Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of 25 monosaccharides, disaccharides, and related ingredients and concluded these are safe in the present practices of use and concentration described in the safety assessment. Many of these ingredients are common dietary sugars, dietary sugar replacements, or very closely related analogs and salts; 7 of the ingredients are listed by the Food and Drug Administration as generally recognized as safe food substances. The most commonly reported cosmetic function is as a skin-conditioning agent; other commonly reported functions are use as a humectant or as a flavoring agent. The Panel reviewed the animal and clinical data included in this assessment, acknowledged that the oral safety of many of these ingredients has been well established, and found it appropriate to extrapolate the existing information to conclude on the safety of all the monosaccharides, disaccharides, and related ingredients.
Introduction
This report addresses the safety of the following 25 monosaccharides, disaccharides, and related ingredients as used in cosmetic formulations: Calcium Gluconate#
Fructose#
Fucose Galactose Galactosyl fructose Galacturonic acid Gluconic acid Glucose#
Isomalt##
Kefiran Lactitol##
Lactose##
Lactulose Maltose Mannose Melibiose Potassium gluconate#
Rhamnose Ribose Sodium gluconate#
Sucralose#
Sucrose#
Trehalose##
Xylobiose Xylose
#Generally recognized as safe (GRAS) food additive or approved direct food additive
##Listed in the Food Chemical Codex
The monosaccharides, disaccharides, and related ingredients have a number of reported functions in cosmetics, with the most common use as a skin-conditioning agent (Table 1). 1 Other commonly reported functions are use as a humectant or as a flavoring agent.
Definitions, Structures, and Reported Functions.a,b
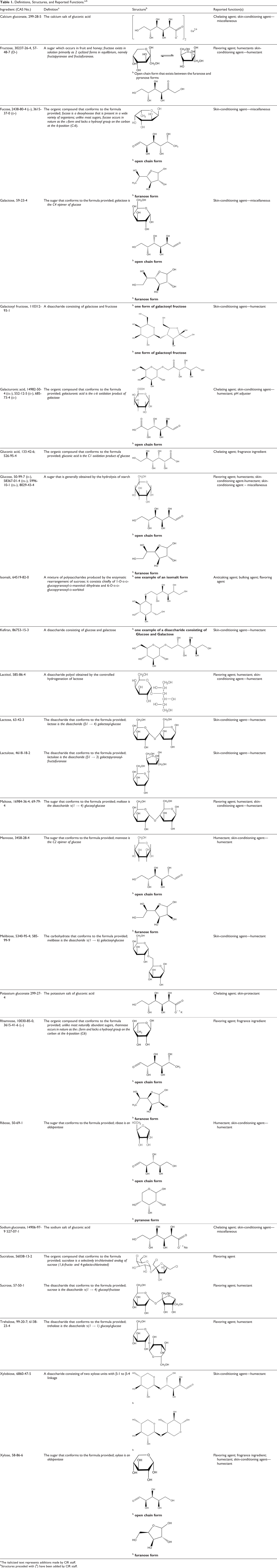
a The italicized text represents additions made by CIR staff.
b Structures preceded with (b) have been added by CIR staff.
Most of these ingredients included in this safety assessment are common dietary sugars, dietary sugar replacements, or very closely related analogs and salts. Several are listed by the Food and Drug Administration (FDA) as generally recognized as safe (GRAS) food additives 2 or direct food additives and/or are listed in the Food Chemicals Codex 3 as used in foods; for these ingredients, the focus of this assessment will be on dermal effects, primarily dermal irritation and sensitization. This approach is supported by the fact that some of these ingredients, namely, fructose, galactose, glucose, lactose, sodium gluconate, and sucrose, are listed in Annex IV of the European Registration, Evaluation, Authorization and Restriction of Chemicals (REACH), 4 which “sets out substances that are exempted from the registration, evaluation, and downstream user provisions of REACH as sufficient information is known about these substances that they are considered to cause minimum risk because of their intrinsic properties.” 5
For those ingredients that are not identified as common dietary substances, that is, fucose, galactosyl fructose, galacturonic acid, kefiran, lactulose, mannose, melibiose, and xylobiose, a search for oral toxicity data was performed. Very limited published data were found.
Chemistry
Definition
A monosaccharide is a carbohydrate that cannot be decomposed to a simpler carbohydrate by hydrolysis and is often called a simple sugar. 6 A disaccharide is a carbohydrate that yields 2 monosaccharides upon hydrolysis. Many of these ingredients exist in equilibrium between an open chain form and one or more ring forms, resulting in a hemiacetal or hemiketal linkage involving the aldehyde (aldose) or ketone (ketose) moiety of the open chain form, with 2 possible stereochemical configurations (Figure 1). The resulting stereoisomers are called anomers, and the stereocenter is referred to as the anomeric carbon. The definition and structure of each ingredient included in this report is provided in Table 1.
