Abstract
The current study was aimed at exploring the protective efficacy of spirulina against the hemato-biochemical alterations and nephrotoxicity induced by lead (Pb). Female rats aged 12 weeks were treated for 4 weeks with Pb (0.344 g kg−1 bw) associated or not with spirulina (5.3 g kg−1 bw).
Renal damage induced by Pb was related to a severe anemia, increases of oxidative stress-related parameters (thiobarbituric acid reactive substances (TBARS) (+29%), protein carbonyl (PCO) (+66.3%), and advanced oxidation protein product (AOPP) (+110%)), plasma lactate dehydrogenase (LDH) (+80%), creatinine and urea levels in plasma, and uric acid concentration in urine, as well as genotoxic changes (+89.3% and +60% for DNA and mRNA levels, respectively). Conversely, LDH and antioxidant enzyme activities in kidney were decreased, as well as the levels of plasma uric acid, and urinary creatinine and urea levels. Spirulina-supplemented rats exhibited normal peripheral blood and renal parameters and renal histology. It can be suggested that Arthrospira platensis alleviates damages induced by Pb, thanks to its high phenolic content and antioxidant capacity.
Introduction
Lead (Pb) is one of the most ubiquitous environmental toxins. It has long been viewed as a risk factor for human health, accounting for 0.6% of the global disease burden in the year 2010. 1 Exposure to Pb produces broadly deleterious outcomes to organism function including hematopoietic systems. 2 In fact, this metal enters into the body by ingestion, inhalation, or through the skin. In low concentrations over a long period, it mainly attacks the hematopoietic system, thus causing alteration of hematological parameters. 3,4 Meanwhile, Pb causes renal damage, especially associated to the anemia’s mechanism, 5 leading to proximal tubular dysfunction or irreversible nephropathy depending on the exposure type. 6
Production of reactive oxygen or nitrogen species and resulting oxidative stress have been reported as the main cause of nephrotoxicity. 7 Although the exact mechanism of Pb biotransformation is not completely understood, numerous experimental data have shown that chronic Pb exposure is known to disrupt the pro-oxidant/antioxidant balance existing in the mammalian cells, through a generation of highly reactive species. 8 In turn, production of reactive oxygen species (ROS) causes alterations in subcellular structures including proteins, DNA, and membrane lipids, and changes in cellular antioxidant system. 9,10
To overcome oxidative stress, some pharmaceutical drugs are currently available for the treatment of kidney disease but with either unreliable efficacies or undesirable side effects. In recent years, there has been an increased interest among phytotherapy researchers to discover novel natural products offering alternative health care options that are more effective and safer against drug and xenobiotics’ toxicity. 11
Spirulina platensis (Arthrospira platensis, Phormidiaceae) is a blue–green alga/cyanobacterium widely used as a nutraceutical and pharmaceutical agent. 12 It has become an interesting subject for many investigations conducted either in vitro and/or in vivo since it is a valuable food source of antioxidants with high-quality proteins, amino acids, vitamins (A, B1, B2, B3, B6, C, E, and K and folate), beta-carotene and other pigments, phenolic acids, minerals (magnesium, zinc, manganese, and selenium) as well as macro- and micronutrients, including unsaturated fatty acids, polysaccharides, and carbohydrates. 12,13 Other findings showed that it is nontoxic, inexpensive, almost without side effects compared to synthetic products, 14 and it is used as a bioactive feed additive. 15 Due to its antioxidant properties, spirulina has been shown to have protective effects against drugs or chemicals-induced cardiotoxicity, hepatoxicity, and nephrotoxicity. 14,16 –18
In view of the above findings, we aimed to assess the protective effects of co-administration of spirulina against sub-acute Pb-induced oxidative damages by evaluating DNA and mRNA levels, alterations in hematological and serum biochemical markers, lipid and protein oxidations, enzyme and non-enzyme antioxidants, as well as histopathological changes related to renal injuries in female rats.
Materials and methods
Compounds and chemicals
Lead-acetate ((CH3COO)2Pb, 3H2O; 99.6% purity) was purchased from SD Fine Chemicals (Mumbai, Maharashtra, India). It was dissolved in sterilized distilled water at a 3% final concentration.
Spirulina was collected in September 2016 from El Alia, a marine bio-algae farm in Mahdia (Tunisia). It was identified by Professor Yousef Krichen (National Agronomic Institute of Tunisia), and one sample was recorded in the herbarium at the Faculty of Pharmacy, Monastir (national list code no. ASP-53). Spirulina blue–green powder was first subjected to heavy metal analyses for Pb, mercury, cadmium, and arsenic using atomic absorption spectrometer to ascertain its safety.
