Abstract
Tavaborole is a topical antifungal agent approved by the US Food and Drug Administration for the treatment of toenail onychomycosis. As part of the nonclinical development program, reproductive and developmental toxicity studies were conducted (rat oral fertility and early embryonic development, rat (oral) and rabbit (dermal) embryo–fetal development). There were no effects on fertility or reproductive performance at doses up to 300 mg/kg/d (107 times the maximum recommended human dose [MRHD] based on mean area under the plasma concentration–time curve comparisons). In the rat embryo–fetal development toxicity studies, teratogenicity was not observed at doses up to 100 mg/kg/d (29 times the MRHD). However, several treatment-related skeletal malformations and variations were observed at 300 mg/kg/d (570 times the MRHD). In rabbit embryo–fetal development toxicity studies dosed via oral or dermal administration, the no observable adverse effect level for maternal toxicity and embryo–fetal toxicity was 50 mg/kg/d (16 times the MRHD) and 5% (26 times the MRHD), respectively.
Introduction
Onychomycosis is a fungal infection of the nail unit (primarily the nail bed and often involving the nail plate and less frequently the nail matrix), most commonly occurring in the toenails. Signs of infection include a thickening of the nail plate and nail bed, discoloration, splitting of the nail plate, and lifting of the nail plate from the nail bed, known as onycholysis. This often results in difficulty in grooming nails, social embarrassment, damage to socks and stockings caused by an abnormal nail plate, secondary bacterial infection, and pain or discomfort due to separation of the nail plate. Typically, the infection is caused by dermatophytes, which are ubiquitous in the environment and are the same organisms associated with athlete’s foot, jock itch, and ringworm. A large proportion of the population is susceptible to dermatophyte infections and there does not appear to be a protective adaptive immune response, so reinfection is common in the susceptible population. 1,2
Treatment options include oral and topical drugs. Limitations of currently approved oral therapies include drug–drug interactions and systemic adverse effects, including hepatotoxicity. 3 -7
The difficulty in treating onychomycosis topically may result from the deep-seated nature of the infection within the anatomically complex nail unit (nail plate, nail bed, and nail matrix) and the inability of some drugs to effectively reach all compartments of the nail unit.
Oral reproduction studies have been reported with other antifungals that inhibit the synthesis of ergosterol, which is a vital component of fungal cell membranes. Lamisil (terbinafine hydrochloride) was administered to rabbits and rats at doses up to 300 mg/kg/d (12-23× the maximum recommended human dose [MRHD] in rabbits and rats, respectively, based on body surface area [BSA] comparisons) and revealed no evidence of impaired fertility or harm to the fetus due to terbinafine. 6 Sporanox (itraconazole) was found to cause a dose-related increase in maternal toxicity, embryotoxicity, and teratogenicity in rats at dosage levels of approximately 40 to 160 mg/kg/d (5-20× the MRHD) and in mice at dosage levels of approximately 80 mg/kg/d (10× the MRHD). In rats, the teratogenicity consisted of major skeletal defects; in mice, it consisted of encephaloceles and/or macroglossia. 7
Tavaborole topical solution, 5% (Kerydin, CAS #174671-46-6; Anacor Pharmaceuticals, Inc, Palo Alto, California), a cyclized boronic acid complex (5-fluoro-1,3-dihydro-1-hydroxy-2,1-benzoxaborole), is approved by the US Food and Drug Administration (FDA) for the topical treatment of onychomycosis of the toenails due to Trichophyton rubrum or Trichophyton mentagrophytes (Figure 1). This novel, low-molecular-weight (152 Da) pharmaceutical compound demonstrates potent antifungal activity against dermatophytes with minimum inhibitory concentration values in the low mg/mL range. 8
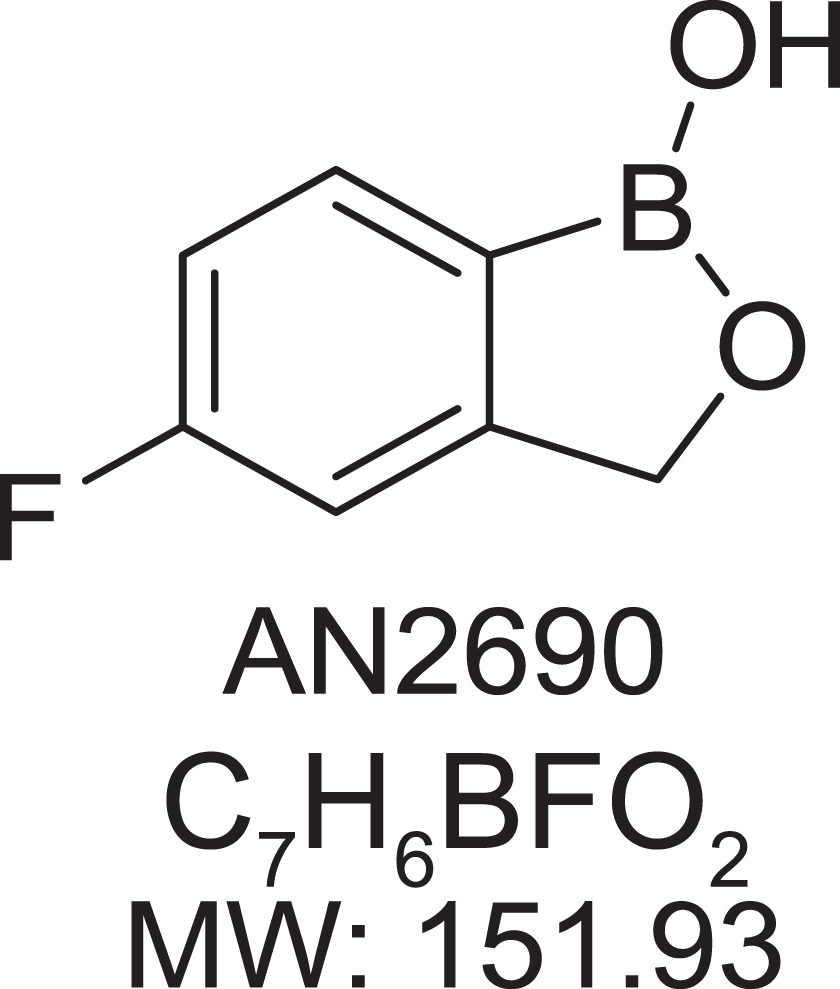
Chemical structure of tavaborole.
The mechanism of action of tavaborole is the inhibition of fungal protein synthesis through inhibition of leucyl-tRNA synthetase, an aminoacyl-tRNA synthetase (AARS). 9 Although tavaborole inhibits an AARS, its mechanism of action is different from that of other previously identified AARS inhibitors. It represents a new class of inhibitors that exert their action on AARSs via the editing domain.
Preclinical studies, including a battery of genetic toxicology studies 10 and carcinogenicity studies, 11 have demonstrated the safety of this compound. Toxicology studies were conducted with a dermal route of administration to mimic the human exposure. Oral toxicology studies were conducted to provide higher systemic levels in order to establish safety margins. For women of childbearing potential, there is always a concern for unintentional exposure of an embryo or fetus. Therefore, the following studies, including fertility and early embryonic development in rats (oral), embryo–fetal development toxicity studies in rats (oral) and rabbits (oral and dermal), and a prenatal and postnatal development study in rats (oral), were conducted. The fertility and early embryonic development toxicity studies in rats were designed to determine the potential toxicity on the female estrous cycle, tubal transport, implantation and development of preimplantation stages of the embryo, and detection of functional effects on male fertility. The embryo–fetal developmental toxicity studies in rats and rabbits were conducted to evaluate the developmental toxicity, including the teratogenic potential, of tavaborole. The results of the prenatal and postnatal development study are described in another publication. 12 That study was designed to evaluate the survival, development, and reproductive capabilities of pups (F1 generation) from implantation through lactation. All the described studies were submitted in support of FDA approval of tavaborole.
Materials and Methods
All studies were conducted as per published International Conference on Harmonization (ICH) guidelines, 13 and protocols were reviewed and approved by the appropriate Institutional Animal Care and Use Committee. Food and tap water were available ad libitum. Certification analysis of each diet lot was performed by the manufacturer. Water was supplied using an automatic watering system. The water supply was monitored for specified contaminants at periodic intervals according to Standard Operating Procedures (SOPs) at each laboratory. There was no indication that contaminants were present in the water or diet that could have interfered with the results of these studies. All laboratories conducting these studies were accredited by the Association for Assessment and Accreditation of Laboratory Animal Care.
Test Substance and Formulation Preparation
Tavaborole powder was manufactured by Anacor Pharmaceuticals, Inc (Palo Alto, California) according to Good Laboratory Practice (GLP) conditions. Tavaborole oral suspensions were prepared in 1% (wt/vol) medium-viscosity carboxymethylcellulose United States Pharmacopeia (USP) grade (Sigma-Aldrich, St Louis, Missouri) at appropriate concentrations to meet dose level requirements. Tavaborole topical dosing solutions were prepared similarly to the marketed formulation (5% tavaborole) in 80% ethanol (USP, 190 proof) and 20% propylene glycol (Spectrum Quality Products, New Brunswick, New Jersey) to achieve the desired concentration (up to 10% tavaborole) and stored at room temperature. The oral formulations were prepared fresh weekly, stored at 2°C to 8°C, and allowed to reach room temperature prior to dosing. Analyses for homogeneity and concentration verification were conducted for all studies. In all cases, formulations were found to be homogeneous and within ±15% of target concentrations.
