Abstract
Pesticides, including organophosphate (OP), organochlorine (OC), and carbamate (CB) compounds, are widely used in agricultural and indoor purposes. OP and CB act as acetyl cholinesterase (AChE) inhibitors that affect lots of organs such as peripheral and central nervous systems, muscles, liver, pancreas, and brain, whereas OC are neurotoxic involved in alteration of ion channels. There are several reports about metabolic disorders, hyperglycemia, and also oxidative stress in acute and chronic exposures to pesticides that are linked with diabetes and other metabolic disorders. In this respect, there are several in vitro and in vivo but few clinical studies about mechanism underlying these effects. Bibliographic databases were searched for the years 1963–2010 and resulted in 1652 articles. After elimination of duplicates or irrelevant papers, 204 papers were included and reviewed. Results indicated that OP and CB impair the enzymatic pathways involved in metabolism of carbohydrates, fats and protein within cytoplasm, mitochondria, and proxisomes. It is believed that OP and CB show this effect through inhibition of AChE or affecting target organs directly. OC mostly affect lipid metabolism in the adipose tissues and change glucose pathway in other cells. As a shared mechanism, all OP, CB and OC induce cellular oxidative stress via affecting mitochondrial function and therefore disrupt neuronal and hormonal status of the body. Establishing proper epidemiological studies to explore exact relationships between exposure levels to these pesticides and rate of resulted metabolic disorders in human will be helpful.
Introduction
Among a large number of different types of pesticides available, there are groups of them that are considered hazardous to human. The major pesticides that are widely used include organophosphates (OP), organochlorines (OC) and carbamate (CB). The main mechanism of action of OP and CB is inhibition of the enzyme acetylcholinesterase (AChE) that results in signs and symptoms of excessive cholinergic stimulation. 1 –3 Unlike OP poisoning, CB poisoning tends to be of shorter duration. Because the inhibition of tissue AChE is reversible and also CB are more rapidly metabolized. OC are neurotoxins, highly lipophilic, chemically stable, and persistent in the environment with enduring half-lives. The lethal mechanism of action of OC is persistent opening of sodium channels in neurons resulting in repetitive firing of action potentials. Reduction of K+ permeability, and inhibition of calmodulin, Na-, K-, and Ca-ATPase occur by OC. 4,5 In human, pesticide poisoning is common and most often fatal. Among pesticides, OP are responsible for more than 50% of total poisoning cases. 6,7 In addition, OP have been used as harmful nerve gases. 2,8,9 Oxidative stress is another mechanism for toxicity of pesticides resulting in cell death (necrosis and apoptosis) and changes in metabolic and vital functions of the cells. 10 –12 Among different signs of toxicity caused by these substances, hyperglycemia is most commonly seen. 13 –16 Chronic exposure to small amounts of OP and OC leads to deleterious effects on carbohydrate metabolism. 17 Several organs such as pancreas, liver, muscles, and brain that are involved in metabolism of carbohydrate, fat, and protein can be affected by pesticides through alterations in glycolysis, gluconeogenesis, glycogensis, and glyconeogenesis. 18 On the other hand, effects of these pesticides on pathways of metabolism, neuronal stimulation, and hormonal secretion (adrenal, gonad, and etc.) can change metabolism of entire body. The aim of this review was to evaluate all pathways that are involved in metabolic effects of OP, CB, and OC pesticides.
Methods
Bibliographic databases including PubMed, Google Scholar, and Scopus were searched between years 1963 and 2010 for the keywords ‘pesticide toxicity and carbohydrate metabolism’ and ‘organophosphate and carbamate and organochlorine and hyperglycemia.’ In the first step, 1652 articles were found. Following elimination of duplicates or irrelevant papers, 195 papers were selected and reviewed. The reference lists of found articles were hand-searched to ensure inclusion of all possible studies as schematically drawn in Figure 1 .
