Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of 11 alkyl betaines as used in cosmetics. These ingredients are reported to function as hair and skin conditioning agents, antistatic agents, surfactants-cleansing agents, and viscosity-increasing agents in cosmetic products. Although there are data gaps, the shared chemical core structure, similar functions and concentrations of use in cosmetics, and the expected similarities in physicochemical properties enabled grouping these ingredients and reading across the available toxicological data to support the safety assessment of each individual compound in the entire group. The Panel concluded alkyl betaines were safe as cosmetic ingredients in the present practices of use and concentration, when formulated to be nonirritating.
Keywords
Introduction
This safety assessment addresses the safety of 11 alkyl betaines as used in cosmetics. The parent compound, betaine, is a naturally occurring N-trimethylated amino acid, also called trimethylglycine, and can be isolated from sugar beets. 1 It is a common component in the human diet. As given in the International Cosmetic Ingredient Dictionary and Handbook, these ingredients are reported to function as hair and skin conditioning agents, antistatic agents, surfactants-cleansing agents, and viscosity-increasing agents in cosmetic products. The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) has reviewed the safety of cocamidopropyl betaine and related amidopropyl betaines as used in cosmetics. 2 The Panel concluded that these ingredients “were safe in cosmetics as long as they are formulated to be nonsensitizing, which may be based on a quantitative risk assessment.”
The common core chemical structure, similar functions and concentrations in cosmetics, and the predicted physicochemical properties enabled grouping of these ingredients and reading across the available toxicological data to support the safety assessment of each individual compound in this group. Toxicological data on betaine and betaine analogs (synonym: betaines, C12-C14 [even numbered] alkyldimethyl, or C12-C14 alkyldimethyl betaines) in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. These data are available on the ECHA website. 3,4
Chemistry
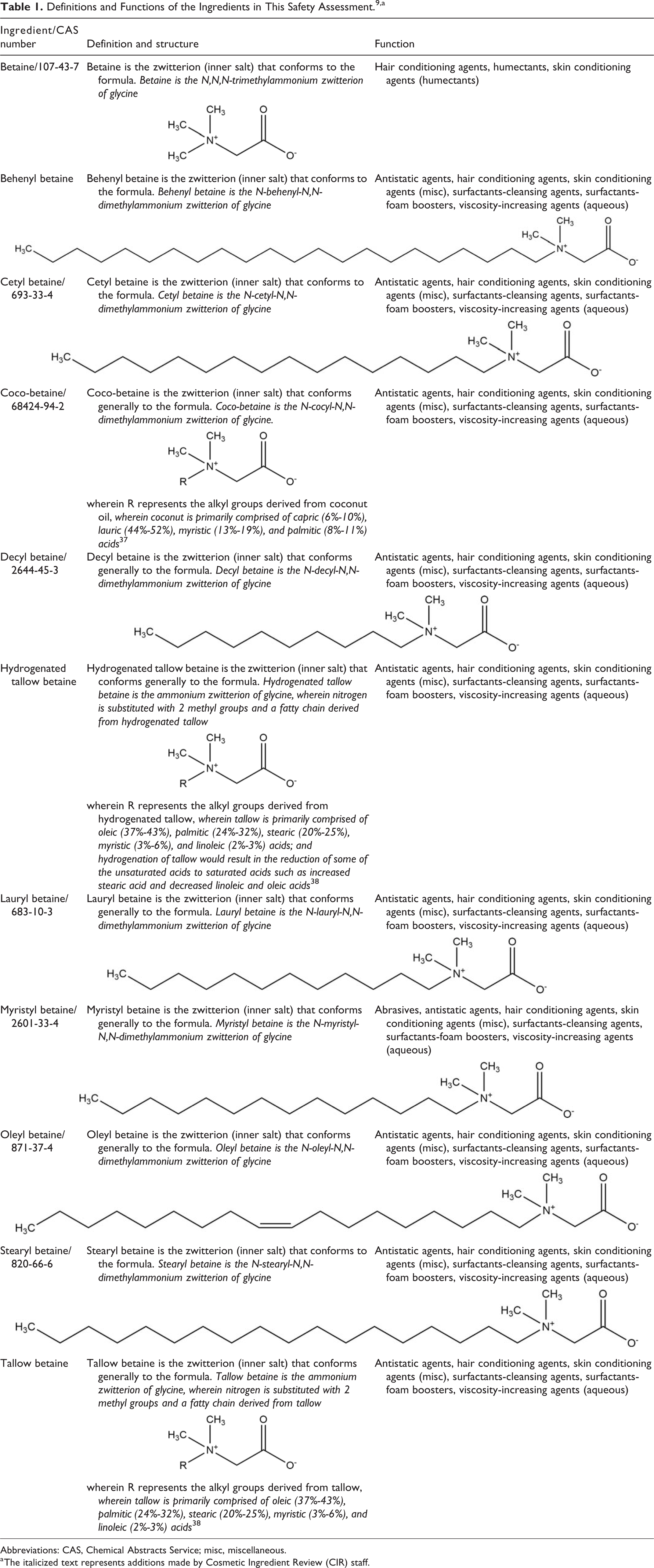
The definitions of the 11 alkyl betaines in this safety assessment and formulas and idealized structures can be found in Table 1. The alkyl betaines are zwitterionic ingredients comprised of tertiary ammonium substituted acetate. These ingredients have a core structure of 2-(alkyldimethylammonio)acetate (ie, N,N,N-trisubstituted glycine; Figure 1).
Definitions and Functions of the Ingredients in This Safety Assessment. 9,a
Abbreviations: CAS, Chemical Abstracts Service; misc, miscellaneous.
a The italicized text represents additions made by Cosmetic Ingredient Review (CIR) staff.
