Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of 10 Rosmarinus officinalis (rosemary)-derived ingredients and concluded these ingredients are safe as used in cosmetics when formulated to be nonsensitizing. The R officinalis-derived ingredients are most frequently reported to function in cosmetics as skin conditioning agents or as fragrance ingredients. The Panel reviewed the available animal and clinical data to determine the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should continue to use good manufacturing practices to limit impurities that could be present in botanical ingredients.
Introduction
This report reviews the use and safety data of the following 10 Rosmarinus officinalis (rosemary)-derived ingredients as used in cosmetics: Rosmarinus Officinalis (Rosemary) Extract Rosmarinus Officinalis (Rosemary) Flower Extract Rosmarinus Officinalis (Rosemary) Flower/Leaf Stem Extract Rosmarinus Officinalis (Rosemary) Flower/Leaf/Stem Water Rosmarinus Officinalis (Rosemary) Leaf Rosmarinus Officinalis (Rosemary) Leaf Extract Rosmarinus Officinalis (Rosemary) Leaf Oil Rosmarinus Officinalis (Rosemary) Leaf Powder Rosmarinus Officinalis (Rosemary) Leaf Water Rosmarinus Officinalis (Rosemary) Water
Most of the ingredients included in this review are extracts, oils, powders, or waters derived from a defined part of the R officinalis (rosemary) plant.
R officinalis (rosemary)-derived ingredients are reported to have a number of functions, and the most common functions in cosmetics are as a skin conditioning agent or use as a fragrance ingredient. 1 Two of the ingredients, that is, rosmarinus officinal is (rosemary) flower extract and rosmarinus officinalis (rosemary) leaf extract, are reported to function as antioxidants. However, rosmarinus officinalis (rosemary) leaf powder is reported to function only as a flavoring agent.
Chemistry
Definition
The definition of each R officinalis (rosemary)-derived ingredient indicates what part(s) of the plant from which the ingredient is obtained (Table 1). In some cases, the definition also gives insight as to the method of manufacture.
Definitions and Reported Functions.

Abbreviation: misc, miscellaneous.
General Characterization
The R officinalis L. plant, from the botanical family Lamiaceae, is a scented, evergreen shrub with a very pungent odor that is native to the Mediterranean region and Portugal; the odor is sometimes defined as camphor-like. 2,3 Rosemary has a spicy, harsh, bitter, aromatic taste. Bluish labiate flowers grow on the upper green part of the branches. Rosemary oil is produced mostly in Spain, France, and Tunisia. 4
R officinalis L. is generally recognized as safe (GRAS) in foods as a spice and as a natural seasoning and flavoring (21CFR182.10). Rosemary has traditional or folk medicine uses, some with reported side effects. 2,5,6 The flowering dried twig tips, the dried leaves, the fresh leaves, the fresh aerial parts, and the flowering branches are considered to be the medicinal parts. 5
Chemical and Physical Properties
R officinalis (rosemary)-derived ingredients are strongly aromatic. Chemical and physical property data are provided in Table 2.
Chemical and Physical Properties.

Preparation/Extraction
Food-grade rosmarinus officinalis (rosemary) extract is prepared by extraction from the leaves of R officinalis. Food-grade acetone, ethanol, hexane, or a combination of hexane and ethanol (in a 2-step process) are used as extraction solvents; the ethanol extract is sometimes deodorized or partially deodorized ethanol. 7,8 Food-grade rosmarinus officinalis (rosemary) extract may also be extracted using supercritical carbon dioxide (CO2). Subsequent production steps include filtration, purification, solvent evaporation, drying, and sieving; the extract may be deodorized, decolorized, and standardized using diluents and carriers that are permitted in foods.
An additional method of manufacturing the cosmetic ingredients includes extraction with absolute ethanol (resulting in what has been called “an absolute”) or a collection of the insoluble waxes (resulting in what has been called “a concrete”). 9
Both rosmarinus officinalis (rosemary) leaf extracts and rosmarinus officinalis (rosemary) leaf oil can be produced by supercritical fluid extraction with natural CO2 and a small amount of ethanol as a solvent. 10 -13 One supplier of the leaf extract reported that the essential oil is removed by multistep separation, 12 and a supplier of the leaf oil adds a small amount (<4%) of sunflower oil to increase solubility when blending. 13
Food-grade R officinalis (rosemary) leaf oil is the volatile oil obtained by steam distillation from the fresh flowering tops or dried crushed aerial parts of R officinalis L. 7 The oil from R officinalis can also be obtained by hydrodistillation of dried crushed aerial parts. 14 Essential oils prepared by a steam distillation process yields 2 distinct fractions, a water-insoluble fraction and a water-soluble fraction. 1 The water-insoluble fraction contains the term oil in the name, and the water-soluble fraction contains the term water in the name. Therefore, rosmarinus officinalis (rosemary) leaf water is the water-soluble fraction of the steam distillation of R officinalis (rosemary) leaves.
Constituents/Impurities
R officinalis L. is composed of an array of constituents, primarily phenolic acids, flavonoids, monoterpenes, diterpenes, diterpenoids, and triterpenes. Structures for some of the principal components according to chemical family are depicted in Figures 1 to 5.
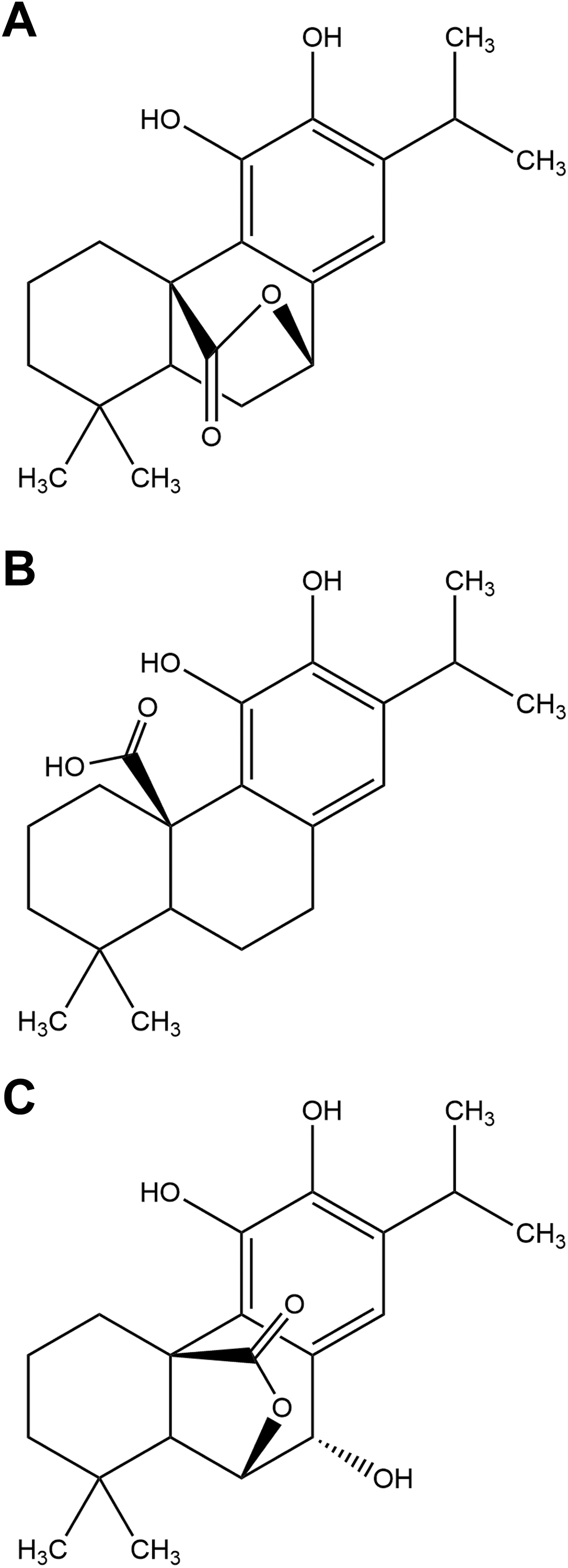
Principal diterpenes. A, Carnosol. B, Carnosic acid. C, Rosmanol.
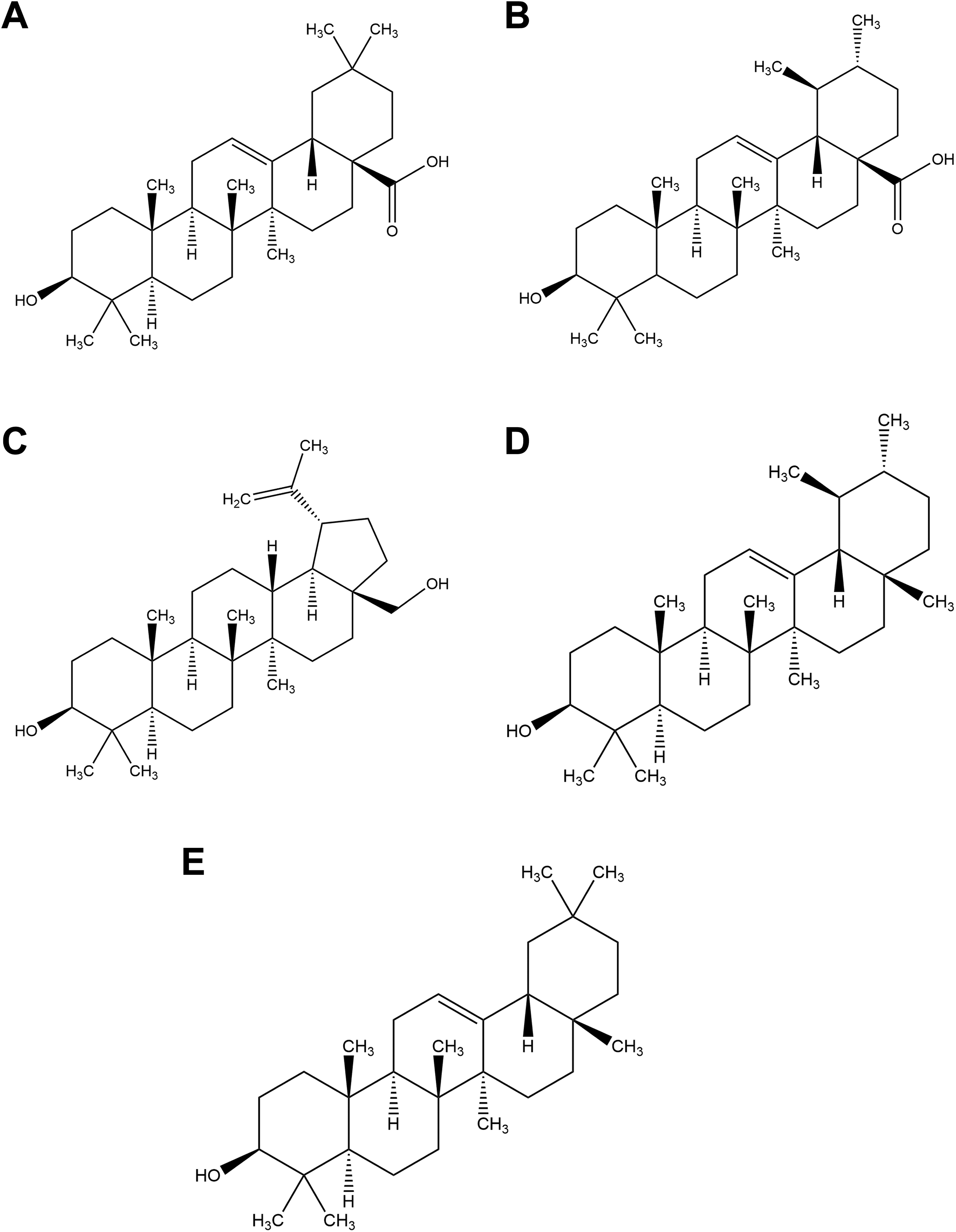
Principal triterpenes. A, Oleanolic acid. B, Ursolic acid. C, Betulin. D, α-Amyrin. E, β-Amyrin.
