Abstract
Purpose:
Aldicarb and methomyl are carbamate pesticides commonly implicated in human poisonings. The primary toxic mechanism of action for carbamate poisoning is cholinesterase (ChE) inhibition. As such, it is logical to assume that the currently accepted therapies for organophosphate poisoning (muscarinic antagonist atropine and the oxime acetylcholinesterase reactivator pralidoxime chloride [2-PAM Cl]) could afford therapeutic protection. However, oximes have been shown to be contraindicated for poisoning by some carbamates.
Methods:
A protective ratio study was conducted in guinea pigs to evaluate the efficacy of atropine and 2-PAM Cl. The ChE activity was determined in both the blood and the cerebral cortex.
Results:
Coadministration of atropine free base (0.4 mg/kg) and 2-PAM Cl (25.7 mg/kg) demonstrated protective ratios of 2 and 3 against aldicarb and methomyl, respectively, relative to saline. The data reported here show that this protection was primarily mediated by the action of atropine. The reactivator 2-PAM Cl had neither positive nor negative effects on survival. Both blood acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activities were significantly reduced at 15 minutes postchallenge but gradually returned to normal within 24 hours. Analysis of cerebral cortex showed that BChE, but not AChE, activity was reduced in animals that succumbed prior to 24 hours after challenge.
Conclusion:
The results suggest that coadministration of atropine and 2-PAM Cl at the currently recommended human equivalent doses for use in the prehospital setting to treat organophosphorus nerve agent and pesticide poisoning would likely also be effective against aldicarb or methomyl poisoning.
Introduction
Organophosphorus (OP) and carbamate compounds are widely used for pest control in agricultural, industrial, and household settings worldwide. Human exposure to either of these 2 classes of readily available pesticides may result from a deliberate act or accidental release, and each class poses a significant acute health risk to humans. 1,2 As such, it is critical that an adequate medical response strategy be available.
Carbamate compounds inhibit cholinesterases (ChEs), specifically acetylcholinesterase (AChE) and butyrylcholinesterase (BChE), preventing them from performing normal physiological functions. 3 At high levels of exposure, inhibition of ChEs by either carbamates or OPs rapidly leads to the accumulation of the neurotransmitter acetylcholine (ACh), the endogenous ligand for muscarinic and nicotinic receptors at synaptic junctions. 3 If left uncontrolled, the sudden and rapid increase in ACh levels at the synapses results in hyperstimulation of the cholinergic receptors and symptoms of cholinergic crisis. Unfortunately, based on symptomology alone, it is difficult to discern whether the observed cholinergic crisis is a result of carbamate or OP intoxication.
The immediate medical response paradigm in the United States for the treatment of intoxication with a ChE inhibitor is the administration of the muscarinic antagonist, atropine, and the oxime AChE reactivator, pralidoxime chloride (2-PAM Cl). 4 In prehospital first responder and military settings, this is typically accomplished using a Duodote autoinjector (Meridian Medical Technologies, Columbia MD), though no more than 3 such administrations are recommended in the absence of adequate supportive care (specifically, ventilator support). 5 It should be noted that the use of this Food and Drug Administration-approved medical countermeasure is primarily based on previous studies conducted using OP chemical warfare nerve agents 4,6 and not carbamates. Additionally, previous studies have reported some evidence that 2-PAM Cl is contraindicated against certain carbamate pesticides, particularly propoxur and carbaryl. 7 -9 Therefore, it is unclear whether the standard US countermeasure regimen of atropine and 2-PAM Cl should be administered following carbamate poisoning. 6
Aldicarb and methomyl are of concern because of the potential serious health effects following an acute exposure. Based on a similar mechanism of toxicity (ie, ChE inhibition), an evaluation of the effectiveness of the current standard of care approved for OP intoxication against members of the carbamate class of pesticides is warranted. As such, whether atropine and 2-PAM Cl are useful therapies against aldicarb and methomyl is the subject of this research. Efficacy studies in both guinea pig in vivo (protective ratio [PR] study and blood–brain barrier [BBB] penetration) and in vitro models (ChE evaluation and reactivation determination) were conducted to address this subject.
Materials and Methods
Materials
The challenge materials (CMs) were aldicarb (Sigma-Aldrich, St Louis, Missouri), methomyl (Chem Service, West Chester, Pennsylvania), and sarin (US Army Edgewood Chemical Biological Center, Edgewood, Maryland). Sarin (GB) was used as a positive control CM for the reactivation experiments. The therapeutics used were atropine (free base) at 1.64 mg/mL in an aqueous solution, pH 4.3, and 2-PAM Cl at 102.8 mg/mL in an aqueous solution, each procured from King Pharmaceuticals (St Louis, Missouri). The CM and test material used in the study are summarized in Table 1. The carbamates were diluted in multisol (a biocompatible solution of 48.5% water, 40% propylene glycol, 10% ethanol, and 1.5% benzyl alcohol, vol/vol).
Identification of Challenge and Test Materials.
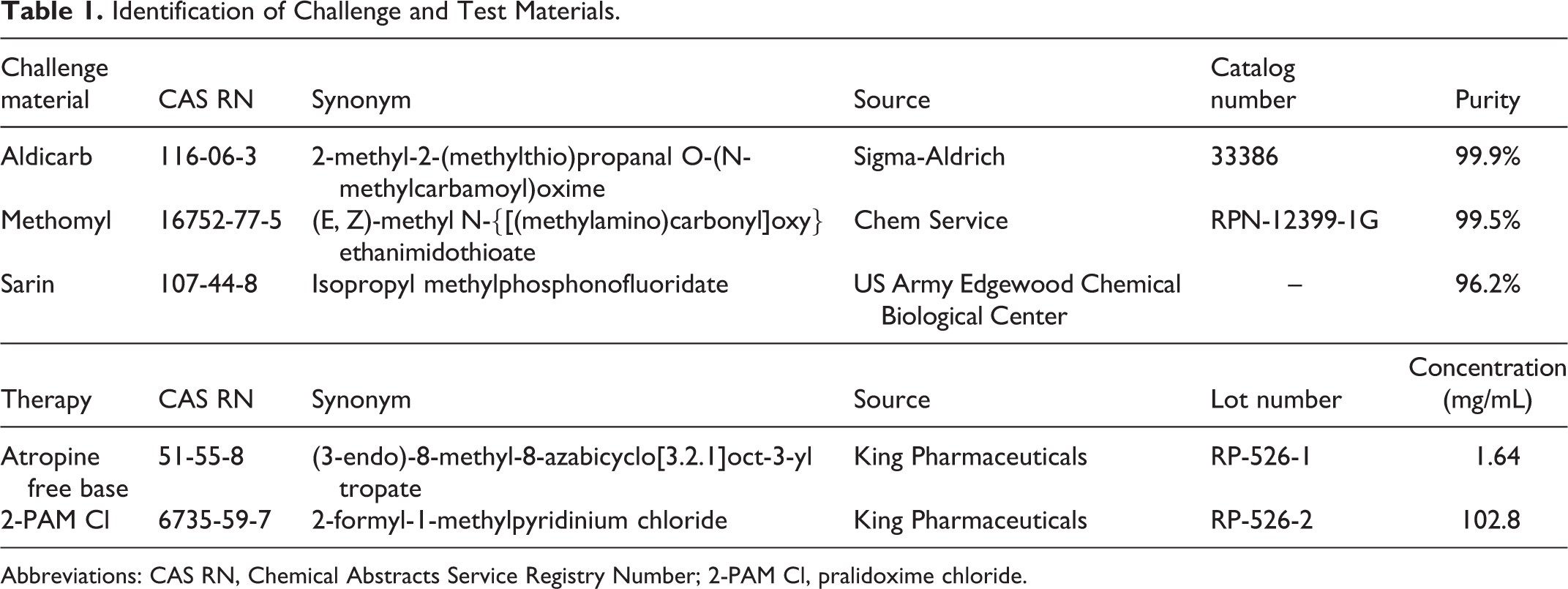
Abbreviations: CAS RN, Chemical Abstracts Service Registry Number; 2-PAM Cl, pralidoxime chloride.
Animals
A total of 292 male Dunkin Hartley guinea pigs (Cavia porcellus) were procured from Charles River facilities (Raleigh, North Carolina; Stone Ridge, New York). Each guinea pig had a vascular access port (VAP) surgically implanted in the jugular vein by the vendor prior to arrival. Animals were singly housed due to VAP implantation, and food and water were available ad libitum. During the 3-day quarantine, the guinea pigs were weighed and randomized by body weight into test days and treatment groups. This study was conducted under an approved protocol from Battelle (2979-CG920832).
