Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of methyl glucose polyethers and esters which function in cosmetics as skin/hair-conditioning agents, surfactants, or viscosity increasing agents. The esters included in this assessment are mono-, di-, or tricarboxyester substituted methyl glucosides, and the polyethers are mixtures of various chain lengths. The Panel reviewed available animal and clinical data, including the molecular weights, log Kows, and other properties in making its determination of safety on these ingredients. Where there were data gaps, similarities between molecular structures, physicochemical and biological characteristics, and functions and concentrations in cosmetics allowed for extrapolation of the available toxicological data to assess the safety of the entire group. The Panel concluded that there likely would be no significant systemic exposure from cosmetic use of these ingredients, and that these ingredients are safe in cosmetic formulations in the present practices of use and concentration.
Introduction
This report assesses the safety of methyl glucose polyethers and esters which are defined in the International Cosmetic Ingredient Dictionary and Handbook as cosmetic ingredients.
1
Data on methyl glucoside (methyl α-
Chemistry
Definition and Structure
The definitions and functions of the methyl glucose polyethers and esters reviewed in this safety assessment are included in Table 1. Because each of these ingredients represents a multitude of substitution arrangements, numbers of substitutions, or chain lengths, one structure would not fully represent these ingredients, therefore, an “idealized” structure that gives the best practical representation of the actual structure and available molecular weight data are included in Table 2.
Definitions and Functions of the Ingredients in This Safety Assessment.a, 1
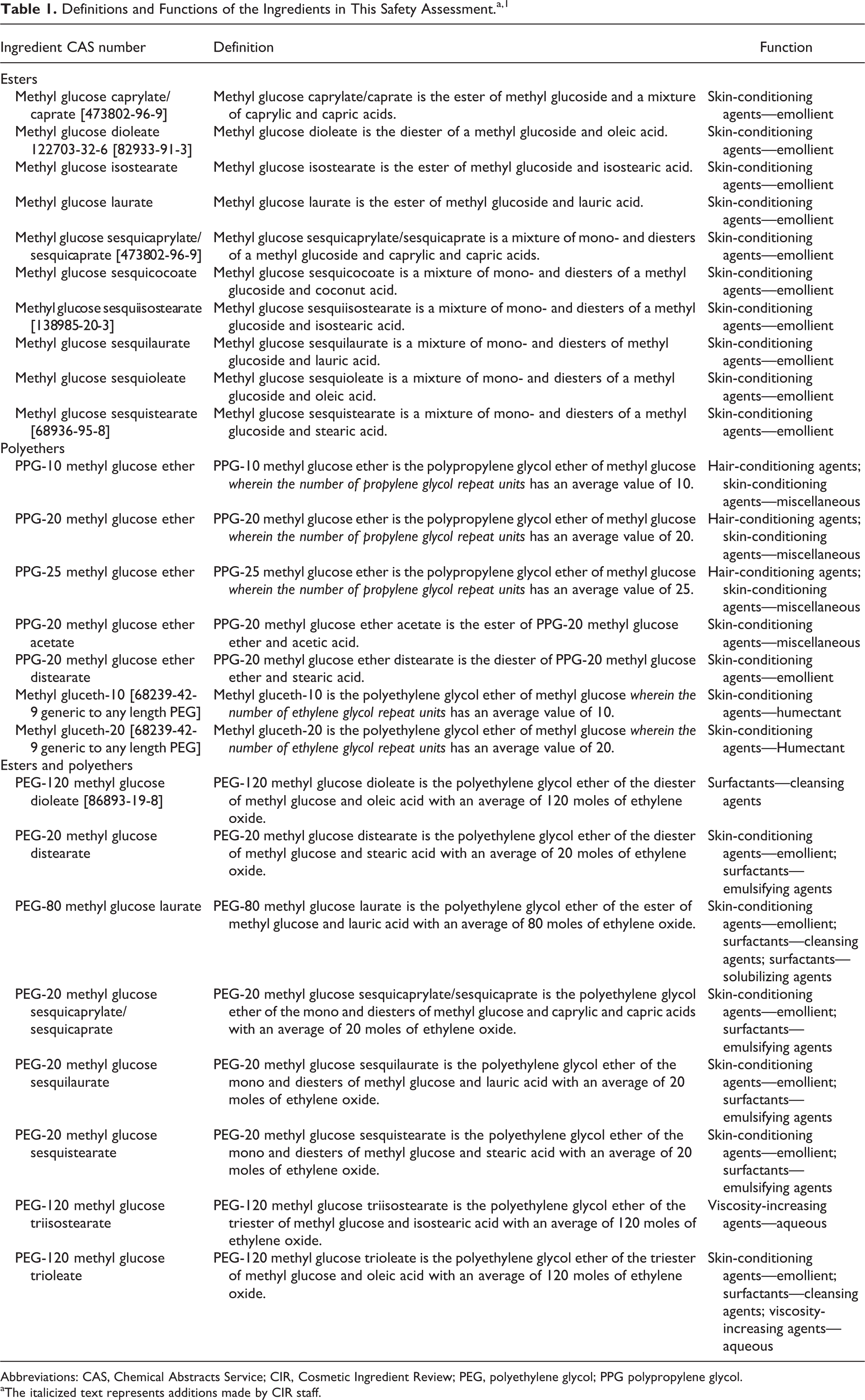
Abbreviations: CAS, Chemical Abstracts Service; CIR, Cosmetic Ingredient Review; PEG, polyethylene glycol; PPG polypropylene glycol.
aThe italicized text represents additions made by CIR staff.
Idealized Structures, Molecular Weights, and LogPow’s of Ingredients in This Safety Assessment.
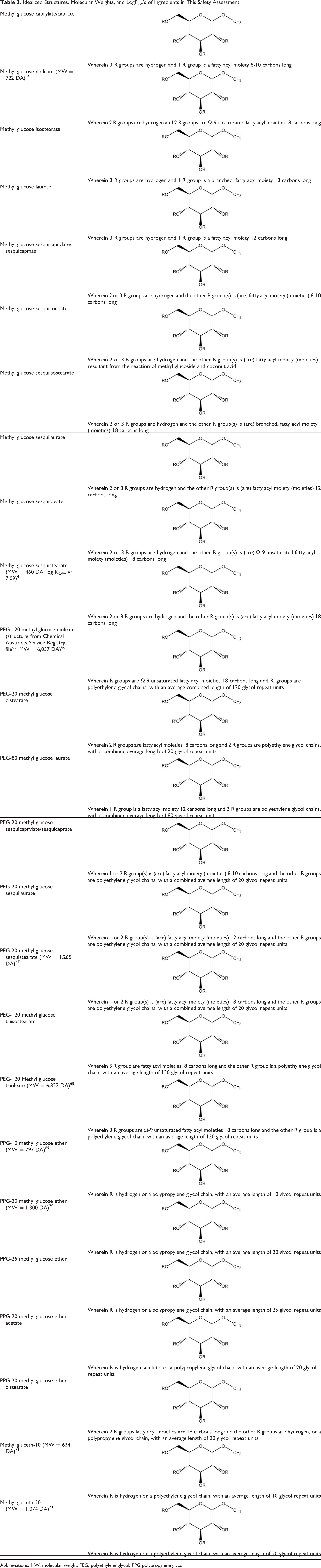
Abbreviations: MW, molecular weight; PEG, polyethylene glycol; PPG polypropylene glycol.
The ingredients in this group each have a methyl glucoside core. Glucose is a common, naturally occurring monosaccharide. Glucosides are those glucose molecules modified at the anomeric alcohol functional group. Accordingly, methyl glucosides are those ingredients composed of glucose molecules with a methyl ether group at the anomeric carbon (Figure 1). These ingredients vary by the identity and quantity of modifications at the other glucose alcohol functional groups, modified via traditional esterification or polyetherification techniques.
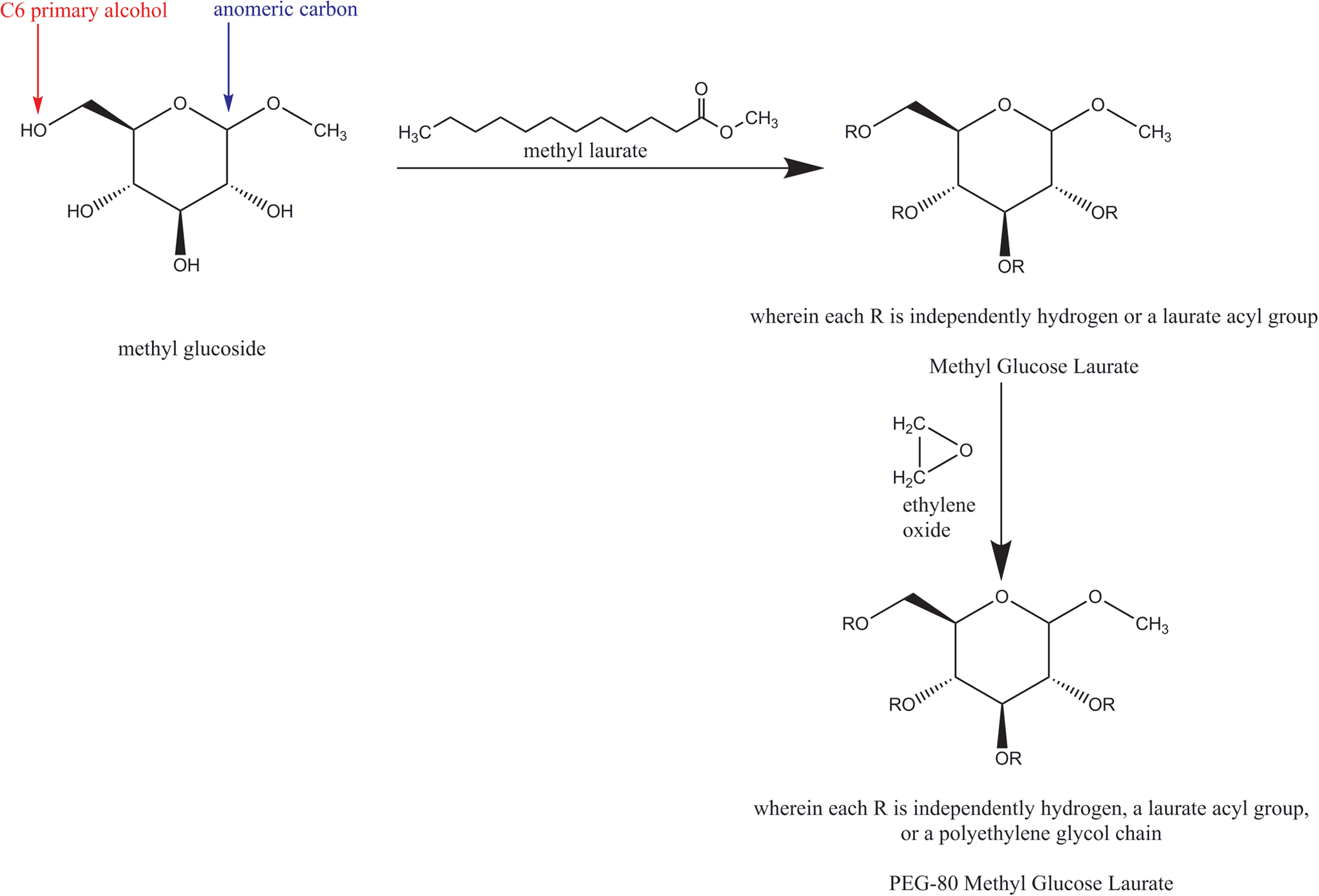
Methyl glucose laurate synthesis and PEG-80 methyl glucose laurate synthesis.
