Abstract
Reestablishment of fertility, after a male contraceptive method, is of great concern. In this context, RISUG (Reversible Inhibition of Sperm Under Guidance) has been evaluated for its mutagenicity following reversibility with dimethyl sulfoxide (DMSO)/sodium bicarbonate (NaHCO3) in Wistar albino rats. Animals were divided into 7 groups, namely, sham-operated control, vas occlusion with RISUG for 90 and 360 days, reversal with DMSO and NaHCO3 after 90 and 360 days, respectively. The testis, cauda epididymis, cauda epididymal spermatozoa, and serum were evaluated for apoptosis and hormonal status through various assays. RISUG was subjected to Ames test at dose levels of 10, 50, and 100 µL. Results of terminal deoxynucleotidyl transferase nick end labeling and caspase-3 assays in testes and cauda epididymis revealed that the percentage of positive cells in the experimental groups was comparable to sham-operated control. Annexin V assay in cauda epididymal spermatozoa showed slight elevation in group II (P < 0.05), whereas in the remaining groups, minimum numbers of positive sperms were found. Hormone profile, namely, testosterone, prolactin, cortisol, prostate-specific antigen, and sperm antibody concentration, remained unchanged. In Ames test, no significant increase was observed in the number of revertant colonies on plates containing RISUG in the presence and absence of S9 mix in all 3 strains. Therefore, the reversal of RISUG-induced contraception by solvent vehicle DMSO/NaHCO3 was successful without any toxicity at the cellular levels.
Introduction
From a global standpoint, the concept of contraception as a method for population control is of paramount importance. The prospects of available contraceptives in male found to be ideal on the basis of 3 main criteria, namely, one-time application, long-term effect, and reversal upon need without any side effects. As the reversal compliance is lacking in most of the approaches and, if present, with less success rate, most men choose vasectomy and condoms as a contraceptive option now. Vasectomy, although long acting, is generally considered permanent due to difficulties of reversal, the expense, and development of antisperm antibodies (ASAs), which bring failure rate in restoring fertility. 1 At present, this has led to lacunae in contraceptive choices to men without toxicity after reversal. However, RISUG that has been already proven to be single-intervention, long-lasting, vas-based reversible contraceptive method in rats, rabbits, and monkeys 2 –7 has successfully completed phase I and II clinical trials and is currently under multicentric phase III clinical trials in India. 8,9 Noninvasive reversal approaches have been studied in langur monkeys. 2,10 The dimethyl sulfoxide (DMSO) and sodium bicarbonate (NaHCO3) solvents were used to dissolve RISUG in rats and rabbits to achieve reversibility. 6,7,11 The current research intended to assess the mutagenicity of the drug and solvents following reversal in rats.
Materials and Methods
Drug
RISUG, a copolymer synthesized through gamma irradiation of the monomers, styrene, and maleic anhydride, dissolved in the solvent vehicle DMSO in 1:2 ratios. It was kindly provided by Prof S. K. Guha, School of Medical Science and Technology, Indian Institute of Technology, Kharagpur, West Bengal, India.
Animals
Seventy adult Wistar albino rats (Rattus norvegicus), aged 10 to 12 weeks, weighing 150 to 180 g and of proven fertility, were used in the present study. Animals were maintained in the Departmental Experimental Animal Facility with the provision of 12-hour/12-hour light and dark schedule in polypropylene cages (size 43 cm × 27 cm × 15 cm). The temperature in animal house during the study period was maintained at 23°C ± 2°C, and the relative humidity ranged between 40% and 70%. Animals were fed with rat pellet diet (Ashirwad Industries Ltd, Chandigarh, India) and water ad libitum. Animals were maintained under perfect veterinary supervision in accordance to the guidelines of the regulation of scientific experiments on animals. The experimental protocol has approval of the Institutional Animal Ethical Committee. 12
Experimental Protocol
Rats were allocated into the following 7 groups containing 10 animals in each: Group I: Sham-operated control Group II: Vas occlusion with RISUG for 90 days Group III: Vas occlusion with RISUG for 90 days and reversal with DMSO Group IV: Vas occlusion with RISUG for 90 days and reversal with 5% NaHCO3
Group V: Vas occlusion with RISUG for 360 days Group VI: Vas occlusion with RISUG for 360 days and reversal with DMSO Group VII: Vas occlusion with RISUG for 360 days and reversal with 5% NaHCO3.
Vas Occlusion
Rats of groups II to VII were subjected to bilateral vas occlusion under sodium thiopentone anesthesia (20 mg/kg body weight intravenously, THIOSOL Sodium; Neon Laboratories Ltd, Mumbai, Maharashtra, India). After skin disinfection and hair shaving of the scrotum region, a single median incision was made just above the scrotum to expose the vas of inguinal region. The therapeutic dose of RISUG, that is, 5 to 7 μL, was injected into each vas deferens sufficient enough to block the pathway. The polymerization of styrene–maleic acid was catalyzed by application of normal saline and solidified on contact with fluid medium in the vas. The syringe was slowly withdrawn. The vas was placed in its original position, and incision was closed by catgut suture in the inner layer and by silken suture outside. The vas deferens of group I animals was exposed in a similar manner; however, no drug was injected. Postoperative care included antibiotic ceftriaxone (Cefoat; A to Z Pharmaceuticals Ltd, Gujarat, India) and anti-inflammatory drug meloxicam (Melonex; Intas Pharmaceuticals Ltd, Gujarat, India), injected in the gluteal muscles, along with topical application of Healex Plus spray (Shreya Life Sciences Pvt Ltd, Mumbai, India). Proper bedding and feed after half an hour was provided to the treated animals. All rats were kept under observation during healing period. The sutures were removed after complete healing. The vas deferens of group I animals were exposed in a similar way, but no drug was injected. Success of vas occlusion was confirmed by cohabitation of the vas occluded animals with the proven fertile females. 5,6 Mating was confirmed by vaginal smears for the appearance of spermatozoa.
