Abstract
Apoaequorin is a bioluminescent calcium-binding apoprotein endogenous to the Aequorea species of jellyfish and is commercially available in a dietary supplement in support of brain and cognitive health. Results from a previous 90-day subchronic oral gavage study established the no-observed-adverse-effect-level (NOAEL) of lyophilized apoaequorin protein powder (LAPP) at 666.7 mg/kg/day. The current 90-day oral gavage study in Sprague-Dawley rats administered dose levels of 1000, 2000, and 4000 mg/kg/day of test substance as received. These doses are expressed as milligram of supplement with the amounts of apoaequorin based on the analysis of the percentage of active ingredient. The corresponding amounts of apoaequorin protein are 603, 1206, and 2412 mg/kg/day. These dose levels target approximately 4221, 8442, and 16,844 times more than the expected human oral intake. There were no mortalities, clinical observations, ophthalmological, clinical pathology, or histopathological changes attributable to LAPP administration. Changes in mean body weight and feed efficiency, without other correlating clinical or pathological or other toxicologically relevant findings, were considered to be of little toxicological significance. Therefore, the NOAEL for LAPP administered orally up to 90 days was 4000 mg/kg/day (2412 mg/kg/day based on 603 mg/g or 60.3% active ingredient, apoaequorin protein), the highest dose tested in male and female rats.
Introduction
Apoaequorin (CAS No. 845410-67-5) is a calcium-binding apoprotein endogenous to coelenterates of Aequorea species, with a molecular weight of 22.3 kDA. It is part of aequorin, a photoprotein isolated from the jellyfish Aequorea victoria. Aequorin emits blue light by an intramolecular reaction when mixed with Ca2+. 1 This light can be recorded in ways that allow the transient luminescence to indicate where calcium is being liberated inside the cell. 2 Due to their bioluminescence properties, recombinant aequorins have been used to monitor calcium levels at both the subcellular and cytoplasmic levels. Engineered recombinant aequorins have led to novel technological approaches to monitor calcium signals in organelles and subcellular domains. 3,4
Apoaequorin is also a member of the EF-hand calcium-binding family of proteins of which include calmodulin, with the ability to form dimers with a potential of eight bound and six bound Ca2+ ions associated with cholesterol binding to apoaequorin domains. 5 The role of calcium in neurobiology is widely recognized. Alterations in calcium homeostasis have been claimed to underlie the altered cellular function characteristic of the aging process. Calcium-binding proteins have been recognized as being susceptible to toxicity via calcium and calcium-mediated events, such as excess calcium-induced cell death or excitotoxicity. For example, xenobiotic-induced neurotoxicity via Ca2+ influx can be mediated by glucose levels, which in turn regulate the expression of a selected family of cation channels referred to as transient receptor potential canonical channels (TRPC). 6 Available information indicates that calcium-channel blocking agents control the flow of calcium ions from crossing calcium channels in order to reduce the signaling by the calcium ion and the adverse effects of calcium crossing through these channels. In some cases, TRPCs joined with metabotropic glutamate receptors may induce excitotoxicity by Ca2+ influx. 6
Recent data show that apoaequorin may be associated with improvements in a quantitative measure of cognitive function, specifically verbal learning, in aging adults, thus becoming a potential tool in addressing age-related decline in cognitive function 7 and to facilitate memory maintenance. 8
The safety evaluation of apoaequorin has become paramount to enabling further research, development, and commercialization efforts. Through more rigorous testing, raising the no-observed-adverse-effect-level (NOAEL) can secure a higher margin of safety for the continued expansion of product development.
Apoaequorin has been used for over 30 years in living systems as an analytical tool for cell biology without evidence of adverse biological effects. 9 The fact that apoaequorin shares homologies with calcium-binding proteins that are also found in the human body 10 suggests that it is unlikely to cause adverse effects. The role of this protein in the human body as a protective agent has also been recognized. 11
The safety profile of apoaequorin has been evaluated in both in vitro and in vivo studies involving animal models. In in vitro studies, the protein apoaequorin has been used in multiple cell types and localized to important intracellular compartments, of which there were no reported indications of any deleterious or toxic effects from exposure. 4
The allergenic profile of apoaequorin has also been investigated. There are currently no known inducers of allergy to A Victoria or other phylogenetically related organisms. To further perform specific risk assessment of apoaequorin allergenicity and capacity of the digested protein to cause sensitization or allergic elicitation, an exhaustive bioinformatic assessment against allergen databases following in vitro simulated gastric digestion of the protein was conducted. Results suggest the in silico assessment support that this protein is not a known allergen or likely to cross react with known allergens and exhibits a digestibility profile similar to other nonallergic dietary proteins. 12
Previous work reported in a 90-day subchronic oral gavage toxicity test, at dose levels of 92.6, 462.9, and 926.0 mg/kg/day (corresponding to 66.7, 333.3, and 666.7 mg/kg/day of apoaequorin), demonstrated that this protein has no toxicity by oral exposure in Sprague-Dawley rodents. Administration of lyophilized apoaequorin protein powder (LAPP) at levels up to 926.0 mg/kg/day did not cause adverse effects in male and female rats with a NOAEL of apoaequorin protein established at 666.7 mg/kg/day, the highest dose tested. 9
Given the interest for continued use of LAPP as a dietary supplement and with the objective of further characterizing the safety profile of apoaequorin, an additional study was conducted administering doses higher than previously tested. The current study assessed the effects of the LAPP preparation when administered daily for 90 days, via oral gavage to Sprague-Dawley rats, up to 4000 mg/kg/day that corresponds to the equivalent dose of apoaequorin received at 2412 mg/kg/day. With the increased attention and popularity of products in the brain health space, additional safety data using as high a safety factor as possible is extremely pertinent to consumers, regulators, and the industry. Consumers are seeking out ingredients/products that exhibit well-established safety profiles.
Materials and methods
Study design
The study was performed at Product Safety Labs (PSL, Dayton, NJ, USA) in the departments of Toxicology and Analytical Chemistry, in accordance with: (a) US Food and Drug Administration (FDA) Redbook (2000): Chapter IV.C.4.a Subchronic Toxicity Studies with Rodents 13 ; (b) Organization for Economic Co-operation and Development (OECD) Guidelines for the Testing of Chemicals, Section 4 Health Effects (Part 408): Repeated Dose 90-day Oral Toxicity Study in Rodents; and (c) standard operating procedures at PSL and as per the mutually agreed study plan with the Sponsor. This study utilized both FDA and OECD guidance taking into consideration animal welfare concerns, with the minimal number of animals used to make accurate inferences about the sample population. The study meets the requirements of 21 CFR 58:US FDA Good Laboratory Practices (GLP) Standards, 1987 and OECD Principles of GLP, 1997.
Test item
Standardized LAPP used in the present study was provided by Quincy Bioscience (Madison, WI, USA) and was the same product as used in the previous 90-day subchronic study (Quincy Bioscience has given permission and financial resources for the use of this test product in the current study). 9 The product is a tan colored powder, containing 60.3% of apoaequorin w/w dry powder, as measured by reversed-phase high-performance liquid chromatography (RP-HPLC). The protein is produced through recombinant fermentation of innocuous prokaryotic species and recovered from the fermentation broth by purification and drying. Fermentation and cell removal are performed by a large-scale producer of biologics and probiotics and follow National Institutes of Health (NIH) guidelines for large-scale fermentation of recombinant organisms (Effective June 24, 1994, Published in Federal Register, July 5, 1994 (59 FR 34472)). Clarification of the fermentation mash through hollow fiber tangential flow filtration removes cells and results in a clear aqueous broth in which the protein is suspended. The purified protein is freeze dried and tested for potency and microbiology. Ash, carbohydrates, moisture, and total fat found in the test substance are food grade components used in the fermentation medium but not consumed by the producing organism. The manufacturing process complies with the good manufacturing practices (GMP, 21CFR 111).
RP-HPLC is used to both quantitate and identify apoaequorin in the final product, using a protein standard developed for this purpose and qualified through rigorous purification and test methods. In addition to HPLC, SDS-PAGE is carried out to confirm the identity of the protein. The molecular weight of apoaequorin (CAS No. 845410-67-5) is 22.3 kDA. Other specification parameters, such as moisture and microbiological analysis for food borne pathogens, are routinely monitored.
