Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) assessed the safety of boron nitride which functions in cosmetics as a slip modifier (ie, it has a lubricating effect). Boron nitride is an inorganic compound with a crystalline form that can be hexagonal, spherical, or cubic; the hexagonal form is presumed to be used in cosmetics. The highest reported concentration of use of boron nitride is 25% in eye shadow formulations. Although boron nitride nanotubes are produced, boron nitride is not listed as a nanomaterial used in cosmetic formulations. The Panel reviewed available chemistry, animal data, and clinical data and concluded that this ingredient is safe in the present practices of use and concentration in cosmetic formulations.
Keywords
Introduction
This report reviews the safety of boron nitride as used in cosmetics. Boron nitride is an inorganic compound reported to function in cosmetics as a slip modifier. Although the International Cosmetic Ingredient Dictionary and Handbook does not specify which crystal form(s) is(are) used in cosmetics, 1 it is presumed that the hexagonal form of boron nitride is used because that form (similar to graphite) matches the functionality in cosmetics.
In the 2011 Associations Survey of Nanomaterials Used in Cosmetic Products, a report prepared for the International Cooperation on Cosmetic Regulation by a working group of the Personal Care Products Council (Council), Canadian Cosmetic Toiletry, and Fragrance Association, COLIPA (now known as Cosmetics Europe), and the Japanese Cosmetic Industry Association, boron nitride is not listed as a nanomaterial used in cosmetic formulations. 2 This safety assessment prepared by the Cosmetic Ingredient Review (CIR) includes (limited) toxicity data on boron nitride nanotubes because although boron nitride is not thought to be a nanomaterial used in cosmetic formulations, these data demonstrate the lack of toxicity of boron nitride.
