Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of magnesium sulfate, which functions as a bulking agent in cosmetic products and is reportedly used at concentrations up to 11% and 25% in leave-on and rinse-off products, respectively. The Panel noted that the history of safe medical use of magnesium sulfate provides further confidence that there are no significant toxicity concerns relating to systemic exposure to this ingredient after cosmetic product application. Furthermore, the extensive clinical experience of the Panel, including the results of numerous patch tests, indicates that magnesium salts do not have the potential to induce sensitization. The Panel also noted that results were negative for 50% magnesium sulfate in a mouse skin irritation study and in an in vitro sensitization assay. The Panel concluded that magnesium sulfate is safe in cosmetics in the present practices of use and concentration described in the safety assessment.
Keywords
Introduction
The safety of magnesium sulfate, an inorganic sulfate, as used in cosmetics, is reviewed in this safety assessment. Magnesium sulfate is reported to function as a bulking agent in cosmetic products.
Chemistry
Definition and Structure
Magnesium sulfate (MgSO4, CAS Nos. 18939-43-0 and 7487-88-9) is the magnesium salt of sulfuric acid. 1
Physical and Chemical Properties
Magnesium sulfate, available in the form of efflorescent crystals, has a molecular weight of 120.37 Da and is soluble in water and sparingly soluble in alcohol. 2
Method of Manufacture
Hydrated magnesium sulfate can be mined as kieserite or epsomite (Epsom salts; magnesium sulfate heptahydrate), or it can be prepared by dissolving magnesium oxide, magnesium hydroxide, or magnesium carbonate in sulfuric acid. 3 Heating of this hydrate reversibly drives off water and produces anhydrous magnesium sulfate (a potent desiccant).
Use
Cosmetic
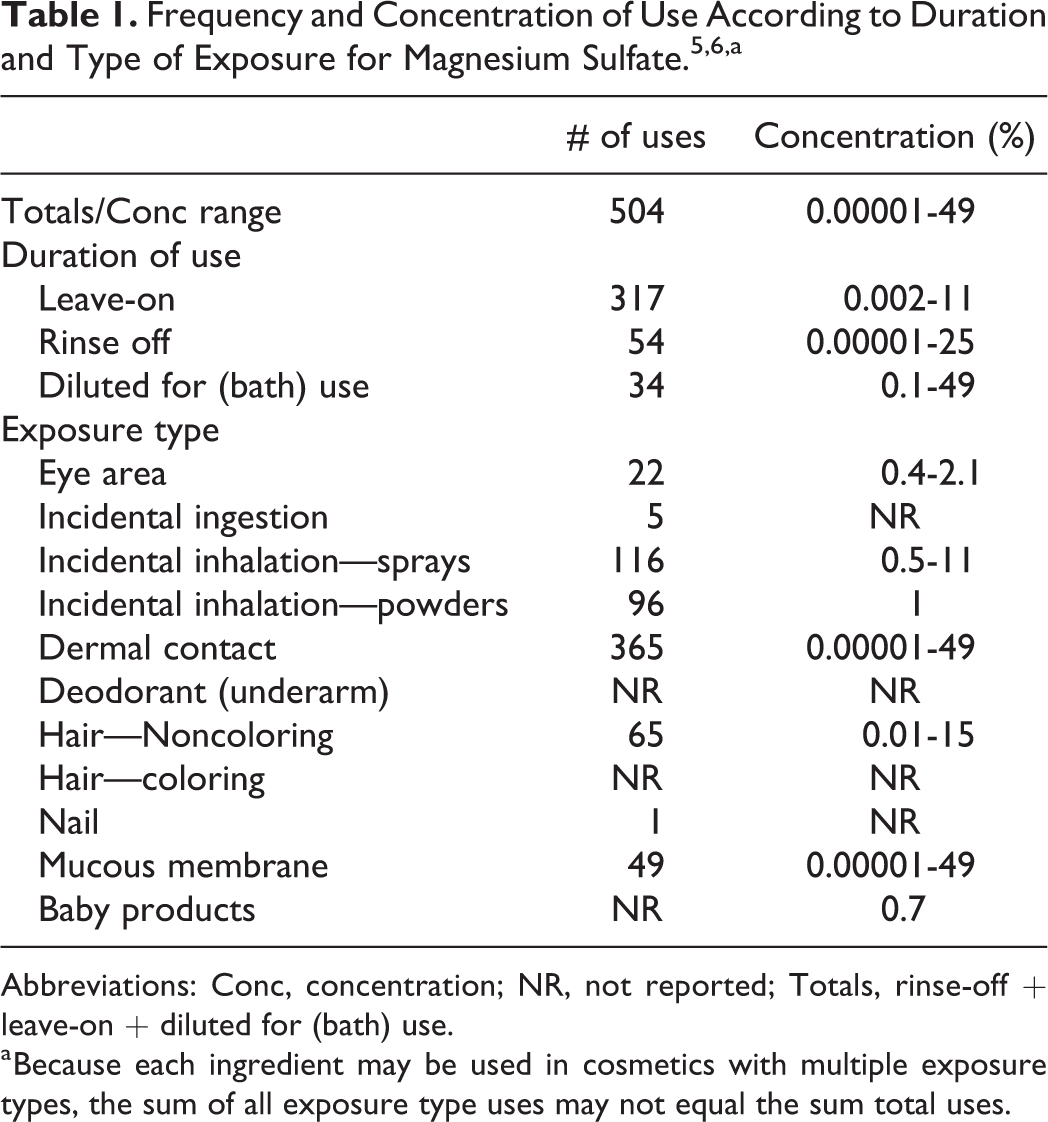
Magnesium sulfate functions as a bulking agent in cosmetic products. 4 According to information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), magnesium sulfate is used in a number of rinse-off and leave-on cosmetic products. 5 Results from a survey of ingredient use concentrations provided by the Personal Care Products Council (Council) in 2013 indicate that this ingredient is used at concentrations up to 25% in rinse-off products (paste masks) and up to maximum use concentrations of 11% in leave-on products (hair sprays). 6 Summarized data on the frequency and concentration of use of magnesium sulfate in cosmetic products are presented in Table 1.
