Abstract
Cryptotanshinone (Cry) has multiple potential functions in treating different diseases. Most studies on Cry focus on its pharmacological effects and mechanisms, but toxicological reports on Cry are rare. Zebrafish is used as a model organism in drug development as it saves costs and time. This work aimed to investigate the toxicity of Cry on zebrafish. Results showed that growth retardation, pericardial edema, and scoliosis occurred when zebrafish embryos were exposed to Cry, indicating its teratogenic effects. Cell apoptosis was observed in the brainstem area of embryos using acridine orange staining, and qPCR showed that caspase-3 was increased in Cry-exposed embryos. The results of locomotor activity and touched-evoke escape reaction experiments showed that Cry significantly reduced the swimming speed and escape reaction time of larvae.
Introduction
Cryptotanshinone (Cry) is one of the active components of Radix Salvia miltiorrhiza (RSM), which has been widely used in clinics for the treatment of cardiovascular, cerebrovascular, and neurodegenerative diseases in many Asian countries for hundreds of years. 1 Several studies have investigated the effects of Cry on various types of cancer, including lung cancer, hepatoma, esophageal squamous cell carcinoma, osteosarcoma, tongue squamous cell carcinoma, ovarian cancer, and renal cell carcinoma.2–10 Cry has many different approaches to fight against cancer. Cry induces cell apoptosis by activating caspase-9/3 and Sub-G1 accumulation in chronic myeloid leukemia K562 cells, esophageal cancer cells, renal cell carcinoma, tongue squamous cell carcinoma, and prostate cancer cells through the activation of the JAK, STAT, SHP, JNK, or p38 MAPK signaling pathway, respectively.2,3,6,9,11 When the expression of MMP-2 and MMP-9 is inhibited, Cry suppresses the migration and invasion of ovarian cancer A2780 cells and tongue squamous cell carcinoma.5,9 By inhibiting the expression of epidermal growth factor receptor, which plays an important role in lung cancer, Cry suppresses the proliferation of non-small cell lung cancer. Further research showed that this effect may be associated with miRNA-146a-5p. 8 Other studies have shown that Cry inhibits cancer by enhancing immunotherapeutic effects, inducing mitochondrial fragmentation, or activating macrophages.4,7,10 Moreover, Cry has been reported to influence multiple factors in rat coronary arteries 12 and ethanol-induced liver injury, 13 inhibit endothelin-1 expression, and stimulate nitric oxide production in human vascular endothelial cells. 14 Through the STAT3 signaling pathway, Cry inhibits the promotion of the early phase of adipogenesis. 15 In addition, Cry has been found to exert various anti-inflammation effects, such as inhibiting cyclooxygenase-2 enzyme activity, LPS-induced inflammatory response, or collagen-induced arthritis, in rats.16–18 These findings suggest that Cry has several functions and has the potential to be used as a leading compound in the development of new drugs for many diseases.
Herbal medicine usually contains several components, and distinguishing the toxicity of each ingredient is complicated. For many centuries, RSM has been widely used throughout Asia as an ethnic medicine. The use of RSM in clinics has already resulted in adverse drug reactions and endangered many patients.19,20 However, the mechanism of toxicity of RSM remains to be fully elucidated. Toxicity assessments of a drug prior to clinical trials are accomplished customarily through laboratory rodent studies, but these studies are expensive and time consuming. Toxicological scientists have searched for an alternative animal to partially or completely substitute rodents. 