Abstract
Introduction
Oxidative stress resulting from the increased formation of reactive oxygen species or the perturbation of antioxidant defenses has been implicated in a number of disease states including cancer, arthritis and inflammatory processes, and cardiovascular, neurologic, and immune deficit, among others.
1
-4
First identified in the sclerotia of the ergot fungus,
Beneficial physiologic properties of
Although numerous reports detail the bioavailability of
Early in vivo studies in rats at 70 to 100 mg/kg confirm
To date, an acceptable daily intake or tolerable daily intake for
In order to qualify safety of this purported antioxidant for its suitability as a dietary supplement, nature-identical ergothioneine, EGT+, manufactured from a proprietary process and derived as
Materials and Methods
Test Substance
The test substance, a synthetic
Dose levels for the bacterial reverse mutation (Ames) test and oral toxicity studies were chosen based on recommended guidelines and an expected human dose of 5 to 10 mg/d, respectively, derived from studies that quantified the average amount of
For the dose-analysis phase of the 90-day study, neat EGT+ samples were collected on days 1, 43, and 92 and dose mixture samples were collected on day 1 for homogeneity and on days 43, 71, and 92 for concentration verification.
Toxicity Studies—Methods
Mutagenicity and 90-day subchronic toxicity were conducted in conformance with Office of Economic Cooperative Development (OECD) Principles of Good Laboratory Practices 49 and US Food and Drug Administration Good Laboratory Practices. 50,51 The genotoxicity and repeated-dose subchronic studies (28- and 90-day studies) were conducted by Eurofins/Product Safety Labs (Dayton, New Jersey) under OECD Guidelines for the Testing of Chemicals, Section 4, No. 471, “Bacterial Reverse Mutation Test”, adopted July 21, 1997 52 ; Commission Regulation (EC) No. 440/2008 B. 13/14 B: “Mutagenicity-Reverse Mutation test using Bacteria, dated May 3, 2008 53 ; and EPA Health Effects Test Guidelines, OPPTS 870.5100, “Bacterial Reverse Mutation Assay” EPA 712-C-98-247, August 1998. 54 The in vivo toxicity study undertaken by the testing laboratory was in accordance with the most recent Guide for the Care and Use of Laboratory Animals, 55 according to the Association for Assessment and Accreditation of Laboratory Animal Care standards and accreditation and the OECD Guidelines for the Testing of Chemicals, Section 4, No. 407, “Repeated Dose 28-day Oral Toxicity Study in Rodents,” adopted October 16, 2008; and the OECD Guidelines for the Testing of Chemicals, Section 4, No. 408 “Repeated Dose 90-day Oral Toxicity Study in Rodents,” adopted September 21, 1998.
Mutagenicity Studies
Nature-identical ergothioneine EGT+ was tested for potential cytotoxicity and mutagenicity in a bacterial reverse mutation assay according to the plate incorporation test (experiment 1) and the preincubation test (experiment 2) at concentrations of 0, 1.58, 5.0, 15.8, 50, 158, 500, 1,580, and 5,000 μg/plate solubilized in distilled water of
Twenty-Eight–Day Oral Toxicity Study
In a preliminary 28-day range-finding study for potential toxicity indicated by any changes in clinical signs, body weight, body weight gain, food consumption, food efficiency, or macroscopic findings at necropsy, groups of 5 CD IGS Sprague Dawley (Crl: SD, Charles River Laboratories, Inc, Raleigh, North Carolina) rats/sex/dose were orally intubated daily with the nature-identical ergothioneine test substance dissolved in water at dose levels of 0 (distilled water vehicle control), 50, 200, or 500 mg/kg bw/d at a dose volume of 5 mL/kg bw/d adjusted weekly for body weight. Prior to treatment, rats were acclimated for 6 days. Rats were approximately 8 weeks of age and males weighed 241 ± 6.05 g and females weighed 189 ± 7.18 g at the start of treatment. Rats were individually housed in suspended stainless steel cages with mesh floors. Litter paper was placed beneath the cages and was changed at least 3 times per week. The room temperature ranged from 19°C to 22°C with a relative humidity of 48% to 78% and a 12-hour light/dark cycle. Humidity was above the targeted upper limit for 1 day during the study. A portable dehumidifier was used to lower the humidity levels during this time. Filtered tap water was available ad libitum from an automatic watering supply system. The dose preparations were prepared daily based on the most recent daily body weights. Neat test substance and dosing preparations were sampled and preserved. At terminal sacrifice, all animals were euthanized by carbon dioxide asphyxiation. All animals in the study were subjected to a gross necropsy, which included examination of the external surface of the body, all orifices, musculoskeletal system, and the cranial, thoracic, abdominal, and pelvic cavities with their associated organs and tissues. Animals were not fasted prior to necropsy. Tissues were not preserved or evaluated further unless indicated for possible toxicological interest.
Ninety-Day Oral Toxicity Study
Nature-identical ergothioneine EGT+ was tested in a 90-day oral toxicity study for potential toxicity in CD IGS Sprague Dawley (Crl: SD) rats following regulatory guidelines. Based on the negative results of the 28-day study and the expected human intake, dose levels of 0 (control), 400, 800, and 1,600 mg/kg bw/d (increased from the 28-day study and 2,400-9,600 times the expected exposure for a 60-kg human) were chosen and orally intubated to 10 rats/sex/group at a uniform volume of 10 mL/kg bw, adjusted weekly for body weight, for at least 90 days daily. The control group received distilled water vehicle only at the same dose volume as the test animals. Prior to treatment, rats were acclimated for 5 days. At the start of treatment (day 0), rats were approximately 7 to 8 weeks of age and males weighed 191 to 243 g and females weighed 139 to 160 g. Rats were individually housed in suspended stainless steel cages with mesh floors. Litter paper was placed beneath the cages and was changed at least 3 times per week. The room temperature ranged between 18°C and 23°C with a relative humidity of 38% to 60% and a 12-hour light/dark cycle. Feed 2016CM Harlan Teklad Global Rodent Diet (Harlan Teklad, Inc, Indianapolis, Indiana) and filtered tap water from an automatic watering system were provided ad libitum except when animals were fasted overnight prior to blood sampling. Test animals housed in the same room for a sentinel health monitoring program evaluated for the absence of viruses near the end of the in-life portion of the study showed no viral contaminants. The nature-identical ergothioneine at the appropriate concentrations was prepared and thoroughly stirred just prior to use daily. Samples of the test substance both neat and in the dosing solution were collected at the beginning, middle, and end of the in-life phase of the study and analyzed by validated HPLC methodology against an ergothioneine standard to evaluate stability and to verify dose concentration and homogeneity. Ophthalmologic evaluations were conducted prior to the commencement of the study and on day 89 by focal illumination and indirect ophthalmoscopy after pharmacologic mydriasis with 1% tropicamide solution. During week 12, blood samples were collected 2 hours postdosing via sublingual bleeding of fasted rats while under isoflurane anesthesia for hematology (erythrocyte count, hemoglobin concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, red cell distribution width, absolute reticulocyte count, platelet count, total white blood cell and differential leukocyte count such as lymphocyte, neutrophil, eosinophil, monocyte, and basophil, and large unstained cell), clinical chemistry (serum aspartate aminotransferase, serum alanine aminotransferase, sorbitol dehydrogenase, alkaline phosphatase, total bilirubin, urea nitrogen, blood creatinine, total cholesterol, triglycerides, fasting glucose, total serum protein, albumin, globulin, calcium, inorganic phosphorus, sodium, potassium, chloride), and plasma analysis for
Statistical Analysis
Bacterial reverse mutation assay
The mutation factor (MF) is calculated by dividing the mean value of the revertant counts by the mean values of the solvent control (the exact and not the rounded values).