Animals
All animal care and experimental protocols were in accordance with the NIHguidelines 14 for the care and use of laboratory animals and were fully approved by the PSL Institutional Animal Care and Use Committee (National Research Council of the National Academies 15 ). Male and female Sprague-Dawley rats (Charles River Laboratories, Wilmington, MA, USA 16 ) were used for the present study. Prior to initiation of testing, the rats were acclimated to laboratory conditions for 7 days. Following acclimatization, animals (n = 80) were selected for the study on the basis of adequate body weight gain, freedom from clinical signs of disease or injury, and a body weight within ±20% of the mean within a sex. Selected rats were randomly distributed by body weight using a stratified randomization procedure. The animals weighed 202–252 g (males) and 162–190 g (females) and were approximately 8 weeks of age at initiation of dosing.
The animals were individually housed in suspended stainless steel caging with mesh floors. Litter paper was placed beneath the cages and was changed at least three times per week. All animals were housed under standard laboratory conditions, with adequate fresh air supply, room temperature 19–23°C, relative humidity 43–69%, with 12-h light–dark cycle, and the room was kept clean and vermin free. The animals were fed (ad libitum) with 2016 Certified Envigo Teklad Global Rodent Diet® throughout the acclimatization and experimental period. All animals were provided with filtered tap water (ad libitum) from an automatic water dispensing system.
Experiment
Rats were divided into four groups (10/sex/group). One group was used for vehicle control while the other three were used as treatment groups and received the test substance, LAPP. All animals were treated orally (gavage) once daily with test substance prepared in a stock solution of 0.005 M tris buffer using deionized, reverse osmosis water at dose levels of 0 (vehicle control group), 1000 (low-dose group), 2000 (mid-dose group), or 4000 (high-dose group) mg/kg/day (dosing volume 10 mL/kg) for at least 90 consecutive days, based on a previous dose range-finding study. 17 Thus, the equivalent dose of LAPP received by each group was 0, 603, 1206, and 2412 mg/kg/day, respectively. Fresh aliquots containing 10% (low dose), 20% (intermediate dose), and 40% (high dose) of the test substance were prepared daily before administration. All dose solutions were prepared fresh daily. The control group received the vehicle at the same volume as the test animals. The dose levels selected were based on the expected human dose levels of LAPP supplement at approximately 10 mg/person/day (i.e. assuming a 70 kg human). These dose levels targeted approximately 4221, 8442, and 16,884 times higher than the expected human oral intake on a milligram per day basis. The high dose was considered the maximum practical dose with this formulation and test system and was not expected to cause marked toxicity. The low and intermediate dose levels were selected to derive a potential dose–response for any effects seen. Based on the stability, homogeneity (HO), and concentration verification results of the test substance, the animals are considered to have received the targeted dose concentrations of LAPP.
Parameters investigated
Chemical analysis
Neat test substance samples were collected on days 1, 43, and 94. Samples of dose solutions were collected from each dose level on day 1 for HO and on days 43 and 94 for concentration verification. The method using HPLC was validated in terms of linearity, system suitability (instrument precision), specificity, accuracy (spike recovery), and precision. All samples were stored frozen and were thawed prior to analysis.
Clinical and ophthalmological examination, body weight, and feed consumption
All animals were observed once daily for clinical signs of toxicity and twice daily for mortality. On day 1 (prior to first treatment with the test substance) and approximately weekly thereafter, a detailed observation was conducted while handling each animal, generally occurring on days that the animals were weighed and feed consumption measurements taken. Potential signs noted included, but were not limited to, changes in skin, fur, eyes, and mucous membranes, occurrence of secretions and excretions, and autonomic activity (e.g. lacrimation, piloerection, pupil size, and unusual respiratory pattern). Likewise, changes in gait, posture, and response to handling, as well as the presence of clonic or tonic movements, stereotypies (e.g. excessive grooming and repetitive circling), or bizarre behavior (e.g., self-mutilation and walking backwards), were also recorded.
Ophthalmological examination was performed by a board-certified veterinary ophthalmologist on all animals before the start of study and on day 81 of the study, which included slit lamp biomicroscopy and indirect ophthalmoscopy (fundoscopy). Individual body weights were recorded at least two times prior to randomization. Test animals were weighed on day 1 (prior to study start) and approximately weekly thereafter (intervals of 7 days ± 1). The animals were also weighed immediately prior to euthanasia in order to calculate organ-to-body/brain weight ratios. Body weight gain was calculated for selected intervals and for the study overall. Individual animal feed consumption was measured and recorded weekly to coincide with body weight measurements. Feed efficiency was calculated and reported. All animals were fasted overnight prior to blood collection.
Clinical pathology
Clinical pathology was performed on all animals for blood chemistry and hematology of the terminal sacrifice animals once toward the end of the in-life phase of the study. Blood samples for hematology (except coagulation samples) and clinical chemistry were collected via sublingual bleeding under isoflurane anesthesia on study day 88 of the test period. Approximately, 500 µL of blood was collected in a precalibrated tube containing K2EDTA for hematology assessments. The whole blood samples were stored under refrigeration and shipped on cold packs. Approximately, 1000 µL was collected into a tube containing no preservatives for clinical chemistry assessments. These samples were centrifuged in a refrigerated centrifuge and the serum was transferred to a labeled tube. Blood samples used to determine the prothrombin time (PT) and partial thromboplastin time (coagulation) were collected via the inferior vena cava under isoflurane anesthesia at termination. All samples were stored in a −80°C freezer and shipped frozen in dry ice to DuPont Haskell Global Centers for Health and Environmental Sciences (Newark, DE, USA) for further analysis. All blood samples were evaluated for quality by visual examination. The day before collection of samples for the clinical pathology evaluation, the animals were placed in metabolism cages. Animals were fasted after 3 pm (at least 15 h prior to necropsy) and urine was collected from each animal.
Hematology
Hematological parameters included erythrocyte count (RBC), hemoglobin concentration (HGB), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), red cell distribution width (RDW), absolute reticulocyte count (ARET), platelet count (PLT), total white blood cell (WBC), absolute neutrophil (ANEU), absolute lymphocyte (ALYM), absolute monocyte (AMON), absolute eosinophil (AEOS), absolute basophil (ABAS), absolute large unstained cell (ALUC), and differential leukocyte count. MCH concentration (MCHC) was calculated. In addition, separate blood smears, stained with new methylene blue or “Wright-Giemsa” stain, were prepared from each animal undergoing hematological evaluation and were examined, if required, to substantiate or clarify the results of hematology findings.
Coagulation assessment
Coagulation parameters included PT and activated partial thromboplastin time (APTT).
Clinical chemistry
Clinical chemistry parameters measured included serum aspartate aminotransferase (AST), serum alanine aminotransferase (ALT), sorbitol dehydrogenase (SDH), alkaline phosphatase (ALKP), total bilirubin (BILI), urea nitrogen (BUN), blood creatinine (CREA), total cholesterol (CHOL), triglycerides (TRIG), fasting glucose (GLUC), total serum protein (TP), albumin (ALB), globulin (GLOB), calcium (CALC), inorganic phosphorus (IPHS), sodium (NA), potassium (K), and chloride (CL).
Urinalysis
Urinalysis parameters included quality (QUAL), pH, ketone (KET), color (COL), glucose (UGLC), bilirubin (UBIL), clarity (CLAR), specific gravity (SG), blood (BLD), volume (UVOL), protein (UMTP), urobilinogen (URO), and microscopic urine sediment examination.
Pathology
Terminal necropsy and tissue collection. At termination, all surviving animals were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia. All animals in the study were subjected to a full necropsy, which included examination of the external surface of the body, all orifices, and the thoracic, abdominal, and cranial cavities and their contents. The following tissues were weighed wet as soon as possible after dissection to avoid drying: adrenals (combined), kidneys (combined), spleen, brain, liver, thymus, epididymides (combined), ovaries with oviducts (combined), uterus, heart, and testes (combined). Over 40 organs and tissues were collected and preserved in 10% neutral buffered formalin for histopathological examinations (in accordance with (a) FDA Redbook (2000): Chapter IV.C.4.a Subchronic Toxicity Studies with Rodents 13 ; (b) OECD Guidelines for the Testing of Chemicals, Section 4 Health Effects (Part 408): Repeated Dose 90-day Oral Toxicity Study in Rodents; and (c) standard operating procedures at PSL. Additionally, epididymides, eyes with optic nerve, and testes were preserved in modified Davidson’s fixative and then retained in ethanol for histopathological examination.
Histopathological examination
The preserved organs and tissues of the animals from both the control and high-dose groups were subjected to histological examination. In addition, gross lesions of potential toxicological significance noted in any of the test groups at the time of terminal sacrifice were also examined. The fixed tissues were trimmed, processed, embedded in paraffin, sectioned with a microtome, placed on glass microscope slides, stained with hematoxylin and eosin, and examined by light microscopy. Slide preparation and histopathological assessment, by a board-certified veterinary pathologist, were performed by Histo-Scientific Research Laboratories (Mt. Jackson, VA, USA).
