Abstract
Anatomically, the kidneys are paired, bean-shaped (in most mammals), excretory organs that lie in the retroperitoneum. High blood flow to the kidneys, together with high oxygen consumption, makes them more vulnerable to exposure, via the circulation, and subsequent injury related to high concentrations of xenobiotics and chemicals. In preclinical drug development and safety assessment of new investigational drugs, changes in kidney structure and/or function following drug administration in experimental laboratory animals need to be put in context with interspecies differences in kidney functional anatomy, physiology, spontaneous pathologies, and toxicopathological responses to injury. In addition, translation to human relevance to avoid premature drug termination from development is vital. Thus, detection and characterization of kidney toxicity in preclinical species and human relevance will depend on the preclinical safety testing strategy and collective weight-of-evidence approach including new investigational drug mechanism of action (MOA), preclinical and clinical interspecies differences, and MOA relevance to humans. This review describes kidney macroscopic and microscopic functional anatomy, physiology, pathophysiology, toxicology, and drug-induced kidney toxicities in safety risk assessment and drug development.
Introduction
Understanding the science of kidney injury with respect to functional anatomy, pathophysiology, mechanisms and biomarkers of nephrotoxicity, and alternative models that allow human translation of preclinical data is very critical for pharmaceutical drug development, chemical hazard risk assessment, and consumer health industries. In this review, the basic anatomy of the normal kidney tubular, interstitial, vascular, and glomerular structures and their function are discussed. In addition, the basic general pathological terms applicable to kidney commonly used in toxicologic pathology are presented. Examples of toxicologically relevant interspecies physiologic and developmental differences are discussed. In addition, some examples of common drug-induced tubular, interstitial, vascular, and glomerular pathologies and toxicities encountered in drug development along with preclinical to clinical translation and human relevance of some of these toxicities are presented. This review is based on a symposium entitled “The Brilliant Kidney in Drug Development: From Basics to Chips” at the Annual Meeting of the American College of Toxicology (West Palm Beach, Florida, 2018). The objective of this symposium was to provide current state, innovation, and anticipated regulatory changes as it relates to kidney efficacy and safety risk assessment. The symposium covered basic, yet relevant kidney general physiology and drug-induced pathologies and case examples to illustrate recent advances in biomarkers and emerging technologies bridging the preclinical to clinical gap. Specifically, 4 talks highlighted: (1) basic kidney histologic anatomy, physiology, and toxicologic pathology; (2) mechanisms of drug-induced kidney toxicity and their interpretations; (3) translational biomarkers for early detection of kidney injury; and (4) application of kidney-on-a-chip microphysiological system for kidney safety and regulatory assessment.
Kidney Macroscopic and Microscopic Anatomy
Kidney Macroscopic Anatomy
The renal system is comprised of 2 kidneys, 2 ureters, a urinary bladder, and a urethra. The kidneys are paired, bean-shaped (in most mammals), excretory organs that lie in the retroperitoneum and are situated in the posterior part of the abdomen on each side of the vertebral column. The right kidney is lower than the left kidney. Kidneys weight varies with body surface area, age, and sex, and both kidneys weigh approximately 0.51% to 1.08% (a mean of 0.65%) of the body weight. 1,2 Despite this low weight relative to body weight, the kidneys receive 25% of the cardiac output, with 85% of renal blood flow in the cortex, 14% in the outer medulla, and only 1% in the inner medulla. 1,2 Such significant kidney microanatomic differences in perfusion and blood flow and oxygen consumption makes the kidneys more vulnerable to exposure, via the circulation, and subsequent injury related to high concentrations of xenobiotics and chemicals. 2 The main renal artery, which originates from the abdominal aorta, branches between kidney parenchymal lobes to form the interlobar arteries and these arteries extend to the corticomedullary junction to form the arcuate arteries (ie, these arteries arch between the cortex and the medulla), which run parallel to the capsule. Interlobular arteries give rise to afferent arterioles that give rise to glomerular capillaries (ie, capillary loops). When these capillaries exit the glomerulus, they exit as efferent arterioles that further branch to form peritubular (ie, interstitial) capillaries. Efferent arterioles extend into the medulla as vasa recta and supply the outer and inner medullae. The vessels of the venous system run parallel to the arterial vessels and form the interlobular vein, arcuate vein, interlobar vein, and renal vein. The vasa recta and peritubular capillaries drain into interlobular veins 3 ; the veins leave the kidneys as renal veins, which empty into the inferior vena cava. Lymphatics run parallel only to the cortical vasculature. 3
Each kidney is surrounded by a fibrous capsule, which is smooth and can be easily removed at autopsy in healthy kidneys. Surrounding the renal capsule is a thick layer of perinephric fat to provide protection from trauma. Macroscopically on cut surface at autopsy, the kidney parenchyma has 4 major anatomical regions: (1) the superficial cortex (a pale outer region) that contains renal corpuscles, (2) the deep medulla (a darker inner region) with conical medullary pyramids, (3) renal papilla, and (4) renal pelvis. The medulla is further subdivided into the outer medulla and the inner medulla. The outer medulla is divided into the outer stripe of the outer medulla and the inner stripe of the outer medulla (Figure 1). The outer stripe contains parts of the proximal tubule (thick descending limbs of Henle’s loop), straight parts of the distal tubule (thick ascending limbs), and collecting ducts. The inner stripe comprises descending thin limbs, ascending thick limbs, and collecting ducts. The inner medulla is established by thin limbs (descending and ascending) and collecting ducts. 4 In the lower urinary tract, ureters transport urine from the kidneys to the urinary bladder. The urinary bladder serves as a temporary storage reservoir for urine before the urethra transports urine out of the body. The urethra of females is short and is longer in males. The structure and function of the lower urinary tract is generally similar in all mammals. 1,2
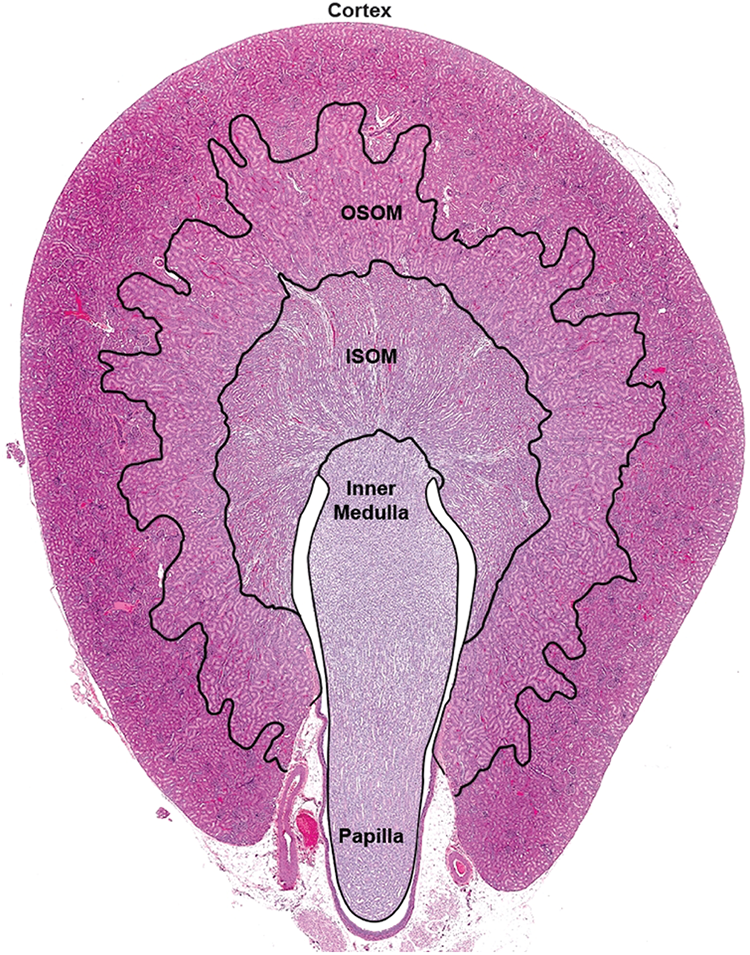
Microscopic anatomical regions of the rat kidney. Note inner stripe of the outer medulla (ISOM), outer stripe of the outer medulla (OSOM), very well-developed OSOM, and unipapillary, single renal pyramid. Hematoxylin and eosin stain. Source: Reprinted from Khan, Hard, and Radi (2012), ©2012.
