Abstract
The Cosmetic Ingredient Review (CIR) Expert Panel (Panel) assessed the safety of ethanolamine and 12 salts of ethanolamine as used in cosmetics. Ethanolamine functions as a pH adjuster. The majority of the salts are reported to function as surfactants, and the others are reported to function as pH adjusters, hair fixatives, or preservatives. The Panel reviewed available animal and clinical data, as well as information from previous relevant CIR reports. Because data were not available for each individual ingredient and because the salts dissociate freely in water, the Panel extrapolated from those previous reports to support safety. The Panel concluded that these ingredients are safe in the present practices of use and concentrations (rinse-off products only) when formulated to be nonirritating, and these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
Keywords
Introduction
This report assesses the safety of ethanolamine and ethanolamine salts which are defined in the International Cosmetic Ingredient Dictionary and Handbook as cosmetic ingredients. 1
In 1983, the Cosmetic Ingredient Review (CIR) Expert Panel issued a safety assessment on triethanolamine (TEA), diethanolamine (DEA), and monoethanolamine (MEA). In that 1983 report, the Panel concluded that ethanolamine, an ingredient that functions in cosmetics as a pH adjuster, is safe for use in cosmetic formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin, and ethanolamine should only be used in rinse-off products. 2
In 2010, the Panel decided to reopen that 1983 safety assessment on TEA, DEA, and MEA as 3 separate reports (1 for each ingredient) and to include additional related ingredients in each of the new reviews. Diethanolamine and its salts and TEA and TEA-containing ingredients have been found safe in the present practices of use and concentration when formulated to be nonirritating, with the caveat that these ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. 3,4
The International Nomenclature Cosmetic Ingredient (INCI) name for MEA is now ethanolamine. Ethanolamine and the acid salt ingredients (listed subsequently) would be expected to dissociate into ethanolamine and the corresponding acid, some of which have been reviewed separately. In most cases, this means that the composition of these salts is stoichiometrically half ethanolamine (ie, as its conjugate acid).
The following 12 salts are included in the rereview of ethanolamine:
Inorganic acid salts
Ethanolamine HCl MEA-sulfite
Organic acid salts
MEA-benzoate MEA-salicylate MEA-cocoate MEA-tallowate MEA-undecylenate MEA-laureth-6 carboxylate MEA PPG-6 laureth-7 carboxylate MEA-PPG-8-steareth-7 carboxylate
Organic-substituted inorganic acid salts
MEA-lauryl sulfate MEA-laureth sulfate
Ethanolamine HCl is reported to function as a pH adjuster and a buffering agent. MEA-sulfite is reported to function as a hair fixative. The majority of the ethanolamine salts are reported to function as surfactants. However, MEA-benzoate and MEA-salicylate are reported to function as preservatives and not as surfactants.
MEA-salicylate also has been reviewed previously by the CIR Expert Panel. In 2003, the Panel concluded that MEA-salicylate is safe as used when formulated to avoid skin irritation and when formulated to avoid increasing the skin’s sun sensitivity or when increased sun sensitivity would be expected, and directions for use include the daily use of sun protection. 5
The definitions and structures of the ingredients in this report are provided in Table 1. The conclusions of the components that have been reviewed previously by the CIR are provided in Table 2.
Definition and Structures of Ethanolamine and Ethanolamine-Containing Ingredients.
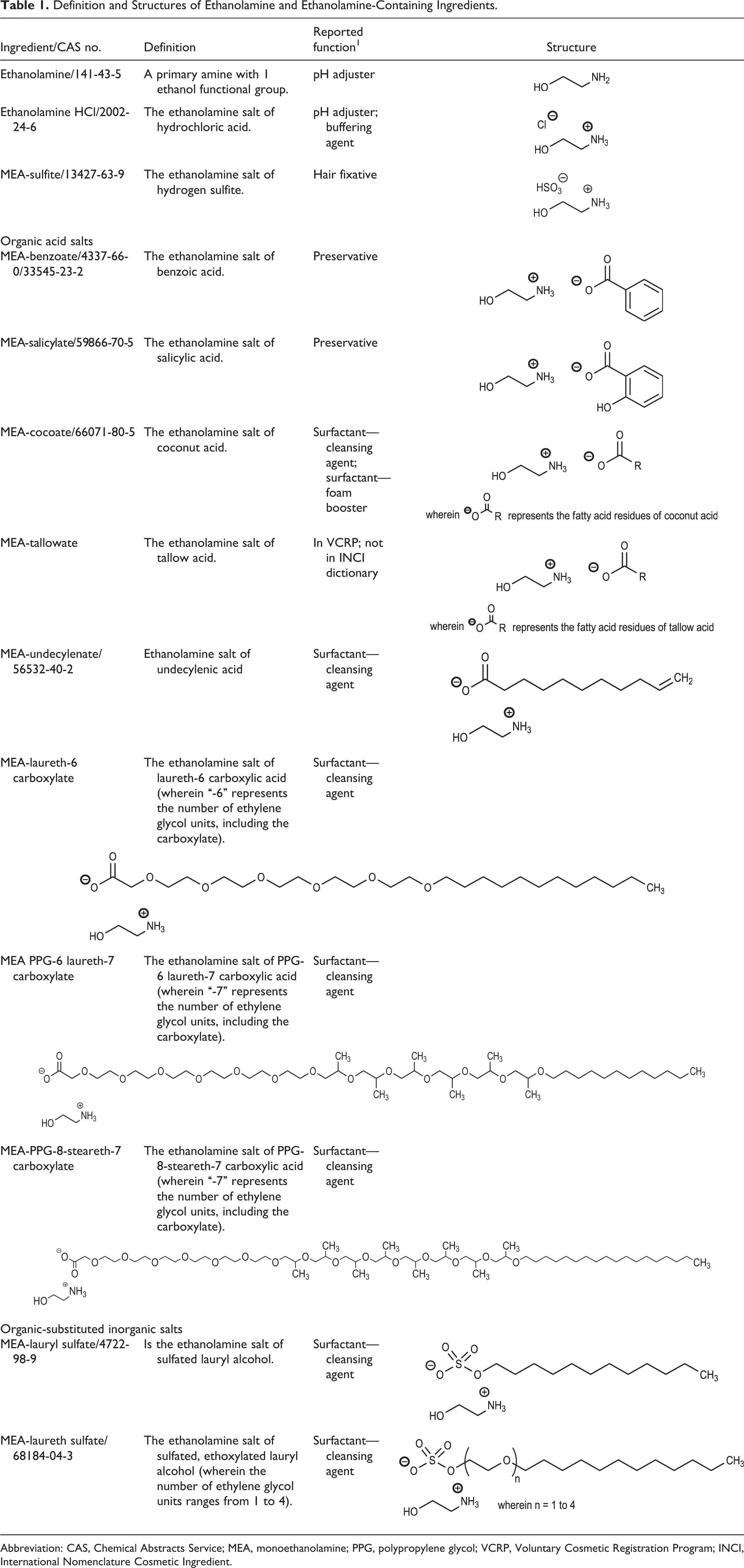
Abbreviation: CAS, Chemical Abstracts Service; MEA, monoethanolamine; PPG, polypropylene glycol; VCRP, Voluntary Cosmetic Registration Program; INCI, International Nomenclature Cosmetic Ingredient.
Conclusions of Previously Reviewed Ingredients and Components.