Abbreviations: Conc, concentration; NR, not reported; Totals, rinse-off + leave-on + diluted for (bath) use.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Cosmetic products containing magnesium sulfate may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Magnesium sulfate is used at concentrations up to 11% and 1% in cosmetic products that are sprayed (hair and foot sprays, respectively) and at concentrations up to 1% in powders (foot powders). Because this ingredient is used in aerosol/pump hair sprays and in powders, it could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared to pump spray. 7 –10 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 7,8
Noncosmetic
Magnesium sulfate is an FDA-approved direct food additive that is generally recognized as safe. 11 Additionally, FDA has stated that magnesium sulfate heptahydrate should be listed on the label of a drug product as Epsom salt, which may be used as a laxative. 12 According to other sources, magnesium sulfate is an anticonvulsant that is used to prevent or treat seizures in obstetric patients with preeclampsia or eclampsia and as a tocolytic agent in patients with premature labor. 13,14 It is also used to prevent early mortality in patients with acute myocardial infarction and, in patients with asthma, as an adjunctive treatment for acute exacerbations of moderate to severe asthma.
Toxicokinetics
Animal
Groups of pregnant female Long-Evans rats were injected subcutaneously (sc) with magnesium sulfate according to the following procedure: One group received a single 270 mg/kg sc injection of magnesium sulfate. Two other groups received a 270 mg/kg loading dose of magnesium sulfate sc, followed by 27 mg/kg every 20 minutes for 2 or 4 hours. The gestation days on which dosing occurred were not stated. Magnesium crossed the placenta and the fetal blood–brain barrier and was concentrated in the forebrain. 15
Wistar rats were exposed (inhalation exposure) to long and short magnesium sulfate whiskers 5 days per week (6 h/d) for 1 year. 16 Long and short whiskers were defined as those having a mean diameter of 1.8 and 1.5 µm, respectively. The exposure groups were defined as follows: long whisker (27 rats), short whisker (27 rats), and controls (26 rats). Long and short whiskers were tested at mean concentrations of 4.0 mg/m3 and 2.3 mg/m3, respectively. Few whiskers were detected in rat lungs, even at day 1 postexposure. This finding suggested that magnesium sulfate whiskers are dissolved and eliminated rapidly from the lungs. Additional results are reported in the Repeated Dose Toxicity section.