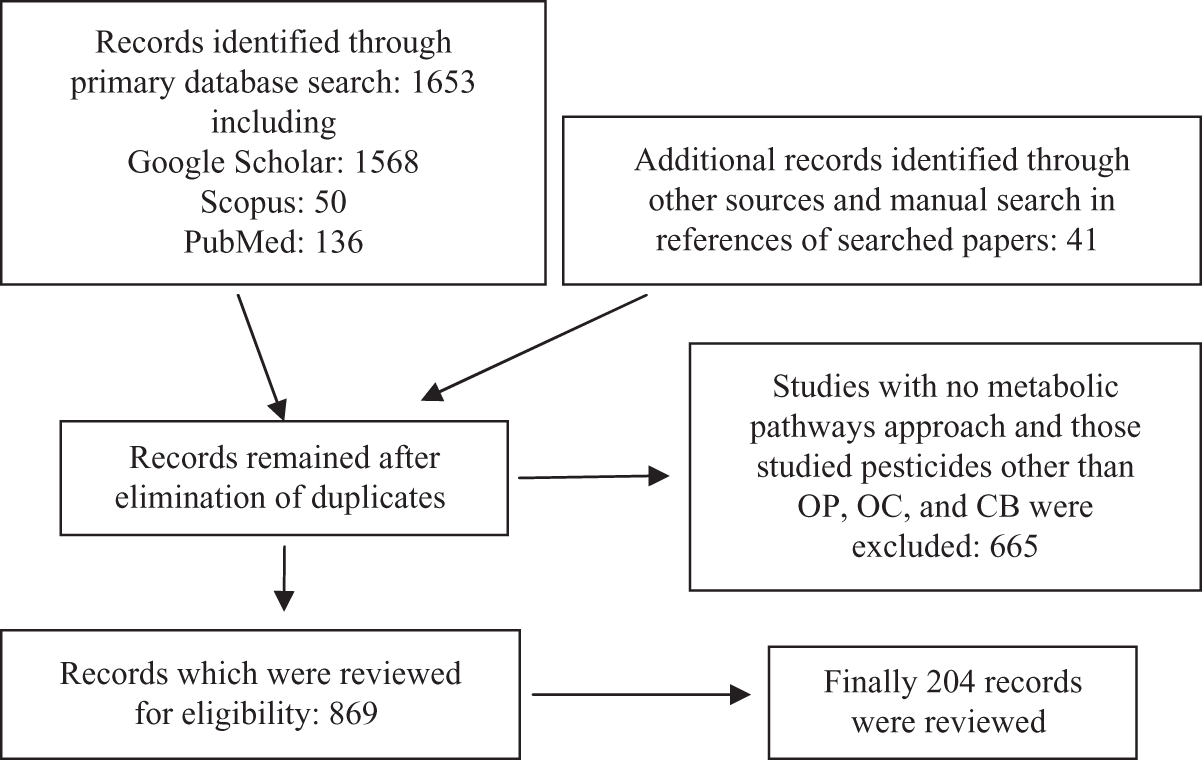
Flow diagram of the literature review process.
Results
Findings are presented in three parts including outcomes on blood glucose levels in laboratory and clinical studies, interactive effects of organs involved in hyperglycemia, and the compounds found to reduce or reverse the effects of pesticides.
Blood glucose alteration in clinical and animal studies
Clinical studies
Clinical studies indicated incidence of hyperglycemia following exposure to OP and other pesticides.
19,20
Polychlorinated dibenzo-
Animal studies
Reversible hyperglycemia occurred 2 hours post administration of acephate and then reversed to normal state as a result of insulin release from pancreas. 36 Repeated exposure to sublethal doses of dimethoate, fenitrothion, and carbofuran induced a significant increase in blood glucose levels. 14,37 –39 Moreover, diazinon, sublethal concentrations of carbaryl, and phorate increased serum glucose content. 40 –42 Abnormal hyperglycemia and glycogenesis occurred in subchronic and acute exposures to malathion. 43 –45 There were also some results in contrary to these findings; for instance, soman (GD) and VX did not affect blood glucose levels. 46 Administration of azynphos methyl, malathion, and diazinon prevented the postprandial hyperglycemia. 47,48 Another report elucidated the induction of hypoglycemia following exposure to malathion. 49 Incidence of hyperglycemia according to target organs is shown in Table 1 .
Summary of animal and clinical studies pesticides and hyperglycemia


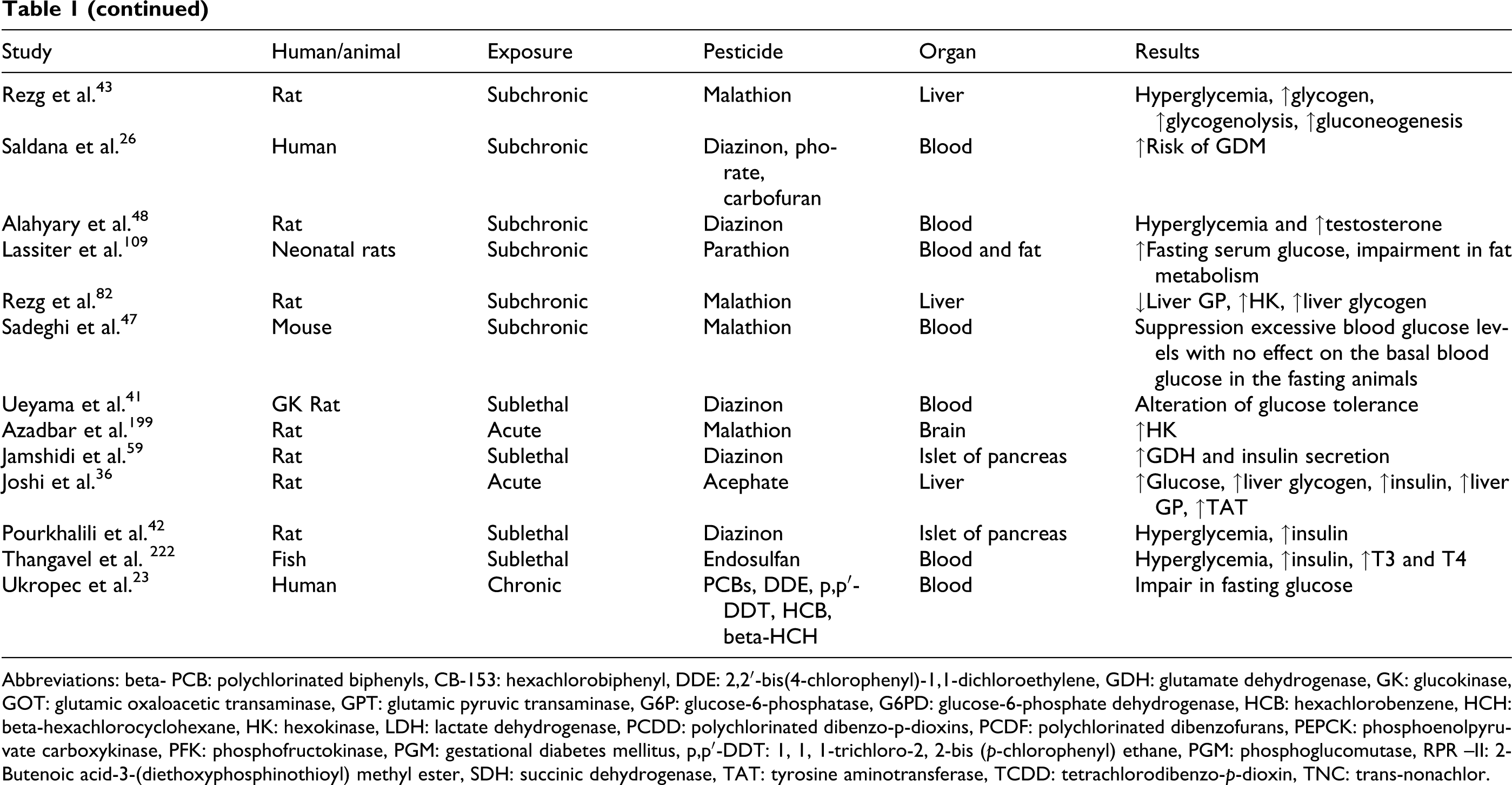
Abbreviations: beta- PCB: polychlorinated biphenyls, CB-153: hexachlorobiphenyl, DDE: 2,2′-bis(4-chlorophenyl)-1,1-dichloroethylene, GDH: glutamate dehydrogenase, GK: glucokinase, GOT: glutamic oxaloacetic transaminase, GPT: glutamic pyruvic transaminase, G6P: glucose-6-phosphatase, G6PD: glucose-6-phosphate dehydrogenase, HCB: hexachlorobenzene, HCH: beta-hexachlorocyclohexane, HK: hexokinase, LDH: lactate dehydrogenase, PCDD: polychlorinated dibenzo-p-dioxins, PCDF: polychlorinated dibenzofurans, PEPCK: phosphoenolpyruvate carboxykinase, PFK: phosphofructokinase, PGM: gestational diabetes mellitus, p,p′-DDT: 1, 1, 1-trichloro-2, 2-bis (
Pancreas
Pancreas, that has a critical role in secretion of insulin, glucagon, and digestive enzymes, is a target organ for toxicity of pesticides. 50 –53 The effect of pesticide on this organ can alter metabolism of carbohydrate, lipid, and protein.
Impairments of glucose metabolism in pancreas