Chemistry
Definition and Structure
Boron nitride (CAS No. 10043-11-5) is an inorganic compound, with the chemical formula BN. 1 The compound can assume 3 crystal polymorphs. One is a flat, hexagonal crystalline structure, similar to graphite, except that the carbon atoms are replaced by boron and nitrogen atoms. 3,4 The alternate boron and nitrogen atoms form interlocking hexagonal rings, each with 3 boron atoms and 3 nitrogen atoms, and the layers are held together by van der Waals forces (Figure 1). 5 There is no boron–nitrogen bonding between the layers. 6 A spherical form, with a hexagonal crystal structure, can also occur. 7 Additionally, boron nitride can be in cubic form in which alternately linked boron and nitrogen atoms form a tetrahedral bond network (Figure 2). 5
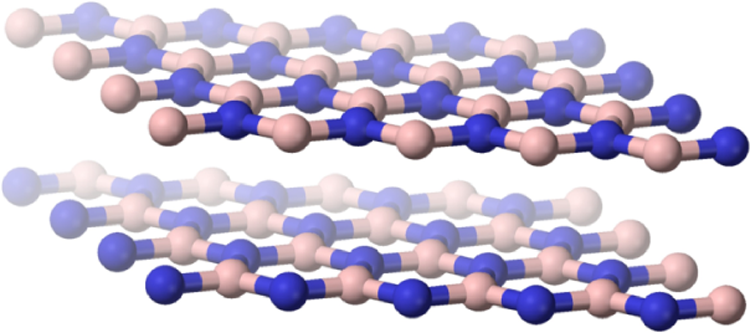
Hexagonal boron nitride. Adapted from Barron. 15
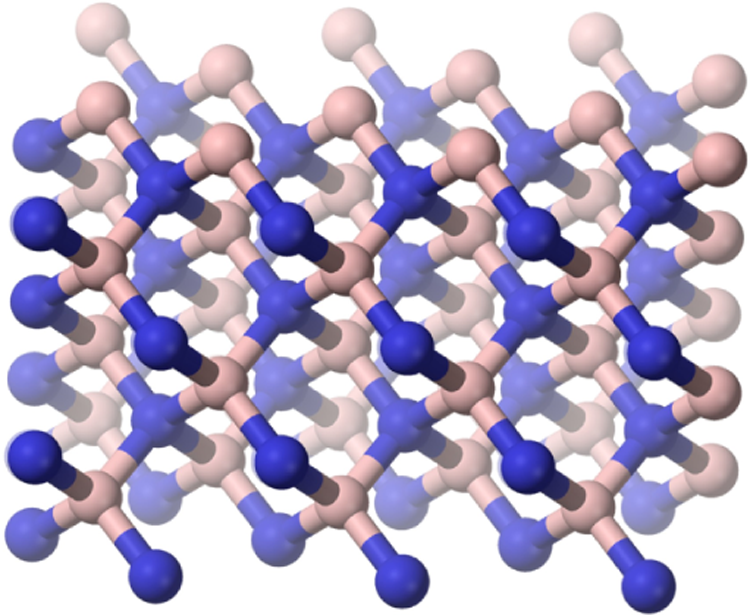
Cubic boron nitride. Adapted from Barron. 15
Physical and Chemical Properties
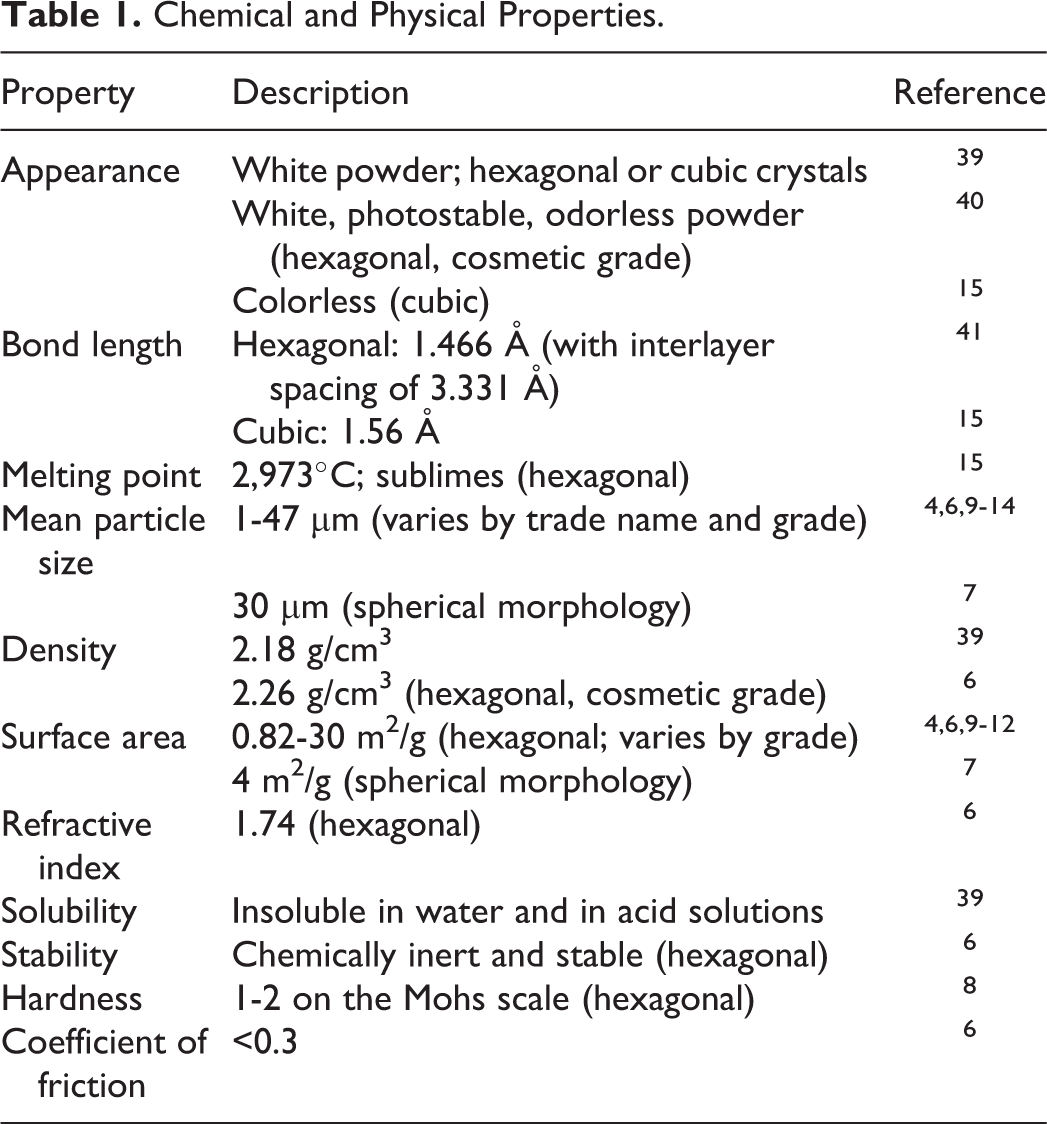
The physical and chemical properties of boron nitride are provided in Table 1. The 3-ring, layered structure gives hexagonal boron nitride a lubricating effect, high thermal stability, and oxidation resistance. 8 The mean particle size of cosmetic-grade hexagonal boron nitride powder varies by trade names and grade, with sizes of 1 to 47 µm reported. 4,6,9 -14 Cubic boron nitride is the second hardest material known, with diamond being the hardest. 5
Chemical and Physical Properties.
