Abstract
Cosmetic ingredients derived from Achillea millefolium function in cosmetics as skin-conditioning agents—miscellaneous, skin-conditioning agents—humectants, and fragrance ingredients. The Cosmetic Ingredient Review Expert Panel (Panel) reviewed relevant animal and human data to determine their safety in cosmetics and raised concerns about cosmetics containing linalool, thujone, quercetin, hydroquinone, or α-peroxyachifolid. Because final product formulations may contain multiple botanicals, each containing similar constituents of concern, formulators are advised to be aware of these components and to avoid reaching levels that may be hazardous to consumers. Additionally, industry was advised to use good manufacturing practices to limit impurities. The Panel concluded that achillea millefolium extract, achillea millefolium flower extract, and achillea millefolium flower/leaf/stem extract are safe in the present practices of use and concentration in cosmetics when formulated to be nonsensitizing.
Introduction
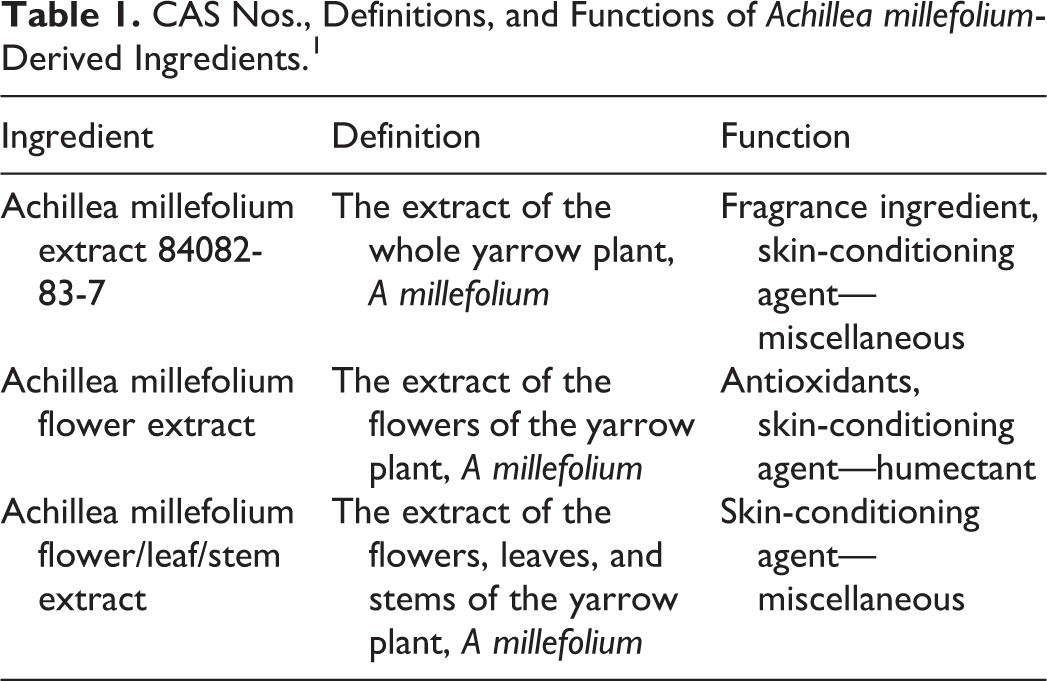
This amended report assesses the safety of Achillea millefolium (yarrow)-derived ingredients as used in cosmetics. The ingredients in this report are achillea millefolium extract, achillea millefolium flower/leaf/stem extract, and achillea millefolium flower extract. These ingredients function in cosmetics as skin-conditioning agents—miscellaneous, skin-conditioning agents—humectants, and fragrance ingredients (Table 1). 1
CAS Nos., Definitions, and Functions of Achillea millefolium-Derived Ingredients. 1
In 2001, Cosmetic Ingredient Review (CIR) published its original safety assessment of achillea millefolium extract as used in cosmetics, 2 concluding that the data reviewed were insufficient to determine the safety of this ingredient. Since that earlier review, additional data were provided and are presented with newly published data in this report. The Expert Panel (Panel) considered both sets of data when evaluating the safety of the A millefolium (yarrow)-derived ingredients.
