Abstract
This is a safety assessment of alumina and aluminum hydroxide as used in cosmetics. Alumina functions as an abrasive, absorbent, anticaking agent, bulking agent, and opacifying agent. Aluminum hydroxide functions as a buffering agent, corrosion inhibitor, and pH adjuster. The Food and Drug Administration (FDA) evaluated the safe use of alumina in several medical devices and aluminum hydroxide in over-the-counter drugs, which included a review of human and animal safety data. The Cosmetic Ingredient Review (CIR) Expert Panel considered the FDA evaluations as part of the basis for determining the safety of these ingredients as used in cosmetics. Alumina used in cosmetics is essentially the same as that used in medical devices. This safety assessment does not include metallic or elemental aluminum as a cosmetic ingredient. The CIR Expert Panel concluded that alumina and aluminum hydroxide are safe in the present practices of use and concentration described in this safety assessment.
Introduction
This report addresses the safety of alumina and aluminum hydroxide as used in cosmetics. According to the International Cosmetic Ingredient Dictionary and Handbook, 1 alumina is reported to function in cosmetics as an abrasive, absorbent, anticaking agent, bulking agent, and opacifying agent; aluminum hydroxide is reported to function as a buffering agent, corrosion inhibitor, and pH adjuster (Table 1).
Definitions and Functions of the Ingredients in This Safety Assessment.a

Abbreviation: CIR, cosmetic ingredient review.
aThe italicized text represents additions made by CIR staff.
These ingredients have been approved by the US Food and Drug Administration (FDA) for use in medical devices and over-the-counter (OTC) drugs (21 CFR 247.10; 21 CFR 331.11; 21 CFR 347.50). 2 –4 The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) concluded that the cosmetic ingredient alumina is chemically equivalent to the alumina used as part of color additives in medical devices such as bone cements and sutures. Alumina is also a material used in the construction of dental and hip implants. The FDA found that the information submitted for the approval of medical devices containing alumina was adequate and determined that alumina is safe for use in devices that come in contact with soft tissue, bone, and internal organs. 2 Additionally, alumina is approved by the FDA as an indirect food additive. 5 The Panel concluded that the FDA’s evaluations of alumina in medical devices, coupled with the Panel’s review of information on aluminum hydroxide, were sufficient to support the safety assessment of alumina.
The Panel also concluded that the aluminum hydroxide used in cosmetics is chemically equivalent to that used in OTC antacid products. The FDA found that the information submitted for the approval of those drugs was adequate to support safe use (21 CFR 247.10; 21 CFR 331.11; 21 CFR 347.50). The FDA also determined that aluminum hydroxide is generally regarded as safe (GRAS) as a direct food additive (21 CFR 176.210, 177.1200, 177.2600). 6 The Panel concluded that FDA’s evaluations of aluminum hydroxide as a food additive and OTC drug, coupled with the Panel’s review of primary scientific toxicity data, were sufficient to support the safety assessment of this ingredient as used in cosmetics.
The CIR has reviewed several cosmetic ingredients that consist of molecules containing aluminum atoms (Table 2). The conclusions were safe as used or safe with qualifications for all of these ingredients.
Cosmetic Ingredients Containing Aluminum That Have Been Reviewed by CIR.
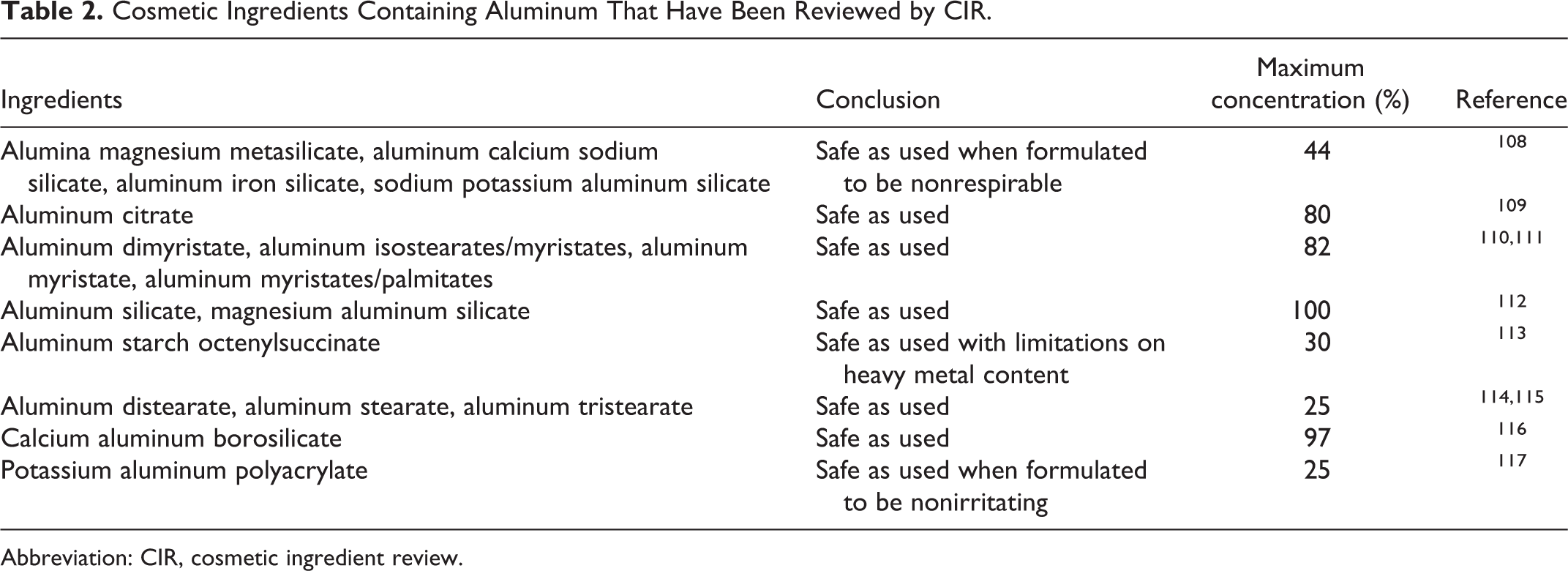
Abbreviation: CIR, cosmetic ingredient review.