Method of Manufacture
Cosmetic-grade hexagonal boron nitride is synthesized using borax and a nitrogen source at a high temperature, resulting in a lamellar structure. 6 Industrial synthesis of hexagonal boron nitride powder is based on a 2-stage process. 8 First, at approximately 900°C, a boron source such as boron oxide or boric acid is converted in a reaction with a nitrogen source, mostly melamine, urea, or possibly, ammoniac, to amorphous boron nitride. In the second step, crystallization occurs at temperatures usually >1,600°C in a nitrogen atmosphere, and a transition into hexagonal boron nitride occurs. At high temperatures (600-2,000°C) and pressures (50-200 kbar), hexagonal boron nitride can be converted to the cubic phase. 15
Composition/Impurities
The typical chemical analysis of hexagonal cosmetic-grade boron nitride is ≥98 to 99.8% BN, 0.2% to 1.6% O2, 0.02% to 0.1% B2O3, and <1 ppm metals (such as Hg, As, and Pb). 4,6,9 -12
Use
Cosmetic
Boron nitride is reported to function in cosmetics as a slip modifier, which means it purportedly helps other substances to flow more easily and more smoothly, without reacting chemically, much like graphite. 1,16 This ingredient is a type of ceramic, having the smoothness and the chemical and thermal inertness associated with most ceramics. 9 According to the information supplied to the US Food and Drug Administration (FDA) by industry, as part of the Voluntary Cosmetic Registration Program (VCRP), the data reported (2013) that boron nitride is used in 643 cosmetic formulations (Table 2). 17 Data received in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council) reported that boron nitride is used at concentrations up to 25%; the highest concentration of use is in eye shadow formulations. 18
Frequency and Concentration of Use According to Duration and Type of Exposure.
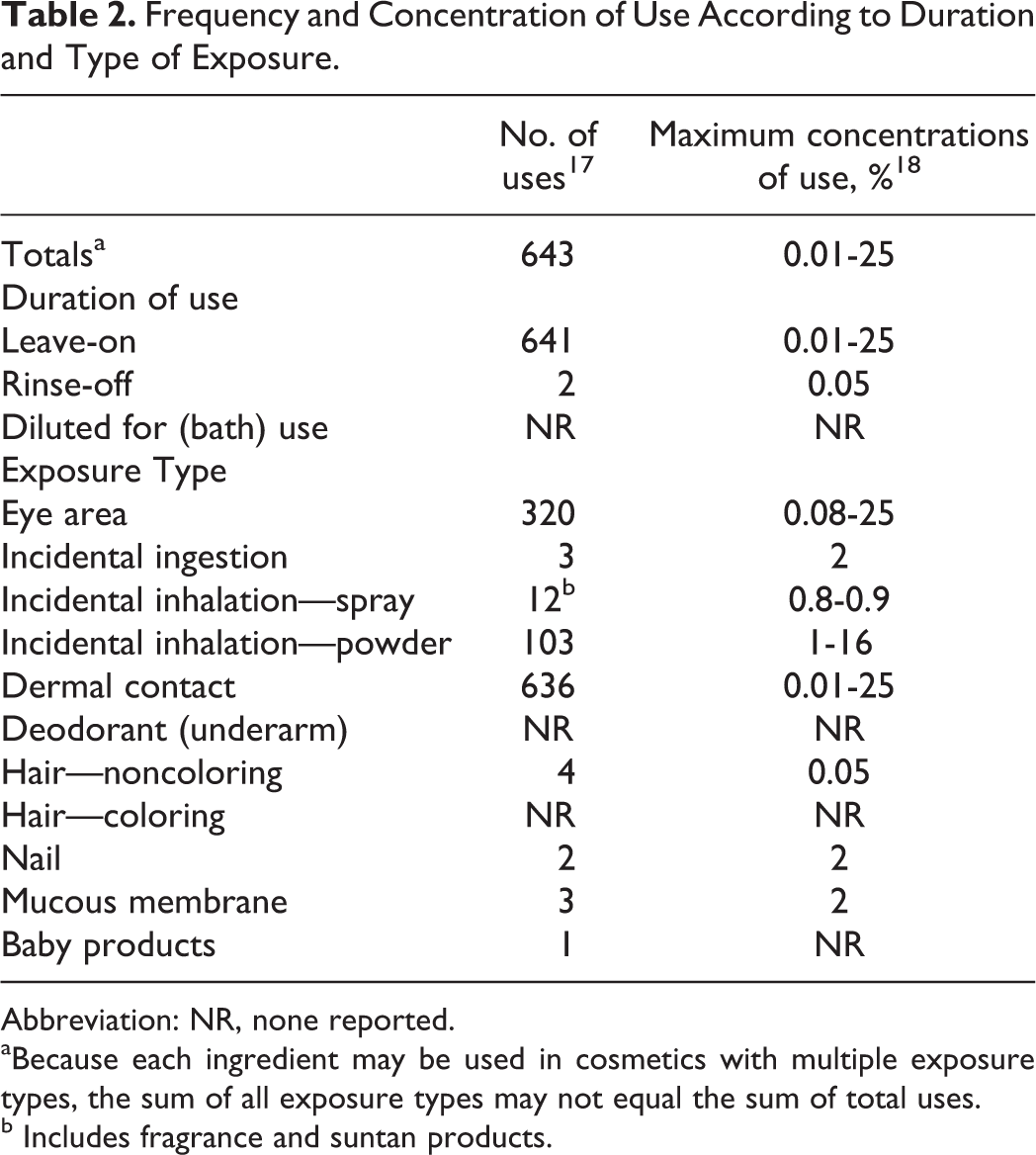
Abbreviation: NR, none reported.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
b Includes fragrance and suntan products.
Although use in baby products was reported to the VCRP, concentration of use data were not reported by industry. Products containing boron nitride may be applied to the eye area (≤25% in eye product formulations), could possibly be ingested (2% in lipstick formulations), or may possibly be inhaled (≤16% in powders and at up to 0.9% in fragrance preparations). In practice, 95% to 99% of the particles released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 μm range. 19,20 Therefore, most particles incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable to any appreciable amount. 21,22 Boron nitride is listed in the European Union inventory of cosmetic ingredients, and no restrictions are specified. 23
