Abstract
Manganese (Mn) complexes are widely studied because of their important catalytic properties in synthetic and biochemical reactions. A Mn (III) complex of an amidoamine ligand was synthesized using a tetradentate amidoamine ligand. In this study, the Mn (III) complex was evaluated for its biological activity by measuring its cytotoxicity in human breast adenocarcinoma cell line (MCF-7). Cytotoxic effects of the Mn (III) complex were determined using established biomarkers in an attempt to delineate the mechanism of action and the utility of the complex as a potential anticancer drug. The Mn (III) complex induces cell death in a dose- and time-dependent manner as shown by microculture tetrazolium assay, a measure of cytotoxic cell death. Our results demonstrated that cytotoxic effects were significantly increased at higher concentrations of Mn (III) complex and with longer time of treatment. The IC50 (Inhibitor concentration that results in 50% cell death) value of Mn (III) complex in MCF-7 cells was determined to be 2.5 mmol/L for 24 hours of treatment. In additional experiments, we determined the Mn (III) complex–mediated cell death was due to both apoptotic and nonspecific necrotic cell death mechanisms. This was assessed by ethidium bromide/acridine orange staining and flow cytometry techniques. The Mn (III) complex produced reactive oxygen species (ROS) triggering the expression of manganese superoxide dismutase 1 and ultimately damaging the mitochondrial function as is evident by a decline in mitochondrial membrane potential. Treatment of the cells with free radical scavenger, N, N-dimethylthiourea decreased Mn (III) complex–mediated generation of ROS and attenuated apoptosis. Together, these results suggest that the Mn (III) complex–mediated MCF-7 cell death utilizes combined mechanism involving apoptosis and necrosis perhaps due to the generation of ROS.
Introduction
Metal complexes, due to their unique structures and reactivates, are attractive in several biomedical applications including anticancer, anti-inflammatory, and antidiabetic reactions. 1 –4 Metal complexes and nanoparticles are highly useful in biological imaging too. 5 Among several biologically prevalent metals, manganese complexes are well known to participate in various important biochemical processes. 6 Particularly, high-valent manganese species are believed to play a role in the oxidation of water in membrane-bound protein (photosystem II) during photosynthesis. 7,8 Some manganese complexes also act as necessary cofactors for enzymatic reactions. 9 It is known that transferrin/transferrin receptor system is responsible for absorption and transportation of manganese ions into the cells where they involve in biological reactions. 10,11 It has also been found that transferrin receptor is highly expressed in some tumor tissues 12 –14 and hence their relevance in cancer cell biology. Interestingly, simple manganese salts 15,16 and different manganese complexes of thiosemicarbazone or hydrazone ligands, 17,18 2-pyridylmethyl amino-2-propionic acid, 19 and thiocyanate ligands 20 have been used for anticancer activities.
Manganese, being a redox active metal, can interfere with the cellular redox homeostasis, thereby producing reactive oxygen species (ROS) that can kill cancer cells. However, in most of the cases, the concentration of manganese used to kill cancer cells was rather high (in mM range). To the best of our knowledge, most previous studies used Mn (II) complex and very limited studies are available on biomedical applications of Mn (III) complex. This is the first attempt to use Mn (III) complex or its salt to test its ability to kill cancer cells. Recently, we have reported the synthesis of a novel Mn (III) complex of an amidoamine ligand, which was characterized using various analytical techniques, including 1H NMR, electrospray ionization mass spectrometer, and single-crystal X-ray diffraction. X-ray crystallography data have demonstrated that the complex is a 6-coordinated species. 21 We used the complex for in situ synthesis of epoxide in the presence of peroxides, and subsequently, the complex also catalyzed the synthesis of cyclic carbonate. We detected rare high-valent manganese (V)-oxo species during our reaction. However, no biomedical application was performed for the water-soluble Mn (III) complex. Taking into account that the Mn (III) complex is capable of producing high-valent oxo species, we hypothesized that the complex may produce ROS, which will cause cellular damage. Cellular damage to cancerous cells, particularly resistant cancerous cells, is a desirable trait for an anticancer drug. In order to develop therapeutic application of the Mn (III) complex in cancer treatment, any toxic effects of this complex need to be determined. Therefore, the goal of this study was to examine if the Mn (III) complex causes any toxic effects, particularly in cancerous cells.
Cancer is the most common cause of death worldwide after cardiovascular diseases. Despite many therapeutic advances such as surgery, chemotherapeutic, radiation, and gene profile, cancer mortality is still high. 22 Hence, direct manipulation of the compounds that regulate apoptosis, a programmed cell death process, is an interesting method of treating cancer. Human breast cancer causes extensive mortality in the Western world, where approximately one-third of women diagnosed with the disease develop metastases. Many breast cancer therapies have limitations such as nonspecific targeting, increased drug resistance, and toxicity to normal tissues. 23 Despite the use of conventional therapy, a 40% recurrence rate has been reported for locally advanced breast cancer, after the diagnosis and removal of the primary tumor. 24,25 Therefore, new therapeutic approaches are required that can inactivate cancer cells without damaging normal cells. Cancer cells tend to escape cell death process and often show resistance for treatment by cytotoxic drugs. Hence, new drugs that induce cancerous cell death by apoptosis are in high demand.
Apoptosis is an important component of multistep process of carcinogenesis and therapy of resistant cells. It includes terminal morphological and biochemical events, 26 leading to cell shrinkage, membrane blebbing, chromatin condensation, and the formation of membrane-enclosed apoptotic bodies that are phagocytosed to dispose of the cellular debris. 27 The apoptotic phases are characterized by DNA fragmentation, loss of mitochondrial membrane integrity, and release of molecules that initiate activation of intracellular proteases. The mitochondria play an important role in the initiation of apoptotic cascade by acting as a convergence center of apoptotic signals. 28 It is also the most active organelle for cellular oxidoreductive reactions, which may compromise mitochondrial membrane potential (MMP) upon severity of the generation of ROS. Thus, mitochondria has been considered as an important factor in the execution of apoptotic cell death. 29,30
The MCF-7 cell line was developed by the Michigan Cancer Foundation (from which it derives its name) in 1973. 31 It has been widely used in in vitro research due to its inherent lack of functional caspase-3. 32 The MCF-7 cell line became highly resistant to most chemotherapeutic agents because of the absence of a crucial effector protease and subsequently led to resistance to apoptosis. 33 Studies have shown that MCF-7 cell line is resistant to classical breast cancer chemotherapeutic agents, for example, paclitaxel, camptothecin, doxorubicin, and etoposide. 34 Therefore, in this biological study, we investigated whether the above synthesized and characterized Mn (III) complex causes any cell death in MCF-7 cells. Further, we determined the mechanism of action of the Mn (III) complex on cell death process.
Materials and Methods
Materials
The MCF-7 cell line, Dulbecco modified eagle medium (DMEM), penicillin, streptomycin, and fetal bovine serum (FBS) were obtained from the American Type Culture Collection (ATCC; Manassas, Virginia). YO-PRO-I Vybrant apoptosis assay kit #4 (V-13243; Molecular Probes, ThermoFisher Scientific, Eugene, Oregon) for labeling with Fluorescein Isothiocyanate (FITC) and propidium iodide (PI) for flow cytometry analysis were purchased from Invitrogen (ThermoFisher Scientific, Carlsbad, California). Microculture tetrazolium (MTT) reagents, Dimethyl Sulfoxide (DMSO), 5′, 5′, 6′, 6′ tetrachloro-1′, 1′, 3′, 3′-iodide (JC-1) dye, 2′, 7′dichlorodihydrofluoresceindiacetate (DCFH-DA) fluorescence dye, ascorbic acid, and other reagents were obtained from Sigma-Aldrich (St Louis, Missouri). Super oxide dismutase manganese 1 (MnSOD1) antibodies were obtained from Cell Signaling Technology (Danvers, Massachusetts). N, N-dimethylthiourea (DMTU) was obtained from Thermo Fisher Scientific (Grand Island, New York).