Statistical analysis
For the in-life and organ weight data, mean and standard deviations were calculated for all quantitative data. If warranted by sufficient group sizes, data within groups were evaluated for HO of variances and normality by Bartlett’s test. 18 Where Bartlett’s test indicated homogeneous variances, treated and control groups were compared using a one-way analysis of variance (ANOVA). When one-way ANOVA was significant, a comparison of the treated groups to control by Dunnett’s test 19,20 for multiple comparisons was performed. Where variances were considered significantly different by Bartlett’s test, groups were compared using a nonparametric method (Kruskal–Wallis nonparametric ANOVA 21 ). When nonparametric ANOVA was significant, comparison of treated groups to control was performed using Dunn’s test. 22 Statistical analysis was performed on all quantitative data for in-life and organ weight parameters using Provantis® version 9, Tables and Statistics, Instem LSS (Staffordshire, UK).
Clinical pathology data were initially evaluated by Levene’s test 23 for HO and Shapiro–Wilk test 24 for normality. If the preliminary test was not significant, a nonparametric one-way ANOVA followed with Dunn’s test was performed, while in the case of significance, a parametric one-way ANOVA followed by Dunnett’s test. Transforms of the data were used to achieve normality and variance HO. The order of transforms attempted were log, square-root, and rank-order. If the log and square-root transforms failed, the rank-order was used. Other statistical methods were used when appropriate. Significance was judged at a probability value of p < 0.05. Male and female rats were evaluated separately.
Results
Analytical chemistry
Neat test substance samples from the beginning, middle, and near the end of the study were analyzed for the active ingredient, apoaequorin. The difference in the active concentration of the test substance over the course of the study was −0.7% and the overall stability was determined to be 99.3% (Table 1). HO analysis of the day 1 dose preparations resulted in a relative standard deviation (RSD) of 1.83, 1.38, and 3.78% for groups 2–4, respectively. Average percent of target concentrations in the top, middle, and bottom samples were 114.0, 109.8, and 99.0% of the target concentrations of 54.6, 109.3, and 218.5 mg/mL, corrected for apoaequorin active in groups 2–4, respectively (Table 1). Concentration verification analysis samples were collected on the day of preparation for initial (day 1, as part of the HO assessment), middle (day 43), and final preparations (day 94). The mean of day 1 samples were 114.0, 109.8, and 99.0% of target, the day 43 samples resulted in 98.6, 99.1, and 92.6% of target, and the day 94 samples resulted in 98.6, 98.2, and 90.7% of the target corrected concentrations of apoaequorin active for groups 2–4, respectively (Table 1). In summary, based on the overall neat test substance stability, HO, and concentration verification analysis, animals were considered to have received accurate target concentrations of LAPP, based on analysis of the active apoaequorin.
Chemical analysis results.
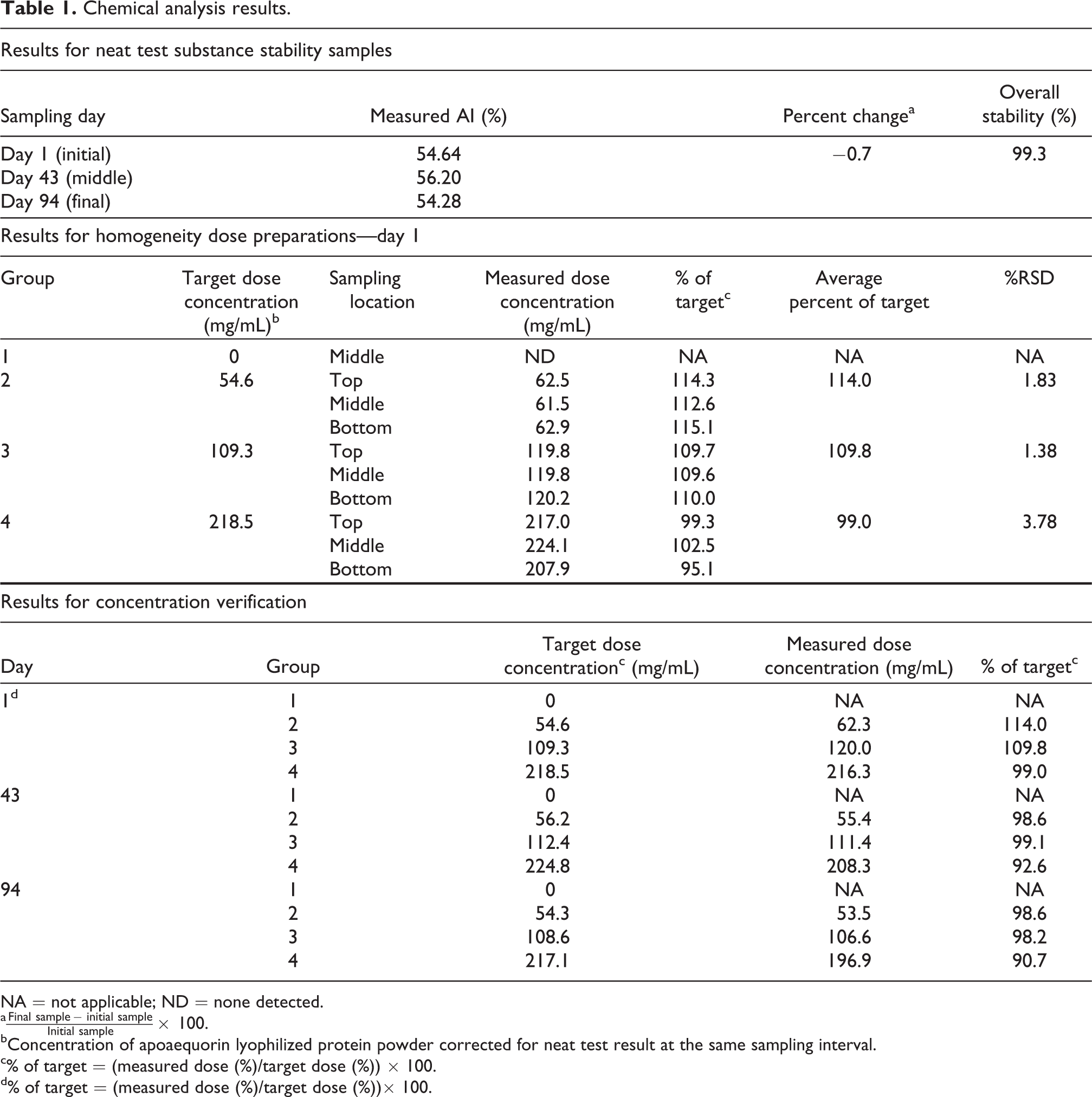
NA = not applicable; ND = none detected.
a
bConcentration of apoaequorin lyophilized protein powder corrected for neat test result at the same sampling interval.
c% of target = (measured dose (%)/target dose (%)) × 100.
d% of target = (measured dose (%)/target dose (%))× 100.
Survival, clinical observations, ophthalmological examination
There were no test substance-related mortalities during the course of study. One high-dose male and one high-dose female were found dead during the study on days 70 and 51, respectively. Prior to death starting on day 63, the high-dose male exhibited gasping, facial and ano-genital staining, moist rales, and nasal discharge. Corresponding detailed observations for this animal included urine stained hair (ano-genital staining) and moist/dry rales. At necropsy, this animal exhibited a thoracic effusion, a collapsed and discolored lung, as well as an enlarged liver. Upon microscopic evaluation, notable lesions included decreased splenic lymphocytes, minimal cardiomyopathy, and mild thymic hemorrhage. The lack of microscopic lung lesions was interpreted to be consistent with aspiration of dose as the cause of death. No abnormal clinical observations were noted for the high dose female prior to death. At necropsy, a 7 mm perforation of the cranial third of the esophagus, associated with red discoloration of the esophageal wall, was observed. This finding was confirmed by microscopic evaluation and was associated with local inflammation and interpreted to be the probable cause of death. Additional notable microscopic findings included decreased splenic lymphocytes and moderate renal tubular necrosis.
