Abstract
PCK rats develop age-related polycystic kidney disease (PKD) and liver disease and have been used to investigate pharmacotherapies to ameliorate hepatorenal lesions for patients with PKD. The PCK rat may be useful to understand the possible susceptibility to hepatotoxicity observed in the patient with PKD having hepatic polycystic lesions. Therefore, the purpose of this study was to investigate the background blood biochemical changes that reflect the hepatorenal function of PCK rats as well as the terminal histopathology in order to determine whether this model would be suitable for extrapolating the susceptibility of hepatotoxicity in patients. The blood biochemical parameters of hepatorenal function and histopathology were investigated in PCK rats at ages 5 to 19 weeks and compared to those outcomes in the Sprague Dawley (SD) rat. There were notable blood biochemical changes possibly due to biliary dysgenesis in the PCK rat as early as 5 weeks of age. High levels of γ-glutamyl transpeptidase, alkaline phosphatase, alanine aminotransferase, and total bile acids persisted throughout the study compared to the SD rat. Increased aspartate aminotransferase, total bilirubin, and hyperlipidemia and a decrease in albumin were also evident at 10 to 19 weeks of age possibly due to progression of cholestatic liver dysfunction secondary to age-related liver cystic progression. Increased liver weights generally correlated with the severity of biliary and hepatic histopathological changes. In male PCK rats, age-related increases in blood urea nitrogen and creatinine at 10 to 19 weeks of age were observed, and the cystic progression was more severe than that in females. These data indicate that the PCK rat showed notable blood biochemical changes reflecting alteration of the liver function compared to the SD rat. Also, there was a large individual variation in these parameters possibly due to variable progression rate of biliary dysgenesis and subsequent liver damages in PCK rats.
Introduction
Polycystic kidney diseases (PKDs) are inherited renal diseases that exhibit profound morphological disorganization that is exemplified by abnormal cellular proliferation and apoptosis of immature epithelial cells, accumulation of fluid within the cysts, and abnormal cell/cell-matrix interactions and cilia function. 1,2 In the human population, the most common type of PKD is inherited as an autosomal-dominant trait (autosomal-dominant polycystic kidney disease [ADPKD]) with an estimated diagnosed prevalence of less than 1/2,000. Approximately 85% of ADPKD is caused by a mutation in the PKD1 gene and 15% by mutation in the PKD2 gene. Although less common but still clinically significant is autosomal-recessive polycystic kidney disease. Autosomal-recessive polycystic kidney disease is caused by mutations in polycystic kidney and hepatic disease 1 (PKHD1) gene, and this gene was identified by comparative genetic analysis with the PCK rat. 3
Tolvaptan, an orally available benzazepine derivative, is a selective vasopressin V2 receptor antagonist 4 used in the treatment of clinically significant hyponatremia, 5 volume overload in heart failure, 6 and liver cirrhosis with edema. 7 Recently, tolvaptan has shown promise for delaying renal cyst progression in ADPKD, 8 –10 with approvals in Japan, Canada, and Europe for this indication. In the pivotal clinical trial, however, tolvaptan was associated with liver injury evident as increased liver enzymes and bilirubin in some patients. In contrast to the ADPKD population, no signals of liver injury have been observed in patient populations treated with tolvaptan for other indications or in nonclinical studies. 11,12 The etiology and/or biological mechanism(s) underlying tolvaptan-associated liver injury in patients with ADPKD have yet to be elucidated, although research into possible mechanism(s) is currently ongoing.
The PCK rat originated spontaneously from a colony of Sprague Dawley (SD) rats 13 and is commercially available as the model of PKD and polycystic liver disease (PLD). The histopathological features resemble human ADPKD, although the pattern of inheritance is autosomal recessive. 14 With the advent of targeted mutation, this model has been developed and is shedding new light not only on the examination of the pathogenesis of these diseases but also on the pharmacotherapeutic approaches. The prevalence of hepatic cysts in patients with ADPKD has been estimated to be as high as 80%. Although hepatic cysts involvement of ADPKD does not result in hepatic dysfunction, 15 Hogan et al recently reported subtle changes in liver laboratory tests that were detected even in the presence of hepatomegaly. 16 We have speculated that patients with PKD having hepatic polycystic lesions might be more susceptible to the drug-induced hepatotoxicity than the normal population. In the present study, therefore, we investigated the background blood biochemical changes that reflect the hepatorenal function of PCK rats as well as the terminal histopathology in order to determine whether this model would be suitable for extrapolating the susceptibility of hepatotoxicity in patients. Hematological evaluations were not included in this study since an effect on hematology was not observed in the population with ADPKD treated with tolvaptan. 10
Materials and Methods
Dosing Substance
Hypromellose (hydroxypropyl methylcellulose [HPMC], substitution type 2910) was purchased from Shin-Etsu Chemical Co Ltd (Tokyo, Japan), and was stored at room temperature (10-30°C) and protected from light. The HPMC was dissolved in water for injection (Otsuka Pharmaceutical Factory Inc (Tokushima, Japan)) to make 1% HPMC solution. The dosing solution was prepared once weekly and stored in a cool place (1-10°C) and protected from light. Animals were treated with 1% HPMC solution via oral gavage daily for 91 days (10 mL/kg). The HPMC had been used as the vehicle in the nonclinical studies with tolvaptan. HPMC is a semisynthetic substance widely used as an excipient in pharmaceutical formulations and was, therefore, used for the current study.
Animals
This study was carried out in strict accordance with the Guidelines for the Animal Care and Use of the Otsuka Pharmaceutical Co, Ltd, which conforms to the international norms stipulated by the Ministry of Health, Labour and Welfare in Japan. The protocol was approved by the Institutional Animal Care and Use Committee of the Otsuka Pharmaceutical Co, Ltd (Permit Number: 13-0159). All efforts were made to maximize animal welfare and minimize suffering.
Ten male and 10 female SD [Crl: CD (SD)] rats and 20 male and 20 female PCK/CrljCrl-PKHD1pck/CRL rats (Microbiological grade: specific pathogen free) were purchased from the Hino Breeding Center (Shiga, Japan) of Charles River Laboratories Japan, Inc. All rats were weighed and inspected for any clinical symptoms on the day of arrival and reweighed on the last day of the 7-day quarantine period. In addition, the rats were daily monitored for general condition during a 12-day acclimation period including the quarantine period. There were no abnormalities in the general condition or body weight changes in any rats. Therefore, based on the observations, all rats could be used in this study.
On the day of the first administration, the rats were 6 weeks old, and the male and female SD rats weighed 156 to 203 g and 150 to 165 g, and the male and female PCK rats weighed 168 to 195 g and 149 to 172 g, respectively. Each rat was uniquely identified. The animals were individually housed in stainless steel cages for rats (222 × 325 × 180 mm; Nippon Cage Co, Ltd (7-7-5, Kamikita, Hirano-ku, Osaka, Japan)) in a room that was lighted for 12 hours (lights on at 07:00 hours) daily. The rats were housed in an air-conditioned room ventilated with an air exchange rate of 14.7 to 15.5 times/hour and maintained at 22.7 to 23.2°C with relative humidity of 53% to 72%. Each rat was allowed free access to pelleted diet (CRF-1, sterilized by radiation; Oriental Yeast Co) and Otsuka’s in-house tap water via an automatic watering device. Each rat was fasted for 16 to 18 hours before necropsy.
Study Procedures
The day of the first administration was designated as day 1 and days 1 to 7 as week 1. The clinical signs were recorded for each rat once daily before administration. The body weight of each animal was recorded before dosing on day 1, at least weekly during dosing, and on the day of necropsy. The average daily food consumption of each animal was calculated by subtracting the amount of uneaten food from the amount of food supplied, which was measured weekly during dosing, and dividing the difference by the number of days between the measurements.
Clinical Chemistry and Terminal Procedures
Prior to dosing and once before dosing in week 4 (males: day 24, females: day 25), week 8 (males: day 52, females: day 53), and week 13 (males: day 87, females: day 88), approximately 0.7 mL of blood was collected from the jugular vein into a 1 mL heparinized syringe (NIPRO, Japan) under unanesthetized condition from all rats for standardized clinical biochemical analysis using an automated clinical analyzer (Hitachi-7180, Japan). Heparinized plasma obtained after centrifugation (3,000 rpm, 20 minutes, 4°C) was stored for 1 day at −80°C prior to evaluation. The parameters evaluated included aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin (TBI), alkaline phosphatase (ALP), γ glutamyl transpeptidase (γ-GT), total cholesterol (CHO), triglycerides (TG), phospholipids (PL), glucose, albumin (ALB), blood urea nitrogen (BUN), creatinine (CRE), and total bile acids (TBA).
