Abstract
There is a current worldwide interest in developing novel sustainable nonanimal nutritional sources, and one such source is the green algae
Introduction
The world’s population has continued to increase while resources are becoming increasingly scarce, resulting in major searches for sustainable sources of nutrition that meet growing demands.
1
–3
Of particular interest is the lack of robust and sustainable protein sources, which continues to generate interest in new nutritional options. In order to provide the proper nutrition to a growing population, novel sources of food must inevitably be examined. One potential source of nutrition is the freshwater green microalgae
There is widespread interest in algal foods and food ingredients. 5 Various macroalgal seaweeds have been consumed as food by humans for thousands of years and some microalgae have been consumed as dietary supplements (eg, chlorella) for at least decades and possibly as food for centuries (although it is difficult to tease out of the literature whether consumption of true algal species or only various cyanobacteria occurred as the terminology, microalgae, is often applied to cyanobacteria without qualification). Nevertheless, there is a wide data gap regarding both safety and nutritional value and other beneficial effects even among widely consumed species with much resting solely on history of use. Perhaps the most valuable contribution of data on historical food uses and the current state of research is solidification of the diversity that exists among algal species and the interspecies and environmental variation in both potentially beneficial and potentially harmful constituents that affirm the need for thorough investigations of novel introductions of algal species to the food supply.
The biomass of 2 other species of green algae,
Material and Methods
Chemicals
All chemical reagents, solvents, pharmaceuticals, and other chemicals used in the studies were of analytical or pharmaceutical grade. Agar bacteriological and nutrient broth number 2 were sourced from Oxoid Ltd (Hampshire, England); minimal glucose agar plates were sourced from Merck Life Science GmbH (Eppelheim, Germany); 2-aminoanthracene (2AA), acetic acid, antibiotic–antimycotic, biotin, colchicine, cyclophosphamide monohydrate, D-glucose-6-phosphate sodium, dimethyl sulfoxide (DMSO), Dulbecco’s modified Eagle’s (DME) medium, ethyl methanesulfonate (EMS), magnesium chloride, methyl methanesulfonate, methylcellulose, N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid, and trypsin ethylenediaminetetraacetic acid solution were sourced from Sigma-Aldrich Co (Saint Louis, Missouri);
Test Item
The test item was
Compositional Analysis.
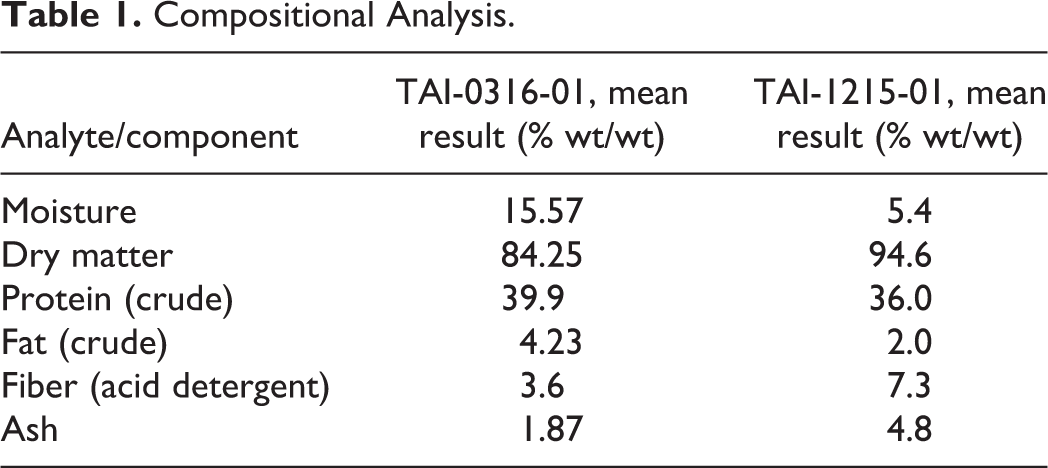
The test item is produced according to current good manufacturing practice for food ingredients using a standard closed aerobic heterotrophic fermentation process under appropriate process controls. The inoculum is
In Vitro Test Systems and Animals
The bacterial reverse mutation assay utilized bacterial tester strains
In the in vivo mammalian micronucleus test, specific pathogen-free (SPF) male Crl: NMRI BR mice (Toxi-Coop, Budapest, Hungary), 8 weeks old and weighing 31.6 to 35.3 g at the start of treatment, were utilized, and for the 28-day repeated-dose oral toxicity study, male and female SPF Hsd.Han Wistar rats (Toxi-Coop), 49 to 52 days old and weighing 207 to 238 g (males) and 124 to 152 g (females), were utilized. Animal acclimatization, housing, and environmental conditions were in accordance with the respective Organisation for Economic Co-operation and Development (OECD) test guidelines. 16,17 Feed (ssniff SM R/M-Z+H complete diet for rats and mice; ssniff Spezialdiäten GmbH, Germany) and potable tap water were provided ad libitum. Both animal studies were conducted according to standard operating procedures (SOPs) for the care and use of animals under a protocol approved by the Institutional Animal Care and Use Committee of Toxi-Coop Zrt. Additionally, the 28-day study was conducted in compliance with the National Research Council Guide for Care and Use of Laboratory Animals 18 and the principles of the Hungarian Act 2011 CLVIII (modification of Hungarian Act 1998 XXVIII) regulating animal protection.
