Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) assessed the safety of 39 polyether lanolin ingredients as used in cosmetics. These ingredients function mostly as hair conditioning agents, skin conditioning agent—emollients, and surfactant—emulsifying agents. The Panel reviewed available animal and clinical data, from previous CIR safety assessments of related ingredients and components. The similar structure, properties, functions, and uses of these ingredients enabled grouping them and using the available toxicological data to assess the safety of the entire group. The Panel concluded that these polyether lanolin ingredients are safe in the practices of use and concentration as given in this safety assessment.
Introduction
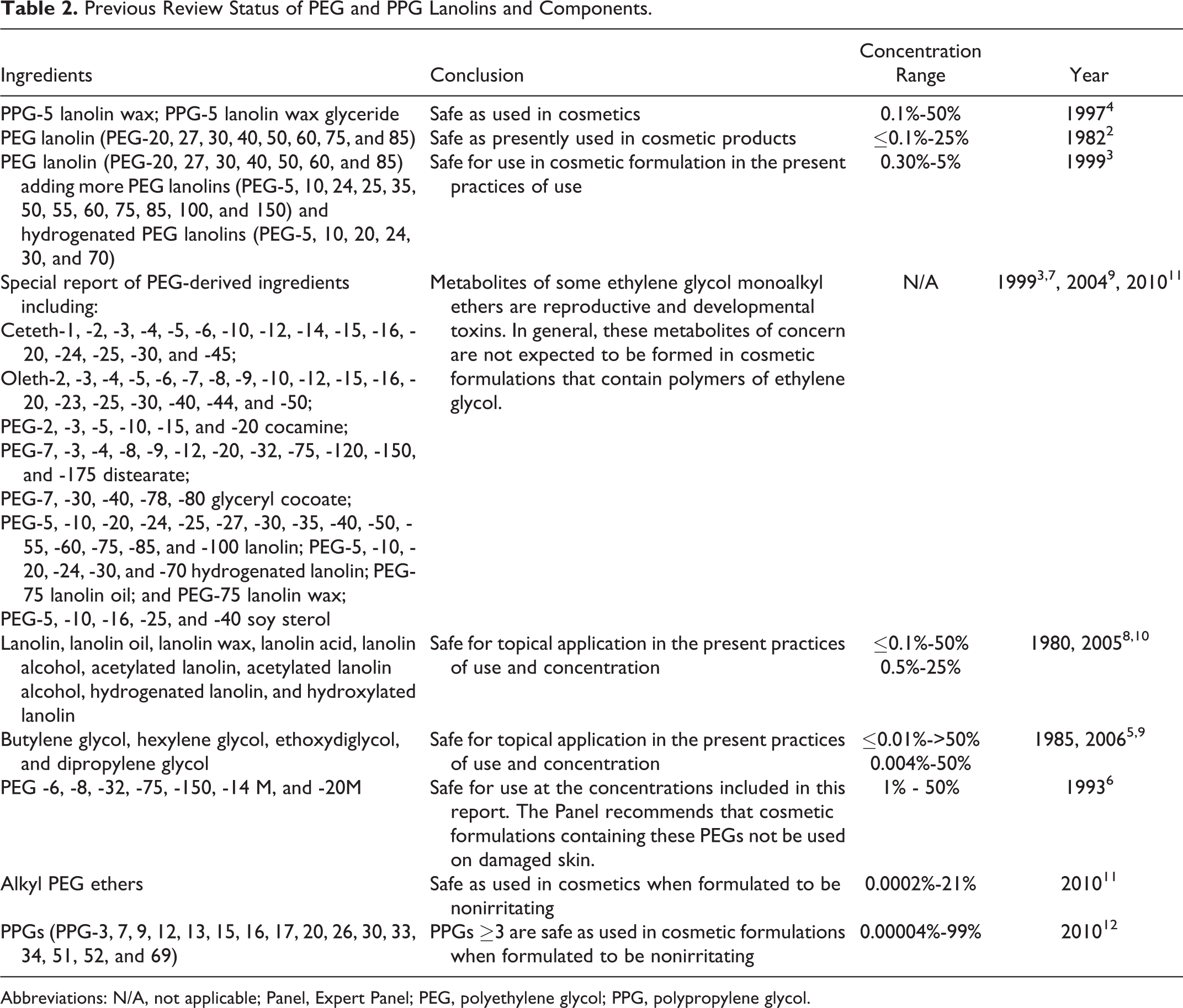
The polyether lanolin ingredients reviewed in this safety assessment mostly function as hair conditioning agents, skin conditioning agent—emollients, and surfactant—emulsifying agents, according to the International Cosmetic Ingredient Dictionary and Handbook. 1 The Expert Panel (Panel) previously reviewed similar polypropylene glycol (PPG) and polyethylene glycol (PEG) lanolins. 2 –4 Because of the similarity in chemical structures and in order to present a complete safety assessment of the polyether lanolin ingredients that are used in cosmetics, these previously reviewed ingredients are included in this report with polyether lanolins that have not yet been reviewed. The full list of the ingredients in this report is provided in Table 1. Because there was an incomplete data set on polyether lanolins in prior Cosmetic Ingredient Review (CIR) reviews, the Panel supplemented those reports with data from existing reports on PEGs, PPGs, and lanolin. 5 –12 (Information from those reports are also used to support the safety of the ingredients included in this report.) That information is described in Table 2. The complete CIR reports are available on the website (http://www.cir-safety.org/ingredients).