A necropsy was performed on all rats on the day following administration of the final oral dose and was also performed on 1 dead male PCK rat as soon as possible. The rats were anesthetized with isoflurane and killed by exsanguination from the posterior vena cava and aorta. Necropsy consisted of macroscopic observations of the external morphology and major internal organs in the abdominal and thoracic cavities. Liver and kidneys obtained from the scheduled killed animals were weighed and fixed in 10% neutral-buffered formalin. Liver and kidneys obtained from 1 dead male PCK animal were also fixed. After fixation, all of the tissue samples were dehydrated with a graded series of ethyl alcohol, dealcoholized with xylene, and embedded in paraffin. The paraffin block tissues were thinly sectioned and stained with hematoxylin and eosin. The kidneys were further stained with periodic acid-Schiff (PAS). Light microscopy was conducted on all specimens of all rats.
Data Analysis
The following data obtained during the dosing period were analyzed by the t test, with F test for homogeneity of variance between groups of SD and PCK rats: body weight, body weight gain, food consumption, and organ weights as well as the results of blood biochemistry. The significance tests were performed in 2 tailed, and statistical significance was presented at either the 5% or 1% level.
Results
One male PCK rat was found dead on day 11, but no clinical signs were observed, and the blood biochemical data obtained before the initiation of dosing were comparable to those of other male PCK rats. Necropsy and histopathology evaluations did not reveal a possible cause of death. The other PCK rats showed no notable symptoms or external findings during the dosing period.
The body weights of male PCK rats were comparable to those of male SD rats, but the body weight of female PCK rats was slightly higher than that of female SD rats during the dosing period, and a 13% increase in mean body weights was noted at week 13 (Figure 1).
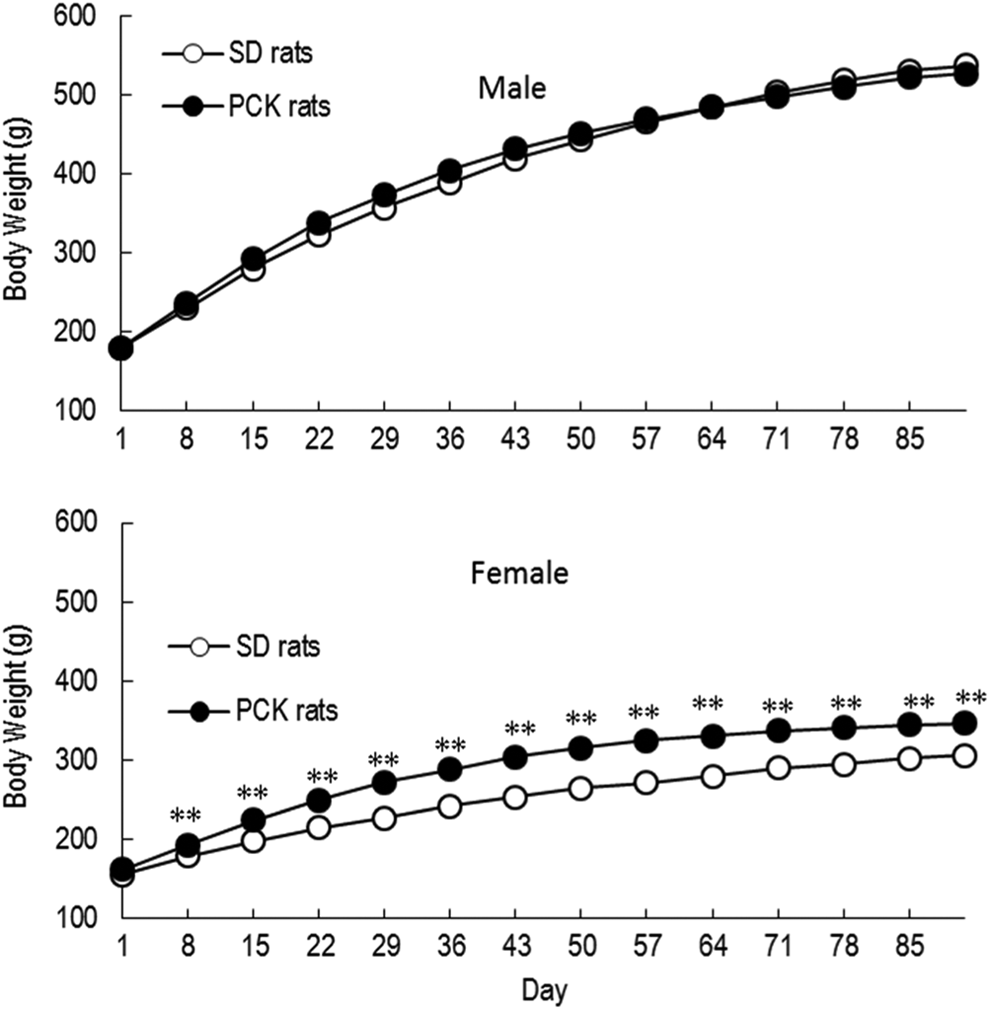
Body weight changes in male and female Sprague Dawley (SD) and PCK rats during the 13-week study. **P < 0.01.
The food consumption of male PCK rats was slightly higher than that of male SD rats at weeks 2, 3, 5, and 6 but was comparable at week 7 and later. In female PCK rats, the food consumption was slightly higher than that of female SD rats during the dosing period, and a 17% increase in mean food consumption was noted at week 13 (Figure 2). The results of blood biochemical parameters reflecting liver function are summarized in Table 1.
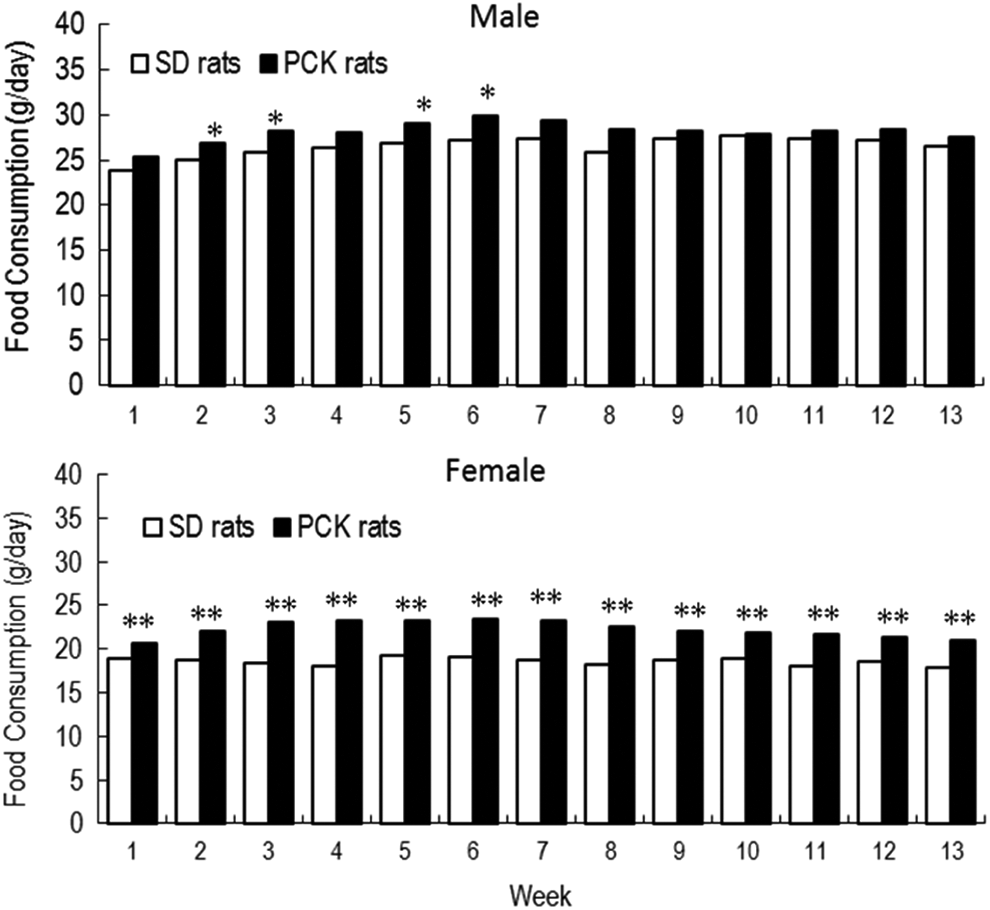
Food consumption in male and female Sprague Dawley (SD) and PCK rats during the 13-week study. *P < 0.05, **P < 0.01.
Summary of Plasma Biochemical Values of Liver Function in PCK and SD Rats.
