Abstract
Tea from the leaves of guayusa (Ilex guayusa) has a long history of consumption by Ecuadorian natives in regions where the plant is indigenous. The tea contains the methylxanthines caffeine and theobromine as well as chlorogenic acids, flavonoids, and sugars. Various studies were performed to evaluate the general and genetic toxicology of a standardized liquid concentrate of guayusa (GC). Guayusa concentrate was found to be negative in in vitro genotoxicity tests including the Ames test and a chromosome aberration study in human lymphocytes. The oral median lethal dose (LD50) of GC was >5,000 mg/kg for female rats. Guayusa concentrate was administered to male and female rats in a 90-day subchronic study at 1,200, 2,500, and 5,000 mg/kg/d of GC and a caffeine-positive control at 150 mg/kg/d corresponding to the amount of caffeine in the high-dose GC group. Effects observed in the GC-treated groups were comparable to those in the caffeine control group and included reductions in body weights, food efficiency, triglycerides values, and fat pad weights and increases in blood chemistry values for serum aspartate aminotransferase, serum alanine aminotransferase, and cholesterol and adaptive salivary gland hypertrophy. No signs of incremental toxicity due to any other components of guayusa were observed. The studies indicate no harmful effects of GC in these test systems.
Introduction
Guayusa (Aquifoliaceae ilex guayusa) is a holly tree found at elevations up to 2,000 m in the countries of Ecuador, Colombia, and Peru. Guayusa is one of several common teas used in the Amazon region, which include Ilex paraguariensis (yerba maté), Paullinia cupana (guarana), and Ilex guayusa (guayusa). 1 These teas have been used by indigenous habitants of the Amazonia rainforest for hundreds of years and perhaps long before that. Although the beneficial effects of I guayusa have been known for centuries in many South American cultures, 1 its potential adverse effects have not been systematically studied or reported.
During the recent past, studies have shown that tea can lower the risk of cardiovascular disease and cancers, 2,3 reduce body weight and plasma levels of free triglycerides (TG), cholesterol, and low-density lipoprotein cholesterol, 4 as well as showing extraordinary antioxidant and antiaging effects 5,6 and anticancer properties. 7 Yerba maté and guarana have been shown to be potentially useful for weight loss and weight maintenance in overweight patients. 8 Yerba maté has been shown to be hypocholesterolemic, hepatoprotective, an effective weight management agent, a central nervous system stimulant, and to benefit the cardiovascular system. 9 There is evidence that it is a DNA protectant and is associated with both the prevention and the cause of some types of cancers. 10 Gambero and Ribeiro 11 recently examined yerba maté and described its beneficial effects in combating obesity by improving the lipid parameters and by modulating the expression of genes that are changed in the obese state and restoring them to more normal levels of expression. Guarana is a permissible food additive classified as a “natural flavoring substances and natural substances used in conjunction with flavors” by the Food and Drug Administration (FDA; 21 CFR 172.510).
For hundreds of years, guayusa leaves have been chewed and consumed outright and/or have been dried and brewed like a tea for their stimulative effects by the Kichwa tribes of Napo Ecuador that is located about 50 miles from Quito. 1 As a result of its longtime use, guayusa has become a central element of local cultural identity and lifestyle. Its use among Ecuadorians was first described by Richard Spruce in the 1800s. 12 Although it has been used for centuries, it was only first recorded in the scientific literature in 1901. 13 Ilex guayusa has also been used as a medicine, an emetic, and as a stimulant. 14 Despite their differences in scientific classifications, yerba maté, guarana, and guayusa have many of the same components as common tea (Camellia sinensis) including polyphenols, such as catechin, epigallocatechin (EGC), epigallocatechin gallate, and theanine, as well as methylxanthines, such as caffeine, theobromine, and theophylline, albeit in varying concentrations. Alikaridis 15 studied the natural constituents of approximately 50 species of Aquifoliaceae worldwide and found the presence of purine bases such as caffeine, theobromine, theophylline, and adenine as well as vitamins (C, B1, and B2), nicotinic acid, and carotene. Radice and Vidari 16 also found the presence of phenols, tannins, reductive sugars, steroids, terpenes, flavonoids, and quinones in I guayusa. Ilex guayusa is now available as a non-Camellia tea in several commercial outlets in the United States, China, and Europe. Yerba maté and other Ilex species have been gaining global popularity as described by Hao et al. 17
One of the prominent common threads among these beverages is methylxanthines. The approximate methylxanthine content of guayusa, yerba maté, guarana, and Camellia sinensis is 3.2%, 1.3% to 2.9%, 3.5% to 10.5%, and 3.7% to 4.7%, respectively. Methylxanthines, well-known phosphodiesterase inhibitors, 18,19 are capable of stimulating the central nervous system, producing diuresis, and relaxing smooth muscles. They have been shown to have a vasoconstricting effect on the brain and a vasodilating effect on peripheral blood flow, leading to a stimulating effect on the central nervous system primarily via antagonism at the level of the adenosine receptors. 20 Phosphodiesterase inhibitors, such as caffeine, elevate the cyclic adenosine monophosphate (cAMP) levels affecting the regulation of cAMP-dependent protein kinases, which are responsible for the regulation of glycogen, sugars, and lipid metabolism. Caffeine is the most prevalent methylxanthine in the guayusa.
Caffeine is commonly consumed by almost 90% of the adults in the United States in forms of coffee, tea, or other caffeinated food products 21,22 due to its ability to enhance alertness, mood, and cognition and to produce stimulatory effects. 23,24 The toxicology of caffeine and its effects on human health have been well studied and controversial over decades. 25
Added caffeine is considered to be generally recognized as safe (GRAS) by the FDA for limited uses (21 CFR 182.1180). Caffeine is also listed in the Food Additives Status List 26 and the Everything Added to Food in the United States list. 27 The Flavor and Extract Manufacturers Association (FEMA) evaluated caffeine safety and determined that caffeine is GRAS as a flavor ingredient in cola-type beverages at levels not to exceed 200 ppm or 0.02%, which approximates 71 mg of caffeine/12 ounces of cola. 28
Early concerns about caffeine consumption have been resolved by continuing study. The 1978 review by the Select Committee of GRAS Substances (SCOGS) for the use of caffeine in foods and cola beverages at levels of 20 mg in 100 mL beverage (0.33 mg/kg/d) or 71 mg in a 355 mL (12 ounces: 1.18mg/kg/d) yielded the conclusion that there were “insufficient data upon which to evaluate the safety of caffeine.” In 2003, in an updated review of the scientific literature, Health Canada concluded that moderate consumption of caffeine up to 400 mg/d (equivalent to 6.7 mg/kg/d for a 60-kg person) was not associated with any adverse effects in healthy adults. 25
Since the review by Health Canada, other government agencies have endorsed the safety of moderate consumption of caffeine. The scientific report of the 2015 Dietary Guidelines Advisory Committee (DGAC) to the United States Departments of Health and Human Services (HHS) and the Department of Agriculture (USDA) stated: “Currently, strong evidence shows that consumption of coffee within the moderate range (3-5 cups per day or up to 400 mg/day caffeine or 6.67 mg/kg/day) is not associated with increased long-term health risks among healthy individuals.”(p8)29 In addition, the European Food Safety Authority (EFSA) published an external scientific report in 2015 entitled “Extensive literature search as preparatory work for the safety assessment of caffeine” and found that in adult participants, no adverse effects are expected following exposure to <400 mg/d (6.67 mg/kg/d in a 60-kg adult). 30 In 2015, the EFSA formally adopted the position that <400 mg of caffeine/d and <200 mg of caffeine in a 2-hour period were safe for the consumer. 31
Here, we aim to examine the in vitro and in vivo general toxicity data, in addition to the available literature on guayusa to determine the safety in use of a standardized liquid concentrate of guayusa derived from guayusa leaves that have been boiled in water. The test material is referred to as guayusa concentrate (GC). Studies were conducted in compliance with Organization for Economic Cooperation and Development (OECD) guidelines and in accordance with Good Laboratory Practices (GLPs) where applicable. The studies included supporting analytical chemistry evaluations, an acute oral toxicity in rats, a 14-day dose range finding repeated dose study in rats, a 90-day repeated dose study in rats, a bacterial reverse mutation assay (Ames test), and an in vitro cytogenetics study using human peripheral blood lymphocytes (HPBL). To our knowledge, this is the first publication presenting a safety analysis of laboratory studies of I guayusa.
Materials and Methods
Test Article/Materials/Analytical Chemistry
Guayusa concentrate lot no. 77903061318 was provided by Runa, LLC, (Quito, Ecuador) and analyzed for this study. Guayusa concentrate is prepared as a concentrated tea by adding dried guayusa leaves to purified water at the ratio of 1.3 to 1.6:1 wt/wt. Leaves are brewed for 2 to 4 hours. The liquid is subsequently cooled to 55°C to 60°C. After further cooling, the filtered liquid is drummed and stored at 0°C to 4°C. The chemical composition of GC was analyzed for caloric content, fats, sugars, metals, chlorogenic acids, flavonoids, theaflavins, anthocyanides, stilbenoids, isoflavones, and phytosterols.
For the 14-day study, the dose preparations (test substance, caffeine reference control, and vehicle control) were sampled for stability on days 1, 7, and 14. Given that the dose preparations were made daily, they were maintained on a stir plate during dose administration and used within approximately 2 hours, and the test substance in the preparation was considered to be stable. The samples were frozen at approximately −20°C immediately following test substance administration. Upon receipt by analytical chemistry, all samples were stored and maintained frozen (approximately −20°C) prior to analysis and analyzed at one time point after the end of the dosing phase. Samples were considered stable from the point at which they were frozen. The dosing solutions were sampled on dosing days 1, 7, and 14 and analyzed for caffeine and chlorogenic acid content.
For the 90-day study, the neat test substance, the negative control, and the 3 GC group dose preparations were analyzed for caffeine and chlorogenic acid content, and the caffeine reference control preparation was analyzed for caffeine. Analyses for the GC caffeine control stability were sampled from the top, middle, and bottom of the dosing solutions at days 1, 43, and 94. Sampling for GC, caffeine, and chlorogenic acid concentration verifications was conducted on days 1, 43, and 94, and homogeneity sampling was conducted on days 1 and 94.
Bacterial Reverse Mutation Assay (Ames Test)
The Ames test (reverse mutation test) was performed at Product Safety Labs (PSL; Dayton, New Jersey) and was conducted in accordance with the US FDA GLP regulations (21 CFR part 58). The assay design was based on the International Council on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) Guidelines S2 (R21) 32 and the US FDA Redbook. 33 The reverse mutation test was performed to investigate the potential of the GC to induce genetic mutation in histidine-requiring strains of Salmonella typhimurium TA98, TA100, TA1535, and TA1537 and a tryptophan-requiring strain of Escherichia coli WP2uvrA (Molecular Toxicology, Inc, Boone, North Carolina) according to the plate incorporation method in the presence and absence of a metabolic activation system (S9 mix). Sterile water served as the negative control, whereas 5 mutagens including sodium azide (NaN3), ICR 191 acridine, daunomycin, methylmethanesulfonate, and 2-aminoanthracene (2-AA; Molecular Toxicology, Inc) were used as the positive controls. Water was also used as the solvent for the positive controls except for 2-AA which was prepared in dimethyl sulfoxide.