Oral Fertility and Early Embryonic Developmental Toxicity in Rats
Animals and test article administration
Male and female naive Sprague Dawley rats (Crl: CD [SD] IGS BR), approximately 6 weeks old, were received from Charles River Laboratories (Portage, Michigan). There were 25 rats/sex/group. Rats were administered tavaborole at 0, 30, 100, or 300 mg/kg/d via oral gavage once daily at a dose volume of 10 mL/kg. Males were dosed beginning 28 days prior to pairing, whereas the females were dosed beginning 14 days prior to pairing. Dosing of the males continued through the mating and postmating period to euthanasia, whereas dosing of the females continued through the mating period to gestation day (GD) 7. Upon completion of the GD 13 uterine examinations, all surviving males were observed externally, euthanized, and subjected to a necropsy and sperm analysis. In repeat-dose toxicity studies of ≥28 days in duration, there were no effects observed in the reproductive tissues of either sex; therefore, the dosing duration of 28 days in males and 14 days in females was justified.
The doses were selected based on the results of 1- and 3/6-month oral toxicity studies in rats. In those studies, tavaborole had been shown to produce hyperkeratosis and hyperplasia of the nonglandular stomach in rats at doses of 30, 50, 100, and 200 mg/kg/d. However, there were no drug-related mortalities in those studies and animals survived until scheduled necropsy. In a range-finding 1-month oral toxicity study in male rats, doses of 500 and 1,000 mg/kg/d produced mortality in 3 of 5 and 5 of 5 rats, respectively. No mortalities occurred at doses of 100 and 200 mg/kg/d.
Observation and evaluations
Parameters of evaluations included female estrous cycle, tubal transport, implantation and development of preimplantation stages of the embryo, and detection of functional effects on male fertility, utilizing treatment of both sexes. Rats were observed for clinical signs, body weights, and food consumption measurements. Males and females of the same treatment group were paired for up to 21 days or until evidence of mating (sperm or copulatory plug) was observed. Females were examined daily for estrous cycle determination during the premating and mating periods until evidence of mating was observed. Mated females were euthanized on GD 13, uterine examinations were conducted, and the number of viable embryos, resorptions, and total number of implantations were recorded. Uteri from females that appeared nongravid were opened and placed in 10% ammonium sulfide solution for the detection of implantation sites. If no foci were seen, the female was considered nonpregnant. 14 The number of corpora lutea on each ovary was recorded. Complete necropsies and organ collection were performed on all rats, and organs and tissues were weighed and preserved. Sperm analyses (motility, concentration, and morphology) were also conducted for male rats.
Resorptions are defined as early and late resorptions. An early resorption is defined as a conceptus that has implanted but has no recognizable embryonic characteristics evident upon examination. A late resorption is defined as fetal remains or tissues that have recognizable fetal characteristics (such as limb buds with no discernable digits present) and are undergoing autolysis. 15
Oral Embryo–Fetal Developmental (Teratology) Toxicity in Rats
Animals and test article administration
Time-mated female Sprague Dawley rats (Crl: CD[SD]), 8 to 10 weeks of age at arrival [GD 0], were received from Charles River Laboratories (Portage, Michigan). There were 25 rats/group for the main study. Rats were administered tavaborole at 0, 30, 100, or 300 mg/kg/d via oral gavage once daily at a dose volume of 10 mL/kg from GD 6 through 19. Individual doses were based on the most recent body weights. Rats in this study were dosed for a longer period (GD 6-19) instead of GD 6 to 16 or 17 as per ICH S5(R2) guidance. However, the longer dosing period had no impact on the study. Dose selection was based on the same criteria described in the fertility study.
Observation and evaluations
Observation of the main study animals included clinical signs, gestation body weights, and gestation food consumption. On the day of cesarean section (GD 20), all main study animals were euthanized and subjected to a complete necropsy, including a uterine examination in which the total number of implantations, early and late resorptions, live and dead fetuses, and the position of the cervix were recorded. Uteri from females that appeared nongravid were opened and placed in 10% ammonium sulfide solution for the detection of implantation sites. If no foci were seen, the female was considered nonpregnant. 14 The total number of corpora lutea on each ovary was also recorded. Gravid uterine weights were recorded, and adjusted terminal body weights were calculated. All fetuses were examined for external malformations and variations. Subsequently, one-half of the fetuses were examined for soft tissue defects, whereas the other half of the fetuses were prepared for subsequent skeletal examination. Malformations and developmental variations were recorded.
Oral and Dermal Embryo–Fetal Development Toxicity in Rabbits
Animals and test article administration
New Zealand white rabbit (Hra:[NZW]SPF) timed-mated females (GD 0), approximately 6 to 7 months of age at the initiation of dosing, were received from Covance Research Products Inc (Kalamazoo, Michigan). In the oral study, groups of 20 rabbits each were dosed once daily on GD 7 through GD 19 at 0 (vehicle), 15, 50, or 150 mg/kg/d at a dose volume of 5 mL/kg. Doses were selected from the results of a pilot embryo–fetal developmental and TK study of tavaborole administered orally by gavage in rabbits.
In the dermal study, groups of 25 rabbits each were dosed once daily on GD 6 to 28 with vehicle and tavaborole topical formulations (1%, 5%, and 10%) at a volume of 5 μL/cm2. Care was taken to avoid abrading the skin. In the dermal study, rabbits were dosed for a longer period to provide maximum exposure duration. The doses were selected based on the results of the dermal range-finding prenatal development study of tavaborole in rabbits and 28-day, 90-day, and 180-day dermal toxicity studies of tavaborole in minipigs where concentrations of 5% and 10% solutions were locally irritating to the application sites of the minipigs.
In the dermal study, at least 24 hours prior to the first administration, the hair was clipped from the back of each animal. The clipped area comprised no less than 10% of the total BSA. The body surface was estimated from the following equation 16 : A = 10·W2/3, where A is the estimated area in square centimeters and W is the body weight in grams. Hair was reclipped as necessary, and the 10% area of application was adjusted weekly based on the mean body weight for each group. The vehicle or tavaborole topical formulation was applied evenly over each designated area (beginning at the scapular region and moving caudally over the application site). Following dermal application, Elizabethan collars were applied and remained on until approximately 2 hours postdose. Once per day, following the dermal irritation scoring and prior to the next dose, the application sites were gently wiped with a cleansing towelette to remove any residual material.
Careful examination of the dosing site for signs of edema and erythema was made and recorded daily for main study animals beginning on GD 6, prior to each daily application and prior to washing, according to the skin reaction scale given below.
The following scores are based upon the Draize 17 scale for scoring skin irritation:

Observation and evaluations
Observation of the main study rabbits included clinical signs, dermal irritation scoring (only for dermal administration), gestation body weights, and gestation food consumption. Blood samples for the determination of the plasma concentrations of tavaborole were collected from TK animals at designated time points on GD 6 and 28. After the final blood collection, the TK animals were euthanized, pregnancy status was determined, and the carcasses were discarded without further evaluation. On GD 29, all main study animals were euthanized and subjected to a complete necropsy, including a uterine examination in which the total number of implantations, early and late resorptions, live and dead fetuses, and the position of the cervix were recorded. Uteri from females that appeared nongravid were opened and placed in 10% ammonium sulfide solution for the detection of implantation sites. If no foci were seen, the female was considered nonpregnant. 14 The total number of corpora lutea on each ovary was also recorded. All fetuses were given an external and fresh visceral examination and processed for skeletal examination. Malformations and developmental variations were recorded.
Plasma and TK Analysis
Blood samples were collected from the embryo–fetal development studies in rats (9 rats/group) and rabbits (4 rabbits/group). The TK sampling time points were predose and 0.25, 0.5, 1, 2, and 4 hours postdose on GD 6 and GD 19 for the rat oral teratology study; 0.25, 0.5, 0.75, 1, 2, and 4 hours postdose on GD 19 for the rabbit oral teratology study; and predose and 0.25, 0.5, 1, 2, and 4 hours postdose on GD 6 and GD 28 for the rabbit dermal teratology study. Samples were collected into tubes containing K2EDTA and stored on an ice block until centrifuging under refrigeration. Serum samples were placed in tightly capped, prelabeled plastic vials and stored frozen at approximately −70°C until analysis using validated liquid chromatography coupled with tandem mass spectrometry (LC–MS/MS) procedures using d2-tavaborole as the internal standard. The TK parameter estimates were determined for tavaborole from mean concentration–time data. A noncompartmental module of WinNonlin software was used to calculate parameter estimates.
