Abstract

Conclusion
In a 1998 safety assessment, the Cosmetic Ingredient Review Expert Panel (Panel) concluded that glycolic and lactic acid, their common salts, and their simple esters (referred to as alpha hydroxy acids [AHA] ingredients) are safe for use in cosmetic products at concentrations ≤10%, at final formulation pH ≥3.5, when formulated to avoid increasing sun sensitivity or when directions for use include the daily use of sun protection. These ingredients are safe for use in salon products at concentrations ≤30%, at final formulation pH ≥3.0, in products designed for brief discontinuous use followed by thorough rinsing from the skin, when applied by trained professionals, and when application is accompanied by directions for the daily use of sun protection. 1
The Panel reviewed newly available studies since that assessment, along with updated information regarding types and concentrations of use (Tables 1 and 2). 2 –41 The Panel determined to not reopen this safety assessment. Therefore, the Panel confirmed the original conclusion as stated above.
Current and Historical Frequency and Concentration of Use of AHAs According to Duration and Exposure.
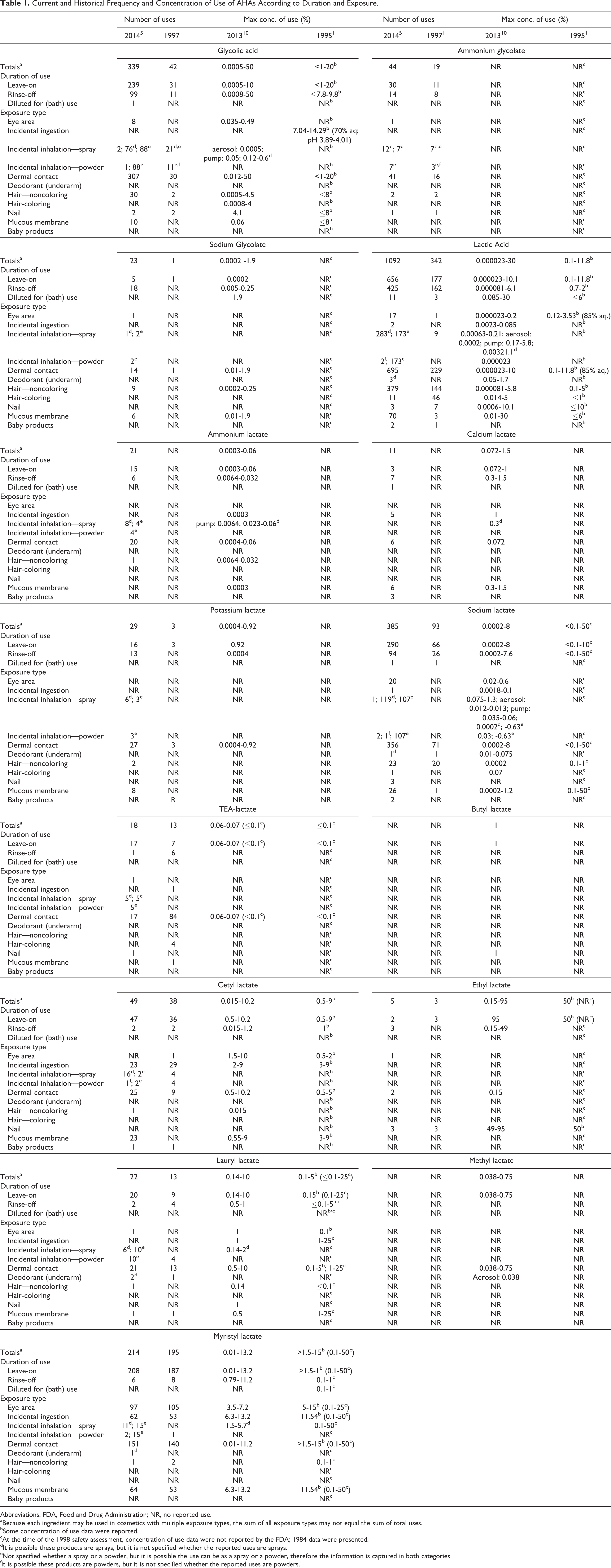
Abbreviations: FDA, Food and Drug Administration; NR, no reported use.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bSome concentration of use data were reported.
cAt the time of the 1998 safety assessment, concentration of use data were not reported by the FDA; 1984 data were presented.
dIt is possible these products are sprays, but it is not specified whether the reported uses are sprays.
eNot specified whether a spray or a powder, but it is possible the use can be as a spray or a powder, therefore the information is captured in both categories
fIt is possible these products are powders, but it is not specified whether the reported uses are powders.
