B and Table 4) in LV/FF group were increased beginning on day 20 compared to that on day 0 in LV/FF group. The values of CK and MYO in LV/FF group were increased also compared to that in day-matched (days 20 and 27) LV and FF groups. There was no change in these values in both LV and FF groups.
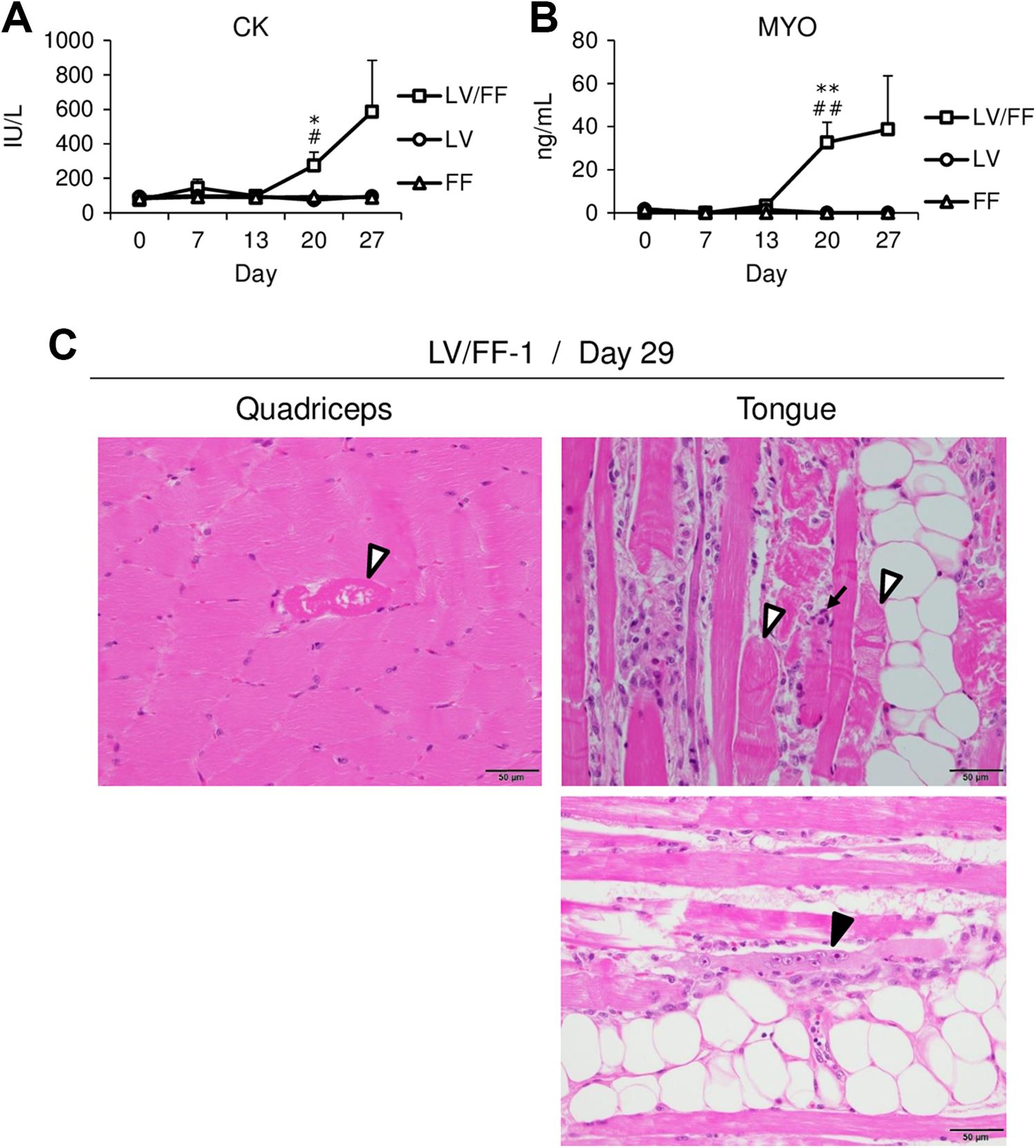
Time-dependent changes in plasma CK (A) and MYO (B) in LV, FF, and LV/FF groups. The plasma CK and MYO were measured on days 0, 7, 13, 20, and 27. The data are shown as the mean values ± SE (n = 4). Differences compared to the LV group were considered significant at *
Activity of CK in Plasma.a
Abbreviations: CK, creatine kinase; FF, fenofibrate; LV, lovastatin.
