Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel assessed the safety of 131 alkyl polyethylene glycol (PEG)/polypropylene glycol ethers as used in cosmetics, concluding that these ingredients are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonirritating. Most of the alkyl PEG/PPG ethers included in this review are reported to function in cosmetics as surfactants, skin-conditioning agents, and/or emulsifying agents. The alkyl PEG/PPG ethers share very similar physiochemical properties as the alkyl PEG ethers, which were reviewed previously by the CIR Expert Panel and found safe when formulated to be nonirritating. The alkyl PEG ethers differ by the inclusion of PPG repeat units, which are used to fine-tune the surfactant properties of this group. The Panel relied heavily on data on analogous ingredients, extracted from the alkyl PEG ethers and PPG reports, when making its determination of safety.
Introduction
This report assesses the safety of 131 alkyl polyethylene glycol (PEG)/polypropylene glycol (PPG) ethers (listed in Table 1) defined in the International Cosmetic Ingredient Dictionary and Handbook as cosmetic ingredients. 1 Most of the alkyl PEG/PPG ethers included in this review are reported to function in cosmetics as surfactants, skin-conditioning agents, and/or emulsifying agents.
Alkyl PEG/PPG Ethers Included in This Assessment.
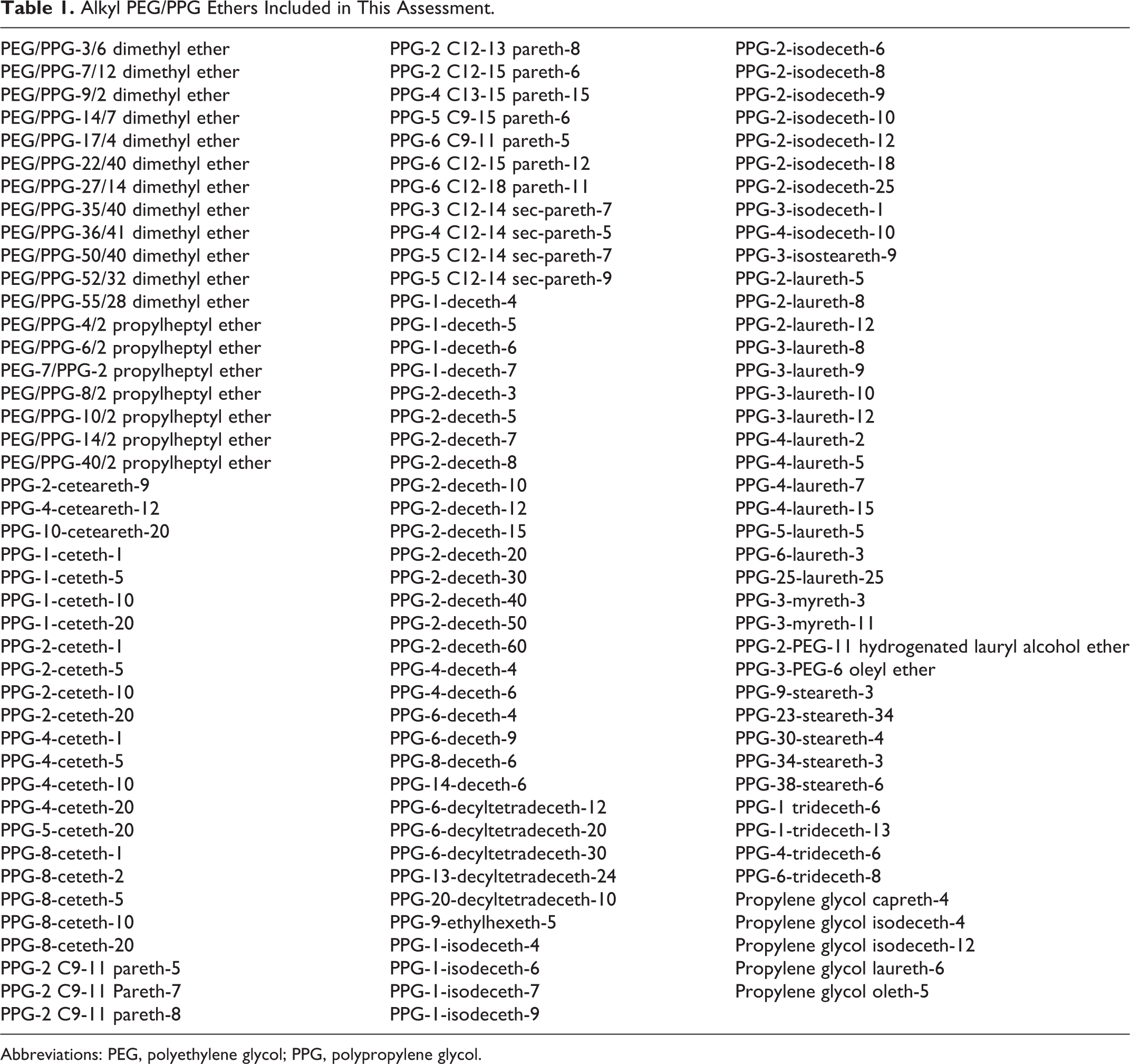
Abbreviations: PEG, polyethylene glycol; PPG, polypropylene glycol.