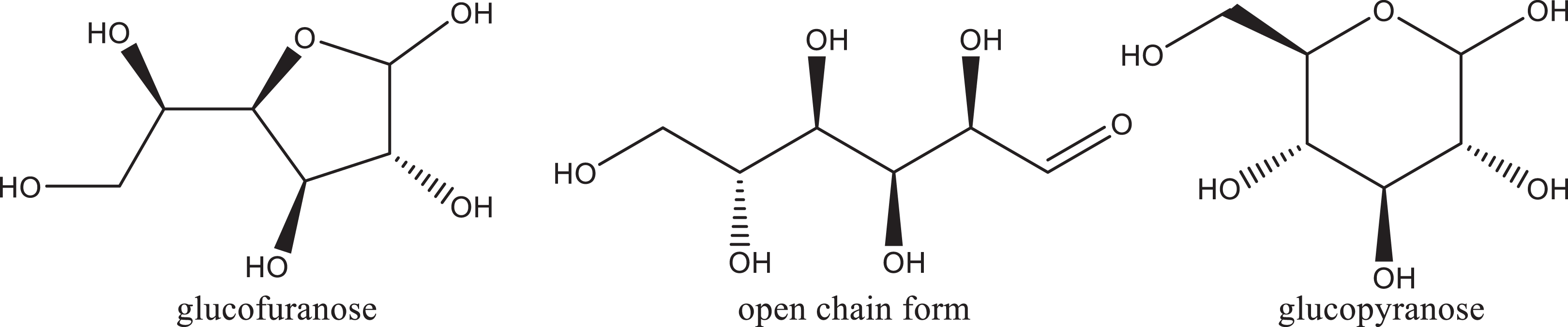
Structural forms of
Chemical and Physical Properties
Due to the high degree of substitution with hydroxyl groups, the monosaccharides and disaccharides are very hydrophilic and readily dissolve in aqueous solvent systems. These sugars have molecular weights ranging from 142 to 391 Daltons and are solids at room temperature, with many having multiple known crystalline forms (Table 2). 3,7 –29
Chemical and Physical Properties.

Natural Occurrence and Methods of Manufacture
The manufacture of the majority of these monosaccharides, disaccharides, and related ingredients is accomplished by extraction from plant sources (Table 3). For instance, the sugar industry processes sugar cane and sugar beets to obtain sucrose. 30 Sugar cane contains 70% water, 14% fiber, 13.3% saccharose (about 10%-15% sucrose), and 2.7% soluble impurities. Sugar cane is extracted with water, clarified to remove mud, evaporated to prepare syrup, crystallized to separate the liquor, and centrifuged to separate molasses from the crystals. Sugar crystals are then dried and may be further refined before bagging for shipment. Sugar beet (75% water and 17% sugar) processing differs in the washing, preparation, and extraction. After washing, the beet is sliced and extracted with water. Sugar refining involves removal of impurities and decolorization. The steps generally followed include affination (mingling and centrifugation), melting, clarification, decolorization (with activated carbon, ion exchange resins, and so on), evaporation, crystallization, and finishing.
Natural Occurrence and/or Methods of Preparation.

Constituents/Impurities
Purity and composition specifications are available for the food and pharmaceutical uses of many of these ingredients (Table 4).
Purity Specifications.

Abbreviations: NMT, not more than; USP, United States Pharmacopeia.
Use
Cosmetic
The ingredients included in this safety assessment have a variety of functions in cosmetics. The most common function is as a skin conditioning agent; many are also reported to function as flavoring agents (Table 1).
The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2014 31 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) in 2013 32 indicate that 22 of the 25 ingredients included in this safety assessment are used in cosmetic formulations.
According to the VCRP data, sucrose has the greatest number of reported uses, 738, followed by trehalose with 474 uses and glucose with 425 uses (Table 5). 31 A concentration of use survey conducted by the Council found that the use of these monosaccharides and disaccharides varies widely by ingredient and use type. 32 Glucose has the highest reported use concentration in a leave-on product; it is reported to be used at 91% in “other” noncoloring hair preparations. It is also used at 97.8% in an ingestible oral hygiene product. Sucrose has the next highest reported use concentration; it is used at up to 58% in leave-on formulations (ie, in other skin care preparations) and 65% in rinse-off products (ie, in other personal cleanliness products). However, most of the ingredients are used at <1% in leave-on products. The 3 ingredients not reported to be used are galactose, galacturonic acid, and lactulose (Table 6).
Frequency and concentration of use according to duration and type of exposure.
