Abstract
A battery of toxicological studies was conducted to investigate the genotoxicity and repeated-dose oral toxicity of Bonolive™, a proprietary water-soluble extract of the leaves of the olive tree (Olea europaea L.), in accordance with internationally accepted protocols. There was no evidence of mutagenicity in a bacterial reverse mutation test and in an vitro mammalian chromosomal aberration test nor was any genotoxic activity observed in an in vivo mouse micronucleus test at concentrations up to the limit dose of 2000 mg/kg bw/d. Bonolive™ did not cause mortality or toxic effects in Crl:(WI)BR Wistar rats in a 90-day repeated-dose oral toxicity study at doses of 360, 600, and 1000 mg/kg bw/d. The no observed adverse effect level in the 90-day study was 1000 mg/kg bw/d for both male and female rats, the highest dose tested.
Introduction
Olives are a traditional component of the Mediterranean diet. Olive fruits and olive oil were used in nutrition as early as 6000 BC without any known side effects, indicating a long history of safe consumption of olive polyphenols in humans. 1,2 Olive leaves have also been consumed traditionally by humans for health purposes 3,4 and have been utilized as animal feed in olive-producing regions. 5,6 The majority of olive trees grown for commercial purposes are still found in countries/regions bordering the Mediterranean Sea. 7
In recent years, olive products, including the olive leaves (which have especially high olive polyphenolic levels), have gained additional interest mainly related to their phenolic composition that has been shown to promote different beneficial effects on health. 8 –13 Virgin olive oil contains at least 36 structurally distinct phenolic compounds. 14,15 Approximately 30% of the total phenolic content of virgin olive oil consists of free forms of tyrosol and hydroxytyrosol (the precursor to oleuropein) and their derivatives while other conjugated forms, such as oleuropein and ligstroside aglycone, makeup nearly half. 16 The major phenolic compounds in olive leaves compromise 5 groups of polyphenols: oleuropeosides, flavones, flavonols, flavan-3-ols, and substituted phenols-like hydroxytyrosol and tyrosol. Oleuropein is the most abundant polyphenol found in olive leaves, followed by hydroxytyrosol, luteolin-7-glucoside, apigenin-7-glucoside, and verbascoside. 8,17,18 Olive-derived products are marketed for a range of beneficial health properties and are most closely associated with evidence for a protective role in degenerative diseases (e.g., cardiovascular disease [CVD]). A cause and effect relationship has been shown between consumption of olive oil polyphenols and protection of low-density lipoprotein (LDL) particles from oxidative damage, which has resulted in a health claim accepted by the European Food Safety Authority: Consumption of olive oil polyphenols contributes to the protection of blood lipids from oxidative damage. 19 Moreover, its cholesterol-lowering activity has been shown in human studies. 10,20 –22
In addition to CVD, a link between consumption of olive polyphenols and osteoporosis was proposed because of epidemiological data indicating a lower incidence of osteoporosis in Southern European countries that are known to adhere to the Mediterranean diet, which is rich in olive oil. 23 A mode of action has been suggested via a combination of in vitro and in vivo studies, 23 –27 indicating that the polyphenols oleuropein, hydroxytyrosol, and tyrosol may protect bone homeostasis by promoting bone-building activity while impairing adipocyte differentiation. The bone-sparing effect of olive polyphenols has also recently been shown in 2 independent studies in humans. While one study provided indirect evidence of such effect using a 2-year intervention with a Mediterranean diet enriched with olive oil, 28 a 12-month supplementation with a proprietary water-soluble olive leaf polyphenol extract (Bonolive™) provided more definitive evidence. 29 Indeed, in this trial, a 32% increase was noted in osteocalcin levels, an osteoblast-specific bone formation marker, in treated subjects compared to those in the placebo group. In addition, Bonolive™ significantly lowered serum triglycerides, total cholesterol, and LDL cholesterol and increased high-density lipoprotein cholesterol.
With respect to safety of olive-derived products, the vast majority of available studies show a very good safety profile. For instance, the toxicological profile of an aqueous olive pulp extract containing 6% olive polyphenols was evaluated in a formal, good laboratory practice (GLP)-compliant (i.e., performed in compliance with the principles of GLP) safety assessment. 30 The 90-day repeated-dose toxicity study demonstrated an oral no observed adverse effect level (NOAEL) in rats of 2000 mg/kg bw/d (the highest dose tested), and this dose also did not produce adverse effects in a dose-range reproduction study or a developmental toxicity study. Similarly, in a study in rats, 7 weeks of consumption of up to 1 mL of a 1200 ppm solution of polyphenols from olive fruit and leaves did not induce liver or kidney toxicity, although there were related findings when animals were given the same volume of a 1600 ppm solution. 31 In humans, oral administration of an ethanol extract of olive leaf containing approximately 20% oleuropein at a dose of 1000 mg/d (500 mg twice daily) for 8 weeks did not induce any clinically relevant changes in hematological parameters, electrolytes, or clinical chemistry parameters related to liver and renal function. 10 In addition, no adverse events related to the 12-month daily administration of 250 mg of the olive leaf extract Bonolive™ were observed in study participants. 29
One study reported some potential negative effects of repeated intake of high concentrations of an olive leaf extract on liver function in an experimental study in mice with a duration of 14 weeks. However, the authors failed to describe the extract in any detail (e.g., type of extract and composition) or the absolute exposure to the test article. After oral consumption of the unknown olive leaf extract, changes in hepatic enzymes and hepatic histopathology were observed at the highest doses (0.50% and 0.75% of the diet). 32 At the same time, daily intraperitoneal administration with 100 or 200 mg/kg oleuropein for 3 consecutive days in mice was shown to improve histological and plasma markers during carbon tetrachloride-induced liver damage. 33
The combination of available evidence suggests that olive-derived products, including olive leaf extracts, may have important health-promoting properties, and the majority of studies, both in animals and in humans, suggest an excellent safety profile. However, as the specific composition of such products may influence both the activity and the safety and because of the high variability in methodology of the studies reported in literature as well as some inconsistencies regarding safety, 31 –33 it is difficult to generalize this conclusion. We herein report a comprehensive preclinical GLP safety assessment of Bonolive™, a proprietary standardized water-soluble olive leaf extract that has been clinically studied with regard to bone health. 29
Materials and Methods
Test Article
The test article, Bonolive™, was supplied by BioActor BV (Maastricht, the Netherlands). This commercially available product is a proprietary extract prepared from olive leaves (Olea europaea L.) cultivated mainly in the Andalusia region of Spain and is manufactured by Ferrer HealthTech (Murcia, Spain) in accordance with food grade and food safety standards as embraced by the Global Food Safety Initiative (FSSC 22000). Olive leaves are milled and extracted using a hydroalcoholic medium, followed by a proprietary process involving multiple extraction and purification (using water and alcohol) steps. Finally, the concentrated extract is vacuum dried to obtain the final product as a powder.
Bonolive™ is a brown–yellowish powder with a high polyphenol content. It is standardized to 40% of olive polyphenols, determined by normal phase liquid chromatography using a commercial standard (Extrasynthese; Genay, France) and a mixture of acetic acid and methanol as the mobile phase. Typical polyphenol analyses of Bonolive™ show that the majority consists of oleuropein, while the following make up a very low to trace percentage of the total: oleoside, hydroxytyrosol, oleoside-11-methyl ester, verbascoside, luteolin-7-glucoside, hydroxytyrosol glucoside, demethyloleuropein, rutin, apigenin-7-glucoside, p-HPEA-EA (ligstroside-aglycone mono-aldehyde), 3,4-DHPEA-EA (oleuropein-aglycone mono-aldehyde), and luteolin. The remainder of the ingredient consists of other major and minor components of olive leaves, such as carbohydrates, proteins, and minerals. Bonolive™ is soluble in water at room temperature.