21 In recent years, zebrafish has rapidly become a popular model organism for translational biomedical research due to their genetic tractability; low cost; rapid reproductive cycle; and ease of behavioral, pharmacological, or genetic manipulation. The testing of chemicals on zebrafish embryos has matured to the point that international standardization, method validation, and broadening of chemical coverage are rapidly occurring. The Organisation for Economic Co-operation and Development fish-testing guidelines acknowledge the importance of fish embryo testing, including fish acute toxicity test, and rules have been established to guide the different phases of the test. 22 Numerous chemicals have been evaluated in zebrafish models, and the testing method was suggested to be valuable in predicting chemical safety in humans.23–25 Although the metabolism of Cry in zebrafish has been reported, 26 few or no side effects of Cry have been reported in animal models. Therefore, the present study aimed to evaluate the toxicity effects of Cry in zebrafish embryos and larvae.
Materials and methods
Materials
Cry was purchased from Xi’an Honson Biotechnology Co., Ltd. (China). Wild-type tuebingen strain zebrafish were obtained from China Zebrafish Resource Center (Wuhan, China). The fish medium (reconstitued water consisting of 2 mM CaCl2, 0.5 mM MgSO4, 0.7 mM NaHCO3, and 0.07 mM KCl, at 28.0 ± 1.0°C) was prepared according to OECD Fish Embryo Test Guideline. 27 All solutions were freshly prepared, and chemicals were dissolved 2 h before incubation. Zebrafish were fed in a recirculating aquaculture system which was purchased from Shanghai Haisheng Biotech Co., Ltd. (Shanghai, China). The water was purified by water purifier (Shenzhen Luoke Water Purification Equipment Co., LTD, China), and the NaHCO3 and NaCl were used to adjust the pH and conductivity at pH 6.8∼7.4 and 500∼550 µS, respectively. The housing system is equipped with temperature control unit, UV light and activated carbon filter system. The day to night photoperiod was 14 h: 10 h.
The lethal and morphological analysis
Based on preliminary experiments, the zebrafish embryos were exposed to the tested solutions from 2 h post fertilization (hpf) to 96 hpf in 35 mm glass culture dish in embryo medium (20 embryos per dish). Cry was treated with different concentrations (1 μM, 2 μM, 5 μM, 10 μM, 20 μM), and 0.5% DMSO (vehicle) in fish medium was used as solvent control group. Twenty embryos were exposed per group and each experiment was repeated 3 times in room with temperature at 28.0 ± 1.0°C.
All embryos were staged as described by Kimmel et al., 28 and lethal (coagulation) or teratogenic effects were recorded (Table 1). Teratogenic effects were considered as fingerprint endpoints, if the following criteria were fulfilled: (i) concentration–response relationship is present and (ii) the endpoint must be observed in ≥50% of all embryos showing teratogenic effects in all test groups of a test substance. The embryos were evaluated and scored for lethal effects, hatching rate and deformities number at 24, 48, 72 and 96 hpf after exposed to the Cry (Figure 6(A)), and half lethal concentration (LC50) and the half effective concentration (EC50) was calculated (GraphPad Prism 6) at 96 hpf (Figure 2). The scoring of teratogenic effects was ended at 96 hpf for all the groups except 20 μM Cry group in which the embryos were all dead at 72 hpf. The teratogenic effects of Cry were observed every 24 h and sum up according to the concentration of Cry, which included scoliosis, malformation of tail and eyes, pericardial edema, deformity of yolk (yolk sac edema) and growth retardation. In order to characterize the teratogenic potential of a test substance, the teratogenicity index (TI), which is traditionally defined as the quotient of LC50 and EC50, was calculated. If the TI of a given substance is >1, the substance is considered to be teratogenic; if the TI is ≤1, the substance produces mainly embryo lethal effects.29,30
Lethal and teratogenic effects observed in zebrafish danio rerio embryos depending on the observation time.

