Abstract
ANX005 is a humanized immunoglobulin G4 recombinant antibody against C1q that inhibits its function as the initiating molecule of the classical complement cascade. The safety and tolerability of ANX005 are currently being evaluated in a phase I trial in healthy volunteers (www.clinicaltrials.gov Identifier: NCT03010046). Inhibition of C1q can be applied therapeutically in a broad spectrum of diseases, including acute antibody-mediated autoimmune disease, such as Guillain-Barré syndrome (GBS), and in chronic diseases of the central nervous system involving complement-mediated neurodegeneration, such as Alzheimer's disease (AD). To support the clinical development of ANX005, several studies were conducted to assess the pharmacology, pharmacokinetics, and potential toxicity of ANX005. ANX-M1, the murine precursor of ANX005, functionally inhibits the classical complement cascade both in vitro and in vivo, to protect against disease pathology in mouse models of GBS and AD. Toxicology studies with ANX005, itself, showed that intravenous administration once weekly for 4 weeks was well tolerated in rats and monkeys, with no treatment-related adverse findings. Serum levels of ANX005 in monkeys correlate with a reduction in free C1q levels both in the serum and in the cerebrospinal fluid. In summary, ANX005 has shown proof of concept in in vitro and in vivo nonclinical pharmacology models, with no toxicity in the 4-week repeat-dose studies in rats and monkeys. The no observed adverse effect level was 200 mg/kg/dose, which is 200-fold higher than the first-in-human starting dose of 1 mg/kg in healthy volunteers.
Introduction
ANX005 is a humanized immunoglobulin G4 (IgG4) recombinant antibody against C1q that blocks the initiation of the classical complement cascade. While the complement cascade is an important part of the innate immune system, inappropriate activation can result in local tissue damage and disease progression.
Guillain-Barré syndrome (GBS) is a rare autoimmune disease in which patients develop antibodies against infectious agents that cross-react with components of the peripheral nerve. 1,2 Pathogenic antibody binding to peripheral nerves leads to engagement of C1q and activation of the classical complement cascade, which can cause severe nerve damage and acute paralytic neuropathy. The annual incidence of the syndrome is approximately 6000 patients in the United States and 100,000 worldwide. The most severe manifestations of GBS include limb paralysis and respiratory failure (25% of patients), with a mortality rate of 3% to 10%, despite the use of best available treatment. 1
Alzheimer's disease (AD) is a neurodegenerative disease characterized by progressive memory loss and dementia, wherein the severity of memory deficits is correlated with the degree of synaptic loss. 3 Complement-mediated neurodegeneration is a process associated with AD and other degenerative central nervous system (CNS) disorders, where C1q interacts directly with synapses in an antibody-independent fashion, leading to inflammation and synaptic removal by microglial cells. 4,5 Alzheimer's disease is the most common cause of dementia in the aging population and is a serious medical, social, and economic problem worldwide. Current therapies provide only modest symptomatic relief, highlighting the need for better, disease-modifying treatments.
Targeting C1q and the classical complement cascade may benefit patients with a wide range of both antibody-dependent and independent disorders. ANX005 binds to C1q to inhibit its interaction with multiple substrates, thereby preventing activation of the classical complement cascade while leaving the lectin and alternative complement pathways intact (Figure 1). Importantly, inhibition of C1q inhibits all downstream components of the classical cascade, thereby preventing (1) immune-cell recruitment by the soluble activation products C3a and C5a (anaphylatoxins), (2) directed immune-cell attack by activation products/opsonins deposited on cell membranes (C1q itself as well as C4b and C3b), and (3) membrane damage caused by activation of the lytic membrane attack complex (C5b-9). ANX-M1, the murine precursor antibody of ANX005, has been shown to inhibit C1q binding in vitro and in vivo, providing benefit in animal models of GBS 6 and AD, 4 as well as in other models of both acute and chronic neurodegeneration (unpublished data).
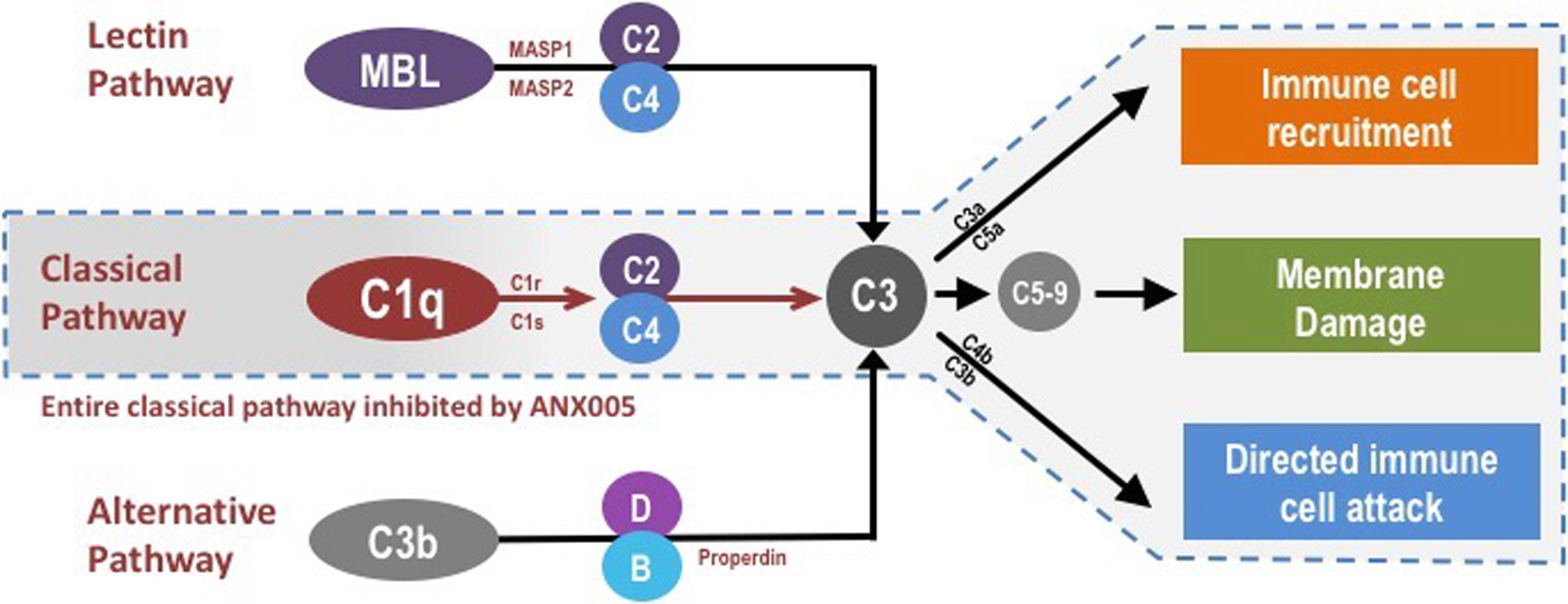
Complement is part of the innate immune system and facilitates the recognition and elimination of pathogens as well as dead and dying cells and other tissue debris. There are 3 main complement pathways—the lectin, classical, and alternative pathways—which are activated by separate initiating molecules but converge on complement component C3. Breakdown products released by activation/cleavage of different complement components lead to (1) immune-cell recruitment (C3a and C5a), (2) directed immune-cell attack (C4b and C3b, which covalently bind to surfaces and function as phagocytic receptors for immune cells), and (3) formation of lytic pores that damage cellular membranes (C5b-9). ANX005 inhibits C1q, the initiating molecule of the classical complement pathway and thereby prevents activation of all complement components downstream of C1q but leaves the full lectin and alternative pathways in place.
To support the clinical development of ANX005, nonclinical studies were conducted to evaluate the pharmacology, pharmacokinetics (PK), and toxicology of the human antibody, ANX005. These studies were conducted in pharmacologically relevant species—Sprague Dawley rats and cynomolgus monkeys. Four-week repeat-dose toxicology studies in rats and monkeys, as well as a tissue cross-reactivity study in a panel of normal human tissues, were completed. This article details the results of these nonclinical studies.
Methods
Test Article
ANX005 is a fully human IgG4 monoclonal antibody produced as separate heavy-chain and light-chain elements in a recombinant Chinese hamster ovary production cell line. ANX005 has a molecular weight of 150 kDa and binds to human C1q with high affinity (described below).
For the pharmacokinetic study conducted in cynomolgus monkeys and the pilot 4-week toxicology studies conducted in Sprague Dawley rats and cynomolgus monkeys, ANX005 was supplied at a nominal concentration of 10.312 mg/mL with pharmaceutically acceptable excipients, pH 6.4, and stored at 4°C. The vehicle control was phosphate-buffered saline (PBS), pH 7.4, which was stored at ambient temperature.
The pivotal 4-week rat and monkey toxicology studies and tissue cross-reactivity study, using a panel of human tissues, were conducted in compliance with US Food and Drug Administration [FDA] Good Laboratory Practice [GLP] Regulations [21 CFR Part 58]). ANX005 used for the GLP studies was supplied at a nominal concentration of 20 mg/mL with pharmaceutically acceptable excipients, pH 6.4, and stored at 2°C to 8°C. The vehicle control was PBS, pH 7.4, which was stored at ambient temperature.
In Vitro Pharmacology
ANX005 binding to serum-derived C1q from multiple species
Serum C1q was captured onto enzyme-linked immunosorbent assay (ELISA) plates using an antibody that binds to an epitope on the collagen tail of C1q that is conserved across all species (JL-1; Abcam ab71940). ANX005 was then titered for binding to the captured C1q.
Activity of ANX005 in an ex vivo hemolysis assay
A standard ex vivo hemolysis assay was used to assess the pharmacological activity of ANX005 for inhibition of C1q activation. Sheep red blood cells (RBCs) were sensitized with surface-reactive antibodies and then coincubated with serum (diluted 100-200×) from different species at 37°C for 1 hour. The serum samples contain C1q and other components of the classical complement cascade, leading to hemolysis of the antibody-sensitized RBCs, as quantified by measuring the release of hemoglobin.
Sequence homology for C1q subunits A, B, and C across species
To aid in the selection of pharmacologically relevant species for toxicity testing, the sequence homology of C1qA, C1qB, and C1qC subunits in human, mouse, rat, rabbit, dog, and cynomolgus monkey were evaluated using sequences from NCBI protein database (https://www.ncbi.nlm.nih.gov/protein/) and compared in NCBI PubMED BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi).
Ex Vivo Study
Human tissue cross-reactivity
ANX005 versus control IgG4 was applied to cryosections from a full panel of normal human tissues, and tissue-bound antibody was identified using a biotinylated anti-human IgG4 secondary reagent. Bound primary/secondary antibody complexes were visualized with a streptavidin-biotin-horseradish peroxidase complex (ABC)/3 3’diamino-benzidine HCl reaction. Staining was assessed on a 5-point intensity grading system (equivocal, minimal, mild, moderate, and marked) as well as staining frequency and pattern. Appropriate positive and negative cell/tissue controls were included in the study.