Under conditions of commercial packaging, the polyphenol concentration of Bonolive™ powder has been shown to be stable up to 60 months (stability testing for percentage olive polyphenols and loss on drying was performed annually on a single batch of the product, which met these specifications through 5 years of testing). Two batches of the test article powder were utilized in the current investigation (03K082 for the 90-day study and 060-L19-1 for the remaining studies). Both batches were analyzed after manufacturing and met all commercial specifications, including standardization to 40% olive polyphenols, 8% maximum loss on drying, 9% maximum residue by calcination and commercial limits for microbial counts and heavy metals, and were used within 1 year of their manufacturing date (which, as discussed above, has been demonstrated stable for at least 5 years). Test solutions were made up daily by careful weight measurement just prior to administration in the current studies.
In Vitro Studies
Bacterial reverse mutation test
The Ames test was conducted to investigate the mutagenic potential of Bonolive™ using Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537 and Escherichia coli strain WP2 uvrA (Moltox, Inc, Boone, North Carolina). Experiments were conducted in the presence and absence of a postmitochondrial supernatant (S9 fraction manufactured by Moltox, Inc, Boone, North Carolina) utilized for metabolic activation. The study was performed following methods previously described by Ames et al, 34 Maron and Ames, 35 Kier et al, 36 and Venitt and Parry, 37 and according to Organization for Economic Co-operation and Development (OECD) Guideline No. 471, 38 Environmental Protection Agency (EPA) Guideline Office of Prevention, Pesticides and Toxic Substances (OPPTS) 870.5100, EC No. 440/2008, 39 and International Conference on Harmonisation (ICH) Guidance S2(R1). 40
A preliminary solubility test was conducted by examining test article mixtures of varying concentrations suspended in ultrapure water (prepared in the laboratory using a Direct-Q 5 water purification system), resulting in an opalescent suspension of the test article at concentrations of 10 to 100 mg/mL and a clear solution in the top agar solution at these concentrations. This was followed by a preliminary range-finding test utilizing the plate incorporation method and conducted in triplicate. Six concentrations, 51.2, 128, 320, 800, 2000, and 5000 µg/plate, were selected for the initial and confirmatory tests based on the preliminary test results. Positive controls for the experiments without metabolic activation were 4-nitro-1,2-phenylene-diamine (4 µg/plate) for TA98, sodium azide (2 µg/plate) for TA100 and TA1535, 9-aminoacridine (50 µg/plate) for TA1537, and methyl-methanesulfonate (2 µg/plate) for WP2. The positive control for experiments with metabolic activation was 2-aminoanthracene (2 µg/plate and 50 µg/plate for all S. typhimurium strains and the E. coli WP2 uvrA strain, respectively).
A standard plate incorporation procedure was used for the initial mutation test. Tester strains were exposed to the test article at each concentration and positive and negative controls, both with and without metabolic activation, and plates were incubated for 48 hours at 37°C. The confirmatory mutation test was conducted using a 20-minute preincubation procedure prior to plating. Plates were again incubated for 48 hours at 37°C. All experiments were conducted in triplicate.
Colony numbers were determined by manual counting, and mean values, standard deviations, and mutation rates were calculated. A result was considered positive if a dose-related increase in revertant colonies occurred and/or a reproducible biologically relevant positive response for at least 1 dose group occurred in at least 1 strain with or without metabolic activation. Biologically relevant increases were considered twice that of negative controls for strain TA100 and 3 times that of negative controls for all other strains.
In vitro mammalian chromosomal aberration test
The in vitro mammalian chromosomal aberration test was performed to determine whether Bonolive™ or its metabolites could induce structural chromosomal aberrations. It was performed in compliance with internationally accepted guidelines: OECD 473, 41 EC No. 440/2008, 39 and US EPA OPPTS 870.5375. 42
Dulbecco’s Modified Eagle (DME; Sigma-Aldrich, Germany) medium was used as the solvent for the test article due to its compatibility with the test system (V79 male Chinese hamster lung cells) and the availability of historical control data. Because a clear solution was obtained up to the concentration of 50 mg/mL Bonolive™ in DME, this concentration was prepared as the stock solution, and test solutions were freshly prepared by diluting with DME to achieve the desired final concentrations. The DME medium also served as the negative control. The positive control for use without metabolic activation was prepared by dissolving ethyl methanesulfonate (EMS) in DME at final concentrations of 0.4 and 1.0 µL/mL. The positive control for use in studies with metabolic activation was prepared by dissolving cyclophosphamide monohydrate in DME to achieve a final concentration of 5.0 µg/mL.
The V79 male Chinese hamster lung cell line was purchased from the European Collection of Cell Cultures (United Kingdom) and grown in supplemented DME medium. A cytotoxicity assay was conducted as a pretest for the purpose of selecting concentrations for the main test. V79 cultures, 1 to 3 days old, were trypsinized, suspended in DME medium, plated (5 × 105 cells/group), and incubated for 24 hours. The cells were then treated with increasing concentrations of the test article in the absence or presence of S9 metabolic activation medium (Moltox, Inc, Boone, North Carolina) and were incubated at 37°C for 3 hours before washing with DME medium and covering with DME containing 10% fetal bovine serum (Sigma-Aldrich Co). After 20 hours (1.5 cell cycles), counts were performed to determine toxicity. Additional groups of cells were treated for 20 hours without or 3 hours with metabolic activation, respectively. These cells were counted after 20 hours without or after 28 hours (2 cell cycles) with or without metabolic activation. Toxicity was determined as percentage of survival in the treatment groups compared to the negative (solvent) control.
Two independent experiments were conducted in the main test. Concentrations were selected based on cytotoxicity parameters per OECD 473 guidelines. In experiment A, V79 cultures (5 × 105 cells/group) were exposed to the negative control or each test article concentration for a 3-hour period with (250, 500, 750, and 1000 µg/mL) and without (250, 500, 750, 1000, and 1250 µg/mL) metabolic activation. Groups of cells were also exposed to the respective positive controls for use with and without metabolic activation. Following the exposure period, the cells were washed with DME medium containing 5% fetal bovine serum, and growth medium was added. Sampling was made 20 hours following the start of treatment. All individual test article and negative and positive control experiments were carried out in duplicate, and concurrent measures of cytotoxicity were also set up and assessed as described for the pretest.
Experiment B was conducted as described for experiment A except that the exposure period without metabolic activation was 20 hours (while exposure with metabolic activation remained 3 hours), and sampling was made after 20 hours for groups treated without metabolic activation and after 28 hours (to cover the potential for mitotic delay) for groups treated both with and without metabolic activation. The test-article concentrations were also slightly different compared to experiment A (62.5, 125, 250, and 500 µg/mL for treatment without and 500, 750, 1000, 1250, and 1500 µg/mL for treatment with S9 metabolic activation).