Human
The concentration in serum and the cumulative renal excretions of magnesium were measured in 3 patients with eclampsia and 7 patients with severe eclampsia given an initial therapeutic dose of magnesium sulfate intravenously (iv; 3 g) and intramuscularly (10 g). 17 The highest single plasma level was 6.0 mEq/L (7.2 mg/100 mL) at 60 minutes posttreatment in a woman with oliguric eclampsia. The average peak level at 60 minutes was 4.5 mEq/L. At the end of 4 hours, the cumulative renal excretions ranged from 38% to 53% of the injected dose.
Magnesium sulfate (USP, 13.9 g) was administered orally to 7 healthy men (ages not stated) in 4 equal hourly increments. 18 Urinary excretion (corrected for baseline excretion rate) was described as an amount of inorganic sulfate equivalent to 30.2% ± 17.2% of the administered dose during the first 24 hours. Excretion during the subsequent 48 hours was negligible.
Toxicology
Acute Toxicity
Dermal
Anhydrous magnesium sulfate (50%) was applied to the ears of mice (number not stated) in a preliminary skin irritation study, performed prior to the sensitization study. 19 The results for these studies are summarized in the Dermal Irritation and Sensitization section of this report. All auricular lymph nodes in test and control animals were considered normal in size. Body weights and body weight gain reported for test animals remained in the same range when compared to controls. The slight body weight loss reported for some animals was not considered toxicologically significant. There were no symptoms of systemic toxicity, and none of the animals died.
Intravenous
Magnesium sulfate was administered iv to groups of Crj:CD(SD) rats (males and females, 6 weeks old) at doses of 90, 130, 200, 300, and 450 mg/kg. 20 Deaths occurred at doses ≥ 200 mg/kg, and the median lethal dose (LD50) values were 206 mg/kg and 174 mg/kg for males and females, respectively. Tonic convulsions, abnormal gait, and tachypnea were observed in surviving animals dosed with ≥130 mg/kg. These signs were transient, and the animals had returned to normal by 15 minutes postdosing. There were no treatment-related changes in body weight or gross pathology in any dose group. In a second experiment, magnesium sulfate was infused into groups of female Beagle dogs (6 months old) at doses of 75, 300, and 1,200 mg/kg (12.5, 50, and 200 mg/kg/h) for 6 hours. Deaths were not observed in any of the dose groups. The following signs were observed in the 1,200 mg/kg dose group: vomiting, decreased spontaneous movement, staggering gait, prone position, and flush of the conjunctiva and ear auricles. These signs were transient, and the animals had returned to normal by 1 hour postdosing. There were no treatment-related changes in body weight, food consumption, or gross pathology.
Repeated Dose Toxicity
Inhalation
Male Wistar rats were exposed (inhalation exposure) to 2 types of magnesium sulfate whiskers 5 days per week (6 h/d) for 4 weeks. 16 Magnesium sulfate whisker is a manmade mineral fiber that is synthesized from magnesium sulfate and magnesium hydroxide by hydrothermal reaction at 100°C to 300°C. In the 4-week study, short whisker (mean diameter = 1.5 µm) was tested at a mean concentration of 2.3 mg/m3 and long whisker (mean diameter = 1.8 µm) was tested at a mean concentration of 4.0 mg/m3. The 124 rats in the 4-week experiment were divided into 3 groups (42, 42, and 40 rats per group for short whiskers, long whiskers, and controls, respectively). There was no indication that adverse effects were observed in this study.