The ester ingredients are mono-, di-, or tricarboxyester substituted methyl glucosides. Those ingredients, wherein a specific degree of esterification is not provided in the definition, are expected to be monoesters.
The polyether ingredients in this report consist of polyethylene glycol (PEG) or polypropylene glycol (PPG) ethers. The number of polyether repeat units specified for each ingredient has 2 possible and distinct meanings, which are recited in the definitions. Under one meaning, the number represents a mixture of polyether chain lengths with that number marking the average, and presumably having a narrow distribution, at one alcohol site (eg, PPG-10 methyl glucose ether). Under the other meaning, the number represents a mixture of polyether chain lengths that are possibly distributed across one or more glucose alcohol sites, with that number marking the sum of all the polyether chain lengths in that molecule (eg, PEG-120 methyl glucose dioleate).
Physical and Chemical Properties
Polypropylene glycol-20 methyl glucose ether acetate is soluble in oils and organic solvents, but is essentially insoluble in water.
2
A log Kow of 13.98 has been reported for
Specifications for methyl glucoside-coconut oil ester (methyl glucose sesquicocoate) as a direct food additive are as follows 5 : acid number (10-20), hydroxyl number (200-300), pH (4.8-5.0, for 5% aqueous), and saponification number (178-190).
Physical and chemical properties associated with methyl glucose polyether and ester trade name materials are included in Tables 3, 4, and 5.
6
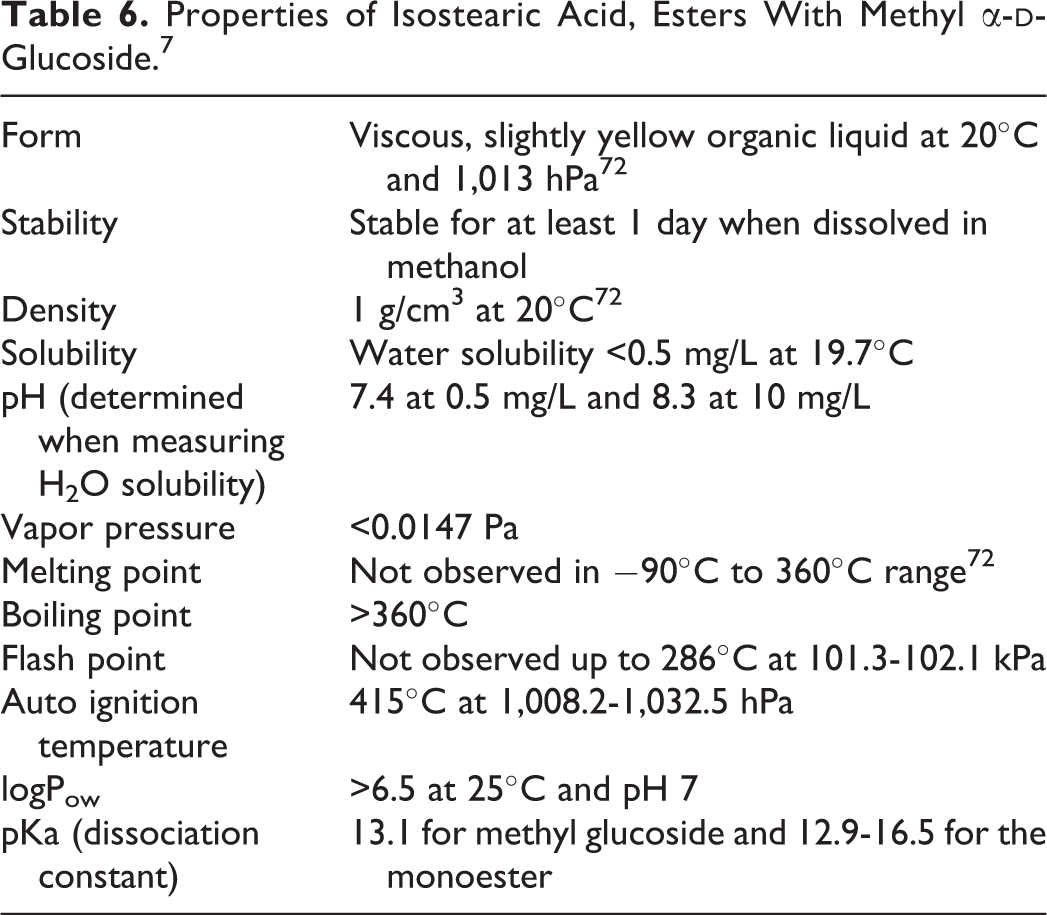
Studies on most of these trade name materials are included in the toxicology section of this article. Additionally, the chemical and physical properties of isostearic acid (esters with methyl α-
Physical Properties of Methyl Glucose Polyether and Ester Trade Name Materials. 6

Abbreviations: PEG, polyethylene glycol; PPG polypropylene glycol.
Properties From Technical Data Sheets on Methyl Glucose Polyether and Ester Trade Name Material.a, 5
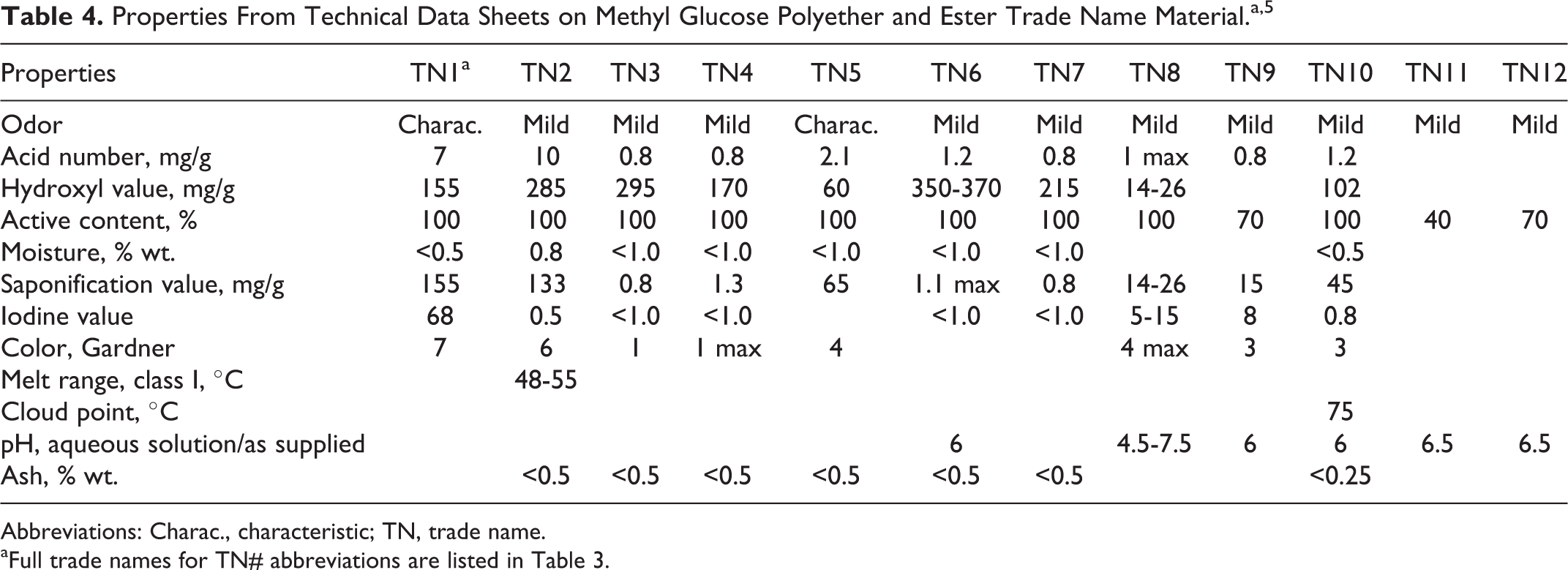
Abbreviations: Charac., characteristic; TN, trade name.
aFull trade names for TN# abbreviations are listed in Table 3.