A statistical evaluation is not regarded as necessary. Toxic effects of the nature-identical ergothioneine are indicated by the partial or complete absence of a background lawn of nonrevertant bacteria (colony counts, if any, should not be reported), a substantial dose-related reduction in revertant colony counts compared with lower dose levels and concurrent vehicle control taking into account the laboratory historical control range or biologically relevant increases in revertant colony numbers. The results were considered positive if the results for the test substance showed a substantial increase in revertant colony counts, that is, response MF ≥ 2 for strains TA98, TA100, and WP2 uvrA or MF ≥ 3 for strains TA1535 and TA1537, with mean value(s) outside the laboratory historical control range. Otherwise, results were considered negative; and the above increase is dose related and/or reproducible, that is, increases were obtained at more than one experimental point (at least one strain, more than one dose level, more than one occasion, or with different methodologies). When the results indicated neither a concentration-related increase in the number of revertant colonies nor a reproducible substantial increase in revertant colonies, the test substance was considered to be nonmutagenic in this test system.
Repeated dose oral toxicology
Group means and standard deviations were calculated for body weight, daily body weight gain, daily feed consumption, daily feed efficiency, organ weight, and organ-to-body/brain weight ratio. Data within groups were evaluated for homogeneity of variances and normality by Bartlett test. Where Bartlett test indicated homogeneous variances, treated and control groups were compared using analysis of variance, followed by comparison of the treated groups to control by Dunnett
Results
Mutagenicity Reverse Mutation Assay
An Ames test using
Results of a Plate Incorporation Test (Experiment 1) of EGT+ on
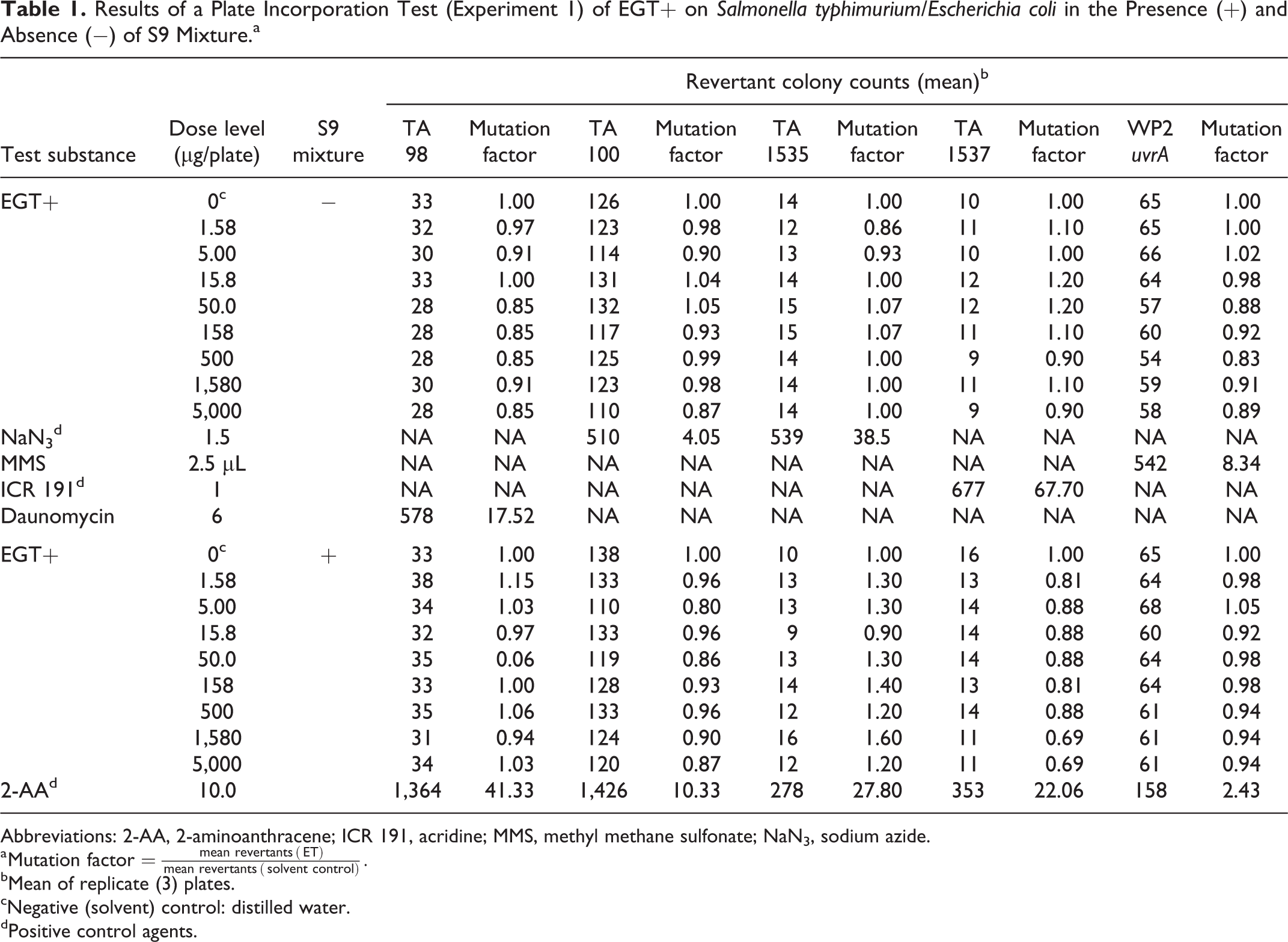
Abbreviations: 2-AA, 2-aminoanthracene; ICR 191, acridine; MMS, methyl methane sulfonate; NaN3, sodium azide.
a
bMean of replicate (3) plates.
cNegative (solvent) control: distilled water.
dPositive control agents.