The ingredient names are written as listed above, without italics and without abbreviations. When referring to the plant from which these ingredients are derived, the taxonomic practice of using italics and abbreviating the genus will be followed (eg, A millefolium).
Chemistry
Definition
The definitions and functions of A millefolium-derived cosmetic ingredients are listed in Table 1. Achillea millefolium is an herbaceous plant with characteristic narrow, oblong, multiple pinnate leaves. 3 The flower heads are small, made up of 5 white or pink florets with a few yellow tubular florets. The plant grows to ∼70-cm tall. Achillea millefolium is a member of the Asteraceae (formerly Compositae) family, which is known to be sensitizing. 4
Physical and Chemical Properties
UV absorbance of a 1% aqueous water achillea millefolium extract peaked at ∼260 nm with small shoulders at 270 and ∼320 nm. 5
Constituents
The constituents of A millefolium are listed in Table 2. A sample of an achillea millefolium extract (aqueous) mixture (water 73.5%, butylene glycol 20%, pentylene glycol 5%, achillea millefolium extract 1%, xanthan gum 0.5%) contained 3.37% polyphenols, 61.25% proteins, and 38.12% sugars.
5
An assay for nitrogen compounds of the same sample showed the possible presence of pipecolic acid,
Constituents of Achillea millefolium. 10

β-sitosterol, 3β-hydroxy-11α,13-dihydro-costunolide, desacetylmatricarin, leucodin, achillin, 8α-angeloxy-leucodin, and 8α-angeloxy-achillin were isolated from the flower heads of A millefolium plants. 6
The essential oil content of A millefolium was lower in the vegetative stage (0.13%) than the full bloom stage (0.34%). 7 Changes in the content of essential oil was found to be related to the maturation of the plant, with increasing amounts of monoterpenes in relation to the sesquiterpene as the plant matures. However, a clear trend could be detected only for the monoterpenic compounds with increasing levels of α- and β-pinene and α-thujone and decreasing levels of sabinene, borneol, and bornyl acetate. Previously reported as major compounds, chamazulene and germacrene D could be found only in trace amounts. The terpenic compounds (sesquiterpenic compounds such as β-bisabolene, α-bisabolol, and δ-cadinene) were detected in greater amounts when using solid-phase microextraction when compared to amounts found in steam-distilled samples.
Gas chromatography–mass spectrometry analysis of the essential oil of A millefolium identified 36 compounds constituting 90.8% of the total oil. Eucalyptol, camphor, α-terpineol, β-pinene, and borneol comprised 60.7% of the oil. 8
A comparison of the aerial parts of A millefolium plants that grew in the Indian Andes at altitudes of 1,600 and 2,850 m was conducted. 9 Of the constituents tested, there was considerable overlap in the content ranges of the major constituents; for example: β-pinene (10.6%-17.7%), 1,8-cineole (3.0%-15.1%), and borneol (0.2%-12.1%).
Constituents of concern
Achillea millefolium is reported to contain linalool (1-4,000 ppm), thujone, quercetin, α-peroxyachifolid, and hydroquinone (Table 2). 10 The potential adverse effects of exposures to these constituents are summarized in Table 3.
Constituents of Concern in Achillea Millefolium.

