Abstract
Caffeine and 1,3-dimethylamylamine (DMAA) are widely used alone and in combination with dietary supplements. No investigation has determined the safety profile of chronic intake of caffeine or DMAA, alone or in combination, within the same study design. A total of 50 young and healthy men completed 12 weeks of daily supplementation with either a placebo (
Introduction
Both caffeine and 1,3-dimethylamylamine (DMAA) are widely used by individuals either as stand alone ingredients or within finished dietary supplements. Like caffeine, DMAA is a central nervous system stimulant and can induce transient sympathomimetic effects. DMAA can be commercially synthesized and has been detected in small quantities in cultivars of
The reports of Gee and colleagues represent adverse outcomes following a single ingestion of very high dosage of DMAA, 4,5 which may have been due to the pressor effects of DMAA. In our early work using DMAA, 6 we noted an acute rise in systolic blood pressure following oral ingestion, which was dose dependent (higher with a 75-mg dosage when compared to a 50-mg dosage). Hence, it is logical to assume that a DMAA dosage of 10 times the amount provided in our early work (and likely the amount ingested by some patients in the Gee et al. reports 4,5 ) would lead to adverse outcomes.
Within the weight loss and sport performance market, many individuals use caffeine and DMAA chronically at a much lower and recommended dosage (i.e. 150–250 mg caffeine and 25–50 mg DMAA). At such dosages and
Methods
Subjects
Men were recruited to participate in this study via informal word of mouth conversations, formal presentations discussing study participation, online postings, and recruitment flyers posted on and off campus. All subject recruitment was performed by the investigators (TMF, ICH, and RJA). A total of 66 healthy men provided written informed consent to participate in this study. However, 16 men failed to complete all aspects of the study (e.g. missed one of the assessment days and decided to cease their participation due to time constraints). Therefore, only 50 men are included in the data analysis. Subject baseline characteristics are presented in Table 1. Women were not enrolled in this study in an attempt to maintain a more homogenous sample. Subjects were not current smokers and were considered to be in good overall health. They did not have a history of cardiovascular, neurological, or metabolic disorders (e.g. hypertension, seizures, and diabetes). Subjects were regular consumers of stimulants (e.g. caffeine) such as beverages or nutritional supplements, who did not report a history of adverse reactions to caffeine or other stimulants. Health history, medication and dietary supplement usage, and physical activity questionnaires were completed by all subjects and reviewed in detail by an investigator to determine eligibility. Prior to participation, each subject was informed of all procedures, potential risks, and benefits associated with the study through both verbal and written form. The study procedures were approved by the University Institutional Review Board for Human Subjects Research (approval document number 2012). Subjects who completed the study protocol were compensated $200 for their time and effort.
Characteristics of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

