Abstract
This study was focused on investigating the anticancer potential of indole-3-carbinol (I3C) against lung cancer H1299 cells via an increase in ROS levels. To investigate the induction of growth arrest and/or cell death in H1299 cells, a cell cycle arrest assay, terminal deoxynucleotidyl transferase (TdT)-mediated dUTP-biotin nick-end labeling (TUNEL) assay, and reactive oxygen species (ROS) detection assay were performed. Through the TUNEL assay, we detected I3C-induced DNA fragmentation. Fluorescence-activated cell sorting (FACS) analysis showed that I3C induced an increase in ROS levels and apoptotic rate in a dose- and time-dependent manner in H1299 cells. Western blotting demonstrated that activated forms of caspase-3, caspase-7, caspase-9, and poly (ADP-ribose) polymerase (PARP) were increased in I3C-treated H1299 cells following treatment with I3C. Furthermore, protein expression levels of FOXO3, bim, bax, and phosphorylated ERK and JNK were increased, while those of pAkt, Bcl-xL, and Bcl-2 were decreased by I3C treatment of H1299 cells. To confirm the relationship between cell apoptosis and ROS generation, H1299 cells were treated with I3C simultaneously with N-acetylcysteine (NAC), and it was shown that ROS levels decreased and viability increased. Moreover, in western blot analysis, expression of anti-apoptotic proteins (thioredoxin1, peroxiredoxin-1, Bcl-2, and Bcl-xL) in I3C-treated cells was evidently downregulated and pro-apoptotic proteins (active ASK1 and cleaved PARP) were upregulated compared to cells co-treated with NAC. The study showed that I3C induced downregulation of ROS regulator proteins and elevation of ROS, thus activating apoptotic signaling cascades in human lung cancer H1299 cells.
Introduction
Lung cancer has been a leading cause of cancer deaths globally in the last two centuries and is estimated to increase. Lung cancer is the most common cancer diagnosed, is a leading cause of cancer death in men, and is the second most common cancer in women. 1 Risk factors for lung cancers include familial factors, excessive intake of vitamin B, and predominantly smoking habits. 2 –5 Surgical resection with adjuvant therapy in the early phases may be the most effective treatment, but there are difficulties in early diagnosis of lung cancer. 6 Therefore, prevention of lung cancer is of high value. It is thought that lung cancers can be prevented via dietary adjustment, and epidemiological and dietary studies have already shown an association between vegetable consumption and reduction of cancer risks. 7,8
Indole-3-carbinol (I3C) is a natural substance that is abundant in cruciferous vegetables, such as cabbage, cauliflower, and radishes. I3C has anti-proliferative and apoptosis-inducing effects in various cancer cells through numerous mechanisms.
9
–11
I3C is readily converted into polyaromatic indolic compounds under acidic conditions, such as 3-(methoxymethyl) indole, N1-nitroso3-nitromethylindole, and 3,3’-diindolylmethane (DIM).
12,13
DIM is the major product of I3C conversion and is also a well-known anticancer agent.
14,15
It was also revealed that I3C and indole derivatives have the potential to prevent cancer. I3C has significant anticancer effects against prostate cancer cells both
Reactive oxygen species (ROS) are oxygen-containing chemical species with reactive chemical properties. ROS include free radicals such as hydroxyl radicals (HO), superoxide (O2), and non-radical hydrogen peroxide (H2O2). At low levels, ROS function as signaling molecules that control redox signaling, but damage DNA and proteins at high levels. 17 ROS are persistently generated within the biological system via various pathways. Mitochondria are considered the main source of ROS generation. 18 –20 Immoderate ROS generation results in oxidative damage in cells; thus, maintaining ROS homeostasis is vital for cell growth and survival. 21
Thioredoxins (Trx) are redox proteins with low molecular weight (10–12 kDa) present in both eukaryotic and prokaryotic cells. Trx1 and Trx2 are the two known forms of thioredoxin in humans. Trx1 is predominantly localized in the cytoplasm and Trx2 in the mitochondria. Trx1 is an oxidoreductase that contains a dithiol-disulfide active site. 22 It is known to reduce other proteins via the cysteine thiol-disulfide exchange and has benefits in the reduction of intracellular oxidative stress. 23 Transgenic mice overexpressing Trx have demonstrated reduced oxidative stress and a strengthened resistance against ROS-mediated inflammation. 24 Moreover, thioredoxin is known to be a negative regulator of apoptosis signal-regulating kinase1 (ASK1), identified as a mitogen-activated protein kinase kinase (MAPKKK), which activates various MAPK pathways and induces apoptosis in cells. 25
Peroxiredoxins (Prdx), a novel group of peroxidases, are a ubiquitous redox regulating protein family with a molecular weight of 22–27 kDa. 26 Prdxs are classified into subgroups based on their functional site sequence similarity and their subcellular locations. Among them, Prdx1 was first discovered as an antioxidant enzyme. Prdx1 reduces hydrogen peroxide (H2O2) into 2H2O and is converted to an oxidized state. 26 –29 In its oxidized form, Prdx1 functions as a molecular chaperone and functions as an oxidoreductase by gaining electrons from Trx. 26,30
The anti-carcinogenic effects of I3C in diverse cancer cells have been reported in previous studies. 9,11,13,16,31,32 However, the anti-carcinogenic effects of I3C in p53-independent pathways have not yet been investigated. The aim of this study was to investigate the anti-proliferative and apoptotic mechanism of I3C against non-small cell lung cancer H1299 cells, which lack p53 expression. 33 This study suggests that I3C induces apoptosis through the activation of ASK1, thus upregulating intracellular ROS generation. Upregulation of ROS induces oxidative stress within H1299 cells, which results in the overexpression and activation of pro-apoptotic proteins. Release of cytochrome c leads to activation of caspase cascades, resulting in apoptosis of human non-small lung cancer H1299 cells.
