Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of fatty acid amidopropyl dimethylamines, which function primarily as antistatic agents in cosmetic products. The relevant animal and human data reviewed for these ingredients indicate that they are potential dermal sensitizers that may be due in part by the sensitizing impurity, 3,3-dimethylaminopropylamine. The Panel concluded that fatty acid amidopropyl dimethylamines were safe as cosmetic ingredients when they are formulated to be nonsensitizing, which may be based on a quantitative risk assessment.
Introduction
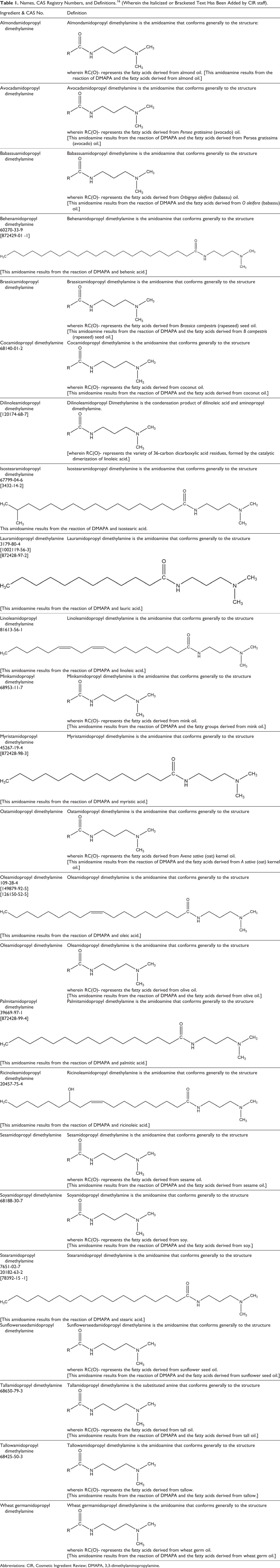
The fatty acid amidopropyl dimethylamines function primarily as antistatic agents in cosmetic products. These chemicals are sometimes referred to as “amidoamines.” The full list of ingredients in this safety assessment is found in Table 1.
Names, CAS Registry Numbers, and Definitions. 16 (Wherein the Italicized or Bracketed Text Has Been Added by CIR staff).
Abbreviations: CIR, Cosmetic Ingredient Review; DMAPA, 3,3-dimethylaminopropylamine.
In December 2010, the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) issued a final amended safety assessment on cocamidopropyl betaine (CAPB) and related fatty acid amidopropyl betaines. 1 The Panel concluded that these ingredients “were safe in cosmetics when they are formulated to be non-sensitizing, which may be based on a quantitative risk assessment.” The Panel was aware of impurities that may exist in the amidopropyl betaines and expressed concern over their sensitizing potential. Those impurities were 3,3-dimethylaminopropylamine (DMAPA) and the fatty acid amidopropyl dimethylamines presented as ingredients in this report. A quantitative risk assessment (QRA) of DMAPA at a concentration of 0.01% in raw CAPB indicated no sensitization in finished cosmetic products; amidoamine at a concentration of 0.5% in raw CAPB may cause sensitization in certain finished cosmetic products. The Panel advised industry to continue minimizing the concentrations of the sensitizing impurities. The summaries of the studies on DMAPA and amidoamine along with a summary of the QRA on these 2 chemicals that the Panel reviewed in the CAPB safety assessment have been incorporated into this safety assessment.
Toxicological data on stearamidopropyl dimethylamine (synonym: N-[3-(dimethylamino)propyl] stearamide) in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. These data are available on the ECHA website. 2
Chemistry
The definitions and CAS registry numbers, where available, and structures of the fatty acid amidopropyl dimethylamines ingredients are presented in Table 1. The available information on the physical and chemical properties of these ingredients are presented in Table 2.
Physical and Chemical Properties.
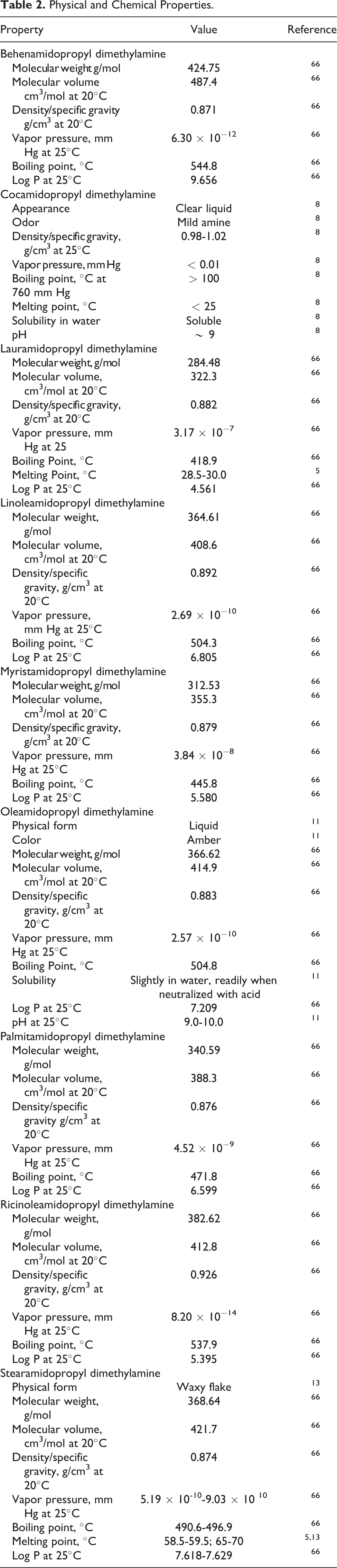
The ingredients in this review each have the same core structure of a fatty acid amide, N-substituted with 3 propyl-N, N-dimethylamine. These ingredients are manufactured by the amidation (ie, amide-forming condensation) of fatty acids with DMAPA, most commonly under alkaline or acidic conditions (Figure 1). 3,4 The resultant ingredients have an identical core, with 2 primary functional groups, a secondary amide and a tertiary amine, separated by a propyl chain. These ingredients differ only by the identity of the fatty acid chain(s) attached to the amide functional group of this core. The synthesis of these ingredients is a clean process with little to no by-products and typically yields products that are 98% to 99% pure fatty acid amidopropyl dimethylamines. 5 Accordingly, starting materials, such as DMAPA, represent the largest concern for impurities.
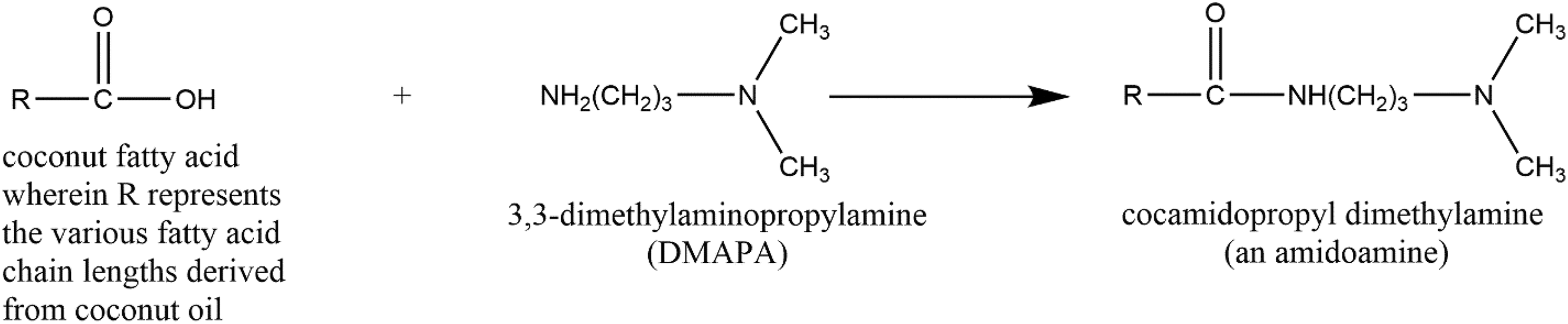
Synthesis of cocamidopropyl dimethylamine.
Despite the long alkyl chain substituents therein, these ingredients are readily solubilized in water, as they are easily converted to ammonium salts (ie, cationic surfactants) at even mildly acidic pH values (ie, the tertiary amines are protonated to form ammonium cations; these ingredients are alkaline materials with pKb values in the range of 5-6). 4,5 Because of their high polarity, both as the free tertiary amines and as the ammonium salts formed in situ, these ingredients are excellent dissipators of triboelectric charges (ie, static electricity), even at low concentrations (eg, 0.1% wt/wt). 5 –7 This property likely accounts for the claimed functions of these ingredients as antistatic agents and, at least in part, as conditioning agents. Although not formally claimed, these ingredients are also known to operate as functional surfactants, thickeners, and bacteriostatic agents. 5
Method of Manufacturing
Cocamidopropyl Dimethylamine
According to a supplier, cocamidopropyl dimethylamine is made by mixing together refined coconut oil with DMAPA and heating the mixture to >75°C and <175°C. 8 The progress of the reaction is followed using standard analytical tests until specifications are met. The product is then filtered and stored in lined steel drums.
N-Nitrosation and Safety Issues
Although nitrosamine content has not been reported, fatty acid amidopropyl dimethylamines are composed of secondary amides and tertiary amines and potentially can be nitrosated. Of the approximately 209 nitrosamines tested, 85% have been shown to produce cancer in laboratory animals. 9 Nitrosation can occur under physiologic conditions. 3 Depending on the nitrosating agent and the substrate, nitrosation can occur under acidic, neutral, or alkaline conditions. Atmospheric NO2 may also participate in the nitrosation of amines in aqueous solution. 4 Accordingly, fatty acid amidopropyl dimethylamines should be formulated to avoid the formation of nitrosamines.