Plates were prepared in triplicate at each experimental point and uniquely identified. The study was conducted with GC at levels of 1.58, 5.0, 15.8, 50, 158, 500, 1,580, and 5,000 μg/plate, with the high level being the standard limit for this test. Following incubation, the number of revertant colonies was counted manually and/or with the aid of a plate counter (colony plate reader, model Colony-Doc-It, Upland, CA). For each experimental point, the mutation factor (MF) was calculated by dividing the mean revertant colony count by the mean revertant colony count for the corresponding concurrent vehicle control group. The results were considered to be positive when MF was increased at least by a factor of 2 for strains TA98, TA100, and WP2 uvrA or by at least a factor of 3 for strains TA1535 and TA1537. In addition, any increases had to be dose related and/or reproducible, that is, increases must be obtained at more than one experimental point (at least 1 strain, more than 1 dose level, more than 1 occasion, or with different methodologies).
Chromosome Aberration Assay in HPBL
The chromosomal aberration assay was used to evaluate the clastogenic potential of neat GC. The study was conducted at BioReliance Corporation (Rockville, Maryland) in accordance with the US FDA GLP regulations (21 CFR part 58). The protocol procedures described were based on the most recent versions of the US FDA Redbook, I.V.C.1 Short-Term Tests for Genetic Toxicity,
33
and OECD 473, in vitro mammalian chromosome aberration test.
34
The experimental procedure followed the study design described in the published literature.
35,36
The study was conducted using HPBL in both the absence and presence of an Aroclor 1254-induced rat liver S9 metabolic activation system (Molecular Toxicology, Inc). Peripheral blood lymphocytes were obtained from 2 healthy nonsmoking 27-year-old female donors who had no recent history of radiotherapy, viral infection, or the administration of drugs. Sterile water (Gibco; Life Technologies, Grand Island, New York) was used to prepare all test samples as well as for vehicle controls. Peripheral blood lymphocytes were cultured in complete medium (RPMI 1640 containing 15% heat-inactivated fetal bovine serum, 2 mmol/L
Acute Oral Toxicity Up-and-Down Study in Rats
This study was conducted at PSL in accordance with the GLP standards and OECD 425 guidelines and was approved by the Institutional Animal Care and Use Committees (IACUC) of PSL. Female nulliparous/nonpregnant Sprague Dawley-derived albino rats 8 to 9 weeks of age (191-204 g) were purchased from Charles River Laboratories, Inc (Kingston, New York). Animals were maintained in a temperature- and humidity-controlled room at 16°C to 20°C and 42% to 70%, respectively, under a 12-hour light–dark cycle and fed a standard Harlan Teklad Global 16% protein rodent diet 2016 (Harlan Laboratories, Inc, Indianapolis, Indiana). Following quarantine and acclimation and prior to each dosing, experimentally naive rats were fasted overnight by removing the feed from their cages. During the fasting period, the rats were examined for health and weighed (initial). Three healthy naive, 8-week-old female rats (not tested previously) were randomly selected for testing. Females were selected for the test because they are frequently more sensitive to the toxicity of test compounds than males. The test substance was administered at an initial limit dose of 5,000 mg/kg of GC to 1 healthy female rat by oral gavage. Due to the absence of mortality in this animal, 2 additional females received the same dose level, simultaneously. Since these animals survived, no additional animals were tested. All animals were observed for mortality, signs of gross toxicity, and behavioral changes at least once daily for 14 days after dosing. A battery of detailed standard clinical observations was made, and body weights were recorded prior to administration and again on days 7 and 14 (termination) following dosing. On day 14, all animals were anesthetized with CO2. Gross necropsies were performed on all animals at terminal sacrifice. Tissues and organs of the thoracic and abdominal cavities were examined.
Fourteen-Day Range Finding Study in Rats
The purpose of this study was primarily to set dose levels for the 90-day study. This study was conducted at PSL following the OECD 407 Guidelines for Testing of Chemicals and Food Ingredients and the US FDA Toxicological Principles for the Safety Assessment of Food Ingredients, 33 IV.C.4.a, and was approved by the IACUC of PSL. Seventy-eight healthy rats (39 males and 39 females 6-7 weeks of age; 240-270 g [males] and 157-179 g [females]) were purchased from Charles River Laboratories and subsequently quarantined and acclimated to the PSL facilities. Animals were maintained in a temperature- and humidity-controlled room at 16°C to 21°C and 42% to 70%, respectively, under a 12-hour light–dark cycle and fed a standard Harlan Teklad Global 16% protein rodent diet 2016 (Harlan Laboratories, Inc). The diet and filtered tap water were supplied ad libitum. Seventy rats (35 males and 35 females) were selected for the test and distributed into 7 groups with 5 males and 5 females each (1 vehicle control, 3 test substance, and 3 caffeine reference control groups). Dose levels of 0 mg/kg/d for control (group 1), 1,200, 2,500, and 5,000 mg/kg/d for GC (groups 2, 3 and 4, respectively), and 36, 75, and 150 mg/kg/d for caffeine controls (groups 5, 6, and 7) were selected. Three caffeine reference control groups were added to the study and formulated to contain caffeine levels equivalent to those expected in the GC test groups based on an expected caffeine concentration of 3% in GC, that is, 1,200 mg/kg/d of GC = 36 mg/kg/d of caffeine; 2,500 mg/kg/d of GC = 75 mg/kg/d of caffeine, and 5,000 mg/kg/d of GC = 150 mg/kg/d of caffeine. Rats were dosed daily via oral gavage for 14 consecutive days. The GC was administered in wt/vol solutions of distilled water at concentration levels of 120, 250, and 500 mg/mL. The caffeine controls were similarly administered in distilled water at concentration levels of 3.6, 7.5, and 15.0 mg/mL. All groups were dosed at a rate of 10 mL/kg.
The animals were observed daily for viability, signs of gross toxicity, and behavioral changes at least once daily during the study and weekly for a battery of detailed observations. Body weights were recorded 2 times during the acclimation period (including prior to dosing on day 1) and on days 3, 7, 11, and 14. Individual food consumption was also recorded to coincide with body weight measurements. The animals were sacrificed on day 15 and evaluated for any macroscopic changes. Food efficiency was calculated by dividing the mean body daily weight gain by the mean daily food consumption.
Ninety-Day Main Study Design
This main study was conducted at PSL in accordance with GLPs and follows the OECD Guidelines for Testing of Chemicals, Section 4 Health Effects (part 408): Repeated Dose 90-day Oral Toxicity Study in Rodents (1998) and the US FDA Toxicological Principles for the Safety Assessment of Food Ingredients 33 IV.C.4. a, and was approved by the IACUC of PSL. One hundred eleven healthy adult Crl: Sprague Dawley CD IGS rats were purchased from Charles River Laboratories and subsequently quarantined and acclimated to the PSL facilities. Animals were maintained in a temperature- and humidity-controlled room at 19°C to 23°C and 41% to 95%, respectively, under a 12-hour light–dark cycle and fed a standard Harlan Teklad Global 16% protein rodent diet 2016 (Harlan Laboratories, Inc). The diet was available ad libitum except during fasting prior to blood collection. Filtered tap water was supplied ad libitum by an automatic water dispensing system. One hundred rats were selected for testing, weighing 231 to 266 g (males) and 174 to 206 g (females), and were 8 weeks of age at initiation of dosing. All doses were administered volumetrically at 10 mL/kg. Dose levels of 0 mg/kg/d for control, 1,200, 2,500, and 5,000 mg/kg/d for GC, and 150 mg/kg/d for the caffeine control were selected for the test. The 150 mg/kg/d caffeine control represented the amount of caffeine found in the 5,000 mg/kg/d GC group.
Prior to study initiation and again on day 81, the eyes of all rats were examined by focal illumination and indirect ophthalmoscopy. The animals were observed for viability, signs of gross toxicity, and behavior changes with observations, occurring at least once daily during the study and weekly for a battery of detailed clinical observations. Body weights were recorded twice during acclimation, including prior to test initiation, weekly thereafter, and prior to terminal sacrifice. Individual food consumption was also recorded to coincide with body weight measurements. Food efficiency was calculated by dividing the mean daily body weight gain by the mean daily food consumption. Urine and blood samples were collected on days 86 (males) and 87 (females) for urinalysis, hematology, and clinical chemistry analyses. Coagulation assessments were performed at study termination prior to necropsy. Gross necropsies were performed on all decedent and surviving study animals, and selected organs and tissues from all dose groups were evaluated histologically.
Clinical pathology was performed on all surviving animals for blood chemistry and hematology at the same intervals noted previously. The animals were fasted overnight prior to each blood collection. Blood samples for hematology (except coagulation samples) and clinical chemistry were collected via sublingual bleeding under isoflurane anesthesia during week 13 of the test period. Approximately 500 μL of blood was collected in a precalibrated tube containing K2EDTA for hematology assessments. The whole blood samples were stored under refrigeration and shipped on cold packs. Approximately 1,000 μL of blood was collected into a tube containing no preservative for clinical chemistry assessments. These samples were centrifuged in a refrigerated centrifuge, and the serum was transferred to a labeled tube. Hematology and clinical chemistry indices examined included the standard list of parameters. All samples were stored in a −80°C freezer and shipped frozen on dry ice to DuPont Haskell Global Centers for Health and Environmental Sciences for evaluation.
At terminal sacrifice, all survivors were euthanized by exsanguination from the abdominal aorta under isoflurane anesthesia. All animals in the study (including the decedents) were subjected to a full necropsy, which included examination of the external surface of the body, all orifices, and the thoracic, abdominal, and cranial cavities and their contents. The standard list of tissues (from all animals sacrificed by design) were weighed wet as soon as possible after dissection to avoid drying: adrenals (combined), kidneys (combined), spleen, brain, liver, thymus, epididymides (combined), ovaries (combined), uterus, oviducts, heart, retroperitoneal fat, and gonadal fat testes (combined). The fixed tissues were trimmed, processed, embedded in paraffin, sectioned with a microtome, placed on glass microscope slides, stained with hematoxylin and eosin, and examined by light microscopy. Histological examination was performed on the preserved organs and tissues of the animals from the vehicle control, high dose, and reference control groups (groups 1, 4, and 5, respectively). With respect to the organs and tissues showing changes that potentially indicated an effect from GC, the adrenal, submandibular, sublingual, and parotid glands from all dose groups (groups 1-5) were also examined. Slide preparation and histopathological assessments were performed by Histo-Scientific Research Laboratories (Mount Jackson, Virginia).