There were no clinical signs in either male or female surviving animals, resulting from the administration of LAPP. All clinical signs and observations were considered incidental and regarded as not toxicologically relevant. These observations included moist rales; red or brown nasal discharge; a broken upper incisor; superficial eschar on the neck, nose/snout, face, back, or right forelimb; alopecia; a laceration and visible swelling in the right forelimb; a broken toenail on a forepaw; ocular discharge; and incisor malocclusion. Additionally, one mid-dose male had a firm, round mass measuring 30 × 20 × 15 mm, which was noted on study day 36, on the right dorsal surface between the shoulder blades during in-life observations. The mass decreased in size to 6 × 12 × 1 mm on study day 50 and was resolved by day 57. No macroscopic observations were noted for this animal.
An ophthalmology evaluation of all animals near experimental completion (day 81) did not reveal any abnormalities. There were no test substance-related microscopic ocular findings (data not shown).
Body weights and feed consumption
In male rats, the mean weekly body weights (Figure 1) for the treated groups at 1000, 2000, and 4000 mg/kg/day of LAPP were generally comparable with control values throughout the study. For all intervals, other than those noted as statistically significant as determined by nonparametric ANOVA (mid dose increased, week 7, p < 0.01), the mean daily body weight gain (Table 2) for all the treated male rats was generally comparable with the control group values throughout the study.
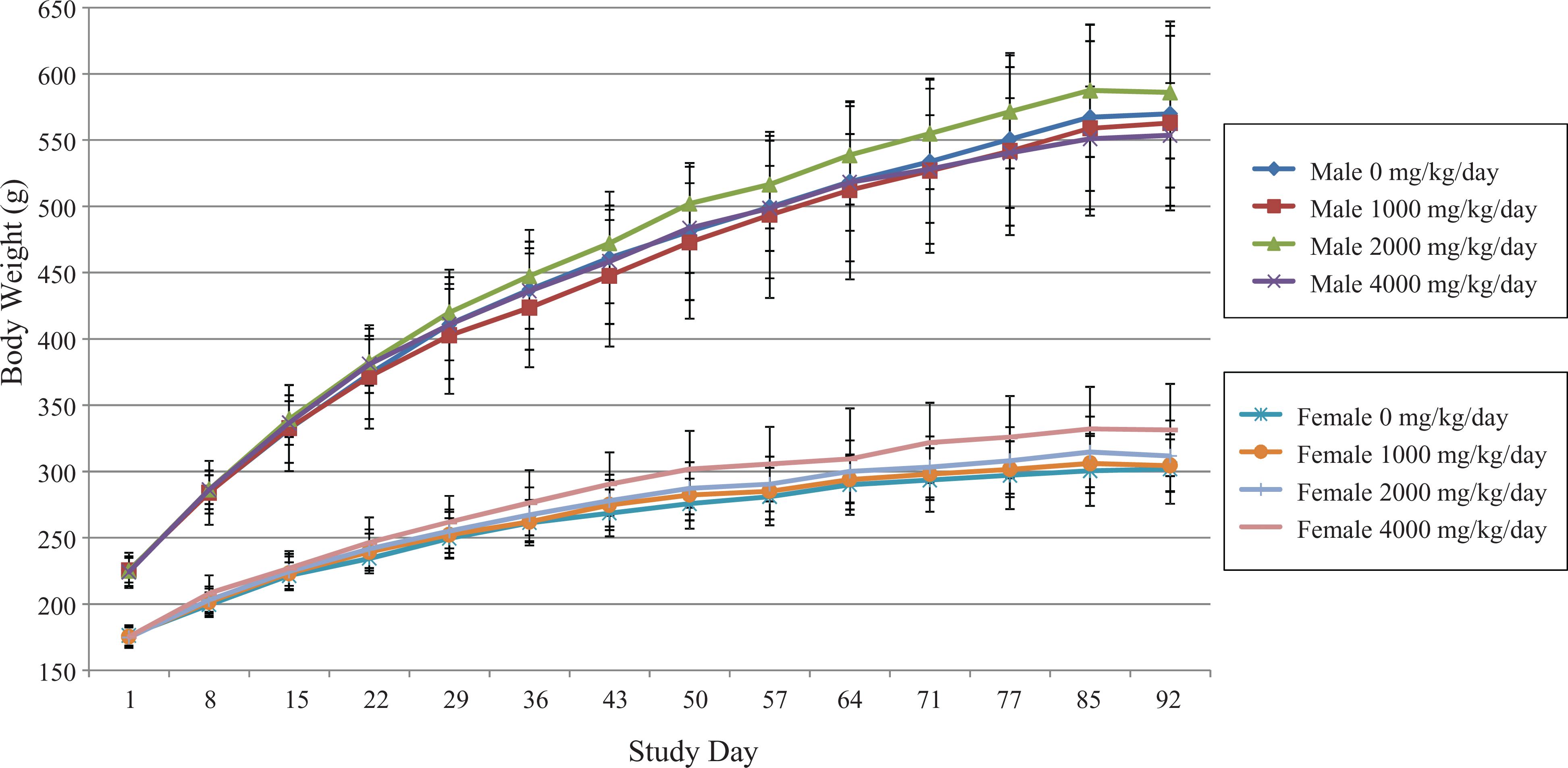
Mean weekly body weights were comparable across treatment groups. Overall, mean weekly body weights for male rats in groups 2–4 were comparable to control group 1 males throughout the study. Mean weekly body weights for females in groups 2–4 were generally comparable to control group 1 females throughout the study, with the exception of a statistically significant increase in group 4 females on day 50 (p < 0.05). There were no statistically significant changes in body weight of male and female rats attributable to administration of LAPP. LAPP: lyophilized apoaequorin protein powder.
Mean daily body weight gain (g) in males and females.
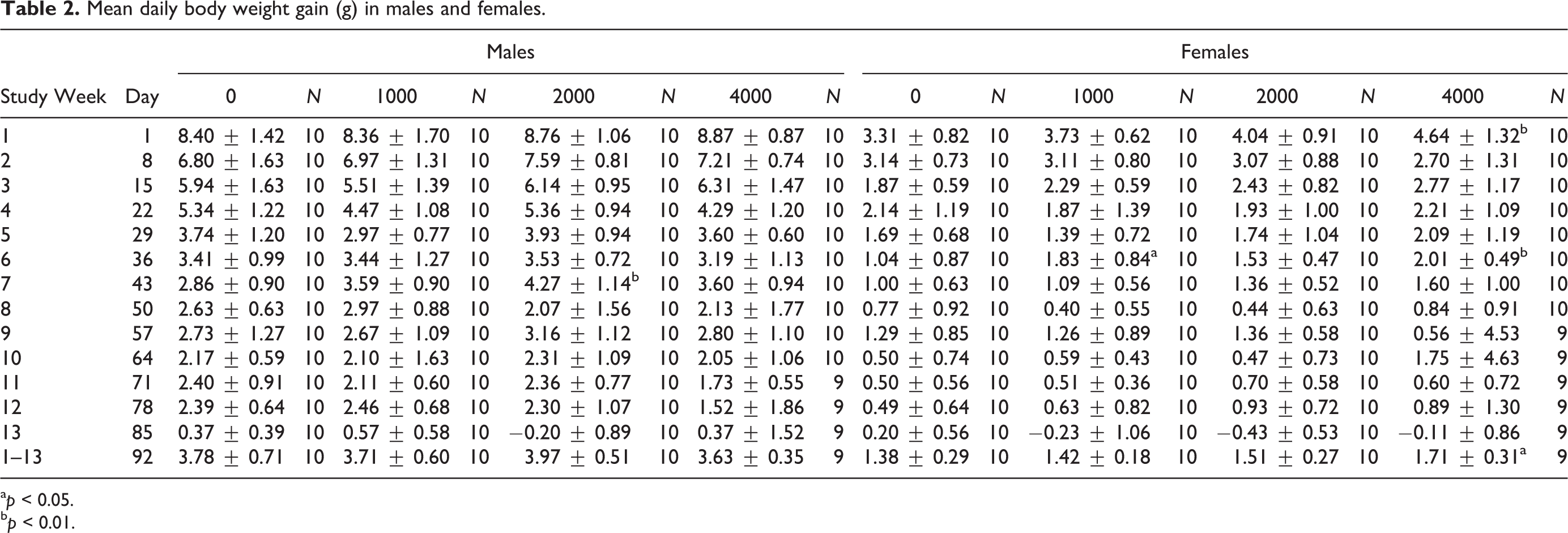
a p < 0.05.
b p < 0.01.