Chromosomes were prepared for analysis by treatment with colchicine (Sigma-Aldrich Co; 0.2 µg/mL) for 2 to 3 hours followed by harvesting, swelling with 0.075 mmol/L KCl, and washing in fixative for approximately 10 minutes before preparing slides, air drying, and staining with 5% Giemsa (Merck & Co, Inc). Two hundred metaphase cells from each experimental group, containing 22 ± 2 centromeres, were evaluated for structural aberrations (slides were coded and scored blind). Chromatid and chromosome type aberrations (gaps, deletions, breaks, and exchanges) were recorded separately. Polyploid and endoreduplicated cells were also scored. Nomenclature and classification of chromosomal aberrations were based on publications by International System for Human Cytogenetic Nomenclature 43 and Savage. 44
Fisher exact and CHI2 tests were utilized for statistical assessments. The test article was considered nonclastogenic if: the number of metaphases with aberrations in all evaluated dose groups was within the range of the laboratory’s historical control data and/or there were no statistically significant increases in the number of metaphases with aberrations compared to the negative control.
The test article was considered clastogenic if: an increased frequency, above the historical control range, of metaphases with aberrations was observed at one or more test concentration; the increase was reproducible between replicate cultures and between tests; and the increase was statistically significant compared to the negative control.
Animal Studies
In vivo mouse micronucleus test
The in vivo mouse micronucleus test was conducted to investigate the genotoxic potential of Bonolive™. The study was conducted in compliance with OECD 474, 45 EC No. 440/2008, 39 and US EPA OPPTS 870.5395. 46 The study was permitted by the Animal Care and Use Committee of Toxi-coop ZRT.
SPF Crl: NMRI BR mice, aged 7 weeks and with body weights of 28.8 to 31.8 g, were utilized for the study. They were acclimatized for 6 days and housed 2 animals per cage in the pretest and 5 to 7 animals per cage in the main test. Housing conditions were 22°C ± 3°C, 30% to 70% relative humidity, and a 12-hour light–dark cycle. All animals received ssniff SR/M-Z+H pellet diet and potable tap water ad libitum.
Humaqua (sterile water, TEVA Pharmaceutical Works Private Ltd, Co, Debrecen, Hungary) was used as the negative control and as the vehicle for administration of the test article and positive control. Test articles were prepared fresh within 2 hours of administration at concentrations of 50, 100, and 200 mg/mL. All treatments were administered at a uniform volume of 10 mL/kg bw.
A non-GLP preliminary toxicity test was conducted to determine the appropriate high dose for the main micronucleus test and whether there were large differences in toxicity between sexes. A single dose of Bonolive™ was administered by gavage to 2 male and female mice at a concentration of 2000 mg/kg bw, and the animals were observed at regular intervals for signs of toxicity and mortality.
Male Crl: NMRI BR mice were randomly divided into 5 groups, including a negative control (n = 10), positive control (n = 5), and 3 test groups. On the basis of the results of the preliminary toxicity test, single oral gavage doses of 500 (n = 5), 1000 (n = 5), and 2000 (n = 10) mg/kg bw were chosen. The positive control, cyclophosphamide 60 mg/kg bw, was administered by intraperitoneal injection. Two extra animals were included in the high-dose group in order to maintain statistical power in case any animals died before the scheduled sacrifices. In the case of no premature deaths, bone marrow slides were not prepared from the extra animals.
All animals were observed immediately after dosing and at regular intervals until sacrifice (by cervical dislocation) for visible signs of reactions to treatment. In the positive control, low- and mid-dose groups, the sacrifices were made once at 24 hours after treatment. In the high-dose and negative control groups, sacrifices were made twice at 24 and 48 hours after treatment (5 animals were used for sampling on each occasion). Bone marrow smears were prepared on standard microscope slides from 2 exposed femurs of the mice from every time point immediately after sacrificing. Two thousand polychromatic erythrocytes (PCEs) per animal were scored blind for percentage of cells with micronuclei. The proportion of immature among total erythrocytes was determined per animal by counting a total of at least 200 immature erythrocytes.
Statistical analysis was performed using Kruskal-Wallis nonparametric analysis of variance (ANOVA) test. Criteria for a positive response were statistically significant (compared to negative controls) increased frequency of micronucleated PCE (MPCE) that were dose-related and exceeded historical control ranges for the laboratory.
90-Day repeated-dose oral toxicity study in rats
The 90-day repeated-dose oral toxicity study in rats was conducted in order to evaluate the possible health hazards, including toxic effects and target organs, of repeated oral exposure to Bonolive™ in male and female rats and to predict a safe consumption level for humans. The GLP study was conducted in compliance with OECD 408 and 47 Food and Drug Administration (FDA) Redbook IV.C.4.a. 48 Care and use of study animals were in compliance with laboratory standard operating procedures under the permission of the laboratory’s Institutional Animal Care and Use Committee as well as in accordance with the National Research Council Guide for Care and Use of Laboratory Animals 49 and in compliance with the principles of the Hungarian Act 2011 CLVIII (modification of Hungarian Act 1998 XXVIII) regulating animal protection.
Healthy CRL:(WI)BR Wistar SPF rats aged 42 to 47 days and weighing 173 to 212 g (males) and 141 to 203 g (females) at the start of the study were randomly divided according to stratification by body weight into 4 groups of 30 animals (15 per sex per dose). Ten animals per sex per group were considered as the main group, while 5 animals per sex per group were considered as the satellite group. Animals were acclimatized for 7 days.
Animals were housed individually in type II polypropylene/polycarbonate cages in a room with 12-hour light–dark cycles, 10 to 15 air exchanges per hour via central air conditioner, a temperature of 22°C ± 3°C, and a relative humidity of 30% to 70%. They received ssniff SM R/M-Z+H complete diet for rats and mice and potable tap water ad libitum.
Bonolive™ was freshly formulated in the vehicle (1% Tween 80 [Sigma-Aldrich Co] prepared in distilled water) at concentrations of 36, 60, and 100 mg/mL. The test item was administered via gavage at doses of 0, 360, 600, and 1000 mg/kg bw/d, at a uniform volume of 10 mL/kg. Each formulation to be administered to the animals was prepared by careful weight measurement just prior to administration each day. The doses were chosen based on data obtained from a previously performed 14-day repeated-dose oral toxicity study (unpublished) and were selected with the aim of inducing toxic effects, if present, but not causing mortality or suffering at the highest dose group, and finding an NOAEL at the lower dose groups.
In the unpublished 14-day study, male CRL:(WI)BR Wistar rats in the 1000 and 2000 mg/kg bw/d groups showed findings of hyaline droplet nephropathy. While this is a common finding in male rats, the fact that the effect appeared dose-dependent was considered reason for further study (see Discussion section). To help determine the significance of this finding, the current study was designed to include a 28-day satellite group for histopathological examination of 5 animals per sex per group with a specific focus on nephropathy, most specifically to observe for the presence of hyaline-like droplets in the epithelial cells of the proximal convoluted tubules, to determine whether the findings of the 14-day study were an artifact or confirm that they were repeatable before continuing with the 90-day study plan. Because of negative results in the satellite groups, the main groups underwent a total of 90 (males) or 91 (females) days of treatment with necropsies performed on the following days. A single additional animal replaced a dead female animal on day 2 in the 1000-mg/kg bw/d group and was treated from that day on.