Specifications for Methyl Glucose Polyether and Ester Trade Name Materials. 5
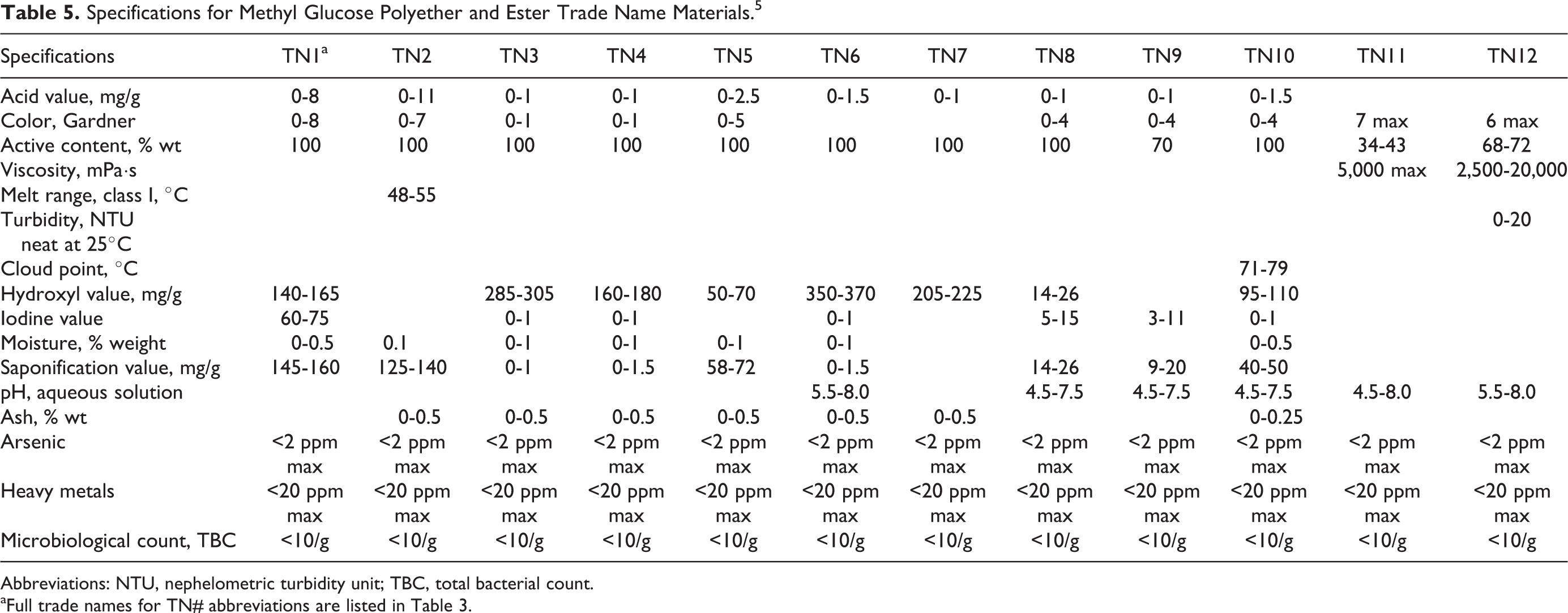
Abbreviations: NTU, nephelometric turbidity unit; TBC, total bacterial count.
aFull trade names for TN# abbreviations are listed in Table 3.
Properties of Isostearic Acid, Esters With Methyl α-
Method of Manufacture
Methyl glucoside (methyl α-
Manufacture of methyl glucoside esters, such as methyl glucose caprylate/caprate, methyl glucose dioleate, methyl glucose isostearate, methyl glucose laurate, methyl glucose sesquicaprylate/sesquicaprate, methyl glucose sesquicocoate, methyl glucose sesquiisostearate, methyl glucose sesquilaurate, methyl glucose sesquioleate, and methyl glucose sesquistearate, is typically achieved via transesterification of an appropriate fatty acid methyl ester (eg, methyl laurate to get methyl glucose laurate) with methyl glucoside (releasing methanol as a by-product). 8 –13 However, esterifications via a variety of other classical techniques, such as reacting the free fatty acids with methyl glucoside and a catalyst, are also known methods of manufacture for these ingredients. 14,15 Under most conditions, the primary alcohol group at C6 of the methyl glucoside core is the most reactive to esterification and is the first site to be substituted.
The polyether methyl glucosides, such as PPG-10 methyl glucose ether, PPG-20 methyl glucose ether, PPG-25 methyl glucose ether, methyl gluceth-10, and methyl gluceth-20, are typically manufactured by reaction of methyl glucoside with the required amount of the appropriate epoxide (eg, propylene oxide is used to produce PPG-10 methyl glucose; ethylene oxide is utilized to produce methyl gluceth-10). 10 For those ingredients with both ester and polyether groups, such as PEG-120 methyl glucose dioleate, PEG-20 methyl glucose distearate, PEG-80 methyl glucose laurate, PEG-20 methyl glucose sesquicaprylate/sesquicaprate, PEG-20 methyl glucose sesquilaurate, PEG-20 methyl glucose sesquistearate, PEG-120 methyl glucose triisostearate, PEG-120 methyl glucose trioleate, PPG-20 methyl glucose ether acetate, and PPG-20 methyl glucose ether distearate, these same methods are utilized, sequentially. An example would be PEG-80 methyl glucose laurate, which is produced in 2 steps: (1) esterification of methyl glucoside with methyl laurate, followed by (2) polyetherification with ethylene oxide.
Impurities
The following impurities data on methyl glucose polyethers and esters are included in Tables 4 and 5: ash (≤0.5% wt.), arsenic (<2 ppm), and heavy metals (<20 ppm).
Use
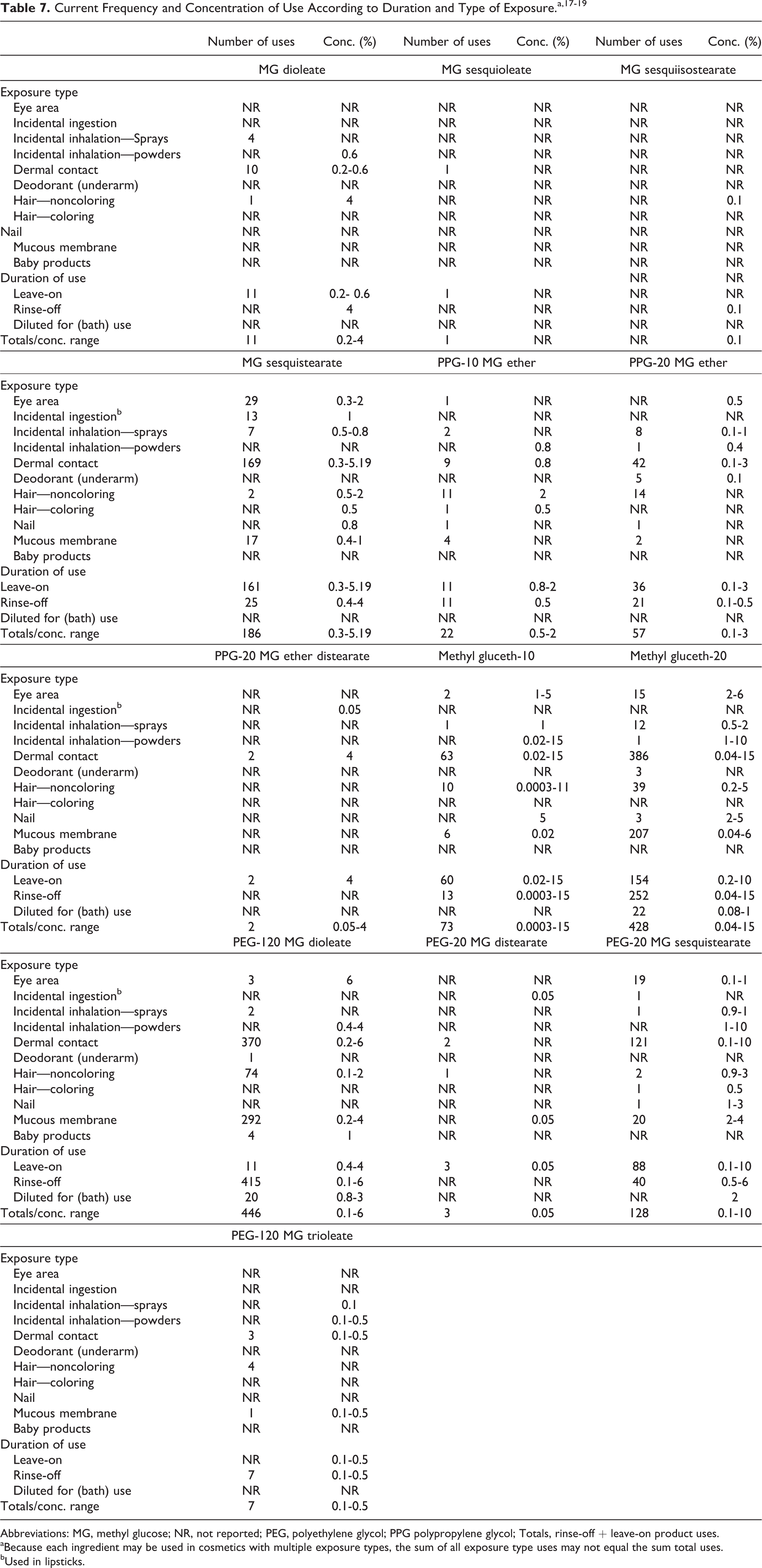
Cosmetic
The methyl glucose polyethers reportedly function as skin and hair-conditioning agents, whereas, the methyl glucose esters reportedly function only as skin-conditioning agents in cosmetic products. 1 Ingredients classified as both methyl glucose polyethers and esters based on their chemical structures function as skin-conditioning agents, surfactants, and viscosity-increasing agents in cosmetic products. According to the information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2013 (summarized in Table 7), methyl glucose dioleate, methyl glucose sesquioleate, methyl glucose sesquistearate, PPG-10 methyl glucose ether, PPG-20 methyl glucose ether, PPG-20 methyl glucose ether distearate, methyl gluceth-10, methyl gluceth-20, PEG-120 methyl glucose dioleate, PEG-20 methyl glucose distearate, PEG-20 methyl glucose sesquistearate, and PEG-120 methyl glucose trioleate are being used in cosmetic products. 17 A survey of ingredient use concentrations that was conducted by the Personal Care Products Council (Council) in 2013 (Table 7) indicates that the polyethers and esters are being used at concentrations up to 15% and 4%, respectively. 18,19 The maximum use concentration was 15% for methyl gluceth-10 and methyl gluceth-20 used in rinse-off skin-cleansing products. For leave-on products, the 15% maximum use concentration was for methyl gluceth-10 used in face and neck creams, lotions, and powders (not sprays). The Council survey results also provided a use concentration for the newly reported VCRP use(s) of methyl glucose sesquistearate (1% maximum use concentration), but not PEG-20 methyl glucose sesquistearate, in lipsticks. Additionally, a maximum use concentration of 0.05% for PEG-20 methyl glucose distearate in lipsticks was reported in this survey. Uses of methyl glucose sesquistearate and PEG-20 methyl glucose sesquistearate, but not PEG-20 methyl glucose distearate, in lipsticks were also reported in FDA’s VCRP.
