Abstract
Glomerulopathy and body weight gain were noted after chronic oral administration of a novel nonsteroidal dissociated agonist of the glucocorticoid receptor compound, fosdagrocorat, to beagle dogs fed an ad libitum diet. To further investigate the role of diet and treatment with either fosdagrocorat or the glucocorticoid comparator, prednisone, on renal safety, a 13-week investigative study was conducted in beagle dogs. Renal histopathology, clinical chemistry, urinalysis, glomerular filtration rate (GFR), body weight, heart rate, blood pressure (BP), and hematology were investigated in restricted- and ad libitum-fed dogs administered prednisone (2.2 mg/kg/d), fosdagrocorat (5 mg/kg/d), or vehicle for 13 weeks. Glomerulopathy was primarily observed in fosdagrocorat- and prednisone-treated ad libitum but not in feed-restricted or ad libitum vehicle-treated dogs. Kidneys in dogs from the prednisone-treated ad libitum had the greatest incidence and severity of tubular degenerative changes. Increased urine volume and decreased urine-specific gravity were present in prednisone- and fosdagrocorat-treated dogs, regardless of diet. These changes were not associated with consistent changes in GFR. Fosdagrocorat or prednisone treatment ad libitum dogs had the greatest increase in body weight gain. Sporadic changes in systolic and diastolic BP were noted in fosdagrocorat- and prednisone-treated groups. Significant reductions in serum cortisol and absolute eosinophils were noted in both ad libitum- and restriction-fed prednisone- and fosdagrocorat-treated dogs. In conclusion, prednisone-treated dogs fed ad libitum had greater glucocorticoid-induced renal effects than those dosed with fosdagrocorat.
Introduction
Glucocorticoids, steroid hormones produced by the adrenal cortex, enter the cell and bind to glucocorticoid receptors (GRs). Upon binding to GR, a glucocorticoid recruits cofactor proteins that either activate or repress transcription of target genes. 1 Transactivation stimulates the expression of some genes via direct DNA binding, while transrepression inhibits the expression of some genes via indirect DNA binding. 1 Many of the adverse metabolic effects on bone and glucose metabolism mediated by glucocorticoids occur via transactivation, whereas many of the desired anti-inflammatory effects occur via transrepression, although there is not a complete segregation along these lines. 1 Thus, the development of selective agonist of GR which triggers transrepression, but does not initiate transactivation, is hypothesized to lead to an anti-inflammatory agent with a reduced side effect profile when compared to conventional glucocorticoid therapies.
Fosdagrocorat, PF-04171327 (CAS No. 1044535-58-1), a phosphate ester prodrug of PF-00251802, is a novel nonsteroidal dissociated agonist of the glucocorticoid receptor (DAGR), developed for the treatment of rheumatoid arthritis. In human cells in vitro, PF 00251802 is a partial agonist (70%-80% efficacy) with effects mediated via transrepression, but is a full antagonist compared to dexamethasone for transactivation. 2 In preclinical in vitro and in vivo assays using prednisolone as a positive control, PF 00251802 demonstrated robust anti-inflammatory activity with reduced effects on markers of bone and glucose metabolism. 2 Fosdagrocorat demonstrated clinical efficacy in patients with rheumatoid arthritis, and dose–response modeling of disease activity scores concluded that fosdagrocorat doses higher than 9 mg daily have efficacy comparable to or greater than that of 10 mg prednisone once daily. 3,4
Glomerulopathy was observed microscopically in dogs after chronic oral administration in a 9-month toxicity study of fosdagrocorat to beagle dogs fed an ad libitum diet (Pfizer, unpublished data). Glomerulopathy was not noted in a previous 13-week toxicity study in dogs on a restricted diet. Proteinuria and/or glomerulopathy have been observed in dogs and other species as a result of exposure to glucocorticoids. 5 –11 Proposed factors that might contribute to glucocorticoid-induced glomerulopathy and proteinuria include direct alterations in glomerular permeability and effects secondary to increased blood pressure (BP) and glomerular filtration rate (GFR), increased blood glucose and insulin resistance, or obesity. 12 The interrelationships between renal disease, diabetes, hypertension, and obesity have been extensively described. 13,14 However, the correlation between obesity and renal hyperfiltration (increased GFR) in the nondiabetic population is poorly understood. There is an association between obesity and renal hyperfiltration in the general population. 15 Nondiabetic obese people are thought to adapt to increased body mass, increased energy intake, and increased tissue turnover. For example, a 51% increase in GFR and a 31% increase in renal plasma flow have been described in severely obese people and considered to be a result of increased arterial BP and renal vasodilation (dilated afferent arteriole) which drive transcapillary hydraulic pressure. 16 However, elevated BP is not always associated with increased GFR decline in the general middle-aged population and additional genetic, environmental, or physiological factors might be necessary cofactors for the development of chronic kidney disease in some individuals. 17 The role of obesity in the development of renal disease was investigated by feeding a high-fat diet to dogs for up to 24 weeks. The increased body weight gain in these dogs resulted in increases in arterial BP, heart rate, circulating insulin concentrations, and GFR, as well as microscopic glomerular changes of increases in Bowman space, mesangial matrix, number of proliferating cells, and thickness of glomerular basement membranes. 18
The present investigative 13-week study was designed to further investigate and appropriately contextualize the renal findings observed in the 9-month toxicity study of fosdagrocorat using a direct comparison with prednisone, an orally administered glucocorticoid prodrug of prednisolone used chronically for rheumatic diseases. To investigate the potential contribution of obesity to glomerular changes, the study included both food-restricted and ad libitum feeding groups. Renal changes and potential contributing factors were investigated by measurements of body weight, body condition (obesity) scoring, urinalysis parameters and urine biomarkers, arterial BP, GFR, glucose tolerance, kidney organ weight, and light microscopic examination of the kidney. Additional end points to further investigate the extent of the expected glucocorticoid pharmacodynamic activity included toxicokinetics, clinical chemistry and hematology parameters, organ weights, and microscopic examination of the liver, adrenal gland, thymus, skeletal muscle, gut-associated lymphoid tissue, and lymph nodes.
Materials and Methods
Dose Selection
The dose of 5 mg/kg fosdagrocorat once daily was chosen based on previous repeat-dose toxicity studies in beagle dogs. In a previous 9-month toxicity study, increased body weight gain, decreased urine-specific gravity, and histopathologic evidence of glomerulopathy were observed at 5 mg/kg/d. The dose of 5 mg/kg/d in dogs was anticipated to result in systemic exposure to PF 00251802 in the clinically relevant range corresponding to administration of fosdagrocorat to humans. 19 Only females were used in the present 13-week investigative study because they exhibited greater body weight gain changes, and to simplify the study design, as in the previous 9-month toxicity study, fosdagrocorat-related effects and exposures were generally similar in males and females. The dose of 1.1 mg/kg prednisone twice daily (2.2 mg/kg/d) was selected based on a preliminary 2-week dose range finding toxicity study in female dogs, comparing 5 mg/kg fosdagrocorat once daily versus 0.55 or 1.1 mg/kg prednisone twice daily. Effects on glucocorticoid-sensitive end points were similar among all groups administered prednisone and included transient body weight loss, decreased serum cortisol, increased serum alkaline phosphatase, decreased eosinophil counts, increased urinary protein/creatinine ratio, and microalbuminuria. Because systemic prednisolone exposure decreased from day 1 to day 14 in both of the prednisone-treated groups, the higher dose of 1.1 mg/kg twice daily was chosen for the present investigation.