Materials and methods
Reagents
Indole 3-carbinol (I3C) was purchased from Sigma-Aldrich (St. Louis, MO, USA) and dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, St. Louis, MO, USA). Then, 300 mM stock solutions of preparation were stored at −20°C. Dimethyl thiazolyl diphenyl tetrazolium (MTT) was purchased from DoGenBio (Seoul, Korea). Caspase-3, caspase-7, caspase-9, Akt, pAkt, ERK, pERK, JNK, pJNK, PARP, cleaved PARP, Bcl-xL, Bim, p21, Prdx1, ASK1, H2aX, and λH2aX primary antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA).
Cell culture
Human non-small cell lung cancer H1299 cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Human colon fibroblast CCD18-Co cells were purchased from the Korean Cell Line Bank (Seoul, Korea). H1299 cells were cultured in RPMI-1640 (GIBCO, Grand Island, NY, USA), while CCD18-Co cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; GIBCO, Grand Island, NY, USA) containing 10% heat-inactivated fetal bovine serum and 1% antibiotics (penicillin/streptomycin). Both cells were maintained in an incubator under standard temperature (37°C), humidity (95%), and CO2 (5%). The cells were harvested at 70–80% confluence and reseeded for expansion and experiments.
Cell proliferation analysis by MTT assay
H1299 and CCD18-Co cells were seeded in 96-well plates at a density of 5 × 103 cells per well and incubated under standard conditions for 24 h. After 24 h of incubation, the cells were treated with I3C at a concentration gradient of 0 to 700 µM and incubated for 24 or 48 h. After treatment for 24 or 48 h, 20 µL of MTT dye solution (5 mg/mL) was added to each well, and the cells were incubated for 2 h at 37°C. After all the media were removed, Formazan was solubilized in 200 µL DMSO, and light absorbance was measured at a wavelength of 570 nm. The viability of I3C-treated H1299 cells was evaluated in the presence or absence of NAC in order to evaluate the relationship between ROS and cell viability.
Cell proliferation analysis by colony formation assay
H1299 cells (500 cells/well) were seeded in 6-well plates and stabilized in an incubator. After 24 h of incubation, stabilized cells were treated with concentrations of 0 and 400 µM of I3C. The media was changed into fresh media after 24 h of treatment, and cells were cultured for 12 days. PBS washing was performed twice for 3 min and cells were fixed with 4% formaldehyde solution for 20 min at 4°C. Following fixation, the cells were washed twice with PBS for 3 min and stained with 1% crystal violet (Sigma-Aldrich) solution for 30 min at room temperature (25°C). Colonies were counted by using hemocytometer.
Terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling assay
Apoptotic cells were detected via terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling (TUNEL) assay using the fluorometric TUNEL system (Promega, Madison, WI, USA). H1299 cells were seeded in 6-well plates at a density of 4 × 105 per well and cultured for 24 h. Media were removed and cells were fixed with 4% formaldehyde solution for 25 min at 4°C, and their membranes were permeabilized using Triton X-100, diluted to 0.5% in PBS for 10 min. Apoptotic cells were treated with 25 µL TdT enzyme buffer. All cells were then stained using Hoechst stain solution (Sigma-Aldrich). Fluorescence-labeled damaged DNA strands were visualized using a fluorescence microscope (Nikon Eclipse TE 2000-U, Tokyo, Japan).