5,5-Dithiobis (2-nitrobenzoic acid) (DTNB),
Pigment composition and antioxidant activity of S. platensis
Measurement of chlorophyll and carotenoid contents
Lipid-soluble pigments such as chlorophyll a, chlorophyll b, and carotenoids were measured in acetone extract of spirulina using a UV-vis spectrophotometer through absorbance at 665.2, 652.4, and 470 nm. Each pigment concentration was then calculated according to the method of Lichtenthaler. 19
Determination of antioxidant activity using β -carotene/linoleic acid assay
Algal extract capacity to inhibit the β-carotene bleaching was estimated according to the method of Koleva et al. 20 slightly modified. The microtiter plates were incubated at 50°C for 120 min, and the absorbance was measured at 470 nm. Readings of all samples were performed immediately and after 120 min of incubation. The antioxidant activity of the extracts was evaluated using the following formula:
where C 0 and C120 are the absorbance values of the control at 0 and 120 min, respectively and S is the sample absorbance at 120 min. The results were expressed as IC50 values in μg mL−1.
Study of the effect of S. platensis in vivo
Feed composition and preparation
Commercial feed provided to the rats consisted of pellets containing a mixture of wheat, alfalfa, soybean, vitamins, and minerals purchased from SNA (Sfax, Tunisia). Alternatively, a diet enriched with 5% spirulina was prepared by mixing plant powder with food pellets in distilled water to obtain a homogenous paste. New pellets were formed and dried at 60°C for 24 h before starting the experiment. This dose was selected on the basis of previous studies, which did not reveal any toxic effects in adult rat, and was effective against toxicity. 21
Ethical approval
The experimental procedures in this study were performed according to the National Institutes of Health Guidelines for Animal Care and approved by the Ethical Committee of Sfax Science Faculty. 22 All animal procedures were conducted in strict conformity with the “Institute ethical committee guidelines” for the care and use of laboratory animals.
Animals and handling
This study was carried out on 24 Wistar female rats, weighing 190 ± 2 g (aged 11–12 weeks), obtained from the Central Pharmacy of Tunis (SIPHAT). The animals were kept in germ-free conditions with free access to standard feed and tap water for 1 week to adapt to the laboratory conditions (21 ± 1°C, 10–h light and 14-h dark cycle, and 40% of humidity) and they were accommodated before being used in the experiments.
The animals were divided into three groups of eight rats each according to their average body weight: First group (C): rats received ad libitum distilled water and normal diet. Second group (Pb): rats were treated through drinking water containing 6 g L−1 Pb acetate, resulting in an average uptake of 343.6 mg Pb kg−
1 body weight day−
1, and a normal diet. Third group (S + Pb): rats were given daily a diet enriched with 5% spirulina (5.3 g kg−1 body weight), and 6 g L−1 Pb acetate through drinking water.
Death was not observed in any experimental group during the whole treatment period (30 days). 17 Figure 1 shows a schematic diagram for the design of the experiment.

Schematic overview of experimental procedures.
The daily food intake was calculated from the difference between the weight of food that remained in the bin (D 1) and the amount placed the day before (D 2) (Table 1).
Antioxidant activity and amounts of chlorophylls and carotenoids in the methanol extract of spirulina.a

DW: distilled water.
a Values represent the means of three replicates ± SE of six samples.
b Antioxidant activity (β-carotene bleaching inhibition) (IC50, µg mL−1).
c Chlorophyll a (µg g−1 DW).
d Chlorophyll b (µg g−1 DW).
e Chlorophyll a + b (µg g−1 DW).
f Carotenoids (µg g−1 DW).
Necropsy and routine blood test
At the end of the experiment and to avoid inter-assay variations, all animals were taken one at a time from each of the three groups, euthanized and subjected to the following examinations:
Blood preparation
First blood sample was withdrawn from brachial artery in tubes without heparin and centrifuged (2500 r min−1 for 15 min) for the efficient separation and recovery of serum. A second whole blood sample was collected in a tube with the anticoagulant ethylenediaminetetraacetic acid, then used for estimation of hematological parameters including the erythrogram (white blood cell (WBC), red blood cells (RBC), packed cell volume (PCV), and hemoglobin (HGB)) and the RBC indices (mean corpuscular hemoglobin concentration (MCHC), mean corpuscular hemoglobin (MCH), and mean corpuscular volume (MCV)). 23
Tissue relative weights
During the period of treatment, the body weight of the rats was measured daily in each group (Table 2). The kidneys were weighed immediately after euthanasia on analytical balance, and relative kidney weight was calculated as follows: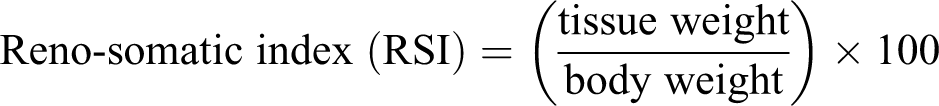
Body and kidney weights, reno-somatic index, daily food, and water consumption by adult rats treated or not with lead, and/or spirulina during 30 days.a

C: Control rats; Pb: rats treated with lead; (S + Pb): rats treated with spirulina and intoxicated with lead; SD: standard deviation; FLSD: Fisher’s protected least significant difference; ANOVA: analysis of variance.
a Values are expressed as mean ± SD of eight rats in each group. One-way ANOVA followed by FLSD as a post hoc test for comparison between groups.
b p < 0.05: comparison between (Pb) and (C) groups.
c p < 0.05: comparison between (Pb + S) and (Pb) groups.
d p < 0.01: comparison between Pb and C groups.
e p < 0.01: comparison between (Pb + S) and (Pb) groups.
f p < 0.001: comparison between Pb and C groups.
g p < 0.001: comparison between (Pb + S) and (Pb) groups.