Intravenous
In a 2-week study, groups of female Beagle dogs received the following iv doses of magnesium sulfate: 12.5, 50, 100, and 200 mg/kg/h. 21 The test substance was administered as 24-hour iv infusions. Dosing was followed by a 2-week observation period. One animal in the 200 mg/kg/h dose group died at 32 hours, and another animal was euthanized because of morbidity at the same time. Treatment-related changes in the 100 mg/kg/h dose group were as follows: decreased food consumption and body weight gain, anemia, mild prolongation of conduction time in the electrocardiogram, and tubular basophilia in the kidneys. Additionally, decreased calcium level was reported for animals that received doses ≥50 mg/kg/h and was considered toxicologically insignificant. These treatment-related changes were not observed at the end of dosing. The no-observed-adverse-effect-level (NOAEL) was considered to be 50 mg/kg/h (for 24 hours) in this study.
Magnesium sulfate was administered (24-hour iv infusion) to groups of 3 female Beagle dogs at doses of 12.5, 50, and 100 mg/kg/h for 2 weeks. 22 Dosing was followed by a 2-week observation period. None of the animals died. The following treatment-related changes were reported (highest dose group): decreased feed consumption and body weight gain, anemia, increased urine volume, decreased serum calcium level, increased inorganic phosphorus level, slight prolongation of conduction time in the electrocardiogram, and tubular basophilia in the kidneys. The NOAEL was considered to be ≤50 mg/kg/h (for 24 hours) in this study.
Reproductive and Developmental Toxicity
Animal
The following doses of magnesium sulfate were administered sc to Crj:CD(SD) female rats 3 times per day on gestation days 15 through 20: 250, 500, and 1,000 mg/kg. 23 The control group and 250 mg/kg group each consisted of 19 rats. The remaining 2 dose groups each contained 20 rats. Effects of the test material on the dams and F1 animals were examined. Dams dosed with 500 and 1,000 mg/kg had decreased food consumption. Hypolocomotion, pronation, bradypnea, and decreased body weight gain were observed in the 1,000 mg/kg dose group. There were no test material-related effects on delivery or lactation, and necropsy results were normal. Results for F1 animals dosed with 1,000 mg/kg were as follows: low body weight, delays in differentiation (eruption of lower incisor and opening of eyelid), and reversible changes in ribs (wavy ribs). However, there were no test material-related effects on viability, functional examinations, behavioral tests, or reproductive ability. It was concluded that the NOAEL for general toxicological effects on the dams was 250 mg/kg/d (3 times per day) and that the NOAEL for reproductive ability and development were 1,000 mg/kg/d (3 times per day) and 500 mg/kg (3 times per day), respectively.
Human
Over a period of 14 years, 7,000 infants were born to mothers who had received magnesium sulfate parenterally because of preeclampsia or eclampsia. 24 A 50% magnesium sulfate (magnesium sulfate heptahydrate; MgSO4·7H2O, USP) solution was injected intramuscularly (30-40 g doses, over 24 hours) into the gravida. This regimen was continued as long as the mother had demonstrable knee jerks, urine output of at least 100 mL over 4 hours, and no depression of respiration. The serum level of magnesium in the fetus rapidly approached the maternal level but could not be correlated with any adverse effect. Dosing did not have any observable deleterious effects on the fetus or newborn.
Five neonates were born to mothers who had been treated iv with magnesium sulfate for tocolysis. 25 The neonates were retrospectively reviewed to assess the presence of radiographic, clinical, and biochemical abnormalities. Two infants had radiographic bony abnormalities; one had frank rachitic changes and dental enamel hypoplasia. One of these patients as well as an additional infant had transient hypocalcemia. It was hypothesized that prolonged infusion of magnesium sulfate, especially when initiated during the second trimester, may lead to fetal parathyroid gland suppression, with consequent abnormalities resembling rickets.
