Abstract
Influx and efflux kidney tubular transporters are major determinants of the disposition of xenobiotics, including pharmaceutical drugs. On the basolateral membrane of proximal tubular cells, there are influx transporters, such as organic cation transporters. On the apical membrane of proximal tubular cells, there are efflux transporters, such as multidrug and toxin extrusion proteins. The secretion process across the apical membrane into the lumen occurs via efflux transporters which plays an important role in serum creatinine (sCr) elimination in urine. The interference of a pharmaceutical drug with transporters can lead to changes in sCr with no alterations in biomarkers or light microscopic evidence indicative of renal injury. Identification of transporters that influence drug disposition, toxicity, and overall nonclinical safety assessment is important in drug discovery and development programs. This mini review describes some key aspects of kidney tubular transporters and drug-induced renal toxicities in safety risk assessment and drug development.
Detection and characterization of renal injury in preclinical species as well as determining the human relevance of the injury is important and will depend on a preclinical safety testing strategy combined with a collective weight of evidence approach. 1 The high activity of transporters along the kidney tubular epithelium contributes to renal injury after exposure to xenobiotics including pharmaceutical drugs. 1 Physiologically, proximal tubular epithelial cells account for approximately 70% of all reabsorptive activity, 1 and these cells express a variety of kidney transporters. 2 Transporters are present on both the luminal (apical; eg, multidrug and toxin extrusion proteins [MATEs]) and blood-facing (basolateral) membrane (eg, organic anion transporters [OATs]) of renal tubular epithelial cells (Figure 1) and can either transport a pharmaceutical drug out of the kidney tubular lumen back into the blood (reabsorption) or into the ultrafiltrate (secretion). 1 In drug development, these transporters can affect the renal clearance of pharmaceutical drugs by regulating drug secretion and/or reabsorption and subsequent drug-induced renal toxicity. 2,3 Examples of investigational drugs or compounds that may cause renal toxicity which is influenced by kidney transporters include some β-lactam antibiotics, cancer therapeutics, antiviral drugs (Figure 1). 2,4
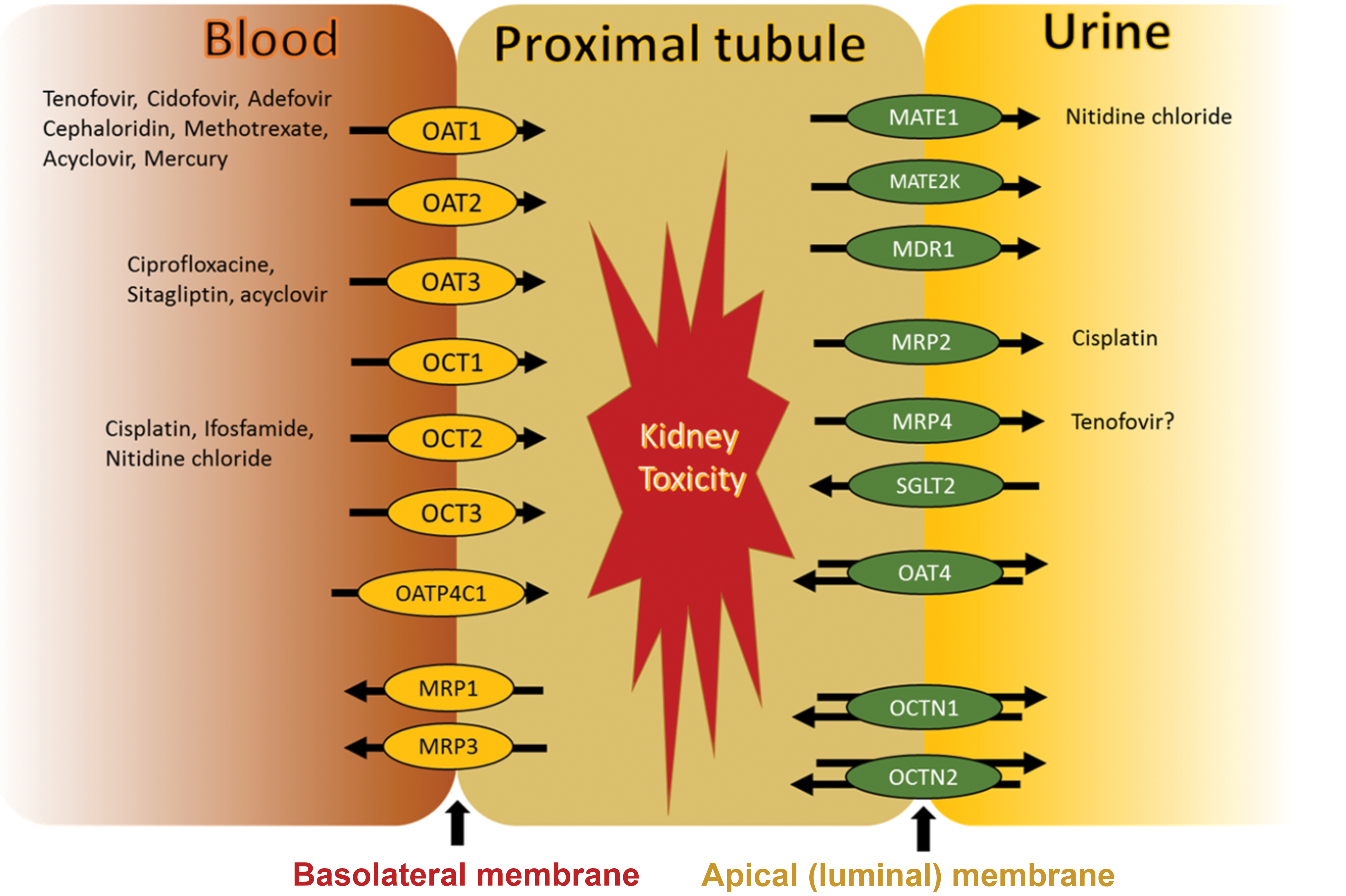
Renal tubular transporters. Kidney proximal tubules contain OAT1-3, OATP4C1, OCT1-3, and MRP1/3 at the basolateral membrane and MATE1, MATE2k, MDR1, MRP2/4, SGLT2, OAT4, and OCTN1/2 at the apical membrane. Both uptake and efflux drug transporters are involved in kidney toxicity of some drugs. Source: reprinted from basit, radi, vaidya, karasu, prasad, ©2019. MATE, multidrug and toxin extrusion; MRP, multidrug resistance–associated protein; OAT, organic anion transporter; OCT, organic cation transporter; SGLT, sodium-glucose cotransporter.
