Abstract
The Cosmetic Ingredient Review Expert Panel assessed the safety of 34 microbial polysaccharide gums for use in cosmetics, finding that these ingredients are safe in cosmetic formulations in the present practices of use and concentration. The microbial polysaccharide gums named in this report have a variety of reported functions in cosmetics, including emulsion stabilizer, film former, binder, viscosity-increasing agent, and skin-conditioning agent. The Panel reviewed available animal and clinical data in making its determination of safety.
Introduction
As given in the Xanthan gum Hydroxypropyl xanthan gum Undecylenoyl xanthan gum Dehydroxanthan gum Xanthan gum crosspolymer Xanthan hydroxypropyltrimonium chloride Gellan gum Welan gum Biosaccharide gum-1 Biosaccharide gum-2 Biosaccharide gum-3 Biosaccharide gum-4 Biosaccharide gum-5 Pseudoalteromonas exopolysaccharides Dextran Carboxymethyl dextran Dextran hydroxypropyltrimonium chloride Sodium carboxymethyl dextran Dextran sulfate Sodium dextran sulfate Sclerotium gum Hydrolyzed sclerotium gum Beta-glucan Beta-glucan hydroxypropyltrimonium chloride Beta-glucan palmitate Hydrolyzed beta-glucan Oxidized beta-glucan Sodium carboxymethyl beta-glucan Pullulan Myristoyl pullulan Levan Rhizobian gum Hydrolyzed rhizobian gum Alcaligenes polysaccharides
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) reviewed other nonmicrobial gums and polysaccharides in 2012 and concluded the galactomannans, a group of 16 legume polysaccharides, are safe as used in cosmetics. 2 In 2009, the Panel reviewed the safety of hyaluronic acid, an amine-derived exopolysaccharide, finding it safe as used. 3 In 1987, the Panel concluded that tragacanth gum (now named Astragalus gummifer gum) was safe as used; the Panel reaffirmed that conclusion in 2006. 4 The Panel also concluded (2005) that Acacia senegal gum is safe as used but that the data are insufficient to support the safety of Acacia catechu gum and Acacia farnesiana gum as used in cosmetics. 5
Three ingredients included in this safety assessment are hydroxypropyltrimonium chloride compounds. Although hydroxypropyltrimonium chloride itself is not a cosmetic ingredient, it is structurally analogous to some of the compounds included in the CIR safety assessment on trimoniums, which the Panel concluded are safe as used in cosmetics when formulated to be nonirritating. 6
Some of these polysaccharide gums can be produced by more than one organism and sometimes by plants. For example, beta-glucans are produced by fungi, yeasts, and grains, 7 and levan can be produced by bacteria, yeasts, or fungi. 8
Many studies have been conducted with some of the microbial polysaccharide gums in regard to health claims, immunomodulatory activity, antioxidant activity, and so on. This safety assessment includes only studies and study types that relate directly to the safety of the cosmetic use of these ingredients.
Chemistry
Definition and Structure
Microbial polysaccharide gums are high-molecular-weight (MW) carbohydrate polymers that makeup a substantial component of the cellular polymers found in and surrounding most microbial cells. 9 These polysaccharide gums are produced by a wide variety of microorganisms and are water-soluble gums that have novel and unique physical properties. Microbial polysaccharide gums are generally divided into 3 groups: exocellular, cell wall, and intercellular. 10 The exocellular polysaccharide gums are those that constantly diffuse into the cell culture medium and are easily isolated. The cell wall (ie, structural) and intercellular polysaccharide gums are integral parts of the cell wall or capsular products and are more difficult to separate from cell biomass.
Microbial polysaccharide gums may be ionic or nonionic and are primarily linear polysaccharides to which side chains of varying length and complexity are attached at regular intervals.
9
Most microbial polysaccharide gums are linear heteropolysaccharides consisting of 3 to 7 different monosaccharides arranged in groups of 10 or less to form repeating units. The monosaccharides may be pentoses, hexoses, amino sugars, or uronic acids. For example, xanthan gum is a polysaccharide produced by a pure culture fermentation of a carbohydrate with
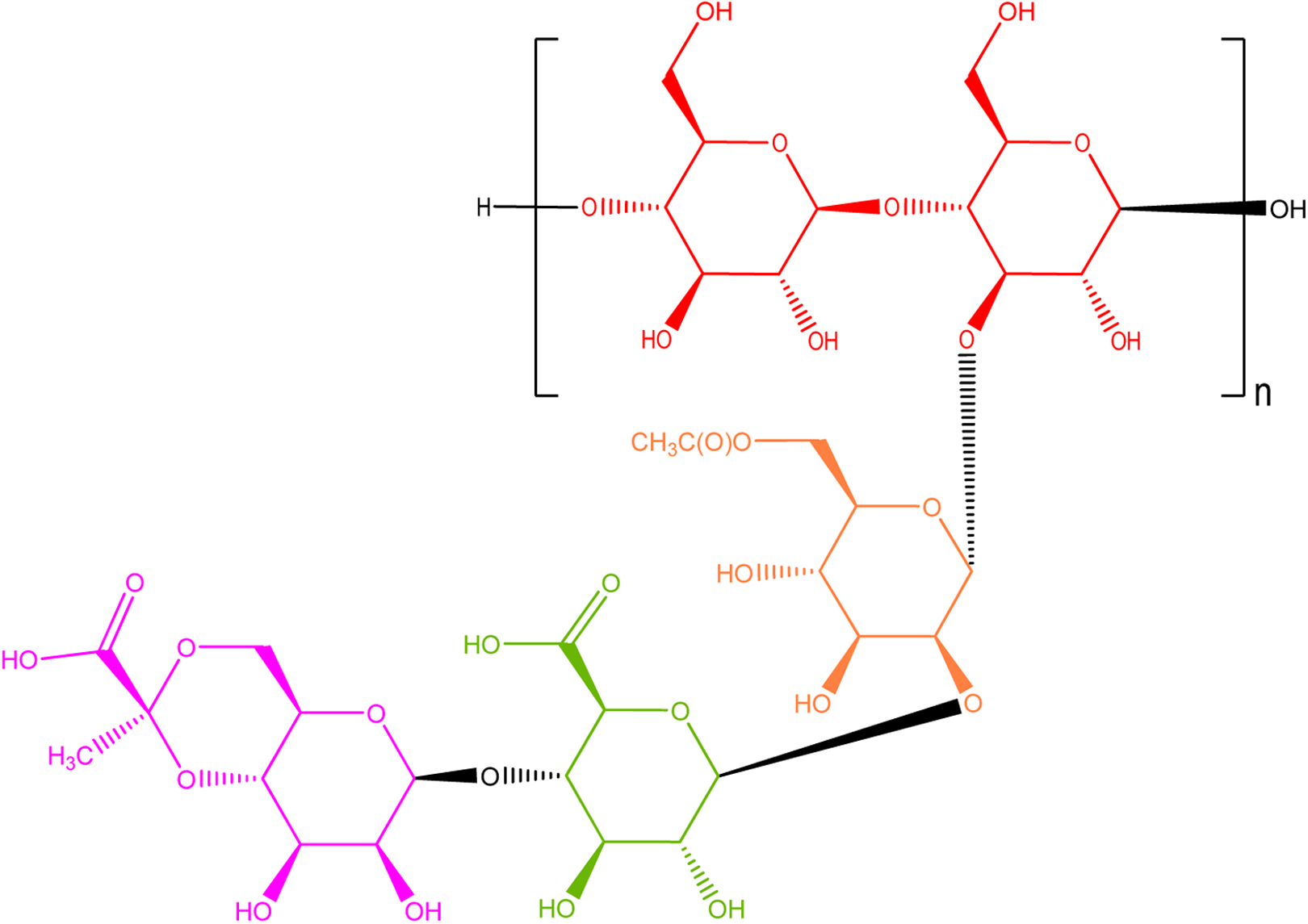
Xanthan gum—a polysaccharide composed of glucose, glucuronic acid, 6-acetyl mannose, and 4,6-pyruvylated mannose.
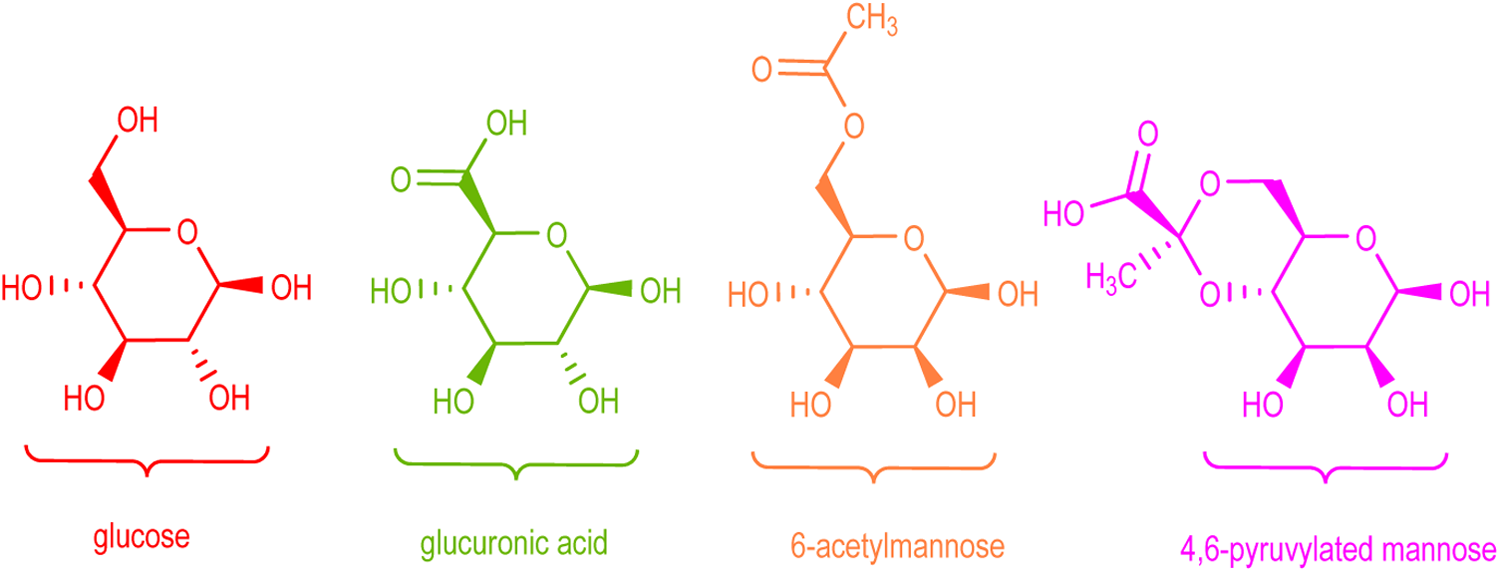
Glucose, glucuronic acid, 6-acetyl mannose, and 4,6-pyruvylated mannose, the monosaccharide components of xanthan gum.
Definition, Function, and Idealized Structure.

Abbreviations: Aq., aqueous; MW, molecular weight.
Physical and Chemical Properties
Available physical and chemical properties are provided in Table 2. The properties of the microbial polysaccharide gums can vary widely based on, among other parameters, the side groups, the ester substituents, or the bacterial strains. 11 –14
Chemical and Physical Properties.