Vas Occlusion Reversal
Reversal was performed following 90 and 360 days of vas occlusion under sodium thiopentone anesthesia. The vas was exposed in a similar way as that of occlusion and injected bilaterally with 250 to 500 µL of DMSO in groups III and VI and 500 to 700 μL of 5% NaHCO3 in groups IV and VII to dissolve RISUG. Dissolution of RISUG was ensured by free flow of liquid droplets from the vas. The vas was returned to its original position, and the incision was closed with catgut and silken sutures. Postoperative care was provided as described previously. Success of reversal was assessed after 15 days of reversal by fertility studies with proven fertile female at 1:2 ratio. 5,6
Sacrification Schedule
Following completion of experimental schedule, all animals were euthanized with an overdose of sodium thiopentone anesthesia.
Parameters
Animals of all groups were examined for the following parameters.
Apoptotic Markers
Terminal deoxynucleotidyl transferase nick end labeling assay
The terminal deoxynucleotidyl transferase nick end labeling (TUNEL) assay was performed using the DeadEnd fluorometric TUNEL system (Promega Corporation, Madison, Wisconsin) following the manufacturer’s instructions with modifications. In summary, sections of testis and cauda epididymis were deparaffinized with xylene, dehydrated in a graded ethanol series, rehydrated, and treated with 3% hydrogen peroxide for 20 minutes to reduce nonspecific staining. Sections were immersed in hot 10 mmol/L citrate buffer solution (pH 6.0) for 5 minutes, then incubated with normal goat serum for 20 minutes. Sections were incubated overnight at 4°C with the p53 primary antibody, washed with phosphate-buffered saline (PBS), and exposed to the avidin–biotin peroxidase complex for 1 hour at room temperature. The chromogenic substrate of peroxidase was developed using a 0.05% solution of 3,3-diaminobenzidine tetrahydrochloride, 0.03% hydrogen peroxide, and imidazole in Tris–HCL buffer (pH 7.6). Sections were counterstained with hematoxylin, and the number of apoptotic and p53-positive cells in each section was analyzed under a nonconfocal fluorescence microscope (Labomed; Labo America Inc, Fremont, CA) using excitation at 480 nm and emission at 535 nm barrier filter to view the green fluorescence of fluorescein and a 525/615 nm excitation/emission spectrum to view the red fluorescence of propidium iodide (PI). Images were generated using the DigiPro 4.0 and Image J program (Image J, Rockville, Maryland). For statistical analysis, 500 cells were counted in randomly selected fields and the numbers of positive cells were counted. 13
Immunofluorescent detection of activated caspase-3
The activity of caspase-3 was measured by tetrapeptide caspase substrate Asp-Glu-Val-Asp (DEVD)-7-amino-4-trifluoromethylcourmarin (DEVD-AFC) caspase-3 kit (Santa Cruz Biotech Ltd, Dallas, TX). In brief, testis and cauda epididymal sections were deparaffinized with xylene, rehydrated in a graded ethanol series, and permeabilized in 0.3% Triton-X 100. DEVD-AFC synthetic substrate was used for estimation and localization of activated caspase-3, which corresponds to the upstream amino acid sequence of the caspase-3 cleavage site in Poly ADP ribose polymerase and the fluorophore AFC. Caspase-3 hydrolyzes the peptide bond between D amino acid and AFC, releasing AFC. Apoptotic cells activate caspase-3, hence emitting high levels of fluorescence compared to control nonapoptotic cells. Free AFC levels were measured at 420 nm excitation and a 535 nm emission barrier filter. Images were generated using the DigiPro 4.0 and Image J software (Image J). For statistical analysis, 500 cells were counted in fields selected by systematic random approach and the numbers of positive cells were counted. 14
Annexin V binding assay
The membrane flip-flop was assessed as described by Shen et al, 15 according to the manufacturer’s protocol (Santa Cruz Biotech Ltd). Briefly, cauda epididymal sperms were washed twice in PBS at 1,200 rpm for 5 minutes and resuspended in 1 mL 1× assay buffer. Samples were redistributed in 100 µL aliquots, stained with annexin V–FITC conjugate and PI, and observed under nonconfocal fluorescent microscope at 480 and 525 nm excitation and 535 and 615 nm emission barrier filters. Results were compared with unstained cauda epididymal cell suspension. Images were generated using the DigiPro 4.0 and analyzed by Image J program (Image J). For statistical analysis, 500 cells were counted in fields selected by systematic random approach and the numbers of positive cells were calculated.
Serum Clinical Biochemistry
Hormone analyses
At the end of the experiment, animals were anesthetized and blood samples were obtained from the hearts of animals and allowed to clot for 20 minutes in laboratory temperature and then centrifuged at 3,000 rpm for 10 minutes for serum separation. Circulatory levels of serum testosterone, cortisol, and prolactin were assayed by enzyme-linked immunosorbent assay (ELISA) kits (DSI, Saronno, Italy). The intra- and interassay coefficients of variation from routine assays were 4.9% and 5.4% for testosterone, 2.95% and 4% for prolactin, 1.9% and 5% for cortisol, respectively.
Prostate-specific antigen
Prostate-specific antigen (PSA) was assayed in the separated serum with commercially available ELISA kits (DSI). The intra- and interassay coefficients of variations from routine assay were 6% and 6.6%, respectively.
Antisperm antibodies
The levels of ASAs were measured in serum using commercially available ELISA kits (DRG International, Inc, Springfield, NJ) in serum. The intra- and interassay coefficients of variations from routine assay were 6.88% and 6.45%, respectively.