In Vivo Studies in Rats and Cynomolgus Monkeys
The pharmacokinetic and toxicology studies were conducted in naïve young adult Sprague Dawley rats (8-9 weeks) and cynomolgus monkeys (2-4 years), which were determined to be pharmacologically relevant species (as described in Results).
The pharmacokinetic and toxicology studies were conducted at the Charles River site in Reno, Nevada, in compliance with all applicable sections of the Final Rules of the Animal Welfare Act regulations (Code of Federal Regulations, Title 9), the Public Health Service Policy on Humane Care and Use of Laboratory Animals from the Office of Laboratory Animal Welfare, and the Guide for the Care and Use of Laboratory Animals from the National Research Council.
Rat Studies
Male and female Sprague Dawley rats were group housed in polycarbonate cages containing appropriate bedding and water valves in a controlled environment (18°C-26°C; 30%-70% relative humidity; 12-hour light and dark cycles; 10 or more air changes per hour) and were offered Certified Rodent Chow and tap water ad libitum. Rats were randomly assigned to treatment groups for all studies. The experimental design of each rat study is outlined in Table 1.
ANX005 Nonclinical Studies in the Sprague Dawley Rat.
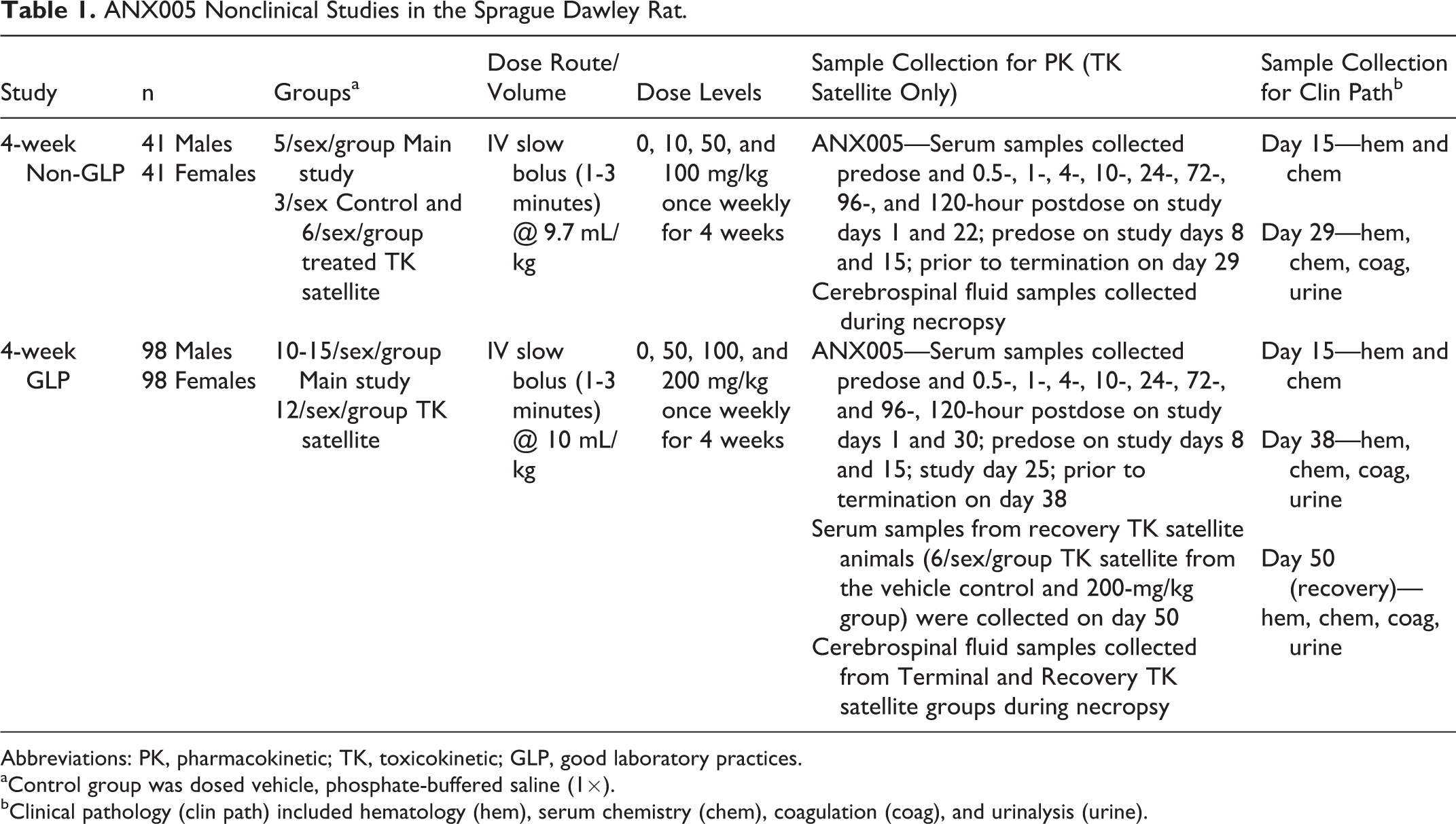
Abbreviations: PK, pharmacokinetic; TK, toxicokinetic; GLP, good laboratory practices.
aControl group was dosed vehicle, phosphate-buffered saline (1×).
bClinical pathology (clin path) included hematology (hem), serum chemistry (chem), coagulation (coag), and urinalysis (urine).
Cynomolgus Monkey Studies
Male and female cynomolgus monkeys were group housed in stainless steel cages in a controlled environment (18°C-29°C; 30%-70% relative humidity; 12-hour light and dark cycles; 10 or more air changes per hour with 100% fresh air) and were offered Certified Primate Chow daily and tap water ad libitum. Cynomolgus monkeys were randomly assigned to treatment groups for all studies. The experimental design of each cynomolgus monkey study is outlined in Table 2.
ANX005 Nonclinical Studies in the Cynomolgus Monkey.
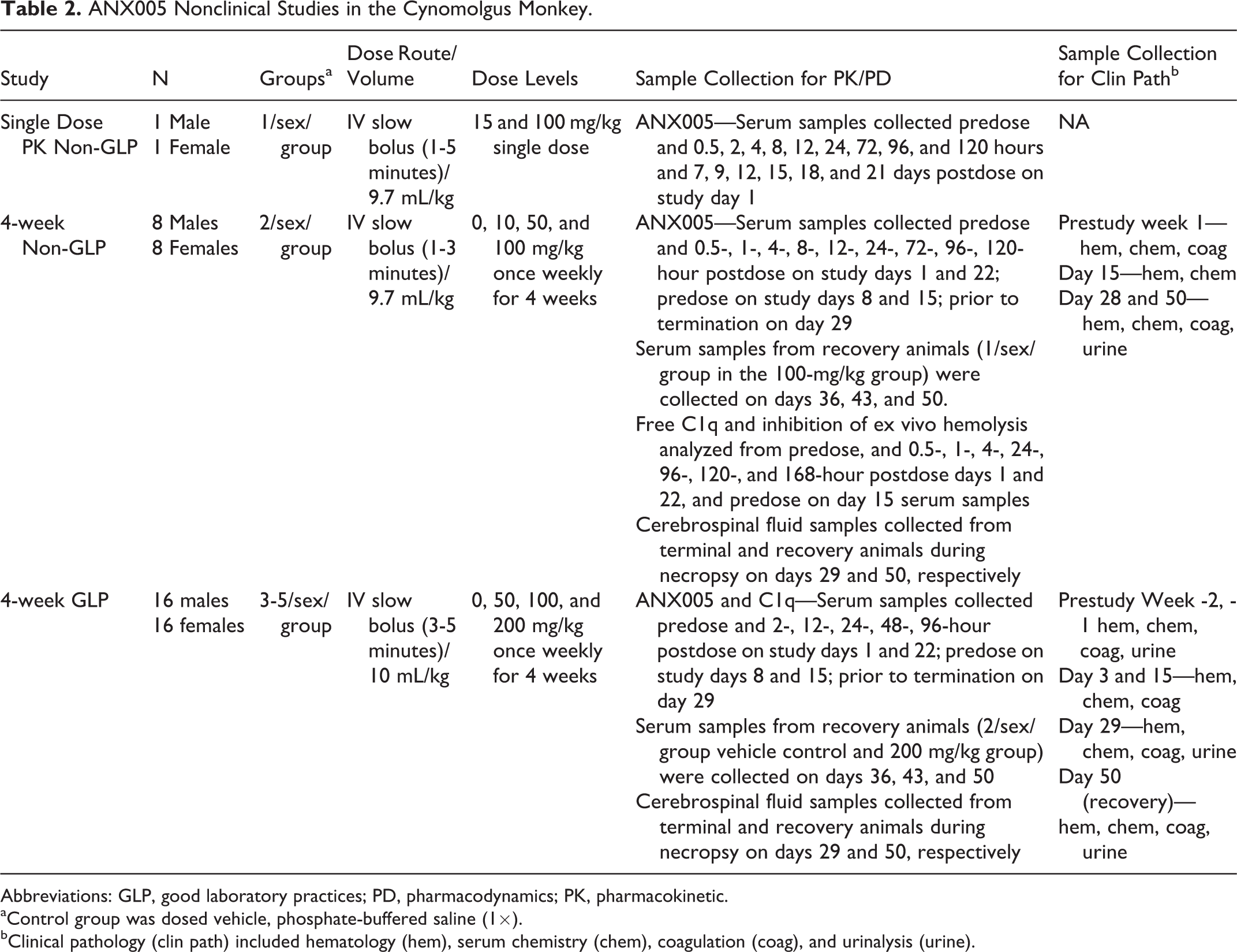
Abbreviations: GLP, good laboratory practices; PD, pharmacodynamics; PK, pharmacokinetic.
aControl group was dosed vehicle, phosphate-buffered saline (1×).
bClinical pathology (clin path) included hematology (hem), serum chemistry (chem), coagulation (coag), and urinalysis (urine).
Measurement of free ANX005 concentrations in monkey and rat serum and cerebral spinal fluid (GLP and non-GLP studies)
Serum and cerebrospinal fluid (CSF) samples were analyzed for ANX005 using a validated ELISA. Plates were coated with purified human complement C1q protein. After capture of ANX005 to the immobilized antibody, peroxidase-conjugated goat antihuman antibody was added for detection of the captured ANX005. Then 3,3’,5,5’-tetramethylbenzidine was added to the plate for an incubation period and the reaction was stopped with 1 M phosphoric acid. The optical density was measured on an Epoch plate reader at 450 nM, with a wavelength correction at 650 nm. The absorbance-versus-concentration relationship was regressed using a 5-parameter logistic Marquardt regression model with a weighting factor of 1/Y2, and concentrations were calculated using a Watson LIMS (v7.3; Thermo, Pennsylvania, USA) data reduction software.
