Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel assessed the safety of cross-linked alkyl acrylates as used in cosmetics. The 23 cross-linked alkyl acrylates included in this safety assessment are reported to function as absorbents, film formers, emulsion stabilizers, viscosity increasing agents, suspending agents, binders, and/or skin-conditioning agents. The Panel reviewed available animal and clinical data, as well as information from previous CIR reports on monomer components. Because data were not available for the individual ingredients, and because residual monomer may be present, the Panel extrapolated from previous reports to support safety. The Panel concluded that cross-linked alkyl acrylates are safe in the present practices of use and concentration, provided that they are not polymerized in benzene. For those ingredients polymerized in benzene, the data available were insufficient to make a determination of safety. A risk assessment for the amount of benzene present would be needed.
Introduction
This draft final report includes information relevant to the safety of 23 cross-linked alkyl acrylates as used in cosmetic formulations. These cross-linked polymers consist of comonomers of at least 1 of the following: acrylic acid, sodium acrylate, methacrylic acid, or alkyl acrylate that share chemical properties, including a general lack of chemical reactivity. The ingredients included in this group are: Acrylates/C10-30 alkyl acrylate cross polymer Acrylates/C12-13 alkyl methacrylates/methoxyethyl acrylate cross polymer Acrylates cross polymer Acrylates/ethylhexyl acrylate cross polymer Acrylates/ethylhexyl acrylate/glycidyl methacrylate cross polymer Acrylates/PEG-4 dimethacrylate cross polymer Acrylates/Steareth-20 methacrylate cross polymer Acrylates/vinyl isodecanoate cross polymer Acrylates/vinyl neodecanoate cross polymer Allyl methacrylate/glycol dimethacrylate cross polymer Allyl methacrylates cross polymer Butyl acrylate/glycol dimethacrylate cross polymer C8-22 alkyl acrylates/methacrylic acid cross polymer Glycol dimethacrylate/vinyl alcohol cross polymer Lauryl methacrylate/glycol dimethacrylate cross polymerr Lauryl methacrylate/sodium methacrylate cross polymer Methacrylic acid/PEG-6 methacrylate/PEG-6 dimethacrylate cross polymer PEG/PPG-5/2 methacrylate/methacrylic acid cross polymer Potassium acrylates/C10-30 alkyl acrylate cross polymer Sodium acrylates cross polymer 2 Sodium acrylates/C10-30 alkyl acrylate cross polymer Sodium acrylates/vinyl isodecanoate cross polymer Stearyl/lauryl methacrylate cross polymer
These ingredients are reported to function in cosmetics as absorbents, film formers, emulsion stabilizers, viscosity increasing agents, suspending agents, binders, or skin-conditioning agents.
In 2002, the Cosmetic Ingredient Review (CIR) published the Final Report on the Safety Assessment of Acrylates Copolymer and 33 Related Cosmetic Ingredients. 1 The Panel concluded that those ingredients were safe for use in cosmetics when formulated to avoid skin irritation. While copolymers are polymers synthesized from 2 or more different monomers, cross polymers are polymers that are cross-linked (ie, individual polymer chains are connected by bridging molecules [cross-linking agents]). Cross-linked polymers are generally less chemically reactive and less soluble (if not totally insoluble) than their respective non-cross-linked counterparts.
A CIR report on another family of polymers is also available. In 1982, the CIR published the Final Report on the Safety Assessment of Carbomers-934, -910, -934P, 940, -941, and -962, in which it was concluded that carbomers are safe as used. 2 That conclusion was reaffirmed in 2003. 3 A carbomer is a homopolymer of acrylic acid cross-linked with an allyl ether of pentaerythritol, an allyl ether of sucrose, or an allyl ether of propylene. 4
Due to the paucity of published safety and toxicity data on these ingredients, this report includes summary information included in technical data sheets, ingredient specification sheets, and material safety data sheets (MSDSs); this information is identified as such.
Chemistry
Definition and Structure
Cross-linked alkyl acrylates are cross-linked polymers in which the comonomers consist of at least 1 of the following: acrylic acid, sodium acrylate, methacrylic acid, or alkyl acrylate. Whereas polymers consisting purely of acrylic acid are often referred to as “carbomers,” copolymers comprised of mixtures of acrylic acid and alkyl acrylate monomers may sometimes be referred to as “alkyl carbomers.” In that vein, most of the ingredients in this report could be classified as cross-linked alkyl carbomers. For example, dodecyl (C12 alkyl) acrylate, acrylic acid, and methacrylic acid could be copolymerized and cross-linked with diallyl sucrose to form an acrylates/C10-30 alkyl acrylate cross polymer with the internal structure.
Accordingly, although all of the monomers and cross-linking agents may be the same, 2 polymers with very different physical properties may share the same name under INCI conventions. The definitions and structures of the ingredients included in this review are provided in Table 1.
Definitions, Functions, and Structures.a
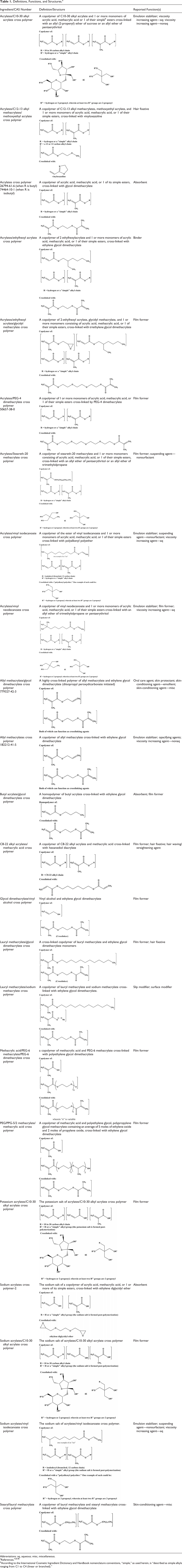
Abbreviations: aq, aqueous; misc, miscellaneous.
Physical and Chemical Properties
The available physical and chemical property information is provided in Table 2. The properties of a single ingredient, such as the above cross polymer, can vary from a highly swellable, soft material to an unswellable, very hard material because of the multitude of possible reaction conditions and the methods involved in the manufacture of these polymers. The nature of these ingredients is highly dependent on the identity of the alcohol radicals of these acrylate esters (eg, the stearyl and lauryl groups of stearyl/lauryl methacrylate cross polymer). 5 Acrylate cross polymers that correspond to 1 INCI name often have many trade names, and production processes may vary for different trade name products bearing the same INCI name. Since the products may have different properties, the trade name is included in parenthesis when available.