The cosmetic ingredients alumina (aluminum oxide) and aluminum hydroxide are stable, oxidized aluminum compounds that differ substantially from aluminum (elemental or metallic) in chemical and physical properties, functions, and potential for toxicity. There has been substantial speculation in the literature that exposure to elemental aluminum or aluminum compounds could play a role in the etiology of Alzheimer’s disease, breast cancer, and other health problems. Overall, scientific research has failed to find cause and effect relationships. 7 –56 Furthermore, systemic exposure to aluminum from the use of alumina and aluminum hydroxide in cosmetics is expected to be negligible because it is poorly absorbed. 7,40,57 –69 The Panel considered the toxicological literature on aluminum and was satisfied that much of the speculation about aluminum toxicity is not relevant to the assessment of the safety of alumina and alumina hydroxide as used in cosmetics. A brief overview of aluminum toxicity studies is attached (Appendix A) to provide supplementary information reflecting the Panel’s consideration of these issues.
Chemistry
Overview
Definitions, CAS numbers, and functions are provided in Table 1. The structures of alumina and aluminum hydroxide are presented in Figure 1.
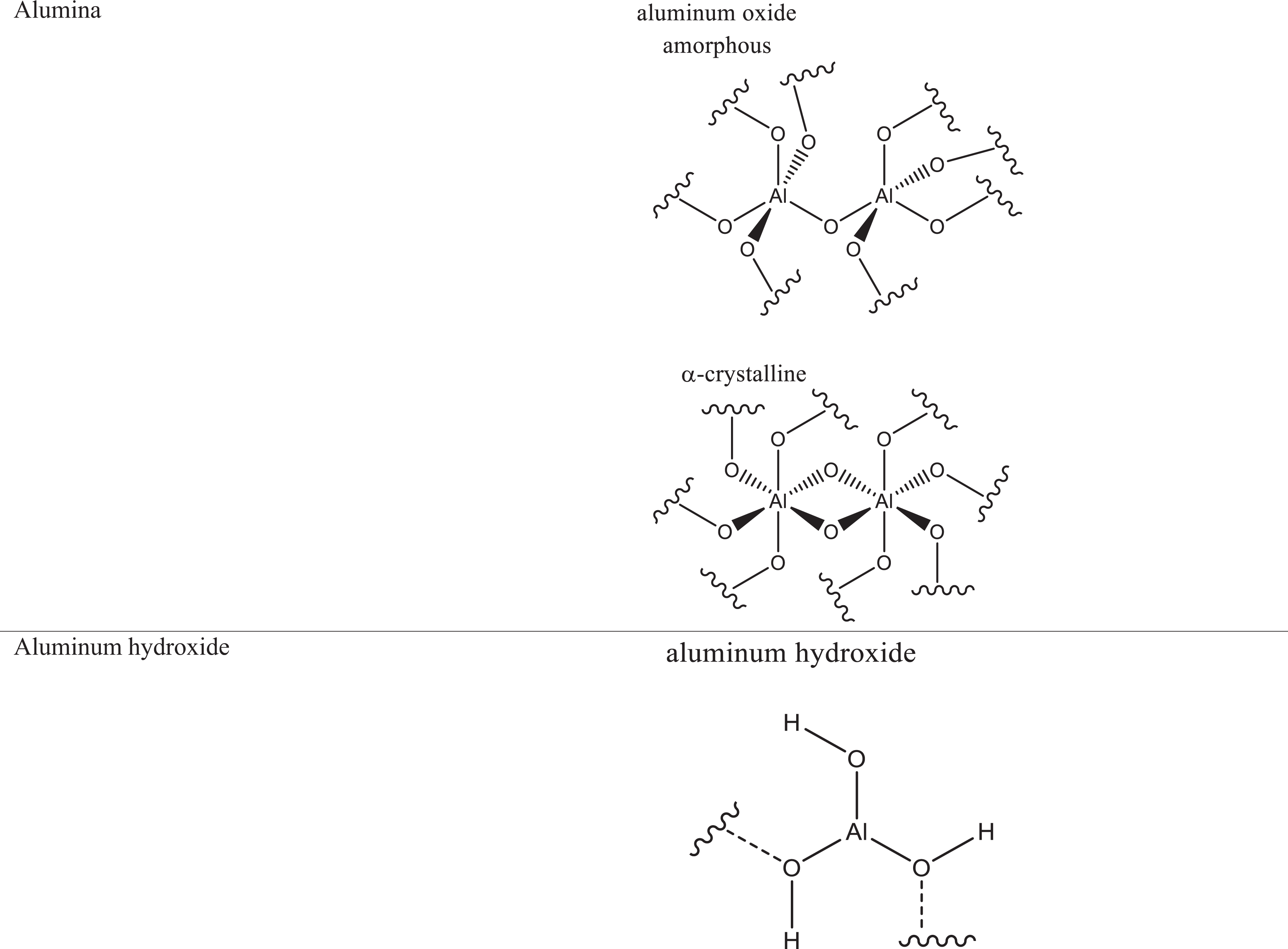
Formulas and idealized structures of the ingredients in this safety assessment.
Alumina, also known as aluminum oxide (Al2O3), is dehydrated (or calcined) aluminum hydroxide. 70 Alumina is also the primary constituent of emerald, ruby, and sapphire (the colors of which come from small impurities of heavy metals). The most common naturally occurring form of alumina is corundum. Corundum is primarily composed of α-alumina, which is crystalline. This water-insoluble, inorganic solid can form a number of other crystalline phases, and an amorphous form as well. Each phase has a unique crystal structure and varies in chemical properties, such as its acid–base reaction rate. When synthetically dehydrated from aluminum hydroxide, a mixture of alumina phases typically forms, unless specific controls are applied. Figure 1 schematically depicts both amorphous and crystalline alumina.
Aluminum hydroxide, also known as hydrated alumina, is most commonly found as the polymorphic mineral gibbsite (a component of the aluminum ore known as bauxite). 70,71 This inorganic, amphoteric solid can also form 3 other polymorphs. However, the chemical formula of Al(OH)3 is the same for all polymorphs, each of which differs from the others only by interlayer spacing and, consequently, by relative acid/base reaction rates.
There are 4 known polymorphs of crystalline aluminum hydroxide: gibbsite, bayerite, nordstrandite, and doyleite, which can have different chemical/physical properties. 72 The properties of the starting materials (pH, presence of anions or salt and mineral surfaces) influence the formation of particular polymorphs from aluminum interlayers and/or hydroxyl aluminum polymers. All the polymorphs of aluminum hydroxide consist of layers of aluminum octahedra with hydroxyl groups on either side, which hydrogen bond the layers together, and differences arising from variations in the stacking sequences of the layers. Of the possible configurations, gibbsite and bayerite represent the 2 ends of the spectrum of types of stacking sequences. Nordstrandite and doyleite have intermediate structures.