Preparation of kidney homogenates
The renal tissue (1 g) was minced and homogenized in 2 mL of ice-cold buffered saline (Tris-HCl, pH 7.4). The homogenates were, then, centrifuged at 9000 r min−1 (4°C, 25 min) to remove any remaining cell debris and stored in aliquots at −80°C until use for various biochemical assays. All samples were analyzed in triplicate.
Biochemical investigations in kidney
Plasma biochemical parameter analysis
All biochemical analyses were carried out using commercial kits according to the manufacturer’s protocol. The levels of urea, uric acid, and creatinine in serum or urine were estimated using commercial reagent kits (Ref. 20151, 20149, and 20090, respectively, Biomaghreb, Tunisia).
Creatinine clearance was calculated as V × U/P, where V is the urinary volume collected within 24 h, and U and P are the urinary and the plasma creatinine levels, respectively. 24 Samples were centrifuged at 5000 r min−1 for 5 min to eliminate the probable presence of excrements.
LDH activities in the kidney and serum were measured using a commercially available diagnostic kit (Ref. 200126, Biomaghreb, Tunisia).
Oxidative damage assays
Total protein level in renal tissues was evaluated according to Lowry et al. 25 using bovine γ-globulin as standard. The level of lipid peroxide in kidney was estimated by measuring the formation of TBARS according to the method of Yagi. 26 PCO content in kidney tissue was measured using 2,4-dinitrophenylhydrazine according to the method by Reznick and Packer. 27 AOPP levels were determined according to the method of Witko et al. 28
Assay of enzyme and non-enzyme antioxidants
Superoxide dismutase (SOD) activity was estimated according to Beauchamp and Fridovich. 29 Catalase (CAT) activity was assayed by hydrogen peroxide consumption following Aebi method. 30 Glutathione peroxidase (GPx) activity was measured according to the method of Flohe and Gunzler. 31 Reduced glutathione (GSH) in plasma was determined by the method of Ellman 32 modified by Jollow et al., 33 based on the development of a yellow color when DTNB was added to compounds containing sulfhydryl groups. Non protein thiol (NPSH) levels were determined according to Ellman 32 and the results were expressed as µmol g−1 tissue.
DNA and RNA evaluations
Total DNA and mRNA were isolated from 100 mg of kidney tissue according to the method of Chomczynski and Sacchi. 34 Each sample was measured at a wavelength of 260 nm and a total DNA content was expressed in μg g−1 of tissue. 35
Histological processing
Some kidney samples, intended for histological examination by light microscopy, was fixed in Bouin solution (30% formaldehyde, 70% saturated picric acid, and 5% glacial acetic acid) for 24 h and lately transferred to 70% alcohol. The pieces were dehydrated through a series of ascending ethanol percentage. Tissues were then embedded in paraffin, serially cut, and stained with hematoxylin–eosin (H-E). 36 Pathological alterations in the kidney were scored according to the following scheme: 0, no damage; +1, mild injury; +2, moderate injury, +3, severe degree of tubular and glomerular injury and necrosis.
Data analyses
Data were analysed using the statistical package program Stat Graphics plus 5.1 (Stats graphics). Statistical analysis was performed using one-way analysis of variance, followed by Fisher’s protected least significant difference test as a post hoc test for the sake of comparing groups [treated groups (Pb) or (S + Pb) vs. (C)], [(S+Pb) vs. (Pb)]. All values were expressed as mean ± SD. Differences were considered significant at various levels (p < 0.05, p < 0.01, and p < 0.001). Pearson’s correlation coefficients were determined using IBMSPSS (version 21; IBM Corp., Armonk, New York, USA) to estimate the statistical relationships between hematological parameters and renal biomarkers.
Results
Pigment composition and antioxidant capacity of S. platensis
Amounts of pigments
The contents of chlorophyll a, chlorophyll b, and lycopene in spirulina extract are shown in Table 1. Chlorophyll a level (264.1 µg g−1 DW) was far higher than those of chlorophyll b and lycopene (23.0 and 36.6 µg g−1 DW, respectively).
Antioxidant activity
The antioxidant assay using β-carotene bleaching inhibition is widely used to measure the antioxidant activity of extracts or natural compounds. In this assay, linoleic acid oxidation produces hydroperoxyl radicals, which cause β-carotene bleaching. The antioxidant activity of spirulina extract is presented in Table 1. The oxidation of β-carotene was effectively inhibited by the methanolic extract of S. platensis with IC50 value of 4.01 μg mL−1.
Effect of S. platensis on food and water intakes
Amounts of food and water consumed by adult female rats in control and tested groups are gathered in Table 2. After 30 days of treatment, Pb exposure of rats caused a significant decrease in the daily food intake (−45.66%) but an increase in water intake (+32.37%) (p < 0.01), respectively. Co-treatment with spirulina improved food and water consumptions by the rats, which reached normal values.