Statistics
Group pairwise comparisons
Levene/Bartlett/analysis of variance (ANOVA)–Dunnett/Welch tests were used for the statistical analyses performed for all parameters except percentage preimplantation loss and percentage postimplantation loss. If sample sizes for all groups were 3 or greater, Levene’s test 18 was used to assess homogeneity of group variances for each specified end point and for each collection interval. If Levene’s test was not significant (P > 0.01), a pooled estimate of the variance (mean square error) was computed from a 1-way ANOVA and utilized by a Dunnett 19 comparison of each treatment group with the control group. If Levene’s test was significant (P < 0.01), comparisons with the control group were made using Welch t test 20 with a Bonferroni correction. In the case where sample size was less than 3 for at least 1 treatment group, Levene’s test could not be implemented. Groups with sample sizes less than 3 were excluded from the analysis, and control–treatment pairwise comparisons that satisfied the sample size assumption (n > 3) were conducted using Welch t test with a Bonferroni correction. If there were only 2 groups involved, the above methodology applied and the Dunnett’s test reduced to a Student’s t test. 21 Results of all pairwise comparisons are reported at the 0.05 and 0.01 significance levels.
Arcsine square root transformation
Arcsine square root transformations were used for statistical analysis of percentage preimplantation and postimplantation loss. 22 The analysis described in the preceding paragraph for group pairwise comparisons was used to analyze the transformed percentage values.
Fisher’s Exact test
An overall test for association between response and treatment was conducted using Fisher’s exact test. 23 Results are reported at the 0.05 and 0.01 significance levels. All end points were analyzed using 2-tailed tests. Overall testing and follow-up pairwise testing were conducted for the control and all treatment groups that had sufficient sample size (n ≥ 3). If all groups had an insufficient sample size (n < 3), or if only the control group had a sample size of less than 3, no testing was performed.
Results
Oral Fertility and Early Embryonic Developmental Toxicity in Rats
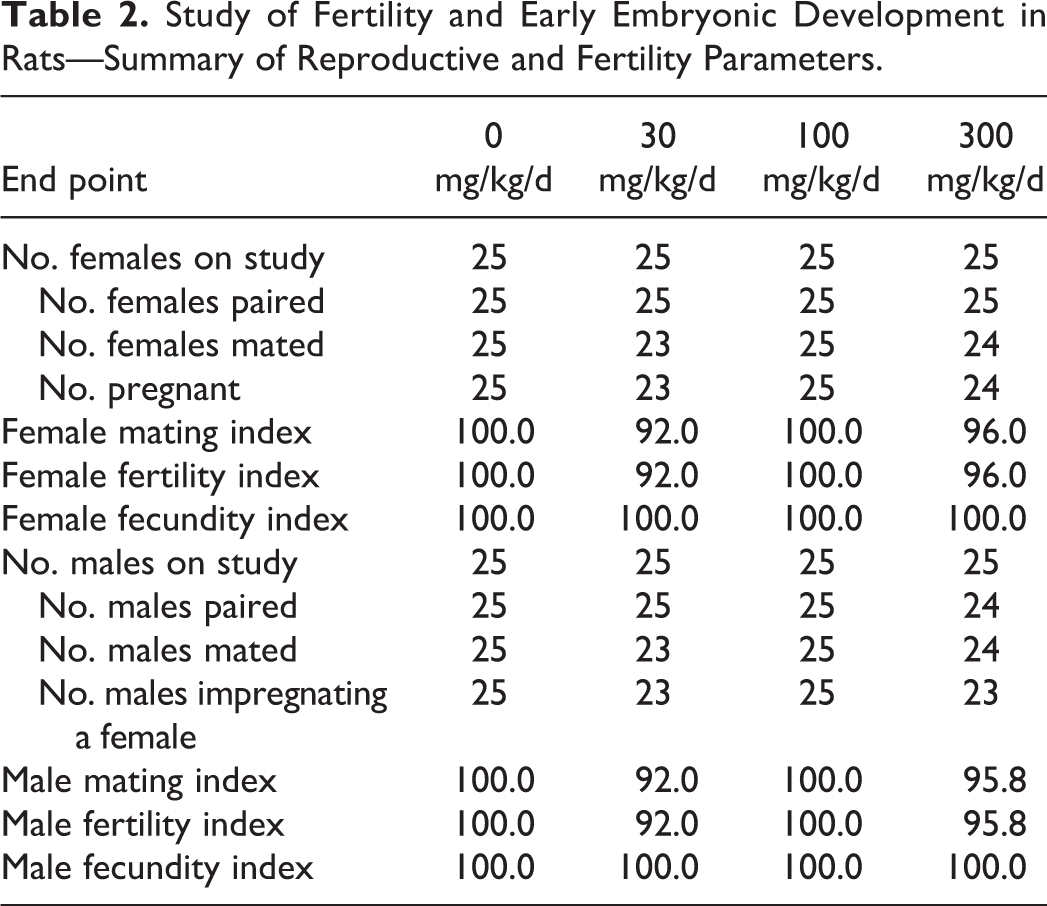
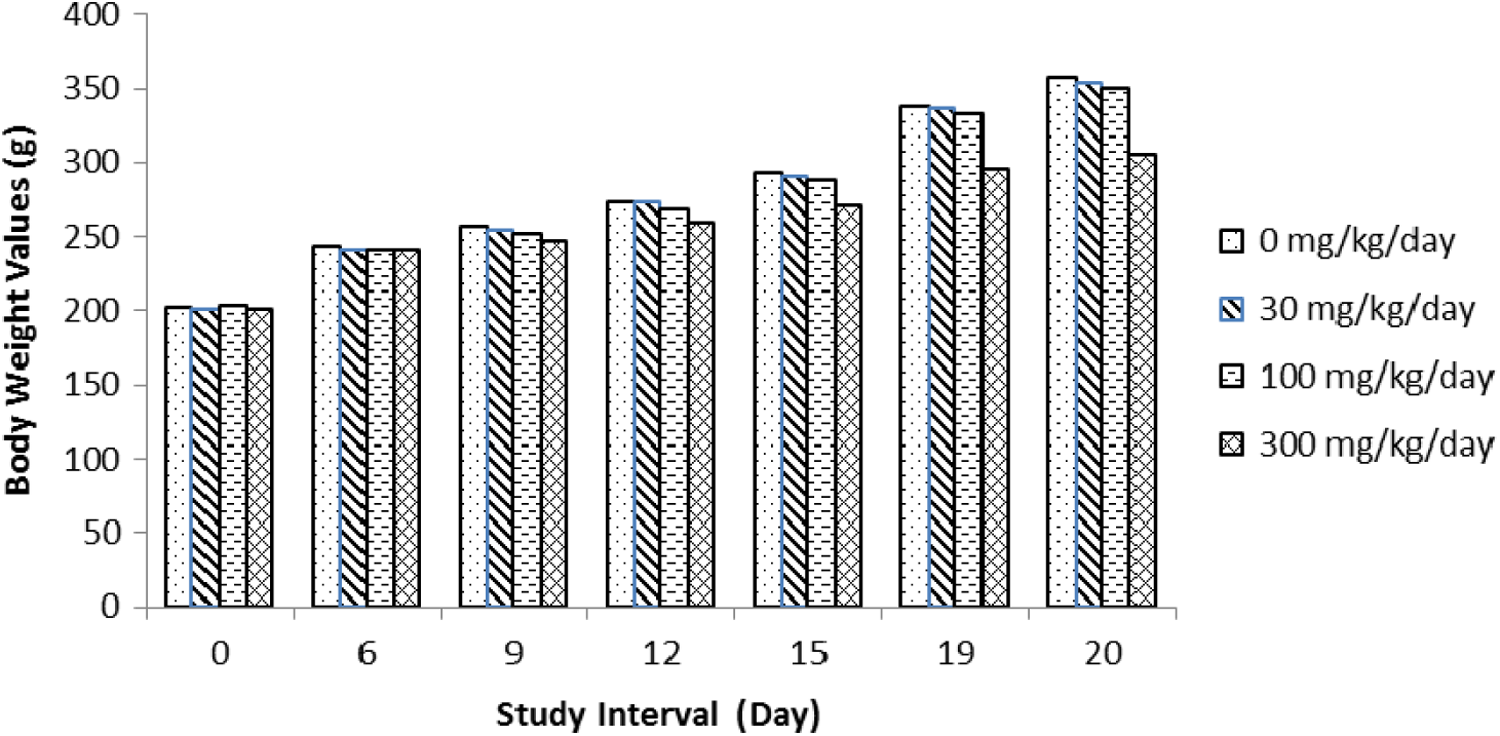
Male and female rats were treated with tavaborole at the oral dose levels of 0, 30, 100, and 300 mg/kg/d. One male at 300 mg/kg/d died during the study. Treatment-related clinical observations were noted prior to death and consisted of decreased activity, difficult breathing, high carriage posture, lacrimation, righting reflex impaired, and salivation. At necropsy, this animal was noted with nonglandular stomach tissue raised, thickened, and white in appearance and smaller than normal seminal vesicles. The findings related to the nonglandular stomach correlated with findings seen in previous studies with tavaborole. Therefore, this death was attributed to treatment with tavaborole. All remaining male and female rats survived to terminal euthanasia. Several treatment-related clinical findings at 100 and 300 mg/kg/d in males and females were observed throughout the study period (Table 1). These findings were most evident at 300 mg/kg/d and to a lesser degree at 100 mg/kg/d and consisted of decreased activity, impaired or lost righting reflex, salivation, low and/or high carriage posture, lacrimation, impaired limb function, red material around the nose and/or mouth, hunched posture, yellow discolored hair around the abdominal and/or anogenital area, skin cold to touch, and breathing abnormalities. Male animals in the 300 mg/kg/d group had lower body weights (Figure 2), body weight gain, and food consumption (data not shown). No effect on body weights (Figure 3), body weight gain, and food consumption (data not shown) was evident in females at 300 mg/kg/d. Gross observation was noted in 300 mg/kg/d males with thickening and/or discoloration of the nonglandular stomach and decrease in absolute seminal vesicle weight. Reproductive organs and uterine implantation parameters were unaffected by treatment with tavaborole at any dose level evaluated. There were no effects on fertility and pregnancy indices (Table 2). Therefore, based on these results, the no observable adverse effect level (NOAEL) for parental and maternal toxicity with tavaborole was 100 mg/kg/d and the NOAEL for fertility and reproductive performance was 300 mg/kg/d. The area under the plasma concentration–time curve (AUC0-4h) value for the 300 mg/kg/d dose in this oral rat fertility study was predicted from a linear extrapolation of the 200 mg/kg/d AUC value for male and female rats combined after 3 months of treatment in a 6-month oral rat toxicity study. This predicted AUC value was 8,077 ng·h/mL, 107 times the MRHD (Table 3).
