Abstract
The peptide product, Valtyron, is obtained via enzymatic hydrolysis of sardine muscle. Although the safety and efficacy of the sardine peptide product have been evaluated in human studies, sardine peptides have not been identified as the subject of toxicological testing. In this study, the sardine peptide product did not exhibit any mutagenic activity in Salmonella typhimurium or Escherichia coli WP2uvrA. Likewise, the sardine peptide product was not associated with clastogenic properties in mouse bone marrow cells in a micronucleus assay. An oral rat LD50 value of greater than 10 000 mg per kilogram of body weight was determined for peptide α-1000, and in rats administered peptide α-1000 by gavage at levels up to 5000 mg per kilogram of body weight per day for 28 days, no compound-related differences were observed in standard toxicological parameters. The results of these studies support the safety of the sardine peptide product for use in food for human consumption as a dietary source of peptides available from sardines.
Short-chain peptides can be produced from sardine protein via enzymatic hydrolysis. To produce peptides from sardines, sardine-derived muscle proteins are subjected to alkaline protease hydrolysis under controlled conditions. The final peptide product contains at least 80% dipeptides, tripeptides, and larger short-chain peptides (up to 5 amino acids in length). Valyl-tyrosine (VY), valyl-tryptophan, isoleucyl-tyrosine, and isoleucyl-tryptophan are some of the dipeptides that have been identified in the sardine muscle hydrolysate product. Dipeptides, including VY specifically, have been shown to possess properties inhibiting angiotensin I converting enzyme. Given these properties, VY-containing sardine peptide products have been reported to exert blood pressure–lowering effects when ingested by humans. 1
In Japan, sardine peptide products have been approved for use as ingredients in Food for Specified Health Uses for blood pressure regulation. 2 In 2008, Valtyron, produced in accordance with current good manufacturing processes and meeting appropriate food-grade specifications, was self-affirmed as generally recognized as safe (GRAS) by Senmi Ekisu Co., Ltd. (Hirano-Cho, Ozu, Japan) for use in a variety of food products at use levels up to 30% or 0.6 g per serving (Senmi Ekisu, personal communication, 2008). Valtyron is intended for use in foods such as breakfast cereals, egg, fish, meat and poultry products, frozen dairy desserts and mixes, grain products and pastas, hard and soft candies, milk and milk products, processed fruits and vegetables, fruit and vegetable juices, and soups and soup mixes. Using a worst-case scenario of estimating intake, it was calculated that a heavy-end (90th percentile) consumer of Valtyron would be exposed to no more than 3 g of Valtyron per day (approximately 64 mg/kg of body weight [BW] per day) under the conditions of intended use. The estimated exposure to sardine peptides under the conditions of use self-affirmed by the manufacturer as GRAS is well below current daily protein intakes and the Recommended Dietary Allowances (RDAs) established for protein by the Institute of Medicine. 3 Specifically, the mean and 90th percentile total population intakes for protein were estimated to be approximately 75 and 114 g/d, respectively, and RDAs for protein were established at 56 and 46 g/d (0.8 g/kg BW per day) for adult males and females, respectively.
Several studies have been conducted in humans to evaluate the potential blood pressure regulation properties as well as the safety of the sardine peptide product. 1,4-7 In human clinical trials, daily ingestion of sardine peptide products at dose levels of 500 mg for 12 to 13 weeks or 1500 mg to 4 g for 2 to 4 weeks was not associated with any significant disturbances in standard hematological, clinical chemistry, or urinalysis parameters. However, a search of the scientific literature databases did not reveal any preclinical toxicological studies pertaining to the safety of sardine peptides for use in food for human consumption.
Accordingly, this series of experiments was undertaken to expand the existing safety database for sardine peptides. The evaluation of sardine peptide safety included an in vitro mutagenicity assay in bacteria (Salmonella typhimurium and Escherichia coli) and an in vivo micronucleus test with ICR mice. In addition, acute single-dose (LD50) and 28-day subchronic oral toxicity studies were conducted with Sprague-Dawley (SD) rats.
Whereas the mutagenicity/genotoxicity assays were performed with the final commercial sardine peptide product (Valtyron), the oral animal toxicology studies were conducted with peptide α-1000, an intermediary product that is obtained during the manufacturing process; however, both products are of very similar amino acid composition, and thus studies conducted using peptide α-1000 support the safety of the end sardine peptide product as an ingredient for use in food. The final sardine peptide product is eluted by passing peptide α-1000 through a chromatography column and extracting fractions of different solubilities to water and ethanol. The fractionation procedure ensures that peptides of high hydrophilicity and hydrophobicity are removed from the final sardine peptide product. Although the final commercial sardine peptide product will be more concentrated in certain peptides compared with peptide α-1000, the relative amino acid composition of peptide α-1000 is largely retained following the elution procedure, with all of the amino acids in the final product present at levels no more than 2-fold greater than the amount in the intermediate product, except tryptophan, which is approximately 2.3-fold greater in the final product. The specifications for peptide α-1000 and the final sardine peptide include a peptide content of not less than 80% and 85%, respectively. The remainder of the products are composed of moisture (≤8%) and ash (≤10%), whereas the intermediate product also contains fat (≤10%). Sodium is present at specification levels of not more than 10% for peptide α-1000 and 2% for the final sardine peptide product. Table 1 summarizes the amino acid compositions of the commercially-available sardine peptide product and of peptide α-1000.
Amino Acid Composition (%) of the Final Sardine Protein Product and Peptide α-1000 a
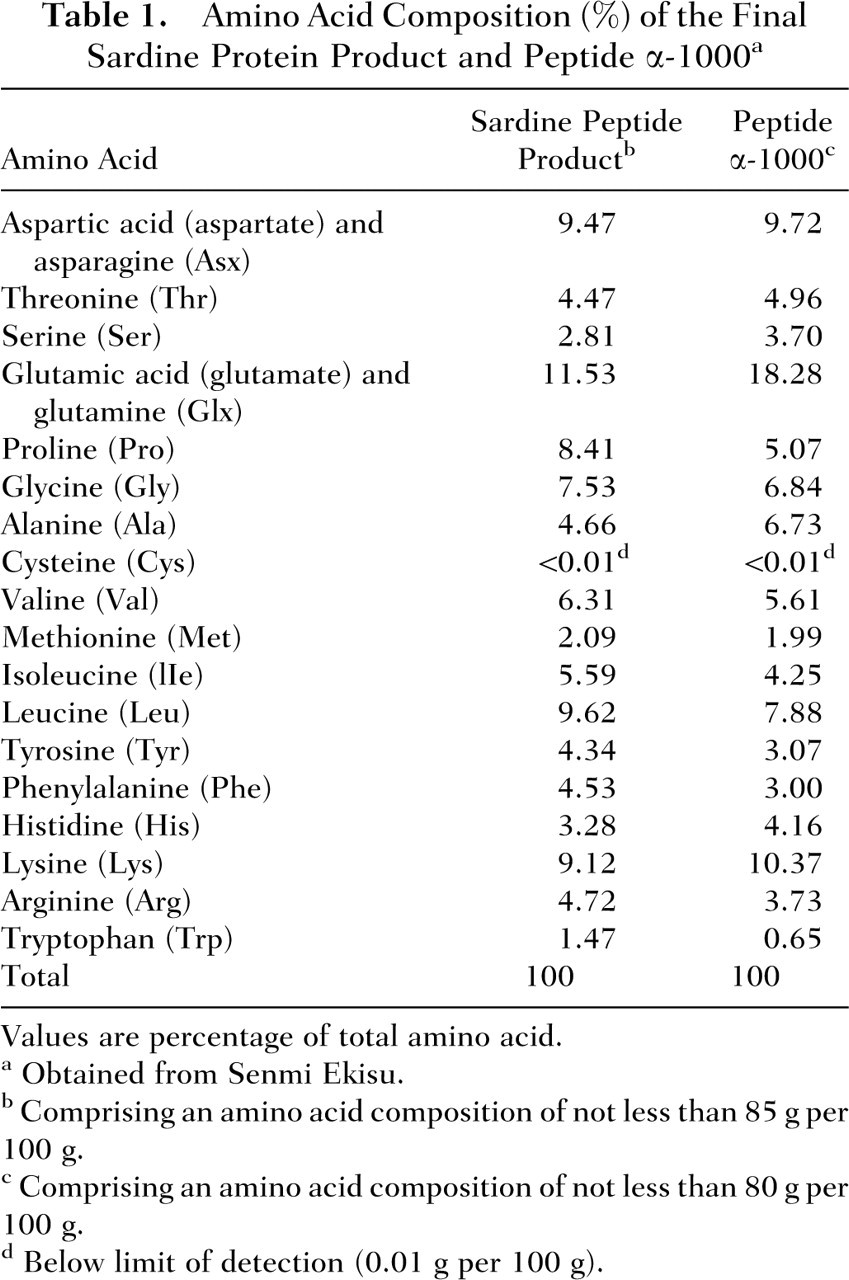
Values are percentage of total amino acid.