There is no universal standard nomenclature for aluminum oxides and hydroxides; thus, there may be inconsistencies in the use of these names among sources. 72 Categorization is based on crystallographic structures found under environmental conditions and cited most often in the literature (Table 3). The α prefix is generally applied to hexagonal, close-packed, and related structures; these are aluminum minerals abundantly found in nature. The γ prefix is generally applied to designate polymorphism, structural alteration, or dehydration of these minerals (originally applied to all aluminum hydroxides and hydrolyzed aluminas other than the α-phase minerals). The γ-phase has cubic close-packed lattices or other related structures.
Comparison of Nomenclature for Alumina and Aluminum Hydroxide. 72
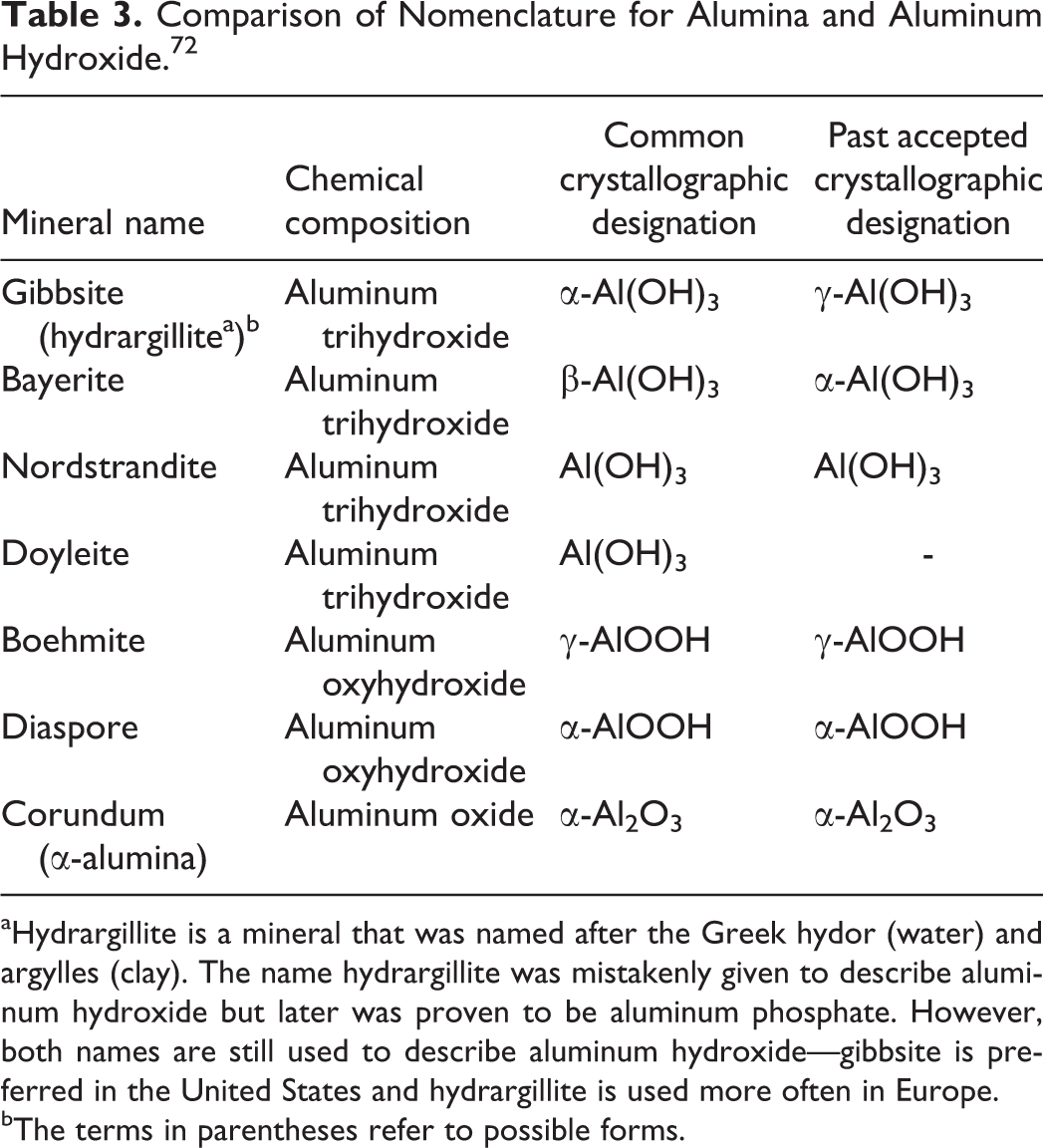
aHydrargillite is a mineral that was named after the Greek hydor (water) and argylles (clay). The name hydrargillite was mistakenly given to describe aluminum hydroxide but later was proven to be aluminum phosphate. However, both names are still used to describe aluminum hydroxide—gibbsite is preferred in the United States and hydrargillite is used more often in Europe.
bThe terms in parentheses refer to possible forms.
Physical and Chemical Properties
Alumina and aluminum hydroxide are white, insoluble solids (Table 4). Alumina is the third hardest naturally occurring substance after diamond and carborundum (SiC). 55 The presence of trace amounts of chromium and cobalt creates ruby and sapphire, respectively.
Chemical and Physical Properties of Alumina and Aluminum Hydroxide.