Impurities
Behenamidopropyl Dimethylamine
A supplier has indicated that the maximum level of DMAPA in behenamidopropyl dimethylamine is 115 ppm. 10 The supplier stated that the typical use level of this material in hair conditioners is 2.3%, which results in a maximum DMAPA level of 2.65 ppm in the finished product.
Cocamidopropyl Dimethylamine
A supplier reported the final composition of the product cocamidopropyl dimethylamine to be 83% to 90% cocamidopropyl dimethylamine, 8.9% to 9.4% glycerin, 1.0% (max) DMAPA, and 5.0% (max) glyceryl esters. 8
Oleamidopropyl Dimethylamine
A product description sheet indicates that oleamidopropyl dimethylamine is at minimum 88% pure and has a maximum concentration of 0.60% DMAPA. 11
Stearamidopropyl Dimethylamine
The maximum level of DMAPA in stearamidopropyl dimethylamine has been reported to be 30 ppm. 12 The supplier stated that, in the typical use concentration of 2.14% stearamidopropyl dimethylamine in hair conditioners, the DMAPA level in the finished product is a maximum of 0.65 ppm. Another supplier indicated that the free DMAPA in stearamidopropyl dimethylamine is less than 0.2%. 13
In another sample of stearamidopropyl dimethylamine, the chemical composition was at minimum 97% of the active matter and contained at maximum 0.002% free DMAPA and 3.0% free fatty acid.
14
The C-chain distribution for this sample of stearamidopropyl dimethylamine was reported as the following: < C16
Use
Cosmetic
All but 1 of the 24 fatty acid amidopropyl dimethylamines included in this safety assessment function as antistatic agents in cosmetic formulations. 16 Brassicamidopropyl dimethylamine is reported to function as a hair- and skin-conditioning agent. In addition to being an antistatic agent, stearamidopropyl dimethylamine is reported to function as a hair conditioning agent.
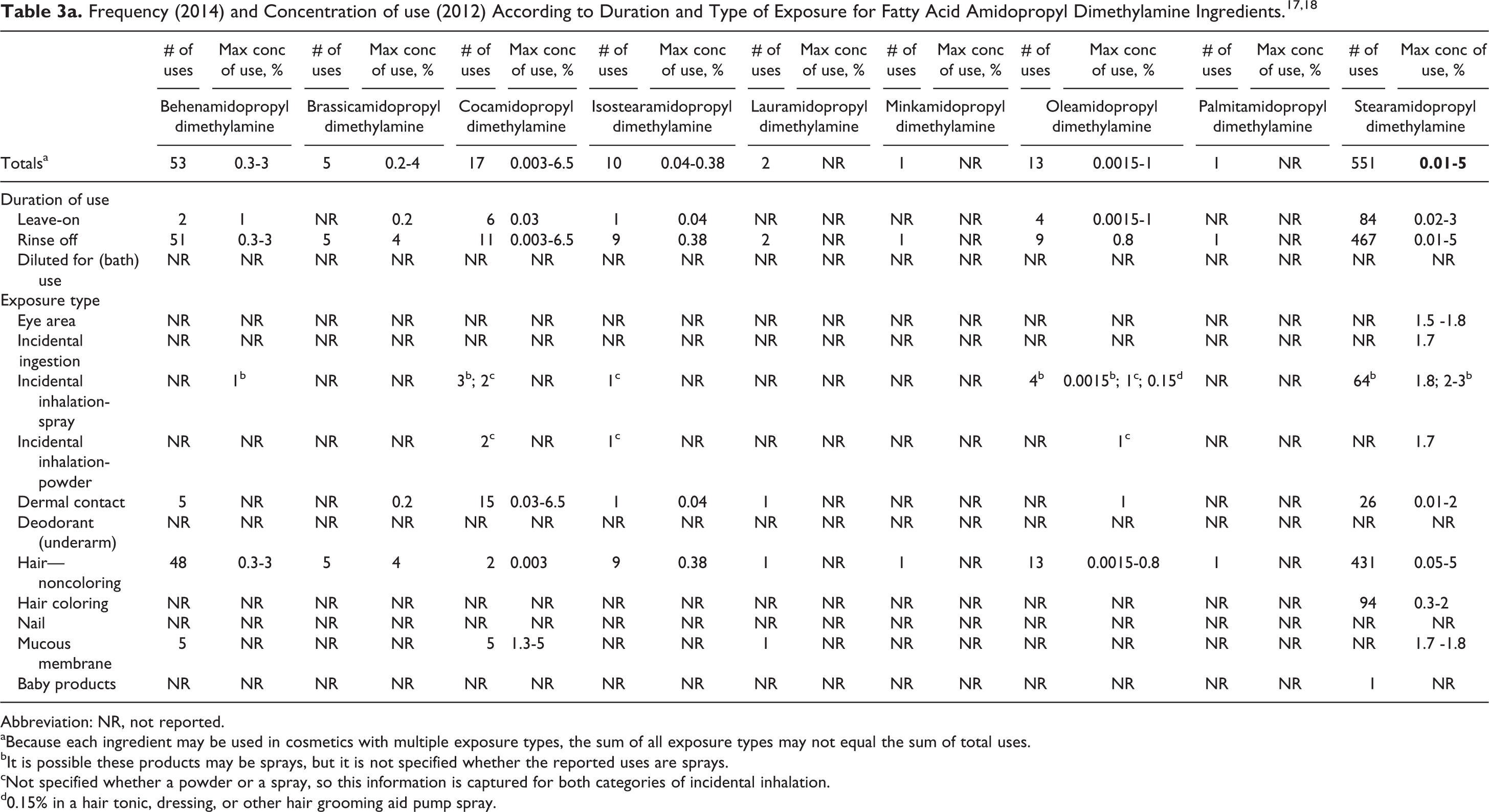
Table 3 presents the frequency and maximum use concentration ranges for fatty acid amidopropyl dimethylamines. According to information supplied to the Food and Drug Administration by industry as part of the Voluntary Cosmetic Registration Program (VCRP), stearamidopropyl dimethylamine has the most reported uses in cosmetic and personal care products, with a total of 427; 355 of those uses are in rinse-off formulations. 17 Most of the rinse-off uses are in hair conditioners. Behenamidopropyl dimethylamine has the second greatest number of overall uses reported, with a total of 35; of those, 32 uses are in rinse-off formulations. Again, most of the rinse-off uses are in hair conditioners. A few uses were reported each for brassicamidopropyl dimethylamine (1), cocamidopropyl dimethylamine (6), isostearamidopropyl dimethylamine (13), lauramidopropyl dimethylamine (1), minkamidopropyl dimethylamine (1), oleamidopropyl dimethylamine (12), and palmitamidopropyl dimethylamine (1). No uses were reported to the VCRP for the remaining fatty acid amidopropyl dimethylamines.
Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
cNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
d0.15% in a hair tonic, dressing, or other hair grooming aid pump spray.
Ingredients Not Reported in Use.
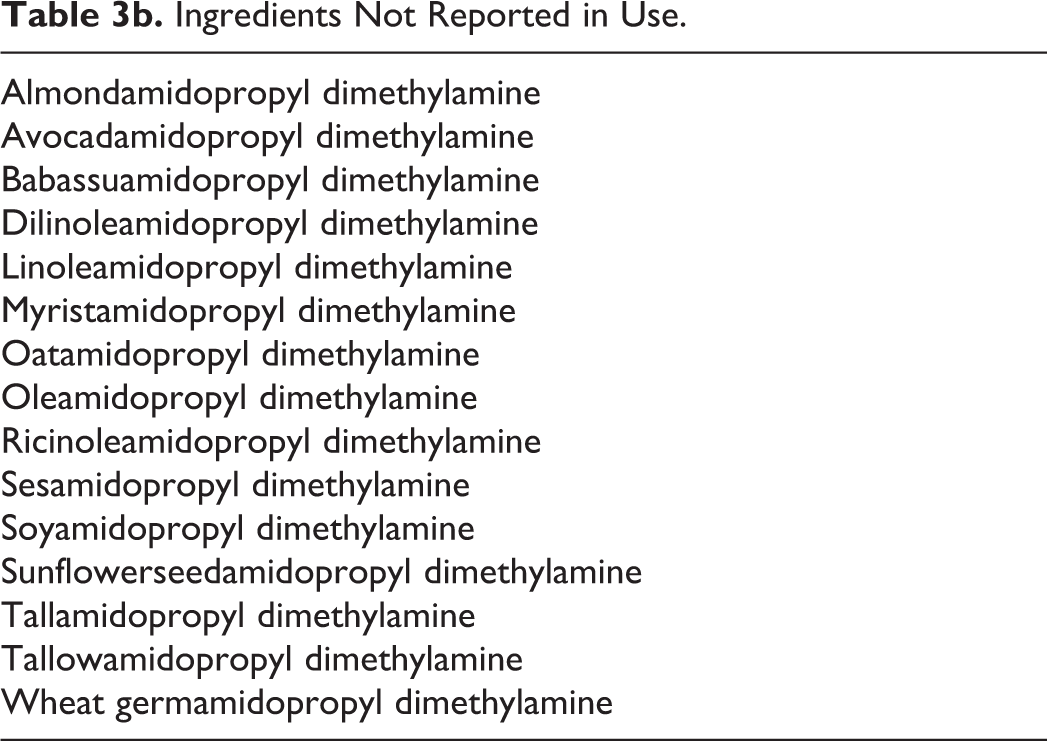
In a survey of use concentrations conducted by the Personal Care Products Council, stearamidopropyl dimethylamine is reported to be used at a range of maximum concentrations of 0.01% to 5%, with 5% reported in noncoloring hair conditioners. 18 In behenamidopropyl dimethylamine, the range of maximum concentrations was reported to be 0.3% to 3%, with 3% reported in noncoloring hair conditioners. A range of maximum concentrations for cocamidopropyl dimethylamine was reported to be 0.003% to 6.5%, with 6.5% reported in skin cleansing products. No use concentrations were reported for almondamidopropyl dimethylamine, avocadoamidopropyl dimethylamine, babassuamidopropyl dimethylamine, minkamidopropyl dimethylamine, oatamidopropyl dimethylamine, oleamidopropyl dimethylamine, sesamidopropyl dimethylamine, and tallamidopropyl dimethylamine. 19
Oleamidopropyl dimethylamine is used in cosmetic sprays, including hair tonics and dressings. Oleamidopropyl dimethylamine and stearamidopropyl dimethylamine may also be used in colognes and indoor tanning products. When used in cosmetic sprays, these ingredients could possibly be inhaled. The maximum concentration of these ingredients reported to be used in a spray product is 0.15% (oleamidopropyl dimethylamine) in a hair tonic, dressing, or other hair-grooming pump spray product. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared to pump sprays. 20,21 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 22,23 The amidoamine ingredients in this safety assessment currently are not restricted from use in any way under the rules governing cosmetic products in the European Union. 24
Noncosmetic
Myristamidopropyl dimethylamine is used as a biocide in contact lens disinfecting solution (concentration reported to be ∼0.0005%) and may have uses as a broad-spectrum therapeutic antimicrobial for keratitis and for surgical prophylaxis. 25 –30
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
In an IH Skin Perm quantitative structure–activity relationship (QSAR) model, the dermal absorption of stearamidopropyl dimethylamine has been estimated to be 0.04 mg and 0.12 mg after 8 and 24 hours, respectively. 2 The maximum dermal absorption rate was calculated to be 2.40 × 10−6 mg/cm2/h. The calculations were based on an instantaneous deposition dose of 9257 mg and a skin area of 2000 cm2. No other studies were found on the absorption, distribution, metabolism, and excretion of fatty acid amidopropyl dimethylamines.