The alkyl PEG/PPG ethers are not expected to metabolize to individual components; therefore, information from previous Cosmetic Ingredient Review (CIR) safety assessments on the individual alcohols is not relevant in this safety assessment. However, the CIR Expert Panel’s determination that 369 alkyl PEG ethers (as well as future alkyl PEG ether cosmetic ingredients that vary from those 369 ethers only by the number of ethylene glycol repeat units) and PPGs ≥3 are safe as used when formulated to be nonirritating 2,3 is relevant because these ingredients share very similar physiochemical profiles, with an internal mixture of various hydrophobicities/hydrophilicities, as expected in these sorts of alkoxyl-based, surfactant-like molecules. 4 The only difference between alkyl PEG ethers and alkyl PEG/PPG ethers is the inclusion of PPG repeat units which is simply used to fine-tune the surfactant properties of these ingredients.
Although there are very little data available on the alkyl PEG/PPG ether ingredients, data on analogous ingredients provide a good indication of the lack of toxicity of these ingredients.
Chemistry
Definition and Structure
Alkyl PEG/PPG ethers are the reaction products of an alkyl alcohol and one or more equivalents each of ethylene oxide and propylene oxide (forming repeats of PEG and PPG, respectively; Figure 1).
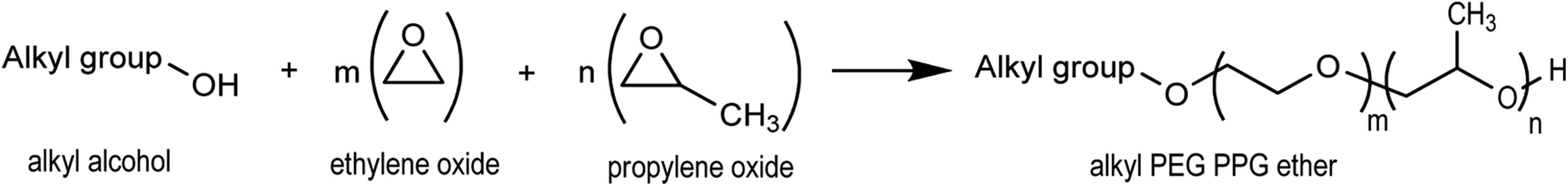
Alkyl polyethylene glycol (PEG)/polypropylene glycol (PPG) ether synthesis.
The definition of each ingredient, as given in the International Cosmetic Ingredient Dictionary and Handbook, is provided in Table 2. 1
Definitions, Structures, and, Functions.

Abbreviation: Cosmetic Ingredient Review.
aCIR staff drew the structures.
PPG-2-laureth-5 represents one of the simplest ingredients in this review as the reaction product of lauryl alcohol, 5 equivalents of ethylene oxide, and 2 equivalents of propylene oxide.
Each of the alkyl PEG/PPG ethers is a surfactant-like molecule, with a chain structure that has a hydrophobic end and a hydrophilic end. Principally, these ingredients differ by variation in the alkyl chain length at the hydrophobic end and the number of alkoxide (PEG and PPG) repeat units at the polyalkoxide, hydrophilic end. The structures in this report are drawn as block-type, alkoxide polymers only for simplicity’s sake. The actual order of alkoxide repeats in each ingredient, and from each source of an ingredient, may be block, alternating, or random.
There are a number of nomenclature conventions to be aware of in this group. For example, PEG-4-PPG-7 C13/C15 alcohol is an ingredient wherein the alkyl chain is variably 13 or 15 carbons long (“C13/C15 alcohol”), and the polyalkoxide end consists of an average of 4 ethylene glycol repeats and 7 propylene glycol repeats (“PEG-4-PPG-7”). PPG-2-laureth-5 is an ingredient (as shown in Figure 2) wherein the alkyl chain is derived from lauryl alcohol (ie, is 12 carbons long; “laur”) and the polyalkoxide end consists of an average of 5 ethylene glycol repeats (“eth-5”) and 2 propylene glycol repeats (“PPG-2”). As an example of a further variation in the naming convention, PEG/PPG-40/2 propylheptyl ether is an ingredient wherein the hydrophobic end is a 7-carbon alkyl chain (“heptyl”), with a 3-carbon branch at the 2-position (“propyl”; this naming convention indicates a Guerbet alcohol and thus substitution at the 2 position), and the poly-alkoxide end is comprised of an average of 40 ethylene glycol repeats and 2 propylene glycol repeats (“PEG/PPG-40/2”).