Results of a Preincubation Test (Experiment 2) of EGT+ on
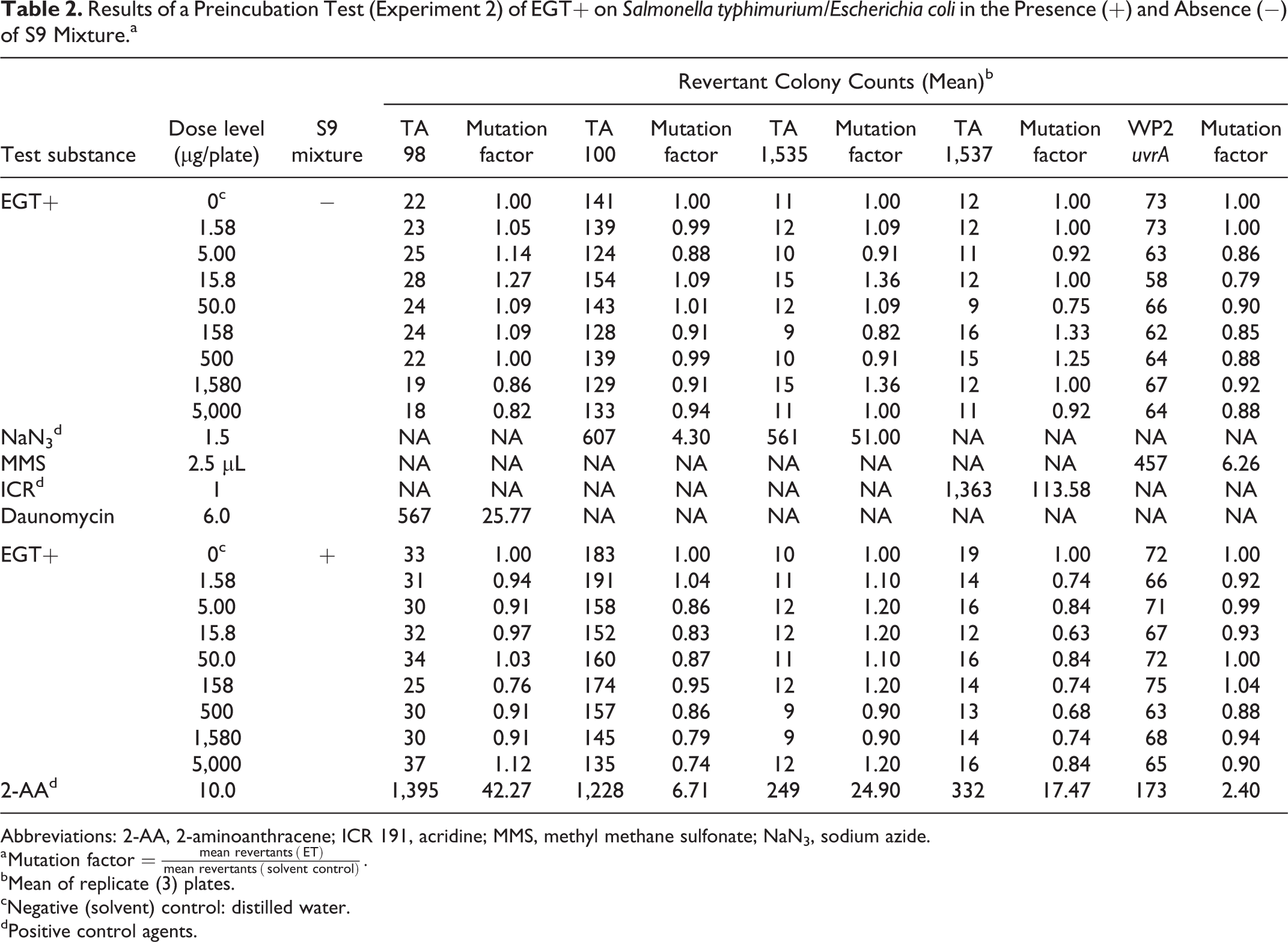
Abbreviations: 2-AA, 2-aminoanthracene; ICR 191, acridine; MMS, methyl methane sulfonate; NaN3, sodium azide.
a
bMean of replicate (3) plates.
cNegative (solvent) control: distilled water.
dPositive control agents.
Twenty-Eight–Day Oral Toxicity Study
Nature-identical ergothioneine was considered stable under the conditions of storage over the course of this study. Similarly, the test substance was considered to be both homogeneously distributed in the preparations and at the targeted concentration throughout the study. There was no mortality in any of the test groups and no treatment-related abnormal clinical findings. There were no daily or detailed clinical observations noted for male or female rats in all dose groups throughout the study attributable to the test administration. Clinical changes of alopecia found intermittently in males and females of all groups, and ocular and nasal discharge in 1 male in the high-dose group was attributed to a malocclusion of the upper incisors at the end of the study. There were no body weight, body weight gain, food consumption, food efficiency, or macroscopic observations at necropsy attributable to test substance administration. Significant, increases in overall mean weekly body weight (10%,
90-Day Oral Toxicity Study
Nature-identical ergothioneine, as measured by ergothioneine recovery by HPLC, was found to be stable, homogeneously mixed, and met the target concentrations in the dosing solution for all dose levels under the storage conditions over the course of the study. The percentage change in stability over the course of the 13-week study was +1.9 (101.9% of initial) for a measured active ingredient percentage of 98.8% overall for the course of the study. The test substance as a prepared solution was reported to be stable and homogenously dispersed (94.4%-101.4%; relative standard deviation 0.6%-5.7%) at measured targeted concentrations (94.4%-104.2%) under the conditions of the study. Ergothioneine, analytically determined in rat plasma from samples collected at terminal sacrifice, resulted in significant dose-proportional increases from control for all test group males and females. The significant increases in mean results of ergothioneine found in terminal rat plasma from treated low-, mid-, and high-dose males of 6,090, 8,869, and 18,256 ng/mL and in females of 7,997, 12,973, and 19,676 ng/mL, respectively, were significantly higher than endogenous levels of
Mean Plasma Analysis of
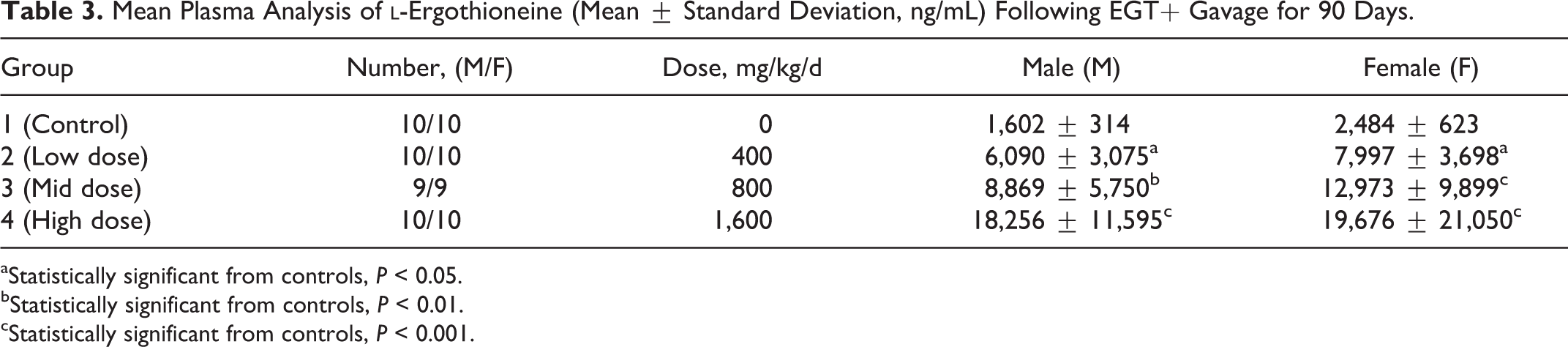
aStatistically significant from controls,
bStatistically significant from controls,
cStatistically significant from controls,
Mortality and clinical observations
There were no mortalities related to the administration of nature-identical ergothioneine. One male in the mid-dose group was found dead on day 66. Microscopic laryngeal lesions associated with inflammatory infiltrate and multifocal, moderate muscle degeneration were consistent with death likely related to gavage administration procedure and unassociated with a test substance effect. One female in the same test group was found dead on day 85 with multifocal red discoloration of the distal aspect of the tail corresponding to locally extensive, moderate epidermal necrosis. There were no other clinical observations. Although the cause of mortality could not be determined for this animal, the lack of a dose-response effect in incidence and the absence of significant macroscopic or microscopic findings suggest that the death of this animal was not related to the nature-identical ergothioneine administration. Clinical observations of surviving animals attributed to oral administration of the nature-identical ergothioneine were restricted to males and consisted of soft feces in 6 of 10 high-dose animals on study days 85 to 95, as well as diarrhea (day 95) observed in 2 other high-dose males. These findings were considered nonadverse. Incidental clinical findings in surviving males included a lesion on the nose/snout (1 of 10 low dose); red ocular discharge in the right eye (1 of 10 high dose); brown staining on the face (1 of 10 mid dose and 1/10 high dose); slight to moderate alopecia on the right/left forelimb/forepaw, abdomen, or anogenital area (1 of 10 low dose and 1 of 10 high dose); enophthalmos (1 of 10 low dose); eschar on the face (1 of 10 mid dose); white oral discharge (1 of 10 mid dose and 1 of 10 high dose); moist rales (1/10 mid dose); a malocclusion of the upper incisors (1 of 10 high dose); a broken tooth of the upper incisors (1 of 10 mid dose); and a broken toenail (1 of 10 mid dose). There were no clinical signs in control males. Clinical signs in females included slight to moderate alopecia on left/right forelimb/hind limb, flank, chest, back, abdomen, head, or anogenital area (3 of 10 control, 4 of 10 low dose, 1 of 10 mid dose, and 4 of 10 high dose); eschar on the back, head, and neck (1 of 10 mid dose); and a broken tooth of the upper left incisor (1 of 10 mid dose). Ophthalmoscopical examinations showed eyes to be normal.