Toxicological Studies
Acute Toxicity
Oral—Nonhuman
Stearamidopropyl dimethylamine
The acute oral toxicity of 10% (wt/wt) stearamidopropyl dimethylamine in propylene glycol was tested in 6 female Wistar rats. 2 The rats received 2 dosages of 1,000 mg/kg body weight of the test material within 24 hours. The rats were observed daily for clinical signs of toxicity for 14 days. Two of the 6 animals died on day 2 and day 3, respectively. Clinical signs observed of both the animals found dead, and the surviving animals included hunched posture, lethargy, uncoordinated movements, piloerection, diarrhea, chromodacryorrhea, pallor, and/or ptosis. Recovery from these symptoms in the surviving animals occurred between days 7 and 10. The 2 animals that died during observation had either slight weight gain or weight loss. Three of the 4 surviving animals had body weight loss between days 1 and 8 but gained body weight between day 8 and the end of the observation period. In 1 dead animal, necropsy showed watery-turbid fluid in the stomach and watery-clear, yellowish fluid in the small intestine. The other dead animal had a spleen of reduced size. In the surviving animals, 1 rat had pelvic dilation of the kidneys. No other abnormalities were observed in the remaining animals. The oral lethal dose 50 (LD50) for stearamidopropyl dimethylamine in this study was determined to be greater than 2,000 mg/kg body weight.
In another oral toxicity study, 40% (wt/wt) stearamidopropyl dimethylamine in deionized water was tested in 20 male and 20 female Sprague-Dawley rats. 2 Dose levels were 420, 1,990, 3,910, and 5,470 mg/kg body weight and were delivered in dose volumes of 1.67, 2.21, 4.44, and 6.22 mL/kg body weight, respectively. Posttreatment, animals were observed for clinical signs and mortality at 1/2, 2, and 4 hours and then daily up to 14 days. No mortalities were observed in the 420 and 1,990 mg/kg dose groups. Two males and 4 females in the 3,910 mg/kg dose group, and all rats in the 5,470 mg/kg dose group died during the observation period and within 8 days of administration of the test material. Clinical signs observed included diarrhea, soft stool, brown-stained abdomen, anal or urogenital region, hypoactivity, hypersensitivity to touch, red-stained nose and mouth, hair loss on abdomen and hindquarters, ataxia, emaciation, bloated abdomen, red stain around eyes, piloerection, lacrimation, high carriage, dyspnea, and hypothermia to touch. At necropsy of the animals that died during the observation period, reddened mucosa (organs not described) was observed in 3 animals from 3,910 mg/kg dose group and 1 animal from 5,470 mg/kg dose group. No other treatment-related changes were reported for any animals in this study. The oral LD50 for stearamidopropyl dimethylamine in this study was determined to be 1,396 mg/kg body weight.
Repeated Dose Toxicity
Oral—Nonhuman
Stearamidopropyl dimethylamine
In an oral 14-day, dose range–finding study performed in accordance to Organization for Economic Cooperation and Development (OECD) guideline 407, stearamidopropyl dimethylamine in propylene glycol (concentration not reported) was administered to 3 Crl: WI(Han) rats/sex/dose via gavage at doses of 0, 50, 200, and 500 mg/kg body weight per day. 2 No mortalities were observed during the treatment period in the low- and mid-dose groups. All animals in the 200 mg/kg/d dose group were observed with piloerection on 2 days during the second week only. No clinical signs of toxicity were observed in the low-dose group. Body weights, body weight gains, and feed consumption were comparable to controls. Hematological changes in the low- and mid-dose groups consisted of slightly lower red blood cell and higher reticulocyte counts in males. No dose-related trend was noted with these changes. Clinical biochemistry changes consisted of higher alanine aminotransferase activity in 2 males in the low-dose group and 2 males and 1 female in the mid-dose group, higher alkaline phosphatase activity in 1 female in the mid-dose group, and higher potassium levels in males in the low- and mid-dose groups. No abnormalities or histopathological changes were noted at necropsy of the low- and mid-dose groups. Slight increases in spleen and thymus weights of the mid-dose group females were comparable to those in control animals.
All animals in the high-dose group were killed for humane reasons between days 6 and 8. From day 4 of treatment and after, these animals were observed with lethargy, hunched posture, labored respiration, abdominal swelling, piloerection, chromodacryorrhea, a lean appearance, and/or ptosis. All animals showed weight loss or reduced body weight gain and reduced food consumption during the treatment period. Necropsy of the high-dose animals found gelatinous contents in the gastrointestinal tract or parts thereof and emaciation. The researchers determined the main cause for moribundity in the high-dose group was fore stomach ulceration and/or hyperplasia of the squamous epithelium of the fore stomach. Other histopathological changes noted at this dose level included lymphoid atrophy of the thymus, correlating with reduced size of the thymus at necropsy; hyperplasia and inflammation of the fore stomach; hyperplasia of the villi in the duodenum and jejunum; foamy macrophages and sinusoidal dilation and congestion/erythrophagocytosis in the mesenteric lymph node; and absence of spermiation and degeneration of spermatids in the testes, oligospermia and seminiferous cell debris in the epididymides, and reduced contents in the prostate and seminal vesicles, which corresponded to a reduced size of seminal vesicles, prostate, and epididymides at necropsy. (Full details on which organs were examined microscopically were not provided in the ECHA summary.) The results of this study were used to determine the doses for a reproduction/developmental toxicity test. 2
Dermal—Nonhuman
Stearamidopropyl dimethylamine
A dermal 90-day repeated dose toxicity study of stearamidopropyl dimethylamine was performed in accordance with OECD Guideline 411 in groups of 5 male and 5 female New Zealand White rabbits. 2 Test solutions were prepared fresh weekly in 30%/70% ethanol/water for each group. The test material was applied at doses of 0%, 0.25%, or 10% wt/vol (equivalent to 0, 5, and 200 mg/kg/d, respectively) in a dose volume of 2 mL/kg/d to intact rabbit skin once daily, 5 d/wk for 13 consecutive weeks. Test sites were not occluded. The animals were collared to prevent oral ingestion of the test material. Clinical signs of toxicity were observed daily, and necropsy and histological examinations were performed at the end of the treatment period.
No mortality was observed during the study. Slight conjunctivitis was observed in 1 control animal and 2 animals in the 0.25% dose group, which was not related to the test material. Animals that received 0.25% test material had moderate or slight erythema, slight edema, slight desquamation, and slight fissuring. Animals that received 10% test material were observed with moderate erythema, slight edema, slight desquamation, and slight fissuring. No treatment-related changes in body weight and body-weight gain were observed during the study. No test-related biologically significant changes were noted in the absolute and relative liver, kidney, and adrenal weight determinations. Statistically significant increases in white blood cell values were noted in the 10% dose group. In addition, there was an increase in platelet values from baseline to necropsy of the 0.25% dose group. The changes in white blood cells of the 10% dose group were attributed to the chronic stress of collaring and not considered to be related to the test material. The significant increase in platelet values of the 0.25% dose group was a result of low baseline values. At necropsy, the treated skin in both the 0.25% and the 10% dose groups had a dry hair coat with an accumulation of test material on the surface. Histopathological examinations revealed minimal acanthosis and hyperkeratosis at the treatment sites of all treated groups. The incidence and severity were similar in both groups. Incidental nontreatment-related histopathological changes were noted in several other tissues such as brain, liver, kidney, prostate, and pancreas. The researchers in this study determined the systemic no observed adverse effect level (NOAEL) of stearamidopropyl dimethylamine was greater than 10% wt/vol in 30%/70% ethanol/water (equivalent to 200 mg/kg body weight per day). 2
Reproductive and Developmental Toxicity
Stearamidopropyl Dimethylamine
The effects of stearamidopropyl dimethylamine (100% active ingredient) on reproduction and development were studied in 10 Wistar rats/sex/dose by oral gavage in accordance with OECD guideline 421. 2 Dose levels tested were 0, 20, 70, and 200 mg/kg body weight per day at a dose volume of 5 mL/kg body weight. Parental males were exposed to the test material 2 weeks prior to mating, during mating, and up to study termination. Parental females were exposed 2 weeks prior to mating, during mating, during gestation, and during at least 4 days of lactation. In the 200 mg/kg males, a weight loss of up to 15% of day 1 weight was observed during the first 2 weeks of treatment, but this effect seemed to recover during the treatment period. The mean body weight and body weight gain of the 200 mg/kg males remained statistically significantly lower throughout treatment. Females of the same dose group had statistically significant reduced body weight gain during the first 2 weeks of treatment as well as during gestation. Food intake was reduced during the entire premating period for males and during the first week of the premating period for the females. Additionally, the feed consumption of the females remained slightly lower throughout pregnancy and lactation. No other treatment-related changes were observed in the parental animals.