Kidney Microscopic Anatomy
The urine-producing functional and structural units of the kidney that are present in the cortex and medulla are called nephrons. The nephron consists of glomeruli, renal tubules (proximal tubules, loop of Henle, distal tubules, connecting segment, collecting ducts), interstitium, and juxtaglomerular apparatus (JGA). The initial plasma filtering portion of the nephron is the glomerulus, which is contained within Bowman capsule. Microscopically, the glomerulus is composed of capillary network lined by a thin layer of fenestrated endothelial cells, glomerular basement membrane (GBM), a central region composed of mesangial cells and matrix, visceral (inner) epithelial cells (podocytes), and parietal (outer) epithelial cells. Afferent arterioles supply blood to the glomerulus, while efferent arterioles exit the glomerulus. The glomerular filtration barrier (size, shape, and charge selective) has 3 layers: the fenestrated endothelium, a negatively charged GBM, and the podocytes. During the filtration process through glomerular capillaries, it is important not to overload such process as this might lead to glomerular lesions and proteinuria such as the case with glomerular calcification that might be seen after administration of dibasic sodium phosphate solution in rats. 5 The glomerulus opens into the proximal convoluted tubules, and in mature male mice, the proximal convoluted tubulars can extend into Bowman capsule. 2 The JGA is located at the vascular pole of the glomerulus. The components of the JGA include the macula densa of the thick ascending limb, afferent and efferent arterioles, granular cells of the afferent arteriole that produce renin and angiotensin II (AII), and the extraglomerular mesangial cells. The JGA is important in tubuloglomerular feedback control of renin secretion produced by the granular cells. 2
Cortical proximal convoluted tubular epithelial cells are cuboidal cells with well-developed, dense surface microvilli (brush border). 3 The first segment of the proximal tubule is convoluted and is called pars convolute, and the subsequent part that passes down toward the medulla to become the descending limb of the loop of Henle is straight and is called pars recta. Thus, the segments (S) of proximal tubules can be subdivided into the S1 and S2 (convoluted) and the S3 (pars recta or straight) in experimental laboratory animals (rats, mice, and monkeys). 2 These segments have different energy and oxygen requirements and can be distinguished using various markers via immunohistochemical techniques. The first part of the proximal convoluted tubules, S1 segment, is short, connects with the glomerular filtration space, contains numerous mitochondria with highest oxidative metabolic rate, and its brush borders and phagosomal system are well developed as this segment is involved in reabsorption and degradation of macromolecules. The S2 segment represents most of the proximal tubules, has a shorter brush border with fewer mitochondria but has more lysosomal bodies, and is the primary site for low-molecular-weight protein resorption. The S3 segment has a tall brush border and peroxisomes and is a primary site for cytochrome P450 metabolizing enzymes. This is important as some drugs might cause tubular hypertrophy in the S3 segment that is related to peroxisome proliferation. Mixed-function oxidase activity occurs primarily in pars recta in the rat. The cortical distal convoluted tubules have very few microvilli and connect the nephron with the collecting duct. In rodents, the distal convoluted tubules contain Tamm-Horsfall protein, a mucoprotein. 2
The spaces among various kidney microscopic anatomic structures (the intertubular, extraglomerular, extravascular spaces) are defined as the interstitium. These spaces in the cortex and medulla (ie, cortical and medullary interstitium) contain different cellular constituents, extracellular matrix, and interstitial fluid. The interstitial space accounts for approximately 7% to 8% of the total parenchymal volume in the cortex and is thicker and accounts for approximately 29% to 40% in the inner medulla. The cells in the interstitium include dendritic cells (DCs), macrophages, lymphocytes, lymphatic endothelial cells, and fibroblasts. The cells in the interstitium of the papilla include stellate cells, monocytes, and pericytes. The proportion of interstitium in the inner medulla increases with age and ischemia. The papillary interstitium is rich in mucopolysaccharides matrix. 2,6
The inner stripe of the outer zone of the medulla consists of Henle loops, thick ascending tubules (thick ascending segment or straight portion of the distal tubule), and collecting ducts. 2 The inner zone of the medulla consists of Henle loops and collecting ducts. The thick ascending limb of the loop of Henle (TAL) is one of the 3 structural segments of the distal tubules. The TAL is located in the outer zone of the medulla and in the cortex. Cells lining the TAL are cuboidal with eosinophilic cytoplasm and round central nucleus. Ultrastructurally, cells of the TAL have prominent mitochondria, rough endoplasmic reticulum, and Tamm-Horsfall protein, which cover the luminal surface of the cells lining the TAL. The collecting duct system (cortical collecting ducts, the outer medullary collecting ducts, and the inner medullary collecting ducts) extends from the connecting segment in the cortex through the outer and the inner medullae to the tip of the papilla. The 2 cell types in this system are the intercalated cell (a dark cell) and the principal cell with amphophilic or clear cytoplasm. In the inner medulla, the intercalated cell is absent. In the papilla, collecting ducts form the ducts of Bellini, which empty into the pelvis. 2
Kidney Interspecies Macroscopic and Microscopic Anatomical Differences
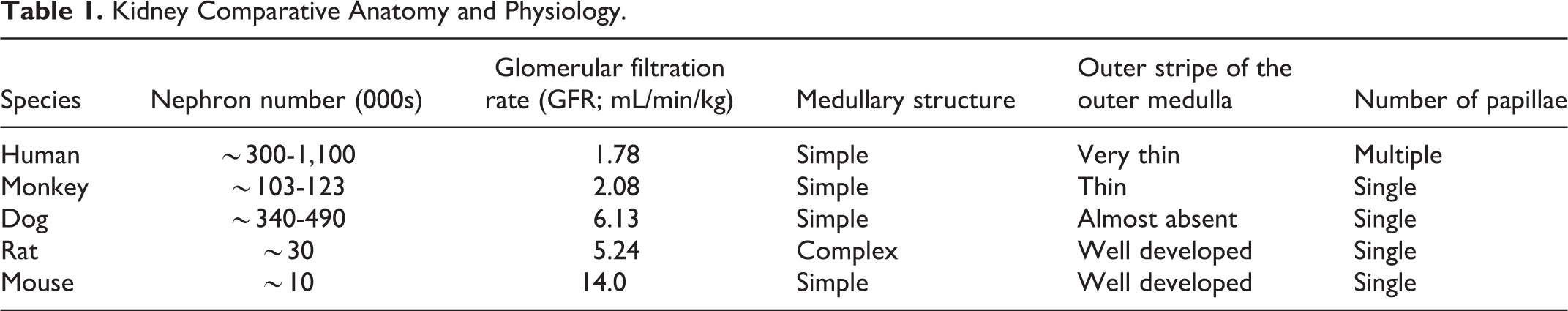
The interspecies and sex differences in kidney macroscopic and microscopic anatomy among experimental laboratory animals used in preclinical safety testing versus humans need to be considered when evaluating and interpreting test article–related kidney changes and pathologies. For example, the kidney medulla in humans is multipapillary (multilobular) because it is divided into 8 to 18 conical masses (pyramids; lobes). Similarly, minipigs have multilobular kidney. Rat, mice, dogs, and cynomolgus monkeys have a unipapillary, single renal pyramid (Figure 1; Table 1). 2,7 In addition, simple (ie, has vascular bundles consisting only of descending and ascending vasa recta) renal medulla anatomical structure is present in most mammals, including humans. However, a complex (ie, descending thin limbs of short loops that are integrated into vascular bundles) renal medulla is present in mammals (eg, rat and mouse) with a high urine concentrating ability with thickest renal medulla 2,4 (Table 1). Therefore, while humans have an osmolality approximately up to 300 mOsm/kg, rats and mice osmolality can be up to 4,300 mOsm/kg and this would, for example, affect drug-related kidney crystalluria formation, a finding in rodents that is not relevant to humans. Mice have long loops of Henle and associated vasa recta in the medulla. In rats, the outer stripe is very well developed and makes up about one-third of the outer medulla (Figure 1) 7 , very thin in humans and nonhuman primates, and is almost completely absent in dogs. 2,4,7 In dogs, all nephrons are of the long-looped variety, whereas in rats and mouse, long-looped nephrons account for 28% of the total nephrons. 2 Normal sexual dimorphism can be noted in both male and female mice (eg, larger kidney corpuscles noted in males than females). The epithelium lining Bowman capsule in the kidney of male mice is cuboidal, while in females the epithelium is simple squamous. This should not be confused with drug-induced epithelial hyperplasia. Also, unlike other species, mice excrete large amounts of protein in the urine under normal physiologic conditions; their urine contains taurine and lacks tryptophan; and they also excrete creatinine in urine. Adult rabbits normally have cloudy urine because most of their dietary calcium is absorbed and they excrete it via the kidneys.