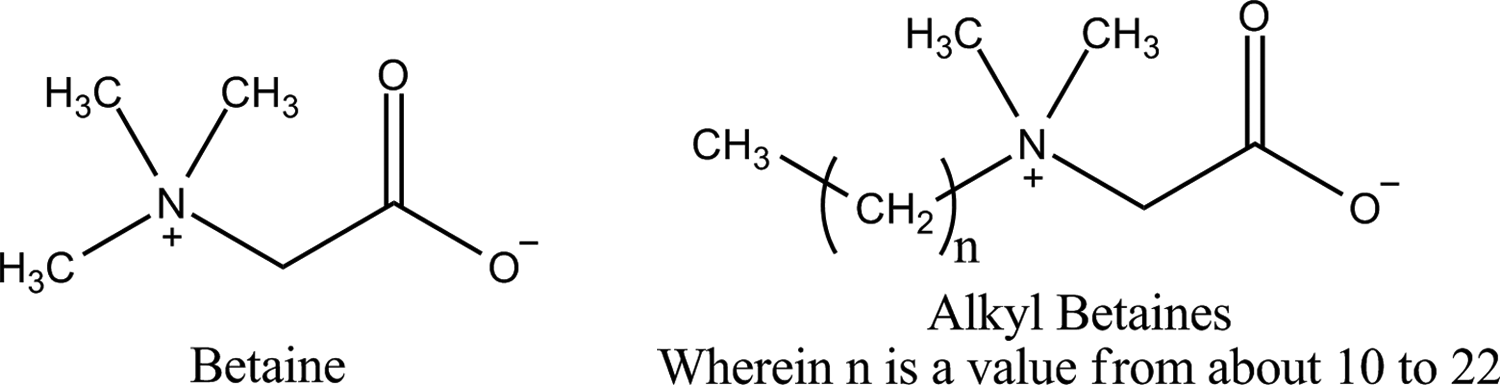
Betaine and the alkyl betaines.
Therein, the “alkyl” is either methyl, as in the case of betaine itself, or a longer chain alkyl group ranging in length from about 10 (eg, decyl betaine) to about 22 (eg, behenyl betaine) carbons. However, C10 to C22 is an estimate, as the compositions of the ingredients derived from plant and animal sources (eg, coco-betaine, tallow betaine, and hydrogenated tallow betaine) are variable.
The zwitterionic structures of these ingredients make them amphoteric, a hallmark characteristic of surfactants. The fatty chains, found on most of these ingredients, add a lipophilic tail to these hydrophilic head groups, further imparting surfactant properties. Those ingredients for which chemical property data were available are colorless, crystalline materials with good solubility in water and polar organic solvents. While ingredients that vary from these only by incremental alkyl chain length changes are likely to have similar profiles, chemical and physical properties were neither publicly available nor submitted by other parties for any of the other ingredients.
Physical and Chemical Properties
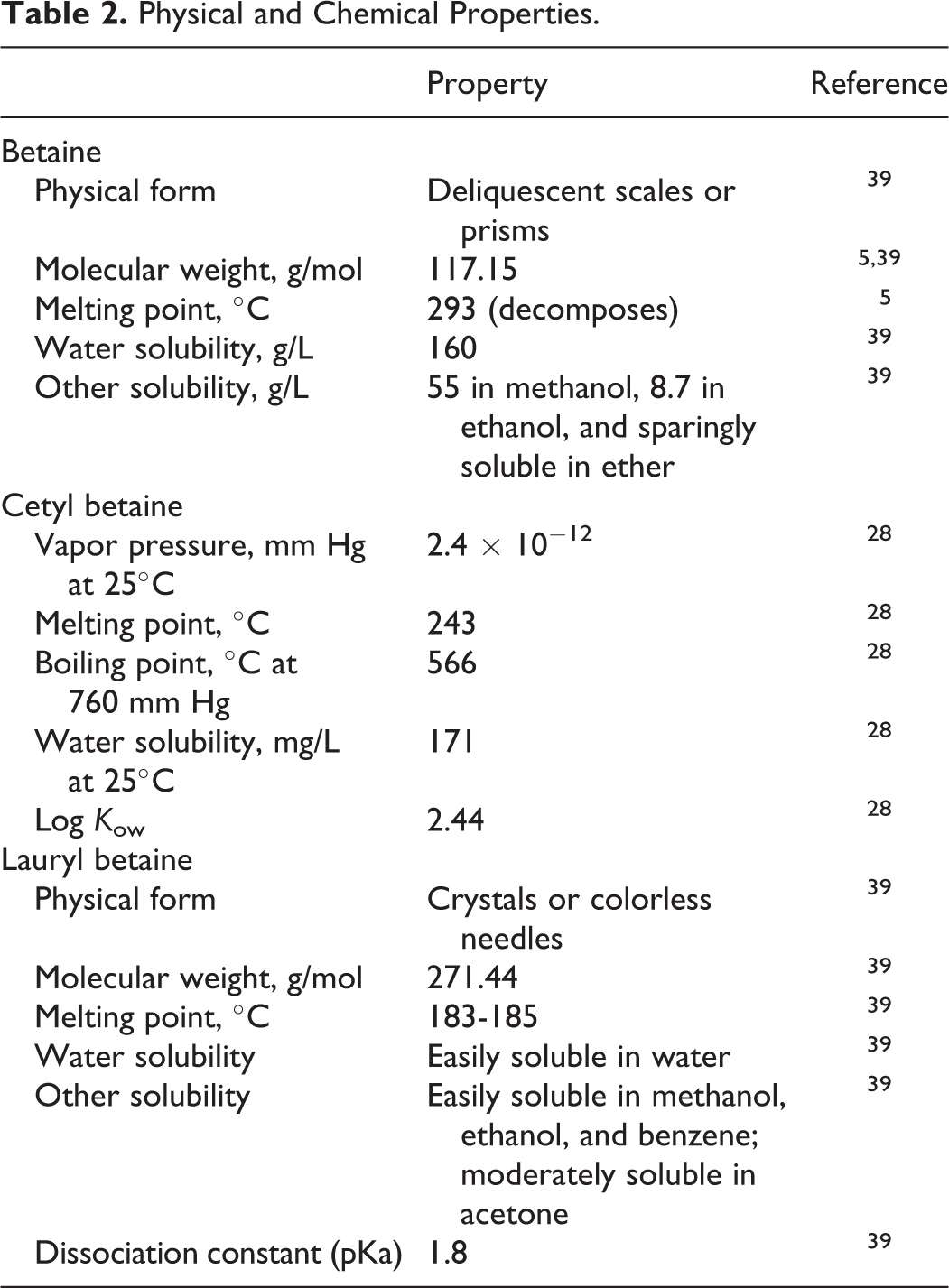
Available chemical properties can be found in Table 2.