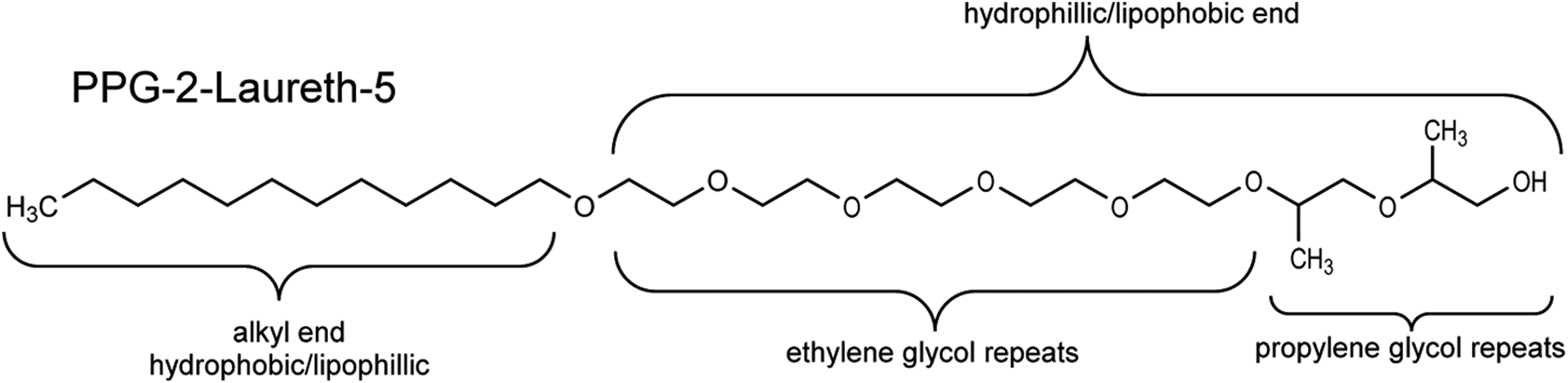
Alkyl polyethylene glycol (PEG)/polypropylene glycol (PPG) ether structure—for example, PPG-2-laureth-5.
The dimethyl ethers are distinct in this group by being capped at both ends with methyl groups, instead of having 1 alkyl chain at 1 end. For example, PEG/PPG-3/6 dimethyl ether is an ingredient wherein 1 carbon is at each end of a polyoxide chain, comprised of an average of 3 ethylene glycol repeats and 6 propylene glycol repeats.
Physical and Chemical Properties
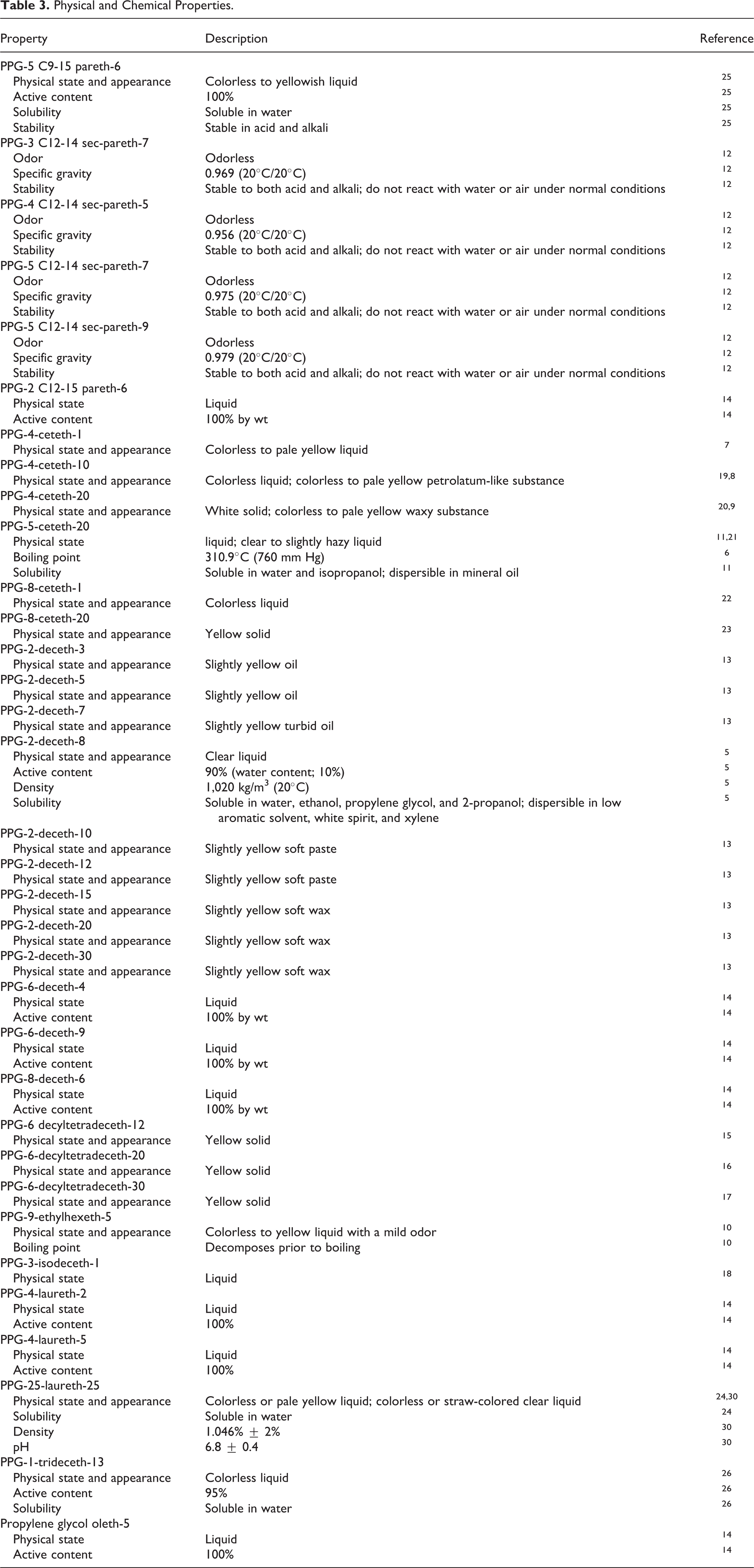
Physical and chemical properties data on the alkyl PEG/PPG ethers are provided in Table 3. 5 –26 Very few published data on specific properties were available, other than most of the alkyl PEG/PPG ethers are clear to slightly yellow liquids.