Kidney Comparative Anatomy and Physiology.
Kidney Physiology
Kidney Glomerular Physiology
The primary functions of kidneys are (1) maintenance and regulation of body’s fluid and electrolytes, (2) maintenance of extracellular fluid volume, (3) endocrine such as elaboration of hormones, (4) regulation of blood pH and pressure, (5) excretion of the waste products of metabolism, and (6) metabolic activities. 7 Adequate blood flow and perfusion affects normal kidney function and about 10% of oxygen consumption occurs in the kidneys. Measurement of kidney function is determined by glomerular filtration rate (GFR) that varies across species (Table 1) and pathologies in afferent and efferent arterioles (eg, drug-induced vasoconstriction) can affect GFR. The plasma is filtered via the glomerulus to produce glomerular ultrafiltrate. Ultrafiltration movement across the glomerulus blood capillary network barrier into the urinary Bowman capsule space involves crossing 3 layers: fenestrated endothelium of glomerular capillaries, GBM, and slit pores between the foot processes of visceral epithelial cells of the Bowman capsule. The fenestrated (ie, have pores) endothelium allows the passage of fluid, blood plasma solutes, and low-molecular-weight proteins and prevents passage of blood cells and platelets. Factors that affect GBM glomerular filtration include molecular size, shape, and charge. Only small-molecular-weight molecules can pass through the glomerular filter and small molecules (eg, amino acids, glucose, sodium, potassium, water) pass through the glomerular ultrafiltrate. Podocytes provide structural support to the capillary loop with their actin cytoskeleton, can express Toll-like receptor-4 (TLR4), and maintain GBM. There are structural proteins (eg, nephrin, podocin, and P cadherin) involved in maintaining the structure of the foot processes, and mutations in these proteins can lead to proteinuria. Mesangial cells are phagocytic and may prevent basement membrane accumulation of macromolecules that escaped from the capillaries. The plasma ultrafiltrate passes down the nephron tubule to form urine. 2
Kidney Tubular Physiology
Physiologically, the proximal tubules account for 70% of all reabsorptive activity. The glomerular ultrafiltrate undergoes reabsorption process of sodium (65%), water (65%), potassium (65%), calcium (90%), phosphate, bicarbonate, magnesium, amino acids (99%), glucose (100%), vitamins, sulfate, and low-molecular-weight proteins in the proximal tubules. These are returned to the circulation by the peritubular and vasa recta capillaries. Sodium reabsorption by the proximal tubule is an active process that is driven by Na+/K+ ATPase at the basolateral cell surface membranes. Reabsorption of NaCl accounts for most of the energy expenditure in the kidney. Obligatory (ie, water is obligated to follow Na+) water reabsorption is a passive process. Approximately two-thirds of calcium reabsorption is passive in exchange for sodium. Approximately two-thirds of phosphate is reabsorbed in the proximal tubules. In the dog, 85% of bicarbonate reabsorption takes place in the proximal tubules. Reabsorption and degradation of macromolecules in the ultrafiltrate takes place in the pars convoluta of the proximal tubule due to its well-developed endocytic-lysosomal apparatus. 2
Maintaining nearly constant blood plasma osmolality and sodium concentration is essential for survival. The urine concentrating mechanism plays an essential role in regulating water and sodium excretion. The descending and ascending portions of the loop of Henle enable recovery of much of the Na+ and water that were filtered by the glomerulus. The urine concentrating and urine forming process in the kidney starts in the descending loop of Henle and depends on the generation of an interstitial osmotic gradient from the corticomedullary border down to the papilla tip. This osmotic gradient is generated by a countercurrent (the descending and ascending loops fluid movement in opposite directions) multiplication (solute pumping leading to multiplication of solutes concentration in the deep medulla) system within the descending and ascending loops of Henle, and passive diffusion in the vasa recta, the countercurrent exchanger. Cells in the descending thick loop (DTL) have aquaporin (AQP) channel proteins (upper 40% of each DTL expresses AQP1) to allow for the high permeability and movement of water from the descending loop lumen into the surrounding interstitium, leading to osmolality increases from approximately 300 mOsmol/kg (isosmotic with blood) in the cortex to about 1,200 mOsmol/kg (hypertonic solution) in the papillary tip region, resulting in hypertonic interstitium. 3,8
There are important species differences in such urine osmolalities with toxicological implications and human risk assessment if there are solubility issues with a test article. For example, rats can concentrate to approximately 3,000 mOsm/kg H2O and mice to approximately 4,300 mOsm/kg H2O. These changes in urine osmolalities are accomplished by osmosis in the descending limb and active epithelial cell transport in the ascending limb and lead to the reabsorption of up to 20% of the water and 10% of Na+ entering this segment of the nephron. Solutes and water recovered from these loops are returned to the circulation via the vasa recta. The ascending loop of Henle lacks AQP proteins and is impermeable to water but is permeable to sodium and other solutes which are actively pumped out of the loop via the Na+/K+ ATPase pump. This leads to the hyperosmotic environment in the kidney medulla. The interstitium is rich in osmotically active urea because of reabsorption by the collecting ducts. 8
The distal convoluted tubule plays a critical role in sodium, potassium, and cation homeostasis. It reabsorbs approximately 5% to 10% of the filtered sodium, 7% to 10% of filtered calcium, 10% of filtered magnesium, and 10% to 15% of water. Water reabsorption in the distal tubules is under the influence of antidiuretic hormone (ADH; vasopressin). In addition, this distal part of the nephron is aldosterone sensitive, a hormone that responds to volume depletion or hyperkalemia. 9 Aldosterone increases the amount of Na+/K+ ATPase in the basal membrane of distal tubules and Na/Cl reabsorption. Distal tubules have receptors for parathyroid hormone to facilitate Ca++ recovery. Hydrogen ions, creatinine, and some drugs such as penicillin are actively secreted and move from the blood into the distal tubules.