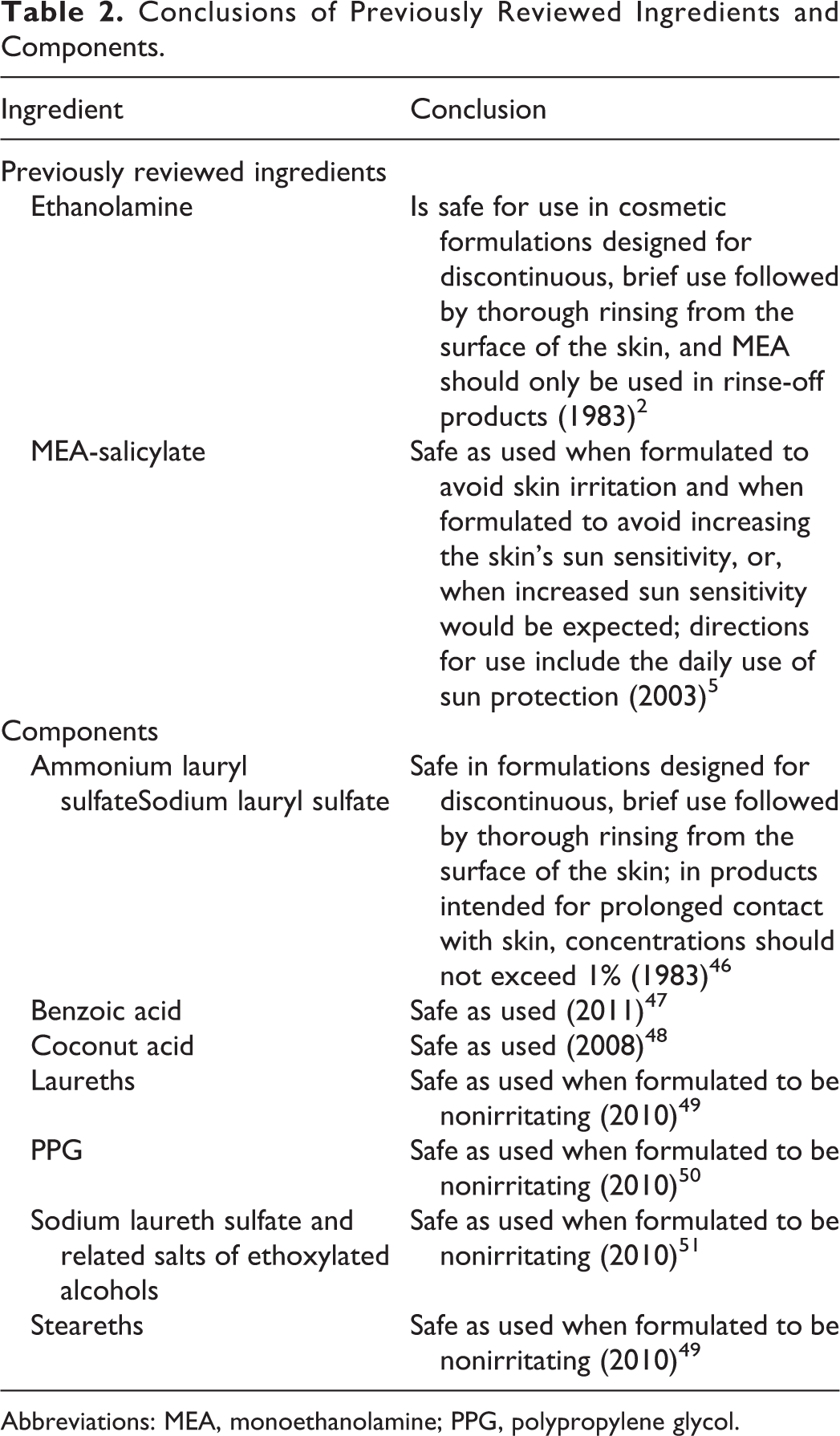
Abbreviations: MEA, monoethanolamine; PPG, polypropylene glycol.
Chemistry
Ethanolamine is an amino alcohol (Figure 1). Ethanolamine is produced commercially by aminating ethylene oxide with ammonia. The replacement of 1 hydrogen of ammonia with an ethanol group produces ethanolamine.

Ethanolamine.
Ethanolamine is reactive and bifunctional, combining the properties of alcohols and amines. At temperatures of 140°C to 160°C, ethanolamine will react with fatty acids to form ethanolamides. Additionally, the reaction of ethanolamines and sulfuric acid produces sulfates, and under anhydrous conditions, ethanolamine may react with carbon dioxide to form carbamates. 2
While many secondary amines are readily nitrosated to form isolatable nitrosamines, primary amines, such as ethanolamine, ultimately yield diazonium salts instead of nitrosamines. 6
Acid Salts: The acid salts (inorganic acid salts, organic acid salts, and sulfate salts) of ethanolamine are ion pairs, which freely dissociate in water (eg, Figure 2). Therefore, these salts are closely related to the corresponding free acids and ethanolamine. In other words, MEA-undecylenate, for example, is comprised of undecylenic acid and ethanolamine. Available physical and chemical properties are summarized in Table 3.
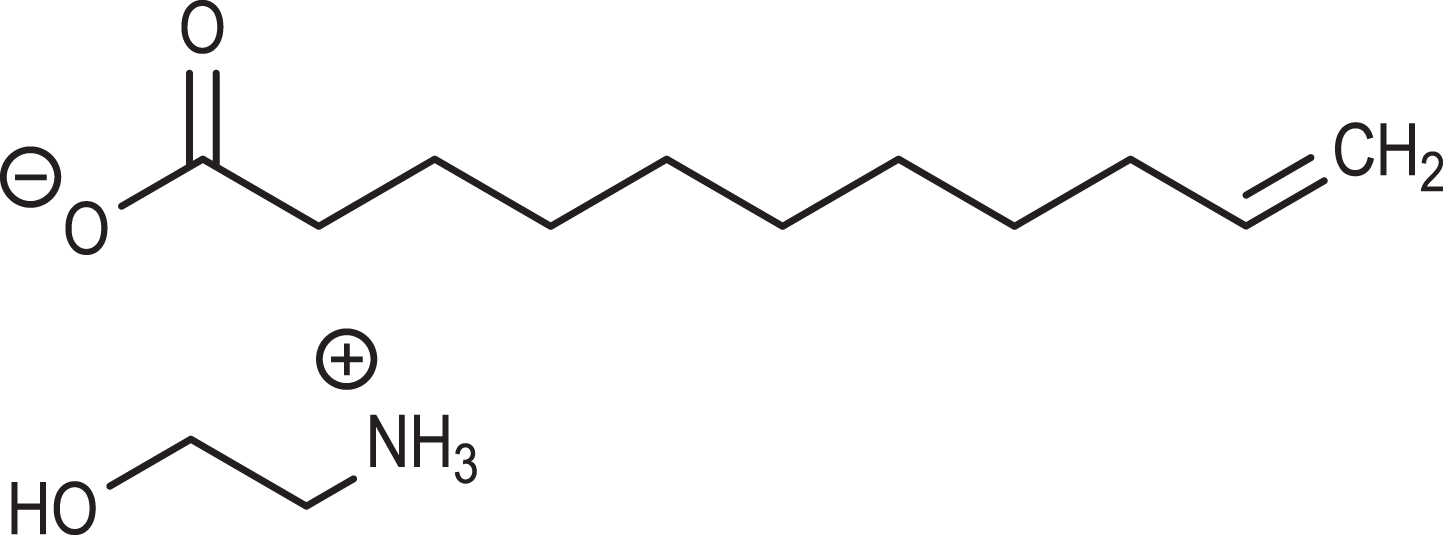
MEA-undecylenate.
Physical and Chemical Properties.