In Vitro, 2-PAM Cl Reactivation of Recombinant Guinea Pig Acetylcholinesterase
Assay buffer (1× phosphate-buffered saline [PBS], pH 7.4 + 0.01% bovine serum albumin + 0.01% glycerol) was added to each well of a 96-well microtiter plate and prewarmed at 37°C for ≥60 minutes. Vehicle or 2-PAM Cl was added to the prewarmed plate for a final concentration of 10, 100, or 500 μmol/L per the test conditions being evaluated. Concurrently, 0.8 mL of assay buffer was added to microtiter tubes and prewarmed at 37°C for ≥60 minutes. To each prewarmed microtiter tube, 0.1 mL of diluted recombinant guinea pig acetylcholinesterase (gpAChE-R, read-through transcript, AA976; Chesapeake PERL, Savage, Maryland) was added. The target enzyme activity was 0.1 U/mL. Subsequently, 0.1 mL of varying concentrations of aldicarb, methomyl, or sarin was added at the experimentally determined concentration of the CM to result in 95% inhibition of the enzyme (IC95). The targeted IC95 for each inhibitor was the inhibition concentration that was determined to yield a final relative activity of ∼5% of the unchallenged control samples on the plate. Samples were incubated at 37°C for approximately 1 minute in order to mimic the in vivo study. Twenty microliters of the challenged gpAChE-R was added to each of the appropriate wells on the plate. At t = 0, 0.5, 1, 2, 4, and 24 hours, 20 μL of a 1:1 mixture of acetylthiocholine (ATC) iodide (A5751; Sigma-Aldrich) and Ellman reagent (5,5′-dithiobis(2-nitrobenzoic acid) [DTNB], D8130; Sigma-Aldrich) was added to each well. The final concentrations of ATC and DTNB were 1.00e−03 mol/L and 5.00 e−04 mol/L, respectively. For all 37°C incubations, the plates and/or microtiter tube boxes were covered with lids to minimize evaporation. At this point, each well of the test plate contained a final volume of 0.2 mL. Each plate was sealed and kinetically analyzed by spectrophotometric readings at a wavelength of 412 nm, obtained using a SpectraMax Plus384 microplate reader (Molecular Devices, Sunnyvale, California) programmed to incubate the plate at 37°C for the duration of the experiment. Readings were taken every 15 seconds for a period of 10 minutes. For analysis purposes, plates were normalized to a 1-cm path length, and the extinction coefficient of DTNB used was 13 600 M−1 cm−1. Furthermore, wells containing identical 2-PAM Cl concentrations but without gpAChE-R were evaluated. These wells served as oximolysis controls, and the values obtained were subtracted during data analysis to determine the effect of 2-PAM Cl reactivation on challenged gpAChE-R. As described by Wille and colleagues, 9 to calculate the reactivation rate constants, relative activity (determined by reference to identically treated control samples) was plotted versus time using GraphPad Prism 5 (GraphPad Software, Inc, La Jolla, California), and plots were fit to a 1-phase exponential, nonlinear regression model.
Protective Ratio Experimental Study Design
Probit analysis of 24-hour lethality rate as a function of challenge dose was used to calculate the median lethal dose (MLD) for each carbamate with and without treatment. The treatments were one of 4 different combinations designated as groups in Table 2. The PR for a particular treatment was defined as the MLD for that treatment divided by the MLD for the saline/saline control group.
Treatment Combinations.
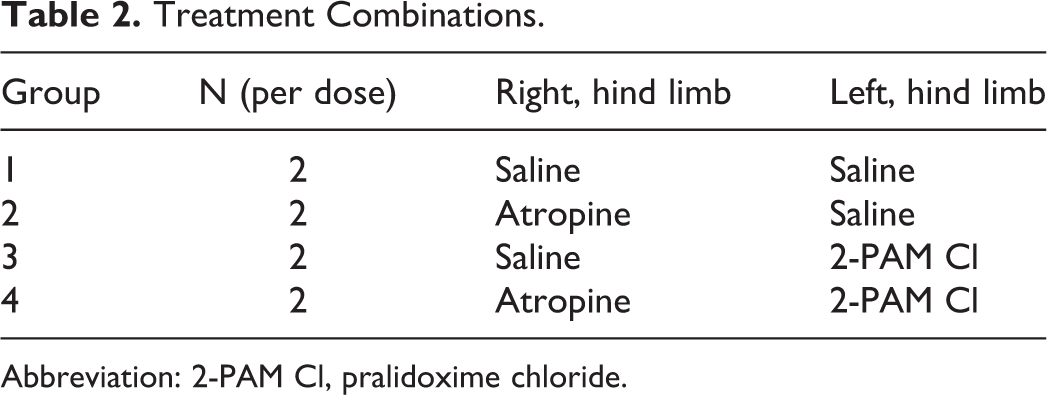
Abbreviation: 2-PAM Cl, pralidoxime chloride.
On the day prior to challenge, each animal was weighed to ensure that it was within the designated weight range (350-500 g) and to provide the basis for calculating the challenge and treatment doses. A blood sample was collected for a baseline ChE analysis, and the animals were clipped of hair on the lateral aspects of the right and left thighs (treatment injection sites) and on the dorsum between the scapulae (challenge injection site).
On each of 5 challenge days, 3 sets of 2 guinea pigs each were administered varying doses of either aldicarb (0.05-25 mg/kg) or methomyl (0.9-450 mg/kg) by subcutaneous injection at the challenge site. Challenge doses were selected based on statistical analysis in order to provide the points needed to complete a dose/lethality curve for each challenge agent/treatment combination while minimizing animal use. At 1 minute postchallenge, one of 4 treatment combinations was administered by intramuscular injection into the hind limbs of the animal as detailed in Table 2. In addition to the carbamate-challenged animals, 2 control animals per challenge day were exposed to the vehicle only.
The standard therapy for OP poisoning a first responder can administer is 3 DuoDote autoinjectors, which contain both atropine free base and 2-PAM Cl. The atropine free base level of 0.4 mg/kg in the guinea pig was selected for this study based on the body surface area-corrected equivalent dose given to a human (the human equivalent dose) victim of OP poisoning in a prehospital setting or given by first responder, 3 DuoDote autoinjectors. 5 The 2-PAM Cl dose administered to the guinea pig was 25.7 mg/kg (146 μmol/kg)—equivalent to the available dosage in 3 DuoDote autoinjectors given to a 70-kg human (the human relevant dose).
The primary end point, 24-hour survival, was used to determine the PR of the treatment groups. Secondary end points included clinical observations and ChE activity measurements in serial blood and terminal brain cortex samples. Clinical observations were recorded prechallenge and at 5, 15, 30, 60 minutes and at 2, 4, 6, and 24 hours postchallenge. If a guinea pig was found dead, the time was recorded upon observation. Using the VAP, blood samples were collected prior to challenge and at 15 and 60 minutes and at 2, 4, and 24 hours postchallenge. In addition, animals that succumbed prior to the 6-hour postchallenge time point were necropsied immediately, and the cerebral cortex was collected for analysis. After the final observation, each surviving animal was euthanized by catheter injection of euthanasia solution, and the cerebral cortex tissue was harvested. The cerebral cortex was rinsed of residual blood; however, it was not perfused.
Blood Processing and ChE Activity Assay
Whole blood samples were processed and analyzed as described by McGarry et al. 10 Briefly, whole blood samples were treated with HemogloBind to remove hemoglobin, which interferes with the ChE activity assay due to spectral overlap. To prepare the HemogloBind treated blood samples for ChE activity analysis, samples were diluted 2-fold in assay buffer (1× PBS). Subsequently, samples were diluted an additional 2-fold into the test plate by adding 100 μL of sample to a total volume of 200 μL in each well of a 96-well plate. Cholinesterase activity was assessed using a spectrophotometric assay conducted in a manner similar to Ellman et al, 11 as described in the in vitro reactivation section above. The relative AChE activity level for each animal (RAAChE) was defined as the ATC turnover rate in the terminal blood sample divided by that in the same animal’s baseline blood sample. A similar calculation was performed using butyrylthiocholine (BTC) turnover rates to determine RABChE.
Tissue Processing and ChE Activity Assay
After brain extraction and resection of the perfused cerebral cortex, the tissue was immediately rinsed to remove any residual blood. The sample was then flash frozen in liquid nitrogen and stored at ≤−70°C until processing. Each tissue sample was later homogenized using one of 2 methods. In the first method, cerebral cortex samples that were used for ex vivo ChE activity analysis and BBB permeability determination were pulverized using a CryoPrep system (Covaris, Woburn MA). Briefly, ∼1 g of frozen tissue was placed into a Kapton tissue tube (Covaris), placed into liquid nitrogen, and pulverized. The resulting coarse powder was then added to appropriately sized homogenization vials and weighed. Tissue homogenization buffer (1× PBS, pH 7.4 + 1% TritonX-100 + 1% protease inhibitor; Sigma-Aldrich) was added to the pulverized cerebral cortex to create a 100 mg/mL solution. The samples were then homogenized for approximately 2 minutes with a PowerGen High-Throughput Homogenizer (Fisher Scientific, Waltham MA). Samples requiring analysis for both ChE activity and BBB penetration studies (described below) were homogenized using the CryoPrep system, as this allowed an equal division of the samples between the 2 analysis teams. In the second method, cerebral cortex samples collected from animals used for the efficacy study were added to appropriately sized homogenization vials and weighed. Tissue homogenization buffer, composed of the same components described for the first method, was added such that the final tissue concentration was 100 mg/mL. The samples were then homogenized for 1 to 2 minutes using an Omni tissue homogenizer (Omni International, Kennesaw GA). Upon completion of either of the 2 aforementioned homogenization methods, the homogenized samples were placed on a rocker at 2°C to 8°C and allowed to rock for ≥1 hour. Samples were then centrifuged at 10 000g for 2 minutes. The supernatant was removed and retained at ≤−70°C for ChE activity analysis. Prior to ChE activity analysis, the homogenate was thawed and diluted 5-fold in assay buffer (1× PBS). Cholinesterase activity was then determined as described previously.
