Abstract
Repeated developmental exposure to the organophosphate (OP) insecticide chlorpyrifos (CPF) inhibits brain fatty acid amide hydrolase (FAAH) activity at low levels, whereas at higher levels, it inhibits brain monoacylglycerol lipase (MAGL) activity. FAAH and MAGL hydrolyze the endocannabinoids anandamide (AEA) and 2-arachidonylglycerol (2-AG), respectively. Peripherally, AEA and 2-AG have physiological roles in the regulation of lipid metabolism and immune function, and altering the normal levels of these lipid mediators can negatively affect these processes. Exposure to CPF alters brain endocannabinoid hydrolysis activity, but it is unclear whether low-level exposure alters this activity in peripheral tissues important in metabolic and immune function. Therefore, rat pups were exposed orally from day 10 to 16 to 0.5, 0.75, or 1.0 mg/kg CPF or 0.02 mg/kg PF-04457845 (a specific FAAH inhibitor). At 12 hours postexposure, FAAH, MAGL, and cholinesterase (ChE) activities were determined. All treatments inhibited FAAH activity in brain, spleen, and liver. CPF inhibited ChE activity in spleen and liver (all dosages) and in brain (highest dosage only). CPF inhibited total 2-AG hydrolysis and MAGL-specific activity in brain and spleen (high dosage only). In liver, total 2-AG hydrolysis was inhibited by all treatments and could be attributed to inhibition of non–MAGL-mediated 2-AG hydrolysis, indicating involvement of other enzymes. MAGL-specific activity in liver was inhibited only by the high CPF dosage, whereas PF-04457845 slightly increased this activity. Overall, exposure to low levels of CPF and to PF-04457845 can alter endocannabinoid metabolism in peripheral tissues, thus potentially affecting physiological processes.
Introduction
Chlorpyrifos (CPF) continues to be one of the most commonly utilized organophosphate (OP) insecticide, despite continuing concern about the dangers of developmental toxicity in children. While the restricted residential usage of CPF has caused a significant decrease in the potential for exposure, children who reside within agricultural communities are still at significant risk. 1 Childhood exposure to CPF has been associated with decreased motor skills and cognitive ability (attention deficit/hyperactivity disorder), decreased IQ and working memory, and altered brain morphology. 2 –4 Following cytochrome P450-mediated bioactivation to its active metabolite chlorpyrifos-oxon, CPF exerts its effects by inhibiting cholinesterase (ChE) activity in the cholinergic system, which is thought to be the cause of the toxic effects associated with developmental exposure. However, even at dosages that yield minimal levels of ChE inhibition, the long-term effects of CPF are still present, suggesting that the cholinergic system is not the only important toxicological target.
The endocannabinoid system has attracted attention because of its susceptibility to inhibition by the OPs and its vital role in brain development. Earlier work demonstrated that many OP insecticides, including chlorpyrifos-oxon, the active metabolite of CPF, are potent in vitro inhibitors of the endocannabinoid-metabolizing enzymes fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL). 5,6 Acute in vivo exposure of adult mice to high levels of CPF resulted in the inhibition of ChE, FAAH, and MAGL. 5 –7 The inhibition of FAAH and MAGL led to increased levels of their respective substrates, the endocannabinoids anandamide (AEA) and 2-arachidonoylglycerol (2-AG), in the brain. 8,9 In developing animals, we have demonstrated that repeated exposure to very low levels of CPF results in the inhibition of FAAH in the brain without inhibition of ChE or MAGL. 10 At higher dosages, inhibition of both ChE and MAGL was noted. 11 Consequently, this inhibition of FAAH and MAGL leads to the accumulation of AEA and 2-AG. 10 –12 In addition to neurological processes, the metabolism of endocannabinoids is also important for regulating immune function 13 –15 and lipid metabolism in peripheral tissues. 16 –18 Therefore, inhibition of endocannabinoid catabolism in peripheral tissues during low-level CPF exposure could potentially deregulate these important functions.
The objective of this study was to determine whether developmental exposure to low-level CPF affects the activities of the endocannabinoid-metabolizing enzymes in peripheral tissues of juvenile rats. Because low levels of CPF can target FAAH, the effect of the specific FAAH inhibitor PF-04457845 in rats was also determined. The tissues selected for study include the spleen and liver because of their respective roles in immune function and metabolic state. The activities of the endocannabinoid-metabolizing enzymes, FAAH and MAGL, were determined as well as the activity of ChE.
Materials and Methods
Chemicals
Chlorpyrifos (>99%) was a generous gift from DowElanco Chemical Company (Indianapolis, Indiana). PF-04457845 was purchased from MedChem Express (Monmouth Junction, New Jersey). All other chemicals were purchased from Cayman Chemicals (Ann Arbor, Michigan) or Sigma Chemical Co. (St Louis, Missouri).