Measurement of free C1q concentrations in monkey serum (non-GLP studies)
Serum levels of C1q-free were measured using a sandwich ELISA. C1q was captured onto ELISA plates using an antibody that binds to the collagen tail of C1q (JL-1; Abcam ab71940) and detected using alkaline-phosphatase conjugated ANX-M1 (murine parent antibody of ANX005, which binds to the same epitope), thereby allowing specific measurement of free C1q (ie, C1q with sites available for drug binding). Plates were read using a luminometer. Standards were fit using a 4-parameter logistic fit and unknowns converted to concentration. Serum C1q-free levels were corrected for dilution and then plotted using GraphPad Prism.
Measurement of free C1q concentrations in monkey serum and cerebral spinal fluid (GLP studies)
Serum and CSF samples were analyzed for C1q using a qualified ELISA assay with anti-C1q JL-1 as the capture antibody. Horseradish peroxidase conjugated ANX-M1 antibody was used for detection. The fluorescent signal, which was proportional to the amount of C1q bound by the capture reagent, was read on a Bio-Tek Synergy 2 plate reader. The luminescence-versus-concentration relationship was regressed using a 5-parameter logistic regression model with a weighting factor of 1/Y2. Data processing was performed using a Watson LIMS (v7.3.0.1; Thermo) data reduction software.
Ex vivo hemolysis measurements in cynomolgus monkeys (GLP and non-GLP studies)
Serum samples from cynomolgus monkeys were used as a source of complement to induce complement-mediated lysis of antibody-sensitized sheep RBCs. Sheep RBCs were presensitized with an anti-RBC antibody (CompTech #B200) and suspended with Gelatin veronal buffered saline containing calcium and magnesium (GVB++; CompTech #B102). Cynomolgus monkey serum samples collected at baseline and following dosing with ANX005 were diluted 50-fold in GVB++. The lysis reaction was triggered by adding an equal volume of RBCs to the serum samples and incubated at 37°C for 1 hour. Supernatants were analyzed by a plate reader for light absorbance at 415 nM. Control samples were run to estimate 100% lysis by adding water to RBCs and background signal in buffer lacking serum to RBCs. Sample signal was background subtracted, normalized to baseline, and then plotted as a percentage of baseline to determine the time course and relative inhibition of hemolysis following dosing with ANX005.
Safety evaluations
General clinical observations of animals were performed twice daily in all the toxicology studies, and cage-side observations were conducted 1 to 2 hours postdose to assess acute toxicity. Other observations performed for all toxicology studies included an assessment of food consumption once daily and measurement of body weight once weekly. The 4-week non-GLP and GLP studies also included clinical pathology (hematology, serum chemistry, coagulation, and urinalysis) and anatomic pathology (gross and microscopic) evaluations (Tables 1 and 2). Cytokine levels were also measured in the non-GLP 4-week monkey study. Other safety evaluations performed for the GLP 4-week studies included ophthalmic examinations; vital-sign measurements; electrocardiograms (ECGs); neurologic exams; and measurements of respiration rate, blood pressure, and heart rate.
Pharmacokinetics
Pharmacokinetic parameters were estimated using a Phoenix pharmacokinetic software (Certara, Princeton, NJ) and a noncompartmental approach consistent with the IV bolus injection route of administration. All parameters were generated from ANX005 individual concentrations in serum (Tables 8, 9). Parameters were estimated using nominal sampling times relative to the start of each dose administration. The concentration at time zero on sampling days was back-extrapolated based on the first 2 observed serum concentrations for parameter estimation. Postdose concentrations reported as below the quantitation limit were treated as absent samples in the toxicokinetics (TKs) analysis.
The area under the concentration-versus-time curve (AUC) was calculated using the linear trapezoidal method with linear interpolation. When practical, the terminal elimination phase of each concentration-versus-time curve was identified using at least the final 3 observed concentration values. The slope of the terminal elimination phase was determined using log linear regression on the unweighted concentration data. Parameters relying on the determination of the terminal elimination phase were not reported if the coefficient of determination was less than 0.800, or if extrapolation of the AUC to infinity represented more than 20% of the total area.
Results
In Vitro Pharmacology
ANX005 binding to serum-derived C1q from multiple species
C1q was captured on ELISA plates from serum of multiple species, and ANX005 was titered to determine the degree of specific binding. ANX005 bound C1q captured from human, cynomolgus, rhesus, dog, and rat serum with comparable affinity, while exhibiting 2- to 3-fold lower affinity for mouse C1q (Table 3). No binding was observed to C1q derived from rabbit serum.
Relative Binding Affinities to Serum-Derived C1q for ANX005.

Activity of ANX005 in an ex vivo hemolysis assay
A standard hemolysis assay was used to assess the pharmacological activity of ANX005 against C1q activation of the classical complement cascade. ANX005 fully inhibited ex vivo hemolysis in serum from humans, cynomolgus and rhesus monkeys, dogs, and rats (Figure 2). Consistent with the lack of binding to rabbit C1q, ANX005 did not inhibit ex vivo hemolysis in rabbit serum (Figure 2). On average, hemolysis was inhibited by ANX005 in the different species, with an IC50 of ∼350 ng/mL (2.5 nM) in this assay system (summarized in Table 4).
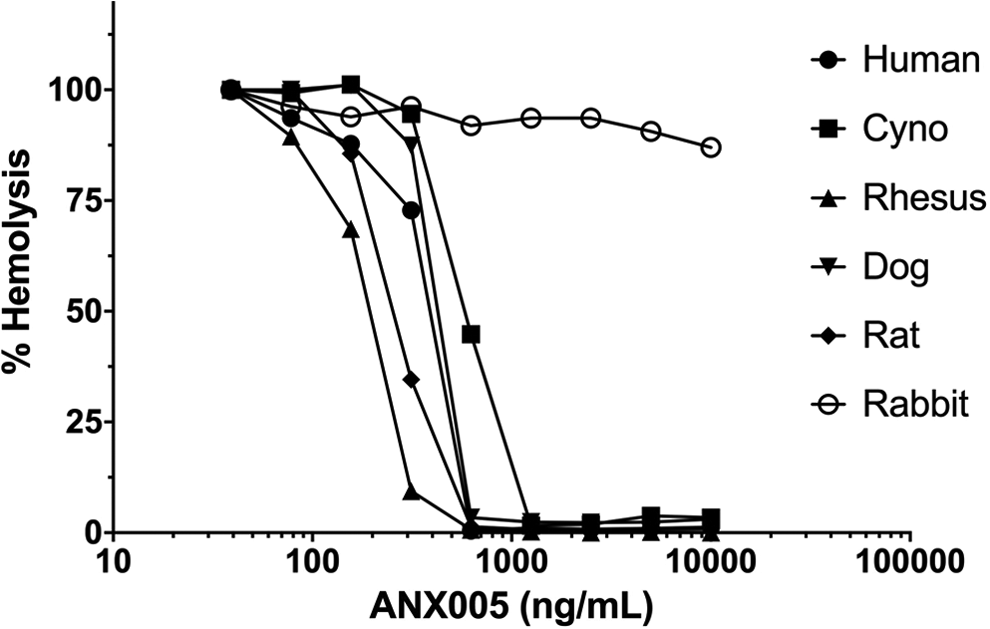
Inhibition of ex vivo hemolysis in serum from humans, cynomolgus monkeys, rhesus monkeys, dogs, rats, and rabbits in the presence of increasing concentrations of ANX005. Sheep red blood cells were sensitized with surface-reactive antibodies and then incubated with sera from different species (final dilution of 100-200). Hemolysis was induced by C1q present in the sera, which interacted with the antibody-coated RBC surface to trigger activation of the classical complement cascade. Hemolysis was monitored by measuring the release of hemoglobin. Data were normalized and plotted relative to baseline hemolysis for each species. RBC indicates red blood cell.
Relative IC50 of ANX005 in RBC Hemolysis Assays.

Abbreviation: RBC, red blood cell.
Sequence homology for C1q subunits A, B, and C across species
Human and cynomolgus monkey C1qA, C1qB, and C1qC share 91%, 95%, and 96% sequence homology, respectively. While the sequence homology for the rat is only 73% to 78% (Table 5), the affinity of ANX005 for rat C1q and its ability to inhibit rat C1q function (described above) support the use of rat as the rodent species for toxicology studies. Therefore, the rat and cynomolgus monkey were selected as pharmacologically relevant species for the pharmacokinetic and toxicology evaluation of ANX005.
Sequence Homology for C1q Subunits A, B, and C Across Species.
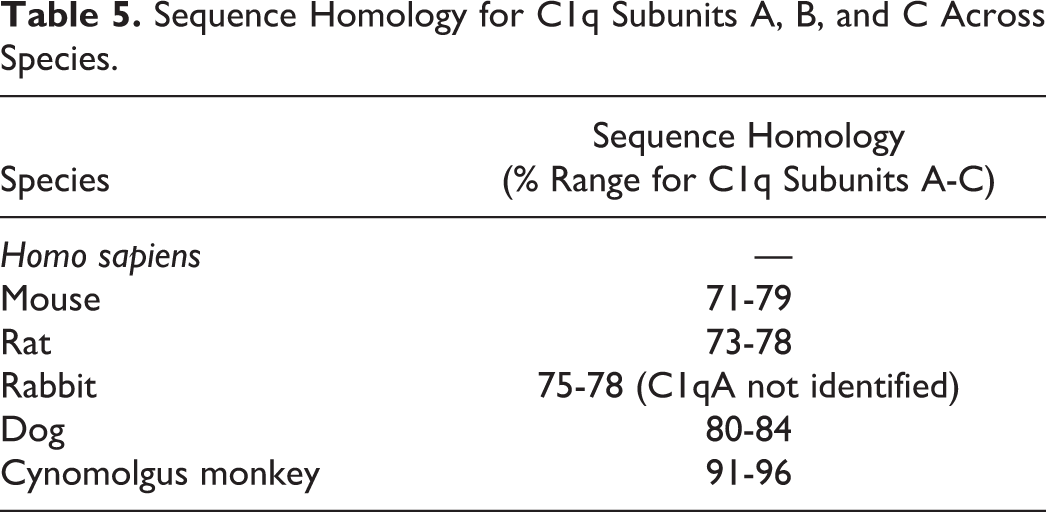
Although the dog could also be considered a pharmacologically relevant nonrodent species, the cynomolgus monkey was selected over the dog because the sequence homology of human and cynomolgus C1q is 91% to 96% versus 80% to 84% with dog C1q. Given the complexity of C1q, a protein comprised 18 subunits, and its interaction with at least a dozen ligands, complement components and complement regulators, it was thought that cynomolgus monkeys would better predict potential immunotoxicity related to ANX005 inhibition.