Blood–Brain Barrier Penetration Determination
Five animals per group were challenged at multiples of the determined MLD: 0.5, 1, and 2 × MLD. A prechallenge and a 30-minute postchallenge blood samples were collected for ChE analysis and determination of carbamate concentration. Following the 30-minute postchallenge blood collection, the brain was perfused with 0.9% saline solution to remove any blood from the tissue for analysis of the CM. Similar experimental design to assess the BBB permeability to an administered compound has been previously described. 12 Briefly, following perfusion, the cerebral cortex was removed and the tissue was prepared using a Covaris CryoPrep system for extraction and analysis. After preparing the tissue samples, acetonitrile was added, the samples were vortexed with a 0.375-in stainless steel homogenization ball for 30 seconds, and then centrifuged at 1,300g for 10 minutes. The resultant clear supernatant was added to approximately 0.2 g of 4:1 magnesium sulfate:sodium acetate. The samples were vortexed and then centrifuged at 1,300g for 1 minute at 4°C. If necessary, the supernatant was further processed by transferring the supernatant to approximately 0.05 g dispersive solid-phase extraction sorbent (3:1:1 magnesium sulfate:primary and secondary amine exchange material/C18). The samples were again vortexed and centrifuged at 1,300g for 1 minute at 4°C. The supernatant was diluted 2-fold with Millipore water, and the samples were directly analyzed for the CM using liquid chromatography–mass spectrometry/mass spectrometry. The aldicarb or methomyl was identified by retention times and specific multiple reaction monitoring transitions. Quantification was performed using the isotopically labeled carbamates as internal standards.
Statistical Analysis
For each compound and treatment, a probit dose–response model was fit to lethality data using the method of maximum likelihood. 13 Estimated parameters of probit dose–response models were used to compute each MLD. The Fieller method or the delta method was used to compute a 95% confidence interval for each MLD. STATA 11.0 (StataCorp, College Station, Texas) was used to analyze the lethality data.
The calculated MLD for the treated groups, when divided by values for the nontreated groups (saline/saline), provided an estimated PR for that particular treatment against each carbamate. The PR is the expected protection that a particular treatment provides against a particular poisoning. The PRs for treatment groups were calculated as
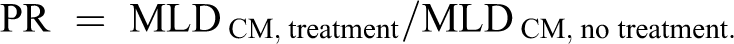
Protective ratios and 95% confidence intervals were calculated for each pair of treatment groups for each compound. The analysis used pairwise tests to compare treatment group means, and significance was adjusted for multiple comparisons using the Bonferroni correction.
Results
Reactivation of AChE Inhibited With Aldicarb or Methomyl
To determine 2-PAM Cl-assisted reactivation of AChE inhibited by aldicarb or methomyl, the IC95 of each was experimentally determined to ensure near-complete inhibition of the gpAChE-R without an excess of the carbamate in solution (Table 3). The capacity of 2-PAM Cl to reactivate AChE inhibited by sarin has been well characterized. 14,15 Therefore, sarin was used as a positive control for the reactivation experiments.
Determination of Aldicarb and Methomyl IC50 and IC95 With gpAChE-R.

Abbreviations: gpAChE-R, recombinant guinea pig acetylcholinesterase; IC95, 95% inhibition of the enzyme; 2-PAM Cl, pralidoxime chloride.
aThe IC95 was used to determine the ability of 2-PAM Cl in reactivating gpAChE-R when inhibited by carbamate (Figure 1).
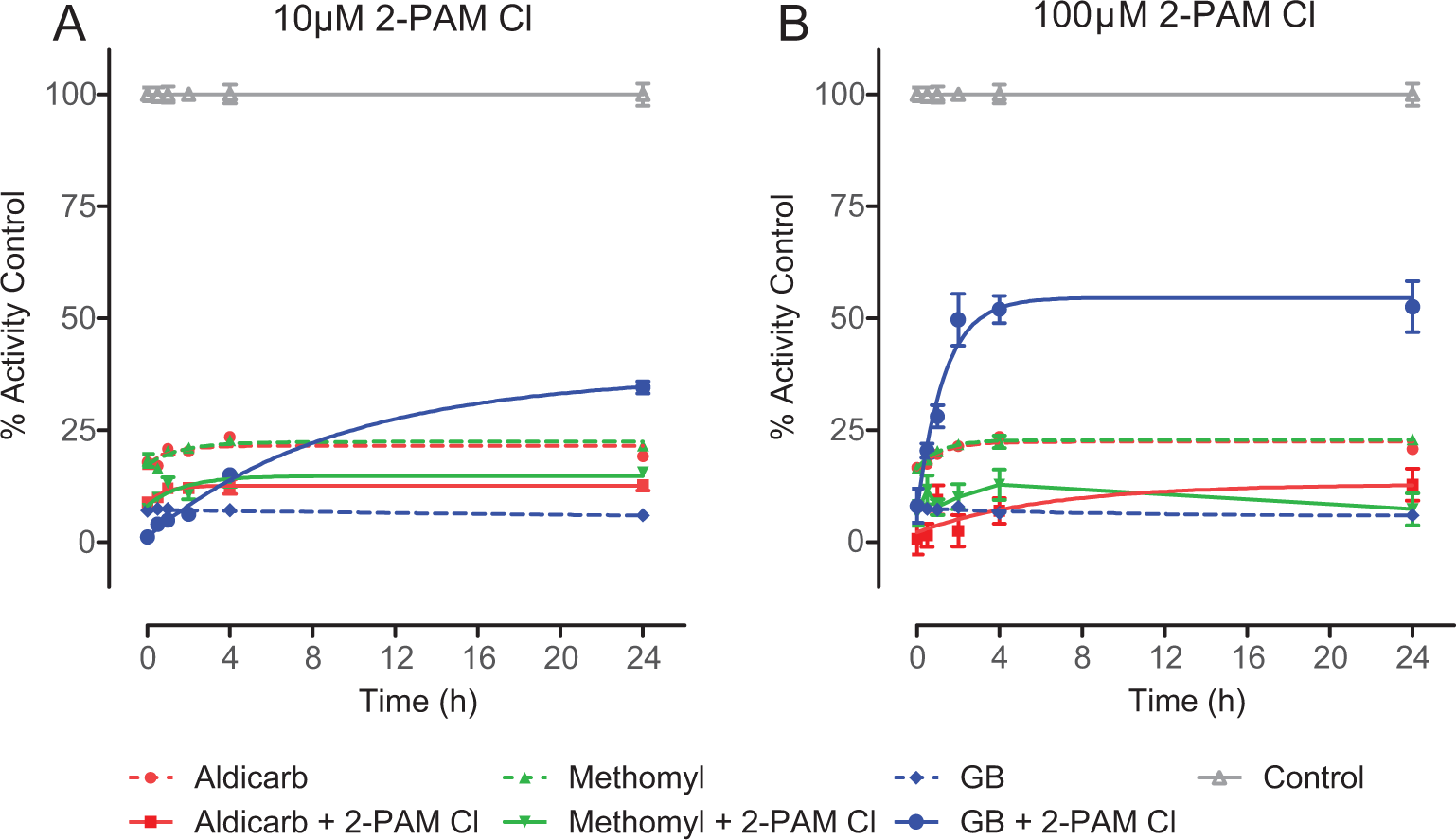
In vitro, pralidoxime chloride (2-PAM Cl) does not reactivate recombinant guinea pig acetylcholinesterase (gpAChE-R) when inhibited by a carbamate. Using the Ellman-based assay, the reactivation of gpAChE-R by 2-PAM Cl, against the experimentally determined 95% inhibition of the enzyme (IC95) of aldicarb (509 μmol/L), methomyl (174 μmol/L), or sarin (GB; 0.475 μmol/L) was evaluated. Unchallenged gpAChE-R was used to determine maximum activity. To more closely mimic the in vivo study, at 1 minute postchallenge, 2-PAM Cl was added at a final concentration of (A) 10 μmol/L or (B) 100 μmol/L. The indicator and substrate were added at 0, 0.5, 1, 2, 4, or 24 hours and read immediately to determine activity. Activity is represented as a percentage of the unchallenged control. Data shown are mean ± standard error of the mean (SEM), representing n ≥ 5. For both carbamates but not Sarin GB, both treatment levels of 2-PAM Cl significantly contributed to additional enzyme inhibition. For GB, both treatment levels of 2-PAM Cl significantly ameliorated enzyme inhibition. The gpAChE-R remained relatively stable for the duration of the 24-hour incubation, as ≤15% decrease in activity was observed as compared to T = 0 hours (data not shown).
The gpAChE-R was challenged at the previously determined IC95 of aldicarb, methomyl, or sarin. One minute postchallenge, 2-PAM Cl was added at either 10 or 100 μmol/L, and samples were analyzed for enzyme activity at specific time points. The 10 and 100 μmol/L 2-PAM Cl levels were selected based on results from a clinical study investigating the effectiveness of 2-PAM Cl at different administered doses against OP pesticide poisoning cases. 16 Notably, 500 μmol/L 2-PAM Cl produced a signal-to-noise ratio that was too low to distinguish specific reactivation effects due to oximolysis, and the data are not reported here.
As shown in Figure 1, aldicarb, methomyl, and sarin elicited inhibition of AChE activity of the gpAChE-R relative to the unchallenged control samples. Although the experimentally determined IC95 was targeted, approximately 80% to 85% inhibition of gpAChE-R was attained in this set of experiments. The reduction in AChE activity due to aldicarb or methomyl was not reversed by the addition of 2-PAM Cl at either 10 μmol/L (Figure 1A) or 100 μmol/L (Figure 1B). In fact, a decrease of ∼10% to 15% activity was observed between the 2-PAM Cl-treated and the 2-PAM Cl-untreated aldicarb and methomyl-challenged gpAChE-R. This decrease in relative activity, which was presumably due to the oxime reversibly inhibiting the gpAChE-R, 17 was observed to be ∼20% for 10 μmol/L 2-PAM Cl alone and ∼35% for 100 μmol/L 2-PAM Cl alone (data not shown). This reduction could account for the difference observed between the carbamate only and carbamate plus 2-PAM Cl. This decrease in activity as a result of addition of the oxime was not observed for the sarin control, presumably because the increase in activity due to reactivation of sarin-inhibited gpAChE-R greatly exceeded the relatively weak inhibition of gpAChE-R by 2-PAM Cl.