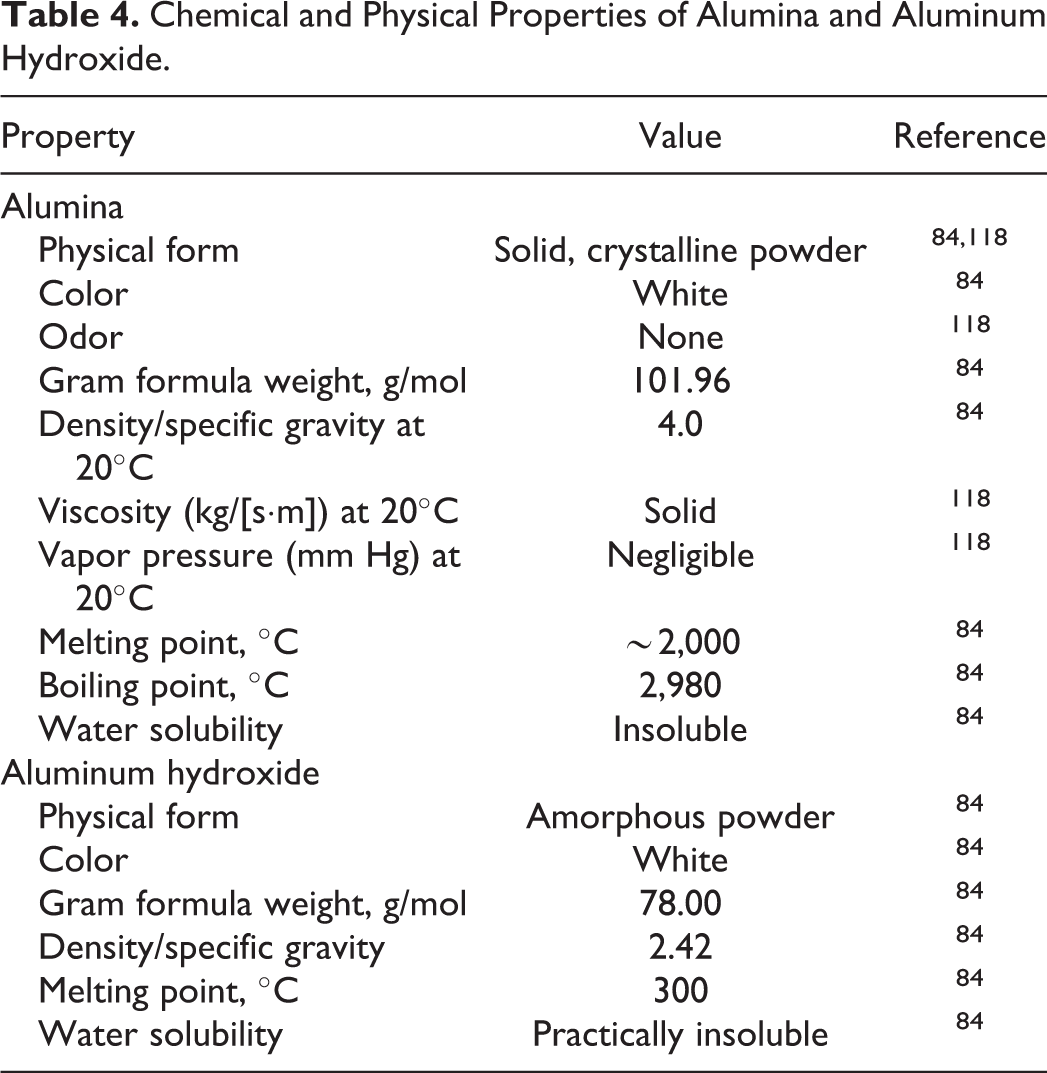
Aluminum compounds cannot easily be oxidized, and thus, atmospheric oxidations generally are not expected to occur. 73 All forms of aluminum hydroxide are amphoteric (eg, they can act as both acids and bases in solution). 74 Accordingly, aluminum hydroxides can serve as buffers to resist pH changes within the narrow pH range of 4 to 5. 75 Aqueous aluminum hydroxide gel has an effective pH of ∼6. 76
Method of Manufacture
Aluminum hydroxide is most commonly produced by aqueous alkaline extraction from bauxite ore, a method known as the Bayer process. 70 Alumina is then produced from the resultant aluminum hydroxide simply by vigorous heating to drive off water. 77
Impurities
Alumina balls used in artificial hips must meet the following specifications: grain size <5 μm and purity >99.7% aluminum oxide. 78 The maximum percentages for trace substances permitted are MgO, 0.2%; SiO2, 0.01%; CaO, 0.03%; Na2O, 0.02%; Fe2O3, 0.03%; and TiO2, 0.01%.
When used in OTC drugs as a color additive, alumina should contain no more than 0.5% insoluble matter in dilute hydrochloric acid. The following are the limits of impurities: lead (as Pb) ≤10 ppm, arsenic (as As) ≤1 ppm, mercury (as Hg) ≤1 ppm, and aluminum oxide (Al2O3) ≥50% (21 CFR 73.1010).
Use
Cosmetic
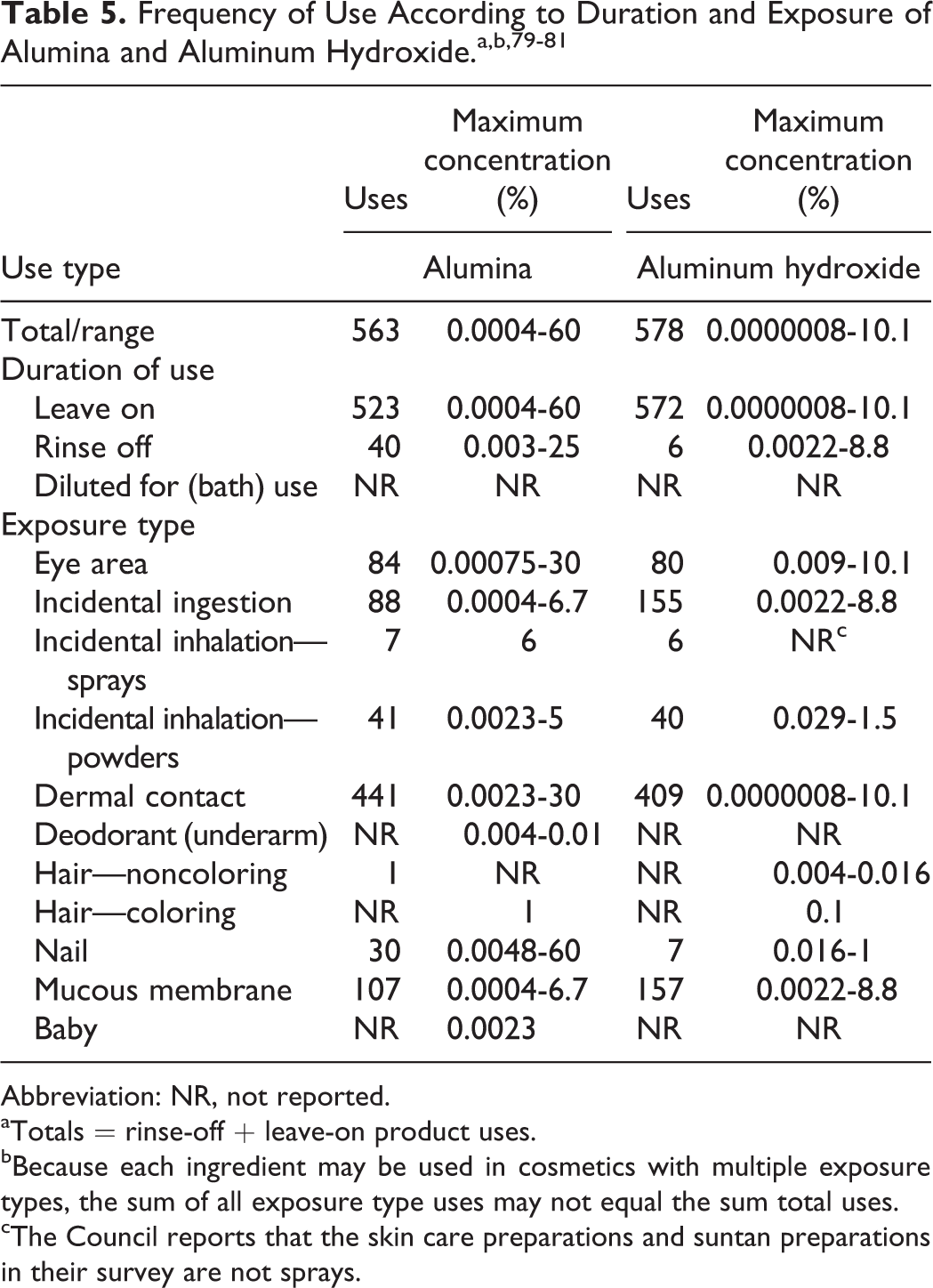
Data on ingredient usage are provided to the FDA Voluntary Cosmetic Registration Program (Table 5). 79 A survey of the maximum use concentrations has been conducted by the Personal Care Products Council. 80,81
Abbreviation: NR, not reported.
