Abstract

Conclusion
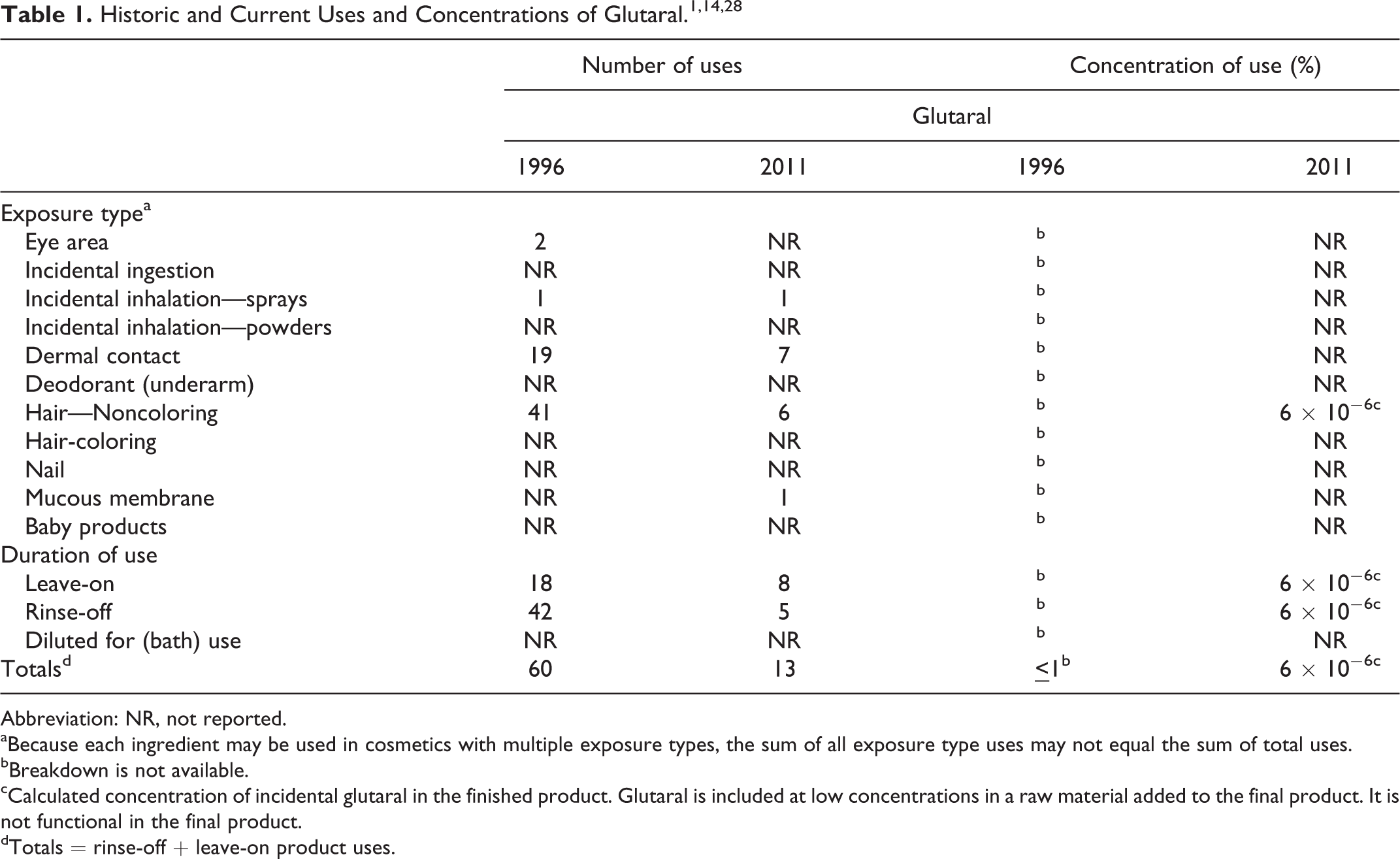
In the 1996 safety assessment of glutaral, the Cosmetic Ingredient Review Expert Panel (Panel) stated that this ingredient is safe for use at concentrations up to 0.5% in rinse-off products. There were insufficient data to determine the safety of glutaral in leave-on products, and this ingredient should not be used in aerosolized products. 1 The Panel reviewed newly available studies since that assessment along with updated frequency and concentration of use information (Table 1). 2 –46 The Panel determined to not reopen this safety assessment and confirmed the original conclusion of glutaral.
Abbreviation: NR, not reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum of total uses.
bBreakdown is not available.
cCalculated concentration of incidental glutaral in the finished product. Glutaral is included at low concentrations in a raw material added to the final product. It is not functional in the final product.
dTotals = rinse-off + leave-on product uses.
Discussion
Since the original conclusion, numerous studies have been published, including a 2-year National Toxicology Program (NTP) study on inhalation. Although the number of uses for glutaral has decreased from 60 to 13, this ingredient is currently being used in an aerosol product and in leave-on products. The current concentration of use is 6 × 10−6% in noncoloring hair products. The Panel received clarification that this concentration of glutaral is incidental and that glutaral is not added to the products for functional use. Additionally, although glutaral did not cause cancerous lesions in the 2-year NTP study, several studies have found that this ingredient does cause damage to the upper respiratory tract in animals. 6,16,25,38,45,46
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.