Chemical and Physical Properties.

The polymers in this group share a general lack of chemical reactivity that renders them nearly impervious to degradation. These ingredients are essentially insensitive to solar ultraviolet light (UV) degradation, as the primary UV absorption of acrylics is at a lower wavelength.
Method of Manufacture
Cross-linked alkyl acrylates are typically produced via free radical, head-to-tail chain-propagation polymerization. 5 The most common method is the emulsion method, but bulk and solution methods are also used. The marked variability in the identity of monomers and cross-linking agents, the ratio of comonomers, the order of addition of comonomers, the level of cross-linking, and other reaction conditions in the polymerization process can significantly alter the polymeric structure and properties of the product. 6 Additionally, postsynthesis, mechanical processing of these products can also significantly affect the consistency of these ingredients. These variables will likely differ from vendor to vendor, and possibly even from batch to batch.
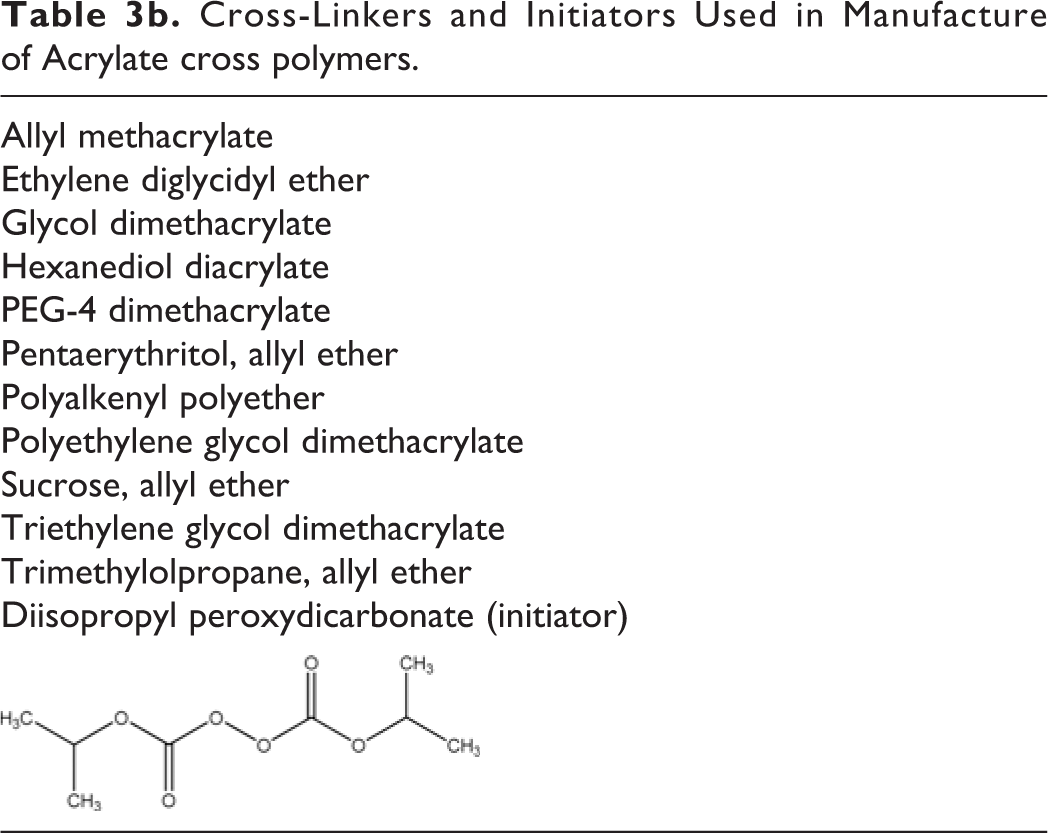
Table 3a lists the monomers used to create these cross polymers (based on INCI definition), and Table 3b names the cross-linking compounds and initiators used. 4
Monomers Used to Create Cross-Linked Alkyl Acrylates.

Cross-Linkers and Initiators Used in Manufacture of Acrylate cross polymers.
Acrylates/C10-30 alkyl acrylate cross polymer
According to a trade product technical data sheet, acrylates/C10-30 alkyl acrylate cross polymer (as Pemulen) is polymerized in an ethyl acetate–cyclohexane mixture. 7 Another source reports that acrylates/C10-30 alkyl acrylate cross polymer may be polymerized in benzene. 8 A third supplier reports that acrylates/C10-30 alkyl acrylate cross polymer is polymerized in n-hexane. 9
Acrylates/steareth-20 methacrylate cross polymer
Acrylates/steareth-20 methacrylate cross polymer (as Aculyn 88 polymer) is manufactured by an emulsion polymerization process. 10
Acrylates/vinyl isodecanoate cross polymer
Acrylates/vinyl isodecanoate cross polymer (as Stabylen 30) is produced synthetically by a free radical polymerization. 11
Acrylates/vinyl neodecanoate cross polymer
Acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) is manufactured by an emulsion polymerization process. 12
Impurities and Residual Monomer or Solvent
Acrylates/C10-30 alkyl acrylate cross polymer
According to product specification sheets from 1 company, acrylates/C10-30 alkyl acrylate cross polymer can contain (total) residual solvent (ethyl acetate + cyclohexane) at a maximum of 0.45% (Carbopol 1382; Carbopol Ultrez 20; Carbopol Ultrez 21) 13 –15 or 0.5% (Pemulen TR1; Pemulen TR2; Carbopol ETD 2020). 16 –18 Another supplier, who uses n-hexane as a solvent, reported that the maximum residual solvent in the polymer is 0.2% n-hexane. 9
As Carbopol 1342, the product specifications state that acrylates/C10-30 alkyl acrylate cross polymer can contain 0.5% (max) residual benzene. 19 A supplier reported that analysis of 40 lots of Carbopol 1342 indicated that the average level of benzene was 0.25%, and the level ranged from 0.04% to 0.41% benzene. 20 (According to the European Commission Cosmetics Directive, benzene cannot be present as a constituent of other substances, or in mixtures, in concentrations equal to, or greater than 0.1% by weight. 21 As another point of reference, US Pharmacopeia limits for benzene for several carbomers manufactured with benzene range from 0.01% to 0.5%. 22 )
One source stated that residual monomer content of acrylates/C10-30 alkyl acrylate cross polymer (trade name not provided) is typically less than 0.25% acrylic acid and less than 0.5% residual ester (C10-30 alkyl acrylate), 8 while another stated that acrylic acid monomer content is <0.1%. 23
Acrylates cross polymer
One source reported that acrylates cross polymer contained <0.005% methyl methacrylate and <0.005% butyl acrylate, 24 and another reported 0.005% (max) of methyl methacrylate, ethylene methacrylate, and isobutyl methacrylate, and that acrylates cross polymer did not contain residual solvents or preservatives. 25
Acrylates/steareth-20 methacrylate cross polymer
The composition of acrylates/steareth-20 methacrylate cross polymer (as Aculyn 88 polymer) is stated as 28.0% to 30.0% acrylates/steareth-20 methacrylate cross polymer, <0.01% residual monomer, 70.0% to 72.0% solvent (water), and 0.195% (max) sodium benzoate. 10 According to actual analytical specifications, the amount of residual ethyl acrylate present is ≤0.0001%.