Obtained from Senmi Ekisu.
Comprising an amino acid composition of not less than 85 g per 100 g.
Comprising an amino acid composition of not less than 80 g per 100 g.
Below limit of detection (0.01 g per 100 g).
Method
In Vitro Microbial Mutagenicity Assays
The in vitro microbial mutagenicity assays were conducted at Biochemical and Pharmacological Laboratories, Inc. (Wakamatsu-cho Higahi, Tondabayashi, Osaka, Japan) during the period of April 26, 2000, and May 15, 2000. For the mutagenicity assays, a sardine peptide product (Lot 00.3.30) with a peptide content of 89.1% was provided by Senmi Ekisu. The potential in vitro mutagenicity of the sardine peptide product was determined using the histidine-dependent auxotrophic S typhimurium strains TA98, TA100, TA1535, and TA1537 and the tryptophan-dependent auxotrophic E coli strain WP2uvrA in the standard plate incorporation assay. The S typhimurium strains were obtained from Professor B. N. Ames (University of California, Berkeley, Calif) and the E coli strain was obtained from Professor T. Kada (National Institute of Genetics, Japan). The microorganisms were compliant with the requirements of the Japanese Ministry of Health, Labor and Welfare’s Article 34-3. 8 The tests were conducted in accordance with the Organization for Economic Cooperation and Development Guidelines for Testing of Chemicals (No. 471: Bacterial Reverse Mutation Test). 9
Five concentrations of the sardine peptide product were tested: 313, 625, 1250, 2500, and 5000 μg per plate. The sardine peptide product was dissolved in 0.1 M sodium-phosphate buffer (pH 7.4) (Wako Pure Chemical Industries, Ltd., Osaka, Japan) to prepare a 50-mg/mL solution, which was further serially diluted with the buffer prior to testing. The 0.1-M sodium-phosphate buffer solution served as the negative control. The following compounds were tested as positive controls: 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (AF-2) (Wako Pure Chemical Industries), sodium azide (NaN3) (Wako Pure Chemical Industries), 9-aminoacridine (9AA) (Merck, Whitehouse Station, NJ), and 2-aminoanthracene (2AA) (Sigma-Aldrich, Tokyo, Japan) (Table 2 ). The reverse mutation assays were performed in the presence and absence of an exogenous metabolic (S9) activation system (Oriental Yeast Co., Ltd., Tokyo, Japan), which was prepared from the livers of male SD rats following 1 injection of phenobarbital at 30 mg/kg BW and 3 injections of phenobarbital at 60 mg/kg BW administered every 24 hours in the morning and a single intraperitoneal injection of 5,6-benzoflavone (80 mg/kg BW) administered in the afternoon of the third day.
Positive Controls for In Vitro Mutagenicity Assays of a Sardine Peptide Product
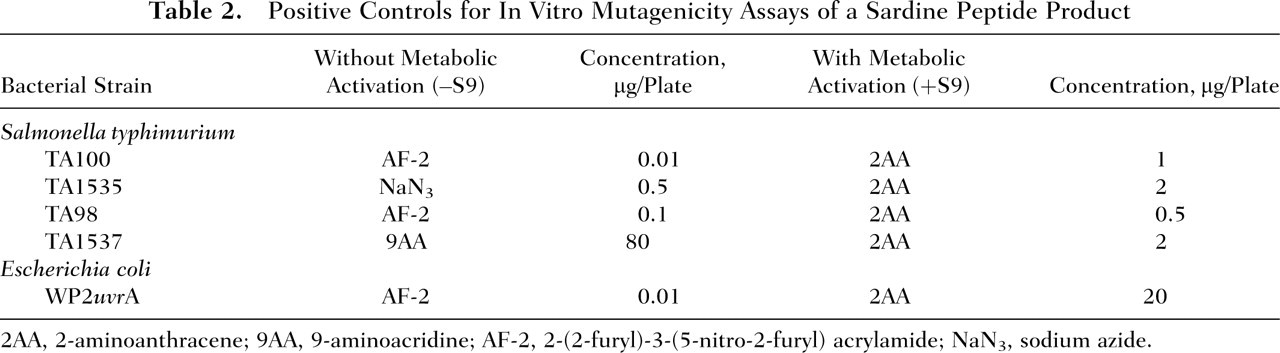
2AA, 2-aminoanthracene; 9AA, 9-aminoacridine; AF-2, 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; NaN3, sodium azide.
The test articles were added to the plates in a volume of 0.1 mL. In the case of assays conducted with metabolic activation, 0.5 mL of the S9 activation system was added to each plate. Three plates were used for the negative control, whereas for the positive controls and for each concentration level of the sardine peptide product tested, 2 plates were included. Plates were incubated at 37°C for 48 hours. Dose-dependent increases of greater than 2-fold compared with the negative control values were considered a positive response.
In Vivo Micronucleus Assay in Mice
The in vivo micronucleus assay was conducted at the Drug Safety Research Laboratories, Shin Nippon Biomedical Laboratories, Ltd. (Miyanoura, Yoshidamachi, Kagoshima, Japan) from August 26 to December 6, 2004. The study was conducted in accordance with the Guideline for Genotoxicity Studies of Drugs (Notification No. 1604 issued by the Director of Evaluation and Licensing Division, Pharmaceutical and Medical Safety Bureau, the Ministry of Health, Labor and Welfare, Japan.) 10 The testing followed OECD Guidelines for Testing of Chemicals (No. 474: Mammalian Erythrocyte Micronucleus Test). 11 A sardine peptide product (Lot AX6.04) with a peptide content of 85.6% was supplied by Senmi Ekisu. Thirty male Crj:CD-1(ICR) mice, purchased from Charles River Laboratories Japan, Inc. (Yokohama, Japan), were acclimated to laboratory conditions for 11 days. The environmental conditions under which the mice were housed were specifically regulated. The temperature and humidity ranges were 19°C to 25°C and 30% to 70%, respectively. The ventilation rate was 15 air changes per hour, and artificial light was provided for 12 h/d (ie, 12-hour light/dark cycle). The animals were housed in aluminum cages, with 4 animals per cage during the acclimation period and 5 animals per cage after group assignment. A standard commercial pellet diet (CE-2; Clea Japan, Inc., Tokyo, Japan) and water were provided ad libitum. The animals were identified by staining with a dye (picric acid solution; Kanto Chemical Co., Inc., Tokyo, Japan) during the acclimation period; after assignment to groups, the ear-punching method was used. At the end of the acclimation period, the animals were randomly distributed to groups (a total of 6 groups with 5 animals per group) according to mean body weight. At the start of the study period, the mice were 8 weeks old and body weights ranged from 32.7 to 38.9 g.
To determine whether the sardine peptides possess clastogenic properties, the sardine peptide product was administered to the mice once daily at dose levels of 250, 500, 1000, or 2000 mg/kg BW by gavage using a stomach tube (Fuchigami Kikai, Kyoto, Japan) and a disposable syringe (Nipro Corporation, Osaka, Japan) for 2 consecutive days at 24-hour intervals (Day 0 denotes the day when testing commenced) (Table 3 ). The dose selection was based on the absence of any signs of toxicity in mice administered sardine peptides orally at a dose level of 5000 mg/kg BW in preliminary dose-finding experimental studies (private communication). The dosing volume was 10 mL/kg BW, and the actual volume administered was calculated based on the body weight of each animal determined immediately prior to dose administration. Required amounts of sardine peptide were dissolved in 0.5% (wt/vol) aqueous solutions of carboxymethyl cellulose sodium (0.5% CMC-Na) to prepare sardine peptide solutions with concentrations of 25, 50, and 100 mg/mL. The solvent solution was prepared by dissolving the required amount of CMC-Na (Wako Pure Chemical Industries) in “water for injection” (Otsuka Pharmaceutical Factory, Inc., Tokushima, Japan). After preparation, the solvent solution was autoclaved at 121°C for 20 minutes (Autoclave: MLS-3750; Sanyo Electric Co., Ltd., Osaka, Japan) and stored under refrigeration (2-6°C) until use. In the case of the highest concentration of the sardine peptide product tested (ie, 200 mg/mL), a suspension was prepared by precipitation. Each sardine peptide test solution or suspension was prepared shortly before use, and because the test article was a peptide mixture, sonification was not required.