a No clear organ structures are recognized.
b Malformation of tail was assessed when the tail was bent.
c At 24 and 48 hpf, when the embryos were not hatched, growth retardation was assessed by optical comparison to the control embryos (e.g. size, development). At 72 hpf growth retardation was assessed when an embryo shows a body length below 2.8 mm.
The number of “+” represents the level of teratogenic.
Acridine orange staining
To detect the apoptotic cells, the embryos were imaged under the fluorescent microscope after staining with acridine orange. Ten embryos were assigned in blank control, solvent control, and 2 μM Cry groups, respectively. At 48 hpf, all embryos were incubated with the acridine orange dye for 30 min at 26°C in the dark, and then washed twice, followed by anaesthesia with 0.02% tricaine (stock solution prepared in 20 mM Tris, pH 7.4). Then, the embryos were dechorionated by tweezers and mounted in 3% methylcellulose on microscope slides with depression. Images of embryos were recorded on the fluorescent microscope Leica DMI8 (Hamburg, Germany) at 40× magnification. 31
Gene expression analysis
Zebrafish embryos were exposed to 2 μM Cry from 2 hpf to 48 hpf. Total RNA was isolated from the 20 embryos or larvae according to the manufacturer's instructions (RNAiso Plus, Takara Biotechnology). The RNA concentrations were determined spectrophotometrically by measuring the absorbances at 260 and 280 nm, and the RNA quality was assessed through gel electrophoresis. The complementary DNA (cDNA) was synthesised from the total RNA using the All-in-one™ first-strand cDNA synthesis kit (Genecopoeia), according to the manufacturerò¥s suggested protocol, and qRT-PCR was conducted using the All-in-one™ qPCR mix system (Genecopoeia) with SYBR green, according to the standard protocol. The following primer sets were used to amplify the following probes (Table 2), qRT-PCR was performed using an Agilent Stratagene 3005P system. In order to evaluate the effect of cell apoptosis, the murine double minute 2 (mdm2), B-cell lymphoma/leukemia- 2 gene (bcl2), p53 tumor suppressor gene (p53), bcl2 associated X protein (bax), apoptotic protease activating factor-1 (apaf1), caspase 3 (casp3) and caspase 9 (casp9), whose primer sequences were published in the apoptosis pathway were investigated. 32 Elongation factor 1α (ef1α) was used as an endogenous control. 33 The PCRs were performed under the conditions recommended by the manufacturer: 95°C for 10 min for the initial denaturation, 40 cycles of denaturation at 95°C for 30 s, 60°C for 20 s for annealing, and 72°C for 20 s for extension. 2−ΔΔCT method was used to analysis the change in expression of the target gene of Cry treated relative to an untreated control.
RT-qPCR primer used in this study.