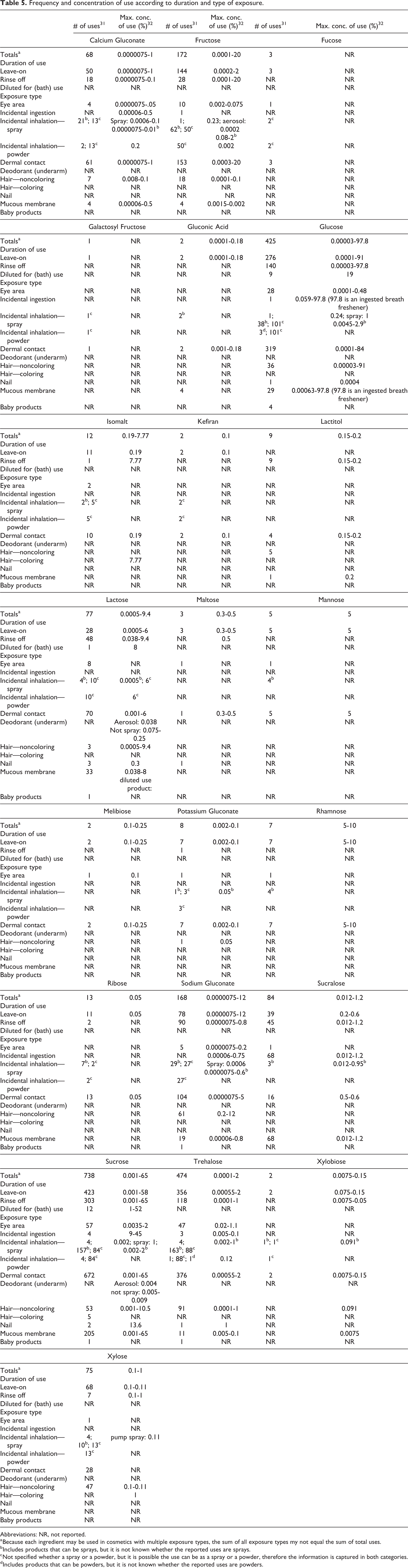
Abbreviations: NR, not reported.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
b Includes products that can be sprays, but it is not known whether the reported uses are sprays.
c Not specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
d Includes products that can be powders, but it is not known whether the reported uses are powders.
Ingredients Not Reported to be Used.

Voluntary Cosmetic Registration Program data indicate that glucose, lactose, sodium gluconate, sucrose, and trehalose are used in baby products; however, concentration of use data for baby products were not reported by industry. Some of the ingredients used are in products that could be incidentally, or are purposely, ingested (e.g., 97.8% glucose in an ingestible oral hygiene product), and some are used near the eye area or mucous membranes (eg, 2% sucrose in eye lotion and 65% in personal cleanliness products, respectively). Additionally, some of these ingredients are used in cosmetic sprays and powders that could possibly be inhaled (eg, glucose is used at 1% in a spray body and hand preparation). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared to pump sprays. 33,34 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 35,36 All of the ingredients included in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 37
Noncosmetic
Several of the ingredients have specific GRAS food and direct food additive uses: Calcium gluconate: GRAS designation; a direct food additive that meets the specifications of the Foods Chemical Codex; it is used as a firming agent, formulation aid, sequestrant, and texturizer at levels not to exceed current good manufacturing practices (GMP); GMP result in a maximum level, as served, of 1.75% for baked goods, 0.4% for dairy product analogs, 4.5% for gelatins and puddings, and 0.01% for sugar substitutes (21CFR184.1199) Fructose: A direct food additive; in high fructose corn syrup (containing approximately 42 or 55% fructose); high fructose corn syrup must conform to the identity and specifications listed in the monograph entitled “High-Fructose Corn Syrup” in the Food Chemicals Codex, with no limitation other than current GMP (21CFR184.1866) Glucose: GRAS direct food additive ( Potassium gluconate: GRAS designation; does not have a CFR citation.
2
The Select Committee on GRAS Substances (SCOGS) concluded there is no evidence in the available information on potassium gluconate that demonstrates or suggests reasonable grounds to suspect a hazard to the public when they are used at levels that are now current or might reasonably be expected in the future.
38
Sodium gluconate: GRAS designation; as a sequestrant in food, with no limitation other than current GMP (21CFR182.6757) Sucralose: Direct food additive as a multipurpose additive that meets the specifications of the Foods Chemical Codex; it is used as a sweetener in foods generally, in accordance with current GMP in an amount not to exceed that reasonably required to accomplish the intended effect (21CFR172.831) Sucrose: GRAS designation; a direct food additive that must be of a purity suitable for its intended use, with no limitation other than current GMP (21CFR184.1854)
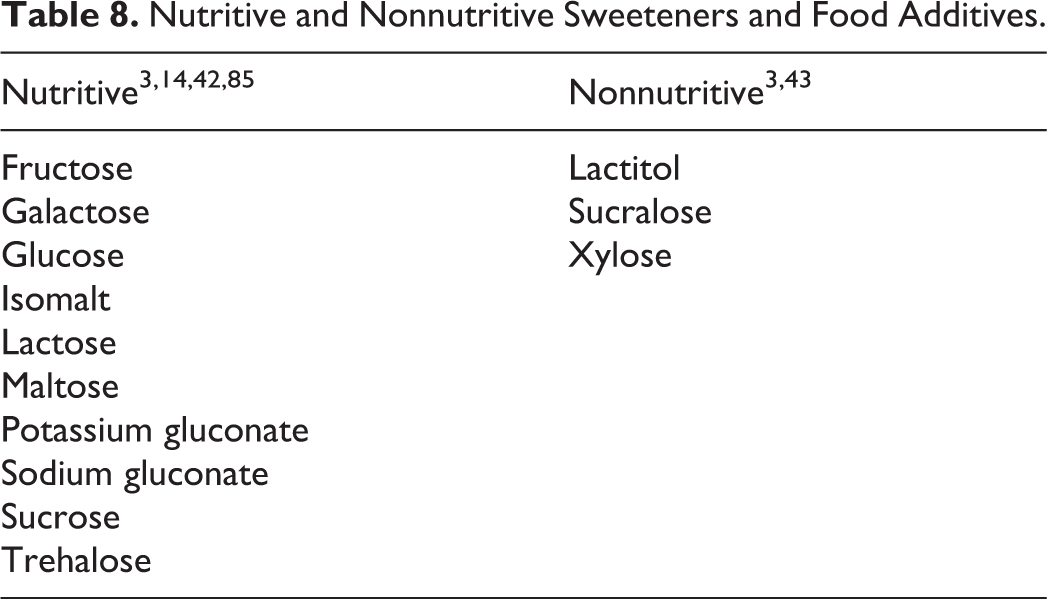
Lactulose is an approved drug used to treat constipation. 39 A general list of noncosmetic uses, including food uses that are not affirmed as GRAS or those that are inactive ingredients in approved drugs, are listed in Table 7. 3,9,10,12,26,39 –41 Table 8 provides a listing of those ingredients that are nutritive and nonnutritive sweeteners. 3,14,42,43
Examples of Noncosmetic Uses.