All animals were observed twice daily for mortality. Dead animals were subjected to gross and histopathological examinations (1 female in the 1000-mg/kg bw/d group on day 2 and 1 male in the 1000 mg/kg bw/d bw group on day 60). General cage-side observations for clinical signs were made twice during the acclimation period and once daily after administration of the test article. Detailed clinical observations were conducted on the day prior to the first treatment and once weekly thereafter. Observations included, but were not limited to, changes in skin, fur, eyes and mucus membranes, occurrence of secretions and excretions, and autonomic activity. Any changes in gait, posture, and response to handling as well as the presence of clonic or tonic movements, sterotypies or bizarre behavior was also observed. A functional observation battery was performed during the final week of the study. Sensory reactivity to stimuli, assessment of grip strength and motor activity, general physical condition, and behavior of animals were tested.
Measurements of body weight were conducted twice during the acclimation period, on the first experimental day prior to treatment, twice weekly during weeks 1 to 4, once per week during weeks 5 to 13, and immediately prior to sacrifice. Food consumption was determined and food efficiency calculated once weekly. Ophthalmological examination was carried out on all animals prior to the experimental period and on control and high-dose group animals at the end of the experimental period.
After an overnight fast (approximately 16 hours) following final administration of the test article, blood samples from both the satellite and main group animals were collected from the retro orbital venous plexus under Isofluran CP anesthesia (CP-Pharma Handelsgesellschaft GmbH, Burgdorf, Germany), after which the animals were euthanized by exsanguination from the abdominal aorta. Blood samples were analyzed for hematologic (white blood cell (WBC), red blood cell, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet, reticulocyte, and WBC differential), blood coagulation (activated partial thromboplastin time and prothrombin time), and clinical chemistry parameters (alanine aminotransferase, aspartate aminotransferase, gamma glutamyl transferase, alkaline phosphatase, total bilirubin, creatinine, urea, glucose, cholesterol, bile acids, inorganic phosphate, calcium, sodium, potassium, chloride, albumin, total protein, and albumin/globulin ratio). Gross pathological examinations and determinations of selected absolute organ weights (liver, kidneys, testes, epididymides, uterus and fallopian tubes, thymus, spleen, brain, heart, adrenals, ovaries, and thyroid/parathyroid) and relative organ weights (compared to body weight and brain weight) were conducted on all animals. Complete histopathological examinations were conducted on the preserved organs and tissues (adrenals, aorta, bone marrow of the femur, cerebrum, cerebellum, pons, medulla, eyes, mammary gland, gonads, heart, kidney, large intestine, liver, lungs, submandibular and mesenteric lymph nodes, quadriceps muscle, esophagus, nasal turbinates, pancreas, pituitary, prostate, submandibular salivary glands, sciatic nerve, seminal vesicle, skin, small intestines, spinal cord at 3 levels, spleen, sternum, stomach, thymus, thyroid and parathyroid, trachea, and urinary bladder) of all animals (satellite and main groups) of the control and high-dose groups. In addition to the high-dose group, the kidneys of male animals in the low- and mid-dose satellite groups were also processed and examined histologically for investigating the possible presence of hyaline-like droplets.
Statistical analysis was done using SPSS PC+ software. The heterogeneity of variance between groups was checked by Bartlett test. Where no significant heterogeneity was detected, a one-way ANOVA was carried out. If the obtained result was positive, Duncan multiple range test was used to assess the significance of intergroup differences. Where significant heterogeneity was found, the normal distribution of data was examined by Kolmogorov-Smirnov test. In case of a nonnormal distribution, the nonparametric method of Kruskal-Wallis one-way ANOVA was used. If there was a positive result, the intergroup comparisons were performed using the Mann-Whitney U test. A P value of <0.05 was considered statistically significant, and statistically significant results were reported at the P < 0.05 and P < 0.01 levels.
Results
Bacterial Reverse Mutation Test
Based on the results of the solubility test and range-finding test, the test item was suspended at a concentration of 100 mg/mL and was tested at concentrations of 51.2, 128, 320, 800, 2000, and 5000 µg/plate in the initial and confirmatory mutation tests.
No substantial increases in revertant colony numbers were observed in any of the strains following treatment with Bonolive™ in the presence or absence of metabolic activation (S9), at any concentration level (Supplemental Tables 1 and 2). Sporadic increases in revertant colony numbers compared to vehicle control were observed in both the experiments, however there was no tendency of dose-related increases beyond the generally acknowledged border of biological relevance in the performed experiments, and mutation rates were well below the threshold of being considered positive.
In conclusion, Bonolive™ was considered nonmutagenic at concentrations up to 5000 µg/plate on bacterial tester strains S. typhimurium TA98, TA100, TA1535, and TA1537 and E. coli WP2 uvrA under the testing conditions of the study.
In Vitro Mammalian Chromosomal Aberration Test
After 3 and/or 20 hours of exposure to the test article with and without S9 metabolic activation, Bonolive™ did not induce an increase in the number of cells with aberrations or rates of polyploidy or endoreduplicated metaphases at any examined concentration (Supplemental Tables 3 and 4). There were no statistically significant differences between treatment and the solvent control groups, and no dose–response relationships were noted.
In the negative control group, the percentage of cells with structural aberrations was ≤5%, confirming the suitability of the V79 cell line used. The concurrent positive controls (EMS and cyclophosphamide) caused the expected biologically relevant increases in cells with structural chromosome aberrations as compared to solvent controls, and thus the study was considered valid.
Bonolive™ did not induce structural chromosome aberrations in V79 Chinese Hamster lung cells under the tested conditions, up to cytotoxic concentrations. Therefore, it was concluded that Bonolive™ is not clastogenic in the applied test system.
In Vivo Mouse Micronucleus Test
No mortality or gender specific effects were observed in the preliminary toxicity test; therefore, the micronucleus test was conducted at the doses described above in males only. On the day of treatment and 24 hours after treatment, a slight decrease in activity and piloerection were observed in the male and female mice treated at the 2000 mg/kg bw dose level, but these signs were not observed 48 hours after treatment.
In the main study, no mortality, clinical signs of toxicity, or adverse reactions to treatment were observed in the positive controls, negative controls, or the 500 and 1000 mg/kg bw dose groups. On the day of treatment, a slight decrease in activity and piloerection was observed in 4 of the 10 male mice treated with 2000 mg/kg bw of Bonolive™. These symptoms were not observed at 24 and 48 hours after treatment. Because there was no mortality, bone marrow slides were not prepared on the 2 extra animals included in the high-dose group, and these animals were symptom free during the study.
No significant differences were observed in frequency of MPCE between the 3 dose groups compared to the negative control (Supplemental Table 5). A large, statistically significant increase in MPCE frequency was observed in the positive control group compared to negative control, demonstrating an acceptable sensitivity of the test. In the 2000 mg/kg bw dose group, the number of PCEs was slightly decreased compared to the negative control group value at the 48-hour sampling time point. This effect demonstrated exposure of the bone marrow to the test item (i.e., the test article reached the target organ) and is not biologically significant.
Overall, Bonolive™ did not show any biologically or statistically significant genotoxic activity at concentrations up to the limit dose of 2000 mg/kg bw in the in vivo mouse micronucleus test.