Ex vivo Human Tissue Cross-Reactivity
A tissue cross-reactivity study was conducted using cryosections from a full panel of human tissues from at least 3 different human donors to evaluate the on target and off target tissue binding of ANX005. Due to the ubiquitous presence of C1q in blood and tissue fluids, and its production by multiple cell types, specific ANX005 staining was observed in all human tissues examined, except on the surface of blood cells. ANX005-specific staining consisted primarily of minimal to moderate intensity extracellular and/or cytoplasmic staining in multiple cell types and tissue elements. No staining patterns were observed to indicate off target cross-reactivity of ANX005 (data not shown).
In Vivo Studies
Two studies in rats and 3 studies in cynomolgus monkeys were performed. The experimental design and results of these studies are discussed below and summarized in Tables 1, 2, and 6–9.
TK Parameters for ANX005 in Sprague Dawley Rats (Males and Females Combined) From the 4-Week Non-GLP and GLP Toxicology Studies.
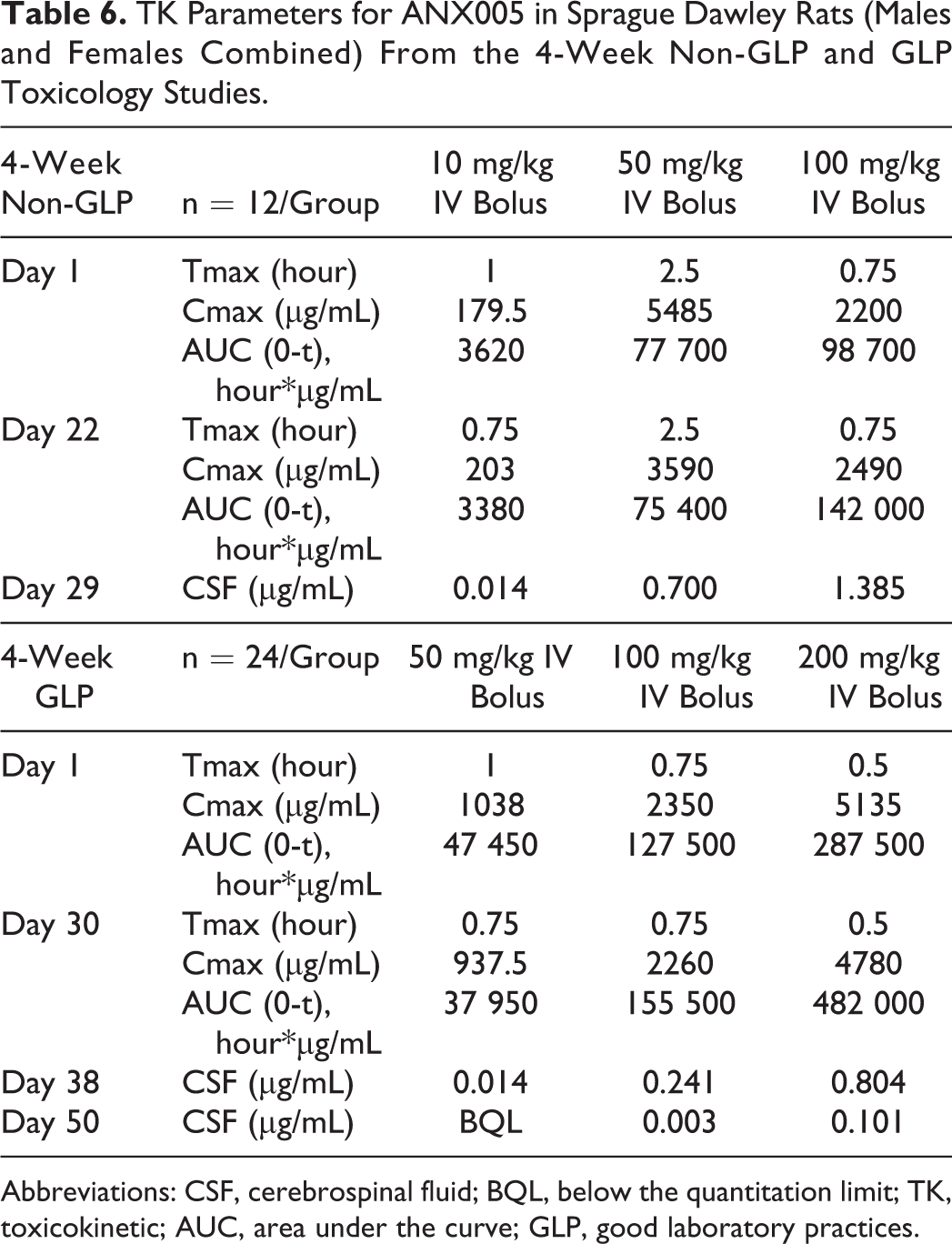
Abbreviations: CSF, cerebrospinal fluid; BQL, below the quantitation limit; TK, toxicokinetic; AUC, area under the curve; GLP, good laboratory practices.
CSF/Serum Ratios From the 4-Week GLP Rat Toxicology Study (GLP).
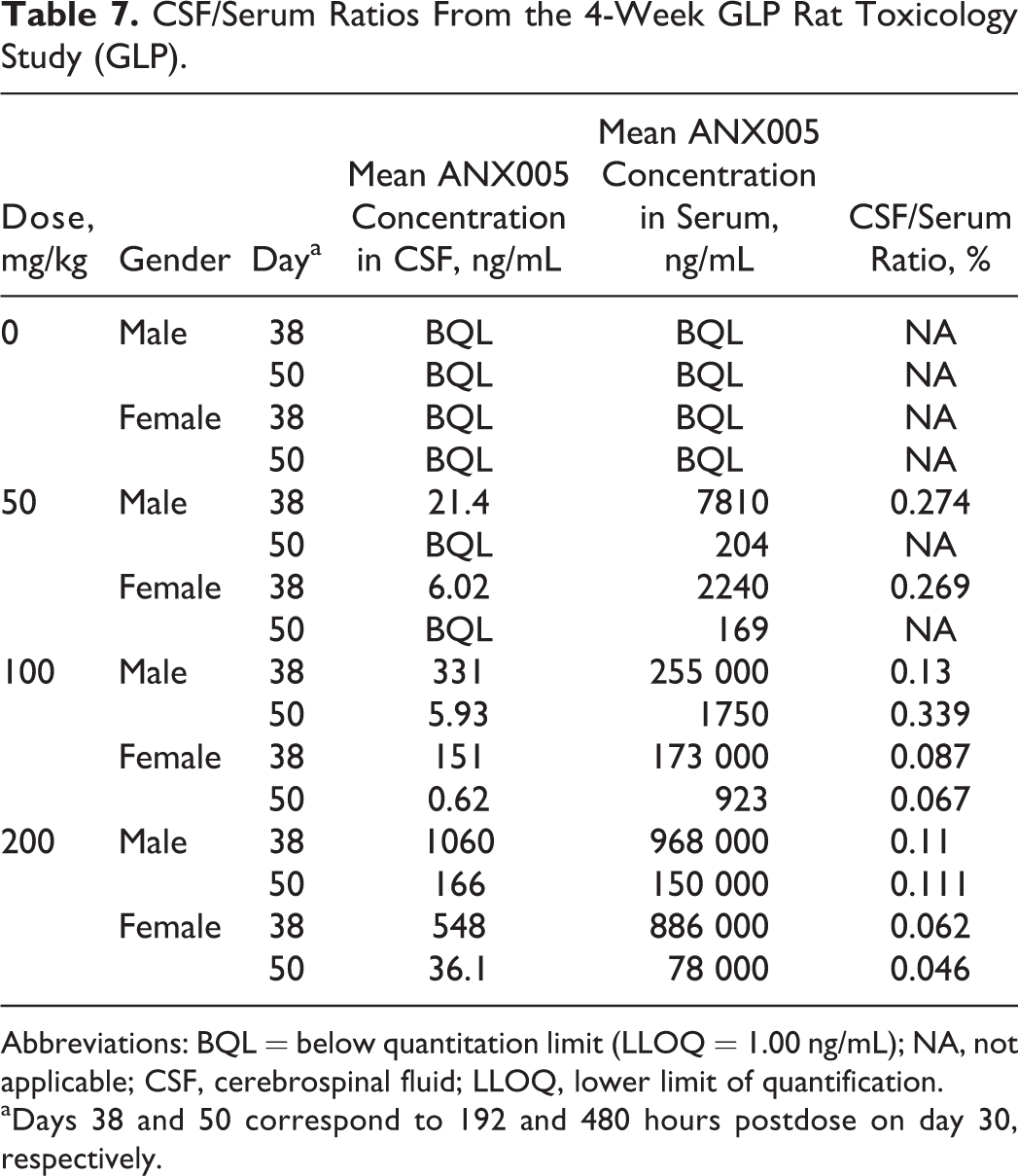
Abbreviations: BQL = below quantitation limit (LLOQ = 1.00 ng/mL); NA, not applicable; CSF, cerebrospinal fluid; LLOQ, lower limit of quantification.
aDays 38 and 50 correspond to 192 and 480 hours postdose on day 30, respectively.
The TK Parameters for ANX005 in Cynomolgus Monkey Serum (Males and Females Combined) From the PK (Non-GLP) and Toxicology Studies (Non-GLP and GLP).
