Abstract
Introduction:
Based on the ICH S7B and E14 guidance documents, QT interval (QTc) is used as the primary in vivo biomarker to assess the risk of drug-induced torsades de pointes (TdP). Clinical and nonclinical data suggest that drugs that prolong the corrected QTc with balanced multiple ion channel inhibition (most importantly the
Methods:
Telemetry data from Beagle dogs given dofetilide (0.3 mg/kg), sotalol (32 mg/kg), and verapamil (30 mg/kg) orally and Cynomolgus monkeys given medetomidine (0.4 mg/kg) orally were retrospectively analyzed for effects on QTca, JTpca, and T-peak to T-end covariate adjusted (Tpeca) interval using individual rate correction and super intervals (calculated from 0-6, 6-12, 12-18, and 18-24 hours postdose).
Results:
Dofetilide and cisapride (IKr or Kv11.1 blockers) were associated with significant increases in QTca and JTpca, while sotalol was associated with significant increases in QTca, JTpca, and Tpeca. Verapamil (a Kv11.1 and Cav1.2 blocker) resulted in a reduction in QTca and JTpca, however, and increased Tpeca. Medetomidine was associated with a reduction in Tpeca and increase in JTpca.
Discussion:
Results from this limited retrospective electrocardiogram analysis suggest that JTpca and Tpeca may discriminate selective IKr blockers and multichannel blockers and could be considered in the context of an integrated comprehensive proarrhythmic risk assessment.
Introduction
Clinical and nonclinical guidelines (ie, ICH E14 and ICH S7B) that outline nonclinical drug proarrhythmia evaluation currently suggest the use of QT interval (QTc) as a primary in vivo biomarker to assess the proarrhythmic risk for development of drug-induced torsades de pointes (TdP) arrhythmias. 1 This approach has been highly successful from a safety perspective, as no newly marketed drugs have been associated with an unacceptable risk of TdP since those guidelines were implemented. However, potentially safe and efficacious compounds may have been inadvertently halted in development due to the potential for QTc prolongation and perceived proarrhythmic risk resulting from blockade of the human Ether-à-go-go-Related Gene (hERG) channel, which encodes IKr. 2,3 The concern for drug development discontinuation of potentially safe and efficacious drugs encouraged the launch of the comprehensive in vitro proarrhythmia assay (CiPA) initiative, a new mechanistic approach to cardiac safety assessment of new drugs based on a more comprehensive understanding of the ionic currents that play a role in QTc prolongation and the development of TdP arrhythmias. 3 -5 Many approved drugs prolong the QTc interval but have been shown to present minimal risk for development of TdP. The class III (Vaughan Williams’ classificiation) antiarrhythmic drug amiodarone, a blocker of IKr but also late sodium and calcium currents, causes QTc prolongation correlated with clinical efficacy. 6 Verapamil and ranolazine are multi-ion channel blockers that also induce QTc prolongation but have not been associated with increased risk of TdP. 7,8 Clinical data have emerged to suggest that an analysis of the J to T-peak (JTp) interval can be considered to differentiate between selective IKr blockers associated with a high proarrhythmic risk and drugs with a balanced multi-ion channel inhibition profile. 5,9
The aim of this retrospective analysis was to evaluate the effects of several well-characterized drugs on the QTc, JTpc, and T-peak to T-end corrected (Tpec) electrocardiogram (ECG) intervals in relevant nonrodent safety pharmacology species and to compare the results with the clinical ECG responses observed with these same drugs. There is limited nonclinical ECG information available regarding drug effects on these intervals in nonclinical species and specifically the manner in which these novel ECG parameters change with drugs that block multiple cardiac ion channels. Thus, the rationale for using these additional ECG biomarkers is that IKr blockers are expected to prolong predominantly JTp, while drugs that block calcium and the late sodium currents are expected to shorten JTp. Dofetilide, sotalol, verapamil, and cisapride were selected for this proof-of-concept analysis based on their known ion channel inhibition profiles and clinically well-characterized ECG effects. Medetomidine was selected for evaluation of its potential unique effects on both the JTp and Tpe intervals. Medetomidine produces changes in body temperature which is known to affect the QTc interval; thus, an assessment of this physiological effect was evaluated in monkeys.
Materials and Methods
Test Facility and Experimental Animals
The data evaluated within this investigation were obtained retrospectively from members of the Health and Environmental Sciences Institute (HESI)-sponsored scientific consortium, and the analysis was undertaken by the Pro-arrhythmia subcommittee of the Cardiac Safety Technical Committee. 10 All in-life phases of the study from 4 institutions, from which the data were collected, were subject to local animal laboratory and welfare guidelines in terms of the vivarium conditions, study conduct, and animal use approval procedures. Male Beagle dogs or male cynomolgus monkeys were surgically instrumented with either a DSI Physiotel Digital L21, a D70-PCT telemetry system (DSI, St Paul, Minnesota) or an easyTEL L-CPTA-0100 (EMKA, Falls Church, Virginia) transmitter to monitor systolic arterial blood pressure, the ECG in a lead II (DII) configuration, body temperature, and activity.
Test Drugs
All drugs or appropriate vehicles were administered orally to telemetered animals in a crossover design. Since this is only a preliminary analysis of these drugs on these ECG measures, the data presented herein only include the vehicle control and high-dose groups of the tested drugs. This proof-of-concept investigation is the first potential application of drug-induced changes in the JTpc and Tpec intervals using standard safety pharmacology methods. The drugs selected to investigate these complementary proarrhythmia biomarkers on cardiac repolarization processes are well characterized in nonclinical, nonrodent species with recognized translation to clinical results. The 5 drugs (dofetilide, verapamil, sotalol, medetomidine, and cisapride) that are reported within this investigation are well characterized in the clinical setting (Table 1). The doses that were selected for the individual studies are in accordance with the ICH S7A and ICH S7B guidelines that suggest the highest doses used exceed the anticipated human exposure.