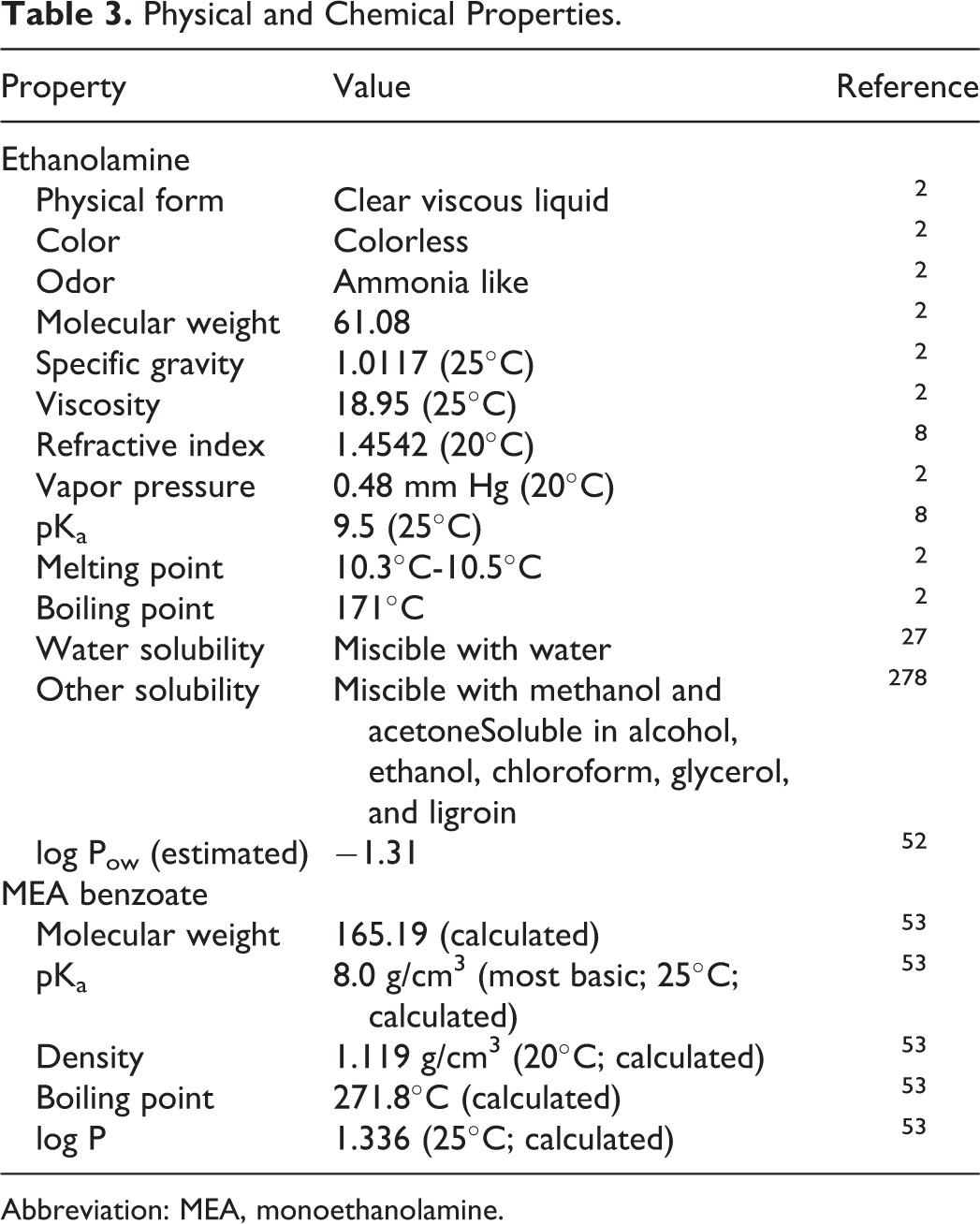
Abbreviation: MEA, monoethanolamine.
Method of Manufacture
Ethanolamine is produced by reacting 1 mol of ethylene oxide with 1 mol of ammonia. Typically, ethylene oxide is reacted with ammonia in a batch process to produce a crude mixture of approximately one-third each ethanolamine, DEA, and TEA, which is then separated, achieving varying degrees of single component purity. 7 Ethanolamine combines with long-chain fatty acids to produce neutral carboxylates, also called alkanolamine soaps. 8
Impurities
A supplier reported in an earlier CIR safety assessment that ethanolamine contains a small amount of DEA. 2 Residual ethylene oxide is not expected during the manufacture of ethanolamine. 7
Use
Cosmetic
Ethanolamine and ethanolamine HCl are reported to function in cosmetics as pH adjusters, and ethanolamine HCl is also reported to function as a buffering agent. 1 MEA-sulfite is reported to function as a hair fixative. The majority of the ethanolamine salts are reported to function as surfactants. However, MEA-benzoate and MEA-salicylate are reported to function as preservatives, not surfactants.
Amines, such as ethanolamine, can be useful in pH adjustment because they can both donate a proton in an alkaline environment and accept a proton in an acidic environment. For ethanolamine HCl, a proton has already been accepted from HCl but ethanolamine HCl can certainly function as a proton donor.
The Food and Drug Administration collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). The VCRP data obtained in 2011 report that ethanolamine is used in 788 formulations. 9 All of the uses are in rinse-off products (Don Havery, personal communication, October 18, 2011), and 772 uses are in hair-coloring formulations. 10 A few of the other ethanolamine-containing ingredients are in use, but all of those are reported to be used in less than 15 rinse-off formulations.
According to the data submitted by industry in response to a survey of the maximum reported use concentration by category conducted by the Personal Care Products Council (Council), ethanolamine is used in rinse-off products (only) at up to 18%. 11 MEA-lauryl sulfate has the highest reported concentration of use, with a concentration of 35% reported in hair dye formulations. Use data for ethanolamine and all other in-use ethanolamine-containing ingredients are provided in Table 4. Ethanolamine ingredients not reported to be in use, according to VCRP data and the Council survey, are listed in Table 5.
Frequency and Concentration of Use According to Duration and Type of Exposure.