Pancreatic muscarinic acetylcholine ACh receptors play an important role in stimulating insulin and glucagon secretion from islet of Langerhans.
54
By activating protein kinase C (PKC) as a consequence of phospholipid hydrolysis, ACh can contribute to secretion of insulin through increasing free cytosolic calcium concentration and subsequently exocytosis of insulin granules.
55
In acute and subacute exposures to malathion, serum levels of insulin, glucagon, and Langerhans islet glucokinase (GK) elevated; however, glucose-stimulated insulin secretion diminished.
56
–58
Nonetheless, rises in insulin level and GK activity did not overcome the elevated glucose levels. Diazinon reduced glucose-stimulated insulin secretion and caused hyperglycemia not only through disturbing the balance between free radicals and antioxidants in the islets of Langerhans but also by induction of toxic stress.
42,59
Tetrachlorodibenzo-

A simplified model outlining potential cellular mechanisms underlying the effects of organophosphate (OP), carbamate (CB), and organochlorine (OC) on pancreatic β-cell. Positive effects are shown by green arrows and negative effects by red.
Pancreatic injury
There are several evidences indicating the relationship between diabetes mellitus and pancreatic disease. 66,67 The risk of pancreatic disease can increase among subjects following exposure to pesticides. Mortality from pancreatic cancer was more frequent after exposure to DDT and OP. 68 –73 Acute pancreatitis was demonstrated as elevated serum amylase and glucose levels in exposed subjects. Dimethoate enhanced lipid peroxidation (LPO) and reactive oxygen species (ROS) and reactive nitrogen species (RNS) in the pancreas indicating significant occurrence of oxidative damage, depletion of glutathione stores, and alteration of enzymatic antioxidant defense. 37 In addition, it caused degenerative changes in many areas of the pancreas, affecting both the pancreatic acini and islets of the Langerhans. Ultrastructurally, some beta cells revealed dense nuclei with wide perinuclear cisternae and reduced number of beta granules. 74 In another study, exposure to dimethoate induced noticeable pancreatitis as identified by increased blood amylase and lipase. 37,50 –52 An association between exposure to pendimethalin and s-ethyl dipropylthiocarbamate with pancreatic cancer has been also reported. 19,75
Liver
This organ plays a major role in metabolism and has a number of functions in the body, including glycogen storage, decomposition of red blood cells, plasma protein synthesis, hormone production, and detoxification of xenobiotics such as pesticides.
