Abstract
The Cosmetic Ingredient Review Expert Panel (the Panel) reviewed the safety of synthetic fluorphlogopite as used in cosmetics. Synthetic fluorphlogopite functions as a bulking agent and a viscosity-increasing agent. The Panel reviewed available animal and human data related to this ingredient along with a previous safety assessment of other magnesium silicates. The Panel concluded that synthetic fluorphlogopite was safe as cosmetic ingredients in the practices of use and concentration as given in this safety assessment.
Introduction
This is a safety assessment of the cosmetic ingredient synthetic fluorphlogopite (sometimes spelled fluorophlogopite). As given in the International Cosmetic Ingredient Dictionary and Handbook, 1 synthetic fluorphlogopite is a synthetic mimic of a natural mineral that functions in cosmetics as a bulking agent and a viscosity-increasing agent—aqueous.
Synthetic fluorphlogopite is partially composed of magnesium aluminum silicate sheets. The silicate clay, magnesium aluminum silicate, as well as magnesium trisilicate, zeolite, and other clays were reviewed previously by the Cosmetic Ingredient Review Expert Panel (the Panel) as part of a group of aluminum silicate clays and found to be safe as used in cosmetic products. 1 Summaries of the relevant data from that report are included in the appropriate sections subsequently. The similar chemical structures and physicochemical properties as well as functions and concentrations in cosmetics of magnesium aluminum silicate and related clays enable referring to these ingredients and reading across the available toxicological data to support the assessment of synthetic fluorphlogopite.
Chemistry
Definition and Structure
Synthetic fluorphlogopite (CAS No 12003-38-2) is a synthetic mimic of a mica-type, fluorine-substituted mineral composed of magnesium aluminum silicate sheets, weakly bound together with potassium (Figure 1). 3
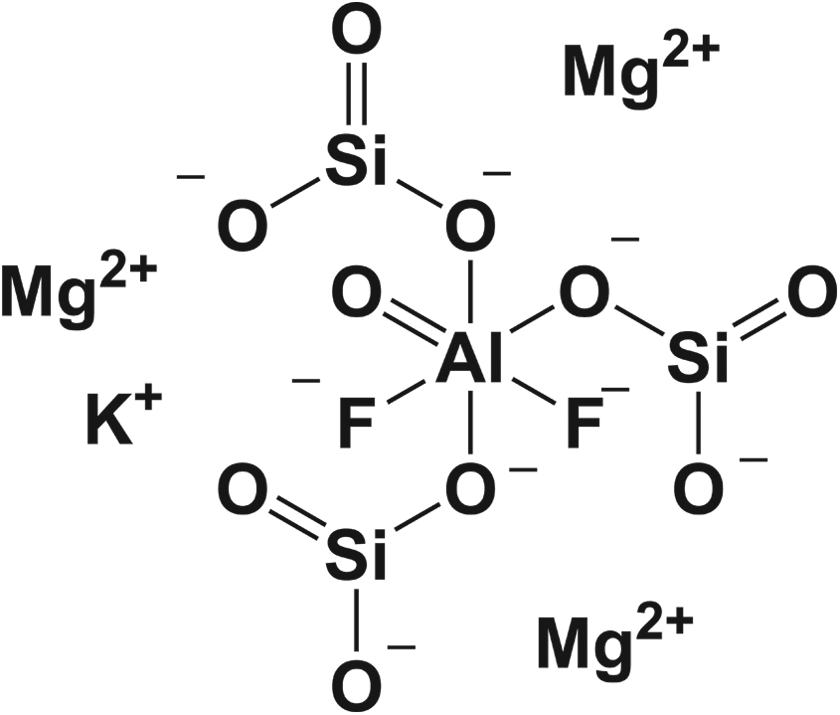
The structure and average formula of synthetic fluorphlogopite. 2
Phlogopite, the nonfluorine-substituted mineral, like other micas, has a layered structure of magnesium aluminum silicate sheets weakly bonded together by layers of potassium ions. 4 These potassium ion layers produce the perfect cleavage. Single large plates or “books” of phlogopite can grow to considerable size.
Fluorphlogopite differs in that 2 of the hydroxyl groups, per aluminum atom, are replaced with fluorine atoms. Fluorine is present in the phyllosilicate mineral group in general and in the micas particularly as a substitute for OH. The presence of fluorine enhances the thermal stability of the trioctahedral mica structure.