Genotoxicity Studies
Bacterial reverse mutation test
In accordance with OECD Principles of Good Laboratory Practice (GLP)
19
and the Hungarian GLP Regulation: 42/2014 (VIII.19.) Emberi Erőforrások Minisztériuma (EMMI) decree of the Minister of Human Capacities, the mutagenic potential of
The GLP initial and confirmatory bacterial reverse mutation tests were conducted in triplicate, with and without metabolic activation, using a standard plate incorporation procedure and a preincubation procedure, respectively. Based on the preliminary test results, the tests were conducted using DMSO as the vehicle at concentrations of 5,000, 1,600, 500, 160, 50, 16, and 5 µg/plate. Positive and negative controls were chosen in accordance with the cited guideline and literature, and the sensitivity, reliability, and promutagen activation potential of the S9 metabolic activation system mix (S9-mix) were certified by the supplier using known controls and further investigated with positive control 2AA by the testing laboratory. The test solutions, positive control solutions, and the S9-mix were prepared freshly at the beginning of each experiment.
In vitro mammalian chromosomal aberration test
The clastogenic potential of
Test solution concentrations for the main chromosomal aberration assay were chosen based on a GLP preliminary cytotoxicity test, and the test solutions, positive controls, and S9-mix were freshly prepared directly prior to treatment of cells in each experiment. We have previously described the procedures for the treatment, harvesting, and preparation of the cells for analysis.
29
The main chromosomal aberration assay consisted of 2 independent experiments conducted in duplicate at the test item concentrations (positive and negative controls were also tested) and treatment/sampling intervals shown below: ▪ Experiment A: 3 -hour treatment/20-hour (∼1.5 cell cycles) sampling time. Without S9-mix: 100, 200, and 400 µg/mL With S9-mix: 150, 300, and 600 µg/mL ▪ Experiment B: 20-hour treatment/20 and 28-hour (∼2 cell cycles) sampling times. Without S9-mix: 50, 100, 200, and 400 µg/mL ▪ Experiment B: 3-hour treatment/28-hour sampling time. With S9-mix: 150, 300, and 600 µg/mL
In vivo mammalian micronucleus test
In compliance with OECD GLP
19
and the Hungarian GLP Regulation: 42/2014 (VIII.19.) EMMI decree of the Minister of Human Capacities and OECD guideline 474,
17
the in vivo genotoxic potential of
In order to determine the high dose and evaluate for possible differences in toxicity between the sexes, a preliminary toxicity test was conducted by administering a single dose of 2,000 mg/kg bw (the limit dose) of the test item by gavage to 2 male and 2 female mice and monitoring for signs of toxicity or mortality over a 2-day period. Based on the results of the preliminary toxicity test, in accordance with OECD 474,
17
the high dose was set at 2,000 mg/kg bw and a declining dose schedule was employed for the mid and low doses. Thirty-five male mice were randomly divided into 5 groups of 5 or 10 animals for dose administration as follows: ▪ The test item was administered by gavage at doses of 0 (negative control), 500, 1,000, and 2,000 mg/kg bw. ▪ The positive control was administered by intraperitoneal injection. ▪ The negative control and 2,000 mg/kg bw groups consisted of 10 analyzable animals each, and all other groups consisted of 5 animals each.
