Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) assessed the safety of 16 alkyl ethylhexanoates for use in cosmetics, concluding that these ingredients are safe in cosmetic formulations in the present practices of use and concentrations when formulated to be nonirritating. The alkyl ethylhexanoates primarily function as skin-conditioning agents in cosmetics. The highest concentration of use reported for any of the alkyl ethylhexanoates is 77.3% cetyl ethylhexanoate in rinse-off formulations used near the eye, and the highest leave-on use reported is 52% cetyl ethylhexanoate in lipstick formulations. The Panel reviewed available animal and clinical data related to these ingredients, and the similarities in structure, properties, functions, and uses of ingredients from previous CIR assessments on constituent alcohols that allowed for extrapolation of the available toxicological data to assess the safety of the entire group.
Introduction
Cetearyl ethylhexanoate was reviewed previously by the Cosmetic Ingredient Review (CIR) Expert Panel (Panel) in 1982, with the conclusion that this ingredient (then named cetearyl octanoate) is safe as used in cosmetics. 1 As reported in 2006, the Expert Panel reaffirmed the conclusion of safe as used. 2 A summary of the data included in these previous reviews is provided in Table 1.
Summaries of Previous Reviews of Cetearyl Ethylhexanoate.

Abbreviation: SIOPT, single insult occlusive patch test.
The Expert Panel determined that the data supporting the safety of cetearyl ethylhexanoate can be extrapolated to support the safety of the 15 additional ethylhexanoates that are used in cosmetics. Therefore, the following 16 ingredients are included in this assessment. C12-13 Alkyl ethylhexanoate C12-15 Alkyl ethylhexanoate C14-18 Alkyl ethylhexanoate Cetearyl ethylhexanoate Cetyl ethylhexanoate Decyltetradecyl ethylhexanoate Ethylhexyl ethylhexanoate Hexyldecyl ethylhexanoate Isocetyl ethylhexanoate Isodecyl ethylhexanoate Isostearyl ethylhexanoate Lauryl ethylhexanoate Myristyl ethylhexanoate Octyldodecyl ethylhexanoate Stearyl ethylhexanoate Tridecyl ethylhexanoate
The safety of the individual constituents of these esters is relevant to the safety of each ester as a whole. The constituent acid that is common to all the alkyl ethylhexanoates, that is, 2-ethylhexanoic acid, is a likely metabolite of the alkyl ethylhexanoates; 2-Ethylhexanoic acid is not a cosmetic ingredient. However, because it is a likely metabolite, the summary document from the first rereview of cetearyl ethylhexanoate discussed the reproductive and developmental toxicity of this compound. 2 This information is recapitulated in this rereview document because it is applicable to all the alkyl ethylhexanoates.
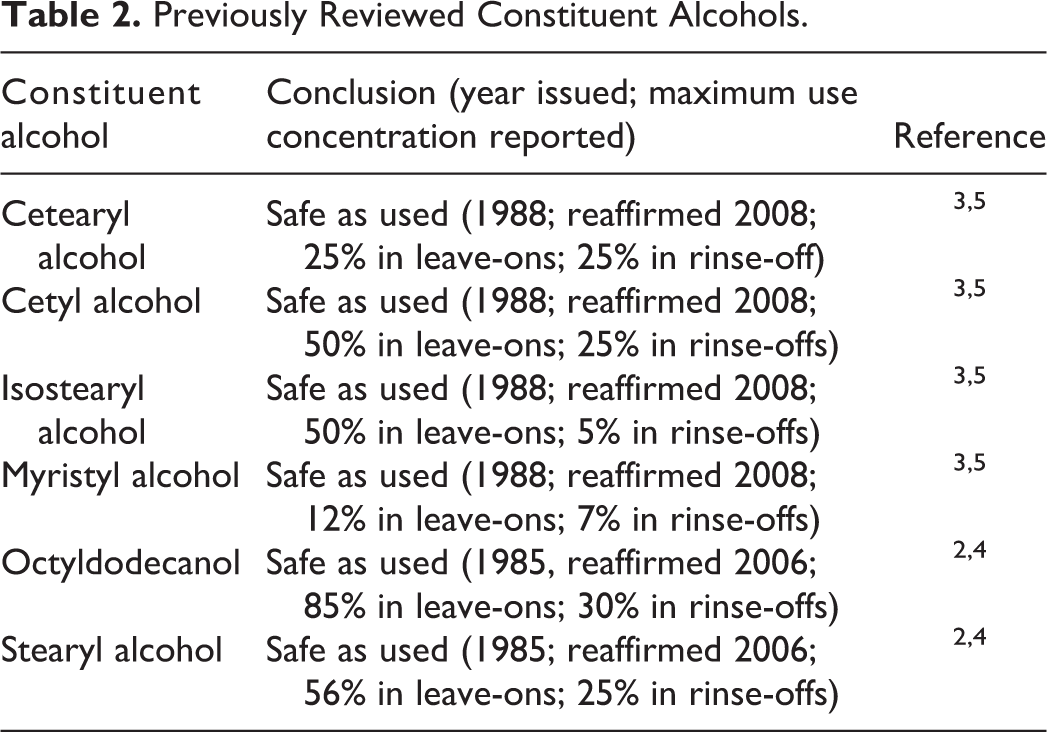
Six of the constituent alcohols are cosmetic ingredients that have been found safe by the CIR. 2 -5 Table 2 provides a listing of the previously reviewed constituent alcohols, that is, cetearyl, cetyl, isostearyl, myristyl, and stearyl alcohol and octyldodecanol. The maximum reported concentration of use of each alcohol at the time of its review is provided in Table 2 so as to reflect contextual constraints.