Abbreviations: Aq., aqueous; MW, molecular weight.
Constituents/Impurities
The available constituent and impurity data are provided in Table 3.
Constituents/Impurities.

Methods of Manufacture
Methods of manufacture for many of the microbial polysaccharide gums are provided in Table 4. Some of the polysaccharide gums discussed in this safety assessment can be produced by more than 1 organism and, in some cases, by plants. For example, beta-glucans are produced by fungi, yeasts, and grains, 7 and levan can be produced by bacteria, yeasts, or fungi. 8
Methods of Manufacture/Organisms Used in Production.

Use
Cosmetic
The microbial polysaccharide gums named in this report have a variety of reported functions in cosmetics that include emulsion stabilizer, film former, binder, viscosity-increasing agent, and skin-conditioning agent. 1 The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2012, 15 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council), 16,17 indicate that 19 of the 34 microbial polysaccharide gums named in this safety assessment are currently used in cosmetic formulations. Xanthan gum is used in almost every category of cosmetic product, with 3,470 reported uses. Biosaccharide gum-1, sclerotium gum, and beta-glucan are reported to be used in 346, 193, and 137 cosmetic formulations, respectively. All other in-use ingredients have less than 70 uses. The ingredient with the highest concentration of use is pullulan; it is used at up to 12% in leave-on formulations (ie, tonics, dressings, and other hair-grooming aids) and 17% in “other” oral hygiene products (a breath freshener that dissolved in the mouth 18 ). Both xanthan gum and biosaccharide gum-1 are used at up to 6% in leave-on formulations, and xanthan gum crosspolymer and biosaccharide gum-4 are used at 5% in leave-on formulations. All other in-use ingredients are used at concentrations of ≤3%.
In some cases, reports of uses were received in the VCRP but no concentration of use is available. For example, sodium carboxymethyl dextran is reported to be used in 10 formulations, but no use concentration data were available. In other cases, no reported uses were received in the VCRP, but a use concentration was provided in the industry survey. For example, hydrolyzed sclerotium gum was not reported in the VCRP to be in use, but the industry survey indicated that it is used in leave-on formulations at up to 1%. It should be presumed that hydrolyzed sclerotium gum is used in at least 1 cosmetic formulation.
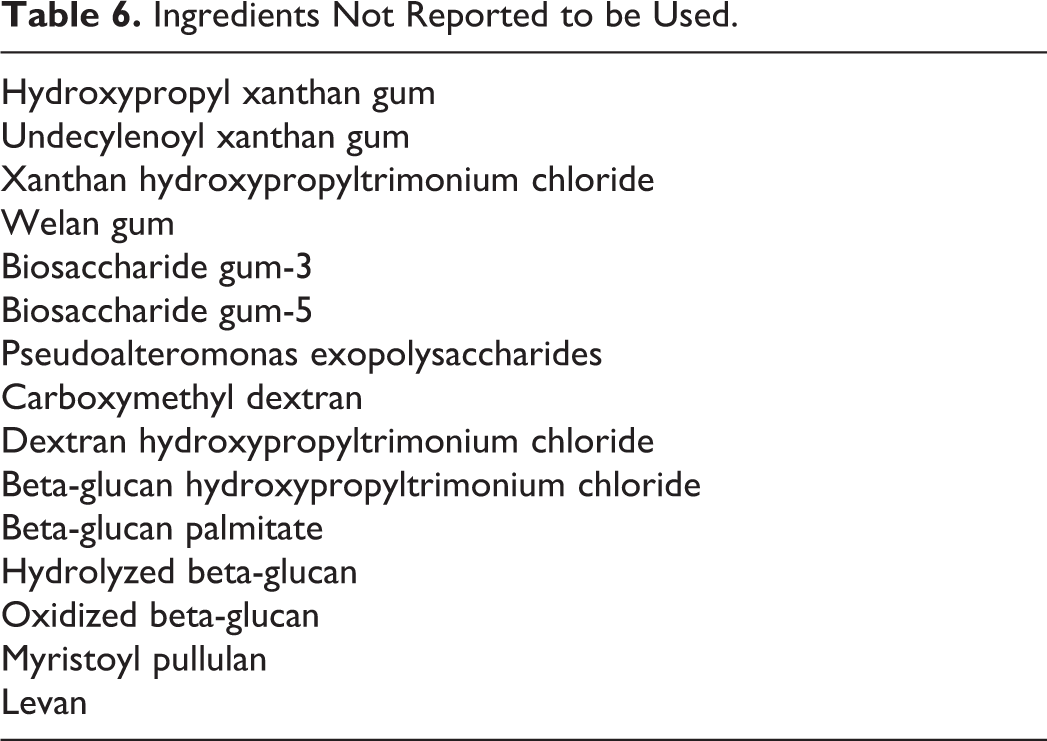
Frequency and concentration of use data are provided in Table 5. The ingredients not listed in the VCRP or by the Council as being used are listed in Table 6.
Frequency and Concentration of Use According to Duration and Type of Exposure.
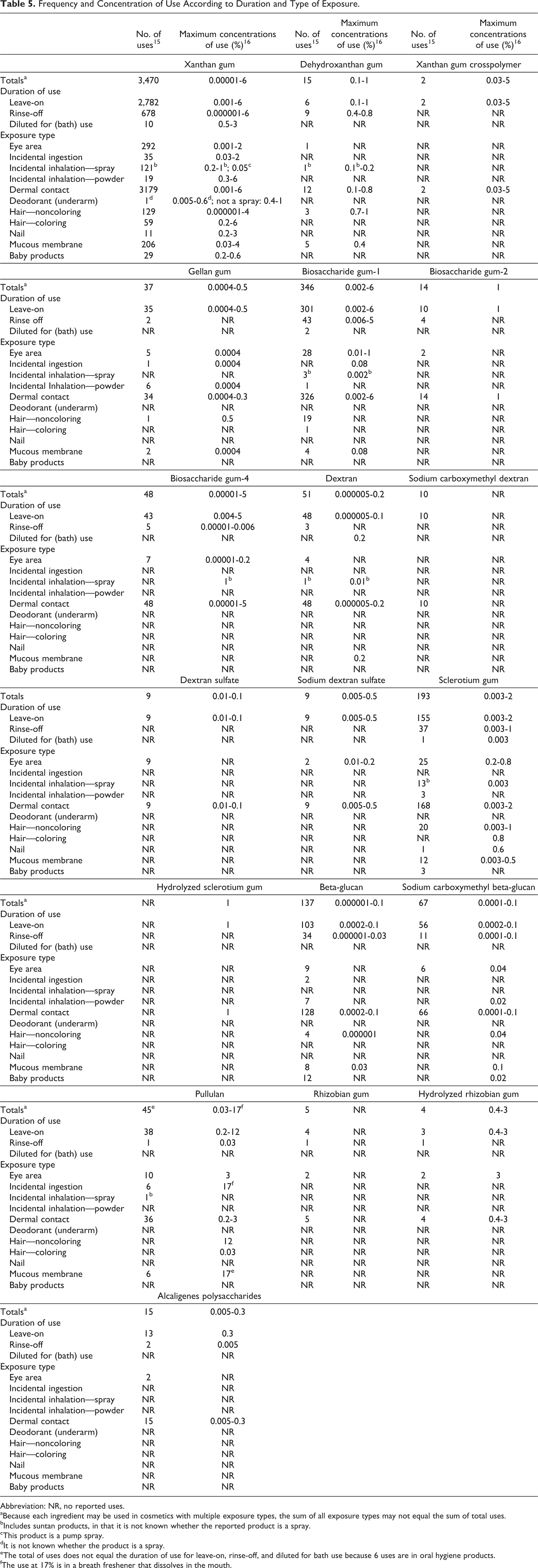
Abbreviation: NR, no reported uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether the reported product is a spray.
cThis product is a pump spray.
dIt is not known whether the product is a spray.
eThe total of uses does not equal the duration of use for leave-on, rinse-off, and diluted for bath use because 6 uses are in oral hygiene products.
fThe use at 17% is in a breath freshener that dissolves in the mouth.
Ingredients Not Reported to be Used.
Products containing some of the microbial polysaccharide gums are reported to be used on baby skin, to be applied to the eye area or mucous membranes, or could possibly be ingested. Some of these ingredients are reported to be used in product types that may be inhaled; for example, dehydroxanthan gum is used in a face and neck spray at 0.2%. In practice, 95% to 99% of the particles released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 μm range. 19,20 Therefore, most particles incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable. 21,22 Xanthan gum is reported to be used in deodorants at up to 0.6%, and it is not known whether these products are sprayed. There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic diameters in the range considered to be respirable. 22 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays compared to other cosmetic sprays.
All of the microbial polysaccharide gums named in the report are listed in the European Union inventory of cosmetic ingredients. 23
Noncosmetic
Noncosmetic uses of microbial polysaccharide gums are summarized in Table 7. Some of the food and medical use information is described in the following paragraphs.
Examples of Noncosmetic Uses.

Xanthan gum 24 and gellan gum 25 are approved as direct food additives in gums, chewing gum bases, and related substances. Xanthan gum is also approved as an indirect food additive. 26 Beta-glucan (as curdlan, a specific beta-glucan that is a linear polymer consisting of β-(1 → 3)-linked glucoside residues) is approved as a direct food additive for multipurpose addition. 27 Dextran is an indirect food additive that is generally recognized as safe. 28 The World Health Organization concluded that studies on the safety of xanthan gum, 29 gellan gum, 30 and pullulan 31 provided sufficient information to be allocated an acceptable daily intake of “not specified.” Pullulan appears in the Japanese List of Existing Food Additives. 32
Xanthan gum is used as a stabilizer, thickener, and emulsifying agent in water-based pharmaceutical preparations. 33 Dextran, as dextran 70, is an approved active ingredient for over-the-counter use as an ophthalmic demulcent at 0.1% when used with another approved polymeric demulcent. 34 Dextran is used as a plasma volume expander (as dextran 70) and as a blood flow adjuvant (as dextran 40). 35 Sodium dextran sulfate is used as a clinical reagent.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Dermal
In Vitro
Beta-Glucan
A single application of 5 mg/cm2 of a 0.5% (oat) beta-glucan solution was applied to human abdominal skin.
36
Beta-glucan penetrated the skin into the epidermis and dermis (no details were provided). Human
Dextran
Fluorescent dextrans (FDs) in aqueous (aq.) solution were used to determine the dermal absorption of different MW dextrans (MW 3,000-70,000, identified as FD-3 and FD-70, respectively) through the skin that has been subjected to mini erosion. 37 Prior to testing, absorption of FD-20 from a test “Cellpatch” (cell) into systemic circulation via a mini-erosion site was verified in 2 male subjects and 1 female subject. The largest of the test molecules filtered from the blood into the urine was 20,000 MW, and with increasing erosion diameter, absorption increasingly occurs via the lymphatic system. Each subject received a single cell containing 0.5 mL solution (5 mmol). After 24 hours, a mean of 54.3% of the total FD-20 dose had been absorbed, and 12.6% of the absorbed dose was recovered in the urine.