Effect of S. platensis on body and kidney weights and reno-somatic indices
Death was not observed during the experimental period. As presented in Table 2, our results showed a significant decrease in the body weight (−25%), but there was an increase in absolute and relative kidney weight by 48.31% (p < 0.01) and 100% (p < 0.001), respectively, when compared with those of controls. When spirulina was supplemented to Pb-treated rats, a partial recovery in body weight and absolute kidney weight was found, compared to the Pb group. Similarly, the reno-somatic index reached normal values (Table 2).
Effect of S. platensis on hematological parameters
To investigate the consequences of spirulina supplementation on Pb toxicity, we first assessed the effects of Pb acetate on blood system in rats. Pb-exposed rats exhibited a significant decrease in RBCs, HGB, PCV, MCV, MCHC, and MCH erythrocyte counts (−24.1%, −39.7%, −33.5%, −12.5%, −9.2%, and −20.7%, respectively) compared to the healthy control animals (Table 3). Conversely, leukocyte parameter like WBC count was significantly increased (+25.8%, p < 0.01). The changes in blood indices shown in Pb-treated group were reverted to near normal levels upon co-treatment with spirulina (p < 0.05).
Effects of Pb and/or spirulina treatment for 30 days on erythrocyte and leukocyte parameters in adult Wistar rats.a

Pb: lead; RBC: red blood cells counts; HGB: hemoglobin concentration; PCV: packed cell volume; MCV: mean corpuscular volume; MCHC: mean corpuscular hemoglobin concentration; MCH: mean corpuscular hemoglobin; WBC: white blood cells counts; C: control rats; Pb: treated rat with lead; S + Pb: rat treated with spirulina and intoxicated with lead; SD: standard deviation; FLSD: Fisher’s protected least significant difference; ANOVA: analysis of variance.
a Values are expressed as means ± SD for eights rats in each group. One-way ANOVA followed by FLSD as a post hoc test for comparison between groups.
b p < 0.01: comparison between (Pb) and (C) groups.
c p < 0.01: comparison between (S + Pb) and (Pb) groups.
d p < 0.001: comparison between (S + Pb) and (Pb) groups.
e p < 0.001: comparison between (Pb) and (C) groups.
f p < 0.05: comparison between (Pb) and (C) groups.
g p < 0.001: comparison between (S + Pb) and (Pb) groups.
Effect of S. platensis on biochemical markers of kidney function
Urine output
Urinary excretion during 24 h was lowered by 22% (p < 0.01) in the Pb group as compared to controls. Co-treatment with spirulina improved this parameter, whereas not reaching normal values (Table 4).
Coefficient correlations among the RBC’s, HGB, WBC, and renal markers (creatinine and urea).a

C: control rats; Pb: treated rat with lead; (S + Pb): rat treated with spirulina and intoxicated with lead; BUN: blood urea nitrogen; SD: standard deviation; FLSD: Fisher’s protected least significant difference; ANOVA: analysis of variance.
a Values are expressed as mean ± SD for eights rats in each group. One-way ANOVA followed by FLSD as a post hoc test for comparison between groups.
b p < 0.01: comparison between Pb and C group.
c p < 0.05: comparison between (Pb + S) and (Pb) groups.
d p < 0.05: comparison between Pb and C group.
e p < 0.01: comparison between (Pb + S) and (Pb) groups.
f p < 0.001: comparison between Pb and C group.
g p < 0.001: comparison between (Pb + S) and (Pb) groups.
h BUN was calculated using the formula conversion: BUN (mmol L−1) = [urea (mmol L−1)]/2.14.
Creatinine levels in plasma and urine
Our results showed a constellation of disorders in the renal function of the Pb-treated group illustrated by changes of creatinine levels on plasma and urine (Table 4). In fact, creatinine levels in Pb-treated female rats were higher in plasma (+15%, p < 0.05) and lower in urine (−57.9%, p < 0.001) than those in control. Likewise, a significant reduction in creatinine clearance, an indicator of glomerular dysfunction, was observed in the Pb-treated female rats (−75%, p < 0.001). Co-administration of spirulina caused a marked improvement of the parameters cited above.
Uric acid levels in plasma and urine
Results indicated that uric acid levels of the Pb-treated group were lower in the plasma (−36%, p < 0.01) but higher in urine (+28.8%, p < 0.01) than in controls (Table 4). This nitrogenous waste product (uric acid) was restored in spirulina supplemented animals compared to the Pb group (Table 4). Moreover, supplementation of the diet of Pb-intoxicated female rats with spirulina significantly decreased blood urea nitrogen (BUN) levels (−38.8%, p < 0.01) (Table 4).
Urea levels in plasma and urine
Other kidney specific biomarkers were analyzed. Our results showed that urea levels of Pb-treated rats were significantly higher in plasma (+71.5%) and lower in urine (−52%) compared to those of controls (Table 4). However, dietary spirulina supplementation to the Pb-poisoned rats markedly improved urea levels to near normal values (Table 4).
Kidney and plasma LDH activities
Our results showed that a 12-week treatment with Pb induced a significant increase in plasma LDH activity (+80%, p < 0.001), while it decreased in the kidney (−63.5%, p < 0.001) (Figure 2). Co-administration with spirulina improved significantly LDH activities in plasma and kidney compared to those of Pb treated rats (Figure 2).