Salmonella typhimurium reverse mutation assay (Ames test)
Ames test was carried out according to the method of Maron and Ames. 16 The S typhimurium tester strains TA97a, TA98, and TA100 were used in the Ames reversion test purchased from Institute of Microbial Technology (Chandigarh, India). Standard plate incorporation assay was used that consisted of exposing the tester strain(s) to the test substance (RISUG) at dose levels of 10, 50, and 100 µL directly on a glucose minimal (GM) agar plate in the presence and absence of a metabolic activation system. The RISUG doses S9 suspension and bacterial suspension were added to sterile test tubes containing 2 mL of molten agar supplemented with limited histidine and biotin. Temperature of top agar was maintained between 43°C and 48°C and to minimize prolonged exposure to avoid killing of the tester strains. Then contents of the tubes were mixed and poured on GM agar plates. After the top agar has solidified, the plates were inverted and incubated at 37°C for 48 hours. After incubation, histidine-revertant colonies were counted on all plates and the results were expressed as the number of revertant colonies per plate. Sodium azide and daunomycin (daunorubicin hydrochloride; Himedia Pvt Ltd, Mumbai, Maharashtra, India) 5.0 μg/plate were used as positive control.
Statistical Analysis
The mean values were compared using respective standard deviation, followed by statistical comparison between sham-operated control, vas occlusion, and vas occlusion reversal for evaluation of significant changes. Power calculation was performed, and difference between groups was analyzed by χ2 test using PS version 3.12 software (Department of Biostatistics, Vanderbilt University, Medical Centre, Nashville, Tennessee). The P values <0.05 were considered as significant.
Results
Apoptotic Markers
The TUNEL assay
The percentage of TUNEL-positive cells in testes and cauda epididymis of all the studied groups was found in the range of 10% to 12% and 6% to 10%, respectively. However, DNAase I-treated respective tissues showed 47.94% (18.16%) and 40.74% (16.45%) of TUNEL-positive cells. Testis showed slightly higher number of positive cells than cauda epididymis (Figure 1). The TUNEL-positive cells were localized to spermatogonial cells and spermatids in testes, whereas principal and basal cells in cauda epididymis. However, the TUNEL values of both short- and long-term reversal groups were normal compared to sham-operated control (Figure 2).
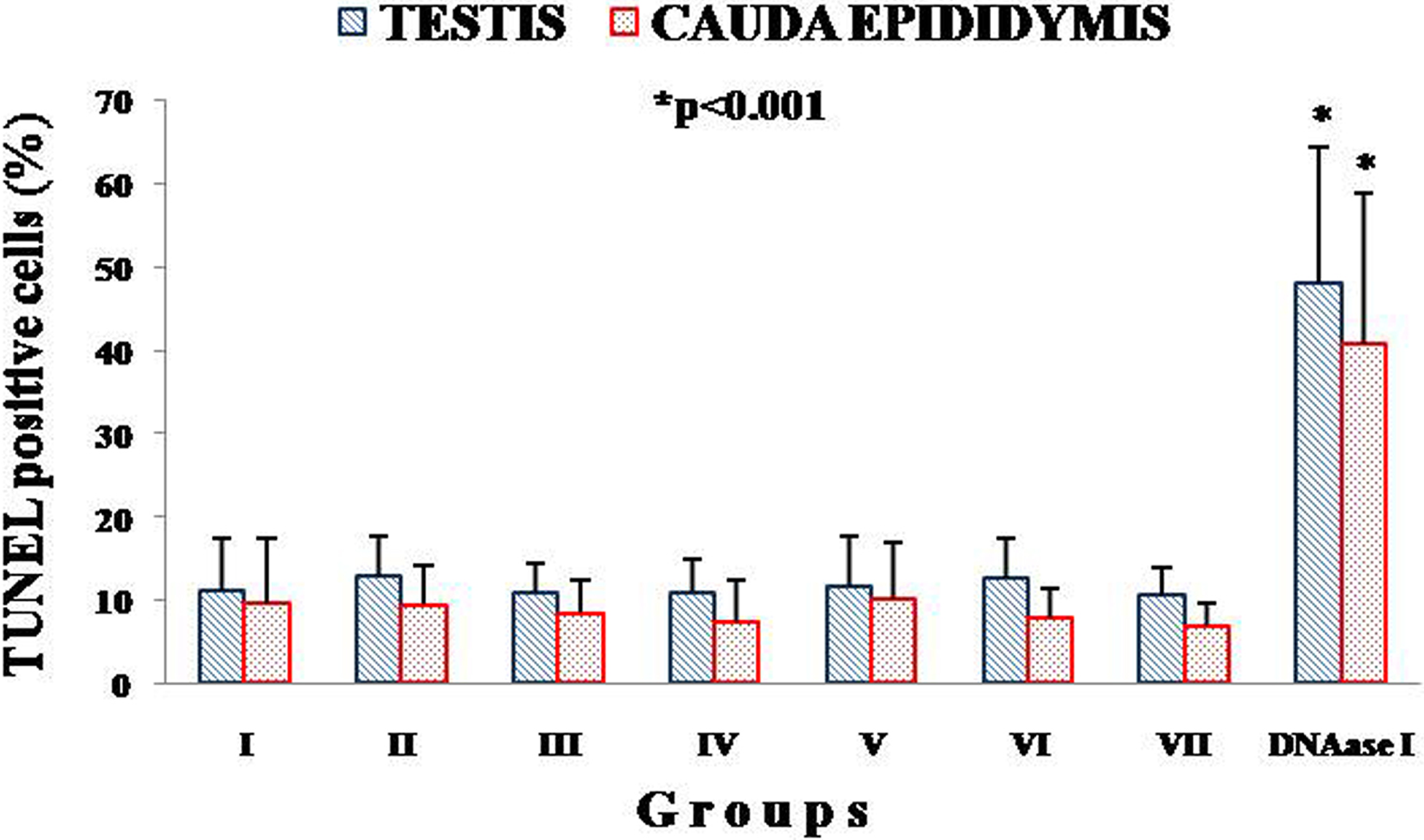
Percentage DNA breakage in experimental groups of testis and cauda epididymis. DNAase I-treated parallel positive controls showed significant variation in percentage of DNA damage comparing to test groups (I-VII). Positive control of testis showed highly significant (P < 0.001) variation comparing to positive controls of cauda epididymis. Images were analyzed by ImageJ and DigiPro software.