Previously Reviewed Constituent Alcohols.
Provided in Table 3 is summary information on ethylhexyl and isodecyl alcohols. These alcohols are not cosmetic ingredients; however, some data on these alcohols were presented in a previous CIR review and are included here because of the relevance to the safety of the alkyl ethylhexanoates. 6
Available Data on Constituent Alcohols not Reviewed by the CIR.

Abbreviations: CIR, Cosmetic Ingredient Review; NOAEL, no observed adverse effect level; bw, body weight; wk, week, mos, months; wt, weight.
Chemistry
Definition and Structure
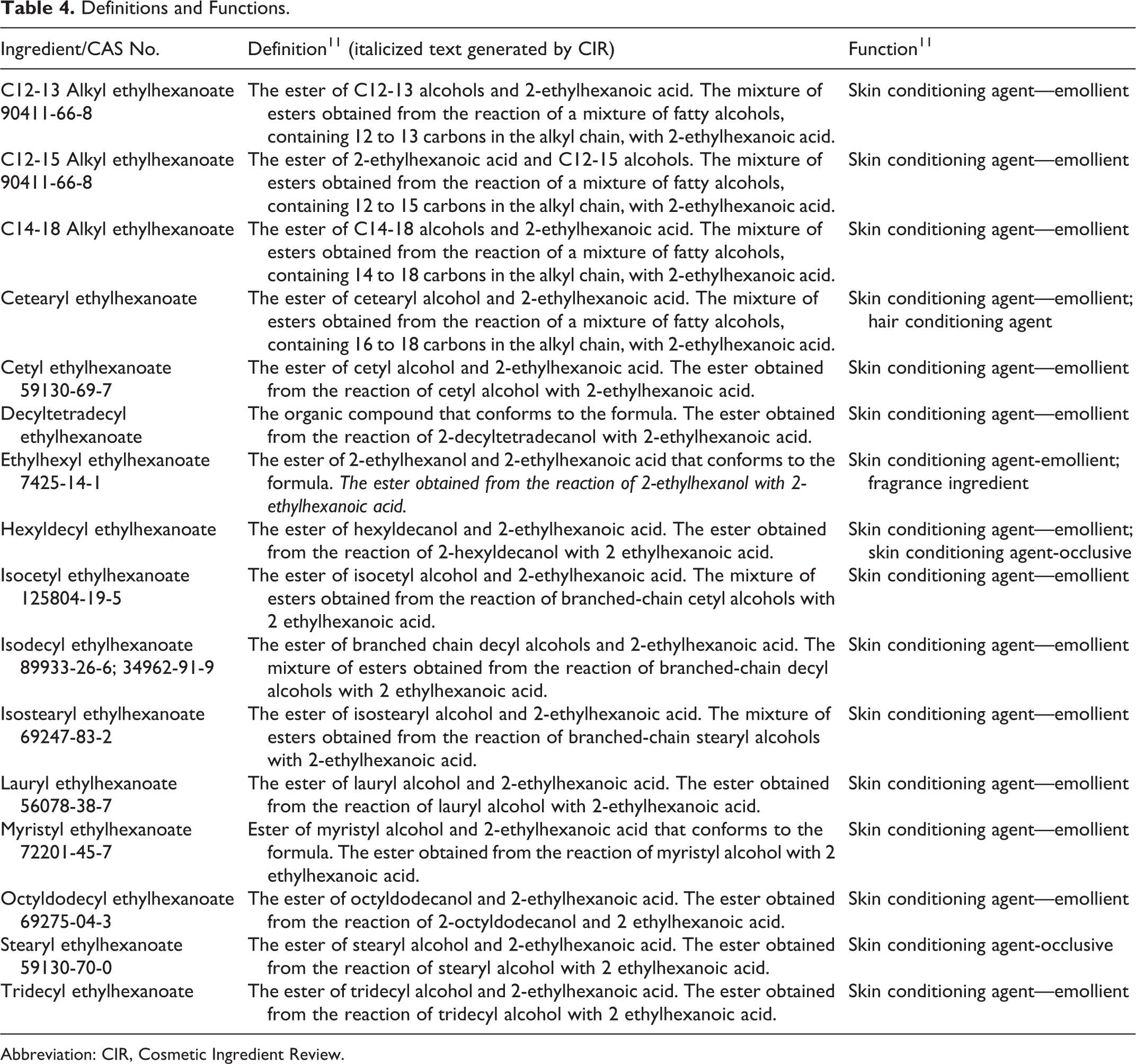
The structure of each ingredient is depicted in Figure 1. The ingredients included in this assessment are defined in Table 4.