Reactivation rate constants (kreact) are presented in Table 4. Neither concentration of 2-PAM Cl had a significant effect on the kreact of gpAChE-R inhibited with aldicarb or methomyl (Table 4). In order to establish that 2-PAM Cl could reactivate AChE in this in vitro model, the OP nerve agent sarin was tested concurrently under similar conditions. Previously reported data have shown that 2-PAM Cl is an effective reactivator of sarin-inhibited AChE. 14,18 In the present study, sarin inhibited gpAChE-R activity and the addition of 2-PAM Cl resulted, as expected, in reactivation in a dose-dependent manner (Figure 1A and B).
Reactivation Rate Constant (k react).
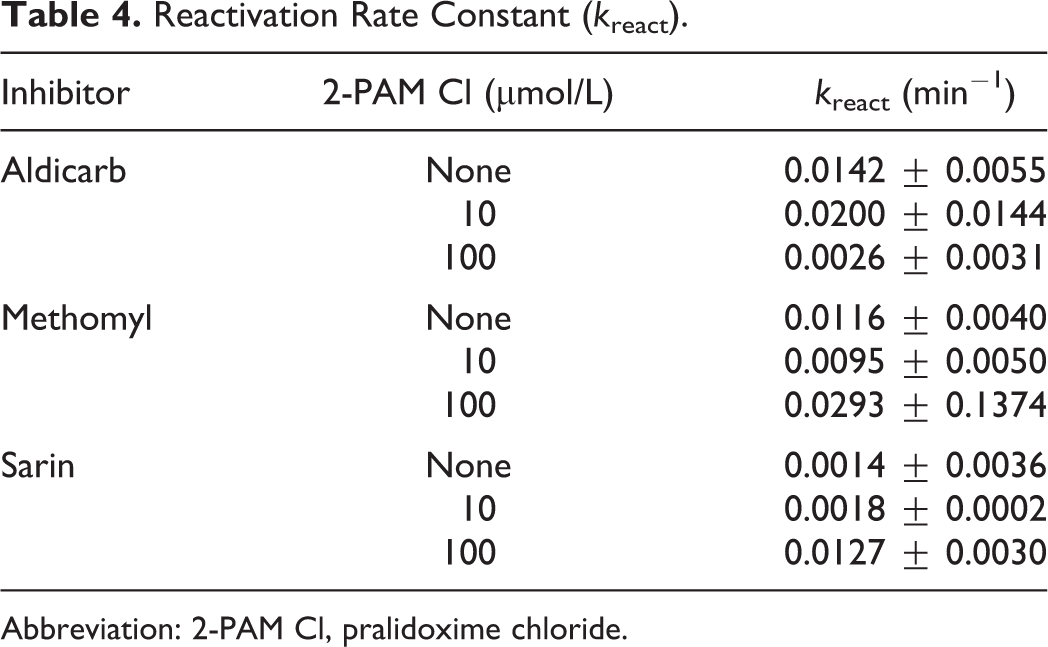
Abbreviation: 2-PAM Cl, pralidoxime chloride.
Protective Ratio Study
The probit dose–response analysis was calculated from the lethality data from each treatment combination (Table 2). Each probit analysis involved 30 guinea pigs to characterize the curves and establish the MLD. Once the probit curves (Figure 2) were established and the MLD was calculated for each treatment group, PRs relative to saline only controls (ie, negative controls) were calculated by dividing the MLD for a treatment group by that of the saline/saline-treated control group (Table 5).
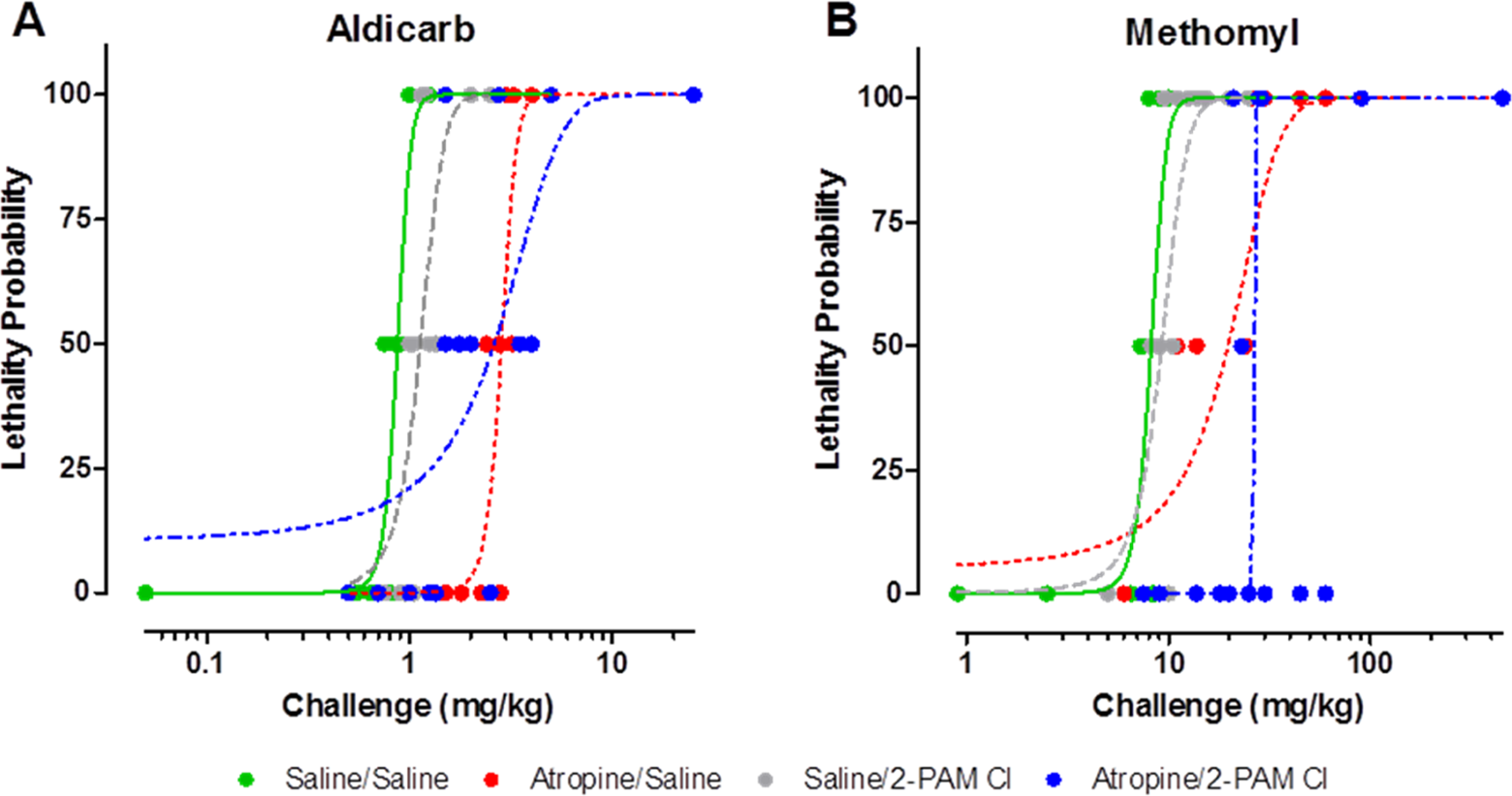
Atropine treatment alone produces a 2- to 3-fold protection over saline treated when challenged with Aldicarb (A) and Methomyl (B). Pralidoxime chloride (2-PAM Cl) afforded no additional protection. Probit analysis was performed based on the lethality data using Dunkin Hartley guinea pigs results from the protective ratio study. Varying doses were used in this in vivo study to determine a probit curve for each treatment group. These probit analyses were then compared to calculate the protective ratios. For each data point, n = 2; for each probit curve, n = 30 per curve. See Table 6 for statistical analysis results.
MLD and Protective Ratios for Treatment Groups.
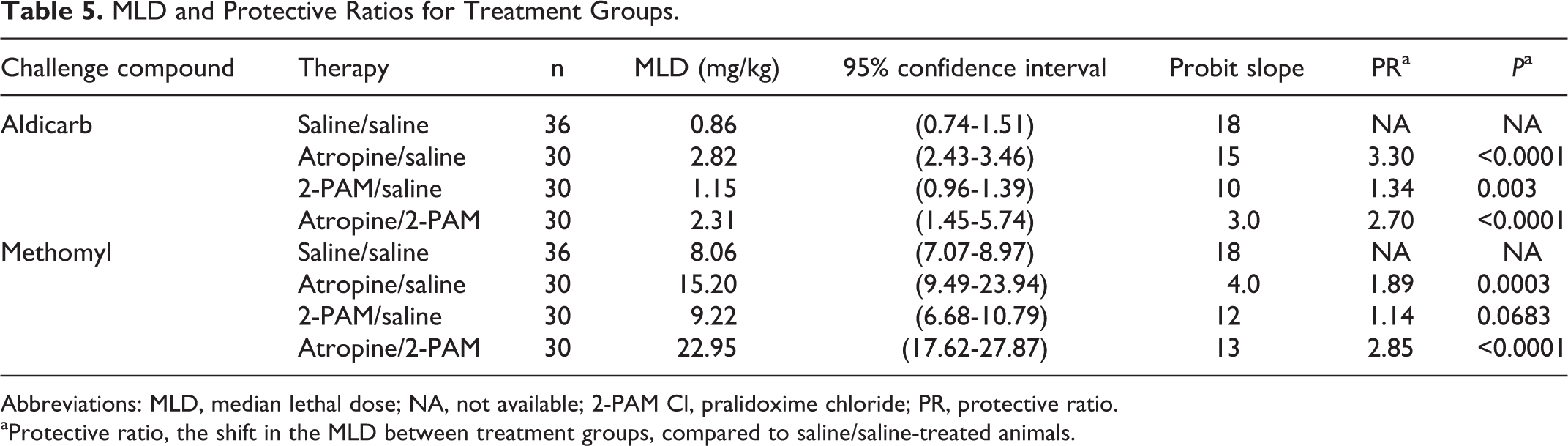
Abbreviations: MLD, median lethal dose; NA, not available; 2-PAM Cl, pralidoxime chloride; PR, protective ratio.
aProtective ratio, the shift in the MLD between treatment groups, compared to saline/saline-treated animals.