Experimental Design
The 13-week investigative, non-Good Laboratory Practice (GLP) study was conducted at a Pfizer Worldwide R&D facility, which is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. In addition, the study was conducted under Institutional Animal Care and Use Committee approved protocols, in accordance with the National Research Council Guide for the Care and Use of Laboratory Animals and in compliance with the Animal Welfare Act and its implementing regulations. Thirty female beagle dogs, greater than 8 months, and experimentally naive, were used in the study and divided into 6 experimental groups (5 females/dose), as summarized in Table 1. Clinically acceptable dogs were manually allocated to study groups based on body weight and prestudy data analysis. The weight range was from 5.6 to 7.9 kg at dose initiation. Fosdagrocorat was administered by oral gavage to 2 experimental groups at 5 mg/kg once daily for 13 weeks (88 consecutive days). For direct comparison with fosdagrocorat, prednisone was also administered by oral gavage to 2 groups at 1.1 mg/kg twice daily (approximately 6 hours between doses) for 13 weeks. Two separate groups of dogs received vehicle (0.5% methylcellulose [wt/vol] and 0.1% polysorbate 80 [vol/vol] buffered with 50 mM phosphate [pH 6.5]) twice daily for 13 weeks and served as control groups. Animals were fed a standard dry food diet (Certified Harlan Teklad 2027) once daily, and for each dosing regimen (fosdagrocorat, prednisone, or vehicle), 1 group was fed a restricted quantity (200-250 g modified when necessary due to body weight loss) and the other group fed ad libitum (350 g).
Experimental Design and Treatment Groups.
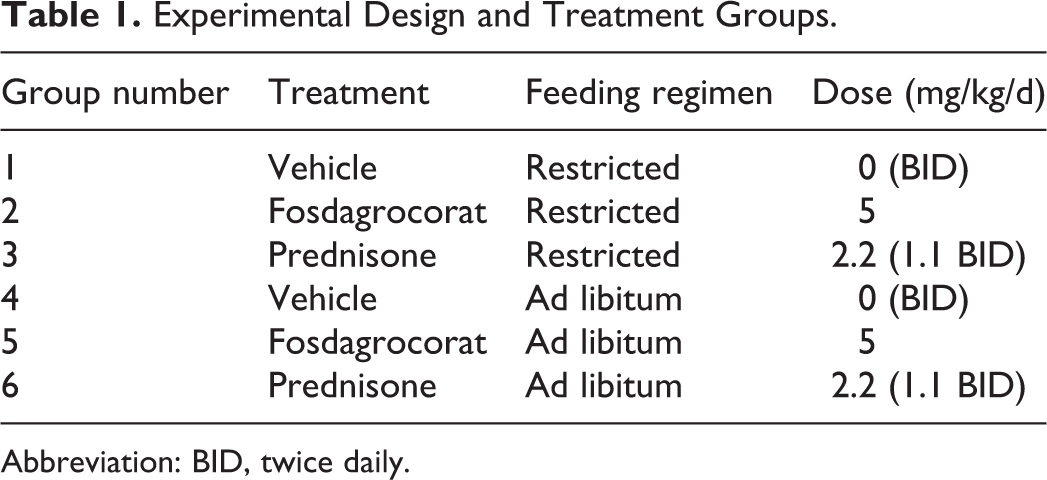
Abbreviation: BID, twice daily.
Investigative Parameters
Quantitative assessment of food consumption (daily) and body weight (pretreatment and twice weekly thereafter) was evaluated. Body condition scoring was assessed by a clinical veterinarian once pretreatment and approximately monthly beginning week 1 and recorded according to a standardized rating scale. 20
For the cardiovascular measurements (BP and heart rate), all animals were instrumented with telemetry transmitters (Data Sciences International (DSI) TA11PA-C40 telemetry implant, St. Paul, MN) via the femoral artery with a period of at least 2 weeks of postoperative recovery prior to the experimental start date. Heart rate and BP data were collected over a 20-hour interval twice pretreatment and weekly beginning day 4. During the pretreatment data collection period, animals were sham dosed, and room entries were limited to those anticipated during the dosing period (eg, food presentation and at the time of dose administration). Heart rate and BP data were collected and transmitted by a Bluetooth device (DSI model JET3ETA-BP) to receivers (DSI model JET Bluetooth Receiver). Signals were recorded using a PC-based data acquisition system (Ponemah, P3 v5.0; DSI). Ponemah review was used for postacquisition analysis of heart rate, BP, and activity measurements. Data were transferred to a Microsoft Excel analysis workspace from which appropriate tabular and graphic data summaries were generated.
Clinical pathology (hematology, clinical chemistry, qualitative urinalysis, and urine chemistry) and renal biomarker parameters were monitored. Hematology and clinical chemistry parameters were evaluated in samples collected from animals fasted overnight once during the pretreatment period and during weeks 4, 8, and 13. Blood samples for final clinical laboratory measurements were collected before blood samples for final toxicokinetic analysis. Qualitative urinalysis and urine chemistry (microalbumin, creatinine, cystatin C, neutrophil gelatinase-associated lipocalin [NGAL], and total protein) were measured on samples collected overnight, twice during the pretreatment period and during weeks 4, 8, and 13. The NGAL assay was validated (non-GLP) using electrochemiluminescence methodology.
The GFR was determined as a function of plasma iohexol clearance for all animals fasted overnight twice pretreatment and prior to the first daily dose administration during weeks 4, 8, and 13. A 300 mg/kg bolus of iohexol (Omnipaque iohexol injection) was infused into the cephalic or saphenous vein at a dose volume of 1 mL/kg over 30 to 60 seconds. Plasma clearance was calculated for all animals by the equation CLplasma =
Intravenous glucose tolerance tests (IVGTTs) were performed on all animals fasted overnight once pretreatment and prior to the first daily dose administration during week 12. A 50% glucose solution was infused into the cephalic or saphenous vein at a dose of 1 mL/kg over a 2-minute period of time. Glucose and insulin concentrations were determined from blood samples collected at once pretreatment and week 12 at 3, 5, 15, 30, 40, and 60 minutes following administration of the glucose solution. The half-life (
For the determination of systemic exposure, blood samples were collected from all animals for measurement of the plasma concentrations for the primary active metabolite of fosdagrocorat (PF-00251802) and prednisone (prednisolone) on day 1 and week 13 (day 85) and subsequent determination of toxicokinetic parameters (
At the end of the treatment period, animals were euthanized and necropsies were conducted. Tissues from kidney, liver, adrenal gland, thymus, skeletal muscles (gastrocnemius and biceps femoris), gut-associated lymphoid tissue (GALT), and popliteal and mesenteric lymph nodes were collected, fixed in 10% formalin, processed for histopathologic examination, and organ weights were collected for the kidney, liver, adrenal gland, and thymus.