Annexin V/PI staining assay
An annexin V-FITC apoptosis staining/detection kit (Abcam, Cambridge, UK) was used to detect cellular apoptosis. H1299 cells (4 × 105 cells) were seeded in 6-well plates and cultured for 24 h. After 24 and 48 h of treatment with I3C, each well was washed twice with PBS and harvested using trypsin-EDTA (Sigma-Aldrich, St. Louis, MO, USA). The supernatant was removed after centrifugation, suspended in 1× binding buffer (5 µL of Annexin V-FITC and 5 µL propidium iodide added) and incubated for 5 min at room temperature in the dark. Apoptotic cells were quantified by flow cytometry (Beckman Coulter Brea, CA). Identical experiments were performed for I3C-treated cells with or without NAC.
Cell cycle arrest
To analyze the cell cycle arrest induced by I3C in H1299 cells, the cells were collected after 48 h of treatment with different concentrations of I3C. Then, the cells were suspended in cold 70% ethanol and fixed at −20°C for 18 h. The fixed cells were centrifuged, and supernatants were carefully removed with a pipette. Pelleted cells were incubated with 1 mL of DNA staining solution (50 μg/mL of propidium iodide and 200 μg/mL of DNase-free RNase in PBS with triton-X100 diluted to 0.2% for permeability) for 30 min. An FC 500 series cytometer (Beckman Coulter) was used for acquisition and analysis. Flow cytometric data were organized using the CXP program (Beckman Coulter).
DCF-DA staining assay
Intracellular ROS generation changes in treated cells were observed using a 20,70-dichlorofluorescein diacetate (DCF-DA) cellular ROS detection assay kit. H1299 cells were treated with 0, 100, 200, and 400 µM I3C. At 400 µM, cells were also treated with or without 5 mM NAC for 24 or 48 h. Treated cells were washed twice with PBS and collected using trypsin-EDTA. The collected cells were incubated at 37°C with 25 μM DCF-DA solution for 30 min. ROS generation in cells was analyzed using flow cytometry. An FC 500 series cytometer (Beckman Coulter) was used for flow cytometric analysis. Flow cytometric data were organized using the CXP program (Beckman Coulter).
Western blot analysis