Structures ordered by chain length and chemical structure.
Definitions and Functions.
Abbreviation: CIR, Cosmetic Ingredient Review.
The alkyl ethylhexanoates are branched alkyl esters that are the result of the esterification of an alkyl alcohol with 2-ethylhexanoic acid (or 2-ethylhexanoic acid chloride). The key similarities between these ingredients are a carboxyl ester functional group (flanked on the ester side by an ethylhexyl group) and an alkyl chain on the alcohol side (Figure 2).
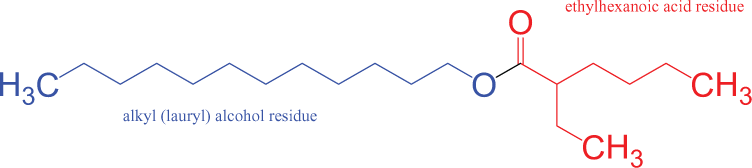
Lauryl ethylhexanoate.
Methods of Manufacture
Most of these alkyl ethylhexanoates are produced synthetically via classical Fischer type esterification methods (ie, reaction of a carboxylic acid with an alcohol to produce a carboxylic ester, Figure 3), although the reaction may be promoted by acid or base catalysis or by the use of an acid chloride.

Synthesis of lauryl ethylhexanoate from lauryl alcohol and ethylhexanoic acid.
For example, cetearyl ethylhexanoate is commercially prepared by catalytic esterification with removal by azeotropic distillation. 1 Cetearyl ethylhexanoate can also be prepared by blending cetyl octanoate and stearyl octanoate in a weight ratio of 7:2. Ethylhexyl ethylhexanoate is prepared by the direct esterification reaction of 2-ethylhexanoic acid and 2-ethylhexanol in n-hexane (solvent) and with Novozym 435 (a commercial immobilized lipase from Candida antartica) acting as the catalyst. 7
Physical and Chemical Properties
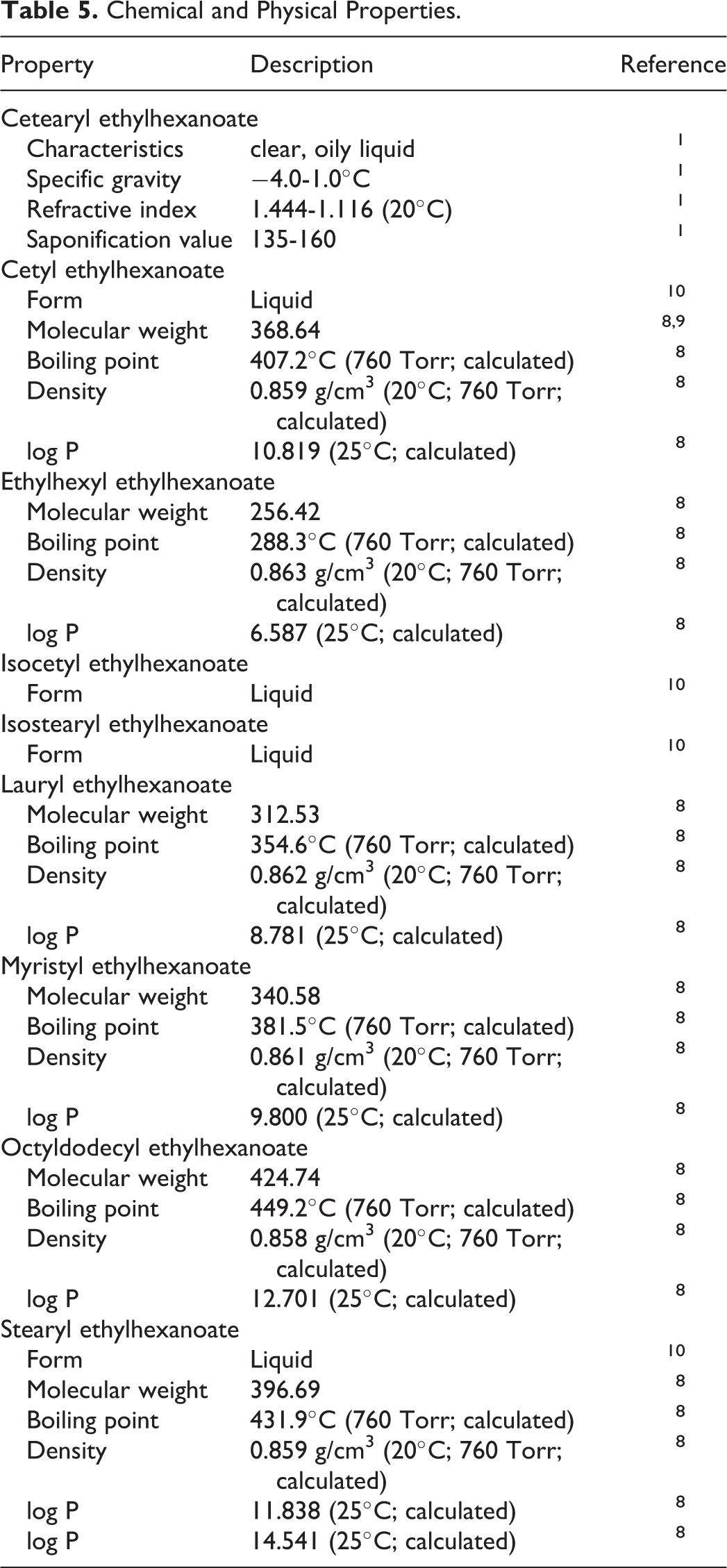
Physical and chemical properties data are provided in Table 5.1, 8 -10
Chemical and Physical Properties.