aData are shown as the mean values ± SE (n = 4).
bDifferences compared with the LV group were considered significant at
cDifferences compared with the FF group were considered significant at
Concentration of MYO in Plasma.a
Abbreviations: FF, fenofibrate; LV, lovastatin; MYO, myoglobin; SE, standard error.
aData are shown as the mean values ± SE (n = 4). The assay value below the lower detection limit (3.91 ng/mL) was regarded as 0.
bDifferences compared with the LV group were considered significant at
cDifferences compared with the FF group were considered significant at
Histopathological Change in Skeletal Muscle
The degeneration/necrosis of myofibers, characterized by marked eosinophilic sarcoplasm, loss of striations, and fragmentation (white arrowhead), infiltration of inflammatory cells (arrow), and regeneration of myofibers, characterized by basophilic sarcoplasm and internal vesiculated nuclei with prominent nucleoli (black arrowhead), were observed in the skeletal muscles in LV/FF-1 dog (Figure 1C). The infiltrated inflammatory cells were mononuclear cells, which did not have segmented nuclei or eosinophil or basophil granule. As shown in Table 5, minimal, slight, or moderate degeneration/necrosis of myofibers was observed in the tongue, femoral quadriceps, intercostal muscle, or diaphragm in all dogs of all groups. Regeneration of myofibers and infiltration of inflammatory cells were observed in the tongue in only LV/FF-1 dog. These histopathological changes were not observed in our historical control slides of dogs that were orally administered empty capsules once daily for 4 weeks (data not shown). As the histological score, “minimal” means that the change was observed in 5% below of myofibers, “slight” means about 10%, and “moderate” means about 25%. None of the histopathological changes were observed in the gastrocnemius muscle and heart in any dogs.
Histopathological Examination.a

Abbreviations: FF, fenofibrate; LV, lovastatin.
a− indicates no change, ±: minimal,
Increase in Skeletal Muscle–Specific miRNAs in Plasma
The plasma miR-1 and miR-133a levels were measured by real-time RT-PCR on days 0, 7, 13, 20, and 27. Plasma miR-1 level in the LV/FF group was increased beginning on day 20 compared to that on day 0 in the LV/FF group and in the day-matched (days 20 and 27) FF group and on day 20 with that in the LV group (Figure 2A and Table 6). The plasma miR-133a level was increased beginning on day 20 in the LV/FF group compared to that on day 0 in the LV/FF group and in the day-matched (days 20 and 27) LV and FF groups (Figure 2B and Table 7). The plasma miR-133a level was highly correlated with plasma CK (Figure 2D). However, the correlation between plasma miR-1 level and CK activity was not as high (Figure 2C).