The effect of molecular size on absorption of FDs was then determined. Four cells with 100 μL of FD-3, FD-10, FD-20, or FD-70 in 0.5 mL isotonic saline were applied for 24 hours to 6 mm mini-eroded sites on 4 male and 3 female subjects. A fifth cell, without FD, was applied as a negative control. The FD concentration in each cell was measured using spectrofluorometry at various times. The dextrans were readily absorbed, but absorption decreased with increasing molecular size. The absorption of FD-3 was 37.9%, but for FD-70, it was 20.1%. Further testing using 3 male and 3 female subjects determined that the degree of transdermal absorption was directly related to the area of erosion; 20.5% of FD-3 absorbed through a 3-mm erosion area, whereas 60.7% of the same molecular size FD absorbed through a 10-mm erosion.
Oral
Nonhuman
Xanthan gum
Rats were fed a diet containing 2% [14C]xanthan gum that was produced by fermentation of uniformly labeled glucose with
Gellan gum
In animal feeding studies using radiolabeled gellan gum, the majority of the gellan gum administered was recovered in fecal matter. 14 This appears to indicate that no endogenous enzymes that are able to break down gellan gum are present in the small intestine (no additional details were provided).
The absorption, distribution, and excretion of gellan gum were determined in studies using a dually radiolabeled gum that was prepared in separate fermentations using [3H]glucose and [ 14 C]glucose as carbon sources. 39 The 3 H product was subjected to multistage purifications for a relatively pure [ 3 H]polysaccharide, which was then added to the 14 C fermentation, giving a polysaccharide fraction that was dual labeled and a nonpolysaccharide fraction labeled only with 14 CO2. In the first study, 1 male and 1 female Sprague Dawley rats were dosed by gavage with a single dose of 960 mg/kg [ 3 H/ 14 C]gellan gum (4 μCi). Expired air was collected for 24 hours after dosing, and <0.55% of the dosed radioactivity was detected as 14 C.
Four male and 3 female Sprague Dawley rats were then given a single dose by gavage of 870 mg/kg [ 3 H/ 14 C]gellan gum (2.9-4.1 μCi 14 C; 0.7-0.9 μCi 3 H). Urine and feces were collected for 7 days. Approximately 86% of the dosed 14 C was excreted in the feces and 2% to 3% in the urine, and approximately 100% of the dosed 3 H was excreted in the feces and 4% was in the urine. Tissue and carcass 14 C radioactivity was approximately 3% to 4% of the dose. The 3 H activities in the tissues were below the limits of accurate quantification.
In the last study, 1 male and 4 female Sprague Dawley rats were dosed with 1 g/kg [ 3 H/ 14 C]gellan gum by gavage, and blood samples were taken at various intervals over a 7-day period (it is not stated but it appears that 1 dose was administered). The peak level of radioactivity in blood, equivalent to 0.4% of the dose, occurred about 5 hours after dosing.
Dextran
Groups of 5 fasted male Sprague Dawley rats were given a single dose by gavage of 50 mg/kg fluorescein-labeled dextrans (FD-4: 4,400 average MW; FD-20: 19,000 average MW; or FD-40: 40,500 average MW). 40 The dextran solution was prepared as 25 mg/mL in isotonic phosphate buffer. Blood samples were taken at various intervals for up to 4 hours after dosing with FD-4, up to 8 hours after dosing with FD-20, and for up to 24 hours with FD-40. Urine samples were taken at intervals for up to 8 hours after dosing with FD-4 and FD-20 and for up to 24 hours after dosing with FD-40. None of the dextrans were detected in the serum after oral administration. Small amounts of the dose, ranging from 0.308% with FD-4 to 0.0138% FD-40, were detected in the urine. The oral bioavailability was 0.398%, 0.0728%, and 0.0431% for FD-4, FD-20, and FD-40, respectively.
Dextran sulfate
Two groups of 5 male Wistar rats were dosed by gavage with 5 mg/mL of 20 mg/kg dextran sulfate containing 12 μCi 3 H-dextran sulfate/kg (8,000 average MW), and each group was killed 3 or 24 hours after dosing. 41 Most of the radioactivity was detected in the feces; 22.5% of the dose was recovered after 24 hours. Metabolites, breakdown products, and 3 H2O were not found in the feces, and the researchers stated that it was most likely that the dose recovered in the feces was unabsorbed dextran sulfate. Approximately 10% of the dose was recovered in the urine after 24 hours, with 6% recovered after 3 hours. Urinary 3 H elution profiles indicated that intermediate MW metabolites or breakdown products were formed and that either intact or partially intact dextran sulfate was absorbed through the epithelium of the gastrointestinal (GI) tract. The 6- to 24-hour samples indicated a marked shift toward smaller MW products.
Beta-glucan
Two male Sprague Dawley rats were dosed orally with 20 mg/kg body weight (BW) [U- 14 C]beta-glucan (as curdlan) in water prepared from [U- 14 C]glucose. 42 Most of the radioactivity was recovered in expired CO2; 77% of the dose was recovered in 24 hours and 89% in 72 hours. A total of 7.7% and 12% of the radioactivity as administered dose was recovered in the feces after 24 and 72 hours, respectively, and 2.6% and 3.3% was recovered in the urine after 24 and 72 hours, respectively.
In another study, 3 male Wistar rats were dosed orally with 20 mg/kg BW [ 14 C] beta-glucan (as curdlan) in water. 42 The excretion of 14 CO2 was low for the first 3 hours, but increased linearly up to 12 hours, plateauing at 39% of the administered radiolabel. A total of 3.4% and 3.8% of the radioactivity as administered dose was recovered in the feces after 24 and 48 hours, respectively, and 1.3% and 1.4% was recovered in the urine after 24 and 48 hours, respectively. In a group of 3 male Wistar rats administered 5 mg/mL tetracycline for 5 days prior to and 2 days following beta-glucan, it was demonstrated that the intestinal microflora are partly responsible for the metabolism of beta-glucan to carbon dioxide.
The effect of dose on metabolism was also examined. 42 Three male Wistar rats were given an oral dose of 2.3, 23, or 230 mg/kg BW [ 14 C]beta-glucan (as curdlan) in water. At the 2 higher doses, excretion of radioactivity as carbon dioxide decreased with increasing dose, while fecal excretion of the radiolabel increased. The researchers stated that this was an indication of limited metabolism at higher doses.
Pullulan
Five fasted male Wistar rats were dosed by gavage with a 2 mL of a 10% solution of pullulan (49,000 MW) in 0.9% saline.
43,44
The animals were killed 1 hour after dosing, and the contents of their stomach and small intestines were collected. Approximately 3% of the pullulan had been hydrolyzed; it was not known whether the hydrolysis products were absorbed by the small intestine. Human
Dextran
Dextran can be depolymerized by α-1-glucosidases (dextranases) that occur in the liver, spleen, kidney, and lower part of the GI tract. 11
Dextran sulfate
Six fasted male subjects were given a single oral dose of 1,800 mg dextran sulfate (7,000-8,000 MW; 17%-20% sulfur), and after 48 hours, a single intravenous (IV) dose of 225 mg dextran sulfate in saline infused over 60 minutes. 45 After oral dosing, no measurable dextran sulfate was found in the plasma using the competitive binding assay, and there was no increase in activated partial thromboplastin time (aPTT). Plasma lipolytic activity did not increase the first 3 hours after oral dosing; at 3 to 4 hours after oral dosing, it increased by 2 times the baseline average. Very little dextran sulfate was recovered in the urine after oral dosing. After IV dosing, peak plasma concentrations were 26 to 35 μg/mL, and the aPTT was increased by an average of 6.9 times over the baseline values. The plasma lipolytic activity increased by an average of 438 times the baseline value. Dextran sulfate was recovered in the urine after IV dosing.
Pullulan
Pullulan is only partially hydrolyzed by salivary and pancreatic amylases of the upper GI tract; essentially no monomeric glucose is released during hydrolysis. 44 Pullulan is largely resistant to digestion in the GI tract because of the occasional presence of 1 → 3-glycosidic linkages and the high percentage of α-1 → 6-glycosidic linkages. 31 The degree of digestion appears to be dependent on molecular mass. Pullulan is fermented in the colon by intestinal microflora to produce short-chain fatty acids; the degree of fermentation is dependent on the degree of polymerization of pullulan.
Six subjects ingested 10 g pullulan (50,000 MW) for 14 days. 44,46 Administered pullulan was fully digested in the intestinal tract and was not detected in the feces. After 14 days, the fecal short-chain fatty acid concentration increased from 6 to 8.8 mg/g. The researchers concluded that pullulan was completely fermented to short-chain fatty acids by intestinal bacteria.
Parenteral
Nonhuman
Dextran
Rabbits were given a daily IV 30 mL dose of a 6% solution of partly hydrolyzed bacterial dextran (75,000 average MW) 6 d/wk for 103 to 113 weeks. 47 Eleven animals were evaluated. Approximately 25% of the dose was excreted in the urine. The plasma concentration of dextran at study termination (0.50 g/100 mL) did not differ from the value at 2 months (0.44 g/100 mL), but it was generally greater than the value reported at 24 hours after a single 30-mL dose (not given). Moderate dextran storage was observed in the spleen without an increase in the total carbohydrates. However, considerable storage was observed in the liver with a marked increase in carbohydrates. Additional details and results are provided in the section on “Repeated Dose Toxicity”.
Groups of 5 male Sprague Dawley rats were given a single IV dose of 5 mg/kg FD-4 (4,400 average MW), FD-20 (19,000 average MW), FD-40 (40,500 average MW), FD-70 (71,000 average MW), or FD-150 (147,800 average MW). 40 The dextran solution was prepared as 5 mg/mL in isotonic phosphate buffer. Blood samples were taken at various intervals for up to 4 hours after dosing with FD-4, up to 8 hours after dosing with FD-20, and for up to 24 hours with FD-40, FD-70, and FD-150. Urine samples were taken at intervals for up to 8 hours after dosing with FD-4 and FD-20 and for up to 24 hours after dosing with FD-40, FD-70, and FD-150. Pharmacokinetic parameters were MW dependent. Concentrations of the 3 highest MW dextrans could be detected in serum for up to 12 hours after dosing, whereas FD-20 and FD-4 were not found in the serum after 3 and 1.5 hours, respectively. The distribution half-life (t1/2α) ranged from 0.0517 to 0.895 hours for FD-4 and FD-150, respectively, and the elimination half-life (t1/2β) ranged from 0.282 to 3.03 hours for FD-4 and FD-150, respectively.