The effects of maternal magnesium sulfate treatment on newborns were studied. 26 The participants in this study were newborn infants, delivered at ≥34 weeks of gestation, whose mothers had received a minimum of 12 hours of iv magnesium sulfate therapy prior to delivery. A total of 26 magnesium-exposed and 26 control infants were enrolled. The mean dose of magnesium sulfate prior to delivery was 51.2 ± 24 g, and the mean duration of therapy was 23.1 ± 10 hours. The mean maternal serum magnesium level before delivery was 5.8 ± 1.1 mg/dL. Infants exposed to magnesium sulfate in utero had a higher incidence of hypotonia and lower median Apgar scores, compared to control infants (P < 0.001). However, there was no association between adverse outcomes and maternal serum magnesium concentrations at the time of delivery, duration of treatment, or dose of magnesium sulfate. Pneumocardiogram data were similar between magnesium sulfate-exposed and control infants (all, P ≥ 0.16).
In a controlled trial, mothers in preterm labor were randomized as follows: magnesium sulfate tocolysis (46 mothers and 55 newborns [9 pairs of twins included]) and saline control (28 mothers and 29 newborns). 27 Magnesium sulfate was administered as a 4 g bolus, followed by infusion of 2 to 3 g of magnesium sulfate per hour. Children with adverse outcomes had higher umbilical cord magnesium levels at the time of delivery. In regression models that controlled for confounders, which included very low birth weight, magnesium remained a significant risk factor (adjusted odds ratio = 3.7; 95% confidence interval [CI] = 1.1-11.9; P = 0.03) for adverse health outcomes in infants. Dosing with magnesium sulfate was associated with 11 composite adverse pediatric outcomes, which included intraventricular hemorrhage and periventricular leukomalacia and cerebral palsy. However, the differences in this trial were not statistically significant (magnesium sulfate: 37% [11 adverse events in 30 infants]; saline solution: 21% [6 adverse events in 29 infants]; P = 0.25).
Between January 2000 and February 2009, 6,654 women with preeclampsia were treated with an iv infusion of magnesium sulfate, with the goal of achieving a therapeutic range of 4 to 7 mE/L (2.0-3.5 mmol/L). 28 Eighty-eight infants (6% of the infants) were diagnosed with hypotonia. Lower 1-minute and 5-minute Apgar scores, intubation in the delivery room, admission to special care nursery, and hypotonia were all significantly increased as maternal serum magnesium concentrations increased prior to birth.
Genotoxicity
In Vitro: Bacterial Cells
Magnesium sulfate was evaluated for genotoxicity in the Ames test using the following Salmonella typhimurium strains: TA92, TA94, TA98, TA100, TA1535, and TA1537. 29 The test substance (in phosphate buffer) was evaluated at doses up to 100 mg/plate with metabolic activation, and results were negative in all bacterial strains tested. In another Ames test, magnesium sulfate was evaluated in the following bacterial strains at doses up to 5,000 µg/plate, with and without metabolic activation: S typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2 uvrA. 30 Magnesium sulfate did not induce an increase in the incidence of reverse mutations in any of the bacterial strains tested in this assay.
The genotoxicity of magnesium sulfate was evaluated in the SOS Chromotest using E coli strain PQ37, with and without metabolic activation. 31 The SOS Chromotest is a colorimetric assay that measures the expression of genes induced by genotoxic agents in E coli by means of fusion with the structural gene for β-galactosidase. Magnesium sulfate (MgSO4·7H2O) was tested at concentrations up to 30,000 nmol/mL and was not genotoxic, with or without metabolic activation.
In Vitro: Mammalian Cells
In a chromosome aberration assay using Chinese hamster ovary cells, 32 the genotoxicity of magnesium sulfate (dissolved in physiological saline) was evaluated at concentrations up to 4 mg/mL in culture. Magnesium sulfate did not induce chromosomal aberrations in this assay. 29 In another chromosomal aberration assay, a Chinese hamster lung fibroblast cell line (CHL/IU) was used (direct and metabolic activation methods), and magnesium sulfate was evaluated at concentrations up to 5.0 mg/mL, with and without metabolic activation. 30 Magnesium sulfate did not induce an increase in the incidence of chromosomal aberrations or genome mutation (polyploidy) in this assay.