aTotals = rinse-off + leave-on product uses.
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
cThe Council reports that the skin care preparations and suntan preparations in their survey are not sprays.
Alumina was reported to be used in 523 leave-on products at concentrations up to 60% (in nail products). It is reported to be used in 40 rinse-off products. Formulations include 84 products used around the eye at concentrations up to 30%, 87 lipsticks up to 6.7%, and 104 skin care preparations up to 25%.
Aluminum hydroxide was reported to be used in 572 leave-on products up to 10.1% and 6 rinse-off products up to 8.8%. Formulations include 80 products used around the eye at up to 10.1%, 154 lipsticks up to 7%, oral hygiene products up to 8.8%, and 6 suntan preparations up to 0.9%.
Noncosmetic
Aluminum salts are incorporated into some vaccine formulations as an adjuvant to enhance the immune response to vaccination. 82 The aluminum compounds used in some US licensed vaccines are aluminum hydroxide, aluminum phosphate, alum (potassium aluminum sulfate), or mixed aluminum salts. Aluminum hydroxide may be used in vaccines up to 25 μg/L in large-volume parenteral drug products (21 CFR 201.323) and up to 1.25 μg in single-dose products (21 CFR 610.15), depending on the calculation method (Table 6).
US Government Regulations for Medical Devices, Drugs, and Other Regulated Uses of Alumina and Aluminum Hydroxide.

Abbreviations: PBP, pharmacy bulk packages; PDP, product development protocol.
The FDA evaluated the safety of aluminum hydroxide in OTC drugs (Table 6). The FDA stated that the oral maximum daily dose of an antacid containing aluminum hydroxide dried gel is 8 g (21 CFR 331.11). A chewable tablet of aluminum hydroxide:magnesium trisilicate (80:20 mg) was approved by the FDA. 3 Two other chewable tablets were approved with aluminum hydroxide:magnesium trisilicate doses of 80:20 mg and 160:40 mg. 4 Liquid suspensions of aluminum hydroxide are also used as antacids. 83
Aluminum hydroxide gel is approved for use in OTC skin protectant drug products as an active ingredient at 0.15% to 5%, with caution to consult a doctor for children younger than 6 months of age (Table 6; 21 CFR 247.10; 21 CFR 347.50).
The safety and effectiveness of aluminum hydroxide for use in OTC drugs has not been established for the treatment of diarrhea or the topical treatment of acne. Aluminum hydroxide has been approved for use in digestive aid drug products and preparations for treating diaper rash (21 CFR 346.14).
Alumina is used as an adsorbent, desiccant, and abrasive. 84 It is used as filler for paints and varnishes. It is also used in the manufacture of alloys, ceramic materials, electrical insulators and resistors, dental cements, glass, steel, and artificial gems. It is used in coatings for metals and other surfaces and as a catalyst or catalyst substrate for organic chemical reactions.
Alumina is approved as an indirect food additive by the FDA. 5 Aluminum hydroxide is considered GRAS as a direct food ingredient by the FDA (21 CFR 176.210, 177.1200, 177.2600). 6
There are many regulations and recommendations for aluminum compounds. Those that are informative for the purpose of this safety assessment are listed in Table 7.