Abbreviations: MG, methyl glucose; NR, not reported; PEG, polyethylene glycol; PPG polypropylene glycol; Totals, rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
bUsed in lipsticks.
Cosmetic products containing methyl glucose polyethers and esters may be applied to the skin and hair, or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Polyethylene glycol-20 methyl glucose sesquistearate (aerosol hair sprays), methyl gluceth-10 (body and hand sprays), and methyl gluceth-20 (pump hair sprays, hair grooming pump sprays, hair preparation spray gel, moisturizing sprays, and indoor tanning aerosol preparations) are used in products that are sprayed (highest maximum use concentration = 2%). Additionally, methyl glucose dioleate, methyl glucose sesquistearate, PPG-10 methyl glucose ether, PPG-20 methyl glucose ether, methyl gluceth-10, methyl gluceth-20, PEG-120 methyl glucose dioleate, PEG-20 methyl glucose sesquistearate, and PEG-120 methyl glucose trioleate may be used in face/body powders (highest maximum use concentration = 15%, for methyl gluceth-10; lower values for remaining ingredients). Because these ingredients are used in aerosol/pump hair sprays or powders, they could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm, compared with pump sprays. 20 –23 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 20,21
Noncosmetic
Methyl glucoside-coconut oil ester (methyl glucose sesquicocoate) is listed among the food additives permitted for direct addition to food for human consumption. 5 This methyl glucose ester is used as an aid in crystallization of sucrose and dextrose at a level not to exceed the minimum quantity required to produce its intended effect. It is also used as a surfactant in molasses, at a level not to exceed 320 ppm. As an indirect food additive, methyl glucose sesquicocoate may be safely used as a processing aid (filter aid) in the manufacture of starch, including industrial starch-modified, intended for use as a component of articles that contact food. 24
Toxicokinetics
Data on the absorption (including percutaneous absorption), distribution, metabolism, and excretion of methyl glucose polyethers and esters were not found in the published literature, and unpublished data were not provided.
Methyl Glucoside
The pulmonary absorption of lipid-insoluble α-methyl-
Methyl-
Toxicology
Acute Toxicity
Oral
Methyl glucose dioleate
The acute oral toxicity of methyl glucose dioleate was evaluated using 10 Wistar-derived albino rats (5 males, 5 females). 28 The animals were dosed orally (by gavage; dose = 5 g/kg body weight), observed for 14 days, and then killed. Complete gross necropsy was performed on each animal. The test material was not toxic when administered orally (LD50 > 5 g/kg).
Methyl glucose sesquistearate
Methyl glucose sesquistearate was evaluated in an acute oral toxicity study involving albino rats (5 males, 5 females). 29 The animals were observed for 14 days after dosing. There was no evidence of gross pathology at necropsy of surviving animals. An LD50 of > 5 g/kg was reported.
In another study, the acute oral toxicity of methyl glucose sesquistearate in rats was evaluated according to the Organization for Economic Cooperation and Development (OECD) 401 test protocol. Additional study details were not provided. An LD50 of >2,000 mg/kg was reported.
Polypropylene glycol-10 methyl glucose ether
An LD50 of > 13.8 mL/kg is reported in an acute oral toxicity study of PPG-10 methyl glucose ether in rats (number and strain not stated). 30 Details relating to the test protocol were not stated.
Polypropylene glycol-20 methyl glucose ether
The acute oral toxicity of PPG-20 methyl glucose ether was evaluated using rats (number and strain not stated). 31 Details relating to the test protocol were not stated. An LD50 of >3 mL/kg was reported.
Polypropylene glycol-20 methyl glucose ether distearate
An LD50 of >5 g/kg was reported for PPG-20 methyl glucose ether distearate in a study involving rats (number and strain not stated). 32 Details relating to the test protocol were not stated.
Polyethylene glycol-120 methyl glucose dioleate
An LD50 of >5 g/kg was also reported for PEG-120 methyl glucose dioleate in a study involving rats (number and strain not stated). 33 Details relating to the test protocol were not stated.
Polyethylene glycol-20 methyl glucose sesquistearate
The acute oral toxicity of PEG-20 methyl glucose sesquistearate was evaluated using 10 Wistar-derived albino rats (5 males, 5 females). 34 The animals were dosed orally (by gavage; dose = 5 g/kg body weight), observed for 14 days, and then killed. Complete gross necropsy was performed on each animal. Gross changes were not observed in any of the animals, and the LD50 was >5 g/kg.
Polyethylene glycol-120 methyl glucose trioleate
The acute oral toxicity of PEG-120 methyl glucose trioleate (and) propylene glycol (and) was evaluated using rats (number and strain not stated). None of the animals died, and the LD50 and no observable effect level (NOEL; for systemic toxicity) were > 12g/kg. 35 Because the test material contains 37% to 43% PEG-120 methyl glucose trioleate, the LD50 for PEG-120 methyl glucose trioleate in this study was actually >4.44 to 5.16 g/kg. 36
Isostearic acid, esters with methyl α-d -glucoside
In an acute oral toxicity study performed according to the OECD TG 423 protocol, a single oral dose (gavage
Dermal
Polyethylene glycol-120 methyl glucose trioleate
The acute dermal toxicity of PEG-120 methyl glucose trioleate (and) propylene glycol (and) water was evaluated using rats (number and strain not stated). A single dose of the test substance (12 g/kg) was applied under occlusion. None of the animals died, and the LD50 and NOEL (for systemic toxicity) were >12 g/kg. 35 Because the test material contains 37% to 43% PEG-120 methyl glucose trioleate, the actual LD50 for PEG-120 methyl glucose trioleate in this study was >4.44 to 5.16 g/kg. 36
Repeated Dose Toxicity
Isostearic acid, esters with methyl α-d -glucoside
A combined repeated dose toxicity study with a reproduction/developmental toxicity screening test was carried out according to the OECD 422 test protocol.
7
Isostearic acid, esters with methyl α-
There were no treatment-related changes in mortality, clinical appearance, functional observations, body weight, food consumption, and macroscopic and microscopic examination found in adult rats. At a dosage of 1,000 mg/kg/d, the parental NOEL was 150 mg/kg/d. The parental no observed adverse effect level (NOAEL) was defined as ≥1,000 mg/kg/d, based on the findings observed at 1,000 mg/kg/d. It was noted that the findings at this dose level were not considered adverse and were without any corroborative findings, such as histopathological changes. Results relating to reproductive and developmental toxicity are included in that section of the report. 7
Ocular Irritation
Methyl glucose dioleate
The ocular irritation potential of methyl glucose dioleate (as 20% gravimetric mineral oil suspension) was evaluated in the Draize test using 6 New Zealand albino rabbits. 28 The test material (0.1 mL; dose not stated) was instilled into 1 eye, and the contralateral eye served as the untreated control. The eyes were not rinsed after instillation. Reactions were scored for up to 72 hours postinstillation. It was concluded that the test material was not an ocular irritant under the conditions of this study.
Methyl glucose sesquistearate
The ocular irritation potential of undiluted methyl glucose sesquistearate was evaluated in the Draize test using 6 New Zealand albino rabbits. 29 The preceding test procedure was used. It was concluded that the test material was nonirritating to the eyes of rabbits.
Polypropylene glycol-10 methyl glucose ether
The ocular irritation potential of 100% PPG-10 methyl glucose ether was evaluated in rabbits (number and strain not stated) using the Draize test. 30 The test substance was classified as a mild transient irritant.
Polypropylene glycol-20 methyl glucose ether
The ocular irritation potential of 100% PPG-20 methyl glucose ether was evaluated in rabbits (number and strain not stated) using the Draize test. 31 The test substance was classified as a mild transient irritant.
Polypropylene glycol-20 methyl glucose ether distearate
In another Draize test, PPG-20 methyl glucose ether distearate was classified as practically nonirritating in rabbits (number and strain not stated) when tested at a concentration of 100%. 32
Polyethylene glycol-120 methyl glucose dioleate
The ocular irritation potential of PEG-120 methyl glucose dioleate was evaluated in the Draize test using 5 male or female New Zealand albino rabbits. 37 The test substance (100 µL) was instilled into 1 eye of each animal, followed by massaging for 30 seconds. Untreated eyes served as controls. Reactions were scored at 24, 48, 72 hours, and 7 days postinstillation, and maximum average Draize scores (MASs; range = 0-110) were determined. PEG-120 methyl glucose dioleate was classified as a slight irritant (MAS = 8.8). An in vitro assay was conducted to determine if there was a correlation with the in vivo Draize test conducted on rabbits. Using sheep red blood cells, this in vitro assay assessed hemolysis and protein denaturation. The extent of hemolysis was determined spectrophotometrically. Assay results for PEG-120 methyl glucose dioleate were as follows: effective concentration that caused 50% hemolysis (H50) = 1,125.56 µg/mL; denaturation index (DI) = 12.82%; H50/DI = 87.80. The Pearson and Spearman correlation coefficients between the log H50/DI and the MAS were 0.752 and 0.705, respectively. Thus, PEG-120 methyl glucose dioleate was also classified as a slight irritant in the in vitro assay.
The ocular irritation potential of 100% PEG-120 methyl glucose dioleate was evaluated in the Draize test using rabbits (number and strain not stated). 33 The test substance did not induce ocular irritation. In comparative irritation tests, an unspecified concentration of the test substance significantly reduced the ocular irritation induced by SLS and AOS in rabbits (number and strain not stated). The 2 abbreviated chemical names were not defined.
Polyethylene glycol-20 methyl glucose sesquistearate
The ocular irritation potential of undiluted PEG-20 methyl glucose sesquistearate was evaluated in the Draize test using 9 New Zealand albino rabbits. 34 The test material (0.1 mL) was instilled into the right eye, and the left eye served as the untreated control. The eyes of 3 and 6 rabbits were rinsed and unrinsed, respectively, after instillation. Reactions were scored for up to 72 hours postinstillation. It was concluded that the test material was a minimal transient ocular irritant.