I3C-treated H1299 cells (3 × 106 cells) were harvested and lysed using RIPA buffer (Sigma-Aldrich) with PMSF phenylmethylsulfonyl fluoride (Sigma-Aldrich). Proteins were separated by SDS-PAGE at 110 V for 110 min and transferred to an Immobilon-P PVDF membrane (Merck Millipore, Burlington, MA, USA) at 60 V for 2 h. The membranes were blocked using 3% bovine serum albumin (Bovogen, Australia) and incubated with primary antibodies on a rocker at 4°C overnight. The membranes were washed with TBS-T three times and incubated with HRP-conjugated secondary antibody (Jackson Laboratory, Bar Harbor, USA). Chemiluminescence was enhanced using ECL (Gendepot, Barker, USA) and detected using a Chemi-doc detection system (Bio-Rad, Hercules, CA, USA).
Statistical analysis
The results are expressed as arithmetic mean ± standard deviation. To compare the statistical meaning between the groups, two-sided unpaired Student’s t test was used. Experiments were repeated three times, and the representative data were shown. A one way ANOVA followed by Bonferroni post hoc test was used for statistical analysis and a p value of <0.05 was considered statistically significant.
Results
Indole-3-carbinol (I3C) inhibited proliferation of lung cancer H1299 cells
To verify the anti-proliferative effects of I3C on H1299 cells and CCD18-Co cells, the cells were treated with 0 to 700 µM I3C for 24 and 48 h. Cell proliferation was analyzed by MTT assay. H1299 cells treated with I3C for 24 h showed only slight decreases or no changes in viability at low concentrations, but drastic decreases at concentrations above 400 µM at 48 h. The result shows that I3C has a significant (

Inhibition of the cell viability and proliferation of H1299 cells treated with I3C. (A) Cell cytotoxicity assay of H1299 and CCD18-Co cells was performed with different concentrations of I3C ranging from 0 to 700 μM for 24 h and 48 h. Cell viability was assessed with MTT assay. Single and double asterisks indicate significant differences from the control cells (*
I3C induced apoptosis in H1299 cells
To determine the alteration of cell cycle distribution and apoptotic effect of I3C in H1299 cells, a cell cycle arrest assay and Annexin V/PI staining assay were performed. Flow cytometric analysis was performed in cells treated with I3C (0, 100, 200, and 400 μM). The results showed that treatment of I3C induced a significant (
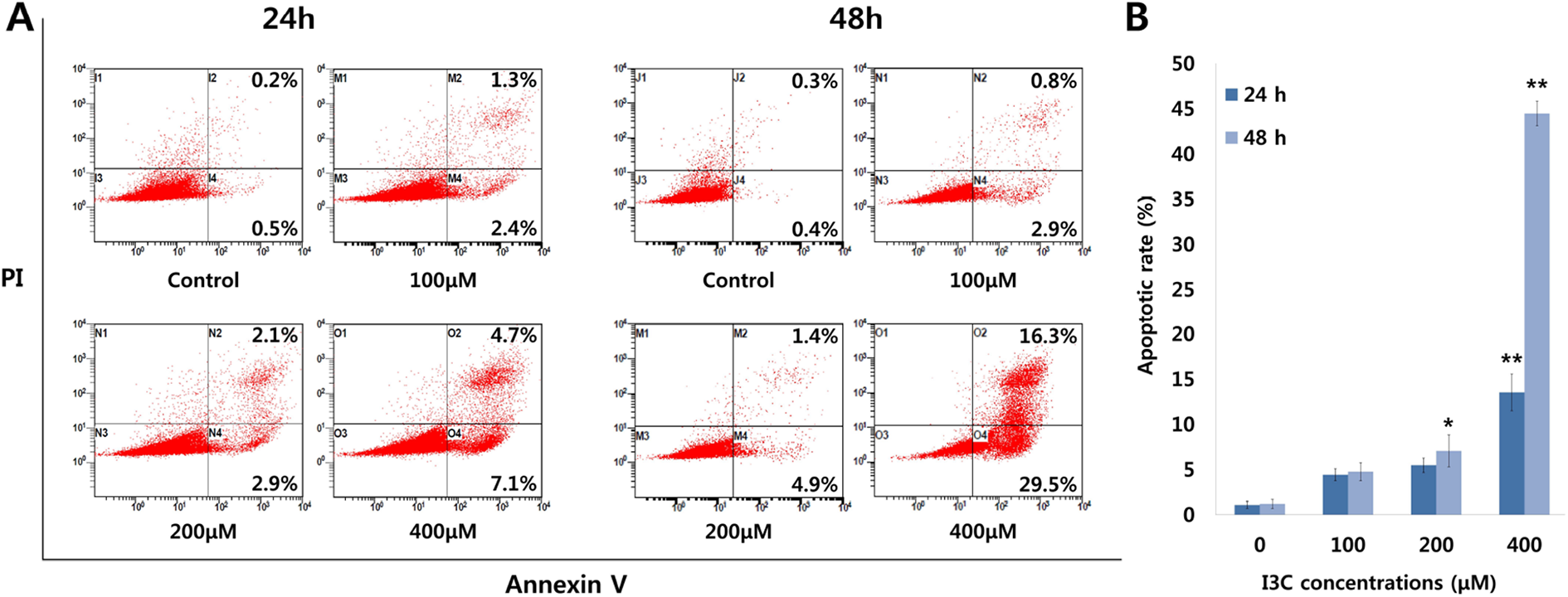