Animals
Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited facility in a temperature-controlled environment (22°C ± 2°C) with a 12-hour dark–light cycle with lights on between 0700 and 1900. LabDiet rodent chow and tap water were freely available during experimentation. All procedures were approved by the Mississippi State University Institutional Animal Care and Use Committee. Adult male and female Sprague Dawley rats (CD IGS; Harlan Laboratories, Indianapolis, Indiana, USA) were utilized for breeding. The day of birth was considered as postnatal day 0 (PND 0). Within each litter, male and female rat pups were assigned to different treatment groups. There were always representative control animals of the same sex present in each litter to match the CPF-treated animals.
Animal Treatment
Beginning on PND10, rat pups were exposed daily by oral gavage (per os) at a volume of 0.5 mL/kg body weight for 7 days. Chlorpyrifos and PF-04457845 were dissolved in corn oil and delivered to the back of the throat using a 25-μL tuberculin syringe equipped with a 1-inch, 24-gauge straight intubation needle (Popper and Sons, Inc., New Hyde Park, New York, USA). Daily body weights were recorded, and weight gain was calculated as the difference between the body weights on PND11-16 and the original body weight at initiation of treatment on PND10. The time frame of exposure, PND10 through PND16, corresponds to the postnatal stage during which significant brain maturation occurs in humans. 19 –21
The CPF treatment groups selected for study were control (corn oil), low CPF (0.5 mg/kg), medium CPF (0.75 mg/kg), and high CPF (1.0 mg/kg). These dosages span the range between no inhibition of brain ChE and low inhibition of brain ChE. 10 –12 These dosages are also below the oral repeated no-observed-effect level (NOEL) for signs of toxicity (4.5 mg/kg) for postnatal rats but span the oral repeated NOEL for inhibition of brain ChE activity (0.75 mg/kg) as reported by Zheng et al. 22 However, it is difficult to extrapolate these dosages to environmental exposure levels in children. The dosage of PF-04457845 utilized was 0.02 mg/kg. This dosage was designed to induce significant inhibition of FAAH without completely eliminating its activity which is the pattern of inhibition similar to CPF.
Rat pups were killed at 12 hours after the last exposure to CPF. Brains were rapidly removed and dissected on ice to obtain the forebrain (entire brain excluding the medulla and cerebellum). The forebrain was immediately frozen on a stainless steel plate on top of dry ice and maintained at −80°C until assay. The forebrain was the region utilized to determine the level of inhibition of endocannabinoid-metabolizing enzymes in our previous studies. 10 –12,23
Enzyme Assays
Forebrain, spleen, and liver were homogenized in cold 0.05 M Tris-HCl buffer containing 0.2 mM EDTA (pH 7.4 at 37°C) in a glass mortar using a Wheaton motorized tissue grinder and a Teflon pestle. For determination of forebrain, spleen, and liver ChE activity, an aliquot of homogenate was diluted in cold 0.05 mol/L Tris-HCl buffer (pH 7.4 at 37°C) to a final tissue concentration of 1.0, 1.25, and 2.5 mg/mL, respectively. The ChE activity was measured spectrophotometrically using a modification 24 of Ellman et al 25 with acetylthiocholine as the substrate (1 mmol/L final concentration) and 5,5-dithiobis(nitrobenzoic acid) as the chromogen.
AEA and 2-AG hydrolysis activity were measured as described previously 12,23 in forebrain, spleen, and liver homogenates at final tissue concentrations of 2.667, 2, and 1 mg/mL, respectively. The amount of arachidonic acid formed by the hydrolysis of exogenous 2-AG and AEA was determined by LC-MS as described previously, 26 correcting for endogenous arachidonic acid levels in the tissue homogenates. For determination of MAGL and FAAH activity, corresponding tubes containing either 10 μmol/L of the specific MAGL inhibitor JZL184 or of the specific FAAH inhibitor URB597, respectively, were included during the 10-minute preincubation period prior to adding the endocannabinoid substrate.
Protein concentrations were quantified with the Folin phenol reagent using bovine serum albumin as a standard. 27 Specific activities were calculated as nmoles (ChE) or pmoles (FAAH and MAGL) of product produced in min−1 mg protein−1.