Physical and Chemical Properties
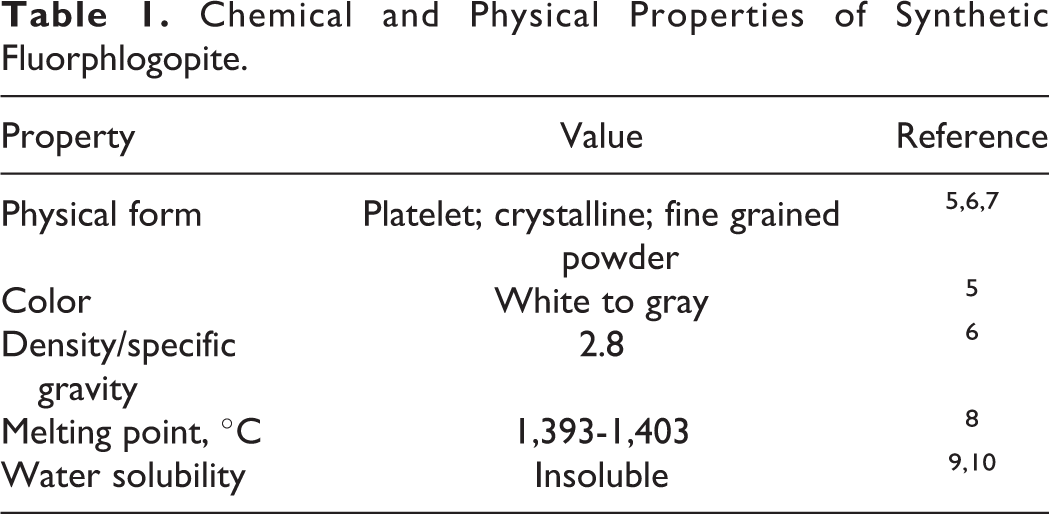
Physical and chemical properties of synthetic fluorphlogopite are presented in Table 1. In large pieces, fluorphlogopite is pale yellow and is transparent and nonfluorescent with vitreous to resinous luster, and it shows yellowish white color in thin section. 11 Mohs hardness is 2 to 3.
Chemical and Physical Properties of Synthetic Fluorphlogopite.
One source describes synthetic fluorphlogopite as a white to gray free-flowing powder with an average particle size of 10 to 15 μm and a pH range of 5 to 8. 5 The particles have a low degree of surface reactivity (in contrast to natural phlogopites). Other sources report that synthetic fluorphlogopite has a pH value of 7.0 to 11.0 (in a 10% aqueous slurry), a bulk density of 0.240 to 0.300 g/cm3, and a particle size distributions of 9.0 to 45 μm 12 and 20 to 150 μm. 9
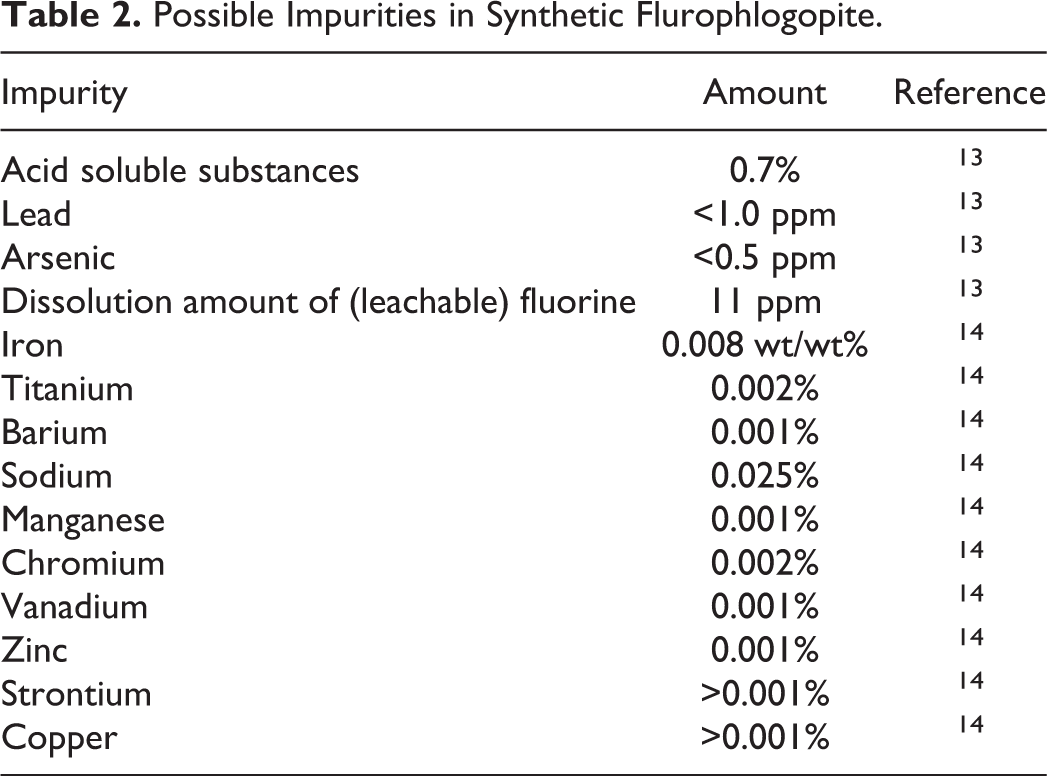
Possible impurities are listed in Table 2. Acid soluble substances are potentially leachable from synthetic fluorphlogopite, including fluorine ions. 14
Possible Impurities in Synthetic Flurophlogopite.