Method of Manufacture
Achillea millefolium extract is processed from the stem, leaves, and other aerial parts of the plant. 5 Under controlled temperature, time, pressure, and pH conditions (not provided), the plant parts are milled before an aqueous extraction. The extract is filtered then combined with butylene glycol (preservative) and xanthan gum. Other solvents (eg, alcohols, propylene, butylene glycol), or series of solvents and additives (preservatives), have also been reported to be used in the extraction process. 2
Use
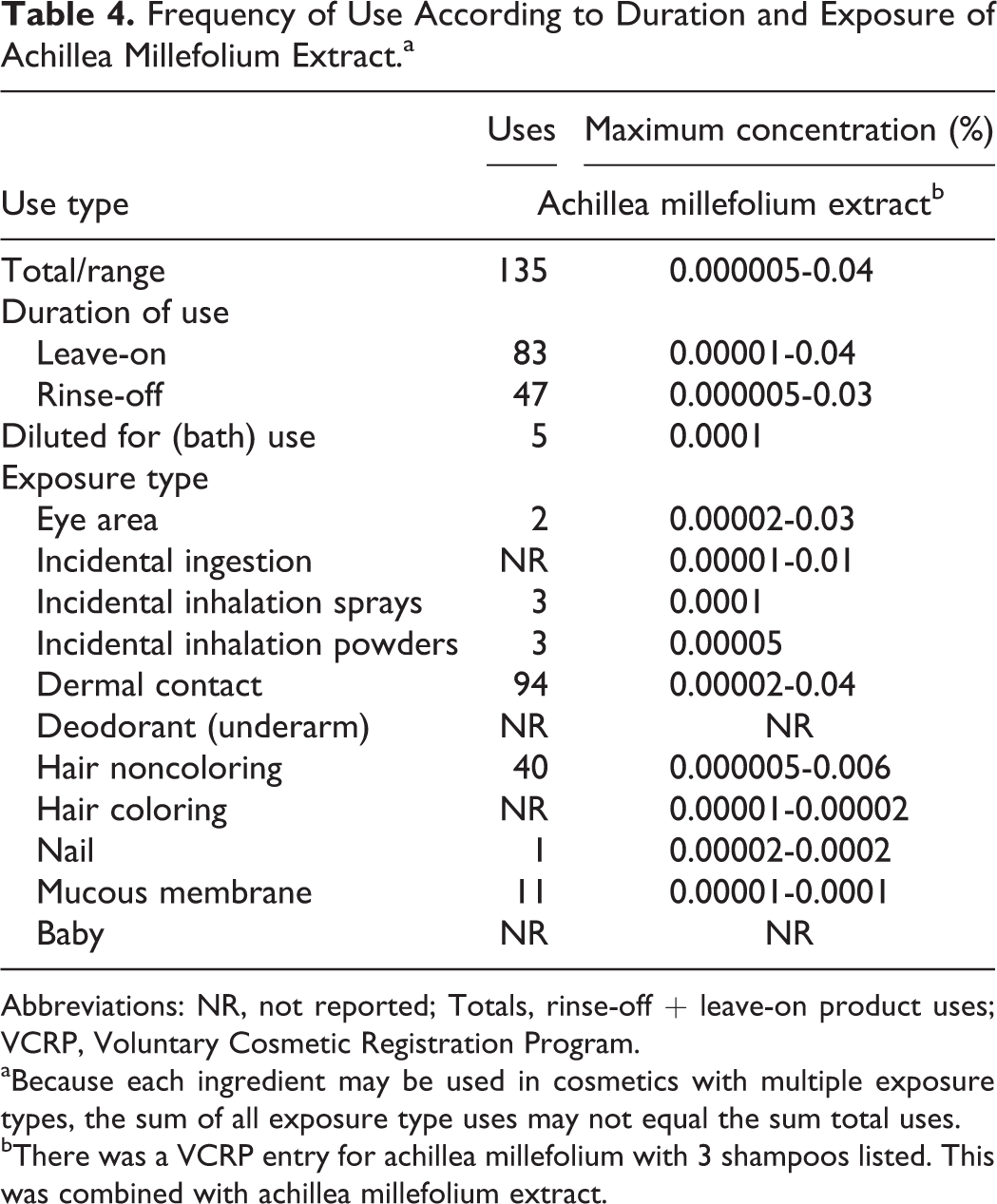
The safety of A millefolium (yarrow)-derived ingredients included in this safety assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported under cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database (Table 4). 11 Use concentration data are submitted by industry in response to surveys conducted by the Personal Care Products Council (Council) of maximum reported use concentrations, by product category. 12
Frequency of Use According to Duration and Exposure of Achillea Millefolium Extract.a
Abbreviations: NR, not reported; Totals, rinse-off + leave-on product uses; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
bThere was a VCRP entry for achillea millefolium with 3 shampoos listed. This was combined with achillea millefolium extract.
The VCRP had an entry for “achillea millefolium.” It was assumed that this entry was actually “achillea millefolium extract,” and data from that entry were combined with the extract data. 11
Achillea millefolium extract was reported to be used in 135 cosmetic products; these include 83 leave-on products and 47 rinse-off products. The extract was reported to be used up to 0.04% in leave-on products and up to 0.03% in rinse-off products. The extract is reported to be used in eye makeup products up to 0.03%, hair preparations up to 0.03%, lipstick up to 0.00001%, and skin care products up to 0.03%.