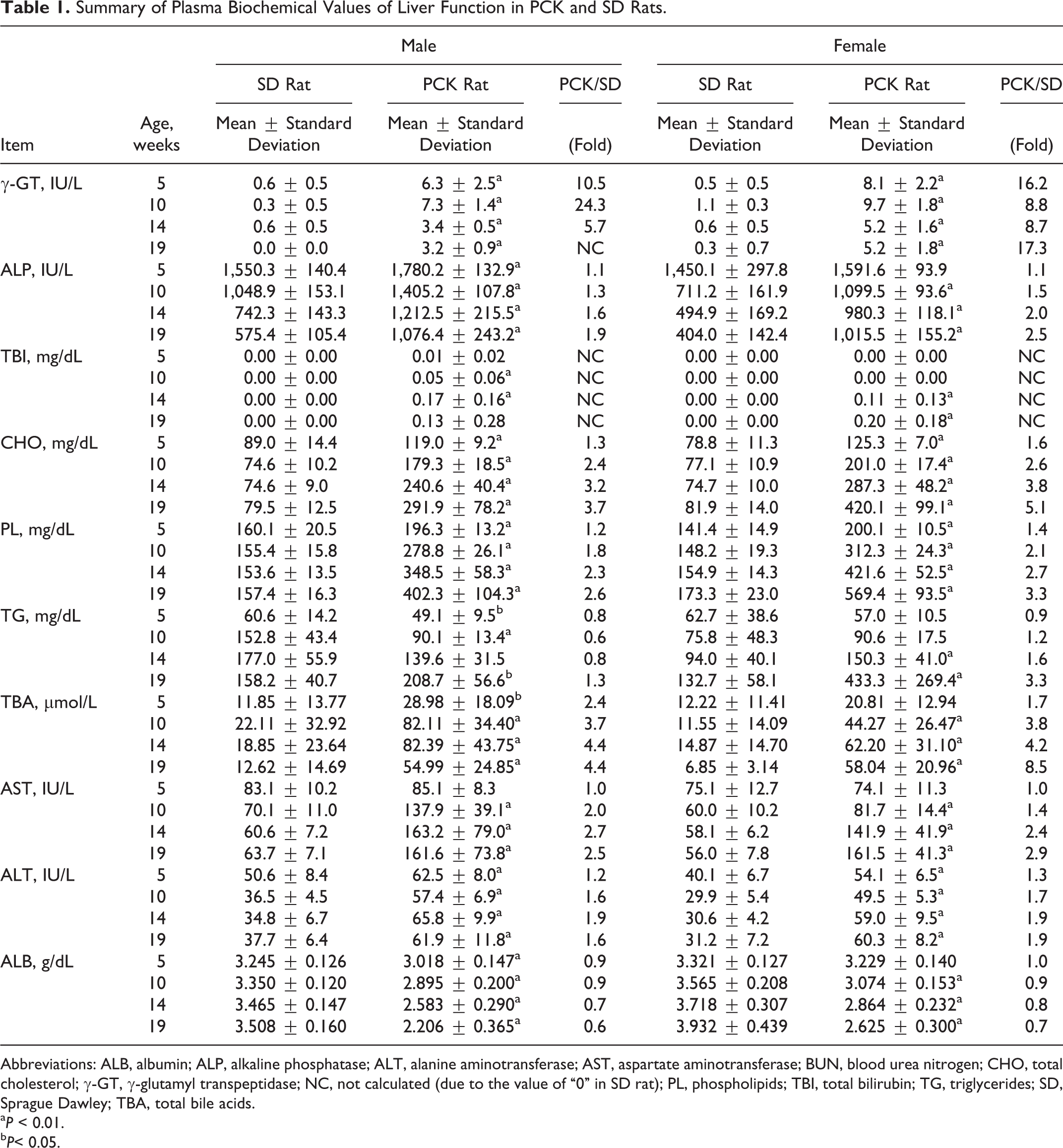
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CHO, total cholesterol; γ-GT, γ-glutamyl transpeptidase; NC, not calculated (due to the value of “0” in SD rat); PL, phospholipids; TBI, total bilirubin; TG, triglycerides; SD, Sprague Dawley; TBA, total bile acids.
a P < 0.01.
b P< 0.05.
Compared to the SD rat, PCK rats of both sexes showed extremely higher values for γ-GT. Large individual variation existed in this value before the initiation of dosing (at age 5 weeks) and continued to be observed throughout the study. The PCK rats also showed higher values of ALT, ALP, and TBA compared to the SD rats, and the higher levels were observed throughout the study (Figure 3).
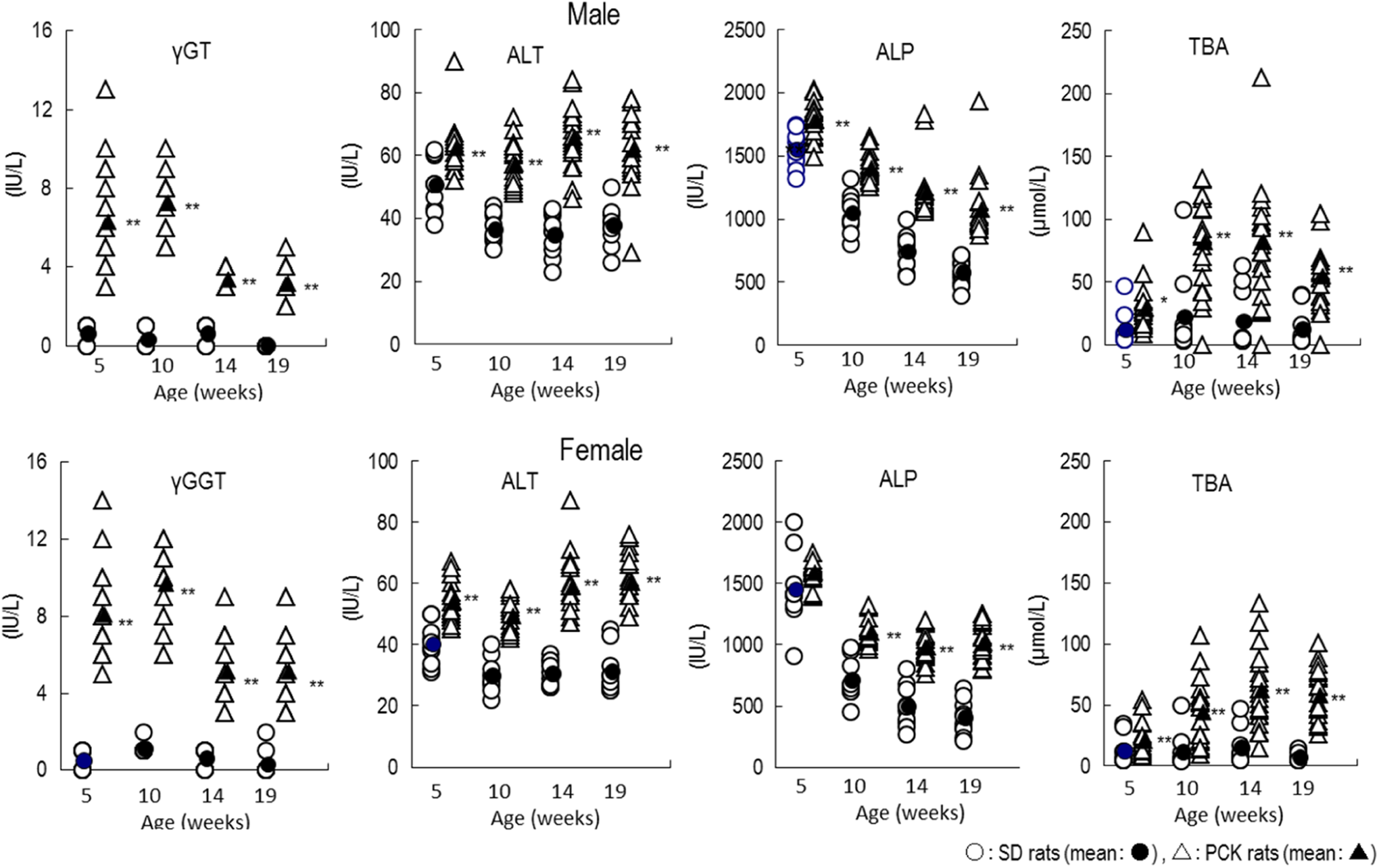
Blood biochemical parameters that maintained high levels at ages 5 to 19 weeks in PCK and SD rats. *P < 0.05; **P < 0.01. ALP indicates alkaline phosphatase; ALT, alanine aminotransferase; γ-GT, γ-glutamyl transpeptidase; TBA, total bile acids; SD, Sprague Dawley.
The ALB levels for PCK rats were lower for both sexes at age 5 to 19 weeks compared to the SD rat and also decreased as time elapsed contrary to the increasing trend observed in SD rats. The AST and TBI for the PCK rats were comparable to that of SD rats at age 5 weeks, but the values increased at age 10 to 19 weeks as time elapsed and also exhibited large individual variation in these values (Figure 4).