Noncosmetic Use
The properties of hexagonal boron nitride allow for its use in a number of technological processes. Different forms of hexagonal boron nitride are used for different applications: boron nitride, compacted by hot pressing or hot isostatic pressing, can be used in making shaped components for various industries; boron nitride-coating systems using suspensions and sprays are used to selectively change the properties of substrate surfaces; and powdered boron nitride-coating systems using powders and granules can be used in thermal management. 8 Hexagonal boron nitride can be used as an electrical insulator; as thermocouple protection sheaths, in crucibles and linings for reaction vessels; and as a coating for refractory molds used in glass forming and in superplastic forming of titanium. 24 It can also be incorporated in ceramics, alloys, resins, plastics, and rubber to give them self-lubricating properties. 5 Hexagonal boron nitride is used in the formulation of coatings and paints for high-temperature applications, and it is also used as a substrate for semiconductors, lens coatings, and transparent windows. 25
Hexagonal boron nitride can be made in single layers to form nanotubes, and bundles of boron nanotubes are used for wire sleeving. 5 Boron nitride nanotubes are being produced commercially. 26
Cubic boron nitride is used as an abrasive and wear-resistant coating; it is used for cutting tools and as abrasive components for shaping/polishing with low carbon ferrous metals. 5
Toxicokinetics
In a pharmacokinetics study, 4 New Zealand white rabbits were given an intravenous (IV) injection of 10 mg/kg body weight (bw) sterilized glycol chitosan (G-chitosan)-coated boron nitride nanotubes in physiological solution. 27 Blood samples were taken at various intervals for up to 10 hours. Boron concentration was measured as an indicator of the presence of boron nitride nanotube. The terminal half-life was 98 ± 37 minutes. The researcher stated that an absence of multiexponential kinetic components indicated no significant temporary accumulation of boron nitride nanotubes in tissues. The researchers also stated that the data suggested a “relatively high” clearance of boron nitride nanotubes from the blood and a quick distribution and/or excretion but also noted that the functionalization of the nanoparticles has a role in their behavior at a systemic level.