Similarly in female rats, for all intervals other than those noted as statistically significant by nonparametric ANOVA with post hoc comparison (high dose increased, week 7, p < 0.05), the mean body weights for the treated female rats were comparable with the control group values throughout the study (Figure 1). Mean daily body weight gain for the treated female rats was generally comparable with the control group values throughout the study (Table 2). The exception for the generally comparable body weight gain includes statistically significant increases in the low-dose group during week 6 ( p < 0.05) and high-dose females during weeks 1 and 6 as determined by one-way ANOVA main group effect (F(3, 39) = 3.467 and F(3, 39) = 3.763, p < 0.01 and p < 0.05), and study overall (weeks 1–13; (F(3,38) = 2.875, p < 0.05). In summary, there were no test substance-related changes in mean body weight or body weight gain in males or low- and mid-dose females. An overall increase in mean body weight gain for high-dose females was associated with a slight increase in body weight that, without other correlating toxicologically relevant findings, is interpreted to be of little toxicological significance and not adverse.
Mean daily feed consumption for treated male rats at dose levels of 1000, 2000, and 4000 mg/kg/day was comparable with the control group values throughout the study. For all intervals other than those noted as statistically significant, as determined by one-way ANOVA main group effect (mid and high dose, increased, week 7, F(3,39) = 4.201, p < 0.05–0.01) with post hoc comparisons, mean feed efficiency for the treated male rats was generally comparable with the control group values throughout the study (Table 3). Similarly, in females, feed consumption was comparable with the control group values throughout the study period (Table 3). Increases in mean feed efficiency in the low dose groups on week 6, as determined by one-way ANOVA main group effect (F(3,39) = 3.893, p < 0.05) and in the high dose females on weeks 1 and 6 (F(3,39) = 4.185 and F(3,39) = 3.893, p < 0.01), with post hoc comparison, and for the study overall (weeks 1–13; F(3,38) = 4.624, p < 0.01), with post hoc comparisons, were noted (Table 3). There were no test substance-related changes in mean feed consumption and feed efficiency in males or low- and mid-dose females. Increases in feed efficiency were observed in high-dose females. This change was not associated with a significant feed consumption increase but alternatively was attributed to an increase in body weight gain for the same intervals, and thus interpreted to be of little toxicological relevance and considered not adverse.
Mean daily food efficiency in males and females.
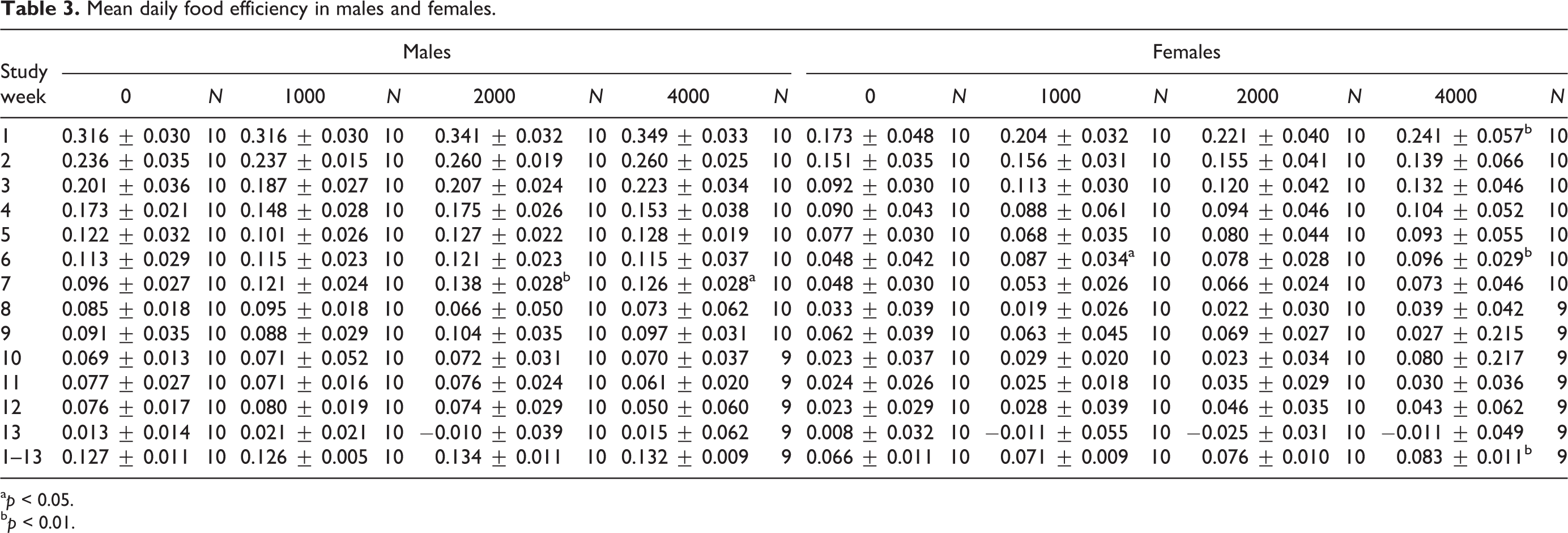
a p < 0.05.
b p < 0.01.
Clinical pathology
Hematology and coagulation parameters
There were no test substance-related effects of the LAPP treatment at dose levels of 1000, 2000, and 4000 mg/kg/day on hematology parameters in male and female rats (Table 4). A statistically significant decrease (p < 0.05) was observed in RCDW (measure of the range of variation of RBC volume) in male rats administered 4000 mg/kg/day of LAPP was not associated with concurrent RBC changes and was interpreted to be not toxicologically relevant. AMONs and ALUCs were increased (p < 0.05) in high-dose males. These minimal changes did not have correlating changes in WBC numbers or histopathological correlates and are interpreted to have no toxicological relevance.
Summary of mean hematology and coagulation values.
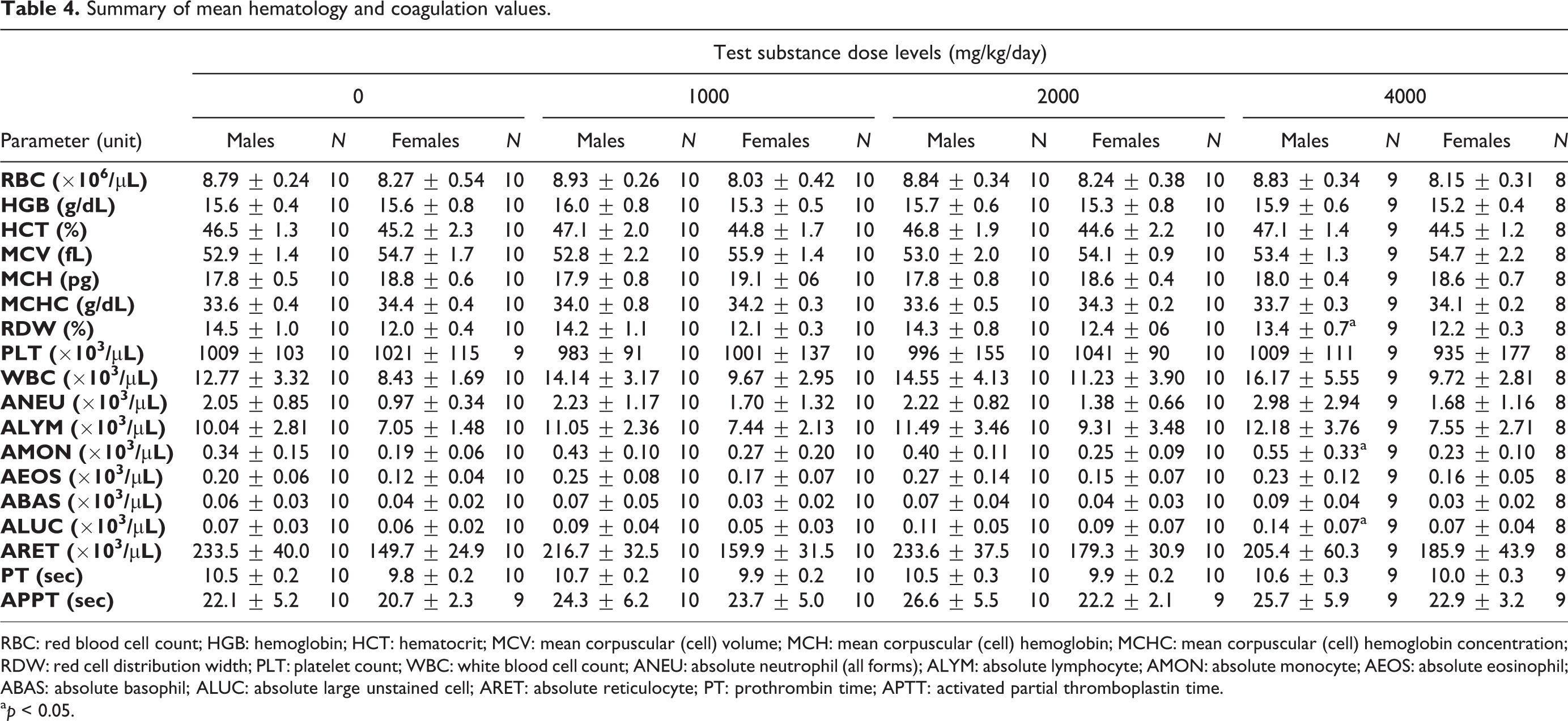
RBC: red blood cell count; HGB: hemoglobin; HCT: hematocrit; MCV: mean corpuscular (cell) volume; MCH: mean corpuscular (cell) hemoglobin; MCHC: mean corpuscular (cell) hemoglobin concentration; RDW: red cell distribution width; PLT: platelet count; WBC: white blood cell count; ANEU: absolute neutrophil (all forms); ALYM: absolute lymphocyte; AMON: absolute monocyte; AEOS: absolute eosinophil; ABAS: absolute basophil; ALUC: absolute large unstained cell; ARET: absolute reticulocyte; PT: prothrombin time; APTT: activated partial thromboplastin time.
a p < 0.05.