Statistical Analysis
Statistical analysis was performed using SAS statistical package (SAS Institute Inc, Cary, North Carolina). The sphericity of the body weight gain data was initially tested by analysis of variance (ANOVA) using the general linear model with a repeated measures paradigm and was found to violate the assumption of sphericity. Therefore, subsequent analysis by ANOVA using the mixed procedure 28 was conducted with a repeat-measures paradigm with a Huynh-Feldt covariance structure, 29 followed by separation of means using least significant difference. The analysis identified significant differences in the main effects (sex, treatment, and day) and all possible interactions. Enzyme-specific activities were analyzed by ANOVA using the mixed procedure to determine the significant differences in sex, treatment, and Sex × Treatment interactions. All analyses included litter and Sex × Treatment × Litter as random effects. Mean separation was performed by least squared means. There was no significant overall effect of sex and no significant Sex × Treatment interaction with any parameter. Therefore, male and female data were combined for analysis.
Results
There was no overall effect of treatment or any significant related interaction on weight gain during developmental exposure to CPF or the PF-04457845 (Figure 1). There was a significant overall effect of day (
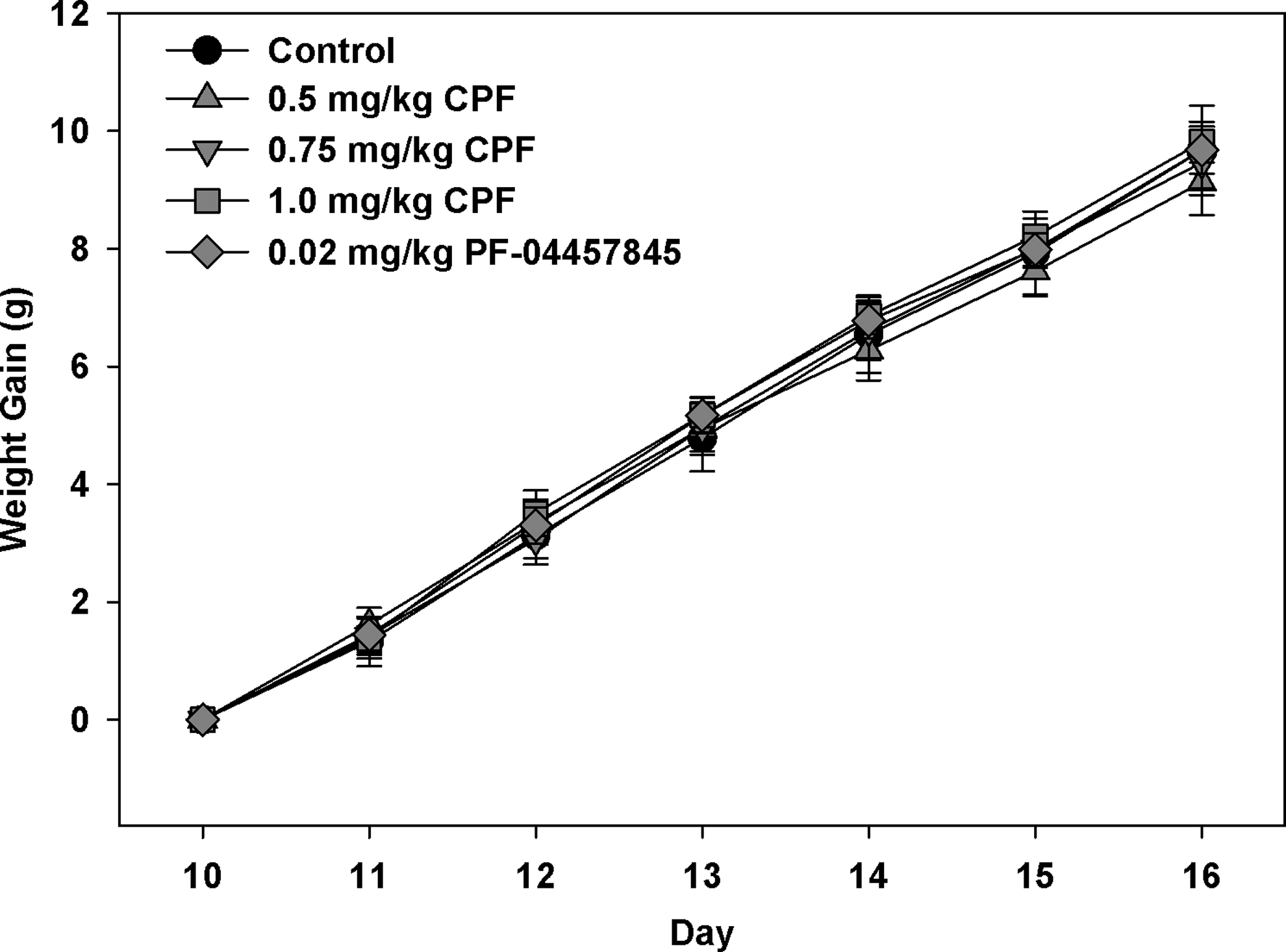
Rates of weight gain of rat pups exposed daily from postnatal day 10 through day 16 to either corn oil (control), 0.5, 0.75, or 1.0 mg/kg chlorpyrifos (CPF) or 0.02 mg/kg PF-04457845. Values are expressed as weight ± standard error (SE; n = 8-12).