Impairments of glucose metabolism in liver
There are many studies investigating the effects of OP on liver metabolism. Fenthion induced hepatic glycogenolysis 76 . Exposure to fenitrothion elevated glycogen phosphorylase (GP) activity leading to a reduction in hepatic glycogen content. 14,77 The same effect was reported following a 48-hour exposure to carbofuran. 78 Subchronic exposure to malathion, enhanced hepatic gluconeogenesis and glycogenolysis, via stimulation of GP and phosphoenolpyruvate carboxykinase (PEPCK); whereas diazinon increased hepatic PEPCK. 79 –81 Dichlorvos reduced the activity of hepatic GK and liver glycogen content while hepatic GK mRNA levels were increased. 65 In other studies, malathion lowered hepatic protein and lipid contents associated with stimulated gluconeogenesis, and increased hexokinase (HK) activity. In contrary to the above studies, malathion decreased hepatic GP activity with a rise in the hepatic glycogen rate (hepatomegaly). 43,82,83 Acute exposure to acephate increased liver glycogen content and activities of gluconeogenesis enzymes, glucose-6-phosphatase (G6P), and tyrosine aminotransferase in the liver. 36 Lactate dehydrogenase (LDH), a key enzyme in glycolysis, was elevated but succinic dehydrogenase (SDH) and ATPase activities that contribute to production of mitochondrial ATP were depleted in the liver post exposure to quinalphos. 84 Moreover, an increase in LDH or a reduction in SDH activity in the livers and brains of exposed subjects to sublethal concentrations of sevin were reported. 85 During exposure to fenitrothion, liver and blood lactate levels increased; however, the protein content decreased. 39 The increase of liver and muscle LDH reflects a possible improvement in tissue glycolytic capacity. In signaling cascade level, diazinon up-regulated adenyle cyclase activity, which is a response to stimulants acting on beta-adrenergic, glucagon, or G-protein receptors. 86
Hepatic injury
Histochemical alteration in the liver can affect metabolism of carbohydrates, lipids, and particularly proteins. Sublethal doses of carbaryl and phorate increased alkaline phosphatase (ALP) and bilirubin. 40 Chlorpyrifos and deltamethrin caused liver damage. 87 Exposure to methidathion phosphorodithioic acid S-[(5-methoxy-2-oxo-1,3,4-thiadiazol-3(2H)-yl)methyl] O,O-dimethyl ester (MD) induced histopathological changes, including recruited mononuclear cells in all portal areas, sinusoidal dilatation, and focal microvesicular steatosis and hydropic degenerations in parenchymal tissues. 88,89 In addition, methylparathion increased liver weight. 90 Leptophos, chlorpyrifos, and diazinon inhibited glutamic oxaloacetic transaminase (GOT), glutamic pyruvic transaminase (GPT), gamma-glutamyl transferase (GGT), and LDH. 91 Two-bButenoic acid-3-(diethoxyphosphinothioyl) methyl ester (RPR-II) increased plasma GPT and GOT activities, but decreased these values in the liver. Meanwhile, it elevated plasma acid phosphatase (AcP) and ALP and reduced hepatic AcP and ALP levels. 92 Finally, dimethoate increased serum urea, total bilirubin, GOT, GPT, and amylase. 38,93
Impairments of protein metabolism
OP are known to alter serum levels of amino acids. 94,95 For instance, carbofuran decreased liver and muscle total protein. Isofenphos-treated myeloid blast cells showed an increase in lactate production and a decrease in glucose oxidation and the synthesis of glutamate, palmitate, and stearate from glucose. 96 It is suggested that pesticides reduce tissue protein content because of glucose production in gluconeogenesis process and also because of inhibition of protein synthesis.
Muscle
Muscular activity accounts for much of the body's energy consumption. All muscle cells perform carbohydrate and lipid metabolism for production of ATP as energy. In addition, muscle stores glucose in the form of glycogen and creatine phosphate to generate ATP. There are two pathways for ATP synthesis; anaerobic process called glycolysis which produces two ATP and two lactic acid molecules and aerobic process resulting in higher production of ATP, without lactate formation. On the other hand, globules of fat in muscle can be used for production of energy. There are several studies showing muscular toxicity of pesticides in the pathways involved in ATP and carbohydrate metabolism. 97,98 Increased availability of carbohydrate precursors resulting from augmented muscle glycogenolysis appears to contribute to the hyperglycemia and liver glycogen deposition following guthion poisoning. 99
Impairments of glucose metabolism in muscle
Both fenthion and malathion induced muscle glycogenolysis while malathion enhanced muscle phosphofructokinase (PFK) activity. 43,76 Ronnel had no metabolic activity in skeletal muscles but decreased rates of glucose oxidation to CO2 in these muscles. 100 Lastly, the activities of LDH and GP increased with depletion of glycogen and elevation of lactate post exposure to dimethoate in the muscles. 101
Muscle injury
Glutathione and cytochrome c oxidase (CcOX) depletion and induction of xanthine oxidase (XO) post exposure to OP suggest overproduction of ROS. 102,103 Induction of Na/K-ATPase and inhibition of Ca-ATPase activity and elevation of heat-shock protein expression as a protective mechanism 104 reveal muscle injury in response to OP. On the other hand, muscle fasciculation after release of ACh made an extra energy demand that stimulates gluconeogenesis. Therefore, the protein and glycogen stores decreased. Accumulation of lipids inside the muscle occurred following exposure to isofenphos 105 probably due to downregulation of metabolic activity in fat tissues.