Abbreviation: IV, intravenous.
Nutritive and Nonnutritive Sweeteners and Food Additives.
In Europe, the following are listed in REACH Annex IV: fructose, galactose, glucose, lactose, sodium gluconate, and sucrose. 4 Substances included in Annex IV are exempted from registration (as well as downstream user requirements and evaluation) for all their possible uses irrespective of the tonnage in which they are manufactured or imported (currently or in the future).
Toxicokinetics
Although many of the ingredients included in this safety assessment are food ingredients, they are not all processed by the body in the same manner (see Tables 8 and 9). Some are nutrients, which are absorbed intact in the small intestines and then metabolized in the body to serve as sources of energy and others are not (Table 8). For example, glucose 44 and potassium gluconate 45,46 are rapidly absorbed in the small intestine (Table 9). 9,12,23,27,38,44 –61 In contrast, isomalt is absorbed only to a limited extent, 12 and lactitol, 12 lactulose, 52 and sucralose 23,55,57 are not absorbed in the gut. Trehalose can be metabolized by trehalase in the gut to produce glucose, which can then be readily absorbed. Some of these ingredients (eg, gluconic acid, potassium gluconate, and sodium gluconate) are important intermediates in carbohydrate metabolism; gluconic acid is a normal metabolic product of glucose oxidation, and the amounts produced endogenously are much greater than what is consumed. 9 Because the absorption, distribution, metabolism, and elimination of most of the ingredients included in this safety assessment have been reviewed to evaluate their use as common dietary substances, only summary information is provided in this report.
Summary Metabolism Data.

Abreviation: GRAS, generally recognized as safe.
Dermal Penetration
In vitro
Glucose
The permeability coefficient for glucose was determined in vitro using full-thickness mouse skin and the dermis of nude mice.
62
Unlabeled glucose, 0.01 mol/L, was first used on both sides of the skin to saturate the sorptive capacity of the cell system. A concentration of 3.3 × 10−6 mol/L
In Vivo
Glucose
The transdermal penetration of glucose through Rhesus monkey skin was measured using optical coherence tomography (OCT). 63 The hair on the right hind leg of 4 anesthetized monkeys was shaved, a probe holder was taped to the shaved skin, and 0.2 mL of 20% concentrated glucose in distilled water was applied topically through the hole in the probe holder during the course of imaging. The skin was imaged using OCT for 8 minutes prior to application of the glucose and then for 2 hours after application. The diffusion process was monitored in a 140-μm thick region 210 μm below the dermis region. The mean permeability rate of 20% glucose was calculated to be (4.41 ± 0.28) × 10−6 cm/s.
Toxicological Studies
Most of the ingredients included in this assessment are found in foods, and the daily exposure from that food use would result in a much larger systemic dose than that resulting from use in cosmetic products. Numerous studies and reviews have been published about the safety of dietary exposure to monosaccharides and disaccharides. Examples of these reviews include the “Evaluation of the Health Aspects of Sucrose as a Food Ingredient” 49 and “Evaluation of the Health Aspects of Sodium, Potassium Magnesium, and Zinc Gluconates as Food Ingredients.” 50 Also, many of the ingredients included in this report are used as inactive ingredients in approved drugs that are administered via numerous routes. Consequently, systemic toxicity is not addressed further in this report for those ingredients that are GRAS food substances, direct food additives, or identified in the Food Chemicals Codex as used in foods. The focus of the safe use of those monosaccharides and disaccharides as cosmetic ingredients is on the potential for irritation and sensitization. When available, dermal toxicity, ocular irritation, and genotoxicity studies are included.
For the ingredients that are not identified as common dietary substances, that is, the monosaccharides fucose, galacturonic acid, and mannose and the disaccharides galactosyl fructose, kefiran, lactulose, melibiose, and xylobiose; a search for oral toxicity data was performed. However, very little published data were found.
