Abstract
The Cosmetic Ingredient Review Expert Panel (the Panel) assessed the safety of nitrocellulose and collodion as used in cosmetics, concluding that these ingredients are safe in the present practices of use and concentration in cosmetic formulations. Both ingredients are used almost exclusively in nail product formulations. The maximum concentration of use of nitrocellulose in nail polish and enamels is 22%; for collodion, the maximum reported concentration of use in nail polish and enamel is 14%. The Panel reviewed available animal and clinical data in making its determination of safety.
Introduction
Nitrocellulose, a cellulose-based ingredient, is used in cosmetics, reportedly functioning as a dispersing agent—nonsurfactant and as a film former. Nitrocellulose is used almost exclusively in nail products. 1 Collodion, a cosmetic ingredient that is a solution of nitrocellulose dissolved in ethanol and ether and that is reported to function as a binder and a film former, is also addressed in this safety assessment.
Flexible collodion, defined by the International Cosmetic Ingredient Dictionary and Handbook as a mixture of collodion, camphor, and castor oil, is not included in this safety assessment.
Chemistry
Definition and Structure
According to the International Cosmetic Ingredient Dictionary and Handbook, nitrocellulose (CAS No. 9004-70-0) is defined as a cellulose derivative that conforms generally to the formula C12H16N4O18, 1 which equates to an average of 2 nitrate groups per mono-sugar repeat unit (see Figure 1). Collodion (CAS No. 9004-70-0) is defined as a solution of pyroxylin (chiefly nitrocellulose) that contains approximately 6% pyroxylin, 24% ethanol, and 70% ether. 1 The Merck Index states that pyroxylin is a variable mixture that consists primarily of cellulose tetranitrate, which also equates to an average of 2 nitrate groups per mono-sugar repeat unit. 2 Based on these definitions, collodion is simply a preformulation solution of nitrocellulose.
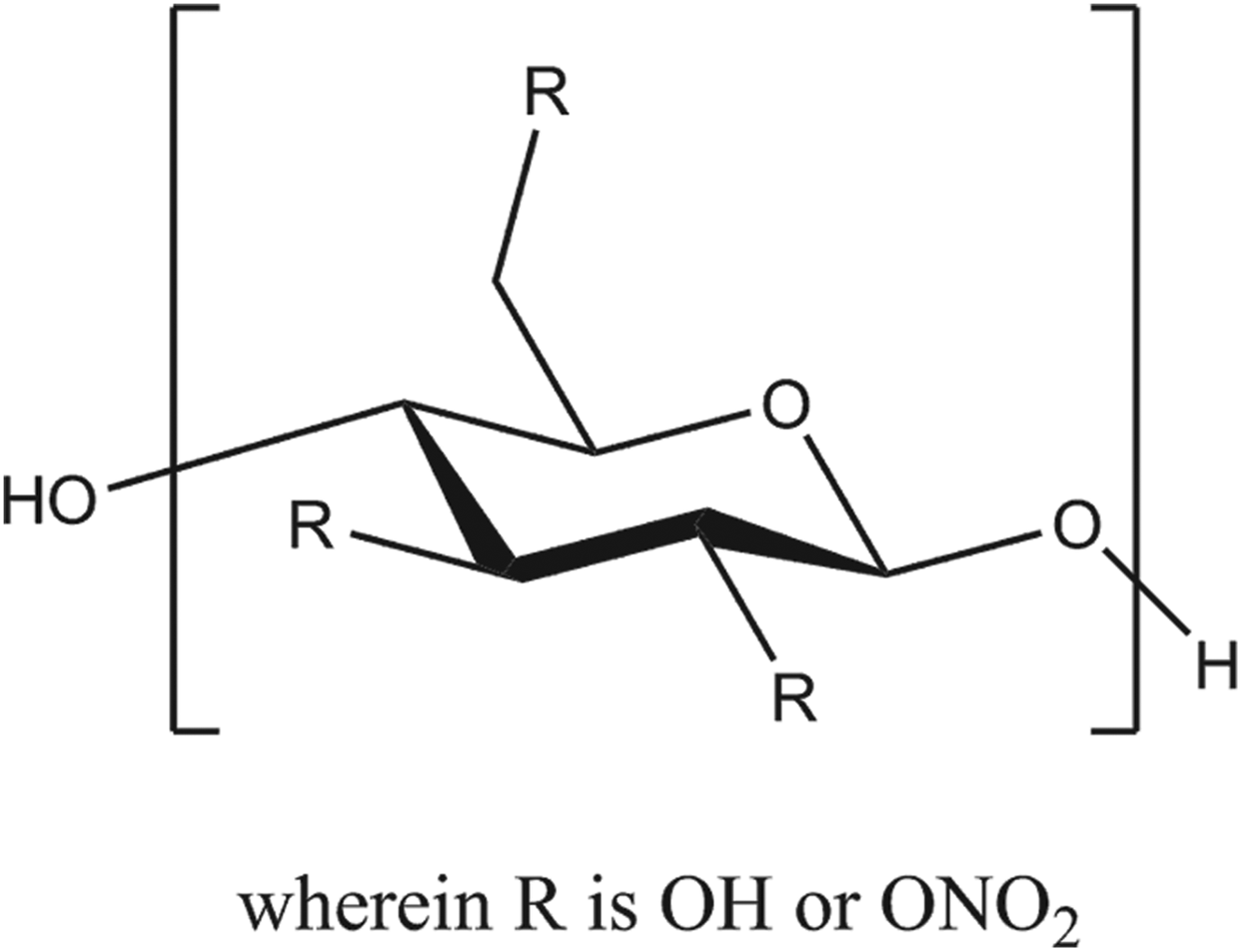
Nitrocellulose.