Reactivity
The alkyl ethylhexanoates can be expected to undergo chemical or enzymatic hydrolysis to 2-ethylhexanoic acid and the corresponding alcohols. 1 Transesterification and other typical ester reactions (such as aminations) may also occur.
Use
Cosmetic
The alkyl ethylhexanoates primarily function in cosmetics as skin-conditioning agents. 11 Ethylhexyl ethylhexanoate also functions as a fragrance ingredient. (Table 4) Data on the usage of ingredients as a function of cosmetic product category are provided by the manufacturers to the Food and Drug Administration’s Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained from the Food and Drug Administration in 2013 12 and data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) 13 indicate that 8 of the alkyl ethylhexanoates named in this safety assessment are currently used in cosmetic formulations.
The current and historical frequency and concentration of use data for cetearyl ethylhexanoate are provided in Table 6; the frequency of use has increased from 229 uses in 2002 2 to 404 uses in 2013 12 ; the maximum concentration of use has not changed and remains at 35% (in dermal leave-on formulations). 2,13 Frequency and concentration of use data for the other 7 in-use ethylhexanoates are provided in Table 7. With the exception of 275 uses for cetyl ethylhexanoate, these ingredients each are used in less than 50 formulations. 12 The highest concentration of use reported for any of the alkyl ethylhexanoates is 77.3% cetyl ethylhexanoate in rinse-off formulations used near the eye, and the highest leave-on use reported is 52% cetyl ethylhexanoate in lipstick formulations. 13 Table 8 lists the 8 alkyl ethylhexanoates that are not reported to be used according to the surveys of both the FDA and the Council.
Current and Historical Frequency and Concentration of Use According to Duration and Type of Exposure.
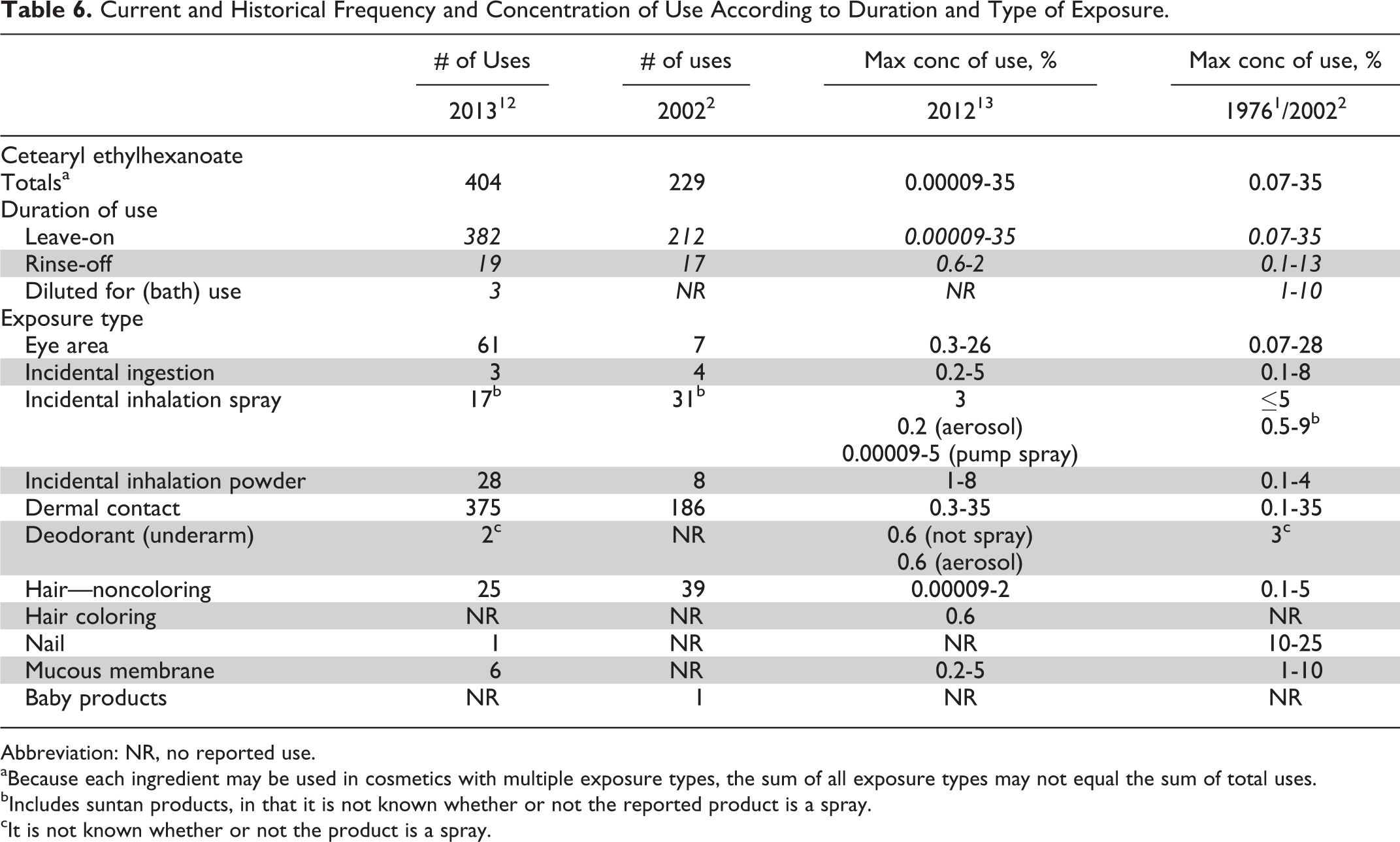
Abbreviation: NR, no reported use.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether or not the reported product is a spray.