Body weight, body weight gain, food consumption, and food efficiency
There were no significant changes in body weight, body weight gain, food consumption, and food efficiency changes in treated male and female rats attributable to the administration of the nature-identical ergothioneine. Changes in male or female body weight gain were transient and without correlated changes in body weight, while decreases observed in male food consumption were transient and without concurrent or dose-dependent body weight, body weight gain, or food efficiency changes. Overall (days 0-92) and weekly mean body weight and mean daily body weight gain (Figure 1) of all treated rats were comparable with controls with the following exceptions: males showed a significant decrease in daily body weight gain during days 8 to 15 (high-dose group); females showed a significant decrease in body weight on days 64 and 71 (high-dose group); and females showed a significant decrease in daily body weight gain during days 36 to 43 (high-dose group). Overall and weekly feed consumption and mean daily feed efficiency of all treated rats were generally comparable to controls with the following exceptions: males showed decreases in food consumption during days 1 to 15 and 22 to 29 (high-dose group); females showed a significant decrease in feed consumption during days 1 to 15 and 64 to 71 (high-dose group) and overall (days 0-92); and females showed a significant decrease in feed efficiency on days 36 to 43.
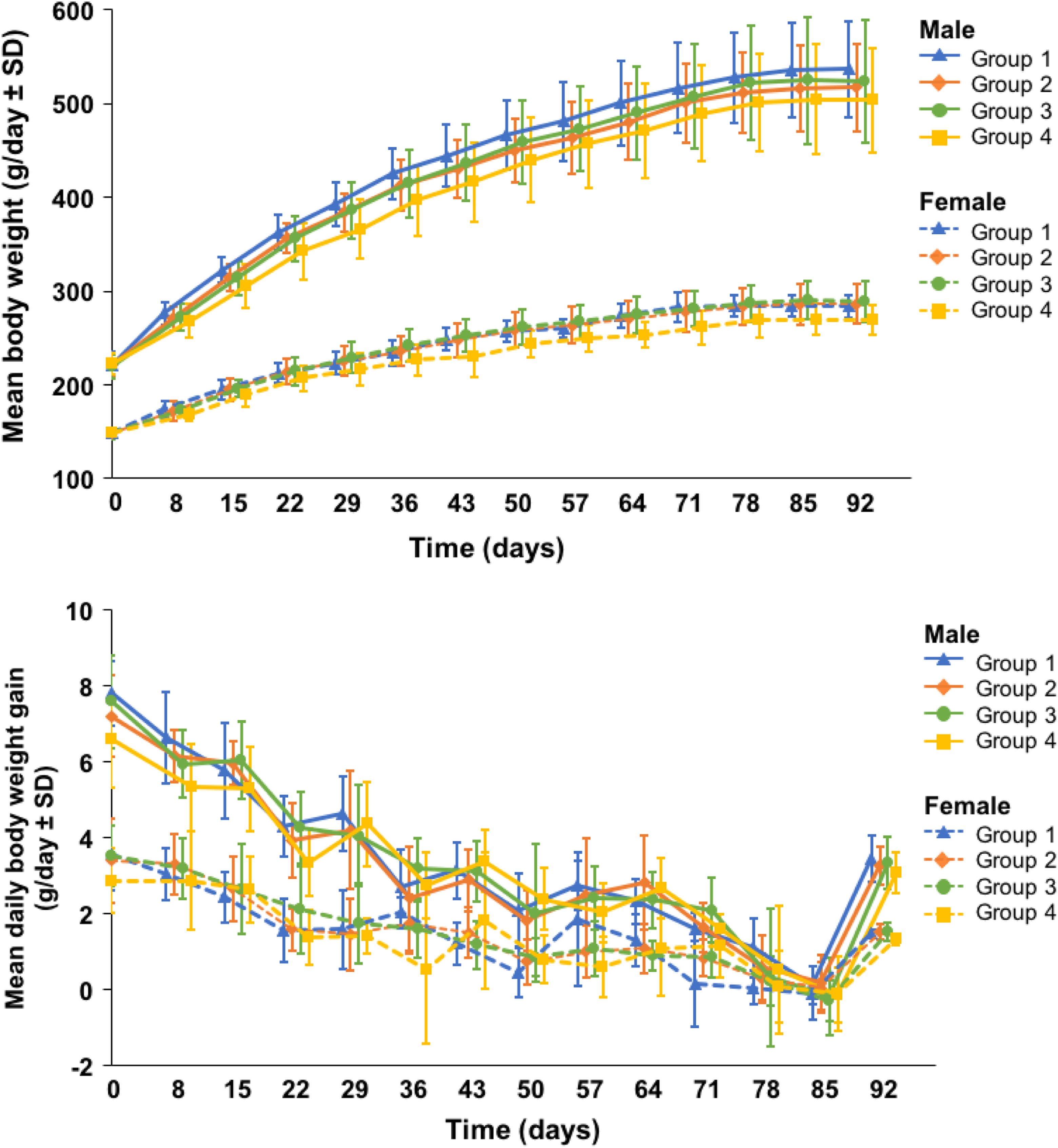
Mean body weight and body weight gain (day ± standard deviation [SD]) of male and female Sprague Dawley rats administered EGT+ gavage for 90 days for groups 1 (control), 2 (400 mg/kg/d), 3 (800 mg/kg/d), and 4 (1,200 mg/kg/d); mean of 9 of 10 animals/sex/group.
Clinical pathology
Hematology, coagulation, and clinical chemistry parameters revealed no adverse changes and no morphologic changes. Statistically significant hematology changes (Table 4) reported included decreased monocyte concentration (mid-dose males), decreased hemoglobin concentration (low-dose females), decreased hematocrit (low- and mid-dose females), decreased mean corpuscular volume (low-, mid-, and high-dose females), increased red cell distribution width and reticulocyte concentration (high-dose females), and decreased absolute eosinophil count (high-dose females). Prothrombin time was minimally decreased in mid-dose males and females.