One source states nitrocellulose is a mixture of polymeric nitrate esters and that 3 types of nitrocellulose are recognized (1) collodion (or pyroxylin), with 8% to 12% nitrogen, (2) pyrocellulose, with 12.6% nitrogen, and (3) guncotton, with a minimum of 13.35% nitrogen. 3 Each name, under each method of nomenclature, relates to a chemical with a cellulose backbone and some degree of nitration (ie, how many R groups in Figure 1 are ONO2 vs OH).
Chemical and Physical Properties
Table 1 lists the data on available chemical and physical properties. Nitrocellulose is a white, amorphous solid that is virtually insoluble in water. When dissolved in an organic solvent (collodion is an example of solubilized nitrocellulose), the solution is clear, either colorless or slightly yellow (the yellowness is the result of decomposition that can occur from heat or ultraviolet irradiation), and precipitates nitrocellulose when mixed with water.
Chemical and Physical Properties.

Abbreviation: UV, ultraviolet.
Method of Manufacture
The nitrocellulose used in nail formulations is a nitrate ester obtained by the reaction of a mixture of nitric and sulfuric acids with an alcohol (ie, 1 of the hydroxyl groups on the cellulose backbone). 4
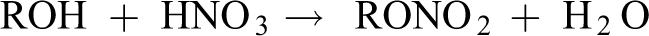
The cellulose used for this reaction is a natural product derived from wood or cotton, wherein the cellulose polymer is a chain of mono-sugar repeat units, each with 3 free hydroxyl groups. The degree of nitration affects some physical properties of this polymer, making it more or less useful for different industries. The formula recited in the International Cosmetic Ingredient Dictionary and Handbook suggests that only 2 hydroxyls are typically esterified with nitro groups, per monomer residue, in the cosmetic ingredient.
For military use, cotton linters or wood pulp are treated with mixed nitric acid and sulfuric acid at 30°C. 3 The resulting slurry is centrifuged to remove most of the acid, treated with boiling water, washed with a heavy stream of water, and then screened to remove most of the water. Mechanistically, this is no different than the method above but likely involves increased equivalents of nitric and sulfuric acids to result in a higher degree of nitration (which comports with the military use as a propellant). No data were available specifically on the method of manufacture of collodion.
Impurities
Published impurity data were not found for either ingredient.
Use
Cosmetic