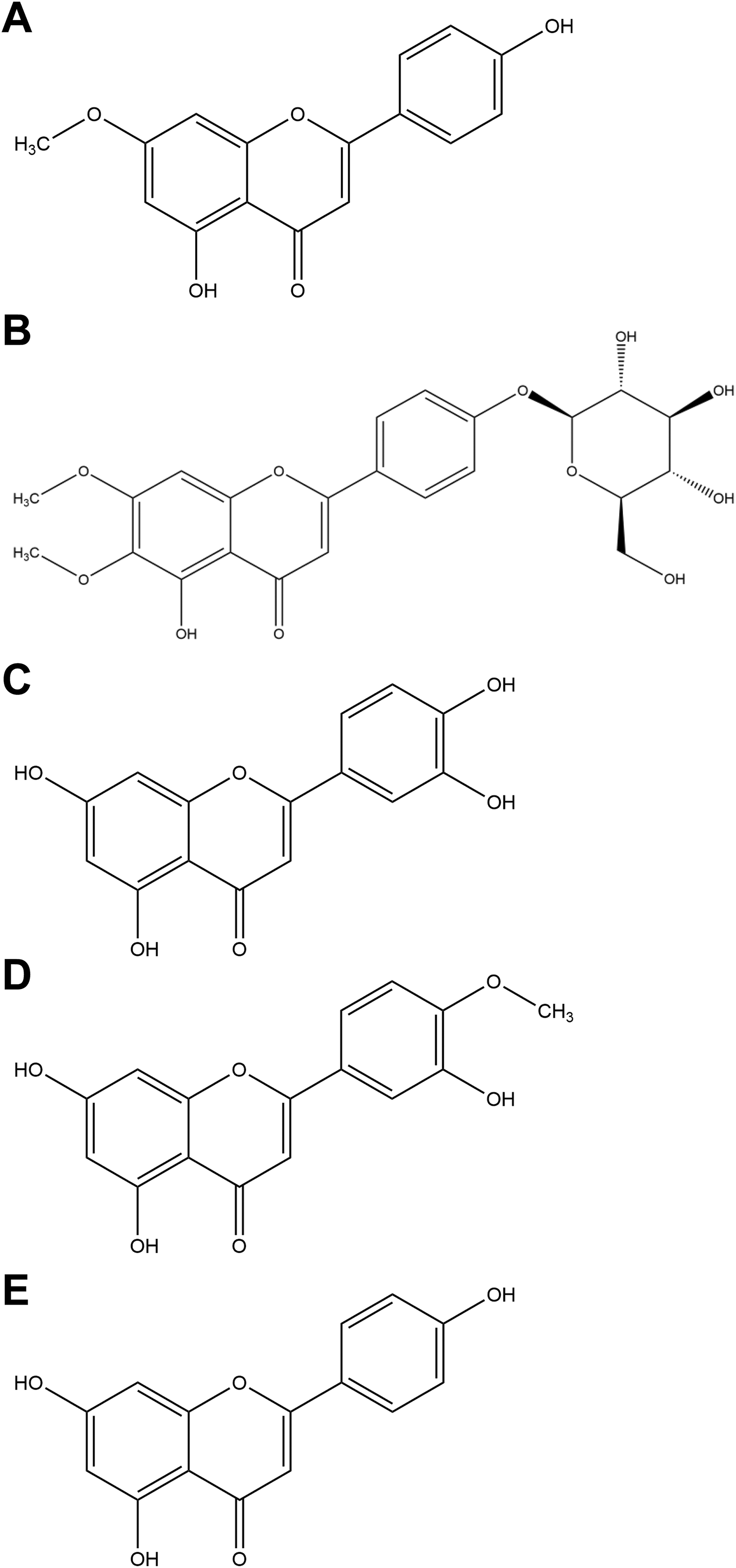
Principal flavonoids. A, Genkwanin. B, Cirsimarin. C, Luteolin. D, Diosmetin. E, Apigenin.
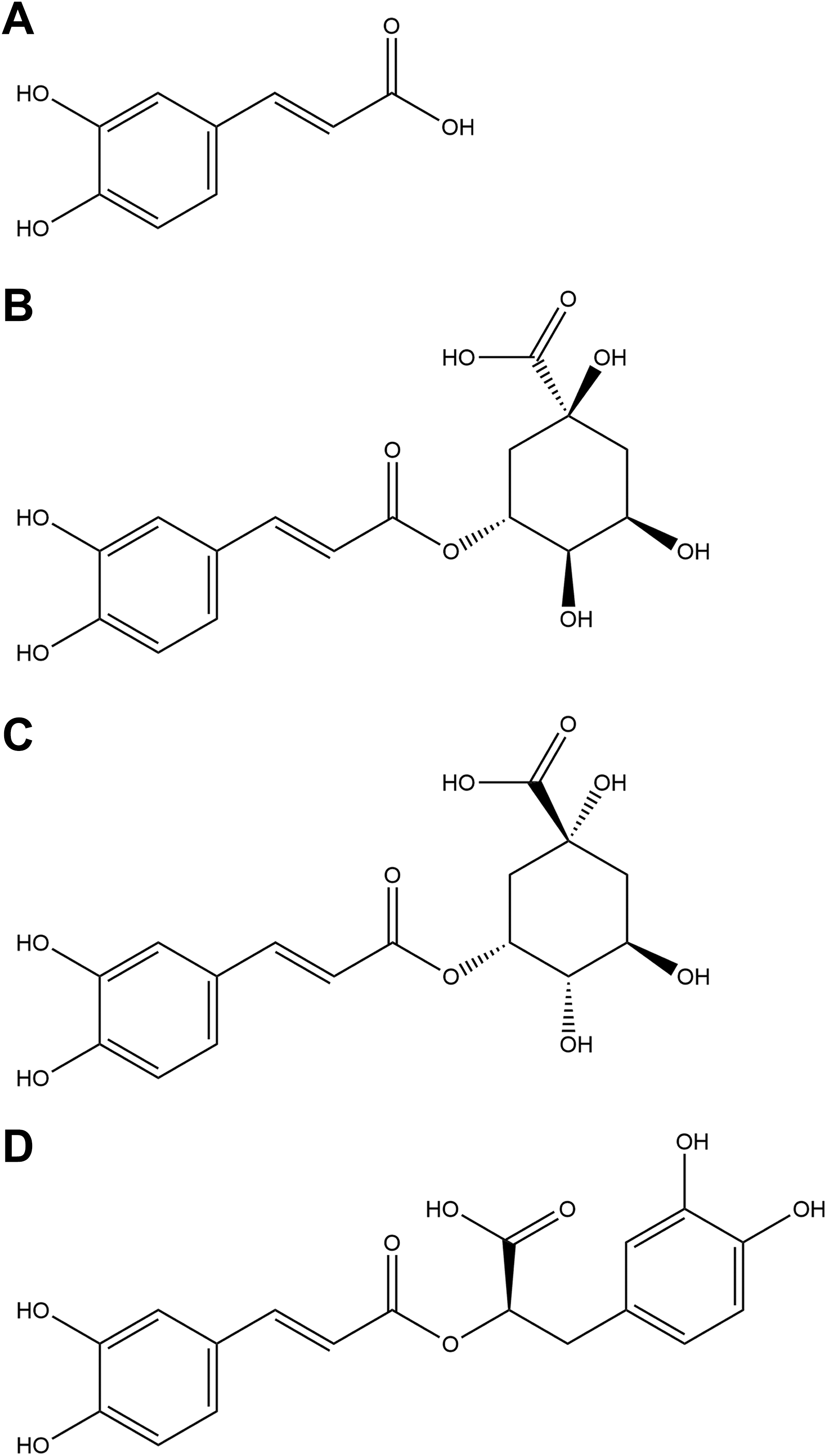
Phenolic acids. A, Caffeic acid. B, Chlorogenic acid. C, Neochlorogenic acid. D, Labiatic acid.
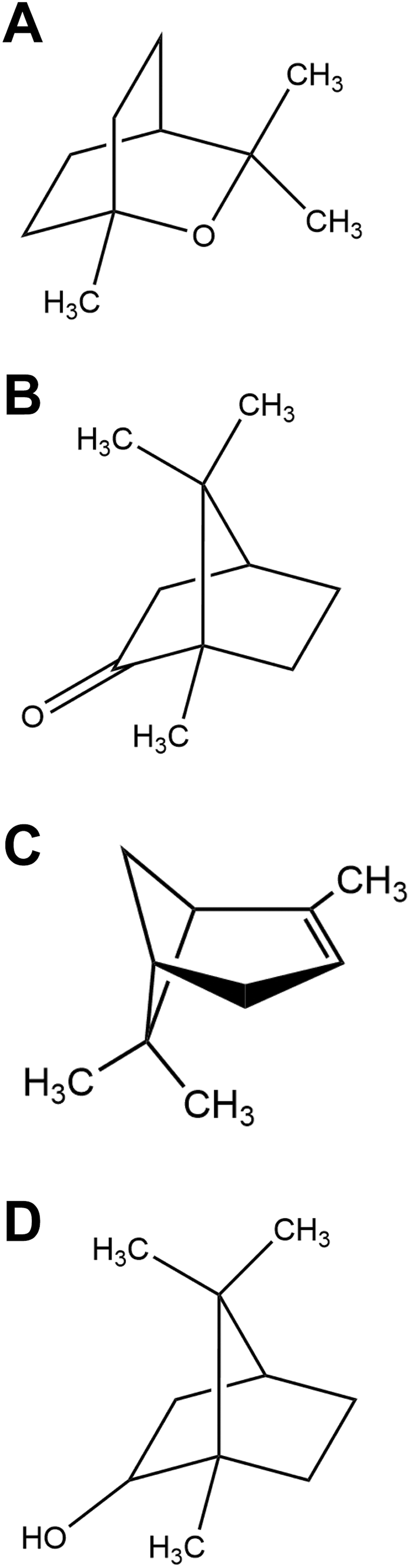
Principal volatiles. A, 1,8-Cineole. B, Camphor. C, α-Pinene. D, Borneol.