Synthesis of Mn (III) Complex
Synthesis of Mn (III) complex was performed according to the literature method. 21 All reactions were performed under inert atmosphere. First, the amidoamine ligand was synthesized and then the ligand was metallated with manganese metal salt precursor to obtain the Mn (III) complex. In short, 2-aminoisobutyric acid was reacted with phthalic anhydride and then the protected amino acid was reacted with thionyl chloride to make the corresponding acid chloride of the protected amino acid. The acid chloride was then reacted with o-phenylenediamine in the presence of triethylamine to obtain a phthalic acid protected diamide-diamine. The resulting compound was then reacted with hydrazine hydrate in ethanol to obtain the diamide–amine ligand. The ligand was taken in dry tetrahydrofuran and reacted with n-butyl lithium. Manganese (II) chloride was added to the solution, and on exposure to air, the complex was converted to Mn (III) complex. It was then crystallized from methanol. The complex was characterized by elemental analysis, 1H-NMR, electrospray ionization mass spectrometry, and X-ray crystallographic technique as mentioned in our previous publication. 21
Cell Culture
Cells were grown following instructions from ATCC. A complete medium containing DMEM with 10% FBS and the antibiotics penicillin (500 U/mL) and streptomycin (500 U/mL) were used. The cells were grown in a humidified CO2 incubator set at 37°C with 5% CO2 atmosphere. For subculturing, cells were washed with phosphate-buffered saline (PBS; 11.9 mmol/L phosphates, pH 7.4, 13.7 mmol/L NaCl, 2.7 mmol/L KCl) and then detached with 3 mL/75-cm2 flask trypsin–EDTA (0.05% trypsin/0.53 mmol/L EDTA in Hank balanced salt solution without sodium bicarbonate, calcium, and magnesium) by incubation at 37°C in 5% CO2 atmosphere for 5 minutes or until cells detached. After detachment, cells were collected by centrifugation at ∼500g for 5 minutes. The cells were resuspended in the complete medium. Cell density was determined by counting the number of cells using hemocytometer following staining with Trypan blue dye.
Cytotoxicity Assay
Microculture tetrazolium assay, a colorimetric method to measure cell viability as an indicator of cytotoxicity, was used to access the toxic effects of the compound. The MTT assay is based on that the yellow water-soluble substrate 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide is reduced into an insoluble colored formazan product by mitochondrial succinate dehydrogenase enzyme in live cells. 35 Since the reduction of MTT can only occur in metabolically active cells, the level of activity correlates with the viability of the cells.
The human breast cancer MCF-7 cells were trypsinized, and the cell count was adjusted to 20 × 103 cells/well in 96-well plates using DMEM containing 10% FBS, 1% penicillin, and 1% streptomycin. When the monolayer was formed after 24 hours, the cells were treated with ethanol as a vehicle control and different concentrations of Mn (III) complex (1.25, 2.5, 6.25 mmol/L) and incubated at 37°C in the 5% CO2 incubator for another 24 hours. After incubation, the medium was removed and cells were washed twice with the FBS-free medium and replaced with a mixture of 10 μL of MTT dye and 90 μL FBS-free medium per well. The 96-well plates were then gently shaken for 3 minutes and incubated for 4 hours at 37°C in a 5% CO2 incubator. Afterward, the supernatant was removed and 100 μL of DMSO was added in each well of 96-well plates. The plates were mixed by pipetting gently to solubilize the formed formazan. By using a fluorescence microplate reader (SYNERGY H4, hybrid technology; BioTek), the absorbance was measured at a wavelength of 570 nm. The percentage of growth inhibition was calculated using the following formula
36
:
% Cell inhibition = (Absorbance value of test compound − Absorbance value of blank)/(Absorbance value of control − Absorbance value of blank) × 100.
Ethidium Bromide/Acridine Orange Staining
The nucleic acid-binding fluorochromes ethidium bromide/acridine orange (EB/AO) were used to determine the cell viability and apoptosis of the MCF-7 cells via fluorescence microscopy. After treatment of cells with ethanol and Mn (III) complex, the cells were washed using 1× PBS (pH 7.4) and then stained with 1:1 ratio of a solution consisting of 100 mg/mL AO and 100 mg/mL EB in PBS. Following washing, cells were immediately visualized under UV light using a Nikon Labophot fluorescence microscope equipped with a digital camera. Fields were selected randomly for photographs. Given that EB stains fragmented nuclear DNA red, whereas AO stains live cells green, counts of live cells (green) and apoptotic cells (red) were obtained from these images. Approximately 200 to 300 cells per treatment were counted for statistical calculation. 37
Flow Cytometry Analysis
The flow cytometry technique was performed for the separation of apoptotic and necrotic cells. The cells were stained with YO-PRO-I and PI dyes using Vybrant apoptosis assay kit #4 (V-13243; Molecular Probes), according to the manufacturer’s protocol. The MCF-7 cells were seeded at 600 × 103 cells/mL and then treated with Mn (III) complex to induce apoptosis. Control cells were treated with corresponding concentrations of ethanol used as vehicle. Next, the cells were harvested by 0.05% trypsin and washed twice with 1× PBS, pH = 7.4. Afterward, 1 μL of YO-PRO-I stock solution (component A) and 1 μL of PI stock solution (component B) were mixed per milliliter of cell suspension. After 30 minutes of incubation at 4°C, the cells were analyzed based on cell fluorescence profiles using BD FasCalibur flow cytometry to sort out fluorescently labeled cells in a population. Fluorescence emissions were measured at 515 to 545 nm for FITC using a FL-1 PMT detector and 564 to 606 nm for PI using FL-2 Pre-menstrual Tension (PMT) detector. In a quadrant, live cells will show little or no fluorescence in the bottom left panel, whereas necrotic cells will reveal red and green fluorescence in the top left and the top right panels, respectively, and apoptotic cells have green fluorescence in the bottom right panel. 37 Yo-Pro-1 has been used as a marker to detect early and late apoptotic events by many investigators including our laboratory. 37 –40 Yo-Pro-1-based flow cytometry detects changes in plasma membrane phospholipid orientation, similar to annexin V-based flow cytometry, and both methods provide comparable results. 40
Determination of ROS
The MCF-7 cells were plated in black, clear-bottomed, 96-well plates at a cell density of 20 × 103 cells per well in order to assess the production of intracellular ROS levels. Cells were treated with Mn (III) complex or ethanol as vehicle control for 24 hours. Afterward, DCFH-DA substrate was added to the medium at a final concentration of 10 μmol/L and incubated in dark for 30 minutes. The cells were then washed twice with 1× PBS (pH 7.4) and resuspended in 100 μL of PBS. The DCFH-DA intensity was determined using a fluorescence microplate reader (SYNERGY H4, hybrid technology; BioTek) at 485/535 nm .The data were obtained from experiments that were performed independently in triplicate. 41 To examine the effects of free radical scavengers on ROS production and its effects on cell death, scavengers were incubated 30 minutes prior to treatment with Mn (III) complex.