Single Dose (Acute) Toxicity
Dermal
Lactitol
The dermal LD50 of lactitol in rabbits is >4500 mg/kg bw. 20
Oral
Lactulose
The oral LD50 of lactulose is 48.8 mL/kg in mice and >30 mL/kg in rats. 21
Repeated Dose Toxicity
Oral
Lactulose
Groups of 8 male albino rats were fed a diet containing 0.0%, 0.5%, 1.0%, 2.0%, or 5.0% (equivalent to 0.0, 1.1, 2.2, 4.0, and 11.3 g/kg bw/d, respectively) of a 50% lactulose syrup for 21 weeks. 64 None of the animals died during the study, and no signs of general toxicity were observed. Mild diarrhea was reported for animals fed >2.2 g/kg bw/d of the test material; diarrhea subsides with 3 to 5 hours of feeding. Feed consumption was not statistically significantly affected at any dose level. The organ weights were similar for treated and control animals. A statistically significant increase in cecal weights in the 2% and 5% groups was considered an adaptive reaction. No toxicologically significant changes in hematology, clinical chemistry, or urinalysis parameters were reported.
Ocular Irritation
In Vitro
Gluconic acid
The ocular irritation potential of a 50% aqueous solution of gluconic acid was evaluated in vitro in enucleated rabbit eyes. 9 The test material was applied to 4 eyes and observed over a period of 4 hours following application. Slight corneal swelling and slight permeability of the superficial epithelial cells were not considered to be of any toxicological significance.
Isomalt
A battery of in vitro tests were performed to determine the ocular irritation potential of isomalt; based on the overall results of each test included in the battery, isomalt was classified as a nonirritant. A neutral red uptake assay was performed in human keratinocytes, and the cytotoxicity of undiluted isomalt to the cells was measured after 24-hour exposure. 65 Two experiments were performed. Undiluted isomalt was classified as a nonirritant in this in vitro test.
A red blood cell (RBC) lysis and denaturation assay, comprised of 2 range-finding and denaturation assays and 2 lysis assays, was performed in calf RBCs. 66 Concentrations of ≤100 000 mg/L isomalt were tested. Isomalt did not induce hemolysis or protein denaturation and was classified as a nonirritant. Based on the lack of induction of hemolysis, the predicted in vivo ocular irritation potential corresponded to a modified maximum average score of 0.
The third test in the battery was the hen’s egg test on the chorioallantoic membrane in which isomalt was tested undiluted according to the end point assessment and at concentrations of 10% and 50% (w/w) in water according to the reaction-time method. 67 Each aspect of the experiment was performed twice. According to the European Cosmetic Toiletry and Perfumery Association (COLIPA; now, Cosmetics Europe) classifications, undiluted isomalt was classified as a slight irritant when tested undiluted in the endpoint assessment; the 10% and 50% concentrations were classified as nonirritant using the reaction-time method.
In vivo—nonhuman
Gluconic acid
A 50% aqueous solution of gluconic acid was not irritating to rabbit eyes. 9 A 50% solution of gluconic acid (pH 1.8; 0.1 mL) was instilled into the conjunctival sac of 1 eye in 9 New Zealand white rabbits; the contralateral eye served as an untreated control. The eyes of 3 animals were rinsed after 2 seconds, and of another 3 animals after 4 seconds; the eyes of the remaining 3 animals were not rinsed. The eyes were examined for irritation 1, 24, 48, and 72 hours and 7 days after instillation. Slight redness and conjunctival swelling were observed initially; however, no signs of irritation were observed after 72 hours.
Lactitol
Lactitol was not irritating to rabbit eyes. 20 The study was performed according to the Organization for Economic Co-operation and Development Test Guideline 405. 68 No other details were provided.
In vivo—human
Lactose
A face and neck formulation containing 2.48% lactose did not produce irritation or hypersensitivity in a 4-week safety-in use ophthalmological evaluation. 69 Thirty-one participants participated in the study.
Reproductive and Developmental Toxicity
According to the package insert for the prescription drug lactulose in studies of mice, rats, and rabbits, doses of lactulose solution up to 6 or 12 mL/kg/d produced no deleterious effects on breeding, conception, or parturition. 21 (Details were not provided.)