Physical and Chemical Properties.
The alkyl PEG/PPG ethers, such as alkoxylate polymers, are generally not defined as a single compound but as a mixture of a homologous series with a medium-range molecular weight and a specific percentage by weight of the hydrophobic tail. 27 The degree of hydrophobicity and hydrophilicity are fine-tuned by the components that make up each ether. The hydrophobicity of the product can be controlled by the fatty alcohol used and the distribution of the propylene glycol block; alternatively, the hydrophilicity is controlled by varying the length and position of the ethylene glycol repeats.
Method of Manufacture
The manufacture of alkyl PEG/PPG ethers consists of a number of variable steps. 28 The first step typically involves activating the alkyl alcohol (eg, lauryl alcohol) with a metal hydroxide (eg, potassium hydroxide), thereby generating an alkoxide (eg, lauroxide; ie, the initiator). This alkoxide is then reacted with ethylene oxide, propylene oxide, or a mixture of both (a mixture for random polyalkoxides and consecutively for block polyalkoxides; ie, propagation). The propagation of the polyalkoxide is then terminated with a Brønsted-Lowry–type acid (eg, hydrochloric acid), or in the case of the dimethyl ethers, a methyl halide (eg, methyl iodide). This synthetic pathway (specifically, the addition of ethoxide) can potentially lead to the generation of some 1,4-dioxane; however, this by-product can be monitored easily by gas chromatography and minimized via suitable process and purification accommodations.
Inclusion of propylene oxide into nonionic surfactants can be accomplished by (1) placement of a single block of propylene oxide between the alcohol and a block of ethylene oxide; (2) by placing a single block of propylene oxide after a single block of ethylene oxide; (3) by direct placement into the polyoxyethylene portion as a propylene oxide block or as an ethylene oxide–propylene oxide mix; or (4) by placing a single propylene oxide block in the middle of the ethylene oxide chain. 29 The propylene oxide placement affects the physical and surface active properties.
Impurities
No published impurities data were found, other than one source stating that PPG-25-laureth-25 contains ≤10 mg/L 1,4-dioxane. 30
It is not expected that there would be any significant amount of the residual starting materials used in the manufacture of the alkyl PEG/PPG ethers (ie, ethylene oxide and propylene oxide) or any significant amount of the residual by-product 1,4-dioxane found in these ingredients. Since these are volatile compounds, any levels present are expected to be low.
Formulators are advised to keep the levels of these starting materials and residual byproducts low. The National Toxicology Program Report on Carcinogens, Twelfth Edition, states that ethylene oxide is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans, including epidemiological studies and studies on mechanisms of carcinogenesis, 31 and that propylene oxide and 1,4-dioxane are reasonably anticipated to be human carcinogens based on sufficient evidence of carcinogenicity from studies in experimental animals. 32,33 The International Agency for Research on Cancer (IARC) concluded there is limited evidence in humans and sufficient evidence in experimental animals for the carcinogenicity of ethylene oxide, with an overall evaluation that ethylene oxide is carcinogenic to humans. 34 For propylene oxide and 1,4-dioxane, the IARC concluded there is inadequate evidence in humans, and there is sufficient evidence in experimental animals for carcinogenicity, with an overall evaluation that propylene oxide is possibly carcinogenic to humans. 34,35
Use
Cosmetic
The alkyl PEG/PPG ethers included in this review are reported to function in cosmetics mostly as surfactants, skin-conditioning agents, and/or emulsifying agents. 1 The function(s) of each ingredient are provided in Table 2.