Nitrocellulose is widely used in nail-polish formulations and is reported to function in cosmetics as a dispersing agent—nonsurfactant and as a film former. 1 Collodion is reported to function as a binder and a film former. The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2013 reported that nitrocellulose is used in 516 nail product formulations and 1 “other” makeup formulation 5 (Table 2). Nitrocellulose is used in 410 of 489 nail-polish and enamel formulations and 67 of 79 basecoat and undercoat formulations reported in the VCRP, as shown in Table 3. The results of a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (the Council) report that nitrocellulose is used at concentrations up to 41% in other manicuring preparations, namely, in nail “stickers” made from dried nail polish. 6 The reported maximum concentration of nitrocellulose use in nail polish and enamels is 22%.
Frequency and Concentration of Use According to Duration and Type of Exposure.
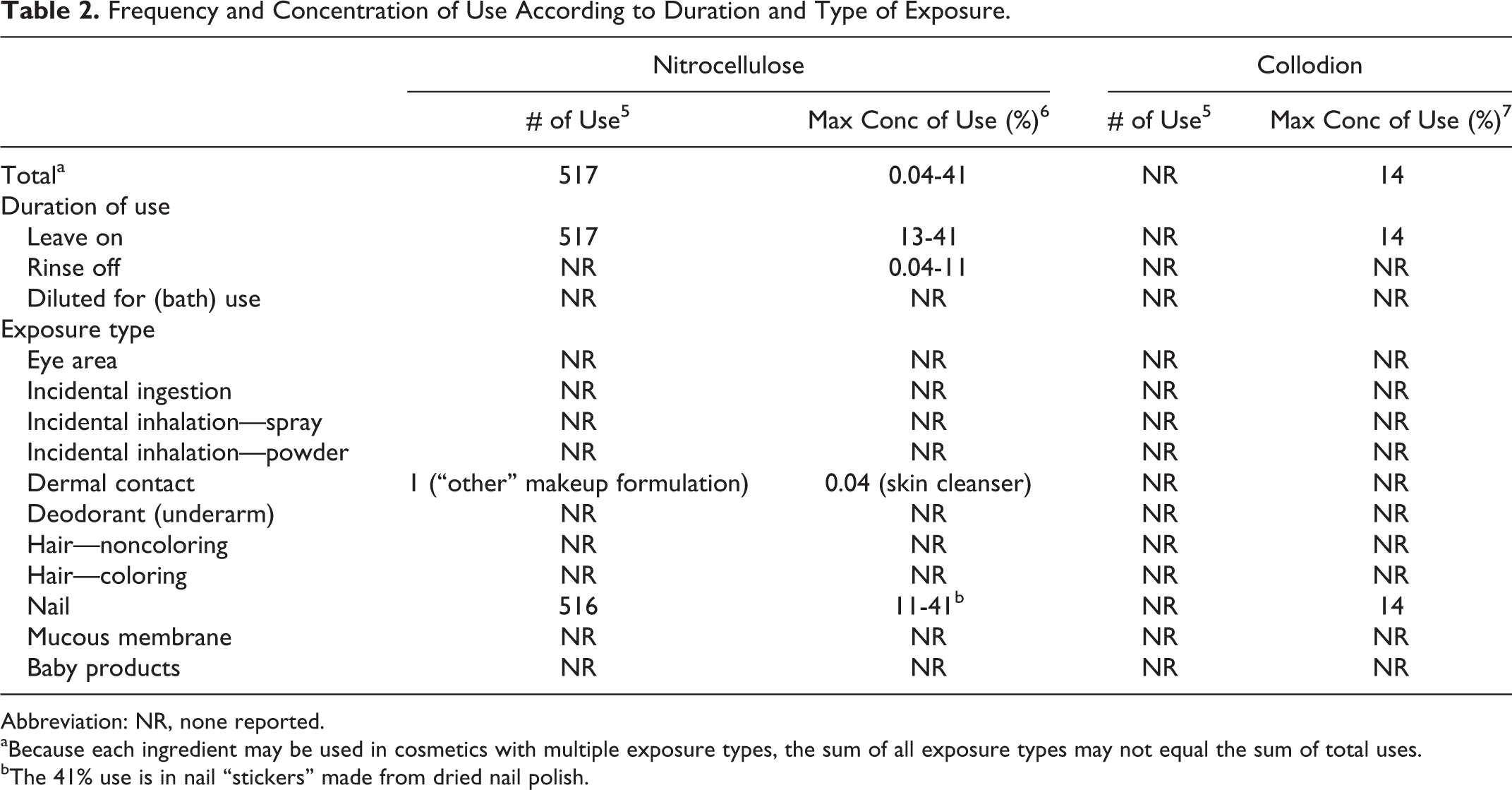
Abbreviation: NR, none reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bThe 41% use is in nail “stickers” made from dried nail polish.
Use of Nitrocellulose in Nail Products by Individual Category.
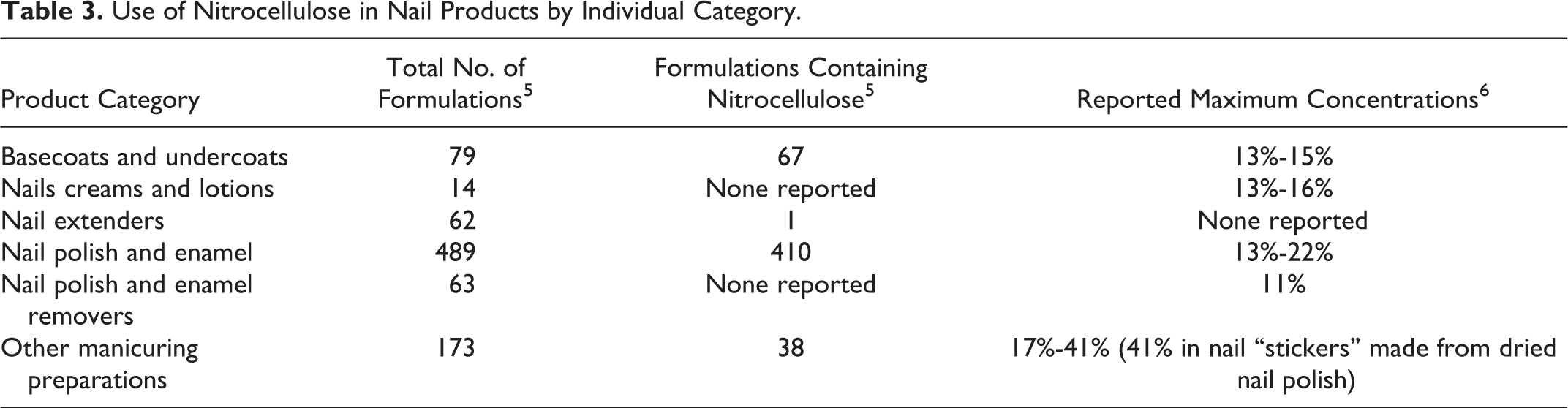
Collodion is not reported to be used by the VCRP. 5 However, it should be presumed that there is at least 1 use of collodion because, according to a concentration of use survey conducted by the Council, collodion is reported to have a maximum concentration of use of 14% in nail polish and enamel. 7
Noncosmetic Use
Nitrocellulose is a prior-sanctioned food ingredient for use in the manufacture of paper and paperboard products used in food packaging (21CFR181.30). Nitrocellulose is also an approved indirect food additive (21CFR175.105, 21CFR175.300, 21CFR176.170, and 21CFR177.1200). Nitrocellulose centrifugation tubes are used in biological work for high-speed centrifugation, 8 and nitrocellulose membranes are used in protein blotting. 9 Nitrocellulose is used for coating slides for embedding media; nitrocellulose slides are useful when mounted sections are immersed in alkalis or hot acids. 10 Another use of nitrocellulose is as a binder in printing inks and wood coatings. 11
Nitrocellulose is a principal ingredient of propellants, smokeless powder, rocket fuel, ball powder, mortar increment, and some explosives. 3 However, these incendiary uses of nitrocellulose are likely related to a polymer with a higher degree of nitration than that of the cosmetic ingredient and would be better classified as pyrocellulose or guncotton.
Corn and callus remover products containing salicylic acid at 12% to 17.6% in a collodion-like vehicle are generally recognized as safe and effective by the FDA for topical application as an over-the-counter drug (21 CFR 358.503). The collodion-like vehicle is described as a solution containing pyroxylin (nitrocellulose) in an appropriate nonaqueous solvent that leaves a transparent cohesive film when applied to the skin as a thin layer. That nonaqueous vehicle would appear to be alcohol and ether, which would evaporate rapidly, leaving the pyroxylin (nitrocellulose).