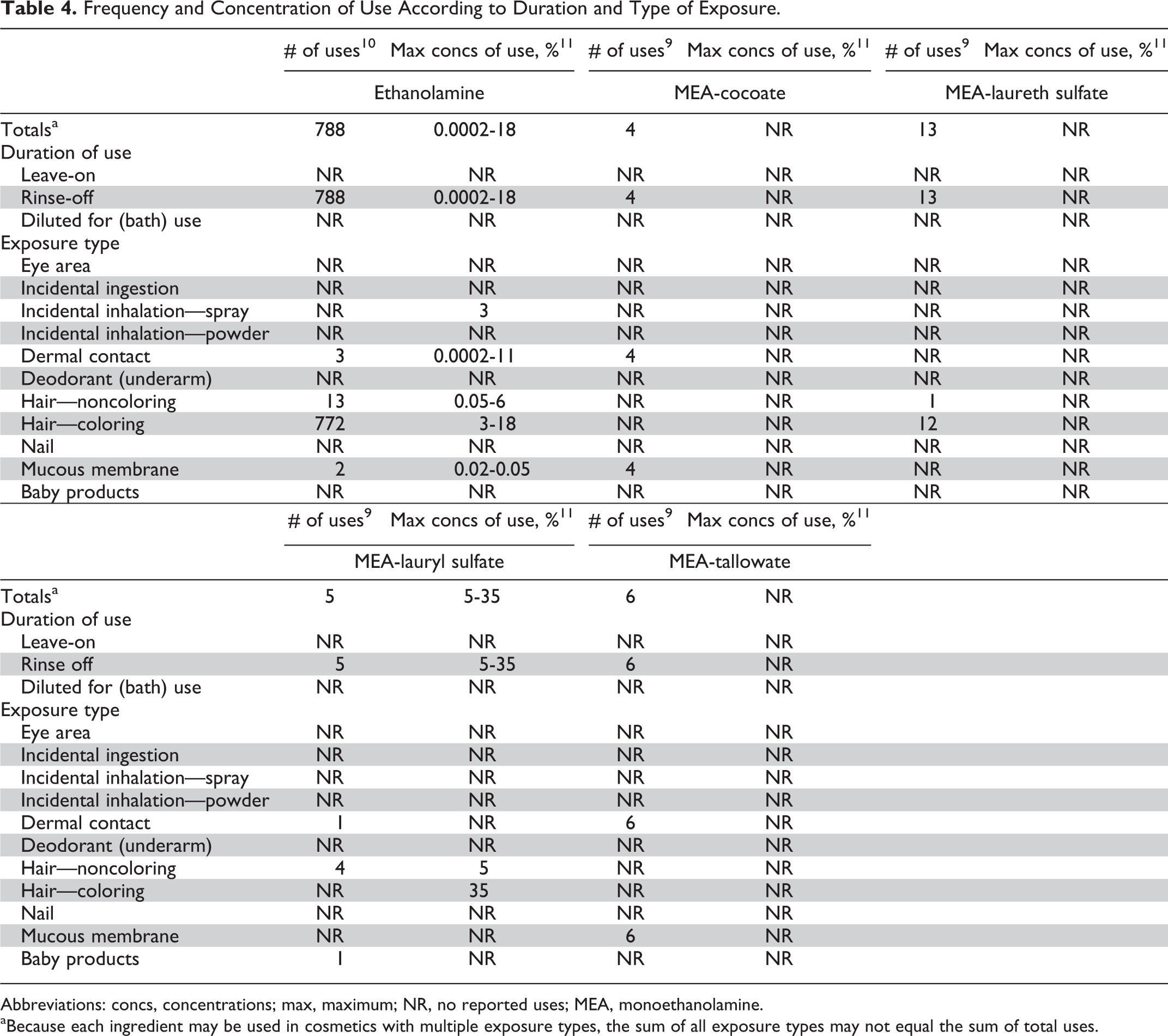
Abbreviations: concs, concentrations; max, maximum; NR, no reported uses; MEA, monoethanolamine.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
Ingredients Not Reported to be in Use.

Abbreviations: MEA, monoethanolamine; PPG, polypropylene glycol.
Ethanolamine is used in a hair color aerosol spray at a maximum concentration 11 of 3% and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 12 –15 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 12,14
Monoalkylamines, monoalkanolamines, and their salts are listed in the Cosmetics Regulation (European Commission [EC]) No 1223/2009 of the European Parliament and of the Council in Annex III: the list of substances which cosmetic products must not contain, except subject to the restrictions and conditions laid down. 16 These restrictions for these ingredients include an allowed maximum secondary amine content of 0.5% in finished product, do not use with nitrosating agents, minimum purity of 99%, maximum secondary amine content of 0.5% for raw materials, maximum nitrosamine content of 50 µg/kg (ppb), and must be kept in nitrite-free containers. MEA-benzoate is listed by the EC in Annex V, a list of preservatives allowed in cosmetics; as a preservative, MEA-benzoate is allowed a maximum concentration in ready-for-use preparations of 0.5% as benzoic acid. MEA-salicylate is also listed in Annex V; as a preservative, MEA-salicylate is allowed a maximum concentration in ready-for-use preparations of 0.5% as salicylic acid, and it is not to be used in products for children under 3 years of age, except shampoos. The ingredients named in this report that have EC restrictions are listed in Table 6.
Status for Use in Europe According to the EC CosIng Database for Ethanolamine Ingredients.

Abbreviations: MEA, monoethanolamine; PPG, polypropylene glycol; EC, European Commission.
Information provided by Health Canada indicates that ethanolamine is used primarily in hair coloring products, with concentrations of use of ≤30% (Health Canada, personal communication, October 29, 2010). Ethanolamine is also reported to be used in Canada at concentrations of ≤30% in noncoloring hair preparations and rinse-off formulations, and at ≤10% in leave-on formulations; and this includes reported use at 0.3% to 3% in baby products and 3% to 10% in lipsticks.
Noncosmetic
Ethanolamine is used in the manufacture of various noncosmetic substances such as those used in textiles and for agricultural purposes. 2 Ethanolamine has uses as an indirect food additive. According to 21 CFR 175.105, ethanolamine is allowed for use as a component of adhesives. 17 Ethanolamine is also used as a rust inhibitor in water-based metalworking fluids. 18
Ethanolamine is approved as an inactive ingredient in drugs in delayed action tablets (maximum potency 1 mg), and ethanolamine HCl is approved for use in intravenous drugs (maximum potency 0.15%). 19
Toxicokinetics
Ethanolamine occurs naturally in phospholipids known as phosphatides. 8 Ethanolamine is a structural component of the phospholipids as part of the headgroups in phospholipid bilayers. In man and other animals, the alcohol group of ethanolamine is phosphorylated, and phosphorylated ethanolamine is transferred to cytidine monophosphate to form cytidine-5′-diphosphoethanolamine and phospholipids via diacylglycerol.
In Vitro
Ethanolamine
Three full-thickness skin preparations from CD rats, CD-1 mice, and New Zealand white rabbits, and 6 samples from female mammoplasty patients were used to compare the dermal penetration of ethanolamine through skin of different species. 20 [14C]Ethanolamine (98.8% purity and specific activity [sp. act.] 15.0 mCi/mmol) was applied to the skin sample undiluted or as an aqueous (aq) solution at a dose of 4 mg/cm2. Dose volumes of 7 µL undiluted [14C]ethanolamine or 32 µL of the aq solution (22% w/w) were applied to the exposed surface of the skin (1.77 cm2) for 6 hours. With undiluted ethanolamine, the cumulative dose absorbed (found in the effluent) was 5.98%, 16.92%, 8.66%, and 0.61% through rat, mouse, rabbit, and human skin, respectively. With aq ethanolamine, the cumulative dose absorbed was 1.32%, 24.79%, 1.87%, and 1.11% through rat, mouse, rabbit, and human skin, respectively. In vitro absorption of undiluted and aq ethanolamine was much greater through mouse skin than human skin, and human skin was the least permeable of all the skin samples. With human skin samples, the cumulative dose absorbed was greater with aq ethanolamine than with undiluted ethanolamine. The researchers hypothesized that enhanced penetration of aq ethanolamine may be attributable to elevated skin hydration.
Ethanolamine HCl
The dermal penetration of [1,2-14C]ethanolamine HCl was evaluated in vitro using split-thickness skin from weanling Yorkshire pigs. 21 An ethanolic solution was prepared so that a 5-µL application to 0.8 cm2 of skin gave a chemical dose of 4 µg/cm2 and a radioactive dose of approximately 0.05 µCi. After 50 hours, 11% of the dose was lost to evaporation, 5% penetrated percutaneously, and 62% was recovered in the skin residue. Seven to nine times more radioactivity was recovered in the upper 100 µm layer compared to recovery from the remaining dermis.
Dermal
Nonhuman
The distribution and metabolism of ethanolamine were determined using groups of 5 male athymic nude mice with human skin grafts and ungrafted athymic nude mice.
21
[1,2-14C]Ethanolamine HCl in ethanol was applied at a dose of 4.0 µg (3.6 µCi) to a 1.45 cm2 area of grafted or nongrafted skin. Penetration appeared similar for both groups. Radioactivity in expired carbon dioxide (C
The radioactivity in proteins and amino acids isolated from the liver, human skin grafts, and mouse skin was determined as an evaluation of the metabolism of ethanolamine. The researchers stated that the appearance of 14C in skin and hepatic amino acids and proteins, and the incorporation of ethanolamine into phospholipids, was evidence of extensive metabolism of the absorbed ethanolamine. The liver was the most active site of ethanolamine metabolism.