Mean Hematology Values, Day 86 (Mean ± Standard Deviation) of Sprague Dawley Rats Following EGT+ Gavage for 90 Days.a
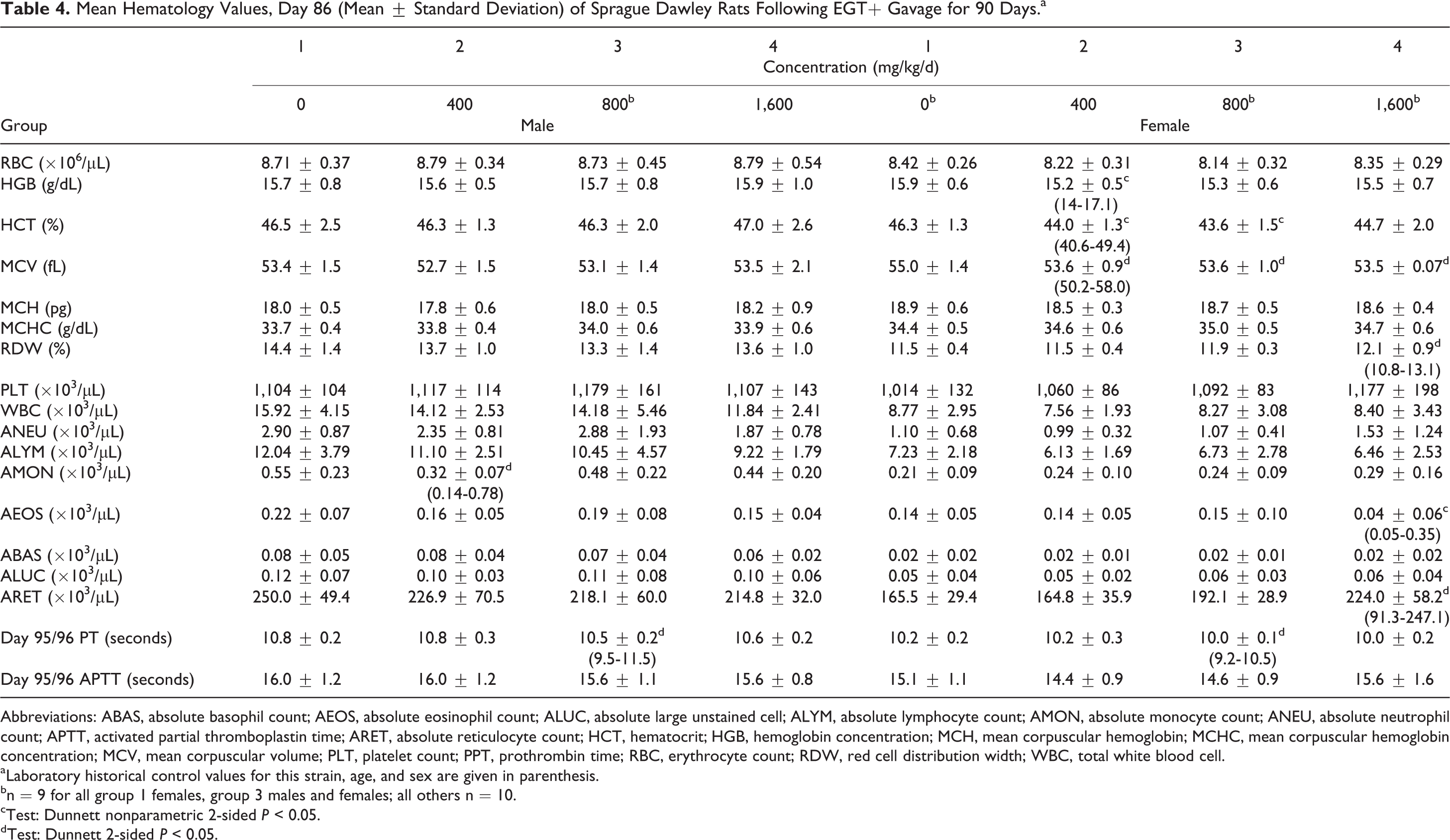
Abbreviations: ABAS, absolute basophil count; AEOS, absolute eosinophil count; ALUC, absolute large unstained cell; ALYM, absolute lymphocyte count; AMON, absolute monocyte count; ANEU, absolute neutrophil count; APTT, activated partial thromboplastin time; ARET, absolute reticulocyte count; HCT, hematocrit; HGB, hemoglobin concentration; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; PLT, platelet count; PPT, prothrombin time; RBC, erythrocyte count; RDW, red cell distribution width; WBC, total white blood cell.
aLaboratory historical control values for this strain, age, and sex are given in parenthesis.
bn = 9 for all group 1 females, group 3 males and females; all others n = 10.
cTest: Dunnett nonparametric 2-sided
dTest: Dunnett 2-sided
Clinical chemistry changes (Table 5) included increased alkaline phosphatase concentration (high-dose males and females), decreased glucose concentration (high-dose males), decreased sodium concentration (mid-dose males), decreased triglyceride concentration (high-dose males), decreased chloride concentration (mid- and high-dose males), decreased creatinine, protein, and albumin concentration (high-dose females), and increased triglyceride concentration (high-dose females). These changes were considered nonadverse and not related to exposure because the magnitude of the changes was considered not clinically significant, fell within historical control range of the laboratory, and were not accompanied by any other corresponding clinical or pathological change. There were no test substance-related changes in blood cell morphology. The only statistically significant changes reported in urinalysis was increased urine volume in mid-dose males, decreased urinary protein in low- and mid-dose males, and decreased urinary pH in high-dose females. These changes, without supporting clinical, macroscopic, or microscopic changes, were not adverse and, as such, considered unrelated to the administration of the nature-identical ergothioneine.