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Oral
Nitrocellulose (containing 12.9% nitrogen by wt) was not absorbed by rats following oral dosing. 12 One fasted male CD rat was dosed by gavage with 1 mL/100 g (∼20,000 dpm/mL) aqueous [14C]nitrocellulose and another with [14C]nitrocellulose suspended in 0.2% methyl cellulose—0.4% Tween 80; each rat was dosed for 4 days. The labeled compound was prepared by nitrating [14C] cotton. For the aqueous dose, the fiber was cut and ground and then concentrated by sedimentation. Only fibers small enough to go through an 18-gauge dosing needle were used. The animals were killed 24 hours after the last dose. Radioactivity was recovered only in the gastrointestinal tract and in the feces. No detectable radioactivity was found in any other tissues or body fluids.
A mass-balance metabolism study was performed in which 1 Beagle dog was fed 90 g wet nitrocellulose (27.9 g on a dry basis). 13 The nitrocellulose contained 13.08% nitrogen. Feces were collected every 24 hours for 4 days. Over a 4-day period, 9.5 g of nitrocellulose (34% of the dose) were recovered; 8.8 g were recovered after 24 hours and 0.7 g after 48 hours.
Toxicological Studies
Single-Dose (Acute) Toxicity
Oral
The oral LD50 of a 5% nitrocellulose suspension was >5,000 mg/kg in mice and rats. 14 The tested material was a military-produced nitrocellulose with a nitrogen content of 13.1%. (This test article is used in multiple studies and will be described simply as 13.1% nitrogen, 65.6% >88 μm). Fasted male and female albino Swiss mice and male and female CD rats were dosed by gavage with 5,000 mg/kg of 5% ground nitrocellulose in water; the dose was administered in 2 portions 30 minutes apart, due to the large volume. (The number of female mice and male and female rats dosed was not specified). Two of 10 male mice died; no gross lesions were observed.
The LD50 of a nitrocellulose-based propellant was >5,000 mg/kg in mice and rats upon oral administration. The test substance (ball powder) is composed of 83.23% nitrocellulose, 10.235% nitroglycerin, 0.685% dinitrotoluene, 1.105% diphenylamine, 5.255% dibutyl phthalate, 1.045% total volatiles, 0.895% moisture and volatiles, 0.49% residual solvent, 0.09% calcium carbonate, and 0.12% sodium sulfate. Ten male and 10 female ICR mice 15 and 7 male and 7 female Sprague-Dawley rats 16 were dosed by gavage with 5,000 mg/kg test article in 1% carboxymethylcellulose. Five male and 5 female mice and 5 male and 5 female rats served as controls and were dosed with 10 mL/kg of vehicle only. None of the mice or the rats died during the study.
Repeated-Dose Toxicity
Oral
In the 13-week repeated-dose dietary studies in mice, rats, and dogs, administration of 1% and 3% nitrocellulose (nitrogen content, 13.1%) in feed (calculated on a dry basis) had no adverse effects; effects seen at 10% were attributed to the fiber content and not the chemical nature of the test article. 12 For each species, a “cotton control” group was fed a diet containing 10% cotton linters and a negative control group was given untreated feed. Also, the reversibility of toxic effects was evaluated by killing half of the animals at the termination of dosing, that is, at 13 weeks, and the remainder after a 4-week recovery period, that is, 17 weeks.
Groups of 8 male and 8 female albino Swiss mice were used. By the end of week 2, 4 cotton control males, 4 cotton control females, 1 low-dose male, and 6 high-dose male mice died; the deaths of the high-dose and cotton-control animals were attributed to intestinal impaction of the fibers. Body weights of mice in the low- and mid-dose groups were similar to the negative controls; severe weight loss was reported for the high-dose animals during week 1 of the study. Feed consumption increased slightly in the low- and mid-dose group and considerably in the high-dose group; however, mice of the high-dose group scattered much of the feed. The absolute and/or relative spleen weights of mice in the 10% group and in the cotton control group killed at 13 weeks were statistically significantly decreased compared to controls; at 17 weeks, the spleen weights were normal. No adverse effects due to the chemical nature of nitrocellulose were observed and no test article–related gross or microscopic lesions, changes in hematological parameters, or alterations in serum immunoglobulin E (IgE) concentrations were found.
Groups of 8 male and 8 female CD rats were fed the test or control diets. Four animals/sex/group were killed at 13 weeks and the remainder at 17 weeks. Blood samples were taken at 0, 4, 8, 13, and 17 weeks. No adverse effects were observed. Because no adverse effects were observed and test article–related lesions were not found during the 13-week necropsy, a 17-week necropsy and blood analysis were not performed. Body weight gains by rats in the 1% and 3% groups were similar to the untreated controls. Body weight gains by rats in the 10% group and the cotton control group were decreased compared to the untreated controls; body weights by the recovery animals in the 10% group, but not the cotton control group, approached those of the negative control animals. Rats in the test groups had increased feed consumption with increased dose. In the low- and mid-dose groups, increased feed consumption was attributed to compensation for the nonnutritive fiber; the high-dose animals scattered their feed. Decreases in liver, kidney, and/or spleen weights in male rats of the high-dose group were attributed to decreased body weight gain. No adverse effects due to the chemical nature of nitrocellulose were observed, and no test article–related gross or microscopic lesions or changes in hematological or clinical chemistry parameters were observed
Two male and 2 female Beagle dogs per group received treated feed for 13 weeks. One male and 1 female from each group were killed at 13 weeks, and the remaining 2 animals were killed at 17 weeks. Blood samples were taken prior to dosing and at 4, 8, 13, and 17 weeks. No adverse effects were observed. As with the rats, because no adverse effects were observed and no test article–related lesions were found during the 13-week necropsy, the 17-week necropsy and blood analysis were not performed. No test article–related changes in weight were observed. Feed consumption was greater in all test animals than in controls; again, this was attributed to nonnutritive bulk. Dietary administration of nitrocellulose did not cause gross or microscopic lesions or changes in organ weights, hematological parameters, or serum IgE concentration in dogs.