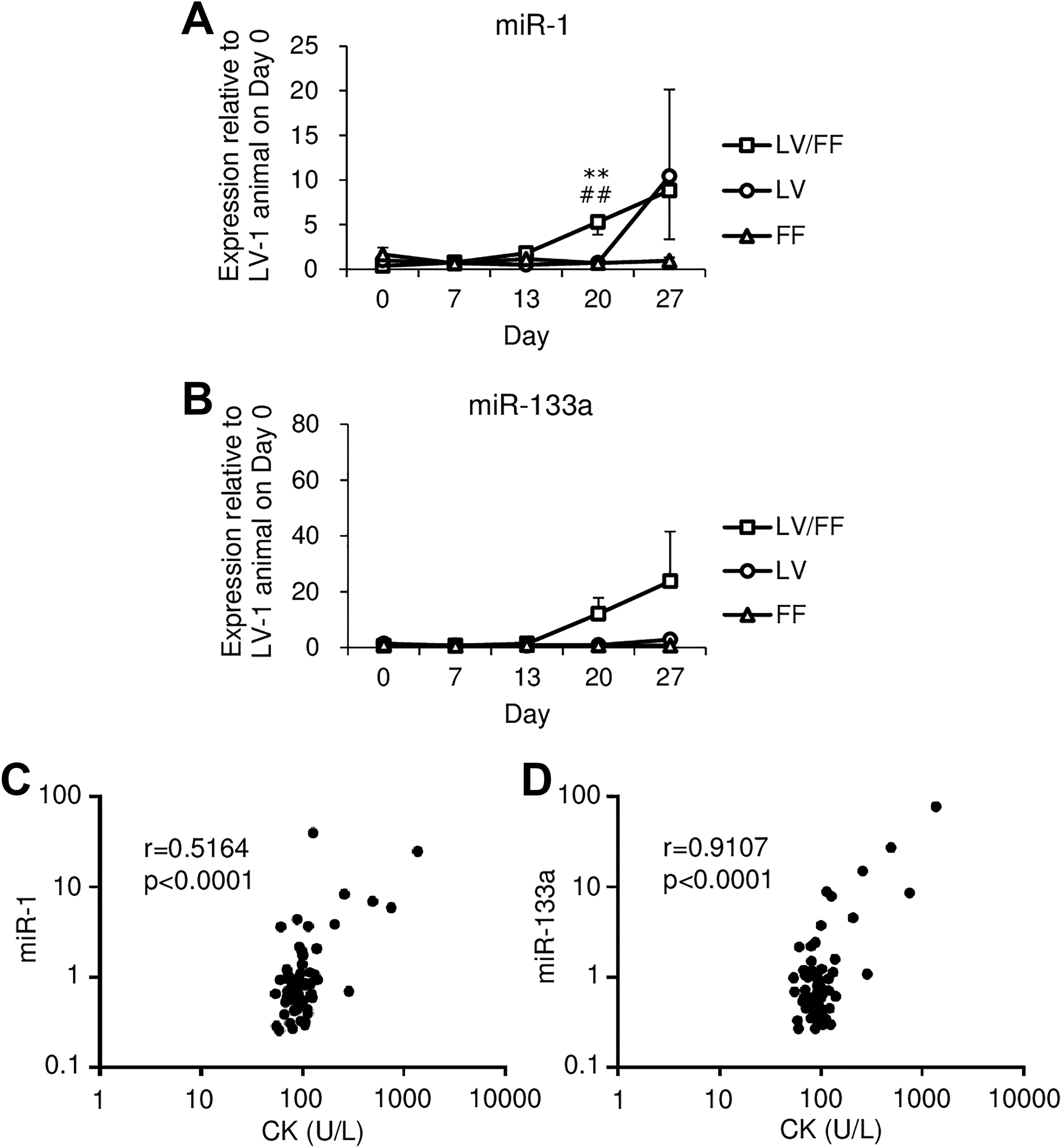
Time-dependent changes in plasma miR-1 (A) and miR-133a (B) levels in LV, FF, and LV/FF groups. The plasma miR-1 and miR-133a were measured on days 0, 7, 13, 20, and 27. The relative expression levels of the plasma miRNAs were measured using real-time RT-PCR and normalized to that of ath-miR159a-3p (spike in control). The fold changes compared with the values of LV-1 animal on day 0 were shown. The data are shown as the mean values ± SE (n = 4). Differences compared with the LV group were considered significant at **
MiR-1 Level in Plasma.a
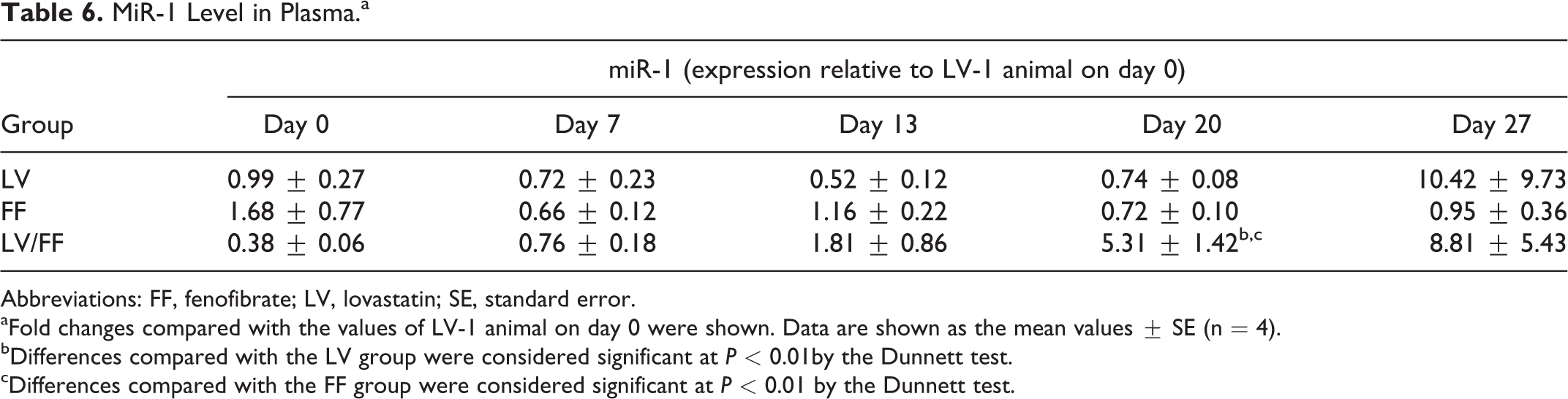
Abbreviations: FF, fenofibrate; LV, lovastatin; SE, standard error.
aFold changes compared with the values of LV-1 animal on day 0 were shown. Data are shown as the mean values ± SE (n = 4).