Statistical Analysis
Statistical analyses for body weight, food consumption, clinical laboratory measurements, organ weight, and cardiovascular measurements were conducted in SAS version 9.2. Statistical tests were conducted at the 5% and 1% significance levels.
Results
Systemic Exposure
Systemic exposure to the pharmacologically active metabolites, prednisolone and PF 00251802, is summarized in Table 2. Plasma concentrations (
Systemic Exposure.a
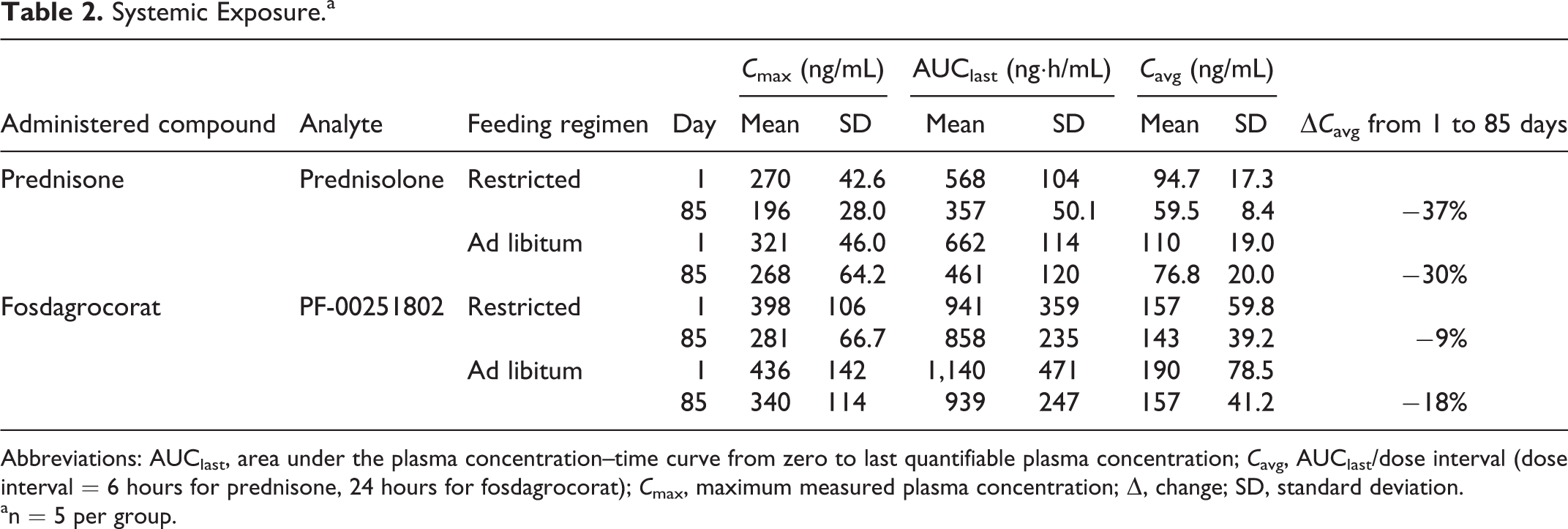
Abbreviations: AUClast, area under the plasma concentration–time curve from zero to last quantifiable plasma concentration;
an = 5 per group.
Effects on Food Consumption, Body Weight, and Body Condition Scores
Food consumption, body weight, and body condition scores are summarized in Table 3. On the restricted feeding regimen, food consumption did not differ among groups; body weights were lower in the prednisone group than in the vehicle control or fosdagrocorat groups. On the ad libitum feeding regimen, food consumption and body weights were greater in both the fosdagrocorat and prednisone groups relative to the vehicle control group. On the restricted and ad libitum feeding regimen, body condition score increased relative to the control groups by 27.29% to 17.24% in the fosdagrocorat- and prednisone-treated groups, respectively. The increase in body condition score is consistent with the increases in body weight; however, it is likely due, in part, to increases in mid-section girth secondary to hepatomegaly as evidenced by significantly increased liver weights compared to controls.
Treatment-Related Effects on Food Consumption, Body Weight, and Body Condition Scores.a
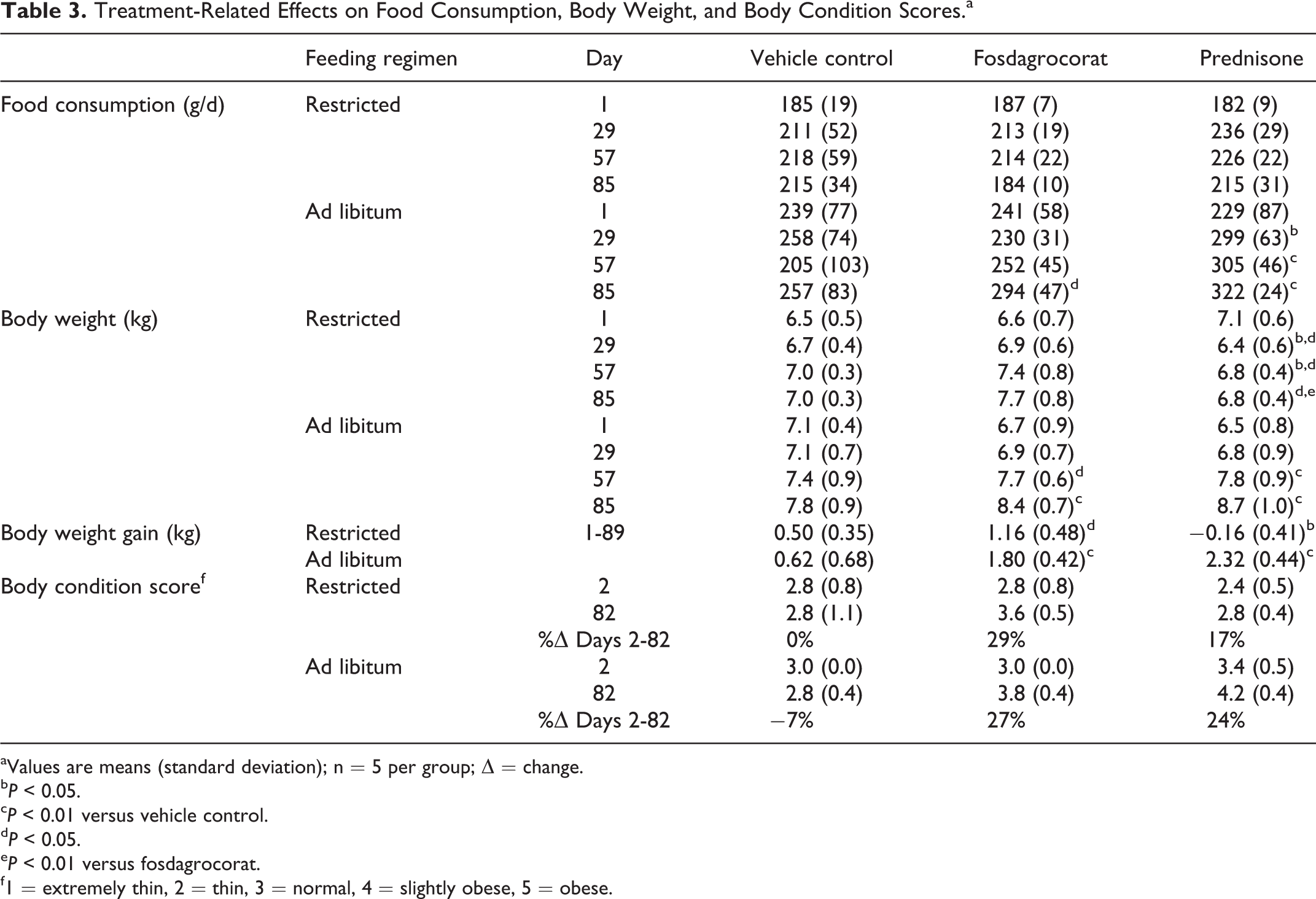
aValues are means (standard deviation); n = 5 per group; Δ = change.
b
c
d
e
f1 = extremely thin, 2 = thin, 3 = normal, 4 = slightly obese, 5 = obese.