A detailed list of chemical constituents by plant part is presented in Table 3, and a more focused listing of constituents of R officinalis is provided in Table 4. Table 5 provides composition data on 3 R officinalis (rosemary) leaf extracts, based on certificates of analysis provided by suppliers of rosmarinus officinalis (rosemary) leaf extract; these certificates report a phenolic diterpenes content of 14% or 25%. 15 -18
Chemical Constituents by Plant Part (ppm). 74,a
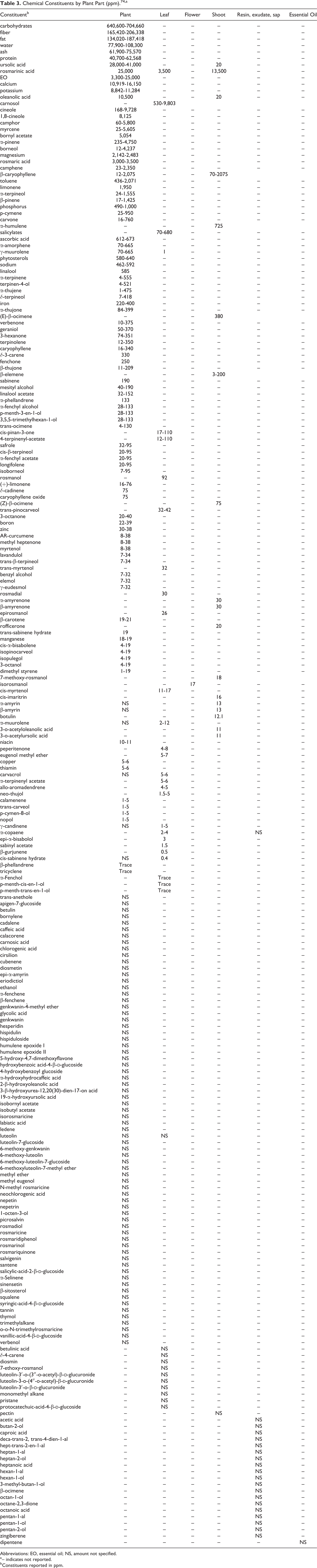
Abbreviations: EO, essential oil; NS, amount not specified.
a – indicates not reported.
b Constituents reported in ppm.
Constituent Data by Plant Part.

Abbreviation: DMSO, dimethyl sulfoxide.
Rosmarinus officinalis (Rosemary) Leaf Extracts (CO2 Extract): Certificates of Analysis.
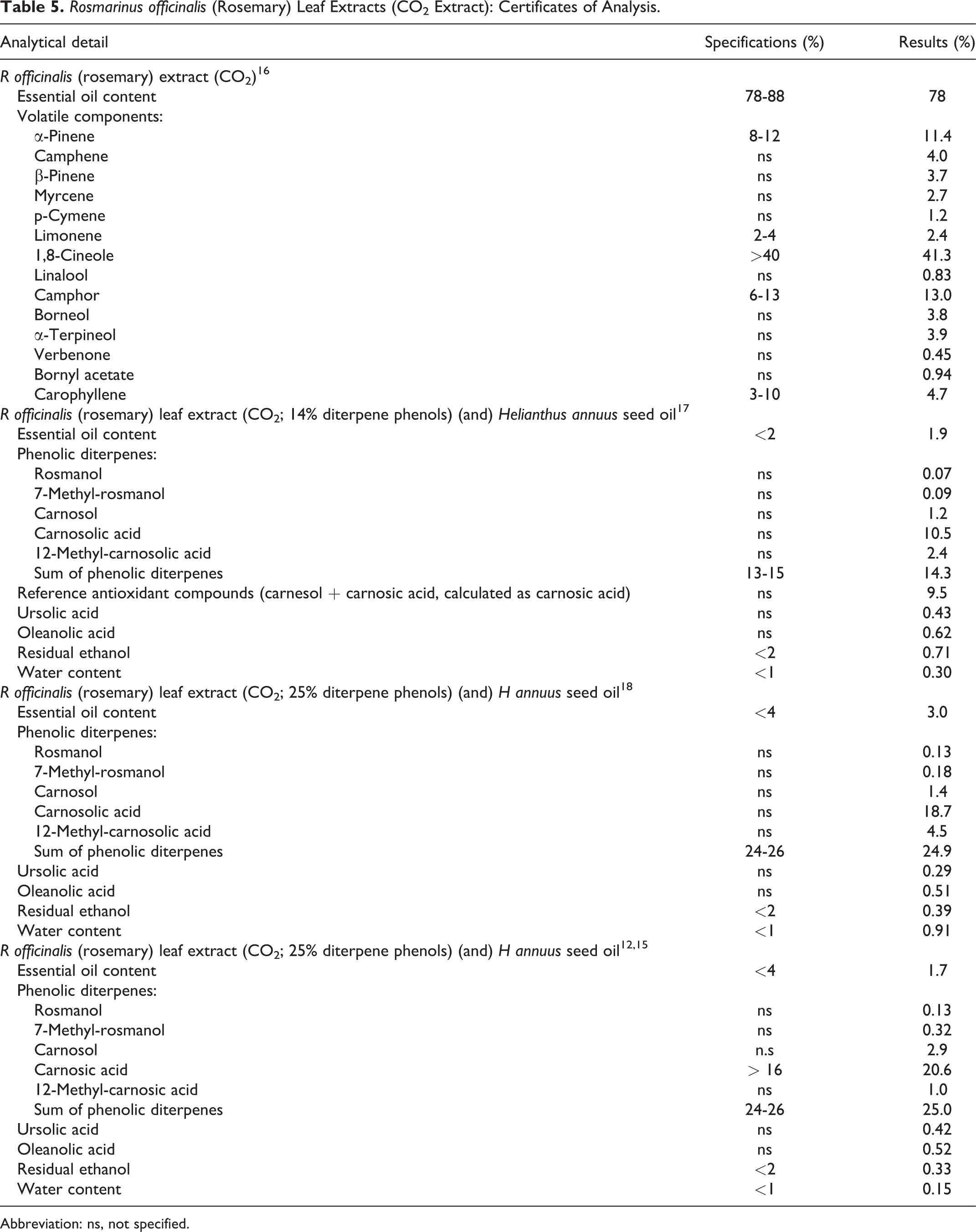
Abbreviation: ns, not specified.
According to the European Cosmetic Regulations, certain fragrance allergen compounds are subject to declaration on the label if the concentration of a specified allergen exceeds 0.001% in leave-on and 0.01% in rinse-off products.
19
One supplier declared the following concentrations of allergen compounds in a rosmarinus officinalis (rosemary) leaf extract: <0.1% linalool and <0.2%
The principal antioxidative components of rosmarinus officinalis (rosemary) leaf extract are the phenolic diterpenes carnosol and carnosic acid. 8 The amount of carnosol and carnosic acid present in the extract varies with the method of extraction, with levels as low as 5% to 7% carnosol plus carnosic acid found in rosemary extract prepared from a partially deodorized ethanol extract of rosemary to as high as 30% carnosol plus carnosic acid in an extract prepared with supercritical carbon dioxide. 2,7
Carnosol and carnosic acid are not the only constituents that vary with extraction method. Table 6 provides a sample of the differences in constituent profiles in rosemary leaves based on extraction method. Some of the studies summarized in this safety assessment provided information on the amount of constituents present in the test article; when this information was available, it is included.
Differences in Constituent Profiles in Rosmarinus officinalis (Rosemary) Leaf Extract Based on Extraction Method.a,8
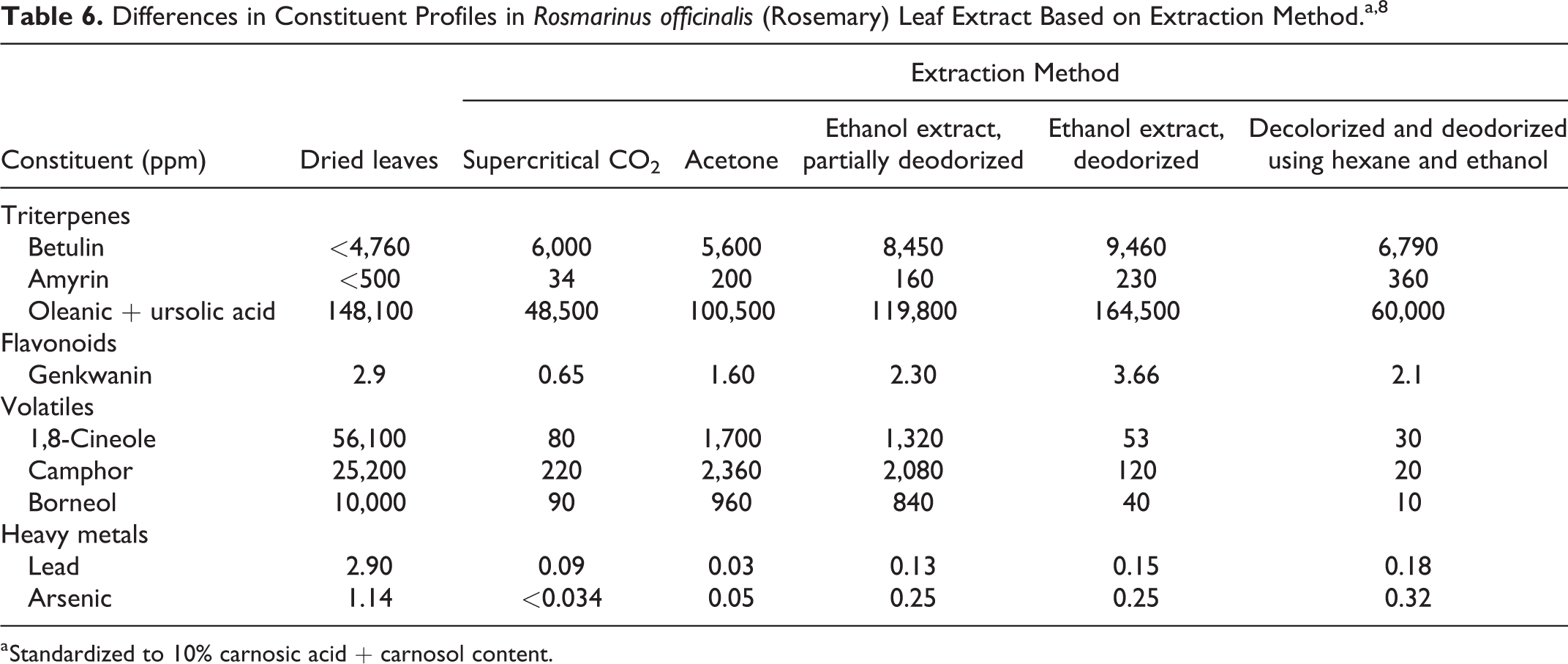
a Standardized to 10% carnosic acid + carnosol content.