With respect to ChE activity, there was a significant effect of treatment (
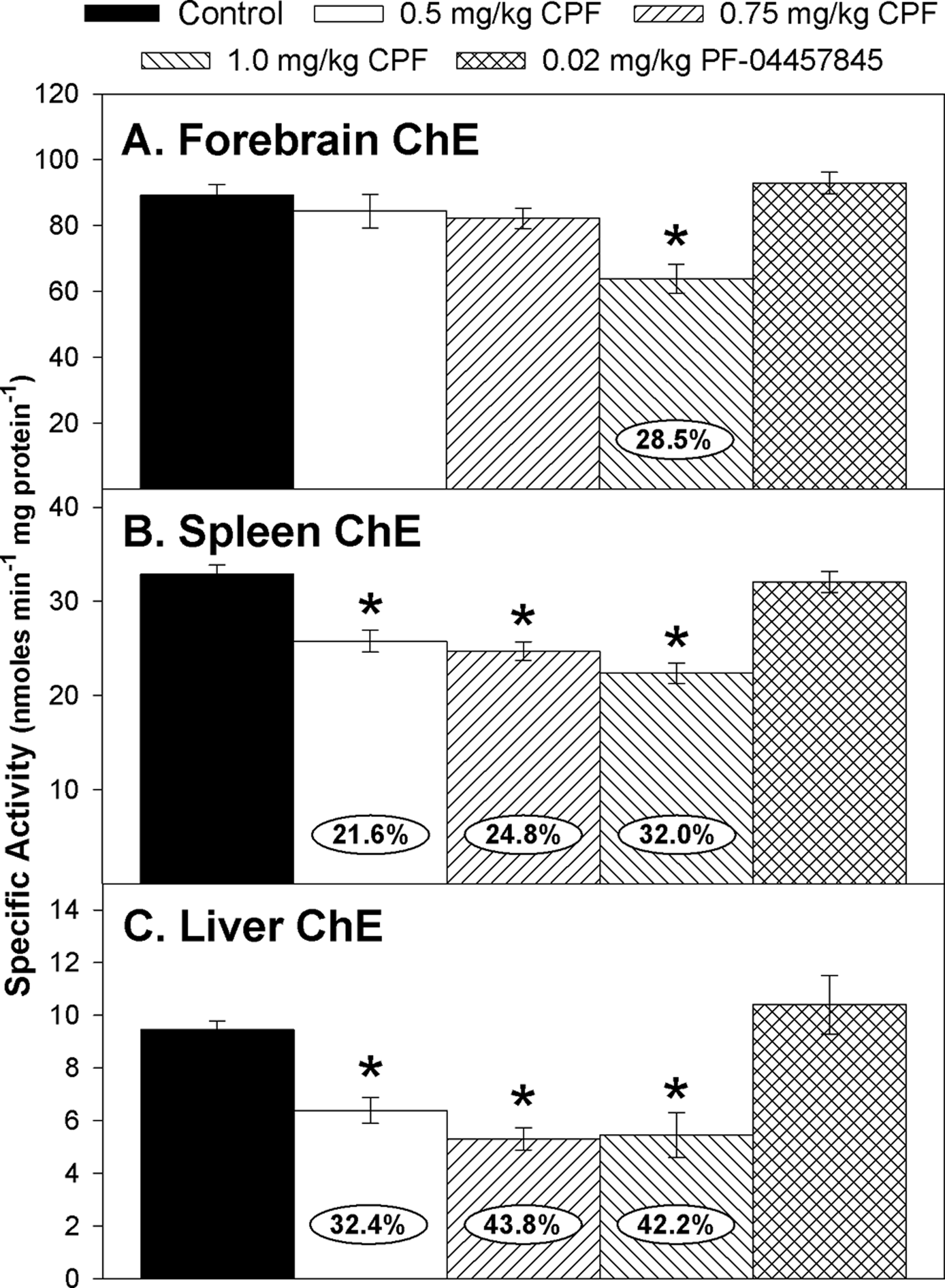
Specific activity of cholinesterase (ChE) in the (A) forebrain, (B) spleen, and (C) liver of rat pups at 12 h after last administration following daily exposure from postnatal day 10 through 16 to either corn oil (control) or 0.5, 0.75, or 1.0 mg/kg chlorpyrifos (CPF) or 0.02 mg/kg PF-04457845. Values are expressed as nmol product min−1 mg protein−1 ± standard error (SE; n = 8-12). Percentage of inhibition for each treatment group as compared to its respective control is presented in the oval overlaying the corresponding bar. Statistical differences from control values are indicated by asterisks above the corresponding bar (*
With respect to FAAH activity in the different tissues, there was a dose-dependent inhibition in forebrain (Figure 3A), spleen (Figure 3B), and liver (Figure 3C). There was a significant effect of treatment (
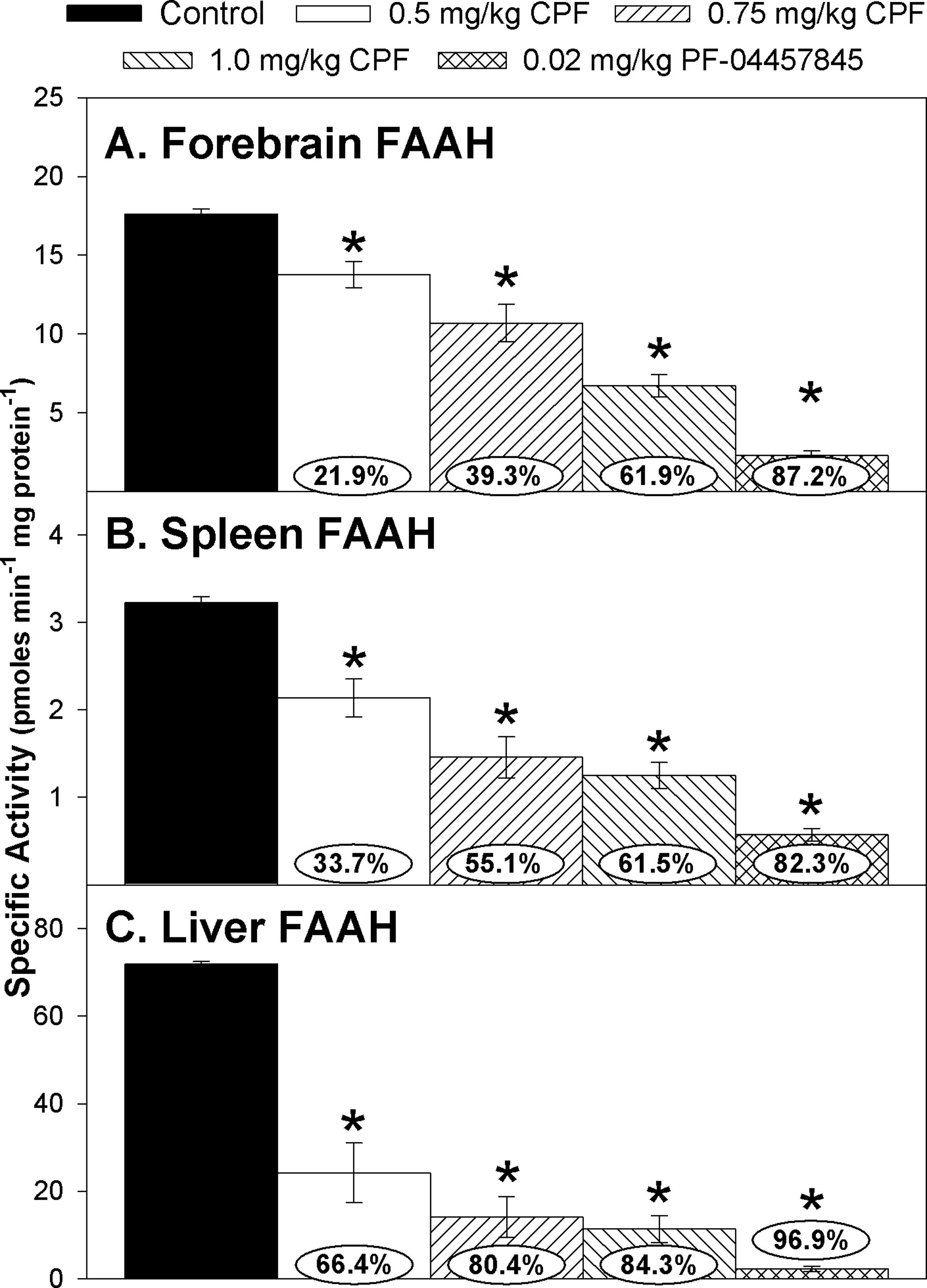
Specific activity of fatty acid amide hydrolase (FAAH) in the (A) forebrain, (B) spleen, and (C) liver of rat pups at 12 hours after last administration following daily exposure from postnatal day 10 through 16 to either corn oil (control) or 0.5, 0.75, or 1.0 mg/kg chlorpyrifos (CPF) or 0.02 mg/kg PF-04457845. Values are expressed as nmol product min−1 mg protein−1 ± standard error (SE; n = 8-12). Percentage of inhibition for each treatment group as compared to its respective control is presented in the oval overlaying the corresponding bar. Statistical differences from control values are indicated by asterisks above the corresponding bar (*