Determination of MMP
The MCF-7 cells were seeded at a density of 20 × 103 cells/well in 96-well plates and incubated overnight. Cells were then treated with Mn (III) complex or ethanol as vehicle control. Staurosporine was used as a positive control. After 24 hours, cells were washed with 1× PBS, pH 7.4, and incubated with medium containing 10 μL of 10 mg/mL 5′, 5′, 6′, 6′-tetrachloro-1′, 1′, 3′, 3′-iodide (JC-1 dye) for 20 minutes at 37°C. Finally, the cells were washed and resuspended in 100 μL PBS for fluorescent measurements using a microplate reader (SYNERGY H4, hybrid technology; BioTek). Due to the electrochemical potential gradient in normal cells, the dye concentrates in the mitochondrial matrix where it forms red fluorescent aggregates. A reaction that affects the MMP prevents the accumulation of the JC-1 dye in the mitochondria; thus, the dye is dispersed throughout the entire cell leading to a shift from red (590 nm) to green (525 nm) fluorescence. Mitochondrial depolarization is indicated by a decrease in the red/green fluorescence ratio. Each experiment was repeated at least 3 times in triplicate. 42
Preparation of Cell Lysates and Western Blotting
After appropriate treatments with ethanol, Mn (III) complex, and H2O2 as a positive control to induce cytotoxicity or apoptosis, the culture media were collected and centrifuged at 500g for 5 minutes to get the cell pellet. Cell lysates were prepared by first washing the attached cells once with ice-cold 1× PBS (pH = 7.4) and then lysed for 20 minutes with a lysis buffer Radioimmunoprecipitation Assay (RIPA) containing a protease inhibitor cocktail, a 1 mmol/L phosphatase inhibitor cocktail, and 0.1 mol/L Phenylmethylsulfonyl Fluoride (PMSF). The floating cells collected were mixed with corresponding cell lysates. Following cell lysis, cell debris was removed by centrifugation at 12 000g (Eppendorf centrifuge) for 15 minutes at 4°C. Protein concentration in cell lysates was determined by a standard Coomassie Bradford protein assay according to the manufacturer’s instructions (Bio-Rad, Hercules, California). Western blotting procedure was followed as described by Agrawal and group. 37
In order to prepare protein samples for sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), cell lysates were mixed with equal volumes of 2× SDS sample buffer (0.5 mol/L Tris-HCl, pH = 6.8, 20% glycerol, 4% [wt/vol] SDS, 0.5% [wt/vol] bromophenol blue) and boiled for 5 minutes at 100°C. Samples containing 100 μg proteins were separated by 10% SDS-PAGE. The proteins from the gels were electrophoretically transferred onto polyvinylidene fluoride membranes (Millipore, Billerica, Massachusetts). The membranes were blocked with 5% skim milk prepared in Tris-buffer saline (150 mmol/L NaCl and 20 mmol/L Tris-HCl, pH = 7.2) containing 0.05% Tween-20 (TBS-T, Sigma–Aldrich, St Louis, Missouri) for 1 hour at room temperature. The membrane was then incubated with diluted primary antibody overnight at 4°C with constant shaking. The primary antibody dilution used for MnSOD1 was 1:1,000 prepared, according to the manufacturer’s recommendations. After primary antibody incubation, the membranes were washed 3 times for 10 minutes each with TBS-T and were incubated for 2 hours with a 1:2,000 dilution of antirabbit immunoglobulin G conjugated to horseradish peroxidase in 2% skim milk in TBS-T. The membranes were then washed 3 times for 10 minutes each with TBS-T. Signal detection was achieved with a Super Signal West Pico Chemiluminescence kit (Pierce Biotechnology, Rockford, Illinois) and high-performance chemiluminescence film (Amersham Biosciences, Piscataway, New Jersey).
Statistical Analysis
The results are expressed as a mean ± SD from 3 independent experiments. One-way analysis of variance test was used to evaluate statistical significance between control and experimental groups. The P values (a P ≤ 0.05 and b P ≤ 0.01) were taken as the value with significant differences as compared with their controls.
Results
Mn (III) Complex Induces Cytotoxicity in MCF-7 Cells
In our experiments, the ability of the Mn (III) complex (Figure 1) to promote cell death or cytotoxicity was evaluated by MTT assay using human breast cancer cell line MCF-7. The results are presented in Figure 2. This method provides comparable cytotoxic effects. Our cytotoxicity assay indicates that the Mn (III) complex causes cytotoxicity in MCF-7 cells in a dose-dependent manner. In further experiments, using a medium dose of 2.5 mmol/L, we determined whether the cytotoxicity in MCF-7 cells was dependent upon the time of the treatment. Results (Figure 3) show that the Mn (III) complex-mediated cytotoxicity was also time dependent. The IC50 value derived from the above experiments for MCF-7 cell line was determined to be 2.5 mmol/L at 24 hours of treatment. The cytotoxicity was increased further with increasing the time of incubation with the complex. Although apparently a high concentration is needed to achieve cytotoxicity in cancer cells, there are no other data available in the literature to compare to study on this compound. Future studies would be to study the effect of this complex in normal breast cells to compare its relative effects on cancer cells.
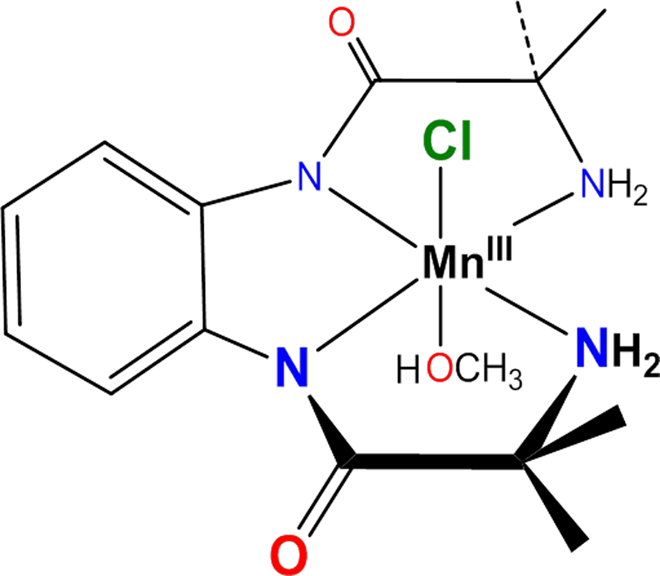
Chemical structure of Mn (III) complex.