Effect of Pb, or S + Pb on plasma (a) and kidney (b) LDH activities of females rats, during 30 days. Data are means ± SD of eight rats per group. Significant differences between groups are mentioned as follows. Comparison between (Pb) vs. control (C) group: *p < 0.05; **p < 0.01; and ***p < 0.001. Comparison between (Pb + S) group vs. lead (Pb): + p < 0.05; ++ p < 0.01; and +++ p < 0.001. Pb: lead; S: spirulina; LDH: lactate dehydrogenase; SD: standard deviation.
Relationships between hematological parameters (RBC, HGB, WBC) and renal biomarkers (urea, creatinine)
Table 5 lists the independent variables identified with Pearson correlation coefficient (R) between RBC, HGB, and WBC with plasmatic renal biomarkers (creatinine and urea). HGB was conversely correlated with urea, and to a lesser extent to creatinine. Similar trends were found between RBC and renal markers. Likewise, statistical study showed a significant inverse relationship between WBC counts and renal biomarkers, but here it was more significant with creatinine than with urea levels.
Plasma level of bio-indices of kidney functions in adult rats treated with lead (Pb) and/or spirulina 30 days.a

RBC: red blood cells counts; HGB: hemoglobin concentration; WBC: white blood cells counts.
a Values represent the means of three replicates ± SE of six samples.
b Significance correlation p < 0.001.
c Significance correlation p < 0.01.
Effect of S. platensis on lipid peroxidation and protein oxidation levels in the kidney
Markers of lipid oxidative damages
Our findings revealed changes in the levels of lipid peroxidation (LPO) in the kidney of the Pb-treated group as evidenced by the enhanced TBARS levels (+29%, p < 0.001) in renal tissues of female rats when compared to controls (Table 6). These modifications were significantly alleviated following co-administration of spirulina.
Oxidative status and antioxidant system activity in adult rats treated with lead (Pb) and/or spirulina for 30 days.a

TBARS: thiobarbituric acid reactive substances; PCO: protein carbonyl; AOPP: advanced oxidation protein product; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GSH: glutathione, NPSH: non-protein thiol; C: control rats; Pb: treated rat with lead; S + Pb: rat treated with spirulina and intoxicated with lead; SD: standard deviation; FLSD: Fisher’s protected least significant difference; ANOVA: analysis of variance.
a Values are expressed as mean ± SD for eights rats in each group, One-way ANOVA followed by FLSD as a post hoc test for comparison between groups.
b p < 0.001: comparison between Pb and C groups.
c p < 0.001: comparison between (Pb + S) and (Pb) groups.
d p < 0.05: comparison between (Pb + S) and (Pb) groups.
e p < 0.01: comparison between (Pb + S) and (Pb) groups.
f p < 0.01: comparison between Pb and C groups.
g p < 0.05: comparison between Pb and C groups.
Markers of protein oxidative damages
In the Pb group, a significant increase of PCO by 66.3% and AOPP levels by 1.1-fold (p < 0.001) (markers of protein oxidative damages) in the kidney of adult rats was observed when compared to the controls (Table 6). Treatment with spirulina resulted in a marked decrease of kidney PCO and AOPP levels (−18.3% and −46.4%, respectively) (p < 0.01) when compared to the Pb group.
Effect of S. platensis on enzymatic and non-enzymatic antioxidants in the kidney
Enzymatic antioxidant status
Table 6 shows the effect of Pb alone or in combination with spirulina on SOD, CAT, and GPx activities in the kidney homogenates of an adult rat. Statistical analysis showed that exposure to Pb caused a significant decrease in SOD activity (by −53.1%) as compared to control animals. Similar observations concerning CAT and GPx activity (−47.2% and −41%, respectively) were observed following Pb intoxication. Supplementation of rats with spirulina improved these enzyme activities, which reached normal values.
GSH and non-protein thiol levels
In the kidney homogenates of Pb-treated rats, GSH and NPSH values decreased significantly (−26.5% and −43%, respectively) (p < 0.01) when compared to negative controls (Table 6). Supplementation of spirulina ameliorated these antioxidant levels.
Effect of S. platensis on DNA and mRNA levels in the kidney
Figure 3 illustrates the DNA and mRNA level patterns in the kidney of the control and treated groups of female rats. Results showed that Pb induced higher contents of DNA (+89.3%) and mRNA (+60%) in the kidney of rats, compared to control values. Conversely, female rats co-treated with Pb and alga exhibited amelioration in liver DNA and RNA contents when compared to the Pb-treated group.

(a) DNA quantity and (b) RNA quantity in the kidney of control and adult females rats treated with Pb or their combination (S + Pb), during 30 days. Data are mean ± SD of eight rats per group. Significant differences between groups are mentioned as follows. Comparison between (Pb) vs. control (C) group: *p < 0.05;**p < 0.01; and ***p < 0.001. Comparison between (S + Pb) group vs. Pb: + p < 0.05; ++ p < 0.01; and +++ p < 0.001. Pb: lead; S: spirulina; SD: standard deviation.