bDifferences compared with the LV group were considered significant at
cDifferences compared with the FF group were considered significant at
MiR-133a level in Plasma.a

Abbreviations: FF, fenofibrate; LV, lovastatin; SE, standard error.
aFold changes compared with the values of LV-1 animal on day 0 were shown. Data are shown as the mean values ± SE (n = 4).
Changes in Plasma Concentration of LVL, LVA, and FFA by LV/FF Coadministration
The plasma concentration of LVL, LVA, and FFA were measured at 1, 2, 4, 8, and 24 hours after the administration on day 1 and at 0, 1, 2, 4, 8, and 24 hours after the administration on day 27. The LVL plasma concentration was significantly increased at 1, 2, 4, and 8 hours on day 1 and at 1, 2, 4, and 24 hours on day 27, and maximum plasma concentration (
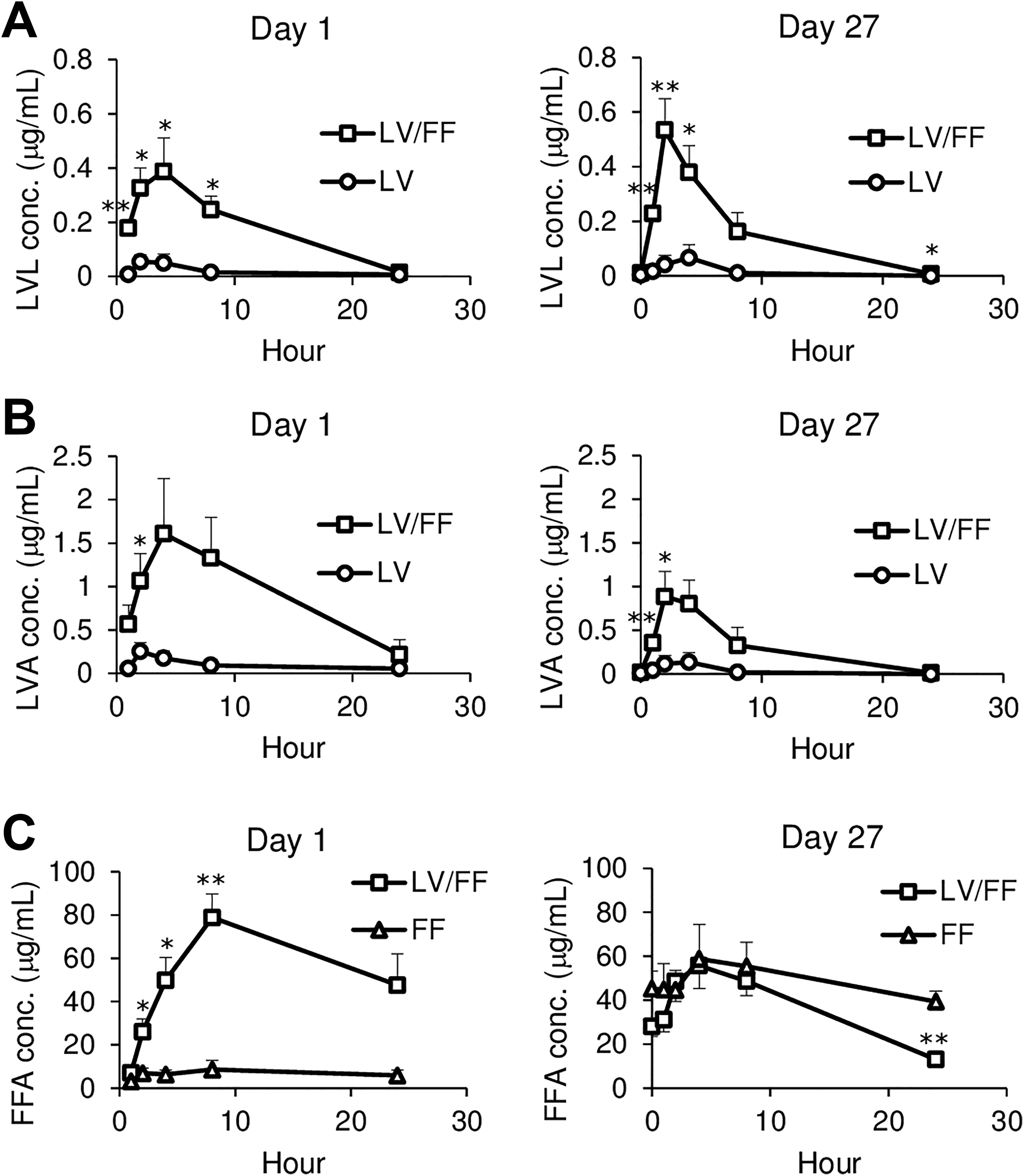
Time-dependent changes in the plasma concentrations of LVL (A), LVA (B), and FFA (C) in LV, FF, and LV/FF groups. The plasma concentrations of LVL, LVA, and FFA were measured at 1, 2, 4, 8, and 24 hours after the administration on day 1 and at 0, 1, 2, 4, 8, and 24 hours after the administration on day 27. The data are shown as the mean values ± SE (n = 4). Differences compared to the LV group (A, B) or FF group (C) were considered significant at *
Pharmacokinetic Parameters of LVL.
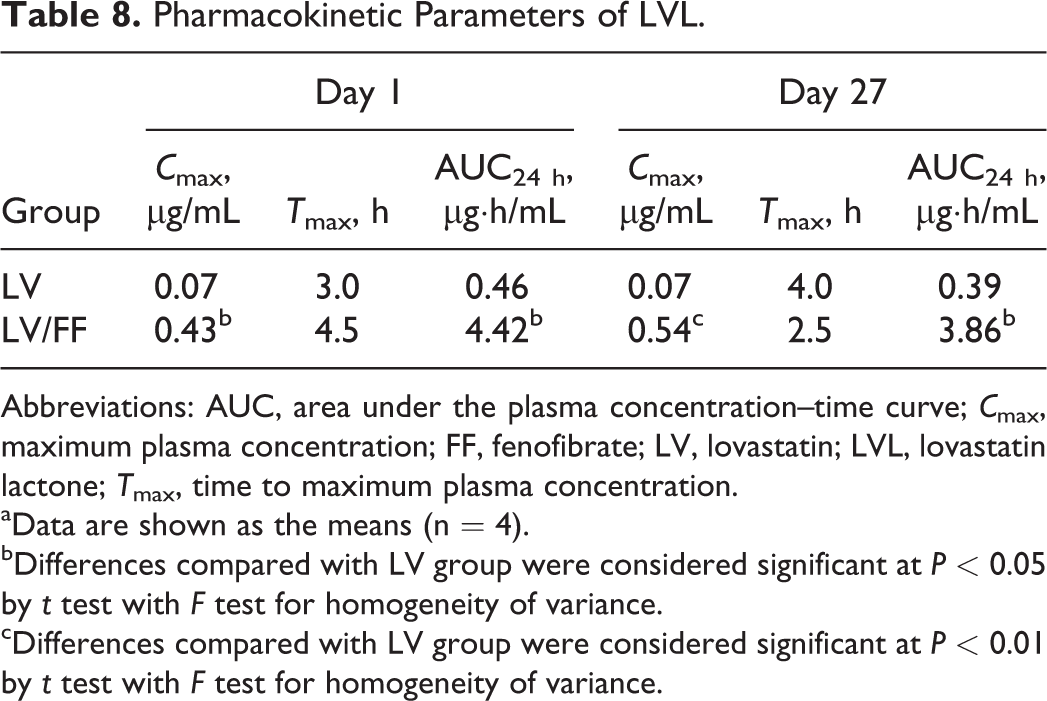
Abbreviations: AUC, area under the plasma concentration–time curve;
aData are shown as the means (n = 4).