Effects on Cardiovascular Measurements of BP and Heart Rate
Cardiovascular end points are presented in Figure 1 as difference versus vehicle control. Systolic BP was increased by fosdagrocorat and prednisone in the ad libitum feeding groups. Statistical significance was achieved for changes in systolic BP for fosdagrocorat on day 67 and for prednisone on days 25, 46, and 67 during 1.75 to 4.25 hours postdose. Statistically significant increases in diastolic BP were observed for fosdagrocorat with the restricted feeding regimen on days 25, 46, 67, and 88 and for prednisone with the ad libitum regimen on days 46 and 67. Increased diastolic BP on the ad libitum regimen reached statistical significance on day 88 (8.25-19.5 hours postdose) for fosdagrocorat or days 46 and 67 (1.75-4.25 hours postdose) for prednisone. There was no change in either systolic or diastolic pressure observed for prednisone with the restricted feeding regimen. There was an initial decrease in heart rate for both prednisone groups (day 4) followed by a gradual increase in heart rate on days 46, 67, and 88 for the prednisone ad libitum regimen.
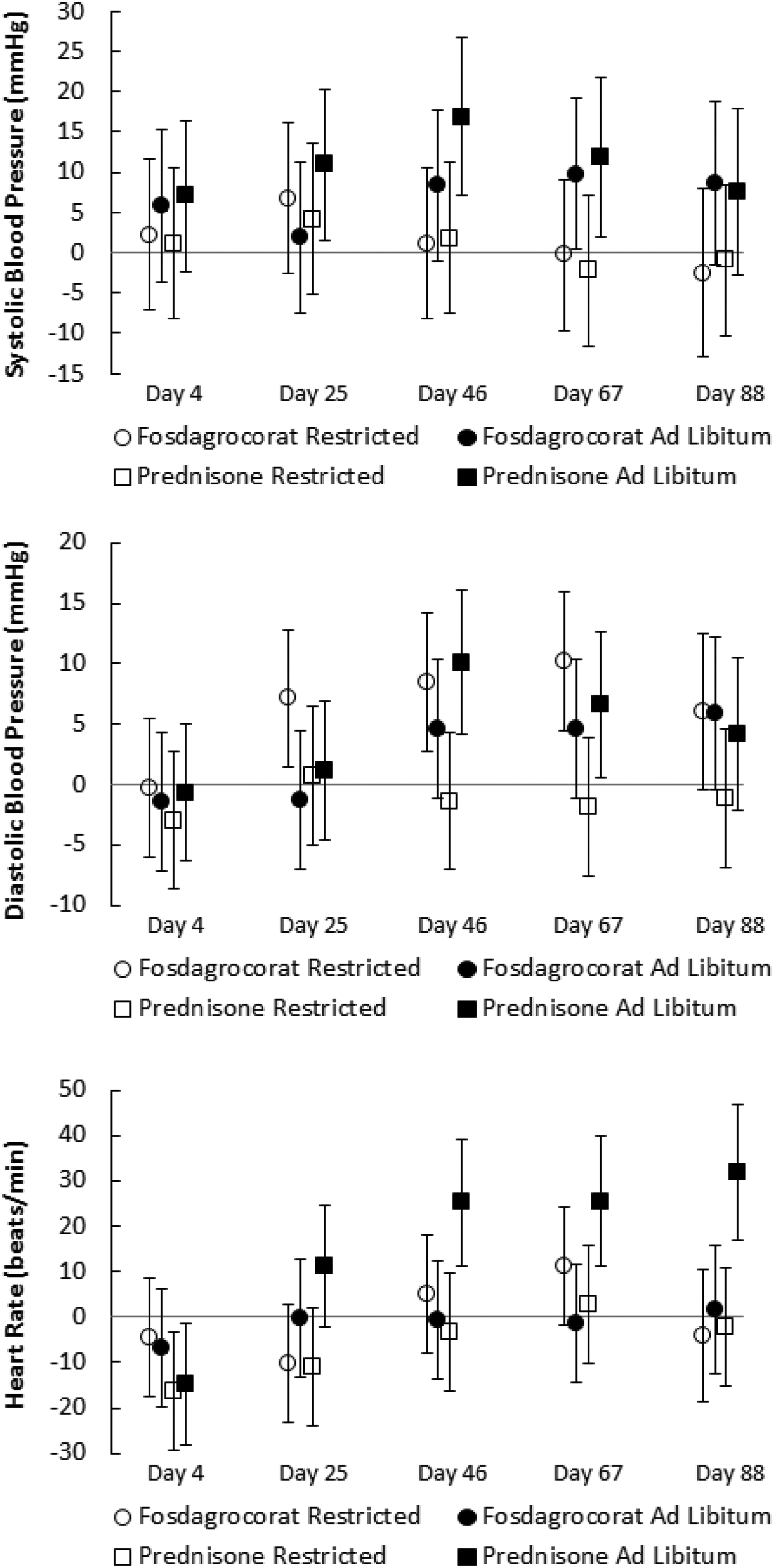
Treatment-related changes in blood pressure and heart rate. Values are expressed as mean difference versus vehicle control ± 95% confidence limits (CLs) during the interval from 1.75 to 4.25 hours postdose for 6 animals per group. Values for which the 95% CL do not cross the horizontal vehicle control zero reference line differ significantly from their respective vehicle control group (
Effects on Clinical Pathology Parameters
Effects on mean clinical pathology (hematology and clinical chemistry) parameters measured at week 13 are summarized in Table 4. In both restricted and ad libitum feeding regimens, fosdagrocorat decreased lymphocytes without a noteworthy effect on neutrophils. Prednisone decreased lymphocytes and increased neutrophils with the ad libitum feeding regimen but did not exhibit statistically significant effects with the restricted feeding regimen. Cortisol concentrations were suppressed below the level of quantitation and eosinophil counts were markedly decreased by fosdagrocorat and prednisone with both feeding regimens. There were no statistically significant differences among groups in serum glucose. Statistically significant decreases in BUN and creatinine were elicited by fosdagrocorat and prednisone with both feeding regimens. Statistically significant increases in alanine aminotransferase (ALT) and gamma-glutamyltransferase (GGT) parameters were observed only with prednisone in the ad libitum feeding regimen. This was associated with higher severity grades of hepatocyte vacuolation as noted histopathologically for prednisone in the ad libitum feeding regimen.