Toxicological Studies
Single-Dose (Acute) Toxicity
Parenteral
A single IV injection of 1, 5, or 10 mg/kg bw coated boron nitride nanotubes did not produce toxicity in rabbits. 27,28 In a pilot study, 3 New Zealand White rabbits received a single 1 mg/kg IV dose of G-chitosan boron nitride nanotubes in physiological saline at a concentration of 1 mg/mL. 28 Two control rabbits were given an IV dose of G-chitosan only. Blood samples were collected at 0, 2, 24, and 72 hours. No signs of toxicity were observed, and with the exception of an increased platelet count at 72 hours (that was still in the reference range), no differences in hematological parameters were observed between treated and control animals.
In a follow-up study, groups of 3 male New Zealand white rabbits were given a single IV dose of 5 or 10 mg/kg sterilized G-chitosan boron nitride nanotubes in physiological solution (average hydrodynamic size 500 nm). 27 Blood samples were collected 0, 1, 3, and 7 days after dosing. No signs of toxicity were observed, and the results of the hematological analysis were within normal limits.
Repeated Dose Toxicity
Parenteral
G-chitosan boron nitride nanotubes also did not produce toxicity in a repeated dose study. 27 Three male New Zealand white rabbits were dosed by IV injection with 5 mg/kg bw/day sterilized G-chitosan boron nitride nanotubes in physiological solution for 3 consecutive days, and blood samples were taken as mentioned previously. As in the single-dose studies, no signs of toxicity were observed and results of the hematological analysis were within normal limits.
Cytotoxicity
Contradictory results have been reported on the cytotoxicity of boron nitride nanotubes; this may be due to that fact the nanotubes were coated in one study but not in the other. In one study, boron nitride nanotubes (not coated) were found to be cytotoxic when evaluated using lung epithelial cells (A549), alveolar macrophages (RAW 264.7), and fibroblast cells (3T3-L1), as well as in human embryonic kidney cells (HEK 293). 26 Tween 80 was used as the dispersing agent and yielded a suspension of individual nanotubes with long-term stability; the typical diameters were less than 80 nm (average of ∼50 nm), and the average length was ∼10 µm. Cells were exposed to 0.02, 0.2, 2, and 20 µg/mL of the nanomaterial. In a 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay of cell proliferation, 2 µg/mL boron nitride nanotubes produced a decrease in the MTT signal when compared to the untreated cells. At 48 hours, this result was time-, dose-, and cell type-dependent, with the greatest effect observed in cells with high endocytic (phagocytic) activity, that is, macrophages, and the lowest toxicity observed in cells with the lowest endocytic activity, that is, HEK 293 cells. A fluorometric DNA assay and fluorometric microculture assay were performed with lung A549 epithelial cells and RAW 264.7 macrophage cells to confirm these results, and exposures of 0.2, 2, and 20 µg/mL nanomaterial were used. Similar results were obtained; a decrease of 60% to 80% in viable cell number was observed with 2 µg/mL in macrophage cells, and a 30% loss was observed with 20 µg/mL in A549 epithelial cells.