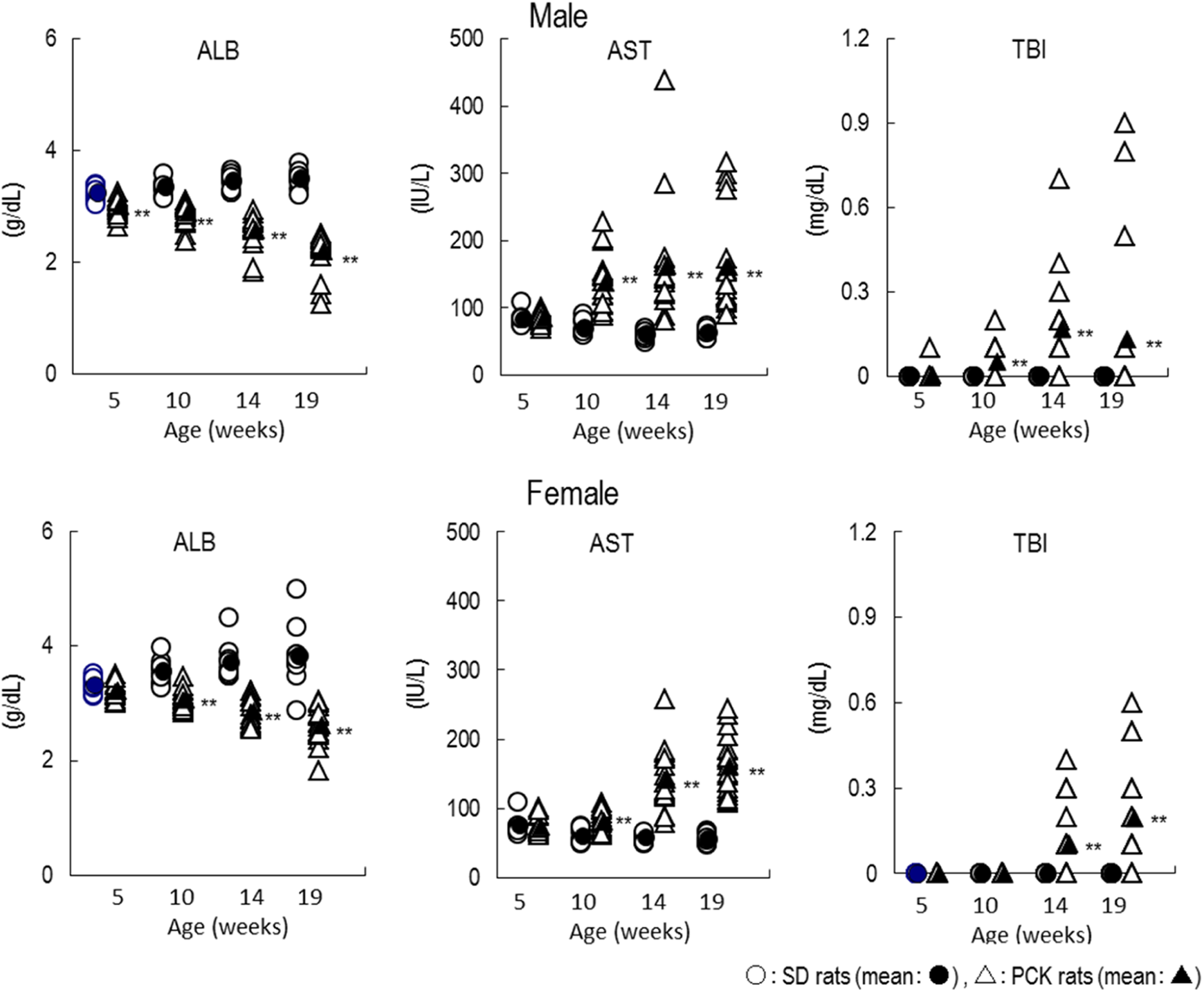
Blood biochemical parameters that decreased/increased as time elapsed in PCK and SD rats. *P < 0.05; **P < 0.01. ALB indicates albumin; AST, aspartate aminotransferase; TBI, total bilirubin.
For CHO and PL, the values in the PCK rats were higher than that of SD rats in both sexes at age 5 to 19 weeks and also increased as time elapsed and exhibited large individual variation. There were no remarkable differences in TG between SD rats and PCK rats in the males, but female PCK rats at ages 14 and 19 weeks showed a higher value than that of female SD rats. As with other parameters, TG increased as time elapsed with large individual variation (Figure 5).
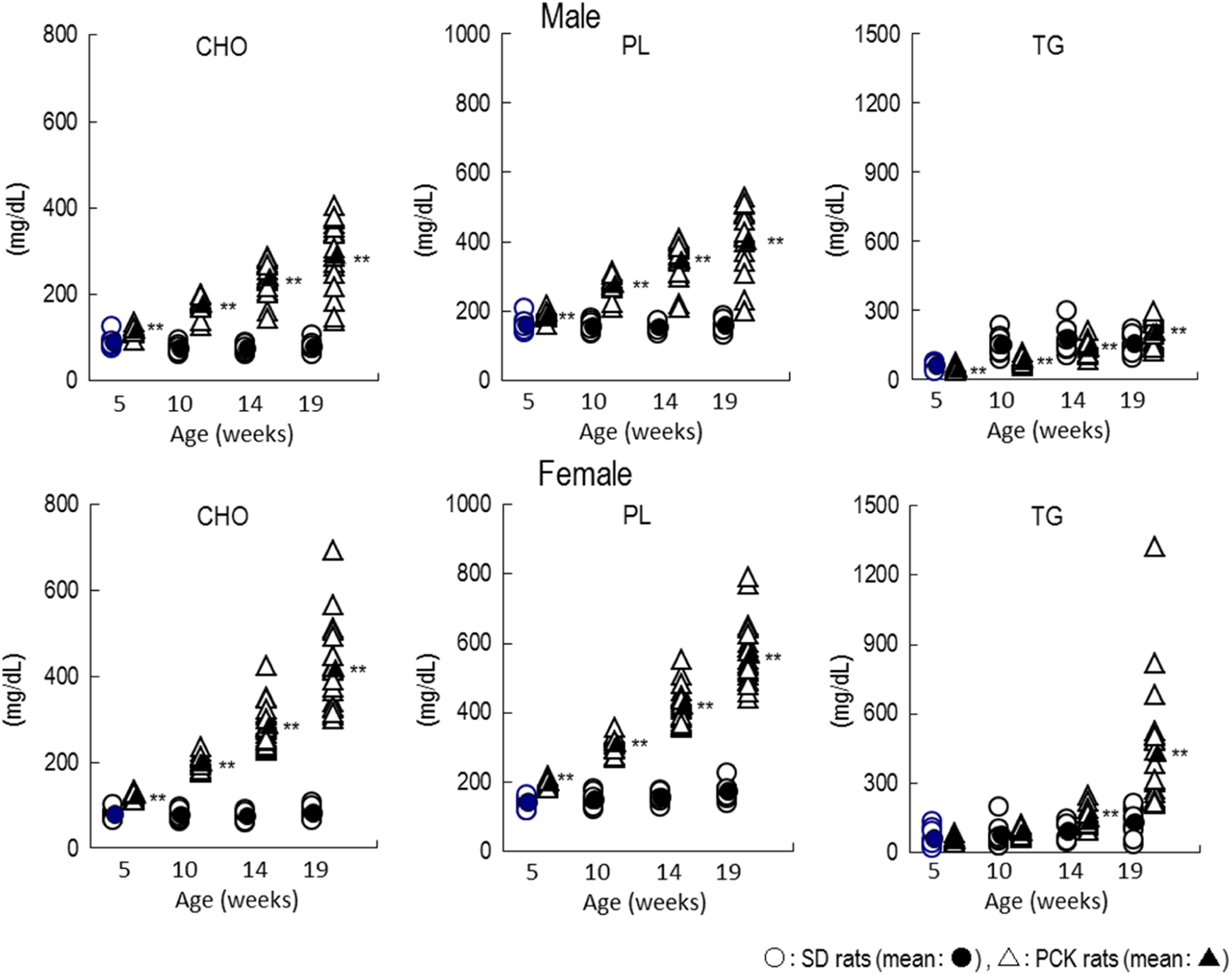
Plasma lipid level changes as time elapsed in PCK and SD rats. **P < 0.01. CHO indicates total cholesterol; PL, phospholipids; TG, triglycerides.
As for BUN and CRE, each value of male PCK rats was slightly higher than that of SD rats at age 10 to 19 weeks, but female PCK rats showed no remarkable changes in BUN and CRE when compared to those of female SD rats (Table 2).
Summary of Plasma Biochemical Values of Kidney Function in PCK and SD Rats.