Oral
Nonhuman
In a dietary 2-generation reproductive toxicity study (described later in the Reproductive and Developmental Toxicity section), blood samples were taken from groups of 10 male and 10 female F0 and F1 Wistar rats that were fed 0, 100, 300, or 1000 mg/kg ethanolamine HCl for 10 weeks. 22 Plasma levels of ethanolamine, calculated as ethanolamine HCl, increased in a dose-dependent manner. The plasma concentration of ethanolamine was <3 mg/kg for control male and female F0 and F1 animals. In the low-dose group, the plasma concentration values of ethanolamine were <4 mg/kg, in the mid-dose animals, the levels were 8 to 11 mg/kg, and in the high-dose animals, the plasma ethanolamine levels ranged from 60 to 81 mg/kg.
Other
Nonhuman
Five male athymic nude mice were dosed intraperitoneally with 4.0 µg (3.6 µCi) of [1,2-14C]ethanolamine HCl in ethanol, and the distribution and metabolism were examined following dosing.
7
Radioactivity was detected in expired C
Toxicological Studies
Acute (Single) Dose Toxicity
Dermal
Female C3H mice were used to determine the in vivo and in vitro morphological response of mouse skin exposed to a single application of 1%, 5%, or 10% ethanolamine in acetone. 23 In vivo, ethanolamine was applied to an area of skin 1 in in diameter, the animals were killed the next day, and the treated skin was removed and processed. In vitro, ethanolamine was applied to a 1-in diameter mouse skin disc for 1 minute, and the skin discs were then cultured for 20 hours. No lesions were observed in vivo or in vitro at any concentration tested. Lactate dehydrogenase activity in the in vitro samples was statistically significantly elevated with 5% and 10% ethanolamine, suggesting that ethanolamine was mildly toxic to the skin at these concentrations.
The dermal median lethal dose (LD50) in rabbits is reported as 1.0 to 2.5 g/kg ethanolamine. 8 In a study determining the acute dermal toxicity of a mixture containing ethanolamine (as well as hydroxylamine, diglycolamine, propylene glycol, catechol, and water; percentages not specified), 2 g/kg of the mixture were applied to the shaved backs of 5 male and 5 female rabbits. 24 The dermal LD50 of the mixture was greater than the 2 g/kg dose. In a similar study with a formulation containing ethanolamine (and hydroxylamine, diglycolamine, gallic acid, and water; percentages not specified), the LD50 of the mixture in rabbits was 1.24 g/kg body weight (bw). 25
Oral
In oral studies, the LD50 of ethanolamine is reported as 0.7 to 15.0 g/kg in mice and 1.0 to 2.9 g/kg in rabbits. 8 In a study determining the acute oral toxicity of a mixture containing ethanolamine (as well as hydroxylamine, diglycolamine, propylene glycol, catechol, glycolic acid, and water; percentages not specified), groups of 5 male and 5 female rats were used. 26 The oral LD50 of the mixture was 0.95 g/kg. In a similar study with a formulation containing ethanolamine (and hydroxylamine, diglycolamine, gallic acid, and water; percentage in formulation not specified), the oral LD50 of the mixture was 0.815 g/kg bw.
Inhalation
The acute inhalation toxicity of a mixture containing ethanolamine (as well as hydroxylamine, 1-amino-propano-2-ol, catechol, and water; percentage of each not specified) was determined using groups of 5 male and 5 female rats. The animals were exposed to 1.11, 1.61, 2.13, or 2.84 mg/L for 4 hours. All animals exposed to 2.84 mg/L died. All animals in the other test groups survived. The inhalation median lethal concentration (LC50) of the mixture was estimated to be 2.48 mg/L. 26
Repeated Dose Toxicity
Dermal
In a previous report, percutaneous application of 4 mg/kg/d ethanolamine to rats resulted in nonspecific histological changes in the heart and lung, fatty degeneration of the liver parenchyma, and subsequent focal liver necrosis. The duration of dosing was not specified. 2
Oral
The report cited previously also included a study group of 10 rats that were fed 0 to 2.67 g/kg/d ethanolamine for 90 days. Heavy livers and kidneys were observed at ≥0.64 g/kg/d, and deaths and “major pathology” occurred with ≥1.25 g/kg/d. In a 2-year dietary study in which groups of 12 Beagle dogs were fed 0 to 0.0975 g/kg/d of a hair dye composite that contained 22% ethanolamine, no toxic effects were observed. 2
Inhalation
The dominant effects of “continuous exposure” of dogs, guinea pigs, and rats to 5 to 6 ppm ethanolamine vapor were skin irritation and lethargy. No dogs or rodents exposed to 12 to 26 ppm ethanolamine vapor for 90 days died, but mortality was reported in dogs exposed to 102 ppm ethanolamine vapor for 2 days and rodents exposed to 66 to 75 ppm ethanolamine vapor for 24 to 28 days. Dogs and rodents exposed to 66 to 102 ppm ethanolamine vapor had behavioral changes, pulmonary and hepatic inflammation, hepatic and renal damage, and hematological changes. 2
Reproductive and Developmental Toxicity
Dermal
The developmental toxicity of dermally applied aq ethanolamine was evaluated using groups of 30 to 45 gravid Sprague-Dawley rats and 15 gravid New Zealand white rabbits. 27 Ethanolamine was applied at doses of 0, 10, 25, 75, and 225 mg/kg bw/d to the backs of rats on days 6 to 15 of gestation, with a dose volume of 1 mL/kg. Significant skin irritation, consisting of erythema followed by necrosis, scabs, and scar formation, occurred with 225 mg/kg ethanolamine but not at the other dose levels. No effects on kidney or liver weights were reported. Despite maternal effects in the 225 mg/kg dose group, no effects on reproductive parameters were observed at any dose, and there were no treatment-related increases in the visceral or skeletal malformations. In rats, the no-observed effect level (NOEL) was 75 mg/kg/d for maternal toxicity and 225 mg/kg/d for embryonal/fetal toxicity.
In rabbits, 0, 10, 25, and 75 mg/kg bw/d ethanolamine was applied to the back (2 mL/kg) on days 6 to 18 of gestation. Severe skin irritation at the application site, consisting of necrosis, exfoliation, and crusting, was observed in the 75 mg/kg group, and crusting, transient erythema, and edema were observed in a few rabbits of the 25-mg/kg dose group. No effects on kidney or liver weights were reported. As with the rats, no effects on reproductive parameters were observed at any dose, and there were no treatment-related increases in the visceral or skeletal malformations. In rabbits, the NOEL was 10 mg/kg bw/d for maternal toxicity. No effect on embryonal/fetal toxicity was observed at the highest dose level of 75 mg/kg bw/d.
Oral
Ethanolamine
The teratogenic potential of ethanolamine was evaluated in a Chernoff-Kavlock postnatal mouse screening assay. 28 Gravid CD-1 mice were dosed orally with 850 mg/kg/d ethanolamine on days 6 to 15 of gestation, resulting in 16% mortality of maternal mice and reduced numbers of viable litters. Litter size, percentage survival of pups, birth weight, and pup weight gains were not affected.
A preliminary study was performed in which gravid Wistar rats were dosed orally with 0 to 500 mg/kg/d aq Ethanolamine, details not provided. 29 Maternal effects were observed in the high-dose animals, but no embryonal/fetal effects were observed at any dose. Based on these results, groups of 40 Wistar rats were dosed by gavage with 0, 40, 120, or 450 mg/kg bw/d aq ethanolamine on days 6 to 15 of gestation. On day 20 of gestation, 25 dams/group were killed and necropsied, while the remaining 15 were allowed to litter and then killed on day 21 of lactation. Despite evidence of maternal toxicity in the 450 mg/kg group, as indicated by statistically significant decreases in maternal body weights and feed consumption, no effects on reproductive parameters, incidences of visceral or skeletal malformations, or postnatal growth were observed at any dose. The NOEL for maternal toxicity was 120 mg/kg/d. No effect on developmental toxicity was observed at the highest dose level of 450 mg/kg/d.