To estimate the contribution of MAGL to the overall 2-AG hydrolysis activity in each tissue, we subtracted the 2-AG hydrolysis activity values obtained in tissue homogenates preincubated with JZL-184 from total 2-AG hydrolysis activity values obtained in tissue homogenates without JZL-184. Therefore, the data are presented in Figure 4 as follows: (1) total 2-AG hydrolysis (defined as activity obtained without any correction using JZL-184) (left side), (2) non-MAGL hydrolysis (defined as activity that is insensitive to inhibition with JZL-184 (middle), and (3) MAGL hydrolysis (right side). With respect to the forebrain (Figure 4A), there was no significant overall effect of treatment on total, non-MAGL, or MAGL-specific 2-AG hydrolysis. However, lower level statistical analysis of the 1.0 mg/kg CPF treatment group suggested a trend toward a significant decrease from control levels for total 2-AG hydrolysis (
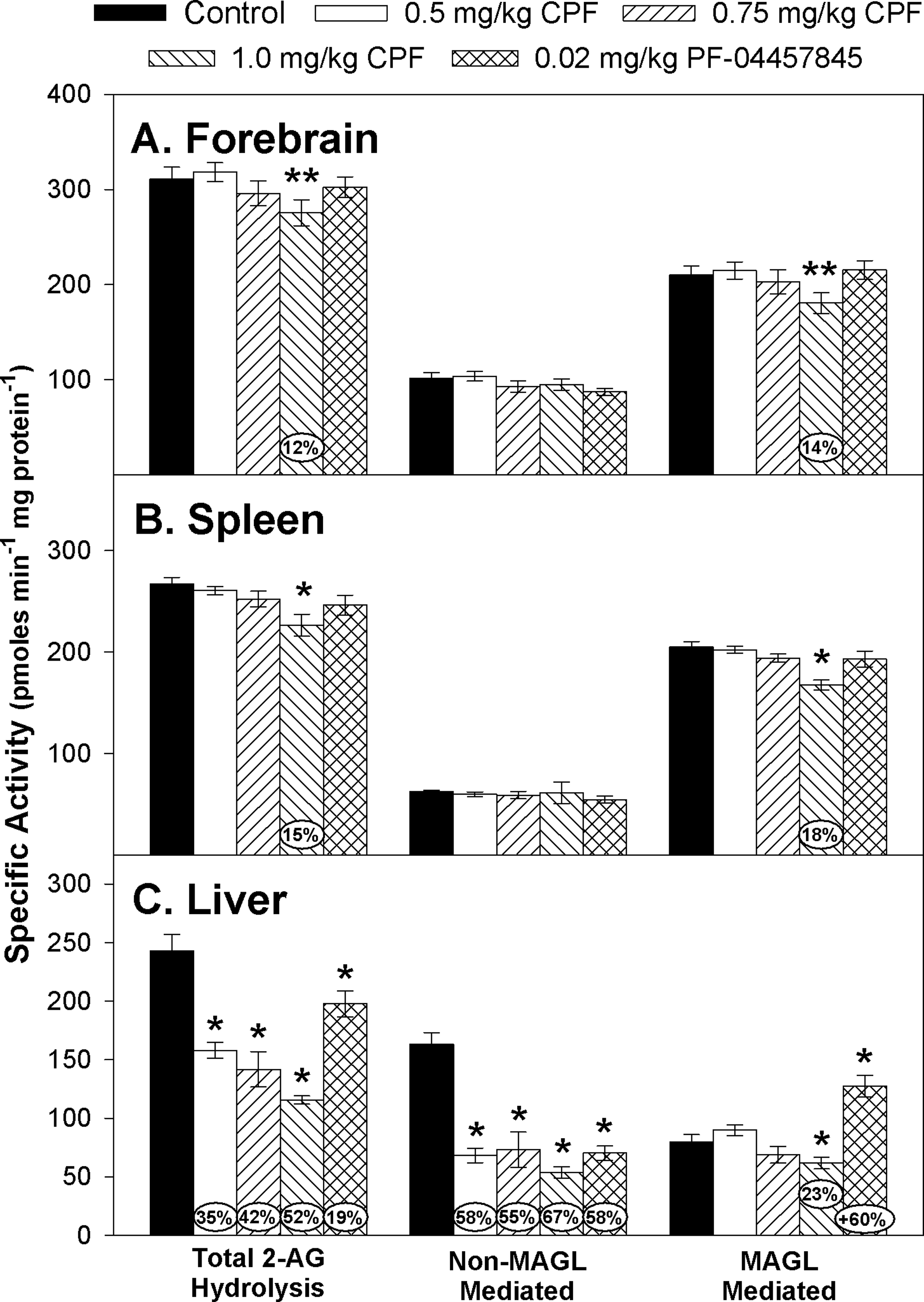
Specific activity of total 2-AG hydrolysis, non–monoacylglycerol lipase (MAGL)-mediated 2-AG hydrolysis, and MAGL-mediated 2-AG hydrolysis in the (A) forebrain, (B) spleen, and (C) liver of rat pups at 12 h after last administration following daily exposure from postnatal day 10 through 16 to either corn oil (control) or 0.5, 0.75, or 1.0 mg/kg chlorpyrifos (CPF) or 0.02 mg/kg PF-04457845. Values are expressed as nmol product min−1 mg protein−1 ± standard error (SE; n = 8-12). Percent inhibition for each treatment group as compared to its respective control is presented in the oval overlaying the corresponding bar. Statistical differences from control values are indicated by asterisks above the corresponding bar (*
Discussion