Evaluation of the apoptotic rate in H1299 cells via Annexin V/PI double staining assay after 24 and 48 h treatment with different concentrations of I3C. I3C induced apoptosis in a dose- and time-dependent manner. (A) H1299 flow cytometry dot plots. The percentage of apoptotic cells is shown. (B) Bar graph shows the increases in relative value of apoptotic rate after treatment of I3C for 24 h and 48 h in H1299 cells. The data represent the mean ± SD of three independent experiments. Single and double asterisks indicate significant differences from the control cells (*
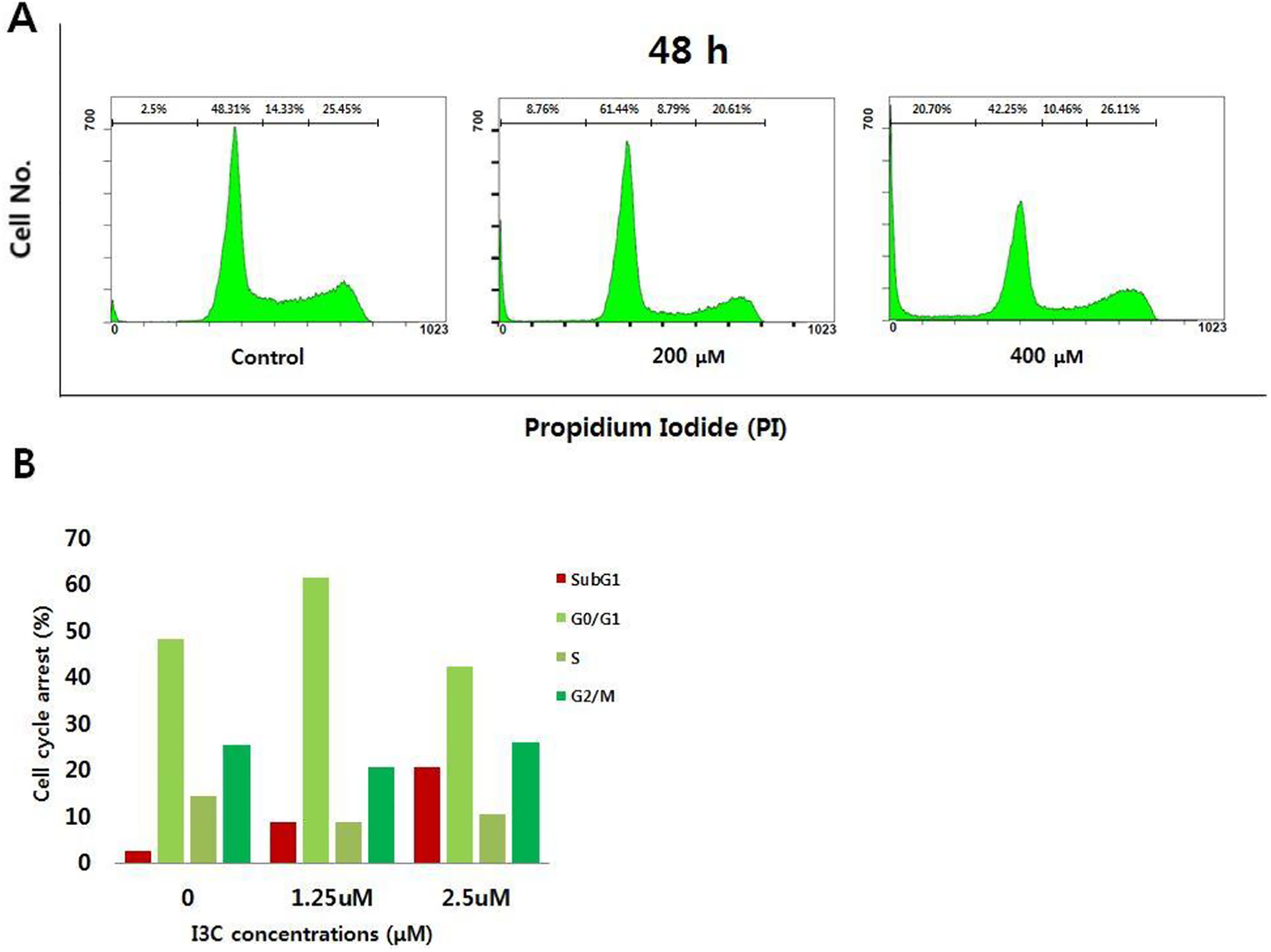
Analysis of the cell cycle progression in I3C-treated H1299 cells. Cells were treated with 400 μM of I3C for 24 or 48 h and then collected, fixed, and stained with propidium iodide (PI) for a flow cytometric analysis of DNA content. The upper panels show representative examples of cell cycle analysis by flow cytometry after PI staining. The low panels represent the results of flow cytometric analyses for the sub-G1 stage of the cell cycle.

Detection of DNA fragmentations in H1299 cells after 24 h and 48 h of treatment with 200 µM and 400 µM of I3C by TUNEL assay. Blue fluorescence shows whole nuclei of cells, stained with Hoechst, while green fluorescence only shows fragmented DNA, labeled with TdT. Hoechst-stained cells images and the TdT-labeled fragmented DNA images were merged to visualize the comparisons of I3C-induced DNA fragmentations in H1299 cells in a dose- and time-dependent manner.
I3C increased ROS levels in H1299 cells
I3C-derived intracellular ROS generation in H1299 cells was quantified by FACS using DCF-DA, permeable fluorescent and chemiluminescent probes. The mean fluorescence intensity (MFI) of H1299 cells treated with 100, 200, and 400 µM I3C was analyzed using a flow cytometer. Treatment with I3C induced a significant (

ROS generation in human lung cancer H1299 cells, treated with I3C. (A) Intracellular ROS generation induced by I3C was measured in H1299 cells using DCFH-DA (10 μM) and flow cytometry after 24 h and 48 h of treatment. Mean fluorescence intensity (MFI) is indicated on each plot. (B) Bar graph shows the quantitation of the MFI. The vector control MFI was set at 100%. The data represent the mean ± SD of three independent experiments. Single and double asterisks indicate significant differences from the control cells (*

Cell viability in H1299 cells treated with I3C in presence or absence of NAC. (A) Cell cytotoxicity assay of H1299 was performed with 400 μM of I3C with or without NAC for 24 h and 48 h. Cell viability was assessed with MTT assay. Single asterisks indicate significant differences from the control cells (*