The actual amount of constituents present also varies according to the stage of development, variety of plant, season harvested, and origin of the leaves. 2,8,21,22 High-performance liquid chromatography analysis of dimethyl sulfoxide extracts of rosemary leaves indicated the highest accumulation rate of the phenolic diterpenes carnosic acid, carnosol, and 12-O-methylcarnosic acid, of rosmarinic acid, and of the flavones genkwanin and isocutellarein 7-O-glucoside was found in the young stages of plant development. 23 The diterpenes and rosmarinic acid, but not the flavones, were found in the flower, stem, and root extracts at lower concentrations than in the leaves during the early stages of plant growth, but the concentration of each, except for 12-O-methylcarnosic acid, tended to increase during flowering. Rosmarinic acid concentrations in the leaves also decreased once flowering started, while the level in the flowers was slightly increased during flowering. The flavones acted similarly to carnosic acid.
Water and light conditions also affect the amount of the constituents found in rosemary plants; for example, highly oxidized diterpenes increase in rosemary plants exposed to drought and high light stress. 24 Although it is generally accepted that the geographical region and stage of growth affects plant composition, some researchers reported that, within one country, the chemical composition of rosemary essential oil (plant parts not specified) did not vary with geographical region or harvest time. 25
Food-grade rosmarinus officinalis (rosemary) leaf extract has acceptance criteria of not more than 3 mg/kg arsenic and 2 mg/kg lead and not more than 8.0% loss on drying. 7 Food-grade rosemary leaf oil is to have not less than 8.0% borneol and not less than 1.5% esters, calculated as bornyl acetate. 7
Table 7 provides toxicity and other information on some constituents of R officinalis (rosemary)-derived ingredients.
Toxicity Information on Constituents of Rosmarinus officinalis (Rosemary).

Abbreviations: IARC, International Agency for Research on Cancer; MMC, mitomycin-C; NOAEL, no-observable-adverse-effect level; TPA, 12-tetradecanoylphorbol-13-acetate; UV, ultraviolet; wt, weight.
Use
Cosmetic
The R officinalis (rosemary)-derived ingredients included in this safety assessment have a variety of functions in cosmetics (Table 1). Most of the ingredients function as a skin conditioning agent and/or as a fragrance ingredient; rosmarinus officinalis (rosemary) leaf powder is reported to function only as a flavoring agent. 1
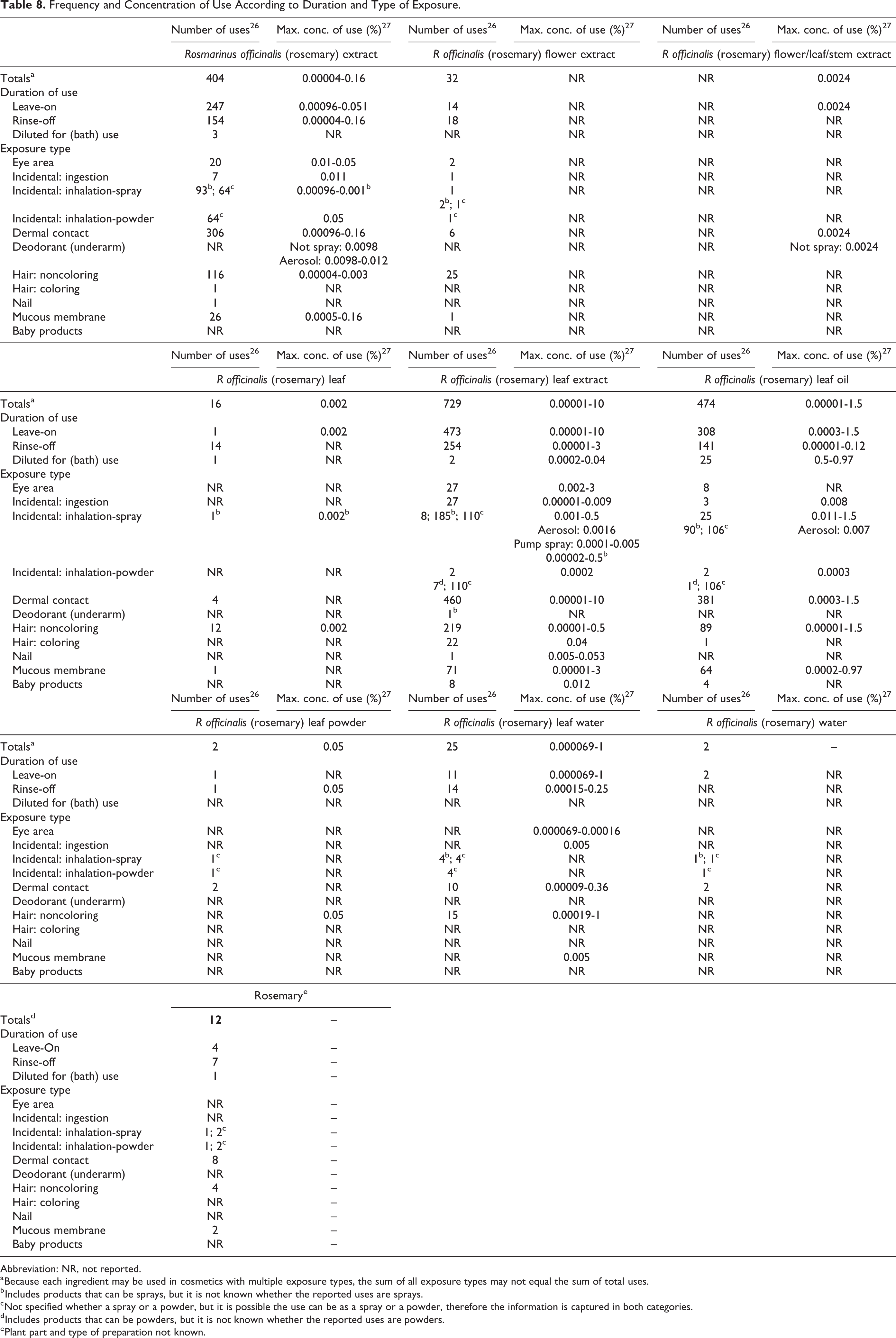
The US Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2014, 26 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 27,28 in 2013, indicating that 9 of the 10 ingredients included in this safety assessment are currently used in cosmetic formulations (Table 8). Rosmarinus officinalis (rosemary) leaf extract has the greatest number of uses, 729, followed by rosmarinus officinalis (rosemary) leaf oil, 474 uses, and rosmarinus officinalis (rosemary) extract, 404 uses. According to the results of the concentration of use survey, most cosmetic formulations contain very low concentrations of the R officinalis (rosemary)-derived ingredients, often much less than 0.1%. However, R officinalis (rosemary) leaf extract is reported to be used at up to 10% in body and hand products and 3% in eye shadow formulations and bath soaps and detergents. Rosmarinus officinalis (rosemary) flower/leaf/stem water is the only ingredient not reported to be used.
Frequency and Concentration of Use According to Duration and Type of Exposure.
Abbreviation: NR, not reported.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b Includes products that can be sprays, but it is not known whether the reported uses are sprays.
c Not specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories.
d Includes products that can be powders, but it is not known whether the reported uses are powders.
e Plant part and type of preparation not known.
In some cases, reports of uses were received in the VCRP, but concentration of use data were not provided. For example, rosmarinus officinalis (rosemary) flower extract is reported to be used in 32 cosmetic formulations, but no use concentration data were reported. In other cases, no uses were reported in the VCRP, but concentration of use data were received from industry; R officinalis (rosemary) flower/leaf/stem extract had no reported uses in the VCRP, but a use concentration in a deodorant was provided in the industry survey. Therefore, it should be presumed there is at least one use in a deodorant formulation.
Products containing R officinalis (rosemary)-derived ingredients may be applied to baby skin (eg, 0.012% R officinalis [rosemary] leaf extract in baby lotion, oils, and creams), used in products that could be incidentally ingested (eg, 0.012% rosmarinus officinalis [rosemary] leaf in lipstick formulations), or used near the eye area (eg, up to 3% rosmarinus officinalis [rosemary] leaf extract in eye shadow formulations) or mucous membranes (eg, up to 3% rosmarinus officinalis [rosemary] leaf extract in bath soaps and detergents). 27 Additionally, R officinalis (rosemary)-derived ingredients are used in cosmetic sprays and powders; for example, rosmarinus officinalis (rosemary) leaf extract is reported to be used in other fragrance preparations at up to 0.5% and rosmarinus officinalis (rosemary) extract is used in face powders at up to 0.05%. These products could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm. 29 -32 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 29,32
Rosmarinus officinalis (rosemary) extract is used in aerosol deodorants at concentrations up to 0.012%. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 29 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
All of the ingredients named in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 33
Noncosmetic
Rosmarinus officinalis L. is GRAS as a spice and as a natural seasoning and flavoring when the intended use is for human consumption (21CFR182.10) and for animal drugs, feed, and related products (21CFR582.10). It is also GRAS as an essential oil, oleoresin (solvent free), and natural extractive (including distillates) for human consumption (21CFR182.20) and for animal drugs, feed, and related products (21CFR582.20). Rosemary oil can be used in the formulation of denatured alcohol and rum (27CFR21.65).
According to The Official Journal of the European Union, extracts of rosemary contain several antioxidant compounds, and although the European Food Safety Authority (EFSA) was not able to establish an acceptable daily intake due to insufficient toxicological data, the EFSA considered the margin of safety was high enough to conclude that dietary exposure was not a concern. 34 Extracts of rosemary are allowed in various food products at amounts of 30 to 1,000 mg/kg, expressed as the sum of carnosol and carnosic acid.
Rosemary leaves are used as a seasoning in cooking. 35 Rosmarinus officinalis (rosemary) leaf oil is used as a condiment and flavoring agent in food; as an antioxidant in edible oils, meats, and other fat-containing foods; and as a dietary supplement. Also, rosemary oil is reported to have antimicrobial activities. 4
Anti-inflammatory, antioxidant, and antimicrobial uses have been reported for rosemary. 21,36 -38 Rosemary has traditional or folk medicine uses, some with negative reported side effects. 2,5,6 Rosemary has been used as an antispasmodic in renal colic and dysmenorrhea, and it has been used for relieving respiratory disorders. The essential oil is used internally as a carminative and as an appetite stimulant; however, large amount of the oil are reported to cause gastroenteritis and nephritis. The essential oil is added to bath water as a circulation stimulant. As the oil or as an ointment, external application use is as an analgesic liniment for rheumatism. Rosemary is used as a poultice for poorly healing wounds and in the treatment of eczema. Additional folk medicine practices include use in lotions to treat baldness 14 and use of the leaves and branches in treating headaches. 4
Toxicokinetics
Penetration Enhancement
The effect of rosemary oil on the permeation of aminophylline was determined in human skin in vivo using attenuated total reflection Fourier transform infrared spectroscopy. 39 Rosemary oil did enhance the permeation of aminophylline; however, the increase in permeation was less than that observed with 50% ethanol.