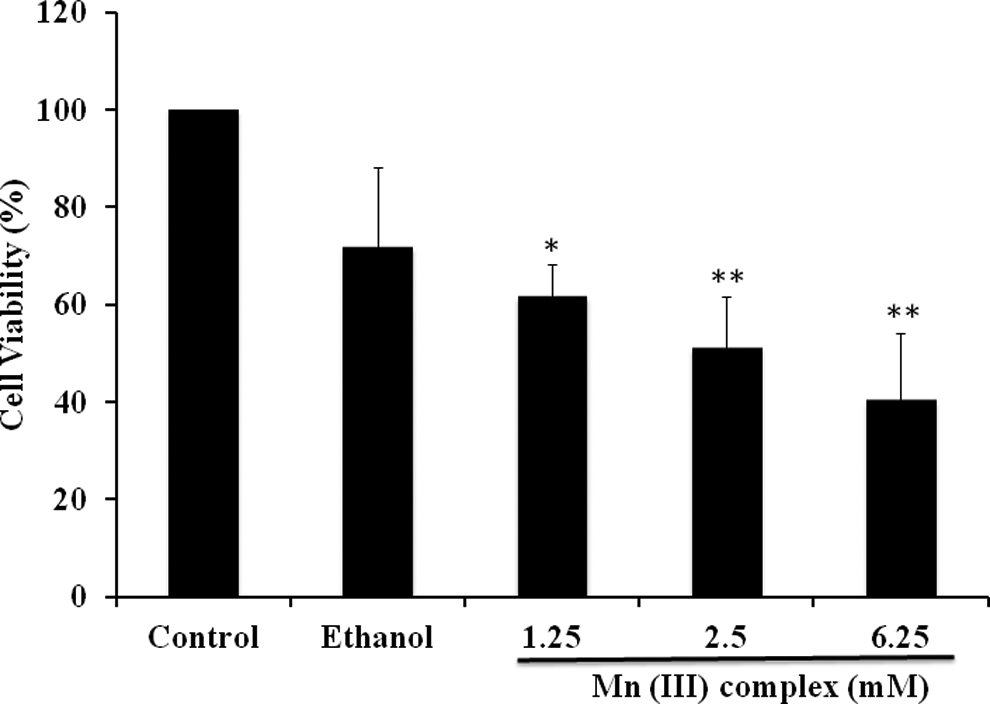
Manganese (III) complex inhibits cancer cell proliferation. Human breast adenocarcinoma cell line (MCF-7) cells were seeded at a density of 20 × 103 cells/well in 96-well plates. After 24 hours, cells were treated with 1.25, 2.5, and 6.25 mmol/L of Mn (III) complex or ethanol as vehicle control. After 24 hours, the cell viability was determined by MTT assay. The values shown are mean ± standard deviation from 3 experiments performed in triplicate. The P value *P ≤ .05; **P ≤ .01, were taken as the value with significant differences as compared with their controls.
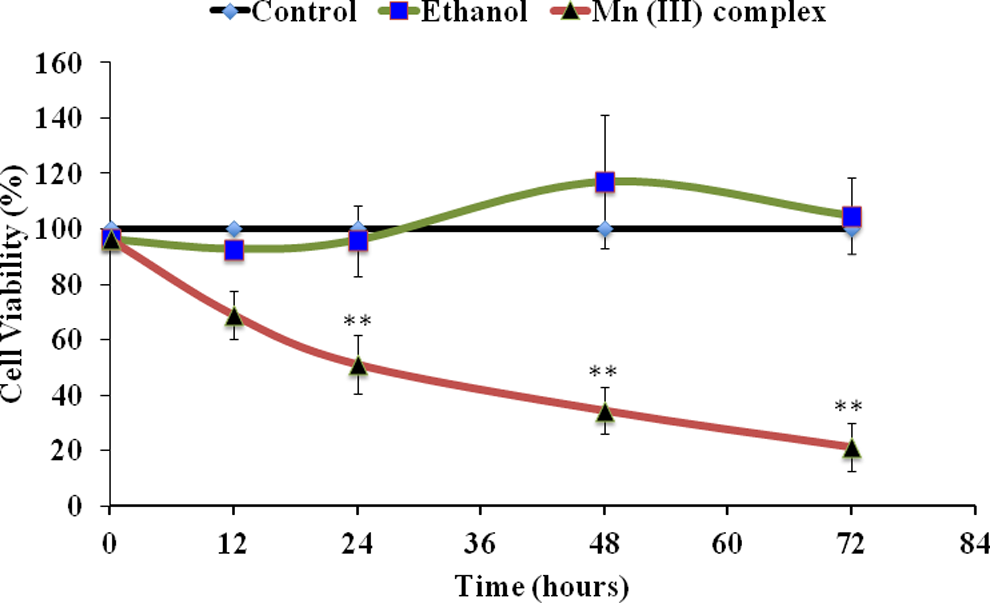
Time-dependent effect of Mn (III) complex. Human breast adenocarcinoma cell line (MCF-7) cells were seeded at a density of 20 × 103 cells/well in 96-well plates. Cells were treated with 2.5 mmol/L of Mn (III) complex or ethanol as vehicle control for indicated times (12, 24, 36, 48, and 72 hours). The cell viability was determined by MTT assay. Results shown are the mean values ± standard deviation determined from triplicate samples repeated 3 times independently. The P values **P ≤ 0.01 were taken as the value with significant differences as compared with their controls.
Cytotoxicity of Mn (III) Complex Is Partly Mediated by Apoptosis
Our goal was to determine whether the Mn (III) complex–induced cytotoxicity was mediated via its apoptotic effects or merely due to nonspecific cell death by necrosis. The AO/EB staining was used to determine the apoptotic cell death. This method is frequently used to study the induction of apoptosis. Characteristic morphological changes due to apoptosis were assessed by fluorescence microscopy using EB/AO staining at 2.5 mmol/L of Mn (III) complex after 24 hours of treatments. The dose selected was close to the IC50 value as determined from dose- and time-dependent experiments. Quantitative analysis of the staining data showed a significantly greater amount of cell death in the MCF-7 cell line (Figure 4) complementing our cytotoxicity data. Therefore, it is likely that the Mn (III) complex–induced cytotoxicity could be at least partly related to apoptotic cell death.
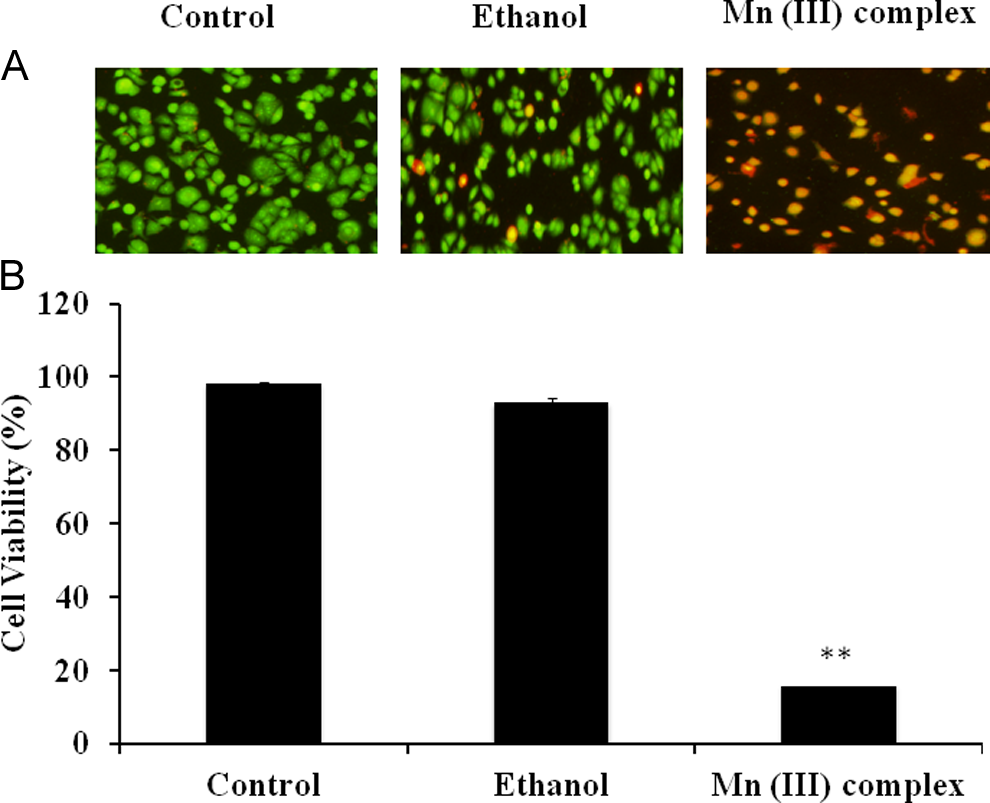
Effect of Mn (III) complex on apoptosis as determined by fluorescence microscopy. Human breast adenocarcinoma cell line (MCF-7) cells were seeded at density 10 × 104 cells/well in 6-well plates. Cells were treated with either 2.5 mmol/L Mn (III) complex or ethanol as vehicle control for 24 hours to induce apoptosis. Cells were then stained with acridine orange/ethidium bromide and visualized under UV light using a fluorescence microscope as described in materials and methods. (A) The percentage of apoptosis was determined by counting live (green) and dead (red) cells. (B) Values in graph are mean ± standard deviation from 3 experiments performed in triplicates. The P value **P ≤ 0.01 was taken as the value with significant differences as compared with their controls.