ROS generation in H1299 cells treated with I3C in presence or absence of NAC. (A) Intracellular ROS generation was measured using DCFH-DA (10 μM) and flow cytometer after 24 h and 48 h of treatment. Mean fluorescence intensity (MFI) indicated on each plot. (B) Bar graph shows quantitation of the MFI. The vector control MFI was set at 100%. The data represent the mean ± SD of three independent experiments. Single asterisks indicate significant differences from the control cells (*

Evaluation of the apoptotic rate in H1299 cell by Annexin V/PI double staining assay after 24 and 48 h of treatment with I3C and NAC. (A) Dot plots represent flow cytometry of I3C- or NAC-treated H1299 cells. The percentages of apoptotic cells are shown. NAC has inhibited the apoptosis induced by I3C. (B) Bar graph shows the variation in relative value of apoptotic rate after treatment of H1299 cells with I3C and NAC for 24 h and 48 h. The data represent the mean ± SD of three independent experiments. Single asterisks indicate significant differences from the control cells (*
I3C induced apoptotic stimulus in H1299 cells
To identify the changes in protein expression levels in apoptotic cells treated with I3C, western blotting analysis was performed. The expression of PARP; cleaved PARP; and caspase-3, -7, and -9, which are important modulators of intrinsic apoptosis, were first measured to verify the apoptotic influence of I3C. The expression of PARP decreased while the cleaved form was upregulated. In addition, inactive caspase-3, -7, and -9 were downregulated, and their active forms were upregulated. Moreover, it was observed that protein levels of Akt and H2AX remained constant, while the expression levels of λH2aX, FOXO3, Bax, and Bim proteins were upregulated and protein levels of pAkt, Bcl-2, and Bcl-xL were downregulated in a dose-dependent manner (Figure 9). These results prove that treatment with I3C induces apoptosis in non-small cell lung cancer H1299 cells.
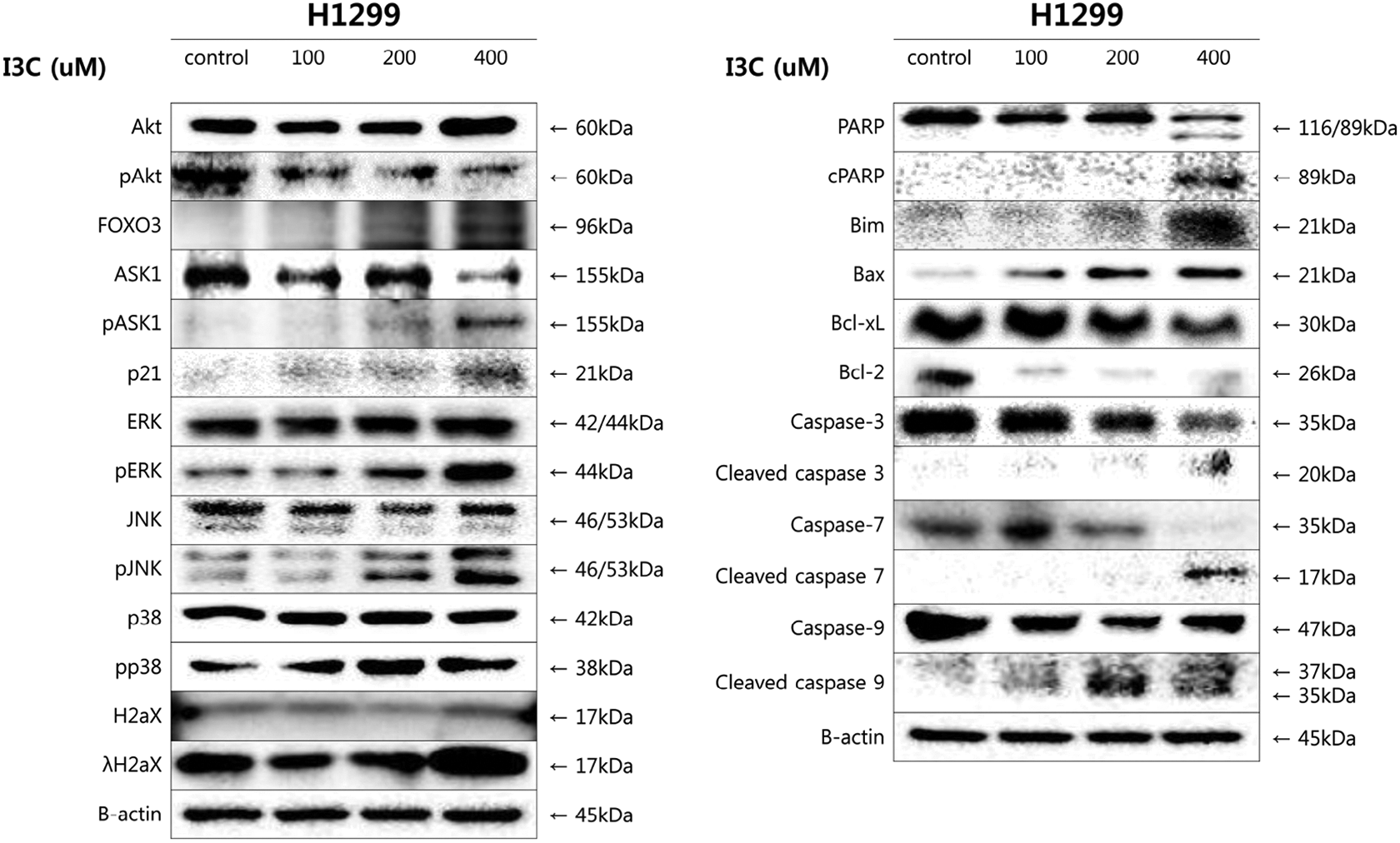
Analysis of protein expression in I3C-treated H1299 cells by western blot analysis. Cells were exposed to 100 μM, 200 μM, and 400 μM of I3C for 48 h. Caspase-3, caspase-7, caspase-9, Bcl-2, Bcl-xL, pAkt, and PARP expressions were decreased in I3C-treated H1299 cells. Expression of FOXO3, JNK, ERK, pp38, p21, Bim, Bax, and cleaved caspases in I3C-treated H1299 cells was increased.