The levels of inhibition observed in forebrain FAAH and ChE induced by the different CPF dosages were similar to our previous observations. 10 –12 As we have previously reported, the pattern of sensitivity to CPF-dependent inhibition from highest to lowest level in forebrain was FAAH activity > ChE activity > MAGL activity. 23 For forebrain MAGL, we did not observe a clear statistically significant decrease. The level of MAGL inhibition (∼14%) at 1.0 mg/kg CPF was similar to what we have observed previously, although it appears to be at the threshold of statistical significance because we previously reported it to be significant in the study of Carr et al (2017) 11 but not significant in the study of Carr et al (2013). 12
Our data also indicate the presence of FAAH activity in juvenile rat spleen homogenates. The fact that PF-04457845, a specific inhibitor of FAAH, markedly inhibited spleen AEA hydrolysis activity strongly supports this. This is also in agreement with previous in vitro work where it was reported that anandamide hydrolysis occurs in adult mouse splenocytes. 30 However, the FAAH activity detected in juvenile rat spleen is in conflict with the finding of others where FAAH activity was limited in adult mouse spleen. 31 It is possible that there may be some species or ontological differences between adults and juveniles, or a combination of the two.
Of the three enzymes measured in spleen, FAAH activity was the most sensitive to inhibition following CPF exposure (up to 62% inhibition following the highest dosage of CPF). At this level of reduced FAAH activity in spleen, it is likely that AEA accumulates. Previous studies have reported that administration of another specific inhibitor of FAAH, URB597, to adult rats results in elevated levels of AEA, palmitoylethanolamide (PEA), and oleoylethanolamide (OEA) in the spleen and hypothalamus. 32 PEA and OEA are also known substrates for FAAH. We have previously reported that CPF exposure also caused the elevation in these lipid mediators in the forebrain. 