To confirm whether the Mn (III) complex–induced cell death was due to apoptosis, we performed flow cytometry analysis. This method distinguishes cells undergoing apoptosis and necrosis from the normal population. Figure 5 suggests that the Mn (III) complex–treated cells had a significantly higher population of both apoptotic and necrotic cells compared with respective controls. These results indicate that the Mn complex brings about apoptotic and necrotic cell death. The overall cell death results, however, confirm our previous findings from the cytotoxicity assay.
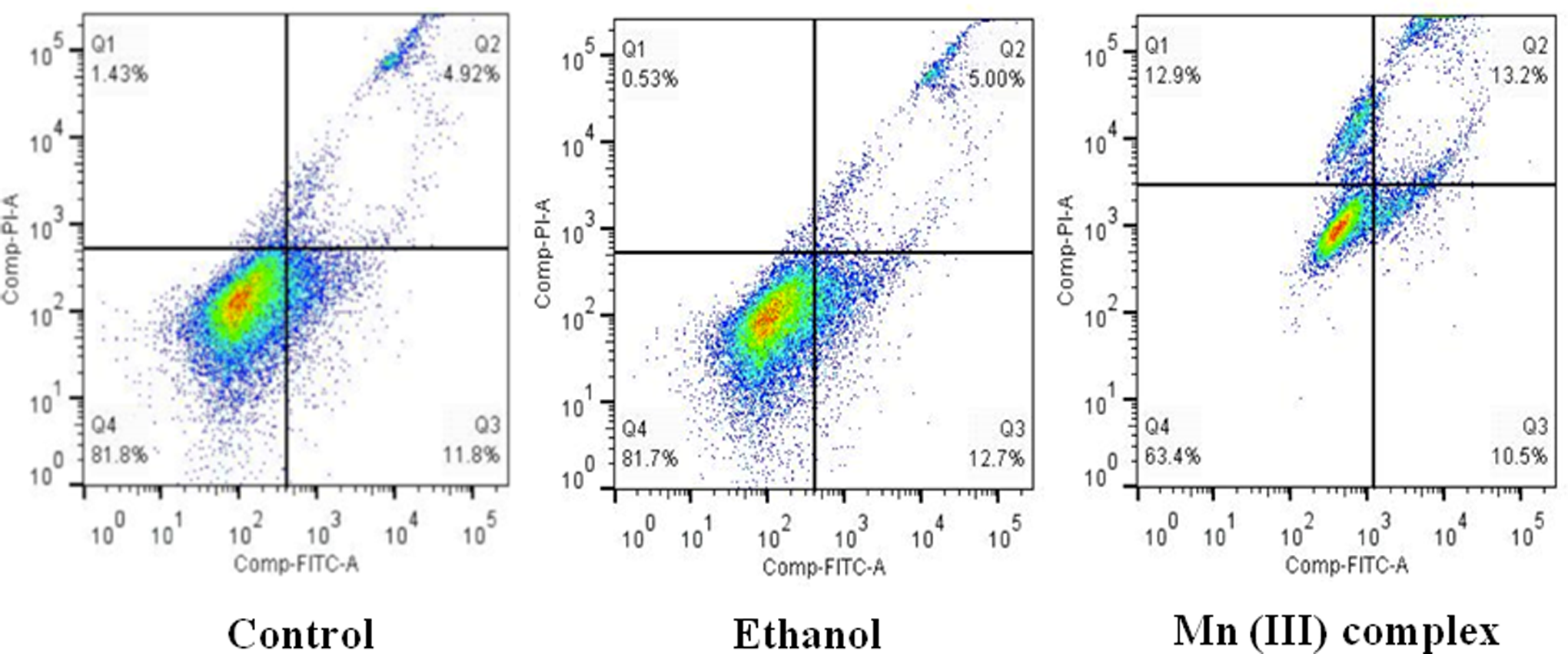
Determination of specificity of Mn (III) complex-induced apoptosis by flow cytometry. Human breast adenocarcinoma cell line (MCF-7) cells were cultured at a density of 600 × 103 cells/mL in 6-well plates. Cells were then treated with or without 2.5 mmol/L of Mn (III) complex for 24 hours. Ethanol was used as a vehicle control. Apoptosis and necrosis were determined by flow cytometry using the vibrant apoptosis assay kit #4 under optimized conditions as per manufacturer’s instructions. Shown in a quadrant are live cells (lower left), necrotic cells (upper left, upper right), and apoptotic cells (lower right). Shown is a representative experiment repeated twice with similar results.
Mn (III) Complex Produces ROS
Since mitochondria are the primary instigators of ROS production, we examined whether the Mn (III) complex affects production of ROS in mitochondria. The ROS is another criterion to evaluate oxidative cellular damage and, thus, cytotoxicity. We, therefore, examined the level of ROS production in treated MCF-7 cells and compared to the level found in the control. Our results suggested that the Mn (III) complex significantly increased ROS production in treated MCF-7 cells (Figure 6).
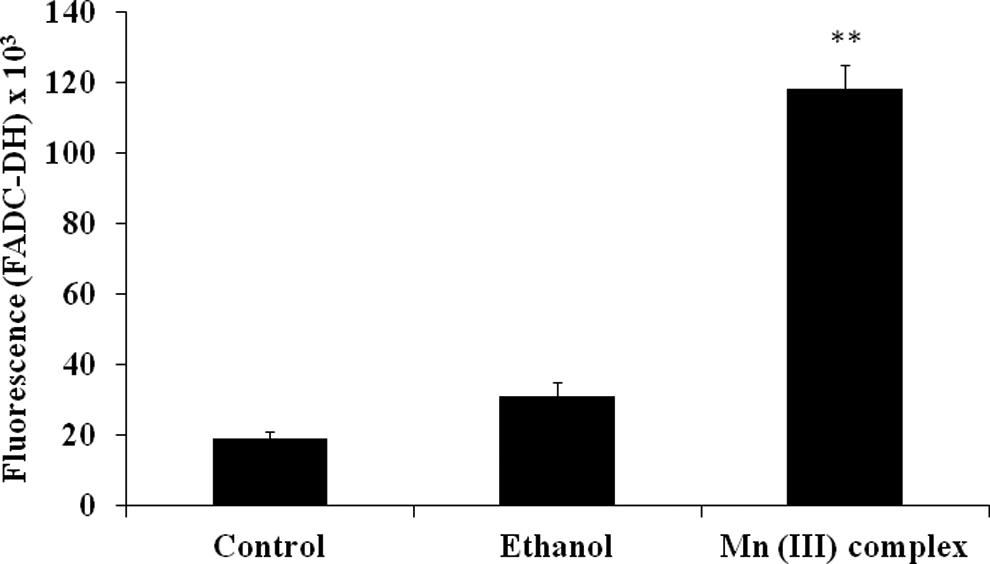
Production of reactive oxygen species (ROS) following Mn (III) complex treatment. Human breast adenocarcinoma cell line (MCF-7) cells were plated at a density of 20 × 103cells/well in 96-well plates for 24 hours. Cells were then treated with or without ethanol as a vehicle control or 2.5 mmol/L of Mn (III) complex for 24 hours. Reactive oxygen species levels were determined using 2’, 7’dichlorodihydrofluoresceindiacetate (DCFH-DA) assay. The values were reported as fluorescence intensities measured at an excitation wavelength of 485 nm and emission wavelength of 535 nm using a fluorescence microplate reader. Data shown are mean ± standard deviation obtained from 3 experiments, each determined in triplicate. The P value **P ≤ 0.01 was taken as the value with significant differences as compared with their controls.