Acrylates/vinyl isodecanoate cross polymer
The residual acrylic acid monomer content of acrylates/vinyl isodecanoate cross polymer (Stabylen 30) is reported to be <0.05% by weight. 11
Acrylates/vinyl neodecanoate cross polymer
The composition of acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) is stated as 28.0% to 30.0% acrylates/vinyl neodecanoate cross polymer, <0.1% residual monomer, and 70.0% to 72.0% solvent (water). 12 According to actual analytical specifications, the amount of residual ethyl acrylate present was ≤0.0001%.
Another source reported the residual monomer level of acrylates/vinyl neodecanoate cross polymer is <0.01%. 26
Lauryl methacrylate/glycol dimethacrylate cross polymer
The residual monomer levels of lauryl methacrylate/glycol dimethacrylate cross polymer are <0.01% lauryl methacrylate and <0.01 ppm ethylene glycol dimethacrylate. 27 Lauryl methacrylate/glycol dimethacrylate cross polymer has a residual solvent level of ≤0.1% isopropanol. The ingredient can contain up to 2% adsorbed water.
Sodium acrylates cross polymer 2
The maximum amount of residual monomer content in sodium acrylates cross polymer 2 (Aqua Keep 10SH-NFC) is 0.02%. 28
Use
Cosmetic
Cross-linked alkyl acrylates are reported to function as absorbents, film formers, emulsion stabilizers, viscosity increasing agents, suspending agents, binders, and/or skin-conditioning agents in cosmetic formulations. 4 Acrylates/C10-30 alkyl acrylate cross polymer functions as a primary emulsifier in oil-in-water emulsions. 7 Voluntary Cosmetic Registration Program data obtained in 2011, 29 and the concentration of use information received in response to a survey conducted by the Personal Care Products Council, 30 indicates that 11 of the 23 cross-linked alkyl acrylates named in this report currently are used in cosmetic formulations. Acrylates/C10-30 alkyl acrylate cross polymer has the greatest number of uses, with 1,696 reported; 1,365 of those uses are in leave-on products. Acrylates cross polymer, acrylates/vinyl isodecanoate cross polymer, acrylates/vinyl neodecanoate cross polymer, allyl methacrylates cross polymer, lauryl methacrylate/glycol dimethacrylate cross polymer, lauryl methacrylate/sodium methacrylate cross polymer, and sodium acrylates/C10-30 alkyl acrylate cross polymer are all used in less than 75 formulations.
Some acrylates/C10-30 alkyl acrylate cross polymers are polymerized in benzene; the highest reported concentrations of use of this ingredient when polymerized in benzene are 0.4% and 1.1% for leave-on and rinse-off products, respectively. 31 The use concentrations for acrylates/C10-30 alkyl acrylate cross polymer not polymerized in benzene are up to 5% in leave-on and rinse-off products; 5% is the highest rinse-off concentration of use of the cross-linked alkyl acrylates. The highest concentration of use reported in leave-on cross-linked alkyl acrylates is 6% acrylates/ethylhexyl acrylate cross polymer. 30 Frequency and concentration of use data are provided in Table 4a. The ingredients not reported to be used are listed in Table 4b.
Frequency and Concentration of Use According to Duration and Type of Exposure.
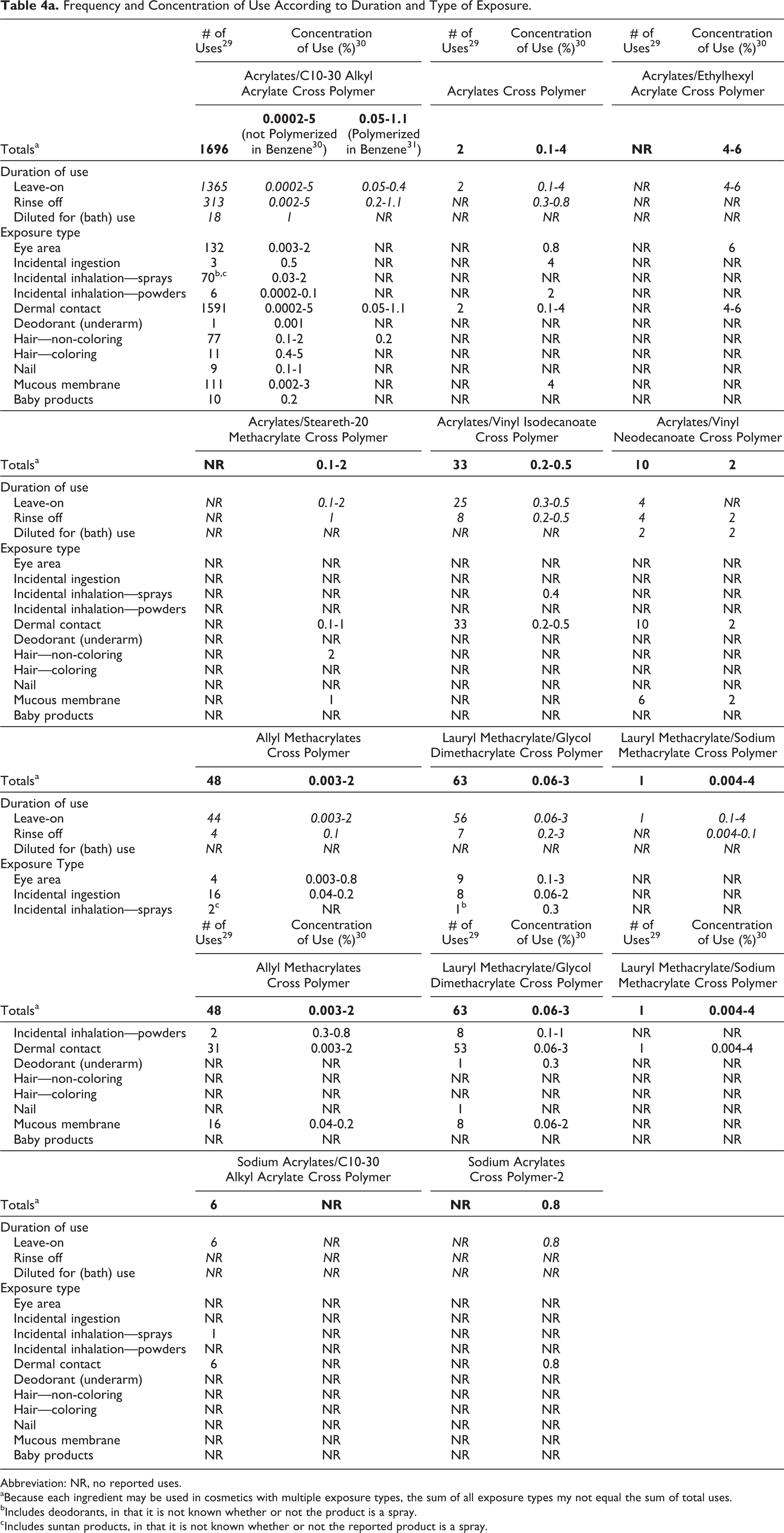
Abbreviation: NR, no reported uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
bIncludes deodorants, in that it is not known whether or not the product is a spray.