Drugs Tested for Effects on the QTc, JTpca, and Tpeca Intervals of the ECG.
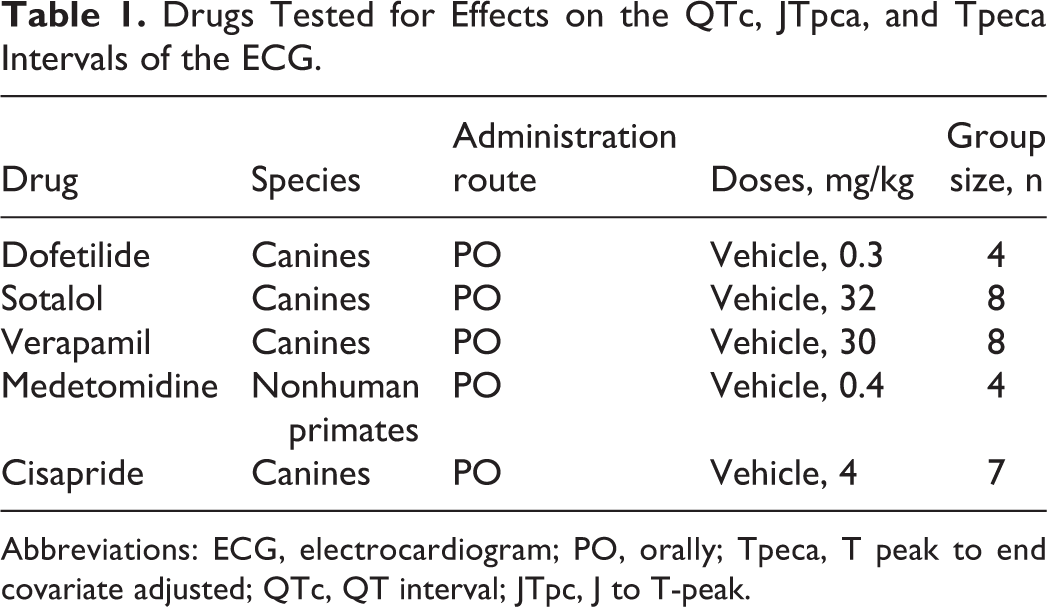
Abbreviations: ECG, electrocardiogram; PO, orally; Tpeca, T peak to end covariate adjusted; QTc, QT interval; JTpc, J to T-peak.
Data Collection and Retrospective Analysis
The ECG signals were continuously acquired from at least 1 hour prior to dosing through 24 hour postdose on each study day. Sampling rates were ≥500 Hz for ECG recordings. Derived parameters were automatically calculated by either ECGauto (EMKA), Ponemah (DSI) or Notocord ECG51a module (Notocord, Newark, New Jersey) data analysis software systems. Derived parameters included heart rate (HR) and the QT, JTp, and Tpe intervals. Although a large number of parameters were available, this article is limited to ECG parameters that are directly or indirectly relevant to the evaluation of cardiac ventricular repolarization. Data were analyzed by scientists unfamiliar with the drugs and doses and presented using Microsoft Excel (Microsoft Corporation, Redmond, Washington). Statistical analysis was undertaken using SAS version 9.2 with analysis of variance (ANOVA) for repeated measures on super intervals 0 to 6, 6 to 12, 12 to 18, and 18 to 24 hours postdose. These super intervals were selected based on expected exposure with oral administration of the selected drugs and included intervals when pharmacological effects were expected to resolve. No bioanalysis of drug exposure levels were available for this retrospective analysis. Inferential analysis was performed on QTca, JTpca, and Tpeca independently for each drug. For each time point, the individual difference between the high dose and control data was calculated. A repeated measure ANOVA was performed with time as a fixed effect. If the time effect was significant (P ≤ 0.05), a 2-sided t test on the least-square means of each time point was used. Otherwise, the 2-sided t test was performed on the least-square means pooled across all time points. Due to the short duration of effects with medetomidine, statistical analysis also included ANOVA for repeated measures for hourly means. Individual HR correction was calculated for QTc, JTpc, and Tpec as described previously. 11 Briefly, analysis of covariance was used to correct QT, JTp, and Tpe intervals for HR using 15-minute data bins. The resulting HR-corrected intervals were identified as QT interval corrected for HR based on analysis of covariance (QTca), J to T-peak interval corrected for HR based on analysis of covariance (JTpca), and T-peak to T-end interval corrected for HR based on analysis of covariance (Tpeca). The baseline ECG, obtained prior to treatment, was used to determine the appropriate correction factors. Baseline QT, JTp, or Tpe (uncorrected) and HR data were used to estimate coefficients in the linear regression: log (JTp or Tpe) = α + β log(HR). The estimated slopes (β) from the baseline data (ie, period of up to 24 hours prior to dosing) were used to rate correctly the JTp or Tpe intervals in the formula: log (JTp or Tpe)ca = log(JTp or Tpe) – β × [log(HR – log(HRm))], in which HRm was the mean heart rate from the baseline period.
Results
All the ECG parameters (ie, QT, JTp, and Tpe) from untreated animals were negatively correlated with HR changes (Figure 1) in both Beagle dogs and cynomolgus monkeys (QTca, JTpca, and Tpeca). The use of individual rate-correction methods minimized the changes associated with HR variation (Figure 1).