Study of Fertility and Early Embryonic Development in Rats– Summary of Selected Clinical Observations
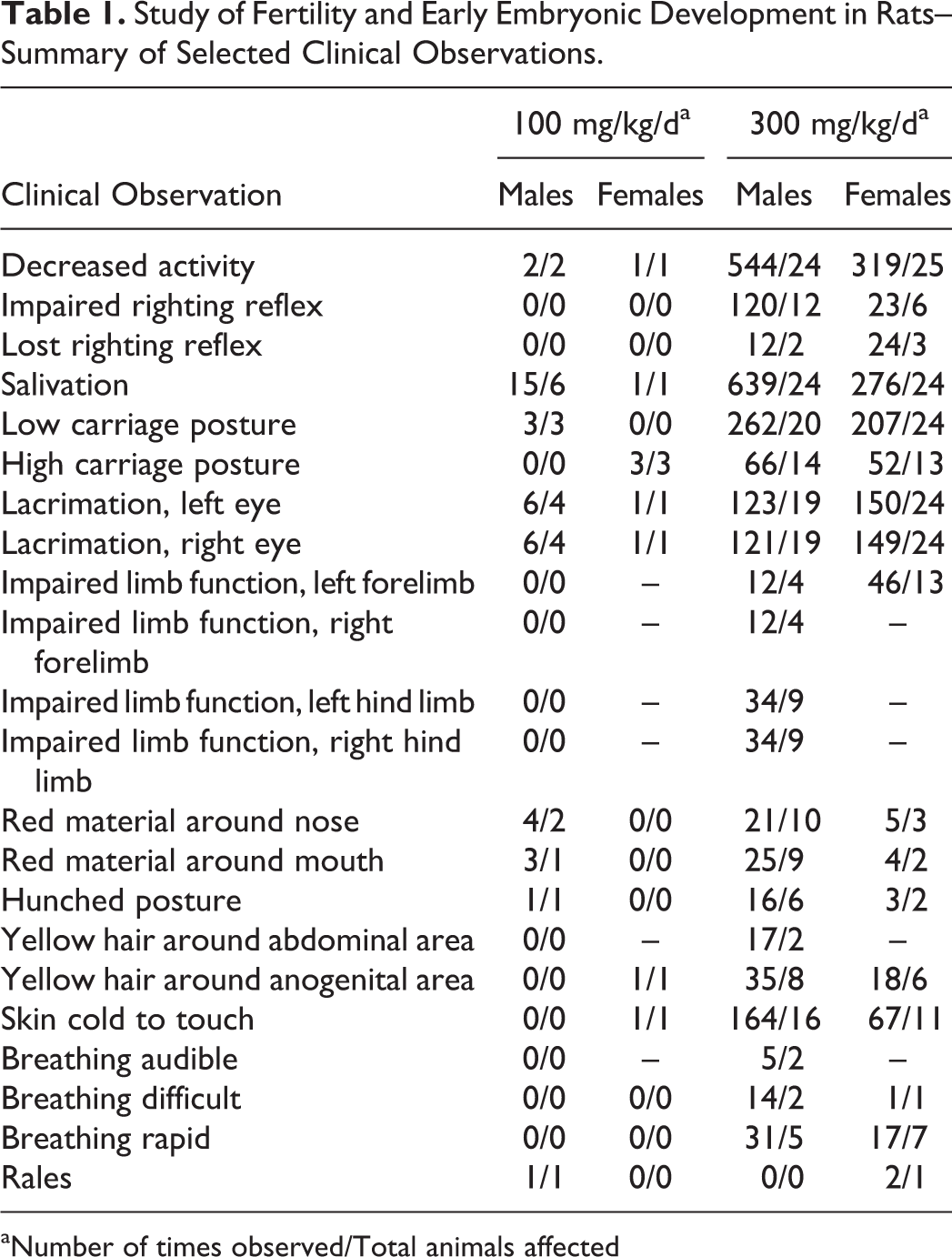
aNumber of times observed/Total animals affected
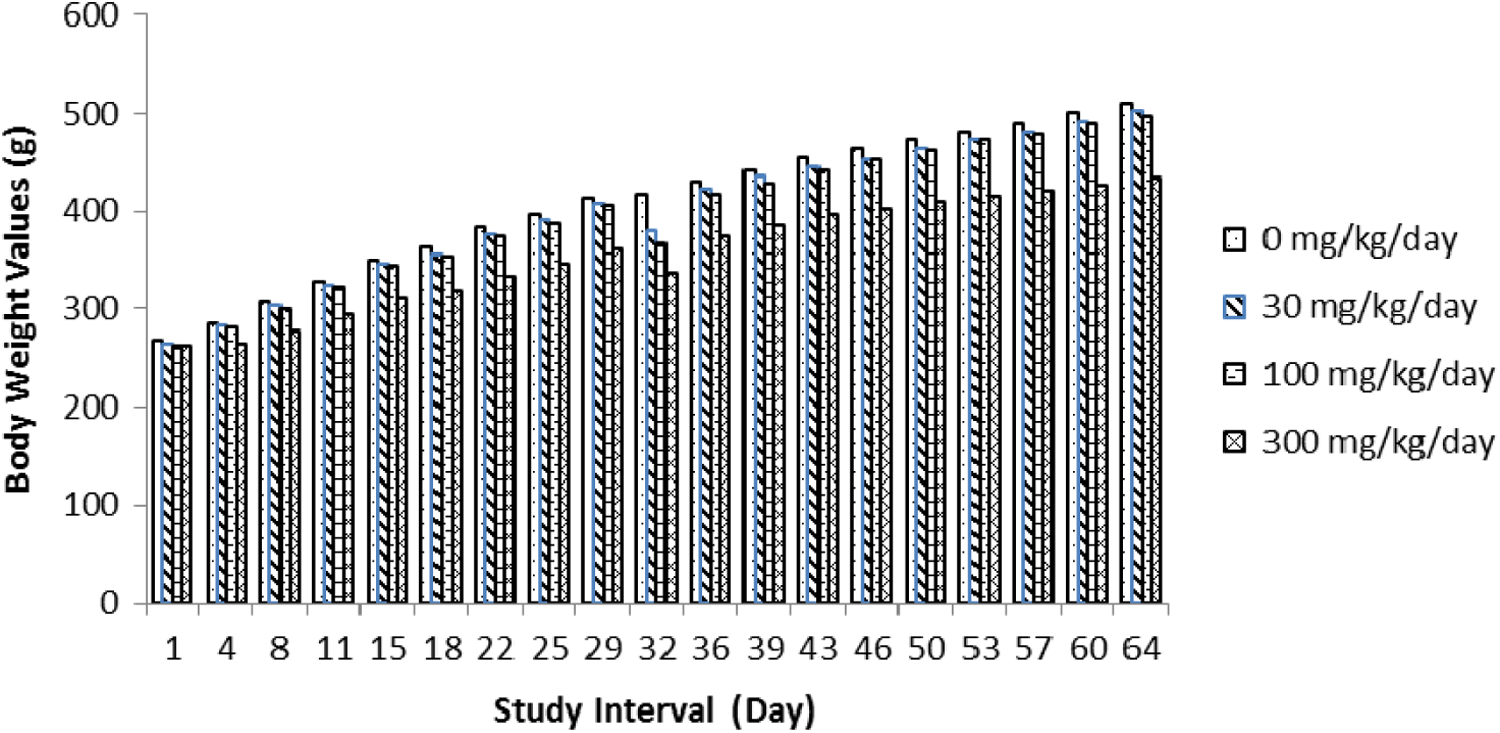
Summary of male body weights in the rat fertility study.