Physical and Chemical Properties.
Method of Manufacturing
Betaine
Betaine (food grade) may be extracted from sugar beets via liquid chromatographic separation from sugar beet molasses. 5,6 It is subsequently refined and crystallized. Betaine anhydrous (as animal feed additive) is produced by reacting chloroacetic acid and sodium hydroxide with heat and stirring. 7 Trimethylamine is then added to the mixture, and the resultant solutions are filtered and purified. Betaine hydrochloride (as animal feed additive) follows the same synthesis pathway as betaine anhydrous, except that hydrochloric acid is added and the filtrate is purified. 7
Coco-betaine
In data supplied by a manufacturer, coco-betaine is produced by reacting fatty dimethyl amines from coconuts with chloroacetic acid in aqueous solution. 8
Impurities
Betaine
Betaine (food grade) contains very small quantities of chloride, sulfate, and heavy metals. 6 Trace analysis shows very small amounts of polychlorinated biphenyl (PCB), polycyclic aromatic hydrocarbon, and dioxins. No pesticide traces have been detected. Betaine does not contain methanol, ethanol, or isopropanol (limits of detections were 5.0, 2.5, and 0.5 ppm, respectively).
Betaine anhydrous and betaine hydrochloride (as animal feed additives) contained <2.0 mg/kg arsenic and <10 mg/kg heavy metals (expressed as lead). 7 Dioxin content was <0.50 ng/kg and PCB content was <0.35 mg/kg. Betaine content for the anhydrous and hydrochloride forms was ≥96% and ≥93%, respectively.
Coco-betaine
According to information supplied by a manufacturer, coco-betaine is composed of approximately 31% coco-betaine, 7% sodium chloride, and 62% water. 8 There are no solvents, preservatives, or other additives. The product may contain a maximum of 100 ppm dichloroacetic acid, 100 ppm monochloroacetic acid, 0.5% free amines, 2% glycolic acid, 20 ppm heavy metals (copper, lead, tin, platinum, palladium, mercury, arsenic, cadmium, antimony, nickel, chromium, and cobalt), 2 ppm arsenic, 10 ppm iron, and <3% volatile organic compounds.
C12 to C14 alkyldimethyl betaines
According to information supplied to ECHA, betaines, C12 to 14 alkyldimethyl (a mixture of lauryl betaine and myristyl betaine) consists of betaine, C12-alkyldimethyl; betaine, C14-alkyldimethyl; (carboxylatomethyl) hexadecyldimethylammonium; sodium chloride; sodium glycolate; and unknown impurities. 4 Percentage composition was not provided, and there are no further details.
Use
Cosmetic
The safety of the cosmetic ingredients addressed in this safety assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
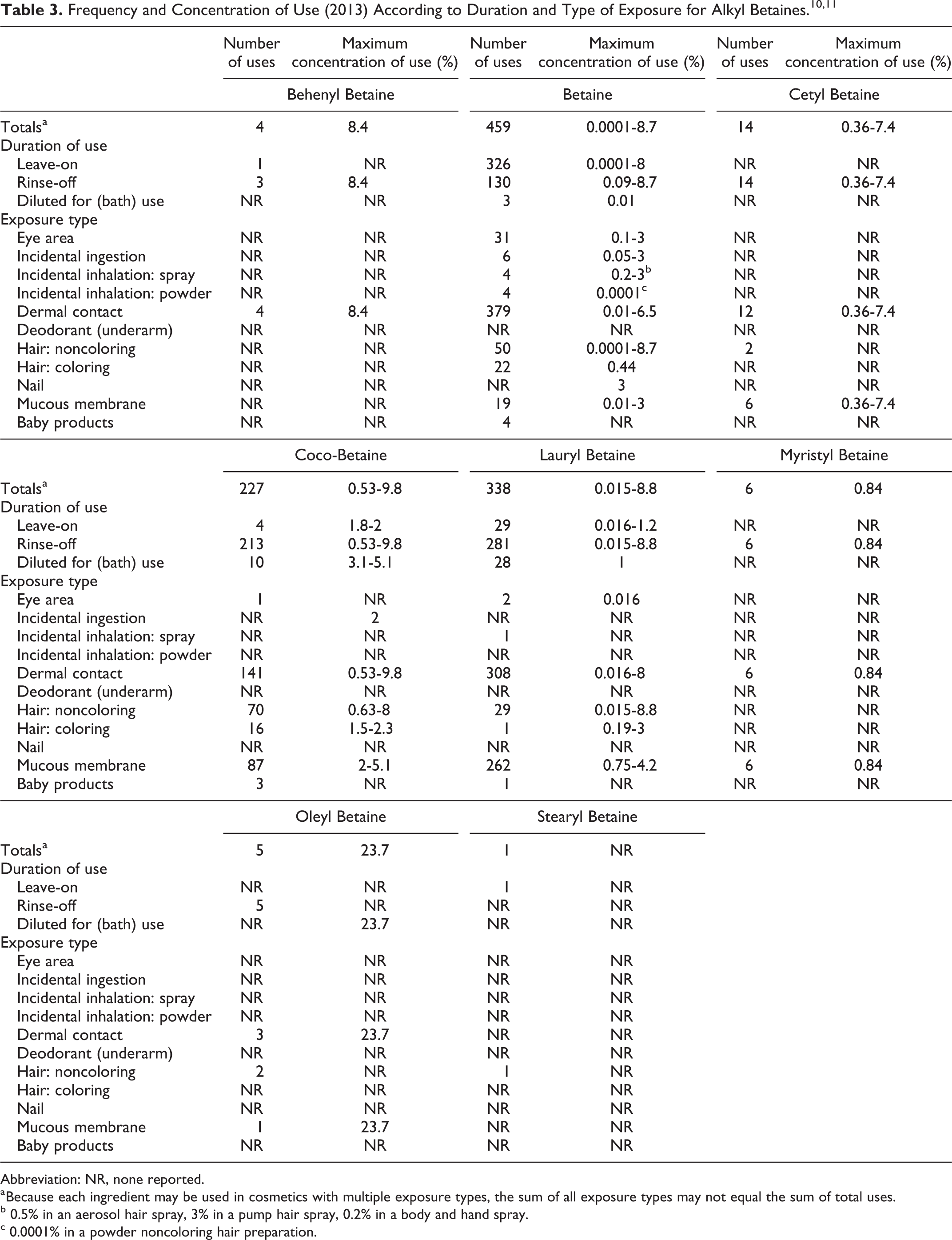
Table 3 presents the current product formulation data for alkyl betaines. Betaine mainly functions as a hair conditioning agent, humectant, and skin conditioning agent—humectant in cosmetic products. 9 The remaining alkyl betaines additionally function as antistatic agents, skin conditioning agents—miscellaneous, surfactants-cleansing agents and foam boosters, and viscosity-increasing agents. According to 2013 VCRP data, betaine has the most reported uses in cosmetic and personal care products, with a total of 459; the majority of the uses are in leave-on skin care preparations. 10 Lauryl betaine has the second greatest number of overall uses reported, with a total of 338; the majority of those uses are in rinse-off personal cleanliness products.
Abbreviation: NR, none reported.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b 0.5% in an aerosol hair spray, 3% in a pump hair spray, 0.2% in a body and hand spray.
c 0.0001% in a powder noncoloring hair preparation.