bDifferences compared with LV group were considered significant at
cDifferences compared with LV group were considered significant at
Pharmacokinetic Parameters of LVA.
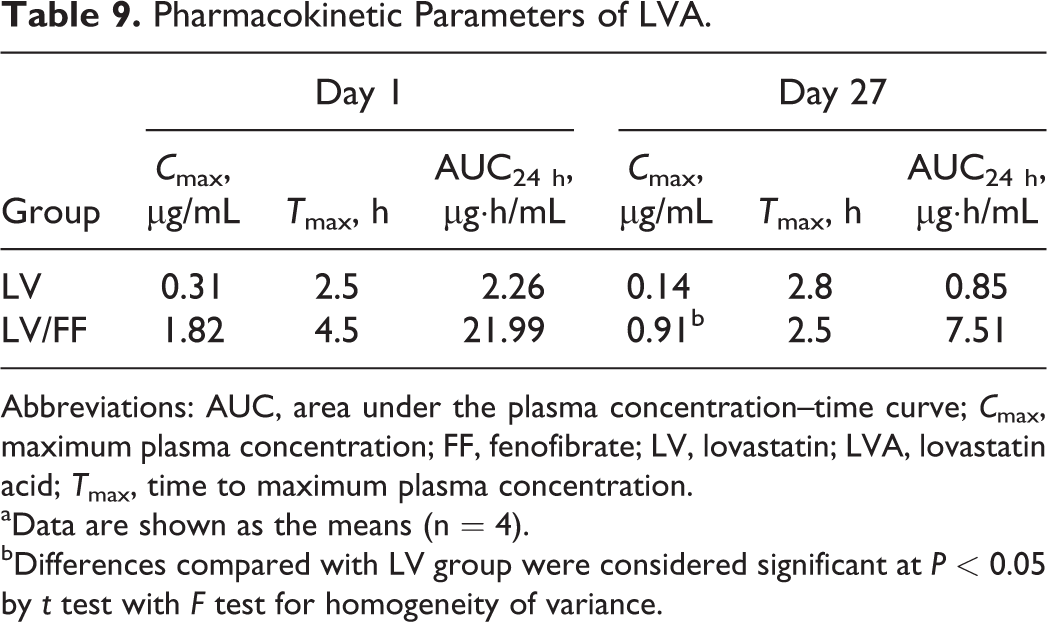
Abbreviations: AUC, area under the plasma concentration–time curve;
aData are shown as the means (n = 4).