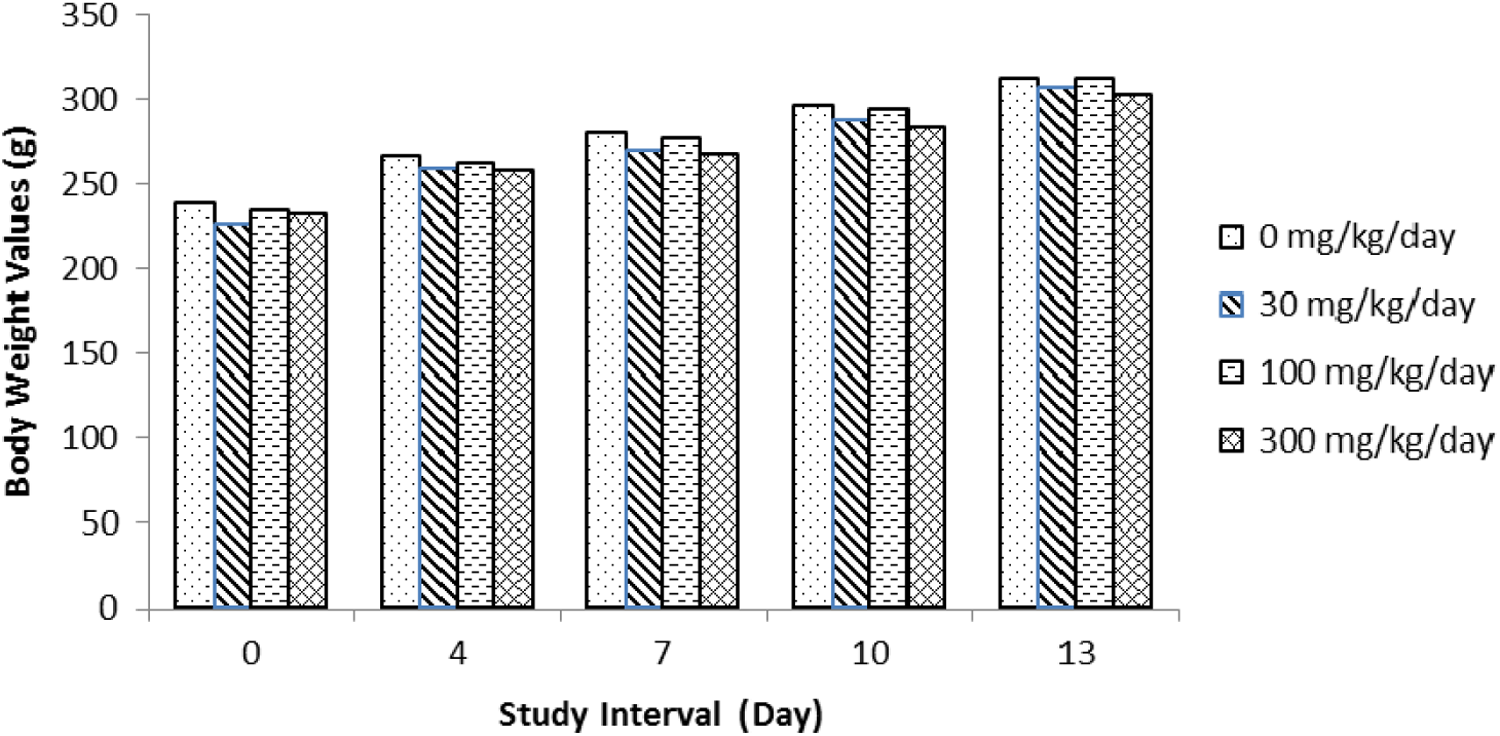
Summary of female body weights in the rat fertility study.
Study of Fertility and Early Embryonic Development in Rats—Summary of Reproductive and Fertility Parameters.
Calculated Safety Margins.
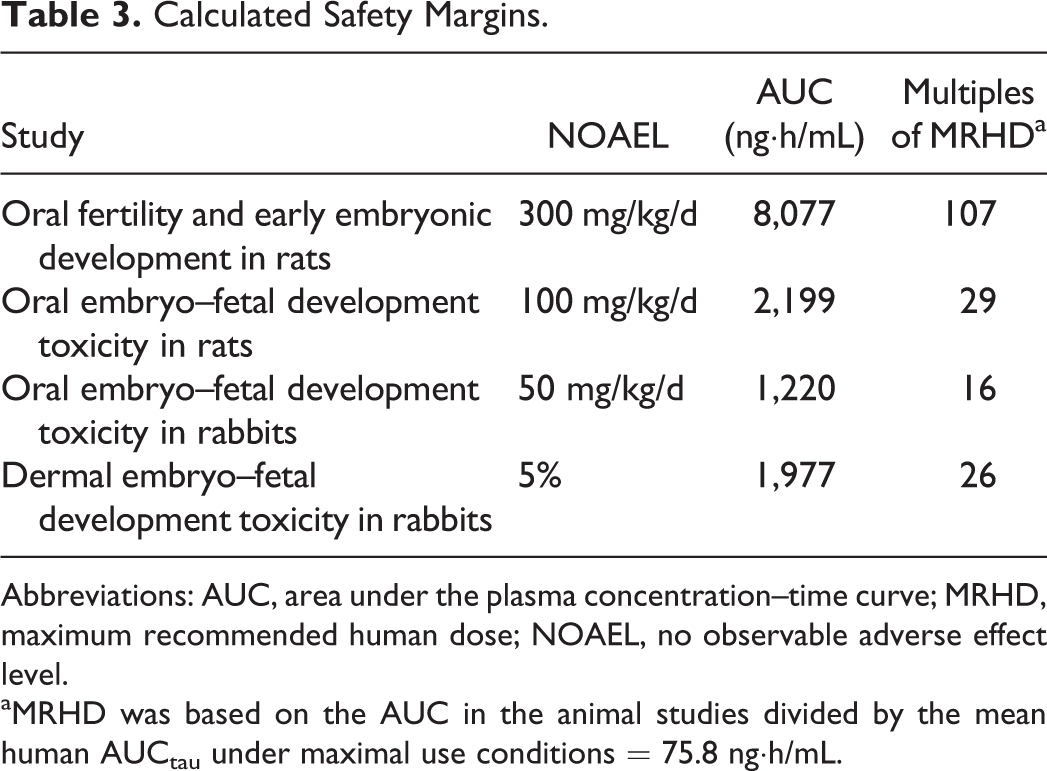
Abbreviations: AUC, area under the plasma concentration–time curve; MRHD, maximum recommended human dose; NOAEL, no observable adverse effect level.
aMRHD was based on the AUC in the animal studies divided by the mean human AUCtau under maximal use conditions = 75.8 ng·h/mL.
Oral Embryo–Fetal Development Toxicity in Rats
Time-mated female rats were treated with tavaborole at the oral dose levels of 0, 30, 100, and 300 mg/kg/d. No treatment-related mortality was observed in tavaborole-treated rats at doses up to 300 mg/kg/d. Several clinical findings (decreased activity, impaired righting reflex, salivation, lacrimation, low and/or high carriage posture, piloerection, and hunched posture) considered related to treatment with tavaborole were observed at 100 and 300 mg/kg/d. Mean gestation body weights were significantly lower than controls in the 300 mg/kg/d group on GD 15, 19, and 20 (Figure 4). In addition, decreased food consumption was also evident at 300 mg/kg/d (data not shown). One female at 300 mg/kg/d died pregnant, and 3 females in the 300 mg/kg/d group had all resorbing fetuses, thereby providing 25, 25, 24, and 20 litters with viable fetuses for evaluation on GD 20 in the control, 30, 100, and 300 mg/kg/d groups, respectively. However, a treatment-related increase in postimplantation loss and a corresponding decrease in viable fetuses were observed at 300 mg/kg/d along with lower gravid uterine weights. Mean fetal body weights (male, female, and sexes combined) were about 38% lower in the 300 mg/kg/d group and considered related to treatment with tavaborole. Mean fetal body weights were also slightly lower (6%) at 100 mg/kg/d, and although statistically significant, these weights were within the historical control data for this laboratory and therefore were not considered adverse. No treatment-related fetal external or visceral malformations were evident at any dose level evaluated (Table 4). However, at 300 mg/kg/d, several treatment-related skeletal malformations (absent, fused, or misshapen neural arches observed at the highest frequency in the thoracic region, absent or fused ribs, larger than normal cranial fontanelle, various bones incompletely or not ossified, and other variations) were observed. The delay in ossification seen at this dose level was consistent with the lower maternal and fetal body weights (Table 4). Therefore, based on these results, the NOAEL for maternal toxicity was 100 mg/kg/d and the NOAEL for developmental toxicity was 100 mg/kg/d (29 times the MRHD; MRHD was based on the mean AUC in the animal studies divided by the mean human AUCtau under maximal use conditions = 75.8 ng·h/mL) (Table 3). The skeletal malformations and variations observed in fetuses of dams dosed at 300 mg/kg/d were at 570 times the MRHD.
Summary of female body weights in the oral embryo–fetal development toxicity study in rats.
Results of the Oral Embryo–Fetal Development Toxicity Study in Rats.