Statistical Analyses
Repeated dose studies
Mean and standard deviations were calculated for all quantitative data. Significance was judged at a probability value of P < 0.05. Male and female rats were evaluated separately. The averages and standard deviations of body weights, food consumption, urine volume, hematology, blood chemistry, and absolute and relative organ weights were calculated and analyzed by Bartlett test for homogeneity of variances and normality. 37 Where Bartlett test indicated homogeneous variances, treated and control groups were compared using a 1-way analysis of variance (ANOVA). When 1-way ANOVA was significant, a comparison of the treated groups to the control group by Dunnett test for multiple comparisons was performed. 38,39 Where variances were considered significantly different by Bartlett test, groups were compared using a nonparametric method (Kruskal-Wallis nonparametric ANOVA). 40 When nonparametric ANOVA was significant, comparison of the treated groups to the control group was performed using Dunn test. 41 Statistical analysis was performed on all quantitative data for in-life and organ weight parameters using Provantis (version 8, Tables and Statistics; Instem LSS, Staffordshire, United Kingdom). Clinical pathology was preliminarily tested via Levene test 42 for homogeneity and via Shapiro-Wilk test 43 for normalcy followed by a 1-way ANOVA followed with Dunnett test. 38,39
Chromosome aberration study
Fisher exact test 44 was used to compare the induction of chromosome aberrations in the treated cultures and solvent control.
Results
Test Article/Materials/Analytical Chemistry
Several lots of GC were extensively analyzed for chemical composition and nutritional value (lot no. 77903061318 for the acute, 14-day, 90-day, and Ames Studies; lot no 195805021422 for the chromosome aberration assay). The basic proximate analyses of GC are summarized in Table 1.
Proximate Analysis of Guayusa Concentrate (GC).
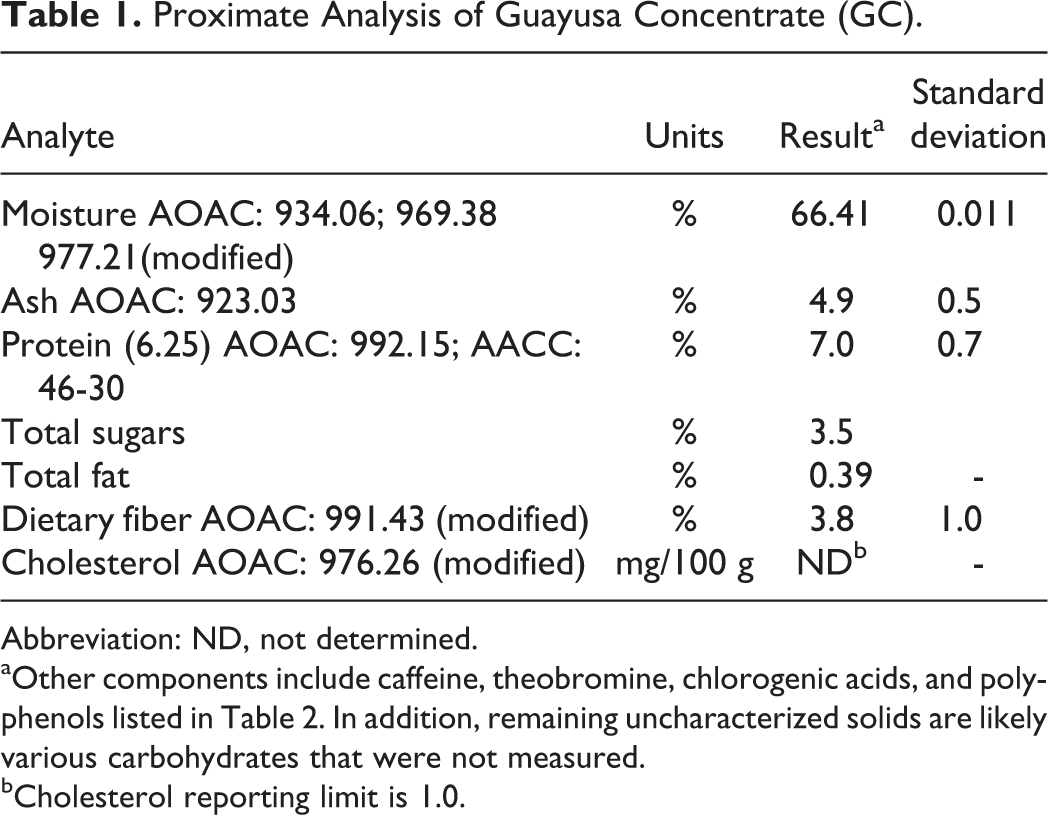
Abbreviation: ND, not determined.
aOther components include caffeine, theobromine, chlorogenic acids, and polyphenols listed in Table 2. In addition, remaining uncharacterized solids are likely various carbohydrates that were not measured.
bCholesterol reporting limit is 1.0.
Lot no. 77903061318 was further analyzed for characteristic components known to have been found in aqueous guayusa extracts and teas. The major compounds found are summarized in Table 2. In addition, no detectable levels of extracts apigenin, β-sitosterol, campesterol, cholesterol, cyanadins, delphinidins, genistein, hesperidin, kuromanin, luteolin, malvidins, naringenin, ononin, peonidins, petunidins, pterostilbene, puerarin, resveratrol, rutin, sissotrin, stigmastanol, stigmasterol, theanine, theophylline, or vitexin were found from several lots tested.
Analysis of Tested Lot of Guayusa Concentrate (GC) for Secondary Metabolites Known to Be in Guayusa.
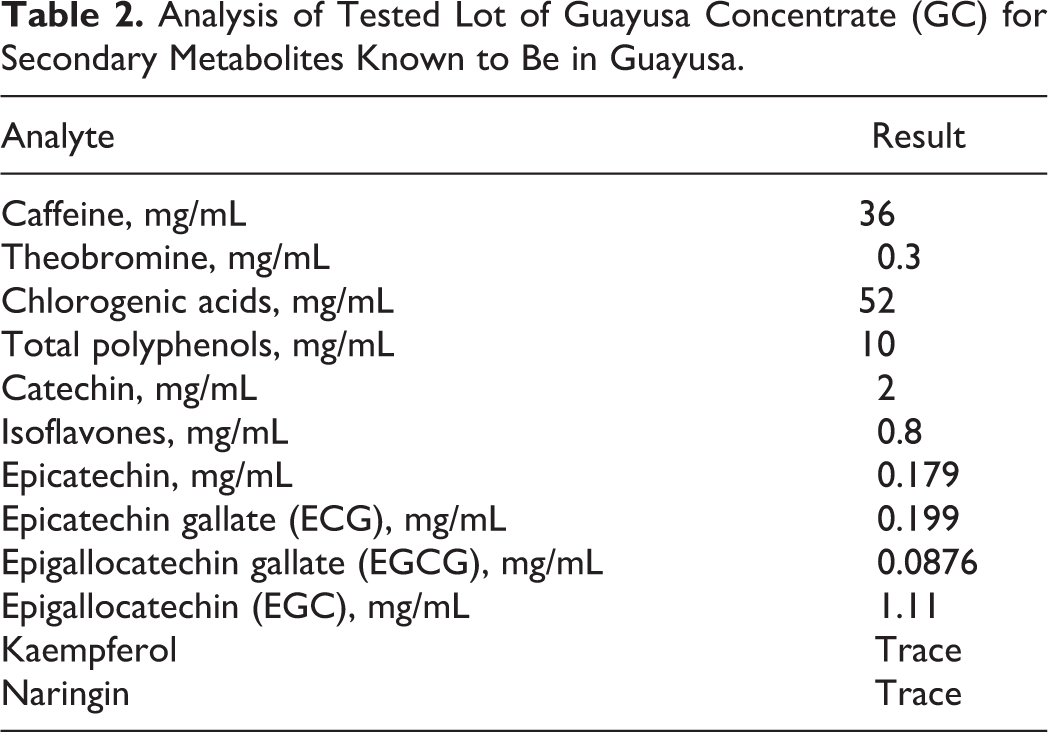
The neat test substance and groups 1 to 4 dose preparations were analyzed for caffeine and chlorogenic acid content, and group 5 caffeine reference control preparation was similarly analyzed for caffeine. Stability of the neat GC was examined at study outset and study termination for comparisons. Each dose preparation (test substance, caffeine control, and vehicle control) was sampled at the beginning, near the middle, and near the end of the study for the verification of concentration and homogeneity at each dose level. Upon sampling, dose preparations and neat test substance samples were stored frozen until the analysis was performed.
Bacterial Reverse Mutation Assay (Ames Test)
This test was designed to determine the mutagenic potential of GC using histidine-requiring strains of S typhimurium TA98, TA100, TA1535, and TA1537 and a tryptophan-requiring strain of E coli WP2uvrA. The mean number of revertant colonies was less than twice, compared to the negative control values, at all concentration levels (1.58, 5.0, 15.8, 50, 158, 500, 1,580, and 5,000 μg/plate), with the high level being the standard limit for this test of the test substance. The mean number of revertant colonies in the positive control groups was markedly increased when compared to the negative control group, confirming the sensitivity of all phases of the test. No toxic effects or precipitation of the assay material were observed in any strain at any dose of the test material, and no contamination was present in any of the assay control plates. A summary of the Ames test data is presented in Table 3.
Summary of Ames Test Revertant Colonies and Mutation Factorsa Without/With Metabolic Activation (S9).
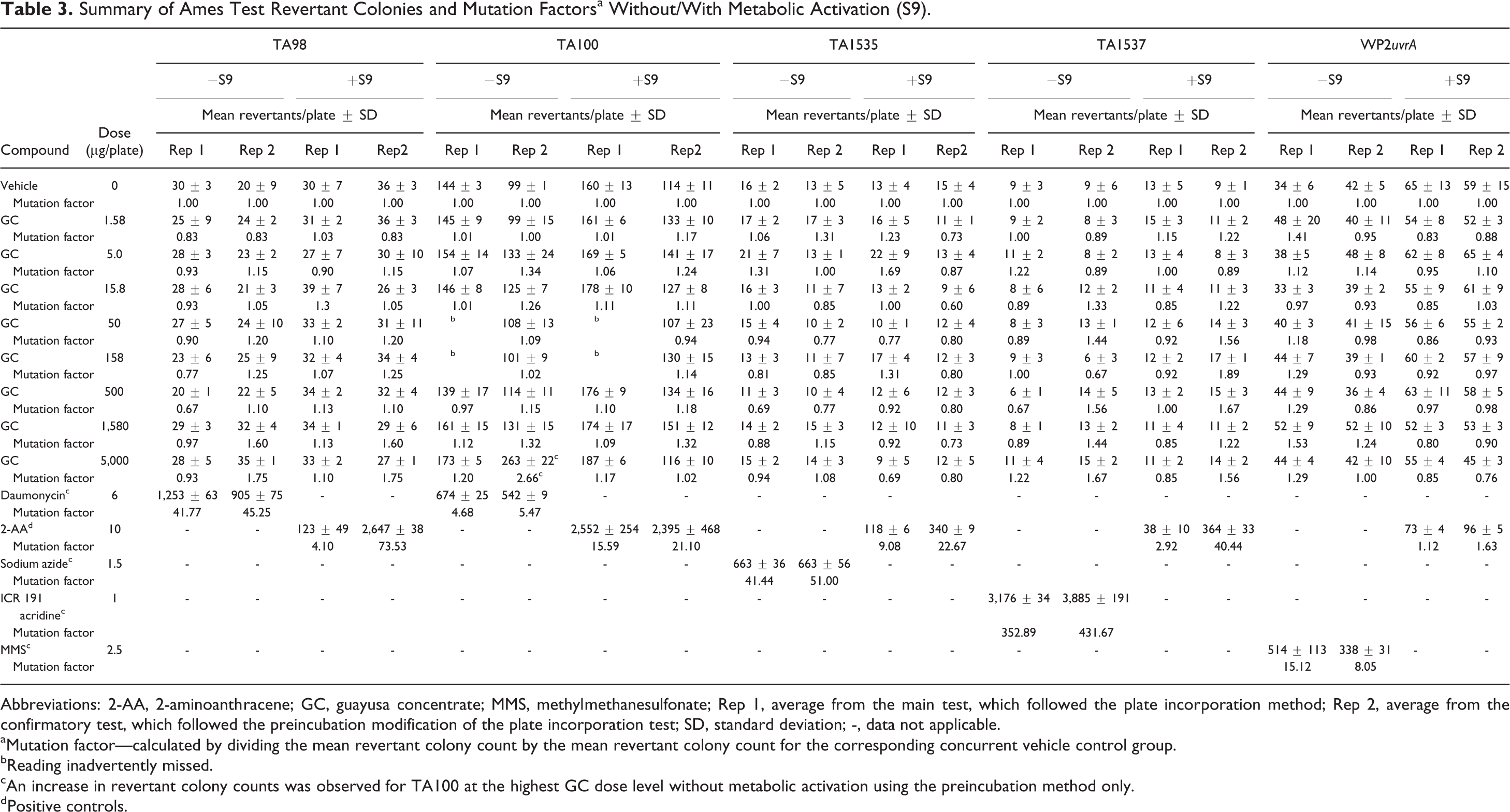
Abbreviations: 2-AA, 2-aminoanthracene; GC, guayusa concentrate; MMS, methylmethanesulfonate; Rep 1, average from the main test, which followed the plate incorporation method; Rep 2, average from the confirmatory test, which followed the preincubation modification of the plate incorporation test; SD, standard deviation; -, data not applicable.