cIt is not known whether or not the product is a spray.
Frequency and Concentration of Use According to Duration and Type of Exposure.
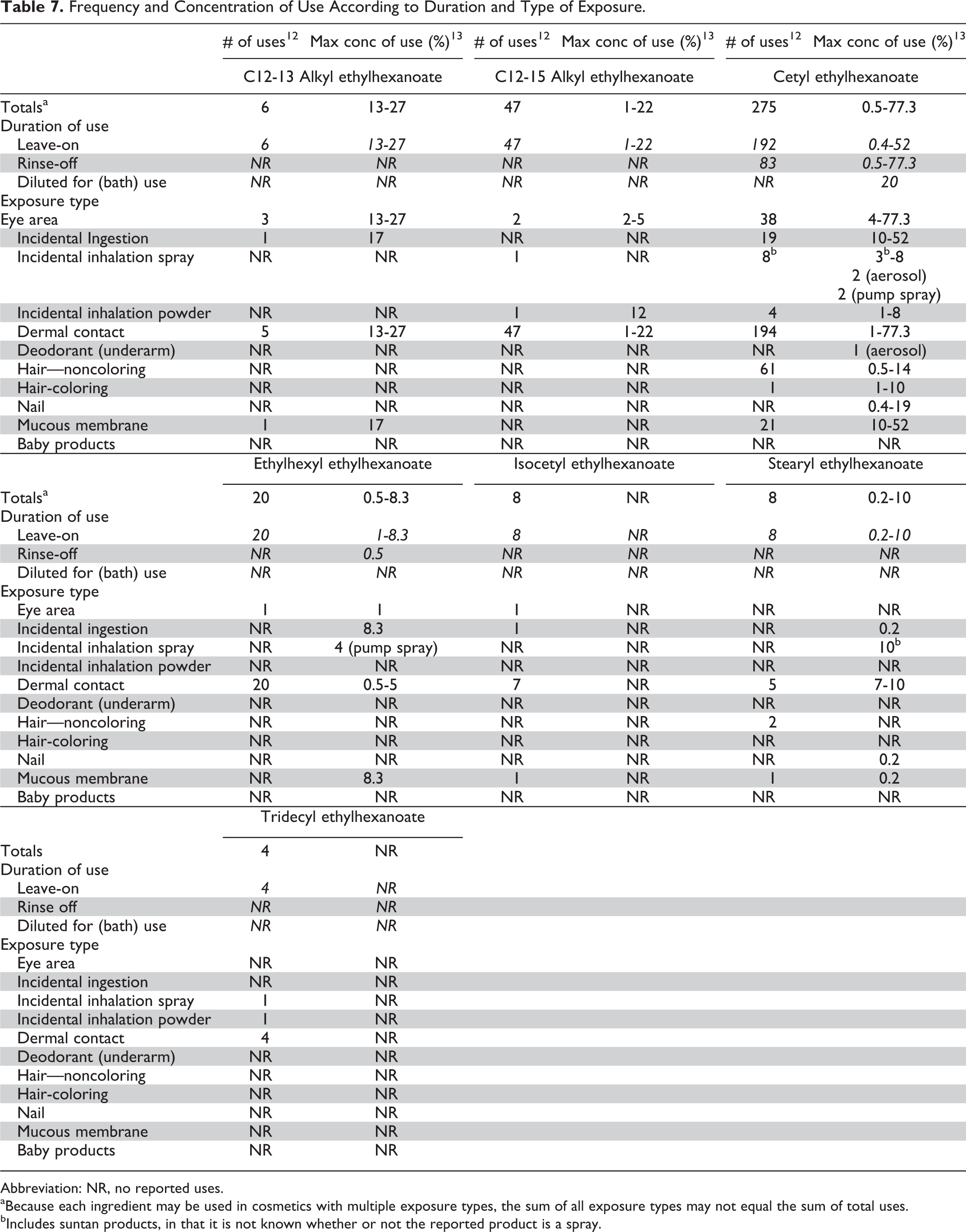
Abbreviation: NR, no reported uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIncludes suntan products, in that it is not known whether or not the reported product is a spray.