A comparison of the calculated PRs based on intercomponent comparison is presented in Table 6. Atropine provided a significant level of protection (P < 0.0083) to animals challenged with aldicarb or methomyl, whereas 2-PAM Cl alone did not have any effect on the PR (P < 0.0083; Table 6).
Protective Ratio Comparison.
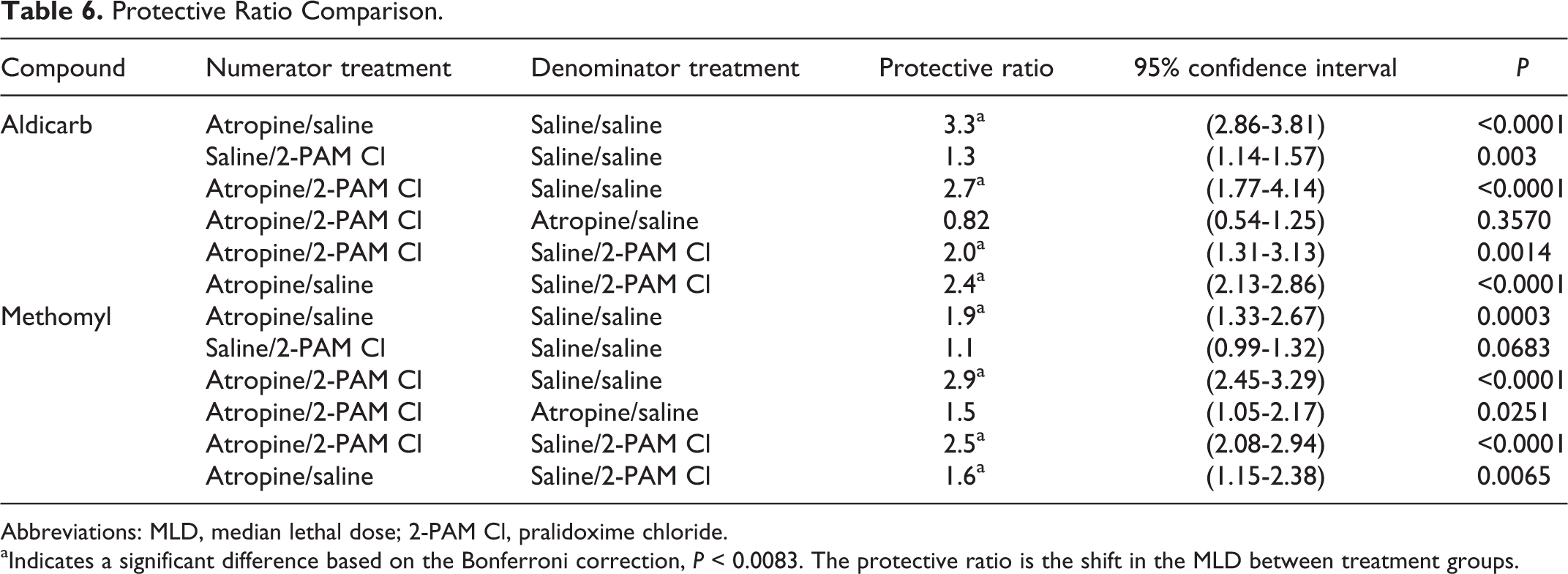
Abbreviations: MLD, median lethal dose; 2-PAM Cl, pralidoxime chloride.
aIndicates a significant difference based on the Bonferroni correction, P < 0.0083. The protective ratio is the shift in the MLD between treatment groups.
Additionally, clinical observations were recorded throughout the 24-hour postchallenge observation period. At least 80% of the animals that showed signs of cholinergic overstimulation had increased salivation, lacrimation, hyperpnoea, and tremors; the severity and time to onset of these signs depend on dose of CM and the presence of treatment (data not shown). More than 90% of those animals that survived for the 24-hour observation period were normal within 6-hour postchallenge (data not shown).
Acetylcholinesterase and BChE Activity in the Blood and Cerebral Cortex Following Carbamate Challenge
For aldicarb challenges, blood enzyme assays indicate that both AChE and BChE (evaluated by ATC and BTC hydrolysis, respectively) were inhibited rapidly (Figure 3A and B). The ChE activity returned to baseline within the 24-hour observation period. There was no difference between the treatment groups with regard to ChE activity trends.
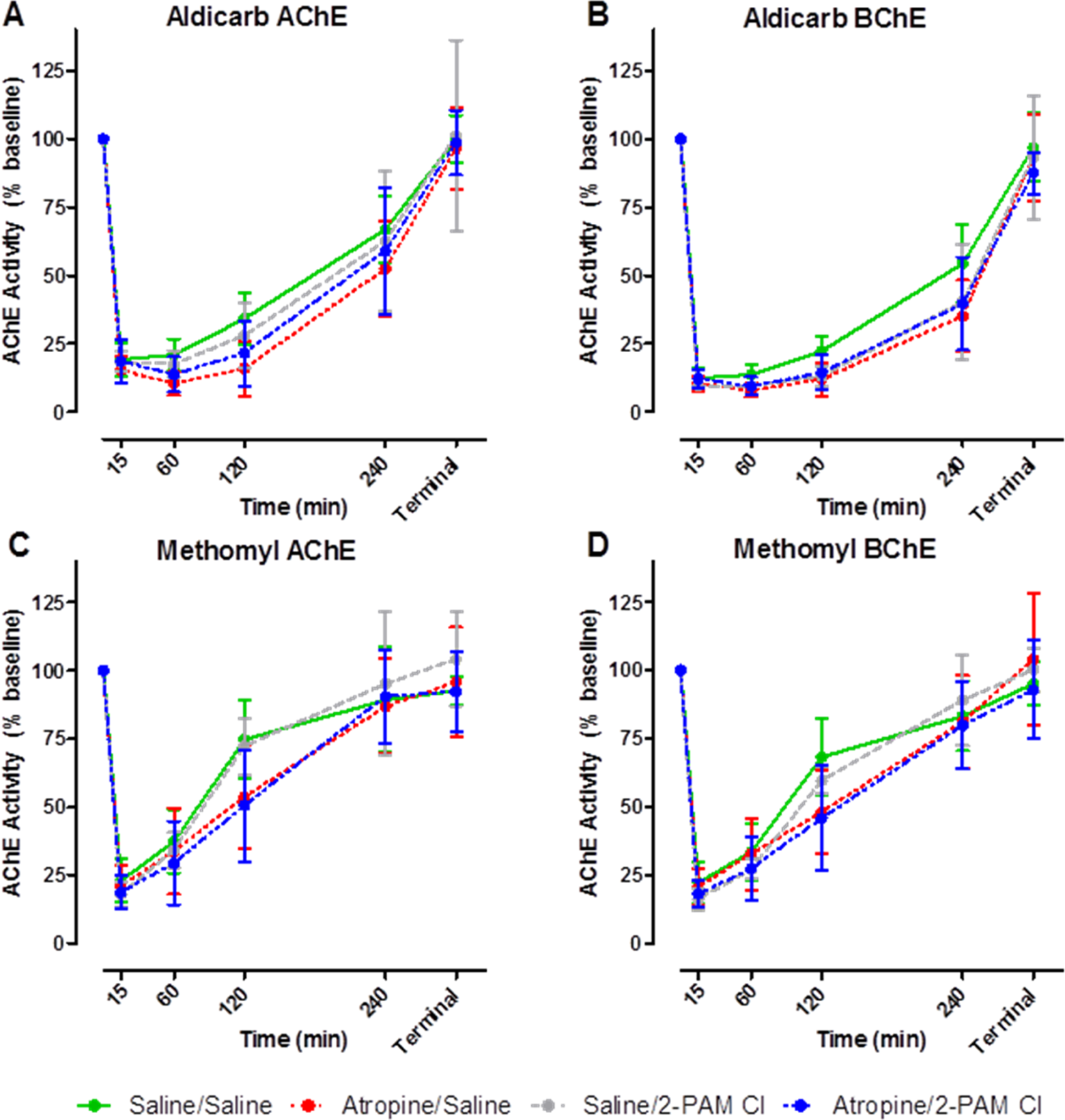
Serial blood samples were taken, and acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activity was determined following challenge with aldicarb (A and B) or methomyl (C and D). Within 15 minutes following challenge, cholinesterase (ChE) activity was reduced by ∼75% but returned to prechallenged levels within 24 hours regardless of treatment. The ChE analysis was performed using an Ellman-based assay. Animals were challenged with varying concentrations of aldicarb or methomyl, and blood was collected at specific time points postchallenge: 0, 0.25, 4, 2, 4, 24 hours. Each time point is the mean ± standard error of the mean (SEM) of all samples collected for that treatment and challenge material (CM) group (n ≥ 6) including varying CM concentrations. Due to the varying challenge doses with each treatment group, these data only reflect trends in ChE activity, and no statistical analysis was able to be conducted.