Synthetic fluorphlogopite is not soluble in water. 15 In a test of the solubility of the ions in distilled water, magnesium, aluminum, and potassium were present at <5 × 10−6, <5 × 10−5, and <5 × 10−4 g/L, respectively, after stirring for up to 72 hours at 30°C. The amount of potassium in the blank was similar to the amount in the test substance, suggesting that the detected potassium was in the water and not from the synthetic fluorphlogopite.
Synthetic fluorphlogopite, as opposed to natural fluorphlogopite, is virtually iron free. 11 However, synthetic fluorphlogopite may be intentionally manufactured with iron to more efficiently absorb UV rays. 14
Fluorphlogopite has some unique properties due to the replacement of most of the hydroxyl groups on aluminum (that are normally present in nonfluorphlogopite) with fluoride. However, the aluminum–fluoride bond is only moderately thermodynamically stable. Over time and exposure, atmospheric oxygen and water can replace those fluorides ions, regenerating the more stable hydroxyl groups.
Synthetic fluorphlogopite is stable for 5 years in a sealed container at <25°C and for at least 1 year once opened. 9 However, fluorine ions (F−) are reported to leach out of synthetic fluorphlogopite particles and bricks. 10,16
The Japanese Standard of Quasi-Drugs requires that synthetic fluorphlogopite has a pH between 5.5 and 7.5, a maximum of 2% acid soluble substance, ≤20 ppm lead, ≤5 ppm arsenic, and ≤20 ppm dissolution amount of fluoride. 17 Synthetic fluorphlogopite meeting these standards are reported to be >99% pure.
Although the previously reviewed magnesium aluminum silicate clays mentioned earlier are natural clays and synthetic fluorphlogopite is not, they both are large, flat particles (0.8 × 0.8 × 0.1 μm) with a layered structure of magnesium aluminum silicate sheets weakly bonded together. They are both chemically inert and insoluble in water.
The previous safety assessment of magnesium aluminum silicate clays described hectorite (magnesium/lithium silicate) clay as containing fluorine, 1 which suggested a structural similarity to synthetic fluorphlogopite. However, current information suggests that fluorine is not a significant component of hectorite.
Method of Manufacture
A reported manufacturing method of synthetic fluorphlogopite designed for industrial scale batches (up to several tons) involved melting oxide–fluoride mixtures (metal; ie, aluminum and manganese) at a given “soak” temperature (wherein the contents are liquid; up to 1,450°C) and then cooling at a continuous rate of a few degrees per hour between 1,400°C and 1,300°C. 18 This technique produced large fluorphlogopite monocrystals (several centimeters).
An alternate method synthesized fluorphlogopite single crystals, several millimeters in size, suited to laboratory uses. 8 A mixture of SiO2, γ-Al2O2, MgO, and K2SiF6 was melted at 1,450°C for 3 hours, cooled to 1,385°C at a rate of 100°C/h, and then quenched into cold water. The resulting charge was loaded back into the furnace, heated from ∼1,000°C to 1,385°C at a rate of 500°C/h, and finally cooled at a rate of 1°C/h down to 1,300°C. This procedure led to the formation of large and detachable monocrystals of synthetic fluorphlogopite up to 1 cm in diameter.
The extent of fluorine substitution for OH groups depends on several factors. 11,19 The most important are (1) hydrofluoric acid activity in the fluid during the crystallization and the postcrystallization phase, (2) temperature, and (3) cation population of the octahedral sheet. Crystal structure may be altered by adjusting the pressure during cooling. 4
In order to minimize the number of fluoride ions available for leaching, the stoichiometric equivalent of fluorine is decreased to less than 1 (ie, less K2SiF6 is added) and the melt temperature is decreased (between 900 and 1,000ºC). 10 To remove any free fluoride, the resulting ingot is pulverized, and the powder is heat treated at 600°C to 1,350°C and washed with an aqueous solution containing one or more acids or chelating agents.