bDifferences compared with LV group were considered significant at
Pharmacokinetic Parameters of FFA.a
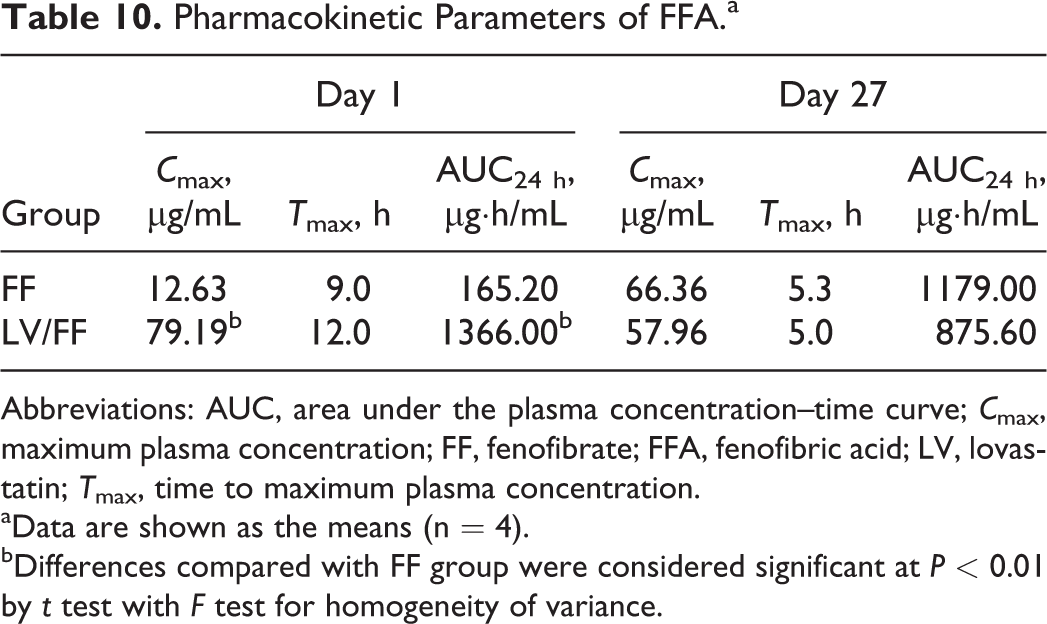
Abbreviations: AUC, area under the plasma concentration–time curve;
aData are shown as the means (n = 4).
bDifferences compared with FF group were considered significant at
Discussion
Because the dog is widely used in drug development as a nonrodent experimental animal, a drug-induced adverse reaction dog model is considered to be useful for an evaluation and investigation of a mechanism and a biomarker of clinical drug-induced adverse reactions. Skeletal muscle injury occurs by various drugs, including statins and fibrates, and it becomes an obstacle in the development of drugs. A drug-induced skeletal muscle injury in dogs has been reported only by long-term administration (12 weeks increasing dose up to 320 mg/kg) of atorvastatin. 10 Therefore, we intended to induce skeletal muscle injury in the dog by a drug administration.
A previous clinical study reported that LV caused skeletal muscle injury in 0.1% to 0.2% of patients in monotherapy, and combination therapies with other drugs (e.g., fibrates, cyclosporine) increased the risk of skeletal muscle injury including rhabdomyolysis. 11,13,16 It was also reported that FF caused rhabdomyolysis in 2.78 per 100,000 patient-years in monotherapy, and combination therapies with statins increased the risk to 15.00 per 100,000 patient-years. 1 Therefore, we selected LV and FF coadministration for inducing skeletal muscle injury in this study.
In dogs, skeletal muscle injury was not observed in any oral mono-administration of LV (up to 28 weeks of repeated dosing at 180 mg/kg) and FF (up to 13 weeks at 900 mg/kg and up to 52 weeks at 400 mg/kg). 13 -15 Therefore, we selected oral coadministration of these drugs, and selected maximum nonlethal daily doses for 4 weeks reported in previous reports as each drug dosage in this study. In the repeated oral dose study of LV in beagle dogs, 180 mg/kg was reported as a lethal dose; however, the dosing period was not mentioned. 13 On the other hand, 180 mg/kg was used in the 14- and 28-week repeated oral dose toxicity study, 14 suggesting that 180 mg/kg is a nonlethal dose for 14 weeks of repeated dosing. Therefore, the dose of 180 mg/kg LV was adopted in the present study. Moreover, the dose of 400 mg/kg FF was considered to be a feasible dose because a dosage of 400 mg/kg was reported to be a lethal dose in a 52-week repeated oral dose study in beagle dogs. 15 Thus, the dose of 400 mg/kg FF was adopted in the present study.