There were no test substance-related changes in coagulation values for male or female rats (Table 4). There were no statistical significant differences from control for PT and APTT parameters throughout the study.
Clinical chemistry
There were no clinical chemistry changes in male or female rats attributable to the administration of LAPP at dose levels of 1000, 2000, and 4000 mg/kg/day (Table 5). ALKP, BUN, and CHOL were increased (p < 0.05) and ALB was decreased (p < 0.05) in high-dose males treated at 4000 mg/kg/day. AST and ALKP were increased (p < 0.05) in high-dose females treated at 4000 mg/kg/day. All changes were of small magnitude, with values that were within PSL’s historical data ranges. Without associated microscopic changes in the liver or the kidney, any correlating serum chemistry or other toxicologically significant findings are interpreted to be of no toxicological significance. There were no test substance-related changes in CALC or phosphorous parameters. Other differences in clinical chemistry values, which were statistically significant, consisted of decreased potassium in low-dose males (p < 0.05) and increased ALKP (p < 0.05) in low-dose females. These were observed in a non-dose-dependent manner, they are within PSL historical data range values, and are interpreted to be within expected biological variation and not toxicologically relevant.
Summary of mean clinical chemistry values.a
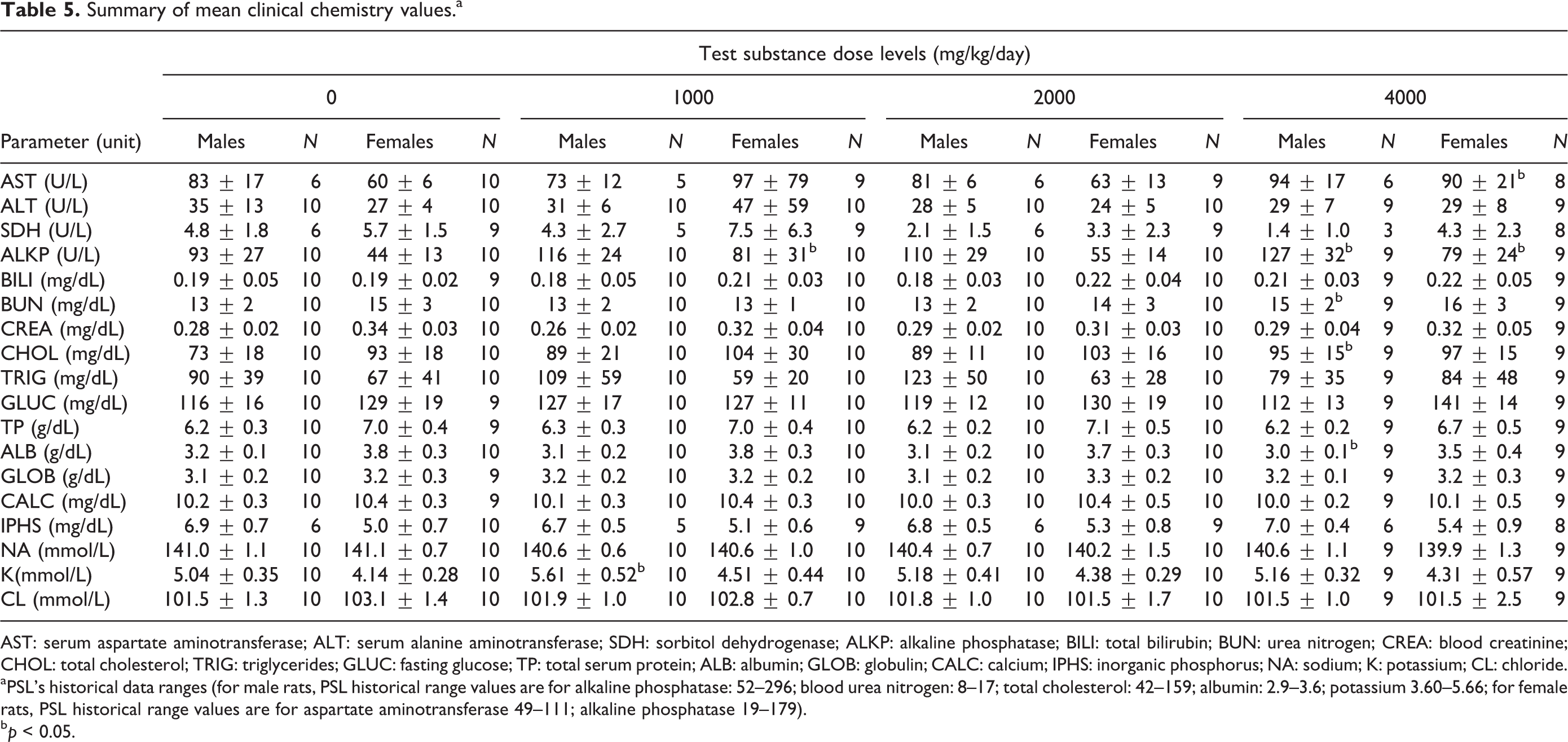
AST: serum aspartate aminotransferase; ALT: serum alanine aminotransferase; SDH: sorbitol dehydrogenase; ALKP: alkaline phosphatase; BILI: total bilirubin; BUN: urea nitrogen; CREA: blood creatinine; CHOL: total cholesterol; TRIG: triglycerides; GLUC: fasting glucose; TP: total serum protein; ALB: albumin; GLOB: globulin; CALC: calcium; IPHS: inorganic phosphorus; NA: sodium; K: potassium; CL: chloride.
aPSL’s historical data ranges (for male rats, PSL historical range values are for alkaline phosphatase: 52–296; blood urea nitrogen: 8–17; total cholesterol: 42–159; albumin: 2.9–3.6; potassium 3.60–5.66; for female rats, PSL historical range values are for aspartate aminotransferase 49–111; alkaline phosphatase 19–179).
b p < 0.05.
Urinalysis
There were no treatment-related effects on urinalysis parameters in male and female rats following administration of LAPP at dose levels of 1000, 2000, and 4000 mg/kg/day for 90 consecutive days. Urinalysis parameters did not reveal any statistically significant difference or physiological abnormalities in males and females in the control and treatment groups.
Organ weights
In male rats, absolute and relative organ weights for all treated males were comparable to control values with the exception of significantly decreased absolute brain weight in low-dose animals as determined by one-way ANOVA main group effect (F(3,38) = 3.329, p < 0.05) with post hoc comparison and increased relative spleen-to-body/brain weights for high dose animals as determined by one-way ANOVA main group effect (F(3,38) = 6.274, p < 0.01 and F(3,38) = 3.336, p < 0.05, respectively) with post hoc comparisons (Table 6). These changes did not have correlated relative or absolute weight changes, respectively (Tables 7 and 8). Additionally, they were not associated with histopathological findings and are interpreted to be of no toxicological significance.
Summary of mean terminal body weights and organ weights (g ± SD).