95°C for 10 min for the initial denaturation, 40 cycles of 95°C for denaturation, 60°C for 20 s for annealing, and 72°C for 20 s for extension.
Behavioral assays
The normally developed 60 embryos were selected and randomly put into three groups. At 2 hpf, the embryos were exposed to Cry at the doses of 0 (solvent control group), 1, 2 µM of Cry, respectively. After 94 h of exposure, each larva was removed and placed per well in a 96-well plate, except the dead or deformity larvae. Before monitoring, the larvae were acclimated at 28°C for 10 min. Movement and the response of dark-to-light transition of each larva was observed by DanioVision™ (Noldus Information Technology, The Netherlands) and analyzed with module of Etho view XT 14 software.
For touched-evoke escape reaction (TEER), embryos were exposed to Cry from 2 to 96 hpf. Then larvae were allowed to adapt experimental environment for 10 min before touching starts. The response of larvea was evoked by gentle touching the dorsal tail region for 20 times with an eyelash probe. The time of escaping from a certain range (circle with a diameter of 1 cm) after touching was counted, and mean escape times were analyzed in each group. Twenty embryos in each treatment group from three replicate experiments were used for the TEER test.
Validity criteria and statistics
Egg batches were only used, if fertilization rates were ≥80%. An assay was considered valid, if the controls did not show >10% teratogenic plus lethal effects at 96 hpf. Statistical analysis was conducted using SPSS 17.0. Data was expressed with mean ± SD. Student’s T-Test was used and the results were evaluated at a significance level of p < 0.05.
Results
Cry decreases the survival rate and increases deformities in zebrafish embryos
Lethal and teratogenic effects were recorded and summarized at 24, 48, 72, and 96 hpf (Table 3). All teratogenic percentages in the solvent control group were under 10% throughout the trial. After incubation with different concentrations of Cry, the abnormal rate and mortality of embryos were shown in a time- and dose-dependent manner (Table 1). Cry obviously affected the developmental process of embryos. Almost no embryos hatched in Cry groups at 48 hpf when the hatching rate of the vehicle group was 50%. The calculated LC50 and EC50 values of Cry were 7.81 and 3.89 μM at 96 hpf (GraphPad Prism 6), respectively, resulting in a teratogenicity index of 2.0. The corresponding concentration–response curves for Cry are given in Figure 1.
Overview of coagulate and teratogenic effects of Cry in zebrafish Danio rerio embryos at 4 dpf.
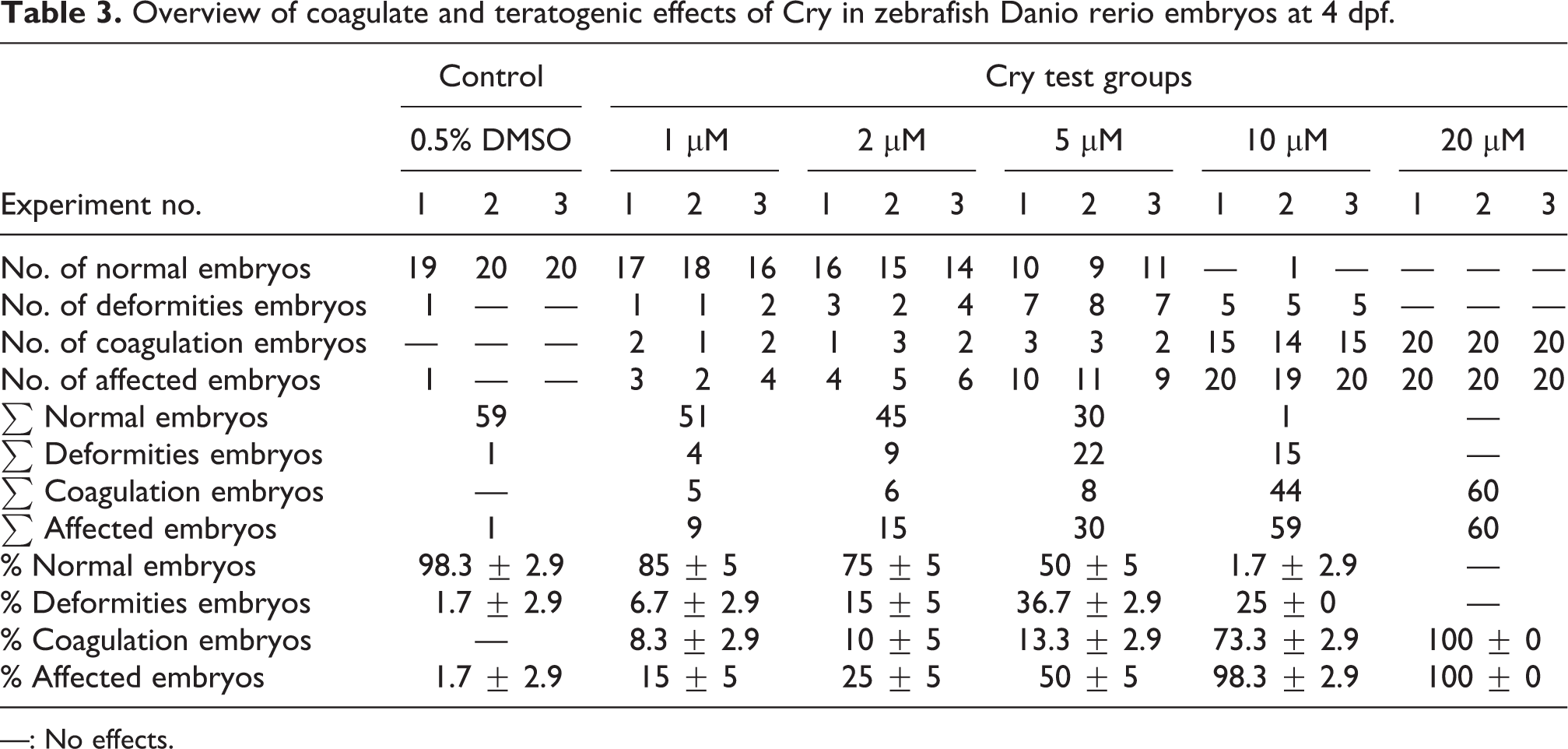
—: No effects.