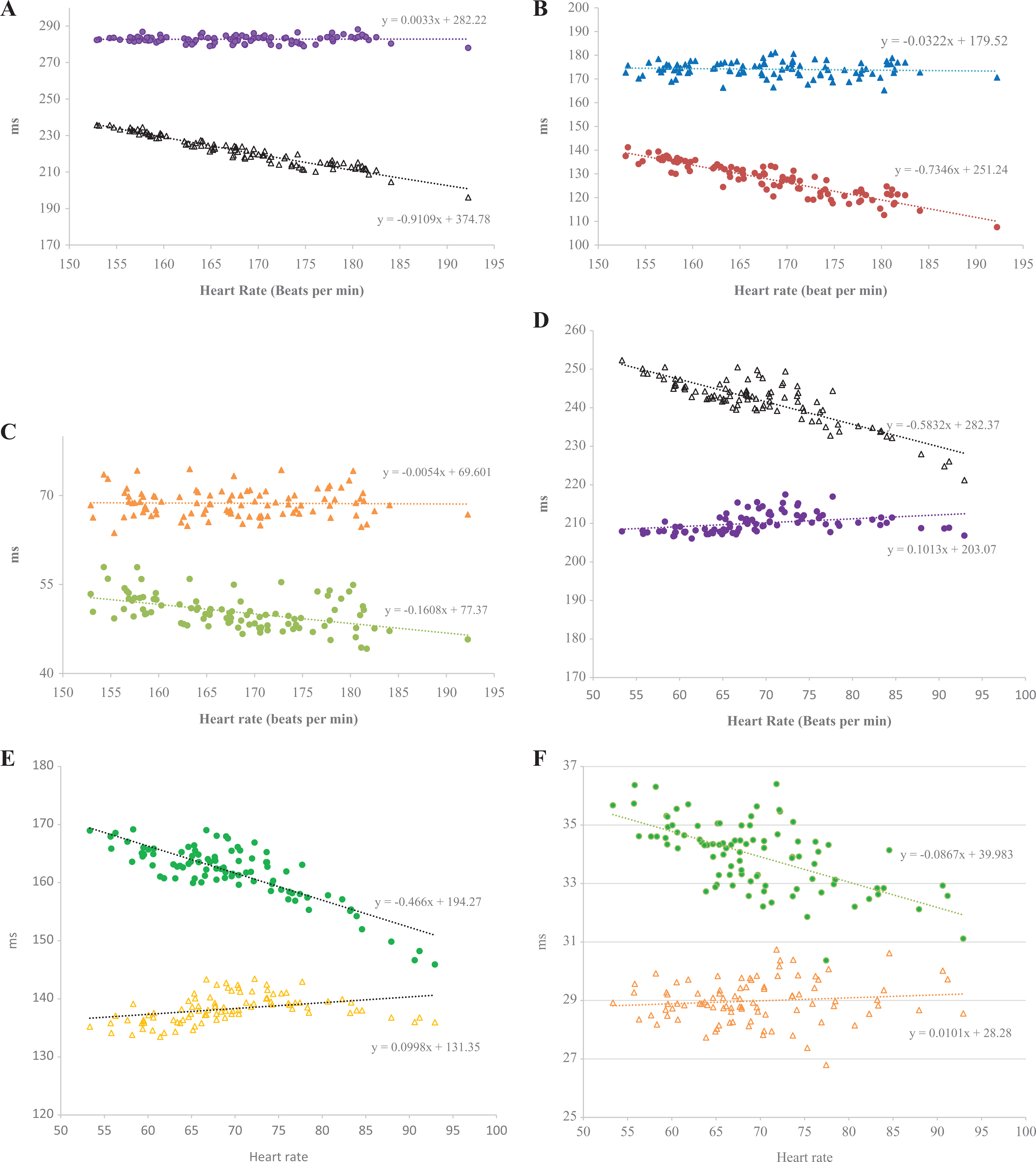
A, The uncorrected QT (black open triangles) and heart rate-corrected QTca (purple circles) intervals as a function of heart rate in conscious untreated cynomolgus monkeys. The 24-hour data are presented as hourly means (n = 4). B, The uncorrected J to T-peak (JTp; red circles) and heart rate-corrected J to T-peak interval corrected for heart rate based on analysis of covariance (JTPca; blue triangles) intervals as a function of heart rate in conscious untreated cynomolgus monkeys. The 24-hour data are presented as hourly means (n = 4). C, The uncorrected T-peak to end interval (Tpe; green circles) and individually rate-corrected T-peak to end interval corrected for heart rate based on analysis of covariance (Tpeca; orange triangles) intervals as a function of heart rate in conscious untreated cynomolgus monkeys. Data are presented as hourly means (n = 4). D, The uncorrected QT (Black open triangles) and heart rate-corrected QT interval-corrected for heart rate based on analysis of covariance (QTca; purple circles) intervals as a function of heart rate in conscious untreated Beagle dogs. The 24-hour data are presented as hourly means (n = 4). E, The uncorrected JTp (green circles) and individual rate-corrected JTpca (orange open triangles) values as a function of heart rate in conscious untreated Beagle dogs. Data are presented as hourly means (n = 4). F, The uncorrected Tpe (green circles) and individually rate-corrected Tpeca (orange open triangles) intervals as a function of heart rate in conscious untreated Beagle dogs. Data are presented as hourly means (n = 4).
Dofetilide
Dofetilide (0.3 mg/kg, po) was associated with a significant increase in the QTca interval (the maximum effect was +18%) for the super intervals ranging from 0 to 18 hours postdose (Figure 2) in Beagle dogs. The JTpca was also significantly increased from 0 to 12 hours postdose (the maximum effect was +18%). No significant change in Tpeca was observed over any interval examined.