90-Day Repeated-Dose Oral Toxicity Study in Rats
No test item-related mortality was observed in any of the groups (0/vehicle control, 360, 600, or 1000 mg/kg bw/d) during the treatment period. One male and one female animal in the 1000 mg/kg bw/d groups were found dead on days 60 and 2, respectively. The cause of death in both cases was determined to be the treatment procedure and not administration of the test article. Shortly after treatment and just prior to death, the male animal showed salivation, convulsion, prone positioning, decreased activity, dyspnea, and narrow eye aperture. Gross pathological examination revealed dark red coloring in the lungs and liver, dark color of the right lobe of the thymus, and yellowish fluid content in the thoracic cavity. This concurred with histopathological findings of acute alveolar emphysema accompanied by acute hemorrhage in the lungs and congestion in the liver and thymus. In the female animal, there were no preceding clinical signs or gross pathological findings. However, histopathological examination revealed acute catarrhal pneumonia and serous-fibrinous pleuritis. Both were considered individual findings related to the gavage procedure and were considered unrelated to treatment with the test article. One female animal was added to the study on day 2 to replace the female that died on that day.
As further described below, no toxicologically relevant findings were noted in the satellite animals after 28 days, including no histopathological findings related to the kidneys. Thus the full 90-day study plan was carried out.
Clinical observations, both general and detailed (including the functional observational battery), showed normal behavior and physical conditions of the animals and did not reveal any toxic signs related to administration of the test article in satellite or main group animals. Several commonly occurring and nontreatment-related findings were noted and occurred with low incidence in the control and/or main treatment groups. These included slight salivation in 2 females (mid-dose) after treatment on days 2 and 10, scabs on the neck of 1 male control and a male and female from the mid-dose groups, alopecia on the limbs and neck of several animals in the low- and mid-dose groups, and red crust around an eye of a female control rat. One female animal from the low-dose group showed extreme sound response at the food measurement on days 14 and 28 (jumping, convulsions, twitches, and lateral position) with recovery after a few minutes. As this sign was only seen in a single animal, and not at higher dose-groups, it was considered an individual occurrence and not related to treatment.
There were no test article related toxicologically relevant differences in body weight or body weight gains or in daily food consumption in any of the test groups compared to control animals (see Supplemental Tables 6 and 7). Statistically significant differences with respect to controls were noted for the lower mean body weight gain of female animals in the high-dose main group between days 11 to 14, 28 to 35, and 56 to 63. However, these changes did not result in overall total body weight or weight-gain differences compared to controls; thus, the changes were not considered biologically or toxicologically relevant. Similarly, at several time points, the mean daily food consumption was slightly less compared to controls in female animals of the mid-dose main group (week 9) and the high-dose main group (weeks 2, 9, 11, and 12; Supplemental Table 8). Several sporadic statistically significant changes in feed efficiency compared to controls occurred (slightly less than controls in males of the low- and high-dose main groups week 5 and in females of the mid-dose main group week 1) but were transient and considered indicative of biological variation and not due to administration of the test article (Supplemental Table 9).
Ophthalmoscopic evaluation did not reveal any test article-related alterations. Laboratory examinations showed no toxicologically relevant changes in hematology, blood coagulation, or clinical chemistry parameters at the end of the 28- or 90-day treatment periods. Slight sporadic alterations in several laboratory parameters were noted and are shown in Tables 1 and 2. However, the differences in values compared to controls were small, and values remained well within or marginal to historical control ranges, were not in a direction of usual concern, and/or had no dose relevance, and were without corresponding histopathological findings. Therefore, they were not considered to be of clinical or toxicological relevance.
Summary of Selecteda Hematological Findings in the 90-Day Repeated Oral Toxicity Study.b
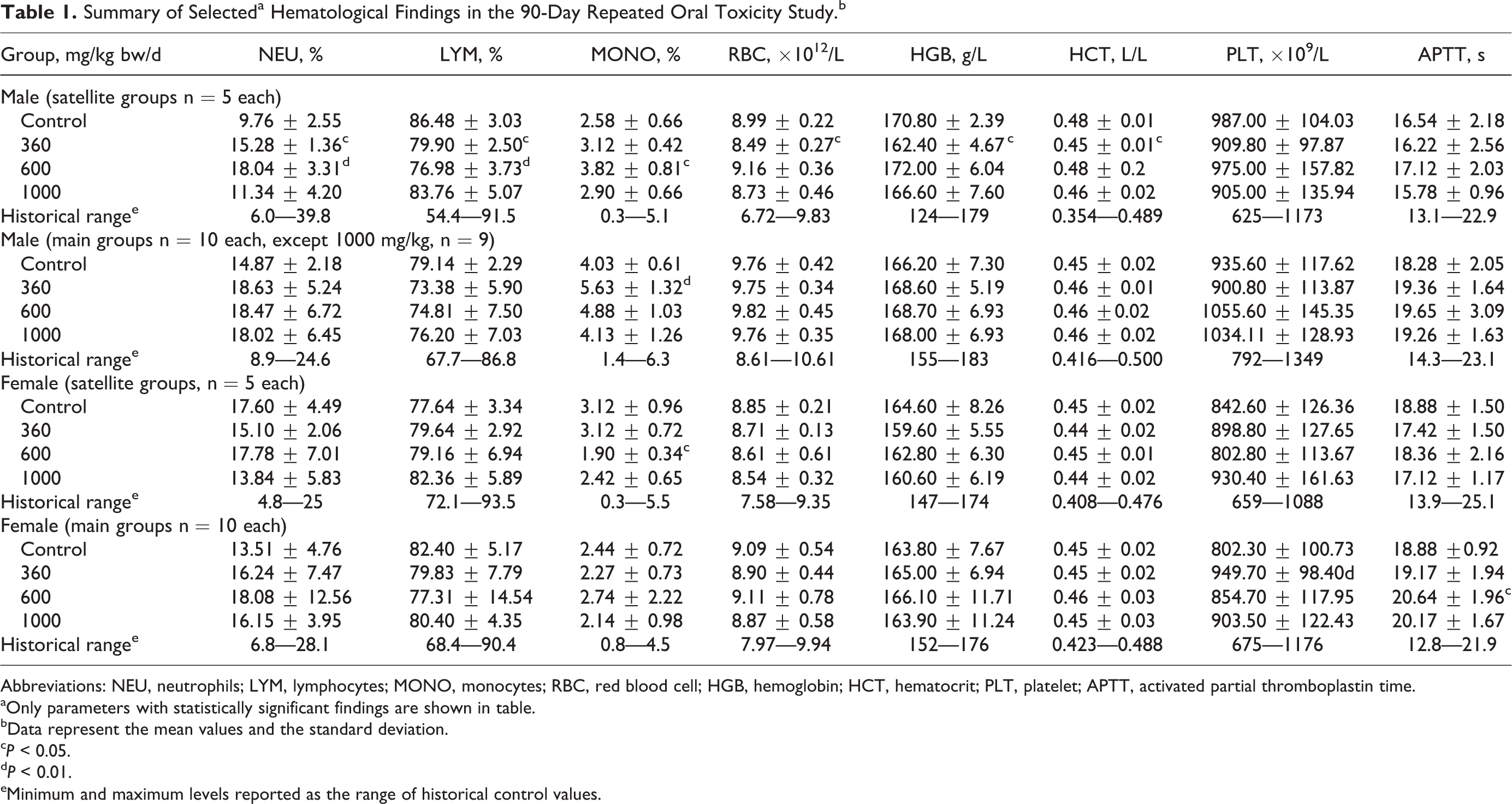
Abbreviations: NEU, neutrophils; LYM, lymphocytes; MONO, monocytes; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; PLT, platelet; APTT, activated partial thromboplastin time.
aOnly parameters with statistically significant findings are shown in table.
bData represent the mean values and the standard deviation.
c P < 0.05.
d P < 0.01.
eMinimum and maximum levels reported as the range of historical control values.