Definition and Function of the PEG- and PPG-Lanolin Ingredients in This Safety Assessment.a

Abbreviations: PEG, polyethylene glycol; PPG, polypropylene glycol.
aPEG-25 lanolin was included in the 1999 report but is currently not listed in the Council’s database.
Previous Review Status of PEG and PPG Lanolins and Components.
Abbreviations: N/A, not applicable; Panel, Expert Panel; PEG, polyethylene glycol; PPG, polypropylene glycol.
Chemistry
Definition
The ingredients in this report are polyalkoxylated (polyether) derivatives of lanolin, a refined derivative of the unctuous fat-like sebaceous secretion of sheep. The polyalkoxyl chains are PEGs, PPGs, or polyglycerols, varying in alkoxyl repeat units from as small as 2 to as long as 100. The definitions of these polyether lanolin ingredients are provided in Table 1.
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by industry in response to surveys, conducted by the Personal Care Products Council, of maximum reported use concentration by product category.
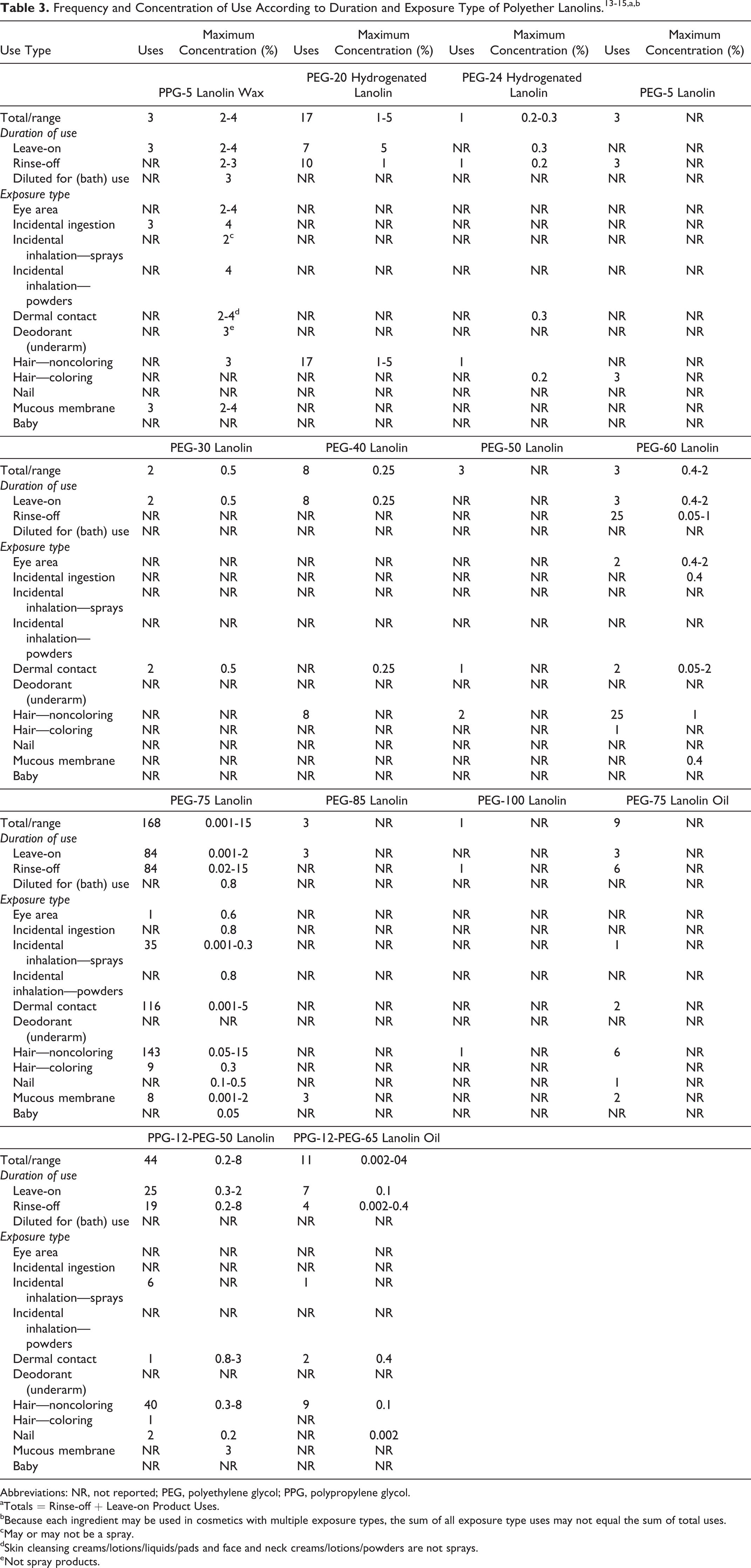
Polyethylene glycol-75 lanolin had the most reported uses at 168 (Table 3). 13 The remaining ingredients with reported uses in the VCRP are reported to be used in 44 or fewer ingredients. These polyether lanolins have a reported maximum concentration of use range of 0.001% to 15%. Polyethylene glycol-75 lanolin is reported to be used in leave-on products (eg, hair straighteners) at up to 15%. Polypropylene glycol-5 lanolin wax is reported to be used in leave-on products that have dermal contact (ie, eye shadow, face powders, and lipstick) at up to 4%. Polypropylene glycol 12 PEG 50 lanolin is reported to be used in rinse-off products (eg, hair conditioners) at up to 8%. 14,15 Ingredients with no reported uses or use concenrations are listed in Table 4.
Abbreviations: NR, not reported; PEG, polyethylene glycol; PPG, polypropylene glycol.
aTotals = Rinse-off + Leave-on Product Uses.
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
cMay or may not be a spray.
dSkin cleansing creams/lotions/liquids/pads and face and neck creams/lotions/powders are not sprays.
eNot spray products.
Abbreviations: PEG, polyethylene glycol; PPG, polypropylene glycol.
Some of these ingredients are used in cosmetic sprays and powders and could possibly be inhaled; for example, PEG-75 lanolin is reported to be used at a maximum concentration of up 0.3% in spray products and PPG-5 lanolin wax is reported to be used at 4% in spray formulations. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared to pump sprays. 16,17 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 18,19 Conservative estimates of inhalation exposures to respirable particles during the use of loose-powder cosmetic products are 400-fold to 1000-fold less compared to protective regulatory and guidance limits for inert airborne respirable particles in the workplace. 20 –22