Genotoxicity
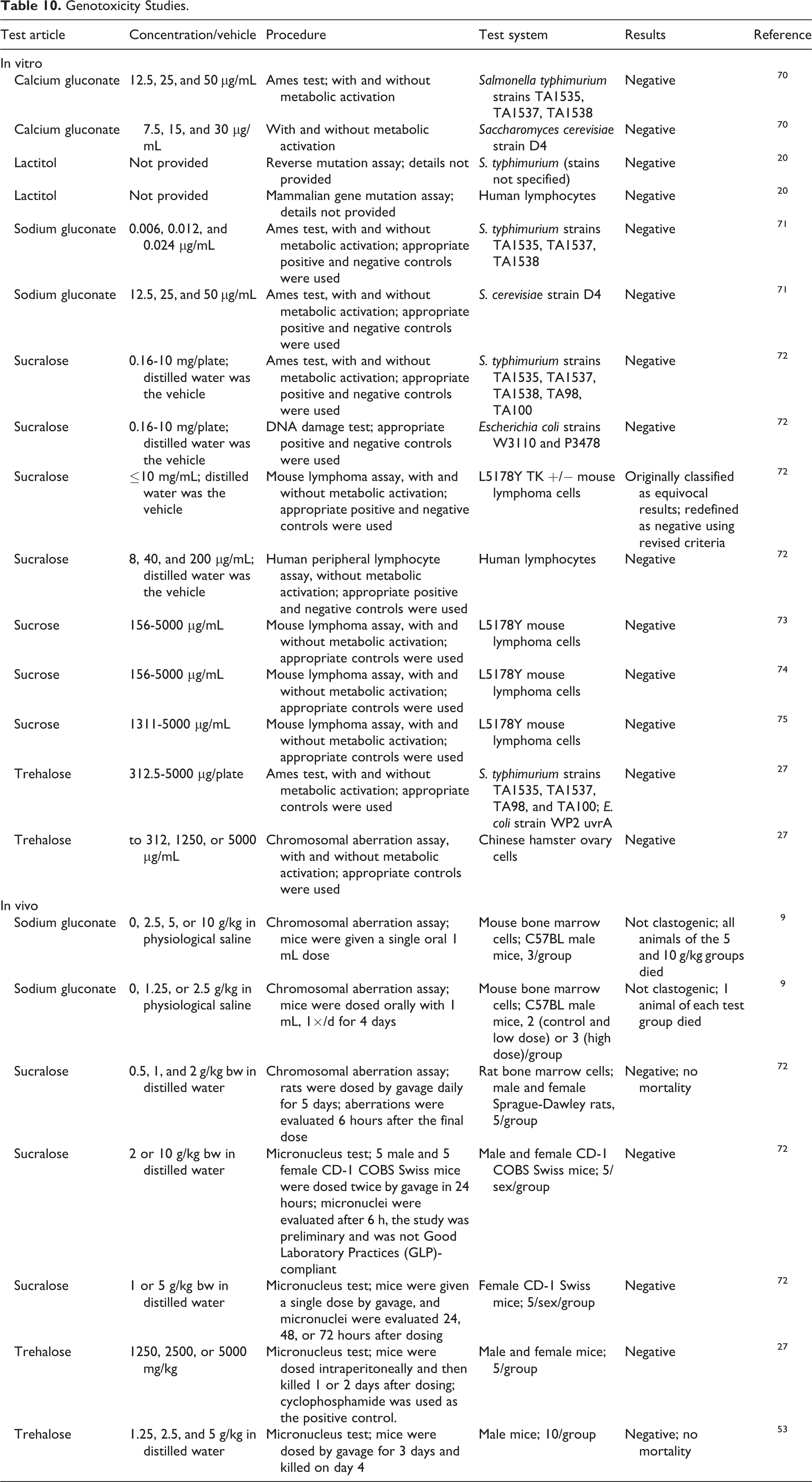
The genotoxicity of a number of the monosaccharides and disaccharides has been evaluated in in vitro and in vivo studies. The results of these studies are overwhelmingly negative (Table 10 9,20,27,53,70 –75 ).
Genotoxicity Studies.
Carcinogenicity
According to the package insert for the prescription drug lactulose, administration of lactulose solution in the diet of mice for 18 months at concentrations of 3% and 10% (v/w) did not produce any evidence of carcinogenicity. 21 (Details were not provided.)
Irritation and Sensitization
Dermal Irritation/Sensitization
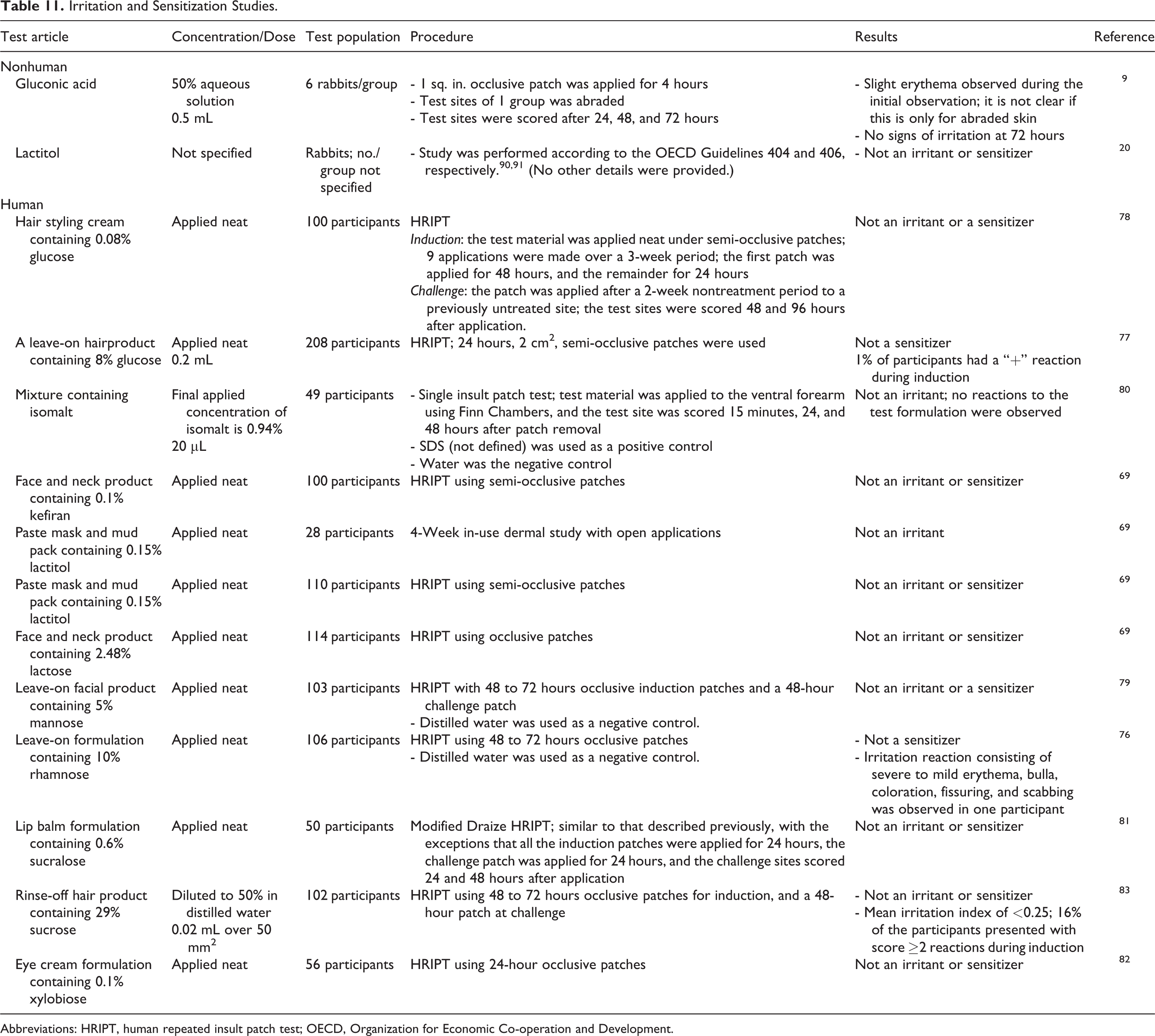
Dermal irritation and sensitization studies are summarized in Table 11. In nonhuman studies, a 50% aqueous solution of gluconic acid was not a dermal irritant 9 and lactitol was not an irritant or sensitizer in rabbits. 20 In human repeated insult patch tests (HRIPTs), formulations containing 10% rhamnose, 76 up to 8% glucose, 77,78 5% mannose, 79 2.48% lactose, 69 and <1% isomalt, 80 kefiran, 69 lactitol, 69 sucralose, 81 and xylobiose 82 were not irritants or sensitizers. A formulation containing 10% rhamnose did induce a significant irritation reaction in 1 participant, 76 and irritation was observed in 16% of the participants during induction in an HRIPT of a rinse-off hair product containing 29% sucrose (tested as a 50% dilution); no sensitization reactions were reported for this product. 83
Irritation and Sensitization Studies.
Abbreviations: HRIPT, human repeated insult patch test; OECD, Organization for Economic Co-operation and Development.
Occupational Exposure Limits
Sucrose
The National Institute for Occupational Safety and Health recommended exposure limit for sucrose is a time-weighted average (TWA) of 10 mg/m3 (total exposure) and TWA of 5 mg/m3 (respiratory exposure).25 The Occupational Safety and Health Administration permissible exposure limit (PEL) is a TWA of 15 mg/m3 (total) and TWA of 5 mg/m3 (respiratory). The American Conference of Governmental Industrial Hygienists threshold limit value is 10 mg/m3 as TWA; it is in category A4, not classifiable as a human carcinogen.