The MnSOD1, a marker for cytotoxic damage by oxygen-free radicals, was also measured in response to the Mn (III) complex treatment. Western blotting results shown in Figure 7 suggest that the Mn (III) complex induced the expression of MnSOD1 in MCF-7 cells as was the increased expression of MnSOD1 by H2O2 used as positive control in this experiment. The expression of actin used as a protein loading control remained constant in all treatments. These results confirm our findings (Figure 6) that the Mn (III) complex induces cytotoxicity due to production of oxidative stress in mitochondria perhaps due to the increases in ROS production in MCF-7 cells.
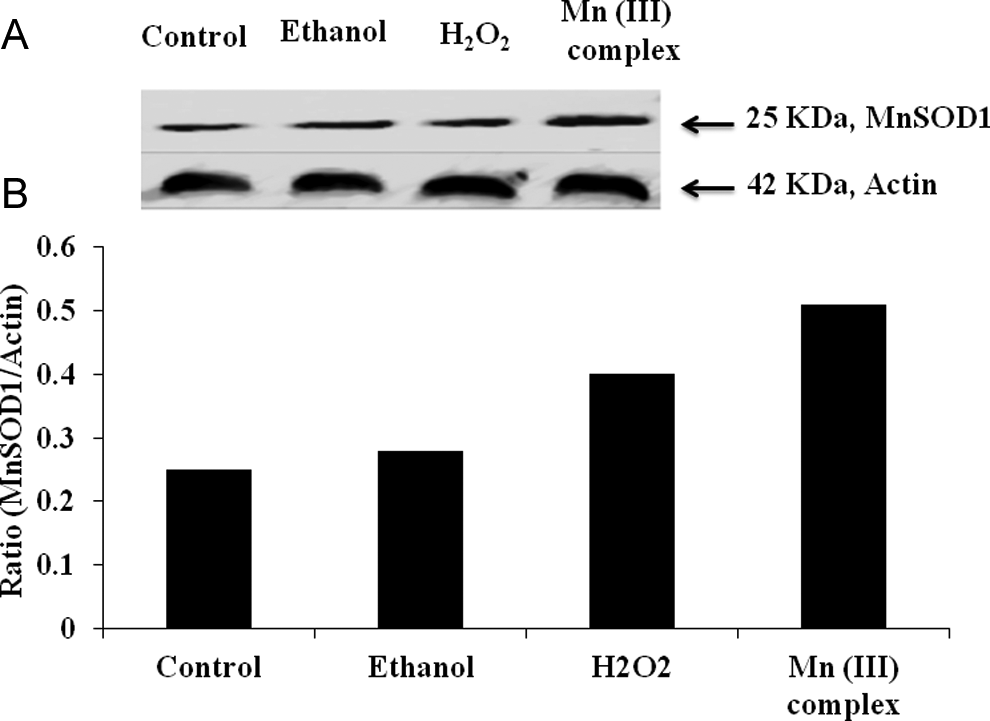
Effect of Mn(III) complex on expression of manganese super oxide dismutase1 (MnSOD-1). Human breast adenocarcinoma cell line (MCF-7) cells were cultured as described in materials and methods. Cells were treated with ethanol as control or 2.5 mmol/L of Mn (III) complex for 24 hours. Cell lysates were prepared in lysis buffer and subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) followed by Western blotting as detailed in materials and methods. Membranes were probed with specific antibodies to MnSOD-1 followed by horseradish peroxidase conjugated secondary antibody. Actin was used as an internal control to account for any variation in protein loading. The blots were visualized with chemiluminescence staining and autoradiography. A, Western blots showing expression of MnSOD-1 and actin proteins. B, Quantitative representation of the ratios of MnSOD1 and actin following densitometry analysis of the bands in (A).
Mn (III) Complex Depolarizes Mitochondria in MCF-7 Cells
Since ROS production is accompanied by changes in MMP, we examined whether the Mn (III) complex affects mitochondrial membrane function by measuring changes in its membrane potential. The MMP is known to be affected by oxidative status, which is likely to be impaired in cancerous cells. The JC-1 dye staining and analysis of the JC-1 stained cells to determine changes in the ratio of red/green fluorescence is a well-established method of determining mitochondrial membrane integrity. The ratio of red/green fluorescence was decreased in cells following the Mn (III) complex treatment (Figure 8). These results indicated that the Mn (III) complex might have depolarized the mitochondrial membrane and decreased the membrane potential in cancerous cells, thus causing cell death.
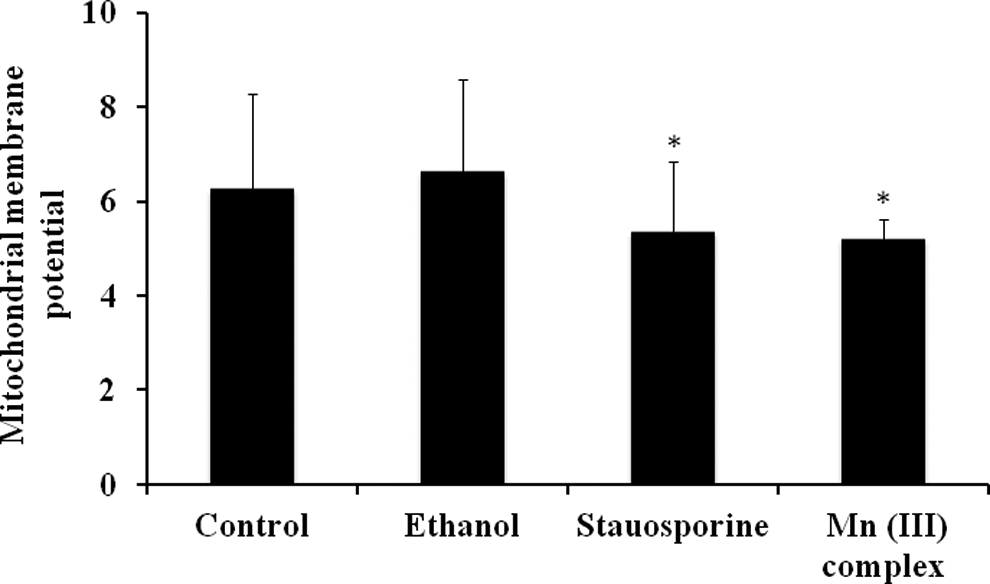
Effect of Mn (III) complex on mitochondrial membrane potential (MMP). Human breast adenocarcinoma cell line (MCF-7) cells were plated at a density of 20 × 103 cells/well in 96-well plates for 24 hours. Cells were then treated with 2.5 mmol/L of Mn (III) complex, 100 nmol/L staurosporine as positive control or ethanol as vehicle control for 24 hours. The MMP was detected using the JC-1 assay as described in materials and methods. The values are reported as fluorescence intensities measured at an excitation wavelength of 590 nm and emission wavelength of 525 nm using a fluorescence microplate reader. Data shown are mean ± standard deviation obtained from 3 independent experiments, each determined in triplicates. The P values *P ≤ 0.05 were taken as the values with significant differences as compared with their controls.