The nonstatistically significant decrease in the mean number of corpora lutea was observed in the 70 and 200 mg/kg dose groups when compared to the control animal; however, a statistically significant lower number of implantation sites were noted in the 200 mg/kg dose group females. A statistically significant lower number of live pups was noted in the 70 and 200 mg/kg dose groups. No other treatment-related changes were noted in any of the remaining reproductive parameters investigated in this study (ie, mating, fertility and conception indices and precoital time, testes and epididymides weights, spermatogenic staging profiles). Based on the results of this study on stearamidopropyl dimethylamine, the researchers determined the paternal NOAEL to be 70 mg/kg body weight per day, the maternal NOAEL to be 70 mg/kg body weight per day, and the developmental NOAEL to be 200 mg/kg body weight per day. 2
In the dermal 90-day repeated dose toxicity study in rabbits described earlier, no treatment-related findings concerning the reproductive organs were observed. 2 The dermal developmental toxicity potential of stearamidopropyl dimethylamine was studied in 80 artificially inseminated New Zealand White rabbits. 2 Groups of 20 rabbits received the test material at 0, 5, 100, or 200 mg/kg body weight per day at a dose volume of 2 mL/kg body weight during days 7 through 18 of gestation. The test material was applied to the clipped backs of the rabbits as a solution in 30% isopropanol and 70% reverse osmosis, membrane-processed deionized water. The test sites were not occluded and were rinsed with water 2 hours after each application. The rabbits were collared to prevent oral ingestion of the test material. The rabbits were observed daily during and after the dosage periods for clinical signs of toxicity, skin irritation, mortality, abortion, delivery, body weight, and feed consumption. All rabbits were killed on day 29, and complete gross necropsy was performed. Reproductive organs that were examined included the prostate, seminal vesicles, testis, epididymis in males and the ovaries, uterus, and vagina in females. The uteri were examined for pregnancy, number of implantations, live and dead fetuses, and early and late resorptions. Corpora lutea were counted. Each fetus was weighed and subsequently examined for gross external variations and gender, prior to examination for soft tissue and skeletal variations.
No mortalities were observed during the course of the study. Clinical signs attributed to administration of the test material included alopecia (5, 100, and 200 mg/kg/d doses), excess lacrimation (100 and 200 mg/kg/d dosages), ungroomed coat and green-colored matted fur around mouth and rump (200 mg/kg/d dosage). Statistically significant (P ≤ 0.05 to P ≤ 0.01) increases in the incidences of rabbits with these signs occurred only in the mid- and high-dose groups when compared to the controls. Dose-dependent skin reactions including atonia, desquamation, and fissuring were observed in mid- and high-dose groups. One high-dose group rabbit had eschar present, attributed to the treatment. Two low-dose group rabbits aborted on day 21 of gestation and 1 rabbit in the high-dose group delivered prematurely; however, these events were not test material–related. Body weight gains were significantly decreased in the mid-dose (P ≤ 0.05) and high-dose (P ≤ 0.01) group animals. High-dose group animals had a significant decrease (P ≤ 0.01) in average body weight during treatment and continued to have lower average body weights than control rabbits during the post-dosage period. Body weights and body weight gain of low-dose group rabbits were comparable to control values. When compared to the control values, maternal feed consumption was affected in the mid- and high-dose groups, with the average daily feed consumption of the high-dose group rabbits significantly decreased (P ≤ 0.05 to P ≤ 0.01) from day 15 through day 21 of gestation.
Slightly impaired implantation was observed, along with slightly decreased litter sizes, in the 200 mg/kg dose when compared to the control group, but this effect was not statistically significant (P > 0.05). All of the values were within expected historical control values. The test material did not adversely affect pregnancy incidence or average numbers of corpora lutea or resorptions. Viable fetuses were present in 20, 14, 17, and 14 litters from control, low-, middle-, and high-dosage groups, respectively. One rabbit each from low- and high-dose group had all implantations resorbed. No treatment-related fetal variations at gross external, soft tissue, or skeletal examination were observed.
The researchers concluded that dermal application of stearamidopropyl dimethylamine in rabbits during gestation days 7 through 18 did not produce evidence for developmental toxicity. The maternal no observed effect level (NOEL) was determined to be 5 mg/kg body weight per day, the maternal NOAEL was determined to be100 mg/kg body weight per day based on variations in body weight and feed-consumption data, and the developmental NOAEL was determined to be 200 mg/kg body weight per day in this study. 2
Genotoxicity
Stearamidopropyl Dimethylamine
The mutagenic potential of 85% stearamidopropyl dimethylamine was studied in reverse mutation assay using Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2 uvrA, with and without S9 metabolic activation.
31
The test concentrations ranged from 5 to 5,000 μg/plate. The positive controls were 2-nitrofluorene, 9-aminoacridine, sodium azide, methyl methane sulfonate, and 2-aminoanthracene. The test material was cytotoxic at
The mutagenic potential of 100% pure stearamidopropyl dimethylamine in ethanol was studied for cell mutation in mouse lymphoma L5178Y TK+/− cells in accordance with OECD guideline 467 in 2 independent experiments. 2 Concentrations tested were 0.003 to 60 µg/mL, and the experiments were performed with and without 8% or 12% S9 metabolic activation. No statistically significant positive effects with or without S9 activation were observed in either experiment. Positive controls yielded the expected results. It was concluded that stearamidopropyl dimethylamine was not mutagenic in this assay.
The genotoxic potential of stearamidopropyl dimethylamine in ethanol was studied in a chromosome aberration study using human peripheral blood lymphocytes in accordance with OECD guideline 473. 2 In experiment 1 of this 2-part study, the test material was tested up to 10 μg/mL, without and with S9 metabolic activation. In experiment 2, the test material was tested up to 25 and 10 μg/mL, without and with S9, respectively. Incubation for cells in the first experiment was 3 hours, and in the second experiment, incubation was 3 hours and 24 hours or 48 hours. In both experiments, no statistically or biologically significant increased number of cells with chromosomal aberrations were observed both with and without metabolic activation. Solvent and positive controls yielded expected results. Under the conditions of this study, stearamidopropyl dimethylamine was not considered clastogenic.
Carcinogenicity
No studies were found on the carcinogenicity potential of fatty acid amidopropyl dimethylamines.
Irritation and Sensitization
The North American Contact Dermatitis Group (NACDG) evaluated 25,813 patients for allergic contact dermatitis with patch tests from 1998 to 2007.
32
“Amidoamine” produced relevant allergic reactions in 0.5% of the seniors (20/4, 215; ages
Ocular irritation studies are summarized in Table 4. 2,33 –38 No minimal irritation was observed in ocular irritation assays of behenamidopropyl dimethylamine and dilinoleamidopropyl dimethylamine. All but 1 ocular irritation study of stearamidopropyl dimethylamine report no minimal irritation; the exception found severe ocular irritation when tested at 100% in rabbit eyes.
Nonhuman Ocular Irritation Studies.