In another Draize test, the ocular irritation potential of PEG-20 methyl glucose sesquistearate (as 25% gravimetric aqueous suspension) was evaluated using 6 New Zealand white rabbits (6 months old). 38 The procedure was similar to the one in the preceding study, except that none of the eyes were rinsed after instillation. The test material was classified as a minimal ocular irritant.
Polyethylene glycol-120 methyl glucose trioleate
In an ocular irritation test, 0.1 mL of PEG-120 methyl glucose trioleate (and) propylene glycol (and) water (contains 34% to 43% active PEG-120 methyl glucose trioleate) were instilled into the eyes of rabbits (number and strain not stated) according to the Draize protocol. 35 None of the animals died. A total maximum average Draize score of 2 (range = 0-110) was reported at 1 hour postinstillation, and a score of 0 was reported at 48 hours postinstillation.
Skin Irritation and Sensitization
Animal and human skin irritation/sensitization studies are summarized in this section and in Table 8. Most of the results are classified as negative.
Skin Irritation and Sensitization Studies.
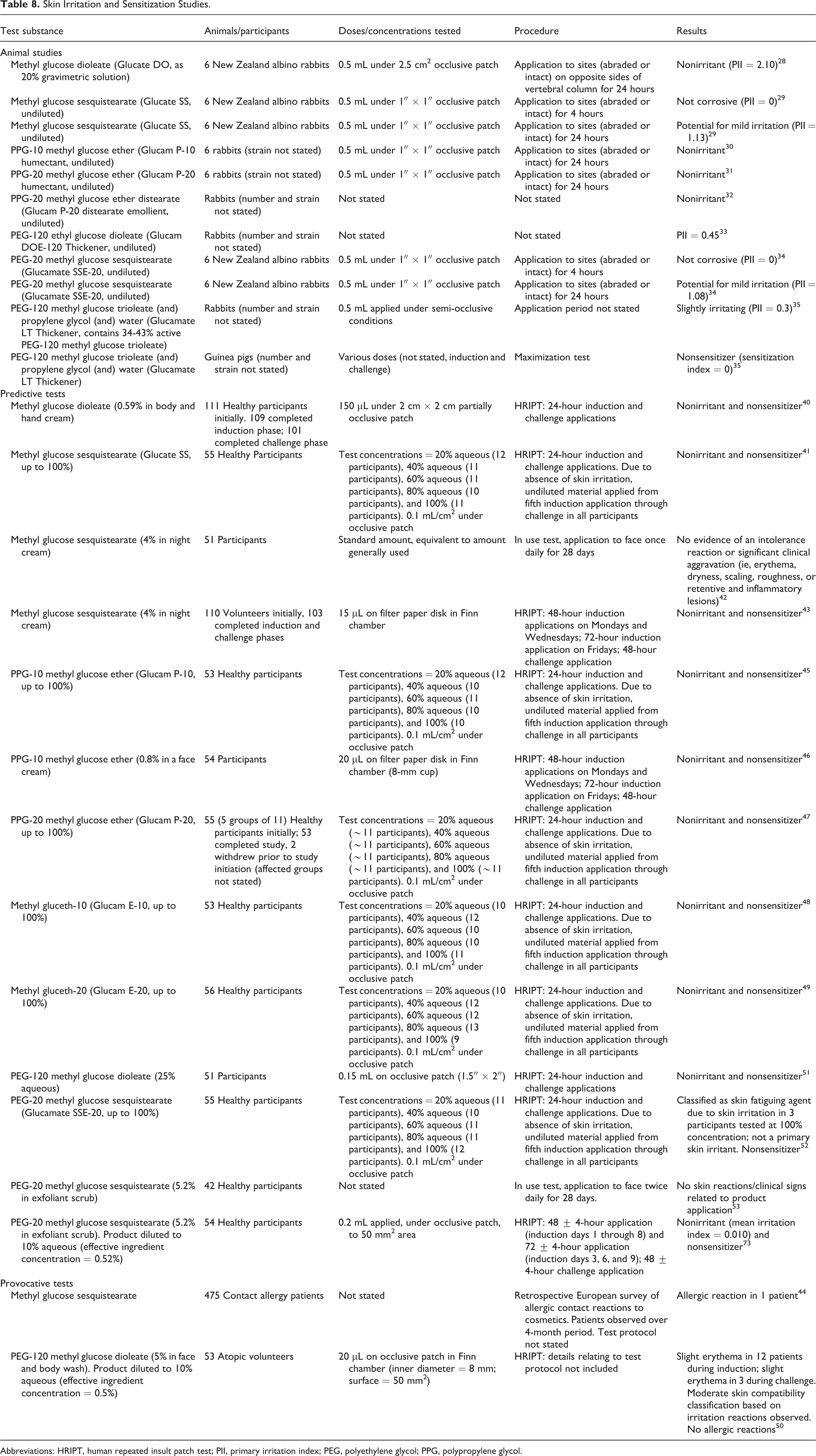
Abbreviations: HRIPT, human repeated insult patch test; PII, primary irritation index; PEG, polyethylene glycol; PPG, polypropylene glycol.
Methyl glucose dioleate
Animal
The skin irritation potential of methyl glucose dioleate (20% w/w mineral oil suspension) was evaluated in a Draize skin irritation test using 6 New Zealand albino rabbits. 28 The test material (0.5 mL) was applied, under a 2.5 cm2 occlusive patch to clipped areas of intact or abraded skin. The 2 test sites were on opposite sides of the vertebral column. The trunk was then covered with an impermeable occlusive wrapping for 24 hours. Reactions were scored at 24 and 72 hours postapplication. It was concluded that the test material was not a primary dermal irritant under the conditions of this test (primary irritation index [PII] = 2.10).
Methyl glucose sesquistearate
Methyl glucose sesquistearate was evaluated in a skin irritation study involving rabbits, using the OECD 405 test protocol. Additional study details were not included. The test substance was classified as a nonirritant. 39
Human
Predictive testing
The skin irritation and sensitization potential of a body and hand cream containing 0.59% methyl glucose dioleate was evaluated in a repeated insult patch test (double-blind conditions) that initially involved 111 healthy participants (64 males, 47 females; 18-74 years old). 40 Of the 111, 109 completed the induction phase and 101 completed the challenge phase. The test procedure comprised 9 sequential 24-hour induction applications and 2 concurrently conducted 24-hour challenge applications (1 at induction site and 1 at naive site). The product was applied using a partially occlusive patching device consisting of a 2 cm × 2 cm absorbent pad (immersed with 150 µL of product), centered on the adhesive-coated surface of a 2 cm × 4 cm plastic film. The product was neither a clinically significant skin irritant nor a sensitizer under the conditions of this study.
Methyl glucose sesquistearate
Animal
Undiluted methyl glucose sesquistearate was evaluated in a Draize dermal corrosion (tissue destruction) test using 6 New Zealand albino rabbits (3 males, 3 females). 29 The test material (0.5 mL) was applied, under a 1″ × 1″ occlusive patch, to clipped areas of intact or abraded skin. The trunk was then covered with an impermeable occlusive wrapping for 4 hours. Test sites were scored for erythema/edema reactions at 4 and 48 hours postapplication. It was concluded that the test material was not corrosive (PII = 0).
The skin irritation potential of undiluted methyl glucose sesquistearate was evaluated in a Draize skin irritation test using 6 New Zealand albino rabbits (3 males, 3 females). 29 The test protocol was similar to the one in the preceding study, except that occlusive patches remained in place for 24 hours and reactions were scored at 24 and 72 hours postapplication. It was concluded that the test material had a potential for mild irritation (PII = 1.13).
The skin irritation/corrosive potential of methyl glucose sesquistearate in rabbits was evaluated according to the OECD 404 test protocol. Additional study details were not included. The test substance was classified as nonirritating. 39
Methyl glucose sesquistearate was evaluated in a skin sensitization study involving guinea pigs, using the OECD 406 test protocol. Additional study details were not included. The test substance was classified as a nonsensitizer. 39
Human
Predictive testing
In a human skin irritation and sensitization study, methyl glucose sesquistearate was evaluated undiluted (100%, as supplied; 11 participants) and at the following concentrations in water: 20% (12 participants), 40% (11 participants), 60% (11 participants), and 80% (10 participants).41 Participants (55 total, all healthy) comprising the 5 groups collectively were >18 years old. During induction, the test material (0.1 mL/cm2, under occlusive patch) was applied for 24 hours, and this procedure was repeated for a total of 4 consecutive exposures per week for 3 weeks. Because there was no visible evidence of skin irritation up to the fourth patch application in any of the test groups, undiluted test material was applied for the remainder of induction and during the challenge phase. For patch applications 5 through 12, visible irritation (1+ reaction) was observed in one participant. This 1+ reaction was not considered significant. During challenge, initiated after a 2-week nontreatment period, an occlusive patch was applied for 24 hours to a new test site. Reactions were scored at 24, 48, and 72 hours postremoval. There was no visible evidence of skin sensitization. The test material did not act as a primary irritant or sensitizer in this study.
The cutaneous (by clinical evaluation and self-assessment) and cosmetic (using a questionnaire) acceptability of a night cream containing 4% methyl glucose sesquistearate was evaluated using 51 female participants. 42 Each was instructed to apply a standard amount of the product (equivalent to amount generally used) to the face once daily for 28 days (± 2 days). Facial skin of the participants were clinically examined by the investigating dermatologist on days 1 and 29, a daily self-evaluation card was completed by each participant on days 1 to 28, and an acceptability questionnaire was completed by each participant on day 29. There was no evidence of an intolerance reaction or significant clinical aggravation (ie, erythema, dryness, scaling, roughness, or retentive and inflammatory lesions in clinical examinations) in any of the participants. Additionally, none of the participants presented subjective signs that were deemed relevant and probably caused by product application. It was concluded that the product was well-tolerated, and, based on questionnaire results, that the product was judged favorably by the participants.