According to the VCRP data, decyl betaine, tallow betaine, and hydrogenated tallow betaine have no reported uses in cosmetics in the United States. In the Council’s use concentration survey, betaine had a maximum use concentration range of 0.0001% to 8.7%, with the 8.7% reported in rinse-off noncoloring hair conditioners. 11 Lauryl betaine had a maximum use concentration range of 0.015% to 8.8%, with 8.8% reported in rinse-off noncoloring hair shampoos. The Council reports that they do not have any suppliers listed for decyl betaine, hydrogenated tallow betaine, stearyl betaine, or tallow betaine. 12
Betaine and lauryl betaine were reported to be used in hair sprays, body and hand products, noncoloring hair powders, and indoor tanning preparations that may be aerosolized or become airborne and could possibly be inhaled. For example, betaine is used in hair sprays at up to 3% and in noncoloring powders at up to 0.0001%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm as compared with pump sprays. 13 –16 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 14,15 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1,000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace. 17 –19 Alkyl betaines are not restricted from use in any way under the rules governing cosmetic products in the European Union. 20
Noncosmetic
A biocide that contains cetyl betaine was concurrently being studied, during the preparation of this report, as a preventative treatment of human immunodeficiency virus type 1 and other sexually transmitted diseases in vaginal microbicides and contraceptives. 21,22 Betaine hydrochloride has been approved by the FDA to treat homocystinuria (by reducing homocysteine levels). 23 It is also present as an active ingredient in over-the-counter digestive aids; however, the FDA has determined that there are inadequate data to establish general recognition for the safety and effectiveness of the ingredient for this specified use (21 CFR §310.545).
Toxicokinetics
Betaine
Low percutaneous permeation for betaine was observed in a percutaneous absorption study using Franz chambers with freshly isolated human epidermis. 3 The study followed Organization for Economic Cooperation and Development Guideline 428, but was not Good Laboratory Practice compliant. Betaine at 5% in saline or emulsion was applied to the epidermis samples. The exposure was observed for 24 hours. Approximately 0.1% of the applied dose in both vehicle types permeated through the epidermis samples.
The pharmacokinetics and acute effects on plasma total homocysteine of orally administered betaine (see “use of betaine hydrochloride in treating homocystinuria”) were assessed in healthy human volunteers (3 men and 7 women). 24 Information on the absorption and elimination of betaine was also developed. In a double-blind crossover study, each patient ingested the betaine in doses of 1, 3, and 6 g mixed with 150 mL orange juice. The doses were ingested 7 days apart following a 12-hour overnight fast. Blood samples for serum betaine concentration measurement were drawn just before receiving the betaine dose, at 20-minute intervals during the first 3 hours and then at 4, 7, and 24 hours postdosing. Urine samples were collected before dosing and during the 24-hour follow-up period. Within 2 hours, a dose-dependent increase in serum betaine concentration was observed. Absorption and elimination of betaine were dose dependent, with urinary excretion of betaine increasing with betaine dose. Only a very small proportion of the ingested betaine was excreted in urine, however, with 3.2%, 4.3%, and 7.4% of the 1, 3, and 6 g doses accounted for, respectively.