Immediately after dosing and at regular intervals until euthanasia, all mice were examined for visible reactions to treatment. As a single treatment was administered, bone marrow sampling (from femurs) was made twice according to the cited test guideline immediately following euthanasia (by cervical dislocation).
Twenty-Eight-Day Repeated-Dose Oral Toxicity Studies in Rats
The study was conducted in general accordance with OECD GLP 19 and the Hungarian GLP Regulation: 42/2014 (VIII.19.) EMMI decree of the Minister of Human Capacities and OECD 407 guideline 31 in order to obtain information on the toxic potential and estimate a no observed adverse effect level (NOAEL) for each sex.
Distilled water (aqua purificata; Parma Produkt Kft., Budapest, Hungary) was chosen as the vehicle and control due to the test item’s solubility. Dose solution concentrations of 50, 100, and 200 mg/mL were prepared freshly each day, by careful weight measurement and dissolving of the test item in the vehicle, and administered at a constant dosing volume of 20 mL/kg bw within 4 hours of preparation. The control group received the same volume of the distilled water vehicle only. Due to a lack of toxicity data in the literature and the anticipated food-like use of the
Statistical Analyses
Statistically analysis of study results, with the exception of the bacterial reverse mutation test, was conducted using SPSS PC+ software, version 4 (SPSS, Inc, Chicago, Illinois). The specific statistical tests applied and algorithms employed are described below, and a
Bacterial reverse mutation test
Colony numbers were determined by manually counting; from this mean values, standard deviations and mutation rates were calculated. No statistical evaluation of the test results was conducted as, in accordance with the guideline, biological relevance was the criterion applied for interpretation of results.
In vitro mammalian chromosomal aberration test
The χ2 test was utilized to analyze the number of aberrations and number of cells with aberrations in comparisons of treated groups, control groups, and historical data.
In vivo mammalian micronucleus test
Kruskal-Wallis nonparametric 1-way analysis of variance (ANOVA) was utilized to analyze micronucleated polychromatic erythrocytes (MPCEs).
Twenty-eight-day repeated-dose oral toxicity studies in rats
For analysis of body weight, food consumption, feed efficiency, clinical pathology, and organ weight data, variance between groups was checked with Bartlett homogeneity of variance test, and if significant heterogeneity was detected, the Kolmogorov-Smirnov test was performed to examine the distribution of data for normality. In the case of a non-normal distribution, the Kruskal-Wallis nonparametric 1-way ANOVA was conducted, and if a significant result was obtained, post hoc intergroup comparisons were conducted using the Mann-Whitney
Results
Genotoxicity Studies
Bacterial reverse mutation test
The concentration range finding test was conducted in
Similar to the concentration range finding test, in the main tests, precipitation of the test item was observed but did not interfere with scoring in any case, and no concentration-related or biologically relevant increases in the number of revertant colonies of any of the tester strains were observed at any test concentration with or without S9 under the conditions of the plate incorporation procedure (see Supplemental Table 1) or the preincubation procedure (see Supplemental Table 2). Additionally, no growth inhibition was observed at any test item concentration, with revertant colony numbers of each tester strain remaining in the range of the biological variability of the applied test system, and the background lawn development remaining unaffected.