Rhabdomyolysis is a syndrome that causes a breakdown of skeletal muscle and leakage of MYO and other intracellular molecules into the circulation. As an increase in serum CK and, in some cases, myoglobinemia and myoglobinuria occur, they are used as a diagnostic index in the syndrome. 17 In this study, increase in plasma CK activity and MYO concentration were observed in the LV/FF group but not in the LV and FF groups (Figure 1A and B and Tables 3 and 4). Increase in plasma CK and MYO also occur in cardiac dysfunction such as myocardial infarction; however, there was no histopathological change observed in the heart of any dogs in this study (Table 5). Therefore, skeletal muscle injury was more involved in the increase in CK and MYO. More than 1,000 U/L CK in serum is generally considered to be the diagnostic criteria of rhabdomyolysis. 17 In LV/FF-1 dog, the plasma CK activity (1,360 U/L on day 27, data not shown) exceeded the criteria, and degeneration/necrosis or regeneration of myofibers and infiltration of inflammatory cells were observed in the skeletal muscles (Figure 1C and Table 5). In the other dogs in all groups, minimal or slight degeneration/necrosis of myofibers was observed in the skeletal muscles (Table 5). In histopathological examination, it was not clarified whether the coadministration of LV and FF caused an exacerbation of skeletal muscle injury such as the injury observed in LV/FF-1 animal due to the limited number of animals for animal welfare concern. However, the coadministration was considered to be somewhat involved in an exacerbation of skeletal muscle injury, since CK and MYO values were higher in the LV/FF group than in the LV and FF groups. Contrary to expectation, the slight histopathological changes of skeletal muscle were observed in the mono-administered groups (1 dog in each LV and FF group). In dogs, skeletal muscle injury was not observed in each oral administration drug study 13 -15 ; thus, this discrepancy needs to be clarified.
MicroRNAs are small noncoding RNA that are being studied as a potential noninvasive biomarker for tissue injury due to their high stability in blood.
18,19
The miR-1, miR-133a, miR-206, and others were reported to be plasma biomarkers of skeletal muscle injury in muscular dystrophy dog models
8,9
; however, a drug-induced injury model is better for a study of biomarkers for a drug safety assessment. Therefore, plasma miRNAs were evaluated in the present study. In the miRNAs that are predominantly expressed in the skeletal muscle in dogs,
20
only miR-1 and miR-133a in the plasma of the LV/FF group tended to increase on day 27 compared to that on day 0 in preliminary miRNA array analysis (data not shown). In the PCR analysis, plasma miR-1 and miR-133a in the LV/FF group were increased compared to those in the LV and FF groups (Figure 2A and B and Tables 6 and 7). In LV/FF-1 dog that had the most severe skeletal muscle injury, plasma miR-1 and miR-133a levels were the highest in all dogs of all groups (on day 27, miR-1: 24.8-fold, miR-133a: 76.9-fold compared with LV-1 animal on day 0, data not shown). Both miR-1 and miR-133a are also predominantly expressed in the dog heart as well as skeletal muscle
20
; however, there was no histopathological change observed in the heart of any dogs in this study (Table 5). Therefore, skeletal muscle injury was more involved in the increase in the miRNAs. Plasma miR-133a was highly correlated with CK (
The changes in plasma LVL, LVA, and FFA levels were measured in this study, because in circulating blood, LV consists of LVL, a nonactivated form, or LVA, an activated form
21
; moreover, almost all FF exists as FFA, an activated form.
15
Plasma LVL and LVA levels in the LV/FF group were increased compared to that in the LV group both on day 1 (LVL;
On day 27, the plasma FFA level in the LV/FF group was not increased compared to that in the FF group (
In conclusion, we induced skeletal muscle injury in dogs by oral coadministration of LV and FF for 4 weeks. In addition, we found that plasma LVL and LVA levels were increased by coadministration of FF in the dogs. These findings may contribute to the evaluation and investigation of a biomarker in drug-induced skeletal muscle injury and provide new important information on a drug interaction.
Footnotes
Authors Contributions
Kawata, R, contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Yokoi, T, contributed to design and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