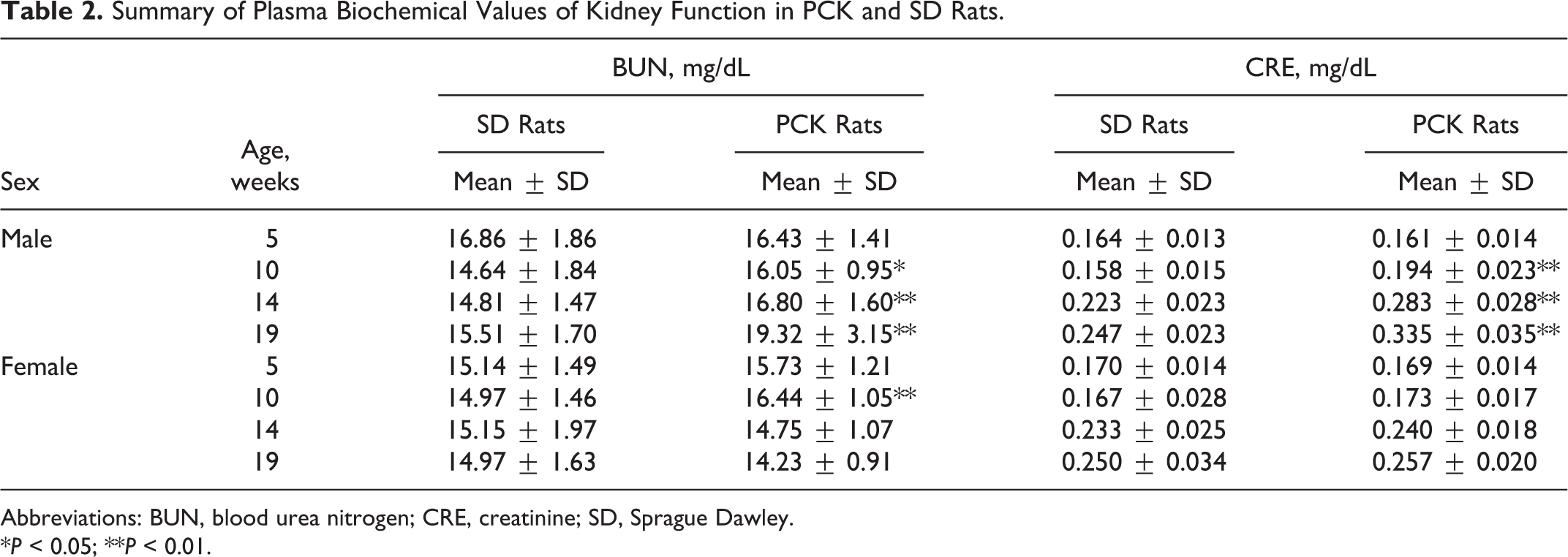
Abbreviations: BUN, blood urea nitrogen; CRE, creatinine; SD, Sprague Dawley.
*P < 0.05; **P < 0.01.
At necropsy, PCK rats showed enlarged liver and an irregular liver surface morphology as the major findings. The absolute liver weights of males and females were 2.5- and 2.9-fold higher compared to SD rats. Histopathologically, all PCK rats showed dilatation/hyperplasia of bile duct, periductal fibrosis, and inflammatory cell infiltration, and the severity of each finding in males was higher than that in females. Periductal lymphoid follicle and inflammatory cell infiltration of lumen/bile duct were also observed in 5 of 19 male PCK rats. PCK rats also showed focal necrosis of hepatocyte and vacuolation of hepatocyte in both sexes, and some male PCK rats showed eosinophilic cytoplasm of hepatocyte (Table 3, Figures 6 and 7).
Hepatic Histopathological Findings in PCK and SD Rats at 19 Weeks of Age.
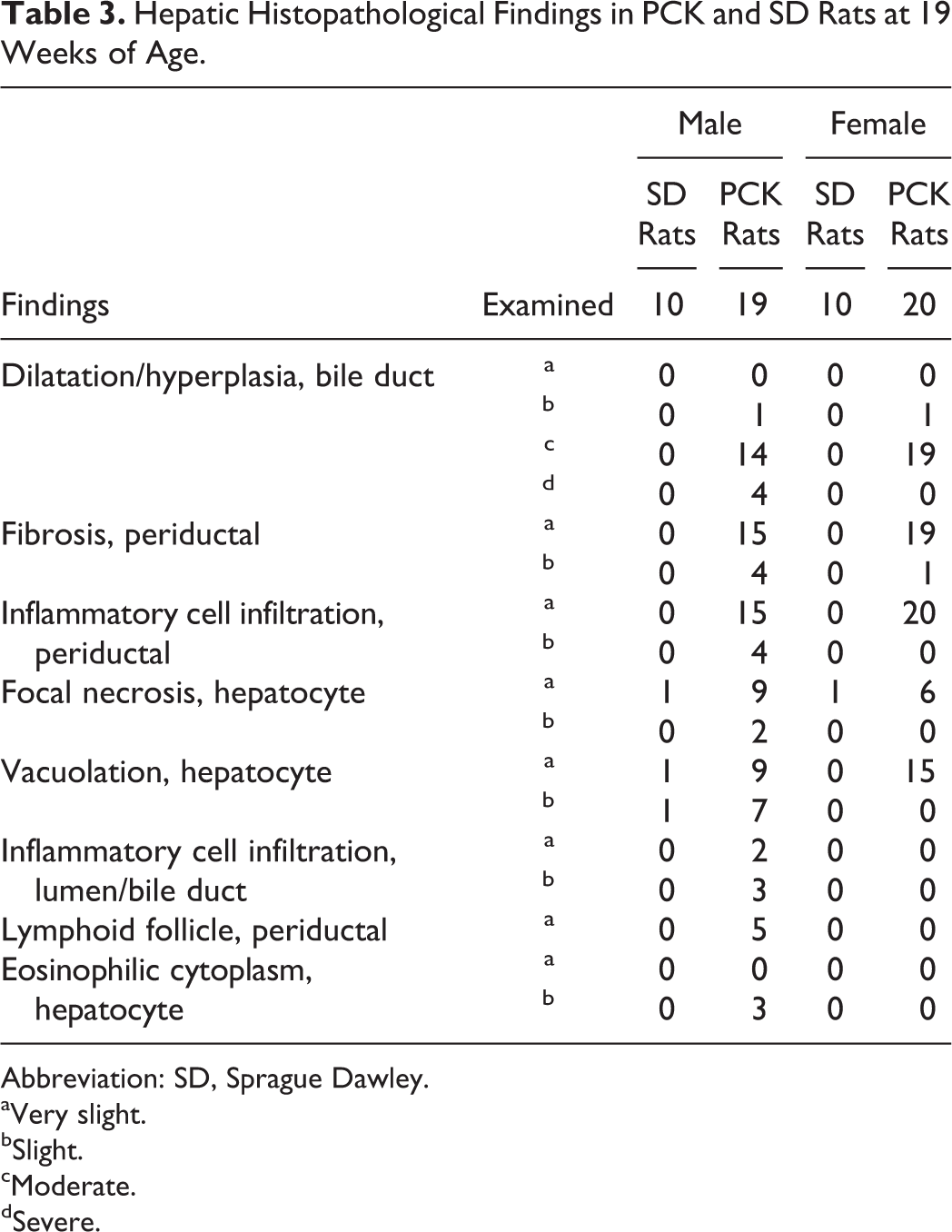
Abbreviation: SD, Sprague Dawley.
aVery slight.
bSlight.
cModerate.
dSevere.