Treatment-Related Effects on Clinical Pathology Parameters in Week 13.a
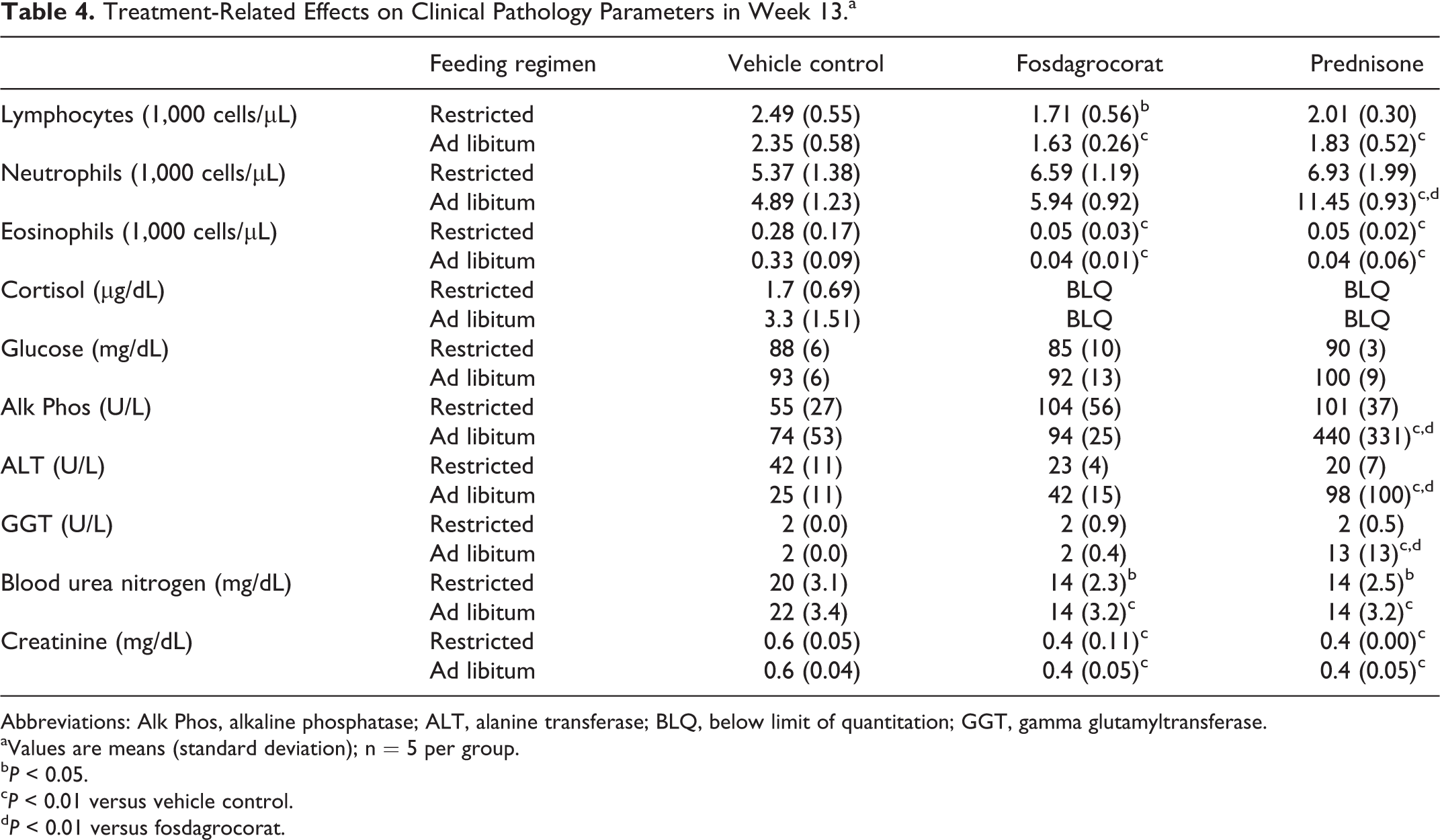
Abbreviations: Alk Phos, alkaline phosphatase; ALT, alanine transferase; BLQ, below limit of quantitation; GGT, gamma glutamyltransferase.
aValues are means (standard deviation); n = 5 per group.
b
c
d
Effects on Urinalysis Parameters, Renal Biomarkers, and GFR
Mean urinalysis parameters, renal biomarkers, and GFR are summarized in Table 5. Independent of feeding regimen, fosdagrocorat and prednisone increased urine volume with decreased specific gravity. The effect on urine volume was more prominent and consistent for prednisone than fosdagrocorat. Urine protein concentration was increased by prednisone. The urine protein effect was transient with the restricted feeding regimen but sustained with the ad libitum feeding regimen. Likewise, microalbumin was significantly increased by prednisone with the ad libitum feeding regimen. No statistically significant increases in urine protein or microalbumin were observed in fosdagrocorat groups. Urine cystatin C was increased by prednisone to a similar extent with restricted and ad libitum feeding regimens, although statistical significance was more consistently observed with the restricted regimen. Cystatin C was significantly increased by fosdagrocorat only in week 4 with the restricted feeding regimen. Statistically significant increases in NGAL were observed only for prednisone with the ad libitum feeding regimen. The GFR tended to be increased relative to baseline in fosdagrocorat groups during weeks 4 and 8 and in control and prednisone groups during week 4 independent of feeding regimen. However, the changes in GFR were variable and statistically significant only sporadically for fosdagrocorat.