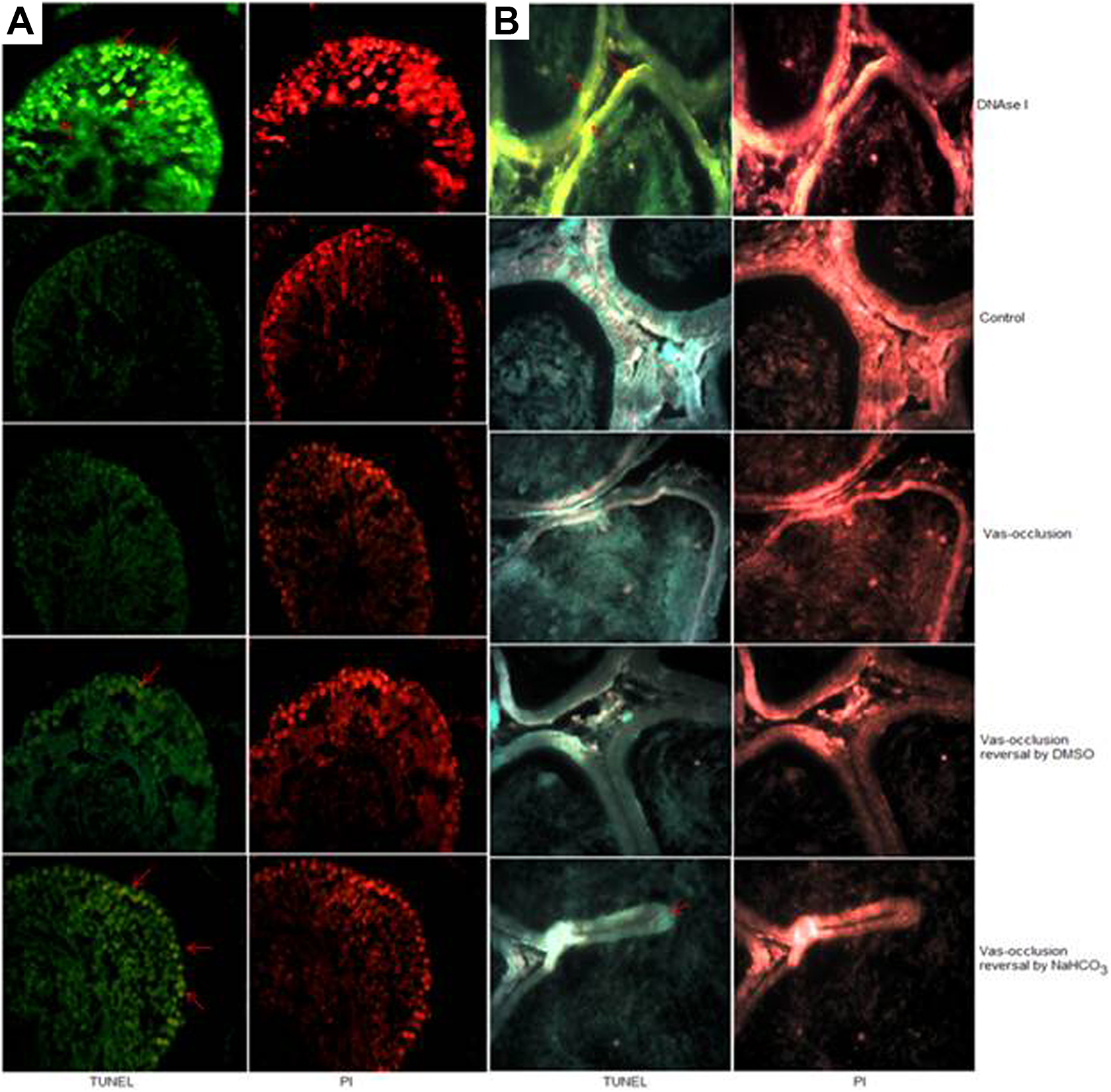
Paraffin sections of (A) testis and (B) cauda epididymis of vas occlusion with RISUG and vas occlusion reversal by dimethyl sulfoxide (DMSO) and sodium bicarbonate (NaHCO3) animals were treated with recombinant terminal deoxynucleotidyl transferase (TdT) and propidium iodide. Pointed red arrows indicate terminal deoxynucleotidyl transferase nick end labeling (TUNEL)-positive cells. DNAase I treatment was used as positive control. Fluorescent images were observed at ×400 and analyzed by ImageJ and DigiPro software.
Immunofluorescent detection of activated caspase-3
The percentages of caspase-3-positive cells in testes and cauda epididymis in all the experimental groups were found in the range of 0.68% to 1.08% and 0.40% to 0.92%, respectively. A slight insignificant elevation was observed in groups II and V. Testis showed slightly higher number of positive cells than cauda epididymis (Figure 3). Caspase-positive cells were localized to spermatogonial cells and spermatids in testes, whereas principal and basal cells in cauda epididymis. However, the percentage of caspase-3 activated cells in experimental groups was comparable to sham-operated control (Figure 4).