There was no use information reported for achillea millefolium flower extract or achillea millefolium flower/leaf/stem extract. Achillea millefolium extract was reported to be used in perfumes and face powders and could possibly be inhaled. This ingredient was reportedly used in face powders at concentrations up to 0.00005% and in perfumes up to 0.0001%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 12,13 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 14,15
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
No new toxicokinetics data were identified or made available for review. However, since botanical extracts are mixtures, toxicokinetic data would only have meaning for individual constituents of the extract, not for the extract as a whole.
Cytotoxicity
Achillea millefolium extract
A product containing an aqueous extract of A millefolium (5.0 μL/mL) was not cytotoxic to L5178Y cells after 3 hours of incubation. 16
Toxicological Studies
Acute Toxicity
Dermal
Acute dermal toxicity data on A millefolium (yarrow)-derived ingredients were not found in the published literature, and no unpublished data were submitted.
Oral—Nonhuman
Achillea millefolium extract
The oral LD50 of the mixture containing an aqueous extract of A millefolium was reported to be >2,000 mg/kg in female rats. 17 There were no mortalities when an aqueous A millefolium extract (10 g/kg; leaves, stalks, stems) was orally administered to male and female Wistar rats. 18
Intraperitoneal
Achillea millefolium extract
There were no mortalities when an aqueous A millefolium extract (3 g/kg; leaves, stalks, stems) was intraperitoneally (IP) administered to male and female Wistar rats. 18
Repeated dose toxicity
Dermal
Repeated dose dermal toxicity data on A millefolium (yarrow)-derived ingredients were not found in the published literature, and no unpublished data were submitted.
Oral—Nonhuman
Achillea millefolium extract
An aqueous A millefolium extract (0.3, 0.6, 1.2 g/kg/d; leaves, stalks, stems) orally administered to male and female Wistar rats (n = 10/sex) for 28 or 90 consecutive days produced no signs of toxicity. 18 The rats were observed for clinical signs and necropsied at the end of the treatment period or after a 30-day recovery period. All rats survived until the end of both treatment periods. Rats in both treatment time groups had mobility, reflex responses, muscular tone, and breathing patterns similar to rats in the control group treated with water. Weight gain was similar in all groups. There were no changes in organ weight observed, with the exception of a decrease in liver weights in females in the long-term/low-dose group, in males in the long-term/mid-dose group, and in both sexes in the mid-dose/long-term and high-dose/short- and long-term groups. Histopathological examination was unremarkable. The authors concluded that the rats had no treatment-related toxicological or histopathological abnormalities.
An ethanol (60%) multiherb mixture (20 mg/kg/d) that included achillea millefolium extract (3.5%; 0.7 mg/kg), orally administered to CBA/HZb mice (n = 6) for up to 6 months, caused no clinical signs. 19 Body weights were similar to controls. No differences were observed in the spleen, kidney, testicles, or liver weights when compared to controls. There was an increase in the serum activity of aspartate aminotransferase on day 7 compared to the activities at 24 hours of treatment. The serum activity of alanine aminotransferase and the concentration of cholesterol did not change during the treatment period. The authors concluded that the test mixture was not toxic to the liver, kidney, spleen, pancreas, testes, and lungs. This mixture also contained Vaccinium myrtillus, Taraxacum officinale, Cichorium intybus, Juniperus communis, Centaurium umbellatum, Phaseolus vulgaris, Morus nigra, Valeriana officinalis, and Urtica dioica.
Reproductive and Developmental Toxicity
Achillea millefolium Extract
An ethanol (45%) A millefolium extract (2.8 g/kg/d; 56 times the equivalent of a human dose of 50 mg/kg/d) was not maternotoxic when orally administered to Sprague Dawley rats (n = 5) but caused decreased body weights in fetuses. 20 The dams were orally administered the test material during gestational days (GDs) 1 to 8 or 8 to 15. The dams were killed on GD 20 and necropsied. There was no increase in preimplantation or postimplantation losses. Placental weights were increased in dams treated with achillea millefolium extract on GDs 8 to 15 compared to the water and ethanol controls and on GDs 1 to 8 compared to water control fetuses. Body weights were reduced in fetuses exposed to achillea millefolium extract on GDs 8 to 15 compared to the water controls. There was no difference in the incidence of external or internal malformations.