aMutation factor—calculated by dividing the mean revertant colony count by the mean revertant colony count for the corresponding concurrent vehicle control group.
bReading inadvertently missed.
cAn increase in revertant colony counts was observed for TA100 at the highest GC dose level without metabolic activation using the preincubation method only.
dPositive controls.
An increase in revertant colony counts was observed for TA100 at the highest dose level without metabolic activation using the preincubation method only. To assess reproducibility, this portion of the test was repeated using the preincubation method without metabolic activation using 6 replicate plates (vs 3) and had a narrower dose interval than originally used. Results from additional testing with TA100 showed no increase in revertant colonies. Because the isolated increase was not clearly dose related, or reproducible between methodologies (preincubation and plate incorporation methods), and was only observed in one experimental point, this observed increase is considered to be the result of normal experimental variation rather than being indicative of bacterial mutagenicity. No increases in the number of revertant colonies observed with the remaining strains (TA1535, TA1537, TA98, or E. Coli WP2uvrA) in either the absence or the presence of S9 using either the plate incorporation or the preincubation method. Therefore, based on these findings and on the evaluation system used, GC is considered negative for bacterial mutagenicity in the Ames test.
Chromosome Aberration Assay in HPBL
The chromosome aberration assay was performed to determine the clastogenic potential of GC in HPBL cells that were exposed for 4 and 20 hours in the nonactivated test system and for 4 hours in the S9-activated test system. In the preliminary toxicity assay using GC, the doses tested ranged from 1% to 10% (vol/vol). Substantial toxicity (≥50% reduction in mitotic index relative to the vehicle control) was observed at dose levels ≥7.5% vol/vol in the nonactivated and S9-activated 4-hour exposure groups and at all dose levels tested in the nonactivated 20-hour exposure group. Based on these findings, the doses chosen for the chromosome aberration assay ranged from 0.5% to 5% vol/vol for the nonactivated and S9-activated 4-hour exposure groups and from 0.01% to 0.5% vol/vol for the nonactivated 20-hour exposure group. Similarly, the caffeine controls were tested using duplicate cultures at the following concentrations (equivalent to the caffeine concentration in GC in culture media), which included nonactivated and S9-activated 4-hour treatments at 155, 310, 620, 930, 1,240, and 1,550 µg/mL and nonactivated 20-hour treatment at 3.1, 7.75, 15.5, 31, 62, 93, 124, and 155 µg/mL. A summary of the cytogenetics data is presented in Table 4.
Summary of Cytogenetic Analysis of Human Peripheral Blood Lymphocytes Treated With GC and Caffeine.
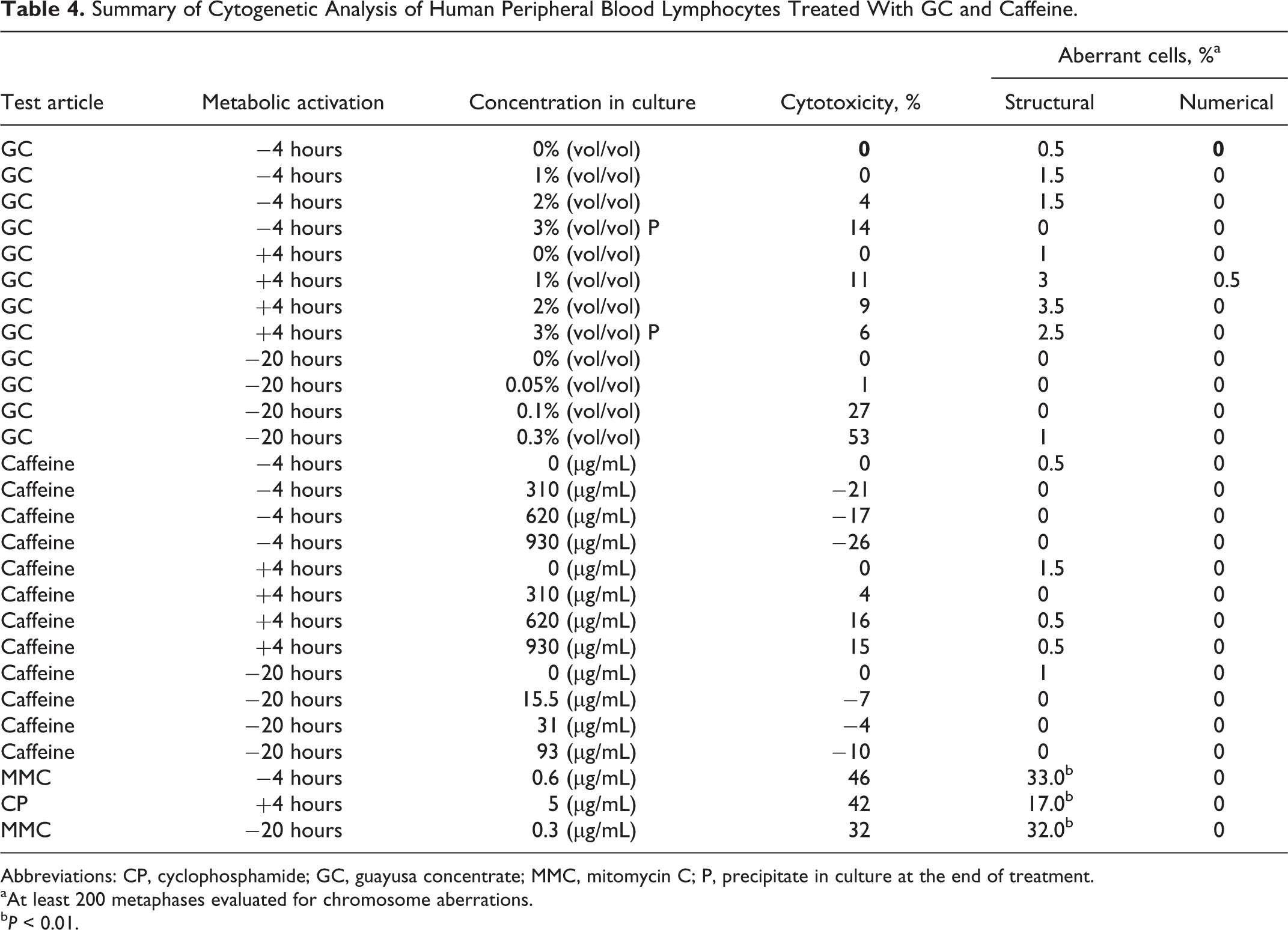
Abbreviations: CP, cyclophosphamide; GC, guayusa concentrate; MMC, mitomycin C; P, precipitate in culture at the end of treatment.
aAt least 200 metaphases evaluated for chromosome aberrations.
b P < 0.01.
In the OECD testing guideline 473 and FDA Redbook guideline for this test, the top concentration analyzed for chromosome aberrations must have 50% ± 5% cytotoxicity or precipitate at the end of treatment or 5 mg/mL (whichever comes first). In the 4-hour exposure, the top concentration was based on precipitate at the end of treatment. In the 20-hour exposure, the top concentration was based on 55% ± 5% condition (53% falls within 55% ± 5%). Thus, the study met the OECD 473 requirements of the assay. No significant or dose-dependent increases in structural or numerical (polyploid or endoreduplicated cells) aberrations were observed in either GC or caffeine control groups with or without S9. These results indicate that GC and the equivalent concentrations of caffeine control were negative for the induction of structural and numerical chromosome aberrations in both the nonactivated and S9-activated test systems in the in vitro mammalian chromosome aberration test using HPBL consistent with previous caffeine studies on human lymphocytes. 45,46,47 Untreated and positive control values were within the historical control range, indicating that the subject assay met the criteria for a valid assay.
Acute Oral Toxicity Up-and-Down Study in Rats
The acute oral toxicity study was conducted to determine the potential of GC to produce toxicity following a single oral dosing in female rats. A limit dose of 5,000 mg/kg was administered to 1 healthy female rat by oral gavage. Due to the absence of mortality in this animal, 2 additional females received the same dose level. Since these animals survived, no additional animals were tested. All animals survived test substance administration through to study termination and gained body weight during the study. Immediately following administration, the animals were hypoactive and exhibited oral discharge, abnormal respiration, hunched posture, reduced fecal volume, and/or soft feces. However, the animals recovered from these symptoms by day 3 and appeared active and healthy for the remainder of the study. No gross abnormalities were noted in any of the animals when necropsied at the conclusion of the 14-day observation period. Under the conditions of this study, the acute oral median lethal dose (LD50) of the test substance is >5,000 mg/kg of body weight in female rats.
Fourteen-Day Oral Dose Range Finding Study in Rats
Analysis of the test substance
The test substance was stable over the course of this study. Based on the overall concentration verification analysis, animals were considered to have received target dose concentrations of GC and caffeine reference control within an expected margin of variation (±15%; data not presented).
Mortality, general appearance, body weight, and food consumption
There were no test substance-related or incidental mortalities in this study. Animals treated with GC at 5,000 mg/kg/d had evidence of salivation and hypoactivity. Dose-dependent hypoactivity was also observed with the intermediate (75 mg/kg/d) and high-dose (150 mg/kg/d) caffeine groups. Statistically significant concentration-dependent reductions in body weights were noted in both sexes; however, they were more pronounced in males. In addition, initial reductions in body weight gain, food consumption, and food efficiency were observed in both males and females in test substance and caffeine-treated groups. Although residual decreases in food efficiency were considered test substance related, they did not adversely affect the animals as indicated by their steady weight gain following initial reductions (data not presented). There were no macroscopic observations at necropsy in male or female rats attributable to the administration of either GC or caffeine.