cIncludes suntan products, in that it is not known whether or not the reported product is a spray.
Ingredients Not Reported to be Used.

Products containing some cross-linked alkyl acrylates may be applied to baby skin, used near the eye area or mucous membranes, or could possibly be ingested or inhaled. In practice, 95% to 99% of the particles released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range. 32,33 Therefore, most particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal region and would not be respirable to any appreciable level. 34,35 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 35 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
All of the ingredients included in this review, with the exception of acrylates/C12-13 alkyl methacrylates methoxyethyl acrylate cross polymer and methacrylic acid/PEG-6 methacrylate/PEG-6 dimethacrylate cross polymer, are listed in the European Union inventory of cosmetic ingredients. 36 The 2 ingredients that are not included in the European Union inventory are in the process of being named and will be added once that process is complete. 37
Noncosmetic
Acrylic ester polymers are used in coatings, textiles, adhesives, and paper manufacture. 5
Toxicokinetics
Published toxicokinetics, absorption, distribution, metabolism, and excretion data were not found for the cross polymers. Large polymeric structures, however, such as cross-linked alkyl acrylates, generally are not absorbed through the skin. Toxicokinetics data on some of the monomers are provided in Table 5.
Relevant Summary Information on Component Monomers.
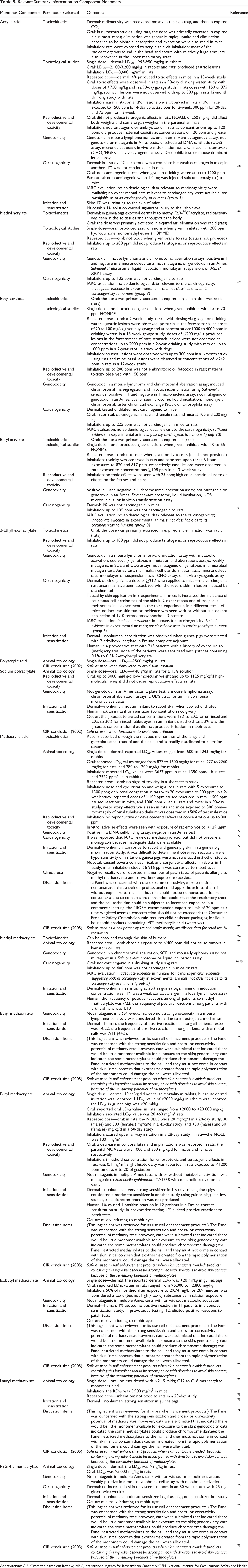
Abbreviations: CIR, Cosmetic Ingredient Review; IARC, International Agency for Research on Cancer; NIOSH, National Institute for Occupational Safety and Health.
Effect on Skin Permeation
Acrylates/C10-30 alkyl acrylate cross polymer
A topical formulation vehicle that included acrylates/C10-30 alkyl acrylate cross polymer (Pemulen TR-2), in combination with PEG 400 and carbomer, reduced the permeation of N,N-diethyl-m-toluamide through skin. 38 Evaluations were made in vitro using excised rat skin and in vivo using Beagle dogs.
Toxicological Studies
To aid in the evaluation of the safety of these cross polymers, Table 5 provides a brief summary of relevant data on a number of monomer components. (This summary is not intended to be an all-encompassing review of these monomers.)
Single-Dose (Acute) Toxicity
Dermal
Acrylates/C10-30 alkyl acrylate cross polymer
According to an industry MSDS, the dermal LD50 of acrylates/C10-30 alkyl acrylate cross polymer (as Pemulen TR1) in rabbits is >2.0 g/kg. 39
Acrylates/vinyl neodecanoate cross polymer
The dermal LD50 of acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) in rabbits is >5.0 g/kg. 12
Oral
Acrylates/C10-30 alkyl acrylate cross polymer
According to an industry MSDS, the oral LD50 of acrylates/C10-30 alkyl acrylate cross polymer (as Pemulen TR1) in rats is >10 g/kg. 39 Another source provided information from an MSDS, stating that the oral LD50 in rats is >2 g/kg. 23
Acrylates/vinyl isodecanoate cross polymer
The oral LD50 acrylates/vinyl isodecanoate cross polymer (as Stabylen 30) in rats is >2 g/kg body weight. 40
Acrylates/vinyl neodecanoate cross polymer
The oral LD50 of acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) in rats is >5.0 g/kg. 12
Sodium acrylates cross polymer 2
According to an industry MSDS, the oral LD50 of sodium acrylates cross polymer 2 (as Aqua Keep 10SH-NFC) in rats is >2 g/kg. 41
Inhalation
Acrylates/vinyl neodecanoate cross polymers
The inhalation LC50 of acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) in rats is >16.34 mg/L air (1 hour). 12
Repeated Dose Toxicity
Inhalation
Acrylates/C10-30 alkyl acrylate cross polymer
In an industry MSDS for acrylates/C10-30 alkyl acrylate cross polymers (as Pemulen TR-1), a 2-year inhalation study in which rats were exposed to a respirable, water-absorbent sodium polyacrylate dust is described under toxicological information. Lung effects such as inflammation, hyperplasia, and tumors were observed. 39 There were no observed adverse effects at exposures of 0.05 mg/m3.