Lipid metabolism impairments and obesities
The association between OC and homeostatic model assessment-insulin resistance (HOMA-IR) tended to strengthen as waist circumference increased. OC and nondioxin-like PCB can elevate triacylglycerol and fasting glucose that along with obesity can increase the risk of type 2 diabetes. 21,106 Diacylglycerol metabolism is the probable target for chlorpyrifos oxon, and in chronic exposure, chlorpyrifos caused an increase in rat body weight via increasing adipose tissue. 107,108 Parathion exposure increased weight gain and evoked signs of a prediabetic state, with elevation in fasting serum glucose and impaired fat metabolism. 109 Chronic exposure to sublethal doses of dimethoate markedly increased cholesterol levels, whereas DDT lowered blood cholesterol and fatty acids. 38,63,110 Ronnel downregulated the metabolic activity in adipose tissue. 100 In addition, accumulation of fatty acids in the cytosol increased oxidation of fatty acids in peroxisomes and the endothelial reticulum, resulting in overproduction of ROS and further injury (Figure 3).
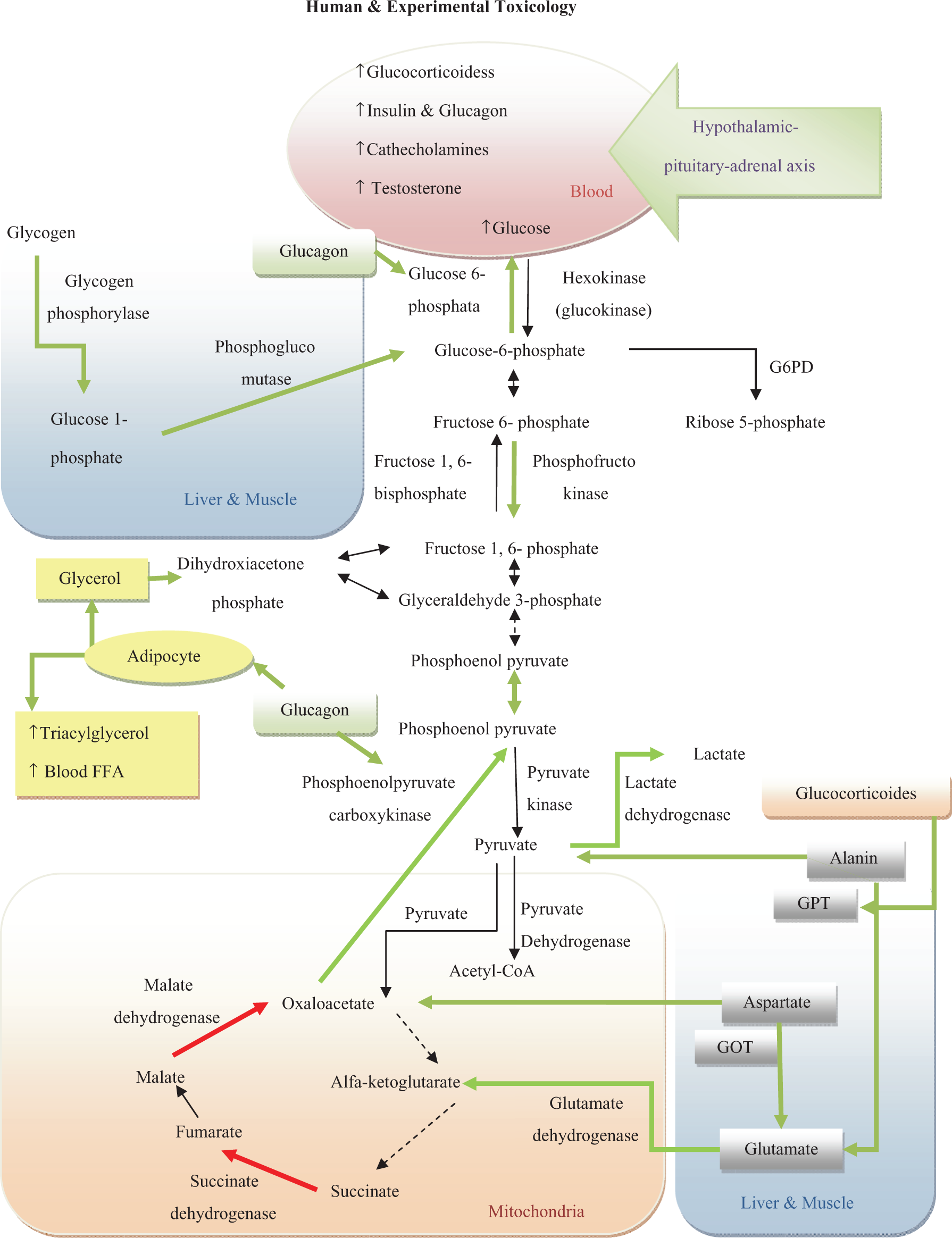
A model of metabolic impairment of carbohydrates, fats, and proteins caused by organophosphate (OP), organochlorine (OC), and carbamate (CB). Positive effects are shown by green arrows and negative effect by red.