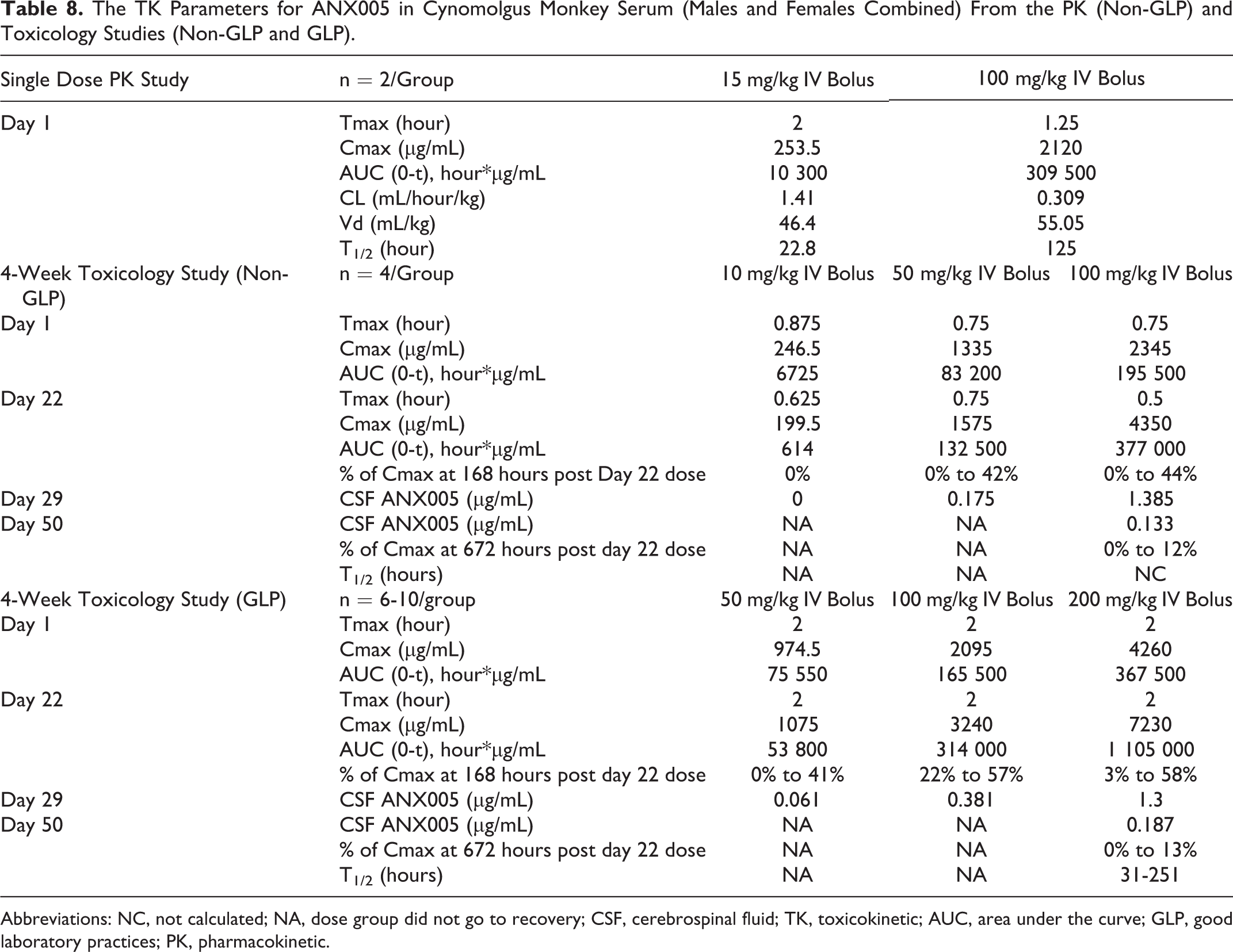
Abbreviations: NC, not calculated; NA, dose group did not go to recovery; CSF, cerebrospinal fluid; TK, toxicokinetic; AUC, area under the curve; GLP, good laboratory practices; PK, pharmacokinetic.
Individual ANX005 CSF and Serum Concentrations on Days 29 or 50 From the 4-Week GLP Monkey Toxicology Study.
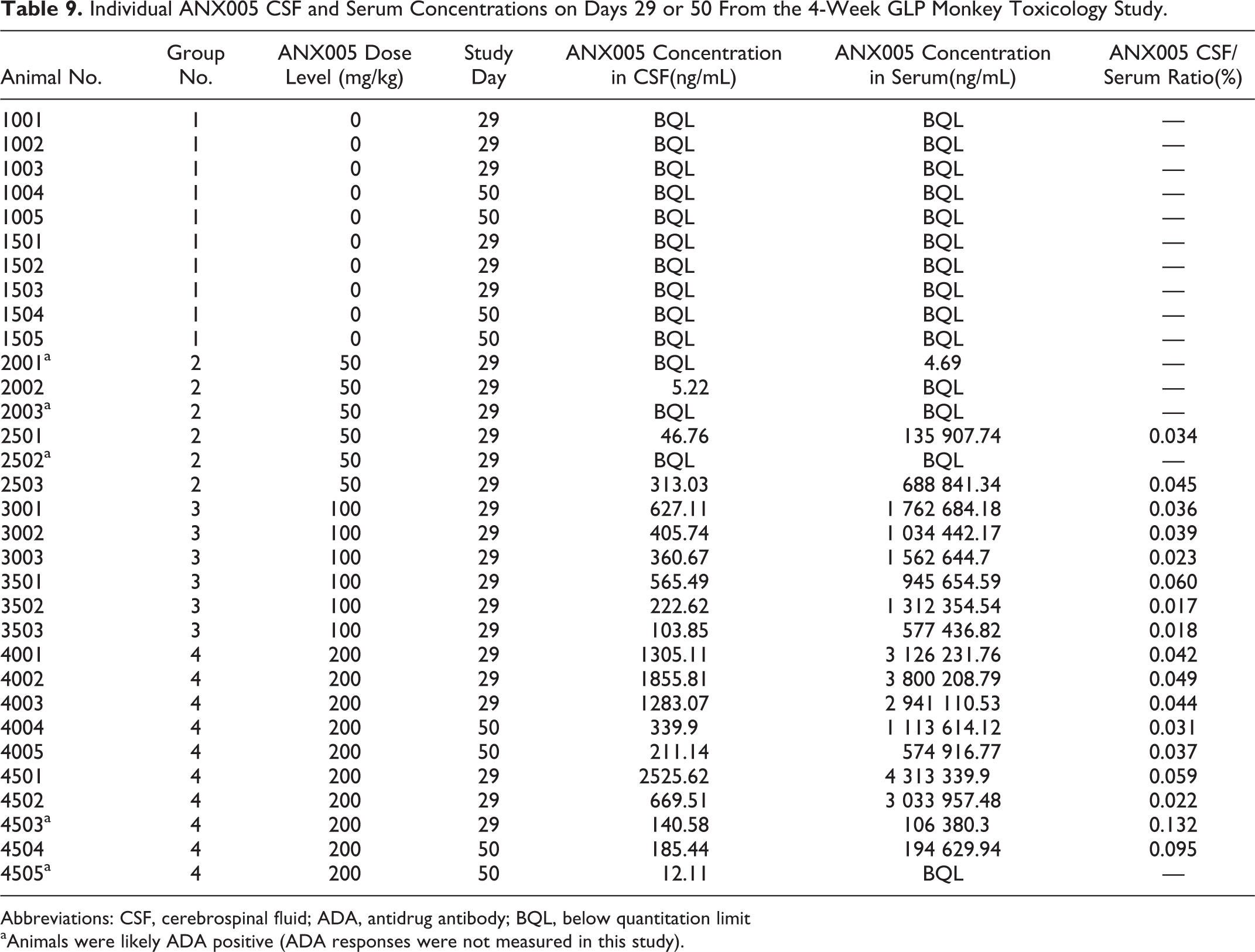
Abbreviations: CSF, cerebrospinal fluid; ADA, antidrug antibody; BQL, below quantitation limit
aAnimals were likely ADA positive (ADA responses were not measured in this study).
Sprague Dawley Rat Studies
Four-Week Rat Non-GLP and GLP Toxicology Studies
Two 4-week toxicology studies were performed (experimental design provided in Table 1; results in Tables 6 and 7).
In the non-GLP study, ANX005 was dosed at 10, 50, and 100 mg/kg versus vehicle, as an intravenous bolus to male and female Sprague Dawley rats once weekly for 4 weeks. All animals survived to the scheduled day of necropsy (day 29). In the GLP study, ANX005 was dosed at 50, 100, and 200 mg/kg versus vehicle on days 1, 8, 15, and 30, followed by a 3-week recovery period. All animals survived in the GLP study to the scheduled days of necropsy—day 38 for the main study animals and day 50 for the recovery animals. In both studies, there were no ANX005-related changes in clinical signs, body weight, body weight gain, food consumption, or clinical pathology parameters. Additionally, there were no ANX005-related changes in gross pathology, organ weight, or histopathology.
ANX005 levels were measured in the serum and CSF, and findings were similar between the 2 studies (Table 6) and are provided graphically for the GLP study in Figure 3. In serum, the AUC generally increased greater than dose proportionally between the lower doses but was approximately dose proportional between 100 and 200 mg/kg. The Cmax showed a dose-proportional increase. The estimated half-life (T½) was up to 18 hours at 10 mg/kg, 50 hours at 50 mg/kg, 80 hours at 100 mg/kg, and 106 hours at 200 mg/kg. There was no apparent sex difference in the TK parameters, and no notable accumulation occurred after the 4 IV doses of ANX005 in either experiment (Table 6). The apparent volume of distribution (Vd) ranged between 14 and 141 mL/kg within the 2 studies, suggesting that ANX005 was not distributed extensively beyond the vasculature.
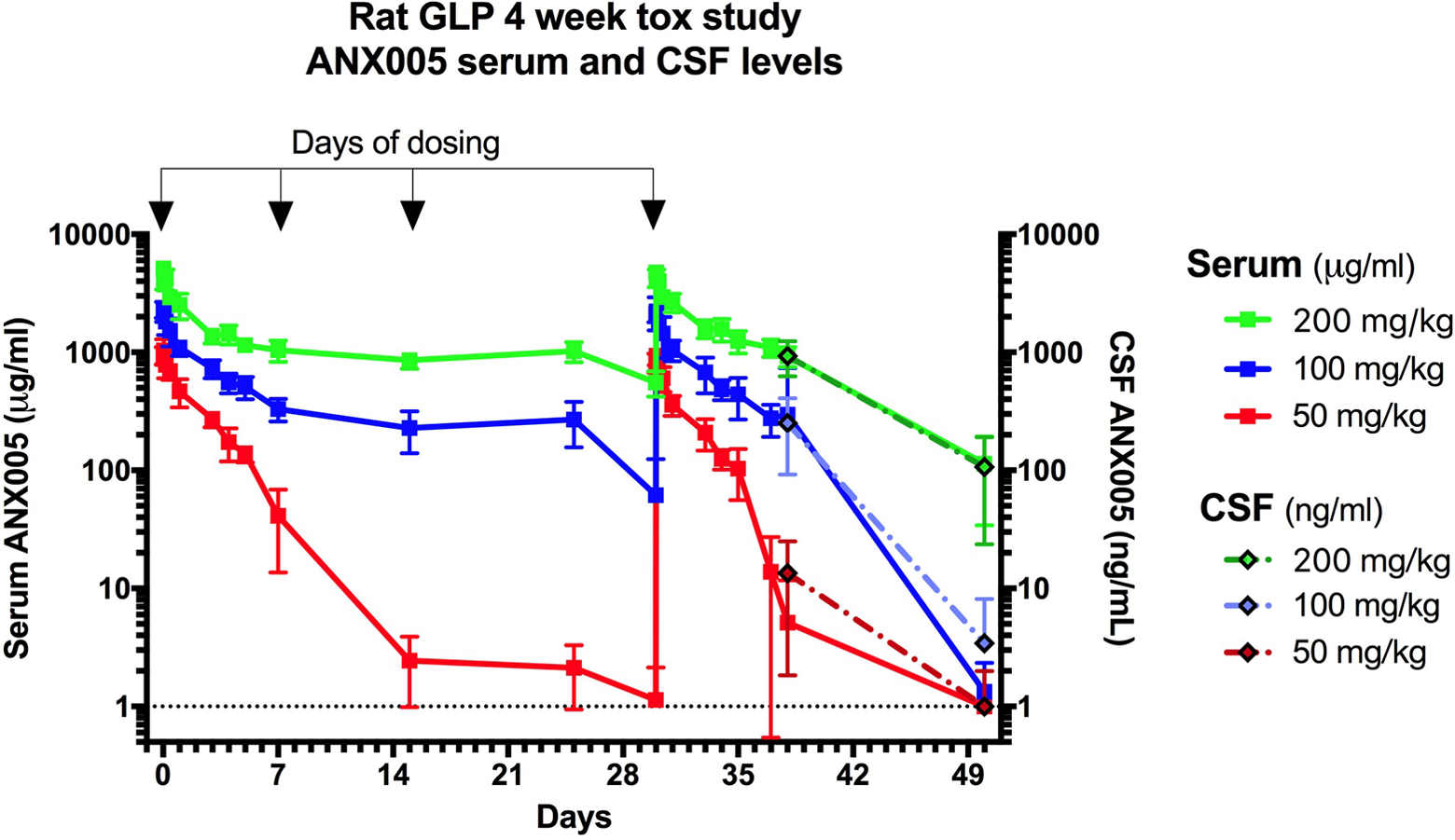
ANX005 was administered to male and female Sprague Dawley rats by IV bolus on days 1, 8, 15, and 30 at doses of 50, 100, or 200 mg/kg versus vehicle. Serum samples were collected at multiple time points within the weeks after the first and fourth doses, and at nadir before the second and third doses. Blood and CSF samples were collected from the main study animals on day 38 and from the recovery animals on day 50. CSF indicates cerebrospinal fluid.