Organizational Findings and Government Regulations With Regard to Aluminum and Related Compounds. 90
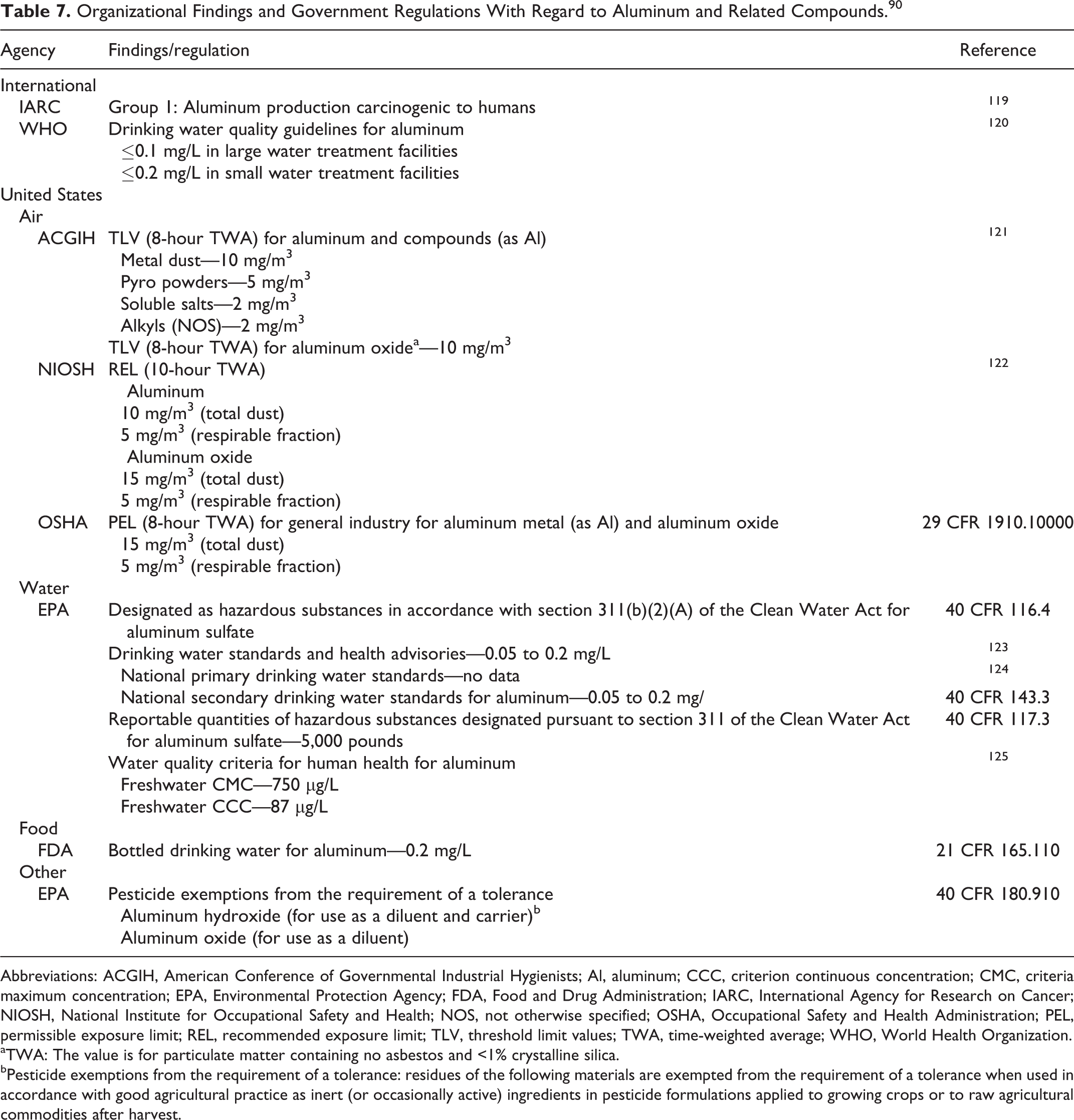
Abbreviations: ACGIH, American Conference of Governmental Industrial Hygienists; Al, aluminum; CCC, criterion continuous concentration; CMC, criteria maximum concentration; EPA, Environmental Protection Agency; FDA, Food and Drug Administration; IARC, International Agency for Research on Cancer; NIOSH, National Institute for Occupational Safety and Health; NOS, not otherwise specified; OSHA, Occupational Safety and Health Administration; PEL, permissible exposure limit; REL, recommended exposure limit; TLV, threshold limit values; TWA, time-weighted average; WHO, World Health Organization.
aTWA: The value is for particulate matter containing no asbestos and <1% crystalline silica.
bPesticide exemptions from the requirement of a tolerance: residues of the following materials are exempted from the requirement of a tolerance when used in accordance with good agricultural practice as inert (or occasionally active) ingredients in pesticide formulations applied to growing crops or to raw agricultural commodities after harvest.
Alumina in Medical Devices
Alumina has been approved by the FDA for use in medical devices. The alumina used in these devices must comply with ASTMF603-12, “Standard Specification for High-Purity Dense Aluminum Oxide for Medical Application.” 2
The FDA considered the safety of alumina when approving the following medical devices that contain this material: Color additives for polymethyl methacrylate (PMMA) bone cement and sutures. Endosseous dental implant abutments. Femoral bearing head of artificial hips.
Color Additives
Colors that contain alumina (eg, FD&C Blue #1 aluminum lake) are approved by the FDA to be used in color cosmetics, food, dietary supplements, drugs for internal and external use, and medical devices (ie, bone cement, surgical sutures). 85 The colors are created by applying the coloring material to an alumina substrate. Alumina has been approved as a color additive for OTC drugs (21 CFR 73.1010).
Ceramic Hip
The use of ceramic femoral heads (ie, CeramTec Alumina Heads, Alumina V40 Head) made of alumina/ceramic composites has been approved for use in hip joint replacements in humans. The materials conform to FDA’s “Guidance Document for the Preparation of Premarket Notifications for Ceramic Ball Hip Systems.” 78,86,87 One of these hip replacement products was reported to consist of ∼75% alumina, ∼25% zirconia, and <1% chromium oxide. 88
Other Devices
Alumina has been approved for use in endosseous dental implant abutments (Table 6; 21 CFR 872.3630). Alumina/ceramic composite is used to make internal stents for treating tracheomalacia. 89 These stents are implanted inside the trachea.
Toxicokinetics
Overview
Aluminum hydroxide, as measured by aluminum content, is poorly absorbed through either oral or inhalation routes and is essentially not absorbed dermally in healthy humans. 90 Orally, the bioavailable forms of aluminum hydroxide are absorbed at only approximately 0.1%. Ingested aluminum hydroxide is excreted as aluminum in the feces. Studies on uptake and elimination rates of aluminum hydroxide indicate that a near steady state is maintained in most healthy adults, with aluminum body burdens varying slightly up and down over time with an overall small rate of increase over a lifespan. High-level, long-term use of antacids containing aluminum hydroxide will cause levels of aluminum to increase in the blood and other tissues. The levels return to normal upon cessation of high-level exposure. Under certain atypical conditions (eg, poor renal function with increased aluminum load), levels of aluminum in the body may rise high enough to cause toxicity in humans.
Blood and tissue (liver, spleen, kidney, brain, bone) levels of aluminum from the ingestion of aluminum hydroxide (100, 281, 1,500, 2,000 mg/kg/d) were increased by concurrent oral administration of citric, lactic, malic, oxalic, or tartaric acids in rats. 91 –93
Dermal
Aluminum salts used in antiperspirants form hydroxide precipitates of denatured keratin in the cornified layer that surrounds and occludes the opening of sweat ducts. 94 This mechanism suggests that there is little or no dermal absorption of aluminum hydroxide or any other form of aluminum.