Summary
This report addresses the safety of 25 monosaccharides, disaccharides, and related ingredients as used in cosmetics. Many of these ingredients are GRAS food substances, direct food additives, or common dietary sugars, dietary sugar replacements, or very closely related analogs; for these ingredients, the primary focus of this review was on local effect, such as dermal irritation and sensitization. For the ingredients that are not identified as dietary substances, oral toxicity data were also searched.
The monosaccharides, disaccharides, and related ingredients are reported to have a number of functions in cosmetics, and the most common function is as a skin-conditioning agent; use as a humectant or flavoring agent was also common. According to VCRP data obtained from the FDA and concentration of use data obtained by the Council, 22 of the 25 ingredients reviewed in this assessment are reported to be in use. Sucrose has the greatest number of reported uses, 738, and glucose has the highest reported use concentration, 97.8% in an ingested breath freshener and 91% in “other” hair coloring products. The number of uses and maximum concentration of use vary widely by ingredient and type of use; most of the ingredients are used in leave-on products at less than 1%. Noncosmetic uses include food use and use as inactive ingredients in approved drugs.
Although many of the ingredients included in this safety assessment are food ingredients, they are not all processed by the body in the same manner; some (eg, glucose) are sources of energy and others (eg, sucralose) are not. Also, absorption is not the same for each of these ingredients; some are absorbed in the intestines (eg, glucose and potassium gluconate), whereas others are not absorbed in the gut (eg, lactitol and sucralose).
In an in vitro study, the permeability coefficient of glucose was 9.5 × 10−5 cm/h through full thickness nude mouse skin and 0.29 cm/h through the dermis (only) of nude mouse skin. In an in vivo study with Rhesus monkeys, using OCT, the mean permeability rate of 20% glucose was calculated to be (4.41 ± 0.28) ×10−6 cm/s.