Ingredients Not reported to be Used.

Some alkyl ethylhexanoates are used in formulations that are reported to be applied to the eye area or mucous membranes or in products that could possibly be ingested. Additionally, some of the alkyl esters are used in cosmetic sprays or powders and could incidentally be inhaled. The highest concentration of known spray use is 5% cetearyl ethylhexanoate in a pump spray formulation. Other uses may or may not be sprays, such as 8% cetyl ethylhexanoate in fragrance preparations and 10% stearyl ethylhexanoate in suntan preparations. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 14,15 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 16,17 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 16 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Ethylhexyl ethylhexanoate is listed in the European Union inventory of cosmetic ingredients, 18 and according to the European Chemical Substances Information System, it has a reproductive risk classification of category 3, substances which cause concern, with risk phrase R63, possible risk of harm to the unborn child. 19 Ethylhexyl ethylhexanoate is not restricted by the European Commission, but the Scientific Committee on Consumer Safety (SCCS) is of the opinion that “the use in cosmetic products of substances classified as carcinogenic, mutagenic or toxic for reproduction, of category 1, 2 and 3, under Annex I to Directive 67/548/EEC shall be prohibited.… A substance classified in category 3 may be used in cosmetics if the substance has been evaluated by the Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers (SCCNFP) [now called the SCCS] and found acceptable for use in cosmetic products.” 20 All other alkyl esters named in this safety assessment are listed in the European Union inventory of cosmetic ingredients. 18
Noncosmetic
Ethylhexyl ethylhexanoate is used in pharmaceutical preparations for improving the spreading behavior of the oil. 7
Toxicokinetics
The alkyl ethylhexanoates are most likely hydrolyzed in the gastrointestinal tract to 2-ethylhexanoic acid and the corresponding alcohols. 1 These products, in turn, would enter their respective metabolic pathways.
Penetration Enhancement
The effect of alkyl ethylhexanoates on the penetration of indomethacin through excised hairless rat skin was examined. 10 The permeation of 1% indomethacin from suspensions and from hydrogenated phospholipid gels containing cetyl ethylhexanoate, isocetyl ethylhexanoate, or stearyl ethylhexanoate was determined. The permeation rate of indomethacin from the esters increased with increased solubility of the drug in the ester. The solubility of indomethacin in liquid paraffin is very low, and there was no permeation of indomethacin from liquid paraffin after 10 hours. Permeation rates (and solubility) were higher in gels formed by hydrogen phospholipid than from suspensions. In all cases, a linear relationship existed between the cumulative amounts of indomethacin that permeated from any ester from 4 to 10 hours.
Animal Toxicology
Although no additional significant dermal, oral, or inhalation toxicity was reported, Table 1 provides summary information from the original safety assessment on cetearyl ethylhexanoate.
Reproductive and Developmental toxicity
2-Ethylhexanoic acid is a possible metabolite of the alkyl ethylhexanoates; therefore, the reproductive and developmental toxicity of 2-ethylhexanoic acid may be relevant to the safety of alkyl ethylhexanoates. Accordingly, the data on the reproductive and developmental toxicity on 2-ethylhexanoic acid, and the mechanism of action for both, which were included in the original rereview of cetearyl ethylhexanoate, are reiterated here. 21 -36
2-Ethylhexanoic acid has been shown to be a liver and a developmental toxicant when administered orally at high-dose levels to rodents. In developmental studies, it has been postulated that 2-ethylhexanoic acid maternal liver toxicity begins a cascade of effects that includes metallothionein (MT) induction, zinc accumulation in the liver due to MT binding, and a resulting zinc deficiency in the developing embryo. In this model, it is the zinc deficiency in the developing embryo that causes developmental toxicity. Support for this mechanism of action comes from several sources. Animal studies have demonstrated that dietary zinc supplementation reduces the toxic effect and that further zinc deficiency makes 2-ethylhexanoic acid more toxic. In vitro studies using embryo cultures have demonstrated that either zinc deficiency or 2-ethylhexanoic acid-treated sera produced developmental toxicity. Zinc supplementation of either or both sera eliminated the effect.
To further examine this question, di-2-ethylhexyl terephthalate (DEHT), which yields 2-ethylhexanoic acid through metabolism, was chosen as a model that would result in 2-ethylhexanoic acid exposures without liver toxicity and MT induction. Di-2-ethylhexyl terephthalate is metabolized in the gut and liver to 2-ethylhexanol and terephthalate. Two moles of 2-ethylhexanol are produced per mole of DEHT. Subsequent hydrolysis of 2-ethylhexanol produces 2-ethylhexanoic acid. It can be hypothesized that this pathway to 2-ethylhexanoic acid production from a precursor would not give rise to acute liver toxicity, MT induction, zinc sequestration, and developmental toxicity.