An aqueous A millefolium leaf extract (0.3, 0.6, 1.2 g/kg/d) orally administered to male Wistar rats (n = 10) for 90 days was not toxic nor caused any clinical or behavioral signs, but there was an increase in abnormal sperm in the males in the high-dose group. 21 The rats were killed and necropsied after 90 days, focusing on the testes, epididymis, prostate, and seminal vesicles (including coagulating glands). Daily sperm production and number of sperm were not affected. Body weight gain was similar in all groups.
An aqueous A millefolium extract (1.0, 5.0, and 10.0 mL/100 mL feed) fed to Oregon-R strain of fruit flies (Drosophila melanogaster) resulted in F1 offspring with a dose-dependent increase in the number of malformations. 22 There were no changes in the number of offspring.
Achillea Millefolium Flower Extract
An ethanolic A millefolium flower extract (200 mg/kg/d) IP administered to male Swiss albino mice (n = 6) for 20 days and an hydroalcoholic extract (300 mg/kg/d) orally administered for 30 days caused exfoliation of immature germ cells, germ cell necrosis, and seminiferous tubule vacuolization. 23 Mice in the treatment groups had an increased number of metaphases in the germ epithelium that might be due to cytotoxic substances or substances stimulating cell proliferation. Neither extract caused any differences in body weight gain or in testis and seminal vesicle weight.
An ethanolic A millefolium flower extract (200, 400, 800 mg/kg) IP or orally administered to male albino Wistar rats (n = 5) every other day for 22 days caused no changes in the low-dose IP group and the low- and mid-dose oral groups; however, there were abnormalities in the development of sperm in the mid- and high-dose groups. 24 There were scattered immature cells on basal membrane in seminiferous tubules in the IP mid-dose group. A decrease in cell accumulation and vacuolization in seminiferous tubules was observed. In the IP high-dose group, thickened seminiferous tubules on basal membrane, decreased cell accumulation in seminiferous tubule, severe disarrangement, degenerative cells, and severe decrease in sperm count were also observed. At the oral high dose, basal membranes were thickened and disarrangement in cells was observed. After a 40-day recovery period, normal physiology was observed in the low- and mid-dose groups compared with controls; however, there continued to be abnormal and damaged cells in the high-dose groups.
Genotoxicity
In Vitro
Achillea millefolium extract
In an Ames test using Salmonella typhimurium (TA98, TA100, TA102, TA1535, TA1537), the mixture containing an aqueous extract of A millefolium (0.06-5 μL) was not mutagenic with or without metabolic activation. 25
In 2 micronucleus tests using V79 cells, the mixture containing an aqueous extract of A millefolium (up to 15 000 μg/mL) was not clastogenic or aneugenic with or without metabolic activation. 26 In a gene mutation assay using mouse lymphoma L5178Y TK+/−, a product (up to 5 μL/mL) that contained an aqueous extract of A millefolium (0.5%) was not mutagenic with or without metabolic activation. 16 The controls had the expected results.
Irritation and Sensitization
Irritation
Ocular
Achillea millefolium extract
In an Epiocular Human Cell Construct assay, a product containing a mixture of an extract of A millefolium (0.00045%) was found to not have irritation potential. 27
Sensitization
Dermal—Nonhuman
Achillea millefolium extract
In a local lymph node assay (LLNA) using mice, a mixture containing an aqueous extract of A millefolium (25%, 50%, and 100% in dimethylformamide) was not a sensitizer. 28 Because this assay was performed on a mixture where the substance of interest was less than 80% of the mixture, the results do not permit a quantitative evaluation of the sensitization potential of achillea millefolium extract. 29
Dermal—Human
Achillea millefolium extract
In a patch test of participants with atopic dermatitis (n = 9), there were no positive reactions to A millefolium extract (1% in petrolatum). 30 Finn chambers were used and the test sites were observed on days 2 and 3.