Reproductive and Developmental Toxicity
Published reproductive and developmental toxicity data were not found. Reproductive and developmental toxicity data on some of the monomers are provided in Table 5.
Genotoxicity
Genotoxicity data on some of the monomers are provided in Table 5.
Acrylates/C10-30 alkyl acrylate cross polymer
Acrylates/C10-30 alkyl acrylate cross polymer, tested at 156 to 500 µg/plate in dimethyl sulfoxide, was not mutagenic in an Ames assay with Salmonella typhimurium TA98 and TA100. 23 It is not stated directly, but it appears that the studies were performed with and without metabolic activation.
Acrylates/Steareth-20 methacrylate cross polymer
The acrylic copolymer of acrylates/steareth-20 methacrylate cross polymer (as Aculyn 88 polymer) was not mutagenic in an Ames test, with or without metabolic activation. 10 (Study performed using good laboratory practices [GLP]; details not provided.)
Acrylates/vinyl neodecanoate cross polymer
The acrylic copolymer of acrylates/vinyl neodecanoate cross polymer (as Aculyn 38 polymer) was not mutagenic in an Ames test, with or without metabolic activation. 12 (GLP study; details not provided).
Sodium acrylates cross polymer 2
According to an industry MSDS, sodium acrylates cross polymer 2 (as Aqua Keep 10SH-NFC) was negative in an Ames test using S typhimurium TA98, TA100, TA1535, and TA1537 and Escherichia coli WP2uvrA. 41
Carcinogenicity
Published carcinogenicity studies were not found. Carcinogenicity data on some of the monomers are provided in Table 5.
Irritation and Sensitization
Irritation and sensitization data on some of the monomers are provided in Table 5.
Skin Irritation and Sensitization
Dermal irritation and sensitization studies, using alternative methods and nonhuman and human test populations, are presented in Table 6.
Dermal Irritation and Sensitization: Alternative, Nonhuman, and Human.
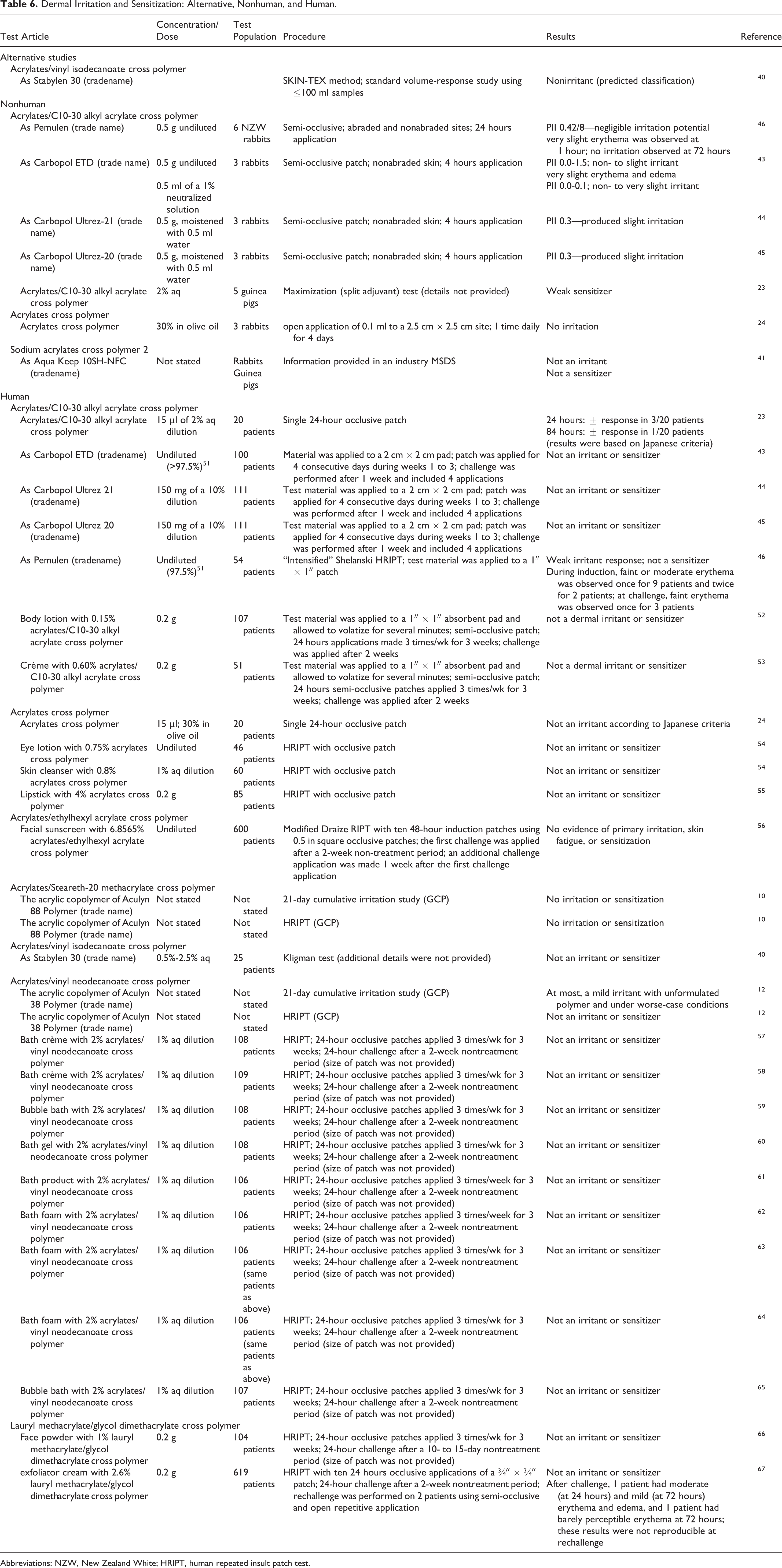
Abbreviations: NZW, New Zealand White; HRIPT, human repeated insult patch test.
In an alternative method study, acrylates/vinyl neodecanoate cross polymer was predicted to be a nonirritant. The nonhuman studies reported no to slight irritation with undiluted and weak sensitization with 2% aq, acrylates/C10-30 alkyl acrylate cross polymer, no irritation with acrylates cross polymer at 30% in olive oil, and no irritation or sensitization with sodium acrylates cross polymer 2 (concentration not specified). Mostly, human testing with undiluted acrylates/C10-30 alkyl acrylate cross polymer, acrylates cross polymer, and acrylates/ethylhexyl acrylate cross polymer, up to 2.5% aq acrylates/vinyl isodecanoate cross polymer, 1% aq dilutions of formulations containing 2% acrylates/vinyl neodecanoate cross polymer, and formulations containing up to 2.6% lauryl methacrylate/glycol dimethacrylate cross polymers do not indicate any dermal irritation or sensitization. The only exception was a weak irritant response noted during an intensified Shelanski human repeated insult patch test (HRIPT) with undiluted acrylates/C10-30 alkyl acrylate cross polymer.