In a reproductive and developmental toxicity study, 0, 0.3, 0.6, and 1% DEHT was provided in the feed of rats. The doses were calculated to be 614 to 823 mg/kg/d for males and 783 to1021 mg/kg/d for females. Reproductive toxicity and developmental toxicity were not seen at any dose level. These findings suggest that the process of metabolic conversion of DEHT to 2-ethylhexanol, and subsequent hydrolysis to 2-ethylhexanoic acid, results in a time course of 2-ethylhexanoic acid appearance that allows clearance before sufficient levels can arise to produce acute liver toxicity.
While the above study was undertaken to understand 2-ethylhexanoic acid developmental toxicity, the Panel considered that it is relevant to the assessment of alkyl ethylhexanoates. Like DEHT, alkyl ethylhexanoates must undergo conversion to produce 2-ethylhexanoic acid. In addition, alkyl ethylhexanoates, as used in cosmetics, would have to pass through the stratum corneum and the epidermis before entering the bloodstream, further moderating the time course of 2-ethylhexanoic acid appearance in the liver.
Genotoxicity
No published genotoxicity data were discovered and no unpublished data were submitted.
Carcinogenicity
No published carcinogenicity data were discovered and no unpublished data were submitted.
Irritation and Sensitization
Dermal Irritation and Sensitization
Summaries of irritation and sensitization data from the original safety assessment on cetearyl ethylhexanoate are available in Table 1. Generally, formulations containing cetearyl ethylhexanoate did not produce significant irritation and were not sensitizers.
Nonhuman
The dermal irritation of cetyl ethylhexanoate was determined using rabbits, guinea pigs, rats, and miniature swine. 37 In rabbits, 0.1 g of undiluted cetyl ethylhexanoate was applied directly to a shaved 3 cm × 3 cm area on the back of 6 albino angora rabbits; n-Hexadecane was used as the control. A collar was used to prevent ingestion of the test substance. The test sites were scored for irritation 24 hours after application. After scoring, the test site was clipped, the test article was applied 30 minutes later, and the area was scored 48 hours after application. Following this reading, all the hair on the dorsal surface was clipped, and the animals were injected with Evans blue in physiological saline and killed 1 hour after injection. The total skin reaction score was assessed by evaluating erythema in live animals and the dilating rate, edema, and bluing rate in skin removed at the end of the study. The relative irritancy score for cetyl ethylhexanoate was 3/3, severely irritating to rabbit skin.
A similar protocol and scoring were followed using groups of 6 male Hartley guinea pigs and 6 male Wistar rats, with the exception that the control was an untreated site. Again, the dose tested was 0.1 g cetyl ethylhexanoate applied to a 3 cm × 3 cm area of shaved skin. The relative irritancy score for cetyl ethylhexanoate was 2/3, moderately irritating, in the guinea pig and 1/3, mildly irritating, in the rat.
Using miniature swine, 0.05 g cetyl ethylhexanoate was applied to the clipped skin of 6 animals for 48 hours using a 15-mm occlusive patch. The test site was then scored as described earlier. The relative irritancy score for cetyl ethylhexanoate was 0/3 (nonirritating) in miniature swine.
Human
The dermal irritation of cetyl ethylhexanoate was evaluated by applying 0.05 g of undiluted test article on a 15-mm patch to 50 male individuals. 37 The patches were removed after 48 hours, and the test sites scored 30 minutes later. Undiluted cetyl ethylhexanoate was mildly irritating (defined as producing 10%-40% positive reactions) to humans skin. (Additional details were not provided.)
Cetearyl ethylhexanoate was not a sensitizer in a human repeated insult patch test completed in 103 individuals in which it was used as a solvent and tested as a control. 38 No reactions were observed during induction or challenge. (Additional details were not provided).
Phototoxicity
Summary information from the original safety assessment on cetearyl ethylhexanoate is found in Table 1. Cetearyl ethylhexanoate was not phototoxic.
Ocular Irritation
Summary information from the original safety assessment on cetearyl ethylhexanoate is found in Table 1. Cetearyl ethylhexanoate was not an ocular irritant in rabbits.
Summary
Cetearyl ethylhexanoate was reviewed previously by the Expert Panel, and in 1982 the Panel concluded that cetearyl ethylhexanoate (then named cetearyl octanoate) is safe as used in cosmetics. The conclusion was reaffirmed, as reported in 2006. The data in the existing safety assessments on cetearyl ethylhexanoate were deemed applicable to an additional 15 alkyl ethylhexanoates that are cosmetic ingredients; therefore, the Panel developed a safety assessment of the alkyl ethylhexanoate group. The alkyl ethylhexanoates are branched alkyl chains that consist of an alcohol and 2-ethylhexanoic acid, and they function in cosmetics primarily as skin-conditioning agents.
The VCRP data indicate that 8 of the 16 alkyl ethylhexanoates are currently in use in cosmetic formulations. Cetearyl ethylhexanoate has the most reported uses, 404, followed by cetyl ethylhexanoate, 275. The remaining ingredients are used in less than 50 formulations. Cetyl ethylhexanoate has the highest reported use concentration, 77.3% in rinse-off formulations used near the eye; it also has the highest leave-on use concentration, 52% in lipstick formulations.
Alkyl ethylhexanoates tended to increase the permeation rate of indomethacin. The increase occurred due to increased solubility.