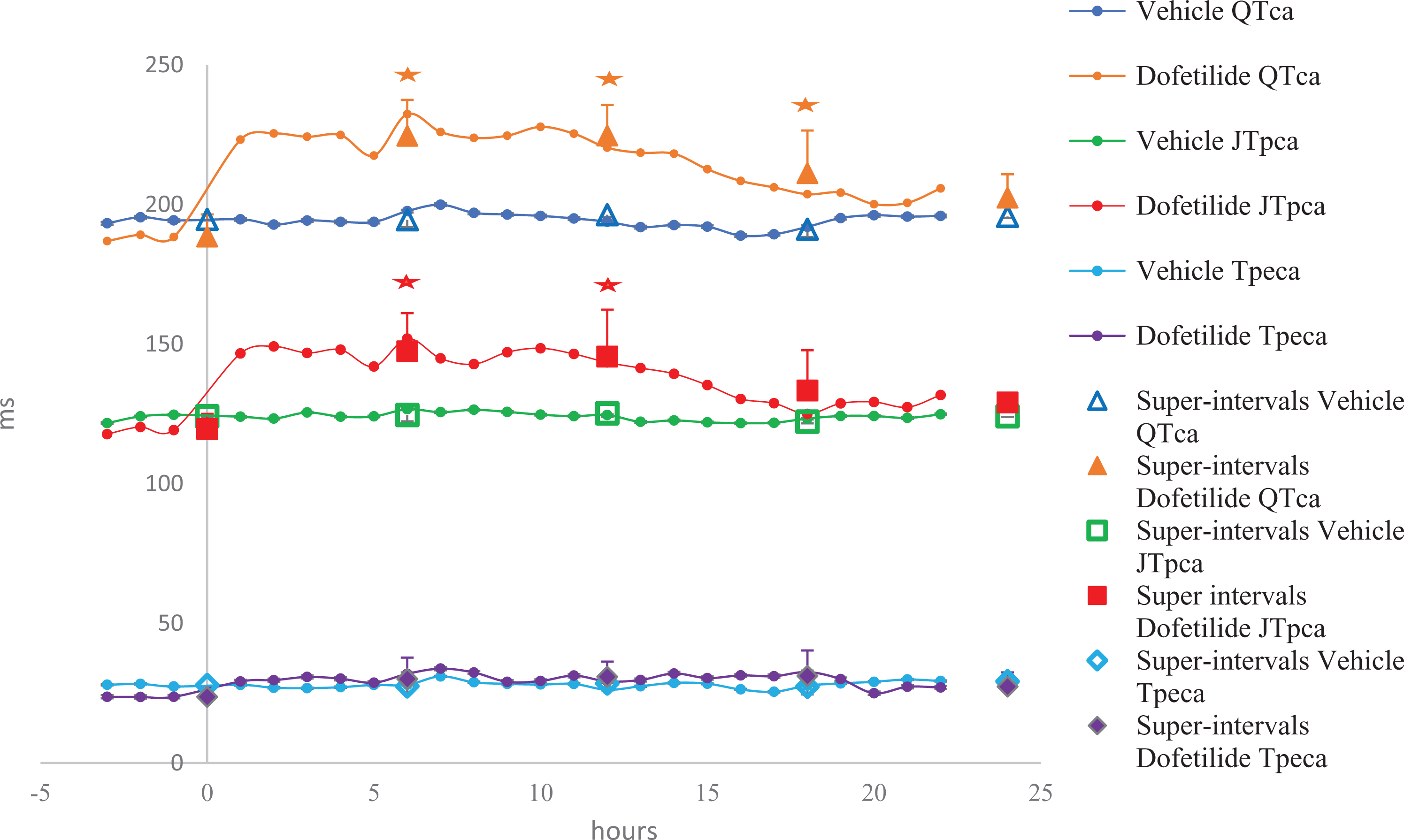
The effects of dofetilide (0.3 mg/kg, po) recorded over 24 hours in telemetered Beagle dogs (n = 4); *P < 0.05 with standard deviation.
Sotalol
Sotalol (32 mg/kg, po) produced an increase in the QTca interval (the maximum effect was +11%) and concommitant increase in the JTpca interval (the maximum effect was +11%) at all time points that were evaluated from 0 to 24 hours postdose (Figure 3) in Beagle dogs. The Tpeca interval was also significantly prolonged (the peak effect was +28%) from 0 to 18 hours postdose.