Concentration–response curves used for the calculations of the LC and EC values (GraphPad Prism 6) for Cry at 96 hpf. The calculated LC50 and EC50 values of Cry were 7.81 and 3.89 μM at 96 hpf, respectively.
The data of morphological changes of zebrafish embryos induced by Cry are listed in Table 4, and the development or teratogenic changes of zebrafish embryos are shown in Figures 2 to 4. The teratogenic rates with high Cry dosages (10 and 20 μM) were high, especially in embryos of the 20 μM Cry group, which were all dead at 72 hpf. The toxicological effect of Cry was observed mainly on the growth process and the heart. Cry delayed the development degree of the embryos by prolonging the hatching process or impeding the development rate (Figure 5). In the heart, pericardial edema frequently occurred in Cry-exposed embryos. Moreover, scoliosis, tail malformation, and yolk sac edema were found in embryos (Figure 4). However, no teratogenic effects were found in the eyes of zebrafish embryos.
Teratogenic effects observed in embryos depending on the different concentrations of Cry at 4 dpf.

* The data of teratogenic of zebrafish at the concentration of 20 μM of Cry were collected at 2 dpf (all of embryos were dead in 3 dpf).

Cry increases deformities and coagulation in zebrafish embryos. Cry (1, 2, 5,10, and 20 μM) was added to the embryos at 2 hpf. Normal and deformed morphological characteristics and coagulation were observed at different time points: (A) 24 hpf, (B) 48 hpf, (C) 72 hpf, and (D) 96 hpf; CG-control group.

Morphology of zebrafish embryos exposed to Cry. The development process of zebrafish embryos were photographed from 1 to 4 dpf of each group. The embryos were obviously deformed in higher concentrations of Cry (10 and 20 μM) and none of them ware survived through 3 dpf in 20 μM Cry.

Major malformations of zebrafish embryo and larvae exposed to Cry. (A) normal embryo, (B) scoliosis, (C) pericardial edema, (D) yolk sac edema, (E) tail curve.

Hatching rate of zebrafish embryos from 1 dpf to 4 dpf. Embryos for the control and experimental groups began to hatch from 24 hpf. Hatching time and hatching success rate decreased with increasing concentrations of 1 μM to 10 μM.
Comparison of human pregnancy category and zebrafsh TI values. 29

Cry induces apoptosis in the brain of zebrafish embryos
Cry-exposed zebrafish embryos were stained with acridine orange. No obvious staining was observed in the solvent control group. On the contrary, a number of apoptotic cells were observed in the embryos exposed to 2 μM Cry at 48 h. Apoptotic cells were observed in the brainstem region (midbrain interneuron area), suggesting possible Cry-induced brain system impairment in zebrafish embryos (Figure 6(A)).

(A) Cry induces apoptosis in zebrafish embryos. Zebrafish embryos (2 hpf) were exposed to 5 μM Cry for 48 h and stained with acridine orange. Representative images are shown. Red arrows point to the apoptotic cells accumulated in the brain of the embryos. (A) blank control, (B) solvent control, (C) 5 μM Cry; (B)Cry alters caspase 3 expression of apoptosis. Zebrafish embryos (2 hpf) were exposed to 5 μM Cry for46 h. mdm2, apaf1, bcl-2, bax, casp3, and casp9 mRNA levels were analyzed by qRT-PCR. Compared with controlled group, *p < 0.05.
Cry alters the expression of apoptotic gene in zebrafish embryos
To further analyze the apoptotic effects of Cry on zebrafish embryos, several markers of apoptosis were selected and detected using qPCR. The expression levels of MDM2, APAF-1, Bcl-2, Bax, and caspase-9 were not significantly changed in the 2 μM Cry-exposed embryos compared with those in the solvent control group. However, a significant increase in caspase-3 (P < 0.05), which is a classical marker of apoptosis, was observed in the Cry-exposed embryos (Figure 6(B)).
Behavioral assays
The locomotor behavior of zebrafish larvae was monitored by a dark–light transition stimulation period. The movement speed of the larvae in each group was significantly decreased when the larvae were exposed from light to dark (Figure 7). During the first dark period, the mean swimming speed of the larvae in the 1 μM Cry-exposed group was significantly reduced compared with that in solvent control group (P < 0.01). In the second dark–light photoperiod, the swimming speed of larvae was significantly reduced in the 2 μM Cry-exposed group. The average duration and frequency of rest, active, and manic behavior in 30 s are shown in Figure 8. Compared with the control group, the Cry-exposed groups showed significantly increased rest duration, but obviously decreased active and manic duration. The TEER test was performed, and the results showed that the escape reaction of larvae was reduced significantly under the effect of Cry. The 2 μM Cry-exposed group had an average escape time of 1.13, which was significantly different from the solvent control group with an average escape time of 12.25 (Figure 9).

Cry change of swimming speed of zebrafish larvae. The zebrafish embryo was exposed to Cry for 94 h, and the larval movement status was measured by the behavioral recorder. (A) The zebrafish larvae movement state during a dark–light–dark–light period stimulation test. (B) Average swimming speed at intervals of 30 s under the dark–light stimulation. *p < 0.05, **p < 0.01 compared with the control group.

Locomotor behavior of zebrafish embryos exposed to Cry. Cry changes the durations and frequency of the rest, active and manic at intervals of 30 s. *p < 0.05, **p < 0.01 compared with the control group.