Toxicological Studies
Single-Dose (Acute) Toxicity
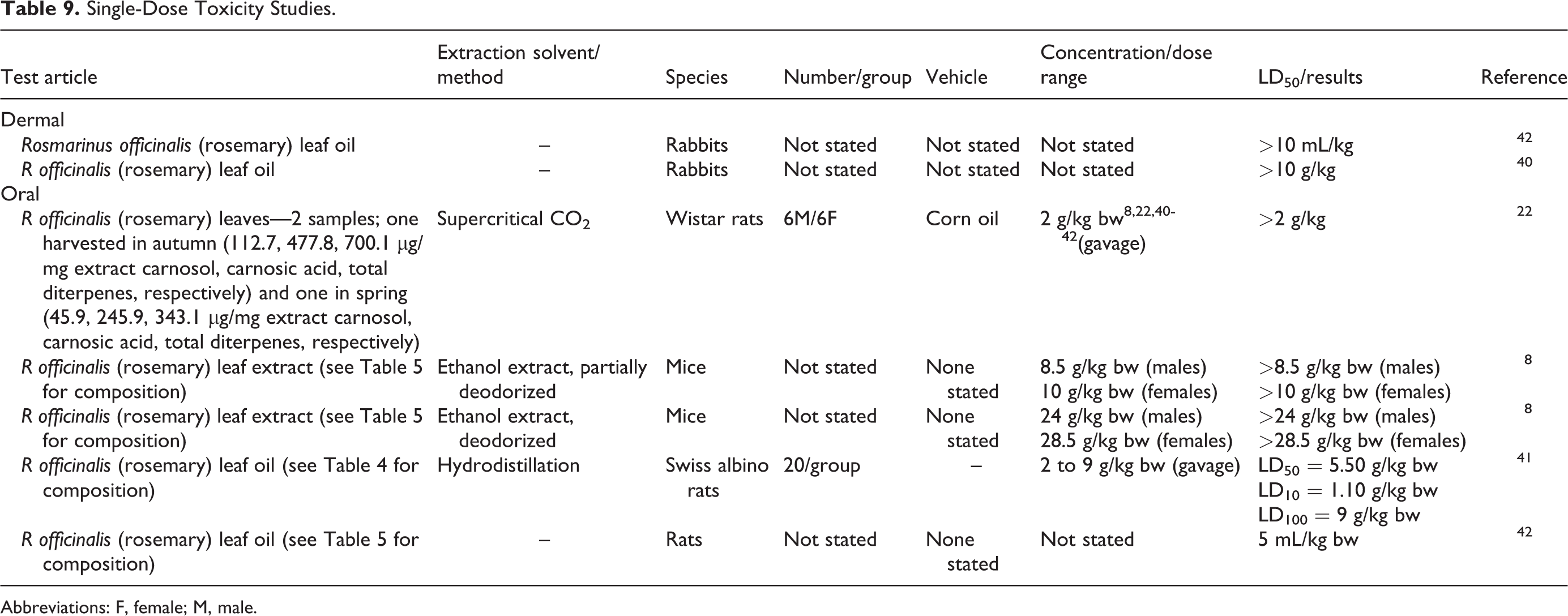
The acute toxicity of R officinalis (rosemary)-derived ingredients is not very remarkable (Table 9). 8,22,40 -42 The dermal the median lethal dose (LD50) of rosmarinus officinalis (rosemary) leaf oil is >10 mL/kg. 42 The oral LD50 of rosmarinus officinalis (rosemary) leaves is >2 g/kg, 22 of rosmarinus officinalis (rosemary) leaf extract is >8.5 g/kg, 8 and of rosmarinus officinalis (rosemary) leaf oil is 5.5 g/kg body weight (bw). 41
Single-Dose Toxicity Studies.
Abbreviations: F, female; M, male.
Repeated-Dose Toxicity
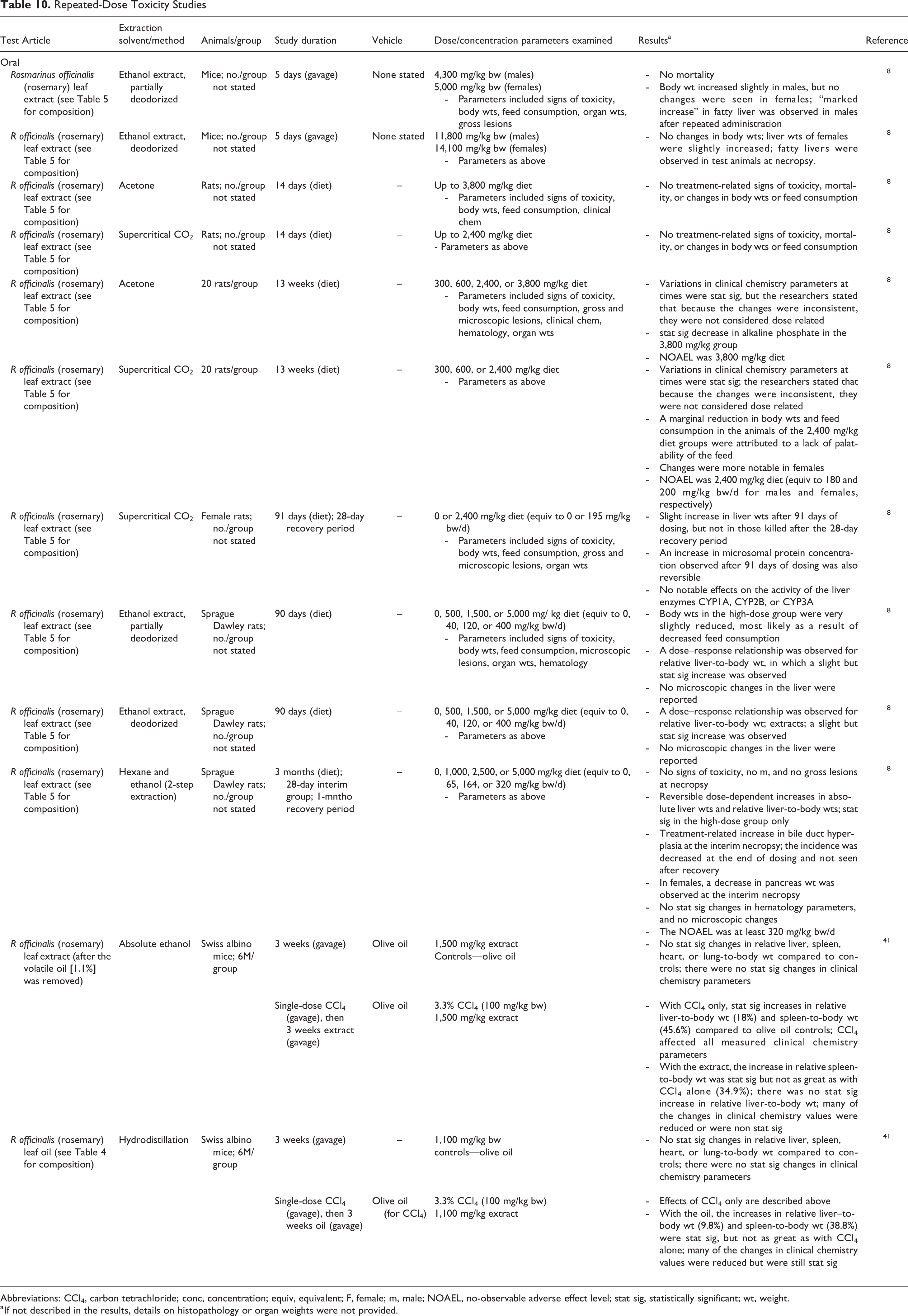
A number of oral repeated-dose toxicity studies were performed in mice and in rats with rosmarinus officinalis (rosemary) leaves extracted in a number of solvents (Table 10). Doses as high as 14.1 g/kg bw rosmarinus officinalis (rosemary) leaf extract were tested (5 days by gavage), and some studies were performed for up to 3 months (dietary) with doses of up to 400 mg/kg bw/d. 8 Increases in absolute and relative liver-to-body weights were observed in many of the studies, independent of the extraction method; these changes were shown to be reversible, and no other signs of toxicity were observed. Oral administration of rosmarinus officinalis (rosemary) leaf oil with carbon tetrachloride, but not without, resulted in an increase in liver weights. 41
Repeated-Dose Toxicity Studies
Abbreviations: CCl4, carbon tetrachloride; conc, concentration; equiv, equivalent; F, female; m, male; NOAEL, no-observable adverse effect level; stat sig, statistically significant; wt, weight.
a If not described in the results, details on histopathology or organ weights were not provided.
Ocular Irritation
Rosemary oil is reported to be a moderate ocular irritant 21 (details not provided).
Anti-Inflammatory Effects
Rosmarinus Officinalis (Rosemary) Leaf Extract
Rosmarinus officinalis (rosemary) leaf extract has been shown to inhibit formaldehyde-induced plantar edema and 12-tetradecanoylphorbol-13-acetate (TPA)-induced and arachidonic acid-induced ear edema. 43,44
In the formaldehyde-induced plantar edema study, groups of 6 male Balb/C mice were given an injection of 20 µL of 3% formaldehyde into the subplantar region of both hind paws. 43 After 2 hours, one hind paw was treated with 10 µL of 12 mg/mL of an ethanol extract of R officinalis (rosemary) leaves topically, as an injection, or both. The mice were killed after 24 hours. Topical administration of the extract reduced edema by 80%, injection reduced it by 22%, and the combined application reduced edema by 24%.
The TPA-induced ear edema study was conducted in groups of 10 male Balb/c mice. 43 The effect of pretreatment with 10 to 1,000 µg/cm2 of an ethanol extract of R officinalis (rosemary) leaves at 30 minutes prior to induction of inflammation with 25 ng/cm2 TPA was evaluated. The mice were killed after 4 hours. Doses of 100, 250, 500, and 1,000 µg/cm2 of the extract statistically significantly reduced inflammation by 38%, 79%, 84%, and 99%, respectively.
In a TPA-induced mouse ear edema study conducted in groups of 6 to 10 female CD-1 mice, a single dose of 20 µL acetone, 0.5 nmol TPA, or TPA and 0.04, 0.12, or 0.36 mg of a methanol extract of R officinalis (rosemary) leaves in 20 µL acetone was applied to one ear of each mouse. 44 The mice were killed after 5 hours, and rosmarinus officinalis (rosemary) leaf extract inhibited TPA-induced inflammation by 17%, 75%, and 92%, respectively. The extract also inhibited TPA-induced erythema.
In the arachidonic acid-induced mouse ear edema study, 0.02, 0.09, and 0.45 mg of a methanol extract of R officinalis (rosemary) leaves in 20 µL acetone was applied to groups of 10 female CD-1 mice at 30 minutes prior to treatment with 0.3 mg arachidonic acid in 20 µL acetone. 44 The mice were killed after 1 hour. Inflammation was inhibited by 12%, 28%, and 54%, respectively.
Effect on Epidermal Hyperplasia
Two hundred microliter acetone, 1 nmol TPA, or 1 nmol TPA and 3.6 mg rosmarinus officinalis (rosemary) leaf extract in 200 µL acetone were applied twice a day for 4 days to the dorsal skin of mice. 44 Three or 4 CD-1 mice were used per group. Topical application of the extract with TPA inhibited a TPA-induced increase in the number of epidermal cell layers and epidermal thickness.