The results presented previously contradict earlier studies on boron nitride nanotubes that did not report cytotoxicity. In one study using human neuroblastoma SH-SY5Y cells, up to 100 µg/mL G-chitosan–coated boron nanotubes were tested in 2 cell proliferation assays, an MTT-assay and a 2-(4-iodophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl-2H-tetrazolium monosodium salt (WST-1) assay, a DNA content assessment, a viability/cytotoxicity assay, an early apoptosis detect assay, and a reactive oxygen species (ROS) assay. 29 A statistically significant reduction in MTT was observed at a concentration of 20 µg/mL boron nitride nanotubes, but this reduction was not observed with G-chitosan alone or in WST-1. The researchers stated that, most likely, the boron nitride nanotubes interact with some tetrazolium salts, such as MTT, but not with others, such as WST-1, suggesting that the interference is due to the water insoluble nature of MTT formazan. The boron nitride nanotubes did not affect the DNA concentration, cell viability, apoptosis, or ROS formation.
In another study, HEK 293 cells were cultured with 100 mg/mL boron nitride tubules, which formed aggregates in culture media. 30 Cells cultured with boron nitride nanotubules were not different from cells cultured in media. Similar results were found with Chinese hamster ovary (CHO) cells.
The Expert Panel noted that all of these studies should be viewed with caution because the very low solubility of the test compounds requires solution vehicles that could bias the results. The exposures to cells in vitro are very different from the ways in which boron nitride might be presented to skin or epithelia in humans.
Reproductive and Developmental Toxicity
Published reproductive and developmental toxicity studies were not found, and unpublished data were not provided.
Genotoxicity
Published genotoxicity studies were not found, and unpublished data were not provided.
Carcinogenicity
Published carcinogenicity studies were not found, and unpublished data were not provided.
Irritation and Sensitization
Dermal
Human
Boron nitride was a weak skin irritant in a 24-hour human patch test. 31 Occlusive patches of boron nitride, 50% in olive oil, were applied to 20 participants. Two participants exhibited a ±response (Additional details were not provided).
Eye shadow formulations containing 13% boron nitride, tested at a concentration of 70% in squalane oil, 32 and 18.7% boron nitride, tested neat, 33 and face powder formulations containing 16% boron nitride 34,35 were not dermal irritants or sensitizers in human repeated insult patch tests (HRIPTs). The details of these studies are presented in Table 3.
Human Repeated Insult Patch Tests.

aThe challenge followed a 2-week nontreatment period in each study.
Ocular
Nonhuman
A short-term exposure (STE) test was performed to evaluate the irritation potential of boron nitride. An STE is an in vitro assay for ocular irritation that assesses cytotoxicity in a rabbit corneal cell line (SIRC cells) following a 5-minute treatment. 36 Boron nitride, tested at 0.05 and 5% in mineral oil, was ranked a minimal irritant in an STE test. 31
Results of chorioallantoic membrane (CAM) assays also predicted that boron nitride is not an ocular irritant. 31 Boron nitride was assayed at 50% in Vaseline using the hen’s–egg test-CAM (HET-CAM; which determines hemorrhage, lysis, and coagulation in the CAM of the egg after exposure to test chemicals 37 ) and the CAM-trypan blue staining method (CAM-TB; which measures the amount of trypan blue adsorbed onto the CAM as the end point of the assay 37 ).
Human
An eye shadow containing 13% boron nitride did not cause any adverse effects in a 2-week use study. 38 Thirty female participants completed the study, 17 of whom wore contact lenses. The eye shadow was applied at least 1 time daily.
Summary
Boron nitride is an inorganic compound that can be hexagonal, spherical, or cubic in form. Hexagonal boron nitride is similar to graphite, whereas cubic boron nitride is similar to diamond. The mean particle size of cosmetic-grade hexagonal boron nitride powder varies by trade name and grade, with mean particle sizes of 1 to 47 μm reported. Boron nitride nanotubes are produced commercially; however, boron nitride is not listed as a nanomaterial used in cosmetic formulations.
Boron nitride is reported to function as a slip modifier in cosmetics, and it is presumed that this means the hexagonal form is used. The VCRP data report 643 uses of boron nitride, and concentrations of use of up to 25% were reported by the cosmetics industry, with the highest use concentration reported in eye shadow formulations.