Brain
Neuronal system regulates energy intake, energy expenditure, 111 and endogenous glucose production sense. Also, it responds to inputs from hormonal and nutrient-related signals on information of both body energy stores and current energy availability. Hypothalamic lactate metabolism lowers synthesis of hepatic glucose and plasma glucose levels. 112 Indeed, changes in brain metabolism can regulate metabolism in other organs. Also, defects in neuronal control system are shown to be associated with obesity and type 2 diabetes. 113
Glucose metabolism impairments in brain
During the first 30 min post exposure to paraoxon, use of glucose in all brain regions was downregulated without causing any consistent change in brain metabolism. However, levels of cerebrum ATP fell down which seemed to be associated with high levels of lactic acid and lower levels of creatine phosphate. 114 Although the activities of GP, phosphoglucomutase (PGM), HK increased while G6P, glucose-6-phosphate dehydrogenase (G6PD), and LDH were not significantly altered by malathion. 115 In quinalphos exposure, LDH elevated but SDH and ATPase were depleted in the brain. 84 Upregulation of gamma-enolase mRNA contributed to production of phosphoenolpyrovate from phosphoglycerate following OP exposure. 116
Other effects
Oxidative stress
During normal conditions, ROS such as superoxide anion, hydrogen peroxide, and hydroxyl radicals are generated at a low rate as a result of mitochondria and microsomes function. However, a reduced capacity of the antioxidant system of the body leads to incomplete reduction of oxygen and, thus, increased production of ROS. 117 –122 OP and OC increase biomarkers of ROS (F2-isoprostanes and F4-neuroprostanes) and RNS (citrulline), likely because of induction of cytochrome P450. In addition, they alter antioxidant enzymes, catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione reductase (GR). Any change in the G6PD, (reduced glutathione) GSH, and glutathione S-transferase (GST) can cause LPO and tissue damage. 11,12,64,80,88,93,123 –133 It should be noticed that antioxidative defense mechanisms and even LPO in various tissues varies according to the type of pesticide and the doses used. 87 Oxidative stress leads to DNA damage and affects genes encoding cytochrome P450s and UDP-glucuronosyl/UDP-glucosyltransferases that are involved in body metabolism. 90,127,134,135 Diazinon caused hyperglycemia in the rats, because of induction of oxidative/nitrosative stress. 42 Furthermore, G6PD transcription elevated due to overproduction of ROS. 133,136 However, many studies have not reported changes in G6PD level 93,115,136 but a reduction in erythrocyte has been observed. 64 G6PD in pentose phosphate shunt uses NADP+ as the electron acceptor to produce NADPH for reduction of oxidized glutathione. Alteration in this enzyme results in oxidative stress and impaired glucose metabolism.
Mitochondrial impairments
Mitochondria as a cellular source of energy has critical role in the production of ATP as energy in direct correlation with metabolism of carbohydrates. Because of its effects on production of ATP, and ROS, and promotion of cell death, this organelle associates with metabolic alterations as a consequence of exposure to pesticides. Mitochondria has long been known to play a critical role in maintaining the bioenergetics status of cells under physiological conditions. Mitochondria produces large amounts of free radicals that contribute to a range of degenerative conditions. 137 For instance, swelling of mitochondrial vacuoles in the cytoplasm, dissolution of mitochondrial matrix, and picnotic nucleus in B cells in the islets of Langerhans were observed following exposure to endosulfan. 138 Serum levels of several mitochondrial toxins, such as persistent organic pollutants, are associated with metabolic syndrome. 139 Aggregation of mitochondria was seen in cerebellar neurons post OP exposure. In addition, impairment in mitochondrial energy metabolism induced a delayed neurodegenerative condition which seems to be a kind of apoptotic neuronal degeneration. 140 –142 Depression of NADH cytochrome c reductase, succinate cytochrome c reductase, and CcOX activity in the mitochondrial respiratory chain, reduction of mitochondrial transmembrane potential and ATP concentration, elevation of ADP/ATP ratio, induction of LDH release, and necrotic cell death occurred following OP poisoning. 143 –146 Moreover, this pesticide induced cholinergic receptor-independent necrotic cell death. Reduction of SDH and elevation of LDH suggest that anaerobic metabolism was favored while aerobic oxidation of pyruvate was impaired after OP exposure. 85
Peroxisome alteration
Peroxisome proliferation receptors (PPARs) are a group of nuclear receptor transcription factor that induce proliferation of proxisome within cells and contribute to metabolism of lipids, carbohydrates, and proteins and also insulin resistance. 147,148 There is evidence suggesting that reduction of the activity of PPAR-gamma may increase insulin sensitivity and adipocyte differentiation. 149,150 OC pesticides methoxychlor can interact with PPARs, increase peroxisomal density values, and induce activity of peroxisomal acyl-CoA oxidase (AOX). 151 The initial reaction in peroxisomal β oxidation is catalyzed by AOX to form hydrogen peroxide through donation of electrons to molecular oxygen. Therefore, in addition to mitochondrial-induced ROS and RNS, accumulation of fatty acids in the cytosol increases oxidation of fatty acids in peroxisomes and the endothelial reticulum. For instance, exposure to eldrin resulted in increased number of hepatic proxisomes and consequently generation of glycogen granules in islets of Langerhans. 152