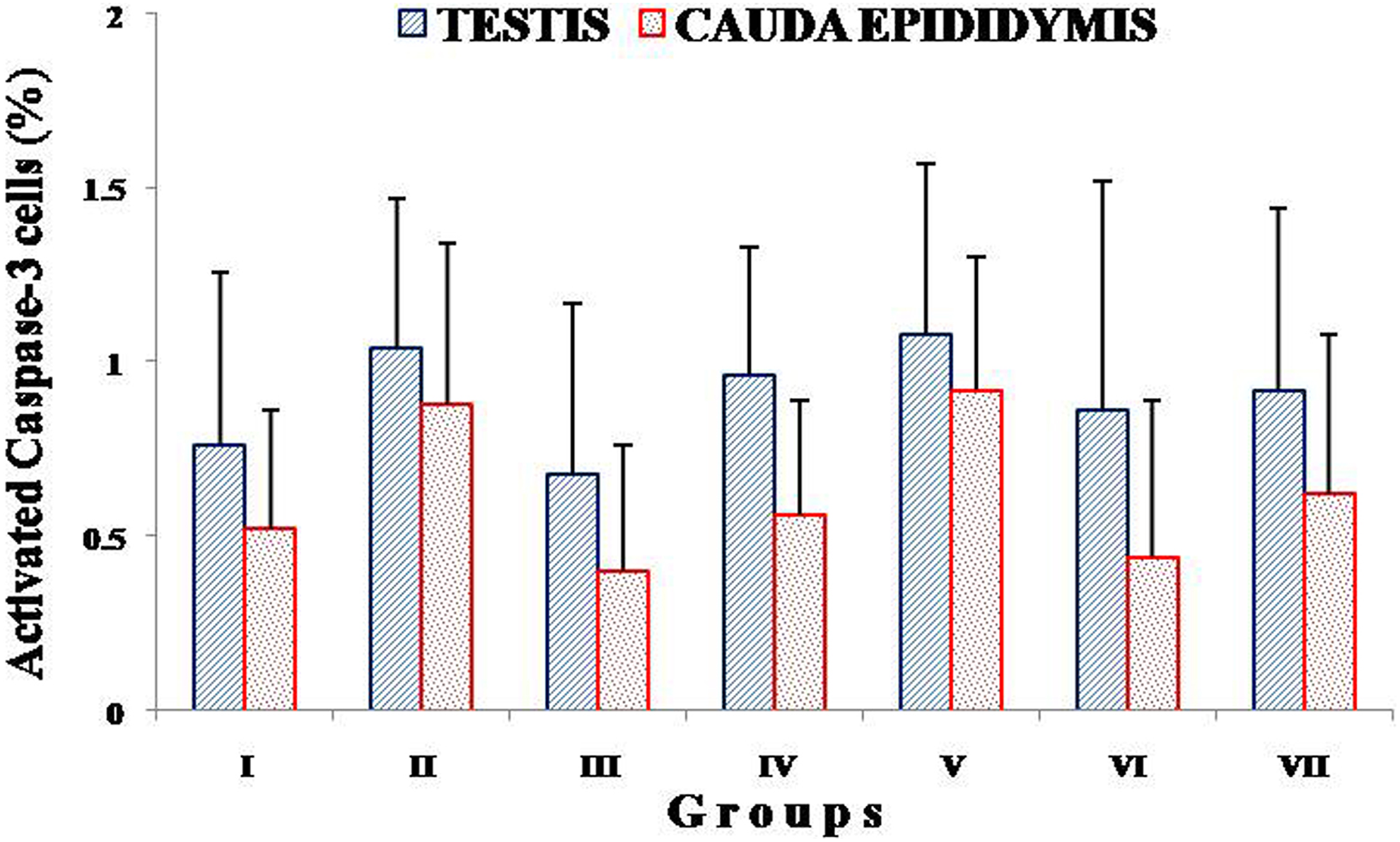
Percentage of positive cells showing activated caspase-3 in cauda epididymis and testis of vas occlusion with RISUG and vas occlusion reversal by dimethyl sulfoxide (DMSO) and sodium bicarbonate (NaHCO3) animals (groups I-VII). Images were analyzed by ImageJ and DigiPro software.
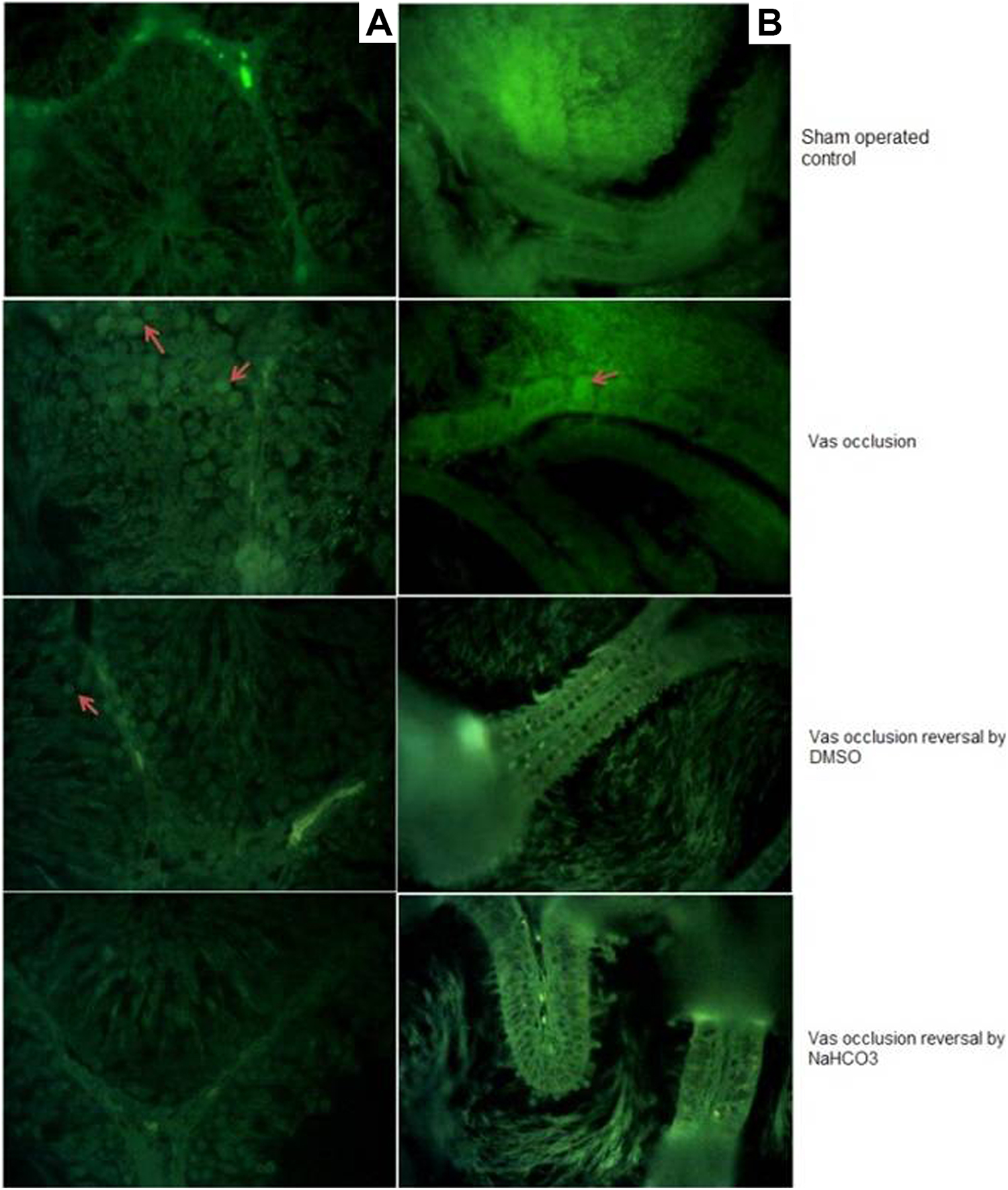
Fluorescent images of (A) testis and (B) cauda epididymis of vas occlusion with RISUG and vas occlusion reversal by dimethyl sulfoxide (DMSO) and sodium bicarbonate (NaHCO3) animals were observed at ×400 and analyzed by ImageJ software. Pointed red arrows indicate the release of fluorophore 7-amino-4-trifluoromethylcourmarin (AFC) in caspase-3 activated cells.
Annexin V binding assay