Summary of Selecteda Clinical Chemistry Findings in the 90-Day Repeated Oral Toxicity Study.b
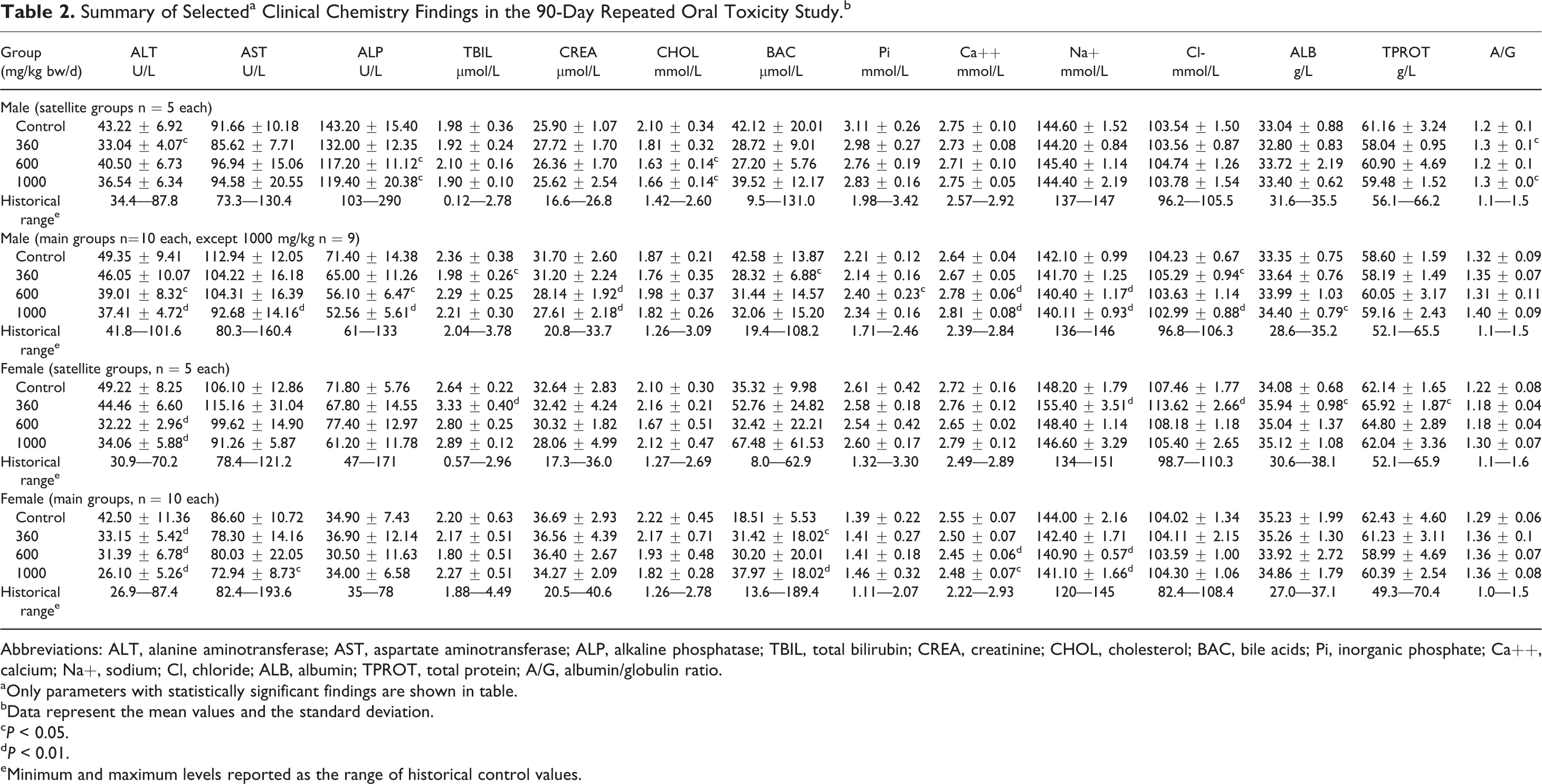
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; TBIL, total bilirubin; CREA, creatinine; CHOL, cholesterol; BAC, bile acids; Pi, inorganic phosphate; Ca++, calcium; Na+, sodium; Cl, chloride; ALB, albumin; TPROT, total protein; A/G, albumin/globulin ratio.
aOnly parameters with statistically significant findings are shown in table.
bData represent the mean values and the standard deviation.
c P < 0.05.
d P < 0.01.
eMinimum and maximum levels reported as the range of historical control values.
No test article-induced gross pathological lesions (Table 3) or organ weight differences (Supplemental Table 10) were observed in any of the organs and tissues at any of the groups (other than in the animal that died on day 60, as mentioned previously) compared to controls. Several individual macroscopic findings are reported in Table 3, none of which were considered toxicologically relevant due to lack of dose-dependence and/or common occurrence in control animals. Statistically significant slightly higher mean liver weight relative to body weight measurements were found in male animals of the mid- and high-dose satellite groups compared to controls (Supplemental Table 11). Similarly, statistically significant increases were also noted with respect to the controls in liver to body weight ratios in the low- and high-dose male animals and mid-dose female animals of the main groups (Supplemental Table 11). No significant differences were found in organ to brain weight ratios (Supplemental Table 12). These differences were of small degree, occurred sporadically, did not show dose–response, and had no related histopathological findings. Therefore, the findings were not considered toxicologically meaningful.
Summary of Necropsy Findings in the Surviving Animals in the 90-Day Repeated Oral Toxicity Study.a
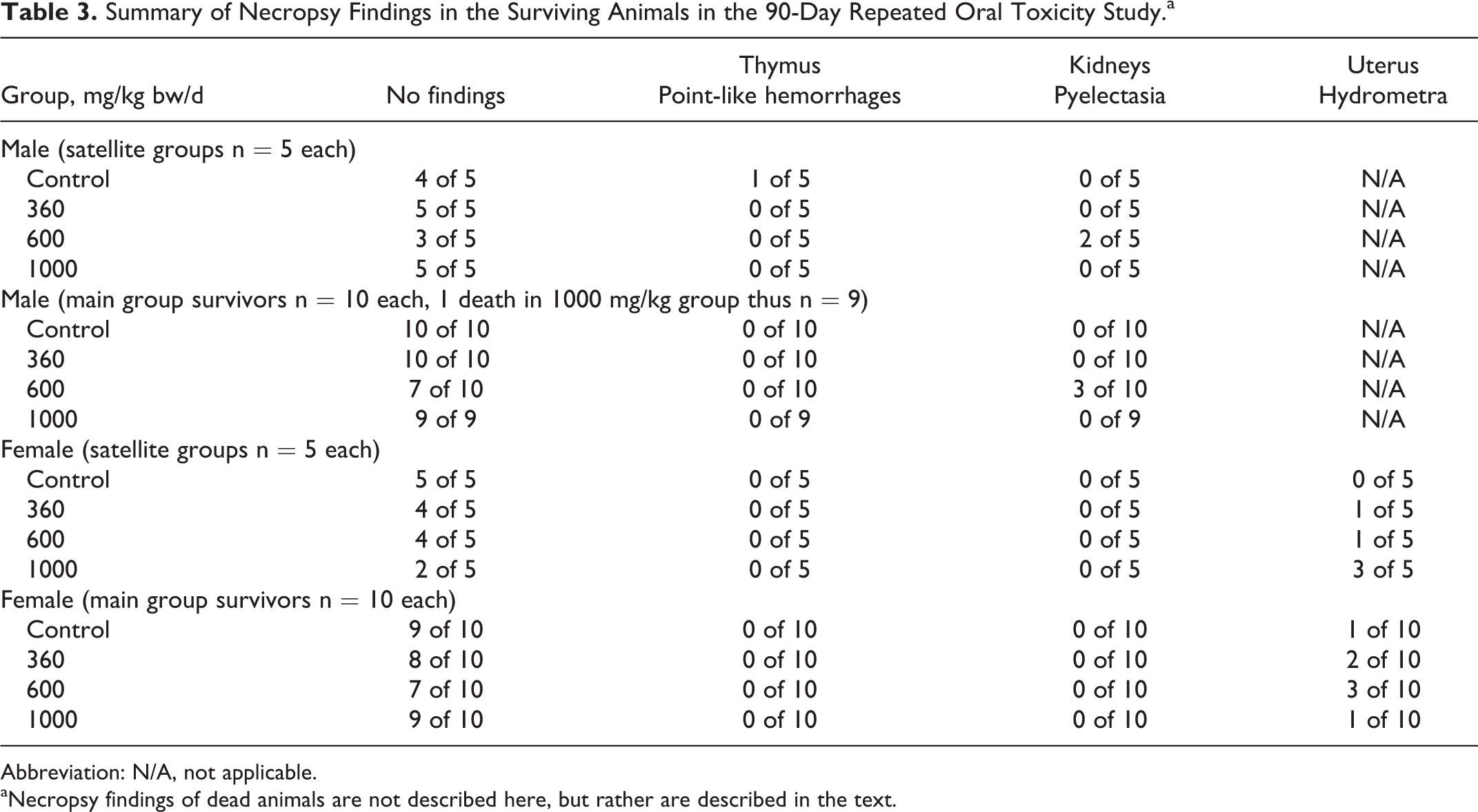
Abbreviation: N/A, not applicable.