Quenching of ROS by Free Radical Scavengers Attenuates Apoptosis
In order to convincingly demonstrate that Mn (III) complex–mediated ROS production is responsible for the observed cell death in MCF-7 cells, we studied the effects of free radical scavengers on ROS production and apoptosis. In initial screaming, both ascorbic acid and DMTU known to quench free radicals diminished the production of ROS and decreased cell cytotoxicity in a concentration-dependent manner (data not shown). Effects of DMTU on ROS quenching were greater than ascorbic acid, and therefore, predetermined concentration of DMTU was used to quench ROS production to examine its effects on apoptosis using Yo-Pro-1 assay kit. As shown in Figure 9A and B, DMTU significantly decreased Mn (III) complex–induced ROS production (Figure 9B), which in turn diminished apoptosis significantly (Figure 9A). These data clearly demonstrate that Mn (III) complex–mediated production of ROS is responsible for the observed cell death in MCF-7 cells.
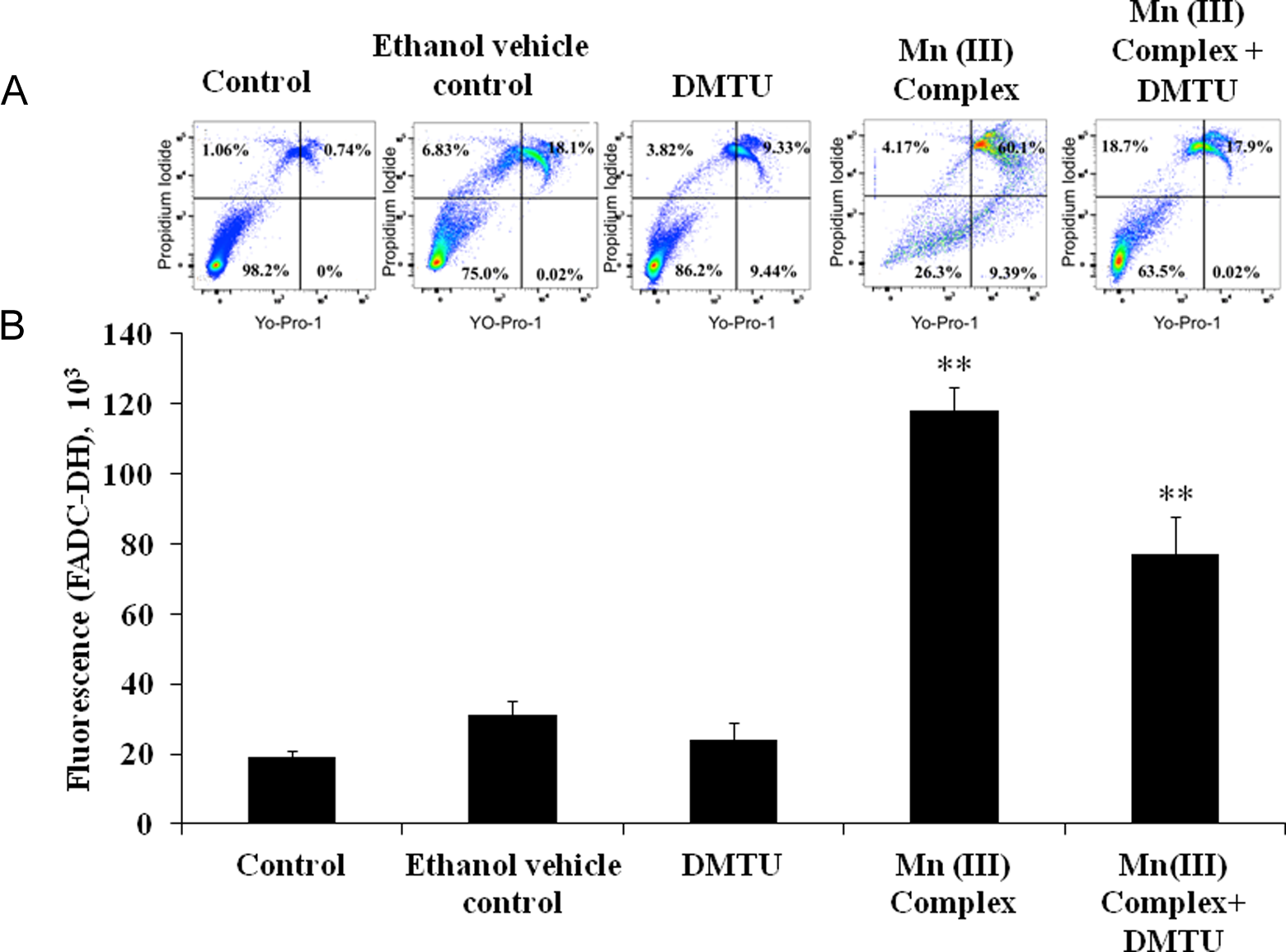
Effect of free radical scavenger DMTU on reactive oxygen species (ROS) generation and attenuation of apoptosis. A, Apoptosis was determined by flow cytometry using the vibrant apoptosis assay kit #4 as described in materials and methods. Human breast adenocarcinoma cell line (MCF-7) cells were cultured at a density 6.0 × 105 cells/mL in 6-well plates. Cells were pretreated with or without DMTU (40 mmol/L) for 30 minutes followed by exposure of Mn III complex (2.5 mmol/L) for 24 hours. Ethanol was used as a vehicle control. Shown in a quadrant are live cells (lower left), necrotic cells (upper left, upper right), and apoptotic cells (lower right). Data are representative experiments repeated at least twice with similar results. B, For ROS measurements, 20 × 103cells/well in 96-well plates were seeded for 24 hours. Reactive oxygen species was detected using 2’,7’dichlorodihydrofluoresceindiacetate (DCFH-DA) dye. Cells were pretreated with or without DMTU (40 mmol/L) for 30 minutes followed by exposure of Mn III complex (2.5 mmol/L) for 24 hours. Ethanol was used as a vehicle control. The values were reported as fluorescence intensities measured at an excitation wavelength of 485 nm and emission wavelength of 535 nm using a fluorescence microplate reader. Data shown are mean ± standard deviation obtained from 3 experiments, each determined in triplicate. The P value **P ≤ 0.01 was taken as the value with significant differences as compared with their control.
Discussion
The Mn (III) complex used in this study has been previously synthesized and characterized for its potential application as a biocatalyst by our group. 23 The aim of this study was to explore the biological effect of the Mn (III) complex using an in vitro cell culture model in order to find its potential application for cancer treatment. The proposed study was particularly directed toward understanding the molecular signaling mechanism, including any role Mn (III) complex plays in the production of ROS as ROS is linked to induce cytotoxicity in cancerous cells.