Treatment-Related Effects on Urinalysis Parameters and Glomerular Filtration Rate.a
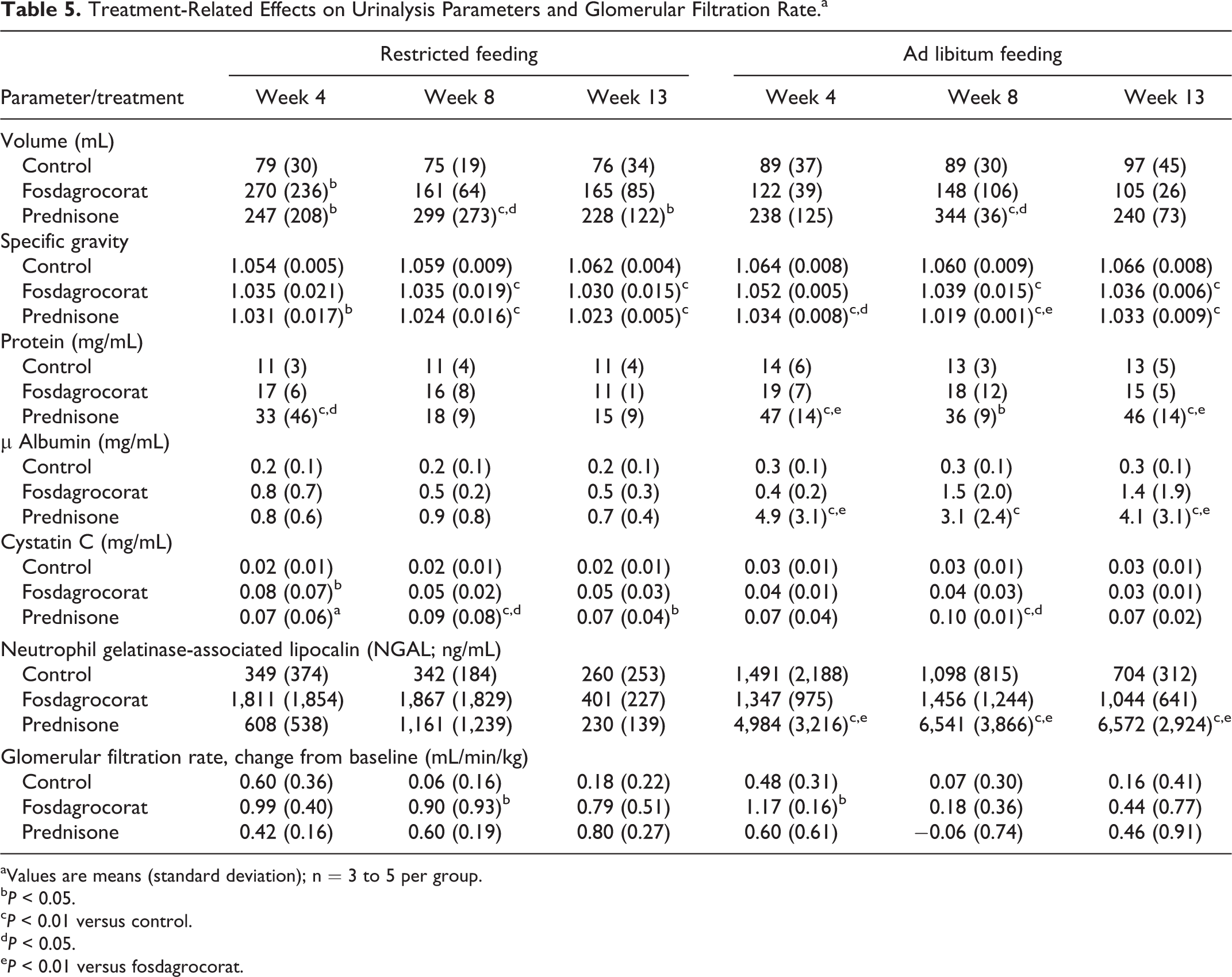
aValues are means (standard deviation); n = 3 to 5 per group.
b
c
d
e
Effects on IVGTT
The IVGTTs were conducted at baseline and during week 12; statistical analysis was performed on changes from baseline. Data are summarized in Table 6. At baseline, glucose half-life (
Treatment-Related Effects on Intravenous Glucose Tolerance Test.a
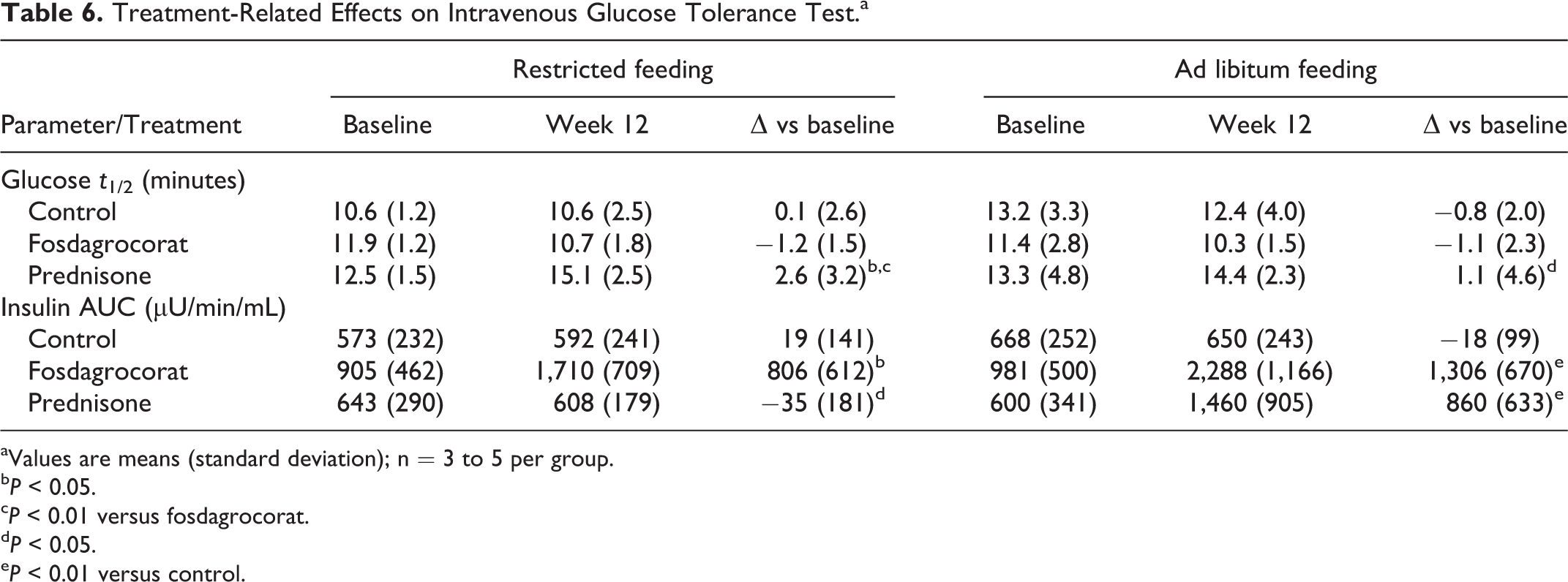
aValues are means (standard deviation); n = 3 to 5 per group.
b
c
d
e
Organ Weights and Histopathology Findings
Organ weights are summarized in Table 7. There were similar decreases in adrenal weights for fosdagrocorat and prednisone with both restricted and ad libitum feeding regimens. Kidney weights were increased in prednisone-treated groups with both restricted and ad libitum feeding regimens; the increase was greater with the ad libitum feeding regimen. Liver weights were increased in fosdagrocorat and prednisone groups on both feeding regimens; the increase was greatest for prednisone on the ad libitum feeding regimen. Thymus weights were numerically lower in the fosdagrocorat and prednisone groups, but the differences were not statistically significant.