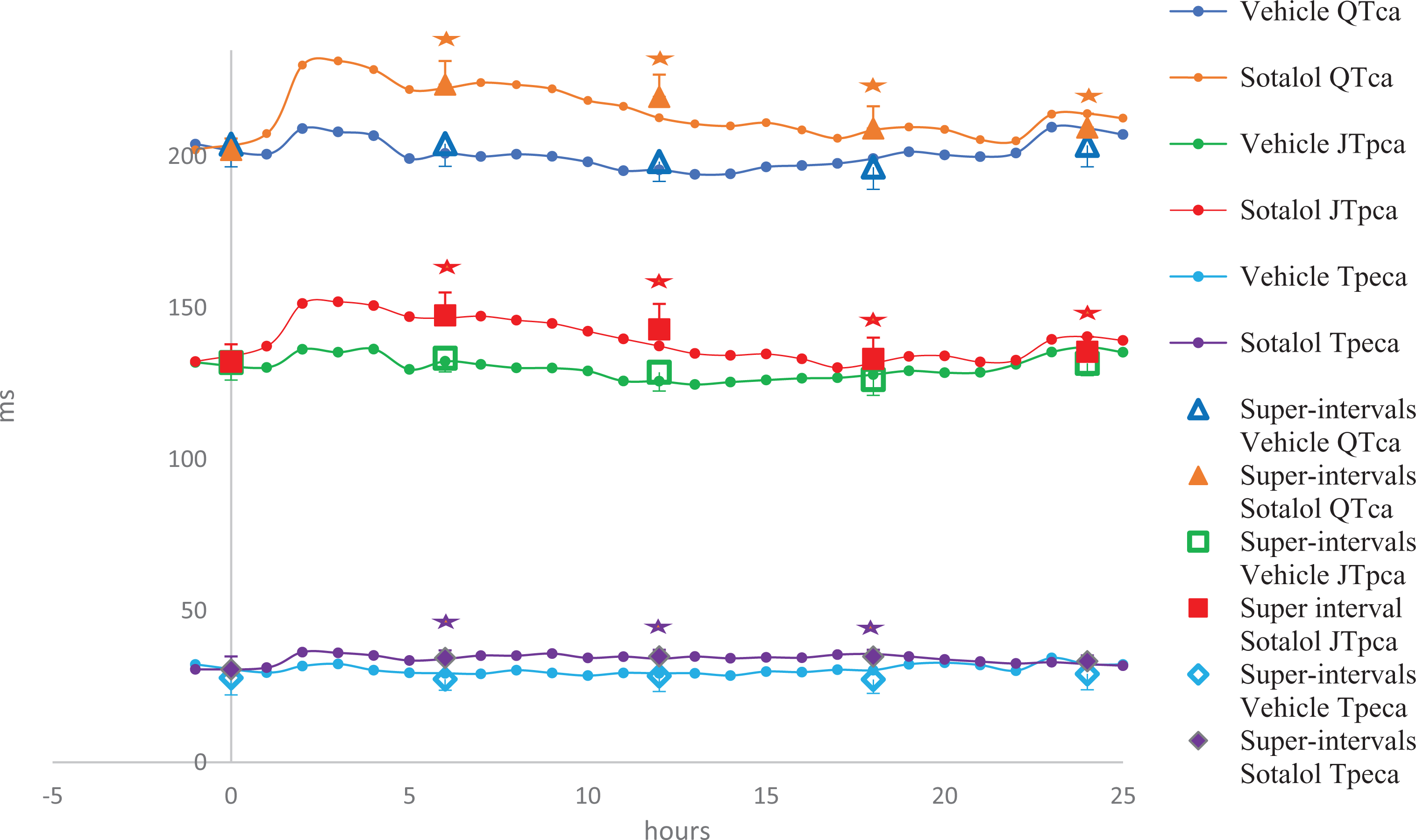
The effects of sotalol (32 mg/kg, PO) recorded over 24 hours in telemetered Beagle dogs (n = 8); *P < 0.05 with standard deviation.
Verapamil
Verapamil (30 mg/kg, po) in Beagle dogs produced a significant decrease in the QTca interval (the maximum effect was −4%) and also in the JTpca interval (the maximum effect was −12%). However, the Tpeca interval was prolonged (the maximum effect was +29%) at the super interval 0 to 6 hours postdose followed by a subsequent recovery to comparable vehicle values (Figure 4).
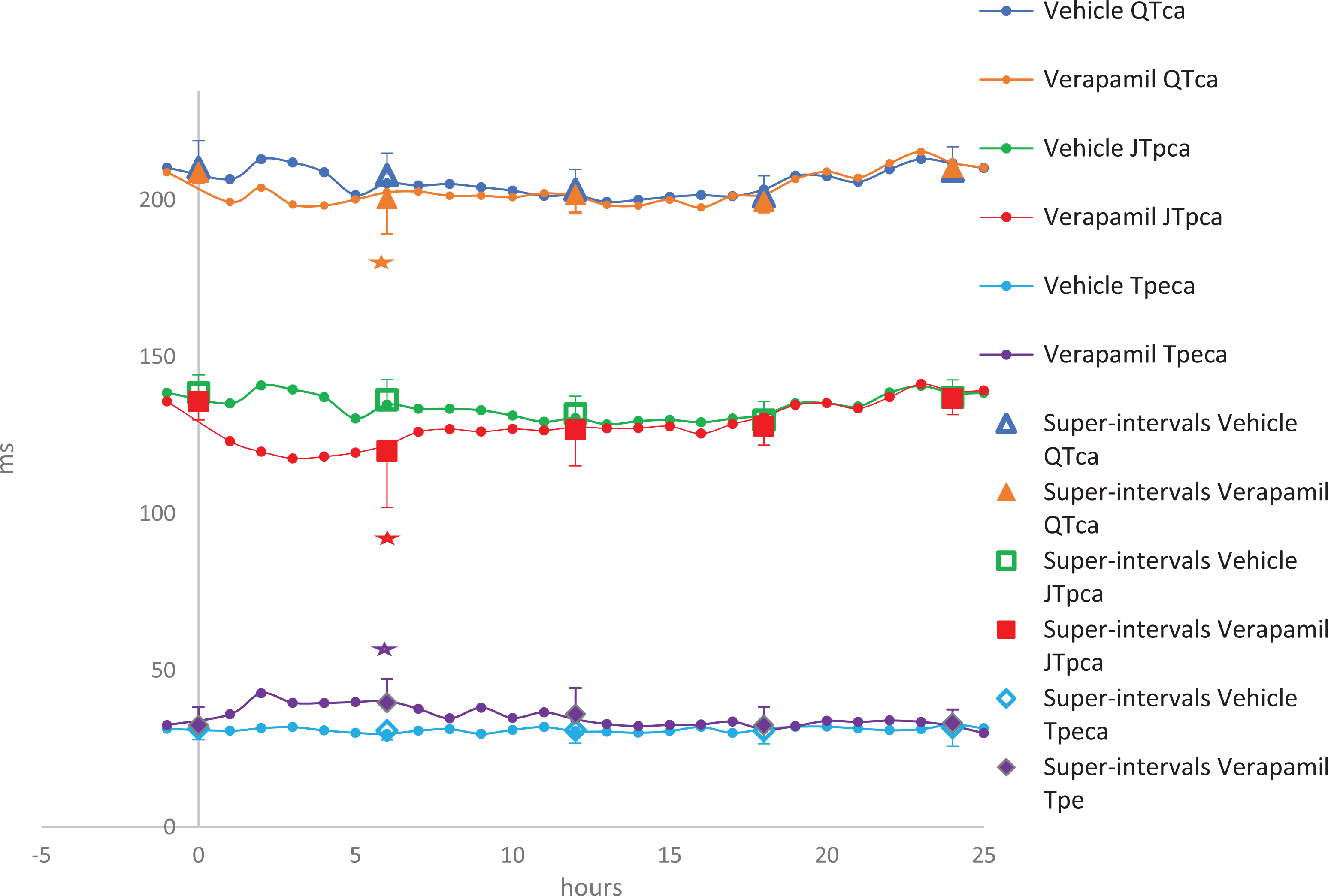
The effects of verapamil (30 mg/kg, PO) recorded over 24 hours in telemetered Beagle dogs (n = 8); *P < 0.05 with standard deviation.