Oral—Nonhuman
Aluminum hydroxide
Bioavailability of orally administered [ 26 Al]aluminum hydroxide (in 2 mL water; pH 7) to male Wistar rats (n = 9) was 0.1%. 95 After administration, the rats were placed in metabolic cages and blood sampled at 20, 45, 60, 90, 150, and 300 minutes. The rats were then killed and necropsied.
The aluminum content returned to normal levels in the tissues of Sprague Dawley rats within 21 days after oral administration of aluminum hydroxide. 96 In the first study, the rats were fed a control diet containing 26 µg Al/g (n = 5) or 989 µg Al/g (n = 15) for 16 days. All rats were then fed the control diet. Five rats were killed and necropsied at the end of the test period and 7 and 21 days thereafter. The treatment group had increased aluminum in the tibiae–fibulae, ulnae–radii, leg muscles, and kidneys. At day 21, all aluminum content measurements were similar to controls.
This experiment was repeated with 9 additional rats (control) and 1,070 µg Al/g in the diet, and the rats were killed and necropsied at 0, 3, and 7 days after treatment. The increase in aluminum content in the test group returned to control levels by day 7. Ingestion of aluminum hydroxide had no effect on the levels of phosphorus, calcium, magnesium, zinc, and iron in the tissues examined.
Only 0.45% ± 0.47% of orally administered aluminum hydroxide (10,000 μmol/kg as concentrated aluminum hydroxide gel with 4 mL water by stomach tube) to renally intact rabbits (n = 10) was absorbed. 97 Renally impaired rabbits absorbed 0.36% ± 0.30%.
Oral—Human
Aluminum hydroxide
Orally administered aluminum hydroxide is poorly absorbed (<0.01%) in humans. 57,63 Using 26Al, the estimated aluminum absorption rates were 0.523%, 0.0104%, and 0.136% in 2 participants receiving a single dose of aluminum citrate, aluminum hydroxide, or aluminum hydroxide dissolved in an aqueous citrate solution, respectively. 98 The test materials were delivered to the stomach through a pediatric feeding tube. Blood was collected at 1, 4, and 14 hours. Feces and urine were collected for 6 days. The uptake of aluminum was greatest for the citrate form and least for aluminum hydroxide. The addition of citrate to aluminum hydroxide increased the 26Al uptake in both participants.
There was no appreciable increase in the amount of aluminum absorbed in participants (n = 8, 10, 7) administered with aluminum hydroxide (equal to 244, 976, or 1,952 mg Al in the form of antacid tablets; pH 9.2). 99 By measuring the amount of aluminum in the urine, the amount of aluminum absorbed was estimated to be 0.001%, 0.004%, and 0.007%, respectively. When the high dose was combined with orange juice (70 mL; pH 4.2) or citric acid (70 g in 1,000 mL distilled water; pH 2.4), absorption increased to 0.03% and 0.2%, respectively.
Intravenous
Aluminum hydroxide
The half-life of intravenously (IV) administered aluminum hydroxide (100 μmol/kg as concentrated aluminum hydroxide gel) in renally intact rabbits (n = 10) was 27 ± 13 hours. 97 In renally impaired rabbits, the half-life was 14 ± 5 hours. Blood was sampled at 24 hours and immediately prior to treatment and at approximately 5, 10, 20, 30, 45, and 60 minutes and 2, 4, 8, 12, 24, and 48 hours after treatment.
Toxicity
Repeated Dose
Oral—Animal
Aluminum hydroxide
When aluminum hydroxide (average 2,400 mg/kg/d in drinking water) was administered to Long Evans male hooded rat weanlings (n = 7 or 8) for 60 days, there was no reduction in cognitive abilities. 100 At necropsy, the highest concentration of aluminum in the brain was in the hippocampus. The test group had decreased weight gain compared to controls, possibly reflecting reduced water intake at the beginning of the test period. The rats were assessed with an open-field activity test biweekly. At the end of the test period, the rats were tested for muricidal behavior by placing an albino mouse with each of the rats. Only 1 treated rat exhibited the behavior.
When aluminum hydroxide (300 mg/kg in carboxymethyl cellulose) and aluminum hydroxide (100 mg/kg) plus citric acid (30 mg/kg) were orally administered to Long Evans rats (n = 10/sex), their learning ability was reduced as measured using a 4-T shaped labyrinth. 101 Control rats learned the way to the goal in an average of 5.1 ± 2.88 times versus 16.0 ± 2.98 and 13.2 ± 5.39 times for the 2 treatment groups, respectively. The aluminum content of the brains of the control rats at necropsy was 6.6 ± 3.01 ppm compared to 18.0 ± 10.20 ppm and 11.0 ± 4.80 ppm in the 2 treatment groups, respectively. There was also increased acetylcholinesterase activity in the aluminum hydroxide plus citric acid group. There was no increase in choline acetyltransferase activity in the brains of either group. No other clinical signs or abnormalities were reported.
Intraperitoneal—Animal
Aluminum hydroxide
Male Wistar rats (n = 12) exhibited decreased weight gain and initial feed efficiency when administered intraperitoneal (IP) aluminum hydroxide (80 mg/kg) 3 times/week for 6 months. 102 However, there were no differences in total feed intake. Aluminum hydroxide did not affect the peak growth rate or the time to reach maturity. The systemic calcium balance in the treated rats was altered, and there was an increase in the amount of calcium excreted in the feces. The rate of skeletal Ca++ accretion was decreased without changes in the bone calcium resorption.
Oral—Human
Aluminum hydroxide
There were no adverse effects observed when participants (n = 9 females, 4 males) were administered aluminum hydroxide (equal to 59 mg Al) 3 times daily for 6 weeks. 103 When compared to the control group (n = 3 females, 2 males), urinary Al was ∼10- to 20-fold greater during treatment. The authors stated that this indicated that ingestion of an Al-containing antacid is associated with Al absorption above that originating from food and drinking water. There were no differences in the lymphocyte subpopulations, lymphocyte proliferation, and in vitro immunoglobulin and interleukin production. There were no differences between groups in the immune parameters examined, except for a slightly smaller CD8+CD45R0+ population (primed cytotoxic T cells) in the test group compared to the referents.
Reproductive and Developmental Toxicity
Aluminum Hydroxide
When aluminum hydroxide (0, 66.5, 133, or 266 mg/kg in distilled water) was administered by gavage on gestation days 6 to 15 to Swiss mice (n = 20), there were no effects attributed to the test substance. 104 There were no differences in maternal weights, feed consumption, appearance, or behavior. There were no differences in the number of total implants, resorptions, number of live or dead fetuses, fetal size parameters, or sex distribution observed at necropsy. There were no differences observed at gross external, soft tissue, and skeletal examinations.