In vitro mammalian chromosomal aberration test
The test item formed a clear homogenous suspension, and no precipitation was observed, in DME medium at 10 mg/mL in the preliminary solubility test. In the preliminary cytotoxicity test, concentrations up to 625 and 1,250 µg/mL were tested without and with metabolic activation, respectively, under all treatment and sampling conditions (data not shown). Higher concentrations were not tested due to excessive cytotoxicity, and the high concentrations for the main test were chosen as 400 and 600 µg/mL without and with metabolic activation, respectively.
In the experiments of the main test, all validation criteria of the assay were met, and no statistically significant or concentration-related increases in the number of cells with aberrations compared to the concurrent negative control or the historical negative control data of the laboratory were observed at any test item concentration under any of the conditions of experiments A or B (see Supplemental Tables 3-7). Inclusion or exclusion of gap-type aberrations did not alter the results. Treatment with the test item did not affect the rate of occurrence of polyploidy or endoreduplicated metaphases, no precipitation of the test item was observed at any of the concentrations, and no significant differences in pH or osmolality were observed in the treated cells compared to the concurrent controls (data not shown).
In vivo mouse micronucleus test
No mortality or adverse reactions to treatment were observed in the preliminary toxicity test at 2,000 mg/kg bw; therefore, the micronucleus test was conducted at doses of 0 (vehicle-control), 500, 1,000, and 2,000 mg/kg bw in male mice only. Likewise, no mortality or adverse reactions to treatment were observed in the treated or negative and positive control groups during the micronucleus test.
Evaluation of micronuclei was valid according to assay criteria. No statistically significant differences in the proportion of polychromatic erythrocytes (PCEs) to total erythrocytes in the treatment groups compared to the concurrent negative control were observed, with all treated groups remaining within 90% of the control value. However, the slight nonsignificant decrease in the ratio of PCEs to total erythrocytes in the 2,000 mg/kg bw group compared to concurrent negative control at both sampling times was considered an indication of bone marrow exposure to the test item. No statistically significant dose-related increases in frequency of MPCEs were observed in the treated groups compared to the concurrent or historical negative controls (see Supplemental Table 8). A statistically significant decrease compared to concurrent negative control was observed in the 1,000 mg/kg bw group; however, frequencies of MPCEs in all treated groups remained within the distribution of historical negative control data at all sampling times.
Twenty-Eight-Day Repeated-Dose Oral Toxicity Study in Rats
No mortality occurred during the treatment period at any of the tested dose levels (0, 1,000, 2,000, or 4,000 mg/kg bw/day) in either sex nor were any clinical signs or abnormal behavior observed during daily or weekly detailed clinical observations or during conduction of the FOB during the last week of treatment (data not shown). Furthermore, grip strength, motor activity, and reactions to stimuli were considered normal, without alterations, during the course of the FOB. No alterations were observed on ophthalmologic examinations of all animals prior to treatment period or during the last week of treatment (data not shown).
No statistically significant differences in mean body weights or food consumption were observed compared to respective controls in the treated male and female groups or in mean body weight gains and feed efficiency in the male groups. A few slight, but statistically significant, transient decreases in mean body weight gains and feed efficiency were observed in various female groups compared to controls; however, these did not affect cumulative body weight gain or feed efficiency (data not shown).
Statistically significant changes in several clinical pathology parameters were observed with respect to controls among the sexes and dose groups as shown in Table 2 (see also, Supplemental Tables 9 and 10 for full hematology and clinical chemistry group data). On gross examination at necropsy, thymic hemorrhages were observed in 2 male animals of the 1,000 mg/kg bw/day group and 1 female control group animal (data not shown). Dilatation of the uterine lumen was observed on examination of the uteri of 1, 2, 3, and 2 female animals of the control, low-, mid-, and high-dose groups, respectively. No other gross lesions were observed in any animals.