Use
Cosmetic
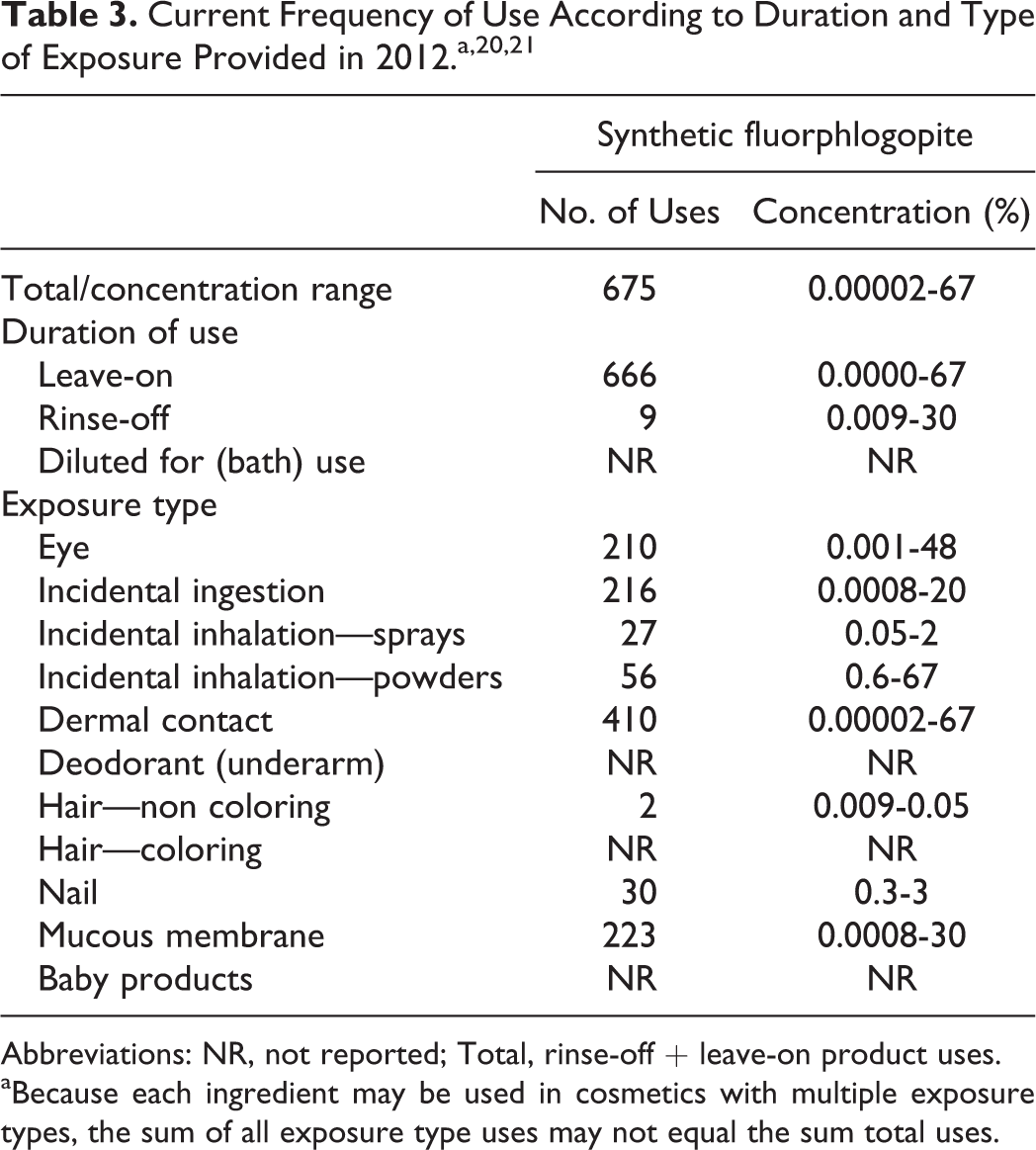
Data on ingredients usage are provided to the Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP). The VCRP reports that synthetic fluorphlogopite is used in 666 leave-on products and 9 rinse-off products (Table 3). 20,21 The Personal Care Products Council survey reports maximum use concentrations of 0.00002% to 67%. It is used up to 67% in leave-on products (face powders) and 30% in rinse-off products (bath soaps and detergents). It is used up to 20% in lipsticks and 15% in eye makeup products.
Abbreviations: NR, not reported; Total, rinse-off + leave-on product uses.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Synthetic fluorphlogopite, as used in perfumes with the maximum reported use concentration of 0.05% and in indoor tanning preparations at 2%, are possible propellant and pump spray products and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 22,23 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal region and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 24,25
Noncosmetic
Fluorphlogopite has been approved as a colorant in all types of food contact polymers at levels not to exceed 5.0% by weight of the finished polymer. 26 Natural phlogopites are used for their heat and electrical insulating properties. 11
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Synthetic fluorphlogopite
Data on the absorption, distribution, metabolism, and excretion of synthetic fluorphlogopite were not found in the published literature, and unpublished data were not provided.
Magnesium aluminum silicate and related clays
Although aluminum was not absorbed, elevated levels of silicon were observed in assayed plasma samples of dogs administered magnesium trisilicate (20 mg/kg) and zeolite (20 mg/kg) orally. 27 The urinary excretion of silica was 5.2% in male subjects given 20 g of magnesium trisilicate. 28 Heat-treated montmorillonite (5, 15, and 45 mg) administered to rats by means of intratracheal instillation was restricted to alveoli within and adjacent to alveolar ducts. 29
Cytotoxicity
Magnesium aluminum silicate and related clays
A sample of aluminum silicate was toxic to pulmonary alveolar macrophages measured by lactate dehydrogenase activity (LDH), β-galactosidase (β-GAL) activity, lactic acid production, cellular adenosine triphosphate activity, and the cellular DNA contents. 30 The LDH activity and β-GAL release were increased at 33.3 and 166.7 μg/mL aluminum silicate. Aluminum silicate had relatively no effect on the hemolysis of rat red blood cells (RBCs) up to 1,000 μg/mL.