The collecting ducts regulate urine volume and osmolarity and contain 2 distinct cell types: principal cells and intercalated cells. Principal cells help maintain body water and salt balance. Plasma osmolarity is regulated by the posterior pituitary hormone ADH. Aldosterone is secreted by the adrenal cortex in response to AII, a vasoconstrictor, stimulation. Principal cells have receptors for ADH and aldosterone to regulate water and Na+ recovery. In fact, the principal cell plays a central role in salt and water transport with transporters such as the epithelial Na+ channel (ENaC), the renal outer medullary K+ channel, and the AQP2 water channel. 10 Control of plasma Na+ and K+ concentrations, extracellular fluid volume, and blood pressure is achieved via the coordinated regulation of ENaC by aldosterone and AQP2 by arginine vasopressin in principal cells. 10 Intercalated cells secrete or absorb acid or bicarbonate and play an important role in regulating blood pH. Intercalated cells reabsorb K+ and HCO3 − and secrete H+. This function lowers the acidity of the plasma while increasing the acidity of the urine.
Kidney Endocrine Functions
The kidneys can be hormonally sensitive as they have high levels of sex hormone receptor expression. Kidney endocrine function involves secreting 3 key hormones: erythropoietin, calcitriol (1,25-dihydroxycholecalciferol), and renin. In addition, kidneys synthesize prostaglandins, growth factors, and arachidonate metabolites. 2,6,11 Erythropoietin is produced by renal cortical peritubular interstitial cells in response to hypoxic conditions. This stimulates erythropoiesis and the production of red blood cells in bone marrow. Hypoxia-inducible factor (HIF) is a key regulator of adaptive responses in renal hypoxic conditions to regulate renal oxygen homeostasis, especially in ischemia–reperfusion injury of acute kidney injury (AKI). 12 Renin is produced within juxtaglomerular (JG) cells after processing and cleavage of circulating prorenin, which is produced in the liver. Reduced renal perfusion pressure and hyponatremia are the primary stimuli for renin release from JG cells that act as sensors of blood pressure. Renin is released by the sympathetic nervous system and AII, ADH, endothelin, and prostaglandins. Renin activates AII, which causes secretion of aldosterone by the adrenal cortical zona glomerulosa. The net effect is systemic vasoconstriction, intrarenal vasoconstriction, and increased aldosterone release. Thus, due to the reduced negative feedback on renin production by JG cells, angiotensin-converting enzyme (ACE) inhibitors can cause hypertrophy and hyperplasia of JG cells. In addition, the kidney is a site of degradation of hormones, such as insulin and aldosterone, and is also responsible for the production of the active form of vitamin D derived from vitamin D3 under the influence of parathyroid hormone. 2 Kidney interstitial cells such as stellate cells, in conjunction with pericytes, act as sensor and have endocrine effector functions for the control of blood pressure, salt balance, and oxygen delivery. 13
Kidney Metabolic Physiology
The kidney is the primary route of xenobiotic excretion and contains xenobiotic biotransformation enzymes. This is because: (1) xenobiotics present in the glomerular ultrafiltrate are concentrated and present at high concentrations, (2) the cortex receives about 80% of total renal blood flow and thus is exposed to the high concentration of xenobiotics coming from the bloodstream, and (3) such high concentrations of xenobiotics may accumulate in proximal tubulars following reabsorption or secretion processes. Xenobiotic-metabolizing enzymes activities such as mixed-function oxidases are present in the proximal tubules and the activity varies along various segments of the nephron. 14 Mixed-function oxidase activity occurs primarily in pars recta region of the proximal tubules in the rat. The kidney plays an important role in the hydroxylation and glucuronidation of fatty acids, arachidonic acid (AA), and 25-hydroxyvitamin D3. 15 Epoxidation of AA is catalyzed by the cytochrome P4502C (CYP2C) and CYP4F subfamilies and these enzymes play an important role in the renal regulation of salt and water balance and in inflammation. The highest activities of cytochromes P450 are present in the proximal tubules and involved in conjugation of glutathione, glucuronic acid, and sulfate. 16 The interspecies and sex differences in kidney physiology among experimental laboratory animals in preclinical safety testing versus humans need to be considered when evaluating and interpreting test article–related kidney changes and pathologies. S1, S2, and S3 tubular toxicological manifestation vary due to differences in kidney metabolic activities, and there is little to no induction of cytochrome P450s following administration of phenobarbital due to the absence of the CYP2B subfamily. In human kidney, CYP2D6 and CYP2E1 are absent. The activity of aldehyde oxidase in humans is higher than that in rats and dogs. Mitochondria in the proximal tubules are the site of production of circulating 1α,25-dihydroxyvitamin D3. 15 Because the blood flow in the renal medulla is much lower than the cortex, the levels of xenobiotic-metabolizing enzymes are lower, and this region have more anaerobic metabolism. Some isoenzymes of P450 and glutathione S-transferases are localized in the thick ascending limb and distal tubules, while prostaglandin H-synthase is localized in the collecting ducts in the medulla. 17 Renal enzyme activities can be different between sexes and cytochrome P450 activity in rodents is often polymorphic by sex and sex hormones (eg, drug-induced acetaminophen toxicity) might influence proximal tubules metabolizing enzyme activity. For example, male mice have more P450 activity than female mice. Induction of kidney metabolizing enzymes activities can lead to increases in kidney absolute weights. 2
In addition, kidney transporters are important consideration because interference with transporters by a drug candidate (eg, inhibition of tubular secretion of creatinine without affecting glomerular function or causing any microscopic changes in the kidney; drug is a substrate for a transporter; transporter affecting drug disposition) might allow for mechanistic understanding of kidney safety signal risk assessment. For example, sex differences in transporter expression were observed with highest in the mouse followed by rat and dog. Organic anion transporter 2 was higher in male mouse than the female. Organic cation transporter 2 was higher in male rat and mouse. Multidrug resistance protein 4 is predominantly expressed in male mouse kidney. 18
Kidney Pathophysiology and Toxicology
Kidney Pathophysiologic Responses