The percentage of annexin V-positive cauda epididymal spermatozoa in groups I to VII was observed to be minimum, that is, below 1% without much variation, except group II, that observed with slight elevation in the value significantly (P < 0.05; Figure 5). Fluorescent images of annexin V–FITC conjugate and PI-treated samples showed green fluorescence as the indication of the presence of few positive cells. However, the results did not show significant differences in all vas occlusion and vas occlusion reversal groups, and the values were comparable to sham-operated control group (Figure 6).
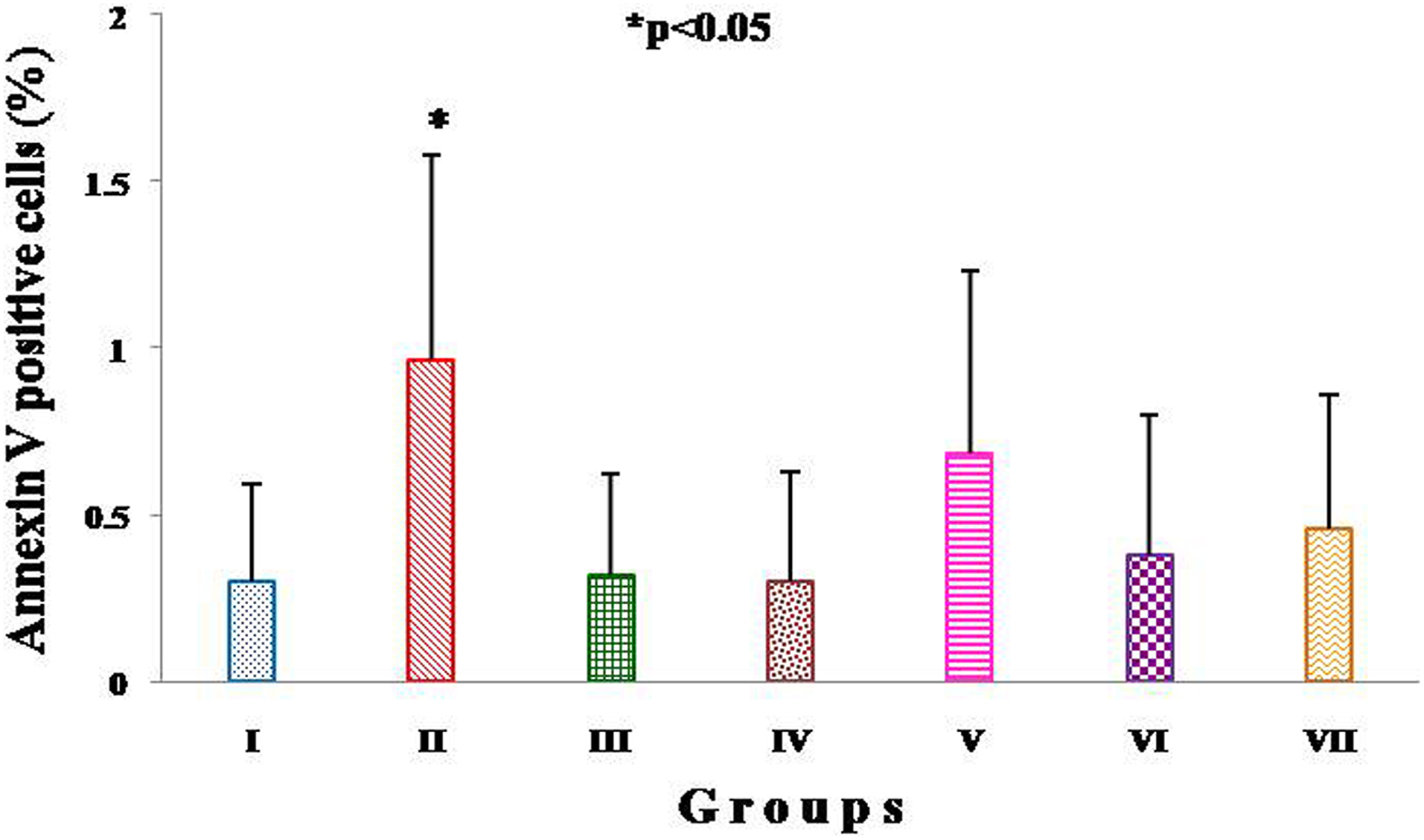
Percentage of annexin V-positive cauda epididymal sperms of groups I to VII. Significant variation was observed in percentage of annexin V-positive cells of group II cauda epididymal sperm when compared with other experimental groups.