Effect of S. platensis on histological aspect of the kidney
The light microscopy examination of kidney tissues in the control group showed normal tubules and intact glomeruli (Figure 4(a) and (b)). Kidney damage and high histological score could be observed in female rats treated with Pb, characterized with a vascular congestion of the epithelial cells and infiltration of inflammatory leukocytes (Figure 4(c) and (d)). Glomeruli fragmentation, associated with the loss of brush border membrane and Bowman’s space enlargement, was also observed in the kidney of Pb-treated rats (Figure 4(e) and (f)). This pathological aspect was alleviated by the administration of spirulina to Pb treated group (Figure 4(g) and (h)). The histopathological changes are graded and summarized in Table 7.
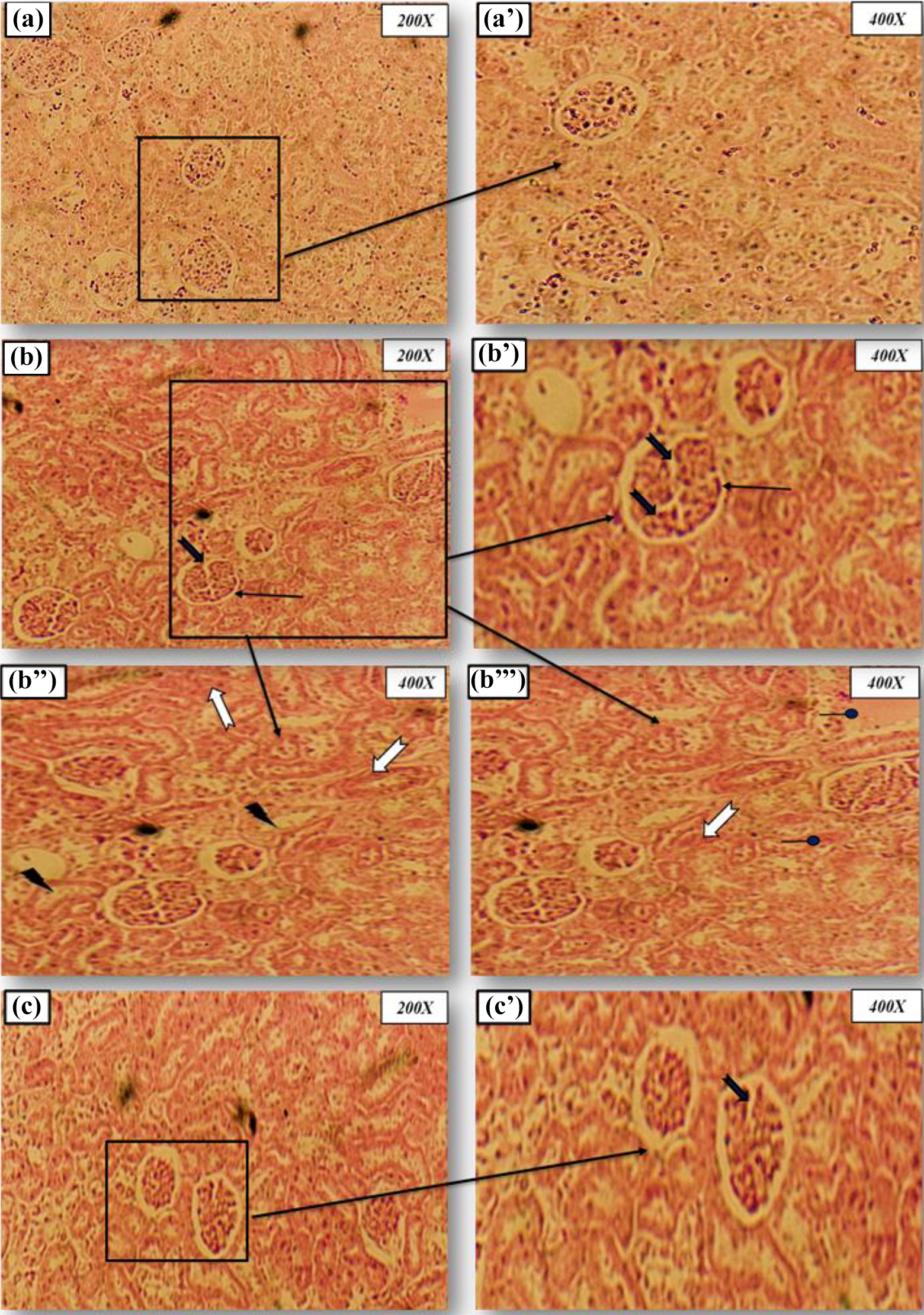
Microscopic observation of females rat kidney were stained with hematoxylin–eosin (magnification: 200× and 400×). (a) and (b): Control group (C) showing normal glomeruli and renal tubules architecture after 30 days of treatment. (c), (d), (e), and (f): Lead-treated group (Pb) showing a severe necrosis in epithelial cells accompanied by glomeruli hypertrophy, glomeruli fragmentation, edema infiltration and a narrowed tubular lumen after 30 days of treatment. (g) and (h): Rat treated with Spirulina plantensis and intoxicated with lead (S + Pb) showing marked improvement in the histological sections with normal glomeruli and tubules after 30 days of treatment. Arrows indicate: Edema infiltration a narrowed tubular lumen glomeruli fragmentation reduced Bowman’s space tubular epithelial cells necrosis. Pb: lead; S: spirulina.