11 It is not clear, however, if similar increases in AEA, OEA, and PEA are induced in peripheral tissues following low-level CPF exposure. In addition, it was reported that the administration of the specific inhibitor of MAGL, JZL-184 to adult rats, inhibited MAGL activity in the spleen by ∼50%, resulting in a doubling of 2-AG levels. 33 Thus, the degree of inhibition of MAGL activity (18%) in spleen caused by the highest dosage of CPF could possibly lead to increased 2-AG levels as well. Future studies will determine the levels of 2-AG, AEA, OEA, and PEA in the spleen following repeated developmental CPF exposure.
Multiple reports describe the importance of the endocannabinoid system in immune function and inflammation. A significant number of these studies indicate that endocannabinoids elicit anti-inflammatory effects. 13,32,34 –37 Thus, increasing the levels of the endocannabinoids could theoretically be beneficial due to their ability to attenuate the negative effects of inflammation. However, if an appropriate inflammatory response is required to combat a pathogen, inappropriate increases, even small increases, in the levels of endocannabinoids and other related mediators induced by CPF exposure might be detrimental. For example, the potential immunosuppressive effects of small increases in endocannabinoid levels in immunocompromised individuals have been identified as a concern. 38 It has also been demonstrated that exposure to CPF and other OPs can induce immunosuppressive effects in both humans 39,40 and animal models. 41 –44 This includes altered immune function following developmental exposure to CPF. 45 However, the mechanism by which this OP-induced immunosuppression occurs is not clear.