Ocular Irritation
Alternative studies
Acrylates/vinyl isodecanoate cross polymer
The EYE-TEX alternative method was used to predict the in vivo ocular irritation classification of acrylates/vinyl isodecanoate cross polymer (as Stabylen 30).40 The results obtained in a standard volume–response study using samples of ≤100 µl test material corresponded to a Draize ocular irritation classification of nonirritant.
Lauryl methacrylate/glycol dimethacrylate cross polymer
The EpiOcular Human Cell Construct (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide [MTT] assay), was used to assess the potential ocular irritation of a face powder containing 1% lauryl methacrylate/glycol dimethacrylate cross polymer. 42 The ET50 (duration of exposure resulting in a 50% decrease in MTT conversion) of the test material was >1,440 minutes, which was the maximum exposure time. (As a reference point, the ET50 of the positive control, 0.3% Triton X-100, was 16.3 minutes.)
Nonhuman
Acrylates/C10-30 alkyl acrylate cross polymer
The ocular irritation potential of acrylates/C10-30 alkyl acrylate cross polymer (as Carbopol ETD) was evaluated using groups of 3 albino rabbits. 43 The test material, undiluted and as a 1% neutralized solution (pH 6.9-7.0), was instilled into the conjunctival sac of 1 eye of each rabbit per group; the contralateral eyes served as a control. The eyes were not rinsed. The undiluted test material produced slight to moderate corneal and conjunctival irritation which cleared by day 7. Slight iridal and conjunctival irritation was observed with the 1% solution. All signs of irritation cleared within 72 hours.
In other studies using the same procedure, the ocular irritation potential of acrylates/C10-30 alkyl acrylate cross polymer (as Carbopol Ultrez 20 and Carbopol Ultrez 21) was evaluated using groups of 3 rabbits. 44,45 The test material was evaluated undiluted and as a 5% dilution in distilled water. The undiluted test material produced moderate corneal irritation and conjunctival irritation which cleared by day 21. (The maximum mean score [MMS] was 37.7/110.) Moderate conjunctival irritation (MMS 9.3/110) was observed with the 5% solution, which was classified as a minimal irritant.
The ocular irritation potential of acrylates/C10-30 alkyl acrylate cross polymer (as Pemulen) was evaluated by instilling 0.021 g of the test article into the conjunctival sac of 1 eye of 9 New Zealand White rabbits. 46 The contralateral eyes were untreated and served as the control. At 30 seconds postinstillation, both eyes of 3 rabbits were rinsed; the eyes of the other 6 rabbits were not rinsed. The eyes were examined for irritation for up to 72 hours following dosing. “Significant” ocular irritation was observed in 3 of the 6 unrinsed eyes. At 24 hours after instillation, corneal opacity was observed in 3 and iritis in 1 unrinsed eye; minimal conjunctivitis was seen in all 6 unrinsed eyes. These observations were resolved by 72 hours. “Less severe responses” were observed in the rinsed eyes. Iritis was observed in 1 and conjunctivitis in 3 of the rinsed eyes at 24 hours after dosing. At 48 hours after dosing, conjunctivitis was observed in 1 rinsed eye. Based on the observations made for the unrinsed eyes, the authors stated that this product was considered a borderline irritant.
Acrylates cross polymer
The ocular irritation potential of acrylates cross polymer was evaluated by instilling 0.1 ml of the test material, at a concentration of 50% in olive oil, into the conjunctival sac of 1 eye of 3 Japanese white rabbits. 24 The Draize score was 1.3. (Additional details were not provided.)
Sodium acrylates cross polymer 2
According to an industry MSDS, sodium acrylates cross polymer 2 (as Aqua Keep 10SH-NFC) is not an ocular irritant in rabbits. 41
Clinical Assessment of Safety
Risk Assessment
Conservative risk assessments were submitted by the Personal Care Products Council’s CIR Science and Support Committee (SSC) and by the CIR to address the carcinogenic endpoint for benzene, because it may be used as a solvent in the manufacture of acrylates/C10-30 alkyl acrylates cross polymer. Both assessments assumed the highest reported concentration of residual benzene in acrylates/C10-30 alkyl acrylates cross polymer used as a raw ingredient, the highest reported use concentration in a leave-on product of the raw ingredient polymerized in benzene, 10% evaporation of the residual benzene during manufacturing of the product, 10% benzene absorbed from the product through the skin, and the reported 50th and 95th percentiles of the amount of product used daily.
CIR SSC Risk Assessment 31
The assumptions used to calculate CIR SSC’s example exposure assessment were: 50th percentile use =7.63 g body lotion used/use day 95th percentile use =16.83 g body lotion used/use day 0.4% acrylates/C10-30 alkyl acrylate cross polymer in body lotion 0.41% benzene in acrylates/C10-30 alkyl acrylate cross polymer 10% benzene absorbed percutaneously
Estimated Exposure 0.41% benzene in raw material × 0.4% acrylates/C10-30 alkyl acrylates cross polymer in a body product = 0.00164% benzene in the product 50th 7.63 g body product used/day × 0.00164% = 0.000125 g/d = 125 µg/d absorb 10% × 125 µg/d = 12.5 µg/d 95th 16.83 g body product used/d 0.00164% = 0.000276 g/d = 276 µg/d absorb 10% × 276 µg/d = 27.6 µg/d
The SSC Comparison to Risk Level
The Environmental Protection Agency (EPA) drinking water concentration associated with 106 cancer risk is 1 and 10 µg/L. 47 Assuming consumption of 2 L of water each day, this results in a value of 2 to 20 µg/d. The estimated exposure from the use of a leave-on body product at the 50th percentile, assuming the greatest concentration of acrylates/C10-30 alkyl acrylates cross polymer polymerized in benzene, is in within the range associated with a 106 cancer risk, while use at the 95th percentile is just above the range associated with a 106 risk. The SSC noted that significant volatilization of benzene would occur during the manufacture of the finished product because the temperatures reached during processing are at or near the boiling point of benzene (80.1°C). They indicated that assuming that only 10% of the residual benzene is volatilized during product manufacture, would yield an exposure within the range associated with a 10− 6 risk for use of a body lotion at the 95th percentile.