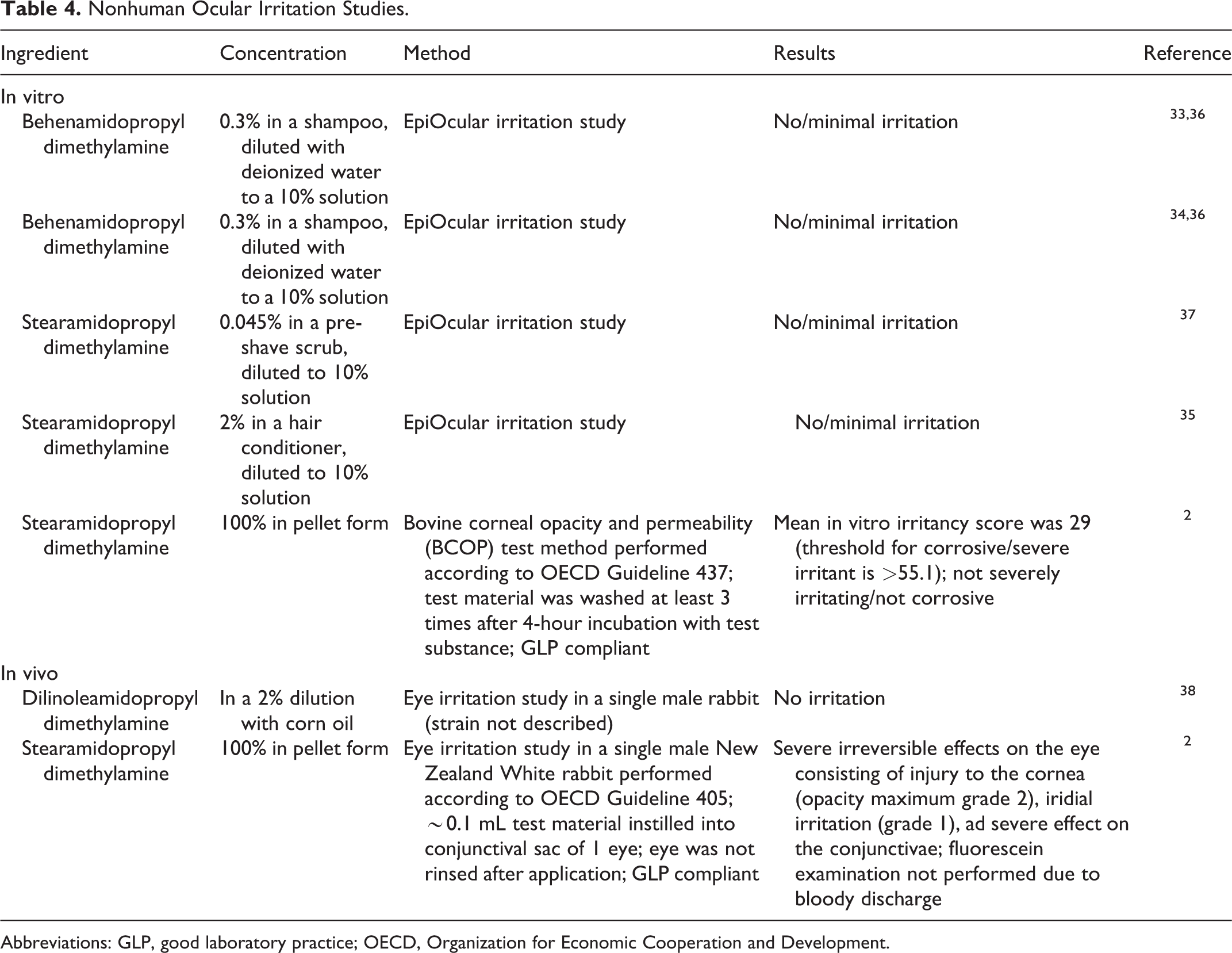
Abbreviations: GLP, good laboratory practice; OECD, Organization for Economic Cooperation and Development.
Dermal irritation studies are summarized in Table 5. 2,39 –42 Stearamidopropyl dimethylamine was considered not irritating in nonhuman studies when tested at 100%. Behenamidopropyl dimethylamine (up to 3%), 0.1% oleamidopropyl dimethylamine, and 0.045% stearamidopropyl dimethylamine in personal care products were not irritating in several in-use studies.
Dermal Irritation Studies.
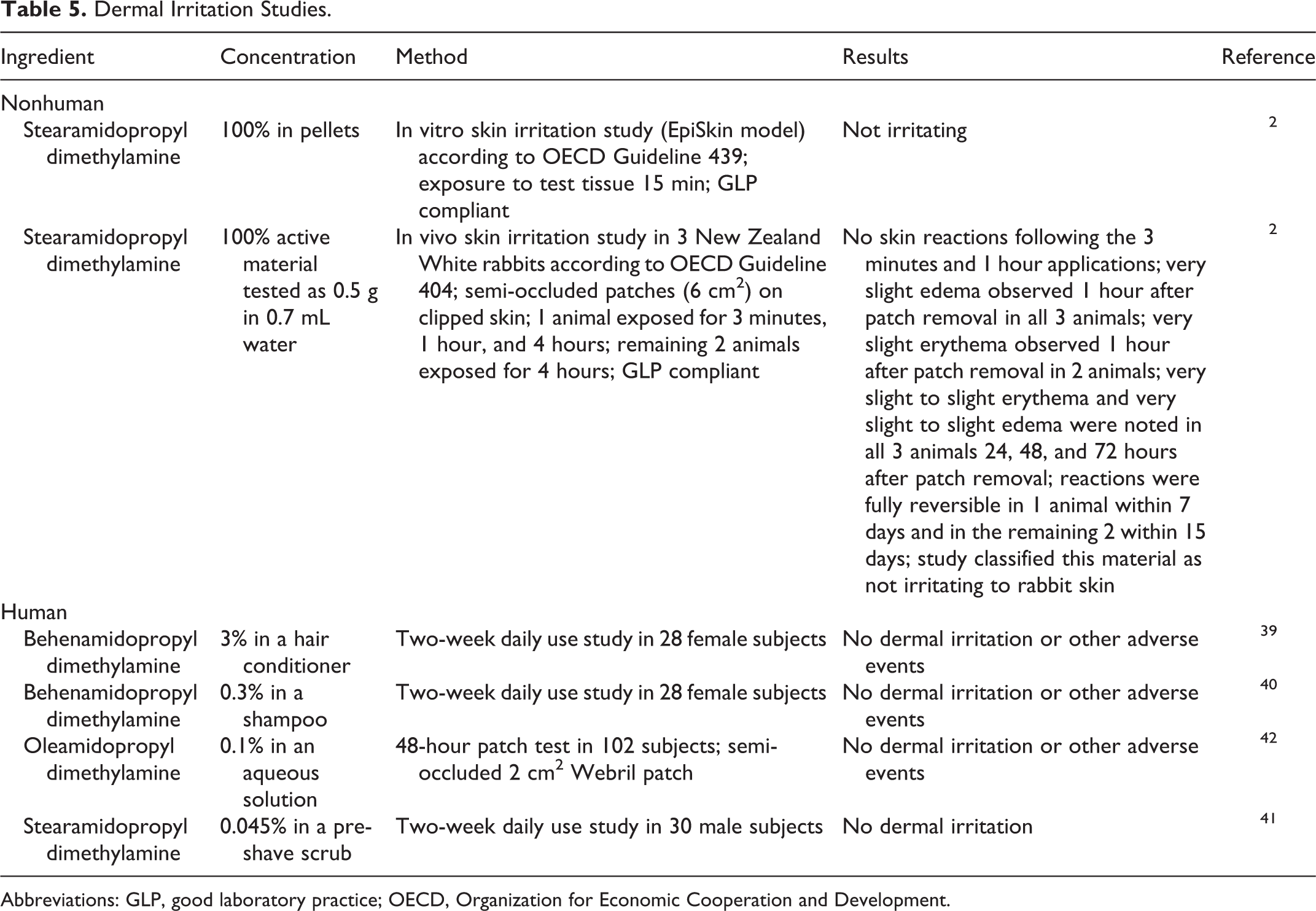
Abbreviations: GLP, good laboratory practice; OECD, Organization for Economic Cooperation and Development.
Dermal sensitization studies are summarized in Table 6. 43 –57 Behenamidopropyl dimethylamine at 0.3% diluted to 1%, 4% brassicamidopropyl dimethylamine, and stearamidopropyl dimethylamine up to 2% in hair conditioners were not contact sensitizers. However, irritation reactions were observed.
The sensitization studies of DMAPA and amidoamine that the Panel reviewed in the safety assessment of cocamidopropyl betaine (CAPB) have been summarized in Table 7. 1 In the tables, amidoamine refers to cocamidopropyl dimethylamine.
Dermal Sensitization Studies.a
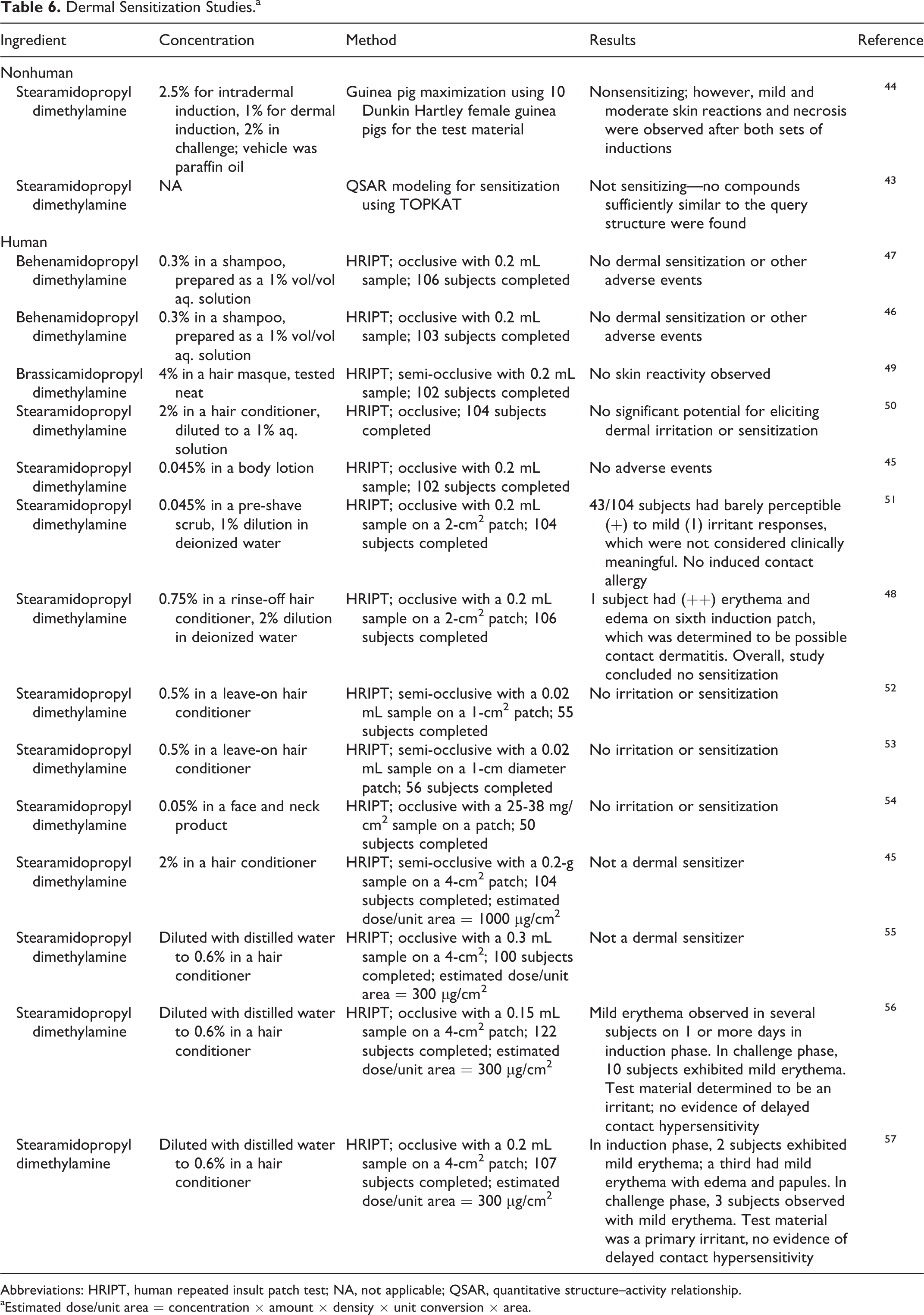
Abbreviations: HRIPT, human repeated insult patch test; NA, not applicable; QSAR, quantitative structure–activity relationship.