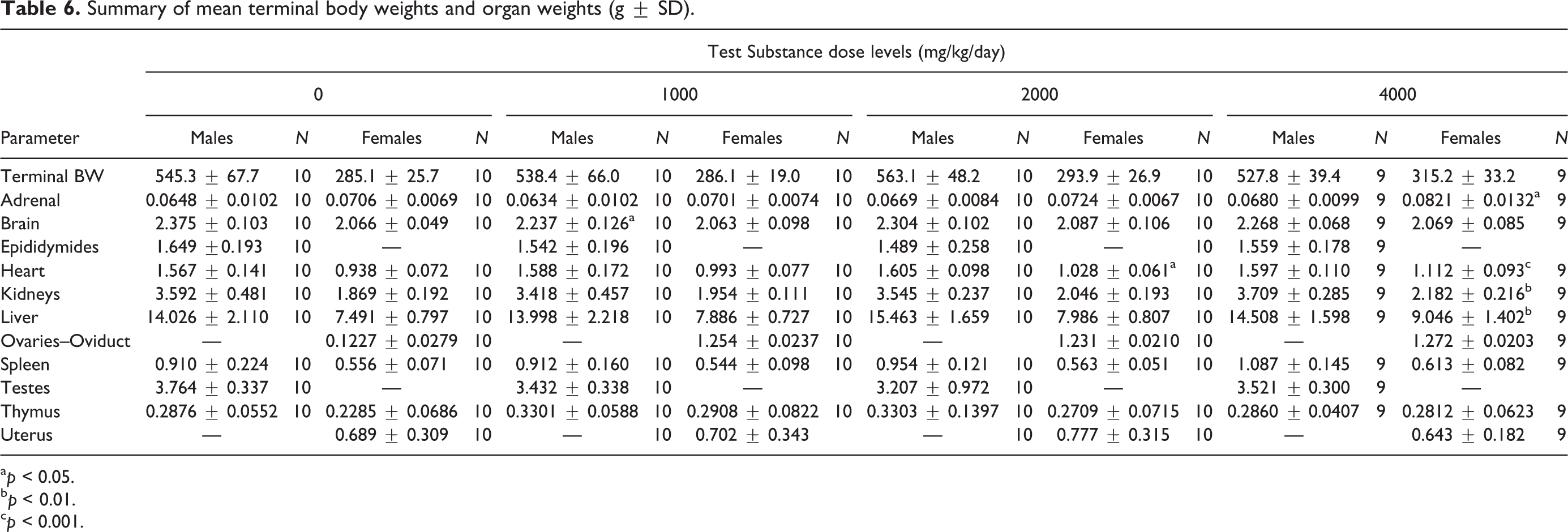
a p < 0.05.
b p < 0.01.
c p < 0.001.
Summary of mean organ-to-body weight ratios.
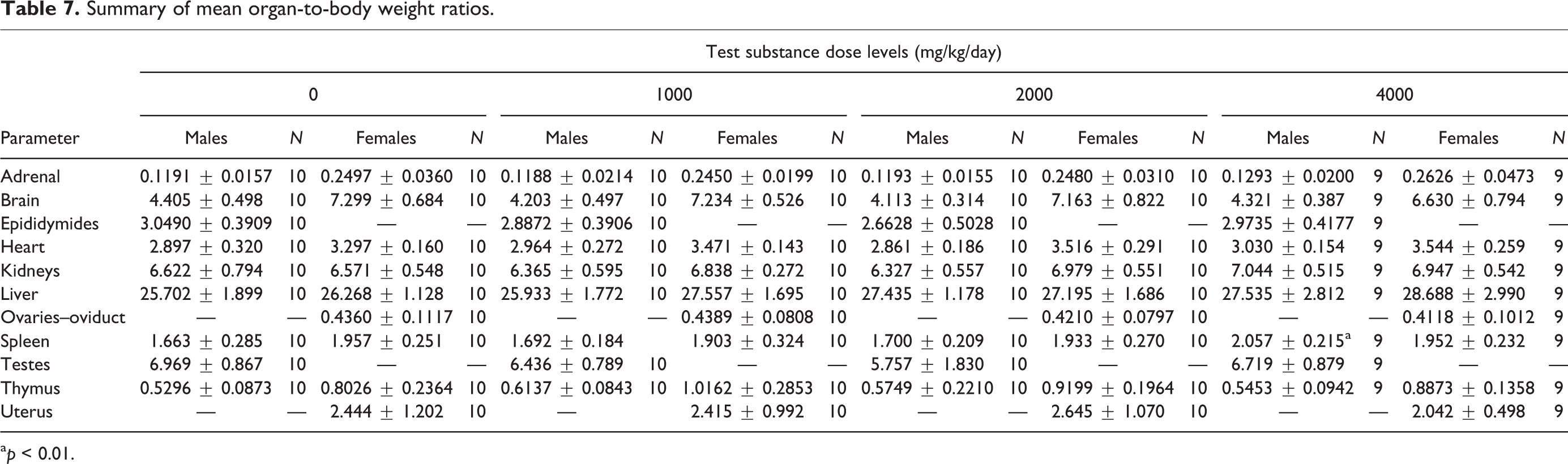
a p < 0.01.
Summary of mean organ-to-brain weight ratios.
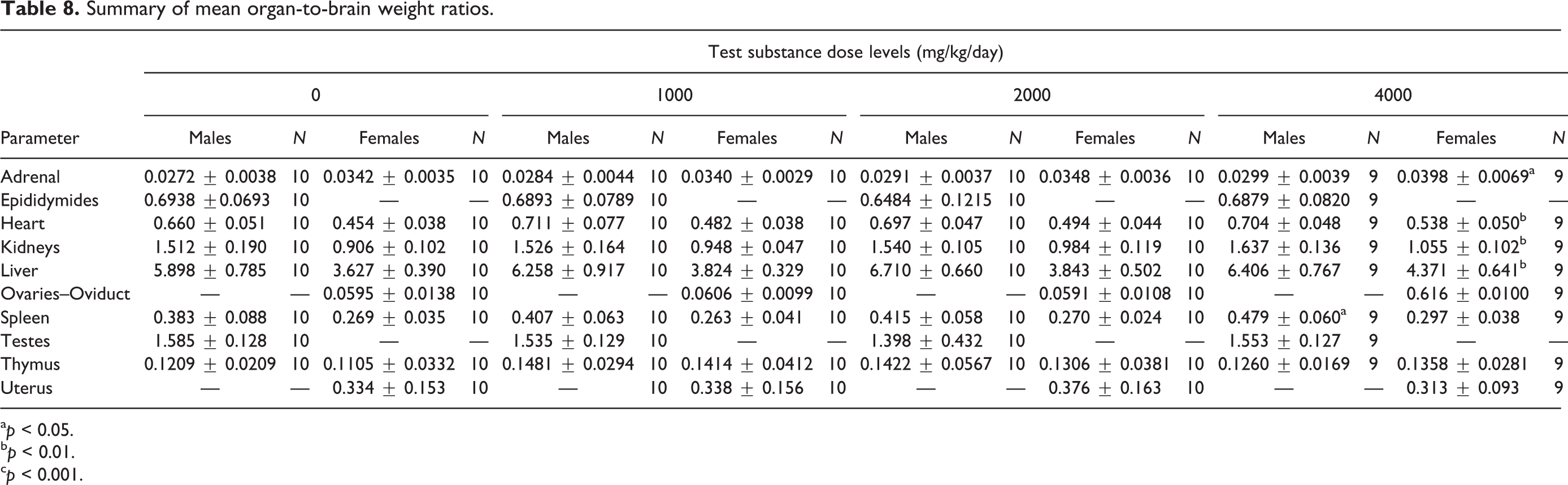
a p < 0.05.
b p < 0.01.
c p < 0.001.
In female rats, absolute heart weights for mid- and high-dose levels were increased as determined by one-way ANOVA main group effect (F(3,38) = 8.651, p < 0.05 and p < 0.001, respectively) with post hoc comparisons (Table 6). In addition, increases (p < 0.05–0.001) were noted in high-dose adrenal, kidney, and liver weights as determined by one-way ANOVA main group effect F(3,380 = 3.747, p < 0.05) and nonparametric ANOVA (p < 0.01) with post hoc analysis. All other absolute organ weights were comparable to controls. Relative adrenal-, heart-, kidney-, and liver-to-brain weights for high dose animals were statistically increased as determined by one-way ANOVA main group effect (F(3,38) = 3.395, p < 0.05; F(3,38) = 6.446, p < 0.001; F(3,38) = 4.119, p < 0.01; and F(3,38) = 4.181, p < 0.01, respectively) with post hoc comparisons (Tables 7 and 8). All other organ weights relative-to-body and to-brain weights for all treated females were comparable to control values. These increases in both absolute and relative-to-brain weight were not associated with concurrent increases in relative-to-body weight or any histological change in these organs but were concurrent with slight increases observed in terminal body weight and interpreted to be of little toxicological relevance and not adverse.