The ChE activity in methomyl-challenged animals was very similar to the aldicarb-treated animals (Figure 3C and D). The only noticeable difference was that the rate of recovery to baseline was faster for methomyl- than for aldicarb-challenged animals. No statistical analysis was able to be conducted between specific data points due to the nature of the varying challenge doses in the development of the probit analysis. All data based on treatment group were pooled at each specific time point, and only trends in ChE activity were observed.
Cortex AChE and BChE activities are presented in Figure 4. There were no observable differences in ChE activity between the treatment groups regardless of CM. However, there appears to be a distinct preference of the carbamate for cortical BChE (Figure 4B and D) over AChE (Figure 4A and C). The AChE activity in the cerebral cortex was not affected by carbamate challenge. Interestingly, in the animals that died during the observation period (ie, prior to 24 hours postchallenge), an elevation (∼15%) in AChE activity from the baseline control animals was observed. The observed elevation in activity could be due to stress-induced read-through AChE in response to the carbamate challenge since it is known that a stress-induced read-through isoform of AChE (AChE-R) can be produced 19 and can be observed within 30 minutes. 20 On the other hand, BChE activity was affected based on time to death. If an animal survived the full 24-hour observation period, its BChE activity levels were observed to be near baseline. However, if the animal died due to challenge, the level of BChE activity was reduced nearly 75%. This difference between the BChE activity based on survivability was statistically significant (P < 0.0001).
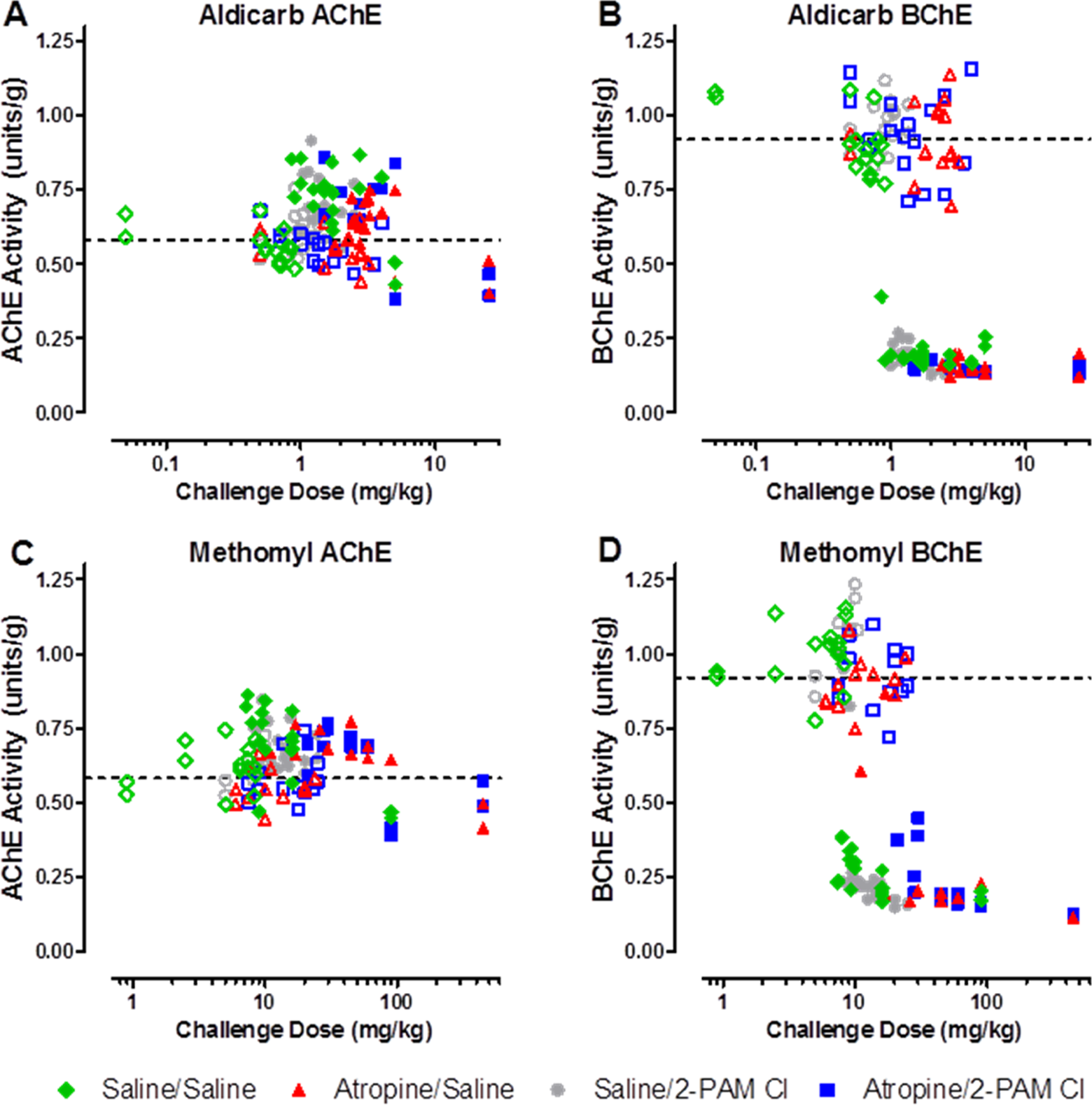
Terminal acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activity in the cerebral cortex. Acetylcholinesterase is unaffected by either aldicarb (A) or methomyl (C), however, BChE activity is significantly reduced by both aldicarb (B) and methomyl (D). Upon death due to challenge or at the completion of the 24-hour observation period, the cortex was removed and ChE analysis was performed using an Ellman-based assay. Closed circles represent animals that succumbed to challenge prior to 24 hours postchallenge. Open circles represent those animals that were euthanized following the 24-hour observation period. The dashed line is the average of all untreated control animals, n = 36. The AChE/BChE activity is recorded in enzyme units per gram of tissue.
Blood–Brain Barrier Permeability of Aldicarb and Methomyl
There were no significant differences between aldicarb levels in the blood and cerebral cortex within 30 minutes of challenge (Figure 5). Methomyl, on the other hand, produced an approximate 40% difference in blood and cerebral cortex levels 30 minutes postchallenge following administration of 1× and 2× MLD (Figure 5). These data indicate that aldicarb and methomyl crossed the BBB within 30 minutes of challenge, and aldicarb crossed to a greater extent than methomyl.
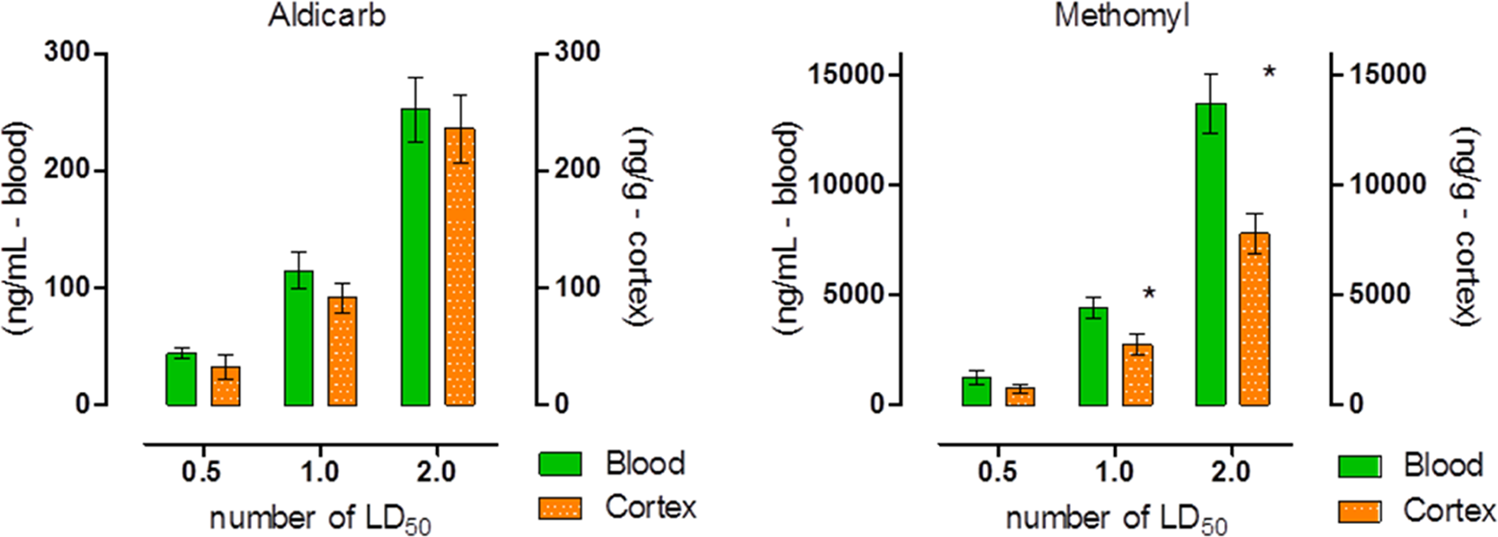
Blood and cerebral cortex concentration of aldicarb (A) or methomyl (B) 30 minutes postexposure. Cerebral cortex was perfused prior to removal to prevent potential cross-contamination between the blood and cortex (Control nonperfused cortexes were collected to determine the impact of this potential cross-contamination by the blood in the tissue, which found that no significate impact was present). Animals were challenged with multiples of the previously determined median lethal dose (MLD): 0.5, 1, or 2. Data shown are mean ± standard error of the mean (SEM), representing n = 5. *P < 0.05, significance reflects difference between levels found in the blood and brain at a particular challenge level. Blood concentration is recorded in nanograms of carbamate per milliliter of blood, and cerebral cortex concentration is recorded in nanograms of carbamate per gram of tissue.