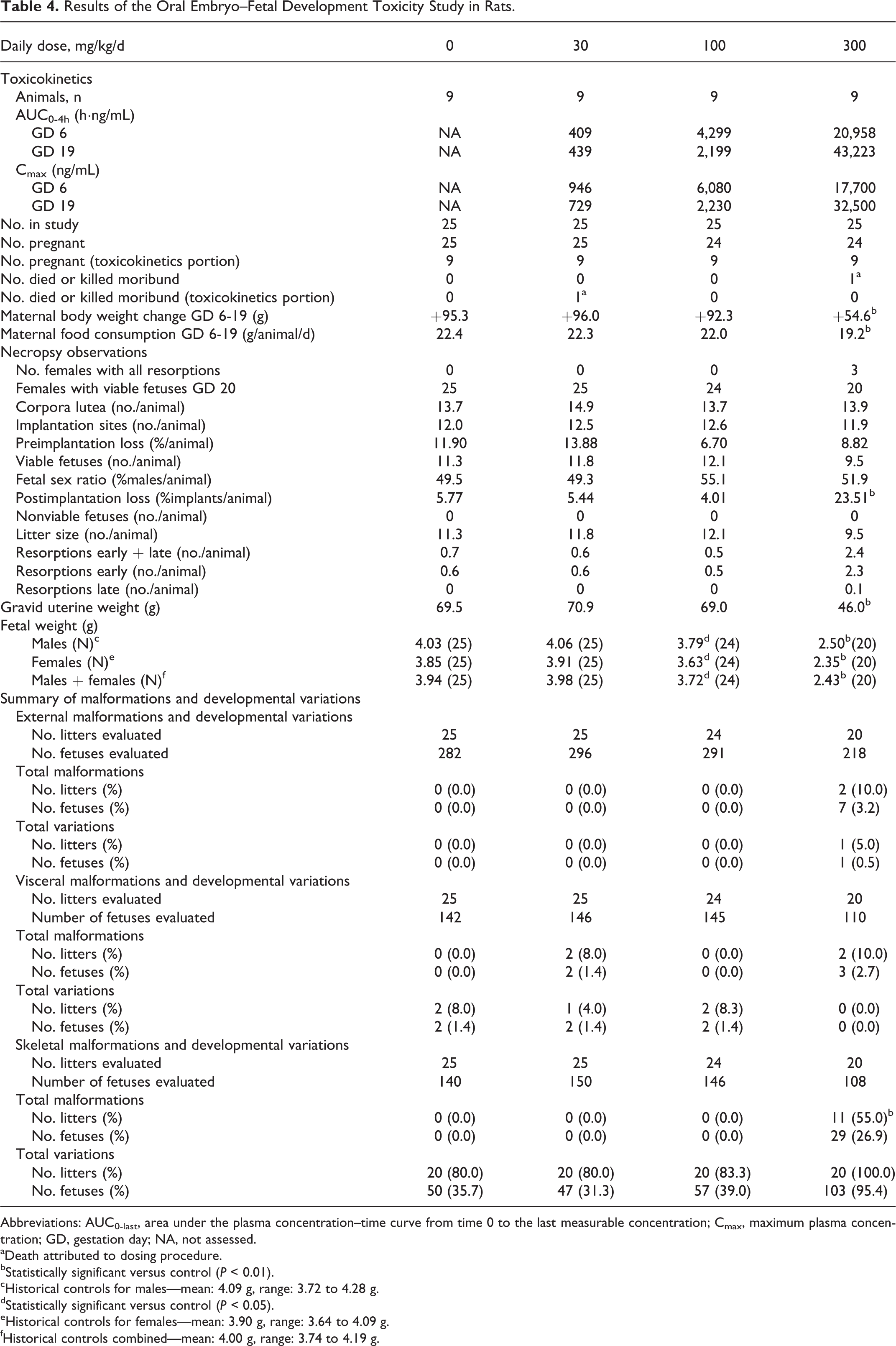
Abbreviations: AUC0-last, area under the plasma concentration–time curve from time 0 to the last measurable concentration; Cmax, maximum plasma concentration; GD, gestation day; NA, not assessed.
aDeath attributed to dosing procedure.
bStatistically significant versus control (P < 0.01).
cHistorical controls for males—mean: 4.09 g, range: 3.72 to 4.28 g.
dStatistically significant versus control (P < 0.05).
eHistorical controls for females—mean: 3.90 g, range: 3.64 to 4.09 g.
fHistorical controls combined—mean: 4.00 g, range: 3.74 to 4.19 g.
The TK evaluation (Table 4) revealed that exposure to tavaborole increased with increasing dose following single-dose (GD 6) and multiple-dose (GD 19) administration of 30, 100, and 300 mg/kg/d tavaborole. Area under the plasma concentration–time curve from time 0 to the last measurable concentration (AUC0-4) for rats dosed at 30, 100, and 300 mg/kg/d on GD 19 was 439, 2,199, and 43,223 ng·h/mL, respectively. Tavaborole was absorbed with a mean time to maximum plasma concentration (tmax) of approximately 0.239 to 1.01 hours on GD 6 and GD 19, respectively. Tavaborole appeared to exhibit nonlinear single- and multiple-dose TK characteristics and exhibited accumulation (∼2×) following the once-daily administration for 13 days at 30, 100, and 300 mg/kg/d. The maximum plasma concentration (Cmax) values on GD 6 were 946, 6,080, and 17,700 ng/mL following administration of 30, 100, and 300 mg/kg/d of tavaborole, respectively. The Cmax values on GD 19 were 729, 2,230, and 32,500 ng/mL following administration of 30, 100, and 300 mg/kg/d tavaborole, respectively.
Oral Embryo–Fetal Development Toxicity in Rabbits
Time-mated female rabbits were treated with tavaborole at dose levels of 0, 15, 50, and 150 mg/kg/d. Doses for the study were based on a range-finding study in nonpregnant rabbits dosed at 20, 100, 300, and 600 mg/kg/d for 10 days. In that study, rabbits in the 600 mg/kg dose group were preterminally euthanized after the first dose due to declining conditions, which consisted of hypoactivity, prostrate, ataxia, moribund, cool to touch, labored breathing, fecal-stained fur, and/or loose and scant stool. There were no test article-related antemortem findings at doses ≤300 mg/kg/d. The NOAEL was 100 mg/kg/d (AUC0-8h = 11,300 ng·h/mL), based on the lack of adverse findings in rabbits in the 100 mg/kg/d dose group.
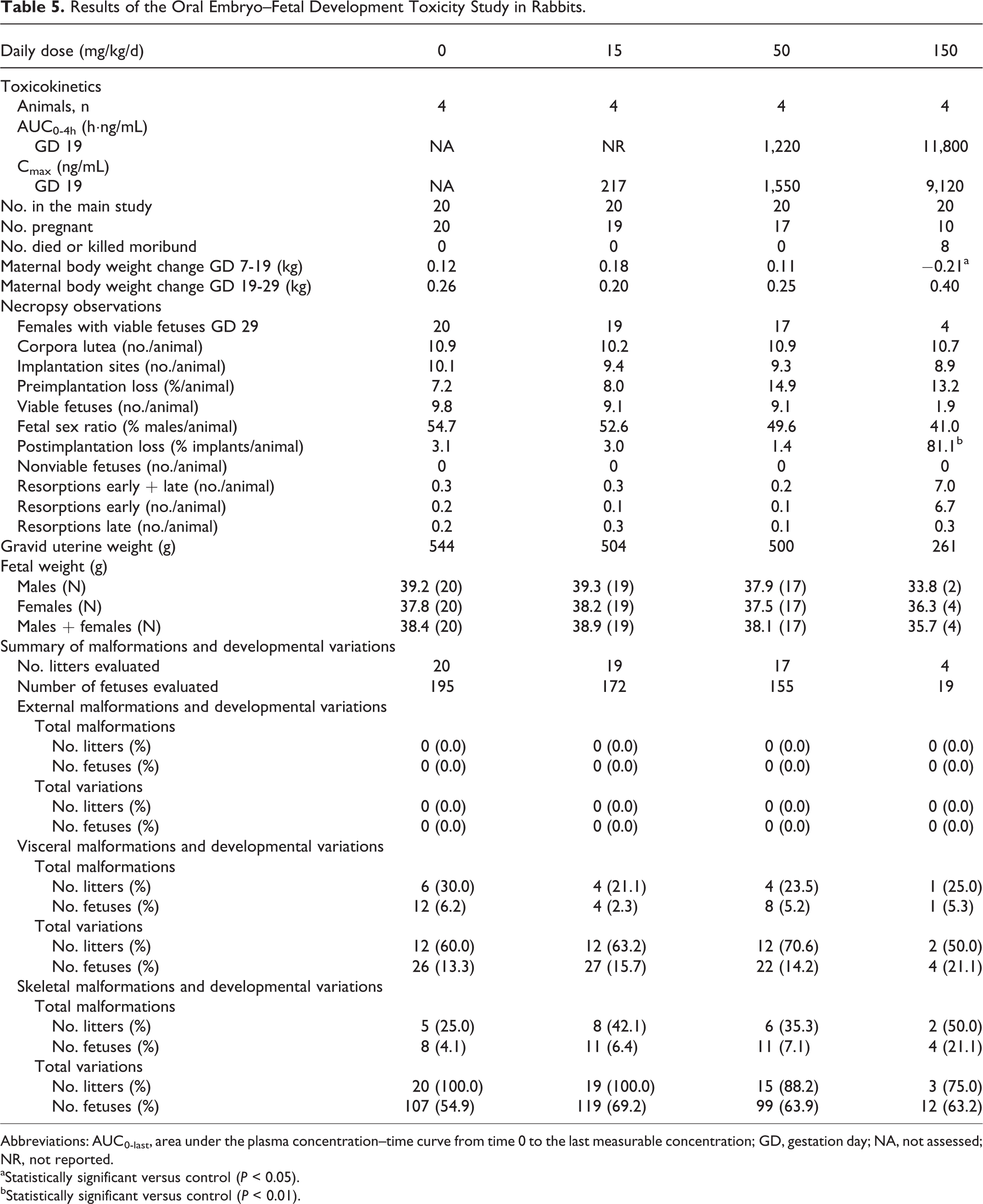
In the definitive study, at 150 mg/kg/d, 8 rabbits died or were killed moribund (Table 5). These rabbits exhibited evidence of abortion, stool findings (scant, no stool, loose, soft stool, and/or small fecal pellets) for 2 or more days between GD 8 and 22, red material in the cage pan for 1 to 10 days between GD 14 and 26, and abortion or no viable fetuses upon cesarean section. During the dosing period (GD 7-19), pregnant rabbits in the 150 mg/kg/d group exhibited statistically significant body weight loss or decreased body weight gain (mean of 210 g loss vs mean of 120 g gain in the control group) (Figure 5). Postimplantation loss in these rabbits likely contributed to these body weight changes. In the 50 mg/kg/d group, 3 rabbits exhibited scant stool and/or small fecal pellets during the dosing period; these stool findings were associated with decreased food consumption.