ANX005 levels in the CSF were below the lower limit of quantification (LLOQ) for the control animals in both studies and for the 10-mg/kg males on day 29 in the non-GLP study but were detectable in all other groups. In the CSF, mean ANX005 concentrations increased with dose and were greater than dose proportional between all dosing levels (Table 6). As shown in the GLP study (Figure 3), there was good correlation between CSF and serum levels on days 38 and 50, with CSF concentrations representing approximately 0.1% to 0.2% those of the serum (Table 7). These results are consistent with the findings for most antibodies in the peripheral circulation, which are present in the CSF at 0.2% serum levels. 7
In conclusion, weekly IV administration of ANX005 to Sprague Dawley rats for 4 weeks provided adequate drug exposure in both studies, with full coverage out to 8 weeks in the recovery animals of the GLP study, including anticipated levels of exposure to the CNS. There was no toxicity at doses up to 200 mg/kg, the highest dose evaluated. Based on these results, the no observed adverse effect level (NOAEL) was 200 mg/kg in the rat.
Cynomolgus Monkey Studies
A PK study and two 4-week toxicology studies were performed (experimental design provided in Table 2; results in Tables 8 and 9).
Single-dose PK study
The PK and pharmacodynamics (PD) of ANX005 were determined following a single IV bolus dose of ANX005 administered at 15 and 100 mg/kg to cynomolgus monkeys (1 male and 1 female/dosing level—Figure 4). The Cmax was 2 hours (except for 1 male in the 100-mg/kg group with a Cmax of 0.5 hours). Peak serum levels of free-ANX005 were ∼250 µg/mL at 15 mg/kg and ∼ 2000 µg/mL at 100 mg/kg. At the low dose of 15 mg/kg, serum levels of ANX005 were cleared over 5 days, while at the top dose of 100 mg/kg, ANX005 levels remained elevated for up to 20 days (last time point evaluated; Figure 4A). Serum levels of free C1q were followed as a PD marker. When ANX005 was present, free levels of C1q were undetectable in all animals, and when drug levels became undetectable, as in both low-dose animals and in one of the high-dose animals, C1q levels simultaneously returned toward baseline (Figure 4B). Consistent with the reduction in serum levels of free C1q, there was full inhibition of classical complement-mediated hemolysis in ex vivo serum samples (Figure 4C). In conclusion, these results demonstrate that intravenous dosing of ANX005 produces serum levels of ANX005 with corresponding reductions in free C1q and classical complement activity.
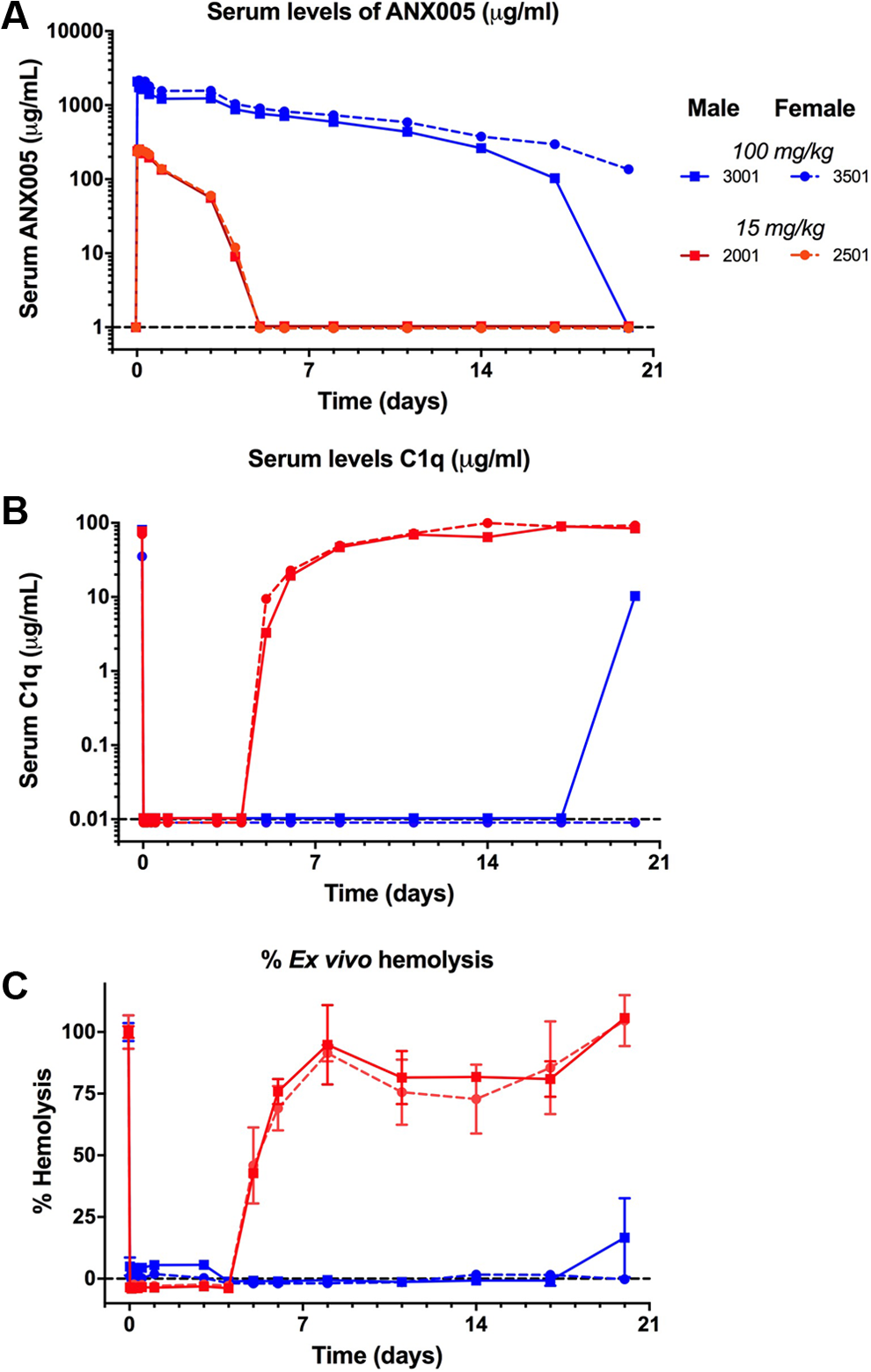
ANX005 was administered to male and female cynomolgus monkeys as a single IV bolus at 15 or 100 mg/kg. Serum and CSF samples were collected at the indicated time points for (A) pharmacokinetics, (B) C1q levels, and (C) evaluation of C1q functional activity/complement-mediated hemolysis ex vivo. Each line represents measurements from individual animals—1 male and 1 female at each dose as marked in the legend. Black dotted lines represent the lower limits of quantitation for each assay. CSF indicates cerebrospinal fluid.
Four-week cynomolgus monkey toxicology study (non-GLP)
In the 4-week non-GLP cynomolgus monkey toxicology study, ANX005 or vehicle (PBS) were administered to male and female cynomolgus monkeys by IV bolus at doses of 10, 50, and 100 mg/kg once weekly for 4 weeks, followed by a 4-week recovery period. All animals survived until the scheduled necropsy (day 29 for main study animals or day 50 for recovery animals). Once-weekly administration of ANX005 for 4 weeks was well tolerated, and there were no ANX005-related changes in clinical signs, body weights, food consumption, hematology, clinical chemistry, coagulation, cytokines, gross necropsy findings, organ weights, or histopathology.
A dose-dependent increase in serum ANX005 levels was observed, with peak exposures of ∼250 µg/mL at 10 mg/kg, ∼1000 µg/mL at 50 mg/kg, and ∼2000 µg/mL at 100 mg/kg (Figure 5A). Maximum serum concentrations of ANX005 were observed between 0.5 and 1 hour postdose. Serum levels of ANX005 in the 10 mg/kg group cleared over 3 to 5 days, while ANX005 levels remained elevated in the 50- and 100-mg/kg groups for the duration of the study. In the 50- and 100-mg/kg groups, trough levels of drug increased with each weekly dose (measured on days 8, 15, 22, and 29), suggesting drug accumulation (Figure 5A). At the 10-mg/kg dose, clearance of ANX005 was more rapid at week 4 than at week 1, suggesting an anti-drug antibody (ADA) response at this lower dose (although ADA responses were not measured in this study). More rapid clearance of ANX005 between weeks 1 and 4 was also observed for 1 male in the 50-mg/kg group (3102) and 1 female in the 100-mg/kg group (4502). Excluding these animals, trough levels of drug increased with each weekly dose in the higher dose groups (measured on days 8, 15, 22, and 29), suggesting drug accumulation (Figure 4A). Drug serum levels remained high within the 50- and 100-mg/kg dosing groups 7 days after the last dose (32% and 38% of Cmax, respectively), indicating substantial exposure throughout the study (Table 8).