The function or activity of transporters can be altered in kidney diseases. For example, administration of the antimicrobial drug, morinidazole, to patients with severe renal impairment resulted in increased plasma drug exposure and reduced renal clearance of morinidazole-conjugated metabolites which are substrates of kidney transporters (eg, OAT). 5 In one study, renal impairment due to accumulation of high amounts of endogenous uremic toxins in 5/6 nephrectomized rats, a model of chronic renal failure in humans, inhibited OAT3-mediated uptake of glucuronides and resulted in increased plasma exposure. 6
Some kidney transporters can be used as pharmacological targets to treat kidney diseases. For example, the healthy kidney reabsorbs approximately 90% of the filtered glucose, a process that is mediated by the sodium-glucose cotransporter 2 (SGLT2) located at the apical membrane of proximal tubular cells to drive glucose into cells together with sodium. Therefore, pharmaceutical drugs such as SGLT2 inhibitors are developed to reduce glucose reabsorption and treat renal glucosuria. Selective SGLT2 inhibitors have demonstrated protective effects in diabetic kidney disease. 7 Collectively, knowledge of kidney transporters and their contribution to kidney health and role in disease is important for renal safety assessment and can be leveraged for pharmaceutical interventions.
Expression, function, localization, and homology of kidney transporters vary by species, 2 and key species differences in kidney transporters are summarized in Table 1. Understanding cross-species differences is important, as it allows for preclinical to clinical translation of transporter-mediated secretary clearance and the distinction between direct drug-induced renal toxicity from indirect effect on kidney transporters. 2 For example, organic cation transporter 3 (OCT3) was only detectable in humans, and the abundance of OAT1, OCTN2, SGLT2, multidrug resistance–associated protein 1 (MRP1), sodium/potassium-transporting ATPase, and MRP3 was highest in rats and lowest in humans 2 (Table 1). Such cross-species differences can contribute to differences in renal toxicity between animals and humans. For example, the renally excreted antiviral drug, tenofovir, where human OAT1 Km value was significantly lower than OAT1 orthologs from preclinical species (including cynomolgus monkeys, mice, rats, and dogs), resulted in significant renal toxicity in humans due to greater accumulation of the drug in the proximal tubules. 8 The OAT2 transporter was localized at the basolateral membrane of proximal tubules of human and nonhuman primate kidneys, while comparatively, its localization was distributed to the apical and basolateral membranes in rats. 9 This is important because OAT2 transporter affects renal elimination of drugs and endogenous compounds. 9 A similar transport rate, efficiency, transporter function, and a high degree of amino acid sequence identity in nonhuman primates versus their human counterparts was reported for OCT2, MATE1, and MATE2K transporters. 10 This suggests that nonhuman primates might be used as a preclinical model to investigate drug-induced renal toxicity related to the inhibition of these kidney transporters. 10
Kidney Cross-Species Key Differences in Transporters.