The frequency of sister chromatid exchanges (SCEs) in cultures of human peripheral blood lymphocytes (from single donor) incubated with magnesium sulfate was evaluated. 33 Cultures containing the following concentrations of magnesium sulfate were incubated for 96 hours: 62.5 µg/mL, 125 µg/mL, 250 µg/mL, 500 µg/mL, and 1,000 µg/mL. At each experimental point and in the corresponding control (unspecified), 40 metaphases of the second mitosis were analyzed. At all concentrations tested, the frequency of SCEs in cultures incubated with magnesium sulfate did not differ significantly (P > 0.05) from that of the control (6.20 ± 0.43 µg/mL). It was concluded that magnesium sulfate was not genotoxic.
Antigenotoxicity
The effect of magnesium sulfate on metal-induced mutagenicity was evaluated in the Ames test using S typhimurium strain TA97. 34 The preincubation mutagenicity test was performed in triplicate by adding the following, in that order, to tubes incubated for 30 minutes: 500 µL buffer or ddH20, freshly prepared metal dilutions (50 µL), bacterial cell culture (100 µL), and MgSO4 (50 µL). One of the following metals was included in one of the tubes (final volume = 700 µL), which was incubated for 30 minutes: Co++ (up to 800 µmol/L), Fe++ (up to 1,000 µmol/L), Mn++ (up to 1,200 µmol/L), Zn++ (up to 1,000 µmol/L), and Cd++ (up to 200 µmol/L). Magnesium sulfate inhibited the mutagenicity of Fe++, Mn++, and Zn++; had only a slight effect on the mutagenicity of Co++; and had no effect on the mutagenicity of Cd++.
Carcinogenicity
The tumorigenicity of magnesium sulfate fibers (diameter = 0.45 µm; length = 22.4 µm) was evaluated using 20 female Syrian hamsters. 35,36 Each 500 mg of fiber was suspended in 50 mL of sterilized saline with 0.25 g of sodium carboxymethylcellulose to delay fiber sedimentation. The magnesium sulfate fiber suspension was sonicated and then injected intratracheally (0.2 mL/animal) once per week for 5 weeks. Vehicle alone was administered to 20 control hamsters according to the same procedure. At 2 years postadministration, the animals were killed and necropsy performed. The following changes were observed in the lungs: fibrosis, pleural thickening, and chronic inflammatory changes. However, these changes appeared to have been too mild to promote the development of pneumoconiosis. Due to solubility, magnesium sulfate fibers could not be detected in the lung tissue of hamsters at 2 years postadministration. There were 9 tumor-bearing animals in the group dosed with magnesium sulfate fibers, and the tumors were defined as follows: adrenal gland (a neuroblastoma, a cortical adenoma, and A & B cell tumor), pleural mesothelioma (2 epithelial types), kidney (a malignant histiocytoma and an anaplastic tumor), lung (1 tumor, unspecified cell type), uterus (1 leiomyosarcoma), and bladder (1 leiomyoma). Malignant histiocytoma of the kidney and leiomyosarcoma of the uterus were observed in the same hamster. Tumors were not observed in vehicle control hamsters.
Wistar rats were exposed (inhalation exposure) to long and short magnesium sulfate whiskers 5 days per week (6 h/d) for 1 year. 16 Long and short whiskers were defined as those having a mean diameter of 1.8 µm and 1.5 µm, respectively. The exposure groups were defined as follows: long whisker (27 rats), short whisker (27 rats), and controls (26 rats). Long and short whiskers were tested at mean concentrations of 4.0 mg/m3 and 2.3 mg/m3, respectively. Few whiskers were detected in rat lungs, even at day 1 postexposure. This finding suggested that magnesium sulfate whiskers are dissolved and eliminated rapidly from the lungs. Histopathological examination indicated that the lung tumor incidence was not significantly different from that of control rats.
Dermal Irritation and Sensitization
Irritation
A preliminary skin irritation study (mice) on magnesium sulfate (in propylene glycol) was performed prior to the skin sensitization study summarized below. 19 The test system, procedures, and techniques used in the skin irritation study were identical to those used during days 1 to 3 in the skin sensitization study, unless otherwise specified. Two young adult mice were used, and the ears were treated with 50% magnesium sulfate on 3 consecutive days. The test sites were evaluated for irritation at approximately 3 to 4 hours after the last application. Skin irritation of the ears was not observed in any of the animals tested with 50% anhydrous magnesium sulfate. Additionally, there was no evidence of macroscopic abnormalities of the surrounding area.