aNecropsy findings of dead animals are not described here, but rather are described in the text.
Histopathological examinations did not reveal any test item-related findings in the examined organs of animals in the 1000 mg/kg bw/d satellite or main groups (including no findings suggestive of hyaline-like droplets in the kidneys), and additionally there were no renal changes in male satellite animals of the 360 or 600 mg/kg bw/d groups (analyzed due to kidney findings in the previously mentioned unpublished 14-day study). Histopathological findings of the 2 animals that died in the study were discussed earlier. Several histopathological findings were noted in surviving animals but were not considered toxicologically relevant (Table 4). They were alveolar emphysema (2 male animals and 1 female animal in the control group and 1 male animal in the high-dose group satellite groups, and 2 males and 2 females of the controls and 1 male of the high-dose group in the main groups), alveolar histiocytosis (1 male animal in the main control group), and hemorrhage of the thymus (1 male animal in the satellite control group). These findings were considered a consequence of exsanguination and not related to the test article. Similarly, hyperplasia of the bronchus-associated lymphoid tissue (BALT) was noted in the lungs of 1 male and 1 female of the controls and 1 male of the high-dose satellite groups, and 2 males and 2 females of controls, and 1 male of the high-dose main groups. Hyperplasia of BALT is considered an immunomorphological phenomenon and is not considered to have toxicological significance. 50,51 Dilation of uterine horns, seen in some female animals (3 in the high-dose satellite group, 1 in the main group control, and 1 in the high-dose main group), is considered a common neurohormonal phenomenon in connection with the proestrus phase of the sexual cycle. 52,53
Summary of Histopathological Findings in the Surviving Animals in the 90-Day Repeated Oral Toxicity Study.a
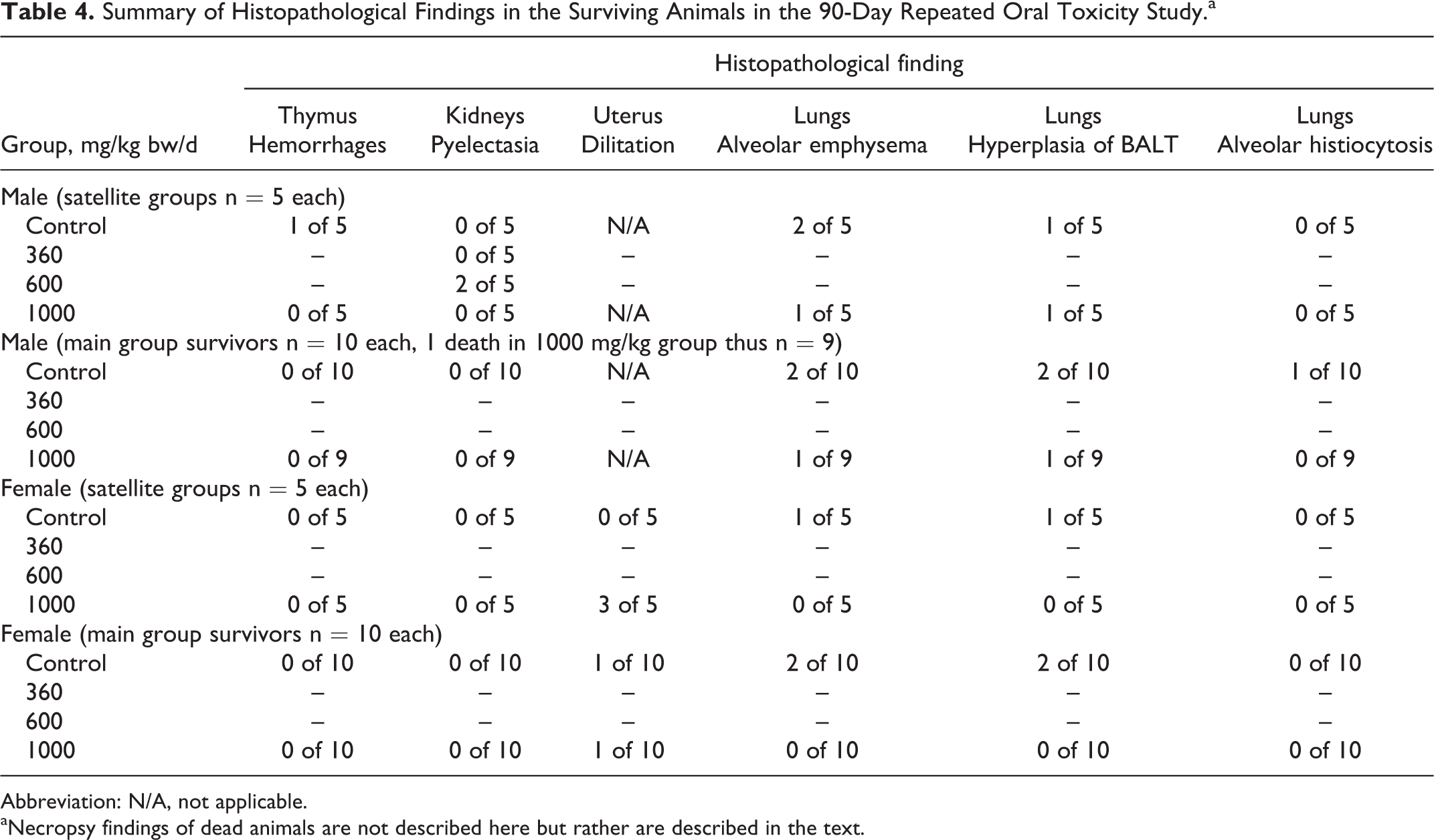
Abbreviation: N/A, not applicable.
aNecropsy findings of dead animals are not described here but rather are described in the text.