Cetyl Betaine and Lauryl Betaine
The absorption of radiolabeled cetyl betaine (5.4 mM) and lauryl betaine (16 mM) was determined using diffusion cells containing excised hairless mouse skin. 25 Lauryl betaine was well absorbed into the receptor phase (approximately 50% of the applied dose within 24 hours), while cetyl betaine partitioned into the skin but slowed transfer to the receptor phase (approximately 1.3% of the applied dose absorbed within 24 hours). Skin digests at the end of the 24 hours found that 15% of the applied dose of cetyl betaine and 25% of the applied dose of lauryl betaine were associated with the tissue. This study also examined the effects of cetyl betaine and lauryl betaine (same concentrations as used in the absorption study) on skin barrier function in hairless mouse skin in vitro. Excised skin was pretreated with each test material for 16 hours. After pretreatment, the permeation of the model compound, nicotinamide, across membranes was measured and the results were compared to the flux across control membranes that were exposed to buffer alone. All surfactants decreased skin barrier function to some extent. The degree of nicotinamide penetration enhancement was correlated with the ratio of the surfactant pretreatment concentration to the surfactant critical micelle concentration. The authors of the study suggested that solubilization of stratum corneum lipids may be an important mechanism explaining the effects observed.
The dermal uptake of cetyl betaine and lauryl betaine was measured in vivo with human skin. 26 Male volunteers received 14C-radiolabeled test materials in aqueous solution on the dorsal upper arm for 30 minutes. The concentrations of cetyl betaine and lauryl betaine applied were 0.14, 1.0, and 5.4 mM and 16, 100, and 800 mM, respectively. The positive control was 50 mM caffeine. At the end of the exposure period, the remaining test materials were rinsed from the skin, and the skin was washed. The stratum corneum at the test sites was removed with repeated tape stripping. Dermal uptake was assessed by measuring the recovered radioactivity from the tape strips and compared to predicted penetration values. The measured uptake of cetyl betaine and lauryl betaine was 28 to 160 nmol/cm2 and 2.3 to 19.5 nmol/cm2, respectively. The predicted penetration values were 51 to 292 nmol/cm2 for cetyl betaine and 3.7 to 35 nmol/cm2 for lauryl betaine. Caffeine penetrated at expected amounts. The tape stripping indicated that the radiolabel was mostly found in the outer layers of the stratum corneum.
The same study also assessed skin barrier function using the same test concentrations for both test materials. 26 Nonradiolabeled cetyl betaine and lauryl betaine were applied to the skin for 30 minutes. The transepidermal water loss (TEWL) was assessed. No changes in TEWL values were observed after treatment of the skin with the betaines or with saline controls.
Toxicological Studies
Acute Toxicity
Acute toxicity studies are presented in Table 4. The oral LD50 values of betaine, cetyl betaine, lauryl betaine, and C12 to C14 alkyldimethyl betaines were 11.1 g/kg, 1.62 g/kg, 0.071 g/kg, and 3 mL/kg, respectively, in rats, and 2.64 g/kg for coco-betaine (30%) in a mouse study. Also in rats, the dermal LD50 values were greater than 16 g/kg for cetyl betaine and 1.3 g/kg for lauryl betaine. The intravenous LD50 of betaine in mice has been reported to be 0.83 g/kg body weight (bw). The LD50 values were 0.15 g/kg for cetyl betaine and 0.053 g/kg for lauryl betaine in an intraperitoneal study in rats. 3 –5,25
Acute Toxicity Studies in Animals.

Abbreviations: bw, body weight; OECD, Organization for Economic Cooperation and Development; GLP, Good Laboratory Practice.
Repeated Dose Toxicity
Repeated dose toxicity data are summarized in Table 5. No effects were observed at the highest dose tested for betaine in rats. No significant toxic effects were observed in rats that received up to 0.35 g/kg/d cetyl betaine in a 91-day oral study. The no observed effect level (NOEL) for coco-betaine was 250 mg/kg/d and the lowest observed effect level (LOEL) was 500 mg/kg/d in a 90-day oral study in rats when tested up to 500 mg/kg/d. In a study of C12 to C14 alkyldimethyl betaines, systemic no observed adverse effects levels (NOAELs) were 50 and 100 mg/kg bw/d in oral rat studies that tested the material up to 300 and 1,000 mg/kg/d, respectively. The systemic lowest observed adverse effects levels (LOAELs) for these 2 studies were 150 (due to increased salivation, increased urea, and nonneoplastic histopathologic changes in the kidney and bladder) and 300 mg/kg bw/d (due to decreased food consumption, bw gain, and absolute bw), respectively. 3,4,27
Repeated Dose Toxicity in Animals.

Abbreviations: bw, body weight; GLP, Good Laboratory Practice; LOAEL, lowest observed adverse effects level; LOEL, lowest observed effect level; OECD, Organization for Economic Cooperation and Development; NOAEL, no observed adverse effects level; NOEL, no observed effect level.