Nitrocellulose was also not toxic in the 2-year dietary studies in mice, rats, and dogs; effects due to fiber content were similar to the 13-week studies described previously. Each species was fed a diet containing 1%, 3%, and 10% nitrocellulose (calculated on a dry basis); as in the 13-week studies, a cotton control group was fed a diet containing 10% cotton linters and a negative control group was given untreated feed. 13 Also in each species, there were 4 subgroups: 1 subgroup was killed after 12 months of dosing, 1 was started on an untreated recovery diet at 12 months and killed at 13 months, 1 subgroup was killed after 24 months of dosing, and 1 was started on a recovery diet at 24 months and killed at 25 months. Details for each species follow.
In the mouse study, 58 male and 58 female CD-1 mice per group were used at study initiation. Four mice/sex/group were killed for the interim 12-month necropsy and for the 13-month recovery group; with the exception of 4 mice/sex/group that were used as the 25-month recovery group, all remaining animals were killed at 24 months. Blood samples were taken at necropsy from the animals killed at 12 months and from 8 mice/sex/group killed at 24 months. During the first 3 weeks of the study, 9 males and 5 females of the 10% nitrocellulose group and 5 males and 1 female of the cotton control group died due to intestinal blockage by the fibers. Additional mice were added to these groups. Also, around month 9 of the study, a number of high-dose and cotton control animals died; no explanation for this cluster of deaths was found. The researchers did state, however, that because there were 3 times more deaths among the high-dose animals than the cotton control mice, the presence of a compound-related effect could not be dismissed. Hyperemia was observed in a number of the cotton controls and some of the 10% nitrocellulose animals; the researchers did not have an explanation for this observation but did hypothesize that it may have been an irritation reaction to the fibers the mice pulled from the feed. Body weight gains by animals in the 10% nitrocellulose and cotton control groups were initially decreased compared to controls. As the study progressed, a dose-relationship for increased feed consumption was observed in the control, low-, and mid-dose groups. Feed scattering in the high-dose group and the cotton controls made it difficult to quantitate actual feed consumption. Other than a treatment-related statistically significant lack of bronchoalveolar carcinomas in high-dose male mice, no test article–related gross or microscopic lesions or changes in organ weights were reported. No effects on clinical chemistry or hematology parameters were found.
For rats, groups of 32 male and 32 female CD rats were used at study initiation. An additional 8/sex/group were added after 6 months; 4 of the 8/sex/group were killed for the interim 12-month necropsy and the other 4 as the 13-month recovery group. With the exception of 4 rats/sex/group that were used as the 25-month recovery group, all remaining animals were killed at 24 months. Blood samples were taken from 4 rats/sex/group at 0, 6, 12, 18, and 24 months. Blood samples were also taken at necropsy from the animals killed at 12 months and from 8 rats/sex/group killed at 24 months. Test article–related toxic effects were not observed. Body weight gains of high-dose and cotton control animals initially were decreased compared to controls; weight gains in these groups were increased, becoming closer to control values, later in the study. Tumors not related to dosing were observed in all groups. A dose-related increase in feed consumption was attributed to the nonnutritive bulk of the fibers; scattering was also observed. No test article–related gross or microscopic lesions or changes in organ weights were found, and there was no effect on clinical chemistry or hematology parameters.
Groups of 6 male and 6 female Beagle dogs were used in the 2-year study. One test animal/sex/group was killed at 12 mos, one recovery animal/sex/group was killed at 13 mos, 2 test animals/sex/group were killed at 24 mos and the remaining 2 recovery animals/sex/group were killed at 25-mos. Blood samples were taken from all dogs at 0, 3, 9, 12, 18, and 24 months. No signs of toxicity were observed. Dose-related differences in body weights were not found. Animals of the 10% nitrocellulose group and the cotton control group had increased feed consumption; this difference was not considered to be toxicologically significant. Test article–related changes in clinical chemistry and hematology parameters or organ weights were not found nor were any test article–related gross or microscopic lesions. Because no changes were found in the animals after 12 or 24 months, the respective recovery groups were not necropsied.
Ocular Irritation
A 33% aqueous solution of nitrocellulose (nitrogen content, 13.1%) was not a primary irritant in rabbit eyes. 14 A modified Draize test was performed in 6 New Zealand White (NZW) rabbits, and the eyes were evaluated for irritation at 24 and 72 hours.