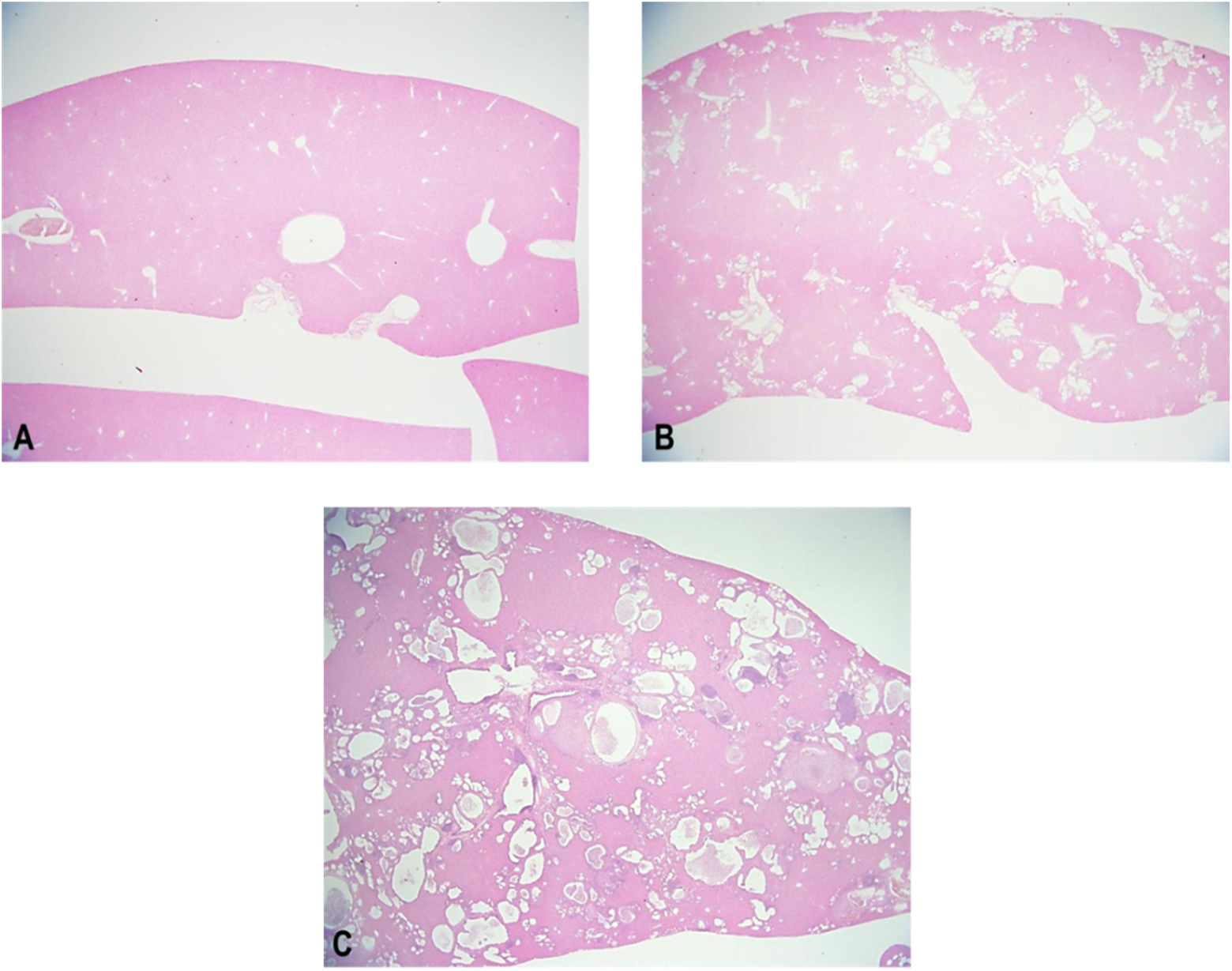
Representative liver section from Sprague Dawley (SD) rat (A) and PCK rat (B, C) at 19 weeks of age. Note the presence of dilation/hyperplasia of bile duct (B: moderate, C: severe)
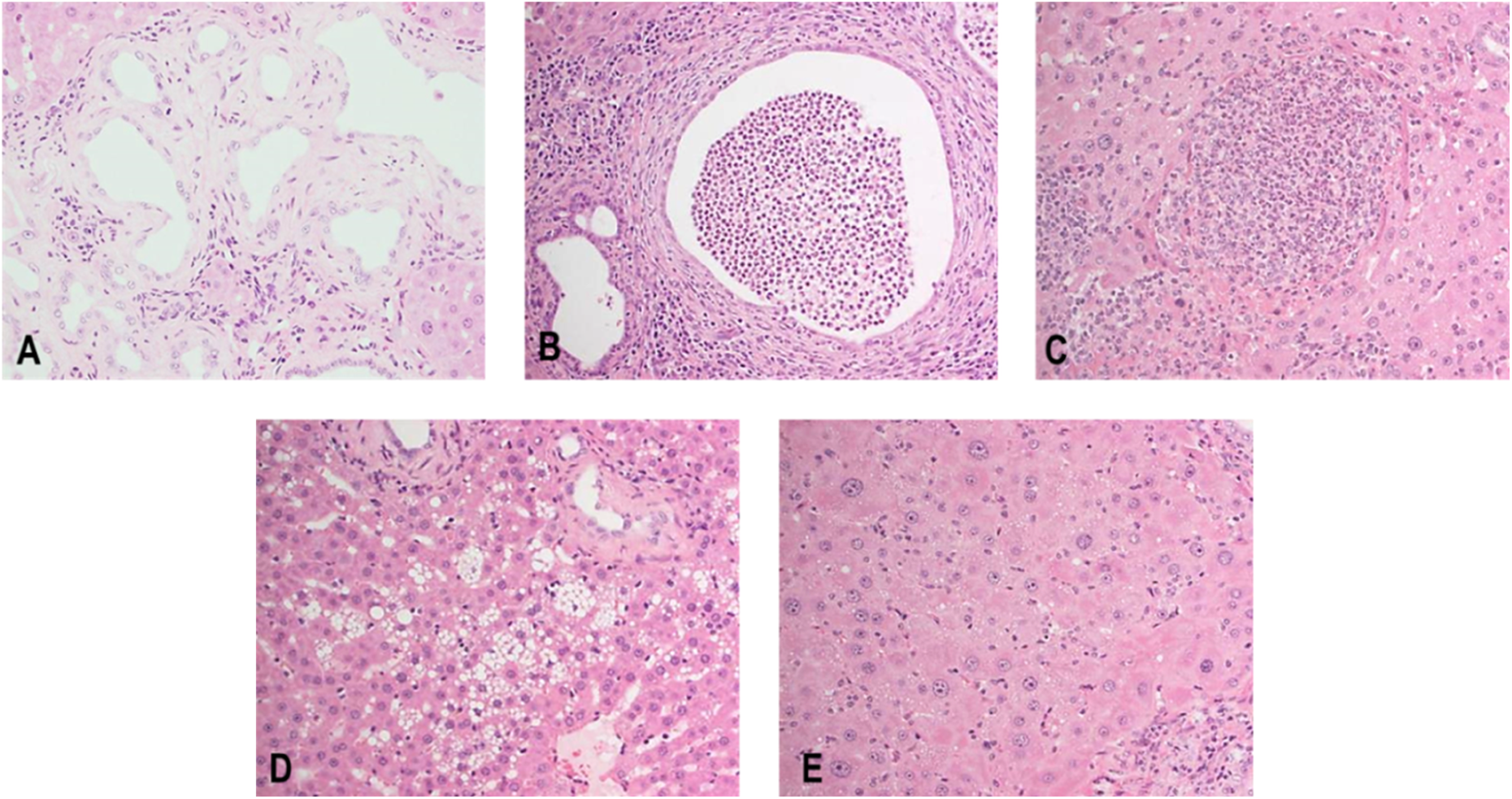
Representative liver section from PCK rat at 19 weeks of age. Note the presence of dilation/hyperplasia of bile duct, periductal fibrosis with inflammatory cell infiltration (A), inflammatory cell infiltration of lumen/bile duct (B), focal necrosis (C), vacuolation (D), and eosinophilic cytoplasm of hepatocyte (E).
Of the PCK rats killed at the end of the study, 4 males showed notably increased liver weight with severe bile duct dilatation and hyperplasia, periductal fibrosis, and inflammatory cell infiltration as well as hepatocyte changes that were more evident than those of the other male PCK rats (Table 4). Furthermore, the degree of increased TBI and AST and decreased ALB correlated with the severity of the biliary and hepatocyte changes observed in these male PCK rats, further reflecting the large variation in end points noted among PCK rats. Interestingly, the degree of change for CHO and PL was conversely lower than those of the other male PCK rats, and vacuolation of hepatocytes was not evident in these male PCK rats. The underlying mechanism was unclear.
Correlation of Plasma Biochemical Changes, Liver Weights, and Histopathology of Male PCK Rats.