Control and Test Groups of Mice Used in the In Vivo Micronucleus Test of a Sardine Peptide Product
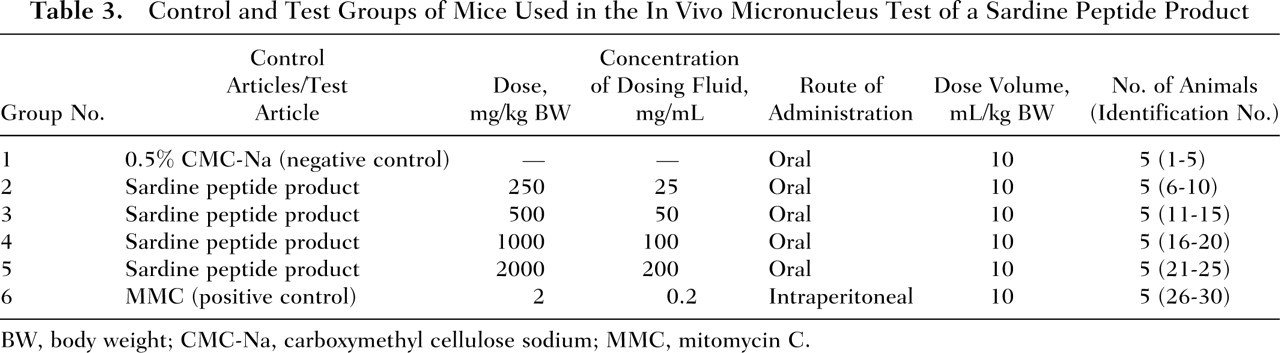
BW, body weight; CMC-Na, carboxymethyl cellulose sodium; MMC, mitomycin C.
The negative control group was administered the solvent solution, 0.5% CMC-Na, once daily for 2 consecutive days (24-hour interval) by oral gavage using a stomach tube (Fuchigami Kikai) and a disposable syringe (Nipro Corporation, Osaka, Japan). The positive control group received a single intraperitoneal injection of mitomycin C (MMC) at a dose level of 2 mg/kg BW using a disposable syringe and needle (Nipro Corporation, Osaka, Japan). To prepare the positive control, MMC (Wako Pure Chemical Industries) was dissolved in physiological saline (Otsuka Pharmaceutical Factory) to make a 0.2-mg/mL solution, which was distributed to tubes and kept frozen (–10°C and lower) until use.
Animals were observed for clinical signs of toxicity and survival prior to dosing and at approximately 1 and 4 hours after dosing on Days 0 and 1 and prior to necropsy on Day 2. Body weights were measured on Days 0 and 1 shortly before dosing and on Day 2 before the animals were killed. Approximately 24 hours after the final dose was administered, the mice were euthanized by dislocation of the cervical vertebrae and the bilateral femurs removed for bone marrow collection and smear preparation. The bone marrow collection time was set in accordance with the Guideline for Genotoxicity Studies of Drugs.
To prepare bone marrow smears, both ends of the femurs were cut and bone marrow fluid was washed out with about 1 mL of fetal bovine serum (Invitrogen Japan K.K., Tokyo Japan). Thereafter, the fluid was centrifuged at 1000 rpm (210 g) for 5 minutes (tabletop multisample centrifuge KS-8300; Kubota Co., Japan) and the supernatant discarded. A small amount of cell suspension was pipetted onto a lipid-free, clean glass slide, and a smear specimen was prepared using a cover-slip. Once the specimen was sufficiently dry, it was fixed with methanol for 5 minutes and immediately stained with 3% Giemsa solution (Merck). Following staining, the specimen was gently washed with a sodium–potassium phosphate buffer solution (Wako Pure Chemical Industries) and subsequently was treated with a 0.004% (wt/vol) aqueous citric acid solution (Wako Pure Chemical Industries) for approximately 3 seconds, washed with distilled water, and finally dried.
Coded specimens were examined using a light microscope (BH-2; Olympus Co., Japan) at a final magnification rate of ×600. For every 2000 polychromatic erythrocytes (PCEs) in each individual sample, the number of micronucleated polychromatic erythrocytes (MN-PCEs) was counted to determine the MN-PCE prevalence. In addition, as an index of growth suppression of bone marrow cells, the numbers of PCEs and normochromatic erythrocytes (NCEs) were scored in 500 cells to determine the ratio of PCE to total erythrocytes (PCE/PCE+NCE).
The statistical significance of differences in the prevalence of MN-PCEs between the negative control group and the test groups or the positive control group was determined by the method of Kastenbaum and Bowman. 12 A difference of P < .05 was considered statistically significant. To determine the statistical significance of differences between the groups in the rate of bone marrow cell proliferation, the Student’s t test was applied. A difference of P < .01 was considered statistically significant.
Acute and Subchronic Toxicity Studies in Rats
The acute and subchronic toxicity studies were conducted at the Tsukuba Research Institute, Jisseiken Co. (Chuo, Ami-cho, Inashiki-gun, Ibaragi, Japan) during the periods from September 11–September 25, 1992, and from October 7, 1992, to April 2, 1993, respectively. These studies were conducted in compliance with the Guidelines for Toxicity Studies on Drugs (Notification No. 24 of the Pharmaceuticals and Cosmetics Division, Pharmaceuticals Affairs Bureau, the Ministry of Health, Labor and Welfare, Japan). 13 The subchronic study also was compliant with the Standards for Conducting Safety Studies on Drugs (GLP) (notification no. 313 of the Pharmaceuticals and Cosmetics Division, Pharmaceuticals Affairs Bureau, the Ministry of Health, Labor and Welfare, Japan). 14 The studies were conducted in accordance with the Japanese Ministry of Environment’s 1973 Act on Welfare and Management of Animals. 15
Test material
Peptide α-1000 (Lot 920304) with a peptide content of 82.4% was supplied by Senmi Ekisu. During the study period, peptide α-1000 was stored in a refrigerator (4-8°C) under light-resistant conditions. Under the recommended storage conditions (cool, dark, and air-tight), the stability of the test material was guaranteed by the sponsor for the duration of the study period.
For the acute and subchronic toxicity studies, the required amount of peptide α-1000 was weighed and dissolved in “water for injection” (Otsuka Pharmaceutical Factory). A 100% (wt/vol) solution was prepared for the acute toxicity study, whereas for the subchronic toxicity study, 12.5%, 25%, and 50% (wt/vol) solutions were prepared twice weekly and were kept under refrigerated (–4°C) and light-resistant conditions. Stability testing of a 50% solution under the specified storage conditions was conducted, and it was determined that the solution was stable for at least 7 days.
Animals and maintenance
A total of 44 male and 31 female 5-week-old Crj:CD(SD) rats, obtained from Charles River Laboratories Japan, were acclimated to the laboratory conditions for 7 days prior to dosing, during which time body weights and general conditions were observed. The animals were identified by marking with Food Blue No. 1 (Tokyo Chemical Industry Co., Ltd., Tokyo, Japan) during the acclimation time and by both ear-punching and marking with Quick Color Paint (Japan SLC, Inc., Hamamatsu, Shizuoka, Japan) after group assignment. The rats were housed in stainless steel bracket cages (360 mm width × 246 mm depth × 180 mm height; Clea Japan). During the acclimation period, 2 or 3 rats were housed per cage, whereas during the study period, animals were caged individually (acute study) or in pairs (subchronic study). Rats were provided a standard commercial pellet diet (CRF-1; Oriental Yeast) and tap water. The tap water was disinfected by the addition of sodium hypochlorite and by irradiation with ultraviolet light. The rat chow was analyzed for possible impurities at the Japan Food Research Laboratories (Kitakyushucity, Japan) at the request of the supplier, and the drinking water quality was determined twice a year at the Ibaraki Environmental Technology Center (Ibaraki, Japan). The animals were maintained under specifically regulated environmental conditions: 12-hour light/dark cycle, temperature 24°C ± 2°C, humidity 55% ± 10%, and ventilation 11 to 15 air exchanges per hour (all-fresh-air system).
Experimental procedure
For the acute study, 8 male rats that were confirmed to be in good health following the acclimation period were randomized according to body weight into 2 groups (4 per group) on the day prior to dosing initiation. On the day of dosing (designated as Day 0 of the study), the rats were 6 weeks old and body weights ranged from 160 to 171 g. Based on the results of a previously conducted acute toxicity study in mice with the sponsor’s material (data not reported), the toxicity of peptide α-1000 was expected to be very low. Therefore, a dose of 10 000 mg/kg BW was selected. Peptide α-1000 was administered orally as a 100% solution by gavage using a metal stomach tube (Natsume Seisakusho Co., Ltd., Tokyo, Japan). The dosing volume was 10 mL/kg BW and the actual volume of peptide α-1000 solution administered was calculated based on body weight measurements obtained on the day of administration. The negative control group was administered the vehicle “water for injection” (Otsuka Pharmaceutical Factory) in a similar manner. Prior to dosing, the animals were deprived of food for 17 hours but had free access to drinking water. Food was provided 3 hours after dose administration and was available ad libitum thereafter.