I3C induced apoptotic stimulus in H1299 cells via the ROS-mediated pathway
Western blotting was conducted to compare the expression levels of ROS-related proteins (Ask1, Prdx-1, and Trx1) with those of apoptotic proteins (Bcl-xL, active and inactive forms of caspase7) in control, NAC-treated, I3C-treated, and co-treated H1299 cells. The results showed that protein levels of Prdx1 and Trx1 in the cells treated with I3C were downregulated, while active ASK1 was upregulated. In contrast, the expression of active caspase-7 was increased and Bcl-xL was decreased in I3C-treated cells. These results support the hypothesis that ROS is firmly related to apoptosis in H1299 cells induced by I3C (Figure 10).

Analysis of ROS-related protein expression in H1299 cells, when treated with I3C in the presence or absence of NAC. The active form of ASK1, which inhibits Trx and Prdx, was increased in I3C-treated cells. Therefore, the expression of Trx and Prdx was decreased. To verify apoptosis induced by ROS generation, PARP, Bcl-2, and Bcl-xL expression was analyzed. The result showed the tendency toward apoptosis after treatment with I3C alone, which is suppressed by co-treatment with NAC.
Discussion
Based on age-adjusted data (2013–2017), the rate of new cases of lung and bronchus cancer was 54.2 per 100,000 per year. The death rate was 40.2 per 100,000 per year. In 2020, it is estimated that 228,820 new cases and 123,720 deaths will occur due to lung and bronchus cancer, which will be the leading cause of cancer death and second leading new cause, right after breast cancer. 34 Since lung cancer is expected to be the leading cause of cancer deaths according to the recently reported data on cancer incidence, 35 it is considered necessary to determine the anticancer mechanisms of various agents. The aim of this study was to identify an effective anti-carcinogenic agent and its effect against H1299 cells, which is p53-null non-small cell lung cancer. 36 –38 Since p53 is a well-known critical factor for tumor suppression, p53 malfunction results in enhanced tumor cell growth. Thus, substantial studies must be conducted to discover anticancer agents that display p53-independent actions. 39 As a candidate, I3C induced apoptosis in human non-small cell lung cancer H1299 cells, where p53 is not present, via upregulation of ROS generation.
Indole-3-carbinol is a subjacent substance of glucobrassicin, which is rich in cruciferous vegetables. It is well known for its beneficial effects and particularly its therapeutic effects against various cancers. 32,40 It was previously reported that I3C has an inhibitory effect on cancer growth at lower concentrations and apoptotic effects at higher doses in lung cancer cells. 31 However, the apoptotic effects of I3C in cancer cells that lack p53 expression or its anti-carcinogenic effects derived by intracellular ROS elevation have not yet been thoroughly studied. In this study, the anti-carcinogenic effect of I3C via intracellular ROS generation elevation and its p53-independent apoptotic pathway were examined. To investigate its mechanisms, a cell viability assay and Annexin V/PI double staining assay were performed to verify its apoptotic potential. In addition, western blotting and ROS detection were performed at different concentrations with or without antioxidant agent (NAC) to observe changes in ROS generation and protein expression in I3C-treated cells.