Sensitization
The skin sensitization potential of anhydrous magnesium sulfate (in propylene glycol) was evaluated using the mouse local lymph node assay, according to Organization for Economic Cooperation and Development Guideline 429. 19 Three groups of 5 mice were used, and the dorsal surface of both ears was epidermally treated with the test substance (10%, 25%, and 50%) at a dose volume of 25 µL/ear. A vehicle control group was also included in the study. The animals were then injected iv with 3H-methyl thymidine, killed, and the draining auricular lymph node of each ear was excised. Lymph nodes were pooled for each animal, and cell suspensions prepared. The stimulation index (SI) was calculated for each group. The SI is defined as the ratio of the DPM/group compared to the DPM/vehicle control group. Because there was no indication that the test substance elicited an SI of ≥3 when tested up to a concentration of 50%, anhydrous magnesium sulfate was considered a nonsensitizer.
Case Reports
Two patients (29 and 32 years old) were treated iv with magnesium sulfate for preterm labor. 37 Both patients were started with a 4 mg iv loading dose of magnesium sulfate. An urticarial reaction, rapid and sudden onset, was observed in both patients, and the eruption cleared when dosing with magnesium sulfate was discontinued.
A 29-year-old female presented with generalized tonic–clonic seizure at 17 hours postpartum. 38 Infusion with magnesium sulfate involved a loading dose of “4 g (16 mmol),” and the patient received 100 mmol over approximately 20 minutes. A peak serum magnesium level of 6.87 mmol/L was reported. The absence of circulatory compromise and arrhythmias was noted. 39
Summary
Magnesium sulfate functions as a bulking agent in cosmetic products. According to information supplied to the FDA by industry in 2014 as part of the VCRP, magnesium sulfate is being used in a number of rinse-off and leave-on cosmetic products. Results from a survey of ingredient use concentrations provided by the Council in 2013 indicate that this ingredient is being used at concentrations up to 25% (in paste masks—rinse-off products) and 11% (in hair sprays—leave-on products).
Hydrated magnesium sulfate can be prepared by dissolving magnesium oxide, magnesium hydroxide, or magnesium carbonate in sulfuric acid. Heating of this hydrate reversibly drives off water and produces anhydrous magnesium sulfate.
Magnesium sulfate crossed the placenta and entered the fetal brain and other tissues, following sc injection into pregnant rats. In patients with eclampsia dosed iv or intramuscularly with magnesium sulfate, cumulative renal excretions ranged from 38% to 53% of the injected dose at the end of 4 hours. Urinary excretion of inorganic sulfate (30.2% ± 17.2% of administered dose) was noted in healthy male participants during the first 24 hours after oral dosing with magnesium sulfate. Excretion during the next 48 hours was negligible.
Anhydrous magnesium sulfate (50%) did not induce systemic toxicity when applied to the skin of mice.
Magnesium sulfate was administered iv to groups of Crj:CD(SD) rats at doses up to 450 mg/kg, and the LD50 values were 206 mg/kg and 174 mg/kg for males and females, respectively. In a second experiment, magnesium sulfate was infused into groups of female Beagle dogs at doses up to 1,200 mg/kg for 6 hours. Deaths were not observed in any of the dose groups.
Male Wistar rats inhaled 2 types of magnesium sulfate whiskers 5 days per week (6 h/d) for 4 weeks. Short whisker (mean diameter = 1.5 µm) was tested at a mean concentration of 2.3 mg/m3 and long whisker (mean diameter = 1.8 µm) was tested at a mean concentration of 4.0 mg/m3. Few whiskers were detected in rat lungs, even at day 1 postexposure, and this finding suggested that magnesium sulfate whiskers are dissolved and eliminated rapidly from the lungs. There was no indication that adverse effects were observed.