The kidney is a common target organ of toxicity and injury as a result of its exposure to drugs, xenobiotics, or chemicals. This is because: (1) the high blood flow rate as they receive more blood flow per gram of tissue (approximately 25% of cardiac output) than any other organ, (2) high doses of test articles typically administered in preclinical toxicity and safety studies, (3) kidney’s key role in excretion through urine, (4) high renal metabolic biotransformation and high oxygen consumption of renal epithelium, (5) high transporter activity of the renal epithelium, and (6) kidney’s role in reabsorption and its ability to concentrate fluids. 2,7 The specific pattern of kidney toxicity and injury is dependent on the physiochemical properties of the drugs (eg, low intrinsic solubility) and their dose, xenobiotics, or chemicals, toxicokinetic properties, renal clearance profile and metabolic attributes, and local kidney tissue concentration and length of time of exposure. In addition, the kidney parenchyma is especially vulnerable to the pro-inflammatory response to infection and subsequent kidney tissue damage. This is because damage-associated molecular patterns (DAMPs), pathogen-associated molecular patterns (PAMPs), and pro-inflammatory mediators gain access to kidney parenchyma and its various compartments via glomerular filtration or peritubular capillaries microcirculation. 12, 19,20 For example, lipopolysaccharides (LPS) are filtered into the tubular fluid and can directly interact with renal tubular epithelial cells (RTECs) via a TLR4-dependent mechanism. 19,21 As a consequence, various compartments of the kidney, including RTECs, glomerular, and endothelial, are injured. This is because the filtered PAMPs and DAMPs, also called “danger signals,” bind to and activate RTECs, renal endothelial, and glomerular cells and can lead to endothelial cell activation and structural changes in the glomerular and peritubular capillaries. 20 Therefore, exogenous and endogenous PAMPs and DAMPs are integral to AKI pathogenesis. 12
Clinical and Anatomic Pathology in Kidney Safety Assessment
Assessment for test article–related kidney pathology and toxicity can include standard clinical pathology parameters such as clinical chemistries for blood urea nitrogen (BUN) and serum creatinine (sCr), along with disturbances in electrolytes and water balance, urine excretion of protein (eg, proteinuria, urine protein to creatinine ratio, and microalbuminuria in nonrodents with glomerular pathologies) and electrolytes, urinalysis (urine volume [eg, oliguria in AKI], appearance, specific gravity, pH, quantitation for microalbumin, and absolute and relative kidney weight determination), and macroscopic and microscopic examination of the kidney tissue. 7,12 Microscopic examination of urine sediment for presence of RBC, glucose, ketones, bilirubin, and crystals can be used to assess for drug-induced changes such as crystalluria and it is important to consider species differences in kidney drug handling. For example, drug-induced crystalluria can be noted in rats without histopathological evidence of kidney injury. In addition, urinary bladder tumors if noted in rodents due to crystalluria mechanism of action (MOA) are not relevant to human cancer risk, and rodent urothelial proliferative lesions secondary to crystalluria are not predictive of human cancer risk. Furthermore, rats are sensitive to the development of irritation-related urothelial tumors as a result of crystalluria, while mice are much less likely to develop urothelial tumors secondary to crystals or uroliths. Drug-induced kidney glomerular pathological changes can manifest clinically as nephrotic syndrome due to albumin loss (ie, plasma proteins leaks into the urine [proteinuria] through abnormally permeable GBM). Such reduction in plasma osmotic pressure leads to edema, pleural effusions, reduced intravascular volume, renal hypoperfusion, and/or secondary hyperaldosteronism. Drug-induced kidney glomerular pathological changes can also lead to nephritic syndrome, which usually presents with hematuria, red cell casts in the urine, azotemia, and/or oliguria. Toxicologists need to be familiar with this language to appropriately communicate preclinical kidney findings with nephrologists and clinicians.
For urine biomarkers, it is essential to collect urine specimens over a 16- to 24-hour period, ideally in metabolism cages on cold packs. For investigative toxicological assessment of rodent crystalluria, it is important not to fast the animals before urine collection. Historical control data from the testing facility might be used to help distinguish test article-related effects from background changes and in assessing individual animal variability. This is especially important in rats and mice because renal function tests may become more variable due to development of spontaneous age-related, species-specific kidney pathologies with no human counterpart such as chronic progressive nephropathy (CPN) as early as 12 weeks of age. 2 Rats with CPN may show increases in kidney weights; increased urine excretion of protein and calcium; decreased urinary excretion of sodium and chloride; decreased creatinine clearance; increased serum concentration of BUN, sCr, sodium, and chloride; and decreased serum concentration of albumin. Castration can result in a lower incidence and severity of CPN. The physiologic status of experimental laboratory animals, diet (eg, imbalance of calcium/phosphorus leading to mineralization in rats), and various physiologic parameters such as age, sex, and interspecies differences in kidney anatomy and physiology are important when assessing kidney responses to injurious test articles and/or chemicals. 2,7
Kidney Spontaneous Pathologies
Histopathology is recognized as the single most appropriate screen for evidence of kidney injury. Familiarity with spontaneous diseases occurring in laboratory animals is essential for the establishment of the nature and cause of morbidity or mortality in an individual animal. For example, rats, especially Sprague Dawley strain, have vault-like folds called fornices in the upper portion of kidney pelvis that extend into the renal medulla and as a result can develop age-related and species-specific spontaneous, random (ie, no dose–response relationship), mineralization, inflammation, and/or hyperplasia in males and/or females in this unique anatomic location. Mechanisms for the development of urothelial proliferative lesions in rodents (ie, genotoxicity, mitogenicity, cytotoxicity) need to be ruled out in the risk assessment paradigm. Most common spontaneous/incidental light microscopic changes in control laboratory animals include CPN in rodents, glomerular amyloidosis in CD-1 mice, glomerular lipidosis/foam cells in beagle dogs with no effects on glomerular function, immature glomeruli in the cortex of dogs 2 and monkeys, corticomedullary mineralization or nephrocalcinosis in female rats, kidney pelvis dilatation in rats, kidney pelvis minimal epithelial hyperplasia and mineralization in rats, increased hyaline droplet accumulation in rats, tubular epithelial vacuolation in mice, mineral deposits, and interstitial mononuclear cell infiltrates in multiple species (Table 2). Mineral deposits, for example, occur at the corticomedullary junction in female rats due to shedding of microvilli and microvesicles from S1 proximal tubules 22 and accumulation in the outer stripe of medulla. 7 In beagle dogs, such deposits can be noted in papillary collecting duct region and in the interstitium of renal papilla in cynomolgus monkeys. The CPN is a spontaneous disease of laboratory rats and mice that represents a significant confounder for interpretation of kidney histopathology in toxicology studies after test article administration. Male gender is a primary risk factor for developing CPN. The CPN is an age-related disease in rats and mice influenced by numerous extrinsic factors such as diet and chemical exposures. 2 Dietary restriction prevented or delayed the development of glomerulosclerosis, tubulointerstitial damage, functional changes, morbidity, and mortality associated with CPN in rats by controlling initial body and kidney growth, glomerular size, and nephron hypertrophy. 2,23 Microscopically, there is tubular atrophy, interstitial fibrosis, tubular dilation, casts, hyperplasia, dilated Bowman space, glomerulosclerosis, glomerular atrophy, casts, and interstitial inflammatory infiltrates. 2,7
Kidney Spontaneous/Incidental Pathologies in Experimental Laboratory Animals.