Grading of the histopathological changes in the renal tissues in rat treated just with lead and those treated with lead or/and spirulina after 30 days.a

OI: edema infiltration; NTL: a narrowed tubular lumen; GF: glomeruli fragmentation; RBS: reduced Bowman’s space; TECN: tubular epithelial cells necrosis; C: control rats; Pb: rat treated with lead; S + Pb: rat treated with spirulina and intoxicated with lead.
a Scoring was done as follows: none (0), mild (+1), moderate (+2), and severe (+3).
Discussion
Heavy metal toxicity is one of the oldest environmental problems and serious health concerns today. Abundant literature describes the deleterious effects of Pb toxicity on animals and human. 37 At low concentrations, Pb may cause harmful effects and tends to accumulate in the food chain. 38 Our results, observed in female rats after 30 days of intoxication, indicated that this metal causes a significant decrease in animal growth, as evidenced by the reduced gain of body weight. This could be related to the decrease of food intake during this period. Our results are in good agreement with previous reports, 39 arguing that Pb has a marked effect on the gastrointestinal tract resulting in the mal-absorption of supplements or the decline in dietary intake. In fact, this metal would affect food satiety signals causing premature termination of food intake during a meal. 40 Likewise, some studies reported a decrease in weight that may be due to anorexia, decreased muscle mass, and cachexia because of the oxidative stress-induced by Pb exposure. 40,41 Co-administration of spirulina to the rats significantly minimized the weight loss. This result is likely due to the microalga content in high-quality proteins, vitamins, amino acids, since these nutrients have been shown to enhance energy digestibility with beneficial effects on the organism’s economy. 42
Pb toxicity symptoms are not only accompanied with a loss in body weight, but also with alterations of the physiological hematologic parameters. Accordingly, our results showed that acute Pb intoxication has a direct effect on RBCs, causing hemoglobin oxidation and RBC hemolysis as a consequence of their membrane’s LPO. 43 Besides, Waldron 44 reported increased osmotic and mechanic susceptibilities of RBCs upon Pb toxicity, accompanied by shortened lifespan of RBCs. These changes were accompanied with a significant decrease in hemoglobin content and PCV. 44,45 Furthermore, our data showed a high increase in leukocyte counts observed in Pb-poisoned female rats. This leukocytosis was related to the stress state and its influence on the immune system. These results are in accordance with previous reports in mice 46,47 or neonates 17 treated with Pb. Similar observations were also made in ruminant and non-ruminant animals. 48,49 Conversely, spirulina supplementation normalized the blood parameters cited above, reversing anemia. A number of compounds have been reported to help in the therapy of anemia in vivo. Accordingly, the hematinic effects upon spirulina consumption might be due to its richness in essential amino acids and biologically active substances including vitamins, carotenoids, chlorophylls, and polysaccharides. Some of these substances have been shown to form complexes with metals, thus reducing the severity of anemia and leukocytosis. 50
Kidney is a complex organ consisting of well-defined components that function in a highly coordinated fashion. Several lines of evidence indicate the relationship between xenobiotics, in particular Pb, and damages in kidney cells. This metal causes renal tubular cells dysfunction or irreversible nephropathy depending on the exposure extent. 9 In our study, the exposure of adult rats to Pb during 30 days profoundly affected the absolute kidney weights. Similar results have been found by Soussi et al. 51 after 15 days of Pb treatment. In this respect, the nutraceutical action of Arthrospira (Spirulina) and some of its metabolites against nephrotoxicity are due to their anti-inflammatory and antioxidant properties. 52,53
The changes in body and kidney weights in Pb-treated rats were accompanied with an alteration of renal markers. Accordingly, our results showed an increase of plasma levels in creatinine, urea, and BUN in rats intoxicated with Pb. Metabolites such as creatinine, urea, uric acid in urine and plasma, on the one hand, and BUN, on the other hand, are generally found after cell death. 54 Moreover, the increase of serum creatinine is considered as the marker leading to nephron damage, 51 and increased urea level is usually correlated with renal ischemia. With that respect, our findings indicate that Pb-intoxicated rats suffer from renal ischemia. Another biochemical marker used in the current study, namely uric acid levels in plasma, indicated a dysfunction of kidneys. Uric acid is the end-product of purine catabolism, and it has been shown to reduce oxidative stress by scavenging ROS like peroxynitrite. 55 Furthermore, a body of evidence have shown that the disturbance of urine biochemical markers in Pb-intoxicated animals is a consequence of oxidative stress, and is associated with glomerular and tubule interstitial changes, hyperuricemia, vacuolization, and renal breakdown. 56 Accordingly, our result may result from a response to an increased production of endogenous ROS. Meanwhile, we observed a marked decrease of urinary volume and creatinine clearance upon Pb treatment, which reflects the diagnosis of renal failure. 51,57,58 The overall data obtained on hematologic parameters and biochemical renal markers upon each treatment have finally been faced with linear regression modeling. Thus, some strong negative correlations appeared between renal biomarkers (urea and creatinine levels) and HGB levels or RBC and WBC counts. Such relationships have been previously established upon chronic kidney disease. 50
In the present study, the changes in LDH activity observed upon Pb treatment (rise of plasma and decrease in kidney) are likely the results of the enhancement of renal cell membrane permeability. This was an additional confirmation of filtration barrier damage caused by Pb intoxication and, therefore, of renal physiological alteration. 19 These changes have also been observed recently upon a short acute Pb exposure. 51 On that point again, co-administration of spirulina to the diet of Pb-treated rats during 30 days improved the level of plasma and renal biomarkers mentioned above. This underlines that alga is responsible for the protection of the kidney structure by inhibiting inflammation and free radical action. Such properties are likely to be related to its composition of phycocyanins and phenolic compounds. 56 Thanks to its radical scavenging activity, spirulina may prevent the discharge of cellular contents into the blood stream.