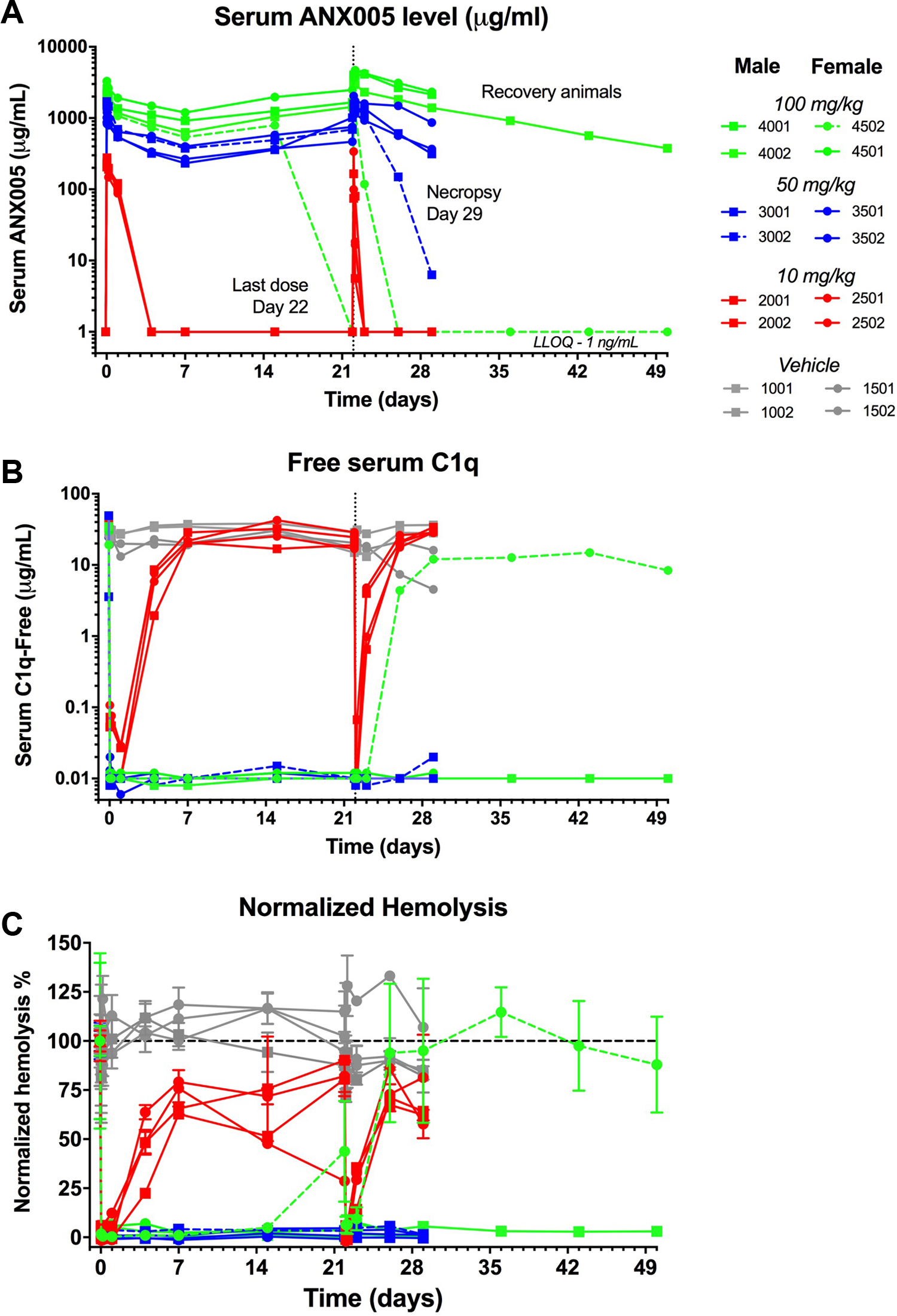
ANX005 was administered to male and female cynomolgus monkeys by IV bolus once weekly for 4 weeks at doses of 10, 50, or 100 mg/kg. Serum samples were collected at various time points for (A) pharmacokinetics, (B) C1q levels, and (C) evaluation of complement-mediated hemolysis ex vivo. Each colored line represents measurements from individual animals. The main study group contained 4 males and 4 females at each dose, as marked in the legend, and were terminated on day 29. One high-dose male and 1 high-dose female proceeded to a 4-week dose-free recovery period and were terminated on day 50.
At time points when ANX005 was measurable in the serum, levels of free C1q were undetectable (Figure 5B). Free C1q returned when ANX005 levels dropped below detection, including all animals in the 10-mg/kg group, as well as the 1 animal in the 100-mg/kg group (4502) with an apparent ADA response. These data indicate that the presence of ADA had a negative impact on ANX005 and corresponding C1q levels. Otherwise, serum levels of free C1q were reduced in the 50- and 100-mg/kg groups over the entire 4-week period (Figure 5B). Additionally, a dose-dependent inhibition of ex vivo complement-mediated hemolysis byANX005 was evident and consistent with ANX005 and free C1q levels (Figure 5C), including the high-dose female (4502) with an apparent ADA response; as ANX005 levels fell below detection limits, C1q levels, and inhibition of complement-mediated hemolysis returned toward pretreatment levels in week 3 and at later time points.
ANX005 concentrations in the CSF were below the lower limit of quantification (LLOQ = 1 ng/mL) for the control group and for the 10-mg/kg group at day 29 (which correlated with undetectable serum levels). ANX005 levels in CSF were approximately 0.04% and 0.11% of those in the serum for the 50- and 100-mg/kg groups, respectively (males and females combined—Table 8). In the 100-mg/kg recovery group, CSF levels were 0.07% of serum levels on day 50 (n = 1, excluding animal 4502 with an apparent ADA response). As in the rat studies, these results indicate that ANX005 penetrated the blood/brain barrier at levels consistent with general antibodies in the circulation. 7 Importantly, these levels of ANX005 CNS penetration were sufficient to fully occupy C1q in the CSF at the higher dose on day 29 (Figure 6).
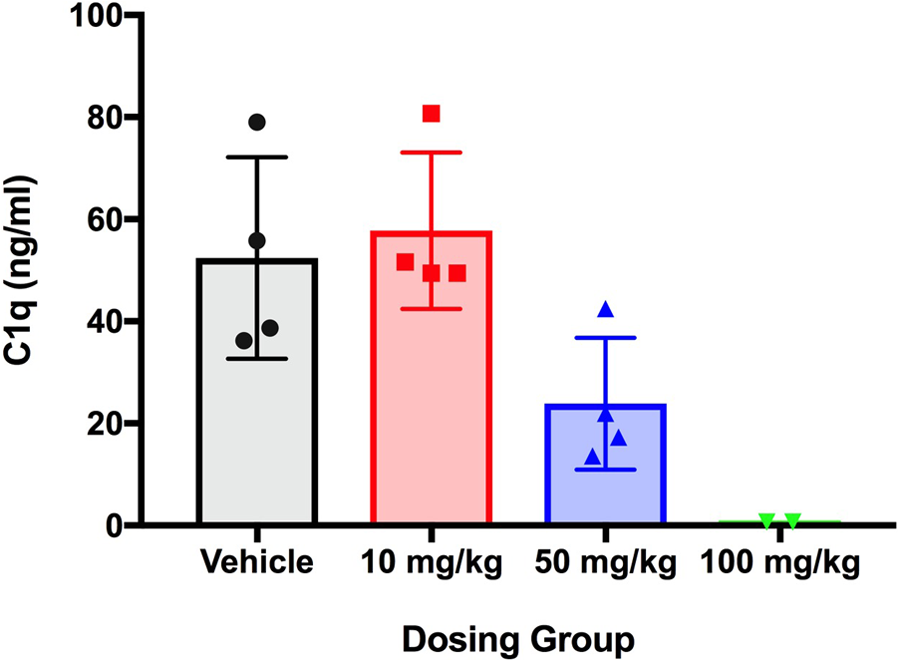
ANX005 was administered to male and female cynomolgus monkeys by IV bolus once weekly for 4 weeks at doses of 10, 50, or 100 mg/kg. CSF samples were collected from the main study animals at day 29, and C1q levels were measured by ELISA. ELISA indicates enzyme-linked immunosorbent assay; CSF, cerebrospinal fluid.
In conclusion, administration of ANX005 via IV bolus injection once weekly for 4 weeks at doses up to 100 mg/kg showed no evidence of toxicity in cynomolgus monkeys. Furthermore, the PD properties of the drug, as measured by C1q levels and inhibition of complement-mediated hemolysis in the serum, were consistent with its PK. Furthermore, the drug penetrated the CNS at expected levels and fully occupied C1q at the high dose.
The 4-week toxicology study (GLP)
ANX005 was administered at doses of 50, 100, and 200 mg/kg versus vehicle (PBS) by weekly IV bolus dosing for 4 weeks to male and female cynomolgus monkeys, followed by a 4-week recovery period. All animals survived until scheduled necropsy (days 29 and 50). There were no ANX005-related clinical observations, body weight changes, ocular effects, or neurologic abnormalities observed in this study. Additionally, no ANX005-related changes were observed for respiratory rate, blood pressure, or heart rate, and all ECGs were qualitatively normal. There were no ANX005-related changes in clinical pathology parameters (hematology, coagulation, clinical chemistry, or urinalysis), organ weights, or microscopic or macroscopic histopathology observations.
In all dosing groups, trough levels of drug increased with each weekly dose (measured predose on days 8, 15, 22, and prior to necropsy on day 29), suggesting drug accumulation (Figure 7). ANX005 concentrations were maintained in the mid- and high-dose groups (Figure 7). Although ADA responses were not measured in this study, several of the low-dose animals (50 mg/kg) and 1 high-dose female (200 mg/kg, 4503) appeared to have an ADA response, based on PK comparisons between the first and fourth weeks, with markedly increased drug clearance. Another high-dose female (4505) exhibited increased drug clearance during the recovery period. Excluding these animals, drug exposure was generally dose proportional between the 100- and 200-mg/kg dosing groups. Drug serum levels remained high within the 50-, 100-, and 200-mg/kg dosing groups 7 days after the final dose (26%, 37%, and 42% of Cmax, respectively, excluding animals that likely had an ADA response), indicating substantial exposure throughout the study (Table 8). Following the 4-week recovery period in the 200-mg/kg animals, 8% of the drug remained in the circulation compared to Cmax, consistent with a mean half-life of 7 to 8 days (excluding animal 4505 with a likely ADA response). Although the half-life was slightly different between males and females, there was no clear gender difference in overall drug exposure. The estimated Vd suggested that ANX005 was not distributed extensively beyond the vasculature.