Many clays (attapulgite, bentonite, hectorite, kaolin, montmorillonite, pyrophyllite, and zeolite) demonstrated cytotoxicity to several macrophage-type cell lines and have hemolytic activity toward several species’ RBCs (as low as 0.2 μg/mL attapulgite to bovine RBCs). 1,30 -63 Toxicity was dependent on particle size, concentration, and mineral composition. Larger particle size causes more adverse effects. In most of the studies, a dose-dependent effect on cytotoxicity or lysis was observed. Most mineral samples were not 100% pure, and many samples already contained toxic dusts or minerals like quartz or cristobalite.
Toxicological Studies
Acute Toxicity
Dermal
Magnesium aluminum silicate and related clays
The acute dermal median lethal dose (LD50) was >3.5 g/kg for rabbits exposed to VEEGUM (magnesium aluminum silicate) (Vanderbilt Minerals, (LLC in Norwalk, CT)). 64
Oral—nonhuman
Synthetic fluorphlogopite
The oral LD50 for female ICR (Crj: CD-1) mice and Sprague Dawley (Crj: CD) rats was >9,000 mg//kg synthetic fluorphlogopite. 65 There were no adverse clinical signs or physical findings at necropsy.
Magnesium aluminum silicate and related clays
The following are a list of acute oral LD50 determinations: calcium silicate, 3,400 mg/kg in rats; magnesium aluminum silicate, 50,000 mg/kg in mice; zirconium silicate, >200 mg/kg in mice; hectorite, >5 g/kg in rats; and kaolin, 149 g/kg in rats (death due to bowel obstruction). 66 -70
Inhalation
Synthetic fluorphlogopite
In a 4-hour acute inhalation study, synthetic fluorphlogopite (4.1 mg/L; median aerodynamic diameter of 2.44 μm with a geometric standard deviation of 2.18 and 2.20 μm) was administered to HanRcc:WIST (SPF) albino rats (n = 5/sex) in a nose-only system. 71 There were no clinical signs during exposure and during the 15-day observation period. There were no findings at necropsy. The LC50 was determined to be >5.1 mg/L.
Repeated Dose Toxicity
Oral—nonhuman
Magnesium aluminum silicate and related clays
In short-term oral toxicity studies, no adverse effects were seen in mice or rabbits dosed up to 5 g/kg magnesium aluminum silicate. 72 Various zeolites (up to 3%) were added to the diets of pigs for 6 weeks. 73 No adverse effects were noted by the supplementation.
A feeding test with dogs and rats ingesting large amounts of VEEGUM (magnesium aluminum silicate, 10% of ration) for 90 days showed that there were no adverse effects, and VEEGUM was considered nontoxic. 74
Guinea pigs had renal lesions after 4 months of drinking magnesium trisilicate (250 mg/L) in their tap water. 75 Rats fed 10% magnesium aluminum silicate had slightly elevated silicon levels of the spleen, and dogs and rats fed 10% VEEGUM had no adverse effects in the 90-day feeding studies. 74 No lesions were found in rats dosed up to 1,000 mg/kg for 104 weeks. 76
Occupational Exposure
Magnesium Aluminum Silicate and Related Clays
Occupational exposure to mineral dusts has been studied extensively. Fibrosis and pneumoconiosis were documented in workers involved in the mining and processing of aluminum silicate, calcium silicate, zirconium silicate, Fuller’s earth, kaolin, montmorillonite, pyrophyllite, and zeolite.
Reproductive and Developmental Toxicity
Synthetic Fluorphlogopite
Data on the reproductive or developmental toxicity of synthetic fluorphlogopite were not found in the published literature, and unpublished data were not provided.
Magnesium Aluminum Silicate and Related Clays
Calcium silicate (250-1,600 mg/kg on days 6-18 of gestation) had no discernible effect on nidation or on maternal or fetal survival in rabbits. 77 Magnesium aluminum silicate (6,000 mg/kg on days 7-12 of gestation) had neither teratogenic nor adverse effects on the mouse fetus. 78
Female rats receiving a 20% kaolin diet for up to 117 days prior to insemination through nidation exhibited maternal anemia, but no significant decrease in birth weight of the pups was recorded. 79 Type A zeolite produced no adverse effects on the dam, embryo, or fetus in either rats (administered days 6-15) or rabbits (administered days 6-18) at any dose level (74 or 1,600 mg/kg). 80 Clinoptilolite (5%) administered in feed for 13 weeks had no effect on female rat reproductive performance. 81
Genotoxicity
Synthetic Fluorphlogopite
In a chromosomal aberration test of synthetic fluorphlogopite (0.02-0.16 mg/mL), there were no chromosomal structural abnormalities observed to Chinese hamster lung-derived fibroblast cells at 24 and 48 hours. 65 Synthetic fluorphlogopite (0.500-5,000 μg) was not mutagenic to Salmonella typhimurium (strains TA98, TA100, TA102, TA1535, TA1537) and Escherichia coli (strain WP2 uvrA) with and without metabolic activation. 82
In a micronucleus test using V79 Chinese hamster cells, synthetic fluorphlogopite (5-100 μL) was not mutagenic with or without metabolic activation. 83 Controls had the expected results.