Paraoxonase
The human paraoxonase-1 (PON1) is a high-density lipoprotein-associated (HDL) phosphotriesterase that is mainly produced by the liver and secreted into the blood. This enzyme is able to hydrolyze toxic OP xenobiotics, endogenous oxidized phospholipids, and homocysteine thiolactone, and protect low-density lipoprotein (LDL) from accumulated lipid peroxides. 153 –156 PON1 mediates metabolism of chlorpyrifos and diazinon as demonstrated in the microsomes of the liver and enterocyte. 155 The specific hydrolytic activity of PON1 in liver and blood provides a natural barrier against entry of chlorpyrifos into the central and peripheral nervous systems. The inherited differences in PON1 enzyme may determine degree of susceptibility to OP injury in human. 156 –160 This enzyme acts as an antioxidant against pesticide-induced LPO, but it was shown that the level of this enzyme decreased by OP. 161 –163 A lower PON1 metabolic capacity of younger animals to acute OP toxicity has been reported. 164 On the other hand, patients with impaired glucose tolerance or non-insulin-dependent diabetes mellitus had slightly higher AChE activities and low serum PON1. 165 In diabetic patients, PON1 is elevated due to several associations between its gene polymorphisms and glucose metabolism. 166,167 Likewise, there are reports of association between common variation in PON2, another isoform of this enzyme, and some metabolic quantitative phenotypes such as plasma lipoproteins, plasma glucose, birth weight, and atherosclerosis. 168
Stress hyperglycemia
The stress linked with critical illness is characterized by alteration of hypothalamic–pituitary–adrenal (HPA) axis hormones, the autonomic nervous system, cytokines, and activation of immune-neuroendocrine axis, 169,170 all in favor of hyperglycemia. Carbohydrates are usually the first energetic supply for various metabolic processes particularly in cases of stress and tissue glycogen depletion. This stress causes release of adrenal hormones via stimulation of sympathetic nervous system during stress leading to enhanced release of catecholamines, glucagon, and growth hormone, which furthermore promote gluconeogenesis, glycogenolysis, and insulin resistance. 171 –179 On the other hand, it has been suggested that insulin release may be suppressed as a result of enhanced activation of the pancreatic alpha receptors. 180 The low insulin levels, together with insulin resistance in the presence of increased secretion of the counter-regulatory hormones, results in a kind of stress hyperglycemia. Inhibition of AChE activity leads to activation of HPA axis and sympathetic autonomic nervous system, which consequently result in hyperglycemia. 181 Malathion decreased hypothalamic corticotrophin releasing hormone in mRNA level that contributed to hypertriglyceridemia, carbohydrate metabolism, and diabetic form of insulin resistance, which are all associated with increased levels of hypothalamic inducible nitric oxide (NO) synthase mRNA. 182 Also, chlorpyrifos and methoxychlor alter gonadotropin-releasing hormone (GnRH) biosynthesis in the hypothalamic cell line in vitro, suggesting to have endocrine disrupting effects on GnRH neurons. 183 Exposure to acephate and other pesticides can increase plasma corticosterone and deplete adrenal cholesterol content. 36,184 Methyl parathion elevated plasma corticosterone concentration. 44 Malathion increased catecholamine and secretion of glucocorticoids that provokes glycogenolysis and gluconeogenesis. 82 Plasma adrenocorticotropes hormone, corticosterone, and aldosterone levels were increased by exposure to acephate and methamidophos. In fact, the effect of methamidophos on the adrenal cortex may be mediated by the pituitary gland, while the effect of acephate may be due to direct gland interaction. 185 Following carbofuran exposure, the level of norepinephrine increased in the olfactory bulbs, the telencephalic hemispheres, and the hypothalamus. An increase in the level of dopamine was also found in hypothalamus. 78 Diazinon increased the synthesis of testosterone, causing changes in carbohydrate metabolism. 48 Fenitrothion blocked dihydrotestosterone-dependent androgen receptor activity in human hepatoma cells. 186,187
Hormones
Thyroxin, the major secretary product of the thyroid and a principal regulator of metabolism, increases oxidation of acetate and synthesis of fatty acids. Thyrotoxicosis is known to induce a broad range of changes in carbohydrate metabolism. 188 Pesticides can influence serum concentrations of reproductive and metabolic hormones, particularly thyroxin. 189 Ronnel was shown to interfere with the metabolic effects of thyroxin and depressed body metabolic activity. 100 In addition, a negative relationship was found between insulin-like growth factor-1 (IGF1) and aldrin and other OC that are involved in the pathogenesis of several diseases like cancer, diabetes, and growth disorders 61,190,191 There are a number of reports indicating estrogenic effect of metoxychlor that can cause alterations of metabolism. 192,193
Prophylactic and therapeutic agents
Antioxidants
Selenium-supplemented diet and combination of vitamins E and C can reduce toxic effects of OP on liver tissue of rats.
88,144
Selenium could restore OP-induced reduction of GPx and increased LPO in white muscle with no effect on the liver. Meanwhile, selenium increased GST activity in all tissues with an increase in GSH levels.
128
Alpha-tocopherol protected the liver and blood from diazinon-induced oxidative stress.