Female Beagle dogs received 24-hour iv infusions of magnesium sulfate at doses up to 200 mg/kg/h. An NOAEL of 50 mg/kg/h was reported. In another similar study, an NOAEL of ≤50 mg/kg/h was reported for female Beagle dogs infused with magnesium sulfate at doses up to 100 mg/kg/h for 2 weeks.
Anhydrous magnesium sulfate (50%) did not induce skin irritation in mice and was classified as a nonsensitizer in the mouse local lymph node assay.
An urticarial reaction was observed in 2 patients who were treated iv with magnesium sulfate (4 mg) for preterm labor. There was no evidence of circulatory compromise or arrhythmias in a female patient with generalized tonic–clonic seizure postpartum who was subsequently infused with magnesium sulfate (100 mmol) over ∼20 minutes.
In a reproductive and developmental toxicity study, groups of pregnant female rats received doses up to 1,000 mg/kg sc 3 times per day on gestation days 15 through 20. The NOAEL for general toxicological effects on the dams was 250 mg/kg/d, and the NOAELs for reproductive and developmental toxicity were 1,000 mg/kg/d and 500 mg/kg/d, respectively. Human studies of infants from mothers dosed parenterally with magnesium sulfate during pregnancy are available. In 2 studies, there were no observable deleterious effects on the fetus/newborn. In an additional study, it was hypothesized that prolonged infusion of magnesium sulfate may lead to fetal parathyroid suppression, with consequent abnormalities resembling rickets.
Magnesium sulfate was not genotoxic in in vitro assays involving bacterial and mammalian cells. Following intratracheal administration of magnesium sulfate manmade fiber (diameter = 0.45 µm; length = 22.4 µm) to 20 hamsters at a dose of 2 mg weekly for 5 weeks, tumors were observed in 9 hamsters. The primary sites of the tumors were the pleural cavity, intracelial organs, kidney, adrenal gland, bladder, and uterus. In another study, the tumorigenicity of magnesium sulfate was also evaluated using 20 hamsters. Each 500 mg fiber was suspended in sterilized saline and sodium carboxymethylcellulose and injected intratracheally (0.2 mL per animal). Again, tumors were observed in basically the same organs of 9 hamsters.
Groups of 27 Wistar rats were exposed (inhalation exposure) to long and short magnesium sulfate whisker 5 days per week (6 h/d) for 1 year. Long and short whiskers had mean diameters of 1.8 µm and 1.5 µm, respectively. These materials were tested at mean concentrations of 4.0 mg/m3 (long whisker) and 2.3 mg/m3 (short whisker), respectively. Histopathological examination indicated that the lung tumor incidence was not significantly different from that of control rats.
Discussion
The Panel noted that the history of safe medical use of magnesium sulfate indicates no significant toxicity concerns relating to systemic exposure to magnesium sulfate. Furthermore, the extensive clinical experience of the Panel, including the results of numerous patch tests, indicates that magnesium salts do not have the potential to induce sensitization. The Panel also noted that results were negative for 50% magnesium sulfate formulation in a mouse skin irritation study and in an in vitro sensitization assay. The highest reported maximum use concentration for magnesium sulfate in any leave-on product category (hair sprays) is 11%.
Magnesium sulfate is being used at concentrations up to 11% in pump hair sprays and at concentrations up to 1% in foot powders and sprays. The Panel discussed the issue of incidental inhalation exposure from propellant and pump sprays and powders and considered pertinent data indicating that incidental inhalation exposures to this ingredient in such cosmetic products would not cause adverse health effects. The data considered include data characterizing the potential for this ingredient to cause acute toxicity, repeated dose (inhalation) toxicity, reproductive and developmental toxicity, and carcinogenicity. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Cosmetic Ingredient Review Expert Panel concluded that magnesium sulfate is safe in cosmetics in the present practices of use and concentration described in the safety assessment.
Footnotes
Author Contributions
Johnson contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Andersen, Bergfeld, Belsito, Hill, Klaassen, Liebler, Marks, Shank, Slaga, and Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The infrastructure of Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.