Immunologic Effects
An aqueous (aq) extract of up to 2.5 mg/mL R officinalis (rosemary) leaves was found to inhibit UV-induced upregulation of matrix metalloproteinase 1 gene transcription in dermal human fibroblasts. 45 The release of the cytokines interleukin (IL) 1α and IL-6 was prevented by the extract.
Reproductive and Developmental Toxicity
Nonhuman
Rosmarinus Officinalis (Rosemary) Leaf Extract
Oral administration of high doses of rosmarinus officinalis (rosemary) leaf extract adversely affected fertility in male rats. 46 Groups of 10 male Sprague Dawley rats were fed a diet with 0, 250, or 500 mg/kg bw/d of an ethanol extract of R officinalis (rosemary) leaves in distilled water. After 53 days of dosing, each male rat was mated with 2 untreated female rats for 10 days; the female rats had been given a subcutaneous dose of 5.0 mg estradiol benzoate 54 hours and 0.5 mg progesterone at 54 and 6 hours, respectively, prior to being placed with the males. The males were dosed during, and killed after, the 10-day mating period, and the reproductive organs were examined. The females were killed 1 week after the mating period, and the reproductive tract of each female was examined to determine pregnancy and the number of implantation sites, viable fetuses, and fetal resorptions.
Body weights of the male rats of the test groups were similar to those of the control group. However, the high-dose group exhibited statistically significantly reduced absolute weights and organ to body weight ratios of testes and male accessory sex organs, diameters of seminiferous tubules and Leydig cell nuclei, height of epithelia of the epididymes and seminal vesicles, germinal and interstitial cell counts, levels of sex hormones, and sperm density and motility when compared to the controls. The numbers of interstitial degenerating cells were statistically significantly increased in the high-dose group. Exposure of the males to the high dose resulted in a reduced number of pregnant females, implantations and viable fetuses, and an increased number of resorptions. Results from the low-dose groups suggested dose–response trends in these parameters, although statistically significant differences were observed only with the high-dose group.
Rosmarinus Officinalis (Rosemary) Flower/Leaf/Stem Extract
A group of 12 gravid female Wistar rats were dosed by gavage with 26 mg/d of a 30% aq extract of rosmarinus officinal (rosemary) flower/leaf/stem extract (13 mg/mL solids) on days 1 to 6 of gestation (preimplantation), and a group of 14 gravid rats were dosed with the extract on days 6 to 15 of gestation (organogenesis). 47 Negative control groups of 12 or 11 gravid rats were given saline by gavage on days 1 to 6 or 6 to 15 of gestation, respectively. All dams were killed on day 21 of gestation. No signs of maternal toxicity were observed, and maternal weight gains were similar for treated and control groups.
In the rats dosed on days 1 to 6 of gestation, a nonstatistically significant increase in preimplantation loss was observed. No changes in postimplantation loss were seen as compared to controls, and no other reproductive parameters were affected. In the group treated on days 6 to 15 of gestation, a nonstatistically significant increase in postimplantation loss rate (2.54%) was reported; analysis of the resorptions found that they occurred during the early postimplantation period. No other changes in reproductive parameters were observed when compared to the negative control group. Developmental effects were not observed in either group.
Human
According to the PDR for Herbal Medicines, rosemary preparations should not be used as a drug during pregnancy; very large quantities of the leaves reportedly can be misused as an abortifacient. 5 According to Herbal Drugs and Phytopharmaceuticals, toxic side effects may occur with components of the essential oil. 48 (Details were not provided.)
Effects on Estrogenic Activity
Nonhuman
Rosmarinus Officinalis (Rosemary) Leaf Extract
Groups of seven or eight 6-week-old ovariectomized CD-1 mice were fed either a diet containing 2% of a methanol extract of R officinalis (rosemary) leaves or the basal diet. 49 After 3 weeks, the animals were given an intraperitoneal injection of 0, 45, or 100 ng/mouse estradiol or estrone in 50 µL corn oil, once daily for 3 days. Eighteen hours after the last injection, the animals were killed and the uterus was removed. In the mice fed the basal diet, estradiol and estrone increased the uterine wet weight in a dose-dependent manner. Rosemary inhibited 35% to 50% of the uterine response; this was statistically significant.
Human
Rosmarinus Officinalis (Rosemary) Leaf Extract
In a study investigating the effects of a botanical supplement on sex steroid hormones and metabolic markers in premenopausal women, a few changes were found; however, the changes were not very remarkable. 50 A group of 15 premenopausal women were asked to take a supplement containing 100 mg R officinalis (rosemary) leaf 5:1 extract; 100 mg Curcurma longa (turmeric) root extract standardized to 95% curcumin; 100 mg Cynara scolymus (artichoke) leaf 6:1 extract; 100 mg Silybum marinum (milk thistle) seed extracted; 100 mg Taraxacum officinalis (dandelion) root 4:1 extract; and 50 mg Schisandra chinensis (berry) 20:1 extract. Four capsules were to be taken twice a day with meals. Rice powder placebo capsules were given to a group of 15 premenopausal women using the same dosing regimen. Blood and urine samples were collected during the early follicular and midluteal phases of study menstrual cycles 1 and 5.
On average, test participants took 6.3 capsules/day and controls took 7.1 capsules/day. Compared to the placebo group, the following changes from cycle 1 to cycle 5 in early follicular phase serum hormone concentrations were statistically significant or borderline significant: decreases in serum dehydroepiandrosterone (−13.2%, P = .02), dehydroepiandrosterone sulfate (14.6%, P = .07), androstenedione (−8.6%, P = .05), and estrone sulfate (−12.0%, P = .08). No other statistically significant changes or trends were observed for other serum sex steroid hormones, serum metabolic markers, or urinary estrogen metabolites at either phase.
Genotoxicity
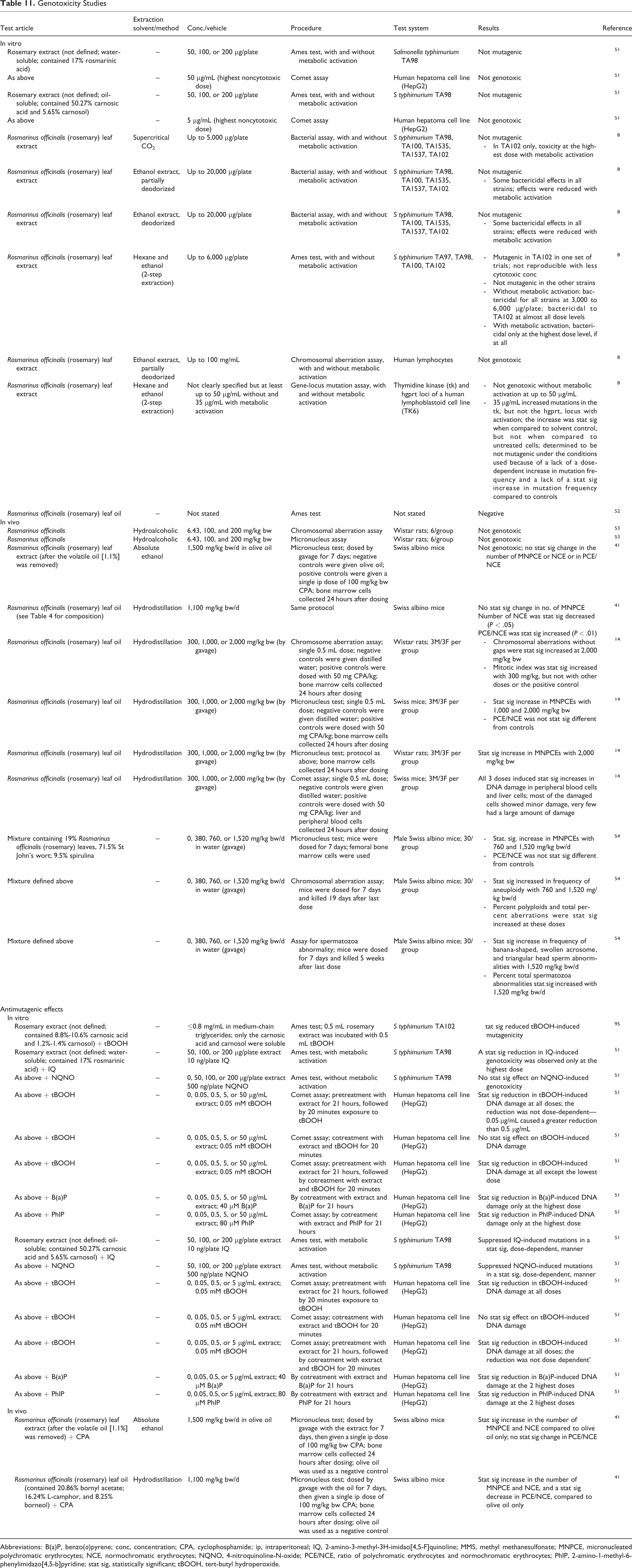
In vitro, rosemary extract (solvent not specified) 51 and rosmarinus officinalis (rosemary) leaf oil 52 were not mutagenic in an Ames test, and rosmarinus officinalis (rosemary) leaf extract was not genotoxic in an Ames test, a chromosomal aberration assay in human lymphocytes, or a gene locus mutation assay in human lymphocytes 8 (Table 11). In in vivo studies in mice and rats, oils that were extracted by hydrodistillation induced statistically significant increases in chromosomal aberrations without gaps in a chromosomal aberration assay at 2,000 mg/kg bw, increases in micronucleated polychromatic erythrocytes (MNPCEs) in several micronucleus tests at 1,000 and 2,000 mg/kg bw, and increases in DNA damage in a comet assay at ≥300 mg/kg bw 14,41 ; however, no genotoxic effects were seen in mice in a micronucleus test at 1,500 mg/kg bw/d with leaves extracted with absolute ethanol. 41 A hydroalcoholic extract of rosemary was not genotoxic in a chromosomal aberration assay or a micronucleus test in rats. 53 A mixture containing 19% R officinalis (rosemary) leaves, 71.5% St John’s wort, and 9.5% spirulina induced in mice statistically significant increases in MNPCEs at 760 and 1,520 mg/kg bw/d in a micronucleus test; in frequency of aneuploidy, percent polyploidy, and total percent aberrations with 760 and 1,520 mg/kg bw/d in a chromosomal aberration assay; and in frequency of banana-shaped, swollen acrosome, and triangular head sperm abnormalities and percent total spermatozoa abnormalities at 1,520 mg/kg bw/d in a spermatozoa abnormality assay. 54
Genotoxicity Studies
Abbreviations: B(a)P, benzo(a)pyrene; conc, concentration; CPA, cyclophosphamide; ip, intraperitoneal; IQ, 2-amino-3-methyl-3H-imidao[4,5-F]quinoline; MMS, methyl methanesulfonate; MNPCE, micronucleated polychromatic erythrocytes; NCE, normochromatic erythrocytes; NQNO, 4-nitroquinoline-N-oxide; PCE/NCE, ratio of polychromatic erythrocytes and normochromatic erythrocytes; PhIP, 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine; stat sig, statistically significant; tBOOH, tert-butyl hydroperoxide.