aEstimated dose/unit area = concentration × amount × density × unit conversion × area.
Sensitization Studies of DMAPA and Amidoamine Previously Reviewed by the CIR Expert Panel. 1

Abbreviations: CAPB, cocamidopropyl betaine; CIR, Cosmetic Ingredient Review; DMAPA, 3,3-dimethylaminopropylamine; HRIPT, human repeated insult patch test; LLNA, Local lymph node assay; SLS, sodium lauryl sulfate;TLC, thin-layer chromatography.
Clinical Use
Case Reports
The case reports of DMAPA and amidoamine that the Panel reviewed in the safety assessment of cocamidopropyl betaine (CAPB) have been summarized in Table 8. 1 In the tables, amidoamine refers to cocamidopropyl dimethylamine.
Case Reports of DMAPA and Amidoamine Previously Reviewed by the CIR Expert Panel. 1
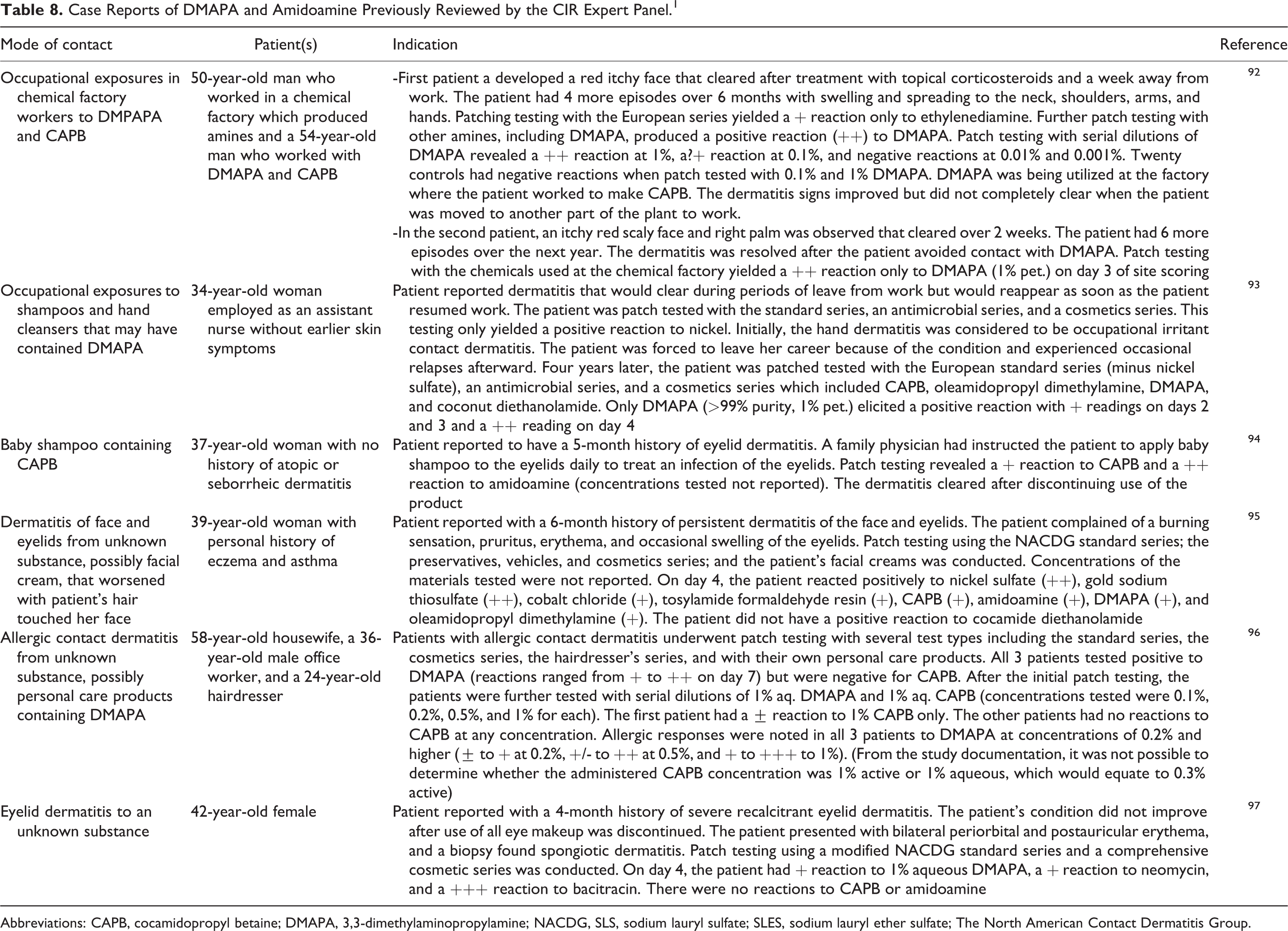
Abbreviations: CAPB, cocamidopropyl betaine; DMAPA, 3,3-dimethylaminopropylamine; NACDG, SLS, sodium lauryl sulfate; SLES, sodium lauryl ether sulfate; The North American Contact Dermatitis Group.
Oleamidopropyl dimethylamine
In the Netherlands, 13 female patients were reported to have allergic contact dermatitis to a baby lotion that contained 0.3% oleamidopropyl dimethylamine. 58,59 Reactions were especially prevalent when the baby lotion was applied to damaged skin and/or the periorbital area. To investigate the possibility of cross-reactions, these patients were patch tested with oleamidopropyl dimethylamine (0.4%), ricinoleamidopropyl dimethylamine lactate (0.5%), stearamidopropyl dimethylamine lactate (0.5%), behenamidopropyl dimethylamine (0.5%), isostearamidopropyl dimethylamine (0.3%), tallowamidopropyl dimethylamine (0.3%), lauramidopropyl dimethylamine (0.2%), myristamidopropyl dimethylamine (0.05%), cocamidopropyl dimethylamine (0.1%), minkamidopropyl dimethylamine (0.1%), and palmitamidopropyl dimethylamine (0.025%). The test solutions were prepared by adding water to the raw material, unless the material was insoluble, then phosphoric acid was added until a clear solution formed. All 13 patients reacted to the oleamidopropyl dimethylamine. One patient had no reactions to any of the other substances, but 12 patients had reactions to at least 4 of the related substances: ricinoleamidopropyl dimethylamine lactate and tallowamidopropyl dimethylamine (11 patients, 85%), lauramidopropyl dimethylamine (9 patients of 12 tested, 75%), and myristamidopropyl dimethylamine (6 patients, 46%). Five patients reacted to isostearamidopropyl dimethylamine, minkamidopropyl dimethylamine, and cocamidopropyl dimethylamine (12 patients tested). The remaining substances elicited a response in only 1 or 2 patients. The author of this study could not rule out that some of these reactions may have been irritant reactions.
In another Dutch report, one medical practitioner reported 3 cases of allergic contact dermatitis in patients who had used a body lotion. 60 In the first case, a 32-year-old female had itchy swelling of the eyelids. Both the upper and the lower lids were edematous, red, and scaly. The symptoms disappeared a few days following use of corticosteroid ointment and avoidance of cosmetics. Patch tests showed the patient was allergic to balsam of Peru and a body lotion that the patient had used around the eyes for several years. When tested with the lotion’s ingredients, the patient had a positive reaction to oleamidopropyl dimethylamine.
In the second case, a 21-year-old was reported to have itchy dermatosis around the eyes and diffuse itching of the body. Upon examination, only mild desquamation was observed on the upper eyelids. The symptoms disappeared within a week of avoiding her cosmetics. Patch tests showed the patient was allergic to nickel cobalt and a body lotion that she had been using. The patient had positive reactions to oleamidopropyl dimethylamine and quaternium-15 when tested with the lotion’s ingredients.