In an in vitro mammalian cell gene mutation test, synthetic fluorphlogopite (15.8-500 μg/mL) was not mutagenic with or without metabolic activation to V79 Chinese hamster cells. 84 Controls had the expected results.
Magnesium Aluminum Silicate and Related Clays
No increase in mutation frequencies was seen in the Salmonella TA1530 or G-46 assay, and no significant increase in recombinant activity was observed in the Saccharomyces D3 assay treated with calcium silicate. 68 Calcium silicate (150 mg/kg) orally administered to rats produced 3% breaks in bone marrow cells arrested in c-metaphase. In a metaphase spread of bone marrow cells, calcium silicate produced no significant increase in the number of aberrations compared to controls. In a dominant lethal assay using rats, calcium silicate (up to 1,500 mg/kg) did not induce any dominant lethal mutations.
In the S. typhimurium LT2 spot test (TA98, TA100, TA1535, TA1537, and TA1538) with or without metabolic activation, magnesium aluminum silicate and hectorite were found to be nonmutagenic. 85,86 In primary hepatocyte cultures, the addition of attapulgite at 10 μg/cm2 had no significant unscheduled DNA synthesis (UDS) response or modulated response to 2-acetylaminofluorene, (AAF) (a positive control), and attapulgite at 10 μg/cm2 caused significant increases in UDS in rat pleural mesothelial cells. 33,87 Zeolite particles (10 μm) produced an increase in the percentage of aberrant metaphases, mostly chromatid breaks. 88
Carcinogenicity
Synthetic Fluorphlogopite
No published carcinogenicity studies of synthetic fluorphlogopite were found, and unpublished data were not provided.
Inhalation
Magnesium aluminum silicate and related clays
Small primary neoplastic lesions were found in 2 of 48 rats exposed to a calcium silicate dust at a concentration of 10 mg/m3 for 7 h/d, 5 d/wk, for a total of 224 days over an elapsed period of 12 calendar months in an inhalation chamber. 89 Moderate to extensive respiratory disease was noted in rats chronically exposed to synthetic zeolite A (20 mg/m3; 5 h/d, 3 d/wk) by inhalation methods. 76
Other exposures
Magnesium aluminum silicate and related clays
Intratracheal injections of aluminum silicate in rats caused lesions in a dose-dependent manner, and the intrapleural injections of 4 different aluminum silicate samples resulted in lesions. 90 One aluminosilicate injection caused 3 malignant mesotheliomas, 1 pleural and 2 peritoneal.
No mesotheliomas developed in rats injected intraperitoneally with 25 mg of calcium silicate dust. 89 Subcutaneous injection into the oral mucosa and into the back, periosteal injections into the periosteal tissue, and intramuscular injections into the thigh of rats and guinea pigs with zirconium silicate resulted in mild inflammatory reactions. 70 Minor inflammatory reactions, but no lesions, were found in rats given intratracheal injections of zeolite (clinoptilolite) and intraperitoneal injections of mordenite, synthetic zeolite 4A, and synthetic zeolite MS5A (1 mesothelioma was seen in rats given MS4A). 76,91 -96 An intrapleural injection of nonfibrous Japanese zeolite caused 2 mesotheliomas in rats, 96 and the subplantar injections of bentonite caused granulomas in rats. 97 In a series of intrapleural injections of rats, kaolin was used as a negative control. 98
Irritation and Sensitization
Irritation
Dermal—nonhuman
Synthetic fluorphlogopite
Synthetic fluorphlogopite (55% in distilled water; 0.3 g) was not irritating to the scratched skin of male Japanese white rabbits (n = 6) after 24 hours under occlusion. 65 In another study, synthetic fluorphlogopite (55% in distilled water; 0.3 g; 5 d/week) was not dermally irritating to Hartley albino guinea pigs (n = 10) in a cumulative irritation test after 4 weeks. 65 There were no effects on body weights, and necropsy did not reveal any changes in the organs. Synthetic fluorphlogopite (0.5 g with a few drops of aqua ad iniectabilia; ∼6 cm2 semiocclusion patch) was not a dermal irritant to female Crl:KBL (New Zealand White Rabbits [NZW]) rabbits after 4 hours. 99
Magnesium aluminum silicate and related clays
Magnesium aluminum silicate (4%) was a weak primary skin irritant in rabbits and had no cumulative skin irritation in guinea pigs. No gross effects were reported in any of these studies. 64
Dermal—human
Synthetic fluorphlogopite
In a dermal patch test (n = 42), a paste of synthetic fluorphlogopite (0.05 g in distilled water; 55%; 1.6-cm-diameter patch) was not irritating after 48 hours under occlusion. 65
A powdery foundation containing synthetic fluorphlogopite (38.5%; diluted in distilled water to 21.2%) was tested in a 48-hour patch test. The test substance was not irritating. 65
In an in vitro human skin model test, tissue treated with synthetic fluorphlogopite (1 mg) had a viability >50%, therefore, was considered to lack irritant potential. 100 Controls had the expected results.