The enzymes in the liver were inhibited by CPF to a greater extent than the other tissues. This is not surprising because the oral route of exposure was utilized and the liver would be the first opportunity for metabolic activation. This was most evident for FAAH activity, which was inhibited by 66% at the lowest dosage of CPF and greater than 80% at the two higher CPF dosages, and it was virtually eliminated by the specific inhibitor of FAAH. It is quite possible that inhibition of FAAH activity at these levels could result in the elevation of AEA as well as other known substrates for FAAH such as PEA and OEA. As with the spleen, future studies will quantify the levels of these compounds in the liver following repeated developmental CPF exposure.
With respect to the hydrolysis of 2-AG in the livers of control juveniles, total 2-AG hydrolysis specific activity (pmol min−1 mg protein−1) was 242.9 ± 14.4, whereas the activity of 2-AG hydrolysis in the presence of JZL-184 was 163.3 ± 9.9. This indicates that only ∼33% of the total 2-AG hydrolysis activity in control juvenile rat liver is sensitive to inhibition by JZL-184 and can be designated as MAGL activity. The remaining 67% of 2-AG hydrolysis activity was not sensitive to JZL-184 and can be designated as non–MAGL-mediated activity (Figure 4C). In adult mice, 85% and 80% of the 2-AG hydrolysis activity in the brain and liver, respectively, is attributed to MAGL. 46 –48 However, we previously observed that in late preweanling rat brain, only 45% of the 2-AG hydrolysis activity could be attributed to MAGL. 23 Thus, it is possible that age-related differences could exist in the liver as well.
Considering total 2-AG hydrolysis in the liver of CPF-treated animals, CPF inhibited total 2-AG hydrolysis activity in a dose-dependent manner (Figure 4C). Because only ∼33% of the 2-AG hydrolysis is due to the activity of MAGL, this suggests that a significant portion of the total 2-AG hydrolysis activity in liver that is inhibited following CPF treatment is due to the inhibition of other 2-AG hydrolases. In addition, our data indicate that in vivo treatment with the specific FAAH inhibitor PF-04457845 could inhibit total 2-AG hydrolysis to a small extent (∼19%). This reduction is due to the inhibition of the non-MAGL-mediated hydrolysis of 2-AG. In fact, the level of inhibition of non-MAGL-mediated 2-AG hydrolysis (Figure 4C) was similar in all the CPF treatment groups and PF-04457845 treatment group. At this point, it is unclear what enzyme(s) account for the remaining non-MAGL-mediated 2-AG hydrolysis activity. Potential candidate(s) are the carboxylesterases, which are sensitive to inhibition by OPs and have been shown to hydrolyze 2-AG in vitro. 26 However, PF-04457845 has not been reported to cross-react with carboxylesterases suggesting a role for FAAH. FAAH was reported to also degrade 2-AG in vitro. 46 Therefore, it is possible that the inhibition of carboxylesterases and FAAH contributed to the disruption of 2-AG metabolism in liver.
The increase in the MAGL-specific hydrolysis in the PF-04457845 treatment group is puzzling. It is possible that the repeated exposure to PF-04457845 resulted in the induction of MAGL activity. Treatment of adult mice to the peroxisome proliferator-activated receptor α (PPAR α) agonist Wy14643 resulted in increased MAGL messenger RNA levels, 49 suggesting that MAGL activity may have been increased as well (it was not measured in this particular study). PEA and OEA are substrates for FAAH and are PPAR α agonists. 50,51 Inhibition of FAAH results in the accumulation of OEA and PEA in the liver, 52 and it is possible that the accumulation of these lipids stimulated the PPAR α resulting in increased MAGL activity. This hypothesis will require more investigation.
The importance of the endocannabinoid system on the regulation of energy homeostasis is becoming increasingly important for understanding obesity and type 2 diabetes. 16,53 –55 Environmental chemicals that disrupt endocannabinoid system function have the potential to exert a role in these diseases. For example, it has been suggested that there is a connection between developmental exposure to OP insecticides and metabolic dysfunction. 56 –60 The mechanism by which this occurs is not yet clear; however, the disruption of endocannabinoid function by OPs could play an important role.
Footnotes
Author Contributions
R. Buntyn contributed to acquisition and analysis and drafted the manuscript; N. Alugubelly contributed to acquisition and interpretation and critically revised the manuscript; R. Hybart contributed to acquisition and drafted the manuscript; A. Mohammed contributed to acquisition and interpretation and critically revised the manuscript; G. Parker contributed to acquisition and critically revised the manuscript; C. Nail contributed to acquisition and interpretation and critically revised the manuscript; M. Ross contributed to conception and drafted and critically revised the manuscript; R. Carr contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research was supported by the Mississippi Agricultural and Forestry Experiment Station (MAFES), the College of Veterinary Medicine, Mississippi State University, by NIH R15ES023162 and NIH R15ES015348-02. This article is Center for Environmental Health Sciences publication #165.