Based on these findings, the dose levels were determined to be 5,000, 2,500 and 1,200 mg/kg/d for the 90-day study. A single high dose of caffeine was used as a positive control at the same amount of caffeine found in the high-dose GC (150 mg/kg/d).
Subchronic 90-Day Oral Dose Toxicity Study in Rats
Analysis of the test substance
Over the course of the study, the percentage change in measured GC caffeine content when compared to the caffeine reference standard was −4.4%, resulted in a test substance stability of 95.6%. The percentage change in measured GC chlorogenic acid content when compared to the chlorogenic acid reference standard over the same time period was 1.1%, resulted in a test substance stability of 101.1%. Based on these findings, the test and reference control substances were determined to be stable and homogeneous over the course of this study. Analyses for caffeine and chlorogenic acid sampled from the top, middle, and bottom of the dosing solutions found all dose preparations to be homogeneously distributed over the course of the study.
Concentration verification analysis results for the study overall averaged 98.4%, 89.1%, and 87.4% of caffeine for groups 2,3, and 4, respectively. Comparison of the overall caffeine concentration results with the targeted nominal concentration indicated that the animals received 100.4%, 90.9%, and 89.2% of the targeted 3% caffeine concentration in GC for groups 2, 3, and 4, respectively. Concentration verification analysis results for chlorogenic acid for the study overall averaged 104.3%, 93.4%, and 92.2% of the targeted concentration for groups 2, 3, and 4, respectively. Neither caffeine nor chlorogenic acid was detected in the vehicle control group 1 dosing solutions. Therefore, based on the stability and concentration verification results, the animals are considered to have received the targeted dose levels of GC (groups 2-4) and caffeine reference substance (group 5).
Mortality, general appearance, body weight, and food consumption
There were no GC-related mortalities in this 90-day rat study. Three animals were found dead during the course of the study: 1 intermediate dose (2,500 mg/kg/d) group male was found dead on day 84 and 2 caffeine reference control animals were found dead on day 47. One high-dose (5,000 mg/kg/d) male was sacrificed in a moribund condition on day 81, due to a decline in general health associated with reductions in food consumption and body weight after sustaining a malocclusion. Clinical observations directly attributed to GC administration for both decedents and surviving animals included salivation in intermediate-dose group males and females and occasional hypoactivity in males of intermediate- and high-dose groups. These findings were also present in the caffeine controls and are considered to be related to the effects of the caffeine present within the GC. There were no ophthalmological changes noted in any test or control animals. Body weight and body weight gain reductions occurred in males in all treated groups. Body weights were slightly lower in females over the course of the study, however, the differences compared to controls were not statistically significant. Table 5 provides a summary of the weekly mean male and female body weights and body weight gains. There were no significant changes in food consumption in either male or female rats in any GC or caffeine groups; however, there were dose-dependent decreases in food efficiency in the GC groups and decreases in the caffeine control (data not presented).
Mean Weekly Body Weight and Body Weight Gain (g ± SD)—90-Day Oral Rat Guayusa Concentrate Study.
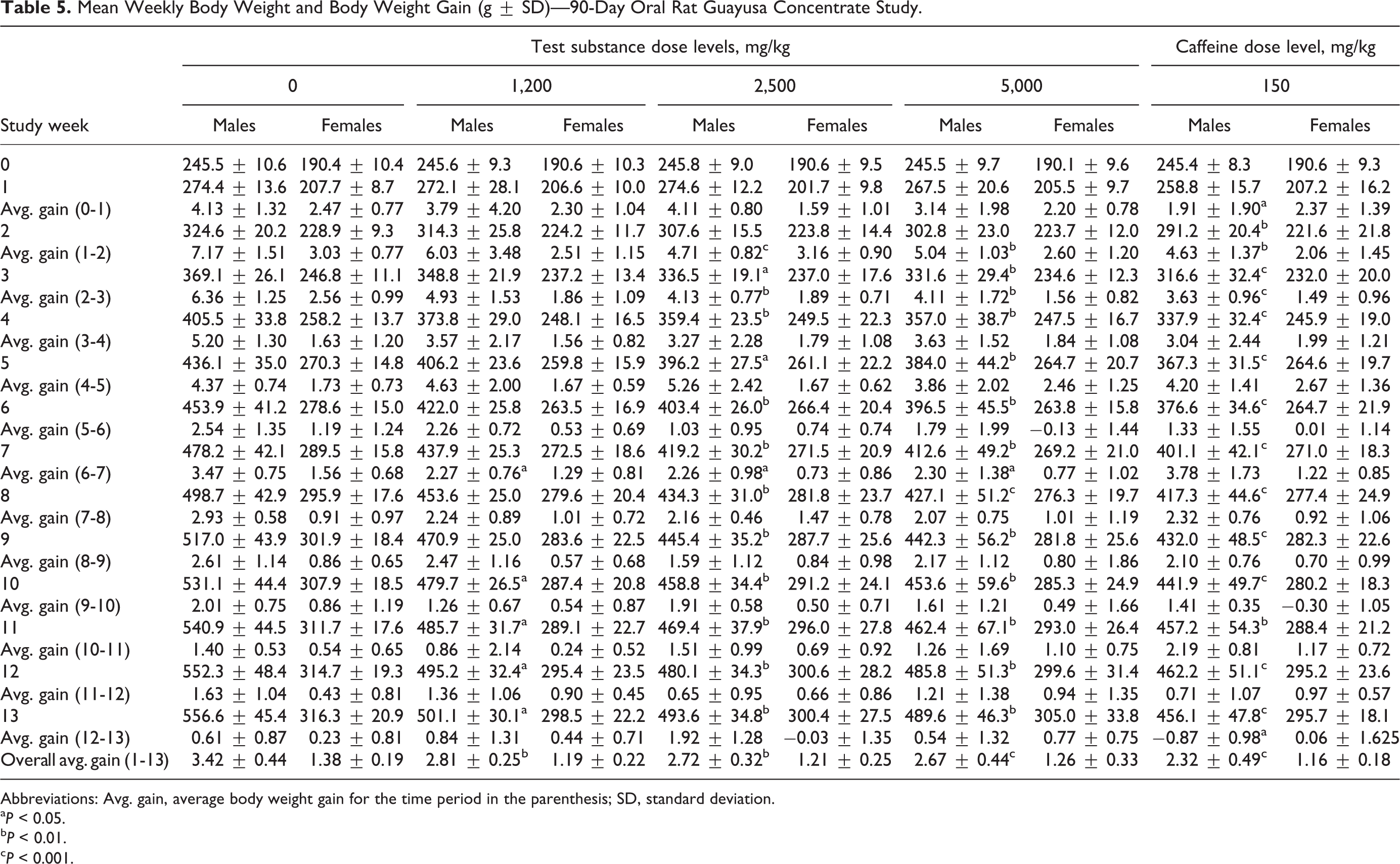
Abbreviations: Avg. gain, average body weight gain for the time period in the parenthesis; SD, standard deviation.
a P < 0.05.
b P < 0.01.
c P < 0.001.
Urinary, hematological, and biochemical findings
There were no GC-related changes in urinalysis parameters in male rats. All urinary parameters were within normal limits for females, except the urinary protein concentration was decreased in females in the high-dose group and in the caffeine control group (data not shown). There were no GC-related red blood cell changes observed in males. Red blood cell changes observed in high-dose females consisted of increased hemoglobin concentration (HGB), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and red blood cell distribution width (RDW). Increased RDW was also observed in the caffeine reference controls. Dose-dependent increases in neutrophil and basophil counts were observed in females in the intermediate- and high-dose groups as well as the caffeine reference controls. There were no GC-related changes in blood cell morphology or in coagulation parameters in females; however, prothrombin times (PTs) and activated partial thromboplastin times were decreased in males at all treatment levels and in the caffeine controls (data not presented).
A summary of the mean clinical chemistry changes interpreted to be associated with test substance administration is summarized in Table 6. Changes in clinical chemistry parameters associated with lipid metabolism included decreased TGs (all test groups and caffeine controls) and increased cholesterol observed in males in the intermediate- and high-dose groups and in females at the high-dose group. The caffeine controls showed decreased TGs in both males and females and increased cholesterol and creatinine in males.
Summary of Mean Clinical Chemistry Values—90-Day Oral Rat Guayusa Concentrate Study.
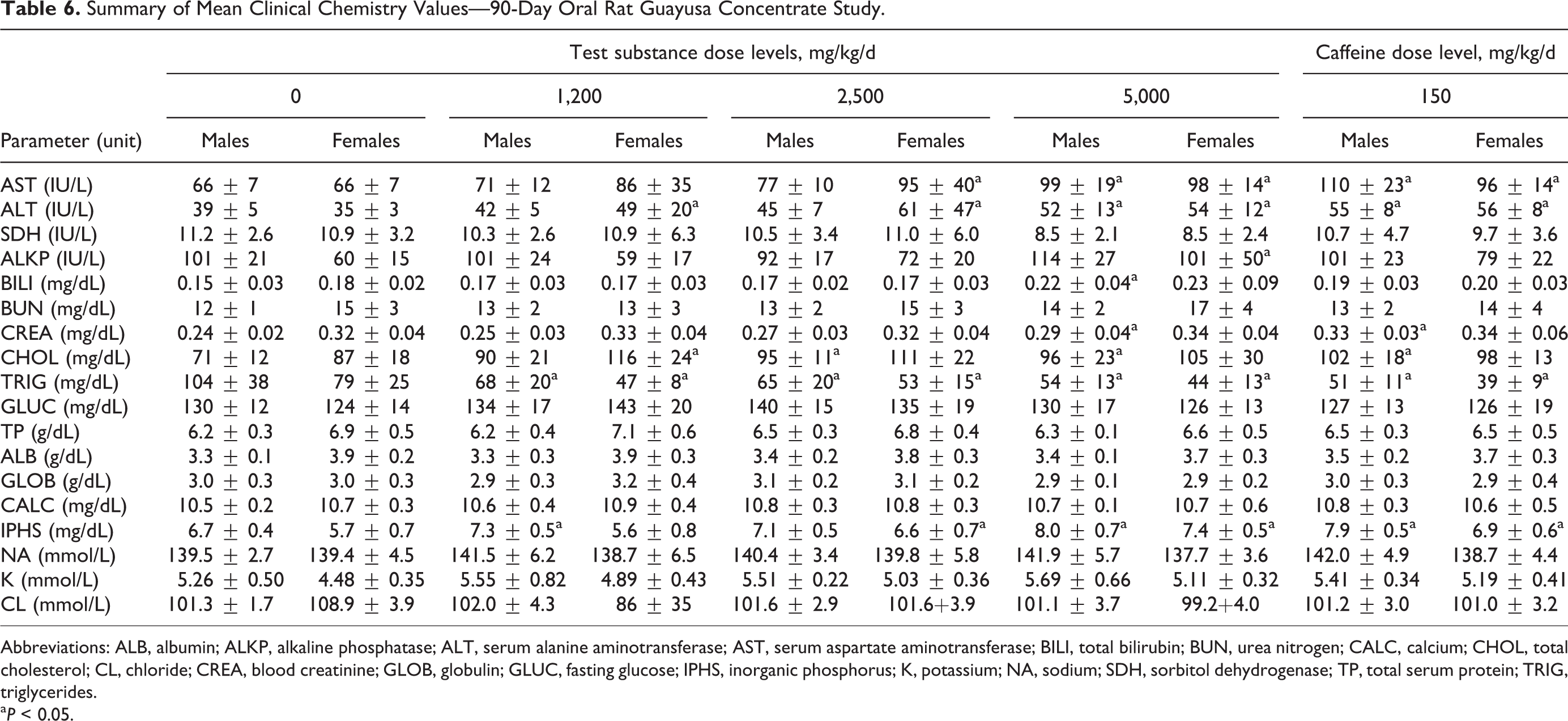
Abbreviations: ALB, albumin; ALKP, alkaline phosphatase; ALT, serum alanine aminotransferase; AST, serum aspartate aminotransferase; BILI, total bilirubin; BUN, urea nitrogen; CALC, calcium; CHOL, total cholesterol; CL, chloride; CREA, blood creatinine; GLOB, globulin; GLUC, fasting glucose; IPHS, inorganic phosphorus; K, potassium; NA, sodium; SDH, sorbitol dehydrogenase; TP, total serum protein; TRIG, triglycerides.
a P < 0.05.