Reproductive and Developmental Toxicity
Reproductive and developmental toxicity studies are summarized in Table 6. Dermal reproductive and developmental toxicity studies of cetyl betaine in rabbits determined the maternal LOAEL to be 10 mg/kg/d due to decreased bw gain and a maternal NOAEL could not be established. The developmental LOAEL was 40 mg/kg/d and the developmental NOAEL was 20 mg/kg/d. In oral reproductive and developmental toxicity studies of cetyl betaine in rats, the LOAEL for the dams was 50 mg/kg due to decreased bw gain and a maternal NOAEL could not be calculated. The developmental LOAEL was 250 mg/kg and the developmental NOAEL was 150 mg/kg. In an oral C12 to C14 alkyldimethyl betaines study, the reproductive NOEL was 150 mg/kg bw/d and the reproductive LOAEL was 300 mg/kg bw/d due to decreased pup weight and litter size and increased postimplantation loss and postnatal loss. Another oral C12 to C14 alkyldimethyl betaines study determined the reproductive NOAEL to be 300 mg/kg/d when the test material was tested up to 1,000 mg/kg/d. 4,28,29
Reproductive and Developmental Toxicity.

Abbreviations: bw, body weight; GLP, Good Laboratory Practice; LOAEL, lowest observed adverse effects level; NOAEL, no observed adverse effects level; OECD, Organization for Economic Cooperation and Development.
Carcinogenicity
Repeated dose toxicity results are presented in Table 5. Betaine was not carcinogenic when tested up to 5% in a 104-week dietary rat study. 3
Genotoxicity
In vitro and in vivo genotoxicity studies are presented in Table 7. Betaine and C12 to C14 alkyldimethyl betaines were not genotoxic in in vitro and in vivo studies. 3 –5
Genotoxicity.
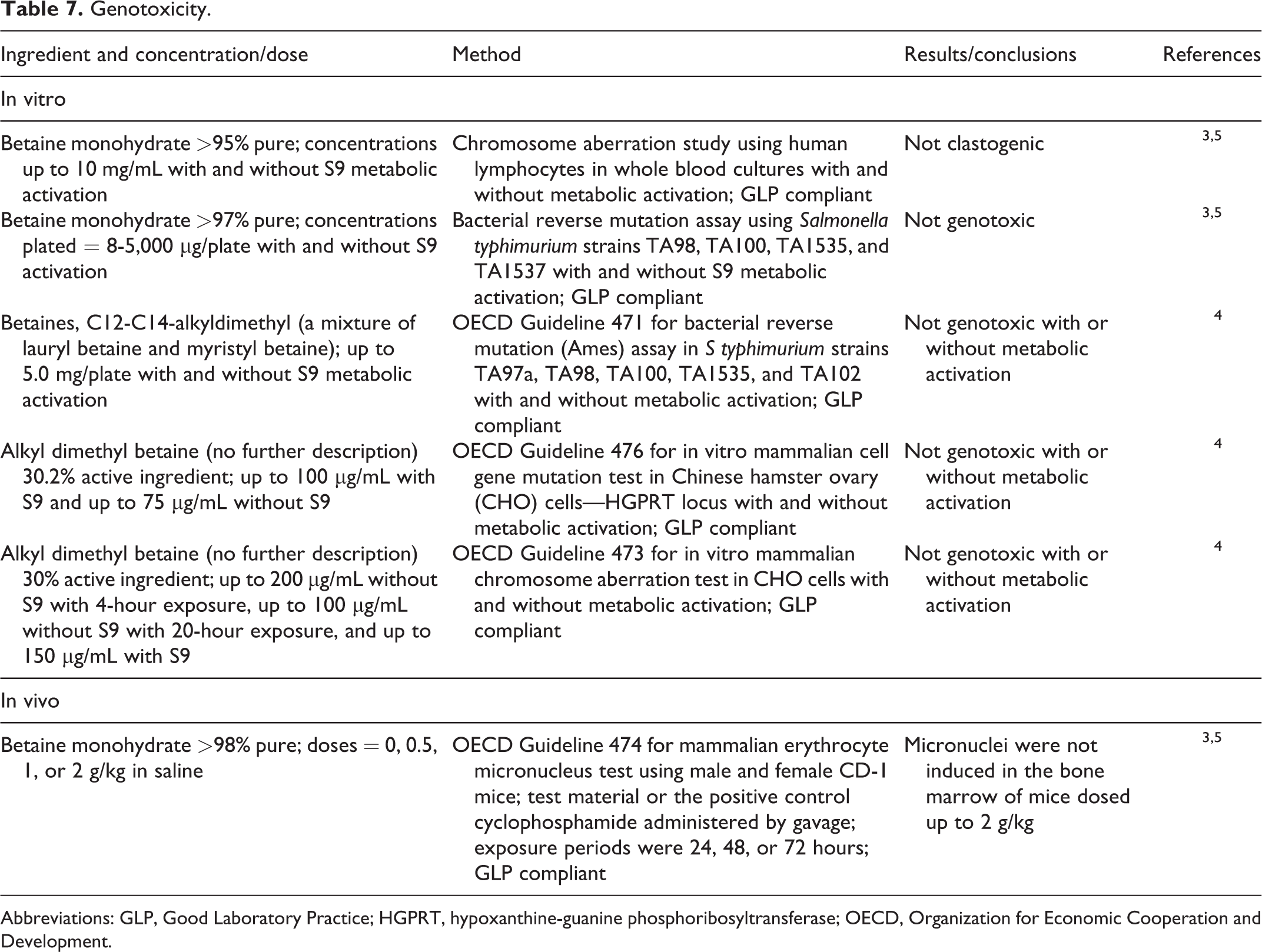
Abbreviations: GLP, Good Laboratory Practice; HGPRT, hypoxanthine-guanine phosphoribosyltransferase; OECD, Organization for Economic Cooperation and Development.