Ball powder (defined previously) was not a primary ocular irritant in rabbits in a modified Draize test. 17 The test material, 0.113 g, was instilled into the lower conjunctival sac of 1 eye of each of the 6 male NZW rabbits, and the eye was not rinsed. The test material, a 0.5 to 1.5 mm spheroidal pellet, was administered neat. The contralateral eye served as the untreated control. The eyes were graded at 1, 4, 24, 48, and 72 hours after dosing; fluorescein staining was used at 24, 48, and 72 hours. Significant amounts of the test article were present in the conjunctival sac of each eye at 1 and 4 hours after instillation. Small pinpoint erosions of the cornea were present in 2 animals at 24, 48, and 72 hours. A small corneal erosion in 1 animal on day 7 of dosing was considered incidental. Slight conjunctival vasodilation (all animals at 4 hours; score of 1) and chemosis (3 animals at 4 hours; score of 1), indicative of mild inflammation, were observed. Ball powder produced minimal irritation.
Reproductive and Developmental Toxicity
Oral
Nitrocellulose did not cause reproductive or developmental toxicity. In a 3-generation reproduction study, the F0 generation, consisting of groups of 10 male rats (from the 2-year study described previously) and 20 female rats, was fed a diet containing 1%, 3%, or 10% nitrocellulose (calculated on a dry basis; nitrogen content, 13.1%). 13 A cotton control group was fed a diet containing 10% cotton linters and a negative control group was given untreated feed. The rats were mated after 6 months of dosing. The initial offspring (ie, the F1a generation) were killed at weaning. The animals of the F0 generation were mated a second time, and 20 to 24 pups/sex (i.e., the F1b generation) were retained at weaning. In all 10 to 12 pairs of F1b animals were mated within their dose group at 3 months of age. As before, the F2a generation was discarded and the F2b rats were retained. The mating procedure was repeated with the F2b rats, and the study was terminated upon weaning of the F3b rats.
At the time of the first matings for males of all parental generations, the mean body weights of the 10% nitrocellulose group and the cotton control group were statistically significantly decreased compared to the controls. In females, the mean body weights were only decreased in the cotton control group. As in other repeated-dose studies, feed consumption in the 10% nitrocellulose and cotton control groups was increased. Statistically significant decreases in the lactation index and pup weight at weaning were observed in the 10% nitrocellulose and the cotton control group; these decreases were primarily observed in the F1b through F2b litters and attributed to a lack of parental nutrition caused by the inert increased bulk in this feed. Fertility was not affected by dosing, and no test article–related effects were seen on reproductive indices.
Genotoxicity
In Vitro
Nitrocellulose (13.1% nitrogen; 65.6% >88 μm) was not mutagenic in the Ames test. 18 Nitrocellulose, prepared as a suspension in 1 to 5 mg/mL distilled water, was evaluated at concentrations of 100, 1,000, and 5,000 μg/plate in Salmonella typhimurium TA1535, TA1537, TA1538, TA98, and TA100 with and without metabolic activation.
In Vivo
Nitrocellulose was not genotoxic in cytogenetic assays. The cytogenetic effect of nitrocellulose was evaluated in lymphocytes and kidney cells from CD rats fed a diet containing 10% nitrocellulose in the previously described 13-week study 12 and in lymphocytes and bone marrow cells from rats fed a diet containing 1%, 3%, or 10% nitrocellulose in the previously described 1- and 2-year studies, respectively. 13 Kidney cells from rats in the long-term study were also analyzed. Nitrocellulose did not produce chromosomal aberrations in any of these cells.
Carcinogenicity
No statistically significant increase in tumors due to the administration of nitrocellulose were reported in the previously described 2-year dietary studies in mice, rats, or dogs when compared to the respective controls. 13
Epidemiology
A matched case–control study nested in a retrospective cohort study examined mortality among workers in a plastics producing plant located in Springfield, Massachusetts. 19 The retrospective cohort study identified 2,490 male workers who were employed a minimum of 1 year between January 1949 and December 1966, and mortality was examined from January 1950 to December 1976. In the case–control study, each case was matched with 4 controls by race and age (series 1 controls) or by race, age, and date of hire (series 2 controls); and in each case of digestive cancer, series 2 controls were also matched with the place of birth. Using series 1 controls, the odds ratio of digestive system cancers (combined) was slightly but not statistically significantly increased for cellulose nitrate production workers (ie, using cellulose nitrate, ethyl alcohol, and camphor) and for cellulose nitrate processing workers (ie, exposed to finished cellulose nitrate). Although statistical significance was not reached, the odds ratio for cellulose nitrate processing increased with increasing exposure times; odds ratios of 1.07, 1.91, and 2.85 were reported for exposures of 1 month, 5 years, and 10 years, respectively. For individual digestive cancers, the only statistically significant increase in odds ratio was that of an odds ratio of 8.90 (P < 0.05) for rectal cancer in cellulose nitrate production workers exposed for >5 years; however, the researchers stated that because very little was known about their other chemical exposures, a reliable interpretation of these data could not be made. Findings using series 2 controls were consistent with those found using series 1 controls. In examining the incidence of genitourinary system cancers, the odds ratios for cellulose nitrate production and for cellulose nitrate processing were not increased.