Lactulose fed to rats at concentrations of up to 5.0% of 50% lactulose syrup for 21 weeks did not result in toxicity. Mild diarrhea was reported for animals fed >2.2 g/kg bw/d of the test material; diarrhea subsides with 3 to 5 hours of feeding. Doses of up to 12 mL/kg/d of lactulose solution produced no deleterious effects on breeding, conception, or parturition in mice, rats, or rabbits. No evidence of carcinogenicity was observed in mice with dosing of up to 10% lactulose solution in the diet for 18 months.
A battery of in vitro tests were performed to determine the ocular irritation potential of isomalt; based on the results, isomalt was classified as a nonirritant. Gluconic acid, as a 50% aqueous solution, and lactitol, concentration not specified, were not irritating to rabbit eyes. A face and neck formulation containing 2.48% lactose did not produce irritation or hypersensitivity in a 4-week safety-in use ophthalmological evaluation
In nonhuman studies, a 50% aqueous solution of gluconic acid was not a dermal irritant and lactitol was not an irritant or sensitizer in rabbits. In HRIPTs, formulations containing 10% rhamnose, 8% glucose, 5% mannose, 2.48% lactose, and <1% isomalt, kefiran, lactitol, sucralose, and xylobiose were not irritants or sensitizers. A formulation containing 10% rhamnose did induce a significant irritation reaction in 1 participant, and irritation was observed in 16% of the participants during induction in an HRIPT of a product containing 29% sucrose (that was a rinse-off hair product tested as a 50% dilution); no sensitization reactions were reported for this product.
Lactitol, sodium gluconate, sucralose, sucrose, and trehalose were not genotoxic in vitro. Additionally, the genotoxic potential of sodium gluconate, sucralose, and trehalose was evaluated in vivo; again negative results were obtained.
Discussion
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed this safety assessment of monosaccharides, disaccharides, and related ingredients. Most of these ingredients are common dietary sugars, dietary sugar replacements, or very closely related analogs and salts. Several are GRAS food additives, direct food additives, listed in the Food Chemicals Codex as used in foods, and/or listed in REACH Annex IV. Because the oral safety of these ingredients has been well documented, systemic toxicity is not a concern of the Panel.
Some of the ingredients, however, are not GRAS food substances or direct food additives; even so, these ingredients are either listed in the Food Chemicals Codex as having a function in foods, listed in the Everything Added to Foods in the United States inventory, and/or listed as an inactive ingredient in oral drugs. Moreover, the leave-on use concentrations of these ingredients are typically <1%. Therefore, the Panel stated that although oral toxicity data are very limited and reproductive toxicity data are mostly absent, the systemic toxicity of these ingredients was not a concern because of the low concentrations of use and their limited systemic exposure from dermal application.
The Panel commented that sucrose is used at high concentrations in some products that come in contact with mucous membranes (ie, 65% in personal cleanliness products). The Panel noted that sucrose is a GRAS food substance, and therefore, the Panel was not concerned about this reported use. Additionally, the Panel observed that glucose is reported to be used at 97.8% in an ingestible oral hygiene product but recognized that glucose is a GRAS direct food additive with no limitations other than following current good manufacturing practice. However, if an ingredient that does not have GRAS food additive status was used at concentrations such as these with similar exposure types, the Panel would most likely want data substantiating the safety of that use, such as metabolism after oral administration.
The Panel discussed an HRIPT of a hair product that contained 29% sucrose, diluted to 50%, which reported irritation during induction. The Panel concluded that the irritation reported was likely attributable to a surfactant effect and was not due to sucrose itself. The Panel acknowledged that sucrose and glucose are used in cosmetics at relatively high concentrations and that data from irritation and sensitization studies at maximum use concentrations of these ingredients are lacking; however, based on the clinical experience of the Panel, there is little concern that these ingredients are irritants or sensitizers.
Because some of the ingredients included in this safety assessment can be used in products that may be aerosolized, the Panel discussed the issue of incidental inhalation exposure. Most of the use concentrations of the ingredients used in cosmetic products that may be aerosolized are <1% (eg, glucose is used at 1% in a spray body and hand preparation). In the absence of inhalation data, the Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. The Panel acknowledged that the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs, but because of the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Finally, because many of these ingredients are obtained from plant sources, the Expert Panel expressed concern regarding pesticide residues and heavy metals that may be present. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
Conclusion
The CIR Expert Panel concluded that the following 25 monosaccharides, disaccharides, and related ingredients are safe in the present practices of use and concentration in cosmetics described in this safety assessment: Calcium gluconate Fructose Fucose Galactose* Galactosyl fructose Galacturonic acid* Gluconic acid Glucose Isomalt Kefiran Lactitol Lactose Lactulose* Maltose Mannose Melibiose Potassium gluconate Rhamnose Ribose Sodium gluconate Sucralose Sucrose Trehalose Xylobiose Xylose
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, Suite 1200, 1620L Street, NW, Washington, DC 20036, USA.
Author Contributions
Monice M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Ronald C. Shank, Thomas J. Slaga, Paul W. Snyder, and Lillian J. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Bart Heldreth contributed to analysis and interpretation and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