Magnesium aluminum silicate and related clays
Applications of 2 g of VEEGUM made to the skin of 2 humans daily for 1 week caused no effects. 65
Ocular—nonhuman
Synthetic fluorphlogopite
In a Hen’s egg test-chorioallantoic membrane (HET-CAM) eye irritation potential test, synthetic fluorphlogopite (100%) demonstrated no irritation potential. 101 In a bovine corneal opacity and permeability assay (BCOP), synthetic fluorphlogopite (20% in 0.9% sodium chloride solution) did not increase opacity or permeability of the treated corneas. 102 The author concluded that synthetic fluorphlogopite did not show ocular irritant or corrosive potential. Controls had the expected results.
In a primary eye irritation test using albino Crl:KBL (NZW) rabbits (n = 3), synthetic fluorphlogopite (0.1 g) was not an ocular irritant. 103 All animals had discharge and redness of conjunctiva 1 hour after administration. One rabbit had chemosis at 1 hour after administration.
Magnesium aluminum silicate and related clays
A 4% solution of magnesium aluminum silicate and a 4% solution of sodium magnesium silicate caused minimal eye irritation in a Draize eye irritation test. 74,74 Bentonite (1-5 mg/mL) caused severe iritis after injection into the anterior chamber of the eyes of rabbits. 104 When injected intralamellarly (0-0.25 mg/mL), widespread corneal infiltrates and retrocorneal membranes were recorded. In a primary eye irritation study in rabbits, hectorite (100 mg) was moderately irritating without washing and practically nonirritating to the eye with a washout. 67 Rats tolerated a single 10 g dose of zeolite A without any adverse reaction in the eye. 76
Sensitization
Nonhuman
Synthetic fluorphlogopite
In a modified guinea pig maximization test (n = 10) with adjuvant exposure, synthetic fluorphlogopite (55% in distilled water; 0.1 g) was not sensitizing. 65 Synthetic fluorphlogopite (5%, 10%, 25% wt/vol in acetone:olive oil 4:1) was not a dermal sensitizer in a local lymph node assay using mice (n = 5). 105
Human
Synthetic fluorphlogopite
A human repeated insult patch test (n = 107) was conducted of a pressed powder that contained synthetic fluorphlogopite (13.824%) under semiocclusion. 106 The authors concluded that this product did not elicit dermal irritation or sensitization.
Photosensitization/Phototoxicity
Synthetic fluorphlogopite
Repeated administrations (days 1-4) of synthetic fluorphlogopite (55% in distilled water; 0.1 g) in an adjuvant strip assay were not dermally photosensitizing to female Hartley albino guinea pigs (n = 10) when exposed to UV-A light (320-400 nm) at 10.2 J/cm2 and then challenged on day 21 (20 mg). 65 When synthetic fluorphlogopite (55% in distilled water) was administered to the shaved skin of male Hartley albino guinea pigs (n = 5) prior to a single exposure to UV-A light (320-400 nm) at 14.0 joules/cm2 for 30 minutes, no skin reaction was observed at 24, 48 and 72 hours after exposure. 65
Summary
Synthetic fluorphlogopite is a synthetic mimic of a natural mica-type mineral that functions in cosmetics as a bulking agent and a viscosity-increasing agent—aqueous. Synthetic fluorphlogopite is composed of magnesium aluminum silicate sheets, weakly bound together with potassium. Magnesium aluminum silicate clays were reviewed previously by Cosmetic Ingredient Review (CIR) with the conclusion that they are safe as used in cosmetic products.
According to information supplied to the FDA by industry as part of the VCRP in 2011, synthetic fluorphlogopite is used in 560 leave-on products and 5 rinse-off products. Furthermore, results from a survey of ingredient use concentrations provided by the Personal Care Products Council in 2011 indicate that synthetic fluorphlogopite was being used at concentrations up to 67% in leave-on products (face powders) and up to 30% in rinse-off products (bath soaps and detergents).
There was no absorption of aluminum, and elevated levels of silicon were observed in assayed plasma samples of dogs administered magnesium trisilicate and zeolite orally. Silica was excreted in the urine of human males administered magnesium trisilicate orally.
The acute dermal LD50 for magnesium aluminum silicate was >3.5 g/kg for rabbits. The oral LD50 for mice and rats was >9,000 mg/kg synthetic fluorphlogopite. The acute oral LD50 for calcium silicate was 3,400 mg/kg in rats; 50,000 mg/kg magnesium aluminum silicate in mice; >200 g/kg zirconium silicate in mice; >5 g/kg hectorite in rats; and 149 g/kg kaolin in rats. The inhalation LC50 of synthetic fluorphlogopite for rats was > 5.1 mg/L.