Undiluted cetyl ethylhexanoate was severely irritating to rabbit skin, moderately irritating to guinea pig skin, mildly irritating to rat skin, and nonirritating to the skin of miniature swine. A 48-hour patch with undiluted cetyl ethylhexanoate produced mild irritation in dermal irritation study in 50 individuals.
Discussion
The Panel began its consideration in the context of the 1982 safety assessment that cetearyl ethylhexanoate was safe as reported to be used in cosmetics. This conclusion was reaffirmed in 2006. Because of perceived structural similarity, cetearyl ethylhexanoate was included in an expansion of that safety assessment to include the entire alkyl esters family of ingredients. The Panel, however, determined that inclusion of ethylhexanoates in that report was not appropriate because 2-ethylhexanoic acid, a suspected liver and developmental toxicant in animal studies at high dose levels, is a possible metabolite of cetearyl ethylhexanoate. The Panel did determine, however, that the data included in the safety assessments of cetearyl ethylhexanoate can be extrapolated to support the safety of the 15 additional ethylhexanoates that are used in cosmetics, thereby creating the alkyl ethylhexanoate family for review.
Regarding the liver and developmental toxicant effects of 2-ethylhexanoic acid in animal studies at high dose levels, the Panel considered that the mechanism is attributed to a cascade of effects that includes MT induction, zinc accumulation in the liver due to MT binding, and a resulting zinc deficiency in the developing embryo. The Panel found that results of testing with DEHT (a 2-ethylhexanoic acid precursor used as a model for exposure without liver toxicity, etc) suggested that the process of metabolic conversion results in a time course that allows clearance of 2-ethylhexanoic acid at rates such that levels cannot attain toxicological significance. The Panel concluded that for alkyl ethylhexanoate compounds that are absorbed through the skin, any metabolism would not generate hexanoic acid at sufficient levels to trigger the MT induction-dependent effects observed in animal studies.
Although there are existing data gaps, the relatedness of molecular structures, physicochemical properties, functions, and/or concentrations in cosmetics allowed grouping these ingredients together and interpolating/extrapolating the available toxicological data to support the safety of the entire group. The similar structure–property relationships and cosmetic product usage suggest that the available data from the previous review of cetearyl ethylhexanoate, as well as from safety assessments on some of the constituent alcohols, can be extrapolated to support the safety of the alkyl ethylhexanoates. For example, the consensus of the Panel was that because dermal penetration of long-chain alcohols is likely to be low, it could be inferred that the dermal penetration for alkyl esters was likely to be even lower. The Panel also noted specifically that 2 of the data gaps were genotoxicity and carcinogenicity data. The Panel relied on the previous CIR safety assessments on the constituent alcohols to alleviate concerns over the lack of these data.
The Panel recognized that some of the alkyl ethylhexanoates can enhance the penetration of other ingredients through the skin, and cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data or when dermal absorption was a concern.
The Panel was also concerned that the potential exists for dermal irritation with the use of products formulated using some of the alkyl ethylhexanoates. They specified that products must be formulated to be nonirritating.
Finally, the Panel discussed the issue of incidental inhalation exposure to alkyl ethylhexanoates from powders and products that may be aerosolized. There were no repeated dose inhalation toxicity data available for the alkyl ethylhexanoates. Cetearyl ethylhexanoate is reportedly used at up to 8% in dusting powders that may become airborne and at known concentrations of up to 5% in pump spray formulations; some of the alkyl ethylhexanoates may be used as high as 10% in some products that may or may not be sprays, such as 10% stearyl ethylhexanoate in suntan preparations. Droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. These ingredients are large molecules, and in most cases, are insoluble in water, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. The Panel considered the data available to characterize the potential for alkyl ethylhexanoates to cause systemic toxicity, irritation, sensitization, or other effects. They noted that cetearyl ethylhexanoate tended not to produce systemic toxicity in single-dose oral, dermal, or inhalation studies or a repeated-dose dermal toxicity study was not a sensitizer and not phototoxic. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products that may be aerosolized is available at
Conclusion
The Panel concluded that the alkyl ethylhexanoates, listed below, are safe in the present practices of use and concentration described in this safety assessment when formulated to be nonirritating. C12-13 Alkyl ethylhexanoate C12-15 Alkyl ethylhexanoate C14-18 Alkyl ethylhexanoate* Cetearyl ethylhexanoate Cetyl ethylhexanoate Decyltetradecyl ethylhexanoate* Ethylhexyl ethylhexanoate Hexyldecyl ethylhexanoate* Isocetyl ethylhexanoate Isodecyl ethylhexanoate* Isostearyl ethylhexanoate* Lauryl ethylhexanoate* Myristyl ethylhexanoate* Octyldodecyl ethylhexanoate* Stearyl ethylhexanoate Tridecyl ehylhexanoate
*Not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
Fiume, M. contributed to conception and design; contributed to acquisition, analysis, and interpretation; and drafted the manuscript. Heldreth, B. contributed to conception and design, contributed to acquisition, analysis, and interpretation; drafted the manuscript and critically revised the manuscript. Gill, L., Andersen, F. Alan, Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Author’s Note
Unpublished data are available from Lillian Gill, Director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