Necropsy findings, organ weights, and histopathological examination
None of the 4 premature deaths that occurred during the course of the study were determined to be related to GC exposure. One intermediate-dose (2,500 mg/kg/d) male was found dead on study day 84 with distended large intestines, red discolored lungs, fluid in the thoracic cavity, and dark thymus. Microscopically, moderate acute inflammation of the thymus and slight diffuse pleuritis were observed. The cause of death of this animal could not be determined. One high-dose (5,000 mg/kg/d) male was sacrificed when found moribund on day 81 due to a subsequent decline in general health caused by reductions in food consumption and body weight after sustaining a malocclusion. This animal presented with small thymus, enlarged adrenal glands, distended small and large intestines, and malocclusion of the upper incisors. These signs correlated with the microscopic findings of moderate atrophy of the thymus and a moderate abscess within the maxillary teeth and surrounding bones, respectively. In the absence of other significant findings, the atrophy of the thymus was considered secondary to morbidity, and the tooth abscess was considered as the cause of morbidity. The morbidity and the findings were considered unrelated to GC treatment. There were no microscopic correlations with the gross findings observed in the adrenal glands and intestines of this animal.
Two animals treated with caffeine were found dead on day 47. One animal exhibited red discolored lungs, small intestines, and kidneys, a dark thymus, and mottled liver. Diffuse slight congestion of the lungs was present microscopically. The other animal presented with enlarged adrenal glands, small intestines filled with a soft, green substance, a distended, fluid-filled stomach, and red/dark discolored liver, lungs, ovaries, uterus, oviducts, thymus, and kidneys. Microscopically, minimal to moderate hemorrhage was present in the adrenal cortex, liver, and thymus. A definitive cause of death for these animals could not be determined. At study termination, there were no macroscopic findings directly attributed to GC administration. A small thymus with associated reduced organ weight, without microscopic correlate, was observed in 1 caffeine-treated group 5 male. In addition, enlarged adrenal glands were observed in 1 group 4 female and 1 caffeine reference control group 5 female, which correlated with slight cortical hypertrophy in the group 4 female only.
Dose-dependent reductions in gonadal and retroperitoneal absolute and relative fat pad weights compared with vehicle control were observed in males and females from all GC treatment groups. Statistically significant decreases occurred in the high dose and caffeine control brain, epididymides, gonadal and retroperitoneal fat pads, liver, spleen, and thymus weights. These changes were slightly more severe in females. Other changes in mean organ weights and mean organ weight ratios were noted; however, they were considered to be secondary to proportional reductions in overall body weight and/or decreased animal health status.
Microscopically, 90-day exposure to GC induced minimal to marked hypertrophy in the salivary glands of all treatment group animals, including those in the caffeine control group. The incidence and severity of the changes in the salivary glands were largely dose dependent, with a greater impact on females and with comparable incidence and severity in the high-dose group and the caffeine control group. Slight hypertrophy was also noted in the adrenal glands, which was considered secondary to treatment-related stress. The remaining microscopic findings were considered incidental—unrelated to treatment because they were sporadic in nature—at incidences consistent with the control group and/or are commonly observed in rats of this age and strain. The summary of the mean terminal body and organ weights is presented in Table 7.
Summary of Mean Terminal Body Weights and Organ Weights (g ± SD)—90-Day Oral Rat Guayusa Concentrate Study.
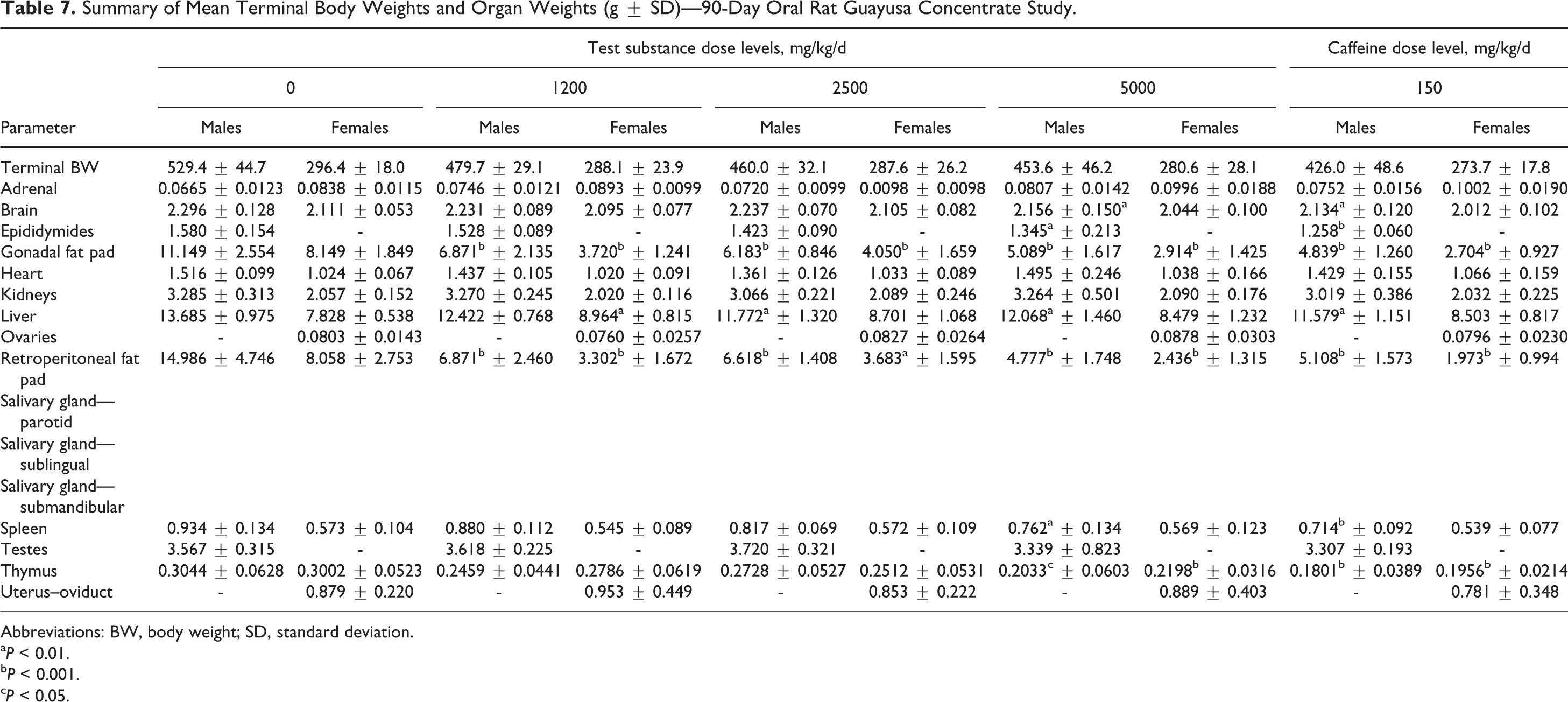
Abbreviations: BW, body weight; SD, standard deviation.
a P < 0.01.
b P < 0.001.
c P < 0.05.
Discussion
The toxicological potential of a concentrate made from I guayusa—GC—was evaluated in a series of genetic and general toxicity assays. Ilex guayusa is a holly tree found in South America whose leaves are steeped in boiling water to prepare a tea-like beverage. The chemical composition is typical of plant extract. Protein and carbohydrates are the primary organic constituents. In addition, GC contains several secondary metabolites found in many other plants, most notably about 3% caffeine as well as chlorogenic acids, theophylline, and polyphenols.
Genetic toxicity of GC was evaluated using an in vitro mutagenicity study (standard Ames test) and an in vitro chromosome aberration study (chromosome aberration test in human lymphocytes). General toxicity was evaluated in an acute up-and-down oral toxicity study in female rats and in a 14-day range finding study and a 90-day study in both male and female rats. In the in vitro mutagenicity study, GC showed no toxic effects or precipitation in any strain at any dose level. Guayusa concentrate was not mutagenic in the presence or absence of metabolic activation in S typhimurium strains TA1535, TA1537, TA98, TA100, or E coli WP2 uvrA. Based on these findings and on the evaluation system used, GC is considered to be negative for bacterial mutagenicity. In an in vitro chromosome aberration study of HPBL cells, neither the GC nor the caffeine-positive controls elicited significant or dose-dependent increases in structural or numerical aberrations with or without S9. Therefore, the study was deemed to be negative for chromosomal aberrations. Therefore, the overall genetic toxicity of GC was negative in both the functional and structural assays. Although there were no genetic toxicity studies available in the literature on guayusa or its extracts, these data are in general agreement with caffeine genetic toxicity studies summarized by. 25,37,48,49 These authors have collectively found that although evidence for the mutagenic potential of caffeine is marginal in lower organisms and may be conflicting in some areas, it appears to be unlikely that at normal, physiologically relevant levels of consumption, caffeine would result in mutagenic effects in humans.
An acute oral toxicity up-and-down procedure was performed in female rats at a single oral dose of 5,000 mg/kg of GC (the equivalent of 150 mg/kg of caffeine), which produced no mortality and some toxic signs that cleared by day 3. The study resulted in an LD50 for GC above 5,000 mg/kg. This dose of GC is equivalent to a dose of 150 mg caffeine/kg as compared to previously reported rat oral caffeine LD50 values ranging from 200 to 400 mg/kg. 50,51
A 14-day repeated oral gavage range finding study was performed at GC doses of 0, 1,200, 2,500, and 5,000 mg/kg/d and doses of 36, 75, and 150 mg/kg/d for caffeine as positive controls—each representing the comparable amounts of caffeine to each successive GC group, respectively. There were reductions in body weights, body weight gain, food consumption (not presented), and food efficiency, which occurred in males in the mid- and high-dose levels of both GC and caffeine controls and in females in the mid-dose GC and the high dose of the caffeine controls. These decreases were not adverse because the animals continued to gain weight following the initial reductions. Additionally, there were no gross necropsy findings in any rats at study termination (no microscopic examination was performed). From these observations, it was determined that no adverse effects occurred in either the GC or the caffeine test groups at the end of the 14-day dosing period.