In short-term oral toxicity studies, no adverse effects were seen in mice or rabbits dosed up to 5 g/kg magnesium aluminum silicate. Long-term feeding and drinking water studies revealed no adverse effects for magnesium aluminum silicate at 10% for dogs and rats and for magnesium trisilicate at 250 mg/L for guinea pigs.
Occupational exposure to aluminum silicate, calcium silicate, zirconium silicate, Fuller’s earth, kaolin, montmorillonite, pyrophyllite, and zeolite has led to fibrosis and pneumoconiosis. There were no reproductive effects for calcium silicate at 1,600 mg/kg for rabbits and for type Z zeolite at 74 or 1,600 mg/kg for rats or rabbits. Magnesium aluminum silicate at 6,000 mg/kg caused neither teratogenic nor adverse effects on the mouse fetus. Clinoptilolite had no effect on female rat reproductive performance.
Synthetic fluorphlogopite was not genotoxic in a chromosomal aberration test up to 0.16 mg/mL using Chinese hamster lung-derived fibroblast cells. It was not mutagenic to S. typhimurium and E. coli up to 5,000 μg, to V79 Chinese hamster cells up to 100 μg/mL, or to mammal cells up to 500 μg/mL. Magnesium aluminum silicate and related clays were not mutagenic in multiple assays. Mice exposed to calcium silicate dust at 10 mg/m3 developed neoplastic lesions.
Synthetic fluorphlogopite was not dermally irritating to rabbits and guinea pigs at 55%. Magnesium aluminum silicate was a weak primary skin irritant in rabbits at 4%. Synthetic fluorphlogopite was not dermally irritating to humans at 0.05 g in two 48-hour assays. It was not predicted to be a dermal irritant in an in vitro human skin model test. VEEGUM was not dermally irritating at 2 g daily for 1 week.
In an HET-CAM and a BCOP assay, synthetic fluorphlogopite was not predicted to be an ocular irritant. It was not an ocular irritant to rabbits. Magnesium aluminum silicate, hectorite, and zeolite A were not ocular irritants to rabbits or rats.
Synthetic fluorphlogopite was sensitizing to guinea pigs or mice. Synthetic fluorphlogopite was not irritating or sensitizing in a product at 13.824%. Synthetic fluorphlogopite was not photosensitizing or phototoxic to guinea pigs at 55%.
Discussion
Synthetic fluorphlogopite particles are large and insoluble in water and are unlikely to penetrate the skin. Although synthetic fluorphlogopite contains fluoride ions, the Panel concluded that these ions are unlikely to leach out of formulation due to the high heat in the manufacturing process.
Because this ingredient can be used in products that may be aerosolized, including perfumes and indoor tanning preparations (possible propellant and pump spray products), the Panel discussed the issue of incidental inhalation exposure. The acute inhalation data on the synthetic fluorphlogopite showed no adverse effects to rats. However, the chronic human industrial exposure data on the magnesium aluminum silicate and related clays resulted in fibrosis and pneumoconiosis.
Although particles appear to have reached the lungs in these studies, the sizes of the particles used were either clearly within the respirable range (ie, ≤10 µm) or were not reported. The Panel determined that the sizes of a substantial majority of the particles of synthetic fluorphlogopite, as manufactured, would be larger than the respirable range and/or aggregate and agglomerate to form much larger particles in formulation. Thus, the adverse effects reported using high doses of respirable particles in the inhalation studies did not indicate risks posed by use in cosmetics.
The Panel considered other data available to characterize the potential for synthetic fluorphlogopite to cause systemic toxicity, irritation, sensitization, or phototoxicity. They noted the lack of systemic toxicity in acute oral exposure and no irritation or sensitization. Also noted were the negative genotoxicity assays for synthetic fluorphlogopite and the magnesium aluminum silicate clays in a chromosomal aberration assay and multiple bacterial assays. In addition, these large macromolecules are insoluble in water and chemically inert under physiological conditions or conditions of use, which supports the view that they are unlikely to be absorbed or cause local effects in the respiratory tract. Further, this ingredient is reportedly used at concentrations of 0.05% to 2% in cosmetic products that may be aerosolized and 0.6% to 67% in powders. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Furthermore, this ingredient is used for viscosity increasing functions, indicating that they tend to swell and aggregate in water and other solvents and would, thus, be too large to be inhaled or respired. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, this information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects.
Conclusion
The CIR Expert Panel concluded that synthetic fluorphlogopite is safe for use in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Author Contributions
L. C. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and gave final approval. L. J. Gill, F. A. Andersen, W. F. Bergfeld, D. V. Belsito, R. A. Hill, C. D. Klaassen, D. C. Liebler, J. G. Marks, R. C. Shank, T. J. Slaga, and P. W. Snyder contributed to conception and design, contributed to acquisition, analysis, and interpretation, critically revised the manuscript, and gave final approval. All authors agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Washington, DC, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.