In a human repeated insult patch test (HRIPT; n = 107), a face moisturizer with self-tanner product containing an extract of A millefolium (0.00045%; 0.2 mL) applied neat was not irritating or sensitizing. 31 The test material was applied to a 2-cm2 occlusive patch. There were transient, barely perceptible to mild nonspecific and specific responses, occasionally accompanied by mild/moderate edema or mild dryness in 9 test participants. Five participants had mild hyperpigmentation without erythema during the induction phase.
In an HRIPT (n = 108), a body splash product containing an extract of A millefolium (0.001133%) applied neat was not irritating or sensitizing. 32 The test material was applied to an occlusive patch and allowed to dry for 20 minutes before administration to the scapula area. There were no adverse events reported. In an HRIPT (n = 53) of a body lotion containing achillea millefolium extract (0.04%), it was concluded that the body lotion was neither irritating nor sensitizing. 33
Clinical Use
Case Studies
A 44-year-old woman with a history of rhinoconjunctivitis and asthma developed rhinitis, asthma, and urticaria symptoms after working seasonally with dried flowers for 6 years. 34 The skin prick test was positive for pollen from Cupressus sempervirens, Olea europaea, Lolium perenne, Salsola kali, Artemisia vulgaris, and Parietaria judaica as well as to cat and dog epithelium. Skin prick tests of aqueous extracts of the dried flowers were positive for A millefolium and safflower. An asthmatic response resulted from a specific inhalation bronchial challenge of A millefolium.
Summary
This amended safety assessment of A millefolium (yarrow)-derived ingredients examines new data submitted to address the needs of the insufficient data conclusion that the Panel reached in the previous safety assessment. These ingredients function in cosmetics as skin-conditioning agents—miscellaneous, skin-conditioning agents—humectants, and fragrance ingredients.
UV absorbance peaked at ∼260 nm with small shoulders at 270 and 320 nm using a 1% aqueous water extract. Achillea millefolium extract was reported to be used in 134 cosmetic products: 83 leave-on products and 47 rinse-off products with use concentrations up to 0.04% in body and hand skin care products. There was no use information reported for achillea millefolium flower extract and achillea millefolium flower/leaf/stem extract.
A millefolium extract was not cytotoxic to L5178Y cells. The oral LD50 for achillea millefolium extract is >2,000 mg/kg for rats; no mortalities were reported at 10 g/kg. There were no mortalities to rats administered IP 3 g/kg achillea millefolium extract.
An aqueous A millefolium extract was well tolerated by rats at up to 1.2 g/kg/d for up to 90 days. An ethanol extract of a herbal mixture that included A millefolium at 3.5% was not toxic to mice when administered orally for up to 6 months. There were no effects to the major organs.
Oral administration of an ethanol A millefolium extract was not maternotoxic at 2.8 g/kg/d when administered on GDs 1 to 8 but did cause reduced body weight in the fetuses when administered on GDs 8 to 15. There was no increase in external or internal malformations. The oral administration of an aqueous A millefolium leaf extract caused an increase in abnormal sperm at 1.2 g/kg/d in rats. Daily sperm production and number of sperm were not affected. Aqueous A millefolium extract caused an increase in the number of malformations in D melanogaster offspring.
Achillea millefolium flower extract administered IP caused damage to the reproductive organs of male mice at 300 mg/kg/d. An ethanolic A millefolium flower extract IP or orally administered to male rats every other day for 22 days caused no changes at 200 mg/kg IP and the 200 and 400 mg/kg oral groups. There were abnormalities in the development of sperm in the 400 and 800 mg/kg IP groups. After a 40-day recovery period, there continued to be abnormal and damaged cells in the 800 mg/kg groups.
Achillea millefolium extract was not genotoxic in an Ames test, 2 micronucleus tests, and a gene mutation assay. Achillea millefolium extract was not irritating to subjects with atopic dermatitis at 1%, and an Epiocular Human Cell Construct assay of a product that contained an extract of A millefolium at 0.00045% was negative for ocular irritation.
An aqueous A millefolium extract was not a sensitizer in an LLNA at 1%. Two products containing Achillea millefolium extract up to 0.001133% were not sensitizing in HRIPTs. A product containing achillea millefolium extract at 0.04% was neither irritating nor sensitizing. A woman was reported to develop an allergic reaction to A millefolium after working with dried flowers.