The skin sensitization potential of a night cream containing 4% methyl glucose sesquistearate was studied using a human repeated insult patch test (HRIPT) involving 110 adult volunteers (between 18 and 70 years of age). 43 Seven of the participants started, but did not complete, the induction phase and were not involved in the challenge phase for reasons unrelated to conduct of the study. During the 3-week induction period, the product (15 µL on filter paper disc in Finn chamber) was applied for 48 hours (Mondays and Wednesdays) and 72 hours (Fridays) to the left upper back 3 times per week. Reactions were scored 15 to 20 minutes after patch removal. The induction phase was followed by a 2-week nontreatment period. During the challenge phase, 1 Finn chamber containing a fresh filter paper disc immersed with the product (15 µL) was applied for 48 hours to the area used for induction (left upper back). A second chamber was applied for 48 hours to a new site on the right upper back. Reactions were scored 30 to 35 minutes after patch removal. Follow-up examinations were performed 24 and 48 hours later. No adverse effects were observed in any of the 110 participants during induction, and the same was true for the 103 participants who completed the challenge phase. It was concluded that the product was neither a skin irritant nor a sensitizer.
Provocative testing
A retrospective European survey of allergic contact reactions to cosmetics was conducted using data on 475 patients with contact allergy to cosmetic ingredients. 44 The patients, treated at 5 European dermatology centers, were observed during a 4-month period (January–April 1996). The test protocol was not stated. One patient, at a center in Belgium, had an allergic reaction to methyl glucose sesquistearate (test concentration not stated).
Isostearic acid, esters with methyl α-d -glucoside
A primary dermal irritation study was performed according to OECD guideline 404 using 3 young adult, male New Zealand white rabbits.
7
Isostearic acid, esters with methyl α-
Isostearic acid, esters with methyl α-
Polypropylene glycol-10 methyl glucose ether
Animal
The skin irritation potential of 100% PPG-10 methyl glucose ether was evaluated according to an occlusive patch test procedure involving at least 6 rabbits (strain not stated). 30 Patches (1″ × 1″) containing the test substance (0.5 mL) were applied for 24 hours to abraded and intact sites that had been clipped free of hair. The patches were secured with adhesive tape, and the entire trunk of each animal was wrapped with an impervious material. The test substance was classified as a nonirritant.
Human
Predictive testing
In a human skin irritation and sensitization study, PPG-10 methyl glucose ether was evaluated, using 53 participants, undiluted (100%, as supplied; 10 participants) and at 20% (12 participants), 40% (10 participants), 60% (11 participants), and 80% (10 participants) in water. 45 During induction, the test material (0.1 mL/cm2, under occlusive patch) was applied for 24 hours, and this procedure was repeated for a total of 4 consecutive exposures per week for 3 weeks. Because there was no visible evidence of skin irritation up to the fourth patch application in any of the test groups, undiluted test material was applied for the remainder of induction and during the challenge phase. During challenge, initiated after a 2-week nontreatment period, an occlusive patch was applied for 24 hours to a new test site. Reactions were scored at 24, 48, and 72 hours postremoval. There was no visible evidence of skin irritation or sensitization during the study.
The skin sensitization potential of a face cream containing 0.8% PPG-10 methyl glucose ether was evaluated using 54 healthy male and female participants in an HRIPT using occlusive patches. 46 A Finn chamber (8 mm aluminum cup, affixed to Scanpor tape) containing a filter disk immersed with the product (20 µL) was used for patch testing. Test sites were either to the left or right side of the infrascapular area of the back. During induction, patches were applied for 48 hours (on Mondays, and Wednesdays) and 72 hours (on Fridays) during 3 consecutive weeks. Reactions were scored by a dermatologist at 15 to 30 minutes after patch removal according to International Contact Dermatitis Research Group (ICDRG) criteria. The challenge phase was initiated following a 2-week nontreatment period. Challenge patches were applied to the original site and a new site for 48 hours. Reactions were scored at 30 minutes and 48 hours after patch removal. The product did not induce skin irritation or sensitization.
Polypropylene glycol-20 methyl glucose ether
Animal
The skin irritation potential of 100% PPG-20 methyl glucose ether was evaluated according to the preceding occlusive patch test procedure involving at least 6 rabbits (strain not stated). 31 The test substance was classified as a nonirritant.
Human
Predictive testing
In a human skin irritation and sensitization study, PPG-20 methyl glucose ether was evaluated undiluted (as supplied) and at the following concentrations in water: 20%, 40%, 60%, and 80%. Five groups of 11 healthy participants were tested. 47 Two of the initial 55 participants withdrew prior to study initiation; the assigned test group for each was not stated. During induction, the test material (0.1 mL/cm2, under occlusive patch) was applied for 24 hours, and this procedure was repeated for a total of 4 consecutive exposures per week for 3 weeks. Because there was no visible evidence of skin irritation up to the fourth patch application in any of the test groups, undiluted test material was applied for the remainder of induction and during the challenge phase. During challenge, initiated after a 2-week nontreatment period, an occlusive patch was applied for 24 hours to a new test site. Reactions were scored at 24, 48, and 72 hours postremoval. There was no evidence of skin irritation or sensitization during the study, and it was concluded that no visible evidence of skin damage was observed in any of the participants tested.
Polypropylene glycol-20 methyl glucose ether distearate
Undiluted PPG-20 methyl glucose ether distearate was classified as a nonirritant in a skin irritation test involving rabbits (number and strain not stated). 32 Details relating to the test protocol were not stated.
Methyl gluceth-10
In a human skin irritation and sensitization study, methyl gluceth-10 was evaluated undiluted (as supplied, 11 participants) and at 20% (10 participants), 40% (12 participants), 60% (10 participants), and 80% (10 participants, 53 total) in water. 48 During induction, the test material (0.1 mL/cm2, under occlusive patch) was applied for 24 hours, and this procedure was repeated for a total of 4 consecutive exposures per week for 3 weeks. Because there was no visible evidence of skin irritation up to the fourth patch application in any of the test groups, undiluted test material was applied for the remainder of induction and during the challenge phase. During challenge, initiated after a 2-week nontreatment period, an occlusive patch was applied for 24 hours to a new test site. Reactions were scored at 24, 48, and 72 hours postremoval. There was no visible evidence of skin irritation or sensitization during the study.
Methyl gluceth-20
Methyl gluceth-20 was evaluated using 5 groups (56 total participants) according to an irritation and sensitization study. 49 The following concentrations (in distilled water) and participants were evaluated in the study: 20% (10 participants), 40% (12 participants), 60% (12 participants), 80% (13 participants), and 100% (undiluted, as supplied; 9 participants). Each concentration was applied at a dose of 0.1 mL/cm2. There was no evidence of skin irritation or sensitization during the study, and it was concluded that no visible evidence of skin damage was observed in any of the participants tested.
Polyethylene glycol-120 methyl glucose dioleate
Animal
The skin irritation potential of 100% PEG-120 methyl glucose dioleate was evaluated using rabbits (number and strain not stated). 33 Details relating to the test protocol were not included. A primary irritation index of 0.45 (range = 0-8) was reported.
Human
Provocative testing
The skin sensitization potential of a face and body wash containing 5% PEG-120 methyl glucose dioleate was evaluated in an HRIPT (occlusive patches) involving 53 atopic volunteers. 50 The product was diluted with water to a concentration of 10% (effective concentration = 0.5%), and a 20 µL volume of diluted product was applied to the skin using a Finn chamber (inner diameter = 8 mm; surface = 50 mm2). Reactions were scored according to ICDRG criteria. Additional details relating to the patch test procedure were not included. Slight erythema was observed in 12 participants during induction; however, these reactions were considered normal for the product type class evaluated. Slight erythema was observed in 3 participants during the challenge phase. The authors concluded that the product had moderate skin compatibility, based on the irritation reactions observed, but that repeated applications did not induce any allergic reactions.
Predictive testing
In a study involving 51 adult participants, the skin irritation and sensitization potential of PEG-120 methyl glucose dioleate was evaluated. 51 An occlusive patch (1.5″ × 2″) containing a 25% aqueous solution of the test material (0.15 mL) was applied to the upper back, between the scapulae, for 24 hours. This procedure was repeated 3 times per week for a total of 10 induction applications. Following a 2-week nontreatment period, a 24-hour challenge patch was applied to the original site and to a new site (volar forearm). Sites were evaluated at 24 and 48 hours postapplication. It was concluded that, under the conditions of this study, the test material did not have skin irritation or sensitization potential.
Polyethylene glycol-20 methyl glucose sesquistearate
Animal
Undiluted PEG-20 methyl glucose sesquistearate was evaluated in a Draize dermal corrosion (tissue destruction) test using 6 New Zealand albino rabbits (3 males, 3 females). 34 The test material (0.5 mL) was applied, under a 1″ × 1″ occlusive patch, to clipped areas of intact or abraded skin. The trunk was then covered with an impermeable occlusive wrapping for 4 hours. Reactions were scored for erythema/edema reactions at 4 and 48 hours postapplication. It was concluded that the test material was not corrosive (PII = 0).
The skin irritation potential of undiluted PEG-20 methyl glucose sesquistearate was evaluated in a Draize skin irritation test using 6 New Zealand albino rabbits (3 males, 3 females). 34 The test protocol was similar to the one in the preceding study, except that occlusive patches remained in place for 24 hours and reactions were scored at 24 and 72 hours postapplication. It was concluded that the test material had a potential for mild irritation (PII = 1.08).
Human
Predictive testing
The skin irritation and sensitization potential of PEG-20 methyl glucose sesquistearate was evaluated on methyl gluceth-20 in an earlier section of this report. 52 The following concentrations (in distilled water) were tested on a total of 55 healthy participants: 20% (11 participants), 40% (10 participants), 60% (11 participants), 80% (11 participants), and 100% (undiluted, as supplied; 12 participants). Each concentration was applied at a dose of 0.1 mL/cm2. Because the 80% concentration induced only very slight erythema (only induction reactions observed; patch applications 2 through 4) during induction, all subsequent patch applications (all participants) were at a concentration of 100%. The reaction classified as very slight erythema (to 80% concentration) was not deemed significant irritation. For patch applications 5 through 12, skin irritation was observed in 3 participants tested with 100%, classifying the material as a skin fatiguing agent at that concentration. Challenge reactions were not observed in any of the participants. The test material did not cause primary skin irritation or sensitization in this study.