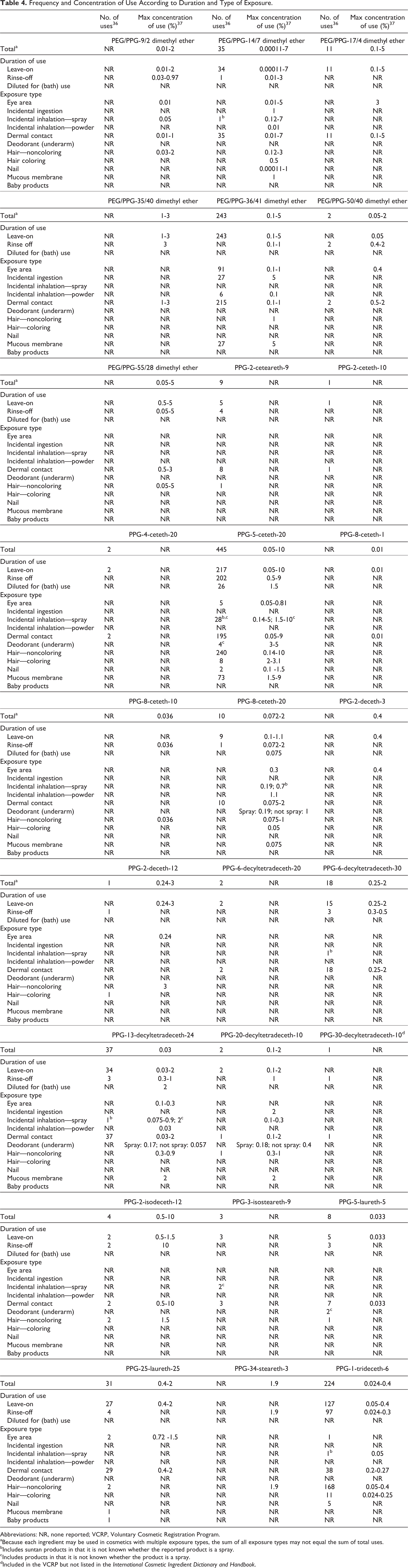
The Food and Drug Administration (FDA) collects information from manufacturers on the use of individual ingredients in cosmetic formulations as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the FDA in 2013 36 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 37,38 indicate that 26 of the 131 alkyl PEG/PPG ethers named in this safety assessment are currently used in cosmetic formulations; additionally, according to VCRP data, PPG-30-decyltetradeceth-10, an ingredient not named in the International Cosmetic Ingredient Dictionary and Handbook, has one reported use. PPG-5-ceteth-20 has the most reported uses (445), followed by and PEG/PPG-36/41 dimethyl ether (243 reported uses) and PPG-1-trideceth-6 (224 reported uses). All other in-use ingredients have less than 40 reported uses (Table 4).
Frequency and Concentration of Use According to Duration and Type of Exposure.
Abbreviations: NR, none reported; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products in that it is not known whether the reported product is a spray.
cIncludes products in that it is not known whether the product is a spray.
dIncluded in the VCRP but not listed in the International Cosmetic Ingredient Dictionary and Handbook.
According to the results of the concentration of use survey, PPG-5-ceteth-20 and PEG/PPG-14/7 dimethyl ether have the highest reported concentrations of use in leave-on formulations; PPG-5-ceteth-20 is used at up to 10% in “other” fragrance preparations and in tonics, dressings, and other hair grooming aids, and PEG/PPG-14/7 dimethyl ether is used at up to 7% in face and neck products and body and hand products. PPG-2-isodeceth-12 has the highest use concentration in rinse-off products; it is used at up to 10% in paste masks and mud packs. All other in-use alkyl PEG/PPG ethers are reported to be used in leave-on products at concentrations of 5% or below (Table 4). The 105 alkyl PEG/PPG ethers not reported to be in use are listed in Table 5.
In some cases, reports of uses were received in the VCRP, but no concentration of use data are available. For example, PPG-2-ceteareth-9 is reported to be used in 9 cosmetic formulations, but no concentration of use data were reported. Additionally, for some ingredients, no reported uses were received in the VCRP, but a use concentration was provided in the industry survey. For example, PEG/PPG-55/28 dimethyl ether was not reported to be in use in the VCRP, but the industry survey indicated that it is used at up to 5% in shampoos and other noncoloring hair preparations and at 3% in face and neck formulations. It should be presumed in these cases that there is at least one use in every category for which a concentration is reported.
Some alkyl PEG/PPG ethers are reported to be used in products that are applied to baby skin (eg, PPG-25-laureth-25 has one reported used in other baby products), to the eye area, or to the mucous membranes (eg, PEG/PPG-14/7 dimethyl ether is used in eye lotions at 5%) or that could possibly be ingested (eg, PEG/PPG-36/41 dimethyl ether is used at 5% in lipsticks). Additionally, some of the alkyl esters are used in cosmetic sprays and could possibly be inhaled; the maximum reported use in spray formulation is 7% PEG/PPG-14/7 dimethyl ether in spray body and hand products. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles <10 μm compared with pump sprays. 39,40 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 41,42
Some of the alkyl PEG/PPG ethers are used in spray deodorant products at low concentrations of use; the highest reported use concentration of this type was 0.19% PPG-8-ceteth-20. 37 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 42 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays compared to other cosmetic sprays.
All of the alkyl PEG/PPG ethers included in this report appear in the European Commission database with information on Cosmetic Ingredients and Substances (CosIng) inventory. 43 Listing in the inventory does not indicate the ingredients are actually used in cosmetic products or approved for such use.
Noncosmetic
In Europe, PPG-4-trideceth-6 can be used at 0.05 mg/kg in food contact materials; it is to be used only in polytetrafluoroethylene (PTFE) items sintered at high temperatures (PTFE is the nonstick coating used on cookware). 44,45
The inclusion of PPG-5-ceteth-20 in the development of dermal 46 and nasal 47 drug delivery systems is being evaluated. 46 In both studies, systems composed of PPG-5-ceteth-20, oleic acid, and water were used to form thermodynamically stable microemulsions that could phase into a liquid crystalline matrix. Other examples of noncosmetic industrial uses are provided in Table 6.5,12,25
Examples of Noncosmetic Uses.