Medetomidine
Medetomidine (0.4 mg/kg, po) in cynomolgus monkeys had no significant effect on the QTca interval but was associated with a significant reduction in the Tpeca interval combined with a trend toward increased JTpca values over the same time points when assessing the super interval data (Figure 5). Statistical analysis performed on hourly means revealed a significant increase in JTpca from 1 to 7 hours postdose, with an increase in Tpeca that reached statistical significance between 1 to 10 hours and 12 to 15 hours postdose. Medetomidine also produced a significant reduction in animal body temperature up to −1°C compared to vehicle control and with a temporal correlation to the observed ECG changes.
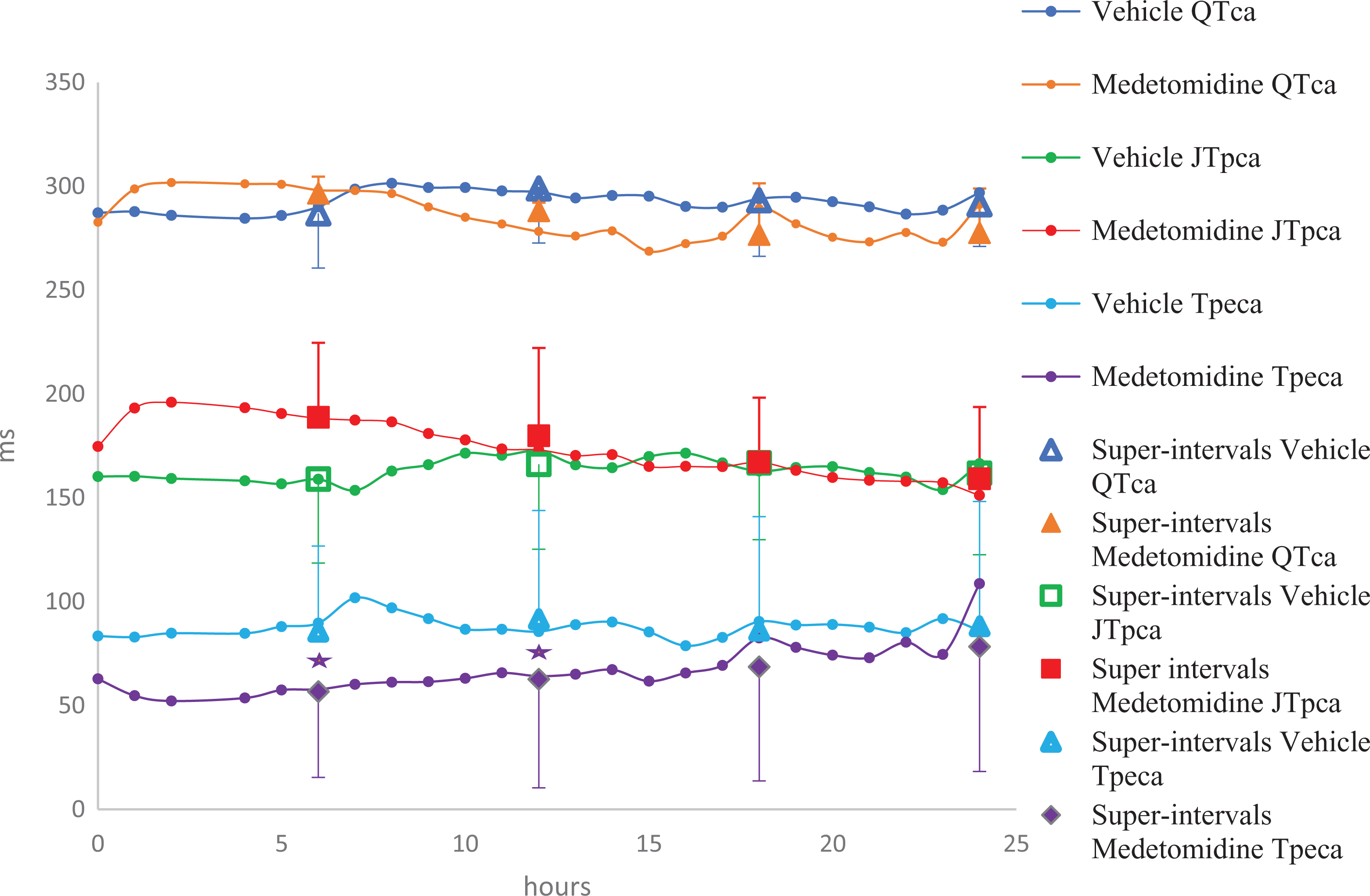
Effects of medetomidine (0.4 mg/kg, PO) recorded over 24 hours in telemetered cynomolgus monkeys (n = 4); *P < 0.05 with standard deviation. A statistically significant decrease in J to T-peak interval corrected for heart rate based on analysis of covariance (JTPca) occurred for hourly means from 1 to 7 hours postdose when compared between vehicle and treated.