A 4-week use test was performed to assess the acceptability (ie, the good tolerance under normal use conditions) of an exfoliant scrub containing 5.2% PEG-20 methyl glucose sesquistearate. 53 Forty-two healthy female volunteers (19-65 years old; Fitzpatrick skin types I to IV) participated in the study. On day 1, the product was applied to wet skin of the face and neck, followed by rinsing, at the investigating center. Subsequent applications were made at home twice per week for 28 ± 2 consecutive days. Acceptability was evaluated daily at home by each volunteer, and visual examinations of application areas (face and neck) were performed by the investigating dermatologist (before day 1 and on day 29). Volunteers also completed a questionnaire at the end of the study. None of the participants had skin reactions/clinical signs that were related to product application. However, the sensation described as very slight pulling in one participants was attributed to product application, and 2% of the volunteers experienced discomfort that was related to product application. It was concluded that the product had “good acceptability” after application under normal conditions of use.
Polyethylene glycol-120 methyl glucose trioleate
Animal
A skin irritation test on PEG-120 methyl glucose trioleate (and) propylene glycol (and) water containing 34% to 43% active PEG-120 methyl glucose trioleate was performed using rabbits (number and strain not stated). 35 A 0.5-mL test solution was applied under semi-occlusive conditions, and additional details relating to the test protocol were not included. None of the animals died. A primary irritation index of 0.3 (range = 0-8) was reported, and the test substance was classified as slightly irritating.
The skin sensitization potential of PEG-120 methyl glucose trioleate (and) propylene glycol (and) water was evaluated in the maximization test using guinea pigs (number and strain not stated). 35 Induction and challenge applications at various doses were made, and additional details relating to the test protocol were not included. None of the animals died. A sensitization index of 0 was reported and the test substance was classified as a nonsensitizer.
Case Reports
Methyl glucose dioleate
A 27-year-old female presented with widespread eczema of the legs, arms, and face, approximately 8 hours after application of an insect repellant. 54 Methyl glucose dioleate is the main component of an ingredient of the repellant that is a water-in-oil emollient and emulsifier. Patch testing with the repellant yielded a positive reaction after 3 days (+D2/++D3). Patch testing with a 3% solution (in paraffin oil) yielded positive reactions after 2 and 3 days ++D2/+++D3). Results were negative for a 3% in paraffin test solution that was used in a patch test on 10 control participants.
Allergic contact dermatitis (widespread, persistent itching dermatitis [erythema and edema]) was observed in a 39-year-old male a day after using the same insect repellant mentioned in the preceding case report. 55 In an open patch test of the repellant, an itching erythematous reaction was observed a few hours (exact time not stated) after patch application. Patch testing with methyl glucose dioleate (10% in petrolatum) revealed positive reactions after 2 and 3 days (+++D2/+++D3).
After self-medication with a paste containing methyl glucose dioleate for treatment of a suspected interdigital mycosis (left foot), a 30-year-old female presented with an itchy dermatitis (erythema and edema) of the legs and abdomen. 56 Patch testing with the paste revealed positive reactions after 2 and 3 days (++D2/+++D3). Patch testing with methyl glucose dioleate (10% in petrolatum) revealed a positive reaction only on day 3 (++D3); results were negative in 5 control participants.
A 60-year-old presented with erythematovesicular lesions on both legs and itch after using a topical antibiotic, for treatment of leg ulcers, for 15 days. 57 After patch testing with individual ingredients of the antibiotic, only the 5% methyl glucose dioleate in petrolatum yielded a positive reaction (++) after 2 and 3 days. In another test (repeated open application test [ROAT]), the patient had a strongly positive reaction to methyl glucose dioleate (5% in petrolatum) after 4 days. This reaction was said to have increased for 2 days after discontinuation at day 4. Test results (ROAT) were negative in 5 control participants.
A 4-day history of a pruritic, erythematovesicular dermatitis of the legs, trunk, and face was reported for a 72-year-old female who used an ointment for treatment of a traumatic leg ulcer. 58 The dermatitis began on the left leg 5 days after initial treatment with the ointment and spread to the other leg, trunk, and face. Patch testing with the ointment yielded a strong positive reaction, which led to further spread of the dermatitis to the face, ears, and upper trunk. Subsequent patch testing identified an ingredient (contains methyl glucose dioleate and oleic acid) of the ointment as the source of the reaction. Patch testing with methyl glucose dioleate (5% in petrolatum) yielded positive reactions after 2 days (+ reaction) and 3 days (++ reaction); reactions were negative in 10 control participants.
A healthy 26-year-old woman, without atopic or contact dermatitis history, presented with a very itchy erythematous papulovesicular eruption on the breast, arms, and the upper part of the abdomen. 59 She had applied a nursing comfort balm on the breasts, and allergic contact dermatitis was suspected. Patch testing was performed using IQ Ultra chambers. Patches were removed prior to the end of the 24-hour application period due to itching, and a positive reaction (+ at day 1) to the balm was observed. This reaction increased in severity (++) at days 2 and 3. Further patch testing with the individual ingredients of the nursing balm revealed a positive reaction only to 5% methyl glucose dioleate in petrolatum (++ on days 2 and 4). Negative patch test results (excluding irritancy) were reported for the 5 control participants tested with methyl glucose dioleate.
Methyl glucose sesquistearate, methyl glucose dioleate, methyl gluceth-20, and PPG-20 methyl glucose ether
A 22-year-old woman presented with a papular/vesicular eruption after using a lotion or facial cream that contained methyl glucose sesquistearate. When the patient was patch tested with this ingredient (5% in petrolatum), results were positive at 96 or 48 hours. 60 Patch-test results for methyl glucose sesquistearate (5% in petrolatum) in 20 control participants were negative. Positive patch-test results were also reported when the patient was patch tested with methyl glucose dioleate (5% in petrolatum). Methyl gluceth-20 and PPG-20 methyl glucose ether (each at 5% in petrolatum) yielded negative patch-test results in this patient.
Reproductive and Developmental Toxicity
Isostearic Acid, Esters With Methyl α-d -Glucoside
A combined repeated dose toxicity study with a reproduction/developmental toxicity screening test was performed according to the OECD 422 test protocol.
7
Isostearic acid, esters with methyl α-
Genotoxicity
Bacterial Cells
Methyl glucose sesquistearate
The mutagenicity of methyl glucose sesquistearate was evaluated in the Ames test. Details relating to the test protocol were not included. The test substance was classified as nonmutagenic. 39
Isostearic acid, esters with methyl α-D-glucoside
Isostearic acid, esters with methyl α-
PEG-120 methyl glucose dioleate
In the Ames plate incorporation test, the genotoxicity of PEG-120 methyl glucose dioleate (in ethanol) was evaluated at doses up to 5,000 µg/plate, with or without metabolic activation, using E coli strain WP2 uvrA and the following S typhimurium strains: TA98, TA100, TA1535, and TA1537. 61 Appreciable toxicity was not observed. It was concluded that PEG-120 methyl glucose dioleate was not genotoxic in any of the bacterial strains tested, with or without metabolic activation.
PEG-120 methyl glucose trioleate
The genotoxicity of PEG-120 methyl glucose trioleate (doses up to 5,000 µg/plate) in water was evaluated with or without metabolic activation using E coli strain WP2 uvrA and the following S typhimurium strains: TA98, TA100, TA1535, and TA1537. 62 The positive controls without activation were: 2-(2-furyl)-3-(5-nitro-2-furyl)acrylamide (AF-2, for strains TA98, TA100, and WP2uvrA), sodium azide (for strain TA1535), and 9-aminoacridine (9-AA, for strain TA1537). With activation, 2-aminoanthracene (2-AA) served as the positive control for all 5 strains. Cytotoxicity was not observed over the range of doses tested. It was concluded that, under the conditions of this test, PEG-120 methyl glucose trioleate was nongenotoxic. All positive controls were genotoxic.
Methyl-α-d -glucopyranoside (methyl glucoside)
The potential of methyl-α-
Mammalian Cells
Isostearic acid, esters with methyl α-d -glucoside
The genotoxicity of isostearic acid, esters with methyl α-
Isostearic acid, esters with methyl α-
Carcinogenicity
Studies on the carcinogenicity of methyl glucose polyethers and esters were not found in the published literature and unpublished data were not provided.
Summary
The safety of methyl glucose polyethers and esters as cosmetic ingredients is reviewed in this report. The methyl glucose polyethers function as skin and hair-conditioning agents, whereas, the methyl glucose esters function only as skin-conditioning agents in cosmetic products. Ingredients classified as both methyl glucose polyethers and esters based on their chemical structures function as skin-conditioning agents, surfactants, and viscosity-increasing agents in cosmetic products.
Data reported to the FDA by industry as part of the VCRP in 2012 indicate that the following methyl glucose polyethers and esters are being used in cosmetic products: methyl glucose dioleate, methyl glucose sesquioleate, methyl glucose sesquistearate, PPG-10 methyl glucose ether, PPG-20 methyl glucose ether, PPG-20 methyl glucose ether distearate, methyl gluceth-10, methyl gluceth-20, PEG-120 methyl glucose dioleate, PEG-20 methyl glucose distearate, PEG-20 methyl glucose sesquistearate, and PEG-120 methyl glucose trioleate.
Results from surveys of ingredient use concentrations provided by the Council in 2012 and 2013 indicate that the polyethers and esters are being used at concentrations up to 15% and 4%, respectively. The 15% maximum use concentration in rinse-off products relates to methyl gluceth-10 and methyl gluceth-20 in skin-cleansing products. For leave-on products, the 15% maximum use concentration relates to methyl gluceth-10 in face and neck creams, lotions, and powders (not sprays).
The survey results provided by the Council also included a use concentration for the newly reported VCRP use(s) of methyl glucose sesquistearate (13 products—1% maximum use concentration), but not PEG-20 methyl glucose sesquistearate (1 product), in lipsticks. Additionally, a maximum use concentration of 0.05% for PEG-20 methyl glucose distearate in lipsticks was reported. Uses of methyl glucose sesquistearate and PEG-20 methyl glucose sesquistearate, but not PEG-20 methyl glucose distearate, in lipsticks were also reported in FDA’s VCRP.