The animals were observed frequently during the first 6-hour period following dose administration and once daily, thereafter, for 7 days, during which animals were evaluated for mortality, clinical signs of toxicity, and behavior. Each animal was weighed on Day 0 (prior to dosing) and on Days 1, 3, and 7 during the observation period. At the end of the observation period, all surviving animals were exsanguinated from the abdominal aorta and posterior vena cava under ether anesthesia and were necropsied, and the main organs of the thoracic and abdominal cavities and the brain were examined macroscopically for gross abnormalities.
For the 28-day subchronic toxicity testing, 24 male and 24 female rats that were found to be in good health at the end of the acclimation period were assigned to the study at the age of 6 weeks. Body weights at the beginning of the study period ranged from 184.8 to 198.2 g for the males and from 139.5 to 158.1 g for the females. The day before the start of dosing (Day 0), the animals were randomly distributed to 4 groups (6 males and 6 females per group) according to body weight. Based on the results of the foregoing single-dose oral toxicity study in which no signs of toxicity were observed at a dose level of 10 000 mg/kg BW, gavage dose levels of 1200, 2500, or 5000 mg/kg BW per day were selected for repeat-dose administration (ie, low-, mid-, and high-dose group, respectively). At each respective dose level, the test substance was administered as a 12.5%, 25%, or 50% (wt/vol) solution of peptide α-1000 at a dosing volume of 10 mL/kg BW. The actual volume of administration was calculated based on body weight measurements obtained on the day of dosing. The doses were administered by gavage using a disposable syringe (Terumo Co., Tokyo, Japan) and a stomach tube (Natsume Seisakusho) daily beginning on Day 1 for a total of 28 consecutive days. The control group animals received gavage doses of equal amounts of “water for injection” (Otsuka Pharmaceutical) (Table 4). Food and water were provided to animals ad libitum throughout the study period.
Control and Test Groups of Rats Used in the 28-Day Oral Toxicity Study of Peptide α-1000
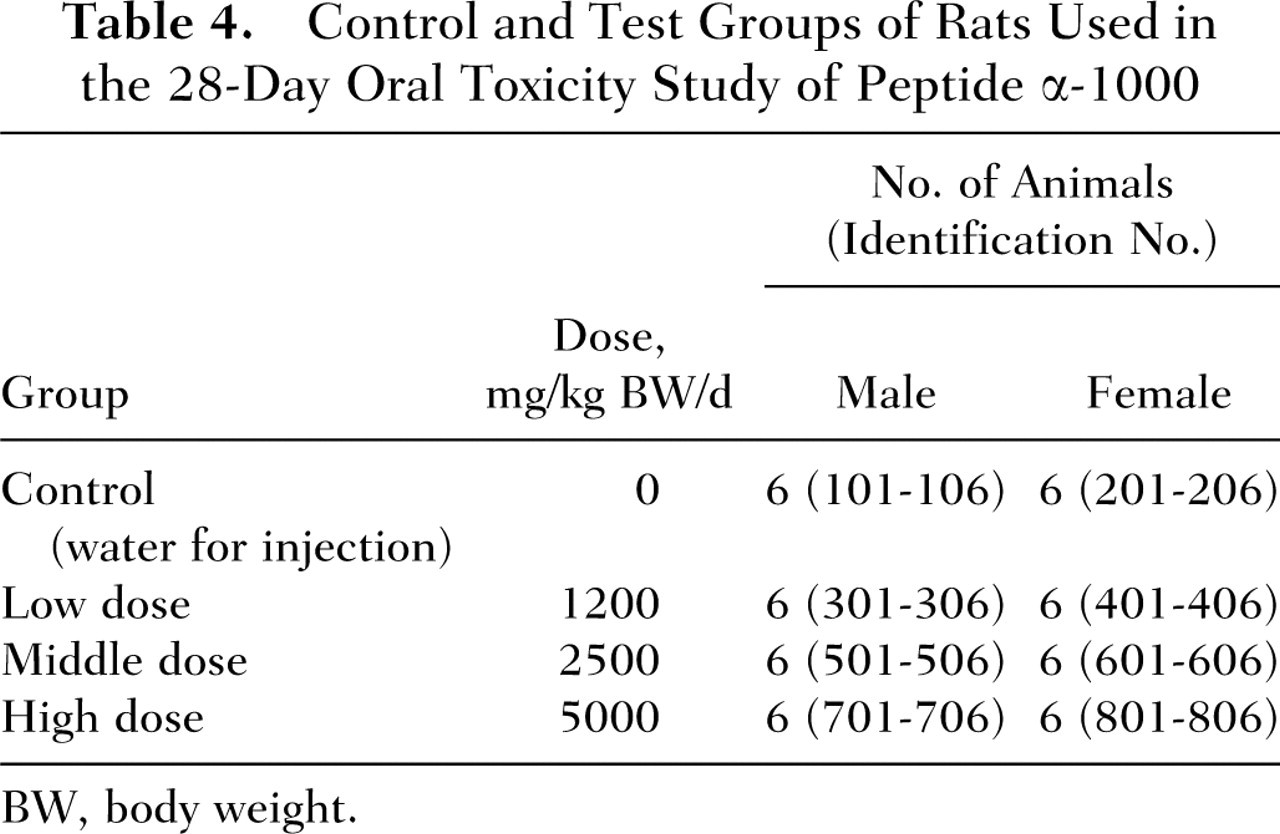
BW, body weight.
The animals were observed once daily for general condition, and body weight measurements were obtained on Days 1, 7, 14, 21, and 28 of the study period. Food consumption was determined weekly (on Days 8, 15, 22, and 29) by measuring the amount of food consumed per cage per week and calculating the mean daily food intake for each animal. On Days 20 and 21, males and females, respectively, were placed into individual metabolism cages for determination of 24-hour water intake and urine collection. Urine samples were analyzed for the following parameters: volume of urine excreted over the 24-hour period, pH, and presence of protein, glucose, ketone bodies, occult blood, and bilirubin (Table 5). On the day following final dose administration (Day 29) and after a fasting period of at least 5 hours, blood samples for clinical laboratory analysis were collected from the abdominal aorta of all animals under pentobarbital anesthesia (Nembutal injection; Dainippon Pharmaceutical Co., Osaka, Japan) and all animals were subsequently exsanguinated. To determine hematology parameters (Table 6), edetic acid dipotassium salt (EDTA-2K; Wako Pure Chemical Industries) was added to the blood samples. For blood chemistry analysis (Table 7), blood samples were kept at room temperature for 15 to 20 minutes and subsequently centrifuged at 2500 rpm at 4°C for 20 minutes to obtain sera. Animals were necropsied; the absolute weights of the lungs, heart, liver, spleen, and kidneys of all the animals were measured; and the weight of each organ relative to 100 g of the rat’s final body weight was calculated. The following organs from all animals were fixed in neutrally buffered 10% formalin solution: stomach, duodenum, jejunum, ileum, colon, rectum, pancreas, lungs, heart, liver, spleen, kidneys, and any other organs with gross lesions identified during macroscopic examination. Specimens of organs in which lesions were found by macroscopic observation were fixed and stained with hematoxylin and eosin and examined microscopically.
Urinalysis Parameters Evaluated in the 28-Day Oral Rat Toxicity Study of Peptide α-1000

Miles Sankyo Co., Ltd., Tokyo, Japan.
Hematological Parameters Evaluated in the 28-Day Oral Rat Toxicity Study of Peptide α-1000
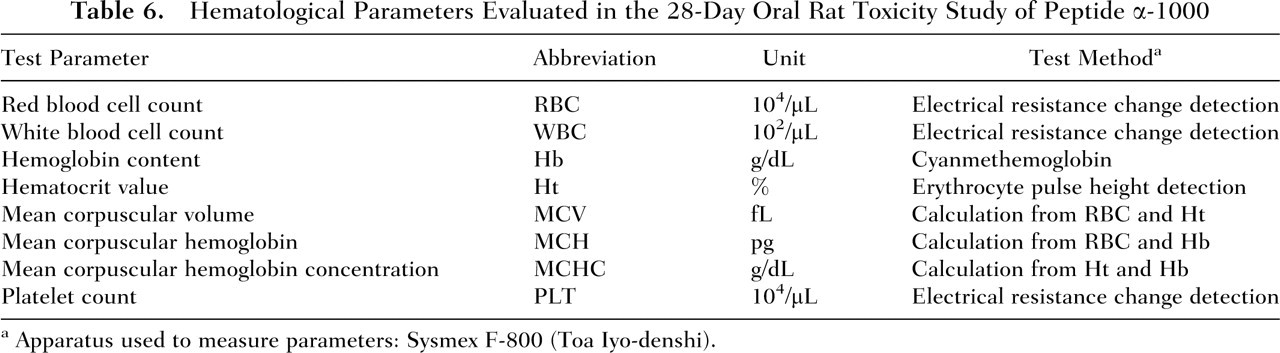
Apparatus used to measure parameters: Sysmex F-800 (Toa Iyo-denshi).