Toxicokinetics
Published absorption, distribution, metabolism, and excretion data were not found. However, data on analogous compounds are available from the previous safety assessments of the alkyl PEG ethers and PPGs. These data suggest that alkyl PEG ethers are readily absorbed through the skin and mucosa of test animals and are quickly eliminated from the body through the urine, feces, and expired air. Dermal metabolism studies of laureth-1 and laureth-10 solutions indicate that in hairless mice, the 4-hour percutaneous absorption decreased from 22.9% for laureth-1 to 2.1% for laureth-10 solutions, 0.25% in ethanol. Compounds analogous to laureth-9 readily penetrated the skin of rats, and approximately 50% of the absorbed dose was excreted. Using humans, the majority of the dose could be wiped away from the test site after 8 hours; less than 2% was found in the urine. With atopic patients, the calculated dermal absorption rate for laureth-9 was 0.0017% for a diluted bath oil and 0.0035% with after-shower application. For PEG-3 methyl ether, however, in vitro absorption data indicated that it would not readily penetrate the skin. 2 Similarly, previous studies using PPG indicated that PPGs are readily absorbed from the gastrointestinal tract and excreted in the urine and feces.
Dermal penetration of propylene glycol from a ternary cosolvent solution through hairless mouse skin was 57% over a 24-hour period. However, in a study in which propylene glycol was applied to the finger tip of a human, the results of thermal emission decay–Fourier transform infrared spectroscopy indicated that propylene glycol did not reach the dermis.
Penetration Enhancement
PPG-4-ceteth-20 did not enhance the penetration of tenoxicam, a nonsteroidal anti-inflammatory drug, through guinea pig skin. 48 An in vitro study was performed in which the permeation through guinea pig skin of a 1.0% tenoxicam suspension containing 10% propylene glycol and 5.0% PPG-4-ceteth-20 was compared to that of tenoxicam without the surfactant. One gram of the test material was applied to the skin sample, and the receptor fluid was sampled every 3 hours for 48 hours. The steady-state flux was 8.11 ± 0.56 × 10−5 μg/s·cm2 for tenoxicam without surfactant and 7.28 ± 0.94 × 10−5 μg/s·cm2 for tenoxicam with PPG-4-ceteth-20; penetration rates were not statistically significantly different.
Toxicological Studies
Published toxicity studies were not found. However, data on analogous compounds are available from previous safety assessments of the alkyl PEG ethers and PPGs. 2,3,5
Reproductive and Developmental Toxicity
No published reproductive and developmental toxicity studies were found. However, data on analogous compounds are available from the safety assessments of the alkyl PEG ethers and PPGs.
Genotoxicity
No published genotoxicity studies were found. However, data on analogous compounds are available from the safety assessments of the alkyl PEG ethers, and PPGs show that these compounds were not mutagenic or genotoxic.
Carcinogenicity
No published carcinogenicity studies were found. Studies from previously reviewed ingredients indicate that compounds analogous to laureth-9 were not carcinogenic to rats, and propylene glycol was not carcinogenic in mice (lifetime dermal study) or in rats (2-year chronic feed study).
Irritation and Sensitization
Nonhuman
PPG-5-ceteth-20 was classified as a moderate-to-mild dermal irritant in an in vitro assay. 49 In a 3-(4,5-dimethylthiazol-2-4)-2,5-diphenyltetrazolium bromide (MTT) cytotoxicity assay performed to predict dermal irritancy, PPG-5-ceteth-20 had an ET50 (ie, time required to reduce tissue viability by 50%) of 9.78 hours; substances with an ET50 in the range of 4 to 12 hours have an expected irritancy of moderate to mild.
A single 24-hour occlusive application of PPG-5-ceteth-20 was not a primary dermal irritant in rabbits. 50 Occlusive patches containing 0.5 mL undiluted PPG-5-ceteth-20 were applied for 24 hours to both intact and abraded skin of 3 rabbits; the test sites were clipped free of hair. The patches were described as 2 × 2; units were not provided. The test sites were evaluated for reactions upon patch removal and 48 hours later. No erythema, eschar formation, or edema was observed at any of the test sites 24 or 72 hours after application.
Nonhuman irritation and sensitization data on several analogous compounds available from previous safety assessments of the alkyl PEG ethers and PPGs indicate that these compounds range from nonirritating to moderately and severely irritating. 2 For example, under semiocclusive conditions, C14-15 alkyl ethoxylate number (AE)7, 0.5 mL at 10%, 25%, or 100%, was not irritating to rabbit skin. However, following a 4-hour occlusive application to rabbit skin, undiluted C12-14AE10 and undiluted C13AE6 were moderately irritating and undiluted C13AE6.5 and undiluted C12-14AE6 were severely irritating. Additionally, 50% propylene glycol may have caused skin irritation in nude mice, whereas 100% propylene glycol was nonirritating to minimally irritating in several species. 3,5
Human
PPG-5-ceteth-20 was not a primary irritant, fatiguing agent, or sensitizer in a human repeated insult patch test (HRIPT). 51 Induction consisted of 10 occlusive patches containing 0.5 mL undiluted PPG-5-ceteth-20 applied to the inner aspect of the arm or forearm of 50 subjects. The first induction patch was applied for 48 hours and the remaining 9 patches for 24 hours; there was a 24-hour rest period between patches. The test site was evaluated for reactions upon removal of each patch. A 48-hour challenge patch was applied 10 to 14 days after the last induction patch. No reactions were observed in any of the subjects during induction or challenge.