The following ingredients are used in cosmetic aerosol/pump sprays: PEG-20 methyl glucose sesquistearate, methyl gluceth-10, and methyl gluceth-20. Additionally, the following ingredients may be used in face/body powders: methyl glucose dioleate (up to 0.6%), PPG-10 methyl glucose ether (up to 0.8%), PPG-20 methyl glucose ether (up to 0.4%), methyl gluceth-10 (up to 15%), methyl gluceth-20 (up to 10%), PEG-120 methyl glucose dioleate (up to 4%), PEG-20 methyl glucose sesquistearate (up to 10%), and PEG-120 methyl glucose trioleate (up to 0.5%). Because these ingredients are used in aerosol/pump hair sprays or powders, they could possibly be inhaled.
Toxicokinetic data on methyl glucose polyethers and esters reviewed in this safety assessment were not found in the published literature. However, the Expert Panel (Panel) expected most of these ingredients to have low potential for skin penetration, based on their molecular weights, log Kows, and other properties.
In a study evaluating the pulmonary absorption of α-methyl-
Acute oral toxicity data (rats) on methyl glucose polyethers and esters (trade name materials) suggest that these ingredients are relatively nontoxic, based on reported LD50 values of >2g/kg or >5 g/kg. In an acute dermal toxicity study (rats) on a trade name material identified as PEG-120 methyl glucose trioleate (and) propylene glycol (and) water (Glucamate LT Thickener), an LD50 of >12 g/kg was reported. Additional acute dermal toxicity data on this ingredient group were not available.
In ocular irritation tests involving rabbits, the following ingredients (all trade name materials) induced no-to-mild ocular irritation: methyl glucose sesquistearate, PPG-10 methyl glucose ether, PPG-20 methyl glucose ether, PPG-20 methyl glucose ether distearate, PEG-120 methyl glucose dioleate, PEG-20 methyl glucose sesquistearate, and PEG-120 methyl glucose trioleate (sold as 34% to 43% active). Methyl glucose dioleate was also nonirritating to the eyes of rabbits at a concentration of 20% or 25%, and PEG-20 methyl glucose sesquistearate was minimally irritating at a concentration of 25%.
In animal (rabbit) studies, methyl glucose dioleate (20% mineral oil suspension), undiluted PPG-10 methyl glucose ether, undiluted PPG-20 methyl glucose ether, undiluted PPG-20 methyl glucose distearate, and undiluted PEG-120 methyl glucose dioleate were classified as nonirritants. Methyl glucose sesquistearate was classified as a nonsensitizer in guinea pigs. Additionally, undiluted methyl glucose sesquistearate was classified as noncorrosive/nonirritating in 2 studies involving rabbits, whereas, in another study (rabbits), it was classified as having mild skin irritation potential. The same was true for undiluted PEG-20 methyl glucose sesquistearate in similar studies involving rabbits. PEG-120 methyl glucose trioleate (sold as 34% to 43% active) was classified as slightly irritating to the skin of rabbits. It was also classified as a nonsensitizer in a guinea pig maximization test; the test concentration was not stated.
A night cream containing 4% methyl glucose sesquistearate was well-tolerated (ie, no erythema or inflammatory lesions) in a 4-week cosmetic use test. This product was also neither a skin irritant nor a sensitizer in an HRIPT. In another 4-week cosmetic use test, an exfoliant scrub containing 5.2% PEG-20 methyl glucose sesquistearate was classified as having good acceptability, that is no skin reactions/clinical signs that were related to product application. The following ingredients were classified as nonirritants and nonsensitizers in human repeated insult patch tests: methyl glucose dioleate (0.59% in body and hand cream), methyl glucose sesquistearate (up to100%), PPG-10 methyl glucose ether (up to100%), PPG-20 methyl glucose ether (up to 80% aqueous), methyl gluceth-10 (up to 100%), methyl gluceth-20 (up to 100%), PEG-120 methyl glucose dioleate (25% aqueous), and PEG-20 methyl glucose sesquistearate (up to 100%). A retrospective European survey of allergic contact reactions to cosmetics was conducted using data on 475 patients with contact allergy to cosmetic ingredients. One patient, at a center in Belgium, had an allergic reaction to methyl glucose sesquistearate (test concentration not stated).
Positive patch-test reactions to methyl glucose dioleate were observed in various case reports. An insect repellant (main component of 1 ingredient = methyl glucose dioleate) induced contact dermatitis in 2 patients. Patch-test results for the repellant were positive in the 2 patients, but negative in 10 control participants. One of the 2 patients was patch tested with methyl glucose dioleate (10% in petrolatum), and results were negative. Dermatitis was observed in 2 additional patients after application of a paste (to treat suspected mycosis) and a topical antibiotic (for leg ulcer), both containing methyl glucose dioleate, respectively. Patch-test results for methyl glucose dioleate (10% in petrolatum) were positive in one patient, but negative in 5 control patients. For the other patient, similar patch-test results at a lower concentration (5% in petrolatum) were reported. A positive patch-test reaction to methyl glucose dioleate (5% in petrolatum) was observed in another patient who had used an ointment containing methyl glucose dioleate to treat a leg ulcer. Patch test results were negative in 10 control participants.
Dermatitis was also observed in a patient after using a lotion or facial cream containing methyl glucose sesquistearate. Patch-test results for the ingredient (5% in petrolatum) were positive, but negative in 20 control participants. Additional patch tests revealed a positive reaction to methyl glucose dioleate (5%) in petrolatum in the patient, and negative reactions to PPG-20 methyl glucose ether and methyl gluceth-20 (both at 5% in petrolatum). Allergic contact dermatitis was also observed in a patient who had used a nursing comfort balm containing methyl glucose dioleate. Patch-test results were positive for 1 ingredient in the product, methyl glucose dioleate (5% in petrolatum). Negative patch-test results were reported for 5 control participants patch tested with methyl glucose dioleate.
Methyl glucose sesquistearate was classified as nongenotoxic in the Ames test. Also, PEG-120 methyl glucose dioleate and PEG-120 methyl glucose trioleate were not genotoxic in the Ames test (S typhimurium and E coli strains) at doses up to 5,000 µg/plate with or without metabolic activation. Negative Ames test (S typhimurium and E coli strains) results were also reported for isostearic acid, esters with methyl α-
Repeated dose toxicity, reproductive and developmental toxicity, or carcinogenicity data on the methyl glucose polyethers and esters reviewed in this safety assessment were not identified in the published literature. However, a mixture (registered with the ECHA) containing on isostearic acid, esters with methyl α-
Discussion
There were limited genotoxicity data; however, robust dermal irritation and sensitization data were available in this assessment. After reviewing the available data, including the molecular weights, log Kows, and toxicity data, the Panel concluded that these ingredients, as used in cosmetics, would not result in significant systemic exposures. The Panel also concluded that the interrelationships between molecular structures and physicochemical and biological characteristics (ie, structure–property and structure–activity relationships), in conjunction with their functions and concentrations in cosmetics, allow grouping these ingredients together and extending the available toxicological data to support the safety of each of the ingredients in the group.
The use of methyl glucose sesquistearate, PEG-20 methyl glucose sesquistearate, and PEG-20 methyl glucose distearate in lipsticks raised the concern about repeated ingestion as a route of exposure. Methyl glucose sesquistearate and PEG-20 methyl glucose distearate are being used in lipsticks at concentrations of 1% and 0.05%, respectively. Use concentration data on PEG-20 methyl glucose sesquistearate in lipsticks were not provided; however, it is assumed that this ingredient is being used at concentrations no greater than 1%. The Panel subsequently agreed that repeated dose oral toxicity data on isostearic acid, esters with methyl α-
The Panel considered the potential effect that methyl glucose would have on glucose metabolism, were these ingredients to be absorbed and metabolized. As previously noted, however, for most of these ingredients, no significant systemic exposure would result from dermal use. The potential for complete deesterification of these ingredients to produce methyl glucose was considered insignificant, and, therefore, should not interfere with glucose metabolism. Regarding the extent of esterification of the methyl glucose esters, the International Cosmetic Ingredient Dictionary and Handbook states that, by convention, esters are monoesters unless stated otherwise.
The Panel discussed the issue of incidental inhalation exposure from aerosol and pump hair sprays and foot powders and sprays. Inhalation toxicity data were not available. However, the Panel considered pertinent data indicating that incidental inhalation exposures to these ingredients in such cosmetic products would not cause adverse health effects, including data characterizing the potential for these ingredients to cause acute oral toxicity, and ocular or dermal irritation or sensitization. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the following methyl glucose polyethers and esters are safe in the present practices of use and concentration, described in this safety assessment, in cosmetics.
Esters
methyl glucose caprylate/caprate*
methyl glucose dioleate
methyl glucose isostearate*
methyl glucose laurate*
methyl glucose sesquicaprylate/sesquicaprate*
methyl glucose sesquicocoate*
methyl glucose sesquiisostearate
methyl glucose sesquilaurate*
methyl glucose sesquioleate
methyl glucose sesquistearate
Polyethers
PPG-10 methyl glucose ether
PPG-20 methyl glucose ether
PPG-25 methyl glucose ether*
methyl gluceth-10
methyl gluceth-20
Esters and polyethers
PEG-120 methyl glucose dioleate
PEG-20 methyl glucose distearate
PEG-80 methyl glucose laurate*
PEG-20 methyl glucose sesquicaprylate/ sesquicaprate*
PEG-20 methyl glucose sesquilaurate*
PEG-20 methyl glucose sesquistearate
PEG-120 methyl glucose triisostearate*
PEG-120 methyl glucose trioleate
PPG-20 methyl glucose ether acetate*
PPG-20 methyl glucose ether distearate
*Ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC, USA.
The 2013 Cosmetic Ingredient Review Expert Panel members are Chair, Wilma F. Bergfeld, MD, FACP; Donald V. Belsito, MD; Curtis D. Klaassen, PhD; Daniel C. Liebler, PhD; Ronald A. Hill, PhD James G. Marks, Jr, MD; Ronald C. Shank, PhD; Thomas J. Slaga, PhD; and Paul W. Snyder, DVM, PhD The CIR Director is Lillian J. Gill, DPA. This report was prepared by Wilbur Johnson, Jr, MS, Senior Scientific Analyst and Bart Heldreth, PhD, Chemist.
Author Contributions
W. Johnson contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised the manuscript. L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. F. A. Andersen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. W. Bergfeld contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. D. Belsito contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. R. Hill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. C. Klaassen contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. D. Liebler contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. J. Marks contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. R. Shank contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. T. Slaga contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.