Clinical Chemistry Parameters Evaluated in the 28-Day Oral Rat Toxicity Study of Peptide α-1000
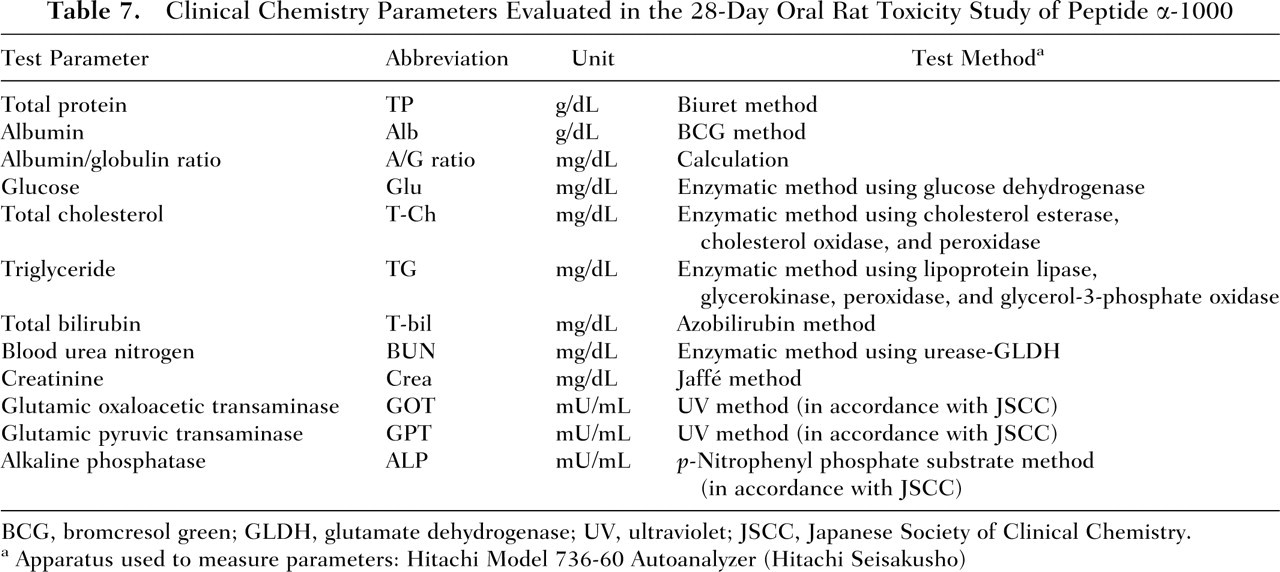
BCG, bromcresol green; GLDH, glutamate dehydrogenase; UV, ultraviolet; JSCC, Japanese Society of Clinical Chemistry.
Apparatus used to measure parameters: Hitachi Model 736-60 Autoanalyzer (Hitachi Seisakusho)
Statistical analysis
For the acute toxicity study, the body weight data measured on Days 1, 3, and 7 were subjected to statistical assessment using Student’s t test and Aspen-Welch’s t test with the level of statistical significance set at 5%. All parameters measured in the 28-day study were analyzed statistically, with the exception of general condition of the rats and findings of the macroscopic and histopathological examinations. The mean and standard deviation were calculated for the quantitative data for each dose group. Individual data were subjected to logarithmic transformation to normalize the distribution, and the homogeneity of variance was tested by Bartlett’s method at a significance level of 5%. When homogeneity of variance was found, one-way analysis of variance was performed. When a significant difference was found in the analysis of variance, Dunnett’s test at 5% was performed to determine which means were significantly different from the control. When homogeneity of variance was not found, the Kruskal-Wallis rank test was used at a significance level of 5%. For semi-quantitative data, the chi-square test was performed.
Results
In Vitro Microbial Mutagenicity Assays
The results of the mutagenicity assay are presented in Table 8 . Compared with the negative control values, exposure to the sardine peptide product did not increase the mean number of revertant colonies in S typhimurium TA100, TA1535, TA98, and TA1537 or in E coli WP2uvrA at any of the concentrations tested (313-5000 mg per plate) with or without an S9 activation system. Conversely, all of the positive control compounds (ie, AF-2, NaN3, and 9AA without S9 activation and 2AA with S9 activation) increased the mean number of reverse mutations by greater than 2-fold relative to the negative control.
Results of In Vitro Mutagenicity Assay Conducted With a Sardine Peptide Product in the Absence (–S9) and Presence of Metabolic Activation (+S9)
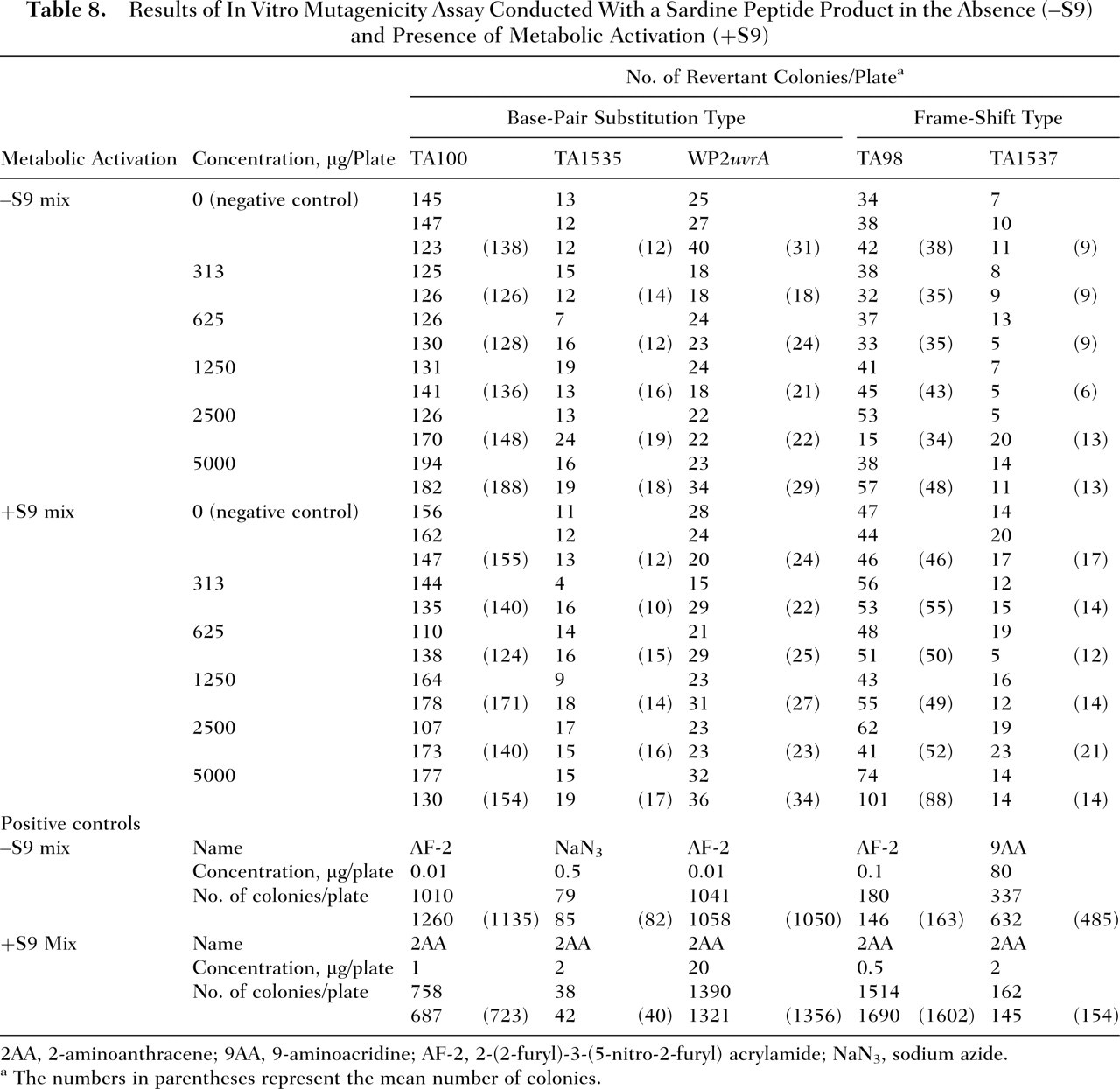
2AA, 2-aminoanthracene; 9AA, 9-aminoacridine; AF-2, 2-(2-furyl)-3-(5-nitro-2-furyl) acrylamide; NaN3, sodium azide.