Groups of 10 gravid Long-Evans rats were dosed by gavage with 50, 300, or 500 mg/kg aq ethanolamine on days 6 to 15 of gestation, and a negative control group of 34 gravid rats was dosed with water only. 30 The animals were killed and examined on day 20 of gestation. Significant maternal toxicity was not noted. Embryolethality was significantly increased in the 500-mg/kg group, and male pups were affected more than female pups. Male pups were also more severely affected than female pups at all dose levels in regard to intrauterine growth retardation and increased gross structural anomalies that were considered indicative of depressed fetal growth. However, pups of either sex who were contiguous to male siblings were more adversely affected that those contiguous to 1 or more female siblings. Male pups contiguous to male siblings (mMm) were, apparently, resorbed (mRm). The number of malformed pups per dam was significantly increased in animals dosed with 300 mg/kg ethanolamine. The number of malformed pups, when evaluated as a percentage of the litter affected, was significantly increased for all 3 dose groups.
Ethanolamine HCl
Groups of 25 male and 25 female Wistar rats (F0 parental generation) were fed 0, 100, 300, or 1000 mg/kg bw/d ethanolamine HCl for at least 75 days prior to mating. 22 Twenty-five male and 25 female F1 pups/group were continued on the test diets of their parents and then mated, becoming the F1 parental generation. The study was terminated after the weaning of the F2 pups. No test substance-related adverse reproductive, developmental, or toxic effects were observed in any of the pups or parental animals of the 100- or 300-mg/kg bw/d groups. In the 1000-mg/kg group, adverse effects on fertility and reproduction were evidenced by statistically significant decreases in absolute and relative weights of the epididymides and cauda epididymidis in male F0 and F1 parents and statistically significant decreased number of implantation sites, increased postimplantation loss, and smaller litters in female F0 and F1 parents. Sperm head count in the cauda epididymidis and absolute and relative liver weights were also statistically significantly decreased in male F0 parents. Body weight gains of both the F0 and F1 parental animals were statistically significantly decreased during gestation when compared to controls, as was feed consumption during lactation. There were no test-related signs of developmental toxicity observed in F1 and F2 pups of this dose group. The NOAEL was 300 mg/kg bw/d for systemic toxicity and for reproductive effects. No effect on developmental toxicity was observed at the highest dose level of 1000 mg/kg/d.
Genotoxicity
In Vitro
Ethanolamine was negative in most Ames tests at concentrations ≤2000 μg/plate 31,32 ; however, “weak mutagenic effects” were reported in 1 study. 33 Ethanolamine, 25 to 500 µg/mL, was negative in a cell transformation assay using hamster embryo cells. 34 Ethanolamine was clearly cytotoxic at 500 µg/mL. Ethanolamine did not produce chromosomal aberrations in rat liver cells, 31 but a “weak positive” response was reported in human lymphocytes. 33 (concentrations tested were not provided.)
Carcinogenicity
Published carcinogenicity data were not found on ethanolamine or ethanolamine salts.
Irritation and Sensitization
Dermal Irritation
Nonhuman
Ethanolamine was corrosive to rabbit skin with single semiocclusive patches of 30%, 85%, and 100% on intact and abraded skin. When applied to rabbit ears using nonocclusive applications and to shaved skin of the abdomen under semiocclusive patches, ≥10% was corrosive, >1% was extremely irritating, and 1% was irritating. 2
Human
In a study in which a formulation containing 5.9% ethanolamine was applied to the backs of 12 female subjects for 23 h/d for 21 days, the formulation was considered an experimental cumulative irritant. Results observed during the induction phase of a patch test of 165 subjects using a formulation containing 11.47% ethanolamine were interpreted by the Panel as irritation. 2
Sensitization
Nonhuman
A local lymph node assay was performed to evaluate the sensitization potential of ethanolamine (as the hydrochloride salt). 18,35 Groups of 6 female CBA/Ca mice were treated on the ear with 10%, 40%, or 70% (w/w) aq ethanolamine HCl. Ethanolamine HCl did not have a skin sensitizing effect.
In a maximization study using 15 Dunkin-Hartley guinea pigs, intradermal and epicutaneous inductions with 0.6% and 10.3% aq ethanolamine, respectively, were performed after 10% sodium lauryl sulfate pretreatment. 18 (Ethanolamine contained less than 0.1% DEA and TEA.) At challenge with 0.41%, 2.05%, or 4.1% ethanolamine, 3, 2, and 3 animals, respectively, reacted positively after 3 days. Of the 15 test animals, 2 reacted to the vehicle, but none of the control animals reacted to ethanolamine or the vehicle. Possible cross-reactions to 5% TEA occurred in 3 animals and to 7% DEA in 2 animals. In a second test using the same protocol, no animals reacted to 0.41% ethanolamine, 2 reacted to 2.05% ethanolamine, and 1 reacted to 4.1% ethanolamine. Additionally, 1 animal reacted to 10% TEA and 2 reacted to 7% DEA. The control animals did not react to any of the ethanolamines.
Human
Based on a study from a previous report, a formulation containing 5.9% ethanolamine, tested undiluted in a 48-hour occlusive patch test with a 10-day nontreatment period prior to challenge, and one containing 11.47% ethanolamine, tested at 5% in 25% alcohol in a repeated insult patch test using semiocclusive patches, were not sensitizing in clinical studies. 2
Provocative testing
Metalworkers with dermatitis were patch tested with 2% ethanolamine in petrolatum (pet). 36 The patches were applied for 1 to 2 days. On day 3, 3 (1.9%) of 155 patients had positive reactions.
Patch testing was performed with 2% ethanolamine in pet in 199 patients with suspected metalworking fluid contact dermatitis. 37 (All patients were metalworkers.) Patches were applied for 1 or 2 days. On day 3, 40 patients had positive reactions, that is, 16 (doubtful), 19 (+), 4 (++), and 1 (+++). The percentage positive reactions was 11.6%.
A patch test using 2% ethanolamine in pet was performed on 370 patients with suspected dermatitis to ethanolamine-containing metal-working fluids and on 452 control subjects. 38 A 10-fold higher reaction was seen in the patient group, with 12.2% of the patients having positive reactions as compared to 1.3% in the control group.
Over a 3-year period, 11 (1.9%) of 595 hairdresser clients and 7 (1.8%) of 401 female hairdressers reacted positively to 2% ethanolamine in pet. 18 In 22 patients with suspected intolerance to oxidative hair dye components, 4 had a positive reaction to 2% ethanolamine in pet. Over a 15-year period, provocative patch testing using ethanolamine was performed on 9602 subjects. There were 363 (3.8%) positive reactions to ethanolamine, and most of the reactions (277; 2.9%) were weak positives. There were 55 (0.6%) irritant reactions reported. Cosensitization was reported, and 38% of the patients who reacted to ethanolamine also tested positive with DEA. Occupational sensitization was reported; of 5884 male patients evaluated, 2.9% who did not work in the metal industry had positive reactions to ethanolamine, as opposed to 7.0% of those working in the metal industry and 15.2% of those exposed to water-based metalworking fluids.
Ocular Irritation
In Vitro
Ethanolamine
The ocular irritation potential of ethanolamine was evaluated in vitro in the rabbit corneal epithelium model. 39 At concentrations of 0.05%, 0.5%, and 1%, ethanolamine was classified as a moderate ocular irritant in this assay.