CIR’s Risk Assessment
The EPA presents the oral slope factor for benzene as a range, based on the assumption that benzene is 100% absorbed after oral exposure. Specifically, the slope factor ranges from 1.5 × 10− 5 to 5.5 × 10− 5 (µg/kg/d)− 1. The EPA drinking water concentration range (1-10 µg/L) representing a 10− 6 lifetime cancer risk was calculated from the slope factor range, rounding down the lowest concentration of the range to 1 µg/L and rounding up the highest concentration to 10 µg/L.
General Equation
[%] benzene in acrylates/C10-30 alkyl acrylates crosspolymer × [%] acrylates/C10-30 alkyl acrylates crosspolymer in body lotion × [g/d] body lotion × [%] benzene absorbed percutaneously × [kg]−1 body weight × 106 [µg/g] conversion factor × slope factor [µg/kg/d]−1 =Cancer Risk Estimate [unitless]
Using the EPA’s highest cancer slope factor in the range (5.5 × 10−5 [µg/kg/d]− 1 ) in accordance with the EPA risk assessment guidelines yields an upper bound lifetime cancer risk estimate of 2.2 × 10−5, assuming the 95th percentile product use and 70 kg body weight:
Upper Bound Risk for 95th Percentile Exposure
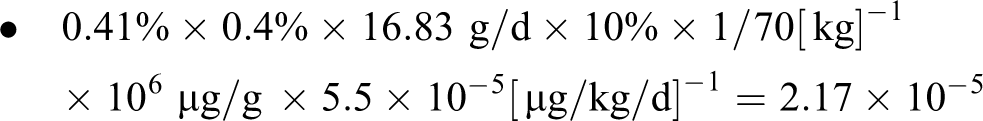
This estimate (2.2 × 10− 5) is 22 times higher than the upper bound risk estimate considered to be de minimis (10− 6).
Assuming that 10% of the benzene evaporates during the product manufacturing process reduces the upper bound estimate to 2 × 10− 5 (2.17 × 10− 5 × 90% =1.95 × 10− 5), which is still about 20 times higher than 10− 6.
Using the EPA’s lowest cancer slope factor in their range (1.5 × 10-5 [µg/kg/d]− 1 ), assuming 50th percentile product use, 10% percutaneous absorption, and 10% evaporation during the manufacturing process yields upper bound cancer risk estimates that still exceed 10− 6 by 2- to 3-fold:
Upper Bound Risk for 50th Percentile Exposure
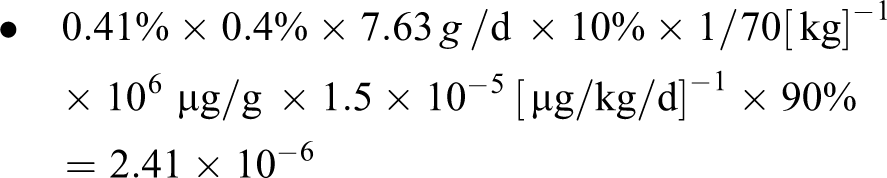
The SSC reported that the cancer risk would <10−6, by comparing the estimated daily absorbed dose of benzene from the product to drinking water concentrations that EPA suggests represents a 10−6 lifetime risk. However, CIR calculated upper bound lifetime cancer risk estimates up to 20-fold greater than 10−6, based on EPA’s cancer slope factors for benzene.
Industrial Exposure Limits
According to an industry MSDS, no exposure limits have been established for acrylates/C10-30 alkyl acrylate cross polymer. 39 The industry-recommended permissible exposure limits for respirable polyacrylate dusts is 0.05 mg/m3. Breathing of dust may cause coughing, mucous production, and shortness of breath. According to an industry MSDS, the exposure limit for respirable sodium acrylates cross polymer 2 dust (particle size <10 µm) is 0.05 mg/m3. 41
Summary
The cross-linked alkyl acrylates are cross-linked polymers and are very large molecules that consist of comonomers of acrylic acid, sodium acrylate, methacrylic acid, and/or alkyl acrylate, and they share chemical properties, including a general lack of chemical reactivity. Cross-linked alkyl acrylates are typically produced via free radical, head-to tail chain-propagation polymerization. Ethyl acetate + cyclohexane, water, n-hexane, and benzene are all named as solvents. Because of the manner in which these polymers are created and the mixture of monomers and cross-linking agents that can be used, 2 polymers that have the same INCI name can have very different physical consistencies. Small amounts of residual monomer and/or solvent may be present in the raw ingredients.
Cross-linked alkyl acrylates are reported to function in cosmetic formulations as absorbents, film formers, emulsion stabilizers, viscosity increasing agents, suspending agents, binders, and/or skin-conditioning agents. In 2011, it was reported that acrylates/C10-30 alkyl acrylate cross polymer was used in 1,696 cosmetic formulations; 1,365 of those uses are in leave-on products, and the reported concentration of use in these leave-on products is up to 5%. According to industry data, acrylates/ethylhexyl acrylate cross polymer had the highest concentration of use in a leave-on product at 6%; the highest concentration of use reported in rinse-off products was 5% acrylates/C10-30 alkyl acrylate cross polymer.
Toxicokinetic data were not found in the published literature. Little toxicity data were available; the acute dermal and oral toxicity data that were found indicated that these ingredients are not very toxic. The little genotoxicity data that were available reported negative results in Ames tests. Carcinogenicity data were not found in the published literature for the polymers, but data were available for the monomers.
In an alternative method study, acrylates/vinyl neodecanoate cross polymer was predicted to be a nonirritant. The nonhuman studies reported no to slight irritation with undiluted and weak sensitization with 2% aq, acrylates/C10-30 alkyl acrylate cross polymer, no irritation with acrylates cross polymer at 30% in olive oil, and no irritation or sensitization with sodium acrylates cross polymer 2 (concentration not specified). Mostly, human testing with undiluted acrylates/C10-30 alkyl acrylate cross polymer, acrylates cross polymer, and acrylates/ethylhexyl acrylate cross polymer, up to 2.5% aq acrylates/vinyl isodecanoate cross polymer, 1% aq dilutions of formulations containing 2% acrylates/vinyl neodecanoate cross polymer, and formulations containing up to 2.6% lauryl methacrylate/glycol dimethacrylate cross polymers do not indicate any dermal irritation or sensitization. The only exception was a weak irritant response noted during an intensified Shelanski HRIPT with undiluted acrylates/C10-30 alkyl acrylate cross polymer.