Human irritation and sensitization data on analogous compounds are available from the safety assessments of the alkyl PEG ethers, and PPGs indicate that generally the ingredients were not strong irritants or sensitizers.
Summary
This report is a safety assessment of 131 alkyl PEG/PPG ethers that can be used in cosmetics. Alkyl PEG/PPG ethers are the reaction products of an alkyl alcohol and one or more equivalents each of ethylene oxide and propylene oxide (forming repeats of PEG and PPG, respectively). Each of the alkyl PEG/PPG ethers has surfactant properties, with a chain structure that has a hydrophobic end and a hydrophilic end; the dimethyl ethers are distinct in this group by being capped at both ends with methyl groups, instead of having 1 alkyl chain at 1 end. The alkyl PEG/PPG ethers are typically manufactured by (1) activating the alcohol by generating an alkoxide; (2) reacting the alkoxide with ethylene oxide, propylene oxide, or a mixture of both; and (3) terminating the propagation. The actual order of alkoxide repeats in each ingredient, and from each source of an ingredient, may be block, alternating, or random, and the propylene oxide placement affects the physical and surface active properties. Based on this chemistry, the potentially carcinogenic compounds ethylene oxide and propylene oxide are present at residual levels, along with the residual byproduct 1,4-dioxane. In practice, these impurities are not present because of steps in the manufacturing process that remove them.
The alkyl PEG/PPG ethers are reported to function in cosmetics mostly as surfactants, skin-conditioning agents, and/or emulsifying agents. The VCRP data obtained from the FDA in 2013 and data received in response to a Council survey of maximum reported use concentration indicate that 26 of the alkyl PEG/PPG ethers named in this safety assessment are currently used in cosmetic formulations. PPG-5-ceteth-20 has the most reported uses (445) and the highest reported concentration in leave-on products (10%). Most of the in-use alkyl PEG/PPG ethers are used in less than 40 formulations and at concentrations of ≤5%.
In Europe, PPG-4-trideceth-6 can be used at 0.05 mg/kg in food contact materials; it is to be used only in PTFE items sintered at high temperatures. PPG-5-ceth-20 is being evaluated for inclusion in dermal and nasal drug delivery systems.
Often, surfactants can be penetration enhancers. However, PPG-4-ceteth-20 did not enhance the penetration of tenoxicam through guinea pig skin. 2
Undiluted PPG-5-ceteth-20 was predicted to be a mild-to-moderate dermal irritant based on the results of an MTT cytotoxicity assay, but it was not a primary irritant in rabbit skin nor was it a primary irritant, fatiguing agent, or sensitizer in a 50-subject HRIPT.
Discussion
Alkyl PEG/PPG ethers are the reaction products of an alkyl alcohol and one or more equivalents each of ethylene oxide and propylene oxide (forming repeats of PEG and PPG, respectively). The alkyl PEG/PPG ethers share very similar physiochemical properties with another family of ingredients that has been previously reviewed by the CIR Expert Panel and found safe when formulated to be nonirritating, that is, the alkyl PEG ethers. The only difference between the alkyl PEG ethers and the alkyl PEG/PPG ethers is the inclusion of PPG repeat units, which are used to simply fine-tune the surfactant properties of these ingredients. The PPGs have also been found safe when formulated to be nonirritating by the Panel.
Although there are little data available on the individual alkyl PEG/PPG ethers, the Panel stated that existing data on analogous ingredients (ie, from the alkyl PEG ethers and PPG safety assessments) support the safety of this ingredient family. These data provided the Panel with a good indication of a lack of toxicity of the alkyl PEG/PPG ethers. Additionally, the alkyl PEG/PPG ethers are larger molecules than alkyl PEG ethers and the PPGs, so they are less likely to penetrate the skin and enter the circulation. And, the maximum use concentration reported for the alkyl PEG/PPG ethers is lower than that reported in the safety assessments of the alkyl PEG ethers or the PPGs.
The Panel was concerned about the possibility of the presence of residual starting materials used in the manufacture of the alkyl PEG/PPG ethers (ie, ethylene oxide and propylene oxide) and of the residual by-product, 1,4-dioxane. These compounds are potentially carcinogenic. The Panel noted these are volatile compounds, and therefore, levels of these compounds in cosmetics are expected to be below the level of toxicological concern. Although levels may be low, the Panel stressed that the cosmetics industry should continue to use the necessary procedures to remove these impurities from the ingredients before blending them into cosmetic formulations.
The Panel recognized that some of the alkyl PEG/PPG ethers can enhance the penetration of other ingredients through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data or when dermal absorption was a concern.
Additionally, the Panel was also concerned that the potential exists for dermal irritation with the use of products formulated with alkyl PEG/PPG ethers. The Panel specified that products must be formulated to be nonirritating.