Annexin V assay of cauda epididymal spermatozoa of experimental groups. Spermatozoa were treated with annexin V-FITC conjugate and propidium iodide (PI). Few spermatozoa with green fluorescence are showing annexin V-positive cells. Images were observed at ×400 and analyzed by ImageJ and DigiPro software.
Serum Clinical Biochemistry
Hormone analysis
The range of concentrations of testosterone (4.54-5.45 nmol/L), prolactin (37.80-43.15 mIU/L), and cortisol (101.36-116.53 nmol/L) in all vas occlusion and reversal groups did not show statistically significant changes compared with those of control levels (data not shown).
Prostate-specific antigen
The levels of serum PSA in all experimental groups were noticed in the range of 0.22 to 0.30 ng/mL with a nonsignificant variation when compared with sham-operated control group (data not shown).
Antisperm antibody
Determination of ASA with the ELISA method offered a range of 35.10 to 43.60 U/mL in all studied groups. The values were comparable to sham-operated control group (data not shown).
Salmonella typhimurium reverse mutation assay (Ames test)
Mutagenicity of RISUG was tested at dose levels of 10, 50, and 100 µL by S typhimurium tester strains TA97a, TA98, and TA100. Sodium azide and daunomycin (daunorubicin hydrochloride) at the dose of 5.0 μg/plate were used as positive controls, whereas DMSO at the doses of 10, 50, and 100 µL were used as a negative control in the presence and absence of the S9 metabolic activation system.
In positive control, a dramatic increase in the number of revertant colonies to >500 and >1,000 was observed on GM agar plates in all 3 strains (TA97a, TA98, and TA100) in the presence of S9 mix, which was significantly higher. In negative control (DMSO), the number of revertant colonies at 10 µL was 12 to 15, at 50 µL was 22 to 33, and at 100 µL was 35 to 46 in the presence of S9 mix, whereas the number of revertant colonies on plates containing RISUG at 10 µL was 17 to 25, at 50 µL was 12 to 62, and at 100 µL was 12 to 68 in the presence of S9 mix (Table 1).
Number of Revertant Colonies Over Glucose Minimal (GM) Agar Plates.a
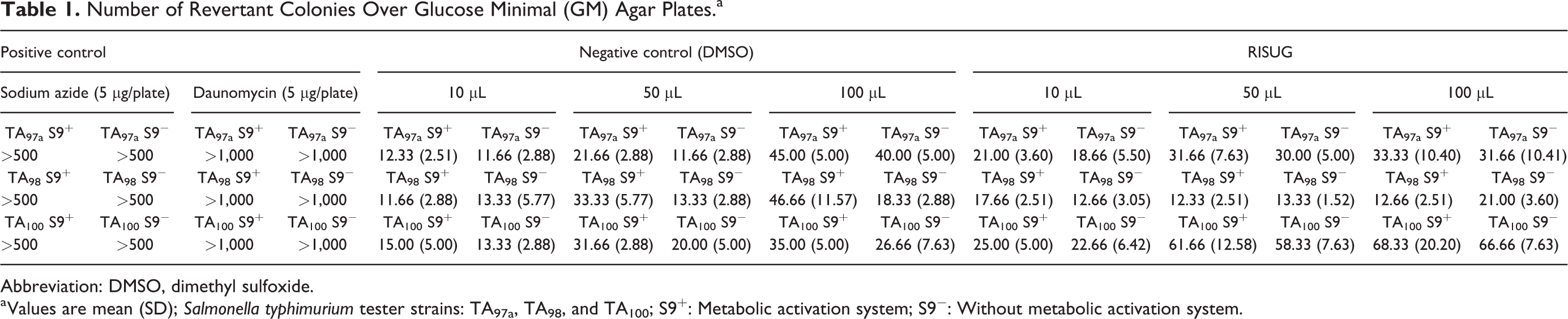
Abbreviation: DMSO, dimethyl sulfoxide.
a Values are mean (SD); Salmonella typhimurium tester strains: TA97a, TA98, and TA100; S9+: Metabolic activation system; S9
However, in the absence of S9 mix, the positive control also showed a remarkable increase in the number of revertant colonies to >500 and >1,000 was observed on GM agar plates in all 3 strains (TA97a, TA98, and TA100), which was considerably elevated. In negative control (DMSO), the number of revertant colonies at 10 µL was 11 to 13, at 50 µL was 12 to 20, and at 100 µL was 18 to 40, while the number of revertant colonies on plates containing RISUG at 10 µL was 12 to 22, at 50 µL was 13 to 58, and at 100 µL was 21 to 67 in the presence of S9 mix (Table 1). No significant increase was observed in the number of revertant colonies on plates containing RISUG in the presence and absence of S9 mix in all 3 strains.