Effect on Antimicrobial Activity
Polypropylene Glycol Lanolin Alcohols
When PPG-2, -5, -10, and -20 lanolin alcohols (0, 1, 2.5, 5, 10, 15, and 20 μg/mL) were added to a petrolatum-liquid paraffin eye ointment, the antimicrobial activity of chloramphenicol and tetracycline were increased in paper disc assays. 23 The increases were greater with concentration and number of propylene oxide units.
Dermal Irritation
Polyethylene Glycol Lanolins
A dermal irritation test using rabbits (n = 3) of 2 samples of PEG lanolin (100% and 10% in a mixture of polysorbate 60, paraffin, and a preservative; length of PEG not provided) was conducted. 24 The test substances were applied over 2 months; however, the frequency and volume of the applications were not provided. Macroscopic and histologic examination showed that both test substances were well tolerated at 10%; 1 sample produced dry and cracked skin on weeks 3 to 4. However, at 100%, 1 sample caused vesicles or blisters at weeks 2 and 4, which resolved by week 5. The skin was dry and cracked and had a slight thickening of the skin at weeks 3 to 8.
Ocular Irritation
Polyethylene Glycol Lanolin
An ocular irritation test using rabbits (n = 3) of 2 samples of PEG lanolin (10% and 100% in a mixture of polysorbate 60, paraffin, and a preservative; length of PEG not provided) was conducted. 24 The low dose was not tested in the second sample. The eyes were irritated at 1 hour for all doses which subsided at 5 and 48 hours. The irritation was almost completely resolved in the 10% group at 48 hours.
Summary
This is an amended safety assessment of 39 polyether lanolins as used in cosmetics. The polyether lanolins group includes both PPG- and PEG-lanolin ingredients that function in cosmetics mostly as hair conditioning agents, skin conditioning agent—emollients, and surfactant—emulsifying agents.
Polyethylene glycol-75 lanolin had the most uses at 168. The remaining ingredients with reported uses in the VCRP are reported to be used in 44 or fewer ingredients. Polyether lanolins are used at a range of 0.001% to 15%. Polyethylene glycol-75 lanolin is reported to be used in leave-on products at up to 15%. Polypropylene glycol-5 lanolin wax is reported to be used in leave-on products that have dermal contact at up to 4%. Polypropylene glycol-12-PEG-50 lanolin is reported to be used in rinse-off products at up to 8%.