The results of For touched-evoke escape reaction (TEER) experiment. The 2 μM Cry-exposed group showed significant decrease in escape time when compared with control group (**p < 0.01).
Discussion
RSM is a popular traditional herb in Chinese, Korean, and Japanese medicine. It has been widely used in the clinic to treat diabetes and cardiovascular diseases.34,35 As one component of RSM, Cry has an identifiable chemical structure, and its purity exceeds 99%, making it possible to evaluate the toxicity mechanism of Cry in vivo. Laboratory rodent studies evaluating the toxicity of drugs are usually expensive and time consuming. Zebrafish is proved to be a successful animal model for discriminating teratogens and nonteratogens. 36 The results of this study showed that Cry is teratogenic as it had a TI value of 2. One of the teratogenic effects of Cry was the growth retardation of embryos during the hatching process in all Cry-exposed groups. However, this effect was gradually relieved over time. Pericardial edema was another effect that was observed. Considering that RSM is mainly used to treat cardiovascular diseases, the suitability of Cry in treating cardiovascular diseases in clinics remains questionable. Other teratogenic effects that occurred after Cry exposure include scoliosis and deformity of the yolk or tail.
The results of acridine orange staining showed that many cells underwent apoptosis in the embryonic brainstem region, indicating that the Cry-induced apoptotic process focused on this specific region and the neurocytes in this area might be damaged. 37 In this study, the mRNA expression of caspase-3 in the 2 μM Cry-exposed group increased by 2.1-fold compared with the control group. Caspase-3 is thought to play a central role as an effect of caspase in the proapoptotic signaling from the caspase cascade to cell death by proteolytic processing of proteins and is essential for certain processes associated with the dismantling of the cell. 38 The tissue-specific developmental expression patterns of the caspase-3 gene regulate the spatial and temporal distribution of apoptotic cells. 39 In the central nervous system, caspase-3 has been identified as a key mediator of neuronal programmed cell death. Strong or uncontrolled activation of caspase-3 might lead to activation of the full apoptotic cascade and neuron death in neurodegenerative diseases. 40 The upregulation of mRNA expression of caspase-3 and apoptosis in the embryonic brainstem suggested that Cry might accelerate the apoptosis of neurons in the brain and finally lead to the growth retardation in embryos.
Further study found that the locomotor ability of embryos was greatly reduced by Cry. The movement of larva depends on the activation of locomotor muscles, which are controlled by spinal motoneurons. 41 As a well-known developmental neurotoxicant, chlorpyrifos has been proved to significantly change the swimming activity of larvae, and the swimming ability of larvae has been proposed as a screening method for neurodevelopmental toxicity studies. 42 Our study found changes in the rest, active, and manic duration, which are also related to symptoms of brainstem injury. 43 Touching the tail activates spinal Rohon–Beard sensory neurons, which project to the hindbrain and innervate reticulospinal Mauthner neurons. 44 The results of TEER and locomotor ability tests suggested that the development of neurons in the brain stem area was damaged, and this damage was confirmed by acridine orange staining. The experiments showed that Cry could cause zebrafish growth retardation, cardiac edema, and nerve cell apoptosis. These toxicological effects might threat the health of some patients when Cry is utilized in clinical, especially for the pregnancy women. Because the intrauterine growth retardation may result in significant fetal morbidity and mortality. 45 However, the experiment focused on the toxicological effects of Cry on the early development stage of zebrafish, which had certain limitations for the appearance of these side effects (Table 5).
In conclusion, Cry altered the growth process of embryos by inducing the apoptosis of neurons in the brainstem area and retarded the locomotor ability of zebrafish at the early stage. However, further studies are necessary to better understand the mechanism of neuronal apoptosis in the brain.
Supplemental material
Supplemental Material, sj-pdf-1-het-10.1177_09603271211009954 - Developmental toxicity of cryptotanshinone on the early-life stage of zebrafish development
Supplemental Material, sj-pdf-1-het-10.1177_09603271211009954 for Developmental toxicity of cryptotanshinone on the early-life stage of zebrafish development by C Wang, T Wang, B-W Lian, S Lai, S Li, Y-M Li, W-J Tan, B Wang and W Mei in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work is supported by Medical Scientific Research Foundation of Guangdong Province (Grant No. A2017172).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