In cancer, there is a loss of balance between cell division and cell death, and cells that should have died do not receive the signals to do so. 43 Inducing apoptosis in resistant cancerous cells is a preferred way to achieve desirable outcome for a potential anticancer drug. Most anticancer drugs induce apoptosis in cancerous cells by activating intrinsic apoptotic pathways or inhibiting the cell survival mechanisms. Although anticancer agents are typically designed to directly kill tumor cells, the effect of such drugs on the host microenvironment may have an impact on their ultimate efficacy. 44
For our purpose, we used the most common cytotoxic assays to assess general cytotoxicity using a widely used model cell line for human breast cancer. We have then incorporated EB/AO and flow cytometry to ascertain whether the cytotoxic effects of the Mn (III) complex were mediated by apoptotic events. A number of well-established apoptotic markers are available. 37,38 These include, besides assessing changes in plasma membrane phospholipid disorientation, activation of caspases, release of cytochrome C from mitochondria, Serine/Threonine-specific Protein Kinase (AKT) phosphorylation, DNA degradation ladder, Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling (TUNEL) staining, and so on. Our laboratory has previously used many of these markers. However, this study was particularly selected to detect changes in membrane integrity that triggers early apoptotic events. Yo-Pro-1 detects both early and late events as well as cells undergoing necrotic death. We used Yo-Pro-1 apoptosis assay kit from Invitrogen specifically designed to assess early and late apoptotic events. Both Yo-Pro-1 and Annexin V detect changes in orientation of phospholipids in plasma membrane. 40 However, the Yo-Pro-1 appears to have more sensitivity and wide application, particularly in multiplexing systems of flow cytometry. 38 Our laboratory has also used this apoptotic assay kit in a number of studies 37 –39 to determine whether the cell death was due to apoptosis or necrosis.
Mitochondrial integrity was measured by ROS production and changes in MMP. We first demonstrated that the Mn (III) complex induces cell death in a dose- and time-dependent manner in in vitro cultures and then determined the IC50 value of the Mn (III) complex (Figures 1 and 9). Additionally, our results establish that the Mn (II) complex synthesized and characterized by our group mediates cytotoxicity using both apoptotic and necrotic cell death mechanisms.
The basis for apparent effects of the Mn (III) complex on the MCF-7 cell line may reside in its influence on mitochondrial function. To further our investigation of the mechanism of action of the Mn (III) complex in inducing cytotoxicity, we looked at generation of ROS and changes in MMP (Δψm). Treatment with the Mn (III) complex reduced the Δψmin MCF-7 cell line (Figure 8). This suggests that the Mn (III) complex might promote mitochondrial respiration in cancerous cells and, consequently, lead to the depolarization of mitochondrial membranes and the induction of cell death by the proximal pathway of mitochondria.
We linked the cytotoxic effects of the Mn (III) complex to the cellular damage caused by the production of ROS. We found that intracellular ROS production was significantly increased in the cell line treated with the Mn (III) complex (Figure 6). This could lead to enhanced glucose oxidation by the mitochondrial electron transport chain, resulting in sustained production of ROS, which, in turn, could inhibit the mitochondrial H+ efflux and ultimately decrease the Δψm. These changes might also lead to the opening of mitochondrial membrane pores by the Mn (III) complex. Mitochondrial pore openings would allow the release of cytochrome c and induction of other apoptosis-inducing factors, resulting in enhanced cell death.
Most early stage tumors occur in the microenvironment and depend heavily on anaerobic glycolysis to produce energy. 45 Dysfunction of mitochondria driven by anaerobic metabolism is also a continuous source of ROS production in tumor cells. As a result, cancer cells are likely to express more MnSOD1. The MnSOD1 converts superoxide radicals (O2 −) into H2O and molecular oxygen (O2). 46 The MnSOD1 is an important enzyme responsible for the exclusion of O2 and is considered a key antioxidant in aerobic cells, whereas a deficiency in this enzyme expression or inhibition of its activity may cause increased amounts of O2 in the cells, which would result in the persistence of the oncogenic phenotype. 47 In Figure 7A, our results showed an increase in the expression of MnSOD1.
Use of free radical scavengers is another criterion to demonstrate whether ROS produced in response to a chemical treatment is in fact responsible for the observed cellular damage.
48,49
A wide variety of free radical scavengers including ascorbic acid, N-acetyl
Although understanding the exact mechanism of action of Mn (III) complex in mediating apoptosis in MCF-7 cells requires further in-depth studies incorporating more signaling molecules of apoptotic signaling pathways, our proposed mechanism of action (Figure 10) is based on our current knowledge about this compound and the data obtained from this article. We propose that Mn(III) complex triggers generation of ROS, primarily in mitochondria, disrupting its function and causing cell death by way of partly inducing apoptosis and partly necrosis.
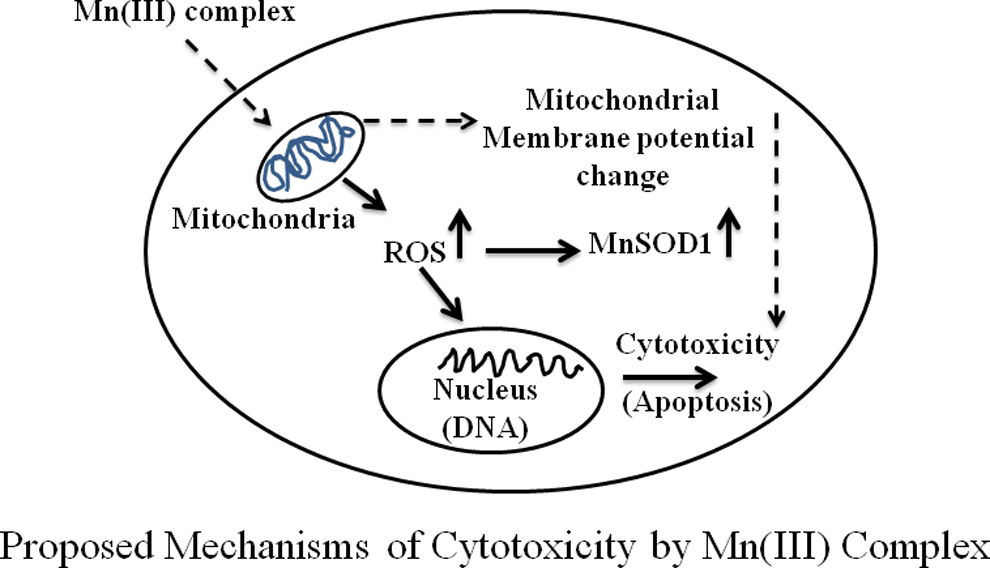
Proposed mechanism of cytotoxicity of Mn (III) complex in human breast adenocarcinoma cell line (MCF-7). Possible mechanism of action of Mn (III) complex-induced cytotoxicity or apoptotic cell death is via the production of reactive oxygen species (ROS) which damages mitochondrial function and triggers apoptosis.
Conclusion
In conclusion, we have investigated the mechanism of action of biological effects of the previously synthesized and characterized Mn (III) complex for its potential utility as an anticancer drug. The data suggest that the Mn (III) complex causes cytotoxicity in human breast cancer cells. Further studies are required to determine whether the complex is useful as anticancer agent. It will also be desirable to study the effects of this compound in normal breast cells.
Footnotes
Acknowledgments
N. Ali and A. Ghosh acknowledge partial support from the Center for Advanced Surface Engineering, under the National Science Foundation Grant No. IIA-1457888 and the Arkansas EPSCoR Program, ASSET III. N. Ali also thanks NASA-EPSCoR for the award of Research Infrastructure Development (RID) grant. A. Ghosh also acknowledges National Science Foundation MRI grant (Award Number CHE 1229149).
Author Contributions
Al-anbaky, Q. contributed to design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Al-karakooly, Z. contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Kilaparty, S. contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Agrawal, M. contributed to conception, contributed to analysis and interpretation, and critically revised manuscript; Albkuri, Y. contributed to acquisition and analysis and critically revised manuscript; Rangu Magar, A. contributed to acquisition and analysis and critically revised manuscript; Ghosh, A. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Ali, N. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