Results of the Oral Embryo–Fetal Development Toxicity Study in Rabbits.
Abbreviations: AUC0-last, area under the plasma concentration–time curve from time 0 to the last measurable concentration; GD, gestation day; NA, not assessed; NR, not reported.
aStatistically significant versus control (P < 0.05).
bStatistically significant versus control (P < 0.01).
Summary of female body weights in the oral embryo–fetal development toxicity study in rabbits.
At necropsy, there were no tavaborole-related gross findings in the surviving rabbits. In the 150 mg/kg/d group, there was an increase in postimplantation loss (81.1% in pregnant rabbits that were killed on GD 29 vs 3.1% in the control group), mainly due to an increased incidence of early resorptions. In this group, there were only 4 dams with viable fetuses, and even among the 4 dams, 2 had a single viable fetus. This resulted in a total of 19 live fetuses in this group. The effects on reproductive parameters in the 150 mg/kg/d group were not consistently associated with maternal toxicity such as decreased food consumption and related stool findings. There were no tavaborole-related placental findings. A decrease in mean fetal body weight was noted in the 150 mg/kg/d male group (13.8% relative to the control male fetuses). There were no tavaborole-related effects on fetal sex ratios, fetal external malformations/variations, or fetal visceral or skeletal findings at any dose (Table 5).
The toxicity observed in the rabbits dosed at 150 mg/kg/d in the definitive study was higher than expected based on the results of the range-finder study in nonpregnant rabbits. Even though the NOAEL in the range finder was 100 mg/kg/d, the definitive study was dosed higher and was conducted in pregnant rabbits that may have been more sensitive to the compound causing a reduction in food consumption and body weight at this dose.
The TK evaluation revealed that systemic exposure to tavaborole increased with increasing dose in a greater than dose-proportional manner as evidenced by a Cmax of 217, 1,550, and 9,117 ng/mL for doses of 15, 50, and 150 mg/kg/d, respectively, on GD 19 (Table 4). AUC0-4h values were 1,220 and 11,800 ng·h/mL for doses of 50 and 150 mg/kg/d, respectively. AUC values were not reported for the 15 mg/kg/d dose. The NOAEL for maternal toxicity and embryo–fetal toxicity was 50 mg/kg/d (AUC0-4h = 1,220 ng·h/mL; 16 times the MRHD) (Table 3).
Dermal Embryo–Fetal Development Toxicity in Rabbits
Time-mated female rabbits were treated with vehicle or tavaborole dose levels of 1%, 5%, and 10% topical formulation. No treatment-related mortality was observed at any dose level evaluated. No effect from treatment with tavaborole was evident in clinical observations, gestation body weights (Figure 6), body weight gain, food consumption (data not shown), and uterine implantation data. However, treatment-related local irritation was evident in 5% and 10% of tavaborole-treated groups and consisted of very slight to severe erythema at the dose site. Slight erythema was seen sporadically over the study period in 1 to 3 animals at 5% tavaborole. In contrast, over half (56%) of the animals treated with 10% tavaborole were observed with severe erythema by GD 24, which was considered to be an adverse effect of the test article. No effect from treatment with tavaborole was evident from fetal evaluations (external, visceral, and skeletal) at any dose level evaluated; however, fetal body weights were lower than controls (7%-10%) at 10% tavaborole, and these differences were considered treatment related (Table 6). Based on these results, the NOAEL and no observable effect level (NOEL) for maternal and developmental toxicity was 5% tavaborole, respectively. The TK evaluation revealed that exposure to tavaborole increased with increasing dose following single-dose (GD 6) and multiple-dose (GD 28) administration of 1%, 5%, and 10% tavaborole (Table 6). Tavaborole was absorbed with a mean tmax of approximately 0.722 to 1.59 hours on GD 6 and 28, respectively. Tavaborole appeared to exhibit linear single-dose pharmacokinetic characteristics following dermal administration of 5% and 10% tavaborole on GD 6 and nonlinear single-dose pharmacokinetic characteristics over the entire dose range studied (ie, 1%, 5%, and 10% tavaborole topical formulation). On GD 28, tavaborole appeared to exhibit linear multiple-dose pharmacokinetic characteristics following administration of 1%, 5%, and 10% tavaborole. No accumulation following the once-daily dermal administration of 1% tavaborole occurred; however, accumulation (∼2×) was observed following administration of 5% and 10% tavaborole once daily for 21 days. The Cmax (ng/mL) levels on GD 6 were 456, 527, and 806 and on GD 28 were 90, 1,043, and 1,855 for the 1%, 5%, and 10% tavaborole groups, respectively. The AUC (ng·h/mL) levels on GD 6 were 628, 970, and 1,928 and on GD 28 were 222, 2,041, and 3,128 for the 1%, 5%, and 10% tavaborole groups, respectively. The NOEL for developmental toxicity was 5% tavaborole solution (AUC0-4h = 1,977 ng·h/mL on GD 28; 26 times the MRHD (Table 3).
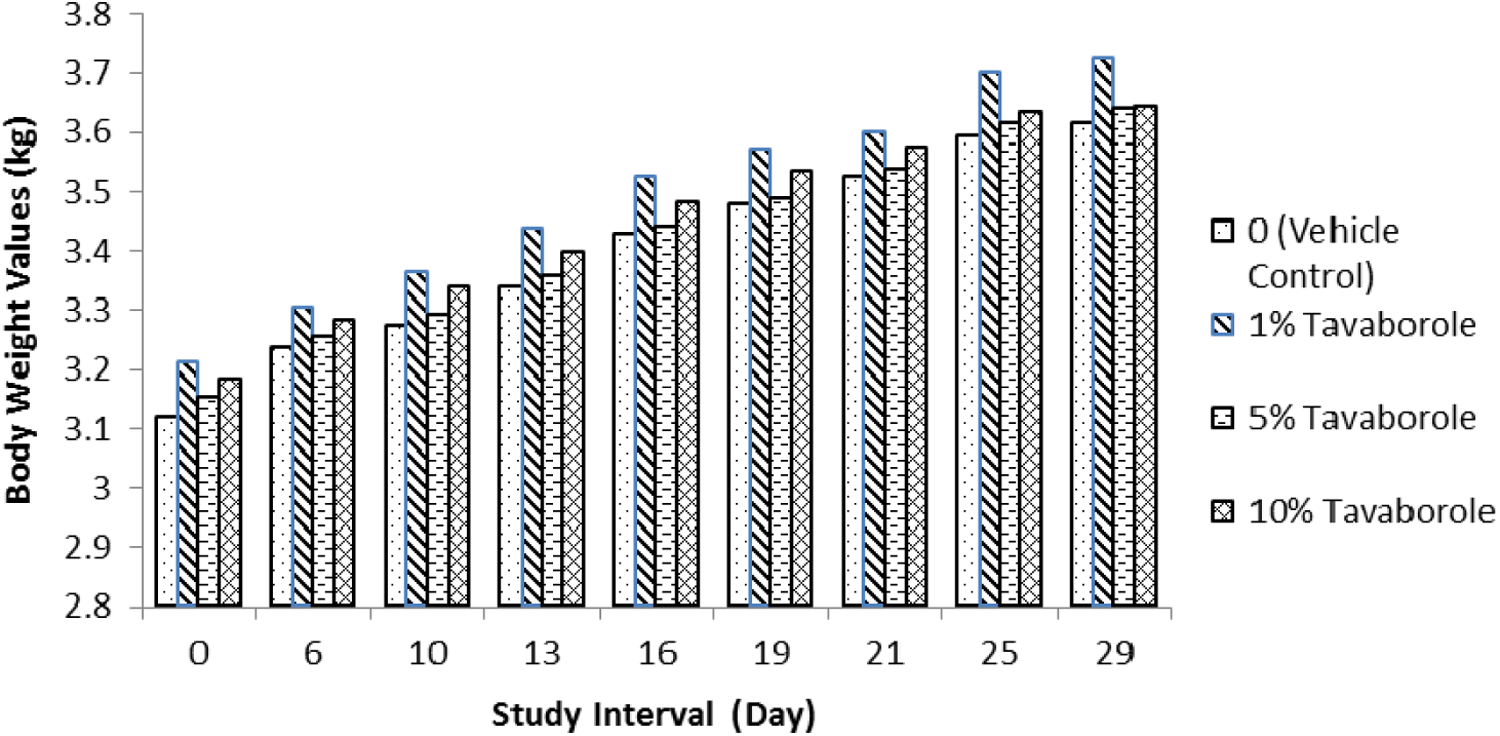
Summary of female body weights in the dermal embryo–fetal development toxicity study in rabbits.