Determination of ChE activity in the blood and the cerebral cortex collected from animals that were killed 30 minutes after challenge (Figure 6) confirmed the results from the PR study (Figure 4), in which AChE activity in the cerebral cortex significantly increased by as much as 50% above baseline, possibly due to stress-mediated production of read-through AChE 20 ; BChE activity was significantly reduced in the brain. In addition, ChE results in the blood indicated a slight, but statistically significant, preference for BChE over AChE for both aldicarb and methomyl at all challenge levels at 30 minutes postexposure. On the other hand, both carbamates appear to have a preference for BChE over AChE in the cerebral cortex at 30 minutes postexposure.
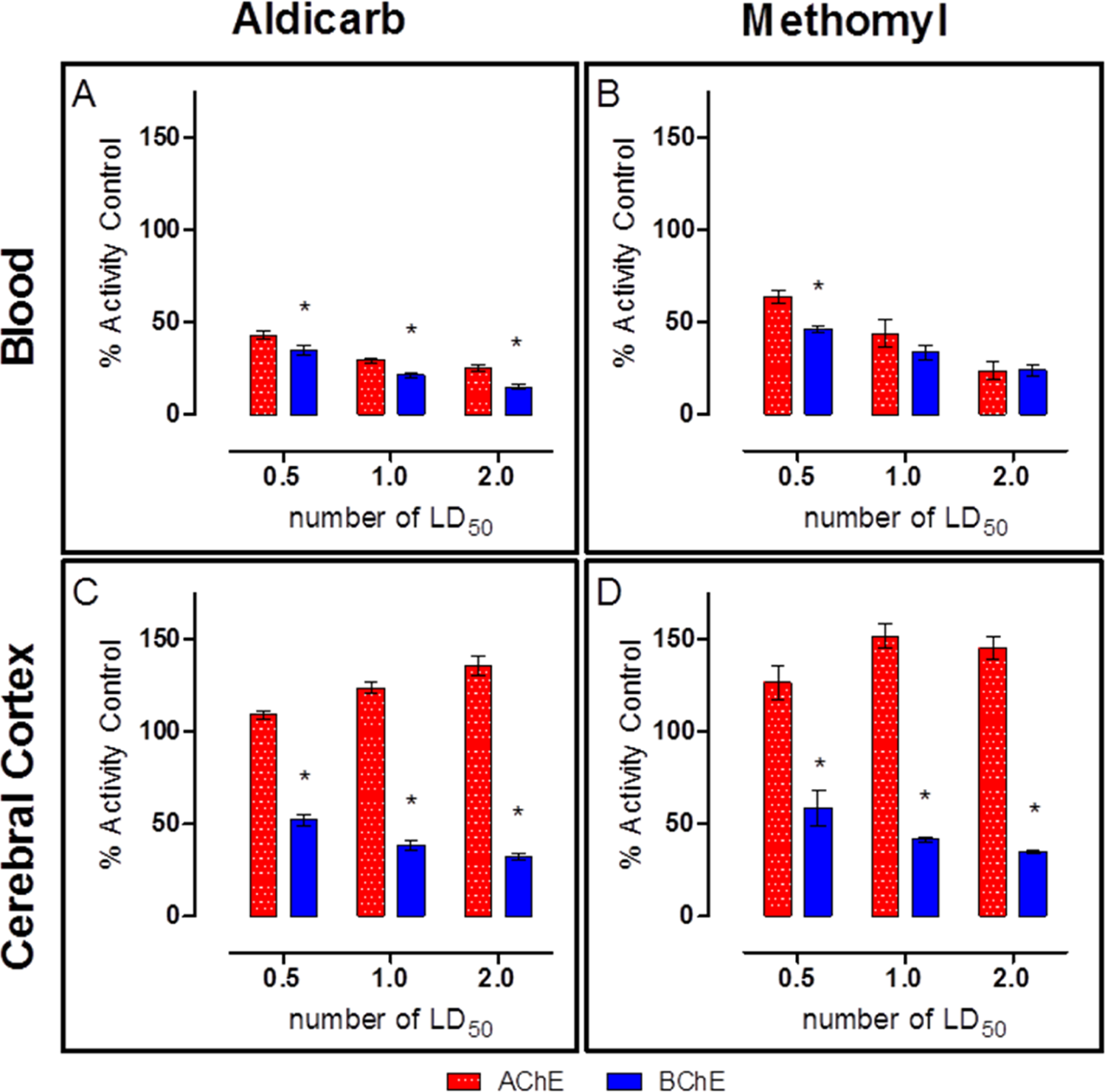
Blood and cerebral cortex cholinesterase (ChE) activity 30 minutes postexposure to aldicarb (A and C) or methomyl (B and D). Blood acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activities were similar, but a statistically significant difference was observed between AChE and BChE activity. Additionally, the cerebral cortex also had a statistically significant difference between AChE and BChE activities, but the difference was greater than in the blood. Cerebral cortex was perfused prior to removal. Animals were challenged with multiples of the previously determined median lethal dose (MLD): 0.5, 1, or 2. Data shown are mean ± standard error of the mean (SEM), representing n = 5. *P < 0.05.
Carbamate Preference for AChE or BChE in the Blood or Cerebral Cortex
Naive blood and cerebral cortex samples were collected and dosed with multiple concentrations of either aldicarb or methomyl and analyzed for ChE activity to confirm whether a preference for AChE or BChE existed. For both aldicarb and methomyl, no preference was detected in the inhibition of AChE or BChE in the blood (Figure 7A and C). However, in the cerebral cortex homogenate, a significant preference for BChE was observed for both compounds at the 3 highest concentrations tested (Figure 7B and D). The concentrations of challenge agent required to produce this BChE preferential response in the cerebral cortex tissue equated to approximately 1 MLD calculated from the PR study (Figure 2). This comparison takes into account the observed carbamate concentration in the cerebral cortex from the BBB assay (Figure 5) following a 30-minute exposure of 1 MLD. These data verified that a preference does exist for BChE over AChE in the cortex, and it also clarifies that this preference is not due to an inability of the carbamates to inhibit synaptic AChE as both cerebral cortex AChE and BChE were inhibited by both carbamates.
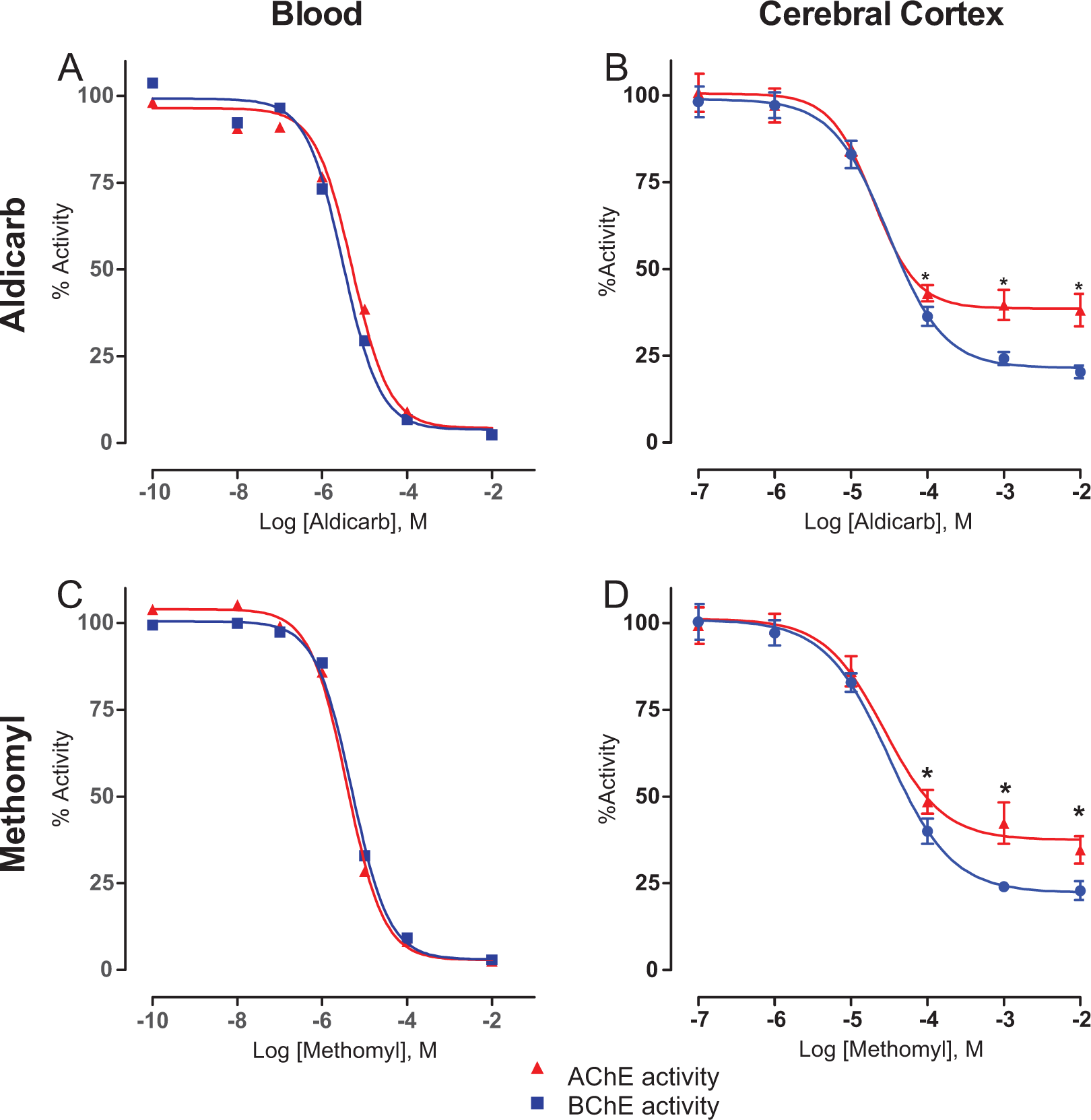
Ex vivo challenge of blood and cerebral cortex homogenate revealed that aldicarb (A and B) and methomyl (C and D) have a preference for cortex butyrylcholinesterase (BChE). Naive blood and homogenized cerebral cortex samples were challenged with varying concentrations of the challenge material for 30 minutes. Cholinesterase (ChE) activity was determined using an Ellman-based assay. Data shown are mean ± standard error of the mean (SEM), representing n = 6. *P < 0.05.