Discussion
In the era of innovations in reproductive health, RISUG is an emerging reversible male contraception method in terms of safety, efficacy, and reversibility. The clinical acceptability based on both reversal approaches for RISUG in higher animals would be increased, as the predicted results of the present study following injection as well as its reversal were in support. The chromatin status and DNA integrity of a cell are critical factors that may affect individual fertility potential. So it is essential to evaluate the effects of chronic stress of RISUG reversal, if occurs, on various reproductive tissues as well as hormone level along with the mutagenicity of the test material. Furthermore, there is no information available about the copolymer reversal on hazardous substances database. Therefore, the present study aimed to through light toward mutagenicity of RISUG followed by its nonlethal or harmless reversal in rat animal model depicting short- and long-term evaluation.
Among all available approaches for men, the vas-based methods are mostly appreciated, while vasectomy accounts for more than 20% of the current methods of contraception in male. After vasectomy reversal, ASAs, abnormal sperm morphology, epididymal dysfunctions, and so on, are the reasons of lower pregnancy rate. 17 Vasectomy with subsequent reversal may also be associated with detectable alterations in sperm DNA integrity. 18 For these reasons, vasectomy cannot be recommended as a truly reversible method of contraception. In the present study, RISUG has surely created a new concept of contraception with great feasibility and long-lasting sterility. As concern with its reversal, RISUG tends to dissolve easily at higher pH (8-9) solution, mainly unstable its components and allows it to unbind from the wall of vas deferens. Hence, DMSO and NaHCO3 are used to flush RISUG plug from the vas deferens. To evaluate the effects after reversal, toxicity-related assays were performed.
Apoptosis is a major regulating factor proposed to cause DNA damage in spermatozoa before and after spermatogenesis. As an antagonist of cell proliferation, apoptosis contributes to keeping the cell number in testicular tissue and helps to remove superfluous and damaged cells, but excessive apoptosis could cause destruction of male reproductive function. Several methods exist for the detection of apoptosis using features of the cell as it undergoes the various stages, leading to the death of the cell. Recently, TUNEL assay, caspase-3, and annexin V have claimed more specificity. The present findings of apoptotic markers in testes, cauda epididymis, and cauda epididymal spermatozoa were pointed out without any evidence of genotoxicity related to drug injection followed by its reversal. There are a variety of etiologic factors that have been associated with sperm DNA fragmentation and/or impaired chromatin integrity. The minimum damage that observed could be due to individual animal response to various internal and external factors. In our recent studies of genotoxicity in rats, the comet assay presented the evidence that vas occlusion with RISUG and its reversal are highly unlikely to produce any genotoxic activity in leukocytes and/or testis, when injected under prescribed dose regimen. 5 Similarly, in rabbits, the overall results of genotoxicity and apoptotic markers indicated that no serious toxic effects were found related to any response of RISUG or its reversal. 11
In addition to apoptotic proteins, hormones have evident role in spermatogenesis. The present investigation based on reversal showed no significant fluctuations in the level of testosterone, prolactin, and cortisol. The values obtained were within control range, which reveals that RISUG reversal does not directly influence the metabolism of reproductive hormones. Prostate-specific antigen and ASA are valuable serum markers for diagnosing and monitoring infertility. Their levels were noticed without appreciable changes in all experimental groups as compared with control animals. Unlike our results, serum clinical chemistry (testosterone and PSA) in langur monkey observed with values fluctuated within control limits, indicating safety of the procedure at the level of accessory reproductive organ. 10,19,20
The Ames test was performed with RISUG to be reported first time in the literature. Presently, the mutagenicity of RISUG as a test compound was examined at different dose levels, that is, 10, 50, and 100 µL. Hence, it suggests that RISUG doses tested against the 3 strains did not meet the criteria for a potential mutagen. The overall results depicted the successful reversal of the test material without any adverse property in concern of toxicity at the cellular level and hormonal imbalance. Although more detailed work is required to bridge the association, the important consideration concerns are the accomplishment of the study in higher animals in future and findings of molecular mechanism of the intrinsic apoptotic pathways including expression and roles of apoptotic proteins that should be highlighted soon. Furthermore, clinical studies should be performed using above reversal solvents to determine its acceptability in men.
Conclusion
The preclinical toxicity testing after reversal of RISUG on various biological samples revealed the dose-specific effects in male albino rats. Conclusively, the present study highlighted RISUG as nonmutagenic with the negative response of genotoxicity markers linked with its reversal. The normal hormonal status, PSA, and ASAs revealed safety of the procedure that did not cause any alteration in the functions of accessory reproductive organs. However, RISUG is considered as safe procedure to be used with its reversibility upon need, even after long duration without any toxic effects.
Footnotes
Acknowledgments
The infrastructural facilities provided by the Head of the Department are gratefully acknowledged. The authors are thankful to Prof Sujoy K. Guha, School of Medical Science and Technology, Indian Institute of Technology, Kharagpur, West Bengal, India, for providing RISUG. The award of NASI Senior Scientist’s assignment to NKL is gratefully acknowledged.
Author Contributions
Abdul S. Ansari and N. K. Lohiya designed the study. Mubarik Hussain and Sadi Rehan Khan performed the experiments and analyzed the data. Ayesha Badar represented the data and wrote the manuscript. N. K. Lohiya finally approved the version to be published. Abdul S. Ansari substantially contributed to conception or design and critically revised the manuscript for important intellectual content. Mubarik Hussain contributed to acquisition, analysis, and interpretation and critically revised the manuscript. Sadi Rehan Khan contributed to acquisition, analysis, and interpretation and critically revised the manuscript. Ayesha Badar contributed to interpretation and drafted the manuscript. N. K. Lohiya contributed to conception and design and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Indian Council of Medical Research, New Delhi.