Treatment-Related Effects on Organ Weights (g).a
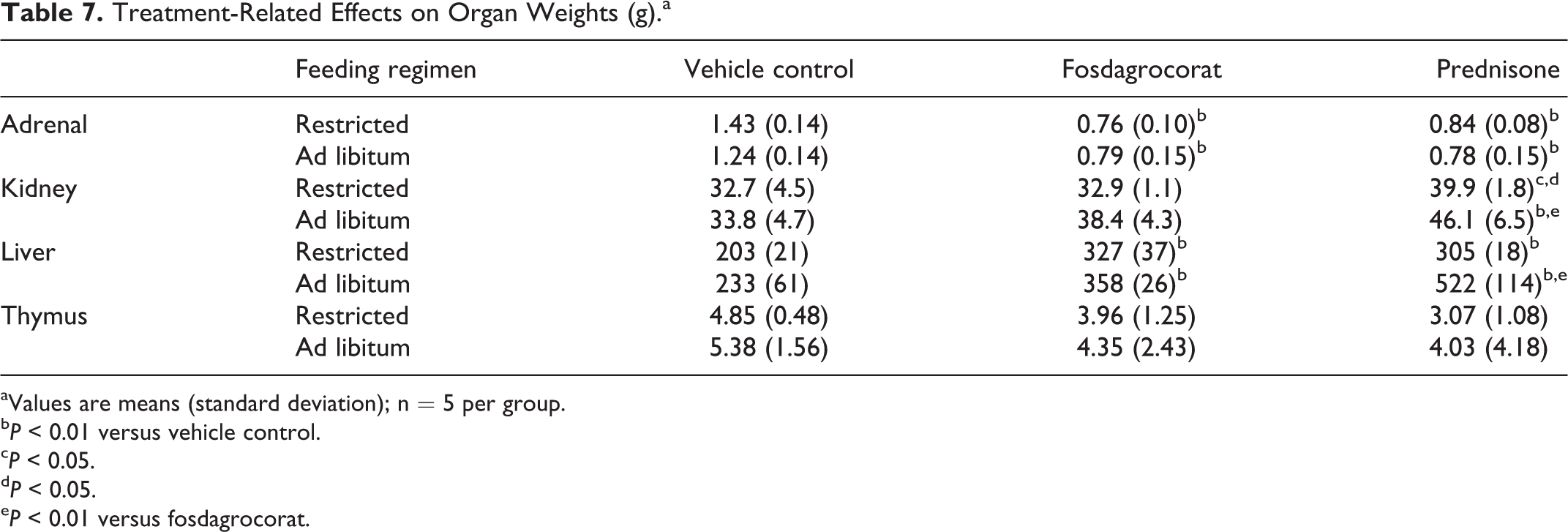
aValues are means (standard deviation); n = 5 per group.
b
c
d
e
Histopathology findings are summarized in Table 8. All animals in the fosdagrocorat and prednisone groups had some degree of atrophy of the adrenal cortex ranging from grade 2 (mild) to grade 4 (marked); there was no clear difference between feeding regimens. All vehicle control animals had minimal (grade 1) decreased lymphoid cellularity of the thymus, which may represent normal thymus involution. All but 1 animal (restricted regimen) treated with fosdagrocorat or prednisone had severe (grade 5) decreased lymphoid cellularity of the thymus. Similar degrees of adrenal cortical atrophy and decreased lymphoid cellularity in thymus indicate that fosdagrocorat and prednisone agonist activity on these expected outcomes was similar. Minimal decreased lymphoid cellularity (grade 1) was observed sporadically with fosdagrocorat and/or prednisone in lymph nodes and GALT.
Treatment-Related Histopathology Findings.a
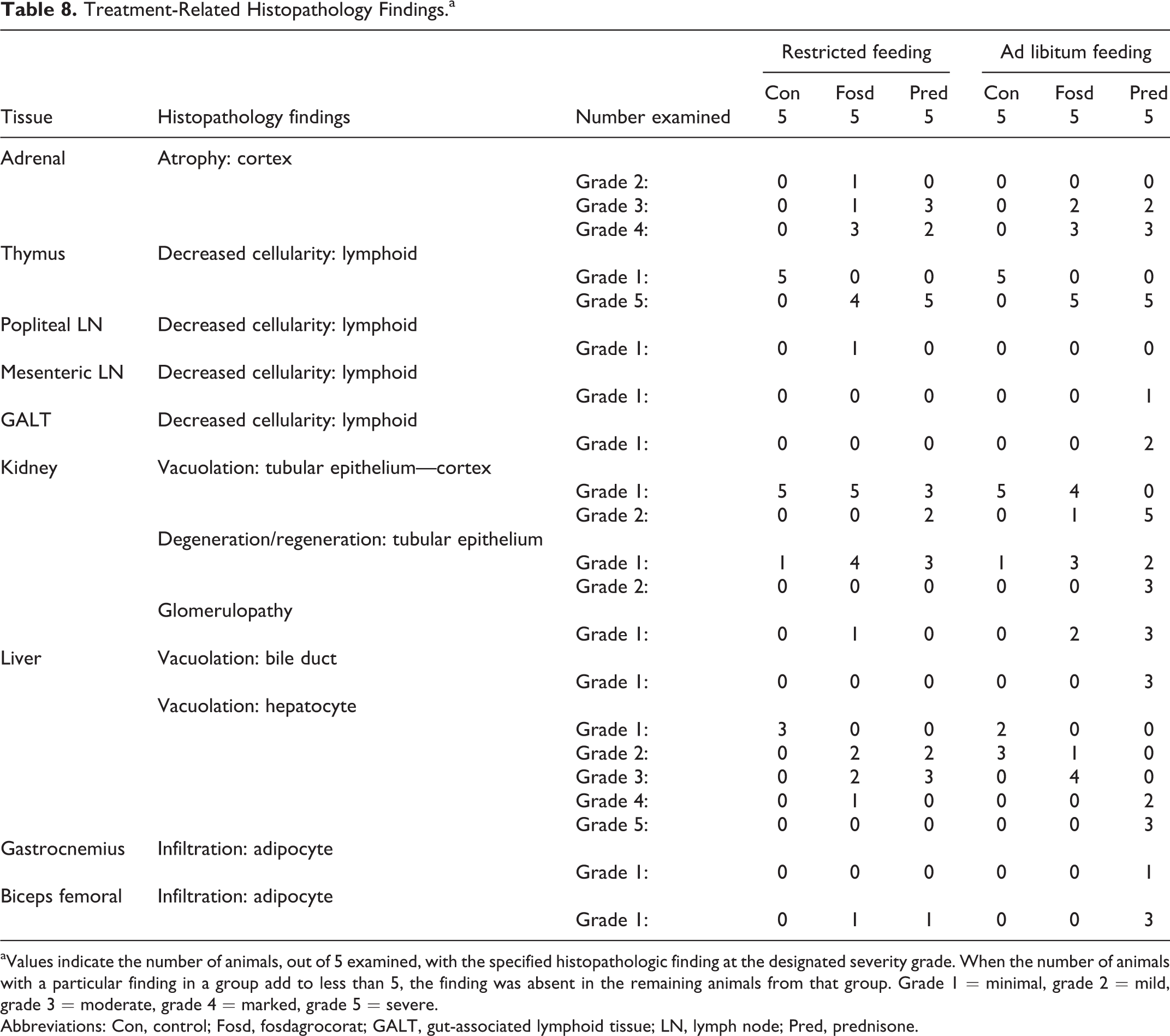
aValues indicate the number of animals, out of 5 examined, with the specified histopathologic finding at the designated severity grade. When the number of animals with a particular finding in a group add to less than 5, the finding was absent in the remaining animals from that group. Grade 1 = minimal, grade 2 = mild, grade 3 = moderate, grade 4 = marked, grade 5 = severe.
Abbreviations: Con, control; Fosd, fosdagrocorat; GALT, gut-associated lymphoid tissue; LN, lymph node; Pred, prednisone.