Finally, the Panel discussed the issue of incidental inhalation exposure of alkyl PEG/PPG ethers in products that could be inhaled. Because the alkyl PEG/PPG ethers are not expected to have chemical activity in biological systems, particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns. Coupled with the small actual exposure in the breathing zone, the expected particle size, and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products that may be aerosolized is available at http://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that the following 131 alkyl PEG/PPG ethers are safe in the present practices of use and concentration in cosmetics described in this safety assessment when formulated to be nonirritating. PEG-4-PPG-7 C13/C15 alcohol* PEG/PPG-3/6 dimethyl ether* PEG/PPG-7/12 dimethyl ether* PEG/PPG-9/2 dimethyl ether PEG/PPG-14/7 dimethyl ether PEG/PPG-17/4 dimethyl ether PEG/PPG-22/40 dimethyl ether* PEG/PPG-27/14 dimethyl ether* PEG/PPG-35/40 dimethyl ether PEG/PPG-36/41 dimethyl ether PEG/PPG-50/40 dimethyl ether PEG/PPG-52/32 dimethyl ether* PEG/PPG-55/28 dimethyl ether PEG/PPG-4/2 propylheptyl ether* PEG/PPG-6/2 propylheptyl ether* PEG-7/PPG-2 propylheptyl ether* PEG/PPG-8/2 propylheptyl ether* PEG/PPG-10/2 propylheptyl ether* PEG/PPG-14/2 propylheptyl ether* PEG/PPG-40/2 propylheptyl ether* PPG-2-ceteareth-9 PPG-4-ceteareth-12* PPG-10-ceteareth-20* PPG-1-ceteth-1* PPG-1-ceteth-5* PPG-1-ceteth-10* PPG-1-ceteth-20* PPG-2-ceteth-1* PPG-2-ceteth-5* PPG-2-ceteth-10 PPG-2-ceteth-20* PPG-4-ceteth-1* PPG-4-ceteth-5* PPG-4-ceteth-10* PPG-4-ceteth-20 PPG-5-ceteth-20 PPG-8-ceteth-1 PPG-8-ceteth-2* PPG-8-ceteth-5* PPG-8-ceteth-10 PPG-8-ceteth-20 PPG-2 C9-11 pareth-5* PPG-2 C9-11 pareth-7* PPG-2 C9-11 pareth-8* PPG-2 C9-11 pareth-11* PPG-2 C12-13 pareth-8 PPG-2 C12-15 pareth-6* PPG-4 C13-15 pareth-15* PPG-5 C9-15 pareth-6* PPG-6 C9-11 pareth-5* PPG-6 C12-15 pareth-12* PPG-6 C12-18 pareth-11* PPG-3 C12-14 Sec-pareth-7* PPG-4 C12-14 Sec-pareth-5* PPG-5 C12-14 Sec-pareth-7* PPG-5 C12-14 Sec-pareth-9* PPG-1-deceth-4* PPG-1-deceth-5* PPG-1-deceth-6* PPG-1-deceth-7* PPG-2-deceth-3 PPG-2-deceth-5* PPG-2-deceth-7* PPG-2-deceth-8* PPG-2-deceth-10* PPG-2-deceth-12 PPG-2-deceth-15* PPG-2-deceth-20* PPG-2-deceth-30* PPG-2-deceth-40* PPG-2-deceth-50* PPG-2-deceth-60* PPG-4-deceth-4* PPG-4-deceth-6* PPG-6-deceth-4* PPG-6-deceth-9* PPG-8-deceth-6* PPG-14-deceth-6* PPG-6-decyltetradeceth-12* PPG-6-decyltetradeceth-20 PPG-6-decyltetradeceth-30 PPG-13-decyltetradeceth-24 PPG-20-decyltetradeceth-10 PPG-9-ethylhexeth-5* PPG-1-isodeceth-4* PPG-1-isodeceth-6* PPG-1-isodeceth-7* PPG-1-isodeceth-9* PPG-2-isodeceth-4* PPG-2-isodeceth-6* PPG-2-isodeceth-8* PPG-2-isodeceth-9* PPG-2-isodeceth-10* PPG-2-isodeceth-12 PPG-2-isodeceth-18* PPG-2-isodeceth-25* PPG-3-isodeceth-1* PPG-4-isodeceth-10* PPG-3-isosteareth-9 PPG-2-laureth-5* PPG-2-laureth-8* PPG-2-laureth-12* PPG-3-laureth-8* PPG-3-laureth-9* PPG-3-laureth-10* PPG-3-laureth-12* PPG-4-laureth-2* PPG-4-laureth-5* PPG-4-laureth-7* PPG-4-laureth-15* PPG-5-laureth-5 PPG-6-laureth-3* PPG-25-laureth-25 PPG-3-myreth-3* PPG-3-myreth-11* PPG-2-PEG-11 hydrogenated lauryl alcohol ether* PPG-3-PEG-6 oleyl ether* PPG-9-steareth-3* PPG-23-steareth-34* PPG-30 steareth-4* PPG-34-steareth-3 PPG-38 steareth-6* PPG-1-trideceth-6 PPG-1-trideceth-13* PPG-4-trideceth-6* PPG-6-trideceth-8* Propylene glycol capreth-4* Propylene glycol isodeceth-4* Propylene glycol isodeceth-12* Propylene glycol laureth-6* Propylene glycol oleth-5*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC.
Author Contributions
M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. B. Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. L. Gill, F. Alan Anderson, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.