Mean Clinical Chemistry Values, Day 86 (Mean ± Standard Deviation) of Sprague Dawley Rats Following EGT+ Gavage for 90 Days.a
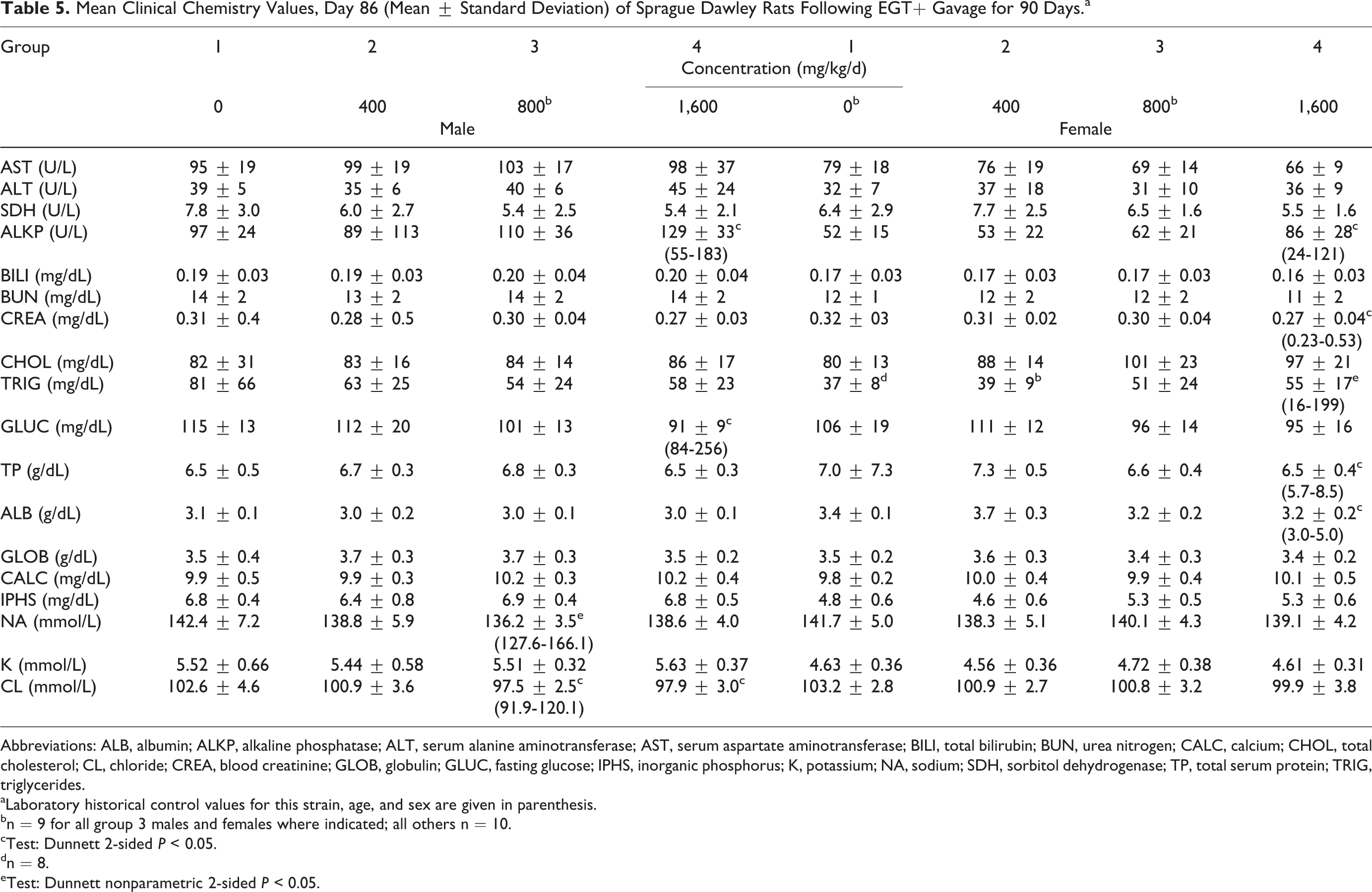
Abbreviations: ALB, albumin; ALKP, alkaline phosphatase; ALT, serum alanine aminotransferase; AST, serum aspartate aminotransferase; BILI, total bilirubin; BUN, urea nitrogen; CALC, calcium; CHOL, total cholesterol; CL, chloride; CREA, blood creatinine; GLOB, globulin; GLUC, fasting glucose; IPHS, inorganic phosphorus; K, potassium; NA, sodium; SDH, sorbitol dehydrogenase; TP, total serum protein; TRIG, triglycerides.
aLaboratory historical control values for this strain, age, and sex are given in parenthesis.
bn = 9 for all group 3 males and females where indicated; all others n = 10.
cTest: Dunnett 2-sided
dn = 8.
eTest: Dunnett nonparametric 2-sided
Macroscopic observations, histopathology, and organ weights
Macroscopic examination of surviving animals revealed no gross abnormalities related to treatment with nature-identical ergothioneine. Incidental findings, confirmed on microscopic analysis, of mild dermal fibrosis in 1 low-dose male, moderate polycystic kidney in 1 mid-dose male, marked auricular chondritis in 1 high-dose female associated with bilaterally enlarged ears at sacrifice, and fluid-filled uterus attributable to variation in the estrus cycle in individual animals of both control and treated rats.
There were no absolute or relative organ weight findings in male rats attributable to the administration of the nature-identical ergothioneine. Decreases in absolute and relative thymus weights along with increases in liver and kidney relative to body weight changes observed in high-dose female rats unassociated with hematological and histological changes were interpreted to be nonadverse and incidental (Tables 6-8). There were no microscopic findings related to the exposure to the nature-identical ergothioneine (Table 9). All microscopic findings were incidental, associated with those observed normally in this age and strain of rat.
Mean Terminal Body and Organ Weights (Mean ± Standard Deviation, g) Days 95 to 96 of Sprague Dawley Rats Following EGT+ Gavage for 90 Days.
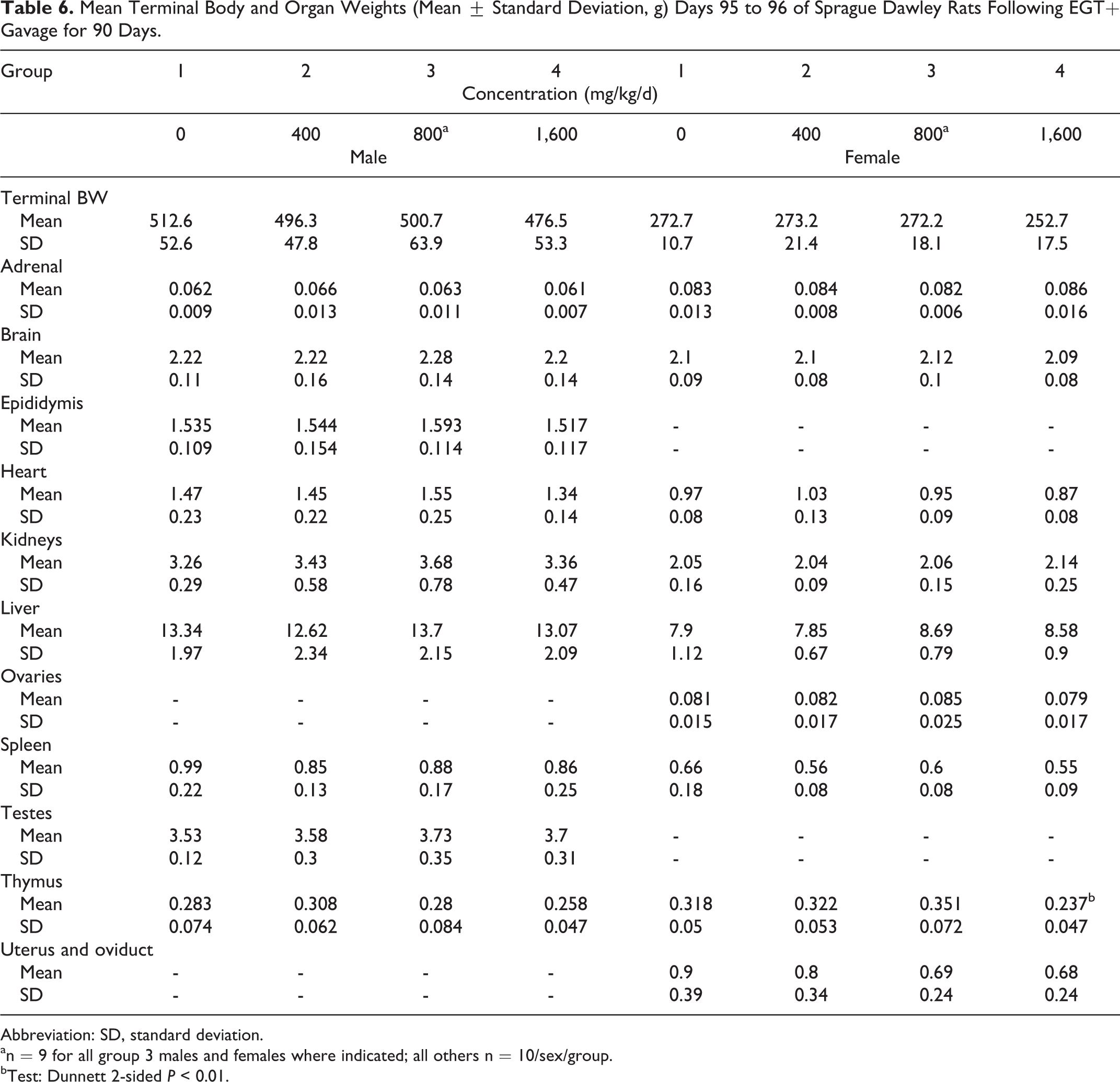
Abbreviation: SD, standard deviation.