DMAA: 1,3-dimethylamylamine.
aValues are mean ± SEM. No other differences of statistical significance noted (
b
Testing and supplementation
Following the initial lab visit in which subjects completed all paperwork, subjects returned to the lab on three occasions (before supplementation, after 6 weeks of supplementation, and after 12 weeks of supplementation) to complete the following assessments. On each occasion subjects reported to the lab following an 8-h overnight fast. Upon arrival, the following was performed: Subjects turned in diet logs (described below). Subjects voided. They were given a standard urine collection cup and provided with instructions for filling. Urine was processed immediately and analyzed within 24 h for a complete urinalysis with microscopic examination (using reagent strips and light microscopy). Subjects were prepped for a resting 12-lead ECG and rested quietly for 10 min. An ECG was then analyzed using automated procedures (Mac 1200; GE Medical System). Respiratory rate (in 60s) was counted by simple observation. Blood pressure was measured using a stethoscope and cuff, while subjects were in a seated position. A blood sample was taken and processed as described below. Body mass and body composition were determined via dual energy x-ray absorptiometry (DEXA) using a 4-min fan array (Hologic QDR 4500W; Bedford, Massachusetts, USA).
Following the above assessments, subjects were randomly assigned to one of four conditions, in double-blind manner: placebo (cellulose), caffeine (250 mg), DMAA (50 mg), or caffeine (250 mg) + DMAA (50 mg). All conditions were produced under standard good manufacturing practices by a dietary supplement contract manufacturer. Quality assurance procedures confirmed the purity and potency of each condition. The study sponsor retained the blinding code until study completion.
For all conditions, subjects were instructed to consume one capsule per day during week 1, in order to acclimate to the condition. Then, beginning with week 2, subjects were instructed to consume two capsules every day of the week for the remainder of the 12-week intervention (one capsule upon rising in the morning and one capsule 4–6 h later). The final capsules were to be consumed on the day prior to the final test day; no capsules were consumed before assessments were performed on the mornings of test days. Capsule counts upon bottle return allowed for the calculation of compliance to intake.
Blood collection and analysis