Rosmarinus officinalis (rosemary) leaf extract was shown to have antimutagenic potential, in vitro, in an Ames test with Salmonella typhimurium and in Comet assays in a human hepatoma cell line. 51 In vivo, in micronucleus assays, rosmarinus officinalis (rosemary) leaf extract did not decrease the number of MNPCEs induced in mice by a genotoxic agent. 41
Carcinogenicity
Effects on Tumor Promotion
Topical application of methanol and double distilled water extracts of R officinalis (rosemary) leaves statistically significantly decreased skin tumors in mice; in these studies, 7,12-dimethylbenz[a]anthracene (DMBA) or benzo[a]pyrene (B(a)P) was used for initiation and TPA 44 or croton oil 55,56 was used for promotion (Table 12). Dietary administration of 1.0% R officinalis (rosemary) leaf extract decreased the incidence of palpable mammary tumors in rats caused by DMBA. 57
Effects on Tumor Promotion.
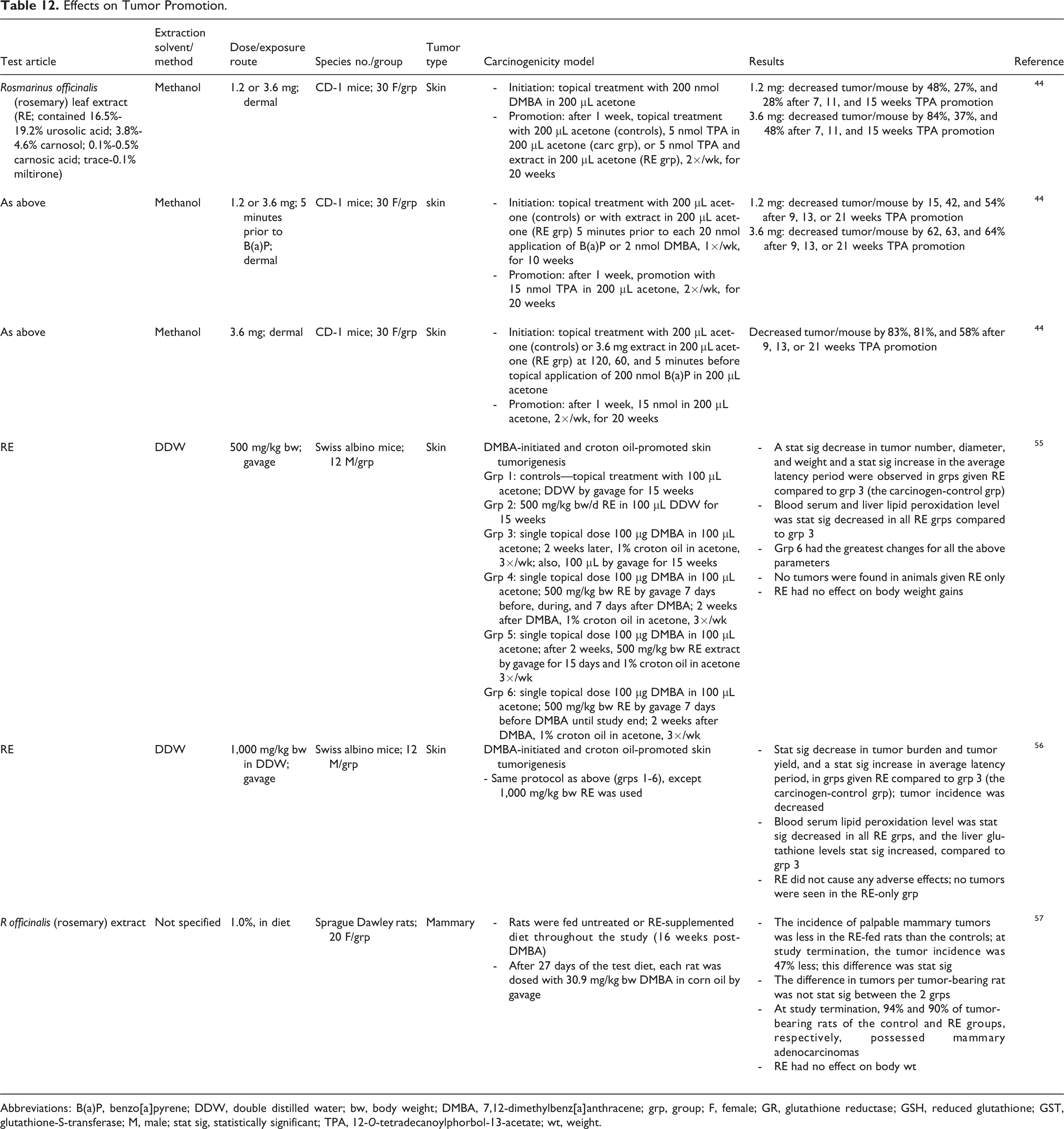
Abbreviations: B(a)P, benzo[a]pyrene; DDW, double distilled water; bw, body weight; DMBA, 7,12-dimethylbenz[a]anthracene; grp, group; F, female; GR, glutathione reductase; GSH, reduced glutathione; GST, glutathione-S-transferase; M, male; stat sig, statistically significant; TPA, 12-O-tetradecanoylphorbol-13-acetate; wt, weight.
Irritation and Sensitization
Skin Irritation/Sensitization
An ointment containing 4.4% rosmarinus officinalis (rosemary) leaf oil (and other essential oils), applied at concentrations up to 40%, was not irritating to rat skin (Table 13). 58 However, in a rabbit study, occlusive application to intact and abraded skin produced moderate irritation. 42
Dermal Irritations and Sensitization
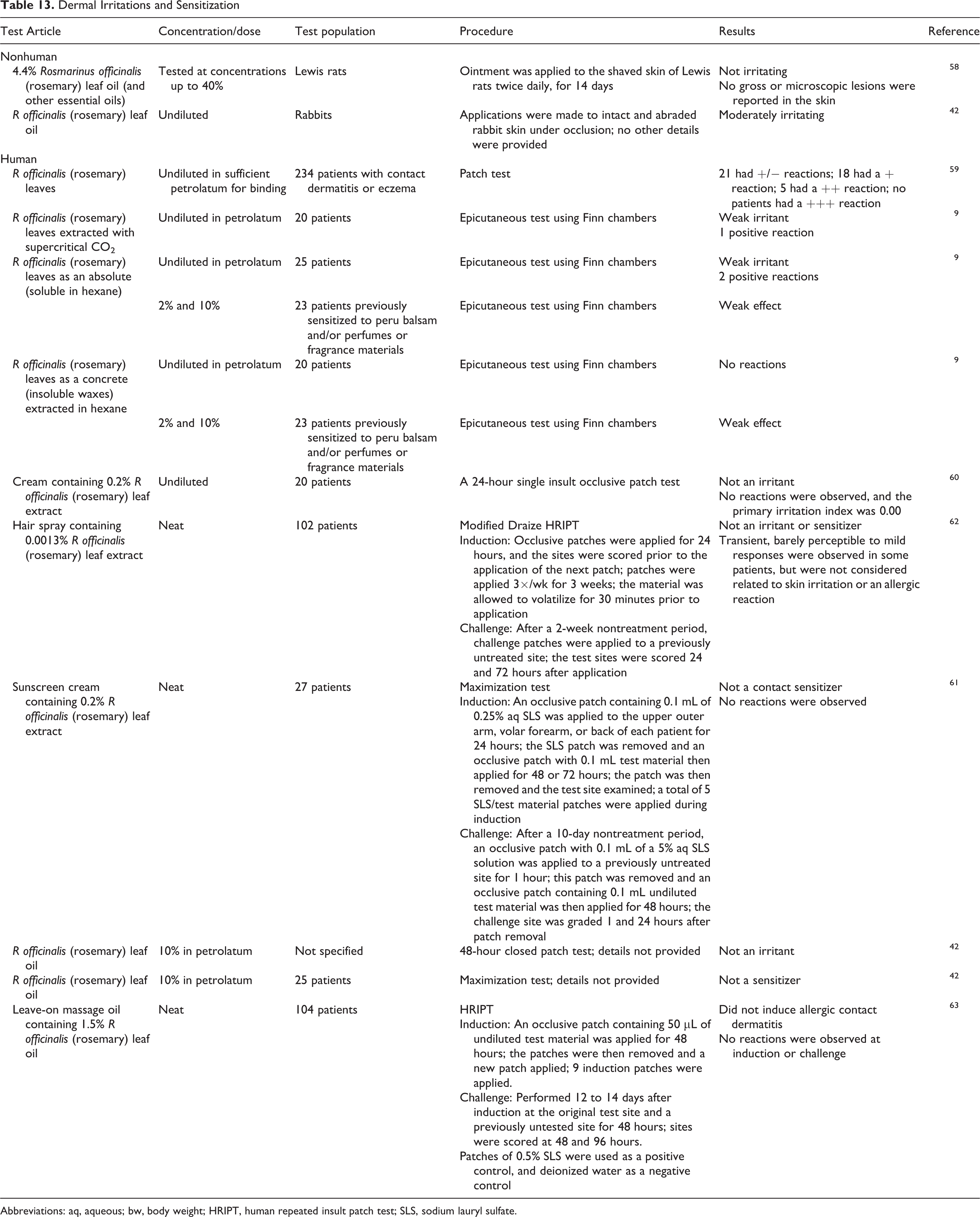
Abbreviations: aq, aqueous; bw, body weight; HRIPT, human repeated insult patch test; SLS, sodium lauryl sulfate.
In clinical testing, R officinalis (rosemary) leaves produced irritation (scores of +/−, +, or ++) in 44 of 234 patients with contact dermatitis or eczema (Table 13). 59 A supercritical extract and the absolute of R officinalis (rosemary) leaves were considered weak irritants in a small study with test populations of 20 to 25 patients; the extracts were not phototoxic. 9 Formulations containing up to 0.2% R officinalis (rosemary) leaf extract were not irritants or sensitizers. 60 -62 Rosmarinus officinalis (rosemary) leaf oil, 10% in petrolatum, was not an irritant in a 48-hour closed patch test or a sensitizer in a maximization study 42 ; a formulation containing 1.5% rosmarinus officinalis (rosemary) leaf oil was not an irritant or a sensitizer in a human repeated insult patch test (HRIPT). 63
Phototoxicity
Rosmarinus Officinalis (Rosemary) Leaf Extract
The phototoxicity of R officinalis (rosemary) leaf extract extracted with supercritical CO2, as a concrete (insoluble wax) extracted in hexane, or as a concrete extracted in hexane, was evaluated. 9 Photopatch tests were performed on 2 of 3 test sites; one site was irradiated with 10 J/cm2 UVA and the second site with 75% of the minimal erythema dose of UVB. The test sites were scored after 48 and 72 hours and were compared to the nonirradiated site. None of the extracts were phototoxic.
Case Reports
Several cases of allergic reactions to R officinalis (rosemary) have been reported (Table 14). 64 -72 In some of the studies, follow-up patch testing included photopatch tests; generally, reactions were stronger in the photopatch tests when compared to standard testing. 68,69 Some of the follow-up patch testing included carnosol; testing with 0.1 and 1.0% carnosol resulted in positive reactions. 65,69
Case Reports With Rosmarinus officinalis (Rosemary).