Summary of Statistically Significant Hematology and Clinical Chemistry Parameters.
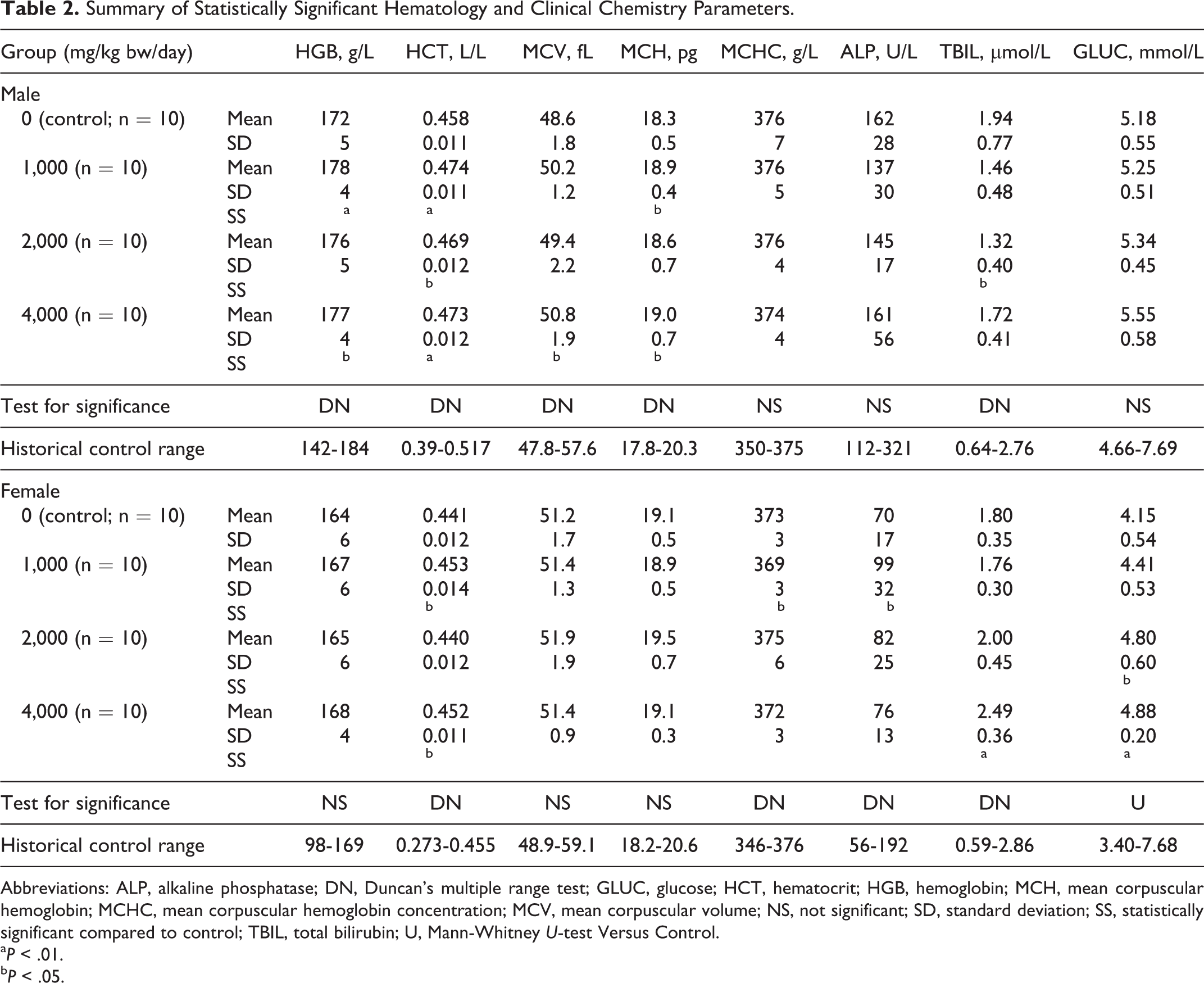
Abbreviations: ALP, alkaline phosphatase; DN, Duncan’s multiple range test; GLUC, glucose; HCT, hematocrit; HGB, hemoglobin; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; NS, not significant; SD, standard deviation; SS, statistically significant compared to control; TBIL, total bilirubin; U, Mann-Whitney
a
b
In female animals, there were no statistically significant differences in organ weights relative to body weights or organ weights relative to brain weights in the treated groups compared to the control groups. A single statistically significant alteration was observed for mean liver weight relative to body weight in 4,000 mg/kg bw/day group males with the appearance of a potential dose relationship (see Supplemental Table 11). Additionally, a few sporadic fluctuations observed in absolute (males and females) and relative (males only) organ weights were statistically significant as follows: decreased mean heart weights (absolute and relative to brain weight) were observed in the 1,000 mg/kg bw/day group males (0.91 ± 0.05 g and 45.41% ± 3.31%, respectively) compared to controls (1.00 ± 0.09 g and 49.93% ± 2.84%, respectively), decreased mean weights of the prostate with seminal vesicles and coagulating glands attached (absolute, relative to body, and relative to brain) were observed in the 4,000 mg/kg bw/day group males (1.53 ± 0.23 g, 0.461% ± 0.057%, and 75.3% ± 9.89%, respectively) compared to controls (1.80 ± 0.18 g, 0.551% ± 0.075%, and 90.8% ± 10.8%, respectively), and decreased mean absolute brain weight was observed in the 4,000 mg/kg bw/day group females (1.79 ± 0.07 g) compared to controls (1.90 ± 0.07 g; data not shown).