The numbers in parentheses represent the mean number of colonies.
In Vivo Micronucleus Assay in Mice
No animals died following administration of the sardine peptide product, and no changes in general appearance or behavior attributable to the administration of the sardine peptide product were observed. Body weights of the sardine peptide-treated animals were comparable to the negative controls.
The prevalence of MN-PCE in bone marrow cells of mice administered the sardine peptide product at dose levels of 250, 500, 1000, and 2000 mg/kg BW was 0.08, 0.06, 0.07, and 0.08%, respectively (Table 9 ). The values observed in the sardine peptide-treated mice did not differ significantly (P < .05) from the value determined in the negative control group (0.04%). The prevalence of MN-PCE observed in the positive control group (3.83%) was significantly higher (P < .05) than the corresponding negative control group value. Of all the erythrocytes examined, the PCE incidence was determined to be 46.88%, 44.76%, 40.52%, and 40.08% in animals administered the sardine peptide product at dose levels of 250, 500, 1000, and 2500 mg/kg BW, respectively. The reductions in the ratios of PCE to total erythrocytes (PCE/PCE+NCE) observed at the two highest dose levels (1000 and 2000 mg/kg BW) were statistically significant (P ≤ .01) compared with the ratio observed for the negative control group (49.96%). The PCE/PCE+NCE value in the positive control group (37.72%) also was significantly lower than that of the negative control group. The prevalence of MN-PCE and the ratio of PCE to total erythrocytes in both the negative and the positive control groups were within the ranges of historical background data for the laboratory (mean ± 3 standard deviation).
Results of the In Vivo Micronucleus Test Conducted in Mice Following Oral Administration of a Sardine Peptide Product (Bone Marrow Cells Collected 24 Hours Following Final Administration)
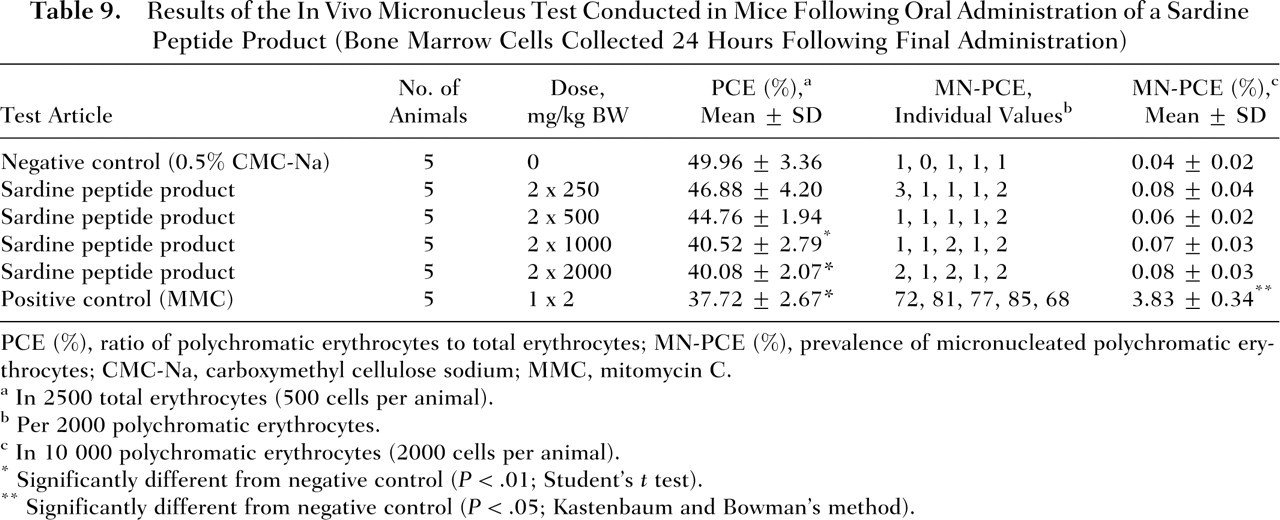
PCE (%), ratio of polychromatic erythrocytes to total erythrocytes; MN-PCE (%), prevalence of micronucleated polychromatic erythrocytes; CMC-Na, carboxymethyl cellulose sodium; MMC, mitomycin C.
In 2500 total erythrocytes (500 cells per animal).
Per 2000 polychromatic erythrocytes.
In 10 000 polychromatic erythrocytes (2000 cells per animal).
Significantly different from negative control (P < .01; Student’s t test).
Significantly different from negative control (P < .05; Kastenbaum and Bowman’s method).
Acute Toxicity Study in Rats
No deaths occurred throughout the 7-day observation period following single administration of peptide α-1000 at a dose of greater than 10 000 mg/kg BW. Mild salivation was observed in 2 of the 4 animals in the peptide α-1000-treated group immediately following dose administration, persisting for approximately 1 hour thereafter. Rats in both the control and treatment groups demonstrated comparable increases in body weight. No macroscopic abnormalities were observed upon necropsy in any of the animals treated with peptide α-1000. Based on these results, oral administration of peptide α-1000 was not associated with any toxic effects, and the LD50 was determined to be greater than the dose level tested in this study (ie, 10 000 mg/kg BW).
Subchronic Toxicity Study in Rats
No deaths and no abnormalities in general condition occurred in either males or females of the control and treatment groups throughout the 28-day study period. Mean body weights and food consumption of rats administered peptide α-1000 by gavage were comparable to controls (Table 10 ). Compared with the control group, the low- and high-dose-level males treated with peptide α-1000 showed reductions in mean water consumption. In females, mean water consumption also appeared to be slightly lower in the test groups compared with the controls. The differences in water intakes in both males and females were not statistically significant. Two test female rats, one at each the mid- and high-dose level (Numbers 603 and 801, respectively), exhibited considerably lower water consumption values compared with the control and low-dose groups (data for individual animals not shown). No statistically significant variations were observed in any of the urinalysis, hematological, and clinical chemistry parameters among males and females of the different groups (Table 11).
Body Weight and Food and Water Consumption for Male and Female Rats Following Daily Gavage Administration of Peptide α-1000 for 28 Days
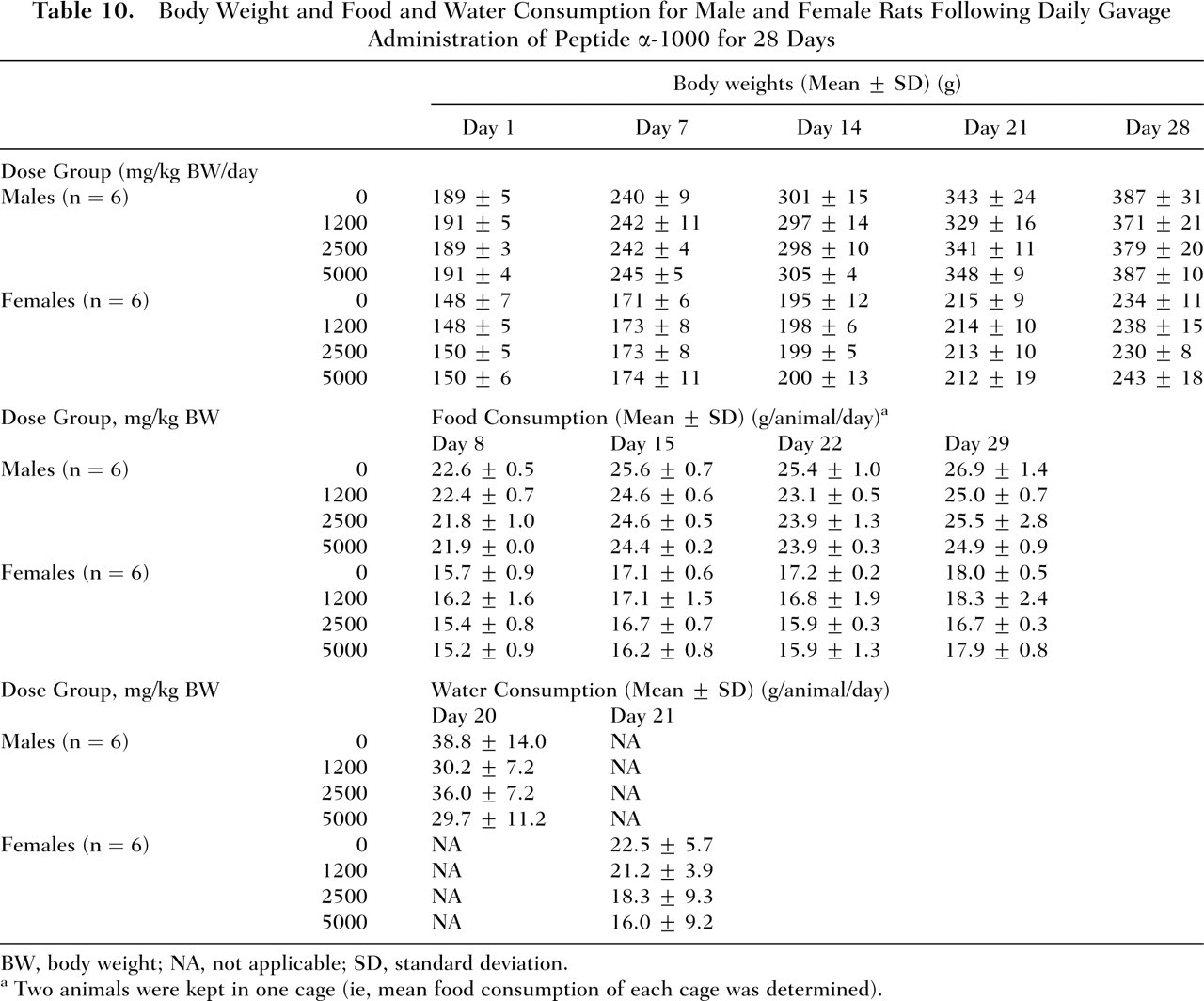
BW, body weight; NA, not applicable; SD, standard deviation.