Abbreviation: aq, aqueous.
Summary
This report addresses the safety of 10 R officinalis (rosemary)-derived ingredients as used in cosmetics. Most of the ingredients included in this review are extracts, essential oils, powders, or waters derived from a defined part of the R officinalis (rosemary) plant. The R officinalis (rosemary)-derived ingredients are reported to have a number of functions in cosmetics, and the most common functions are as a skin-conditioning agent or as a fragrance ingredient. According to VCRP data obtained from the FDA, rosmarinus officinalis (rosemary) leaf extract has the most uses, 729, followed by rosmarinus officinalis (rosemary) leaf oil, 474 uses, and rosmarinus officinalis (rosemary) extract, 404 uses. Most of the reported use concentrations for R officinalis (rosemary)-derived ingredients are well below 0.1%. However, rosmarinus officinalis (rosemary) leaf extract has higher concentrations of use reported, specifically, use at up to 10% in body and hand products and 3% in eye shadow formulations and bath soaps and detergents. Rosmarinus officinalis (rosemary) flower/leaf/stem water is the only ingredient not reported to be used.
Rosmarinus officinalis (rosemary) extract is prepared by extraction from the leaves of R officinalis with acetone, ethanol, hexane, a combination of hexane and ethanol (in a 2-step process), or supercritical CO2; it can also be prepared from a deodorized or partially deodorized ethanol extract of rosemary. Additional methods include extraction with absolute ethanol (resulting in an absolute) or a collection of the insoluble waxes (resulting in a concrete).
Rosmarinus officinalis L. is composed of an array of constituents, primarily phenolic acids, flavonoids, monoterpenes, diterpenes, diterpenoids, and triterpenes. The principal antioxidative components of rosmarinus officinalis (rosemary) leaf extract are the phenolic diterpenes carnosol and carnosic acid. The actual amount of constituents present varies according to the stage of development, variety of plant, season harvested, origin of the leaves, and extraction method.
Rosemary oil increased the permeation of aminophylline through human skin, but the increase was not as great as that seen with 50% ethanol.
The acute toxicity of R officinalis (rosemary)-derived ingredients is not very remarkable. The dermal LD50 of rosmarinus officinalis (rosemary) leaf oil is >10 mL/kg. The oral LD50 of rosmarinus officinalis (rosemary) leaves is >2 g/kg, of R officinalis (rosemary) leaf extract is >8.5 g/kg, and of rosmarinus officinalis (rosemary) leaf oil is 5.5 g/kg bw.
A number of oral repeated-dose toxicity studies were performed in mice and in rats with R officinalis (rosemary) leaves extracted in a various solvents. Doses as high as 14.1 g/kg bw rosmarinus officinalis (rosemary) leaf extract were tested (5 days by gavage), and some studies were performed for up to 3 months (dietary) with doses of up to 400 mg/kg bw/d. Increases in absolute and relative liver-to-body weights were observed in many of the studies, independent of the extraction method; these changes were shown to be reversible, and no other signs of toxicity were observed. Oral administration of rosmarinus officinalis (rosemary) leaf oil with carbon tetrachloride, but not without, resulted in an increase in liver weights.
Rosmarinus officinalis (rosemary) leaf extract has been shown to have anti-inflammatory activity. Rosmarinus officinalis (rosemary) leaf extract inhibited a TPA-induced increase in the number of epidermal cell layers and epidermal thickness in mouse skin.
A high dose (500 mg/kg/d) of rosmarinus officinalis (rosemary) leave extract was a reproductive toxicant in a dietary study in male rats. In a study in gravid female Wistar rats, no statistically significant changes were observed after oral dosing with 26 mg/d of a 30% aq rosmarinus officinalis (rosemary) flower/leaf/stem extract during preimplantation or during organogenesis. In a dietary study in ovariectomized CD-1 mice, 2% of a methanol extract of R officinalis (rosemary) leaves inhibited the uterine response in a statistically significant manner.
In a clinical study investigating the effects on sex steroid hormones and metabolic markers of a botanical supplement containing 100 mg R officinalis (rosemary) leaf 5:1 extract (and other botanical ingredients) in premenopausal women, a few changes were found. Overall, the changes were not remarkable.
In vitro, rosemary extract (solvent not specified) and rosmarinus officinalis (rosemary) leaf oil were not mutagenic in an Ames test, and rosmarinus officinalis (rosemary) leaf extract was not genotoxic in an Ames test, a chromosomal aberration assay in human lymphocytes, or a gene-locus mutation assay in human lymphocytes. In in vivo studies in mice and rats, oils that were extracted by hydrodistillation induced statistically significant increases in chromosomal aberrations without gaps in a chromosomal aberration assay at 2,000 mg/kg bw, increases in MNPCEs in several micronucleus tests at 1,000 and 2,000 mg/kg bw, and increases in DNA damage in a comet assay at ≥300 mg/kg bw; however, no genotoxic effects were seen in mice in a micronucleus test at 1,500 mg/kg bw/d with leaves extracted with absolute ethanol. A hydroalcoholic extract of rosemary was not genotoxic in a chromosomal aberration assay or a micronucleus test in rats. A mixture containing 19% R officinalis (rosemary) leaves, 71.5% St John’s wort, and 9.5% spirulina induced in mice statistically significant increases in MNPCEs at 760 and 1,520 mg/kg bw/d in a micronucleus test; in frequency of aneuploidy, percent polyploidy, and total percent aberrations with 760 and 1,520 mg/kg bw/d in a chromosomal aberration assay; and in frequency of banana-shaped, swollen acrosome, and triangular head sperm abnormalities and percent total spermatozoa abnormalities at 1,520 mg/kg bw/d in a spermatozoa abnormality assay.
Rosmarinus officinalis (rosemary) leaf extract was shown to have antimutagenic potential in vitro. In vivo, in micronucleus assays, rosmarinus officinalis (rosemary) leaf extract did not decrease the number of MNPCEs induced by a genotoxic agent.
Topical application of methanol and double distilled water extracts of rosmarinus officinalis (rosemary) leaves statistically significantly decreased skin tumors in mice; in these studies, DMBA or benzo[a]pyrene was used for initiation and TPA or croton oil was used for promotion. Dietary administration of 1.0% rosmarinus officinalis (rosemary) leaf extract decreased the incidence of palpable mammary tumors in rats caused by DMBA.
An ointment containing 4.4% rosmarinus officinalis (rosemary) leaf oil (and other essential oils), applied at concentrations up to 40%, was not irritating to rat skin. However, in a rabbit study, occlusive application to intact and abraded skin produced moderate irritation.
In clinical testing, R officinalis (rosemary) leaves produced irritation (scores of +/−, +, or ++) in 44 of 234 patients with contact dermatitis or eczema. A supercritical extract and the absolute of R officinalis (rosemary) leaves were considered weak irritants in a small study with test populations of 20 to 25 subjects; the extracts were not phototoxic. Formulations containing up to 0.2% rosmarinus officinalis (rosemary) leaf extract were not irritants or sensitizers. Rosmarinus officinalis (rosemary) leaf oil, 10% in petrolatum, was not an irritant in a 48-hour closed patch test or a sensitizer in a maximization study in 25 subjects; a formulation containing 1.5% rosmarinus officinalis (rosemary) leaf oil was not an irritant or a sensitizer in an HRIPT in 104 patients.
Several cases of allergic reactions to R officinalis (rosemary) have been reported. In some of the studies, follow-up patch testing included photopatch tests; generally, reactions were stronger in the photopatch tests, compared to standard testing. Some also evaluated the effect of carnosol; testing with 0.1% and 1.0% carnosol resulted in positive reactions.
Discussion
Rosmarinus officinalis is GRAS as a spice, and although that mitigates much of the concern of oral exposure with cosmetic use, data on local effects, such as dermal irritation and sensitization data, are necessary to determine safety.
Additional information on the deodorizing process that is part of the preparation of some of the ingredients was not received. After further discussion, however, the Panel stated that because the deodorizing process is part of the preparation of food-grade rosmarinus officinalis (rosemary) extract, and because data are included in this safety assessment on some ingredients that were deodorized and no adverse effects were found, the Panel was not concerned with obtaining additional information on this process or the by-products that might form.
The Panel did note that because botanical ingredients, derived from natural plant sources, are complex mixtures, there is concern that multiple botanical ingredients may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of plant constituents that may cause sensitization or other adverse effects. Specific examples of constituents that could possibly induce sensitization are linalool or monoterpenes, and those that could possibly cause adverse effects are caffeic acid and terpenes, such as thujone, limonene, and methyleugenol.
The Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities.
One study evaluated the irritation potential of R officinalis (rosemary) leaves in patients with contact dermatitis or eczema. The Panel stated that because the test participants were patients with eczematous skin, the report of irritation could not be interpreted for relevance to cosmetic use.
The Panel discussed the positive results observed in a reproductive toxicity study in male rats fed 500 mg/kg/d rosmarinus officinalis (rosemary) leaf extract, as well as the caution in the PDR for Herbal Medicines stating that rosemary preparations should not be used as a drug during pregnancy. The effects in the rat study were observed at exposure concentrations that would be well above those used in cosmetic products, and the PDR refers to the use of rosemary as a drug at very high concentrations. Because these effects were observed only at very high concentrations, and because no statistically significant effects were reported in a study in rats dosed orally with 26 mg/d of a 30% aq extract of rosmarinus officinalis (rosemary) flower/leaf/stem extract, reproductive and developmental toxicity is not a concern with cosmetic use of R officinalis (rosemary)-derived ingredients, which are mostly used at very low concentrations.
Finally, the Panel discussed the issue of incidental inhalation exposure to R officinalis (rosemary)-derived ingredients. The Panel stated that although there were no inhalation data available, the R officinalis (rosemary)-derived ingredients are used at very low concentrations in products that could incidentally be inhaled; for example, rosmarinus officinalis (rosemary) leaf extract is used in other fragrance preparations and rosmarinus officinalis (rosemary) extract is used in face powders. The Panel noted that in aerosol products, 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Cosmetic Ingredient Review Expert Panel concluded that the following 10 R officinalis (rosemary)-derived ingredients are safe in cosmetics in the present practices of use and concentration described in this safety assessment when formulated to be nonsensitizing: Rosmarinus Officinalis (Rosemary) Extract Rosmarinus Officinalis (Rosemary) Flower Extract Rosmarinus Officinalis (Rosemary) Flower/Leaf/Stem Extract Rosmarinus Officinalis (Rosemary) Flower/Leaf/Stem Water* Rosmarinus Officinalis (Rosemary) Leaf Rosmarinus Officinalis (Rosemary) Leaf Extract Rosmarinus Officinalis (Rosemary) Leaf Oil Rosmarinus Officinalis (Rosemary) Leaf Powder Rosmarinus Officinalis (Rosemary) Leaf Water Rosmarinus Officinalis (Rosemary) Water
*Not reported to be in current use. If this ingredient was to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author contributions
M. Fiume and B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. L. Gill, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