Cisapride
Oral administration of cisapride (4 mg/kg) to Beagle dogs produced no significant effect on the JTpca interval; however, a significant increase in QTca was noted from 3 to 5 hours postdose correlated with an increase in JTpca achieving statistical significance at 4 and 5 hours postdose (the maximum effect was +17%; Figure 6).
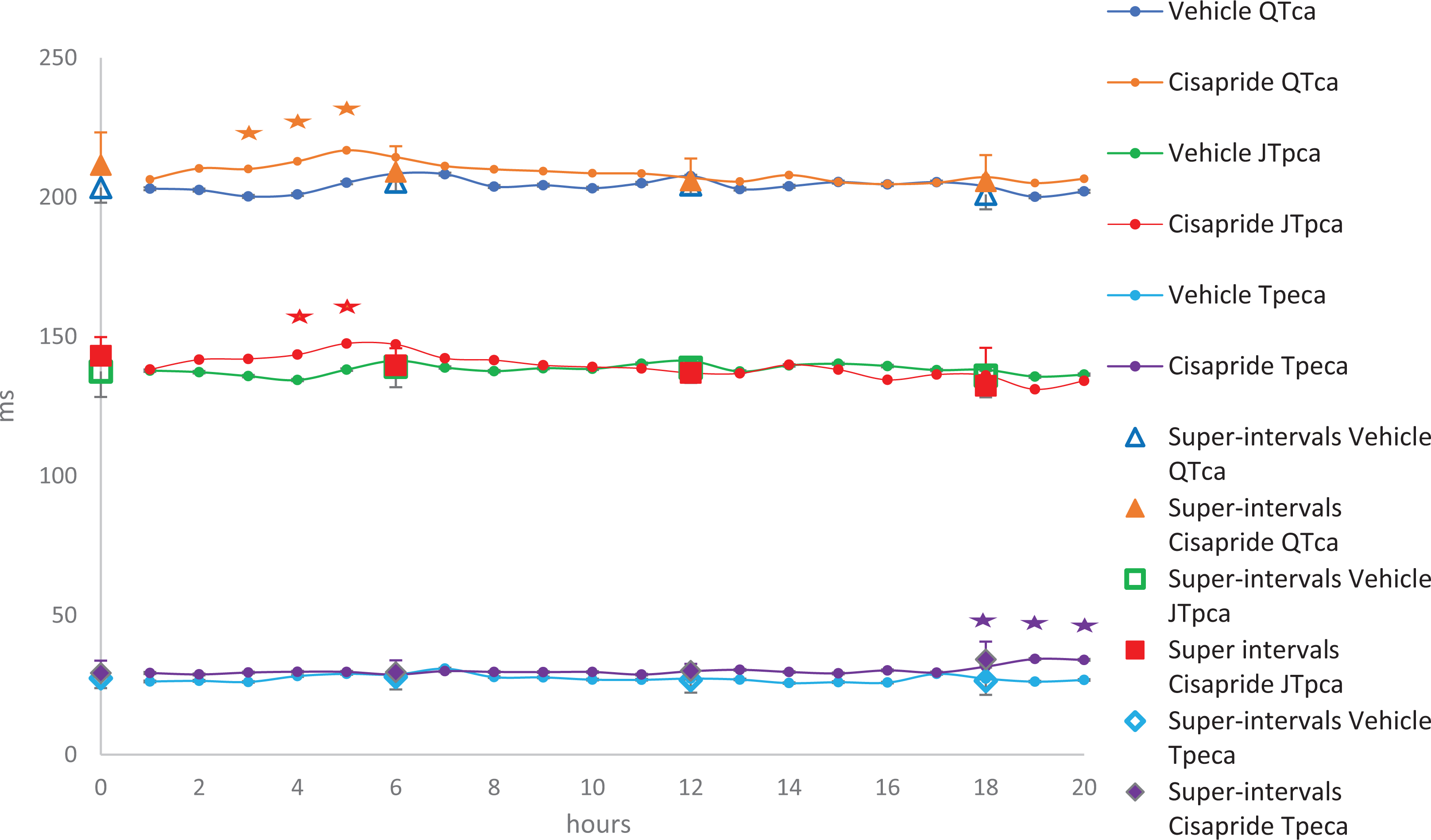
Effects of cisapride (4 mg/kg, PO) recorded over 20 hours in telemetered Beagle dogs (n = 7); *P < 0.05 with standard deviation. A statistically significant increase in QT interval-corrected for heart rate based on analysis of covariance (QTca) occurred for hourly means from 3 to 5 hours postdose and for J to T-peak interval corrected for heart rate based on analysis of covariance (JTPca) from 4 to 5 hours when compared to vehicle. Statistical significance was also noted achieved from 18 to 20 hours, but this change was considered incidental.