Mature male rats spontaneously develop prominent hyaline droplets in the cytoplasm of the S2 segment of proximal tubules. Ultrastructurally, these hyaline bodies consist of alpha2µ globulin and represent glomerular filtration and resorption and catabolism of low-molecular-weight proteins (eg, albumin and immunoglobulins) in secondary lysosomes. Alpha2µ-globulin is androgen regulated and synthesized in the liver of male rats only, which is rapidly cleared in the glomerular filtrate. Alpha2µ-globulin is lost in the urine in the normal mature male rat, creating a physiologic proteinuria. Thus, alpha2µ-globulin nephropathy is a sex- and species-specific entity that can be exacerbated by test article administration and is not relevant to humans. 2,7 Xenobiotics or their metabolites can reversibly bind to alpha2µ-globulin and decrease effectiveness of lysosomal degradation leading to accumulation. The hyaline droplets can be readily visualized with histochemical stains including the Mallory-Heidenhain stain. In addition, a wear-and-tear lipofuscin pigment can be noted in the RTECs in rats and increases with age without kidney functional sequela or clinical significance. Idiopathic necrotizing polyarteritis (beagle pain syndrome) is a spontaneous condition that can be seen in beagle dogs. Clinically, it can be associated with fever, weight loss, and cervical pain (“beagle pain syndrome”). Pathologically, there is involvement of medium-sized muscular arteries including those in the kidney leading to thickened, tortuous vasculature with fibrinoid necrosis, mixed cell infiltrates, and thrombosis. Beagle pain syndrome is presumed to be an immune-mediated condition. Other presentations of vasculitis in dogs might include dermatomyositis and similar disorders with ischemic cutaneous and vascular lesions (ie, dermatopathy).
Drug-Induced AKI and Kidney Toxicology
Acute Kidney Injury
Acute kidney injury is one of the most common causes of kidney toxicity. In AKI, impairment in kidney function (eg, rise in sCr and/or sustained oliguria) leads to retention of nitrogenous waste products. Pathophysiologically, the classification of AKI can be divided into 3 categories: (1) prerenal, (2) intrinsic [renal], and (3) postrenal. 12 In prerenal AKI, kidney perfusion decreases as a result of hypovolemia (eg, hemorrhage, vomiting, or diarrhea), decreased cardiac output (eg, heart failure or pulmonary embolism), impairment of kidney vasomodulation (eg, nonsteroidal anti-inflammatory drugs [NSAIDs], ACE inhibitors, or cyclosporine), and/or vasodilation (eg, sepsis or hepatorenal syndrome). 12 Intrinsic AKI etiopathogenesis could be related to specific insults in kidney vascular, glomerular, tubular, or interstitial compartments. Cellular insults in intrinsic AKI can lead to various cell death modalities such as apoptosis, autophagy, or regulated and genetically controlled cell death (necroptosis). 24 Vascular causes of AKI include vascular stenosis or vasculitis (eg, disseminated intravascular coagulation or drug induced). Glomerular AKI includes immune complex diseases (ICD), glomerulonephritis, and postinfectious glomerulonephritis. Tubular and interstitial AKI can be caused by ischemia (shock), inflammation (eg, sepsis, infections), rhabdomyolysis, hemolysis, crystals, ethylene glycol (EG), cisplatin toxicity, or medications (eg, aminoglycosides, rifampin, NSAIDs, prednisone). 12,25 Postrenal AKI can be caused by bladder obstruction or urinary stones.
Drug-Induced Kidney Glomerular Pathologies
As the initial plasma filtering portion of the nephron, the glomerulus plays an integral role in AKI and drug-induced injury such as ICD. The glomerulus selective barrier to filtered molecules based on their size, shape, and/or charge is facilitated by its structural anatomical features of fenestrated endothelium, a negatively charged GBM, and podocytes. These features and the presence of complement regulatory proteins are contributing factors in ICD. For example, the presence of in situ granular deposits of immunoglobulins and/or complement at the site of tissue injury in kidney sections, using immunohistochemical staining, even in few animals, is generally consistent with ICD. In the kidney of humans and/or rodents, cell surface and soluble complement regulatory proteins (eg, decay accelerating factor [CD55], membrane cofactor protein [CD47], complement receptor type 1 (CR1), CD59, clusterin, and Factor H) are expressed by various microanatomic kidney compartments, including glomerular capillaries, peritubular capillaries, proximal tubules, collecting ducts, medullary interstitium, and glomerular cells (endothelial, epithelial, and mesangial). 12,26 -28 Thus, immune complex-mediated type 3 hypersensitivity reactions and ICD are relatively common in toxicology studies, especially with biotherapeutics after repeated dose administration to monkeys. 29 The reader is strongly encouraged to refer to comprehensive reviews on ICD pathophysiology, especially as it relates collective weight-of-evidence approach, time course of complement formation, and antidrug antibody (ADA) formation and its correlation with exposure in toxicity studies. Immune complex-mediated glomerular pathology related to immunogenicity in animals is generally not considered relevant for predicting immunogenicity in humans. For example, administration of obinutuzumab, anti-CD20 monoclonal antibody, in a 26-week toxicity study in cynomolgus monkeys resulted in dose-independent ICD-mediated hypersensitivity and chronic inflammation in tissues including the kidneys. Kidney glomerulus pathological changes included mesangial hypercellularity and thickening, proliferation of parietal epithelial cells in Bowman space with crescents formation, and periglomerular mononuclear infiltrates. Subepithelial electron-dense deposits in glomerular capillary basement membrane consistent with ICD were noted by transmission electron microscopy. 30
In dogs, glomerulopathy and/or proteinuria has been observed in dogs with Cushing syndrome, a syndrome caused by excessive adrenocorticotropic hormone production (hyperadrenocorticism) and in dogs treated with prednisone. 25 Increased urine volume, decreased urine specific gravity, proteinuria, and glomerulopathy were observed in 10 male beagle dogs after oral administration of prednisone at 2.2 mg/kg, twice daily, for 6 weeks. Similarly, drug-induced glomerulopathy was observed microscopically in dogs in a 9-month toxicity study after oral administration of fosdagrocorat, a dissociated agonist of the glucocorticoid receptor, to beagle dogs. It is interesting to note that obesity resulting from ad libitum feeding exacerbated the severity of the kidney findings observed in both the fosdagrocorat- and prednisone-treated dogs, with the prednisone-treated dogs showing greater overall glucocorticoid-induced renal effects than those dosed with fosdagrocorat. 25
It is important to note that drug-induced minimal change disease (MCD) can be noted in various experimental laboratory animals. The MCD is by characterized by normal glomerular morphology by light microscopic examination and lack of tubulointerstitial or vascular pathologies. However, animals can be proteinuric with nephrotic syndrome and glomerular changes of podocyte foot process effacement and protein resorption droplets, and cell lysis can be noted only by electron microscopic examination. 5-Lipooxygenase inhibitors in rats and masitinib mesylate (tyrosine-kinase inhibitor) in dogs are examples of drug-induced MCD 31,32 (Table 3). Toxicologists need to consider alternative assessment methodologies such as electron microscopy if kidney microscopic examination is within normal limits, yet there is evidence of drug-induced proteinuria.