Male Sprague Dawley rats were also given a single IV dose of 1, 25, or 100 mg/kg FD-4 (average MW 4,300) and FD-150 (average MW 145,000). 48 The dextran solution was prepared as isotonic phosphate buffer at a concentration that would result in a test volume of 2 mL/kg. Blood, urine, and tissue samples were taken at various intervals for up to 6 hours from groups of 4 rats dosed FD-4 and for up to 96 hours in rats dosed with FD-150. Renal excretion was a major excretion pathway for FD-4 but not FD-150. Urinary recovery ranged from 79% to 82% of the dose with FD-4 and only from 1.1% to 2.1% of the dose with FD-150; at each MW, the dose administered did not have a statistically significant effect on the amount excreted. Renal clearance ranged from 344 to 360 mL/h/kg with FD-4 and from 0.131 to 0.245 mL/h/kg for FD-150, and systemic clearance ranged from 420 to 457 mL/h/kg for FD-4 and from 8 to 20 mL/k/kg for FD-150. The highest concentrations of FD-4 were found in the kidneys at 1 minute after dosing (9.31%, 10.0%, and 10.4% of the dose with 1, 25, and 100 mg/kg, respectively) was linear with dose. The highest concentrations of FD-150 were found in the liver (68.5% at 5 hours, 51.6% at 24 hours, and 41.5% of the dose at 24 hours with 1, 25, and 100 mg/kg, respectively) and the spleen (11.5% at 12 hours, 2.09% at 48 hours, and 1.21% of the dose at 96 hours with 1, 25, and 100 mg/kg, respectively); recovery of dextran in the liver and spleen was nonlinear, with a greater difference seen in the spleen than in the liver. The researchers reported that the MW of recovered FD-4 remained relatively constant but that of recovered FD-150 changed significantly. The MW of FD-150 recovered in the urine was <40,000, and in the liver, the average MW at 96 hours was ∼70,000. The estimated MW of the recovered FD-150 in the liver appeared to be dose dependent, with higher doses having a higher average MW. The researchers concluded that excretion of lower MW dextrans was independent of dose, whereas excretion of higher MW dextrans was dose dependent.
Another study also found that MW affected the distribution and excretion of dextran. 49 Female BALB/cCrSlc mice were dosed IV with 0.1 mL of 0.1 wt% 125 I-labeled dextran, MW ranging from 4,980 to 220,000, in phosphate-buffered saline (PBS); dextran was labeled through radioiodination of tyramine residues. Blood samples were taken at various intervals for some groups, and tissues were collected from others (the number of animals/group was not specified). High MW dextran remained in the blood longer than lower MW dextran. The t1/2β also increased with increasing MW, with a pronounced change occurring around 30,000 MW. After 3 hours, 83% of the dose of the lowest MW dextran was excreted, whereas only 41% of the highest MW dextran was found in excrement. Most of the high MW dextran was found in the liver; after 3 hours, 5.2% of the high MW dextran was found in the liver, whereas only 0.7% of the lowest MW was recovered in that tissue. At 3 hours, 3.5% versus 19% of the dose of the lowest and highest MW dextran, respectively, was recovered in the carcass. Two percent to 10% of the dose was recovered in the GI tract, but the amount recovered did not appear to be related to MW.
Dextran sulfate
A preliminary study was performed in which 2 male Wistar rats were dosed by IV injection with 20 mg/kg dextran sulfate 8,000 average MW containing 10 μCi [ 3 H]dextran sulfate/kg via the penile vein (5 mg/100 μL). 41 The rats were killed 1 or 3 hours after dosing. The total 3 H excreted in the urine accounted for approximately 50% of the administered dose. Within 1 hour after IV administration, rapid excretion of intact dextran sulfate occurred.
In the main study, groups of 5 male Wistar rats were dosed by IV injection via the penile vein with 5 mg/100 μL of 20 mg/kg dextran sulfate containing 12 μCi [ 3 H]dextran sulfate/kg, and each group was killed 3 or 24 hours after dosing. Urine was the major route of excretion following IV dosing, with approximately 46% of the dose excreted within 3 hours and 51% within 24 hours. Approximately 2% of the dose was recovered in the feces after 24 hours. Based on the 3 H elution profiles, it was hypothesized that dextran sulfate or its metabolites were incorporated into higher MW compounds, such as glycogen, at 3 to 6 hours and smaller MW at 6 to 24 hours. In the plasma, the highest concentration of dextran sulfate was found at 3 hours; the amount decreased with time.
In the tissues, the highest amounts of radioactivity were distributed in the liver, kidney, and spleen. The amount of radioactivity recovered in the tissues following IV administration was compared to that found following oral administration (the oral study was described previously in this safety assessment). Concentrations of 3 H in all tissues and fluids were statistically significantly higher in the animals dosed by IV injection compared to those in animals dosed orally.
Beta-glucan
Groups of 2 male Sprague Dawley rats were dosed by intraperitoneal (IP) injections with 20 mg/kg BW [ 14 C]beta-glucan (as curdlan) in water, and the animals were killed 0.5, 3, 6, or 24 hours after dosing. 42 After 24 and 48 hours, only 1.8% and 4.1% of the radioactivity was recovered in CO2, respectively, 0.05% and 0.12% was recovered in the feces, respectively, and 3.5% and 4.1% of the radioactivity as percentage of dose was recovered in the urine, respectively. Whole-body radioautography showed that the radiolabel was distributed in the intestinal fluids.
Pullulan
The effect of MW on the distribution and excretion of pullulan was examined. 49 Female BALB/cCrSlc mice were dosed IV with 0.1 mL of 0.1 wt% [ 125 I]pullulan, MW ranging from 5,800 to 853,000, in PBS; pullulan was labeled through radioiodination of tyramine residues. Blood samples were taken at various intervals for some groups, and tissues were collected from others (the number of animals/group was not specified). High MW pullulan remained in the blood longer than lower MW pullulan. The t1/2β also increased with increasing MW. After 3 hours, 96% of the dose of the lowest MW pullulan was excreted, whereas only 15% of the highest MW pullulan was found in excrement. At 3 hours, most of the high MW pullulan, 50% of the dose, was recovered in the liver; only 1% of the dose of the lowest MW pullulan was recovered in the liver after 3 hours. The highest percentage of the dose recovered in the GI tract was 5.3% pullulan with a MW of 100,000 MW, and in the carcass, it was 10.1% pullulan with a MW of 48,000.
Fasted male Wistar rats (number not specified) were injected with a single IV dose 2 mL of 0, 6, 12, 18, or 24 mg/kg fluorescein-labeled pullulan (MW 58,200) in saline in the jugular vein.
50
Pullulan was rapidly eliminated from the blood; however, elimination decreased as dose increased. Hepatic uptake was also dose dependent; the hepatic uptake clearance of pullulan decreased with increased dose. In the liver, distribution of pullulan in the parenchymal cells was 2.5 times greater than in the nonparenchymal cells. The researchers did state that with a higher MW pullulan, average 70,000 MW, uptake was greater in the nonparenchymal cells. Human
Dextran
Four male and 3 female subjects received an IV injection 100 mL, a partially degraded dextran (40,000 average MW; 10% wt/vol in 0.9% saline). 51 Blood samples were taken at various intervals after dosing. There was an initial rapid decrease in the serum in the first hour after dosing. Different fractions of the dextran were eliminated from the plasma at different rates; higher MW fractions remained in the plasma longer.
Toxicological Studies
Single-Dose (Acute) Toxicity
Acute toxicity studies are summarized in Table 8. The acute toxicity of xanthan gum, gellan gum, beta-glucan, sodium carboxymethyl beta-glucan, and pullulan was assessed orally in mice, rats, and/or dogs, and dextran sulfate and beta-glucan were tested by IP and IV dosing in mice and rats. There was no notable toxicity observed in these studies. In acute inhalation studies, the LC50 of xanthan gum was >21 mg/L in rabbits and of gellan gum was >5.06 mg/L in rats. A single 180-minute exposure of humans office dust containing 10 mg curdlan/g dust resulted in decreased nasal volume, swelling in the nasal turbinates, and an increase in nasal eosinophils when compared to “clean” dust.
Acute Toxicity Studies.
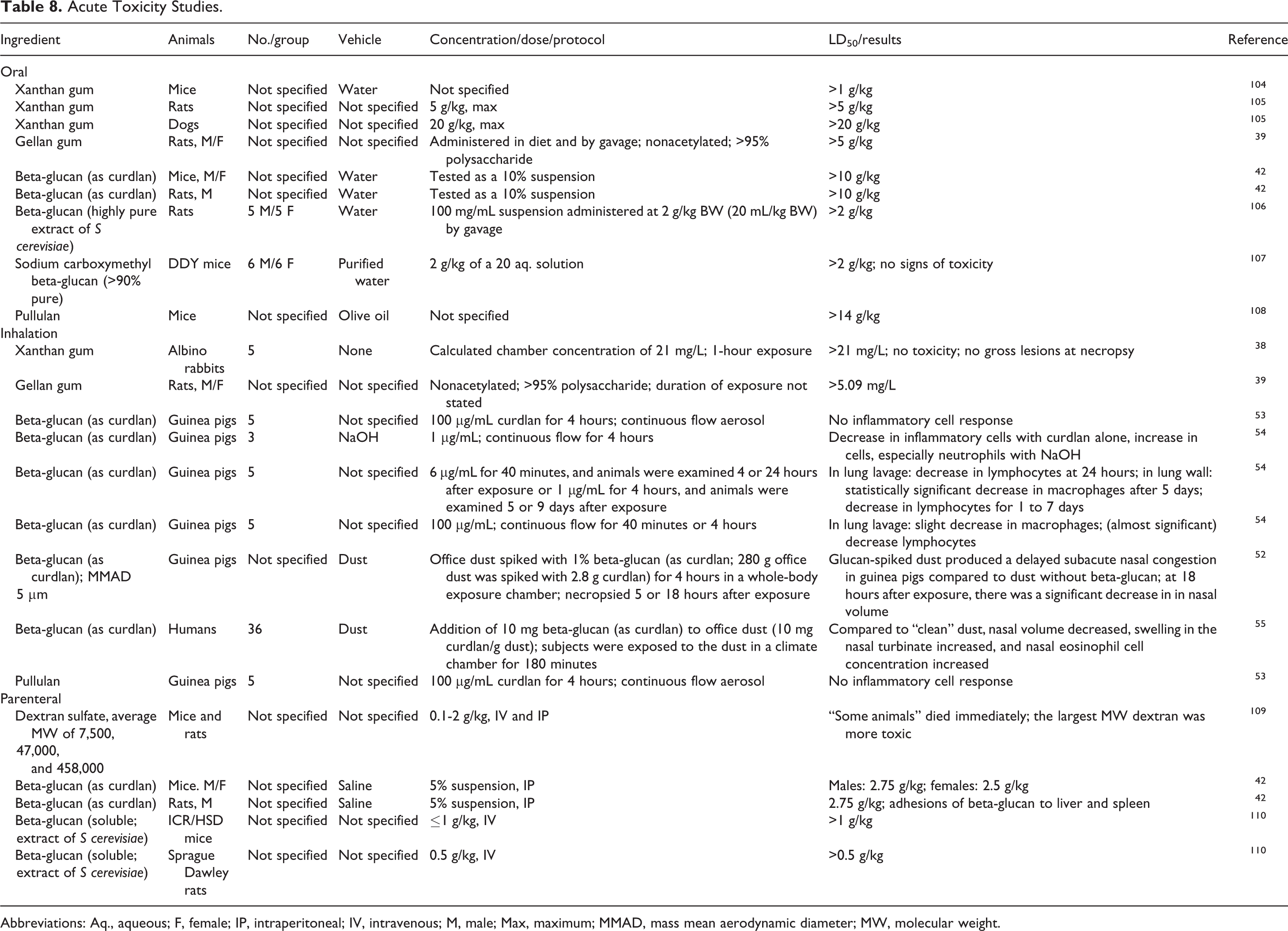
Abbreviations: Aq., aqueous; F, female; IP, intraperitoneal; IV, intravenous; M, male; Max, maximum; MMAD, mass mean aerodynamic diameter; MW, molecular weight.