A few sporadic changes were observed histologically with similar frequencies among controls and treated animals and are summarized in Table 3. No evidence of toxic injury (eg, inflammation, degeneration, necrosis) was observed in the histological morphology of any other tissues or organ systems (eg, cardiovascular, endocrine, hematopoietic, immune, musculoskeletal, central or peripheral nervous systems, or male or female reproductive systems) of the control or 4,000 mg/kg bw/day groups (data not shown).
Summary of Histopathology Findings.a
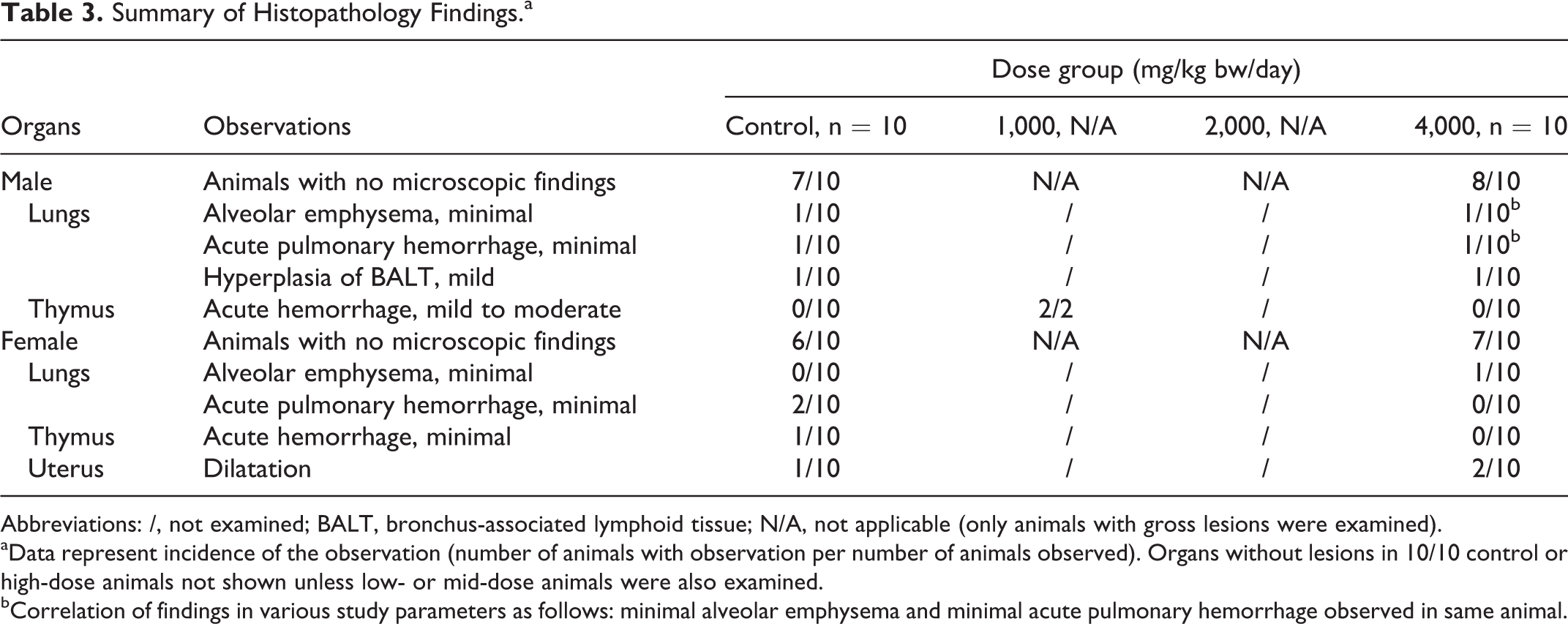
Abbreviations: /, not examined; BALT, bronchus-associated lymphoid tissue; N/A, not applicable (only animals with gross lesions were examined).
aData represent incidence of the observation (number of animals with observation per number of animals observed). Organs without lesions in 10/10 control or high-dose animals not shown unless low- or mid-dose animals were also examined.
bCorrelation of findings in various study parameters as follows: minimal alveolar emphysema and minimal acute pulmonary hemorrhage observed in same animal.
Discussion and Conclusions
Toxicological investigations of several members of the green algae genus
Nonetheless, the test item would be a novel introduction to the food supply, and to the best of our knowledge, this is the first toxicological evaluation of
The ability to score the bacterial reverse mutation test was not affected by observed precipitates, and observed variations in the initial and confirmatory tests were within the range of biological normality. Therefore, the test was considered unequivocally negative, as we have demonstrated that the test item lacks the ability to cause point or frameshift mutations under the applied conditions.
Likewise, the experimental results of the in vitro chromosomal aberration test and the in vivo mammalian micronucleus test were unequivocally negative, demonstrating a lack of potential of the test item to induce chromosomal damage or genotoxicity, respectively, under the applied conditions.
No findings of toxicological interest were observed in the current 28-day oral toxicity study. The observed clinical pathology changes (Table 2), although statistically significant, remained within the historical control ranges of the laboratory, were absent of correlating pathological findings, and with few exceptions (bilirubin and glucose in females), occurred without clear dose relationships; thus, they were considered reflective of normal biological variation and unrelated to administration of the test item.
While toxic alterations in organ weights can occur early on, prior to morphological changes, and the liver to body weight ratio can be predictive of toxicity, 42 in the current study the statistically significant, dose-related elevation of liver weight relative to body weight observed in high-dose females was low in magnitude relative to the controls (approximately 8%) and well within the laboratory’s historical control range (see Supplemental Table 11), and a concomitant significant rise in absolute liver weight was absent but may have been expected if a toxic effect was present given the lack of significant differences in body weight or food consumption. Because of this and the lack of correlating findings in histopathology or clinical chemistry parameters, the alteration was judged as occurring without toxicological relevance. Although statistically significant decreases with respect to controls were observed in both absolute and relative heart and prostate weights in the male low- and high-dose groups, respectively, these changes remained well within historical control ranges and were not dose related or correlated with findings in other study parameters. Therefore, the observed variations were not considered to be test item-related.