Examples of Drugs That Induce Kidney Pathologies in Experimental Laboratory Animals.
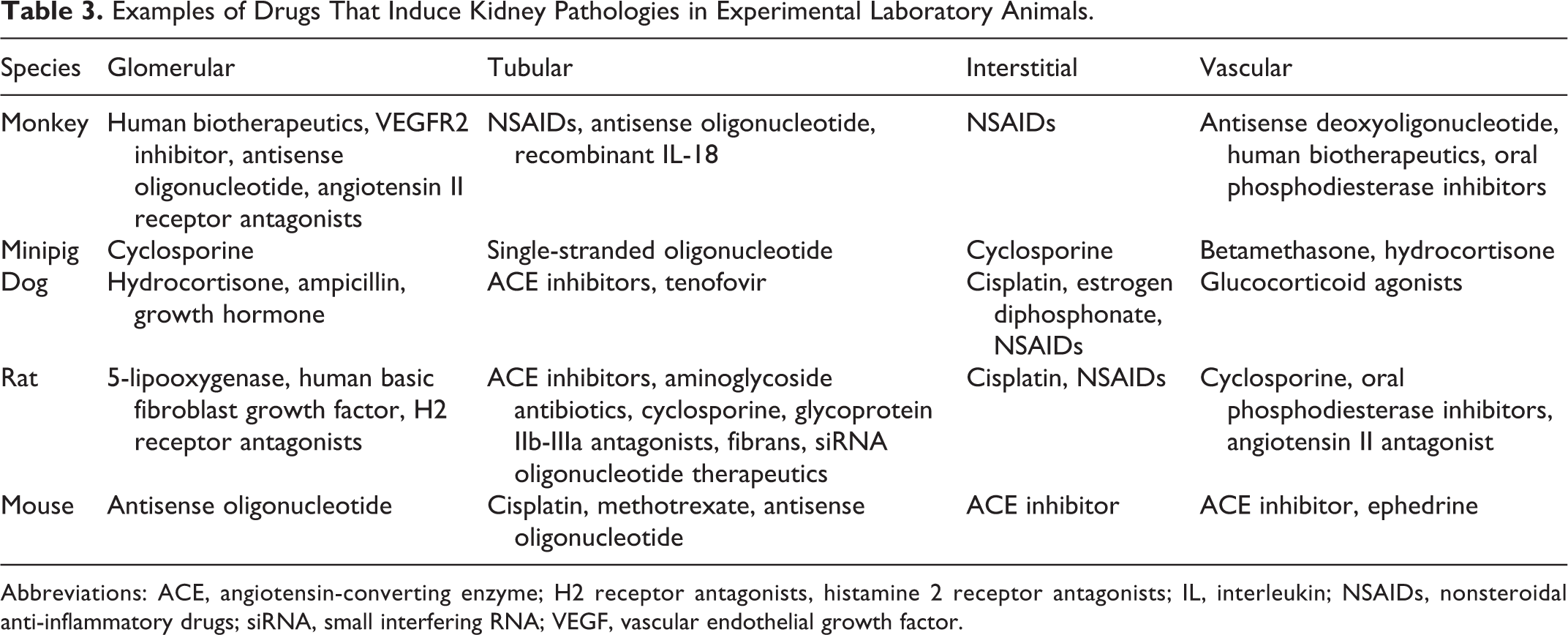
Abbreviations: ACE, angiotensin-converting enzyme; H2 receptor antagonists, histamine 2 receptor antagonists; IL, interleukin; NSAIDs, nonsteroidal anti-inflammatory drugs; siRNA, small interfering RNA; VEGF, vascular endothelial growth factor.
Drug-induced segmental glomerulosclerosis is rare in dogs but has been noted in preclinical safety studies with administration of growth hormone and synthetic progesterone. 33 Focal segmental glomerulosclerosis (FSGS) is a kidney disease in humans characterized by progressive glomerular scarring and a clinical presentation of nephrotic syndrome. In primary FSGS, immunologic podocyte injury takes place. In secondary FSGS, pathologic glomerular hyperfiltration/hypertension takes place in patients with an underlying etiology (eg, obesity, diabetes mellitus). Examples of drug-induced FSGS-like pathologies include Adriamycin, puromycin, and streptozotocin. In the early stages of FSGS-like pathologies, glomerular hypertrophy/enlargement and/or mesangial hypercellularity might be noted morphologically. Interestingly, glomerular tufts collapse and extracapillary proliferation with reticular inclusions can be noted by electron microscopy with some viral infections (eg, Simian immunodeficiency virus) or some drugs [anabolic steroids and interferons]). Differentiating primary versus secondary FSGS-like glomerular pathologies in experimental laboratory animals administered a test article is important in understanding drug MOA and differentiating primary versus secondary glomerular insults.
Drug-induced glomerular vacuolation and proteinuria has been noted after intravenous administration of human basic fibroblast growth factor to rats and monkeys. Histopathologically, enlargement, vacuolation, and karyomegaly of podocytes in glomeruli, hypertrophy/hyperplasia of the parietal epithelium of Bowman capsule in the glomeruli, tubular dilatation and cast formation, thickening of the media in the lobular arteries, arteriopathy, and/or hyperplasia of the epithelium of the papilla and collecting ducts were noted. 34
Anti-vascular endothelial growth factor (VEGF) therapies can induce glomerular thrombotic microangiopathy, mild proteinuria, and hypertension in humans. 35 Strong VEGF expression in mice is demonstrable using immunohistochemistry in glomerular podocytes. 36 In addition, VEGF plays a role in glomerular endothelial cell fenestration formation, at least in vitro. 37 The VEGF is released by podocytes and binds to its receptors on endothelial cells (eg, VEGFR-2). This helps maintain podocyte health and slit diaphragm integrity because inhibition of VEGF signaling downregulates the expression of nephrin, a protein involved in the maintenance of the glomerular slit diaphragm. Reduction in VEGF after administration of some anti-VEGF drugs (eg, VEGFR-2 inhibitor) can lead to dose-dependent, yet reversible, glomerular filtration barrier injury in rats and/or monkeys after test article administration (Table 3). Histopathologically, eosinophilic, hyaline droplets can be noted in podocytes, mesangial, and/or endothelial cells and podocyte and parietal epithelial cell hypertrophy and mesangial cell hyperplasia. Similar droplets might be seen in the S3 segment of proximal tubules.
Kidney Drug-Induced Tubular, Interstitial, and Vascular Pathologies
The immunopathogenesis of AKI is an important consideration as the kidneys contribute to the immune system homeostasis by removing circulating cytokines and bacterial toxins (eg, LPS) and by continuously sampling blood-borne proteins. 38 Emerging evidence supports the involvement of RTECs and the innate and adaptive arms of the immune system in the pathogenesis of intrinsic AKI. Pro-inflammatory DAMPs, PAMPs, TLRs, oxidative stress, HIF, complement system, adhesion molecules, cell death, resident renal DCs, neutrophils, T and B lymphocytes, macrophages, natural killer T cells, secreted cytokines, and chemokines contribute to the immunopathogenesis of AKI. Thus, loss of the immune system homeostasis can have direct or indirect effects on kidney pathology. If the immunopathologic processes in AKI continue, this can lead to renal fibrosis and/or chronic kidney disease. 12
Sepsis is one of the most common causes of AKI.