Based on the 14-day data, a subchronic 90-day oral gavage study was conducted in rats at 0, 1,200, 2,500 and 5,000 mg/kg/d of GC and a single caffeine positive control at 150 mg/kg/d, which reflected the amount of caffeine in the 5,000 mg/kg/d dose of GC (3%). A number of findings were noted during the course of the 90 days, which are described in detail in the Results section. Table 8 summarizes the relevant significant findings from all areas by treatment group. Where laboratory historical control ranges were available, they were entered into the appropriate column for comparison.
Summary of Effects of Guayusa Concentrate and Caffeine in 90-Day Rat Study.
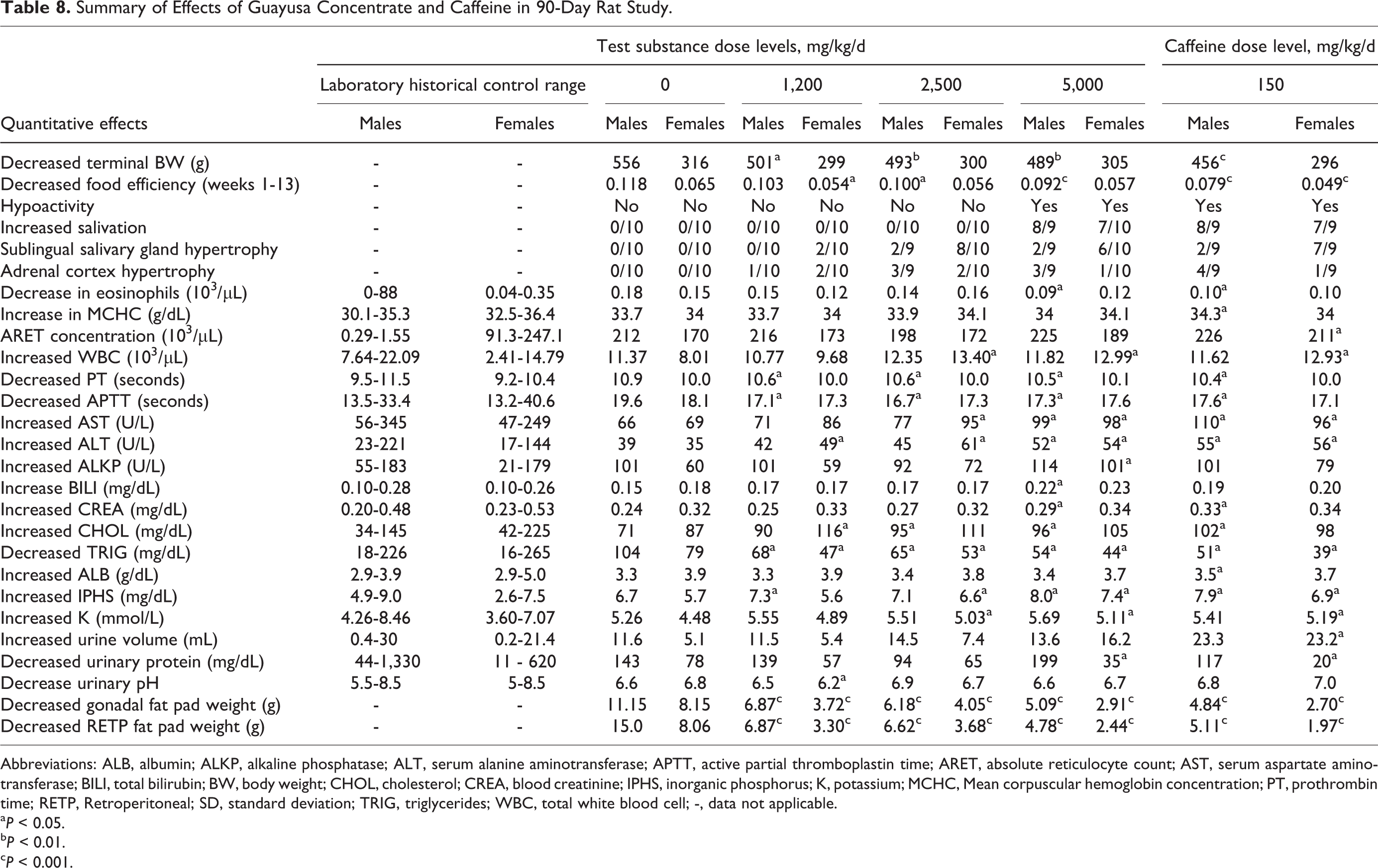
Abbreviations: ALB, albumin; ALKP, alkaline phosphatase; ALT, serum alanine aminotransferase; APTT, active partial thromboplastin time; ARET, absolute reticulocyte count; AST, serum aspartate aminotransferase; BILI, total bilirubin; BW, body weight; CHOL, cholesterol; CREA, blood creatinine; IPHS, inorganic phosphorus; K, potassium; MCHC, Mean corpuscular hemoglobin concentration; PT, prothrombin time; RETP, Retroperitoneal; SD, standard deviation; TRIG, triglycerides; WBC, total white blood cell; -, data not applicable.
a P < 0.05.
b P < 0.01.
c P < 0.001.
Composite Table 8 illustrates that several changes appear to be related to the treatment with GC for a period of at least 90 days. The most prominent dose-related effects were decreased body weight gain, salivary gland hypertrophy, reduced serum TGs, and reduced weight of gonadal and retroperitoneal fat pads. Other minor effects of GC concentrate such as hypoactivity, increased salivation, decreased eosinophil counts, reduced thymus weights, adrenal cortex hypertrophy, increased bilirubin, increased white blood cells (WBCs), and PT in females were also seen in the caffeine control group.
Reductions in body weight and body weight gain occurred in males in all treated groups by week 6. Without alteration in food consumption, these changes had corresponding dose-dependent decreases in food efficiency. Although not statistically significant, suggestive trends in decreased female body weights were also noted after the second week of administration. However, these reductions resolved over the subsequent weeks, resulting in no significant changes or trends in body weight or body weight gains (data not presented) observed in females for the study overall. These body weight and food efficiency effects were also noted in the caffeine control group males and females, which are interpreted to be associated with the caffeine content of the GC. Previous studies have identified decreased body weights in rodents as an effect of caffeine ingestion. 52 -57 In addition, Zheng et al 58 showed that caffeine, catechins, and theanine can result in body weight reductions in mice. Similar effects were noted in a 90-day rat study in the methylaxanthine theobromine at doses up to 250 mg/kg/d. 59 These changes were also consistent with previous studies showing caffeine effects on food intake. 60 Overall, the reductions in body weight, body weight gain, and food efficiency were most profound in males. Clinical studies have shown similar findings in studies on caffeine where males manifest greater positive effects than females. 61 Although the precise pharmacologic effects remain unknown, Temple and Zeigler 61 postulate that levels of circulating steroid hormones may play a role in the gender disparity on the effects of caffeine.
Clinical observations considered attributable to the administration of GC included slight–moderate salivation and hypoactivity. The increased salivation, noted in most animals in the high dose, correlates microscopically with hypertrophy in the submandibular, sublingual, and parotid salivary glands. Sporadic hypoactivity was noted in males from the mid- and high-dose groups. These findings were also present in the caffeine control animals. Similar hypoactivity in rats has been noted postcaffeine exposure in rats by Herz and Beninger 62 and Zimmerberg et al. 63 Caffeine and theophylline are also known hypersalivation agents in humans. 64
Although there are statistically significant increases in PTs and activated partial prothrombin times in all male GC and caffeine groups, the effects were within the historical control range of the performing laboratory, revealed no correlating clinical or pathological data, and were not considered to be related to the administration of GC. None of these effects were noted in females. The changes noted in this study do not indicate that GC or caffeine affected changes in coagulation times in rats. An early study revealed no evidence that caffeine, theobromine, and/or theophylline increased the prothrombin level of the blood in dogs and rabbits when fed in large doses up to 200 mg/kg/d, 65 whereas a more recent study provided evidence that caffeine in combination with taurine may decrease PTs in human volunteers. 66
There were no GC-related red blood cell count changes noted in males. Red blood cell changes observed in females in the high-dose GC group consisted of increased HGB, MCV, MCH, and RDW. Increased RDW was also observed in the 150 mg/kg caffeine reference control. Although of potential toxicological relevance, these changes were generally within PSL historical range, of very low magnitude, and not associated with any other hematological, histopathological, or clinical findings.
Dose-dependent increases in neutrophil and basophil counts were observed in mid-dose, high-dose, and caffeine control females. Because these changes were observed in the concurrent caffeine-treated group, they are interpreted to be potentially associated with caffeine content of the test substance. Additionally, absolute monocyte counts were increased in females administered 5,000 mg/kg GC. Increases were also observed in WBC, lymphocyte, and large unstained cell counts in females in the mid doses and high doses as well as in the caffeine control females. Since these changes were not dose dependent, were of very low magnitude, and were within the PSL historical range, they are interpreted not to be related to treatment. Eosinophil counts were decreased in the males in the high-dose group and in the caffeine control males. Because this finding was also within the PSL historical control range and not associated with any other hematology changes, it was not considered to be toxicologically relevant.
Clinical chemistry changes were observed in male rats at all treatment levels and in females at the mid- and high-dose groups. Increases observed in males at the high dose and in the caffeine control included AST, ALT, creatinine, and phosphorus. Changes in liver enzymes in both males and females may be a result of processes associated with caffeine metabolism. 67 Liver function enzymes, ALT, AST, and alkaline phosphatase were reported as elevated in studies conducted by Khayyat et al 68 and Crişan et al 69 after treatment with energy drinks containing caffeine. A 90-day oral study in rats with caffeine conducted by National Toxicology Program (NTP) at doses up to 272 mg/kg/d for males and 287 mg/kg/d in females also revealed significant changes in AST and ALT blood chemistry values, which were not considered to be adverse since there was no dose response noted. 50,55 In 2011, Akande and Banjoko 70 reported an increase in the serum AST, ALT, and alkaline phosphatase in rats treated with caffeine-containing energy drinks that typically contain 80 to 141 mg of caffeine per 8 ounces. 71 Ebuehi et al 72 reported that caffeine-containing energy drinks significantly affected liver enzyme activities in rabbits, whereas Cheul Do et al 73 reported that caffeine administration caused a significant increase in the levels of AST and ALT in rats. Bukhar et al 74 reported that rats receiving a caffeine-containing energy drink also showed elevated AST, ALT, and alkaline phosphatase values when compared to controls. Ugwuja 75 reported that the consumption of energy drinks alone or in combination with alcohol is associated with significant alterations in various biochemical parameters. On the other hand, Ruhl and Everhart 76 and Cadden et al 77 found decreases in ALT values upon exposure to caffeine.