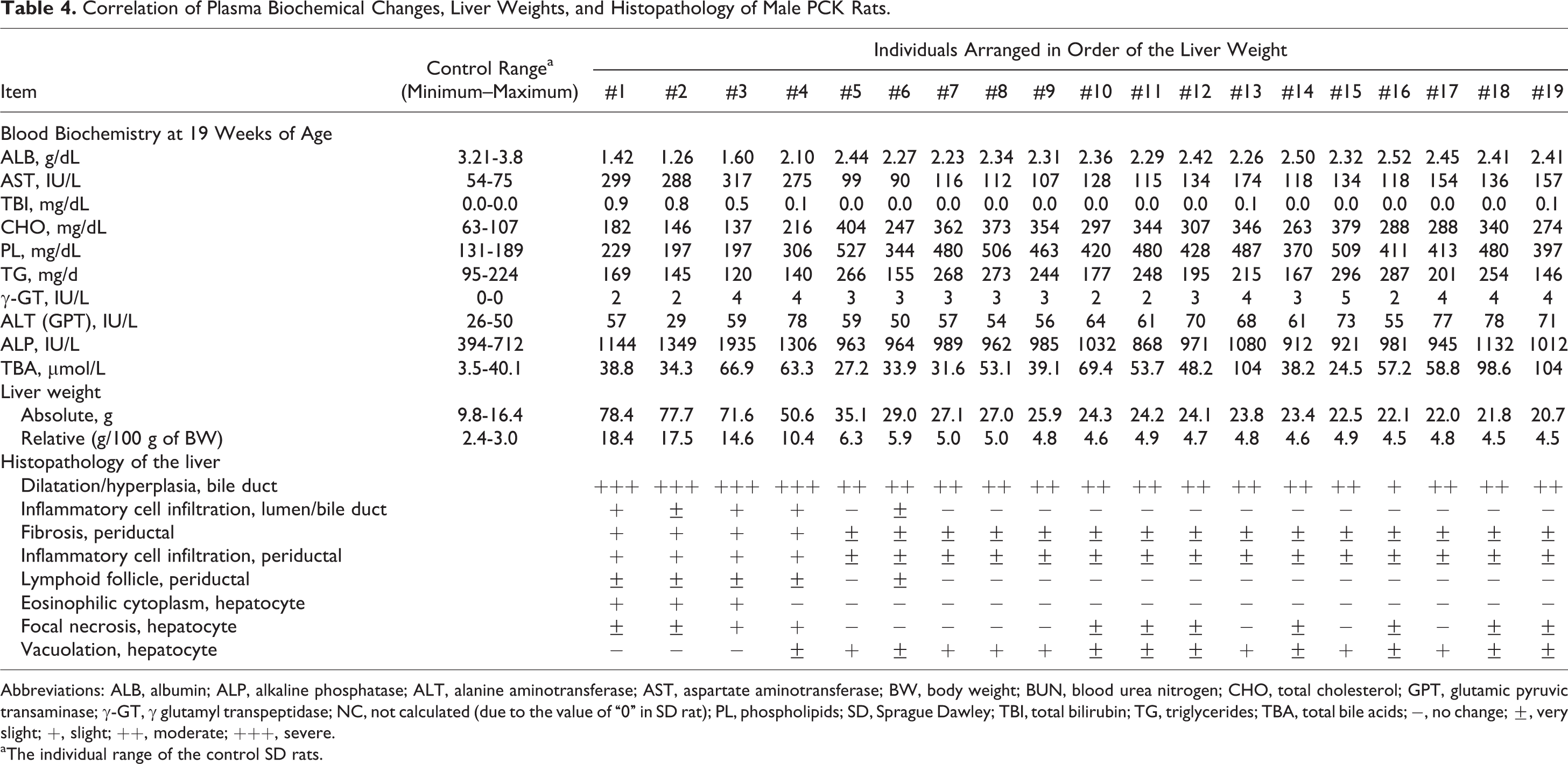
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BW, body weight; BUN, blood urea nitrogen; CHO, total cholesterol; GPT, glutamic pyruvic transaminase; γ-GT, γ glutamyl transpeptidase; NC, not calculated (due to the value of “0” in SD rat); PL, phospholipids; SD, Sprague Dawley; TBI, total bilirubin; TG, triglycerides; TBA, total bile acids; −, no change; ±, very slight; +, slight; ++, moderate; +++, severe.
aThe individual range of the control SD rats.
PCK rats also showed enlarged kidney and cysts at necropsy, and the absolute kidney weights of males and females were 2.4- and 1.8-fold, respectively, higher than those of SD rats. Histopathologically, all PCK rats showed dilatation and degeneration/regeneration of the renal tubule (Table 5, Figure 8). All male and 18 female PCK rats showed hyaline casts in the renal tubule. In the glomeruli, all PCK rats showed PAS-positive granules deposition in both sexes. The renal tubular and glomerular findings showed that the severity of each finding in the males was greater than that of females. Interstitial fibrosis and inflammatory cell infiltration were observed in all male PCK rats, and in 10 female and 4 female PCK rats, respectively. In addition in the PCK rat, PAS-positive granule (hyaline droplets) deposition in the renal tubule and inflammatory cell infiltration of lumen/tubule were observed and considered associated with basement membrane damage and protein leakage into urine, further indicating an age-related renal dysfunction. The severity and incidence of each finding were comparable in males and females.
Kidney Histopathology Findings in PCK and SD Rats at 19 Weeks of Age.
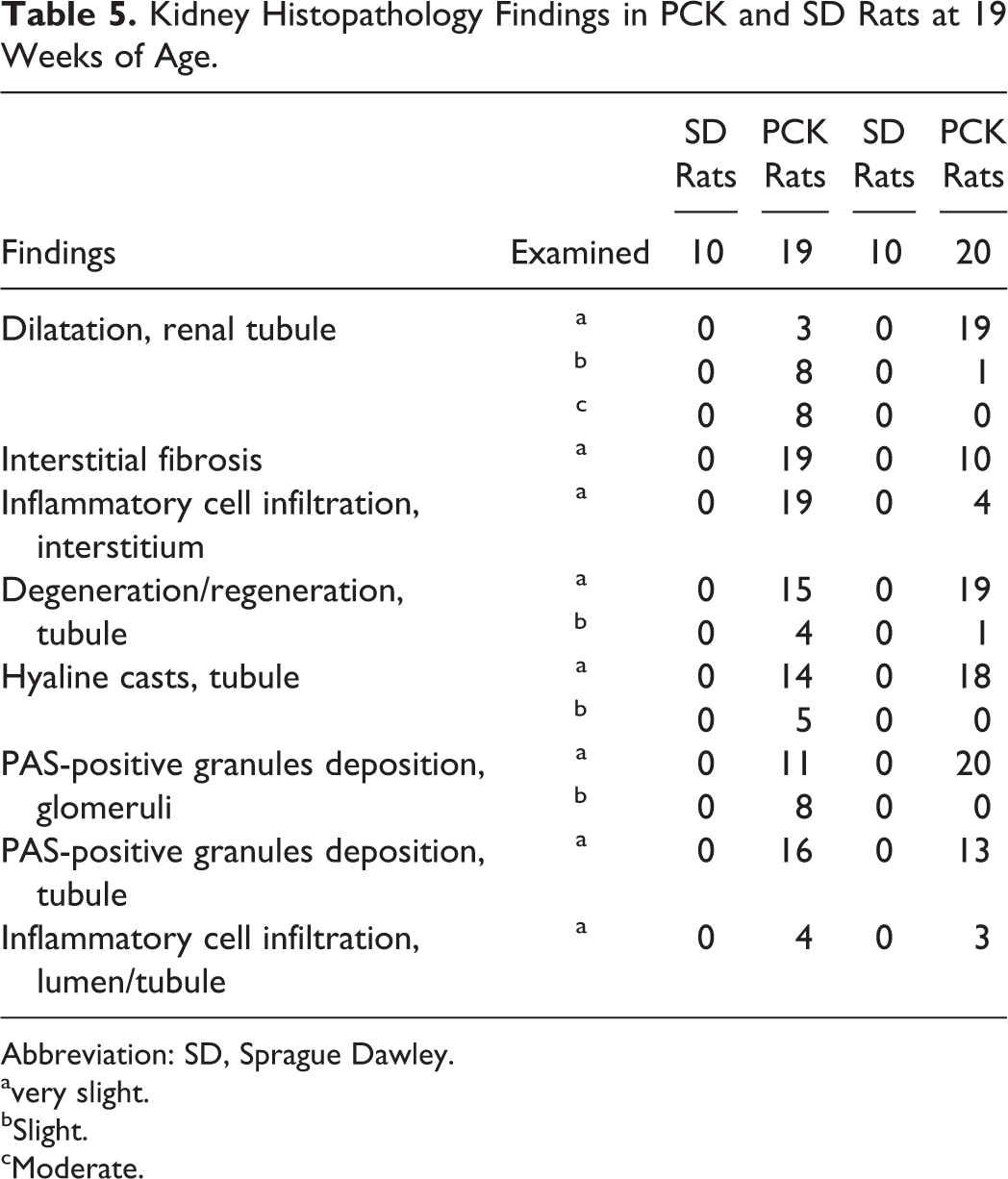
Abbreviation: SD, Sprague Dawley. avery slight.
bSlight.
cModerate.