Discussion
The ultimate goal of this work was to determine whether the current medical countermeasures approved for use in the United States to protect against OP intoxication, namely atropine (free base) and 2-PAM Cl 6 via the DuoDote, are efficacious against the carbamates aldicarb and methomyl in a guinea pig model. Previous studies have shown that 2-PAM Cl was contraindicated with the carbamate pesticide carbaryl. 7,8 This has led to questions regarding the safe and effective use of oxime AChE reactivators for carbamate poisonings in general. 7 It has been theorized that the contraindication is a unique response of carbaryl to the oxime due to structural variations inherent to N-methylcarbamates. 7 However, physostigmine, which has a similar N-methylcarbamate structure, does not share the adverse effects observed in the presence of 2-PAM Cl. 21 Similarly, a recent report also contradicted the notion that oximes are contraindicated against carbamate poisonings, including carbaryl. 22 In light of these conflicting reports regarding the effectiveness of oximes against carbamate ChE intoxication, further investigation is necessary to evaluate the safety and efficacy of atropine and 2-PAM Cl for carbamate (ie, aldicarb and methomyl) poisoning.
When administered at the maximum recommended prehospital human equivalent dose for OP poisoning, concurrent administration of atropine and 2-PAM Cl provided 2- to 3-fold greater protection than vehicle (saline) treatment in guinea pigs challenged with aldicarb or methomyl. The results also indicated that protection was primarily due to the action of atropine, as the addition of 2-PAM Cl did not improve the protection provided by atropine alone. Interestingly, this result did not align with results previously reported in male rats. 23 In that study, which utilized substantially higher levels of the antidotal compounds, administration of 17.4 mg/kg atropine sulfate offered 5-fold protection following an aldicarb challenge and the addition of 50 mg/kg 2-PAM methane sulfonate increased overall protection to 6-fold. In that same report, atropine sulfate offered a 1.6-fold protection against methomyl, whereas the addition of 2-PAM methane sulfonate also elevated overall protection to 1.9-fold. 23 The differences in the results reported by Natoff and Reiff and those of the current study may be due to the bioavailability/distribution of the different salts of 2-PAM, differences in dose levels, interspecies variability between rats and guinea pigs, or some combination thereof.
The general aim of oxime AChE reactivators is to reactivate inhibited AChE. The results of this study indicated that 2-PAM Cl did not restore the activity of gpAChE-R inhibited with aldicarb or methomyl. Additionally, the rate of observed ChE activity recovery in the blood was not altered by any of the treatments tested. This contrasts previous research investigating the effect of 2-PAM on carbamylation and decarbamylation rate constants in membrane-bound bovine erythrocyte AChE. 24 In that report, the authors showed slight, but statistically significant, reductions in both carbamylation and decarbamylation rate constants attributable to the presence of the oxime. 24 It is possible that the observed reduction in both carbamylation and decarbamylation rates by 2-PAM Cl determined by Dawson may offset each other, thus explaining the lack of an effect in both the gpAChE-R and the blood samples observed in the current study. It is also plausible that interspecies variability could at least in part account for the observed difference, since differences in the kinetics of AChE derived from different species are well documented. 14,25,26 The different results are not likely to be due to differences in erythrocyte AChE versus read-through AChE, since the core of each variant of AChE is identical 27 and previous studies have reported similar kinetics between erythrocyte AChE and synaptic AChE. 25,28
The observed recovery of the ChE activities in the serial blood samples was expected based on the combination of (a) short half-lives of carbamates (30-40 minutes for aldicarb 29 and 2-3 hours for methomyl 30 ) and (b) minimal effect of oximes. 22 However, the preferential inhibition of BChE over AChE by both carbamates in the cerebral cortex was not expected based on blood ChE inhibition where no such preference was observed. In fact, AChE and BChE maintain ∼50% amino acid homology within species and share a conserved active site catalytic triad. 31,32 Although there is only one presumed isoform for BChE (although multiple oligomeric forms exist), various AChE isoforms (erythrocytic [-E] and synaptic [-S]) variants exist—all retaining a conserved active site gorge. This conservation between AChE isoforms, as well as the identification of only a single isoform of BChE, should, in theory, maintain continuity in terms of enzyme–substrate affinity for carbamate regardless of the source tissue, that is, blood or cerebral cortex. 27 That said, it is still worthwhile to note that even though the enzymes share a great deal of homology, differences in the active site gorge do exist. For example, BChE only has 6 aromatic amino acids within its gorge compared to 14 in AChE. 33 Fewer aromatic amino acids lining the active site gorge results in a more open gorge configuration, which allows for BChE to accommodate a much larger array of substrates than AChE.
Another parameter that could influence the different BChE and AChE inhibition levels observed in the central nervous system (CNS) is the BBB penetrability of the carbamates. Previous reports have shown that both aldicarb and methomyl do readily cross the BBB and enter the CNS. 34,35 In this study, both aldicarb and methomyl readily crossed guinea pig BBB as they were detectable in the cortex at 30 minutes after a subcutaneous challenge at levels that were at least half of those detected in the blood. In these experiments, the cortex tissue was perfused prior to collection to prevent any cross-contamination of these results with the carbamate found in the blood. Additionally, the finding that CNS BChE was inhibited to a greater extent than AChE was also observed with the perfused cerebral cortex samples, further demonstrating that this observation is not an artifact of ChEs in the blood.
Both aldicarb and methomyl were observed to preferentially inhibit brain BChE over AChE in ex vivo samples (Figure 7). It is important to note that the concentration of carbamate needed to produce this preference is similar to the calculated MLD found in the PR study. Furthermore, this preference for BChE had previously been reported with aldicarb in starlings. 36
Preferential inhibition of BChE by the carbamates in the brain suggests the enzyme can sequester or scavenge the challenge agent, thus protecting AChE. Furthermore, clinical observations from carbamate-poisoned individuals found minimal CNS depression, 34,37 whereas peripheral effects, such as lacrimation, hyperpnoea, and tremors, predominated. 38
Conclusion
In conclusion, the data presented here suggest that the current US prehospital therapeutic regimen for OP chemical warfare nerve agent and pesticide exposures, namely atropine and 2-PAM Cl in the form of DuoDote kits, is also effective against 2 important carbamate insecticides, aldicarb and methomyl, which have been commonly implicated in human poisonings. However, protection against these 2 carbamate compounds is primarily due to the effects of atropine, and 2-PAM Cl provides neither beneficial nor harmful contributions. Further research is needed to elucidate the interactions of 2-PAM Cl and other oximes with other carbamate pesticides.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Jill Harvilchuck and Thomas Snider for their scientific input and technical review of this manuscript. The authors, also, wish to thank all technical staff here at Battelle who contributed to this research, especially Julie Lucas, Laura Hines, and Tyson Winters as the lead technicians, Dr Patrick DeArmond as the lead chemist, and Benjamin Carper and Nancy Niemuth for statistical support.
Author Contributions
M. Brittain contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. K. McGarry contributed to acquisition and analysis and critically revised the manuscript. R. Moyer contributed to conception, contributed to interpretation, and critically revised the manuscript. M. Babin contributed to conception and critically revised the manuscript. D. Jett critically revised the manuscript. G. Plattoff critically revised the manuscript. D. Yeung contributed to conception and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work, ensuring integrity and accuracy.
Authors’ Note
The views expressed in this article are those of the authors and do not reflect the official policy of the NIH, Department of Health and Human Services, or the US Government. No official support or endorsement of this article by the NIAID, NINDS, NIH, or DoD is intended or should be inferred. The experimental protocol was approved by the Institutional Animal Care and Use Committee at Battelle. All procedures were conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals and the Animal Welfare Act of 1966 (PL 89-544), as amended.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH Office of the Director through an interagency agreement between the NIAID and Department of Defense (DoD) and prepared under the auspices of both the NIH and the DoD Defense Technical Information Center (DTIC) under the Chemical, Biological, Radiological & Nuclear Defense Information Analysis Center (CBRNIAC) program, Contract No. SP0700-00-D-3180, Delivery Order Number 0687, CBRNIAC Task 832/CB-IO-OOI2.