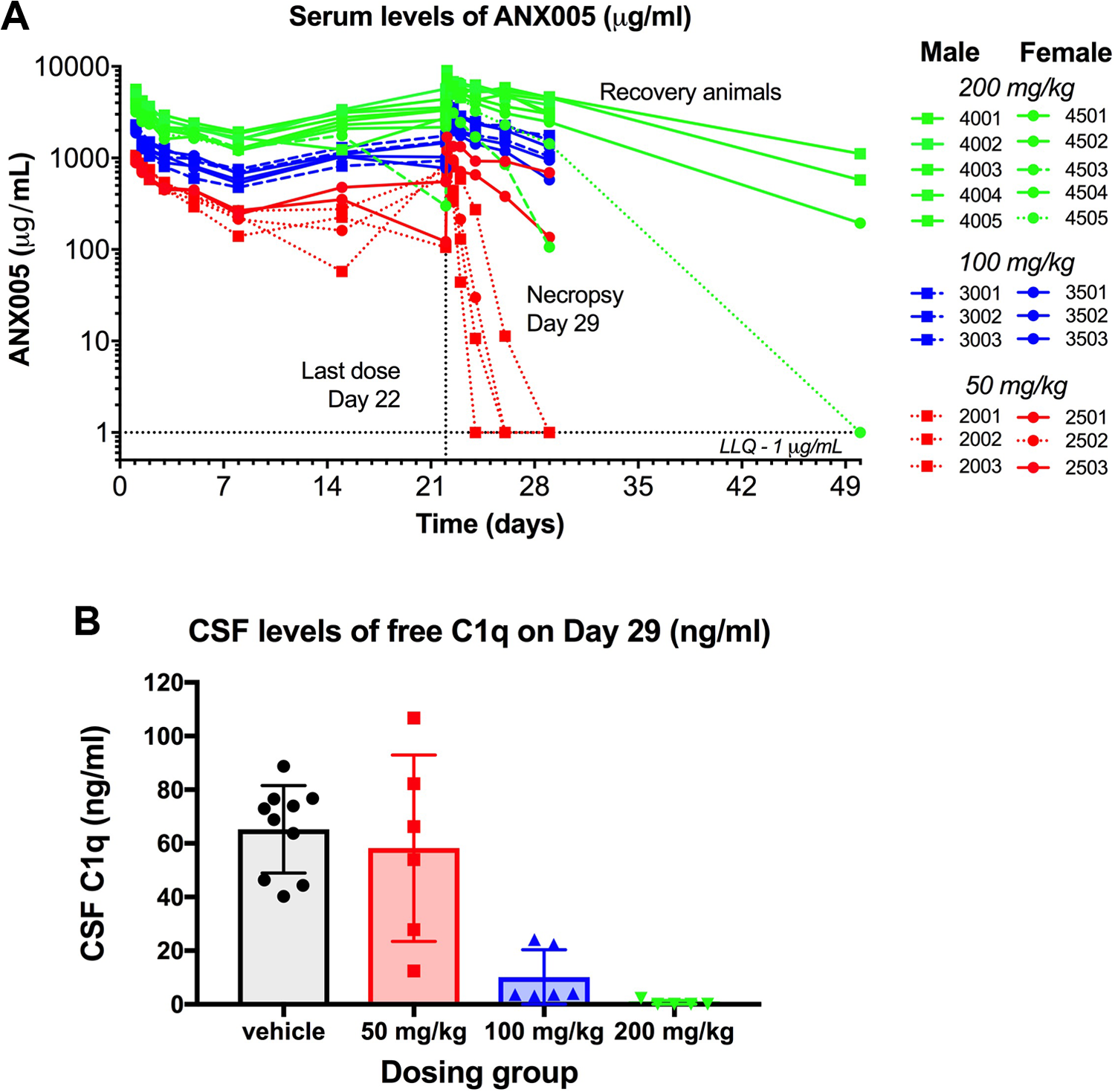
ANX005 was administered to male and female cynomolgus monkeys by IV bolus once weekly for 4 weeks at doses of 50, 100, or 200 mg/kg. (A) Serum samples were collected at multiple time points during the weeks after the first and fourth doses for pharmacokinetics, and at nadir prior to the second and third doses. The main study animals were terminated on day 29, and 4 high-dose animals (2 males, 2 females) proceeded to a dose-free 4-week recovery period prior to termination on day 50. (B) CSF samples were collected from the main study animals on day 29 for analysis of free C1q levels. Female 4503, with an apparent ADA response, was eliminated from the analysis. CSF indicates cerebrospinal fluid; ADA, antidrug antibody.
On days 29 and 50, CSF concentrations of ANX005 were quantifiable from all animals in the 100- and 200-mg/kg groups, indicating the presence of measurable ANX005 in the CNS following repeated IV administration in monkeys. On day 29, the ANX005 in the CSF relative to the serum was 0.03% to 0.05% at 50 mg/kg (n = 2 females), 0.02% to 0.06% at 100 mg/kg (n = 6), and 0.02% to 0.13% at 200 mg/kg (n = 6). By day 50 (4 weeks following the final dose on day 22), ANX005 concentrations in the CSF from the recovery animals in the 200-mg/kg group remained detectable at 0.03% to 0.04% for the males (n = 2) and 0.1% for the females (n = 1; Table 9). Overall, CSF exposures were within range but somewhat lower than the 0.2% anticipated for circulating antibodies. As before, these levels of CNS penetration were sufficient to occupy C1q in the CSF on day 29 in the 100-mg/kg and 200-mg/kg dosing groups, providing full saturation in most animals (Figure 7B).
In summary, IV bolus administration of ANX005 once weekly showed no adverse toxicity in cynomolgus monkeys at dose levels up to 200 mg/kg, the highest dose evaluated. As in the prior studies, the PD properties of ANX005 were consistent with the PK. ANX005 administered via IV was able to fully occupy C1q in the CSF at higher doses. Based on these results, the NOAEL was 200 mg/kg, the highest dose tested, in cynomolgus monkeys.
Discussion
Autoimmune and neurodegenerative diseases are complex disorders involving multiple components of the immune system. In GBS, autoantibodies react with components of the peripheral nerve, recruiting C1q/activation of the classical complement cascade and resulting in peripheral nerve damage. 1,2 In AD and other neurodegenerative disorders, C1q interacts directly with synaptic membranes to activate the classical cascade, resulting in synaptic elimination and CMND. 8 Antibody-mediated diseases, such as GBS, are often treated with intravenous immunoglobulin (IVIG) or plasmapheresis, which provide some measure of benefit; however, complement activation and its associated damage continue to occur. 1,2,9 Thus, unfortunately, many GBS patients treated with IVIG continue to decline, resulting in a higher potential for prolonged or permanent disability. 10 In CNS disorders involving CMND, few treatment options exist, and prolonged activation of the classical complement cascade contributes to progressive loss of neuronal function with cognitive decline.
ANX005 is a human monoclonal IgG4 antibody that targets C1q and is currently being developed as a potential therapeutic agent for treatment of antibody-mediated autoimmune disease and neurodegenerative disorders. To support the clinical development of ANX005, several nonclinical studies were conducted, including in vitro pharmacology assays, a tissue cross-reactive study in a panel of normal human tissues, and studies in rats and cynomolgus monkeys to evaluate the PK, PD, and safety of ANX005. ANX005 showed high-binding affinity to C1q derived from sera across multiple species and exhibited full inhibition of ex vivo complement-mediated hemolysis in human, monkey, dog, and rat serum. Based on these data, the cynomolgus monkey and the rat were selected as pharmacologically relevant species for toxicological evaluation of ANX005. The tissue cross-reactivity study in normal human tissues showed minimal to moderate staining with all tissues, consistent with the fact that C1q is a plasma protein and ubiquitously present within the vasculature and interstitial fluid throughout the body; however, no distinct staining patterns were visible to indicate cross-reactivity with other tissue components.
The toxicology studies in rats and cynomolgus monkeys showed that ANX005 was well tolerated when administered as an IV bolus injection once weekly for 4 weeks at a dose range of 10 to 200 mg/kg. No toxicity was identified. Thus, the NOAEL for the rat and monkey studies was 200 mg/kg, the highest dose evaluated, which is 200-fold higher than the proposed first-in-human starting dose of 1 mg/kg. Pharmacokinetics and TK showed that, at higher doses, ANX005 serum levels generally increased in rats and monkeys in a dose-proportional manner. In vivo PK/PD evaluations showed that, at measurable serum levels of ANX005, C1q was not detectable in the serum of rats and monkeys. Complete reduction of free C1q in the serum was observed at doses in the range of 50 to 200 mg/kg with ANX005 levels in the range of 300 to 4000 µg/mL. Based on serum measurements with the same assays, C1q levels in humans are similar to those in monkeys (20-70 µg/mL; data not shown). Therefore, doses of 50 to 200 mg/kg in humans should achieve full inhibition of C1q, and serum levels of drug should be maintained for 2 to 4 weeks based on the single-dose PK study in monkeys. These results support advancement of ANX005 into the clinic to evaluate its potential therapeutic benefit for patients with GBS.
ANX005 was also present in the CSF of rats and monkeys at levels consistent with those of typical antibodies within the circulation. CSF levels of ANX005 in the range of 100 to 2000 ng/mL (700-14,000 pM) were achieved in monkeys at doses of 50 to 200 mg/kg. Importantly, these levels of CNS penetration were sufficient to fully deplete free C1q in the CSF at doses of 100 and 200 mg/kg. As with monkeys, our assays indicate that normal levels of C1q in the CSF of humans are in the range of 20 to 200 ng/mL (40-440 pM; data not shown), which suggests that intravenous doses of 50 to 200 mg/kg ANX005 in humans will allow full occupancy of C1q in the CSF. As above, these results support advancement of ANX005 into the clinic for evaluation of therapeutic benefit in patients with neurodegenerative disorders such as AD.
In summary, our studies demonstrate that ANX005 is a potent anti-C1q-targeting antibody with the potential of therapeutic benefit in treatment of autoimmune and neurodegenerative diseases.
Footnotes
Acknowledgments
The authors thank Barbara Mounho-Zamora and Deborah Proctor for their critical reviews and Rick Nelson for editorial assistance. The authors also thank the following Study Directors at Charles River for the successful conduct of these studies: Courtney Olson (Single-dose Cynomolgus Monkey PK Study), Subramanya Karanth (Non-GLP 4-week Cynomolgus Monkey Study), Deanna Newcomb (Non-GLP 4-week Rat Study), Rob Kaiser (GLP 4-week Rat Study), Steve Wilson (GLP 4-week Cynomolgus Monkey Study), and Curtis Chan (Tissue Cross Reactivity in Human Tissues).
Author Contributions
Lansita, J.L. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Mease, K.M. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Haiyan, Q. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Yednock, T. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Sankaranarayanan, S. contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised manuscript; Kramer, S. contributed to conception and design, contributed to interpretation, critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All contributors are employees or contractors of Annexon Biosciences.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by an SBIR (Small Business Innovation Research) grant from the National Institutes of Health (1R44AG054404-0). The remaining portion was funded by Annexon Biosciences.