Venous blood samples (∼20 mL) were taken from subjects via needle and Vacutainer®. Blood samples were collected before starting supplementation (Pre) and after 6 and 12 weeks of supplementation. Following collection, samples were processed accordingly and fresh samples were analyzed for complete blood count (Coulter LH750, Beckman Coulter, Inc.), comprehensive metabolic panel (Roche/Hitachi Modular), and lipid panel (Roche/Hitachi Modular). In addition, fresh samples were analyzed for C-reactive protein (using a high-sensitivity, particle-enhanced turbidimetric immunoassay; Roche Integra 800) and cardiac troponin I (using an electrochemiluminescence immunoassay). Remaining blood was stored at −70°C and later analyzed for plasma malondialdehyde (using reagents purchased from Northwest Life Science Specialties, Vancouver, Washington, USA), advanced oxidation protein products (using reagents purchased from Cell Biolabs, San Diego, California, USA), and Trolox Equivalent Antioxidant Capacity (using reagents purchased from Sigma Chemical, St Louis, Missouri, USA).
Dietary records and activity
All subjects were instructed to maintain their normal diet throughout the study period, with the following exceptions: subjects were instructed not to consume caffeinated beverages such as “energy drinks”, coffee, tea, or soda during the study period. They were also advised not to use dietary supplements containing caffeine or other stimulants (other than their assigned condition). Subjects were asked to record food and beverage intake during 72 h prior to each test day (pre, 6 weeks, and 12 weeks). Dietary records were reviewed with each subject for accuracy and then analyzed using Food Processor SQL, version 9.9 (ESHA Research, Salem, Oregon, USA). Subjects were asked to refrain from strenuous physical activity for 48 h prior to each test day, but they were to otherwise maintain their usual exercise training during the entire course of the study.
Statistical analysis
Data were analyzed using a 4 (condition) by 3 (time) analysis of variance. Tukey’s post hoc testing was used as needed. The data are presented as mean ± SEM. All analyses were performed using JMP statistical software. Statistical significance was set at
Results
Of the 50 subjects who completed the study, none reported a significant adverse event. Compliance to capsule intake was not different (
Cardiorespiratory data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a
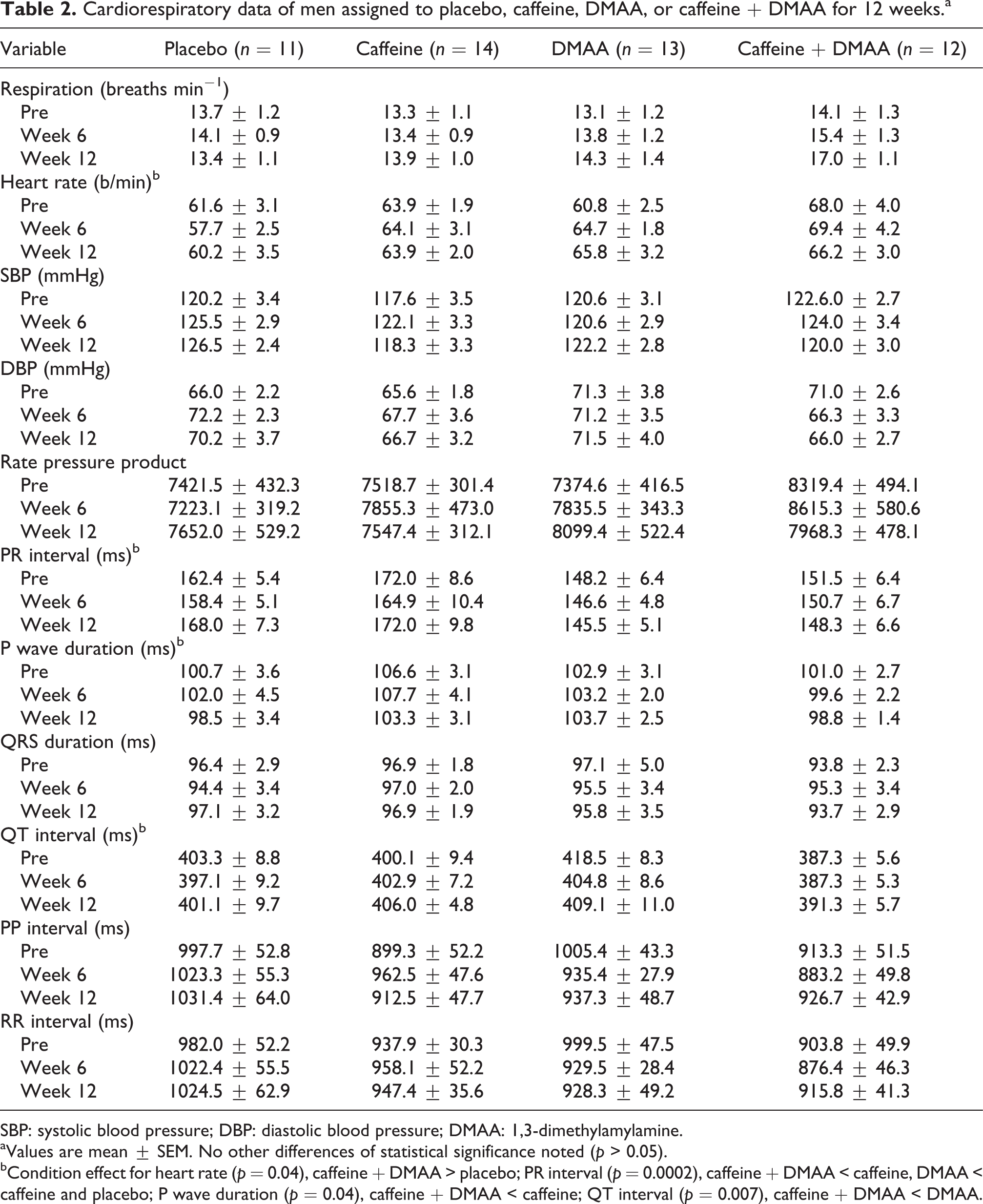
SBP: systolic blood pressure; DBP: diastolic blood pressure; DMAA: 1,3-dimethylamylamine.
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect for heart rate (
Urinalysis data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.