Vacuolation and degeneration/regeneration of the renal tubular epithelium were observed in all groups including vehicle controls. Tubular degeneration/regeneration was characterized by tubular basophilia, epithelial cell degeneration/death, or presence of red blood cells within the lumen. The severity of vacuolation was increased from grade 1 (minimal) in control groups to grade 2 (mild) in 2 of 5 dogs treated with prednisone on the restricted feeding regimen, 5 of 5 dogs treated with prednisone on the ad libitum regimen, and 1 of 5 dogs treated with fosdagrocorat on the ad libitum regimen. Peroidic Acid Schiff (PAS) stains were performed on kidney sections from all animals in the study. Tubular hyaline cast was the only specific kidney finding which corresponded with this special stain.
Glomerulopathy was observed at minimal severity (grade 1) in 3 of 5 dogs in the prednisone group and in 2 of 5 dogs in the fosdagrocorat group with the ad libitum feeding regimen and in 1 fosdagrocorat-treated dog in the restricted feeding regimen. Glomerulopathy was characterized by increased mesangial matrix and cellularity and/or displacement of a portion of the glomerular tuft by homogenous eosinophilic material.
Vacuolation of the bile duct was observed only in 3 of 5 dogs from the prednisone group with ad libitum feeding. Minimal to mild (grades 1 and 2) hepatocyte vacuolation was observed in vehicle control dogs in both feeding regimens. The incidence and severity of diffuse hepatocyte vacuolation were increased in fosdagrocorat and prednisone (restricted and ad libitum) groups, with the greatest severity (grades 4 and 5, marked and severe) in the prednisone group with ad libitum feeding.
Adipocyte infiltration of skeletal muscle was observed for both fosdagrocorat and prednisone but was most pronounced for the prednisone group with ad libitum feeding. Biceps femoral was affected to a greater degree than gastrocnemius.
Discussion
Glomerulopathy has been observed in dogs with Cushing syndrome, a syndrome caused by excessive production adrenocorticotropic hormone, and in dogs treated with prednisone. 5 In a retrospective clinical study in dogs with glomerulonephritis, there was a high prevalence of glucocorticoid excess, including chronic treatment with glucocorticoids and spontaneous hyperadrenocorticism. 6 Twice-daily administration of hydrocortisone to dogs for 12 weeks resulted in reversible increased arterial BP, increased urine protein to creatinine ratio, and microalbuminuria. 7 Increased urine volume, decreased urine-specific gravity, proteinuria, and glomerulopathy were observed in 10 male beagle dogs after oral administration of prednisone at 2.2 mg/kg, twice daily, for 6 weeks. 8 In another study with a smaller number of dogs (2/sex/dose), urine volume increased and specific gravity decreased after daily oral administration of prednisolone, methylprednisolone, or triamcinolone, for 6 weeks, but no histopathologic evidence of glomerulopathy was observed. 9 Proteinuria and/or glomerulopathy have also been observed after administration of dexamethasone to mice and cortisone to rabbits. 10,11 The increased body condition scores despite feeding regimen may represent increased abdominal fat distribution or increased hepatic mass as evidenced by increased liver weights.
In the study presented here, clinical laboratory parameters were selected to characterize the effects of the experimental treatments on end points known to be affected by glucocorticoid agonists. For example, glucocorticoid agonists typically decrease circulating lymphocyte counts and increase neutrophils. 9,21 In this study, decreased lymphocyte counts were observed with both fosdagrocorat and prednisone; however, increased neutrophils were only observed with prednisone. Because food consumption during the current study was similar among groups on the restricted feeding regimen, the decreased body weight in the prednisone group may reflect the catabolic effect of glucocorticoids. 9,21 Statistically significant decreases in BUN and creatinine were elicited by fosdagrocorat and prednisone with both feeding regimens, likely reflective of the catabolic effects of glucocorticoids. 9,22 Glucocorticoids can increase circulating alkaline phosphatase, and transaminases such as ALT and GGT also likely reflective of the catabolic effects of glucocorticoids. 9,22
In the current study, independent of feeding regimen, fosdagrocorat and prednisone increased urine volume with decreased specific gravity. Increased urine volume, lower urine-specific gravity, and decreased urine creatinine were all attributed to the known diuretic effects of glucocorticoids. 23 Decreased urine creatinine could also represent muscle wasting. 24,25 Increases in urine microalbumin were associated with the histopathologic observations such as glomerulopathy, renal tubular degeneration/regeneration, and/or renal tubular hyaline casts. Overall, the glomerular changes observed in this study were interpreted to be of small magnitude and early stage. For instance, glomerulopathy was characterized by expansion of the glomerular mesangium and/or increased glomerular cellularity and/or displacement of a portion of the glomerular tuft by eosinophilic material interpreted to be fibrin. These histopathologic changes typically precede more advanced glomerular injury, including those phenotypes observed in a previous 9-month study (ie, enlargement of glomeruli, segmental glomerulosclerosis, hypertrophy and/or hyperplasia of the visceral and parietal epithelium of the Bowman capsule, adhesions of the glomerular tuft to Bowman capsule, and thickening of the Bowman capsular basement membrane). Also, only 1 to 2 glomeruli per kidney (unilateral or bilateral) per dog were impacted in this study; the exception was a single dog administered prednisone and fed ad libitum, which presented with 3 affected glomeruli. No consistent or significant changes in GFR were noted for any treatment group in this study. Most of other clinical pathology findings associated with the administration of fosdagrocorat or prednisone were consistent with anticipated pharmacological effects of GR agonism. 26,27
Hematological changes, including increased neutrophils, monocytes, white blood cell, and platelets and decreased lymphocytes and eosinophils, are all well documented to be associated with glucocorticoid administration. 28,29 The decreases in cortisol were attributed to negative feedback on the hypothalamic/pituitary/adrenal axis and correlated with a decrease in adrenal gland weights as well as the histopathologic observation such as adrenal cortex atrophy. Clinical chemistry changes including increases in the liver enzymes ALP, ALT, and GGT were likely the result of enzyme induction by the glucocorticoids 30 and, in the case of ALP, were associated with increased liver weights as well as histopathologic observation such as hepatocellular vacuolation. Increases in bilirubin were present in some dogs indicating mild cholestasis, but there was no histopathologic correlate. Increases in total protein and albumin are not well documented with the administration of corticosteroids, but similar increases were seen in a previous study in dogs administered fosdagrocorat. Decrease in serum creatinine was present in a subset of dogs and may be associated with decreased muscle mass induced by corticosteroids, 24,25 although in this study histopathologic evidence of skeletal muscle atrophy was not present. Adipocyte infiltration in skeletal muscle was observed with both compounds and correlated with liver, bile duct, and renal tubular vacuolation (suggestive of lipid accumulation). In conclusion, there were no major differences in renal parameters between beagle dogs dosed with either fosdagrocorat or prednisone when they were fed a restricted diet. However, obesity resulting from ad libitum feeding exacerbated the severity of the renal findings observed in both the fosdagrocorat- and prednisone-treated dogs, with the prednisone-treated dogs showing greater overall glucocorticoid-induced renal effects than those dosed with fosdagrocorat.
Footnotes
Author Contributions
Z. Radi and M. Vogel contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. T. LaBranche contributed to design, contributed to analysis and interpretation, and drafted the manuscript. J. Dybowski and M. Peraza contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. S. Portugal contributed to analysis and interpretation and critically revised the manuscript. D. Lettiere contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Pfizer Worldwide R&D.