Two animals were kept in one cage (ie, mean food consumption of each cage was determined).
Hematology, Clinical Chemistry, and Urinalysis Results for Male and Female Rats Following Daily Gavage Administration of Peptide α-1000 for 28 Days
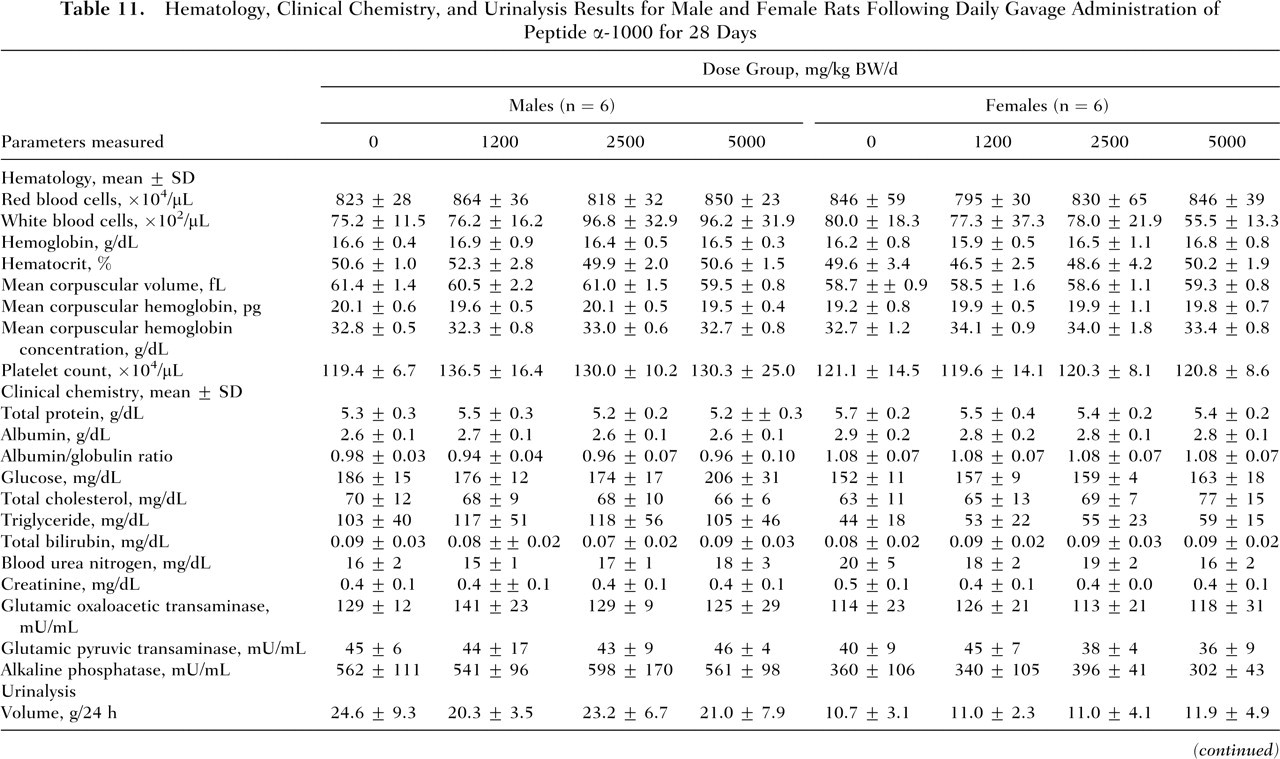
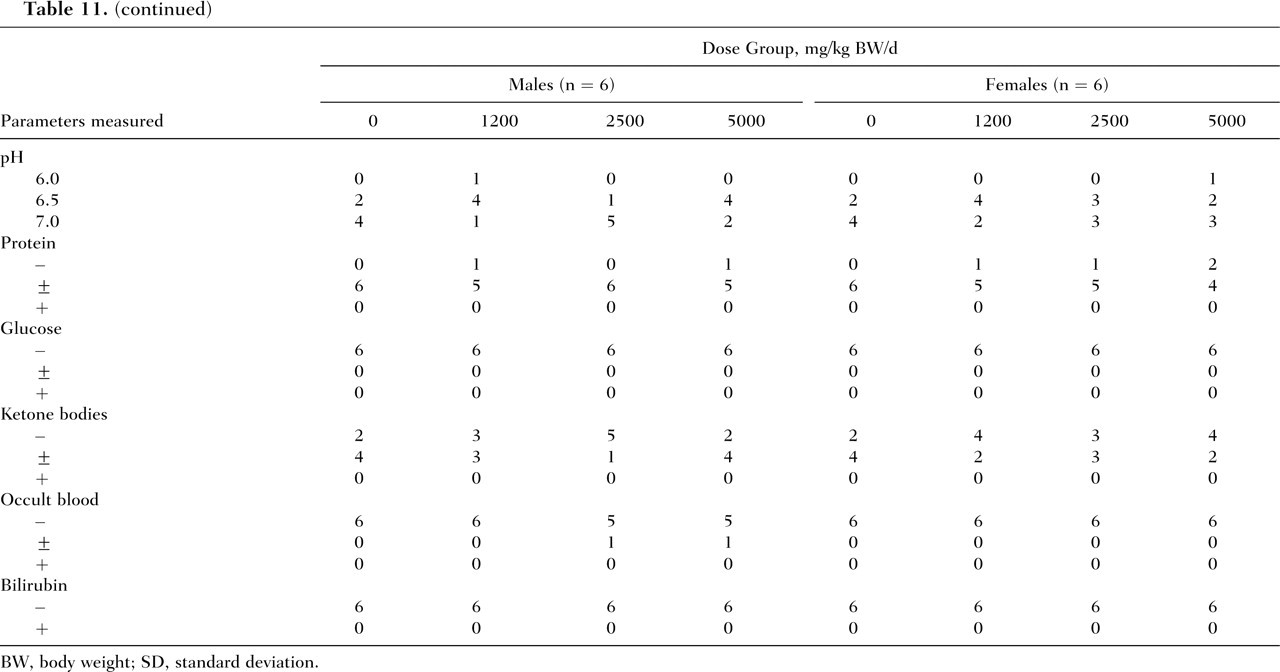
BW, body weight; SD, standard deviation.
Relative to the control group, no significant differences in absolute or relative organ weights were observed in either the males or females of all test groups, with the exception of increased absolute and relative left kidney weights in males of the high-dose group (5000 mg/kg BW per day) (Table 12 ).