DMAA: 1,3-dimethylamylamine.
aValues are mean ± SEM. No other differences of statistical significance noted (
aTime effect (
Oxidative stress, inflammatory, and cardiac biomarker data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

AOPP: advanced oxidation protein products; TEAC: trolox equivalent antioxidant capacity; DMAA: 1,3-dimethylamylamine.
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect for AOPP (
Complete blood count data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

DMAA: 1,3-dimethylamylamine;
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect for WBC (
Comprehensive metabolic panel data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

DMAA: 1,3-dimethylamylamine; BUN: blood urea nitrogen; AST: aspartate transaminase; SGOT: serum glutamic oxaloacetic transaminase; ALT: alanine transaminase; SGPT: serum glutamic pyruvate transaminase; GGT: γ-glutamyl transpeptidase; A: adenine; G: guanine.
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect for creatinine (
cTime effect (
Lipid panel data of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

DMAA: 1,3-dimethylamylamine; HDL-C: high-density cholesterol; LDL-C: low-density cholesterol.
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect cholesterol (
Dietary intake of men assigned to placebo, caffeine, DMAA, or caffeine + DMAA for 12 weeks.a

DMAA: 1,3-dimethylamylamine; RE: Retinol equivants.
aValues are mean ± SEM. No other differences of statistical significance noted (
bCondition effect for protein (
cInteraction effect for vitamin C (
Discussion
Findings from the present study indicate that 12 weeks of supplementation with caffeine and DMAA, whether alone or in combination, does not result in statistically significant changes in any measured variables. These data are specific to a sample of healthy, young men consuming moderate, and recommended daily dosages of caffeine (250 mg) or DMAA (50 mg). These findings confirm our initial results obtained from subjects ingesting finished dietary supplements containing these two ingredients. 7,8 However, our findings are in conflict with those of Gee and colleagues, 4,5 who reported adverse outcomes in four patients, who ingested dosages of DMAA that far exceeded the dosage provided in the current design. As with other widely consumed stimulants (e.g. caffeine), ingestion of multiple times the recommended dosage is highly unadvisable and could result in severe consequences.
Subject characteristics were very similar between conditions, with the exception of the number of years of aerobic exercise training—which was significantly higher for subjects in the caffeine + DMAA condition when compared with those in the placebo condition (Table 1). Although we do not have maximal oxygen uptake (VO2max) data to confirm this, it is possible that these subjects (as a group) may have been more conditioned from a cardiorespiratory perspective than subjects in the other conditions. Likewise, it should be noted that subjects in this study were relatively young and healthy—and were regular users of stimulants prior to enrolling in the study. It is possible that the inclusion of older and sedentary adults (men or women), who are unhealthy and who do not regularly use stimulants, may yield different findings. The same may be true for younger individuals (preteens and teens), who appear to be regular users of “energy drinks” and other products containing stimulants. For these populations, the use of stimulants would be strongly ill advised.
In addition to subject characteristics, dietary intake was very similar between conditions, with intake as expected for younger and healthy men (Table 8). Therefore, we do not believe that our outcome measures between groups or across time were influenced by dietary intake.
With regard to the cardiorespiratory data presented in Table 2, a few variables deserve attention. First, subjects’ heart rate increased in a nonstatistically significant manner (approximately 5 b/min) from pre to week 12 in the DMAA condition. Second and by default, the increase in heart rate produced a similar percentage increase in rate pressure product, as heart rate is used in the calculation of rate pressure product (systolic blood pressure × heart rate). Interestingly, no change was observed in the heart rate for subjects in the caffeine or caffeine + DMAA conditions. Third, respiratory rate for subjects in the caffeine + DMAA condition increased over time, which appeared somewhat additive when considering the small increase observed for both caffeine and DMAA alone. It is possible that a larger sample size, providing more statistical power, may have yielded statistically significant findings in the above measures. Future studies may include a larger sample of subjects ingesting DMAA alone or in combination with caffeine (and possibly other widely utilized stimulants) in an effort to extend and/or replicate our findings and to determine what, if any, clinical meaning these increases might have.
With regard to the oxidative stress, inflammatory, and cardiac biomarker data, a few variables deserve attention (Table 4). First, advanced oxidation protein products demonstrated a rather random increase (in a nonstatistically significant manner) for subjects in both the caffeine and DMAA conditions, but did not do so in subjects in the caffeine + DMAA condition. Second, although not of statistical significance, C-reactive protein increased slightly in subjects in the caffeine condition but decreased in a similar manner in subjects in the DMAA and caffeine + DMAA conditions. Third, troponin I was below the limit of detection in all subjects, in all conditions, at all times of measurement—indicating that none of the conditions impacted cardiac muscle damage as determined by this marker.
The remaining measures related to complete blood count, metabolic panel, and lipid panel were unremarkable, with values remaining very stable across time for subjects in all conditions. These findings agree with our prior work involving subjects’ ingestion of finished dietary supplements containing caffeine and DMAA. 7,8
When considering the typical user of dietary supplements containing caffeine and DMAA, it is important to note that most individuals will not use these products daily. Specifically, many individuals use the ingredients/supplement exclusively on exercise training days, which might consist of 3–5 days per week. Moreover, the dosage of caffeine and DMAA contained within one serving of many dietary supplements sold on the market is less than the amount ingested by subjects in the present study. Therefore, unless individuals are abusing the dietary supplements (which is certainly possible), the cumulative 12-week dosage provided in the present study should have allowed for a robust assessment of the health effects of caffeine and DMAA.
We conclude that 12 weeks of supplementation with caffeine and DMAA, whether alone or in combination, does not result in a statistically significant change in any of the measured outcome variables. Data are specific to a sample of healthy, young men consuming daily dosage of caffeine (250 mg) and DMAA (50 mg) that are considered moderate and recommended. With the above understanding, it is possible that DMAA alone and in combination with caffeine may impact certain cardiorespiratory variables. Additional study may consider the use of a larger sample of subjects, perhaps of both genders and of various age groups, to more fully elucidate the safety profile of these ingredients when used alone and in combination for extended periods of time.
Footnotes
Funding
Funding for this work was provided in part by USPlabs, LLC, and the University of Memphis. RJB has been a Consultant for and/or Principal Investigator on research studies funded by various dietary supplement companies.
Authors’ contributions
RJB was responsible for the study design, biochemical work, statistical analyses, and manuscript preparation. TMF, ICH, and RJA were responsible for subject recruitment, data collection, data entry, and assistance with manuscript preparation. All authors read and approved the final manuscript.