Irritation and Sensitization
Irritation and Anti-Irritation
Nonhuman and human irritation and anti-irritation studies are presented in Table 8. Betaine had anti-irritating effects on the skin in several efficacy studies in humans. In dermal studies, coco-betaine was not irritating in a rabbit study when tested at 16% and was less irritating than sodium lauryl sulfate (SLS) in a human study at an unknown concentration. No dermal irritation reactions were observed in human studies of lauryl betaine at 0.1%, but were observed at concentrations of 1% and 10%. Dermal irritation results were mixed in rabbit studies of C12 to C14 alkyldimethyl betaines, with irritation observed at 30% and at an unknown concentration in 2 studies, and no irritation was observed in 2 other studies at unknown concentrations. Betaine at 10% was not an ocular irritant in rabbits nor were C12 to C14 alkyldimethyl betaines at unknown concentrations in several rabbit studies; however, coco-betaine at 16% and 30% and lauryl betaine at 10% were ocular irritants. In human mucosal studies testing the efficacy of toothpaste, betaine did not produce adverse effects. 1,3 –5,30 –33
Irritation and Anti-Irritation Studies.

Abbreviations: GLP, Good Laboratory Practice; OECD, Organization for Economic Cooperation and Development; .
Sensitization
Nonhuman and human sensitization studies are presented in Table 9. Betaine (up to 50%), coco-betaine (up to 5%), lauryl betaine (0.1%), and C12 to C14 alkyldimethyl betaines (up to 100%) were not sensitizing in nonhuman and human dermal studies. 3 –5,34,35
Dermal Sensitization Studies.

Abbreviations: GLP, Good Laboratory Practice; LLNA, local lymph node assay; OECD, Organization for Economic Cooperation and Development.
Phototoxicity
No relevant published phototoxicity studies on alkyl betaines were discovered and no unpublished data were submitted.
Clinical Use
Case Reports
Coco-betaine
Two cases of eczematous lesions were reported following exposure to shampoos containing coco-betaine. 36 In the first case, a 44-year-old woman presented with acute eczematous lesions with erythema, edema, and vesiculation on the backs and palms of her hands a few days after using a shampoo with chestnut leaf extract. Her scalp also itched and was slightly red. Previous patch tests showed positive reactions to paraphenylenediamine, benzocaine, wool alcohols, parabens, chinoform, perfumes, nickel sulfate, and cobalt chloride. Patch tests with the shampoo and individual components showed a ++ reaction to the shampoo in open test as is, and in patch test at 2% aqueous (aq), ++ reaction to parahydroxybenzoic acid esters (5% pet.) and +++ reaction to coco-betaine (2% aq). No reactions were observed to the perfume component. The dermatitis cleared when the patient changed shampoos.
In the second case, a 22-year-old woman presented with red, swollen face and weeping eczematous lesions. Red, oozing, and crusted acute lesions were also observed on her shoulders and scalp. The symptoms occurred after using a new shampoo. Patch tests with the shampoo and the individual components showed a +++ reaction to the shampoo in open test as is, and in patch test at 2% aq, ++ reaction to coco-betaine (2% aq) and ++ reaction to sodium lauryl ether sulfate (2% aq). The symptoms cleared when the patient changed shampoos. 36
Summary
The alkyl betaines are zwitterionic ingredients comprised of tertiary ammonium substituted acetate. These cosmetic ingredients mainly function as hair and skin conditioning agents. With the exception of betaine, alkyl betaines may also function as antistatic agents, surfactants-cleansing agents, and viscosity-increasing agents. The common core chemical structure, similar functions and concentrations in cosmetics, and the expected absorption, distribution, and metabolism enabled grouping these ingredients and reading across the available toxicological data to support the safety assessment of each individual compound in the entire group.
According to information supplied to FDA’s VCRP in 2013, betaine has the most reported uses in cosmetic and personal care products, with a total of 459; the majority of the uses are in leave-on skin care preparations. Lauryl betaine has the second greatest number of overall uses reported, with a total of 338; the majority of those uses are in rinse-off personal cleanliness products. In an industry survey, betaine was reported to have a maximum use concentration range of 0.0001% to 8.7%, with the 8.7% reported in rinse-off noncoloring hair conditioner. Lauryl betaine was reported to have a maximum use concentration range of 0.015% to 8.8%, with 8.8% reported in rinse-off noncoloring hair shampoos.
Aside from use in cosmetics, betaine is a common food component and is used to treat homocystinuria. Absorption and elimination of betaine in humans were dose dependent, with urinary excretion of betaine increasing with betaine dose.
Cetyl betaine and lauryl betaine were observed to decrease skin barrier function in hairless mouse skin in vitro. Cetyl betaine and lauryl betaine absorbed into mouse skin in vitro, with lauryl betaine absorbing at a faster rate. Dermal penetration rates, measured from the stratum corneum collected from tape stripping, for cetyl betaine and lauryl betaine were 51 to 292 nmol/cm2 and 3.7 to 35 nmol/cm2, respectively, in 30-minute exposures to human skin in vivo.
The oral LD50 values of betaine, cetyl betaine, lauryl betaine, and C12 to C14 alkyldimethyl betaines were 11.1 g/kg, 1.62 g/kg, 0.071 g/kg, and 3 mL/kg, respectively, in rats, and 2.64 g/kg for coco-betaine (30%) in a mouse study. Also in rats, the dermal LD50 values were greater than 16 g/kg for cetyl betaine and 1.3 g/kg for lauryl betaine. The intravenous LD50 of betaine in mice has been reported to be 0.83 g/kg bw. The LD50 values were 0.15 g/kg for cetyl betaine and 0.053 g/kg for lauryl betaine in an intraperitoneal study in rats.