Inflammatory response
Beta-glucan
The inflammatory response following a single exposure to beta-glucan (as curdlan) was evaluated in guinea pigs. 52 –54 Mostly, no effect or a slight decrease in inflammatory cells in lung lavage was observed. A 4-hour inhalation exposure to 1% beta-glucan in dust (mass mean aerodynamic diameter [MMAD] 5 μm) by guinea pigs produced a delayed subacute nasal congestion when compared to dust without beta-glucan (MMAD 6.5 μm) and resulted in decreased nasal volume; after 18 hours, there was a significant decrease in nasal volume. 52 In humans, inhalation exposure to beta-glucan in dust for four 3-hour exposures resulted in increased nasal swelling and decreased nasal volume and an immediate increase in nasal eosinophil/mL count. 55
Pullulan
The effect of pullulan on inflammation was investigated in ICR mice using the xylene-induced acute inflammatory mouse ear model. 56 Groups of 9 mice were dosed orally with 0, 62.5, 125, or 250 mg/kg pullulan in distilled water 30 minutes prior to topical application of xylene to 1 ear. Two hours after xylene application, all animals were killed. Compared to xylene-treated controls, ear weights were statistically significantly decreased in a dose-dependent manner. Additional histological indicators of inflammation were not observed.
Levan
An interleukin (IL) 1α release assay was used to determine the anti-inflammation effect of a 5% aq. levan solution on artificial skin. 57 Primary skin irritation was first induced using sodium lauryl sulfate. A dose of 0.01 or 0.05 mg/mL of the solution was applied to the skin. Levan decreased IL-1α release, indicating an anti-inflammatory effect.
Cytotoxicity
Levan
The cytotoxic effect of 5% levan (wt/wt) was determined using human fibroblasts and keratinocytes. 57 The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay was used to measure cell viability and proliferation after 24-hour incubation with levan. Levan, ≤100 μg/mL, was not cytotoxic to human fibroblasts. Levan had a proliferative effect in keratinocytes; proliferation was >30% at concentrations of >1 mg/ml.
Repeated Dose Toxicity
Repeated dose toxicity studies are summarized in Table 9. The oral toxicity of xanthan gum was evaluated in rats and dogs, of gellan gum in rats, dogs, and monkeys, of dextran in rats, of beta-glucan in mice, rats, and dogs, and of pullulan in rats. Most of the studies were dietary, and study durations lasted up to 2 years. Most observations were related to changes in feed consumption and intestinal effects. In guinea pigs, inhalation exposure to 100 μg/mL beta-glucan (as curdlan), 4 h/d, 5 d/wk for 4 weeks, did not have an effect on the cells of the lung lavage or cell wall, and there were no microscopic lesions of the lung. With IP administration, no toxicity was reported when mice were dose 10 times over 2 weeks with 5 mg xanthan gum in 0.5 mL water or mice or guinea pigs were dosed with 250 mg/kg BW beta-glucan for 7 days. Intravenous administration of 40 and 1,000 mg/kg BW beta-glucan for 30 days resulted in hepatosplenomegaly in mice.
Repeated Dose Toxicity Studies.
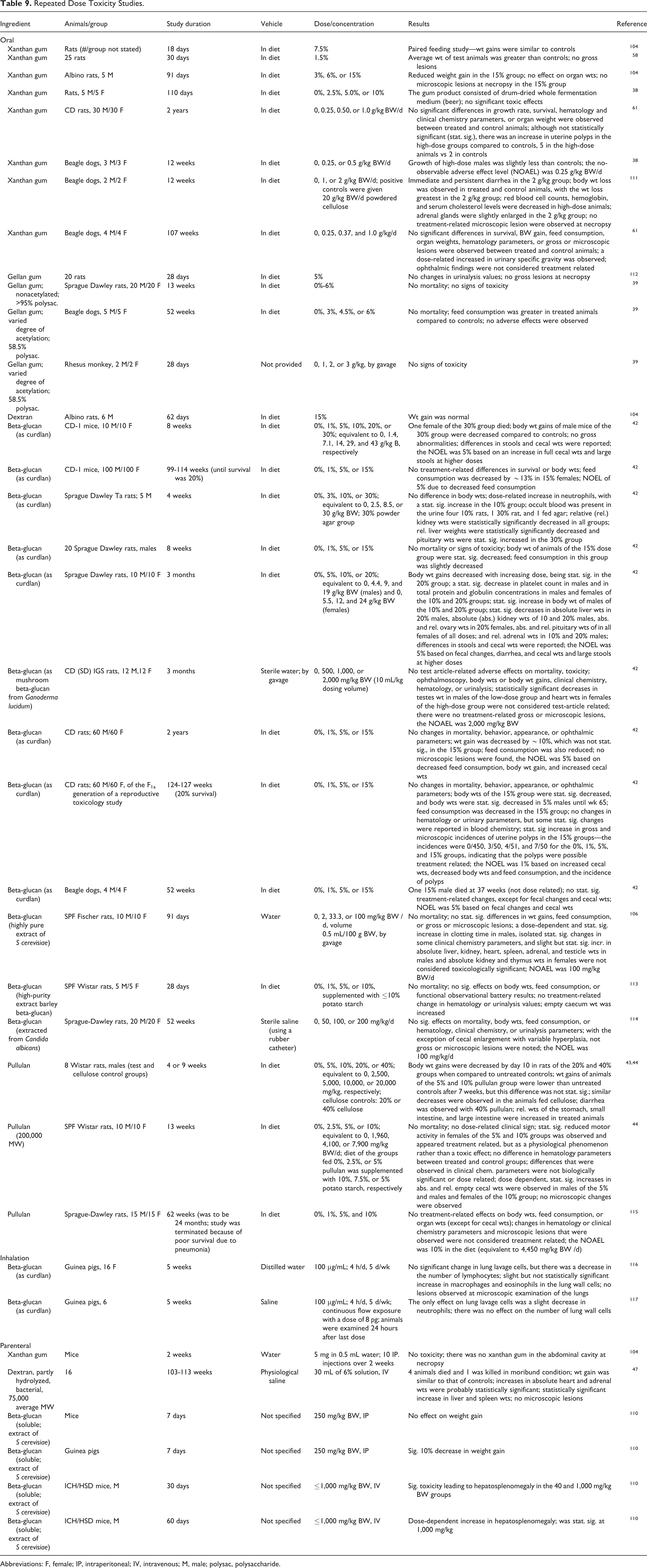
Abbreviations: F, female;
Oral intake by humans
Xanthan gum
Five male subjects consumed 150 mg/kg BW/d xanthan gum as 3 measured portions daily for 23 days. 58 Ingestion of xanthan gum had no significant adverse effect on hematology, clinical chemistry, or urinalysis parameters.
Gellan gum
Five male and 5 female subjects consumed a daily dose of 175 mg/kg gellan gum for 7 days and 200 mg/kg/d for the next 16 days. 58 No adverse dietary or physiological effects and no allergenic effects were reported.
Dextran and pullulan
No adverse effects were seen in a study in which 10 g pullulan, dextran, and soluble starch were ingested by 8 male volunteers. 42,59 Each ingredient was administered for 14 days, and there was a 14-day washout period between treatments.
Beta-glucan
Six male subjects ingested milkshakes with or without beta-glucan (as curdlan) for 28 days. 42 The test subjects were given 6 g/d for 5 days, 35 g/d for 2 weeks, and 50 g/d from days 21 to 28. No evidence of toxicity was observed.
Pullulan
In a tolerance study, 13 subjects ingested 10 g/d of pullulan (50,000 MW) for 14 days. 44,46 There were no effects on clinical chemistry parameters, and no adverse effects were reported.
Industrial exposure
Xanthan gum
The relationship between the handling of xanthan gum powder and adverse symptoms was examined using exposure groups based on average percentage of time spent in a plant that uses a fermentation process to manufacture xanthan gum, not on the basis of expected intensity of exposure to xanthan gum. 60 The exposure of interest was to the employees exposed to milling, blending, and packaging of the product; a total of 39 employees were surveyed. Analysis of the results found no significant acute or chronic effects in pulmonary function in any of the exposure groups.
Reproductive and Developmental Toxicity
Oral
Xanthan gum
A 3-generation reproductive toxicity study was performed in which albino rats were fed dietary levels of 0, 0.25, and 0.5 g/kg BW/d xanthan gum. 61 Ten males and 20 females were used for the first generation, and 20 males and 20 females were used in the next 2 generations. Rats were mated to produce 2 litters per generation, and the successive generations were selected from weanlings of the second litter. Survival and reproductive parameters were similar for treated and control parental rats. Body weights of treated parental rats were slightly decreased compared to controls in each generation. There were no significant differences in developmental parameters between test and control litters, and no malformations were observed in any of the offspring.
Gellan gum
Groups of 25 gravid female Sprague Dawley rats were fed a diet containing 0%, 2.5%, 3.8%, or 5.0% gellan gum (varied degree of acetylation; 58.5% polysaccharide) on days 6 to 15 of gestation. 39 No fetotoxic or teratogenic effects were reported (no other details were provided).
Beta-glucan
A developmental study was performed with 0%, 5%, or 15% beta-glucan (as curdlan) with a control group of 40 male and 80 female CD rats and test groups of 20 male and 40 female rats. 42 The animals, which were mated twice, were fed the test diet throughout the study. Twenty of the treated dams nursed their own litters; the other 20 treated dams switched litters with the control dams so that treated animals would nurse control pups and control animals would nurse test pups. The F1a offspring were killed prior to the second mating.
No changes in mortality, behavior, or appearance were observed. Male sires of the 15% beta-glucan group had decreased growth rates compared to controls, and males and females of the 15% group had decreased feed consumption. At birth, there were no differences in fertility or lactation among the groups, and no abnormalities were reported. However, survival of the F1a, but not the F1b, pups of the 5% group was statistically significantly decreased compared to controls. Weight gain of all F1a litters of treated dams that nursed their own pups was statistically significantly decreased compared to controls; for the F1b litters, the difference was statistically significant only in the 15% group. Statistically significant decreases in weight gain were also observed for pups of treated dams that were nursed by control dams, but the effect was reduced. Statistically significant decreased weight gain at some intervals was also observed for control pups nursed by treated dams. A no observable effect level (NOEL) was not established.
The researchers also examined whether there would be decreased weight gain by the pups if dosing was discontinued during lactation. The protocol was similar to that just described, except that all groups consisted of 20 male and 40 female CD rats, and there was no crossover at lactation. Weight gain by all pups during lactations was similar, although the researchers did state that the pups could have consumed parental diet from day 10+. The NOEL for parental toxicity and embryotoxicity was 15% beta-glucan.
A 3-generation reproductive study was performed in which groups of 20 male and 40 female CD rats were fed a diet containing 0%, 1%, 5%, or 15% beta-glucan (as curdlan) for 100 days. 42 The F0 parents were mated twice, and the number of parents was halved after weaning of the first litter. The F1 parents were mated 3 times and the F2 parents were mated twice. The F1b and F2b litters were used to produce the next generation. After the third mating of the F1 parents, half of the F1 dams were killed on day 13 of gestation, and the remaining dams were killed on day 20 of gestation.