A population-based case–control study was performed using 497 male workers (various occupations) from Montreal, who had histologically confirmed cases of colon cancer that were diagnosed between 1979 and 1985; 1,514 cancer controls (with cancers at other sites) and 533 population-based controls were used. 20 Face-to-face in-depth interviews using a structured questionnaire for numerous possible confounders and a semistructured questionnaire regarding job details were performed. Concentration of exposure was assessed on a relative scale of low, medium, or high exposure. When the odds ratio was adjusted for age and other nonoccupational risk factors, there were 9 cases of colon cancer in workers with substantial exposure to nitrocellulose, and the nonoccupationally adjusted odds ratio was 2.6 with a 95% confidence interval of 1.0 to 6.4. With nonsubstantial exposure to cellulose nitrate, there were 5 cases of colon cancer and the nonoccupationally adjusted odds ratio was 0.5 with a 95% confidence interval of 0.2 to 1.3. When the odds ratio was adjusted for nonoccupational risk factors and occupational exposure, the fully adjusted odds ratio in the 9 cases of colon cancer with substantial exposure to cellulose nitrate was 2.8 and the 95% confidence interval was 1.1 to 7.5. With nonsubstantial exposure to cellulose nitrate, the fully adjusted odds ratio was 0.4 with a 95% confidence interval of 0.1 to 1.2 for the 5 cases of colon cancer. The researchers stated that although the relative risk was significantly high with substantial exposure to cellulose nitrate, a number of associations with occupational substances had less than 10 colon cancer cases with substantial exposure, so there was considerable statistical variability associated with the estimate of relative risk.
Irritation and Sensitization
Nonhuman
A 33% aqueous solution of nitrocellulose (13.1% nitrogen; 65.6% >88 μm) was not a primary skin irritant in rabbits. 14 A modified Draize test was performed in 6 NZW rabbits, and the test solution was applied to intact and abraded skin. The primary irritation score was <0.2.
Ball powder (defined previously) was not irritating in rabbits in a modified Draize irritation test. 21 Two occlusive patches, 1-inch each, containing a thick paste of 0.5 g of the test article in approximately 0.5 mL isotonic saline were applied for 4 hours to the clipped skin on the back of 4 male and 4 female NZW rabbits. Sham and vehicle controls were used. The test sites were wiped with saline upon removal and scored for erythema and edema at 30 and 60 minutes and 24, 48, and 72 hours after patch removal. Very slight erythema (score of 1) was observed at the test and the control sites in some of the rabbits; therefore, the test product had a peak net mean score of 0 and was classified as a nonirritant.
Human
A nail enamel containing 10.5% nitrocellulose was not a primary irritant in a 48-hour patch test completed in 51 participants. 22 Semiocclusive patches, 1” × 1” in size, containing approximately 0.2 mL of the test material were applied to the back of each participant for 48 hours. The test site was evaluated upon patch removal and 24 hour later. No signs of irritation were observed in any of the participants.
A nail lacquer containing 8.85% nitrocellulose was not an irritant or a sensitizer in a human-repeated insult patch test (HRIPT) completed in 108 participants. 23 The nail lacquer was applied to semiocclusive patches as received and was allowed to dry prior to application. Patches were applied to the upper back for 24 hours 3 times/week for 3 weeks, for a total of 9 induction applications. The tests sites were scored 24 or 48 hours after patch removal. Following a 2-week nontreatment period, a 24-hour challenge patch was applied to a previously untreated site on the back, and the site was evaluated upon patch removal and at 48 and 72 hours. No visible skin reactions were observed at any site during induction or challenge.
Provocative Testing
Human
Patch tests with nitrocellulose solution in butyl acetate as well as other nail-polish ingredients were performed in 25 patients with various dermatoses and 19 patients with nail-polish dermatitis; details of the test procedure (for this study performed in the early 1940s) were not provided. 24 In the patients with various dermatoses, erythema was seen in 17 of 25 participants; erythema and edema were observed in 1 participant; and vesiculation was observed in 1 participant. In the patients with nail-polish dermatitis, there was a marked increase in the irritation reactions; all of the participants reacted, and vesiculation or vesiculation with marked erythema was observed in many of the participants. It was determined that benzol was present in the solution.
Thirty patients with reactions to nail polish were patch tested with 29 nail lacquers and some of the component ingredients (details of the patch testing were not provided; this study was also performed in the early 1940s). 25 Nitrocellulose elicited a reaction in 9 of the patients and was a primary irritant. Ten control participants had negative reactions to the nail polishes and the individual ingredients.
A study was performed from September 1977 to August 1983 to examine contact dermatitis in dermatology patients. 26 During that period, 281 100 patients were seen by 12 dermatologists; 13 216 were determined to have contact dermatitis, and in 713 cases, it was determined to be cosmetic dermatitis. In those 713 cases, patch testing found only 1 reaction to nitrocellulose.
Case Reports
Nitrocellulose
A female patient presented with eczema of the neck that had been recurrent for 15 years and permanent for 3 months. 27 Patch testing found contact sensitivity to 2 of her nail varnishes, and further testing reported that she was allergic to toluenesulfonamide–formaldehyde resin, which was an ingredient in those nail varnishes. Subsequent use of a nail varnish that did not contain this ingredient resulted in eczema of the forearm, face, and neck. Additional patch testing reported the patient had contact sensitivity to nitrocellulose; positive reactions were observed with testing of nitrocellulose at the same concentration used in the product (ie, 13.3%), when diluted as 10% or 50% of that found in the product, and when either a mixture of 88.7% ethyl/butyl acetate and 11.3% isopropyl alcohol or a mixture of 49.45% ethyl/butyl acetate, 41.35% toluene, 8.1% isopropyl alcohol, and 1.1% diacetone alcohol was used as the solvent. Subsequent testing of the same concentrations in the same solvents in 100 participants did not elicit any reactions.
Collodion
Two female participants presented with contact sensitivity to a wart paint vehicle (ie, colophony). 28 Patch tests and repeated open application tests with collodion BP (a solution of ∼10% pyroxylin in a mixture of 90% alcohol [1 volume] and solvent ether [3 volumes]) were negative.