Triglycerides were decreased in all treatment groups in the 90-day study, and the cholesterol was increased in the mid- and high-dose as well as the caffeine control groups. These latter 2 parameters are associated with lipid metabolism and are interpreted to be associated with the caffeine content of the GC as was also described by Hongu and Sachan. Indeed, increased cholesterol has been shown to be associated with caffeine consumption, 78,79 and decreased TG concentration has also been attributable to the pharmacological effect of caffeine. 80,81 A study by Zheng et al 58 concluded that catechins and caffeine were synergistic in their antiobesity effects. Black tea studies conducted by Wang et al 82 revealed a dose-dependent decrease in TG in low- and high-dose group females and decreased ALT and cholesterol values in the high dose. Further, the high-dose males in that study had decreased cholesterol and TG values. Other xanthines found in GC, such as theophylline, can change blood chemistry values, which correlate with increases in glucose, uric acid, and cholesterol. 83 Tea catechins alone have been shown to decrease cholesterol, cholesterol ester, total cholesterol, and high-density lipoprotein cholesterol. 84 Sugiura et al 85 also reported decreased TG levels with exposure to catechins, EGC, and epicatechin gallate. Flavonoids, which are also constituents of GC, possess a wide spectrum of biological actions including hypoazotemic, hypotension, estrogenous, spasmolytic, cholagogue, anti-inflammatory, and antioxidant activities. 86 Since the changes in the current study occurred in both the GC- and caffeine-treated groups—and some of these same effects have been noted by numerous researchers studying the changes mediated by methylxanthines—we postulate that the changes in the guayusa and caffeine groups are associated with pharmacological effects of methylxanthines/caffeine metabolism as suggested by Kot and Daniel 67 and Thorn et al. 87 Specifically, decreased TG concentration is likely related to the pharmacological effect of caffeine on adipose tissue, which resulted in reduction of fat pad weights observed at necropsy in males and females at all dose levels. Additional clinical chemistry changes observed in the intermediate-dose groups were similar to increases in the caffeine controls and, therefore, are most likely associated with pharmacological effects of caffeine.
There were no GC-related changes in urinalysis parameters in male rats noted at study termination. Urinary protein concentration was decreased in the high-dose females and in the caffeine control females. These values are considered secondary to caffeine intake, and since the results were within the range of historical control values of the performing laboratory and unaccompanied by any other corresponding clinical or histopathologic changes, they are interpreted to be nonadverse. Urinary pH was decreased in the low-dose females; however, due to a lack of dose relationship, this finding was not considered toxicologically relevant. Microscopic evaluation of the urine revealed no changes from controls in any treated group.
At study termination, no macroscopic findings were noted that could be attributed to the administration of GC. Incidental findings included a small thymus and an enlarged adrenal glands, which were considered secondary to treatment-related stress, a finding consistent with the Scientific and Regulatory Policy Committee’s recent review on this subject authorized by the Society of Toxicologic Pathology. 88 Other incidental sporadic macroscopic observations were within physiological range for rats of this strain and age and were therefore considered unrelated to GC administration.
Microscopic examination revealed minimal to marked hypertrophy in the salivary glands of all treatment groups, including both male and female caffeine control groups. The incidence and severity of the changes in the salivary glands were largely dose dependent, with a greater impact on females. The submandibular and sublingual salivary glands were affected at all dose levels, but changes in the parotid gland were only observed in the mid- and high-dose levels. The salivary gland hypertrophy in the high-dose females was similar to that of the caffeine control females.
Effects of caffeine and other phosphodiesterase inhibitors on salivary glands are well known. 89 -99 In a 1-year study in Wistar rats, hyperemia was observed at doses above 181 mg/kg. 100 In the NTP study on caffeine in Fischer rats, a dose-dependent effect on cellular enlargement in salivary glands was noted and considered to be adaptive. Reversible effects in the salivary gland are a well-known sympathomimetic effect of caffeine. 50 The astringent nature of the more concentrated test material used in this study as opposed to the drinking water solutions used in the NTP study may have contributed to the effect on salivary glands.
Chemicals that perturb the cAMP or any of the accompanying chain of regulatory events involved have been shown to modify epithelial cell proliferation in the gastrointestinal tract. 101 Recently, Inoue et al 102 has shown that hypertrophy induced by astringent types of substances, such as grape skin extract and tannic acid, creates an adaptive, nonadverse effect, which is reversible upon removal of the irritant. The changes noted with these investigations were not degenerative changes and were considered to be an adaptive physiological response. Therefore, we conclude this is an adaptive change with potential reversibility that is indicative of any systemic toxicity of GC.
Significant differences in absolute and relative organ weights were observed in gonadal and retroperitoneal absolute and relative fat weights in males and females from all GC treatment groups and in male and female caffeine controls. Changes in gonadal and retroperitoneal fat weights in the current study were slightly more severe in females and were considered to be related to caffeine administration as shown by many other published studies. Caffeine has been reported to break down fat in in vitro studies on 3T3-L1 mouse adipose cells. 103
Studies in humans reported that caffeine ingestion elevated the metabolic rate and fat oxidation in vivo through lipolysis in fat cells and the release of catecholamines. 104 -106 Vassalli and Jeanrenaud 107 reported that caffeine promotes lipolysis in mouse adipocytes. Verdy 108 reported similar findings in human adipocytes. Bukowiecki et al 109 reported that intake of 0.057% and 0.2% of dietary caffeine for 9 weeks in rats reduced the weight of parametrial white adipose tissue. Chen et al 110 showed that intake of 4 mg/d of caffeine in mice decreased the weight of body fat in genetically obese mice. Kobayashi-Hattori et al 111 examined Sprague Dawley rats administered 0.025%, 0.05%, and 0.1% caffeine in diets over 21 days and report significant reductions in body fat mass and body fat percentage. These authors report that 5 mg/kg/d of caffeine in Sprague Dawley rats revealed an increase in the serum concentrations of catecholamines and free fatty acids, suggesting that caffeine ingestion reduced body fat by lipolysis via catecholamines. Muroyama et al 112 reported that the intake of caffeine reduces the body fat mass and body fat percentage in mice at doses of 37.5 mg/100 g diet, suggesting that caffeine is effective in reducing adipose tissue mass as well as improving disorders in lipid metabolism. In a detailed summary report on the adverse effects of caffeine for the FDA, Milanez 48 indicates subchronic studies (21-210 days in duration) in rats and mice found that caffeine intake decreased food intake and body weight gain and decreased body fat. Among the studies cited is one conducted by Choi et al 113 in which pancreatectomized male Sprague Dawley rats exposed to a 0.01% solution of caffeine (7.6 ± 0.9 mg/d caffeine) revealed suppressed body weight gains and epididymal fat pads. Another cited study was by Michna et al 114 who reported that a solution of 0.4 mg/mL caffeine administered to SKH-1 mice for 15 weeks decreased weights of the parametrial fat pads. Zheng et al 58 was also cited where it was reported that mice exposed to 0.05% dietary caffeine for 16 weeks significantly suppressed body weight gains and fatty accumulation. Hongu and Sachan 115 reported that Sprague Dawley rats were fed 0.1 g/kg/d of caffeine (estimated intake of 6 mg/kg/d) in the diet for 28 days, and this resulted in significant reductions in fat pad weights and total lipids of epididymal, inguinal, and perirenal regions. Wilcox 57 administered 5 mg/kg/d of caffeine to male rats for 9 weeks, in which epididymal and retroperitoneal fat pad weights and epididymal fat cell size were found to be significantly reduced in the caffeine-treated groups.
Slight hypertrophy was also noted in the adrenal glands, and this is considered to be secondary to treatment-related stress and is not a direct effect of GC. Absolute or relative reductions in thymus, spleen, or epididymides weights, along with absolute and relative increases in adrenal gland weights in either males or females, are also interpreted to be secondary to treatment-related stress as described by Everds et al 88 and/or reductions in body weight. Other changes in mean organ weights and mean organ weight ratios with and without statistical significance do not have a macroscopic or microscopic correlate and were considered to be secondary to proportional reductions in overall body weight and/or decreased animal health status. The other microscopic findings occurred sporadically or at a similar incidence to control and other test-treated groups and were generally of the type commonly seen in rats of this strain and age 116 -118 and were considered to be incidental and unrelated to treatment with the test substance or its caffeine content.
In conclusion, both mutagenic and clastogenic testing of liquid GC revealed no genotoxicity. The acute oral toxicity study in female rats done by up-and-down study methodology resulted in no mortality and a predicted LD50 of >5,000 mg/kg. The 14-day repeated dose rat study showed no effects up to 5,000 mg/kg/d. There were no confirmed test substance-related mortalities during the 90-day repeated dose study. The oral administration of GC resulted in increased salivation and hypoactivity in the high dose and controls of both males and females. Reductions in body weight and food efficiency occurred in males of all treated groups and caffeine. There were also changes in hematology, clinical chemistry, and urinary parameters in both male and female GC-treated groups. These findings were also present in the caffeine controls and are most likely attributed to the pharmacologic effects of caffeine present in GC. Organ weight changes attributed to GC administration included reductions in the gonadal and retroperitoneal fat pads at all treatment levels; however, these effects were also associated with caffeine administration. Although there were no macroscopic changes noted, microscopic findings were reported in both males and females as acinar hypertrophy of the salivary glands of all treated groups and the caffeine controls and were considered related to the caffeine content in the GC. These effects in salivary glands are typical of caffeine exposure and have been noted in previous caffeine studies, and they are likely adaptive changes that would be reversible if a recovery period would have been used once exposure had been terminated. Certain effects were observed at all doses (decreases in fat pad weight, salivary gland hypertrophy, serum cholesterol, adrenal cortex hypertrophy, and eosinophils counts). All these effects have been seen in other studies with caffeine or were found to occur in the caffeine control group. Based on the review of data on caffeine in the available literature compiled over the past 50 years, the types and degree of effects observed in this study are likely due to caffeine. Based on this review and the similar percentage incidence and magnitude of effects seen at 5,000 mg GC kg/d compared with effects observed with 150 mg caffeine/kg/d and the paucity of any other effects in the GC groups that were not seen with caffeine, we conclude that there were no incremental toxicological effects attributable to GC at these levels in Sprague Dawley rats.
Based on the results of the studies conducted on GC and information on the documented historical consumption of guayusa tea, there is no evidence of any general or genetic toxicity nor is there evidence of any concern to consumers of guayusa tea or beverages formulated with GC provided their total caffeine consumption does not exceed 400 mg/d as recommended in several reviews including the 2015 EFSA Opinion on Caffeine 30 and the 2015 DGAC Report to the USDA. 29
Footnotes
Author Contributions
Kapp contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised the manuscript. Kraska and McQuate contributed to conception and design; contributed to acquisition, analysis, and interpretation; and critically revised the manuscript. Mendes and Roy contributed to analysis and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Robert Kapp, Richard Kraska, and Robert McQuate are independent consultants to Runa, LLC. Odete Mendes is an employee of Product Safety Labs. Shambhu Roy is an employee of BioReliance Corporation.
Funding
The authors disclosed receipt of direct or indirect financial support for the research, authorship, and/or publication of this article: Supported by Runa LLC (a privately held company that specializes in the organic beverage market and processes and sells guayusa). The company is based in Brooklyn, New York, with offices in Quito and Archidona, Ecuador.