Mean growth and feed consumption were slightly decreased in male parental rats of the F0 and F1 generations of the 15% group. No gross or microscopic changes were observed in F2 parents. No treatment-related effects on reproductive and developmental parameters were observed, but BWs of pups in almost all litters in all generations were statistically significantly decreased during lactation in the 15% group. Biologically significant differences in BWs were not seen in litters of the other dose groups. No gross or microscopic lesions were observed in the F3b pups of the 15% group. In the F1 parents killed after the third mating, no reproductive or developmental effects were observed. Mean fetal weights in all groups were statistically significantly decreased compared to controls; however, there was no dose response. The NOEL for parental animals was 5%, based on decreased growth and increased cecal weights at 15% beta-glucan, and the NOEL for embryotoxicity was also 5%, based on decreased weight gain during lactation in the 15% beta-glucan group.
The teratogenic potential of beta-glucan (as curdlan) was determined using groups of 15 to 20 gravid Dutch-belted rabbits. 42 The rabbits were dosed orally with 0, 1, 2, or 5 g/kg BW/d beta-glucan in a gelatin capsule delivered using a syringe. The 5 g/kg dose was administered as 2 divided doses, and the controls received 2 empty capsules. The rabbits were killed on day 28 of gestation. None of the controls died, but 1, 1, and 3 dams of the 1, 2, and 5 g/kg BW/day groups, respectively, died during the study. Eleven resorptions were observed in the high-dose group, as compared to 4 in the control group, 6 in the 1 g/kg group, and 5 in the 2 g/kg group. The researchers stated, however, that the number of dams with resorptions was similar in all groups and that no teratogenic effects were observed. The NOEL for both maternal and embryotoxicity was 5 g/kg BW/d.
Genotoxicity
Genotoxicity studies are summarized in Table 10. The in vitro genotoxicity of gellan gum (≤20 mg/mL), sodium dextran sulfate (≤25 mg/plate), beta-glucan (≤5,000 μg/plate or μg/mL), sodium carboxymethyl beta-glucan (≤50,000 μg/plate or μg/mL), and pullulan (≤12 mg/mL) was evaluated using Ames test, chromosomal aberration assays, and/or DNA repair tests, with and without metabolic activation. The results were negative in these tests. The only nonnegative result was a weak positive outcome with 20 mg/plate pullulan in a rec assay using
Genotoxicity Studies.
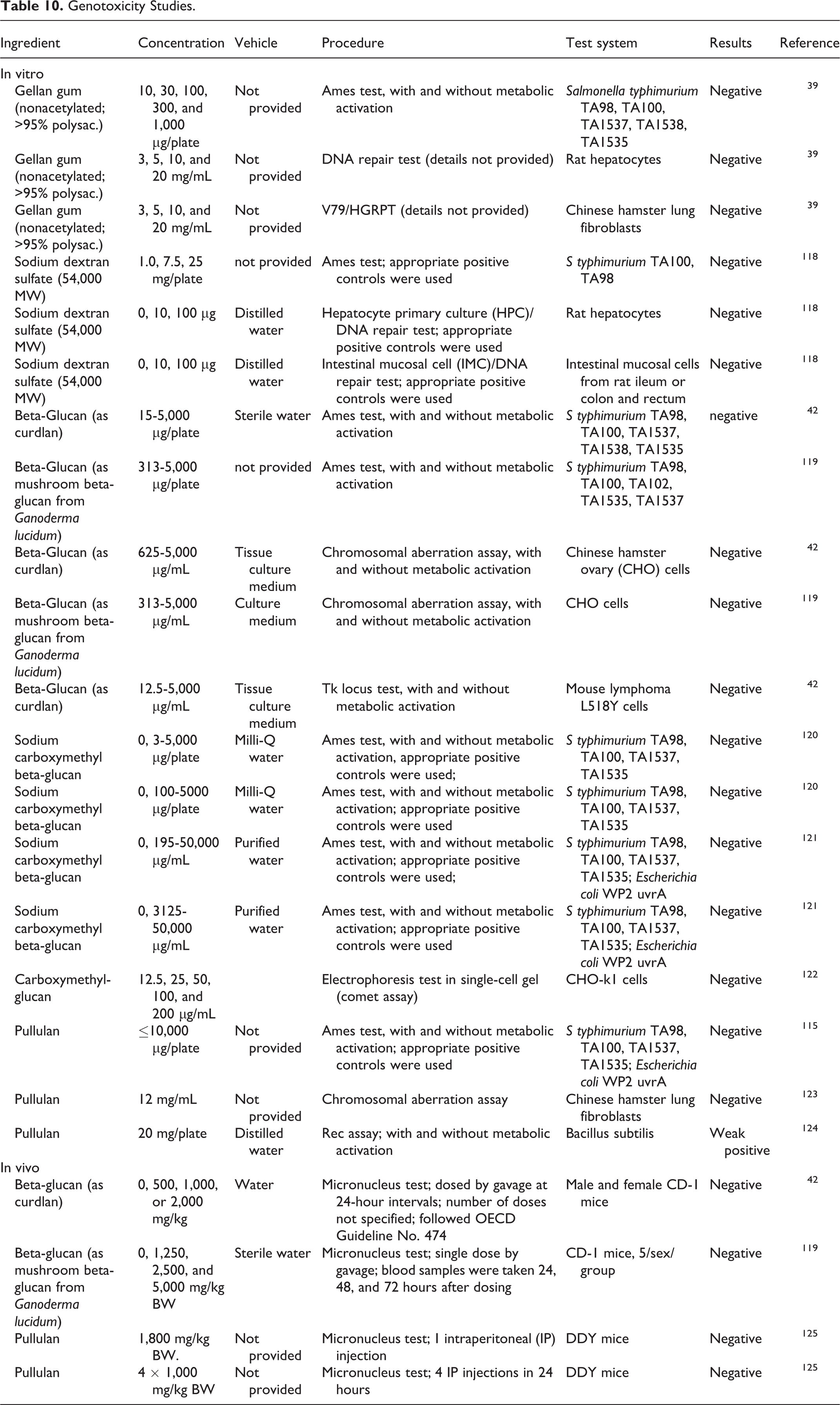
Carcinogenicity
Oral
Gellan gum
Groups of 50 male and 50 female Swiss Crl mice were fed a diet containing 0%, 1%, 2%, or 3% gellan gum (varied degree of acetylation; 58.5% polysaccharide) for 96 weeks (males) or 98 weeks (females). 39 No treatment-related effects on BWs or feed consumption were observed. No treatment-related neoplastic or nonneoplastic lesions were reported.
In another study, groups of 50 male and 50 female F1 generation Sprague Dawley rats were exposed in utero to gellan gum and were maintained on a diet containing 0%, 2.5%, 3.8%, or 5.0% gellan gum (varied degree of acetylation; 58.5% polysaccharide) for 104 weeks. 39 Survival of treated male rats was decreased when compared to controls, but survival of treated female rats was better than the concurrent controls. Male rats of the 3.8% and 5.0% test groups had decreased BWs compared to controls initially and after 76 weeks. However, the researchers stated that the growth pattern of these test animals was the same as that of the controls, and the lower BWs were not indicative of toxicity. There were no neoplastic or nonneoplastic lesions associated with dosing, and gellan gum was not carcinogenic when fed to Sprague Dawley rats.
Dextran
A group of 15 male and 15 female ACI rats were fed a diet containing 2.5% dextran (21,500 MW) for 480 days, and the control group of 20 males and 20 females was fed a basal diet. 62 Body weight gains of treated male rats were statistically significantly decreased compared to controls. An increased incidence in tumors was not reported, and no intestinal tumors were found.
Sodium dextran sulfate
A number of studies have demonstrated that oral exposure to sodium dextran sulfate produces colon cancer in rats; the mechanism is not genotoxic. Oral administration has been shown to induce colonic inflammation, and a 2-day study in which female Fischer 344 rats were given 3% or 6% sodium dextran sulfate in the drinking water indicated that oxidative DNA damage occurred in the colonic mucosa. 63 The MW of sodium dextran sulfate has been found to be a factor in carcinogenic activity. Although an extensive number of studies are available in the published literature, just a few representative studies are summarized below. An example of an inflammation-related mouse colon carcinogenesis model is described, with an indication of strain dependency.
In one study evaluating the carcinogenic potential of sodium dextran sulfate (54,000 average MW; sulfur content 18.9%) in ACI rats, 10 males were fed a diet containing 10% sodium dextran sulfate, 14 males and 12 females were fed 5% in the diet, and a control group of 9 males and 9 females were given a basal diet. 64 All animals were necropsied at natural death or when killed due to moribund condition. All animals in the 10% group died 6 to 14 days after initiation of dosing, and all had severe acute nephrosis. Two animals of the 5% group died on day 14, but most of the remainder of this group lived for more than 130 days. Blood was observed on the surface of the stools of these animals at 2.5 months. The weight gain of animals of this group was decreased compared to controls. Of the 23 rats that survived for more than 130 days, 15 rats developed intestinal tumors; tumors included 5 adenomas, 5 adenocarcinomas, and 3 papillomas in the colon and 6 adenomas and 2 adenocarcinomas in the cecum. Although most rats had a single tumor upon gross observation, microscopic examination found multicentric foci of atypical hyperplasia of the glandular epithelium. No intestinal tumors were reported in the control group.
In a second study testing the same sodium dextran sulfate, 15 male and 15 female ACI rats were fed 1% in the diet for 660 days; the average daily intake was 0.15 g/d/animal. 65 A control group of 10 males and 10 females were fed a basal diet. All but 2 treated male rats survived for 350+ days. Body weight gains of the test group were similar to that of the controls. Intestinal tumors were observed in 22 treated rats; 16 papillomas, 4 squamous cell carcinomas, 2 adenomas, and 5 adenocarcinomas were reported in the colon and rectum and 1 adenocarcinoma was found in the cecum. Thirteen tumors were reported at miscellaneous sites. No intestinal tumors were reported in the control group.
To examine the effect of MW, 3 groups of 15 male and 15 to 16 female ACI rats were fed for 480 days a diet containing 2.5% of a sodium dextran sulfate, MW 520,000, 54,000, or 9,500; each sodium dextran sulfate had a sulfur content of 18% to 19% 62 (the 54,000 MW substance was synthesized using the dextran described previously). A control group of 20 male and 20 female rats was fed a basal diet. There was no significant difference in survival time between any of the groups. Body weight gains of male rats of the 54,000 MW diet were statistically significantly decreased compared to control males; BW gains in the other 2 groups were similar to controls. The 54,000 MW sodium dextran sulfate had the strongest carcinogenic activity, with tumors being reported similar to the studies described previously. The other 2 MW substances did not show significant carcinogenic activity; only 2 colorectal adenocarcinomas were observed in the 520,000 MW group. Colorectal squamous metaplasia was observed in most rats in all test groups. Other miscellaneous tumors were reported, but there was no statistically significant difference between treated and control groups.
A colitis-related mouse colon carcinogenesis model was developed. 66 When 8 male Crj: CD-1 mice were given a single IP injection of 10 mg/kg azoxymethane (AOM), followed by 7 days of 2% sodium dextran sulfate in the drinking water 1 week later, there was a 100% incidence of colonic adenocarcinomas and 38% of adenomas at week 20. No adenocarcinomas were observed in any of the mice dosed with AOM and sodium dextran sulfate simultaneously, 1 week of sodium dextran sulfate, and then AOM, AOM only, or sodium dextran sulfate only, and only 2 adenomas (in 10 mice) were observed when AOM was given during sodium dextran sulfate administration. All of the adenocarcinomas were positive for β-catenin, cyclooxygenase 2, and inducible nitric oxide synthase and negative for immunoreactivity of p53.