Nonhuman
In a study using 6 rabbits, 30% aq ethanolamine was moderately irritating to rabbit eyes. Undiluted ethanolamine, instilled as a volume of 0.005 mL, produced severe injury to rabbit eyes. A hair preparation containing 5.96% ethanolamine had a maximum average irritation score of 0.7/110 for rinsed and unrinsed eyes. 2
Occupational Exposure
A case of occupational asthma in an industrial worker exposed to a detergent containing 8% ethanolamine was reported. 40 Since the detergent was used in hot water, release of ethanolamine vapor was greater than if the water temperature was lower. Exposure to vapors from ethanolamine in cleaning solutions can irritate the nose, throat, and lungs. 41
Occupational Exposure Limits
The Occupational Safety and Health Administration permissible exposure level for ethanolamine is 3 ppm (6 mg/m3) as an 8-hour time-weighted average concentration. 42 The National Institute for Occupational Health and Safety has a recommended exposure limit of 3 ppm (8 mg/m3) for a 10 hours workday and 40 hours work week, and the short-term exposure limit is 6 ppm (15 mg/m3), for periods not to exceed 15 minutes. 43
Miscellaneous Studies
Effect on Hepatic Choline
The effects of ethanolamine HCl on hepatic choline was determined in the F0 and F1 parental rats from the 2-generation feeding study described earlier in this report. 44 Liver weights were determined for all parental rats at necropsy, and the livers of control and high-dose animals (1000 mg/kg bw/d) were examined microscopically. Feeding ethanolamine HCl to rats did not affect liver weights of any of the animals. No adverse microscopic affects in the livers of high-dose rats were reported. Liver samples of F1 female rats of the test and control groups were analyzed for choline, phosphocholine, glycerophosphocholine, and phosphatidylcholine. A statistically significant increase in phosphocholine and phosphatidylcholine was observed in the 100- and 300-mg/kg bw/d dose groups, but no other statistically significant difference in choline content was reported. In that there was no dose–response, these effects were not considered dose related. The researchers concluded that “based on the combined hepatic observations, ethanolamine hydrochloride does not affect hepatic choline metabolism in the rat.”
Effect on Bronchoconstriction
The effect of ethanolamine on bronchoconstriction was investigated using a group of 4 anesthetized male Hartley guinea pigs. 45 When an aerosol of 0.1 mL/kg of 3.3% ethanolamine solution was inhaled through a tracheal cannula, bronchoconstriction was observed. The results were compared to those obtained using an aerosol of potassium hydroxide. Bronchoconstriction induced by ethanolamine was greater than that induced by potassium hydroxide. Additional testing suggested that ethanolamine-induced bronchoconstriction may not result from induction of acetylcholine or histamine release, but that it may result partly from an agonistic effect of ethanolamine at the histamine-H1 receptor and the muscarinic receptor.
Summary
This report assesses the safety of ethanolamine and its salts as used in cosmetics. The ethanolamine salts are expected to dissociate into ethanolamine and the corresponding acid. Ethanolamine typically contains a small amount of DEA as an impurity. (The Panel has previously concluded that DEA is safe in the present practices of use and concentration when formulated to be nonirritating; it should not be used in cosmetic products in which N-nitroso compounds may be formed). Ethanolamine and ethanolamine HCl function in cosmetic formulations as pH adjusters. The organic salts and the organic substituted inorganic salts mostly function as surfactants, with the exception of MEA-benzoate and MEA-salicylate, which are preservatives. MEA-sulfite is reported to function as a hair fixative.
In 2011, ethanolamine was reported to be used in 788 formulations, all of which were rinse-offs and 772 of which are hair coloring formulations. Of the ethanolamine-containing ingredients that are in use, none have more than 15 uses. Reported maximum use concentrations of ethanolamine are 0.0002% to 18%. The MEA-lauryl sulfate has the highest reported maximum use concentration at 35% in rinse-off formulations. In Europe, monoalkylamines, monoalkanolamines, and their salts are on the list of substances which must not form part of the composition of cosmetic products, except subject to restrictions and conditions laid down. These restrictions include a maximum secondary amines contaminant content of 0.5% in finished products, a maximum secondary amines content of 0.5% in raw materials, and a maximum nitrosamine content of 50 µg/kg. The MEA-benzoate and MEA-salicylate are on the list of preservatives in Europe and both, as preservatives, are allowed a maximum concentration in ready-for-use preparations of 0.5% of the acid.
Ethanolamine occurs in phospholipids known as phosphatides. In an in vitro study, absorption of undiluted and aq ethanolamine was much greater through mouse skin than it was through human, rat, or rabbit skin; and human skin was the least permeable. The cumulative dose absorbed through mouse and human skin (found in the effluent) was greater with aq ethanolamine than with undiluted ethanolamine; 16.92% of the undiluted and 24.79% of the aq ethanolamine absorbed through mouse skin while 0.61% undiluted and 1.11% aq ethanolamine absorbed through human skin. In split-thickness pig skin, 5% of the dose of ethanolamine HCl in ethanol penetrated percutaneously. In a metabolism and distribution study, ethanolamine HCl was applied dermally to human skin-grafted and ungrafted athymic nude mice. Radioactivity was recovered in the skin (18.4% recovered in grafted skin and 12.1% in ungrafted skin), liver (∼24%-26%), and kidneys (∼2%). Approximately 5% of the radioactivity was recovered in the urine, and 10% of the radioactivity recovered in the urine was unchanged ethanolamine, and the major urinary metabolites were urea and glycine. In an oral study in which Wistar rats were fed a diet containing 100 to 1000 mg/kg ethanolamine HCl for 10 weeks, plasma ethanolamine concentrations increased in a dose-dependent manner from <3 mg/kg in control animals and <4 mg/kg in low-dose animals to 60 to 81 mg/kg in high-dose animals.
Single applications of 5% and 10% ethanolamine were mildly toxic to mouse skin. The dermal LD50 in rabbits was 1.0 to 2.5 g/kg ethanolamine. Oral LD50 values were 0.7 to 15.0 g/kg in mice and 1.0 to 2.9 g/kg in rabbits. The 4-hour inhalation LC50 of a mixture containing ethanolamine, hydroxylamine, 1-aminopropanolo-2-ol, catechol, and water was estimated as 2.48 mg/L. Percutaneous application of 4 mg/kg/d ethanolamine to rats for an unspecified length of time resulted in nonspecific histological changes in the heart and lung, fatty degeneration of the liver parenchyma, and subsequent focal liver necrosis. In a dietary study in which rats were fed 0 to 2.67 g/kg/d ethanolamine for 90 days, heavy livers and kidneys were observed at ≥0.64 g/kg/d, and deaths and “major pathology” occurred with ≥1.25 g/kg/d. In a 2-year dietary study in which Beagle dogs were fed 0 to 0.0975 g/kg/d of a hair dye composite that contained 22% ethanolamine, no toxic effects were observed. In repeated-dose inhalation studies, the dominant effects of “continuous exposure” of dogs, guinea pigs, and rats to 5 to 6 ppm ethanolamine vapor were skin irritation and lethargy; mortality was observed in dogs exposed to 102 ppm ethanolamine vapor for 2 days and in some rodents exposed to 66 to 75 ppm ethanolamine vapor for 24 to 28 days. Dogs and rodents exposed to 66 to 102 ppm ethanolamine vapor had pulmonary and hepatic inflammation, hepatic and renal damage, and hematological changes.