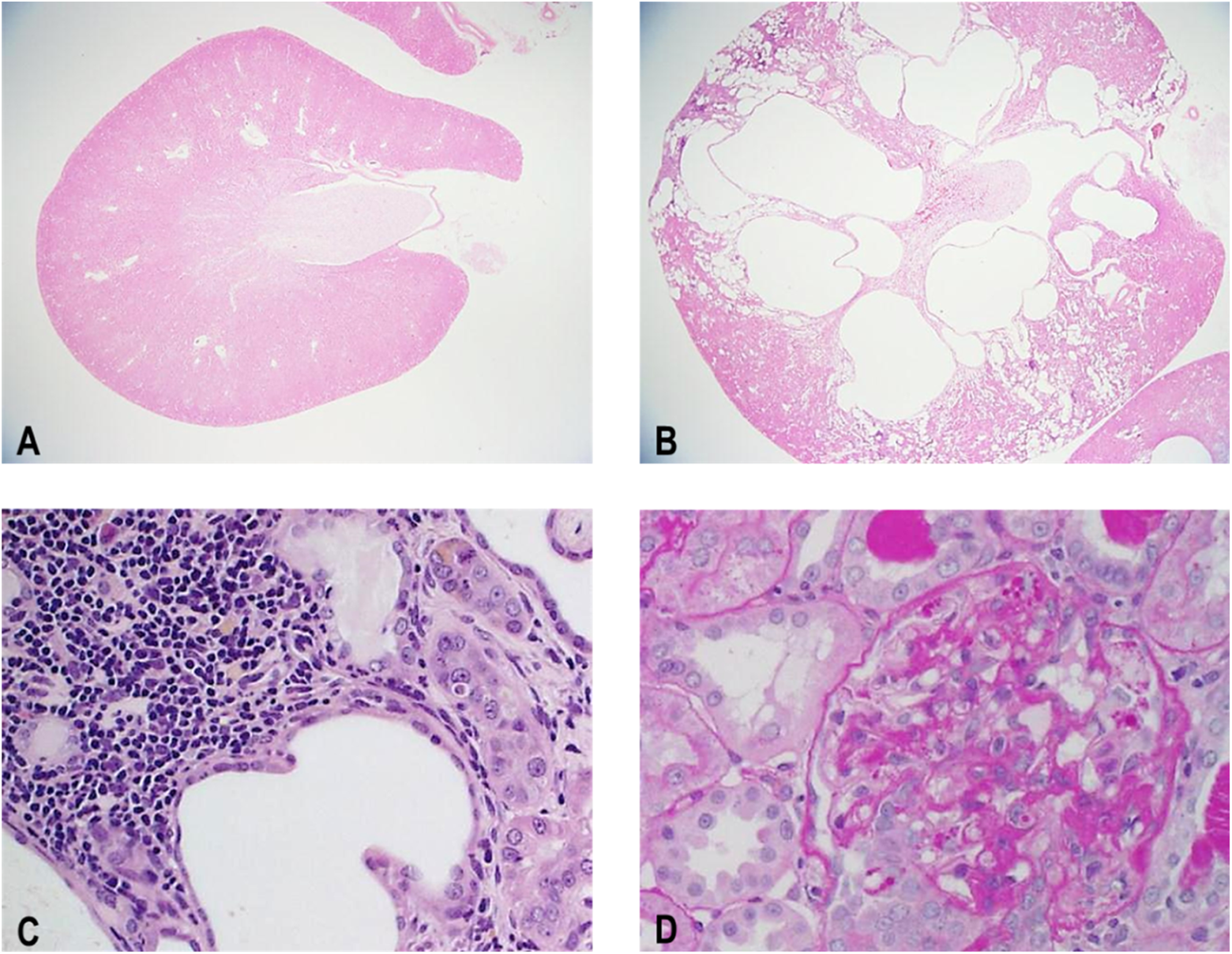
Representative kidney section from Sprague Dawley (SD) rat (A) and PCK rat (B, C, D) at 19 weeks of age. Note the presence of dilation/hyperplasia of bile duct (B, moderate), dilatation and degeneration/regeneration of the renal tubule and inflammatory cell infiltration (C), and PAS-positive granules deposition in glomeruli (D).
Discussion
The PCK rat develops age-related PKD and PLD with features that resemble human ADPKD. 14 This model has made valuable contributions not only to the examination of the pathogenesis of these diseases but also to the pharmacotherapeutic approaches for patients with ADPKD.
The vaptans, vasopressin V2 receptor agonists, were shown to be effective in ameliorating renal pathology using the PCK rat. 17,18 Recently, tolvaptan was approved for the treatment of ADPKD in several regulatory regions, but periodic blood testing for hepatic transaminases was required since idiosyncratic liver injury was observed in the clinical trial for the treatment of ADPKD. 10
Tolvaptan has been previously approved for the treatment of dilutional hyponatremia in various countries and volume overload in heart failure or liver cirrhosis in Japan, but no evidence of liver injury has been observed among these patients without ADPKD. Therefore, it is conceivable to speculate that patients with ADPKD having hepatic cysts are more susceptible to the hepatotoxicity than other patient populations since the prevalence of hepatic cysts in patients with ADPKD has been estimated to be as high as 80%. 15 Therefore, the mechanism(s) and a determination of possible genetic and nongenetic biomarkers of potential susceptibility of this patient population are being investigated.
In the present study, we investigated the features of blood biochemical changes reflecting the hepatorenal function of PCK rat as well as the terminal histopathology in order to determine whether this model would be suitable for extrapolating the susceptibility of hepatotoxicity in patients with ADPKD.
As expected, the growth of PCK rat was comparable to that of SD rat, although female PCK rats showed slightly higher body weight and food consumption than female SD rat. All PCK rats showed fibrocystic hepatorenal lesions at termination. Interestingly, PCK rats showed notable blood biochemical changes as early as 5 weeks of age. High levels of γ-GT, ALP, ALT, and TBA persisted with large individual variation throughout the study period. The high level of γ-GT was possibly due to a consequence of biliary dysgenesis. 19 It is known that PCK rat develops mild bile duct dilation as early as 1 day of age, 14 and the elevation in γ-GT is closely associated with cholangiolar proliferation or bile duct hyperplasia. 20,21 Although there was little preexisting comparable data in the PCK rat compared to the SD rat, development of polycystic lesions might also affect the synthesis of ALP, ALT, or TBA in the liver.
Time-elapsed increases in AST, TBI, and hyperlipidemia and a decrease in ALB were also evident in PCK rat at 10 to 19 weeks of age, and the individual variation in these parameters became more evident with age. These findings suggest that cholestatic liver dysfunction progressed secondary to age-related development of PLD, and the progression results in large individual variation. The clinical chemical profile in the PCK rat is different from those in patients with ADPKD. 15,16 The baseline values reported by Hogan et al for ALT, AST, and TBI in the patient with ADPKD 16 are generally consistent with normal values reported for humans. 22 In contrast to the biochemical features observed in patient with ADPKD, several of the clinical chemistry values, including ALT, TBI, and in particular, γ-GT obtained from PCK rat were higher than the SD rat throughout the study.
Lager et al reported that the plasma concentrations of TBI and ALP in PCK rat were similar to those in normal SD rats at 10 and 26 weeks of ages, 14 and Masyuk et al reported that serum levels of AST and ALT were not different between SD and PCK rats at 3 months of age. 19 These data are in contrast to those obtained in the present study. Although the cause of discrepancies was unclear, it might be related to the differences in the colony of PCK rats used. These other investigators used subcolonies maintained in their laboratories, but the PCK rats used in the current study were commercially purchased.
Measuring AST, TBI, and ALB might be useful to identify severely affected PCK rats, since the individual changes in these parameters at the termination were correlated with the degree of liver weight increase and the severity of biliary and hepatic histopathological changes. In the present study, 4 male PCK rats showed extremely high liver weight and severely affected biliary and hepatocyte changes, although the existence of gender dimorphism was unclear. Mason et al reported that hepatic fibrocystic volume in the female PCK rats tended to be greater than that in the males. 23
In addition, the male PCK rats showed the time elapsed increases in BUN and CRE at age 10 to 19 weeks. The developmental progression of kidney pathology in males was also more evident than that in the females at the termination, and this gender dimorphism (male > female) was similar to that previously described by Lager et al and Mason et al. 14,23
In conclusion, the PCK rat showed notable blood biochemical changes reflecting alteration of the liver function compared to the SD rat. Also, there was a large individual variation in these parameters possibly due to variable progression rate of biliary dysgenesis and subsequent liver damage in the PCK rat. The present study suggests that extrapolation of the susceptibility of hepatotoxicity to the patients with ADPKD using this PCK animal model may lead to an inappropriate conclusion. Therefore, interpretation and extrapolation of clinical chemistry findings observed in the PCK rat to the population with PKD need to be cautiously considered.
Footnotes
Acknowledgments
The authors would like to acknowledge the technical staff at TRI for conducting the in-life portion of the study, performing the necropsy, preparing and analyzing the clinical pathology samples, and preparing the tissue sections for histopathological examination.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported entirely by Otsuka Pharmaceutical Co, Ltd.