The third case, a 29-year-old female with a history of atopic dermatitis and no active dermatitis, reported dry and itchy skin. Scratch tests were positive for several inhalant allergens. Patch tests showed a positive reaction to a body lotion she had been using. Doubtful reactions were noted for hydroxycitronellal and quaternium-15. Further tests showed a positive reaction to oleamidopropyl dimethylamine. The itching improved after the patient discontinued using the body lotion. 60
Oleamidopropyl Dimethylamine and Cocamidopropyl Dimethylamine
A 10-year retrospective study of patients with allergic eyelid dermatitis investigated the possible allergens. 61 Patch testing was performed in these patients with the NACDG’s standard screening tray and other likely allergen trays. Of 46 patients with confirmed allergic eyelid dermatitis, 5 (10.9%) had relevant reactions to oleamidopropyl dimethylamine, and 2 (4.3%) had relevant reactions to cocamidopropyl dimethylamine.
Quantitative Risk Assessment
3,3-Dimethylaminopropylamine and Cocamidopropyl Dimethylamine (“Amidoamine”) in CAPB
The Council’s Task Force on Sensitization Risk from CAPB Impurities (Task Force) conducted a QRA of DMAPA and cocamidopropyl dimethylamine (“amidoamine”) in support of the CIR safety assessment of CAPB. 1,62 –64 Both of these substances are reactants used in the synthesis of CAPB and are, thus, present as impurities in CAPB used as ingredients in cosmetic formulations. 3,3-dimethylaminopropylamine is present in CAPB ingredients, as supplied (“raw”; typically 30% CAPB) to cosmetics manufacturers at 0.00025% to 0.01%, and “amidoamine” is present at 0.05% to 5.0% (0.5% was “typical” and 1.5% was the suggested maximum concentration). The Task Force derived conservative weight-of-evidence no expected sensitization induction levels (WoE NESILs) of 425 µg/cm2 for DMAPA, based on data from 8 local lymph node assay (LLNAs) performed with a variety of vehicles, and 180 µg/cm2 for “amidoamine,” based on data from an human repeated insult patch test (HRIPT) and an LLNA. The NESILs were then used to calculate margins of safety (MOSs) for the potential for sensitization from dermal exposure to these impurities in cosmetic ingredients, assuming 0.01% DMAPA and 0.5% “amidoamine” in “raw” CAPB and default safety assessment factors. Cocamidopropyl betaine ingredients, as supplied, are used at concentrations up to 11% in cosmetic products. 1 The MOSs calculated for DMAPA were acceptable for all 35 product categories addressed in the QRA and for “amidoamine” were acceptable for 30 of the 35 categories. 62 The Task Force recommended refining the QRA for “amidoamine” in the remaining 5 product categories and that “raw” CAPB users should set the CAPB concentrations in finished products based on QRAs for these impurities.
Stearamidopropyl Dimethylamine
The Council’s CIR Science and Support Committee (SSC) performed a QRA of potential dermal sensitization for stearamidopropyl dimethylamine in accordance with the procedure described above for DMAPA and “amidoamine” in CAPB. 1,65 Table 9 presents the QRA calculations from the CIR SCC. The CIR SSC derived a conservative WoE NESIL of 1000 µg/cm2 based on data from 11 HRIPTs, a guinea pig maximization test, a Buehler guinea pig test, and an LLNA. Greater weight was given to the HRIPT data than the animal data, and the 1000 µg/cm2 NESIL reflects the highest dose tested in the HRIPTs. The MOSs calculated for stearamidopropyl dimethylamine were acceptable for 9 of the 22 product categories addressed, assuming the highest maximum use concentration reported for each category. 62 This includes use at 5% in hair conditioners but not at 1.5% in eye make-up removers, for example. The CIR SSC concluded that further justification is needed for current use levels of this ingredient in the remaining 13 product categories.
Quantitative Risk Assessment of Stearamidopropyl Dimethylamine in Cosmetic Products. 65,a
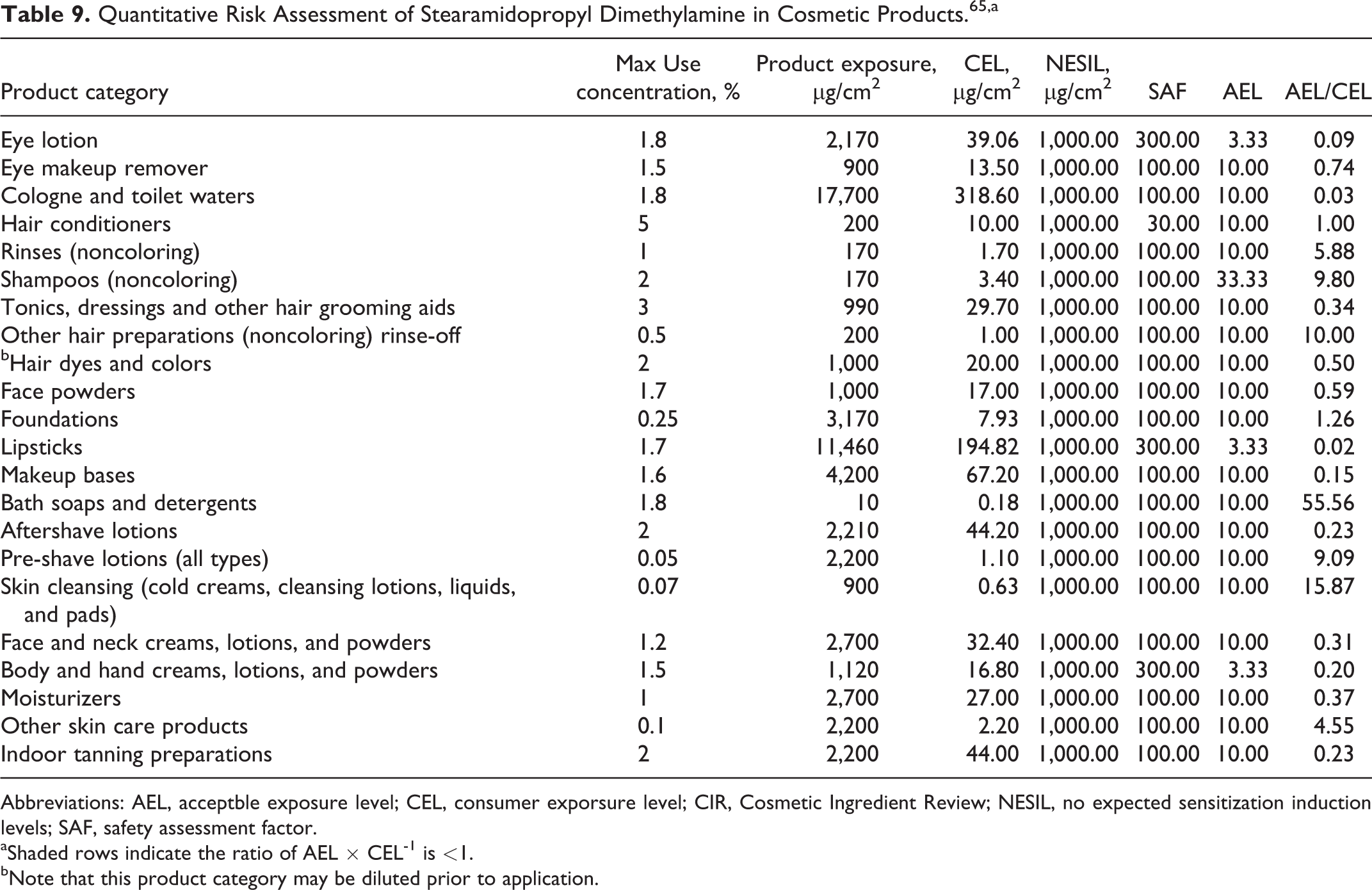
Abbreviations: AEL, acceptble exposure level; CEL, consumer exporsure level; CIR, Cosmetic Ingredient Review; NESIL, no expected sensitization induction levels; SAF, safety assessment factor.
aShaded rows indicate the ratio of AEL × CEL-1 is <1.
bNote that this product category may be diluted prior to application.
Summary
The fatty acid amidopropyl dimethylamines, referred to as “amidoamines,” function primarily as antistatic agents in cosmetic products. The CIR Panel has expressed concern about these chemicals in a safety assessment of fatty acid amidopropyl betaines, in which fatty acid amidopropyl dimethylamines were noted as impurities with sensitizing potential.
Fatty acid amidopropyl dimethylamines have the core structure of a fatty acid amide, N-substituted with 3 propyl-N’, N’-dimethylamine. These ingredients are manufactured by the amidization (ie, amide forming condensation) of fatty acids with DMAPA, most commonly under alkaline or acidic conditions. Fatty acid amidopropyl dimethylamines are composed of secondary amides and tertiary amines and potentially can be nitrosated. Therefore, fatty acid amidopropyl dimethylamine should be formulated to avoid the formation of nitrosamines.
Of the ingredients in this safety assessment, stearamidopropyl dimethylamine has the most reported uses in cosmetic and personal care products, with a total of 427; of those uses, 355 are in rinse-off formulations. Behenamidopropyl dimethylamine has the second greatest number of overall uses reported, with a total of 35; of those uses, 32 are in rinse-off formulations. For both ingredients, most of the rinse-off uses are in hair conditioners. A few uses were reported each for brassicamidopropyl dimethylamine, cocamidopropyl dimethylamine, isostearamidopropyl dimethylamine, lauramidopropyl dimethylamine, minkamidopropyl dimethylamine, oleamidopropyl dimethylamine, and palmitamidopropyl dimethylamine. No uses were reported to the VCRP for the remaining fatty acid amidopropyl dimethylamines.