Summary
Nitrocellulose is a cellulose derivative produced by nitrating cellulose. Collodion is a preformulation solution of nitrocellulose and is defined in the International Cosmetic Ingredient Dictionary and Handbook as a solution of pyroxylin (chiefly nitrocellulose) that contains approximately 6% pyroxylin, 24% ethanol, and 70% ether. Nitrocellulose is reported to function in cosmetics as a dispersing agent—nonsurfactant and as a film former and collodion is reported to function as a binder and a film former. The VCRP data report that nitrocellulose is used in 516 nail product formulations and 1 other makeup formulation. According to an industry survey, nitrocellulose is used at concentrations of up to 41% in other manicuring preparations, that is, in nail stickers made from dried nail polish; the reported maximum concentration of use in nail polish and enamels is 22%. Collodion is not reported to be used according to VCRP data; however, there must be at least 1 use of collodion because the results of an industry survey report the maximum concentration of use of collodion is 14% in nail polish and enamels.
Nitrocellulose was not absorbed in rats following dosing by gavage with 1 mL/100 g (∼20 000 dpm/mL) aqueous [14C]nitrocellulose or [14C]nitrocellulose suspended in 0.2% methyl cellulose—0.4% Tween 80. In a mass-balance metabolism study performed in a Beagle dog, 9.5 g of nitrocellulose (34% of the dose) were recovered in the feces over a 4-day period.
The oral LD50 of a 5% nitrocellulose suspension and of a nitrocellulose-based propellant (ie, ball powder) was >5,000 mg/kg in mice and rats. In 13-week and 2-year repeated-dose dietary studies in mice, rats, and dogs, 1% and 3% nitrocellulose in feed (calculated on a dry basis) had no toxic or carcinogenic effects; effects seen at 10% were attributed to the fiber content and not the chemical nature of the test article. No tumors due to the administration of nitrocellulose were reported in the 2-year study. Nitrocellulose was not a reproductive or developmental toxicant and did not affect fertility in a 3-generation dietary study in rats; in testing with 1%, 3%, and 10% nitrocellulose, the only adverse effect observed, a statistically significant decrease in the lactation index and in pup weight at weaning in the 10% F1b through F2b litters, was attributed a lack of parental nutrition.
A 33% aqueous solution of nitrocellulose was not a primary skin or ocular irritant in rabbits in Draize tests. Ball powder also was not a skin or ocular irritant. A nail enamel formulation containing 10.5% nitrocellulose was not a primary irritant in a 48-hour semiocclusive patch test completed in 51 participants, and a nail lacquer containing 8.85% nitrocellulose was not an irritant of a sensitizer in an HRIPT completed in 108 participants and tested using semiocclusive patches. Provocative testing in the early 1940s with a nitrocellulose solution in butyl acetate that contained benzol resulted in reactions in 17 of 25 patients with various dermatoses and 19 of 19 patients with nail-polish dermatitis; reactions were much greater in the patients with nail-polish dermatitis. In another study (in 1940s) of patients with allergic eczema to nail polish, patch testing with nitrocellulose elicited a reaction in 9 of the 30 patients tested. In a 64-month (1977-1983) study, a reaction to nitrocellulose was observed in only 1 of the 713 cases with cosmetic dermatitis.
Nitrocellulose (100-5,000 μg/plate) was not mutagenic in the Ames test with or without metabolic activation. Nitrocellulose did not produce chromosomal aberrations in lymphocytes or kidney cells of rats fed a diet containing 10% nitrocellulose for 13 months or in lymphocytes (1 year), bone marrow cells, or kidney cells from rats fed a diet containing 1%, 3%, or 10% nitrocellulose for 1 or 2 years.
In epidemiology studies, although some increases in odds ratios for digestive system cancers were observed with occupational exposure to cellulose nitrate, no definitive link was identified.
Discussion
As a cosmetic ingredient, nitrocellulose is used almost exclusively in nail product formulations. The molecular weight and chemical properties of nitrocellulose support an absence of significant dermal absorption, and there is little possibility of biotransformation. Nitrocellulose did not produce toxic effects in single- or repeated-dose studies, was not a reproductive or developmental toxicant, and was not carcinogenic in animal studies. Nitrocellulose was not found to be mutagenic in the Ames test or genotoxic in chromosomal aberration assays. A human repeat insult patch test in which patches of a nail formulation containing 8.85% nitrocellulose were applied to the skin of participants did not produce any irritation or sensitization reactions, and no visible irritation was observed in another human study when patches of a nail formulation containing 10.5% nitrocellulose were applied for 48 hours. Nitrocellulose is used in formulations at higher concentrations than those tested in the human irritation and sensitization studies, because nitrocellulose is typically used in nail products and significant exposure to the skin would be minimized, it is the opinion of the Panel that neither irritation nor sensitization will be a concern.
Collodion, although listed in the International Cosmetic Ingredient Dictionary and Handbook as a separate cosmetic ingredient, simply is a solution of nitrocellulose in alcohol and ether. As used in formulations, the volatile components would be expected to evaporate, leaving nitrocellulose. Therefore, from a toxicological standpoint, the Panel determined that there are no concerns relative to the potential use of collodion in cosmetics and considered this ingredient safe for use in cosmetics in the same practices of use and concentration as given for nitrocellulose.
Conclusion
The Cosmetic Ingredient Review Expert Panel concluded that nitrocellulose and collodion are safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Authors’ Note
M. Fiume contributed to the conception and design, acquisition, analysis, and interpretation and drafted the manuscript; L. Gill, F. Alan Andersen, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to the conception and design, analysis and interpretation and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