AHAs Not in Current Use According to VCRP and Council Survey Data.
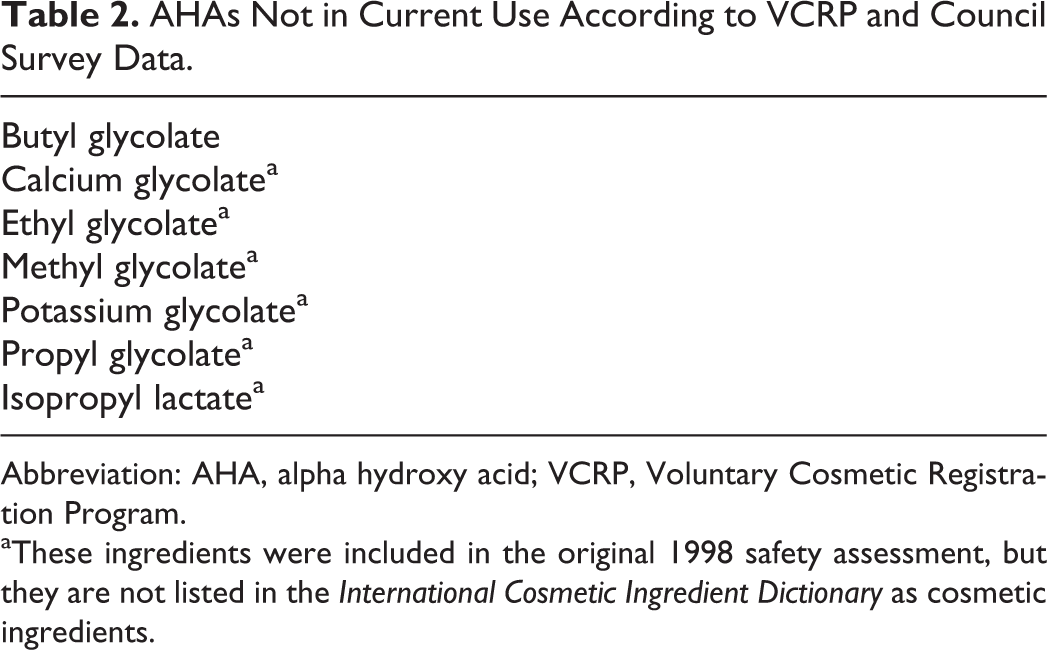
Abbreviation: AHA, alpha hydroxy acid; VCRP, Voluntary Cosmetic Registration Program.
aThese ingredients were included in the original 1998 safety assessment, but they are not listed in the International Cosmetic Ingredient Dictionary as cosmetic ingredients.
Discussion
The use of AHAs has increased considerably since the original assessment. Glycolic acid had been used in 42 cosmetic formulations in 1997, and lactic acid was reported to be used in 342 cosmetic formulations. In 2014, the US Food and Drug Administration (FDA) reported that glycolic acid is used in 339 formulations and lactic acid is used in 1092 cosmetic formulations. A survey of current use concentrations conducted by industry reported that leave-on use concentrations of glycolic and lactic acid are similar to those reported in the 1998 assessment; however, the highest maximum use concentrations in rinse-off products have increased. 10
The Panel acknowledged the FDA’s “Guidance for Industry: Labeling for Cosmetics Containing Alpha Hydroxy Acids” that was issued in 2005, which also addressed the use of sun protection with AHA products. The FDA recommended that the labeling of a cosmetic product that contains an AHA ingredient and that is topically applied to the skin or mucous membrane bear a statement, prominently and conspicuously placed on the cosmetic product, which conveys the following information: Sunburn Alert: This product contains an AHA that may increase your skin’s sensitivity to the sun and particularly the possibility of sunburn. Use a sunscreen, wear protective clothing, and limit sun exposure while using this product and for a week afterwards.
Although AHA ingredients are in products for consumer, salon, and medical use, the Panel stated that this safety assessment does not address the medical use of AHA ingredients; it addresses only the consumer and salon use, that is, those products available to the general public and those applied by trained estheticians, respectively.
Finally, the Panel reviewed the photocarcinogenicity studies that have been published since the original safety assessment. 8,26 In these studies, the dermal application of glycolic acid to mouse skin did not increase the incidence of skin tumors in mice. The Panel stated these studies provided additional evidence to confirm the safety of AHAs for use in cosmetic formulations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