Dermal developmental toxicity studies were performed using rats and rabbits. In a study in which gravid rats were dosed with 10 to 225 mg/kg bw/d ethanolamine on days 6 to 15 of gestation, the NOEL was 75 mg/ kg/d for maternal toxicity and 225 mg/kg/d for embryonal/fetal toxicity. In rabbits that were dosed dermally with 10 to 75 mg/kg bw/d ethanolamine on days 6 to 18 of gestation, the NOEL was 10 mg/kg bw/d for maternal toxicity and 75 mg/kg bw/d for embryonal/fetal toxicity. Significant skin irritation was observed in the high-dose groups for both rats and rabbits. In a Chernoff-Kavlock postnatal mouse screening assay, oral dosing with 850 mg/kg/d ethanolamine on days 6 to 15 of gestation resulted in 16% mortality of maternal mice and a reduced number of viable litters. In a study in which Wistar rats were dosed by gavage with 40 to 450 mg/kg bw/d aq ethanolamine on days 6 to 15 of gestation, the NOEL was 120 mg/kg/d for maternal toxicity and 450 mg/kg/d for developmental toxicity. In an oral study in which Long-Evans rats were dosed with 50 to 500 mg/kg aq ethanolamine on days 6 to 15 of gestation, there was no significant maternal toxicity, but embryolethality was increased in the 500-mg/kg group and more reproductive and developmental effects were observed involving male pups compared to female pups. There was an increase in malformed pups in all dose groups. In a dietary 2-generation study in rats with 100- to 1000-mg/kg bw/d ethanolamine HCl, the NOAEL was 300 mg/kg bw/d for systemic toxicity and for reproductive effects, and the NOAEL was 1000 mg/kg bw/d for developmental toxicity. The effect of ethanolamine HCl on hepatic choline was investigated in the F0 and F1 parental rats, and it was concluded that ethanolamine HCl did not affect the hepatic choline metabolism in the rat.
Ethanolamine was mostly negative in Ames tests (≤2000 μg/plate). Ethanolamine (≤500 μg/mL) was negative in a cell transformation assay. Ethanolamine did not produce chromosomal aberrations in rat liver cells but did produce a “weak positive” response in human lymphocytes. Carcinogenicity data were not found in the published literature.
Semiocclusive application of 30% to 100% ethanolamine was corrosive to rabbit skin. Irritation responses ranging from irritating to corrosive were observed with 1% to 10% ethanolamine, respectively, applied nonocclusively to rabbit ears and under semiocclusive patches to rabbit skin. The degree of irritation increased with the concentration applied. In human studies, a formulation containing 5.9% ethanolamine was a cumulative irritant in a 21-day study, and an irritant response was reported during the induction phase of a patch test of a formulation containing 11.47% ethanolamine. Ethanolamine HCl, ≤70%, did not have a sensitizing effect in a local lymph node assay. In a maximization study in guinea pigs with intradermal and epicutaneous inductions of 0.6% and 10.3% aq ethanolamine, respectively, positive reactions were observed at challenge with 0.41%, 2.05%, and 4.1% ethanolamine. Possible cross-reactions to 5% TEA and 7% DEA were reported. In provocative testing of metalworkers with dermatitis, a higher reaction rate to ethanolamine was observed for the patients as compared to control subjects. In a study of hairdressers and clients, cosensitization to DEA was reported for 38% of the patients who reacted to ethanolamine.
In ocular irritation studies in rabbits, 30% aq ethanolamine was moderately irritating to rabbit eyes, and undiluted ethanolamine produced severe injury. A hair preparation containing 5.96% ethanolamine had a maximum average irritation score of 0.7/110 in rinsed and unrinsed eyes. Ethanolamine, tested at 0.05%, 0.5%, and 1%, was classified as a moderate irritant in the in vitro rabbit corneal epithelial model evaluating ocular irritation potential.
One-tenth mL/kg of a 3.3% ethanolamine aerosol solution induced bronchoconstriction in guinea pigs. Ethanolamine-induced bronchoconstriction may result partly from an agonistic effect of ethanolamine at the histamine-H1 receptor and the muscarinic receptor.
Discussion
This amended safety assessment originated as a rereview of ethanolamine and was expanded to include 12 ethanolamine salts, the safety of which were supported by the data available in the original safety assessment and by other published and unpublished studies. These ingredients are reported to be used only in rinse-off products; therefore, the CIR Expert Panel addressed the safety as used in rinse-off products only.
While the Panel noted gaps in the available safety data for the ethanolamine salts included in this group, the Panel relied on the information available for ethanolamine in conjunction with previous safety assessments of the components of these ingredients. Those data could be extrapolated to support the safety of these ingredients. For example, coconut acid has been found safe as used, and the Panel was able to extrapolate these data to support the safety of MEA-cocoate (ie, the ethanolamine salt of coconut acid).
The Panel noted that small amounts of DEA could be present in ethanolamine and was concerned with levels of free DEA that could be present as an impurity. Therefore, ethanolamine and ethanolamine-containing ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed.
Also, because DEA might be present as an impurity in ethanolamine, the Panel reiterated its discussion regarding the positive findings reported in a dermal carcinogenicity study of DEA. The hepatocarcinogenicity that was reported in mice was considered to have little relevance to the safety of DEA in personal care products. Additionally, renal lesions reported in mice could have been the result of DEA-induced choline deficiency, a mechanism that has little relevance in humans. If DEA-induced choline deficiency was not the cause of the renal lesions, it was thought there was still no carcinogenic risk to humans because DEA does not appear to penetrate human skin to any significant extent at concentrations relevant to human exposures from the use of personal care products.
Because ethanolamine can be used in products that may be aerosolized, that is, a hair color spray, the Panel discussed the issue of incidental inhalation exposure. The limited inhalation data included a single dose study of a nondefined mixture that included ethanolamine and a repeated dose study of ethanolamine did not suggest potential respiratory effects at relevant doses. Since inhalation data were limited, the Panel considered other available data and noted that there was lack of relevant systemic toxicity in dermal and oral single and repeated dose toxicity and reproductive toxicity studies and mostly negative results in genotoxicity studies. Further, this ingredient is reportedly used at concentrations of ≤3% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. However, the potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. Inhaled droplets/particles deposited in the nasopharyngeal and bronchial regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. Nevertheless, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects.
Finally, the potential exists for dermal irritation with the use of products formulated using ethanolamine or ethanolamine salts. The Panel specified that products containing these ingredients must be formulated to be nonirritating.
Conclusion
The CIR Expert Panel concluded that ethanolamine and the 12 related ethanolamine salts, listed subsequently, are safe in the present practices of use and concentration described in this safety assessment (rinse-off products only) when formulated to be nonirritating. The Panel cautioned that ingredients should not be used in cosmetic products in which N-nitroso compounds may be formed. Ethanolamine Ethanolamine HCl* MEA-benzoate* MEA-cocoate MEA-laureth-6 carboxylate* MEA-laureth sulfate MEA-lauryl sulfate MEA-PPG laureth-6 carboxylate* MEA-PPG-8-steareth-7 carboxylate* MEA-salicylate* MEA-sulfite* MEA-tallowate MEA-undecylenate*
Were the ingredients not in current use (as indicated by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the director, Cosmetic Ingredient Review, 1620L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
M. M. Fiume contributed to conception and design; acquisition, analysis, and interpretation; and drafted the article. B. A. Heldreth contributed to conception and design; acquisition, analysis, and interpretation; drafted the article, and critically revised the article. L. J. Gill, W. F. Bergfeld, D. V. Belsito, C. D. Klaassen, J. G. Marks, R. C. Shank, T. J. Slaga, and P. W. Snyder contributed to conception and design, analysis and interpretation, and critically revised the article. R. A. Hill contributed to conception and design, analysis and interpretation, and critically revised the article, and gave final approval. D. C. Liebler contributed to conception and design, analysis and interpretation, and critically revised the article. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