In a pharmacokinetic study in which 4 New Zealand white rabbits were given an IV injection of 10 mg/kg bw sterilized glycol chitosan-coated (G-chitosan) boron nitride nanotubes in physiological solution, no significant temporary accumulation of boron nitride nanotubes in tissues was found. The researchers concluded there was a relatively high clearance of boron nitride nanotubes from the blood and a quick distribution and/or excretion.
A single IV injection of 1, 5, or 10 mg/kg G-chitosan boron nitride nanotubes did not produce toxicity in rabbits. Toxicity also was not observed following repeated dosing with 5 mg/kg bw/day sterilized G-chitosan boron nitride nanotubes in physiological solution for 3 consecutive days.
Contradictory results have been reported on the cytotoxicity of boron nitride nanotubes. One study found that boron nitride nanotubes were cytotoxic when evaluated using lung epithelial A549 cells, alveolar macrophages RAW 264.7 cells, fibroblast 3T3-L1 cells, and human embryonic kidney HEK 293 cells, whereas another study reported that boron nitride was not cytotoxic to HEK 293 or CHO cells. In human neuroblastoma SH-SY5Y cells, up to 100 µg/mL G-chitosan–coated boron nanotubes caused a statistically significant reduction in MTT at a concentration of 20 µg/mL boron nitride nanotubes, but this reduction was not observed with G-chitosan alone or in WST-1 and was attributed to the water insoluble nature of MTT formazan. Boron nitride nanotubes did not affect DNA concentration, cell viability, apoptosis, or ROS formation in these cells.
Boron nitride, 50% in olive oil, was a weak skin irritant in a 24-hour human patch test. Eye shadow formulations containing 13% boron nitride, tested at a concentration of 70% in squalane oil, and 18.7% boron nitride, tested neat, and face powder formulations containing 16% boron nitride were not dermal irritants or sensitizers in HRIPTs.
In nonhuman testing, boron nitride, tested at 0.05% and 5% in mineral oil, was ranked a minimal irritant in an STE test. Additionally, boron nitride, assayed at a concentration of 50% in Vaseline, was negative in a HET-CAM assay and a CAM-TB assay. In human testing, an eye shadow containing 13% boron nitride did not produce any adverse effects in a 2-week use study.
Discussion
Human repeat insult patch test data were available on formulations that contained boron nitride at concentrations up to 18.7%. Although sensitization data were not provided at the maximum reported leave-on use concentration of 25%, the Expert Panel concluded that the existing data were adequate. At a 50% concentration, boron nitride caused minimal irritation. At 18.7%, it was not a sensitizer nor would it be expected to sensitize given its inability to penetrate the stratum corneum. Additionally, boron nitride is not expected to be an ocular irritant at the maximum reported concentration of use; in a 2-week use study, an eye shadow containing 13% boron nitride did not cause any adverse effects.
The Expert Panel stated that although there are data gaps, this ingredient is stable, chemically inert, and completely insoluble, and the extended hexagonal crystal lattice structure makes boron nitride a very large molecule that is not expected to penetrate the stratum corneum. Even if it were to penetrate, it would not be expected to have chemical activity in biological systems. Although boron nitride is not reported to be used in nanomaterial form in cosmetic formulations, the lack of toxicity observed in single- and repeated-dose IV studies with coated boron nitride nanotubes further supported the safety of boron nitride in other forms. Also, the fact that boron nitride is not expected to penetrate the skin precluded the need for repeated dose toxicity, reproductive and developmental toxicity, and carcinogenicity studies to complete this safety assessment.
The Panel discussed the issue of incidental inhalation exposure to boron nitride from powders and from products that may be aerosolized. Boron nitride is reportedly used at up to 16% in products that may become airborne (ie, in face powders) but only at very low concentrations in cosmetic products that may be aerosolized (ie, up to 0.9% in fragrance preparations). Because boron nitride is not expected to have chemical activity in biological systems, particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products that may be aerosolized is available at
Conclusion
The CIR Expert Panel concluded that boron nitride is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author Contributions
M. Fiume contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. L. Gil, F. A. Andersen, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Lillian Gill, Director, Cosmetic Ingredient Review, Washington, DC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