Absolute and Relative Organ Weights for Male and Female Rats Following 28-Day Gavage Administration of Peptide α-1000
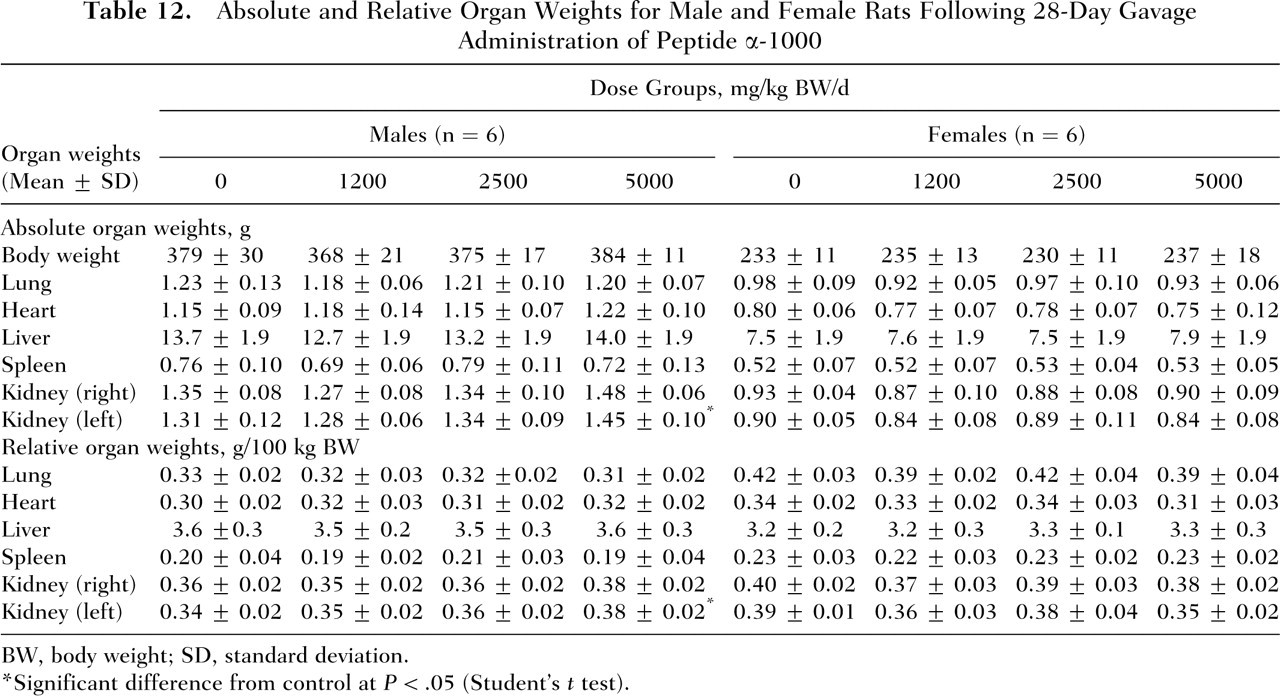
BW, body weight; SD, standard deviation.
Significant difference from control at P < .05 (Student’s t test).
The macroscopic examination did not reveal any gross abnormalities in male rats of either the control or treatment groups. Similarly, in the female rats, with the exception of a single occurrence of hemorrhage in the glandular stomach of 1 female in the mid-dose group, no other macroscopic lesions were noted. The left kidneys of males in the high-dose and control groups were examined histopathologically as a result of the reported weight increase. Microscopic examination of the left kidneys of both the control and high-dose males revealed eosinophilic microparticles, hyaline droplet-like changes, thickening of the basal membrane, calcification in the left renal tubules, infiltration of lymphocytes and fibroblasts into the interstitium, and thickening of the Bowman’s capsule. All of the changes were determined to be of only slight severity; none occurred at levels of moderate severity. The prevalence and severity of these histopathological findings in the high-dose group males were comparable to those observed in the control group males (Table 13).
Histopathological Findings in the Left Kidney of Male Rats Following 28-Day Gavage Administration of Peptide α-1000 at Dose Levels of 0 (Control) or 5000 mg/kg BW/d

BW, body weight; SD, standard deviation.
Severity of histopathological findings: –, no lesions; +, slight; ++, moderate.
Discussion
Consumption of short-chain peptides, including peptides derived from the muscle of sardines, and specifically VY has been associated with blood pressure–lowering effects. As such, there is growing interest in the addition of short-chain peptides to food to control mild hypertension in humans. Several human clinical trials have been conducted to determine the efficacy of sardine peptides for use as blood pressure–lowering agents as well as to assess their safety in humans. 1 4-7 However, no traditional toxicological studies were identified in the published literature that evaluated the safety of sardine peptides in laboratory animals. To further develop the body of evidence supporting the safety of sardine peptides, we assessed the toxicology of a commercial sardine peptide product and a closely related intermediate production product, peptide α-1000.
In the current series of studies, the sardine peptide product was determined to be nonmutagenic in the standard Ames assay in both the presence and the absence of metabolic activation. Similar results were obtained in E coli, in which the sardine peptide product also failed to induce increases in the incidence of reverse mutations. Examination of mice erythrocytes following administration of 2 successive gavage doses of the sardine peptide product at dose levels of up to 2000 mg/kg BW did not elevate the incidence of micronuclei compared with the solvent control. No clinical signs of toxicity were observed in any of the mice following administration of the test material. However, comparison of the treatment groups to the negative control group revealed a dose-dependent reduction in the PCE/PCE+NCE ratio, indicating a potential effect on erythropoiesis. Although statistically significant reductions in the PCE/PCE+NCE ratio were observed in mice following administration of only 2 doses of sardine peptides at 1000 and 2000 mg/kg BW, no variations in hematological parameters were observed in the 28-day rat toxicity study. Likewise, in clinical trials in which indices of hematological relevance were examined, significant variations were observed only between pretreatment and posttreatment values and often occurred in both placebo and test group subjects. 5-7 Apart from a significant reduction in the mean corpuscular volume in study participants consuming sardine peptide products at dose levels of 500 mg/d for 12 weeks compared with placebo-control values, which were decreased at study end relative to baseline levels, no significant differences in hematological parameters were observed between test and placebo subjects. 5 No remarkable differences in any hematology parameters assessed, including mean corpuscular volume, were observed between placebo and test groups in other 12- or 13-week studies in which subjects also consumed 500 mg of sardine peptide daily. 4 7 Considering that the effect on erythropoiesis observed in mice was not reproduced in longer term rat and clinical studies, it is not likely that sardine peptides would adversely affect normal blood cell development in humans. It may be that the decrease in the number of PCEs relative to the total number of erythrocytes reflects a species-specific response.
In the single-dose oral toxicity study of peptide α-1000, no animals died, all animals gained weight, and pathological examination was unremarkable. Transient salivation was observed briefly in 2 of 4 animals following single administration of the test compound at a dose level of 10 000 mg/kg BW. Given that the salivation occurred for only a short period of time immediately following dosing, it may have been attributable to the physical effect of administering a large bolus oral dose via gavage rather than to a direct effect of the test substance. Based on the results of the study, peptide α-1000 exhibited a very low order of toxicity, with an oral LD50 value estimated to be greater than 10 000 mg/kg BW.
In the 28-day repeat-dose oral toxicity study of peptide α-1000, reductions were observed in water consumption of test male rats; however, the decreases were not dose related and not statistically significant and, thus, did not seem to be related to treatment with peptide α-1000. The slight reduction in water consumption in the mid- and high-dose test females appeared to be mediated by isolated incidences of considerably lower individual water intakes of single females at each of the affected dose levels. Since the water intakes of all other animals in these groups were within the normal range, the changes were not attributed to peptide α-1000 treatment. Moreover, urinalysis did not reveal any abnormalities in these animals. No animals died during the study period, and no statistically significant deviations from control values were observed in body weight gains, food consumption, or hematology and clinical chemistry parameters of rats treated with peptide a-1000.
At necropsy, hemorrhage was observed in the glandular stomach of a single female in the group of rats administered peptide α-1000 at a dose of 2500 mg/kg BW per day; however, in light of the isolated nature of this effect, occurring only at the mid-dose level, this chance was considered to be a fortuitous finding, possibly caused by gavage error. In high-dose males (5000 mg/kg BW per day), a statistically significant increase was noted in the absolute and relative weights of the left kidney compared with controls; however, this change was not associated with any compound-related histopathological findings and thus was considered to be toxicologically insignificant. Although histopathology of the affected kidneys revealed inflammation and various other changes in the renal tissues (hyaline droplet degeneration, thickening of the Bowman’s capsule and basal membrane, calcium-like deposits), these occurred with similar incidence and severity in the control males. Vacuolar hyaline cast formation and degeneration and necrosis of the tubular epithelium were only observed in control animals. The renal lesions noted in the control and test males were consistent with male-specific spontaneous nephropathy commonly observed in rats 16 and, as such, were not considered to be related to the oral administration of peptide α-1000. Furthermore, occurrence of such renal nephropathy in the male rat is not considered to be predictive of a similar response in humans. 17 The highest dose level tested (5000 mg/kg BW per day) was therefore determined to be the no-observed-adverse-effect level for peptide α-1000 in rats under the conditions of this study.
This series of studies confirms the safety of the oral sardine peptide product, including specifically the absence of any sardine peptide–related mutagenic or genotoxic effects or any systemic toxicity associated with the oral administration of peptide α-1000, a closely related intermediate of the commercial product with an essentially similar amino acid composition. The results of these studies on the commercially available sardine peptide product and the closely related intermediate, combined with numerous previously conducted clinical trials demonstrating that sardine peptides do not produce any adverse health effects in humans, support the safe use in food of the peptide product derived from sardine protein hydrolysates as a dietary source of peptides under the conditions of intended use by Senmi Ekisu.