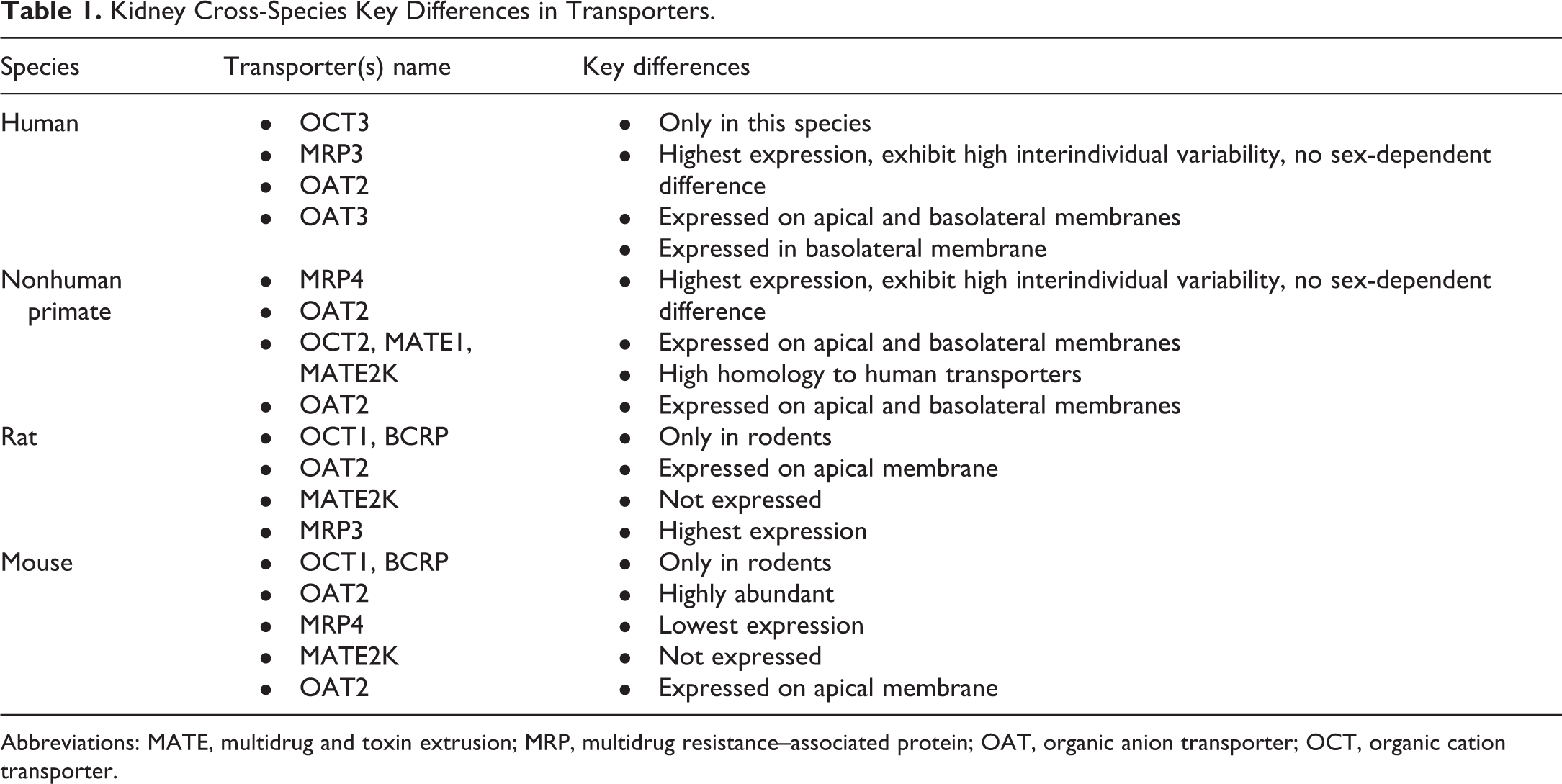
Abbreviations: MATE, multidrug and toxin extrusion; MRP, multidrug resistance–associated protein; OAT, organic anion transporter; OCT, organic cation transporter.