Strain sensitivity to the above model was then examined. 67 In testing with male Balb/c, C3H/HeN, C57BL/6N, and DBA/2N mice, Balb/c mice were very sensitive to the model. C57BL/6N mice also developed colonic adenocarcinomas, but to a lesser extent. C3H/HeN and DBA/2N mice developed only a few colonic adenomas. However, the greatest inflammation response was observed in C3H/HeN mice, followed by Balb/c mice. The researchers hypothesized that the strain differences in susceptibility of colon carcinogenesis induced by AOM and sodium dextran sulfate might be influenced by the response to nitrosation stress due to inflammation as determined by the genetic background.
Antitumor Effects
Xanthan gum
Groups of C57BL/6 mice were inoculated subcutaneously (SC) with 1 × 106 B16Kb melanoma cells. 68 A suspension of 10 mg/mL xanthan gum, 100 μL, or PBS was given by gavage once every 5 days, starting 1 day prior to inoculation. Rapid tumor growth occurred in PBS-treated mice, but tumor growth was statistically significantly reduced in xanthan gum-treated mice. Spleen cell composition was analyzed 22 days after inoculation. There was no change in overall cellular composition, but natural killer cells and tumor-specific cytotoxic T-lymphocyte activity were increased by xanthan gum. All PBS-treated mice died by day 46 after inoculation; 40% of the xanthan gum-treated mice were alive at day 100. The researchers demonstrated that the antitumor effect of xanthan gum was highly dependent on Toll-like receptor 4–mediated signaling.
Pullulan
Male Balb/c mice were used to determine the antitumor and antimetastatic potential of pullulan (water-soluble low-MW β-(1 → 3) with 50%-80% branched β-(1 → 6); MW 100,000). 69 Colon-26 cells were implanted into the spleens of mice on day 0, and groups of mice were dosed orally with 25 or 50 mg/kg or IP with 5 or 15 mg/kg pullulan for 14 consecutive days, starting 12 hours after implantation. Control groups consisted of sham-operated mice (not implanted with colon-26 cells) and mice implanted with the cells but given physiological saline or distilled water instead of pullulan. Splenic tumor weights were reduced with the 50 mg/kg oral and 15 mg/kg IP doses of pullulan but not with the other doses. Liver metastasis was significantly inhibited with 50 (but not 25) mg/kg oral and 5 and 15 mg/kg IP pullulan.
Levan
The antitumor activity of levan produced from 4 different microorganisms, that is,
Irritation and Sensitization
Nonhuman and human dermal irritation and sensitization studies are summarized in Table 11. The dermal irritation and sensitization potentials of xanthan gum, beta-glucan, and sodium carboxymethyl beta-glucan were evaluated in animal studies. Xanthan gum, up to 1%, and beta-glucan, concentration not specified, were not irritating to rabbit skin. One study with 5% aq. xanthan gum on shaved rabbit skin produced localized irritation; study details were not provided. Neither xanthan gum, tested at 0.1%, nor beta-glucan (concentration not specified) were sensitizers in guinea pigs. Sodium carboxymethyl beta-glucan was at most a slight skin irritant in guinea pigs at a concentration of 50% aq.; a 10% aq. solution was not a sensitizer in guinea pigs.
Dermal Irritation and Sensitization.
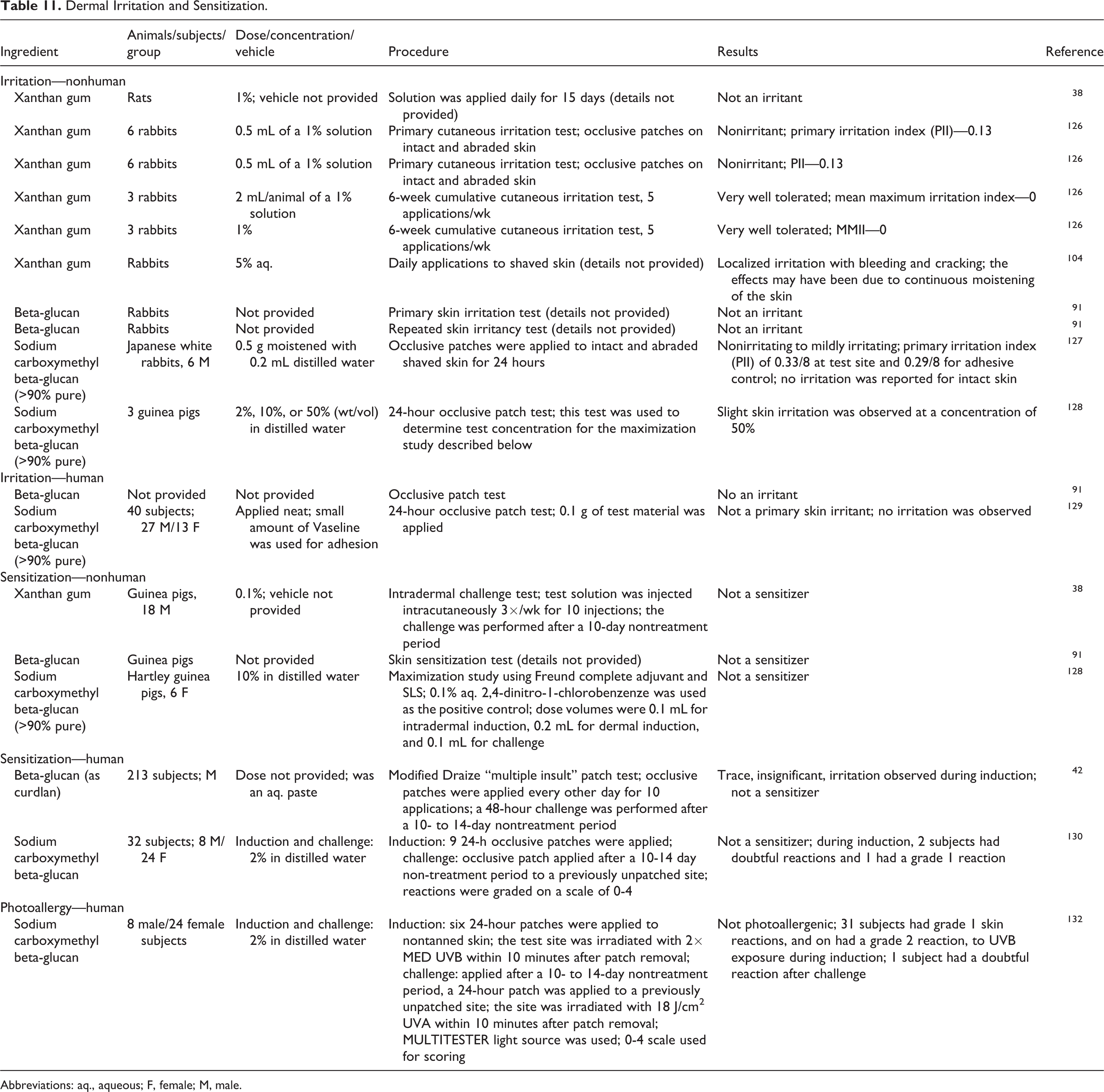
Abbreviations: aq., aqueous; F, female; M, male.
In humans, neither beta-glucan nor sodium carboxymethyl beta-glucan were irritants or sensitizers. The test concentration of beta-glucan was not specified. Sodium carboxymethyl beta-glucan was applied neat in the irritation study and as a 2% aq. solution in the sensitization study.
Phototoxicity
A human photoallergenicity study is also summarized in Table 11. A 2% aq. solution of sodium carboxymethyl beta-glucan was not photosensitizing in clinical studies.
Adverse Reactions
Dextran
Over a 5-year period (1981-1986), 12,646 dextran 70 U were administered to 5,745 patients (mean of 2.2 U/patient) undergoing gynecological surgery or a cesarean section as a plasma volume expander. 71 Fifteen immediate dextran-induced anaphylactoid reactions were reported, with an incidence of 1 reaction/383 patients treated. Life-threatening reactions occurred in 7 of these patients.
Ocular Irritation
Ocular irritation studies are summarized in Table 12. Xanthan gum, 1%, and up to 0.8% gellan gum were not ocular irritants in rabbit eyes, and up to 0.5% gellan gum was not irritating to human eyes. The ocular irritation potential of sodium carboxymethyl beta-glucan was described as weakly irritating in a HET-CAM assay but was practically nonirritating in rabbit eyes.
Ocular Irritation.

Summary
Microbial polysaccharide gums can be produced intercellularly, by the cell wall, or exocellularly. Exocellular polysaccharide gums constantly diffuse into the cell culture medium and are easily isolated, while cell wall and intercellular polysaccharide gums are more difficult to separate from cell biomass. Many of the 34 microbial polysaccharide gums discussed in this safety assessment are produced exocellularly. They are reported to have numerous functions in cosmetics, including emulsion stabilizer, film former, binder, viscosity-increasing agent, and skin-conditioning agent.
The same microbial polysaccharide gums can often be produced by more than 1 organism. For example, beta-glucan can be produced by fungi, yeasts, and grains and levan can be produced by bacteria, yeasts, or fungi. The properties of the microbial polysaccharide gums can vary widely based on, among other parameters, the side groups, the ester substituents, or bacterial strains. These polysaccharide gums are generally very large molecules, and the MW of each ingredient can vary considerably.
Xanthan gum is reported to be used in almost every category of cosmetic product, with 3,470 reported uses. Biosaccharide gum-1, sclerotium gum, and beta-glucan are reported to be used in 346, 193, and 137 cosmetic formulations, respectively. All other in-use ingredients have less than 70 uses. The ingredient with the highest concentration of use is pullulan; it is used at up to 12% in leave-on formulations and 17% in a breath freshener. Both xanthan gum and biosaccharide gum-1 are used at up to 6% in leave-on formulations and xanthan gum crosspolymer and biosaccharide gum-4 are used at 5% in leave-on formulations. All other in-use ingredients are used at concentrations of ≤3%.
Xanthan gum, gellan gum, and beta-glucan are approved as direct food additives, and xanthan gum and dextran are approved indirect food additives. Xanthan gum and dextran also have pharmaceutical applications.
Xanthan gum, orally administered, is slowly broken down in the gut by enzymatic and nonenzymatic mechanisms and can be absorbed in some form to some extent. The absorbed fraction does not accumulate in the tissues and can be completely metabolized to CO2. Gellan gum, orally administered, does not breakdown to any substantial extent in the gut and is only very poorly absorbed.
Dextrans, 3,000 to 20,000 MW, are not absorbed through intact skin in humans. They are absorbed through the dermis (up to 38% for 3,000 MW) if the epidermis is removed (eg, via mini erosion); absorption in this case is inversely proportional to MW. Orally administered dextrans, 4,400 to 40,500 MW, are not absorbed to any appreciable extent in the gut (very low bioavailability). If dextrans were absorbed to a significant extent through the skin, animal studies in which dextrans were administered IV indicate that their half-lives in blood plasma would be directly related to the MW; they would be excreted in the urine to an extent that is inversely related to the MW; the lower MW dextrans would tend to be readily excreted unchanged in the urine; and the higher MW dextrans would have the potential to accumulate and to be broken down in the liver and other tissues and would be more likely to be excreted in the urine in a dose-dependent manner than the lower MW dextrans. Dextran sulfate, 7,000 to 8,000 MW, orally administered, is only very poorly absorbed in the human GI tract. After IV administration, it is rapidly excreted, mostly intact, in the urine, although at least some of it can accumulate in the tissues (to a substantially greater extent than observed after oral exposure), and some of it appears to be incorporated into glycogen and other substances in the body.