Macroscopic findings
At the scheduled necropsy, there were no treatment-related gross pathological observations in any of the groups of rats following administration of the LAPP, containing apoaequorin protein as the active ingredient. Among the changes noted, the uterus was noted as fluid-filled across all treated groups and control animals with similar incidence. Fluid-filled uteri usually corresponded microscopically to luminal dilation of the uterus, attributable to variation in the estrous cycle in individual animals, and this finding was not associated with test substance administration.
The remaining macroscopic observations were of sporadic incidence and showed no trends/patterns to suggest a relationship to administration of apoaequorin protein. These remaining macroscopic observations included small testes, which were noted affecting 1 of 10 males at 1000 mg/kg/day (unilaterally in one low-dose male) and 1 of 10 males at 2000 mg/kg/day (bilaterally in one mid-dose male) LAPP. Microscopically, these testicular findings correlated to atrophy of seminiferous tubules; this finding is a common spontaneous lesion among animals of this age and species and was not considered related to test substance administration. Small epididymides present in the same animals were due to severe oligospermia, consistent with the lack of sperm production from affected testes. The following gross observations did not have a microscopic correlate: low-dose female (uterus fluid filled), mid-dose male (seminal vesicles small, left), and mid-dose male (prostate soft).
Microscopic findings
The incidence and severity of histopathological findings following treatment with LAPP at dose levels of 1000, 2000, and 4000 mg/kg/day for 90-days were comparable between high-dose and control animals. All microscopic findings were unrelated to administration of the test substance and are known incidental lesions that can be observed in the age and strain of rats used in this study. 25 –27 Incidental histopathological findings observed consisted of sperm granulomas and oligospermia in the epididymides, minimal cardiomyopathy observed mostly in control animals; nephropathy; minimal to mild inflammation/inflammatory cell infiltrates, present in multiple organs; hepatic Kupffer cell hyperplasia; focal-pulmonary fibrosis and evidence of splenic hematopoiesis or lymph node lymphoid hyperplasia.
Several rats of both sexes (5 of 10 control males, 3 of 10 control females, 1 of 9 high dose males, and 5 of 9 high dose females) had minimal to mild inflammation of the larynx, usually observed in the ventral pouch (although both connective tissues and glands in the ventral larynx were involved). This lesion was interpreted as procedural, arising from trauma during gavage.
Discussion
The safety profile of apoaequorin is pivotal to its success as a dietary supplement. Since it is homologous with calcium-binding proteins that are also found in the human body, the low probably to cause adverse or allergic effects 10 is supported by both in vitro 4,9 and in vivo studies. In previous work, we reported a NOAEL of apoaequorin protein at 666.7 mg/kg/day after 90-day administration in Sprague-Dawley rodents. 9 A higher NOAEL at 2412 mg/kg/day for apoaequorin protein was established in this study, of similar duration in the same test system.
The results of the present repeat-dose animal toxicity study demonstrates that oral (gavage) administration of recombinant LAPP preparation at levels up to 4000 mg/kg/day (equivalent to 2412 mg of apoaequorin/kg/day) to male and female Sprague-Dawley rats for 90-days was not associated with adverse effects, as evaluated by the general condition and appearance of the animals, growth, feed consumption, clinical observations, ophthalmoscopy, hematology, clinical chemistry, urinalysis, organ weights, and histopathological findings.
Both cases of mortality observed in this study were indicative of incidental oral gavage trauma or accidental administration of test substance into the respiratory tract, and not a result of the test substance exposure and not impacted by its absorption rate/state. Incidents after oral gavage dosing of rodents in toxicity studies may be associated with unexpected respiratory symptoms and spontaneous death and may occur in single animals attributable to technical gavage errors. These incidents are not uncommon and have been reported in the literature. 28 All clinical signs in either male or female surviving animals were considered incidental and not uncommon in rodent toxicology studies. Because they were not observed to appear in a dose-related proportion, were isolated events, had no test substance-related histopathological or clinical pathological correlates, and were also observed with similar incidence in the control group animals, they were interpreted to be not toxicologically relevant.
In male rats, the mean weekly body weights and body weight gain were comparable with the control group values throughout the study. Increased mean body weight gain observed in mid-dose males was transient, not associated with a toxicologically significant increase in body weight, and is interpreted to be of no toxicological relevance. Feed consumption and mean feed efficiency for male rats were also generally comparable to control males. Statistically significant increases observed on week 7 for mid- and high-dose feed efficiency values were transient and without correlating relevant increases in body weight or body weight gain and are also interpreted to be of no toxicological relevance. Mean weekly body weight gain in high-dose females were statistically significant increased. This increase was associated with a very slight increase in body weight, that, without other correlating clinical or pathological toxicologically relevant findings, is interpreted to be of little toxicological significance and not adverse. Overall, mean daily feed consumption for female rats in all treated groups was comparable to control females throughout the study. Mean feed efficiency for female rats in low- and mid-dose groups was generally comparable to control females throughout the study, with the exception of a statistically significant increase on week 6 for low-dose females that was observed transiently and without a correlating relevant increase in body weight and is interpreted to be of no toxicological relevance. Statistically significant increases in feed efficiency observed in high-dose female rats were not associated with a significant feed consumption increase and were attributed to increased body weight gain for the same intervals, with little toxicological relevance.
There were no changes in clinical pathology parameters attributable to LAPP treatment. Statistically significant minimal changes observed in hematology parameters, such as decreased RDDW in high-dose male rats for the measure of the range of variation of RBC volume to supplement RBC and reticulocyte counts for adequate evaluation of RBC changes, or an increase in AMONs and ALSCs, failed to have correlates with erythrocyte or WBC numbers or histopathological correlates are interpreted to be within expected biological variability and have no toxicological relevance. Also, minimal statistically significant changes were observed for ALKP, BUN, CHOL (increased), and ALB (decreased) in high-dose males and AST and ALKP (increased) in high-dose females. All changes were of very minimal magnitude and were within PSL and published historical data. 16 They did not correlate with relevant clinical pathology parameters, such as changes in ALT (which are indicators of a hepatic test substance-related effect) or BUN (which may indicate a renal test substance-related effect). Additionally, there were no associated microscopic changes in the liver or the kidney that would support that any of these minimal changes have toxicological significance. Therefore, all changes are interpreted to be due to biological variation observed for this strain and age of rat and therefore have no toxicological relevance.
In general, organ weights in all treated male rats were comparable to control values. Statistical changes observed for absolute brain weight (decreased) in low-dose animals and relative spleen weights (increased) for high-dose animals need to be interpreted in light of the absence of a correlating relative and absolute concurrent change and the lack of association with histopathological findings. Taken together, these are interpreted to be of no toxicological significance. In females, high-dose increases in either absolute weight or relative-to-brain weight changes observed in heart, adrenal, kidney, and liver weights were not associated with concurrent increases in relative-to-body weight and are thus indicative of an effect secondary to the slight increase observed in the terminal body weight. Because there were no test substance-related histological changes in any of these organs, the differences noted are not considered adverse.
Macroscopic findings observed at necropsy were largely limited to the reproductive tract of female rats’ uterus that was noted as fluid filled and corresponded, in most cases, microscopically to luminal dilation of the uterus that was attributable to variation in the estrous cycle in individual animals. This finding was not associated with test substance administration. Additionally, changes observed in the testes were of sporadic incidence and showed no trends/patterns to suggest a relationship to administration of apoaequorin protein. Further incidental macroscopic observations consisted of a unilateral small seminal vesicles and a soft prostate. Inflammation of the larynx observed in both high-dose and control animals was usually observed in the ventral pouch to indicate its ventral location, although both connective tissues and glands in the ventral larynx were involved, and was interpreted as procedural, arising from trauma during gavage. Lesions in the larynx including ulcerations, squamous metaplasia, and/or erosions have been associated with technical gavage errors. 28
In summary, the results of present subchronic toxicity study suggest that oral administration of apoaequorin protein at levels up to 2412 mg/kg/day does not cause adverse effects in male and female Sprague-Dawley rats. The NOAEL for this study is established at 4000 mg/kg/day of LAPP, with a corresponding active dose of 2412 mg/kg/day for apoaequorin protein, the highest dose tested.
Footnotes
Authors’ note
OM and MRB are employees of Product Safety Labs, where the study was performed.
Acknowledgements
The authors are thankful to Quincy Bioscience, Madison, WI, USA, for supplying standardized LAPP preparation for use in this study and the financial resources. The authors would also like to acknowledge the Management and Celeste Dunn of Product Safety Labs, Dayton, NJ, USA, for designing, conducting, and reporting of findings in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