Discussion
The Panel considered the data from the original 2001 safety assessment of A millefolium (yarrow)-derived ingredients in addition to the new data presented in this report. Achillea millefolium extract is reported to be used up to 0.04% in body and hand creams, lotions and powders, and in eye lotion. An LLNA was performed on an aqueous A millefolium extract at 1%, and an HRIPT was performed at 0.04%. However, the Panel considered that LLNA testing of mixtures containing a small fraction of any constituent of concern may not reliably predict sensitization. The HRIPT data were available at use concentrations demonstrating an absence of dermal irritation and sensitization.
The Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit these impurities in the ingredient before blending into cosmetic formulations.
Cosmetic formulations may contain multiple botanical ingredients, each of which can contribute to the total concentration of constituents of concern. For example, the Panel was concerned that cosmetics containing linalool and α-peroxyachifolid may result in sensitization. Other constituents such as thujone, quercetin, and hydroquinone may result in carcinogenicity, genotoxicity, or depigmentation, respectively. The Panel noted that plants in the Asteraceae (formerly Compositae) family, such as A millefolium, are associated with dermal sensitization. Among the constituents of A millefolium plants are linalool (1-4,000 ppm), thujone, quercetin, α-peroxyachifolid, and hydroquinone. Linalool, a dermal sensitizer, is safe up to 4.3%. α-Peroxyachifolid is a dermal sensitizer at 0.01%. Thujone has been reported to cause neurological toxic effects; the suggested acceptable daily intake was not more than 3 to 7 mg/kg/d. Quercetin has been reported to have some genotoxic effects in in vitro assays but not in oral studies. Hydroquinone has been reported to cause skin depigmentation starting at 0.4%. These constituents are present in the plant. Data show that thujone and other constituents were not present in an extract. The levels of constituents of concern in the cosmetic ingredients derived from plants can vary widely and may even be undetectable in the ingredients, depending on the growing conditions of the plant, the methods of manufacturing of the ingredient, and other factors. The maximum concentration of use of A millefolium-derived extracts in cosmetics was reported to be 0.04%.
The use of other botanical ingredients that may contain constituents of concern (eg, potential sensitizers) in combination with A millefolium ingredients in a single formulation could result in exposures that exceed levels of concern. Thus, cosmetic products containing multiple botanical ingredients should be formulated to ensure that total exposures to such constituents remain below the levels of toxicological concern when used as intended. Manufacturers should employ good manufacturing practices to ensure that constituents of concern are below the levels of toxicological concern, including sensitization. It is important for formulators to be aware that even though the assays in this report revealed no sensitizers, these ingredients may still contain sensitizers, such as sesquiterpene lactones. Products that contain such sensitizers need to be formulated at nonsensitizing levels.
The Panel discussed the issue of incidental inhalation exposure from perfumes and face powders. There were no inhalation toxicity data available. However, the Panel believes that the sizes of a substantial majority of the particles of the products containing these ingredients, as manufactured, are larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. However, these ingredients are reportedly used at concentrations up to 0.0001% in cosmetic products that may be aerosolized and up to 0.00005% in other products that may become airborne.
The Panel noted that 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the very low concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products are available at http://www.cir-safety.org/cir-findings.
The Panel considered other data available to characterize the potential for A millefolium-derived ingredients to cause irritation, sensitization, reproductive and developmental toxicity, and genotoxicity. They noted the lack of systemic toxicity at high doses in acute and subchronic oral exposure studies, no irritation or sensitization at use concentrations in tests of dermal and ocular exposure, as well as the absence of genotoxicity in an Ames test, 2 micronucleus tests, and a gene mutation assay. Although A millefolium-derived ingredients caused an increase in abnormal sperm and damage to male organs in rats, these effects were observed at levels much greater than any from exposure to cosmetics.
Conclusion
The CIR Panel concluded that achillea millefolium extract, achillea millefolium flower extract (Note 1), and achillea millefolium flower/leaf/stem extract (Note 1) are safe in the present practices of use and concentration in cosmetics when formulated to be nonsensitizing.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author contributions
L. C. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. L. J. Gill, F. A. Andersen, W. F. Bergfeld, D. V. Belsito, R. A. Hill, C. D. Klaassen, D. C. Liebler, J. G. Marks, R. C. Shank, T. J. Slaga, and P. W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