The addition of PPG-2, -5, -10, and -20 lanolin alcohols (0, 1, 2.5, 5, 10, 15, and 20 μg/mL) to a petrolatum-liquid paraffin eye ointment resulted in an increase in the release rate and the antimicrobial activity of chloramphenicol and tetracycline. In another study, PEG lanolin (length of PEG unknown) was reported to be “slightly dermally irritating” at 100% in 1 sample, and not irritating in another. PEG lanolin was also reported to be slight ocular irritants.
Because very little new data were found in the published literature, this report includes summaries of previous reviews of lanolin, PPG-5 lanolin wax and PPG-5 lanolin wax glyceride, PEG lanolin, dipropylene glycol, and alkyl PEG ethers, as well as information from the special report on the reproductive and developmental toxicity of ethylene glycol. All of the supporting ingredients were found to be safe, but with an added proviso for the alkyl PEG ethers, which were determined to be safe when formulated to be nonirritating.
Discussion
Although there are data gaps for the polyether lanolin ingredients, the similar chemical structures, physicochemical properties, and functions and concentrations in cosmetics allow for the grouping of these ingredients with other related ingredients, thereby extending the available toxicological data available on any of the related ingredients to support the safety of members of the group.
The Panel acknowledged that a safety assessment of diethylene glycol (aka PEG-2) has not been completed. However, they noted that PEG-3 is actually a mixture that includes PEG-2, and therefore the Panel concluded that PEG-3 and all PEGs with ≥ 4 ethylene glycol repeat units were safe in the present practices of use and concentration.
A wide range of alkyl ethers of polyethylene glycols have been assessed by the Panel as presenting little or no potential toxicity. The Panel concluded that the likelihood of these polyether ingredients being metabolized to reproductive or developmental toxins was very low. 7 The Panel concluded, based on the structures of each polyether lanolin reviewed, that none of these ingredients were likely to be mutagenic or carcinogenic.
The Panel discussed the issue of incidental inhalation exposure from colognes and toilet waters, powders, and body and hand sprays. The limited data available from 1 short-term exposure study on PEG-27 lanolin at 200 mg/L (well above any use concentration in this report) suggest little potential for respiratory effects at relevant doses. These products are used up to 2% in products that may be aerosolized and up to 4% in products that may become airborne. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Furthermore, these ingredients are not likely to cause any direct toxic effects in the upper respiratory tract, based on the properties of the polyether lanolins and on data that show that these ingredients are not irritants. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for polyether lanolins to cause systemic toxicity, irritation, sensitization, or other effects. They noted the lack of systemic toxicity at high doses in several acute and repeated dose dermal and oral exposure studies. There was little or no irritation or sensitization in multiple tests of dermal and ocular exposure, as was the absence of genotoxicity in multiple tests. A detailed discussion of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Panel concluded that the following ingredients are safe in the present practices of use and concentration described in this safety assessment: PPG-5 lanolin wax PPG-5 lanolin wax glyceride* PEG-75 lanolin wax* PEG-5 hydrogenated lanolin* PEG-10 hydrogenated lanolin* PEG-15 hydrogenated lanolin* PEG-20 hydrogenated lanolin PEG-24 hydrogenated lanolin PEG-30 hydrogenated lanolin* PEG-40 hydrogenated lanolin* PEG-70 hydrogenated lanolin* PEG-5 lanolin PEG-10 lanolin* PEG-20 lanolin* PEG-24 lanolin* PEG-27 lanolin* PEG-25 lanolin* PEG-30 lanolin PEG-35 lanolin* PEG-40 lanolin PEG-50 lanolin PEG-55 lanolin* PEG-60 lanolin PEG-70 lanolin* PEG-75 lanolin PEG-85 lanolin PEG-100 lanolin PEG-150 lanolin* PEG-75 lanolin oil Polyglyceryl-2 lanolin alcohol ether* PPG-2 lanolin alcohol ether* PPG-5 lanolin alcohol ether* PPG-10 lanolin alcohol ether* PPG-20 lanolin alcohol ether* PPG-30 lanolin alcohol ether* PPG-20-PEG-20 hydrogenated lanolin* PPG-12-PEG-50 lanolin PPG-12-PEG-65 lanolin oil PPG-40-PEG-60 lanolin oil*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Lillian C. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Bart Heldreth contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Heldreth, B. and Fiume, M. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Jr, Ronald C. Shank, Thomas J. Slaga, and Paul W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Unpublished sources cited in this report are available from the Executive Director Bart Heldreth, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The infrastructure of Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.