In repeated dose studies, no effects were observed at the highest dose tested (5%) for betaine in rats. No significant toxic effects were observed in rats that received up to 0.35 g/kg/d cetyl betaine in a 91-day oral study. The NOEL for coco-betaine was 250 mg/kg/d and the LOEL was 500 mg/kg/d in a 90-day oral study in rats when tested up to 500 mg/kg/d. In C12 to C14 alkyldimethyl betaines, systemic NOAELs were 50 and 100 mg/kg bw/d in oral rat studies that tested the material up to 300 and 1,000 mg/kg/d, respectively. The systemic LOAEL for these 2 studies were 150 (due to increased salivation, increased urea, and nonneoplastic histopathologic changes in the kidney and bladder) and 300 mg/kg bw/d (due to decreased food consumption, bw gain, and absolute bw), respectively.
Dermal reproductive and developmental toxicity studies of cetyl betaine in rabbits determined the maternal LOAEL to be 10 mg/kg/d due to decreased bw gain and a maternal NOAEL could not be established. The developmental LOAEL was 40 mg/kg/d and the developmental NOAEL was 20 mg/kg/d. In oral reproductive and developmental toxicity studies of cetyl betaine in rats, the LOAEL for the dams was 50 mg/kg due to decreased bw gain and a maternal NOAEL could not be calculated. The developmental LOAEL was 250 mg/kg and the developmental NOAEL was 150 mg/kg. In an oral C12 to C14 alkyldimethyl betaines study, the reproductive NOEL was 150 mg/kg bw/d and the reproductive LOAEL was 300 mg/kg bw/d due to decreased pup weight and litter size and increased postimplantation loss and postnatal loss. Another oral C12 to C14 alkyldimethyl betaines study determined the reproductive NOAEL to be 300 mg/kg/d when the test material was tested up to 1,000 mg/kg/d.
Betaine was not carcinogenic when tested up to 5% in a 104-week dietary rat study. Betaine and C12 to C14 alkyldimethyl betaines were not genotoxic in in vitro and in vivo studies.
Betaine had anti-irritating effects on the skin in several efficacy studies in humans. In dermal studies, coco-betaine was not irritating in a rabbit study when tested at 16% and was less irritating than SLS in a human study at an unknown concentration. No dermal irritation reactions were observed in human studies of lauryl betaine at 0.1% but were observed at concentrations of 1% and 10%. Dermal irritation results were mixed in rabbit studies of C12 to C14 alkyldimethyl betaines, with irritation observed at 30% and at an unknown concentration in 2 studies and no irritation observed in 2 other studies at unknown concentrations. Betaine at 10% was not an ocular irritant in rabbits nor were C12 to C14 alkyldimethyl betaines at unknown concentrations in several rabbit studies; however, coco-betaine at 16% and 30% and lauryl betaine at 10% were ocular irritants. In human mucosal studies testing the efficacy of toothpaste, betaine at 4% did not produce adverse effects.
Betaine (up to 50%), coco-betaine (up to 5%), lauryl betaine (0.1%), and C12 to C14 alkyldimethyl betaines (up to 100%) were not sensitizing in nonhuman and human dermal studies. Allergic reactions to coco-betaine have been reported in case reports.
Discussion
The Panel considered the available data on alkyl betaines and noted the low systemic toxicity at high doses in single dose and repeated dose oral animal studies, no reproductive/developmental toxic effects in animal studies, no genotoxicity in in vitro and in vivo studies, and no sensitization in multiple tests. A dermal uptake study of cetyl betaine and lauryl betaine that included tape stripping indicated that these ingredients were mostly found in the outer layers of the stratum corneum. The Panel noted that most surfactants exhibit some irritancy, as was noted in dermal and ocular studies of coco-betaine, lauryl betaine, and C12 to C14 alkyldimethyl betaines. Thus, the Panel stated that products that include these ingredients should be formulated to be nonirritating.
Although there are data gaps, the shared core chemical structure, similar functions and concentrations in cosmetics, and the expected similarities in physicochemical properties enabled grouping these ingredients and reading across the available toxicological data to support the safety assessment of each individual compound in the entire group.
The Panel noted that there were no data available on the UV absorption or phototoxicity of alkyl betaines; however, because none of the structures that comprise these ingredients are chromophores, the Panel felt that there was no concern that these ingredients would cause adverse effects from UV exposure.
The Panel expressed concern about animal-derived ingredients, namely, the transmission of infectious agents. They stressed that these ingredients must be free of detectible pathogenic viruses or infectious agents (eg, bovine spongiform encephalopathy prions). These ingredients should be produced according to current good manufacturing procedures (cGMPs) and should conform to regulations for producing substances from animal-derived materials.
The Panel also expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. The Panel stated that the cosmetics industry should continue to use cGMPs to limit impurities.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, body and hand products, noncoloring hair powders, and indoor tanning preparations. There were no inhalation toxicity data available. Betaine is reportedly used at concentrations up to 3% in cosmetic products that may be aerosolized and up to 0.0001% in cosmetic products that may become airborne. Although the Panel noted that droplets/particles from spray and loose powder cosmetic products would not be respirable to any appreciable amount, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Panel concluded that the following 11 alkyl betaines are safe in cosmetics in the present practices of use and concentration described in the safety assessment, when formulated to be nonirritating. Behenyl betaine Betaine Cetyl betaine Coco-betaine Decyl betaine* Hydrogenated tallow betaine* Lauryl betaine Myristyl betaine Oleyl betaine Stearyl betaine Tallow betaine*
*Indicates not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director Bart Heldreth, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
C. Burnett contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, P. Snyder, and F. A. Andersen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The infrastructure of Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.