In a survey of use concentrations conducted by the Personal Care Products Council, stearamidopropyl dimethylamine is reported to be used at a range of maximum concentrations of 0.01% to 5%, with 5% reported in noncoloring hair conditioners. In behenamidopropyl dimethylamine, the range of maximum concentrations was reported to be 0.3% to 3%, with 3% reported in noncoloring hair conditioners. A range of maximum concentrations for cocamidopropyl dimethylamine was reported to be 0.03% to 6.5%, with 6.5% reported in skin-cleansing products.
No use concentrations were reported for almondamidopropyl dimethylamine, avocadoamidopropyl dimethylamine, babassuamidopropyl dimethylamine, minkamidopropyl dimethylamine, oatamidopropyl dimethylamine, oleamidopropyl dimethylamine, sesamidopropyl dimethylamine, and tallamidopropyl dimethylamine. The amidoamine ingredients in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union. Myristamidopropyl dimethylamine has reported uses as a biocide in contact lens disinfecting solution.
In a QSAR model, the dermal absorption of stearamidopropyl dimethylamine has been estimated to be 0.04 mg and 0.12 mg after 8 and 24 hours, respectively. The maximum dermal absorption rate was calculated to be 2.40 × 10−6 mg/cm2/h. The LD50 values in 2 acute oral toxicity studies of stearamidopropyl dimethylamine in rats were > 2,000 mg/kg body weight and 1396 mg/kg body weight, respectively.
Systemic toxicity was observed in an oral 14-day dose range-finding rat study of stearamidopropyl dimethylamine at a dose of 500 mg/kg body weight per day. In rabbits, the systemic NOAEL of stearamidopropyl dimethylamine in a dermal repeated dose study was greater than 10% wt/vol in 30%/70% ethanol water (equivalent to 200 mg/kg body weight per day).
In an oral reproduction and developmental toxicity study of stearamidopropyl dimethylamine tested up to 200 mg/kg body weight per day in rats, the researchers determined the paternal NOAEL to be 70 mg/kg body weight per day, the maternal NOAEL to be 70 mg/kg body weight per day, and the developmental NOAEL to be 200 mg/kg body weight per day. The dermal application of stearamidopropyl dimethylamine tested up to 200 mg/kg body weight per day in rabbits during gestation days 7 through 18 produced no evidence of developmental toxicity. The maternal NOEL was determined to be 5 mg/kg body weight per day, the maternal NOAEL was determined to be100 mg/kg body weight per day based on variations in body weight and food consumption data, and the developmental NOAEL was determined to be 200 mg/kg body weight per day in this study.
No studies were found on the carcinogenicity of fatty acid amidopropyl dimethylamines. Stearamidopropyl dimethylamine was not genotoxic in a reverse mutation assay, a cell mutation assay in mouse lymphoma, or a chromosome aberration study in human peripheral blood lymphocytes.
No to minimal irritation was observed in ocular irritation assays of behenamidopropyl dimethylamine and dilinoleamidopropyl dimethylamine. All but 1 ocular irritation study of stearamidopropyl dimethylamine report no to minimal irritation; the exception found severe ocular irritation when tested at 100% in rabbit eyes.
In an NACDG retrospective analysis, “amidoamine” produced relevant allergic reactions in 0.5% to 0.7% of seniors, adults, and children tested, respectively.
Behenamidopropyl dimethylamine at concentrations up to 3% and 0.045% stearamidopropyl dimethylamine in personal care products were not irritation in several in-use studies. Behenamidopropyl dimethylamine at 0.3% diluted to 1%, 4% brassicamidopropyl dimethylamine, and stearamidopropyl dimethylamine at 2% neat or diluted to 30% were not contact sensitizers. However, irritation reactions were observed.
Possible cross-reactions to several fatty acid amidopropyl dimethylamines were observed in patients who were reported to have allergic contact dermatitis to a baby lotion that contained 0.3% oleamidopropyl dimethylamine.
A 10-year retrospective study found that of 46 patients with confirmed allergic eyelid dermatitis, 10.9% had relevant reactions to oleamidopropyl dimethylamine and 4.3% had relevant reactions to cocamidopropyl dimethylamine. Several cases of allergic contact dermatitis were reported in patients from the Netherlands that had used a particular type of body lotion that contained oleamidopropyl dimethylamine.
Researchers have included the CAPB impurities, DMAPA and amidoamine, in the scope of sensitization and case studies and have found that one or both of the impurities may be the responsible agent for contact allergy to CAPB. Quantitative risk assessments of these ingredients may be performed to ensure acceptable levels of risk in consumers.
Discussion
The CIR Panel expressed concern in the previous Cocamidopropyl Betaine and Related Fatty Acid Amidopropyl Betaines safety assessment about impurities that have sensitizing potential. These impurities of CAPB include the ingredients discussed in this safety assessment. In addition, the Panel recognizes that there are increasing concerns about the contact sensitization potential of oleamidopropyl dimethylamine, which was recently added to the NACDG’s test panel. The Panel noted that, although a safe conclusion was reached for fatty acid amidopropyl dimethylamines, DMAPA in oleamidopropyl dimethylamine appeared to be present at concentrations greater than those reported for the other amidopropyl dimethylamines. Based on the data submitted, DMAPA in oleamidopropyl dimethylamine could exceed the limit recommended by the Panel in the CAPB safety assessment (ie, 0.01% DMAPA in “raw” CAPB used at the highest reported maximum use concentration of 11% in the product category “other shaving preparations”). The Panel requested that industry provide additional information on DMAPA in oleamidopropyl dimethylamine.
The Panel also noted that, for stearamidopropyl dimethylamine, the highest reported maximum use concentration in leave-on products may result in DMAPA concentrations that exceed the limit for this impurity recommended by the Panel for CAPB. Eleven HRIPT studies of normal human participants indicated that no sensitization was induced by stearamidopropyl dimethylamine applied to the skin at concentrations of use; 2 guinea pig sensitization studies were also negative. However, an LLNA yielded an EC3 of 1.4% (350 µg/cm2), indicating that stearamidopropyl dimethylamine is a potential sensitizer. A QRA based on the HRIPTs and rodent studies yielded a conservative, WoE NESIL of 1,000 µg/cm2 for stearamidopropyl dimethylamine and confirmed that this ingredient has the potential to cause sensitization at reported use concentrations in many categories of finished cosmetic products.
Additionally, the Panel acknowledged the QRAs for the CAPB impurities DMAPA and cocamidopropyl dimethylamine (“amidoamine”), which were originally submitted to support the CIR safety assessment of CAPB. WoE NESILs derived for DMAPA and cocamidopropyl dimethylamine were 425 µg/cm2 and 180 µg/cm2, respectively.
The Panel concluded that nonsensitizing concentrations of fatty acid amidopropyl dimethylamine ingredients in finished products can be determined by formulators based on the QRAs for cocamidopropyl dimethylamine (for the ingredients with fatty acid chain lengths < C18), for stearamidopropyl dimethylamine (for the ingredients with fatty acid chain lengths ≥ C18), and for DMAPA (for all of the ingredients), using appropriate NESILs for these substances. The Panel advised industry to continue minimizing the concentrations of the sensitizing impurity DMAPA.
The Panel expressed concern about the possible ability of amidopropyl dimethylamines with fatty-acid chain lengths <C18 to be absorbed through the skin and into the systemic circulation. However, the high NOAELs in toxicity tests of amidopropyl dimethylamines with longer fatty acids alleviated this concern. The Panel felt that the overall toxicological data and low expected exposures supported the safety of the amidopropyl dimethylamines ingredients.
In past ingredient safety assessments, the CIR Panel had expressed concern over N-nitrosation reactions in ingredients containing amine groups. Fatty acid amidopropyl dimethylamines contain secondary amides and tertiary amines that may be N-nitrosated. Additionally, these ingredients may contain secondary amine impurities which may serve as substrates for N-nitrosation. Therefore, the Panel recommended that these ingredients should not be included in cosmetic formulations containing N-nitrosating agents.
The Panel also expressed concern about pesticide residues and heavy metals that may be present in botanical-derived ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities.
The Panel discussed the issue of incidental inhalation exposure from cologne, indoor tanning products, and other propellant and pump spray products. No inhalation toxicology data were identified in the published literature or provided by industry. These ingredients reportedly are used at concentrations up to 0.15% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects upon exposure by incidental inhalation. The Panel considered other data available to characterize the potential of fatty acid amidopropyl dimethylamines to cause systemic toxicity, irritation, sensitization, or other effects. They noted no safety concerns for these substances from the results of acute and repeated dose toxicity studies and genotoxicity studies. Additionally, little or no irritation was observed at use concentrations in multiple tests of dermal and ocular exposure. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Panel concluded that the 24 fatty acid amidopropyl dimethylamines ingredients listed below are safe in cosmetics when they are formulated to be nonsensitizing, which may be based on a QRA. almondamidopropyl dimethylamine* avocadamidopropyl dimethylamine* babassuamidopropyl dimethylamine* behenamidopropyl dimethylamine brassicamidopropyl dimethylamine cocamidopropyl dimethylamine dilinoleamidopropyl dimethylamine* isostearamidopropyl dimethylamine lauramidopropyl dimethylamine linoleamidopropyl dimethylamine* minkamidopropyl dimethylamine myristamidopropyl dimethylamine* oatamidopropyl dimethylamine* oleamidopropyl dimethylamine oleamidopropyl dimethylamine* palmitamidopropyl dimethylamine ricinoleamidopropyl dimethylamine* sesamidopropyl dimethylamine* soyamidopropyl dimethylamine* stearamidopropyl dimethylamine sunflowerseedamidopropyl dimethylamine* tallamidopropyl dimethylamine* tallowamidopropyl dimethylamine* wheat germamidopropyl dimethylamine*
*Not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic, Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Burnett, C. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Boyer, I. contributed to analysis and interpretation. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Gill, L. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.