an = 9 for all group 3 males and females where indicated; all others n = 10/sex/group.
bTest: Dunnett 2-sided
Mean Organ-to-Body Weights (Mean ± Standard Deviation, g) Days 95 to 96 of Sprague Dawley Rats Following EGT+ Gavage for 90 Days.
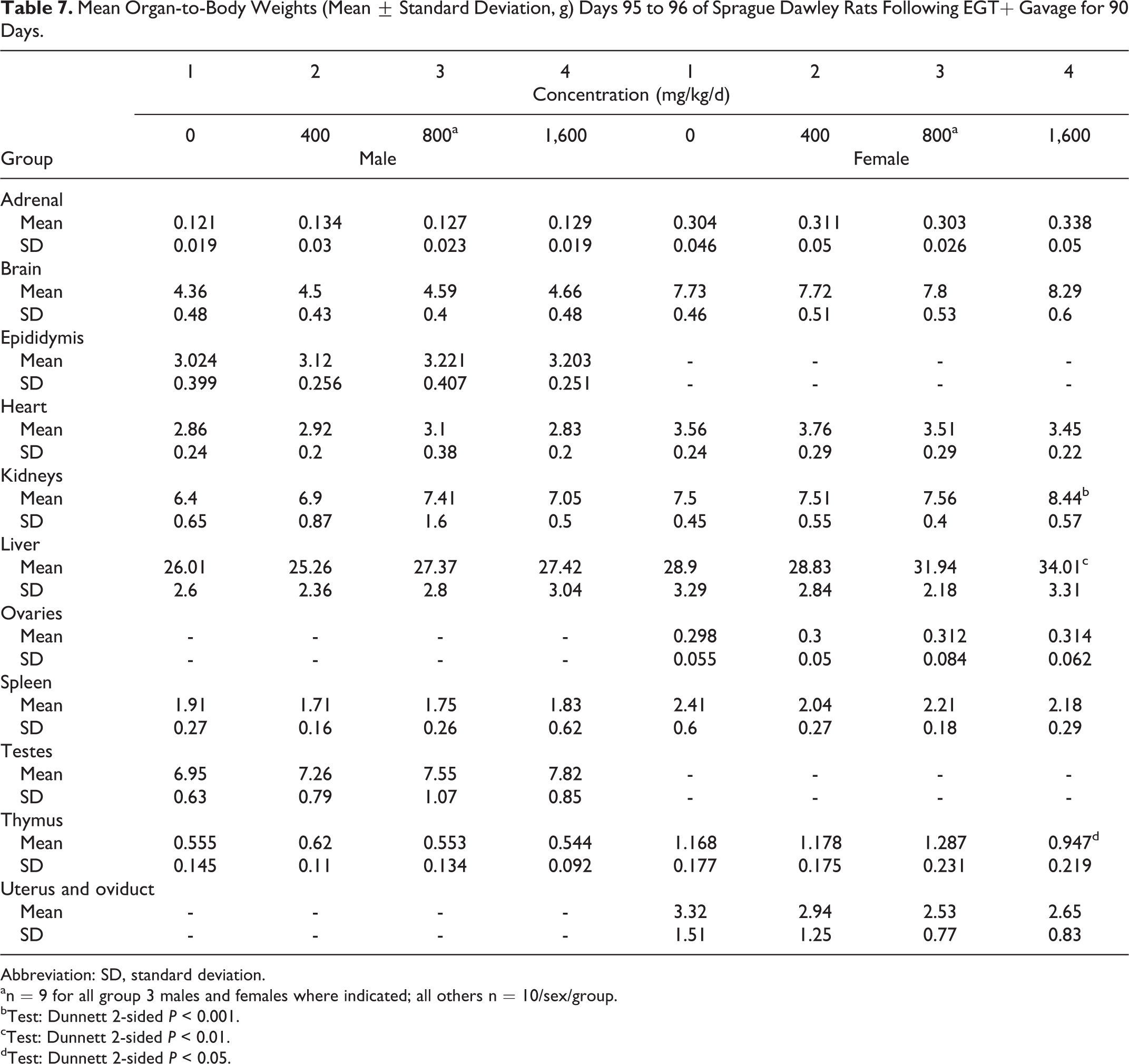
Abbreviation: SD, standard deviation.
an = 9 for all group 3 males and females where indicated; all others n = 10/sex/group.
bTest: Dunnett 2-sided
cTest: Dunnett 2-sided
dTest: Dunnett 2-sided
Mean Organ-to-Brain Weights (Mean ± Standard Deviation, g) Days 95 to 96 of Sprague Dawley Rats Following EGT+ Gavage for 90 Days.