Acute hemorrhages of the thymus, observed at necropsy and also on histopathological examination, and acute pulmonary hemorrhages and alveolar emphysema observed on histopathological examination were considered due to the exsanguination procedure during which circulatory disturbances, dyspnea, and associated hypoxia occur. These finding are commonly observed in untreated animals in our experience as well as documented in the literature 43 –45 and, in the current work, were observed with low frequencies among the sexes and also in control animals.
Dilatation of the uterine lumen was observed in a few animals in all female dose groups on macroscopic examination, but was not dose-related, and was also observed with similar frequencies in control and high-dose females microscopically. This was considered a normal estrogenic effect associated with the estrous cycle, 46,47 rather than an estrogenic effect of the test item, due to its low frequency of occurrence, lack of dose–response, similar occurrence in control animals, and the lack of correlating pathological effects in related organs.
Hyperplasia of the bronchus-associated lymphoid tissue (BALT) is an antigenic response that also occurs in untreated animals. 48,49 In the current work, hyperplasia of the BALT was observed with mild degree and without associated inflammatory lesions in only a single animal each of the control and high-dose male groups and was considered likely due to commensal flora or nonliving antigens. Based on these reasons, the sporadic lesions observed on the gross and histopathological examinations were considered lacking in any toxicological relevance and unrelated to the test item.
In summary, on consideration and interpretation of the results of this toxicological evaluation, we conclude that
Footnotes
Acknowledgments
The authors thank the following individuals for their contributions to the work: participating investigators Erzsébet Biczó, Erika Major Biermanné, Ibolya Bogdán, Katalin Böröczki, Tamás Buda, Tímea Csörge, Kata Eszter Diószegi, Jánosné Dózsa, Stella Fekete, Zsuzsanna Frank, Irén Somogyi Háriné, Ildikó Hermann, Brigitta Horváth, Istvánné Horváth, Kornélia Sereg Jurácsikné, Klára Fritz Kovácsné, Nóra Pongrácz Kurdiné, Adrienn Laczó, Marcell Madár, Edit Kövári Mesterháziné, Mónika Fekete, Viktória Polgár-Balogh, János Stáhl, Beatrix Szilágyi, Eva Láng-Szabó, Zsuzsanna Szabó, Mariann Lennert Szabóné, Edit Szám, Márta Tenk, and Ruth Weimer for the performance of experimental tasks, collection of data, and/or quality assurance; and Jared Brodin for administrative support in preparation of the manuscript.
Authors’ Contributions
T. S. Murbach contributed to conception, contributed to analysis and interpretation of data, drafted the manuscript, and critically revised the manuscript. R. Glávits contributed to analysis and interpretation of data and critically revised the manuscript. J. R. Endres contributed to conception and design, contributed to analysis and interpretation of data, and critically revised the manuscript. G. Hirka contributed to conception and design, contributed to analysis and interpretation of data, and critically revised the manuscript. A. Vértesi contributed to conception and design, contributed to acquisition, analysis, and interpretation of data, contributed to drafting of the manuscript, and critically revised the manuscript. E. Béres contributed to conception and design, contributed to acquisition, analysis, and interpretation of data, drafted the manuscript, and critically revised the manuscript. I. P. Szakonyiné contributed to conception and design, contributed to acquisition, analysis, and interpretation of data, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AIBMR Life Sciences, Inc (Seattle, Washington) was contracted by the study sponsor, as an independent third party, to determine appropriate study protocols and dose selections, place the studies, approve the study plans, and monitor the toxicological studies herein described and to analyze and interpret the resulting data and prepare the manuscript. Toxi-Coop Zrt. (with test facilities in Budapest [28-day study] and Balatonfüred [genotoxicity studies], Hungary) was contracted by AIBMR to develop the study plans and conduct, analyze, and interpret, and report the results of the toxicological studies herein described.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose that financial support for the research described herein was provided by Triton Algae Innovations, San Diego, California.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