Results of the Dermal Embryo–Fetal Development Toxicity Study in Rabbits.
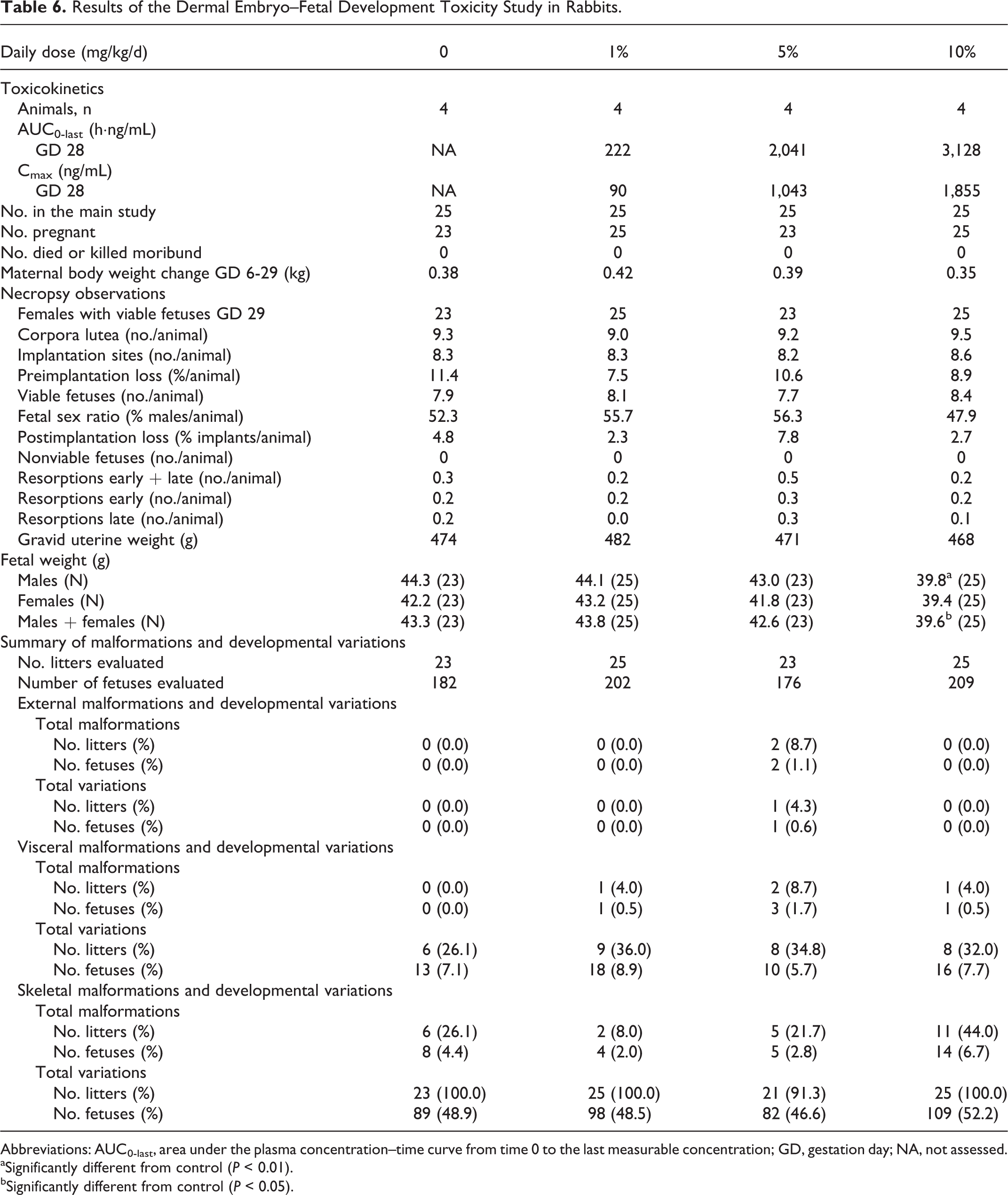
Abbreviations: AUC0-last, area under the plasma concentration–time curve from time 0 to the last measurable concentration; GD, gestation day; NA, not assessed.
aSignificantly different from control (P < 0.01).
bSignificantly different from control (P < 0.05).
Discussion
Tavaborole topical solution, 5% is FDA approved for the topical treatment of onychomycosis of the toenails due to Trichophyton rubrum or Trichophyton mentagrophytes. The reproductive toxicity potential of tavaborole, belonging to a new class of antifungal compounds (oxaborole), was investigated in reproductive and developmental toxicity studies including fertility and early embryonic development in rats (oral) and embryo–fetal development in rats (oral) and rabbits (oral and dermal). A summary of the results of these studies is shown in Table 7.
Summary of Toxicity Findings.
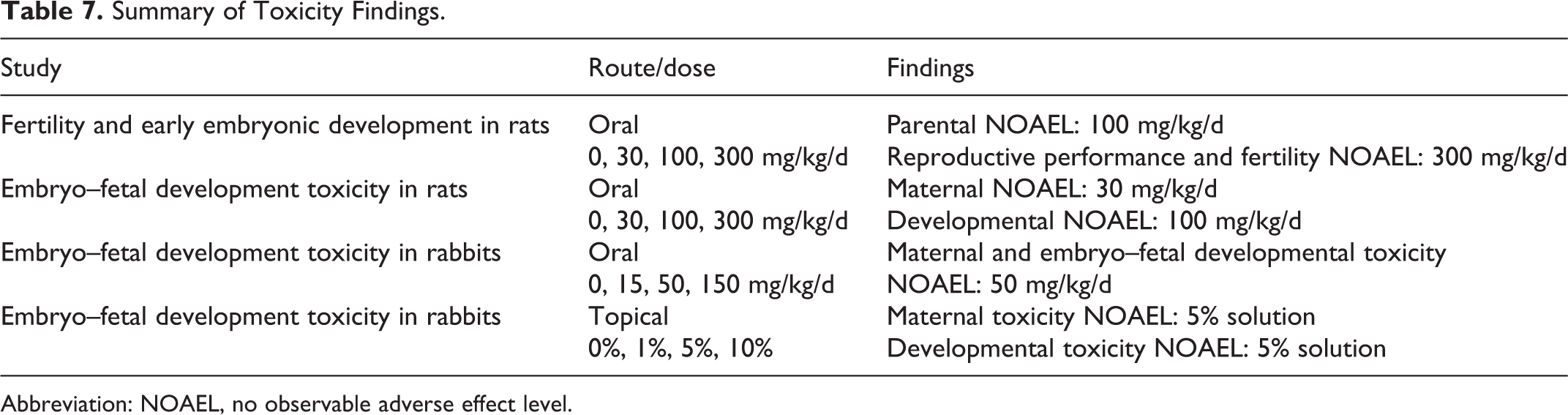
Abbreviation: NOAEL, no observable adverse effect level.
Compared to other approved antifungals, the results of the reproductive toxicology studies conducted with tavaborole exhibit higher safety margins. Oral reproduction studies with Lamisil (terbinafine hydrochloride) in rabbits and rats at doses up to 300 mg/kg/d resulted in safety margins that were 12 to 23× the MRHD, respectively, based on BSA comparisons. 6 Another antifungal, Sporanox (itraconazole) was found to cause a dose-related increase in maternal toxicity, embryotoxicity, and teratogenicity in rats at doses approximately 5 to 20× the MRHD and in mice at doses approximately 10× the MRHD. In rats, the teratogenicity consisted of major skeletal defects; in mice, it consisted of encephaloceles and/or macroglossia. 7
Studies revealed that under the conditions of the present studies, tavaborole was not considered a reproductive toxicant at doses that provided exposures up to 107 times the MRHD nor was it found to be a teratogen at doses that provided up to 29 times the MRHD of tavaborole (Table 3).
Footnotes
Authors’ Note
Studies were conducted at MPI Research, Inc, Mattawan, Michigan, Charles River Laboratories, Horsham, Pennsylvania, and Schering-Plough Research Institute, Summit, New Jersey.
Author Contributions
V. Ciaravino contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. D. Coronado contributed to analysis and interpretation and critically revised the manuscript. C. Lanphear contributed to analysis and interpretation and critically revised the manuscript. A. Hoberman contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. S. Chanda contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vic Ciaravino, Dina Coronado, and Sanjay Chanda are employees of Anacor Pharmaceuticals, Inc, Palo Alto, California.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