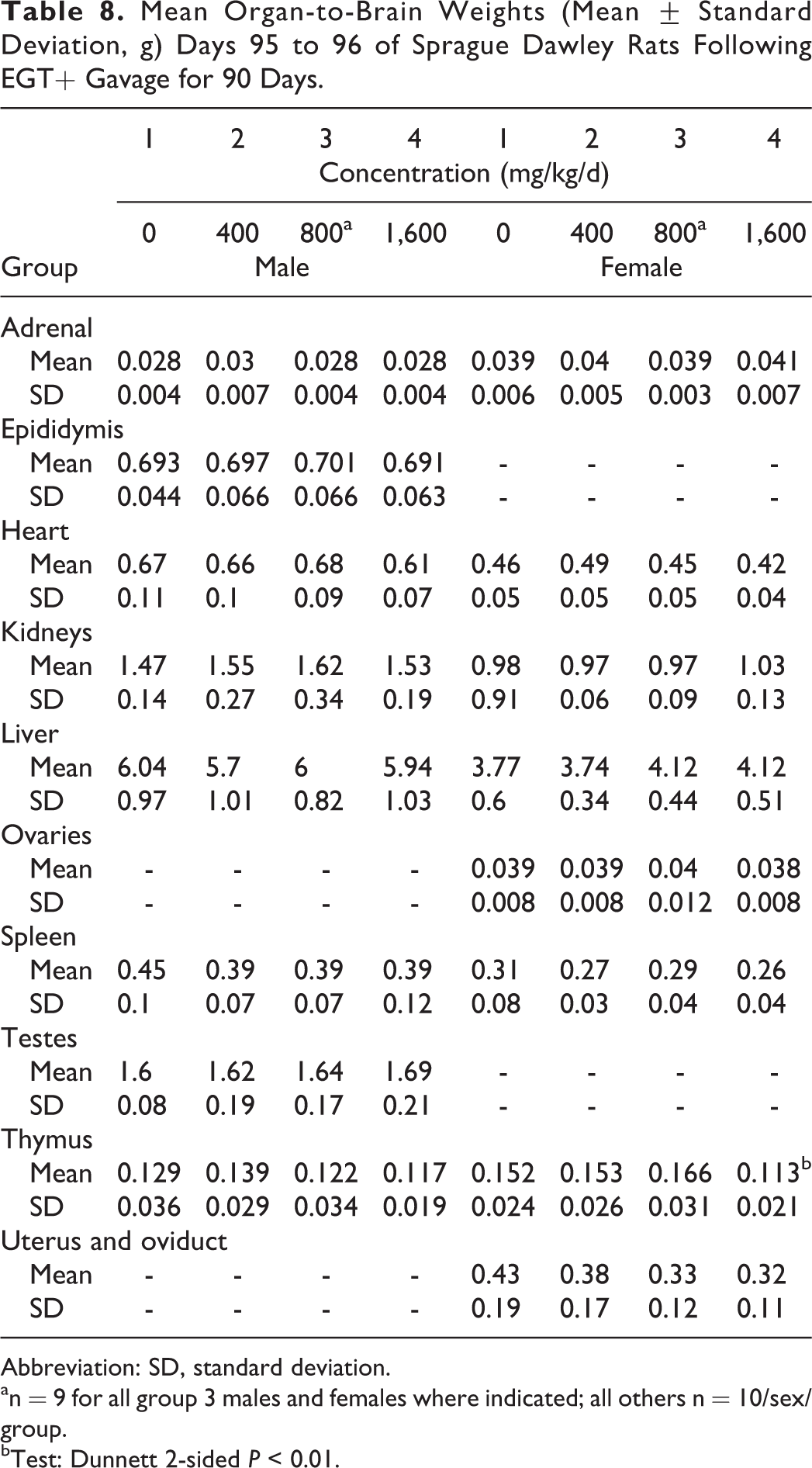
Abbreviation: SD, standard deviation.
an = 9 for all group 3 males and females where indicated; all others n = 10/sex/group.
bTest: Dunnett 2-sided
Incidence and Severity of Microscopic Findings for EGT+ in Sprague Dawley Rats Following EGT+ Gavage for 90 Days.
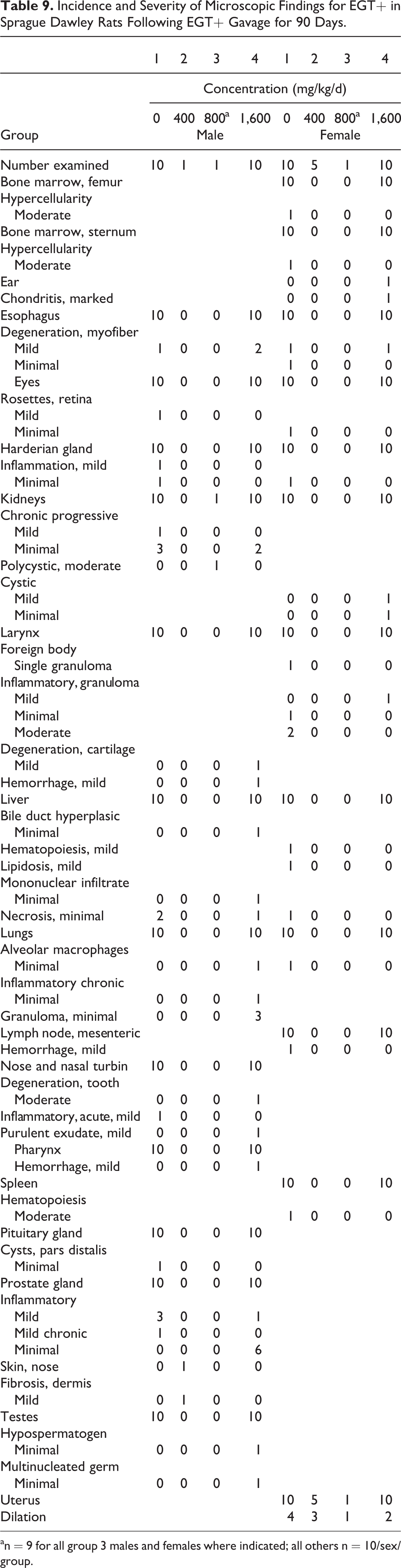
an = 9 for all group 3 males and females where indicated; all others n = 10/sex/group.
The highest concentration tested of 1,600 mg/kg bw/d produced no treatment-related adverse effects and was regarded as the no observed adverse effect level (NOAEL). Therefore, under the conditions of this study, the clinical and anatomic pathology, the NOAEL for nature-identical ergothioneine, EGT+ was 1,600 mg/kg bw/d following daily oral gavage to male and female Sprague Dawley rats for at least 90 days.
Discussion
The present study examined
The short-term, range-finding oral toxicity study further supported that the test substance, EGT+, the highly pure (>99%), biomimetic compound was well tolerated by male and female rats at doses of 500 mg/kg bw/d. There were no test substance-related mortalities during the course of the study. There were no clinical signs, body weight, body weight gain, food consumption, food efficiency, or macroscopic observations at necropsy attributable to the nature-identical ergothioneine administration. Significant increases in mean weekly body weight and mean daily body weight gain in low-dose males over the course of the 28-day study were consistent and associated with increased food consumption and without dose dependency.
Based on the 28-day range-finding study, dose selection of 400, 800, and 1,600 mg/kg bw/d for the 90-day study, equivalent to a substantial safety margin over the estimated human exposure, was well tolerated by the animals. In the longest and highest dosage study of a synthetic ergothioneine to be reported to date, the test substance was determined to be stable, homogeneously distributed, and at targeted dose levels in the distilled water vehicle under the conditions of the study. Plasma analysis at the end of the study confirmed the dose proportionality of the received test substance and its systemic exposure. These results differ with those of Heath et al who reported steady
Oral administration of up to 1,600 mg/kg bw/d nature-identical ergothioneine to CD IGS Sprague Dawley rats for 90 days did not produce any adverse effects under the conditions of the present study. Since the NOAEL represents a large multiple of expected human exposure and a highly suitable safety margin from the intended use of 5 to 10 mg/d for an adult, it is concluded that from a critical evaluation of the available information on EGT+, a nature-identical ergothioneine, the results are supportive of appropriate levels for the safety of the intended use of this product as a dietary supplement.
Footnotes
Author Contributions
Palma Ann Marone contributed to design, contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. Jan Trampota contributed to conception and design, contributed to acquisition and analysis, and critically revised the manuscript. Steven Weisman contributed to conception and design, contributed to acquisition and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Drs Marone and Weisman received payment from Mironova Labs Inc. for preparation of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All research reported was funded by Mironova Labs Inc., Fairfield, New Jersey.