When aluminum hydroxide (384 mg/kg/d; n = 18), aluminum citrate (1,064 mg/kg/d; n = 15), or aluminum hydroxide (384 mg/kg/d; n = 19) plus citric acid (62 mg/kg/d) was orally administered to Sprague Dawley rats (during gestation day 6 to 15), there were no differences among the groups in pre- or postimplantation loss, number of live fetuses per litter, or sex ratio. 105 Fetal body weight was reduced and skeletal variations (delayed ossification of occipital bone and sternebrae; absence of xiphoids) were increased in the aluminum hydroxide plus citric acid group. The absence of xiphoids was also observed in the aluminum citrate group. The dams exhibited decreased weight gain in the aluminum hydroxide plus citric acid group during treatment but recovered and caught up to the other groups posttreatment. There was increased aluminum in the livers, bones, and placentas of the dams in the aluminum citrate group; there were no differences in aluminum content in the kidneys and brains. Aluminum accumulation was not detected in whole fetuses of the treated mice compared with those in the control group (n = 17), which were administered water.
Irritation
Aluminum Hydroxide
Aluminum hydroxide (10% wt/vol in 0.2% Tween-80) was not irritating when applied to the shaved backs of female TF1 strain albino mice (n = 5; 0.5 mL), New Zealand white rabbits (n = 3; 0.5 mL), and large white strain pigs (n = 2; 1.0 mL) for 5 consecutive days. 106 The test substance was applied uncovered. The animals were restrained until the substance was dry.
Clinical Use
Clinical Trials
There are multiple clinical trials of artificial hips (with alumina-on-alumina ball and socket contact or alumina ceramic hips), alumina/ceramic composite stents, and dental implants. There were no adverse reactions reported. None of the failures reported were attributable to adverse health effects of the alumina but were related to mechanical or implantation technical issues (Table 8).
Clinical Trials of Medical Devices Containing Alumina.

aSurvival refers to how long the prosthesis is functional.
In a review of 4 case studies of alumina ceramic hip implant failures, it was determined that all problems were due to design issues, implementation issues, or surgical issues. 107 None of the failures were attributed to adverse reactions to the alumina.
Summary
Alumina functions in cosmetics as an abrasive, absorbent, anticaking agent, bulking agent, and opacifying agent; aluminum hydroxide functions as a buffering agent, corrosion inhibitor, and pH adjuster.
The alumina and aluminum hydroxide produced for cosmetics are chemically equivalent to the materials used in color surgical sutures and to the alumina in other medical devices, as well as to the alumina in OTC drugs. The safety information submitted for those medical devices and drugs was reviewed by the FDA, including the results of acute and long-term biocompatibility testing for cytotoxicity, irritation and intracutaneous reactivity, sensitization, systemic toxicity, implantation effects, and hematocompatibility studies. The FDA found the data to be adequate and determined that alumina was safe and effective for use in hip and dental implants, as well as for coloring PMMA bone cement and surgical sutures. Alumina is approved as an indirect food additive. Aluminum hydroxide is GRAS as a direct food additive and safe for use in OTC drugs.
Alumina was reported to be used in 523 leave-on products at concentrations up to 60% (in nail products). It is reported to be used in 40 rinse-off products at concentrations up to 25%. Aluminum hydroxide was reported to be used in 572 leave-on products up to 10.1% (in eye products) and in 6 rinse-off products up to 8.8% (in oral hygiene products).
Alumina is used in color additives for sutures and is a material used in the construction of endosseous dental implant abutments and femoral bearing heads of artificial hips. In clinical trials of artificial hips, dental implants, and esophageal stents, all adverse effects were from mechanical or installation problems, not attributable to exposure to alumina.
Orally administered aluminum in aluminum hydroxide has low bioavailability and is excreted primarily in the feces; the systemically absorbed aluminum in aluminum hydroxide is excreted primarily in the urine.
Aluminum hydroxide orally administered to rats at 2,400 mg/kg had no effect on cognitive abilities, but 100 mg/kg administered with citric acid reduced the rat’s learning ability. Rats exhibited decreased weight gain and decreased initial feed efficiency when administered with aluminum hydroxide IP at 80 mg/kg 3 times/week for 6 months.
There were no effects on immunological parameters in humans when orally administered with aluminum hydroxide (equal to 59 mg Al) 3 times daily for 6 weeks. There were no reproductive effects in mice when orally administered with 266 mg/kg aluminum hydroxide during gestation days 6 to 15. There were also no reproductive effects in rats at 384 mg/kg aluminum hydroxide orally administered during gestation days 6 to 15. However, when administered to rats with citric acid, there was reduced weight gain in the dams and increased skeletal abnormalities in the pups. Aluminum hydroxide at 10% was not dermally irritating to mice, rabbits, or pigs (n = 2; 1.0 mL).
Discussion
The CIR Panel emphasized that this is a safety assessment of alumina and aluminum hydroxide and that these ingredients are not to be confused with elemental aluminum. The Panel noted that the scientific literature provides no plausible evidence linking Alzheimer disease or breast cancer to the use of these ingredients.
The Panel was not concerned with the potential for incidental ingestion of alumina when used in lipsticks or oral hygiene formulations. The amounts of aluminum ion that could be released in the digestive tract through the incidental ingestion of such cosmetic products are far below the levels of toxicological concern. There was no concern about dermal penetration or cosmetic application around the eye because these ingredients are practically insoluble and are not irritating to the skin.
The Panel discussed the issue of incidental inhalation exposure to alumina and aluminum hydroxide in cosmetic powders and fragrance preparations. These ingredients are reportedly used at concentrations up to 6% in cosmetic products that may be sprayed and up to 5% in other products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable in any appreciable amounts. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. The Panel considered other data available to characterize the potential for alumina and aluminum hydroxide to cause dermal irritation and systemic toxicity in multiple clinical trials of medical devices consisting of alumina. Alumina and aluminum hydroxide are insoluble in water, thus not systemically available. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Panel concluded that alumina and aluminum hydroxide are safe in the present practices of use and concentration described in this safety assessment.
Footnotes
Appendix A
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC.
Author Contributions
Lillian C. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. Ivan Boyer contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Lillian J. Gill, F. Alan Andersen, Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Ronald C. Shank, Thomas J. Slaga, and Paul W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.