The results showed that the cytotoxic effects of I3C in H1299 cells were mediated by an increase in ROS levels, thus activating apoptosis signaling. I3C-treated H1299 cells showed an over-generated ROS level, induced upregulation of Bax and Bim, and downregulation of Bcl-2 and Bcl-xL. These are the key modifications in cytochrome c release when ROS levels increase and thus programmed cell death is enforced. 41,42 As a sign of apoptosis, the activity of caspase-3, -7, -9, and λH2AX increased and induced upregulation of FOXO3, cleaved PARP, and p21. Although it is known that p21 is a downstream gene of p53, it was revealed that p21 expression can also be upregulated by FOXO3 overexpression. FOXO3 overexpression due to downregulation of maternal embryonic leucine zipper kinase (MELK) induces upregulation of p21 and results in cell cycle arrest. 43 Furthermore, a previous study showed that silencing FOXO3 expression resulted in the reduction of caspase-3-mediated PARP cleavage. 44
Due to immoderate cell proliferation and metabolism, ROS generation in cancer cells is much higher than in normal cells. However, the Trx system of cancer cells allows tumor cells to adapt and survive under oxidative stress conditions, while normal cells do not. 45,46 Therefore, signaling pathways based on the Trx system are also of interest for use in drug targeting techniques. 45,47,21 Thioredoxin (Trx) and related proteins included in the Trx system have been shown to regulate intracellular ROS levels and signal cascades that mediate apoptosis. 21,48 The Trx system consists of multiple proteins and molecules, including apoptosis signal-regulating kinase 1 (ASK1). ASK1 is also known as mitogen-activated protein 3 kinase 5 (MAP3K5) that activates JNK and p38 and tumor necrosis factor-α (TNF-α) to induce stress- and cytokine-induced apoptosis. 49 In this study, treatment with I3C induced the activation of ASK1 and thus activation of JNK and p38 (Figure 6), while Trx1 was downregulated (Figure 10).
Previous studies have shown that peroxiredoxin (Prdx) is also a molecule involved in redox homeostasis regulation and an anti-inflammatory antioxidant in neurodiseases. 50,51 Previous studies have elucidated various types, locations, and mechanisms of Prdx as an antioxidant as well as coordinative function with the Trx system in reducing oxidative stress in mitochondria. 28,52 –54 Prdx is persistently oxidized and recycled (reduced) by Trx and Trx reductase. 55 The catalytic residues and mechanisms of Prdx have been revealed in previous studies; they possess 2 essential cysteine residues that function as peroxidase, peroxidatic cysteine (CP). 56,57 2-Cys catalytic residues are reversibly oxidized (inactivated) by hyperoxidation, from Prdx-SH to Prdx-S-S-Prdx. Inactive Prdxs can be recycled via 2 pathways. In the catalytic cycle, Prdx is reduced by oxidizing Trx, and Prdx uses sulfiredoxin as the reducing agent in the inactivation–reactivation cycle. In both ways, H2O2, Trx, Trx reductase, and NADPH were necessary for hyperoxidation and reactivation. 54,58,59
We investigated the protein expression of Prdx, Trx, and ASK1 in cells treated with I3C with or without an antioxidant agent (NAC). The results showed that ASK1 was activated in H1299 cells treated with I3C, while Prdx was downregulated along with Trx expression (Figure 10). I3C induced the activation of ASK1 in H1299 cells; thus, Trx and Prdx activities were inhibited. Consequently, ROS homeostasis collapses and results in an increase in intracellular ROS. Increased ROS stress leads to the release of cytochrome c from mitochondria and thus the progression of caspase cascades. Inactivation of PARP, which was induced by activation of caspase-3, resulted in persistent DNA damage and thus expression of FOXO3. FOXO3 increases the expression of Bax and Bim, which are necessary for cytochrome c release. Consequently, apoptotic signals are continuously exerted, resulting in apoptosis of H1299 cells (Figure 11). These results show that I3C induces apoptosis via the ROS-mediated pathway.

Schematic representation of I3C-induced apoptosis signaling pathway.
In this study, we investigated the ROS-mediated apoptotic effects of I3C in H1299 cells via FACS analysis and western blotting. However, the experiments were dome only in in vitro and no other supportive data with different cell lines, those share similar features. The further investigations are still required to elucidate the anticancer effects against human lung cancer cells. More underlying mechanisms are to be analyzed with more of cell lines and in animal experiments. We believe that this study may provide alternative therapeutic choice and development of advanced anticancer therapies.
Conclusion
In summary, I3C effectively induced apoptosis in human lung cancer H1299 cells. The possible mechanisms involved in I3C-induced apoptosis in H1299 cells are Trx1 and Prdx-1 downregulation via ASK1 activation. Therefore, the increase in ROS levels induces activation of apoptotic proteins such as the Bcl-2 family; caspase-3, -7, -9; and FOXO3.
Footnotes
Author contributions
HML, MJN, and SHP conceived the presented idea, carried out the experiments, and wrote the manuscript. HML, MJN, and SHP contributed to analysis and interpretation of the results. HML, MJN, SHP provided critical feedback, discussed the results, and contributed to writing the first draft and final manuscript.
Availability of data
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Gachon University (Grant Number: GCU-2019-0823).