194
The fruit of date palm (
Bioscavengers
Basically, bioscavengers hydrolyze OP catalytically and thus render them non-toxic, such as OP hydrolase and OP anhydrase. Different organs have different capacities for hydrolyzing pesticides; for example highest aliesterase activities were observed in the small intestine and liver, and low activities were noted in brain, spleen, and skeletal muscles in exposure of rats to parathion and chlorpyrifos. 196 Also, compounds that stoichiometrically bind to OP such as AChE and related enzymes are categorized here. In another study conducted in vitro, free radical scavengers like alpha-tocopherol protected rat islets of Langerhans from diazinon-induced hyposecretion of insulin better than those of calcium channel blockers and autonomic nervous system blockers. 42
Phosphodiesterase inhibitors
Pentoxifylline, a phosphodiesterase-5 inhibitor, is a good candidate for control of malathion-induced toxic stress and mitochondrial damage in rat brain 197 –199 and islets of Langerhans. 200 In addition, the same benefit has been reported for rolipram as a phosphodiesterase-4 inhibitor in malathion-induced toxic stress in rat blood and brain mitochondria. 201 Of course, neither pentoxyfilline nor rolipram was able to recover OP-induced hyperglycemia. New natural sources of phosphodiesterase inhibitors have been introduced in the recent years that need to be tested in the same types of studies. 202
Hormones
Melatonin administration led to a significant increase in antioxidant activity and inhibited LPO in most tissues. 130
Cytoprotectives
Interleukin-10 could prevent or diminish pathological signs of tissue damages in organs such as liver, kidney, and lung caused by OP poisoning. 203 Moreover, zinc prevented hepatotoxic effect of chlorpyrifos in disruption of the membranous organelles and narrowing/blocking of biliary channels. 204
P450 inhibitors
Inhibition of brain, liver, and plasma AChE activity by tribufos was antagonized by piperonyl butoxide. 205
Conclusion
There are a lot of evidences on the effect of pesticides on lipid, protein, and carbohydrate metabolisms. OP and CB increase ACh that contributes to secretion of insulin and glucagon by activation of PKC and increasing the efficiency of free cytosolic calcium concentration on exocytosis of insulin granules. In addition, OP decreased secretion of insulin, specially in response to glucose. Muscle fasciculation occurred by high amount of ACh, can elevate energy demand, and consequently the gluconeogenesis leading to cardiovascular consequences. 206 Therefore, muscle-stored energy (glycogen, lipid, and protein) starts decreasing. In the initial stages of poisoning, carbohydrates are used to supply energy to meet the stress situation, while in the later phases, lipids and proteins serve as the major source of energy. Stress situation occurs as a result of alteration in HPA axis hormones, the autonomic nervous system, cytokines, and activation of immune-neuroendocrine axis by pesticide, that all are in favor of hyperglycemia. Therefore, the levels of some of the serum amino acids decrease due to this energy demands. By decreasing mitochondrial ATP production, both OP and CB provide background for hyperglycemia and subsequently breaking glycogen, lipid, and finally protein, in order to supply energy. Also, they are able to alter enzymatic pathways involved in production of glucose from proteins, lipids, and glycogen in cells like liver and in muscle cells. This hyperglycemia cannot be eliminated because of low insulin secretion. Furthermore, the level of PON1 is reduced by OP that results in lower hydrolyzing capacity of OP.
In addition, oxidative stress caused by these pesticides, injures organs like pancreas, brain, and liver leading to impairments in metabolism of lipids, carbohydrates, and proteins. Furthermore, mitochondrial impairment disrupts activity of respiratory chain and Krebs cycle enzymes and depletes ATP and thus promotes anaerobic metabolism rather than aerobic oxidation of pyruvate.
Among these pesticides, OC are the most lipophilic and thus accumulate in body adipose tissues and brain, and elevate levels of triacylglycerol and fasting glucose. In addition, OC depress metabolic activity in adipose tissue that is linked to obesity to increase the risk of type 2 diabetes. In addition, accumulation of fatty acids in the cytosol increases oxidation of fatty acids within peroxisomes and endothelial reticulum, resulting in overproduction of ROS and further injury. This effect is due to interaction of this pesticide by PPARs, a receptor that is involved in metabolism of lipids, carbohydrates, and proteins and also insulin resistance. Furthermore, OC alter concentrations of sex hormones, IGF1, and thyroxin. OC also reduces secretion of insulin in response to glucose and reduce muscular glucose oxidation.
Taking collectively, differences in the methods of various studies such as exposure pattern, type of animals used, and doses limit power of final conclusion. Gathering more trustful information from clinic, forensic, occupational, and hospital sources would be very much useful to reach a convincing conclusion in future. Also, conducting proper epidemiological studies regarding relationships between exposure level and rate of metabolic disorders in human will clarify the issue. At this stage, we believe this review will be helpful for planning more coherent strategies and studies for better understanding the mechanism of toxicity of these chemicals and maneuvers to reduce their toxic effects in the environment and human being. For better understanding, readers are requested to focus on the Figures 2 and 3, and study the summarized information of all papers that appeared in Table 1 even those that have not been cited inside text. 207 –222
Footnotes
Acknowledgment
This paper is the outcome of an in-house study. Authors would like to thank Dr Azadeh Hosseini-Tabatabaei from Department of Surgery, University of British Columbia, Vancouver, Canada for her kind assistance in language editorial of the paper.
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