Beta-glucan in a topically applied solution can penetrate into the epidermis and dermis. There appears to be no information about its absorption through the skin into the bloodstream. Orally administered, it is readily metabolized to CO2, at least partially by microflora in the gut. Beta-glucan is not well absorbed or eliminated after IP injection. Pullulan, orally administered, is hydrolyzed to some small extent in the gut. It is partially hydrolyzed by amylases in the upper GI tract of human subjects and is subject to breakdown by intestinal microflora to form short-chain fatty acids; the rate of the latter depends on the degree of polymerization. However, pullulan can be essentially completely broken down to short-chain fatty acids in the human gut. It has not been determined to what extent, if any, the products of hydrolysis can be absorbed.
The acute toxicity of xanthan gum, gellan gum, beta-glucan, sodium carboxymethyl beta-glucan, and pullulan was assessed orally in mice, rats, and/or dogs, and dextran sulfate and beta-glucan were tested by IP and IV dosing in mice and rats. There was no notable toxicity observed in these studies. In acute inhalation studies, the LC50 of xanthan gum was >21 mg/L in rabbits and of gellan gum was >5.06 mg/L in rats. A single 180-minute exposure of humans office dust containing 10 mg curdlin/g dust resulted in decreased nasal volume, swelling in the nasal turbinates, and an increase in nasal eosinophils when compared to “clean” dust.
The inflammatory response following a single exposure to beta-glucan (as curdlan) and to pullulan was also evaluated in guinea pigs. Mostly, no effect or a slight decrease in inflammatory cells in lung lavage was observed. A 4-hour inhalation exposure to beta-glucan in dust by guinea pigs produced a delayed subacute nasal congestion when compared to dust without beta-glucan and resulted in decreased nasal volume. Industrial exposure to xanthan gum powder did not appear to cause significant acute or chronic pulmonary effects. Using artificial skin, 5% levan had an anti-inflammatory effect in irritated skin.
Repeated dose oral toxicity of xanthan gum was evaluated in rats and dogs, of gellan gum in rats, dogs, and monkeys, of dextran in rats, of beta-glucan in mice, rats, and dogs, and of pullulan in rats. Most of the studies were dietary, and study durations lasted up to 2 years. Most observations were related to changes in feed consumption and intestinal effects. Inhalation exposure to 100 μg/mL beta-glucan (as curdlan), 4 h/d, 5 d/wk for 4 weeks, did not have an effect on the cells of the lung lavage or cell wall, and there were no microscopic lesions of the lung. With IP administration, no toxicity was reported when mice were dosed 10 times over 2 weeks with 5 mg xanthan gum in 0.5 mL water or mice or guinea pigs were dosed with 250 mg/kg BW beta-glucan for 7 days. Intravenous administration of 40 and 1,000 mg/kg BW beta-glucan for 30 days resulted in hepatosplenomegaly in mice.
No toxic effects were observed in human subjects with oral ingestion of 150 mg/kg/d xanthan gum or 175 to 200 mg/kg/d gellan gum for 23 days, 10 g of dextran or pullulan for 14 days, or 6 to 50 g/d beta-glucan for up to 28 days. No significant or chronic effects in pulmonary function were reported in groups exposed occupationally to xanthan gum.
Dietary reproductive and developmental toxicity studies were conducted with xanthan gum, gellan gum, and beta-glucan. No reproductive or developmental effects were reported in a 3-generation reproductive study in which rats were fed diets containing up to 5 g/kg BW/day xanthan gum. Gellan gum, up to 5%, did not have a fetotoxic or teratogenic effect on rats. Dietary administration of beta-glucan in rats in reproductive and developmental studies did not have any reproductive effects, but there were statistically significant decreases in BWs and BW gains in offspring and parental animals. In a teratogenicity study in which rabbits were dosed with 5 g/kg BW/d beta-glucan, an increase in resorptions in the 5 g/kg group was considered similar to the other test groups and the controls, and 5 g/kg beta-glucan was not teratogenic in rabbits.
The in vitro genotoxicity of gellan gum (≤20 mg/mL), sodium dextran sulfate (≤25 mg/plate), beta-glucan (≤5,000 μg/plate or /mL), sodium carboxymethyl beta-glucan (≤50,000 μg/plate or /mL), and pullulan (≤12 mg/mL) was evaluated in Ames test, chromosomal aberration assays, and/or DNA repair tests, with and without metabolic activation. Results were negative in all of these tests. The only nonnegative result was a weak positive result with 20 mg/plate pullulan in a rec assay using
Dietary studies examining the carcinogenic potential of ≤5% gellan gum and 2.5% dextran reported that neither of these ingredients caused an increase in tumors. However, a number of studies have demonstrated that oral exposure to sodium dextran sulfate produces colon carcinogenesis in rats; the mechanism is nongenotoxic. In one study, the MW of sodium dextran sulfate was a factor in carcinogenic activity; a 54,000 MW sodium dextran sulfate produced colorectal tumors, but 9,500 and 520,000 MW sodium dextran sulfate did not have significant carcinogenic activity. Oral administration has been shown to induce colonic inflammation, and a 2-day study in which female Fischer 344 rats were given 3% or 6% sodium dextran sulfate in the drinking water indicated that oxidative DNA damage occurred in the colonic mucosa. An inflammation-related mouse colon carcinogenesis model indicated that the development of colonic tumors is strain dependent and that Balb/c mice were very sensitive to the model. C57BL/6N mice also developed tumors, but to a lesser extent, whereas C3H/HeN and DBA/2N mice only developed a few tumors.
Oral administration of 10 mg/mL xanthan gum had an antitumor effect in mice inoculated with melanoma cells, and 50 mg/kg pullulan, but not 15 mg/kg, significantly inhibited tumor growth in mice following implantation of colon-26 cells. Levan did not have an antitumor effect in mice.
Nonhuman and human dermal irritation and sensitization studies are summarized in Table 11. The dermal irritation and sensitization potentials of xanthan gum, beta-glucan, and sodium carboxymethyl beta-glucan were evaluated in animal studies. Xanthan gum, up to 1%, and beta-glucan, concentration not specified, were not irritating to rabbit skin. One study with 5% aq. xanthan gum on shaved rabbit skin produced localized irritation; however, study details were not provided. Neither xanthan gum, tested at 0.1%, nor beta-glucan (concentration not specified) was sensitizers in guinea pigs. Sodium carboxymethyl beta-glucan was at most a slight skin irritant in guinea pigs at a concentration of 50% aq.; a 10% aq. solution was not a sensitizer in guinea pigs.
In humans, neither beta-glucan nor sodium carboxymethyl beta-glucan was irritants or sensitizers. The test concentration of beta-glucan was not specified. Sodium carboxymethyl beta-glucan was applied neat in the irritation study and as a 2% aq. solution in the sensitization study. A 2% aq. solution of sodium carboxymethyl beta-glucan was not photosensitizing in clinical studies.
Xanthan gum, 1%, and gellan gum, up to 0.8%, were not ocular irritants in rabbit eyes, and up to 0.5% gellan gum was not irritating to human eyes. The ocular irritation potential of sodium carboxymethyl beta-glucan was described as weakly irritating in a HET-CAM assay, but the ingredient was practically nonirritating in rabbit eyes.
Discussion
Microbial polysaccharide gums are produced by a wide variety of microorganisms, and some can also be isolated from plants, for example, beta-glucan can be isolated from barley and oats. Although these ingredients are produced primarily by microbial sources, the cosmetic ingredients are purified during manufacture, thus, microbial contamination is not a concern.
The Panel determined that, even though there are some data gaps, the available data on polysaccharide gums included in this safety assessment may be extrapolated to support the safety of the entire group. And although there was no specific data on the hydroxypropyltrimonium chloride compounds, data included in a previous CIR safety assessment on trimonium ingredients are applicable in determining the safety of the 3 hydroxypropyltrimonium chloride compounds included in this report.
Parenterally administered polysaccharides appear to be biotransformed to a limited but variable extent in animal and human studies. However, these very large compounds appear not to be significantly absorbed through the skin and would have negligible bioavailability. Coupled with a lack of significant toxicity associated with other routes of exposure, the CIR Expert Panel determined that systemic effects were unlikely to result from topical application of cosmetics containing these ingredients.
One study reported that 5% aq. xanthan gum caused irritation. However, this was the only finding of irritation among almost 20 studies on microbial polysaccharide gums. Given the absence of study details, including no mention of a control group, the Panel concluded that the irritation likely was the result of the study methodology (eg, shaved skin) and not by the xanthan gum. There was no evidence of sensitization in human or nonhuman testing.
The Panel also remarked on the induction of colon cancer in rodents with oral exposure to sodium dextran sulfate. Sodium dextran sulfate is a commonly used model for induction of colitis in a well-characterized mouse model to study colitis, and while the mode of action is unknown, it is not relevant to human cosmetic exposure.
Finally, the Panel discussed the issue of incidental inhalation exposure to microbial polysaccharide gums from powders and other products that may be aerosolized. These ingredients are reportedly used at concentrations up to 1% in cosmetic products that may be aerosolized and up to 6% in other products that may become airborne. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of this ingredient. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. Results of acute inhalation studies with xanthan gum and gellan gum produced no significant toxicity nor did results from a 4-week inhalation study of beta-glucan in guinea pigs. Industrial exposure to xanthan gum powder caused no significant acute or chronic effects in pulmonary function. The Panel also considered the data available to characterize the potential for microbial polysaccharide gums to cause systemic toxicity, irritation, sensitization, or other effects. The Panel noted that testing with a number of microbial polysaccharide gums demonstrated they did not produce systemic toxicity in oral studies; they are not reproductive or developmental toxicants, are not genotoxic, and are not considered irritants or sensitizers. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded the microbial polysaccharide gums listed below are safe in the present practices of use and concentration in cosmetics. Xanthan gum Hydroxypropyl xanthan gum* Undecylenoyl xanthan gum* Dehydroxanthan gum Xanthan gum crosspolymer Xanthan hydroxypropyltrimonium chloride* Gellan gum Welan gum* Biosaccharide gum-1 Biosaccharide gum-2 Biosaccharide gum-3* Biosaccharide gum-4 Biosaccharide gum-5* Pseudoalteromonas exopolysaccharides* Dextran Carboxymethyl dextran* Dextran hydroxypropyltrimonium chloride* Sodium carboxymethyl dextran Dextran sulfate Sodium dextran sulfate Sclerotium gum Hydrolyzed sclerotium gum Beta-glucan Beta-glucan hydroxypropyltrimonium chloride* Beta-glucan palmitate* Hydrolyzed beta-glucan* Oxidized beta-glucan* Sodium carboxymethyl beta-glucan Pullulan Myristoyl pullulan* Levan* Rhizobian gum Hydrolyzed rhizobian gum Alcaligenes polysaccharides
*Ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC, USA.
Author Contributions
M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. F. Alan Andersen, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