Discussion
Results from the current retrospective data analysis of dofetilide, verapamil, sotalol, medetomidine, and cisapride suggest that drug-mediated pharmacological effects on cardiac ventricular repolarization appear to be characterized by changes in not only the QTca interval but also the novel JTpca and Tpeca intervals in relevant nonrodent species. Although it is recognized that the ECG remains an imperfect modality for cardiac electrophysiology monitoring in nonclinical studies owing to some limitations associated with the use of a single biopotential vector with only the conventional derivation of a Lead II configuration, it is the methodological standard. The predictive value of these novel ECG biomarkers to complement changes in the QTc interval and assist in the assessment of the risk of drug-induced TdP remains a central question that requires further investigation. The Tpe interval has been previously considered an index of transmural dispersion of repolarization, since it is important for the genesis of the T wave and induction of arrhythmias. 12 The JTp and Tpe from anesthetized dogs were previously considered to assess inward and outward current modifications, 13 but overall, limited data are available from conscious nonclinical models.
Heart rate correction of ECG parameters has a long clinical and nonclinical history, 14 -16 with standard approaches that suffer from substantial intersubject variability in healthy volunteers. 17 In nonclinical studies, the QT–HR relationship has been characterized in canines, 18 minipigs, guinea pigs, and nonhuman primates. 19 The use of individual correction methods has been considered in nonclinical studies for more than 2 decades. 20 -22 The preliminary analysis undertaken herein suggests that the JTp and Tpe intervals, similar to the QT interval, are negatively correlated with HR in Beagle dogs and cynomolgus monkeys. Clinical study results have not discussed a significant heart rate dependence for Tpe, and the current study was not powered to definitively test whether the slopes that were identified in canines and nonhuman primates were significantly different from zero. The individual correction methods using an analysis of covariance developed for JTpca and Tpeca resulted in consistent values with changing HR that allowed for the interpretation of potential dug-mediated pharmacological effects. The results presented herein suggest that the additional ECG parameters investigated, and in particular Tpe, occurred with a high degree of variability in cynomolgus monkeys.
The use of super intervals has been proposed to reduce the high degree of natural variability in cardiovascular parameters obtained from canines.
23
Use of this methodology was applied to the current study dataset. Similar to clinical findings,
24
-27
the IKr blockers evaluated dofetilide, sotalol, and cisapride significantly increased JTpca in Beagle dogs. However, in contrast to the clinical data with dofetilide,
26
the Tpe interval was not significantly changed in the canine model with this agent which highlights the potential for different results between clinical and nonclinical studies. Despite the differences between clinical and nonclinical data, the risk assessment based on a JTp prolongation for these drugs supports the translational potential of these biomarkers. Verapamil, which also inhibits IKr, is a more potent blocker of the cardiac
Nonclinical studies require elements of drug safety assessments based on the ICH M3(R2), ICH S7A, and ICH S7B regulatory guidance’s which provide the opportunity to evaluate drug-mediated ECG effects 20 in an integrated in vivo model prior to the conduct of human phase 1 clinical trials. Utilization of the JTpca and Tpeca intervals in relevant nonclinical species may provide supplemental data to drug effects on the QT interval. Also, these additional ECG measures may be important additions in the context of the CiPA initiative, since they could assist in providing in vivo findings to complement in vitro and in silico CiPA data. The CiPA paradigm is currently being considered as a mechanistic approach to cardiac safety testing based on a thorough characterization of drug-induced blockade of cardiac ion channels primarily using ion channel testing, as well as effects in human stem cells in conjunction with in silico modeling of the cardiac action potential and changes in the ECG morphology. 4,32,33,34 Several comprehensive studies recently conducted 35,36,32 report that QT/QTc interval data derived from relevant nonrodent models have good (>80%) predictive value toward QT findings in humans. However, further nonclinical investigation with additional positive and negative control drugs will be needed to fully define the predictive value, sensitivity, and specificity of the JTpca and Tpeca intervals obtained from nonclinical studies in proarrhythmia assessments.
Footnotes
Authors’ Notes
All companies represented are members of the HESI Cardiac Safety Technical Committee.
Acknowledgments
The authors wish to thank HESI and partner institutions for allowing access to the data that were used for this analysis and also Ana Gueorguieva for statistical analysis.
Author Contributions
Emmanuel Boulay contributed to acquisition and analysis and drafted the manuscript; Matthew M. Abernathy contributed to conception and design and critically revised the manuscript; Ray Chui contributed to conception and design, contributed to acquisition and interpretation, and critically revised the manuscript; Gregory S. Friedrichs contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript; Nicolas Gendron-Wilke contributed to conception and design and critically revised the manuscript; Andrea Greiter-Wilke contributed to conception and design and critically revised the manuscript; Jean-Michel Guillon contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript; John E. Koerner contributed to conception and design and critically revised the manuscript; Ariane Menard contributed to analysis and critically revised the manuscript; Jill Steidl-Nichols contributed to conception and design and critically revised the manuscript; Jennifer Pierson contributed to conception and design, contributed to interpretation, and critically revised the manuscript; Michael K. Pugsley contributed to conception and design, contributed to interpretation, and critically revised the manuscript; Eric I. Rossman contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript; David Strauss contributed to analysis and critically revised the manuscript; Eric Troncy contributed to analysis and critically revised the manuscript; Jean-Pierre Valentin contributed to conception and design and critically revised the manuscript; Todd Wisialowski contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript; Simon Authier contributed to conception and design, contributed to acquisition, analysis, and interpretation and analysis, and drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