39
In sepsis, there is a systemic inflammatory response, triggered by the innate immune system, to attack foreign bacterial pathogens (eg, LPS) and toxins to limit their spread.
20
Such systemic inflammatory response leads to the release of PAMPs and DAMPs into the circulation.
20
The PAMPs are released from pathogens and DAMPs are released from damaged and injured tissues at sites of infection, and both PAMPs and DAMPs prime and signal the immune system to fight back the invading pathogens and toxins. Interactions of PAMPs and DAMPs with sensors of the innate immune system called pattern recognition receptors (eg, TLRs, nucleotide-binding oligomerization domain-like receptors, and retinoic-acid-inducible-gene-I [
The kidney parenchyma is especially vulnerable to the pro-inflammatory response to infection and subsequent kidney tissue damage because it receives approximately 25% of the cardiac output. This is because the toxin-rich blood, PAMPs, DAMPs, and pro-inflammatory mediators gain access to kidney parenchyma and its various compartments via glomerular filtration or peritubular capillaries microcirculation. 19,20 For example, LPS is filtered into the tubular fluid and can directly interact with RTECs via a TLR4-dependent mechanism. 19,21 As a consequence, various compartments of the kidney including RTEC, glomerular, and endothelial are injured. This is because the filtered PAMPs and DAMPs, also called “danger signal,” bind to and activate RTECs and kidney endothelial and glomerular cells and can lead to endothelial cell activation and structural changes in the glomerular and peritubular capillaries. 20 It is important to distinguish adaptive kidney response (eg, drug-induced medullary tubular hypertrophy, tubular hypertrophy secondary to increased GFR, potassium depletion [chronic hypokalemia], high dietary sodium chloride, increased sodium loss, testosterone treatment, adrenocorticotropic hormone-mediated hyperadrenocorticism 1,2 ) from direct injurious tubular damage. Collectively, exogenous and endogenous PAMPs and DAMPs are integral to AKI pathogenesis.
Examples of drug-induced tubular injury include aminoglycoside antibiotics (eg, gentamicin), cephalosporins, cisplatin, antiviral agents, and calcineurin (CNI) inhibitors (Table 3). Pathophysiologic factors in aminoglycoside kidney toxicology include alterations in tubular cells reabsorption, tubular obstruction and tubular malfunction, reduced glomerular filtration, tubuloglomerular feedback activation, renal vasoconstriction, and mesangial contraction. 42 Local kidney factors (eg, variability in P-glycoprotein and CYP3A4/5 expression or activity, older age, salt depletion, use of NSAIDs, and genetic polymorphisms in genes) are important for susceptibility CNI kidney toxicity. 43
Unless associated with morphologic evidence of tubular cell injury of necrosis and/or degeneration, antisense oligonucleotides (ASO) can cause nonadverse and adaptive kidney changes in rats evident histologically by accumulation of basophilic or eosinophilic and granular, amorphous, or vacuolar material in the cytoplasm or nuclei of renal epithelial cells. This is consistent with lysosomal accumulation and endosomal localization of ASO. Oligonucleotides can be absorbed rapidly and distributed in tissues, most commonly in the kidneys, after systemic or local administration. In rats, exacerbation of CPN and proteinuria might be observed after ASO chronic administration. Pathophysiologically, once oligonucleotides interact with and taken by the brush boarder of proximal tubules via polyanion transporter, they are internalized by endocytosis, leading to incorporation into endosomes and eventually lysosomes and phagolysosomes. Some reports suggest that although there are high concentrations of ASO and cytoplasmic vacuolation, no evidence of kidney functional abnormalities (eg, sCr or BUN) were noted, but low incidence of reversible increase in urine protein to creatinine ratio was also noted in monkeys. 44
Drugs that can induce crystal nephropathy in humans and/or experimental animals include quinolone antibiotics (eg, ampicillin, norfloxacin, and ciprofloxacin), sulfonamides and fluoroquinolones, glycoprotein IIb-IIIa antagonists, ganciclovir, sulfadiazine, and dopamine autoreceptor agonist (Table 3). Intratubular precipitation of xenobiotics/crystals can cause or promote AKI and chronic kidney injury. A number of xenobiotics/drugs are known to precipitate within renal tubules, due to their insolubility (parent drug or its metabolite) within urine. Risk factors for xenobiotic/crystal precipitation within the kidney tubules include intravascular volume depletion, underlying kidney disease, and metabolic disturbances that promote changes in urine pH. EG-mediated crystal nephropathy requires metabolic transformation of EG to oxalic acid that takes place primarily in the liver via alcohol dehydrogenase. When oxalic acid is concentrated in the urine, calcium oxalate crystals form, leading to crystalluria, nephrolithiasis, metabolic acidosis, low urine specific gravity, and proteinuria. The EG-induced crystal deposition in kidney proximal tubules was detected unbound in the lumen and attached to the surface of epithelial cells. 45 Crystal formation can lead to chronic interstitial nephritis with glomerulosclerosis, tubular atrophy, and interstitial fibrosis. 46 Sulfonamides have caused crystal nephropathy, affecting the distal nephron in animals and humans. As the pH of murine urine generally varies between 5 and 8, and is greatly influenced by diet, this would influence crystal nephropathy formation. In addition, a major difference between murine and human urine is the overall osmolality. Murine urine is usually highly concentrated, with male urine having higher protein concentrations than females. It is important to note that for human risk assessment, murine urinary crystalluria MOA is not relevant to human urinary bladder cancer risk.
Cyclooxygenase-mediated prostaglandins are involved in renal functions and their inhibition is associated with potential adverse renal effects. 11 Analgesic nephropathy is characterized by chronic interstitial nephritis and renal papillary necrosis (RPN) or calcifications. Some laboratory animals (eg, rats, dogs) are unusually susceptible to RPN. Kidney changes in monkeys associated with administration of NSAIDs-containing carboxylic acid, such as naproxen, and carboxylic acid moiety (eg, fibrans) were shown to induce formation of casts with crystalline material that precipitates in the kidneys. 29 This can lead to obstructive nephropathy in rats because of accumulation of intratubular crystal precipitates (eg, glycoprotein IIb-IIIa antagonists). 46 Obstructive nephropathy can be seen secondary to co-precipitation of administered IL-18 and Tamm-Horsfall protein in the distal nephron (Table 3). 47
Drug-induced inclusion bodies can be noted in rats with norepinephrine/serotonin reuptake inhibitors and propiverine (anticholinergic drug). Such inclusion bodies can be related to
Conclusions
Detection and characterization of direct and indirect kidney changes and/or toxicity in preclinical species and human relevance will depend on the preclinical safety testing strategy and collective weight-of-evidence approach including new investigational drug MOA, drug physiochemical properties, distinguishing effects related to the parent drug or its metabolite, changes in sCr related to renal transporters, preclinical and clinical interspecies, and sex differences in kidney macro- and micro-anatomy, physiology, and metabolic and hormonal activities, background pathologies, drug-induced, yet sex- and/or species-specific kidney toxicity, and MOA relevance to humans. Various investigative toxicology and pathology tools can be employed on a case-by-case scientific basis to better characterize pharmaceutical drug-induced kidney toxicities.
Footnotes
Author Contributions
Z. Radi contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author is a Pfizer Inc. employee.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