Key sex differences in kidney transporters are summarized in Table 1. Significant sex differences in transporter expression are noted in mice, followed by rats, then dog; however, no such sex difference in expression is noted in humans or nonhuman primates 2 (Table 1). Higher OAT2 and OAT5 transporter expression was noted in female rats and mice versus males. 11,12 Humans and nonhuman primate kidney transporters have also shown a high degree of interindividual variability with no sex differences 2 (Table 1). In summary, there are key cross-species differences in kidney transporters that will help in the preclinical to clinical translation of drug-induced renal toxicity.
The physiochemical properties of drugs are important attributes because they influence the function of kidney transporters, drug disposition, and drug-induced renal toxicity. For example, pharmaceutical drugs that are relatively hydrophilic and more polar have a high affinity to kidney transporters. Such hydrophilic and ionized drugs with hydrogen-bonding abilities, determined by the drug polar surface area as well as the number of hydrogen bond donors and acceptors, are most likely to be secreted mainly into the urine because of their ability to interact with kidney transporters, such as OAT1, OAT3, and OCT2. 3 In addition, kidney transporters contribute to drug concentration variability and exposure response following administration of a drug, metabolite, or coadministration of a drug. For example, renal tubular uptake of cisplatin from the systemic circulation is mediated via OCT2. 13 Oral administration of imatinib, a cationic anticancer agent, significantly increased (approximately 120%) the area under the plasma concentration-time curve of the intravenously administered cisplatin. 13 Furthermore, coadministration of imatinib with cisplatin prevented cisplatin-induced renal toxicity in rats by inhibiting cisplatin uptake and accumulation into renal tubules from the bloodstream by OCT2. 13 In conclusion, knowledge of physiochemical properties and transporters that affect the renal clearance of pharmaceutical drugs and subsequent drug-induced renal toxicity or drug–drug interactions can be used to guide renal safety assessment.
Serum creatinine (sCr), a small organic cation, is formed from the breakdown of creatine phosphate. Approximately 2% of total creatine is converted to creatinine daily and is eliminated renally, via glomerular filtration and tubular secretion, and drug-induced increases in sCr can be noted without microscopic changes in the kidney. 1,14 Although there are known factors that influence sCr levels such as weight, age, sex, muscle metabolism, hydration state, and protein intake, kidney transporters can also play a major role in the uptake of creatinine from the blood. 14 For example, the renal efflux transporters, MATE1 and MATE2K, and uptake transporters, OCT2 and OAT2, play an important role in sCr elimination in urine. 9,14,15 Creatinine clearance based on sCr is used to estimate glomerular filtration rate. Furthermore, drugs can inhibit renal secretion of sCr. Thus, drug-induced increases of up to 30% in sCr can be noted in preclinical species in a dose-dependent manner and could be related to either a direct tubule damage (ie, drug-induced kidney injury) or an indirect effect (ie, without evidence of kidney injury) related to the inhibition of kidney transporters without effects on renal function biomarkers.
In multiple dose studies in healthy volunteers, oral administration of a Janus kinase inhibitor, INCB039110, resulted in dose-dependent and reversible rises in sCr levels. 15 A follow-up renal function clinical study in healthy volunteers revealed that INCB039110 did not affect the clearance of iohexol, a marker of glomerular filtration. Increases in sCr were attributed to interference with renal uptake transporters, such as OCT2, OCT3, and OAT2 and efflux transporters, such as MATE1 and MATE2K. 15 Thus, understanding which transporters are potentially involved in the uptake or efflux of pharmaceutical drug candidates and knowing kidney transporters across nonclinical species and humans will allow better interpretation of a renal injury in a clinical setting.
Conclusions
Kidney transporters contribute to kidney health and disease, and influence drug disposition, drug–drug interactions, toxicity, and overall safety assessment in drug discovery and development programs. Knowledge of cross-species differences in kidney transporters, and factors that influence their function and drug disposition (eg, physiochemical properties) can be used to guide renal safety assessment.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