Alternative test methods for ocular irritation indicated that acrylates/vinyl isodecanoate cross polymer and a formulation containing 1% lauryl methacrylate/glycol dimethacrylate cross polymer are not likely ocular irritants. In studies using rabbits, undiluted acrylates/C10-30 alkyl acrylate cross polymer produced minimal to moderate irritation, and it was considered a borderline irritant in unrinsed rabbit eyes. Acrylates cross polymer, at 50% in olive oil, and sodium acrylates cross polymer 2 did not appear to be ocular irritants in rabbit eyes.
Two different risk assessments evaluating the carcinogenic endpoint for benzene that may be present in acrylates/ C10-30 alkyl acrylates cross polymer resulted in different lifetime risk. One found that the risk was within the range associated with a 106 cancer risk, while the other reported a 20-fold greater risk.
Discussion
Few published data were available on the cross-linked alkyl acrylates. The CIR Expert Panel was provided with some summary information on the monomers for their use in evaluating these cross polymers.
The Panel noted that these cross-linked alkyl acrylates are macromolecules that are not expected to pass through the stratum corneum of the skin, so significant dermal absorption is not expected. Therefore, topically applied cosmetics are not expected to result in systemic or reproductive and developmental toxicity or to have genotoxic or carcinogenic effects upon use.
The Panel noted that cosmetic products containing these ingredients are reportedly used around the eyes, on the lips, and on other mucous membranes. Thus, cross-linked alkyl acrylates could be absorbed systemically through the relatively moist, thin stratum cornea of the conjunctiva, lips, and other mucous membranes, and through ingestion when applied to the lips. However, the Panel noted that any absorption through healthy intact mucous membranes is likely to be not significant, primarily because of the relatively large molecular sizes. Furthermore, the chemically inert nature of the polymers precludes degradation to smaller absorbable species. Absorption of the polymers and their residual monomers in cosmetic products also would be limited after application to the lips or eye area based on the relatively small fractions of the applied products that might be inadvertently ingested or make direct contact with the conjunctiva.
The Panel addressed the concern of residual monomer or solvent that might be present in the cross polymers. In most cases, taking into consideration the low amount of residual monomer in the cross polymers and the low use concentration of the polymers themselves, the Panel was not concerned that the presence of residual monomer would result in adverse effects. However, the use of benzene as a solvent is an exception and did cause concern. It cannot be predicted with certainty what quantity of benzene would be volatilized/leached from acrylates/C10-30 alkyl acrylates cross polymer during manufacture, formulation, or use. While some benzene is inevitably volatilized during manufacture, some benzene may be trapped in the polymer matrix and may leach out during formulation and use, but there is no way of knowing how much (or if any) benzene would leach out without appropriate data from a representative product formulation.
Conservative risk assessments were submitted by industry and by the CIR to address the carcinogenic endpoint for benzene, because it may be used as a solvent in the manufacture of acrylates/C10-30 alkyl acrylates cross polymer. Both assessments assumed the highest reported concentration of residual benzene in acrylates/C10-30 alkyl acrylates cross polymer used as a raw ingredient, the highest reported use concentration in a leave-on product of the raw ingredient polymerized in benzene, 10% evaporation of the residual benzene during manufacturing of the product, 10% benzene absorbed from the product through the skin, and the reported 95th percentile of the amount of product used daily. Industry reported that the cancer risk would <10− 6, by comparing the estimated daily absorbed dose of benzene from the product to drinking water concentrations that EPA suggests represents a 10− 6 lifetime risk. However, CIR calculated upper bound lifetime cancer risk estimates up to 20-fold greater than 10− 6, based on EPA’s cancer slope factors for benzene. Given the uncertainty of the assumptions used in the risk assessment, the Panel was not comfortable with using a risk assessment in evaluating the carcinogenic endpoint. Therefore, the Panel found the data insufficient to conclude that the residual benzene levels are safe.
Because these ingredients can be used in products that may be aerosolized, including sprays and powders, the Panel discussed the issue of potential inhalation toxicity. The limited data available from an acute exposure study suggested little potential for pulmonary overload or other respiratory effects at relevant doses. The Panel considered other data available to characterize the potential for cross-linked alkyl acrylates to cause systemic toxicity, irritation, sensitization, or other effects. They noted the lack of systemic toxicity at high doses in several acute oral exposure studies, little or no irritation or sensitization in multiple tests of dermal and ocular exposure, and the absence of genotoxicity in Ames tests. In addition, these ingredients are macromolecules, insoluble in water, and chemically inert under physiological conditions or conditions of use, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. Further, these ingredients are reportedly used at concentrations ≤4% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of particles produced in cosmetic aerosols would not be respirable to any appreciable extent. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information indicates that inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that the cross-linked alkyl acrylates listed below are safe in the present practices of use and concentration described in this safety assessment, except when they are polymerized in benzene. Acrylates/C10-30 alkyl acrylate cross polymer may be polymerized in benzene, and the available data are insufficient to make a determination of safety for this cross-linked alkyl acrylate when it is polymerized in benzene. Acrylates/C10-30 alkyl acrylate cross polymer Acrylates/C12-13 alkyl methacrylates/methoxyethyl acrylate cross polymer* Acrylates cross polymer Acrylates/ethylhexyl acrylate cross polymer Acrylates/ethylhexyl acrylate/glycidyl methacrylate cross polymer* Acrylates/PEG-4 dimethacrylate cross polymer* Acrylates/Steareth-20 methacrylate cross polymer Acrylates/vinyl isodecanoate cross polymer Acrylates/vinyl neodecanoate cross polymer Allyl methacrylate/glycol dimethacrylate cross polymer* Allyl methacrylates cross polymer Butyl acrylate/glycol dimethacrylate cross polymer* C8-22 alkyl acrylates/methacrylic acid cross polymer* Glycol dimethacrylate/vinyl alcohol cross polymer* Lauryl methacrylate/glycol dimethacrylate cross polymer Lauryl methacrylate/sodium methacrylate cross polymer Methacrylic acid/PEG-6 methacrylate/PEG-6 dimethacrylate cross polymer* PEG/PPG-5/2 methacrylate/methacrylic acid cross polymer* Potassium acrylates/C10-30 alkyl acrylate cross polymer* Sodium acrylates cross polymer 2 Sodium acrylates/C10-30 alkyl acrylate cross polymer Sodium acrylates/vinyl isodecanoate cross polymer* Stearyl/lauryl methacrylate cross polymer* *Were the ingredients not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Fiume, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Heldreth, B. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Boyer, I. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Gill, L., Andersen, F. Alan, Bergfeld, W., Belsito, D. Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to analysis and interpretation, contributed to conception and design, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