In conclusion, Bonolive™ did not cause adverse effects in CRL:(WI)BR Wistar rats after consecutive 28- or 90-day administration via gavage at doses of 360, 600, or 1000 mg/kg bw/d.
Discussion and Conclusion
Bonolive™ is an extract prepared from olive leaves and is considered to have health-promoting effects. 29 While previous studies suggest that olive polyphenols and extracts of olive leaves are generally safe, not all studies have been in agreement, 31 –33 and safety cannot necessarily be generalized. Until now, a comprehensive safety analysis performed according to international standards and protocols had not been conducted or published on this ingredient. In the current bacterial reverse mutation, in vitro mammalian chromosomal aberration, and in vivo mouse micronucleus tests, Bonolive™ was considered nonmutagenic, nonclastogenic, and nongenotoxic, respectively.
In a previous (unpublished) GLP 14-day repeated oral dose study that followed OECD 407 guidelines, which was performed to obtain initial information on the toxic potential of Bonolive™, findings resembling hyaline droplet nephropathy were noted in male CRL:(WI)BR Wistar rats at doses of 1000 and 2000 mg/kg bw/d. The effect appeared to be dose dependent and was not seen at 300 or 600 mg/kg bw/d in males nor in female animals at any dose level (up to 2000 mg/kg bw/d). No other test article-related significant findings were noted.
Hyaline droplet nephropathy describes a spectrum of morphologic changes in the kidneys of male rats induced by a variety compounds and conditions and may not be relevant to humans. 54 –56 There is generally abnormal accumulation of α-2μ-globulin phagolysosomes of the tubular epithelium in this condition. The finding is common in male rats and is not seen in humans although occasionally its severity can occur in a dose-related manner after administration of a test article, suggesting a possible effect. One proposed mechanism of interaction is that a chemical or metabolite may bind with α-2μ-globulin or alter its structure so that the tubular cell lysosomal enzymes cannot degrade the protein complex. Other proposed mechanisms include direct cytotoxic effects. 57 It is unlikely that the various chemicals associated with hyaline droplet nephropathy in the male rat throughout the literature act by the same mechanism. Some chemicals that produce hyaline droplet nephropathy in male rats also produce renal toxicity (unassociated with α-2μ globulin) in female rats, whereas others produce no effects in the kidney of female rats.
Because of this finding in the 14-day study, a satellite study of animals (5 per sex per group) was added to the 90-day study plan. These satellite animals were terminated on day 28 (as opposed to day 90) to obtain preliminary data, and the kidneys of male animals in all dose groups were processed and examined histopathologically to investigate the possible presence of hyaline-like droplets in the epithelial cells of proximal convoluted tubules before the remainder of the 90-days of exposure in the main groups. Examinations of the satellite groups, as well as the main groups, did not reveal any toxicologically relevant findings in the kidney (including hyaline-like droplets) suggesting that the finding in the 14-day study was likely an anomaly.
The deaths of 2 animals in the high-dose group (1 female on day 2, and 1 male on day 60) of the current study were considered a result of gavage injury and unrelated to systemic effects of the test article. Consumption of Bonolive™ via gavage for up to 90 days did not result in adverse effects in CRL:(WI)BR Wistar rats. The NOAEL for the study was 1000 mg/kg bw/d in both males and females, the highest dose tested.
A total of 64 female subjects were enrolled in a published human clinical study that also suggests long-term consumption of Bonolive™ is safe. 29 Treated subjects were given 250 mg of Bonolive™ plus 1000 mg calcium (to avoid potential confounding effects related to calcium deficiencies) and were compared to control subjects given 1000 mg of calcium alone. The treatment period was 12 months, and the incidence of adverse events in the study was similar across both groups. Only 2 serious adverse events occurred (a fracture of the right forearm and an incorrect mammography finding resulting in suspicion of breast cancer), both within the control group. There were no clinically relevant treatment-related adverse events recorded during the study.
In conclusion, the current compilation of toxicological studies is significant in supporting the safety of oral consumption of the olive leaf extract Bonolive™. The ingredient was not found to be mutagenic or genotoxic in the bacterial reverse mutation test, in vitro mammalian chromosomal aberration test, or in vivo mouse micronucleus test. Based on the results of the 90-day study, the NOAEL is considered 1000 mg/kg bw/d, the highest dose tested. The safety of Bonolive™ is also corroborated by the lack of treatment-related adverse events in the long-term (12 months) clinical trial, the history of safe ingestion of olive leaves as animal feed, and consumption of olive-based products by humans that contain many of the same constituents, such as table olives, olive oils, and other olive-derived products with a lack of known toxicity.
Supplemental Material
Supplemental Material, sj-xlsx-1-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-1-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-2-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-2-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-3-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-3-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-4-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-4-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-5-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-5-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-6-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-6-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-7-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-7-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-8-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-8-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-9-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-9-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-10-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-10-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-11-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-11-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Supplemental Material
Supplemental Material, sj-xlsx-12-ijt-10.1177_1091581815619764 - A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™)
Supplemental Material, sj-xlsx-12-ijt-10.1177_1091581815619764 for A Comprehensive Toxicological Safety Assessment of an Extract of Olea Europaea L. Leaves (Bonolive™) by Amy E. Clewell, Erzsébet Béres, Adél Vértesi, Róbert Glávits, Gábor Hirka, John R. Endres, Timothy S. Murbach and Ilona Pasics Szakonyiné in International Journal of Toxicology
Footnotes
Acknowledgments
The authors would like to thank the following participating investigators for their contributions to the work: Viktória Balogh, Erzsébet Biczó, Ibolya Bogdán, István Buda, Tamás Buda, Monika Csatári, Tímea Csörge, Erika Major Biermanné, Zsuzsanna Frank, Zoltánné Háda, Ildikó Hermann, Isvánné Horváth, Zoltán Jakab, Katalin Kégl, Anita Forintos Kékesiné, Klára Fritz Kovácsné, Éva Steinbach Köváriné, Kornélia Sereg Jurácsikné, Adrienn Laczó, Viktória Matina, Edit Kövári Mesterháziné, Mónika Fekete Pápainé, János Stáhl, Ágota Jó Schüllerné, Beatrix Szilágyi Sümeginé, Éva Szabó, Ferenc Szabó, Zsuzsanna Szabó, Mariann Lennert Szabóné, Edit Szám, Judit Szilák, Márta Tenk, Zsuzsanna Vuleta, and Levente Zoltán for the performance of experimental tasks and/or collection of data; and Jared Brodin for administrative support in preparation of the manuscript.
Author Contributions
Clewell, A. and Murbach T. contributed to acquisition, analysis and/or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content; Beres, E., Vertesi, A., Glavits, R., Hirka, G., Endres, J., and Pasics Szakonyiné, I. substantially contributed to conception or design, contributed to acquisition, analysis, and/or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AC, JE and TM are employed by AIBMR Life Sciences, which was contracted by the study sponsors as an independent third party to place and monitor toxicological studies on the test article, and to publish the results. The remaining authors are employed by Toxi-coop Zrt, an independent contract research organization that was hired to perform the toxicological studies and interpret and report the results.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclose that financial support for the research described herein was provided by Ferrer HealthTech (Barcelona, Spain) and BioActor (Maastricht, the Netherlands).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