To substantiate our biochemical findings, histological examination of kidney was undertaken. Marked histological alterations could be seen in Pb-treated rats, including tubular necrosis, hypertrophy of glomeruli cells with reduction of Bowman’s space, hemorrhage, swelling of tubules, and vacuolization. Similar features were recently reported in mice upon acute exposure to Pb 59 and they could be the consequences of the Pb-induced accumulation of free radicals due to increased LPO in the renal tissues. Alternatively, Pb tends to accumulate in the kidney, predisposing it to nephrotoxicity. 36 Here again, it is suggested that the mechanism underlying kidney protection by spirulina involves the radical scavenging properties of the alga. In turn, the above described histological changes were confirmed by nucleic acid studies. In fact, DNA constitutes another major target of ROS deleterious actions. In the present study, the kidney DNA and RNA levels dramatically increased as a result of Pb intoxication. These results are consistent with other studies, 60,61 corroborating strong correlations between Pb exposure, kidney DNA damages, ROS levels, and LPO. We recently reported that Pb induces a loss of nucleotide bases in the DNA, thus causing mutagenic lesions, as a consequence of excessive ROS formation. 19 It is also well-known that LPO causes a loss in membrane integrity and membrane damage. 62 TBARS assay, taken as an index of LPO, allowed us to confirm that oxidative stress occurred concomitantly to nephrotoxicity in Pb-exposed rats. 51,63
Moreover, we found that inexorable generation of ROS during Pb exposure may also affect the cellular protein expression profile, including PCO and AOPP levels, leading to a severe failure of their biological functions and even cell death. Such oxidative protein damages might be one of the mechanisms by which Pb-induced nephrotoxicity. Conversely, we showed that supplementation of rats with spirulina alleviates protein oxidation and, as a consequence, kidney injury. These observations are consistent with previous ones reported recently. 19,51 Besides, antioxidant enzyme proteins have been shown to scavenge ROS, thus appearing as the first line of defense system against oxidative stress. 64
In the present study, a prolonged exposure (30 days) to Pb caused a dramatic decrease of SOD, CAT, and GPx enzyme activities, whereas renal tissues underwent oxidative damages. These results confirmed previous observations. 51,64 Interestingly, we also found a marked drop in the renal levels of non-enzymatic antioxidants (e.g. GSH and NPSH) after 1 month of Pb treatment. GSH is a crucial component of the antioxidant defense mechanism that protects cells against exogenous and endogenous toxins. 65 This reduced level of non-enzymatic antioxidants could be the result of their conjugation and/or their consumption upon radical scavenging action. These results confirm previous work on male Wistar rats. 18,51 Conversely, supplementation with spirulina protected the enzymatic and non-enzymatic antioxidants status of female rats exposed to Pb. This is likely related to the antioxidant compounds of the alga (polyphenols, β-carotene, vitamins C and E, etc.), which are efficient radical scavengers. 66 In the present study, spirulina alone did not cause any kidney toxicity as previously described 67,68 and a schematic representation of the protective effect of spirulina against Pb-induced oxidative damage in blood as well as in the kidney may be drawn (Figure 5).

Conclusive graph for the protective effects of Arthrospira platensis on blood cells and kidney of female rat. Defects are caused by lead acetate.
Conclusions
Taking these facts together, the nutraceutical use of spirulina against kidney damages induced by Pb is practicable thanks to its safe consumption and bioactive properties. In particular, its nephroprotective effect is likely due to its bioactive components such as carotenoids, chlorophylls, and other antioxidants reported elsewhere. These have individual or synergistic effects that contribute to the protection or improvement of renal structure and functions. It is also able to ameliorate hematological profile, biochemical parameters, nucleic acids and membrane integrity, and to scavenge a variety of toxic free radical species. Above all, spirulina is the safer natural product, and could therefore be used as a therapeutic agent with nephroprotective potential against any pro-oxidative toxic agent.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Laboratory of Animal Ecophysiology in the Faculty of Sciences of Sfax [Grant No. UR/13 ES-73]. The authors are grateful to Dr Xavier Dauvergne for his help in data analyses and to Naziha Khidiwi, translator and English teacher, for proofreading the manuscript. This work has been funded by Ministry of Higher Education and Research, Scientific Research and Technology, Tunisia.

